Abstracts
Background and objectives:
Few investigations have addressed the safety of oxygen from concentrators for use in anesthesia in association with nitrous oxide. This study evaluated the percent of oxygen from a concentrator in association with nitrous oxide in a semi-closed rebreathing circuit.
Methods:
Adult patients undergoing low risk surgery were randomly allocated into two groups, receiving a fresh gas flow of oxygen from concentrators (O293) or of oxygen from concentrators and nitrous oxide (O293N2O). The fraction of inspired oxygen and the percentage of oxygen from fresh gas flow were measured every 10 min. The ratio of FiO2/oxygen concentration delivered was compared at various time intervals and between the groups.
Results:
Thirty patients were studied in each group. There was no difference in oxygen from concentrators over time for both groups, but there was a significant improvement in the FiO2 (p < 0.001) for O293 group while a significant decline (p < 0.001) for O293N2O. The FiO2/oxygen ratio varied in both groups, reaching a plateau in the O293 group. Pulse oximetry did not fall below 98.5% in either group.
Conclusion:
The FiO2 in the mixture of O293 and nitrous oxide fell during the observation period although oxygen saturation was higher than 98.5% throughout the study. Concentrators can be considered a stable source of oxygen for use during short anesthetic procedures, either pure or in association with nitrous oxide at 50:50 volume.
Oxygen inhalation therapy; Nitrous oxide; Equipment and supplies; Anesthesia; Inhalation; Developing countries
Justificativa e objetivos:
poucas pesquisas abordaram a segurança do oxigênio a partir de concentradores para uso em anestesia em associação com óxido nitroso. Este estudo avaliou a porcentagem de oxigênio a partir de um concentrador em associação com óxido nitroso em um circuito de reinalação semifechado.
Métodos:
pacientes adultos submetidos à cirurgia de baixo risco foram alocados aleatoriamente em dois grupos e receberam um fluxo de gases frescos de concentradores de oxigênio (O293) ou de concentradores de oxigênio e óxido nitroso (O293N2O). A fração inspirada de oxigênio e a porcentagem do fluxo de gases frescos de oxigênio foram medidas a cada 10 minutos. O razão da concentração liberada de FiO2/oxigênio foi comparada em diferentes intervalos de tempo e entre os grupos.
Resultados:
foram avaliados em cada grupo 30 pacientes. Não houve diferença em oxigênio a partir dos concentradores ao longo do tempo para ambos os grupos, mas houve uma melhora significativa da FiO2 (p < 0,001) no grupo O293, enquanto houve uma queda significativa (p < 0,001) no grupo O293N2O. A razão FiO2/oxigênio variou em ambos os grupos e atingiu um patamar no grupo O293. A oximetria de pulso não caiu abaixo de 98,5% em ambos os grupos.
Conclusão:
a FiO2 na mistura de O293 e óxido nitroso caiu durante o período de observação, embora a saturação de oxigênio tenha ficado acima de 98,5% durante todo o estudo. Os concentradores podem ser considerados uma fonte estável de oxigênio para uso durante procedimentos anestésicos de curta duração, tanto puro quanto em associação com óxido nitroso em volume de 50:50.
Oxigenoterapia; Óxido nitroso; Equipamentos e suprimentos; Anestesia; Inalação; Países em desenvolvimento
Justificación y objetivos:
pocas investigaciones han abordado la seguridad del oxígeno a partir de concentradores para su uso en anestesia en asociación con el óxido nitroso. Este estudio evaluó el porcentaje de oxígeno de un concentrador en asociación con el óxido nitroso en un circuito de reinhalación semicerrado.
Métodos:
pacientes adultos sometidos a cirugía de bajo riesgo fueron asignados aleatoriamente en 2 grupos, para recibir un flujo de gases frescos de concentradores de oxígeno (O293) o de concentradores de oxígeno y óxido nitroso (O293NO). La fracción inspirada de oxígeno y el porcentaje del flujo de gases frescos de oxígeno fueron medidos cada 10 min. La relación concentración liberada de FiO2/oxígeno fue comparada en diferentes intervalos de tiempo y entre los grupos.
Resultados:
treinta pacientes fueron evaluados en cada grupo. No hubo diferencia en el oxígeno de los concentradores a lo largo del tiempo para ambos grupos, pero sí hubo una mejoría significativa en la FiO2 (p < 0,001) en el grupo O293, mientras que hubo una caída significativa (p < 0,001) en el grupo O293NO. La relación FiO2/oxígeno varió en ambos grupos, alcanzando una meseta en el grupo O293. La oximetría de pulso no descendió por debajo del 98,5% en ningún grupo.
