Abstracts
BACKGROUND AND OBJECTIVES: The study of the vasomotor effect of local anesthetics (LA) is of paramount importance for the analysis of the occurrence of cardiotoxic and neurotoxic effects, and drug interactions. In order to find a safer drug than racemic bupivacaine, this study aimed to analyze digital infrared imaging of acute vasomotor effect of bupivacaine and levobupivacaine in rats intraperitoneally. METHOD: We used 30 male Wistar rats distributed into three groups (n = 10) and subjected to an intraperitoneal injection of LA. In Group C (control) 1 mL 0.9% saline was injected intraperitoneally. In Group B (bupivacaine), intraperitoneal injection of 0.5% of racemic bupivacaine (S50-R50), dose of 20 mg.kg-1 of body weight. In Group L (levobupivacaine), intraperitoneal injection of levobupivacaine 0.5% enantiomeric excess (S75-R25) in dose of 20 mg.kg-1 of body weight. The procedure was thermographicly continuously filmed from the time of pre-injection until 30 minutes after injection. The results of the recordings were analyzed in graphical form, verifying the maximum temperature of each rat and the average temperature of the system that housed the animal. RESULTS: The results of graphic analysis showed no difference between Group L and Group C, and the average temperature remained stable through-out the experiment in both groups. In Group B, there was a phenomenon of temperature increase after intraperitoneal injection of bupivacaine. CONCLUSIONS: The results demonstrated that the vasomotor effect of the acute toxicity of levobupivacaine was similar to Group C with saline, through macroscopic studies by infrared digital filmmaking, and that there were vasomotor changes (vasoconstriction), with bupivacaine intoxication in relation to both Group C and Group L.
Bupivacaine, levobupivacaine; Vasoconstriction; Thermography; Rats
JUSTIFICATIVA E OBJETIVOS: O estudo do efeito vasomotor dos anestésicos locais (AL) é de suma importância para a análise da ocorrência de efeitos cardiotóxicos, neurotóxicos e interações medicamentosas. Com a finalidade de encontrar um fármaco mais seguro do que a bupivacaína racêmica, o presente estudo teve por objetivo a análise por imagem infravermelha digital do efeito vasomotor da intoxicação aguda da bupivacaína e da levobupivacaína via intraperitoneal em ratos. MÉTODO: Utilizaram-se 30 ratos machos da linhagem Wistar, alocados em três grupos (n = 10) e submetidos a uma injeção intraperitoneal de AL. No Grupo C (Controle), foi realizada injeção intraperitoneal de soro fisiológico 0,9% 1 mL. No Grupo B (bupivacaína), injeção intraperitoneal de bupivacaína racêmica a 0,5% (R50-S50), dose de 20 mg.kg-1 de peso. No Grupo L (levobupivacaína), injeção intraperitoneal de levobupivacaína a 0,5%, excesso enantiomérico (S75-R25) em dose de 20 mg.kg-1 de peso. Procedeu-se à filmagem termográfica contínua desde o momento da pré-injeção até 30 minutos após a injeção. Os resultados das filmagens foram analisados em forma gráfica, verificando-se a temperatura máxima de cada rato e a temperatura média do sistema que abrigava o animal. RESULTADOS: Os resultados da análise gráfica revelaram que não houve diferença entre o Grupo L e o Grupo C, e a temperatura média permaneceu estável durante todo o experimento em ambos os grupos. No Grupo B, houve um fenômeno de aumento de temperatura após a injeção intraperitoneal de bupivacaína. CONCLUSÕES: Os resultados demonstraram que o efeito vasomotor da toxicidade aguda da levobupivacaína foi semelhante ao Grupo C com soro fisiológico, por meio de estudos macroscópicos por filmagem digital infravermelha, e que houve alterações vasomotoras (vasoconstrição) com a intoxicação por bupivacaína em relação ao Grupo C e em relação ao Grupo L.
