Abstracts
Biossays were performed to assess the effects of different levels of growth medium supplementation with fetal bovine serum (FBS), fish fry extract (FE), combinations of FBS and FE, and addition of insulin-like growth factor I (IGF-I) and fibroblast growth factor (FGF) on the proliferation of brown bullhead catfish cells (BB line). Treatments (n = 4) were: 2.5, 5, 10, and 15.0% FBS or FE and 5/2.5, 5/5, 10/2.5, and 10/5 of a FBS/FE combination as supplement to the growth medium, or the addition of 0.1, 1, 2.5, 10, 25, and 75 ng/ml of either IGF-I or FGF to the growth media. Initial cell density was 1.1 x 10(6) cells per well on uncoated 24-well plates. Incubation temperature was 29.5 ± 0.7ºC. Six hours after plating, initial culture medium was removed, plates rinsed with Dulbecco’s phosphate buffered saline, treatment media added, and cells allowed to proliferate for 24 hours. Another bioassay was performed with rat myoblast omega cells (RMo) using the same levels of growth medium supplemented with FBS, FE and FBS/FE. Base growth medium was Dulbecco’s MEM. The initial cell density was 7.2 x 10(6) cells per well, and the bioassay was carried out at 36.0 ± 0.5ºC, on a 95% air, 5% CO2 incubator. Increasing levels of FBS had a positive effect (P < 0.05) on the proliferation of both BB and RMo cells. Increasing levels of FE had a negative effect (P < 0.05) on the proliferation of BB cells and totally inhibited the proliferation of RMo cells at any level of supplementation. Higher levels of FE on the FBS/FE combinations presented a negative effect on the proliferation of both BB and RMo cells (P < 0.05). Insulin-like growth factor I had a positive quadratic effect (P < 0.05) on the proliferation of BB cells. Apparently, mammalian growth factors slightly stimulated mitogenic activity in fish cells, while FE contained factors which inhibited the mitogenic activity of RMo and BB cell lines.
mitogenic activity; growth factors; catfish; BB cell line
Foram avaliados os efeitos de diferentes níveis de suplementação do meio de cultivo com soro fetal bovino (FBS), extrato de larvas de peixe (FE), combinações de FBS e FE e adição de fator de crescimento semelhante à insulina I (IGF-I) e fator de crescimento fibroblástico (FGF), na proliferação da linhagem de células somáticas do bagre "brown bullhead" (BB). Os tratamentos (n = 4) utilizados foram: suplementação do meio de cultivo com 2,5, 5, 10% e 15% FBS ou FE, ou 5/2,5, 5/5, 10/2,5 e 10/5 da combinação FBS/FE ou, ainda, a adição de 0,1, 1, 2,5, 10, 25 e 75 ng/ml de IGF-I ou FGF ao meio de cultivo. Foi utilizada uma densidade inicial de 1,1 x 10(6) células/câmara, em placas não recobertas de 24 câmaras. A temperatura de incubação foi de 29,5 ± 0,7ºC. Seis horas após plaqueamento, o meio de cultura inicial foi removido das placas; estas foram enxaguadas com solução salina fosfatada tamponizada de Dulbecco, o meio experimental foi adicionado e as células ficaram em proliferação por 24 horas. Um bioensaio paralelo foi conduzido utilizando-se a linhagem celular mioblástica Omega de ratos (RMo), utilizando-se os mesmos níveis de suplementação do meio com FBS, FE e FBS/FE, tendo por meio básico de cultivo o meio Dulbecco’s MEM. A densidade inicial de células/câmara era de 7,2 x 10(6) células e o bioensaio foi conduzido a 36 ± 0,5ºC em uma incubadora com atmosfera de 95% de ar e 5% de CO2. O aumento nos níveis de suplementação com FBS tiveram um efeito positivo (P < 0,05) na proliferação das linhagens BB e RMo. Níveis crescentes de FE tiveram um efeito negativo (P < 0,05) na proliferação de BB e inibiram totalmente a proliferação de RMo em qualquer nível de suplementação. O aumento nos níveis de FE nas combinações FBS/FE tiveram um efeito negativo na proliferação tanto de BB como RMo (P < 0,05). O IGF-I teve um efeito positivo quadrático (P < 0,05) na proliferação das células BB. Aparentemente, fatores de crescimento de mamíferos estimulam discretamente a atividade mitogênica de células de peixes, enquanto os fatores de crescimento contidos no FE inibem a atividade mitogênica tanto da linhagem BB quanto da linhagem RMo.