Conclusión:
la FiO2 en la mezcla de O293 y óxido nitroso cayó durante el período de observación, aunque la saturación de oxígeno quedó por encima de un 98,5% durante todo el estudio. Los concentradores pueden ser considerados una fuente estable de oxígeno para uso durante procedimientos anestésicos de corta duración, tanto puro como en asociación con el óxido nitroso en volumen de 50:50.
Oxigenoterapia; Óxido nitroso; Equipos y suministros; Anestesia; Inhalación; Países en desarrollo
Introduction
The high price of oxygen can encourage the installation of oxygen concentrators.11. Friesen RM, Raber MB, Reimer DH. Oxygen concentrators: a primary oxygen supply source. Can J Anaesth. 1999;46: 1185-90. and 22. Arrowsmith LW. Oxygen concentrators for medical gas pipeline systems. J Inst Hosp Eng. 1989;43:6-8. In Brazil a previous investigation suggested that savings related to the use of a concentrator over 10 years in a non-profit hospital reached 19.5 million dollars.33. Moll JR, Moll AVS, Guttman A, et al. Usinas concentradoras de oxigênio: evolução da fração inspirada de oxigênio e repercussões no paciente anestesiado em sistema com absorvedor de CO2. Estudo piloto. Rev Bras Anestesiol. 2007;57:649-57 [Portuguese]. The oxygen from concentrators represents a cost of US$ 0.0015 mL-1. It is worth to notice the offering of this system as an oxygen source set up a perfect competition in this market in Brazil and reduced its final price nationwide.
A number of studies using oxygen concentrators with open ventilation systems concluded that the efficacy and reliability of such devices make them a suitable alternative to oxygen cylinders in the developing world.44. Carter JA, Baskett PFJ, Simpson PJ. The 'Permox' oxygen concentrator. Its mode of action, performance and potential application. Anaesthesia. 1985;40:560-5. , 55. Fenton PM. The Malawi anaesthetic machine. Experience with a new type of anaesthetic apparatus for developing countries. Anaesthesia. 1989;44:498-503. and 66. Shrestsha BM, Singh BB, Gautam MP, et al. The oxygen concentrator is a suitable alternative to oxygen cylinders in Nepal. Can J Anaesth. 2002;49:8-12.
Oxygen concentrators use zeolite molecular sieve technology, which can produce up to 95% pure O2. Briefly, room air is drawn into the oxygen concentrator through a series of filters to remove dust and bacteria. The concentrator contains two columns of the zeolite molecular sieve within a canister. The sieve adsorbs nitrogen from the air as it is forced through under pressure. The sieve allows O2 to pass through along with the 0.93% argon present in the air. Synthetic zeolite is used for the production of oxygen. The concentrator has been shown to be reliable and cost effective for providing oxygen in locations where cylinders may not always be available.77. Whitley E, Ball J. Statistics review 3: sample size calculation. Crit Care. 2002;6:335-41. However, little is known about the association of oxygen from concentrators with nitrous oxide during mechanical ventilation in anesthesia.
This study evaluated the percent of oxygen from a concentrator in association with nitrous oxide for procedures lasting at least 100 min in a semi-closed rebreathing circuit.
Methods
After approval by the Ethics Research Committee of Hospital Geral de Bonsucesso do Ministério da Saúde, city of Rio de Janeiro, adult patients undergoing low risk surgery were invited to participate, gave written informed consent and were randomly allocated into two groups, either receiving a fresh gas flow (FGF) of 1000 mL min-1 oxygen from concentrator (O293) or a FGF of 500 mL min-1 of oxygen from concentrator plus 500 mL min-1 nitrous oxide (O293NO). The only exclusion criterion was the presence of any lung disease. The oxygen used in this investigation was provided by oxygen concentrator connected to medical gas pipeline systems, providing an output pressure of 4.08-5.09 kg cm- 22. Arrowsmith LW. Oxygen concentrators for medical gas pipeline systems. J Inst Hosp Eng. 1989;43:6-8. (Eniplan Ferri - Engenheiros Associados Ferri Lt., COE 2x20 model, Rio Grande do Sul, Brazil). Nitrous oxide was provided from gas pipeline system originated from standard tanks.
