Abstracts
BACKGROUND AND OBJECTIVES: Combined spinal-epidural (CSE) is a very common obstetric technique. However, the literature does not present a standardization regarding the technique, doses, and anesthetics used, besides there is also the controversy about the possibility that the addition of opioids to the local anesthetic causes fetal bradycardia and affects its vitality. The aim of this study was to identify the techniques and anesthetics used in the Anesthesiology Service of the Hospital Universitário of Universidade de São Paulo (USP) and determine whether the use of sufentanil associated with the local anesthetic affects Apgar scores of newborns. METHODS: The anesthesiology charts of patients submitted to CSE for labor analgesia over a 12-month period at the Hospital Universitário of USP were analyzed. The use and dose of sufentanil, the type of delivery, and Apgar scores in the 1st, 5th, and 10th minutes were recorded. RESULTS: Of the 635 CSE analyzed, 307 used sufentanil and local anesthetic (SUF Group) and 328 only local anesthetic (LA Group). One hundred and twenty-seven (20%) were done using the needle through the needle technique and the other 508 (80%) used the double puncture technique. There were no differences in the Apgar scores in the 1st, 5th, and 10th minutes between both groups. CONCLUSIONS: The use of sufentanil in the combined spinal-epidural did not change Apgar scores of the newborns.
ANALGESIA, Obstetric; ANALGESICS, Opioids; NEWBORN EVALUATION; ANESTHETIC TECHNIQUES, Regional
JUSTIFICATIVA E OBJETIVOS: O bloqueio combinado (BC) é uma técnica muito utilizada em anestesia obstétrica. Porém não há na literatura padronização com relação à técnica, doses e anestésicos utilizados, além da controvérsia sobre a possibilidade da adição do opióide ao anestésico local causar bradicardia fetal e alteração de sua vitalidade. O objetivo deste estudo foi identificar as técnicas e anestésicos utilizados no Serviço de Anestesiologia do Hospital Universitário da Universidade de São Paulo (USP) e avaliar se a utilização de sufentanil associado ao anestésico local no BC altera o Apgar dos recém-nascidos. MÉTODO: Foram analisadas as fichas de anestesia em que foram realizados BC para analgesia de parto durante 12 meses no Hospital Universitário da USP. Foram registrados o uso e dose de sufentanil, a via de parto utilizada e os escores de Apgar do 1°, 5° e 10° minutos dos recém-nascidos. RESULTADOS: Dos 635 BC avaliados, 307 utilizaram sufentanil e anestésico local (Grupo SUF) e 328, só anestésico local (Grupo AL). Cento e vinte e sete (20%) foram realizados através da técnica de agulha por dentro de agulha e os outros 508 (80%) foram realizados pela técnica de duas punções. Não foi verificada diferença entre o Apgar dos grupos estudados no 1°, 5° e 10° minutos. CONCLUSÕES: O sufentanil utilizado no bloqueio combinado não alterou o Apgar dos recém-nascidos.
ANALGESIA, Obstétrica; ANALGÉSICOS, Opióide; AVALIAÇÃO RECÉM-NASCIDO; TÉCNICAS ANESTÉSICAS, Regional
JUSTIFICATIVA Y OBJETIVOS: El bloqueo combinado (BC) es una técnica muy utilizada en anestesia obstétrica. Sin embargo no existe en la literatura una estandarización con relación a la técnica, dosis y anestésicos utilizados, además de la controversia sobre la posibilidad de la adición del opioide al anestésico local causar una bradicardia fetal y la alteración de su vitalidad. El objetivo de este estudio fue identificar las técnicas y anestésicos utilizados en el Servicio de Anestesiología del Hospital Universitario de la Universidade de São Paulo (USP) y evaluar si la utilización de sufentanil asociado al anestésico local en el BC altera el Apgar de los recién nacidos. MÉTODO: Se analizaron las fichas de anestesia en que se realizaron BC para la analgesia de parto durante 12 meses en el Hospital Universitario de la USP. Se registraron el uso y la dosis de sufentanil, la vía de parto utilizada y las puntuaciones de Apgar del 1°, 5° y 10° minutos de los recién nacidos. RESULTADOS: De los 635 BC evaluados, 307 utilizaron sufentanil y anestésico local (Grupo SUF) y 328, solo anestésico local (Grupo AL). Ciento veinte y siete (20%) fueron realizados a través de la técnica de aguja por dentro de aguja y los otros 508 (80%) realizados por la técnica dos punciones. No se verificó diferencia entre el Apgar de los grupos estudiados en el 1°, 5° y 10° minutos. CONCLUSIONES: El sufentanil utilizado en el bloqueo combinado no alteró el Apgar de los recién nacidos.
