Abstracts
BACKGROUND AND OBJECTIVES: The relative density of a local anesthetic in relation to that of the cerebrospinal fluid (CSF) at 37° C is one of the most important physical properties that affect the level of analgesia obtained after the subarachnoid administration of the drug. The objective of this study was to determine the density of local anesthetic solutions, with and without glucose, and the combination of the local anesthetic with adjuvants at 20° C, 25° C, and 37° C. METHODS: The density (g.mL-1) was determined by using a DMA 450 densimeter with a sensitivity of ± 0.00001 g.mL-1. The densities, and variations, according to the temperature were obtained for all local anesthetics and their combination with opioids at 20ºC, 25°C, and 37°C. The solution is hyperbaric if its density exceeds 1.00099, hypobaric when its density is lower than 1.00019, and isobaric when its density is greater than 1.00019 and lower than 1.00099. RESULTS: The densities of both local anesthetics and adjuvants decrease with the increase in temperature. At 37° C, all glucose-containing solutions are hyperbaric. In the absence of glucose, all solutions are hypobaric. At 37°C, morphine, fentanyl, sufentanil, and clonidine are hypobaric. CONCLUSIONS: The densities of local anesthetics and adjuvants decrease with the increase in temperature and increase when glucose is added. The knowledge of the relative density helps select the most adequate local anesthetic to be administered in the subarachnoid space.
ANALGESICS; ANALGESICS; ANALGESICS; ANESTHETICS, Local; ANESTHETICS, Local; ANESTHETICS, Local; PHARMACOLOGY
JUSTIFICATIVA E OBJETIVOS: Uma das mais importantes propriedades físicas que afetam o nível da analgesia obtida após a injeção subaracnoidea de um anestésico local é sua densidade relativa à densidade do líquido cefalorraquidiano (LCR) a 37°C. O objetivo deste trabalho foi determinar a densidade das soluções de anestésicos locais com e sem glicose e a combinação de anestésico local com adjuvantes a 20°C, 25°C e 37°C em avaliação laboratorial. MÉTODO: A densidade (g.mL-1) foi medida como auxílio de um densímetro DMA 450, sensível a ± 0,00001 g.mL-1. A densidade e suas variações com a temperatura foram obtidas de todos os anestésicos locais e suas combinações com opioides a 20°C, 25°C e 37°C. A solução é hiperbárica se sua densidade excede a 1,00099, a solução é hipobárica quando a densidade está abaixo de 1,00019 e é isobárica quando a densidade é maior que 1,00019 e menor que 1,00099. RESULTADOS: Ambos, anestésicos locais e adjuvantes, exibem diminuição da densidade quando se aumenta a temperatura. A 37°C, todas as soluções que contêm glicose são hiperbáricas. Na ausência de glicose, todas as soluções são hipobáricas. A 37°C, morfina, fentanil, sufentanil e clonidina são hipobáricas. CONCLUSÕES: A densidade dos anestésicos locais e adjuvantes diminui com o aumento da temperatura e aumenta com a adição de glicose. O conhecimento da baricidade, densidade relativa, ajuda na seleção do anestésico local mais adequado e dos adjuvantes para uso subaracnoideo.
ANALGÉSICOS; ANALGÉSICOS; ANALGÉSICOS; ANALGÉSICOS; ANESTÉSICOS, Local; ANESTÉSICOS, Local; ANESTÉSICOS, Local; FARMACOLOGIA
JUSTIFICATIVA Y OBJETIVOS: Una de las más importantes propiedades físicas que afectan el nivel de la analgesia obtenida después de la inyección subaracnoidea de un anestésico local, es su densidad relativa a la densidad del líquido cefalorraquídeo (LCR) a 37°C. El objetivo de este trabajo fue determinar la densidad de las soluciones de anestésicos locales con y sin glucosa y la combinación de anestésico local con adyuvantes a 20ºC, 25ºC y 37ºC en evaluación laboratorial. MÉTODO: La densidad (g.mL-1) se midió con la ayuda de un densímetro DMA 450 sensible a ± 0.00001 g.mL-1. La densidad y sus variaciones con la temperatura se obtuvieron de todos los anestésicos locales y de sus combinaciones con opioides a 20°C, 25°C y 37°C. La solución es hiperbárica si su densidad excede a 1.00099, la solución es hipobárica cuando la densidad está por debajo de 1,00019 y es isobárica cuando la densidad es mayor que 0,00019 y menor que 1,00099. RESULTADOS: Ambos anestésicos locales y los adyuvantes, arrojan una reducción de la densidad cuando se aumenta la temperatura. A 37°C, todas las soluciones que contienen glucosa son hiperbáricas. Con la falta de glucosa, todas las soluciones son hipobáricas. A 37°C, morfina, fentanil, sufentanil y clonidina son hipobáricas. CONCLUSIONES: La densidad de los anestésicos locales y adyuvantes se reduce con el aumento de la temperatura y aumenta con la adición de glucosa. El conocimiento de la baricidad, densidad relativa, ayuda a la selección del anestésico local más adecuado y de los adyuvantes para uso subaracnoideo.