ANESTÉSICO, Local; ANIMAL, Rato; DROGAS, Vasodilatadores; TÉCNICAS DE MEDIÇÃO, Temperatura
JUSTIFICATIVA Y OBJETIVOS: El estudio del efecto vasomotor de los anestésicos locales (AL), es de suma importancia para el análisis del aparecimiento de efectos cardiotóxicos, neurotóxicos e interacciones medicamentosas. Con el fin de encontrar un fármaco más seguro que la bupivacaína racémica, el presente estudio se propuso analizar por imagen infrarroja digital, el efecto vasomotor de la intoxicación aguda de la bupivacaína y de la levobupivacaína vía intraperitoneal en ratones. MÉTODO: Fueron usados 30 ratones machos de la raza Wistar, divididos en tres grupos (n = 10) y sometidos a una inyección intraperitoneal de AL. En el Grupo C (Control), fue realizada una inyección intraperitoneal de suero fisiológico al 0,9% 1 mL. En el Grupo B (bupivacaína), una inyección intraperitoneal de bupivacaína racémica al 0,5% (R50-S50), dosis de 20 mg.kg-1 de peso. En el Grupo L (levobupivacaína), una inyección intraperitoneal de levobupivacaína al 0,5%, con exceso enantiomérico (S75-R25) en dosis de 20 mg.kg-1 de peso. Después de procedió a la filmación termográfica continua desde el momento anterior a la inyección hasta 30 minutos después de ella. Los resultados de las filmaciones se analizaron de forma gráfica, verificando la temperatura máxima de cada ratón y la temperatura promedio del sistema que abrigaba al animal. RESULTADOS: Los resultados del análisis gráfico revelaron que no hubo diferencia entre el Grupo L y el Grupo C, y que la temperatu-ra promedio se mantuvo estable durante todo el experimento en los dos grupos. En el Grupo B, se produjo un fenómeno de aumento de temperatura después de la inyección intraperitoneal de bupivacaína. CONCLUSIONES: Los resultados demostraron que el efecto vasomotor de la toxicidad aguda de la levobupivacaína fue similar al Grupo C con suero fisiológico, por medio de estudios macroscópicos por filmación digital infrarroja, y que se produjeron alteraciones vasomotoras (vasoconstricción), con la intoxicación por bupivacaína respecto del Grupo C y con relación al Grupo L.
ANESTÉSICO, Local; ANIMALE, Raton; FÁRMACO, Vasodilatadores; TÉCNICAS DE MEDICION, Temperatura
SCIENTIFIC ARTICLE
Vasomotor effect after acute intoxication with bupivacaine and levobupivacaine in rats via intraperitoneal route analyzed via digital infrared imaging
Angelo Manoel Grande CarstensI; Elizabeth Milla Tambara, TSAII; Jorge Eduardo Fouto MatiasIII; Marcos Leal BrioschiIV; Daniel ColmanV; Márcio Grande CarstensVI
IAnesthesiologist Post-Graduate in Occupational Health, M.Sc. in Surgery, UFPR, Ph.D. Student, Graduate Program in Clinical Surgery, UFPR
IIChief Professor in the Department of Anesthesiology,PUC-PR,Associate Professor in the Department of Anesthesiology, UFPR; Ph.D. in Clinical Surgery, UFPR
IIIAssociate Professor, Department of Surgery, UFPR; Ph.D. in Digestive Surgery at the University of Montpellier, France
IVVisiting Professor at the Graduate Department of Surgery UFPR; Medical Researcher, Center for Pain HCFMUSP, Ph.D., Department of Mechanical Engineering UFPR; Post-doc at the Department of Neurology - Pain Center HCFMUSP
VAnesthesiologist and Medical Examiner, PhD in Clinical Surgery, UFPR
VIME3 CET / SBA do Hospital de Clínicas, UFPR
Correspondence to Correspondence to: Dr. Angelo Manoel G. Carstens Rua Coronel Menna Barreto Monclaro, 510 Centro 80420-070 - Curitiba, PR, Brazil E-mail: angelocarstens@pop.com.br
SUMMARY
BACKGROUND AND OBJECTIVES: The study of the vasomotor effect of local anesthetics (LA) is of paramount importance for the analysis of the occurrence of cardiotoxic and neurotoxic effects, and drug interactions. In order to find a safer drug than racemic bupivacaine, this study aimed to analyze digital infrared imaging of acute vasomotor effect of bupivacaine and levobupivacaine in rats intraperitoneally.
METHOD: We used 30 male Wistar rats distributed into three groups (n = 10) and subjected to an intraperitoneal injection of LA. In Group C (control) 1 mL 0.9% saline was injected intraperitoneally. In Group B (bupivacaine), intraperitoneal injection of 0.5% of racemic bupivacaine (S50-R50), dose of 20 mg.kg-1 of body weight. In Group L (levobupivacaine), intraperitoneal injection of levobupivacaine 0.5% enantiomeric excess (S75-R25) in dose of 20 mg.kg-1 of body weight. The procedure was thermographicly continuously filmed from the time of pre-injection until 30 minutes after injection. The results of the recordings were analyzed in graphical form, verifying the maximum temperature of each rat and the average temperature of the system that housed the animal.