SCIENTIFIC ARTICLE
Combined spinal-epidural for labor analgesia: does the addition of sufentanil to the local anesthetic influence apgar scores of the newborns?* * Received from Serviço de Anestesiologia do Hospital Universitário da Universidade de São Paulo (HU-USP), São Paulo, SP
Bloqueo combinado para analgesia de parto: ¿la adición de sufentanil al anestésico local influye en el apgar de los reciÿn nacidos?
Domingos Dias Cicarelli, TSA; Renata Veloso Silva, TSA; Elke Frerichs, TSA; Marcelo Lacava Pagnocca, TSA
Anestesiologista do Hospital Universitário da USP
Correspondence to Correspondence to: Dr. Domingos Dias Cicarelli Av. Piassanguaba, 2933/71 Planalto Paulista 04060-004 São Paulo, SP E-mail: dcicarelli@uol.com.br
SUMMARY
BACKGROUND AND OBJECTIVES: Combined spinal-epidural (CSE) is a very common obstetric technique. However, the literature does not present a standardization regarding the technique, doses, and anesthetics used, besides there is also the controversy about the possibility that the addition of opioids to the local anesthetic causes fetal bradycardia and affects its vitality. The aim of this study was to identify the techniques and anesthetics used in the Anesthesiology Service of the Hospital Universitário of Universidade de São Paulo (USP) and determine whether the use of sufentanil associated with the local anesthetic affects Apgar scores of newborns.
METHODS: The anesthesiology charts of patients submitted to CSE for labor analgesia over a 12-month period at the Hospital Universitário of USP were analyzed. The use and dose of sufentanil, the type of delivery, and Apgar scores in the 1st, 5th, and 10th minutes were recorded.
RESULTS: Of the 635 CSE analyzed, 307 used sufentanil and local anesthetic (SUF Group) and 328 only local anesthetic (LA Group). One hundred and twenty-seven (20%) were done using the needle through the needle technique and the other 508 (80%) used the double puncture technique. There were no differences in the Apgar scores in the 1st, 5th, and 10th minutes between both groups.
CONCLUSIONS: The use of sufentanil in the combined spinal-epidural did not change Apgar scores of the newborns.
Key Words: ANALGESIA, Obstetric: labor; ANALGESICS, Opioids: sufentanil; NEWBORN EVALUATION: Apgar; ANESTHETIC TECHNIQUES, Regional: combined.
RESUMEN
JUSTIFICATIVA Y OBJETIVOS: El bloqueo combinado (BC) es una técnica muy utilizada en anestesia obstétrica. Sin embargo no existe en la literatura una estandarización con relación a la técnica, dosis y anestésicos utilizados, además de la controversia sobre la posibilidad de la adición del opioide al anestésico local causar una bradicardia fetal y la alteración de su vitalidad. El objetivo de este estudio fue identificar las técnicas y anestésicos utilizados en el Servicio de Anestesiología del Hospital Universitario de la Universidade de São Paulo (USP) y evaluar si la utilización de sufentanil asociado al anestésico local en el BC altera el Apgar de los recién nacidos.
MÉTODO: Se analizaron las fichas de anestesia en que se realizaron BC para la analgesia de parto durante 12 meses en el Hospital Universitario de la USP. Se registraron el uso y la dosis de sufentanil, la vía de parto utilizada y las puntuaciones de Apgar del 1°, 5° y 10° minutos de los recién nacidos.
RESULTADOS: De los 635 BC evaluados, 307 utilizaron sufentanil y anestésico local (Grupo SUF) y 328, solo anestésico local (Grupo AL). Ciento veinte y siete (20%) fueron realizados a través de la técnica de aguja por dentro de aguja y los otros 508 (80%) realizados por la técnica dos punciones. No se verificó diferencia entre el Apgar de los grupos estudiados en el 1°, 5° y 10° minutos.