SCIENTIFIC ARTICLE
Assessment of the densities of local anesthetics and their combination with adjuvants. An experimental study*
Evaluación de la densidad de los anestésicos locales y de la combinación con adyuvantes. Estudio en laboratorio
Luiz Eduardo Imbelloni, TSAI; Adriano Dias MoreiraII; Flávia Cunha GasparIII; Marildo A. Gouveia, TSAIV; José Antônio CordeiroV
IDiretor do Instituto de Anestesia Regional, Hospital de Base, São José do Rio Preto, SP
IIFarmacêutico Industrial; Supervisor de Controle de Qualidade, Cristália Produtos Químicos e Farmacêuticos Ltda
IIIFarmacêutica Industrial; Analista de Controle de Qualidade Pleno, Cristália Produtos Químicos e Farmacêuticos Ltda
IVDiretor do Instituto de Anestesia Regional, Hospital de Base, São José do Rio Preto, SP
VProfessor Livre Docente em Probabilidade e Estatística da FAMERP
Correspondence to
SUMMARY
BACKGROUND AND OBJECTIVES: The relative density of a local anesthetic in relation to that of the cerebrospinal fluid (CSF) at 37° C is one of the most important physical properties that affect the level of analgesia obtained after the subarachnoid administration of the drug. The objective of this study was to determine the density of local anesthetic solutions, with and without glucose, and the combination of the local anesthetic with adjuvants at 20° C, 25° C, and 37° C.
METHODS: The density (g.mL-1) was determined by using a DMA 450 densimeter with a sensitivity of ± 0.00001 g.mL-1. The densities, and variations, according to the temperature were obtained for all local anesthetics and their combination with opioids at 20ºC, 25°C, and 37°C. The solution is hyperbaric if its density exceeds 1.00099, hypobaric when its density is lower than 1.00019, and isobaric when its density is greater than 1.00019 and lower than 1.00099.
RESULTS: The densities of both local anesthetics and adjuvants decrease with the increase in temperature. At 37° C, all glucose-containing solutions are hyperbaric. In the absence of glucose, all solutions are hypobaric. At 37°C, morphine, fentanyl, sufentanil, and clonidine are hypobaric.
CONCLUSIONS: The densities of local anesthetics and adjuvants decrease with the increase in temperature and increase when glucose is added. The knowledge of the relative density helps select the most adequate local anesthetic to be administered in the subarachnoid space.
Key Words: ANALGESICS: clonidine, fentanyl, morphine, sufentanil; ANESTHETICS, Local: bupivacaine, 50% enantiomeric excess bupivacaine, lidocaine; PHARMACOLOGY: density
RESUMEN
JUSTIFICATIVA Y OBJETIVOS: Una de las más importantes propiedades físicas que afectan el nivel de la analgesia obtenida después de la inyección subaracnoidea de un anestésico local, es su densidad relativa a la densidad del líquido cefalorraquídeo (LCR) a 37°C. El objetivo de este trabajo fue determinar la densidad de las soluciones de anestésicos locales con y sin glucosa y la combinación de anestésico local con adyuvantes a 20ºC, 25ºC y 37ºC en evaluación laboratorial.
MÉTODO: La densidad (g.mL-1) se midió con la ayuda de un densímetro DMA 450 sensible a ± 0.00001 g.mL-1. La densidad y sus variaciones con la temperatura se obtuvieron de todos los anestésicos locales y de sus combinaciones con opioides a 20°C, 25°C y 37°C. La solución es hiperbárica si su densidad excede a 1.00099, la solución es hipobárica cuando la densidad está por debajo de 1,00019 y es isobárica cuando la densidad es mayor que 0,00019 y menor que 1,00099.