atividade mitogênica; fatores de crescimento; bagre; linhagem celular BB
MITOGENIC ACTIVITY OF FETAL BOVINE SERUM, FISH FRY EXTRACT, INSULIN-LIKE GROWTH FACTOR-I, AND FIBROBLAST GROWTH FACTOR ON BROWN BULLHEAD CATFISH CELLS BB LINE
CYRINO, J. E. P.1 and MULVANEY, D.R. 2
1Setor de Piscicultura, Departamento de Produção Animal, ESALQ-USP, C.P. 09, CEP 13418-900, Piracicaba, SP, Brazil
2Department of Animal and Dairy Sciences, Alabama Agricultural Experiment Station,
Auburn University, AL 36849, USA
Correspondence to: José Eurico Possebon Cyrino, Setor de Piscicultura, Departamento de Produção Animal, ESALQ-USP,
Av. Pádua Dias, 11, C.P. 09, CEP 13418-900, Piracicaba, SP, Brazil, e-mail: jepcyrin@carpa.ciagri.usp.br
Received July 20, 1998 Accepted November 18, 1998 Distributed September 10, 1999
ABSTRACT
Biossays were performed to assess the effects of different levels of growth medium supplementation with fetal bovine serum (FBS), fish fry extract (FE), combinations of FBS and FE, and addition of insulin-like growth factor I (IGF-I) and fibroblast growth factor (FGF) on the proliferation of brown bullhead catfish cells (BB line). Treatments (n = 4) were: 2.5, 5, 10, and 15.0% FBS or FE and 5/2.5, 5/5, 10/2.5, and 10/5 of a FBS/FE combination as supplement to the growth medium, or the addition of 0.1, 1, 2.5, 10, 25, and 75 ng/ml of either IGF-I or FGF to the growth media. Initial cell density was 1.1 x 106 cells per well on uncoated 24-well plates. Incubation temperature was 29.5 ± 0.7oC. Six hours after plating, initial culture medium was removed, plates rinsed with Dulbeccos phosphate buffered saline, treatment media added, and cells allowed to proliferate for 24 hours. Another bioassay was performed with rat myoblast omega cells (RMo) using the same levels of growth medium supplemented with FBS, FE and FBS/FE. Base growth medium was Dulbeccos MEM. The initial cell density was 7.2 x 106 cells per well, and the bioassay was carried out at 36.0 ± 0.5oC, on a 95% air, 5% CO2 incubator. Increasing levels of FBS had a positive effect (P < 0.05) on the proliferation of both BB and RMo cells. Increasing levels of FE had a negative effect (P < 0.05) on the proliferation of BB cells and totally inhibited the proliferation of RMo cells at any level of supplementation. Higher levels of FE on the FBS/FE combinations presented a negative effect on the proliferation of both BB and RMo cells (P < 0.05). Insulin-like growth factor I had a positive quadratic effect (P < 0.05) on the proliferation of BB cells. Apparently, mammalian growth factors slightly stimulated mitogenic activity in fish cells, while FE contained factors which inhibited the mitogenic activity of RMo and BB cell lines.
Key words: mitogenic activity, growth factors, catfish, BB cell line.
