Abstracts
The origin of avian endothermy is a long-held question the answer of which cannot be provided by first level observations. Oological and reproductive characters have collectively provided a new source of data useful for phylogenetic analyses and paleobiological inferences. In addition, the observations of reproductive and oological evolutionary trends in saurischian dinosaurs lead to the interpretation that not only, the thermophysiology of these dinosaurs progressively became more avian-like but after re-examination allows to infer that deinonychosaurians represented here by three troodontids and one dromaeosaurid might already have developed an avian-like endothermy, thus predating the rise of avians. These results based on reproductive traits are independently corroborated by the discoveries of troodontid dinosaurs 1) in high latitudes, 2) covered with feathers in Chinese Lagerstätten, and recently 3) fossilized in a death pose identical to an avian sleeping posture.
endothermy; avian; troodontid theropod dinosaurs; oology; reproductive behaviors
A origem da endotermia nas aves é uma questão há muito discutida e sua resposta não pode ser encontrada através de observações superficiais. Caracteres oológicos e reprodutivos surgiram como uma nova fonte de dados relevantes tanto para análises filogenéticas quanto para inferências paleobiológicas dos dinossauros Saurischia. Além disso, as observações das tendências evolutivas reprodutivas e oológicas nos dinossauros Saurischia nos levam a inferir que não apenas, como anteriormente a termofisiologia deste grupo de dinossauros tornou-se progressivamente ornítica, mas um posterior reexame nos permitiu concluir que os deinonicossauros, representados aqui por dois troodontídeos e um dromeossaurídeo, provavelmente já haviam desenvolvido uma endotermia semelhante a das aves, anterior, portanto, ao seu surgimento. Estes resultados baseados em características reprodutivas são independentemente corroborados pela descoberta dos dinossauros troodontídeos 1) em altas latitudes, 2) recobertos com penas, no chinês Langerstäten, e, recentemente, 3) fossilizados em posição idêntica à postura de repouso das aves.
endotermia; aviano; troodontídeo dinossauro terópodo; oologia; comortamentos reprodutivos
Oology and the evolution of thermophysiology in saurischian dinosaurs: homeotherm and endotherm deinonychosaurians?
Gerald Grellet-Tinner
Museu de Zoologia, Universidade de São Paulo, Caixa Postal 42494-970, CEP 04218-970, São Paulo, SP, Brasil. Email: deinonychus@swissinfo.org
ABSTRACT
The origin of avian endothermy is a long-held question the answer of which cannot be provided by first level observations. Oological and reproductive characters have collectively provided a new source of data useful for phylogenetic analyses and paleobiological inferences. In addition, the observations of reproductive and oological evolutionary trends in saurischian dinosaurs lead to the interpretation that not only, the thermophysiology of these dinosaurs progressively became more avian-like but after re-examination allows to infer that deinonychosaurians represented here by three troodontids and one dromaeosaurid might already have developed an avian-like endothermy, thus predating the rise of avians. These results based on reproductive traits are independently corroborated by the discoveries of troodontid dinosaurs 1) in high latitudes, 2) covered with feathers in Chinese Lagerstätten, and recently 3) fossilized in a death pose identical to an avian sleeping posture.
Keywords: endothermy, avian, troodontid theropod dinosaurs, oology, reproductive behaviors.
RESUMO
A origem da endotermia nas aves é uma questão há muito discutida e sua resposta não pode ser encontrada através de observações superficiais. Caracteres oológicos e reprodutivos surgiram como uma nova fonte de dados relevantes tanto para análises filogenéticas quanto para inferências paleobiológicas dos dinossauros Saurischia. Além disso, as observações das tendências evolutivas reprodutivas e oológicas nos dinossauros Saurischia nos levam a inferir que não apenas, como anteriormente a termofisiologia deste grupo de dinossauros tornou-se progressivamente ornítica, mas um posterior reexame nos permitiu concluir que os deinonicossauros, representados aqui por dois troodontídeos e um dromeossaurídeo, provavelmente já haviam desenvolvido uma endotermia semelhante a das aves, anterior, portanto, ao seu surgimento. Estes resultados baseados em características reprodutivas são independentemente corroborados pela descoberta dos dinossauros troodontídeos 1) em altas latitudes, 2) recobertos com penas, no chinês Langerstäten, e, recentemente, 3) fossilizados em posição idêntica à postura de repouso das aves.