Patients breathed under a mask with oxygen from concentrator at a flow of 8.0 L min-1 for 3 min in a semi-closed rebreathing circuit. Following this period, anesthesia was induced with sequential intravenous administration of Fentanyl 5.0 mcg kg-1, propofol 1.5 mg kg-1 and atracurium 0.5 mg kg-1. After tracheal intubation, a controlled mechanical ventilation system with a CO2 absorber was used to provide a tidal volume sufficient to maintain exhaled CO2 between 30 and 35 mmHg. Maintenance of anesthesia was achieved with isoflurane and the FGF of 1 L min-1. One side-stream valve (CardioCap5 - GE Datex-Ohmeda Helsink, Finlandia) between the inspiratory unidirectional valve and the Y-piece to connect with the patient allowed the inspired and expired fractions of volatile anesthetics, carbon dioxide (ETCO2), oxygen (FiO2) and nitrous oxide (FiN2O) to be monitored, and another side-stream valve (Capnomac - GE Datex-Ohmeda Helsink, Finlandia), located at the delivery port through which the gases are delivered from the machine to the systems, monitored the percentage of oxygen delivered from concentrator by means of paramagnetic oxygen analysis. The samples derived from these sidestream valves were vented to atmosphere (Fig. 1). The measurements of oxygen ratios in either group were expected to reach stability, considering the consumption of oxygen should be at least 200 mL min-1 throughout the intraoperative period.
Monitoring of gases in the respiratory system. Places for gases monitoring: CardioCap5 measured fractions of volatile anesthetics, carbon dioxide (ETCO2), oxygen (FiO2) and nitrous oxide (FiN2O), and Capnomac measured the gases delivered from the machine to the systems. The samples derived from these sidestream valves were vented to atmosphere.
The variables studied were the concentration of inspired oxygen, as well as the fraction of inspired oxygen measured every 10 min after intubation until the end of anesthesia. Arterial pressure, heart rate and pulse oximetry values were obtained at the beginning of anesthesia as well as every 10 min until the end of the procedure. The ratio of FiO2 to oxygen concentration delivered by the concentrator (O293) was compared at intervals between the groups. The results are presented as median and 25-75 percentiles or mean and standard deviation when normality was achieved. Analysis of variance for repeated measures was used to compare data from the different time intervals within groups. Student's t test was used to compare the age, weight, height and body mass index. Chi-square was used to compare the gender ratio. The hypothesis considered a previous investigation with oxygen concentrator as the sole source and where the mean FiO2 difference was considered the target difference and the use of a nomogram for calculating sample size give a sample of 25-30 patients.33. Moll JR, Moll AVS, Guttman A, et al. Usinas concentradoras de oxigênio: evolução da fração inspirada de oxigênio e repercussões no paciente anestesiado em sistema com absorvedor de CO2. Estudo piloto. Rev Bras Anestesiol. 2007;57:649-57 [Portuguese]. and 77. Whitley E, Ball J. Statistics review 3: sample size calculation. Crit Care. 2002;6:335-41. All comparisons were considered statistically significant when p < 0.05. The statistical package Sigma Stat for Windows, version 2.03, SPPS Inc. was used.
Results
Sixty patients of both sexes formed the sample, with 30 in each group, including ASA physical status one and two. There was no statistically significant difference between the two groups for gender, age, weight, height or body mass index (Table 1).
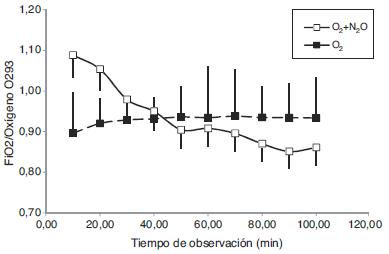
The mean values and standard deviations of delivered oxygen concentration and inspired oxygen percent were registered for both groups (Fig. 2). There was no difference in the delivered oxygen concentration from concentrator over time (Friedman repeated measures ANOVA, p = 0.084). There was a significant improvement in the FiO2 (p < 0.001), which was lower at 10 min compared to 40 min and up, and from 20 compared to 50 min and up (Tukey test, p < 0.05). There was also no difference in the delivered oxygen concentration over time for the fresh oxygen flow from concentrator in the group receiving oxygen and nitrous oxide (p = 0.153). However, FiO2 showed a significant decline, from 52% at 10 min to 40% at the end of anesthesia (p < 0.001). There was no difference within interval 10 throughout 40 min, but from 50 up to 100 min all values registered from 10 to 40 min were significantly different (Tukey, p < 0.05).