RESULTADOS: Ambos anestésicos locales y los adyuvantes, arrojan una reducción de la densidad cuando se aumenta la temperatura. A 37°C, todas las soluciones que contienen glucosa son hiperbáricas. Con la falta de glucosa, todas las soluciones son hipobáricas. A 37°C, morfina, fentanil, sufentanil y clonidina son hipobáricas.
CONCLUSIONES: La densidad de los anestésicos locales y adyuvantes se reduce con el aumento de la temperatura y aumenta con la adición de glucosa. El conocimiento de la baricidad, densidad relativa, ayuda a la selección del anestésico local más adecuado y de los adyuvantes para uso subaracnoideo.
INTRODUCTION
The relationship between the density of the local anesthetic and the cerebrospinal fluid (CSF), known as baricity, is one of the most important determinants of the distribution of the local anesthetic in the leptomeningeal space 1-3. The baricity of the solution being injected and the position of the patient are the primary determinants of the dispersion of the drug 3,4. Several expressions (specific mass, absolute density, specific weight, relative density) are frequently used to describe the characteristics of the solutions used in spinal blocks. The absolute density of a solution is determined by the coefficient between the mass of the solution and the volume it occupies. Baricity is the relative density of local anesthetics when compared to that of the CSF. The temperature of the CSF is always the same as the human body (± 37°C) while the temperature of most solutions administered in the leptomeningeal space is almost always that of the operating room (± 20°C). The temperature of the local anesthetic reaches an equilibrium with the body temperature (37°C), very quickly, before being fixed at the nerve roots. To determine the baricity and predict the dispersion of local anesthetics, the density of the CSF and that of the local anesthetic should be measured at 37-38°C. Several authors define the density of the solution that exceeds 95% 5 or 99% 6 of the confidence limit for the CSF as hyperbaric, and the density below this limit as hypobaric. Opioids and other drugs are frequently used as adjuvants in spinal blocks; however, their densities are not reported in several situations. The first report on the measurement of the specific density of the CSF at 37°C used a specific scale 5. In 2000, the density and baricity of the mixtures used in spinal blocks were determined for the first time in Brazil using the straight-line equation 7. The density of any drug in a solution cannot be determined by a simple formula or physicochemical table since the density depends on the physical status of the solution. The objective of this experimental study was to determine the density of the solutions injected in the leptomeningeal space at 20°C, 25°C, and in clinical conditions, at 37°C, and in association with adjuvants, using a latest-generation densimeter.
METHODS
The densities of all drugs tested were determined at 20°C, 25°C, and 37°C using a densimeter (Anton Paar DMA 4500; Paar Scientific, Ltd., London, UK).
Isobaric solutions of 2% lidocaine (Batch #08042620), 0.5% bupivacaine (Batch #07129084), and enantiomeric excess (S75:R25) 0.5% bupivacaine (Batch #08031702); solutions containing 8% glucose and 1.5% lidocaine (Batch #094/03), 2% lidocaine (Batch #095/03), 0.5% bupivacaine (Batch #0803208), and enantiomeric excess (S75:R25) 0.5% bupivacaine (Batch #07064070); hypobaric solutions of 0.6% lidocaine (Batch #08042620), 0.15% bupivacaine (Batch #07129084), and enantiomeric excess (S75:R25) 0.15% bupivacaine (Batch #08031702); and the following adjuvants: morphine, fentanyl, sufentanil, and clonidine. The mixtures of hyperbaric 1.5% and 2% lidocaine with isobaric 2% lidocaine, hyperbaric 0.5% bupivacaine and hyperbaric enantiomeric excess (S75:R25) 0.5% bupivacaine with the isobaric solutions of the same substances at a proportion of 50% of each one were also evaluated. The first part of the study was designed to measure the density of the drugs removed from a batch at 20°C, 25°C, and 37°C. For each drug, several vials were mixed to complete a volume of 20 mL, and the median density was obtained from the measurement of three samples.