CONCLUSIONES: El sufentanil utilizado en el bloqueo combinado no alteró el Apgar de los recién nacidos.
INTRODUCTION
The combined spinal-epidural (CSE) is an association of two well-known anesthetic techniques: spinal anesthesia and continuous epidural anesthesia 1. It is widely accepted because it allows the use of small doses of local anesthetic associated or not with opioids in spinal anesthesia, with reduced hemodynamic effects and the guarantee of an epidural catheter in case higher levels of anesthesia are needed or the procedure lasts longer 1,2. Its indication as an anesthetic technique for labor was immediate due to the low doses of local anesthetic used and the great perineal relaxation obtained 1,3-6. Several anesthesiologists were excited with the evolution of labor with this technique when compared with the continuous epidural anesthesia 3. However, one cannot find in the literature a standardization regarding the better technique for the combined spinal-epidural, the doses, and the drugs. The techniques described vary between: two punctures in different spaces, one for the spinal anesthesia and the other for the epidural anesthesia; or the needle through needle technique, in which the epidural puncture is done first and a long spinal anesthesia needle is introduced through the epidural needle. The anesthetic is injected in the subarachnoid space, followed by the introduction of the epidural catheter 1. Regarding the anesthetic drugs used, hyperbaric bupivacaine and lidocaine, associated or not with opioids (fentanyl, sufentanil, morphine), 1 can be used. Reports in the literature on the possibility of the addition of opioids to the local anesthetic causing fetal bradycardia, changing fetal vitality, and influencing Apgar scores of the newborns are controversial 5,7,8. This is also a source of concern for pediatricians and obstetricians who question the use of sufentanil in the anesthesia and its possible repercussions on the fetus.
The objective of this study was to identify the techniques and anesthetics used by the Anesthesiology Service of the Hospital Universitário of USP and to evaluate whether the use of sufentanil associated with the local anesthetic on the combined spinal-epidural could affect Apgar scores of the newborns.
METHODS
After approval by the Ethics Committee, a retrospective, observational study was undertaken. The anesthesiology charts of patients who underwent combined spinal-epidural for labor analgesia over a 12-month period (from May, 2004 to May, 2005) at the Hospital Universitário of USP were reviewed. The following data were recorded: local anesthetic (LA) used in the spinal anesthesia and the dose, as well as the dose of sufentanil (when it was used), and the type of delivery (normal delivery, forceps, or cesarean section). Patients were divided in two groups: local anesthetic with sufentanil (SUF group) and local anesthetic (LA group). The anesthetic, associated with sufentanil or not, was chosen by the anesthesiologist responsible for the procedure according to his preference. Both groups were compared regarding weight, age, and incidence of normal delivery, cesarean section, and forceps. As per the routine of the service, episodes of hypotension were treated with a bolus of 5 mg of ephedrine whenever systolic blood pressure was 20% lower than baseline values or below 100 mmHg. Complementary analgesia after the installation of the CSE consisted of 5 mL bolus of 0.125%, 0.25%, or 0.5% bupivacaine with vasoconstrictor, according to the dilation of the uterine cervix.
The Apgar scores of the newborns in the 1st, 5th, and 10th minutes, defined by the neonatologist, were compared in both groups using the ANOVA, and a p < 0.05 was considered significant. The relative risk of the fetuses of patients who received sufentanil to have Apgar scores below 7 in the 1st, 5th, or 10th minutes was determined. The relative risk was calculated by the ratio between the incidence of an Apgar score below 7 in the newborns exposed to sufentanil and the same incidence in those that were not exposed to this drug 9. The types of delivery in each group were compared using the Chi-square test.
RESULTS
Six hundred and ninety-eight anesthesias using the CSE technique were identified, but 63 patients (9%) were excluded from the study because their charts were incomplete. Of the 635 valid anesthesias, 307 combined local anesthetic with sufentanil (SUF group) and 328 used local anesthetic alone (LA group).
Of the 635 blocks evaluated, 127 (20%) were performed using the needle through the needle technique, performing the epidural puncture with a 16G Tuohy needle using the loss of resistance technique, and spinal anesthesia with a 27G (116 mm, Whitacre) needle introduced through the epidural needle and introduction of the 18G epidural catheter. The remaining 508 CSE (80%) were done using two punctures: spinal anesthesia with a 27G (90 mm, Whitacre) needle, and the epidural puncture was done in the best space immediately above or below with a 16G needle (loss of resistance technique) and introduction of an 18G epidural catheter.