These measurements reflected in the ratio of fraction inspired and fresh oxygen flow (FiO2/oxygen flow) in both groups. In the group receiving oxygen from the concentrator (O293) the ratio improved over time (p < 0.001). In the group receiving oxygen and nitrous oxide, the FiO2/oxygen flow ratio decreased over time (p < 0.001). The inhalational anesthetics percentiles were kept at the anesthesiologist discretion and exhaled CO2 were kept within the expected margins, 32.47 ± 1.59 mmHg in the O293 group, and 32.63 ± 1.69 mmHg in the O293NO group.
Pulse oximetry was lower before the induction of anesthesia and values differed between groups (97.7 ± 1.2 vs. 99.1 ± 0.5 for O293NO and O293, respectively; p < 0.001, Student t). Following induction the hemoglobin (Hb) saturation did not fall below 98.5% in either group.
Discussion
This study showed that the FiO2 in the mixture of O293 and N2O fell from 52% to 41% during the observation period. Oxygen saturation was higher than 98.5% throughout the study and the ratio of oxygen supplied and inspired became stable over time, but was lower than one.
This investigation confirms stability of oxygen saturation showed by previous studies using oxygen from concentrators in an anesthesia circuit with a carbon dioxide absorber and a FGF of 500 mL per minute.88. Parker CJ, Snowdon SL. Predicted and measured oxygen concentrations in the circle system using low fresh gas flows with oxygen supplied by an oxygen concentrator. Br J Anaesth. 1988;61:397-402.
and
99. Friesen RM. Oxygen concentrators and the practice of anaesthesia. Can J Anaesth. 1992;39:R80-9. Although a fresh oxygen flow of up to 0.5 L min-1 may result in a significant accumulation of argon compared to higher oxygen flows of 1.0 and 2.0 L per minute,1010. Grano JT, Roberts AL, Bigley AJ. Determination of the mini-mal fresh gas flow to maintain a therapeutic inspired oxygen concentration in a semi closed anesthesia circle system using an oxygen concentrator as the oxygen source. Technical Report. Houston: Texas Univ. Health Science Center at Hous-ton. School of Nursing; 2001. HSC-SN-00-022. Available at: http://handle.dtic.mil/100.2/ADA426737
http://handle.dtic.mil/100.2/ADA426737...
this study suggests that both FiO2 and Hb saturation are stable over time when using a oxygen flow equal to or greater than 0.5 L min-1 during general anesthesia of healthy adults in semi-closed rebreathing circuit system.
The association of oxygen from a concentrator and nitrous oxide has previously been studied under low FGF and it was suggested that the fraction of argon gas was small and prone to falling between 60 and 120 min but that the nitrous oxide concentration increased.1010. Grano JT, Roberts AL, Bigley AJ. Determination of the mini-mal fresh gas flow to maintain a therapeutic inspired oxygen concentration in a semi closed anesthesia circle system using an oxygen concentrator as the oxygen source. Technical Report. Houston: Texas Univ. Health Science Center at Hous-ton. School of Nursing; 2001. HSC-SN-00-022. Available at: http://handle.dtic.mil/100.2/ADA426737
http://handle.dtic.mil/100.2/ADA426737...
A mixture composed of 33% oxygen could threaten the final concentration due to the accumulation of argon.88. Parker CJ, Snowdon SL. Predicted and measured oxygen concentrations in the circle system using low fresh gas flows with oxygen supplied by an oxygen concentrator. Br J Anaesth. 1988;61:397-402. An adjusted inspiratory oxygen concentration of 35-70% in a fresh gas flow as low as 0.5 L min-1 showed no complications and could be considered safe and without additional risk of hypoxia, in rebreathing systems and closed circuits, according to measurements of O2 concentration in the inspired gas.1111. Rathgeber J, Züchner K, Kietzmann D, et al. Efficiency of a mobile oxygen concentrator for mechanical ventilation in anesthesia. Studies with a metabolic lung model and early clinical results. Anaesthesist. 1995;44:643-50. As pointed out, if the oxygen used as fresh gas is from oxygen concentrators, argon concentration may increase as much as 2%. Argon is inert and has no toxic effect on other gases, e.g., oxygen and other potent inhaled agents.1212. Brattwall M, Warrén-Stomberg M, Hesselvik F, et al. Brief review: theory and practice of minimal fresh gas flow anesthesia. Can J Anaesth. 2012;59:785-97.