The second part of the study was designed to determine the effects of adding morphine (100 µg) (Batch #08042484), fentanyl (20 µg) (Batch #08042635), sufentanil (5 µg) (Batch #07107202), and clonidine (30 µg) (Batch #08032077 to the anesthetics described above at 37°C, and comparing them with the density of the solutions in the same temperature. In a 20 mL syringe, local anesthetics were mixed with the adjuvants, at proportions that correspond to the clinical values determined for each adjuvant. The median density was obtained by measuring three samples.
Baricity was determined by the relationship between the density of the anesthetic or anesthetic with adjuvant at 37°C and the density of CSF, based on a recently published study 8. Solutions were considered hypobaric when the density was below the lower limit of the confidence interval (1.000019) of the CSF density. The solution was considered hyperbaric when its density exceeded the 99% confidence limit of the CSF density (1.000099). Solutions were considered isobaric when their densities varied between 1.000019 and 1.000099.
The density of all solutions tested was expressed as median and interquartile, and comparisons were made with the Mood test for medians, since some tests between non-variable measurements do not allow the use of analysis of variance. A p < 0.05 indicates a significant difference.
RESULTS
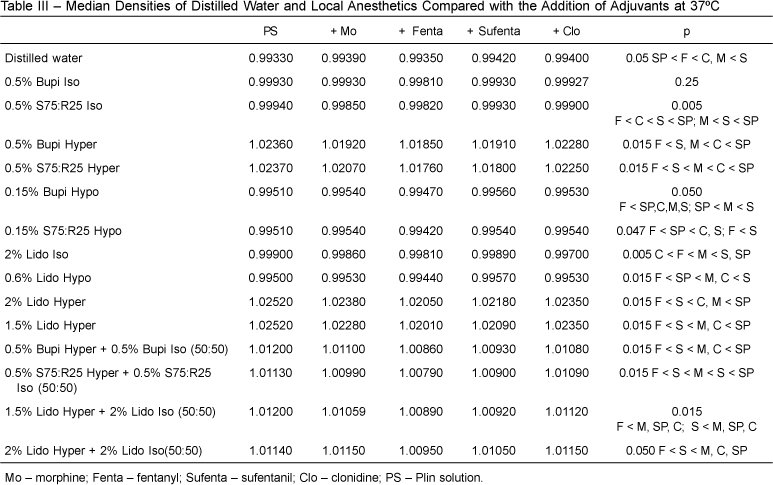
Table I shows the median of the density and standard deviation for water, local anesthetics, and adjuvants at 20°C, 25°C, and 37°C. Water density decreases significantly with the increase in temperature (p = 0.011, δ20°C > δ25°C > δ37°C). The median density of each local anesthetic and adjuvant was significantly different at the different temperatures: the highest value corresponded to 20°C (δ20°C > δ25°C > δ37°C). The density of the adjuvant (morphine, fentanyl, sufentanil, and clonidine) decreased with the increase in temperature (δ20°C > δ25°C > δ37°C).
Increasing the temperature decreased the densities of the hyperbaric solutions, but they remained hyperbaric in all temperatures (Table I). The density of the solutions prepared as "isobaric" (2% lidocaine, 0.5% bupivacaine, and 50% enantiomeric excess 0.5% bupivacaine) fell below the lower limit of the CSF. Those solutions are hypobaric in all measurements (Table I and Figure 1). The increase in temperature reduced significantly the density of the hypobaric solutions, but they all remained hypobaric (Table I).
Table II shows the densities of the anesthetics and their mixture with the adjuvants, at 37°C and the doses commonly used in the institution where the study was conducted. At 37°C, all hypobaric anesthetics are hypobaric. The addition of any adjuvant to hypobaric anesthetics reduced the density of those solutions significantly, maintaining them hypobaric. At 37°C, all isobaric anesthetics are hypobaric. The addition of any adjuvant to isobaric anesthetics reduced even more the density of those solutions, making them hypobaric. At 37°C, all hyperbaric anesthetics are hyperbaric. The addition of any adjuvant to hyperbaric solutions reduced their densities, but they all remained hyperbaric.
Table III compares pure local anesthetics with the addition of adjuvants at the usual doses at 37°C, showing that this addition reduced significantly the density of all solutions.