The SUF group was composed of 307 patients who received 0.5% hyperbaric bupivacaine (2.5 mg) and sufentanil (2.5 µg) for the spinal anesthesia. The LA group had 328 patients who received 0.5% hyperbaric bupivacaine (5 mg) for the spinal anesthesia.
There were no differences in age between both groups; the mean age in the LA group was 23.69 ± 5.56 years and in the SUF group it was 23.05 ± 5.68 years (t test with p = 0.15). There were no differences in weight between both groups: 69.4 ± 11.2 kg in the LA group and 71.6 ± 10.4 kg in the SUF group (t test with p = 0.42).
The relative risk (RR) of the fetus of a woman who received sufentanil to have an Apgar score in the 1st minute lower than 7 was 0.78 (95% IC, 0.60 to 1.02) when compared to the fetuses who were exposed only to the local anesthetic. The relative risk remained below 1 for the Apgar scores in the 5th minute (RR = 0.43/ 95% IC, 0.19 to 0.99) and 10th minute (RR = 0) (Table I).
There were no significant differences in Apgar scores of both groups in the 1st, 5th, and 10th minutes (Table II).
Nineteen patients (2.9%) presented persistent fetal bradycardia shortly after the CSE, and for this reason underwent cesarean sections. Among those patients, eight (2,6%) belonged to the SUF group and 11 (3.3%) to the LA group, which was not statistically significant.
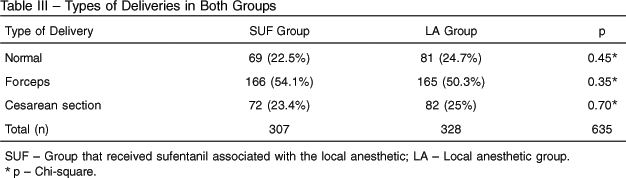
As for the type of delivery, in the SUF group there were 23.4% cesarean sections, 54.1% forceps, and 22.5% normal deliveries. In the LA group, there were 25% cesarean sections, 50.3% forceps, and 24.7% normal deliveries (Table III), without statistically significant differences by the Chi-square test.
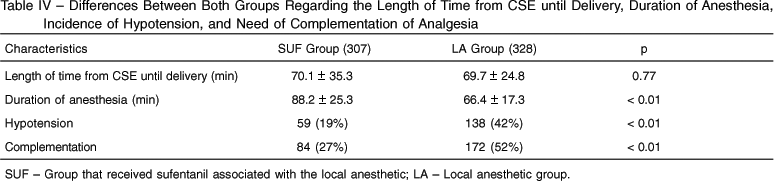
Table IV shows the length of time from the CSE until delivery, the duration of anesthesia, incidence of hypotension in both groups, and the need to complement the blockade. There were statistically significant differences between the groups regarding the duration of anesthesia after the initial dose, with greater duration in the sufentanil group, as well as a decreased incidence of hypotension and decreased need for complementation of analgesia in the sufentanil group.
Five patients (0.7%) had obstruction of the epidural catheter or failure of the complementation through the catheter; there was accidental puncture of the dura mater in six patients (0.9%), and the patients presented headache due to spinal fluid hypotension, being treated with autologous blood patch. One patient (0.16%) presented symptoms compatible with aseptic meningitis.
DISCUSSION
This study did not confirm the negative influence of sufentanil, used in the CSE, on the Apgar scores of newborns.
An Apgar score below 7 is considered an indication of perinatal asphyxia by pediatricians, explaining the reason for the choice of this value to determine fetal distress 9-11. The relative risk of the newborns exposed to sufentanil to present and Apgar score below 7 was below 1, which does not indicate an increased risk 9. This result is confirmed by Lo WK et al. 5 who also did not find differences in the evolution of the newborns after the subarachnoid administration of 10 µg of sufentanil with 2.5 mg bupivacaine when compared with 2.5 mg of bupivacaine alone. Another author 4 reported that the use of fentanyl (100 µg) in spinal anesthesia did not influence the evolution of the newborns. Vertommen et al. 12 stated that sufentanil (30 µg) used in epidural anesthesia could be detected in the plasma of up to 12% of newborns. However, those newborns did not show any changes in vitality. This is confirmed by Scherer et al. 13, who used epidural sufentanil (50 µg) and did not observe deleterious effects on the newborns.