Palavras-chave: endotermia, aviano, troodontídeo dinossauro terópodo, oologia, comortamentos reprodutivos.
INTRODUCTION
Oological and reproductive characters known to be useful for phylogenic analyses (Grellet-Tinner, 2000; Makovicky and Grellet-Tinner, 2000; Grellet-Tinner and Chiappe, 2004; Buffetaut et al., 2005; Grellet-Tinner in press) also provide a source of new data for the paleobiology of saurischian dinosaurs (Grellet-Tinner et al., 2006). Investigation of the physiology in fossilized vertebrates seems a doubtful query, as it cannot be observed directly. However, the discoveries of troodontids species in high paleolatitudes (Fiorillo and Gangloff, 2000), or with feathers in Chinese Lagerstätten suggest a metabolism different than the typical reptilian ectothermy. More importantly, recent evidence of an avian behavior in a non-avian dinosaur led to the interpretation that a 130 millions years old basal troodontid (Xu and Norell, 2004) would have already acquired a homeothermic physiology. Independent paleobiological interpretations founded on oological data allowed Grellet-Tinner et al. (2006) to unravel the evolution of thermophysiology in saurischians. A concise review of key elements is presented here with new conclusions.
Abbreviations
Institutional abbreviations: AMNH, American Museum of Natural History, New York; IGM, Institute of Geology, Mongolia, Ulaan Baatar; MCZ, Museum of Comparative Zoology, Harvard; MOR, Museum of the Rockies; YPM, Yale Peabody Museum of Natural History, New Haven; USC, University of Southern California; LX09XLOZLX00, identification number for US Dept. of Interior.
Technical abbreviations: BSEM, backscattered scanning electron microscopy; CL, cathodoluminescence; DPI, dots per inches; MT, Membrana Testacea; microscopy; PLM, polarized light microscopy; SEM, scanning electron microscope; TLM, transmitted light microscopy.
MATERIAL AND METHODS
Material
Fossil eggs can only be identified taxonomically with certainty either by observing an egg-laying female or by the embryos they contain. In the fossil record, the former requires the rare preservation of a female with eggs in its reproductive system (Sato et al., 2005), the latter is relatively more often recorded (Currie and Horner, 1988; Horner and Weishampel, 1988; Horner and Currie, 1994; Norell et al., 1994, 2001; Horner, 1997; Manning et al., 1997; Mateus et al., 1997; Chiappe et al., 2001a; Schweitzer et al., 2002). These discoveries have also allowed the association of eggs of several extinct dinosaurs to species-specific nest structures (Varricchio et al., 1997; Chiappe et al., 2004) and in some instances, to species-specific nesting behaviors (Norell et al., 1995; Dong and Currie, 1996; Clark et al., 1999; Varricchio et al., 1999; Grellet-Tinner et al., 2006). Nevertheless, taphonomic and taxonomic levels of confidence (two different indices) might differ for a given oological specimen according to its proximity to specific skeletal remains and it's posteriori taxonomic identification, thus could foster erroneous phylogenetic and paleobiological interpretations. As such, the highest level of taphonomic confidence in the association of fossil eggs with skeletal remains occurs when diagnostic embryos are found in ovo (Norell et al., 1994, 2001; Chiappe et al., 2001a) or when eggs are preserved inside a gravid female (Sato et al., 2005). However, a high taphonomic level does not always correspond to a first level of taxonomic confidence. The identification of the skeletal remains could be uncertain, controversial (Horner, 1997; Manning et al., 1997; Mateus et al., 1997), rebutted (Chiappe et al., 2001b), or at a low taxonomic hierarchical level that does not allow an identification at the species or family level (Schweitzer et al., 2002, Buffetaut et al., 2005), leading to a lower level of taxonomic confidence. Although such occurrences do not by essence discriminate against their use for phylogenetic analyses or paleobiologic interpretations, the descriptions of their discoveries and identifications need to be reported. For descriptions of the various taphonomic and taxonomic levels of confidence assigned to the studied material (Table 1), see Grellet-Tinner et al. (2006).
In order to better understand the evolution of eggs, eggshell structures, nesting behaviours, and reproductive patterns in saurischian dinosaurs, crocodilians and modern birds, the two living clades that bracket this group of dinosaurs, need to be considered. This because many aspects of the reproduction, nesting structures, and behaviours of these living clades reflect two radically opposite thermophysiologies (Table 2).