RESULTS: The results of graphic analysis showed no difference between Group L and Group C, and the average temperature remained stable through-out the experiment in both groups. In Group B, there was a phenomenon of temperature increase after intraperitoneal injection of bupivacaine.
CONCLUSIONS: The results demonstrated that the vasomotor effect of the acute toxicity of levobupivacaine was similar to Group C with saline, through macroscopic studies by infrared digital filmmaking, and that there were vasomotor changes (vasoconstriction), with bupivacaine intoxication in relation to both Group C and Group L.
Keywords: Bupivacaine, levobupivacaine; Vasoconstriction; Thermography; Rats.
INTRODUCTION
Clinical activity of local anesthetics (LA) varies according to their structure and can be modulated by many factors, physicochemical nature, such as lipid solubility, pKa, percentage of protein binding. The application of stereoisomerism in the pharmaceutical industry enabled the emergence of local anesthetics increasingly elaborated 2.
Bupivacaine is one of the most commonly used LA in blocks, because of its potency related to lipid solubility, the sensory block of excellent quality and the absence of pronounced motor block by average concentration (0.5%). Its therapeutic index is small, because, unlike lidocaine, the differences in cardiotoxic and neurotoxic concentrations are very low, so that a cardiotoxic accident may occur without neurological prodrome, according to some reports of unexpected accidents with their clinical use 3. Bupivacaine is an equimolar mixture of two enantiomers: R (+) and S (-) bupivacaine. Starting from purified isomers of local anesthetic molecule, it is possible to manipulate the enantiomeric ratio of a racemic compound. This might contribute to their effectiveness and decrease potential toxicity, with increased therapeutic index. Levobupivacaine is constituted by enantiomeric mixture of bupivacaine (S75-R25) 0.5% 4. Levobupivacaine is less toxic if compared to bupivacaine 5-7, but there are also reports of accidents with levobupivacaine 8.
The effects of bupivacaine toxicity in peripheral vascular resistance are controversial, being reported both vasodilation and vasoconstriction. A study successfully tested the hypothesis that the use of vasodilators allows lower neurological toxicity in a rat model subjected to continuous infusion of bupivacaine 9. It is suggested that vasoconstrictors accentuate the neurotoxicity by increased plasma concentration of bupivacaine and, perhaps, vasodilators have a protective effect. Other studies were conducted with the use of vasoconstrictors 10 and demonstrated that the increased peripheral vasoconstriction significantly increases the toxicity, requiring lower doses of LA for seizures and cardiac arrest. The use of vasodilators associated with LA, as nicardipine and phentolamine resulted in a protective effect on acute venous LA 11. Several other studies were conducted with the combination of vasoconstrictors, vasodilators, intravenous or inhalational anesthetics, showing controversial results on the study of the toxicity of local anesthetic 12,13.
Interactions with other drugs are probably more common than expected and can be both beneficial and harmful. Also, the hemodynamic effects of local anesthetics are offered in four anatomical sites: direct action on cardiac or vascular smooth muscle; direct action on the autonomic nerves, evoked reflexes; direct action in the CNS 14.
Adding to the wide range of variables that can alter the vasomotor and, moreover, the possible interactions between drugs, the creation of a study model is justified in order to comparatively evaluate the vasomotor effect of the acute toxicity of bupivacaine as compared to levobupivacaine intraperitoneally in rats, using infrared thermography.
METHODS
The experimental protocol used in this study was conducted according to ethical principles of the Brazilian College of Animal Experimentation (COBEA, 1999). The experimental protocol was approved by the Ethics Committee of Hospital de Clínicas, UFPR.
We used 30 male Wistar rats (Rattus norvegicus albinos, Rodentia mammalia), aged between 120 and 153 days (mean 135.9 days). Preoperatively, a standard ration and water ad libitum are provided until 12 hours before the procedure.