Fetal bradycardia observed immediately after analgesia is frequently attributed to the use of opioids associated with the local anesthetic on CSE. There are several explanations: the fast relief of pain would decrease the concentration of plasma epinephrine 14, the patient would lose the tocolytic effect of epinephrine, developing uterine hypertonia and fetal bradycardia 8,14-16. D'Angelo et al. 17 stated that the use of sufentanil is not associated with uterine hypertonia and that the fetal bradycardia would be secondary to maternal hypotension after the CSE. In our study, the prevalence of persistent fetal bradycardia, which was an indication for cesarean section, was of 2.9%, while in the literature it varies from 5 to 8% 18. Although we did not observe a relationship between fetal bradycardia and the use of sufentanil, the number of patients with bradycardia in the SUF group (2.6%) was smaller than in the LA group (3.3%). This observation would support the affirmation of D'Angelo et al. as the most likely explanation for this effect.
The results demonstrated that CSE lasts longer when sufentanil is associated with the local anesthetic. This is corroborated by the studies of other authors, although the duration in our study was longer 19. The group who underwent CSE only with local anesthetic also had greater prevalence of hypotension as soon as the block was performed, because the dose of local anesthetic in this group was greater than in the sufentanil group. Apparently, this did not influence Apgar scores, since hypotension was promptly corrected with ephedrine. The need for more complementation in the local anesthetic group is compatible with the shorter duration of the anesthesia observed in this group. The influence of this fact on Apgar scores of the newborns also seems unlikely, since the duration of anesthesia in the LA group was very close to the duration of the CSE until delivery. Therefore, despite the need for complementation being greater in the LA group, it happened close to delivery or shortly after, being treated with low doses of bupivacaine (5 mL of 0.5% with vasoconstrictor).
The length of time from the beginning of CSE and the delivery was similar in both groups. This indicates that sufentanil is not responsible for a faster evolution of labor. There are controversial results in the literature that always compare CSE with the classic epidural anesthesia. Tsen et al. 3 demonstrated a faster evolution of labor in the CSE group, which was not confirmed by Norris et al. 18.
There were no differences between the techniques used for CSE regarding side effects. The prevalence of postpuncture headache was 0.9%, which is comparable with the results of other authors, who reported prevalence varying from 0.82% 1 to 2.3% 4. We observed one case of aseptic meningitis among 635 patients evaluated (0.16%). Wee et al. 20 reported two cases among 1,500 patients evaluated (0.13%). Some authors reported an increase in the number of aseptic meningitis after the introduction of the CSE technique 1,12,20-22, especially when the needle through the needle technique is used. In the case observed in the present study, the needle through the needle technique was used for the CSE.
We can make a few critics about this study. Since it was a retrospective study, there might have been a lack of uniformity of the data recorded. Besides, every time a pediatrician reanimated a newborn with a low Apgar score in the first minute, he asked about the presence of opioids in the anesthesia. Therefore, there might have been a bias, i.e., pediatricians may have assigned lower Apgar scores to newborns whose mothers received sufentanil. However, this bias would tend to decrease the classification of the vitality of newborns exposed to sufentanil, leading to the conclusion of a negative influence of sufentanil on Apgar scores of newborns. This bias can be ignored, since that was not the conclusion of this study. Nonetheless, the presence of confounding variables is possible, i.e., other factor could have lowered Apgar scores. Any pregnant woman whose fetus presented altered vitality interpreted as fetal distress after the CSE underwent a cesarean section. Since the incidence of cesarean sections was similar in both groups, it was possible to conclude that the possibility of any confounding variable having jeopardized the results was minimal.
According to the results, it was not possible to attribute to sufentanil any changes in the Apgar scores of newborns, as well as any influence in fetal bradycardia observed after the CSE.
REFERENCES
Submitted em 6 de julho de 2006
Accepted para publicação em 21 de fevereiro de 2007
- 01. Cook TM Combined spinal-epidural techniques. Anaesthesia, 2000;55:42-64.