Methods
Eggshell samples were examined under SEM at the AMNH and Field Museum facilities. Additionally, radial thin sections of eggshell were studied using transmitted light (TRM) and polarized light (PLM) microscopy at the AMNH and Earth Sciences Department of USC. Cathodoluminescence (CL) analysis was performed at the Earth Sciences Department of USC to determine possible diagenetic changes of composition or structure.
Readers are referred to Grellet-Tinner et al. (2006) for more detailed information.
RESULTS
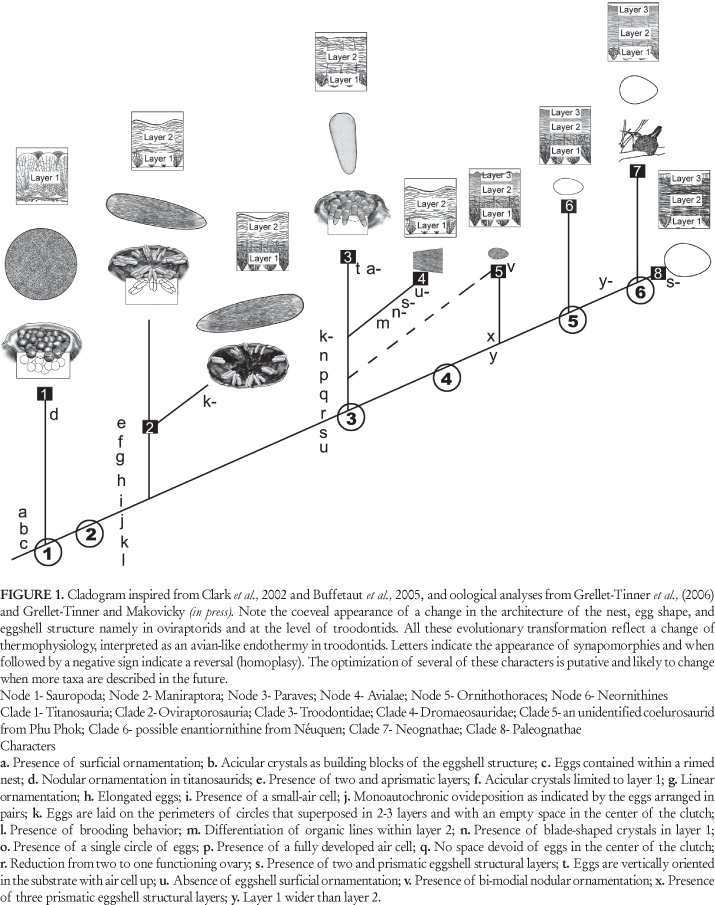
The saurischian taxa studied (Table 1) by Grellet-Tinner et al. (2006) clearly reveal a reproductive evolutionary cline (Fig. 1) from basal archosaurs (as inferred from crocodilians) to modern birds, namely a successive acretion of eggshell structural layers, an monoautochronic ovideposition, the atrophy of one ovary or the evolutionary development of a single functioning ovary, clutch spatial arrangements and nest structures that reflects a change of parental care, which is itself related to a successive and gradual change in thermophysiology in saurischians. Without paraphrasing Grellet-Tinner et al. (2006), a brief review of reproductive transformations in the saurischian lineage with explanation of key features is provided below with discussion and new interpretations.