The experiment was conducted at the Laboratory for Research in Postgraduate Surgical Clinic UFPR. A minimum thermal variation with ambient temperature was assumed, with environmental temperature maintained at 21ºC and relative humidity at 75%. Measurements were checked with dry and wet bulb thermo-hygrometer (Incotherm, Br). Heat loss by convection was minimized by keeping doors and windows closed and minimal movement around the animals. The airflow was controlled with a digital anemometer with rotation blades model HHF 300 A (Omega Engineering, Inc), a distance of 10 cm from the animal, keeping the velocity of air flow less than 0.2 ms-1. This is the value of transition between heat loss by free and forced convection.
The animals were distributed into three groups:
Group C - Control: intraperitoneal injection of saline 1 mL 0.9%
Group B - intraperitoneal injection of bupivacaine 0.5% at 20 mg.kg-1
Group L - intraperitoneal injection of levobupivacaine 0.5% at 20 mg.kg-1
The animals were placed in cages with five animals each. At the time of the experiment, one animal was allocated to a single cage. Once free of environmental interference with anesthesia and other drugs, thermographic continuous filming was performed at three times:
Pre-injection continuously documented by the imager;
Intraperitoneal injection of local anesthetic, according to the groups;
Observation and filming of the vasomotor effects of the drug injected intraperitoneally for a period of 30 minutes.
We used a sensor ThermoVision AGEMA550 (FLIR SystemsTM, Sweden) to capture the spectral range of electromagnetic waves emitted between 3.5 to 5 µm (waves in the middle infrared spectrum). Maximum spatial resolution obtained was 0.1 to 0.2 mm.
The infrared radiation naturally emitted by objects in the environment is captured and converted into an electrical signal through a PTSI detector, cooled with liquid nitrogen (steerling cycle). This signal is processed in a spreadsheet number of 76,000 points of absolute temperature calibrated by frame, represented instantly on the thermal image with a resolution of 320x240 pixels and thermal sensitivity greater than 0.1ºC.
The camera was positioned in a vertical stand 1m away from the animal.
The images were processed in 3.0 GHz Pentium IV computer coupled to a PCMCIA card. Through the specific program ThermaCAMTM Researcher, FLIR Systems (Sweden), the images were recorded up to seven frames per second, throughout the time of the pre-injection, followed during injection and for 30 minutes under the same environmental conditions already described. All images were represented by infrared thermograms in a video monitor and recorded on CD Rom for analysis with the software.
The images were plotted using thermal amplitude (range) between 32.9ºC and 19.2ºC, average (level) of 30ºC and colorimetric scale (palette) continuous "RAINBOW900" in which white, red, yellow, green, blue and black represented, respectively, a sliding scale of temperature areas, equally distributed in the range from warmer to colder. This was maintained until the end of the experiment.
For purposes of the thermodynamic study, the results of the filming were pre-analyzed in graphical form as follows:
1. Y axis - two variables:
The maximum temperature was captured inside the acrylic cage where the mouse was. Once the animal moved unrestrictedly in the cage, the points of highest temperature would be mapped, corresponding to the central compartment, i.e., eye and ear.
The average temperature, captured in the entire area of the experiment, that is, the mouse inside the cage. It had the purpose of demonstrating the phenomena intrinsic to the animal that were significantly influenced by this semi-closed system of heat exchange.
2. X-axis - time in minutes.
After the procedure, the animals were transferred to appropriate cages and observed until recovery. The cages were identified according to the group, being distributed five animals in each cage. Free access to water was provided, and power was restored after 12 hours. We observed the clinical status of animals on a daily basis, evaluating motor activity, behavior and any signs suggestive of disease. After the experiment, animals were sacrificed with a lethal dose of inhaled ether.
For the analysis of thermal changes, Gaussian attributes, independence and homogeneity of variances were tested.
When the previous tests met the proposed criteria, we used univariate ANOVA and Tukey's parametric test to establish the significance of the results.
In all tests, the range stood 0.05 or 5% (p < 0.05) as a rejection of the null hypothesis. To this purpose, we used the specific program Statistica 5.1.
Initially, the accuracy limits (PT) for measurements of temperature were calculated as twice the standard deviation of each set of observations of temperature T (PT = 2sT) and intrinsic sensor error (Bias limit), BT = 0.001 K for each temperature point. Thus, the limit of precision in the measurement of T was estimated using the propagation equation proposed by Kline and McClintock quoted in Kline and Steele (1953):
UT =  PT2 + BT2
PT2 + BT2
UT = temperature measurement uncertainty; PT = limit precision, BT = intrinsic sensor error (bias limit)
As BT is much smaller than PT, the term is negligible in the presence of the equation. Consequently, the uncertainties of the temperatures for each point were computed as follows:
In the graphs of the results of this study, each point of temperature is represented as T ± UT. The first law of Thermodynamics was applied to this model for a closed system in steady state.