- 02. Shaw IC, Birks RJS A case of extensive block with the combined spinal-epidural technique during labour. Anaesthesia, 2001; 56:346-349.
- 03. Tsen LC, Thue B, Datta S et al. Is combined spinal-epidural analgesia associated with more rapid cervical dilation in nulliparous patients when compared with conventional epidural analgesia? Anesthesiology, 1999;91:920-925.
- 04. Collis RE, Baxandall ML, Srikantharajah ID et al. Combined spinal epidural (CSE) analgesia: technique, management and outcome of 300 mothers. Int J Obst Anesth, 1994;3:75-81.
- 05. Lo WK, Chong JL, Chen LH Combined spinal epidural for labour analgesia duration, efficacy and side effects of adding sufentanil or fentanyl to bupivacaine intrathecally vs plain bupivacaine. Singapore Med J, 1999;40:639-643.
- 06. Abouleish A, Abouleish E, Camann W Combined spinal-epidural analgesia in advanced labour. Can J Anaesth, 1994; 41:575-578.
- 07. Westmore MD Epidural opioids in obstetrics a review. Anaesth Intens Care, 1990;18:292-300.
- 08. Clarke VT, Smiley RM, Finster M Uterine hyperactivity after intrathecal injection of fentanyl for analgesia during labor: a cause of fetal bradycardia? Anesthesiology, 1994;81:1083.
- 09. Greenberg RS, Daniels SR, Flanders WD et al. Medical Epidemiology, 3rd Ed, Nova York, McGraw-Hill, 2001;146-162.
- 10. Van De Velde M, Vercauteren M, Vandermeersch E Fetal heart rate abnormalities after regional analgesia for labor pain: the effect of intrathecal opioids. Reg Anesth Pain Med, 2001;26:257-262.
- 11. Nageotte MP, Larson D, Rumney PJ et al. Epidural analgesia compared with combined spinal-epidural analgesia during labor in nulliparous women. N Engl J Med, 1997;337:1715-1719.
- 12. Vertommen JD Opioids in combination with local anesthetics for epidural analgesia during labor. Eur J Obstetr Gynaecol Reprod Biol, 1995;(Suppl 59):S35-S38.
- 13. Scherer R, Holzgreve W Influence of epidural analgesia on fetal and neonatal well-being. Eur J Obstetr Gynaecol Reprod Biol, 1995;(Suppl 59):S17-S29.
- 14. Wong CA, Scavone BM, Peaceman AM et al. The risk of cesarean delivery with neuraxial analgesia given early versus late in labor. N Engl J Med, 2005;352:655-665.
- 15. Van De Velde M, Teukens A, Vandermeersch E Regional anesthesia and analgesia for labor and delivery. N Engl J Med, 2003;348:1818-1820.
- 16. Eisenach JC Combined spinal-epidural analgesia in obstetrics. Anesthesiology, 1999;91:299-302.
- 17. D'Angelo R, Eisenach JC Severe maternal hypotension and fetal bradycardia after a combined spinal epidural anesthetic. Anesthesiology, 1997;87:166-168.
- 18. Norris MC, Fogel ST, Conway-Long C Combined spinal-epidural versus epidural labor analgesia. Anesthesiology, 2001; 95:913-920.
- 19. Yamaguchi ET, Carvalho JC, Fonseca US et al. Sufentanil subaracnóideo associado à bupivacaína hiperbárica para analgesia de parto: é possível reduzir a dose do opióide? Rev Bras Anestesiol, 2004; 54:145-152.
- 20. Wee M, Morgan BM, Collis RE et al. Meningitis after combined spinal-extradural anaesthesia in obstetrics. Br J Anaesth, 1995; 74:351-353.
- 21. Oliveira AS, Côrtes CAF Meningite após técnica combinada para analgesia de parto. Relato de caso. Rev Bras Anestesiol, 2002; 52:515-516.
- 22. Harding SA, Collis RE, Morgan BM Meningitis after combined spinal-extradural anaesthesia in obstetrics. Br J Anaesth, 1994; 73:545-547.
Publication Dates
-
Publication in this collection
10 May 2007 -
Date of issue
June 2007
History
-
Accepted
21 Feb 2007 -
Received
06 July 2006