Considering the entire body of oological and nesting evidence, titanosaurid dinosaurs, a clade of saurischian dinosaurs, would have possessed a quasi similar reproductive system and nesting behaviours as crocodilians. The similarities include an en masse egg laying strategy, symmetrical eggs without air cells, mono-layered eggshell structure, and a very limited nesting parental care (Grellet-Tinner, 2005, Grellet-Tinner et al., 2006). Unlike titanosaurs, the oviraptor C. osmolskae IGM 100/979 (Norell et al., 1994, 2001) and other oviraptorid fossils have been found sitting on their nests in an avian posture (Dong and Currie, 1996). In such life assemblages, oviraptors sit atop an egg clutch with their axial skeleton posited in the clutch center that lacks eggs (Clark et al., 1999; Norell et al., 2001), an important feature for the interpretation of nesting parental care. Commonly, the eggs are paired and arranged in a sub-horizontal position forming up to three superposed layers (Clark et al., 1999; Norell et al., 2001). While this type of life assemblage documents a brooding activity, here solely recognized as protective behavior, it falsifies at the same time the possibility of any heat transfer (incubation) by the presence of multiple rows of eggs and the absence of evidence of egg post-partum manipulation (Grellet-Tinner, 2005; Grellet-Tinner et al., 2006). Nevertheless, the oval and elongated eggs of C. osmolskae display an avian trend by having a faint polar asymmetry not yet as pronounced as that of extant birds but that still indicates the presence of a small air cell (Grellet-Tinner et al., 2006), a feature that is absent in the symmetrical crocodilian (Iverson and Evert, 1995) and titanosaurids eggs (Grellet-Tinner et al., 2004, 2006). Another feature that characterizes C. osmolskae is its paired eggs, a spatial arrangement indicative of two functioning ovaries (Clark et al., 1999; Norell et al., 2001), as titanosaurs and all primitive saurischians. At the same time this advocates for a monoautochronic ovideposition (Clark et al., 1999; Norell et al., 2001, Sato et al., 2005), unlike the en masse ovideposition of crocodilians and titanosaurids. In addition to this mosaic of characters, the eggshell of C. osmolskae is structurally divided in two layers, an intermediate character between the mono-layered crocodilian (Grellet-Tinner and Chiappe, 2004) and tri-layered avian eggshell structures (Grellet-Tinner, 2000, Grellet-Tinner and Chiappe, 2004, Grellet-Tinner et al., 2006; Grellet-Tinner, 2006). Kamat et al. (2000) argued that molluscs achieve a greater mechanical equilibrium by increasing the number of structural layers of their shell and emphasized that differences in crystallographic orientation among layers enhances this trend, a type of biological plywood. In that respect, it is worth noting that the volume of C. osmolskae eggs closely matches that of the Auca Mahuevo titanosaurs but its bi-laminated eggshell is twice as thin and its eggs need to support the weight of overlying eggs and the partial weight of the appendicular skeleton of a brooding adult.
Dozen of small asymmetrical eggs some with embryonic remains in ovo have been found in Cretaceous exposures of the Bajo de la Carpa Formation of Neuquén city, Patagonia (Schweitzer et al., 2002). Although the taphonomic level of confidence of this material is extremely high by the presence of embryos in ovo, the phylogenetic position of this material has been only bracketed between the Ornithothoraces and Ornithuromorpha avian nodes with a likelihood that it could belong to enantiornithines (Schweitzer et al., 2002). As expected by their phylogenetic proximity to extant birds, the fully asymmetric shape of these ornithothoracine eggs from Neuquén shaped like those of modern galliform birds is indicative of the presence of a fully developed air cell (Grellet-Tinner et al., 2006), and their eggshell bears no surficial ornamentation (Schweitzer et al., 2002; Grellet-Tinner et al., 2006).
Most importantly, these eggs share with modern birds a tri-laminated eggshell structure with prismatic inter layers contacts (sensu Grellet-Tinner and Norell, 2002) and short blade-shaped calcite crystals in layer 1. However, the thickness of layer 1 exceeds that of layer 2 (by a ratio close to 1.4) in these enantiornithines (Grellet-Tinner, 2005; Grellet-Tinner et al., 2006) and Phu Phok eggs (Buffetaut et al., 2005). This character is so far unique to these two groups and not recorded in any other positively identified non-avian theropod or modern birds (Buffetaut et al., 2005; Grellet-Tinner et al., 2006). Several eggs in Neuquén were discovered within a limited area but only a few were complete and the rest consists of the halves with a tapered pole (without air cell). This taphonomic feature is entirely congruent with the hatching process in modern seagull colonies where the same egg sections are left intact resting on the ground surface (Hayward et al., 2000). The blunt polar region (with the air cell) and the region immediately adjacent to this pole are often destroyed during the hatching process in modern birds (Hayward et al., 2000). All the oological characters and hatching patterns advocate that basal ornithothoraces would have had a modern avian nesting and hatching behaviors, thus an avian thermophysiology.