The volume-control system was defined as the region between the central portion and periphery of the animal, that is, from the internal organs to the external skin surface. We adopted the measurement of a constant temperature and uniform heat distribution representative for this volume-control, which is the core temperature. We measured the heat loss from the mouse to the external environment, taking into account the mass and specific heat of the animal (3.8 kJ / kg.ºC), divided by total experiment time:
Q = m.c. DT / Dt
Q = heat loss [W]; m = animal mass [kg]; c = specific heat of the animal [J / (kg.ºC)]; DT = difference between the initial and final temperature [ºC]; Dt = time interval of each experiment [s].
Thus, we determined the power (or rate of heat transfer) in each group. The results of the heat loss from the central compartment were expressed in Watts (Watts), that is, heat loss (Joule) per unit time (seconds).
RESULTS
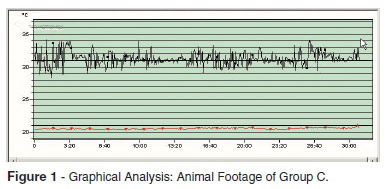
Environmental variables (temperature, humidity and air velocity) remained stable throughout the procedure and no animal died during the experiment. The average temperature analysis captured the entire area of the experiment, that is, monitoring the mouse (the object of study) in the cage (environment), and was represented by variable A in the Y axis. That plotted the filming of the thermodynamic study and served to demonstrate the phenomena intrinsic to the animal that were significantly influenced by the heat exchange system. Due to the constant movement of the animal, it was not always possible to have an orthogonal angle with the thermal imager, which recorded the highest temperature points, the eyes and ears. The graphic analysis of variable B showed an oscillating pattern of uptake of the maximum temperature in the thermogram, eye and ear (Figure 1 variable black line B Y-axis). In this study, eyes and ears were identified as points of higher temperature, corresponding to the central compartment.
GRAPHICAL REPRESENTATION OF THE THERMOGRAMS
Group C or Control Group
Y-axis: variable (A) graphically represents the average temperature of the thermal changes of the rat in the cage; variable (B) represents the maximum temperature of eyes and ears of the animal.
Group B or Group Bupivacaine
In group B, there was a temperature rise of about 1ºC, 5 minutes after the intraperitoneal injection of bupivacaine, which persisted until 18 minutes after the experiment, decreasing after 20 minutes, according to Figure 2. Differences in vasomotor response in Group B compared to C were observed.
The graphic analysis of the variable, which represents the average temperature of bupivacaine group, showed increase in average temperature in the cage (dashed red Figure 2), evidence that the use of bupivacaine altered the physiology of the animal, promoting change in temperature average of all rat-cage.
Group L or Group Levobupivacaine
The results of graphic analysis showed no difference between groups Levobupivacaine and Control. The average temperature of the cage remained stable throughout the experiment (variable A, red line on the Y axis).
Thermograms
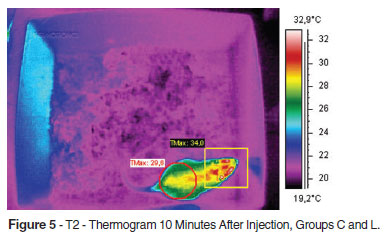
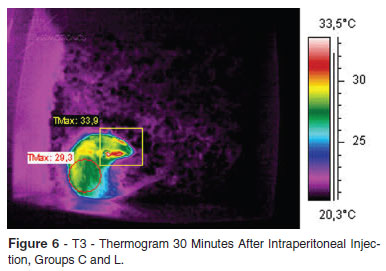
After the identification of the graphical difference, the thermograms specific for the analysis were searched in the database. Here were shown the T1 (intraperitoneal injection), T2 (10 minutes after the initial thermogram) and T3 (30 minutes after injection) in relation to groups C, B and L.
The initial thermograms T1 were homogeneous in the three groups, with no temperature change in the opening minutes in either group.
There was no difference between groups C and L at all times, as shown in Figures 4, 5 and 6.
Therefore, thermograms T1, T2 and T3 are represented only once, without reference to group C or L.
Group B was different in relation to Groups C and L in T2 and T3. The increase of temperature in the region of eyes and ears in T2 and T3 is noteworthy.