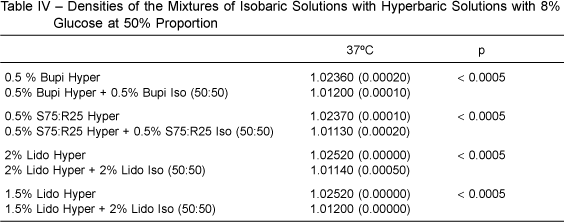
Table IV shows the results of the mixture of isobaric solutions (2% lidocaine, 0.5% bupivacaine, and 50% enantiomeric excess 0.5% bupivacaine) with hyperbaric solutions (1.5% lidocaine, 2% lidocaine, 0.5% bupivacaine, and 50% enantiomeric excess 0.5% bupivacaine, all of them with 8% glucose) at a 50% proportion, showing that the reduction of glucose to 4% corresponded to a significant reduction in density (p < 0.0005), but maintaining the final solution hyperbaric. The addition of adjuvants to the mixture of isobaric with hyperbaric solutions at the proportion of 50% reduced significantly the density of the mixtures, which still remained hyperbaric (Table III).
Figure 1 shows the densities of all medications studied and their hypo- and hyperbaricity.
DISCUSSION
This study measured the densities of the different local anesthetics and adjuvants frequently used in spinal anesthesia, in different temperatures, using a latest generation densimeter (DMA4500) that uses mechanical oscillation resonance, and has a precision of ± 0.00001 g.mL-1 (range of 0 to 3 g.mL-1). The DMA 4500 is calibrated daily with dry air and distilled water. The increasing of temperature from 20°C to 25°C or 37°C led to a significant reduction in the density of all solutions used. Many of the substances studied are isobaric at the temperature of the operating room, but when they are evaluated at 37°C or when they are warmed up by the encounter with the CSF they become hypobaric.
The baricity and temperature of local anesthetics are important factors that affect the cranial dispersion of spinal anesthesia 1-3. The CSF is an isotonic aqueous fluid similar in constitution to the interstitial fluid. The terms density, specific gravity, and baricity define physical characteristics, but they are frequently used in a less than precise manner leading to confusion. Density is defined as the weight by unit of volume (g.mL-1) of a solution at a specific temperature, while the specific gravity of a solution is calculated by the density of water. The density of the human CSF is not uniform, and it can vary with age, gender, pregnancy, and several diseases; similarly, the baricity can also vary. The dynamics of the CSF flow and the pharmacokinetics of the drugs after spinal administration are not completely understood. When referring to solutions injected in the leptomeningeal space, the densities provide more precise information than baricities.
Baricity is the relationship between the density of a solution and the density of the CSF. By definition, when the baricity of a solution is 1.0000, the solution is isotonic; > 1.0000, it is hyperbaric; and if < 1.0000, it is hypobaric. Some authors 8 suggest that the solutions used in spinal blocks should be considered hypobaric when their densities are lower than the lower limit of the confidence interval of the density of the human CSF. This seems mathematically correct.
Several of the values of density, specific gravity, and baricity of local anesthetics solutions or adjuvant mentioned in the literature are done so without mentioning the temperature they were measured at, hindering comparison of the results. For this reason, densities were measured at three different temperatures (20°C, 25°C, and 37°C); baricity, however, was calculated using the density at 37°C published recently 8. The results of density assessment at the three temperatures (20°C, 25°C, and 37°C) showed a significant reduction with the increase in temperature for all local anesthetics and adjuvants studied. At the temperature of the human body (37°C), all hypobaric solutions were hypobaric when evaluated by the densimeter. However, the body temperature changed the density of all isobaric solutions, turning them into hypobaric solutions, similar to the results of another study 9. The body temperature did not change the density of the hyperbaric solutions. The addition if adjuvants changed significantly the densities of all solutions studied.
The results of the density of local anesthetics and their mixtures were consistent with the pycnometer 1, straight-line equation 7, and oscillometric method 9. However, the results of the present study differ somewhat from another study 10 that determined the density of local anesthetics and adjuvants using similar methodology. The densimeter used in that study 10 had a precision of 0.0001 g.mL-1, which is not adequate for small differences in the density of different solutions. For this reason, in the present study, a densimeter with a precision of 0.00001 g.mL-1 was used. All pure solutions of lidocaine, bupivacaine and enantiomeric excess (S75:R25) bupivacaine are hypobaric at 37°C. Those results are in agreement with those of studies with bupivacaine and ropivacaine using the same technique and with the same degree of precision 8,9,11. All hypobaric solutions (0.6% lidocaine, 0.15% bupivacaine, and 50% enantiomeric excess 0.15% bupivacaine) are hypobaric at body temperature.