In many aspects, troodontids, here phylogenetically bracketed between Oviraptorids and enantiornithines (Hwang et al., 2004; Novas and Pol, 2005), offer intriguing and intermediary characters between C. osmolskae and the ornithothoracine eggs from Neuquén. First, the troodontid eggs of T. formosus are still elongated but conical and extremely asymmetrical (Figs. 2A, B). One pole is extremely rounded, which offers a greater mechanical stress resistance as it approximates the shape of a sphere (Bain, 1991). This asymmetry is far more pronounced than that of C. osmolskae and closely resembles the condition commonly observed in ornithothoracines. This feature also suggests that troodontids already possessed an avian-like air cell instead of a small air cell as inferred for oviraptorids and other elongatoolithid eggs (Grellet-Tinner et al., 2006). Second, the short and blade-shaped calcite crystals present in the eggshell structural layer 1 (Figs. 2C, 3C, D) are similar to those observed in known Avialans (Grellet-Tinner, 2000; Grellet-Tinner and Chiappe, 2004) and depart from the acicular crystals (Fig. 3A) observed in this layer of oviraptorid eggshell (Grellet-Tinner et al., 2006) and Deinonychus antirrhopus (Makovicky and Grellet-Tinner, 2000 and Grellet-Tinner and Makovicky; in press). Third, in contrast to the aprismatic contact between the two structural layers observed in oviraptorids and D. antirrhopus (Fig. 3A) and other well-identified Mesozoic theropod eggshells (per. obs, Lourinhã allosauroid material) and in resemblance to ornithothoracine, the transition between layers 1 and 2 (Figs. 2C, 3C, D) is prismatic (sensu Grellet-Tinner and Norell, 2002). Fourth, troodontid eggshells lack surficial ornamentation (Figs. 2C, 3C, D) and fifth, the novel spatial arrangement of troodontid eggs (Fig. 2A, B) mimics that of modern pluvianids (e.g. Grant, 1982; Howell, 1979) by having eggs nearly vertically embedded with their round poles (with air cell) up and barely exposed above the sediment (Figs. 2A, B), a nesting behavior compatible with the hot environments where this troodontid species reproduced (Varricchio, 1993, Loope et al., 1998) and that optimizes the space in the nest (Barta and Szckely, 1997). Lastly, the combined absence of space devoid of eggs in the clutch center (Figs. 2A, B) and the presence of a partial adult skeletal atop the eggs in MOR 748, 750 (Varricchio et al., 1997) fundamentally separate troodontid from oviraptorid nesting behaviors as the axial skeleton of brooding troodontid is in direct contact with eggs placed on a single level. Remarkably, a recently discovered clutch of the Asian troodontid (in prep), Byronosaurus jaffei, from the Upper Cretaceous layers of Djadokhta Formation of Ukhaa Tolgod (Mongolia) and clutches of recently acquired troodontid eggs from China (per.obs) corroborates what has been documented in T. formosus. Moreover, as for the Neuquén ornithothoraces eggs (Grellet-Tinner et al., 2006), the B. jaffei eggs consist of only the tapered halves and adjacent region, indicative of a hatching strategy similar to that of modern birds (Hayward et al., 2000). Above all, Reptilian and avian nests are regarded as microenvironments that optimize egg incubation and hatching (Proctor and Lynch, 1993; Barta and Szckely, 1997). As behaviors of extinct animals are extrapolated from observations of their living relatives, pluvianid nesting and incubating behaviors provide valuable biological clues about the possible nesting behavior and thermophysiology of troodontids. The pluvianid Pluvianus aegyptius partially buries its eggs by digging a depression in sandbars (Howell, 1979). Egg Mountain and Egg Island, the two localities where T. formosus have been recovered were islands in local lacustrine systems (Varricchio, 1993) and B. jaffei paleoenvironments is best described as semi-desertic with sand dunes and oasis. Pluvianus aegyptius eggs fill a bowl-like depression without leaving any empty space, are positioned in a sub-vertical position pointed pole down with only the blunt pole of these eggs exposed periodically. Eggs partially buried or buried only a few centimeters below the surface in hot environments could easily reach lethal levels of 40º centigrade for birds but only 28º centigrade for other extant reptiles during the day if not attended by the parents and experience severe cooling periods during the