There was no statistical difference between the values found in the temperature difference between T1 and T3 in the control group and levobupivacaine.
There was statistical difference between the values found in the temperature difference between T1 and T3 in the control groups compared to levobupivacaine and bupivacaine, as the Chart 1.
The values expressed in W, that is, J/s are obtained by dividing the values from the table above for 1,800 seconds (30 minutes of experiment).
DISCUSSION
Through this model, it was found that the group intoxicated with levobupivacaine had a behavior similar to control. In the group intoxicated with bupivacaine, there was difference in results from the mean temperature of the cage+rat complex, and increase of temperature of the animals, mainly around the eyes and ears, and also the back. This phenomenon could be explained by vasoconstriction, with diversion of blood circulation from the periphery to the heart-lung-brain. It explained, thus, hyperthermia found in eye-ear region. Another plausible hypothesis is that there are muscular contractions, to the extent that there is intraperitoneal absorption of AL.
In general, studies on the effects of vasomotor symptoms of local anesthetic toxicity are controversial and are influenced by several variables. The effects of bupivacaine on the peripheral vasculature are also controversial, being described both vasodilation and vasoconstriction.
The vasodilatory effects of nicardipine and phentolamine increase the cumulative dose, but do not affect the plasma level to trigger seizures induced by bupivacaine. Bernards et al. 11 observed that rats protected with nicardipine and phentolamine required, respectively: 7.6 ± 1.5 and 8.1 ± 1.1 mg.kg-1 compared to control 5.8 ± 1.5 mg.kg-1 of bupivacaine infused continuously to provoke convulsion. At the time of seizure, brain and plasma levels were similar in terms of statistics, which support the thesis that the vasoactive phenomena interfere in the dynamics of the distribution of AL in the model of acute toxicity by intravenous infusion.
A protective effect of propofol and sevoflurane in models of acute toxicity by bupivacaine in rats was observed, suggesting that these mechanisms of protection are not sufficiently clarified and point to a central action of bupivacaine on the GABAergic system 12.
Regarding the assessment of the influence of clonidine on bupivacaine EV toxicity, there was an effect due to the antiarrhythmic properties of a2 agonist. Considering significant the acute intoxication with epidural bupivacaine with accidental intravascular injection in bolus, there is a plausible association with the use of clonidine in the block instead of opioids. The nicardipine has also shown to be protective in acute venous intoxication, having postulated a mechanism of protection by inhibition of calcium channels and reduction of interactions with bupivacaine 15.
A study of bupivacaine toxicity in pigs found that the addition of epinephrine significantly increases peripheral vasoconstriction and toxicity, being necessary the administration of a lower dose to seizures and cardiac arrest 10.
The monitoring parameters such as mean arterial pressure, electroencephalography, and electrocardiography were not part of this protocol, in order to avoid contact from the animal with the monitoring equipment that could influence the results of this research.
When a group intends to study the vasomotor state of a living being, there must be adequate preparation of the animals, ability to control the atmospheric phenomena in the laboratory, and experimental use of high precision equipment. These items have been carefully examined and checked for the establishment of experimental validation of this model.
In this study, the animals were kept in a 12-hour fasting period before the experiment, since some studies have revealed that part of the energy gained from food is released as heat 16.
After a meal, the body surface temperature may increase from 1ºC to 2ºC, reaching its peak 30 minutes after the meal 17.
The animals were kept hydrated ad libitum, as water balance is another important factor that affects surface temperature. If the body's water content is reduced, its thermal conductivity will also be reduced 18.
The temperature of the body depends on its blood flow when the body produces excessive heat during intense exercise, and rectal temperature may temporarily increase to 1.3 up to 3ºC more than the normal core temperature 19. So it was avoided that the animals increased their physical and metabolic activity before the experiment. They were picked up gently by the back to prevent escapes or higher stress before the intraperitoneal injection. In physical activity, heat production may increase 10 to 20 times, increasing muscle temperature higher than the core temperature 20. The release of hormones under stress such as adrenaline can cause cutaneous vasoconstriction and reduce skin temperature, which leads to a heterogeneity in the initial temperature in mice of the same group, if precautions are not taken 21.
Therefore, this study found that the vasomotor effect of the acute toxicity of levobupivacaine was similar to the control group with saline, through macroscopic studies by digital infrared footage.
There were vasomotor alterations with bupivacaine intoxication (vasoconstriction), more than in relation to the control group and in relation to the Levobupivacaine Group.