Adjuvants are frequently added to local anesthetics to improve anesthesia and prolong postoperative analgesia. Opioids (morphine, fentanyl, and sufentanil) and clonidine showed to be hypobaric at 37°C and, when added to local anesthetics, they reduce the density of the new solution, making it more hypobaric, according to another study 12, but it does not seem to have any effect on clinical practice 10,13 suggesting that the change in density is very small. Although the change in density is minimal and clinically unimportant, a reduction in density of 0.0006 g.mL-1 can influence the dispersion of the local anesthetic 2.
When mixing equal parts of the isobaric solution of 2% lidocaine with the hyperbaric solution of 1.5% or 2% (8% glucose) lidocaine, the concentration of glucose decreased to 4%, changing significantly the density of the new mixture, but still maintaining it hyperbaric. The same happened when isobaric solutions of 0.5% bupivacaine and enantiomeric excess 0.5% bupivacaine were mixed with their corresponding hyperbaric solutions (8% glucose). Thus, it is possible to decrease the amount of glucose by 50% while the solutions remain hyperbaric.
The DMA4500/5000 is the first densimeter to use the U-tube oscillation method and measure densities over a wide range of viscosities and temperature. Since the density of the CSF does not show wide variation in clinical conditions, it is possible to predict with precision the baricity of local anesthetics and adjuvants in relation to the CSF. This experimental study showed that all hyperbaric or hypobaric local anesthetics are changed by the addition of adjuvants; however, they remain hyper- or hypobaric. The results demonstrated that some solutions commonly called isobaric are, in reality, hypobaric.
REFERENCES
- 01. Horlocker TT, Wedel DJ - Density, specific gravity, and baricity of spinal anesthetic solutions at body temperature. Anesth Analg, 1993;76:1015-1018.
- 02. Stienstra R, Gielen M, Kroon JW et al. - The influence of temperature and speed of injection on the distribution of a solution containing bupivacaine and methylene blue in a spinal canal model. Reg Anesth, 1990;15:6-11.
- 03. Greene NM - Distribution of local anesthetic solutions within the subarachnoid space. Anesth Analg 1985;64:715-730.
- 04. Stienstra R, Greene NM - Factors affecting the subarachnoid spread of local anesthetic solutions. Reg Anesth, 1991;16:1-6.
- 05. Levin E, Muravchick S, Gold MI - Density of normal human cerebrospinal fluid and tetracaine solutions. Anesth Analg, 1981;60:814-817.
- 06. Davies H, King WR - Densities of cerebrospinal fluid of human beings. Anesthesiology, 1954;15:666-672.
- 07. Cangiani LM - Determinação da densidade e da baricidade das misturas para anestesia subaracnóidea. Rev Bras Anestesiol, 2000;50:92-94.
- 08. Lui ACP, Polis TZ, Cicutti NJ - Densities of cerebrospinal fluid and spinal anaesthetic solutions in surgical patients at body temperature. Can J Anaesth, 1998;45:297-303.
- 09. Richardson MG, Wissler RN - Densities of dextrose-free intrathecal local anesthetics, opioids, and combinations measured at 37°C. Anesth Analg, 1997;84:95-99.
- 10. Nicol ME, Holdcroft A - Density of intrathecal agents. Br J Anaesth, 1992;68:60-63.
- 11. Hallworth SP, Fernando R, Stocks GM - Predicting the density of bupivacaine and bupivacaine-opioid combinations. Anesth Analg, 2002;94:1621-1624.
- 12. Parlow JL, Money P, Chan PS et al. - Addition of opioids alters the density and spread of intrathecal local anesthetics? An in vitro study. Can J Anaesth, 1999;46:66-70.
- 13. Patterson L, Avery N, Chan P et al. - The addition of fentanyl does not alter the extent of spread of intrathecal isobaric bupivacaine in clinical practice. Can J Anaesth, 2001;48:768-772.
Publication Dates
-
Publication in this collection
17 Apr 2009 -
Date of issue
Apr 2009
History
-
Received
22 Sept 2008 -
Accepted
24 Nov 2008