night. The thermoregulation strategy of the parents is to cool down their clutch with water from nearby sources (Grant, 1982) and supply body heat during the cool hours by exposing the rounded pole of their eggs to incubate them. The over all suite of preserved oological and nesting structure in extinct troodontids closely resembles those extant pluvianids and favorably argues that troodontids could possess similar nesting behaviors. In turn, these reproductive and oological characters highly suggest that the North American and Asian troodontids already acquired an avian-like incubating behaviour, the latter been only feasible by acquiring an endothermic ther-mophysiology. When considering the total evidence that include oology with the associated nesting behaviour, the discovery of a basal troodontid (Xu and Norell, 2004) preserved with a tuck-in avian posture, other troodontids recovered with feather-like integument, and troodontid remains discovered in high paleolatitudes, it seems parsimonious to argue that troodontid dinosaurs were endothermic and homeothermic. Whether this thermophysiological innovation was limited to this theropod family remains unanswered. Yet, according to Clark et al. (2002) Troodontidae is the sister taxon to Dromaesauridae, a clade that contains D. antirrhopus (AMNH 3015) a theropod discovered with a partial egg (Makovicky and Grellet-Tinner, 2000; in press). The convex external eggshell surface of AMNH 3015 is apposed to the outer surface of well-preserved gastralia suggesting that, like oviraptorids, this dromaesoraurid would have brooded its eggs. Moreover, the contact of the axial skeleton (gastralia) with the egg, which is similar to troodontids, could be equally interpreted as a heat transfer mechanism, thus suggesting incubation and endothermy in D. antirrhopus. In turn, if confirmed, this thermophysiological feature present in both Troodontids and D. antirrhopus would be a synapomorphy of Deinonychosauria and have predated the divergence of Avialans.
ACKNOWLEDGMENTS
SEM work was done at The FM and The AMNH, TLM, PLM and CL examinations were performed at the Carbonate Laboratory of the Department of Earth Sciences at USC. I would also like to acknowledge E. Buffetaut, R. Coria, J. Cracraft, P. Currie, G. Dyke, J. Gauthier, J. Horner, C. Magovern, M. Norell, P. Makovicky, L. Salgado, P. Sweet, M. Vianey-Liaud, and Zhao, Zi-Kui for lending specimens. This research was funded by the Department of Earth Sciences at USC, the Geological Society of America, AAPS, the Society of Vertebrate Paleontology for work in China, two Collections study grants at the FM and AMNH, and the State of São Paulo Research Foundation (FAPESP) within the BIOTA/FAPESP program (grants 02/136024 and 05/551003). I would like to express my gratitude to Doyle Trankina for his artistic rendering of figure 2 and to Luciana Moreira Lobo for her translation of the abstract in Portuguese. I would also like to thank the two reviewers (C. Maria Magalhães Ribeiro and M. Langer) for their insightful comments.
Recebido em: 17.01.2006
Aceito em: 03.04.2006
- Bain, M.M. 1991. A reinterpretation of eggshell strength. In: Solomon, S.E. (Ed.), Egg and Eggshell Quality. Wolfe Publication, London, p.131-145.
- Barta, Z. & Szekely, T. 1997. The optimal shape of avian eggs. Functional Ecology, 11(5):656-662.
- Buffetaut, E.; Grellet-Tinner, G.; Suteethorn, V.; Cuny, G.; Tong, H.; Koir, A.; Cavin, L.; Chitsing, S.; Griffiths, P.J.; Tabouelle, J., & Le Loeuff, J. 2005. Very small eggs and embryo from the Early Cretaceous of Thailand and the dinosaur-bird transition. Naturwissenschaften, DOI 10.1007/s00114-005-0022-9.
- Chiappe, L.M.; Salgado, L. & Coria, R.A. 2001a. Embryonic skulls of titanosaur sauropod dinosaurs. Science, 293(5539):2444-2446.
- Chiappe, L.M; Norell, M.A. & Clark, J. 2001b. A new skull of Gobipteryx minuta (Aves: Enantiornithines) from the Cretaceous of the Gobi Desert. American Museum Novitates, 3346:1-15.
- Chiappe, L.M.; Schmitt, J.G.; Jackson, F.D.; Garrido, A.; Dingus, L. & Grellet-Tinner, G. 2004. Nest Structure for Sauropods: Sedimentary Criteria for Recognition of Dinosaur Nesting Traces. Palaios, 19: 89-95.
- Clark, J.M.; Norell, M.A. & Chiappe, L.M. 1999. An oviraptorid skeleton from the Late Cretaceous of Ukhaa Tolgod, Mongolia, preserved in an avian like brooding position over an oviraptorid nest. American Museum Novitates, 3265:1-36.