In the present investigation the use of 50% oxygen from a concentrator prompted the FiO2 to fall from 52% to 41%, although the ratio of offered and inspired oxygen reached a plateau. The nitrous oxide uptake by the patient is high initially but decreases over time - this means that there will be a preponderance of nitrous oxide over oxygen in the gas remaining in the breathing system after gas uptake by the patient. This gas will be vented to waste (off the circuit), but the oxygen concentration in the system will fall towards the fraction of oxygen that remains after the gas uptake. The N2O alters the final oxygen offered in the breathing system, since the percentage of oxygen from the concentrator did not fall at anytime in either group.
In contrast, the exclusive use of oxygen from a concentrator caused the FiO2 to increase, with a stable ratio of offered and inspired oxygen. This behavior is the result of nitrogen brought to the breathing system by the patient from dissolved volumes in muscle and fat, which comes out of solution over the following hour after the intubation. This rate of nitrogen excretion decreases over time - and this may be a reason why the FiO2 increased in the pure oxygen group.
These results may add to the literature that oxygen from concentrators are cost-effective, reliable and convenient for supplying oxygen, especially in underdeveloped regions and on low budgets,1313. Mokuolu OA, Ajayi OA. Use of an oxygen concentrator in a Nigerian neonatal unit: economic implications and reliability. Ann Trop Paediatr. 2002;22:209-12. presenting results that suggest oxygen from concentrator provide a stable FGF when added to nitrous oxide 50:50 volume. Pressurized oxygen cylinders are expensive when including the cost of transportation, while properly maintained oxygen concentrators can provide a highly effective low-cost and easy-to-use solution for health facilities in developing countries. A return on investment may be achieved within 1-2 years.1414. L'Her P, Tchoua R, Hutin R, et al. The problem of oxygen in developing countries. Med Trop (Mars). 2006;66:631-8. In describing a modern practice management, an anesthesia department in an underserved country hospital established as a norm the use of oxygen concentrator in order to reduce oxygen costs.1515. Shankar KB, Moseley HS, Mushlin PS, et al. Anaesthesia in Barbados. Can J Anaesth. 1997;44:559-68.
The use of nitrous oxide remains a source of controversy in anesthesia. The Evaluation of Nitrous oxide In a Gas Mixture for Anaesthesia (ENIGMA) trial reported that the use of nitrous oxide increases postoperative complications and may contribute to neurocognitive dysfunction in the young and elderly. However, current evidence in support of a more widespread proscription in clinical practice is unconvincing.1616. Sanders RD, Weimann J, Maze M. Biologic effects of nitrous oxide: a mechanistic and toxicologic review. Anesthesiology. 2008;109:707-22. and 1717. Myles PS, Leslie K, Chan MT, et al. Avoidance of nitrous oxide for patients undergoing major surgery: a randomized controlled trial. Anesthesiology. 2007;107:221-31. Given that nitrous oxide-related anesthesia casualties are rare but usually prosecute, they almost invariably attract significant media attention.1818. Herff H, Paal P, von Goedecke A, et al. Fatal errors in nitrous oxide delivery. Anaesthesia. 2007;62:1202-6. Sweden has thus ceased the use of nitrous oxide with no dissatisfaction from anesthesiologists and no increase in the use of other anesthetic agents, as expected.1919. Enlund M, Edmark L, Revenäs B. Ceasing routine use of nitrous oxide - a follow up. Br J Anaesth. 2003;90:686-8. Notwithstanding, a recent large study in non-cardiac surgery suggests the use of this gas in the intraoperative period was associated with decreased odds of 30-day mortality and decreased odds of in-hospital mortality/morbidity.2020. Turan A, Mascha EJ, You J, et al. The association between nitrous oxide and postoperative mortality and morbidity after noncardiac surgery. Anesth Analg. 2013;116:1026-33.
Oxygen concentrators provide a consistent and less expensive source of oxygen in health facilities where power supplies are reliable. The threat of a hypoxic mixture arising from accumulation of argon should be guarded against using an oxygen analyzer in the inspired gas circuit, as well as a pulse oximetry.