REFERENCES
Submitted on February 27, 2010.
Approved on October 23, 2010.
Received from Universidade Federal do Paraná (UFPR), Paraná, Brazil.
- 01. Berde CB, Strichartz GR - Local Anesthetics, em: Miller RD - Anesthesia, 5th Ed, Philadelphia, Churchil Livingstone, 2000;491-517.
- 02. Simonetti MPB, Batista RA, Ferreira FMC - Estereoisomeria: a interface da tecnologia industrial de medicamentos e da racionalização terapêutica. Rev Bras Anestesiol, 1999;48:390-399.
- 03. Simonetti MPB, Ferreira FMC - Does the d-isomer of bupivacaine contribute to the improvement of efficacy in neural block? Reg Anesth Pain Med, 1999;24(3/suppl):43.
- 04. Delfino J, Vale NB, Magalhães Filho E - Ropivacaína e levobupivacaína a 0,45% associadas a opióides em anestesia peridural para cesariana: estudo comparativo. Rev Bras Anestesiol, 1999;49:244-248.
- 05. Chang DH, Ladd LA, Wilson KA et al. - Tolerability of large-dose intravenous levobupivacaine in sheep. Anesth Analg, 2000;91:671-679.
- 06. Kopacz DJ, Allen HW, Thompson GE - A comparison of epidural levobupivacaine 0.75% with racemic bupivacaine for lower abdominal surgery. Anesth Analg, 2000;90:642-648.
- 07. Pirotta D, Sprigge J - Convulsions following axillary brachial plexus blockade with levobupivacaine. Anaesthesia, 2002;57:1187-1189.
- 08. Crews JC, Rothman TE - Seizure after levobupivacaine for interscalene brachial plexus block. Anesth Analg, 2003;96:1188-1190.
- 09. Fukuda H, Hirabayashi Y, Shimizu R et al. - Sevoflurane is equivalent to isoflurane to attenuating bupivacaine induced arrhythmias and seizures in rats. Anesth Analg, 1996;83:570-573.
- 10. Bernards CM, Carpenter RL, Rupp SM et al. - Effect of midazolam and diazepam premedication on central nervous system and cardiovascular toxicity of bupivacaine in pigs. Anesthesiology, 1989;70:318-323.
- 11. Oda Y, Funao T, Tanaka K et al. - Vasodilation increases the threshold for bupivacaine-induced convulsions in rats. Anesth Analg, 2004;98:677-682.
- 12. Ohmura S, Ohta T, Yamamoto K et al. - A comparison of the effects of propofol and sevoflurane on the systemic toxicity of intravenous bupivacaine in rats. Anesth Analg, 1999;88:155-159.
- 13. Badgwell JM, Heavner JE, Kytta J - Bupivacaine toxicity in young pigs is age-dependent and is affected by volatile anesthetics. Anesthesiology, 1990;73:297-303.
- 14. Blair MR - Cardiovascular pharmacology of local anaesthetics. Br J Anaesth, 1975;47(suppl):247-252.
- 15. Matsuda F, Kinney WW, Wright W et al. - Nicardipine reduces the cardio-respiratory toxicity of intravenously administered bupivacaine in rats. Can J Anaesth, 1990;37:920-923.
- 16. Becker DP - Ein neues Konzept der Ernährungswissenschaft: Termogenese. Bild der Wissenschaft, 1980; 12:60-65.
- 17. Zahorska-Markiewicz B - Thermic effect of food and exercise in obesity. Eur J Appl Physiol, 1980;44:231-235.
- 18. Ekblom B, Greenleaf CJ, Greenleaf JE et al. - Temperature regulation during exercise dehydration in man. Acta Physiol Scand, 1970;79:475-483.
- 19. Guyton AC - Human Physiology and Mechanism of Disease, 5th Ed, New York, Saunders, 1992;946-961.
- 20. Astrand PO, Rodahl K - Textbook of Work Physiology. New York, McGraw-Hill, 1970.
- 21. Kirsch KA - Physiology of Skin Surface Temperature, em: Engel JM, Ring EFJ - Thermological Methods. Wwinheim, Verlagsgesellschaft, 1985;1-9
Correspondence to:
Publication Dates
-
Publication in this collection
13 Apr 2011 -
Date of issue
Apr 2011
History
-
Accepted
23 Oct 2010 -
Received
27 Feb 2010