- Clark, J.M.; Norell, M.A. & Makovicky, P.J. 2002. Cladistic Approaches to the Relationships of Birds to other Theropod Dinosaurs. In: Chiappe, L.M. & Witmer, M. (Eds.), Mesozoic Bird: Above the Heads of Dinosaurs., University of California Press, London, p.31-61.
- Currie, P.J. & Horner, J.R. 1988. Lambeosaurine hadrosaur embryos (Reptilia: Ornithischia). Journal of Vertebrate Paleontology, 8: (Supplement to Number 3), 13A.
- Dong, Z. & Currie, P.J. 1996. On the discovery of an oviraptorid skeleton on a nest of eggs at Bayan Mandahu, Inner Mongolia, People's Republic of China. Canadian Journal of Earth Sciences, 33:632-636.
- Fiorillo, A. & Gangloff, R.A. 2000. Theropod teeth from the Prince Creek Formation (Cretaceous) of Northern Alaska, with speculation on Arctic dinosaur paleoecology. Journal of Vertebrate Paleontology, 2:675-682.
- Grant, G.S. 1982. Avian incubation: egg temperature, nest humidity, and behavioral thermoregulation in a hot environment. Ornithological Monographs, 30:1-75.
- Grellet-Tinner, G. 2000. Phylogenetic interpretation of eggs and eggshells. In: Bravo, A.M. and Reyes, T. (Eds.), First International Symposium on Dinosaur Eggs and Babies, Isona I Conca Dellà Catalonia, Spain, p. 61-75.
- Grellet-Tinner, G. 2005. A phylogenetic analysis of oological characters: A case study of saurischian dinosaur relationships and avian evolution. Ph.D. Dissertation, University of Southern California, Los Angeles.
- Grellet-Tinner, G. 2006. Phylogenetic interpretation of eggs and eggshells: implications for oology and Paleognathae phylogeny. Alcheringa, 30:139-180.
- Grellet-Tinner, G. & Norell, M. 2002. An avian egg from the Campanian of Bayn Dzak, Mongolia. Journal of Vertebrate Paleontology, 17:719-721.
- Grellet-Tinner, G. & Chiappe, L.M. 2004. Dinosaur eggs and nestings: implications for understanding the origin of birds. In: Currie, P.J.; Koppelhus, E.B.; Shugar, M.A. & Wright, J.L. (Eds.), Feathered dragons: Studies on the Transition from dinosaurs to Birds, Indiana University Press, Bloomington, p.185-214.
- Grellet-Tinner, G. & Makovicky, P.J. In press. A possible egg of the theropod Deinonychus antirrhopus: Phylogenetic and biological implications. Canadian Journal of Earth Sciences.
- Grellet-Tinner, G.; Chiappe, L.M. & Coria, R. 2004. Eggs of titanosaurid sauropods from the Upper Cretaceous of Auca Mahuevo (Argentina). Canadian Journal of Earth Sciences, 41(8):949-960.
- Grellet-Tinner, G.; Chiappe, L.M.; Bottjer, D. & Norell, M.A. 2006. Paleobiology of dinosaur eggs and nesting behaviors. Palaeogeography, Palaeoclimatology, Palaeoecology, 232(24):294-321.
- Hayward, J.M.; Zelenitsky, D.K.; Smith, D.L.; Zaft, D.M. & Clayburn, J.K. 2000. Eggshell taphonomy at modern gull colonies and a dinosaur clutch site. Palaios, 15:343-355.
- Horner, J.R 1997. Rare preservation of an incompletely ossified fossil embryo. Journal of Vertebrate Paleontology, 17:431-434.
- Horner, J.R. & Weishampel, D.B. 1988. A comparative embryological study of two ornithischian dinosaurs. Nature, 332(6161):256-257.
- Horner, J.R. & Currie, P.J. 1994. Embryonic and Neonatal Morphology and Ontogeny of a New Species of Hypacrosaurus (Ornithischia, Lambeosauridae) from Montana and Alberta. In: Carpenter, K.; Hirsch, K.F & Horner, J.R. (Eds.), Dinosaur Eggs and Babies, Cambridge University Press, New York, p.312-336.
- Howell. T.R. 1979. Breeding Biology of the Egyptian Plover (Pluvianus aegyptius). University of California Publications in Zoology, 113:1-76.
- Hwang, S.H.; Norell, M.A.; Qiang, J. & Kegin, G. 2002. New specimens of Microraptor zhaoianus (Theropoda: Dromaeosauridae) from northeastern China. American Museum Novitates, 3381:1-44.