The FiO2 in the mixture of O293 and nitrous oxide fell during the observation period although oxygen saturation was higher than 98.5% throughout the study. Concentrators can be considered a stable source of oxygen for use during short anesthetic procedures, either pure or in association with nitrous oxide at 50:50 volume.
References
-
1Friesen RM, Raber MB, Reimer DH. Oxygen concentrators: a primary oxygen supply source. Can J Anaesth. 1999;46: 1185-90.
-
2Arrowsmith LW. Oxygen concentrators for medical gas pipeline systems. J Inst Hosp Eng. 1989;43:6-8.
-
3Moll JR, Moll AVS, Guttman A, et al. Usinas concentradoras de oxigênio: evolução da fração inspirada de oxigênio e repercussões no paciente anestesiado em sistema com absorvedor de CO2. Estudo piloto. Rev Bras Anestesiol. 2007;57:649-57 [Portuguese].
-
4Carter JA, Baskett PFJ, Simpson PJ. The 'Permox' oxygen concentrator. Its mode of action, performance and potential application. Anaesthesia. 1985;40:560-5.
-
5Fenton PM. The Malawi anaesthetic machine. Experience with a new type of anaesthetic apparatus for developing countries. Anaesthesia. 1989;44:498-503.
-
6Shrestsha BM, Singh BB, Gautam MP, et al. The oxygen concentrator is a suitable alternative to oxygen cylinders in Nepal. Can J Anaesth. 2002;49:8-12.
-
7Whitley E, Ball J. Statistics review 3: sample size calculation. Crit Care. 2002;6:335-41.
-
8Parker CJ, Snowdon SL. Predicted and measured oxygen concentrations in the circle system using low fresh gas flows with oxygen supplied by an oxygen concentrator. Br J Anaesth. 1988;61:397-402.
-
9Friesen RM. Oxygen concentrators and the practice of anaesthesia. Can J Anaesth. 1992;39:R80-9.
-
10Grano JT, Roberts AL, Bigley AJ. Determination of the mini-mal fresh gas flow to maintain a therapeutic inspired oxygen concentration in a semi closed anesthesia circle system using an oxygen concentrator as the oxygen source. Technical Report. Houston: Texas Univ. Health Science Center at Hous-ton. School of Nursing; 2001. HSC-SN-00-022. Available at: http://handle.dtic.mil/100.2/ADA426737
» http://handle.dtic.mil/100.2/ADA426737 -
11Rathgeber J, Züchner K, Kietzmann D, et al. Efficiency of a mobile oxygen concentrator for mechanical ventilation in anesthesia. Studies with a metabolic lung model and early clinical results. Anaesthesist. 1995;44:643-50.
-
12Brattwall M, Warrén-Stomberg M, Hesselvik F, et al. Brief review: theory and practice of minimal fresh gas flow anesthesia. Can J Anaesth. 2012;59:785-97.
-
13Mokuolu OA, Ajayi OA. Use of an oxygen concentrator in a Nigerian neonatal unit: economic implications and reliability. Ann Trop Paediatr. 2002;22:209-12.
-
14L'Her P, Tchoua R, Hutin R, et al. The problem of oxygen in developing countries. Med Trop (Mars). 2006;66:631-8.
-
15Shankar KB, Moseley HS, Mushlin PS, et al. Anaesthesia in Barbados. Can J Anaesth. 1997;44:559-68.
-
16Sanders RD, Weimann J, Maze M. Biologic effects of nitrous oxide: a mechanistic and toxicologic review. Anesthesiology. 2008;109:707-22.
-
17Myles PS, Leslie K, Chan MT, et al. Avoidance of nitrous oxide for patients undergoing major surgery: a randomized controlled trial. Anesthesiology. 2007;107:221-31.
-
18Herff H, Paal P, von Goedecke A, et al. Fatal errors in nitrous oxide delivery. Anaesthesia. 2007;62:1202-6.
-
19Enlund M, Edmark L, Revenäs B. Ceasing routine use of nitrous oxide - a follow up. Br J Anaesth. 2003;90:686-8.
-
20Turan A, Mascha EJ, You J, et al. The association between nitrous oxide and postoperative mortality and morbidity after noncardiac surgery. Anesth Analg. 2013;116:1026-33.
Publication Dates
-
Publication in this collection
May-Jun 2014
History
-
Received
18 Mar 2013 -
Accepted
10 June 2013