- Iverson, J.B. & Evert, M.A. 1995. Physical characteristics of reptilian eggs and a comparison with avian eggs. In: D. C. Deeming, D.C. & M. W. J. Ferguson, M.W.J (Eds.), Egg Incubation: Its Effects on Embryonic Development in Birds and Reptiles, Cambridge University Press, New York, p.87-101.
- Kamat, S.; Su, X.; Ballarini, R. & Heuer, A.H. 2000. Structural basis for the fracture toughness of the shell of the conch Strombus gigas. Nature, 405(6790):1036-1040.
- Loope, D.B.; Dingus L.; Swissher, III,C.C. & Minjin, C. 1998. Life and death in the Late Cretaceous dune field, Nemegt Basin, Mongolia. Geology, 26:27-30.
- Makovicky, P. & Grellet-Tinner, G. 2000. Association between a specimen of Deinonychus antirrhopus and theropod eggshell. In: Bravo, A.M. & Reyes, T. (Eds), First International Symposium on Dinosaur Eggs and Babies, Isona I Conca Dellà Catalonia, Spain, p.123-128.
- Manning, T.W.; Joysey, K.A. & Cruickshank, A.R.I. 1997. Observations of microstructures within dinosaur eggs from Henan Province, Peoples' Republic of China. In:Wolberg, D.L.; E. Stump, E. & G.D. Rosenberg, G.D. (Eds.), Dinofest International, The Academy of Natural Sciences, Philadelphia, p.287-290.
- Mateus, I.H.; Mateus, M.T.; Antunes, O.; Mateus, P.; Taquet, V.; Ribeiro, & Manuppella, G. 1997. Couvée, oeufs et embryons d'un dinosaure théropode du Jurassique supérieur de Lourinhã (Portugal). Comptes Rendus de l'Académie des Sciences de Paris, Série 2, 325(A):71-78.
- Norell, M.A.; Clark, J.M.; Dashzeveg, D.; Barsbold, R. Chiappe, L.M.; Davidson, A.R,; McKenna, M.C.; Altangerel, P. & Novacek, M.J. 1994. A theropod dinosaur embryo and the affinities of the Flaming Cliffs dinosaur eggs. Science, 266:779-782.
- Norell, M.A.; Clark, J.M.; Chiappe, L.M., & Dashzeveg, D. 1995. A nesting dinosaur. Nature, 378(6559):774-776.
- Norell, M.A.; Clark, J.M. & Chiappe, L.M. 2001. An embryonic oviraptorid (Dinosauria: Theropoda) from the Upper Cretaceous of Mongolia. American Museum Novitates, 3325:1-17.
- Novas, F.E. & Pol, D. 2005. New evidence on Deinonychosaurian dinosaurs from the late Cretaceous of Patagonia. Nature, 433(7028):858-861.
- Proctor, N. & Lynch, P. 1993. Manual of Ornithology, Avian Structure and Function. Yale University Press, New Haven.
- Sato, T.; Cheng, Y-N.; Wu, X-C.; Zelenitsky, D.K. & Hsiao, Y-F. 2005. A pair of shelled eggs inside a female dinosaur. Science, 308(5720):375.
- Schweitzer, M.; Jackson, F.; Chiappe, L.M.; Schmitt, J.G.; Calvo, J.O. & Rubilar, D.E. 2002. Late Cretaceous avian eggs with embryos from Argentina. Journal of Vertebrate Paleontology, 22:191-195.
- Varricchio, D.J. 1993. Montana climatic changes associated with the Cretaceous Claggett and Bearpaw transgressions. In: Hunter, L.D.V. Energy and mineral resources of Central Montana. Montana Geological Society, Billings, p.97-102
- Varricchio, D.J.; Jackson, F.; Borkowski, J.J. & Horner, J.R. 1997. Nest and egg clutches of the dinosaur Troodon formosus and the evolution of avian reproductive traits. Nature, 385(6613):247-250.
- Xu, X. & Norell, M.A. 2004. A new troodontid dinosaur from China with avian-like sleeping posture. Nature, 431(7010):838-841.
Publication Dates
-
Publication in this collection
14 Sept 2006 -
Date of issue
2006
History
-
Accepted
03 Apr 2006 -
Received
17 Jan 2006