RESUMO
Atividade mitogênica do soro fetal bovino, extrato de larvas de peixe, fator insulínico de crescimento-I e fator de crescimento de fibroblastos na linhagem celular BB do bagre brown bullhead
Foram avaliados os efeitos de diferentes níveis de suplementação do meio de cultivo com soro fetal bovino (FBS), extrato de larvas de peixe (FE), combinações de FBS e FE e adição de fator de crescimento semelhante à insulina I (IGF-I) e fator de crescimento fibroblástico (FGF), na proliferação da linhagem de células somáticas do bagre brown bullhead (BB). Os tratamentos (n = 4) utilizados foram: suplementação do meio de cultivo com 2,5, 5, 10% e 15% FBS ou FE, ou 5/2,5, 5/5, 10/2,5 e 10/5 da combinação FBS/FE ou, ainda, a adição de 0,1, 1, 2,5, 10, 25 e 75 ng/ml de IGF-I ou FGF ao meio de cultivo. Foi utilizada uma densidade inicial de 1,1 x 106 células/câmara, em placas não recobertas de 24 câmaras. A temperatura de incubação foi de 29,5 ± 0,7oC. Seis horas após plaqueamento, o meio de cultura inicial foi removido das placas; estas foram enxaguadas com solução salina fosfatada tamponizada de Dulbecco, o meio experimental foi adicionado e as células ficaram em proliferação por 24 horas. Um bioensaio paralelo foi conduzido utilizando-se a linhagem celular mioblástica Omega de ratos (RMo), utilizando-se os mesmos níveis de suplementação do meio com FBS, FE e FBS/FE, tendo por meio básico de cultivo o meio Dulbeccos MEM. A densidade inicial de células/câmara era de 7,2 x 106 células e o bioensaio foi conduzido a 36 ± 0,5oC em uma incubadora com atmosfera de 95% de ar e 5% de CO2. O aumento nos níveis de suplementação com FBS tiveram um efeito positivo (P < 0,05) na proliferação das linhagens BB e RMo. Níveis crescentes de FE tiveram um efeito negativo (P < 0,05) na proliferação de BB e inibiram totalmente a proliferação de RMo em qualquer nível de suplementação. O aumento nos níveis de FE nas combinações FBS/FE tiveram um efeito negativo na proliferação tanto de BB como RMo (P < 0,05). O IGF-I teve um efeito positivo quadrático (P < 0,05) na proliferação das células BB. Aparentemente, fatores de crescimento de mamíferos estimulam discretamente a atividade mitogênica de células de peixes, enquanto os fatores de crescimento contidos no FE inibem a atividade mitogênica tanto da linhagem BB quanto da linhagem RMo.
Palavras-chave: atividade mitogênica, fatores de crescimento, bagre, linhagem celular BB.
INTRODUCTION
The farming of channel catfish, Ictalurus punctatus, is the most important fish industry in the United States (Wellborn & Tucker, 1985; Stickney, 1990). Although a significant research basis has been established on nutrition (Robinson & Wilson, 1985; Lovell, 1989) and health management of channel catfish (MacMillan, 1989), there is very little information available on the physiology of growth of the species.
True growth in fish can be characterized as an increase in the protein content of the body. Because muscle, organs and demineralized bone tissue are primarily protein (Lovell, 1986), increases in weight gain of these tissues more appropriately reflect true growth than fat deposition (Mulvaney, 1991). Muscle is the primary deposit site of protein in animals (Pearson & Young, 1989), and in fish it can account for about 60% of the weight of an adult (Millward, 1989). Thus, adoption of practices for manipulation of fish growth is conditioned to the understanding of the protein synthesis and muscle growth regulating processes. Most information on the regulation of muscle growth in animals has been gathered from measurements in rodents, but limited research data indicate that the mechanism of muscle growth regulation in fish is comparable (Millward, 1989).
The regulation of myogenic cell differentiation and proliferation and of the rates of muscle protein synthesis and degradation are the mechanisms of control of muscle growth. Those mechanisms are controlled by several factors. The genetic make-up, nutrition, peptide hormones, peptide growth factors, and level of activity are all involved in the regulation of both normal and (or) abnormal growth processes of animals. Growth hormone (somatotropin), insulin, thyroid hormones, exemplify some of the peptide hormones involved in the growth process in animals. Insulin-like growth factor I and II (IGF-I, II), fibroblast growth factor (FGF), and transforming growth factor-b (TGF-b) are considered the major peptide growth factors (Goldberg et al., 1980; Vandenburgh, 1983; Beitz, 1985; Young, 1987; Black, 1988; White & Esser, 1989; Florini & Magri, 1989; Ewton and Florini, 1990; Florini et al., 1991).
Considerable attention has been devoted recently to the role of the peptide growth factors in the regulation of poultry and mammalian growth. The insulin-like growth factors, or somatomedins, are small peptides secreted by several cell types. The IGF-I, with 70 amino acids and a molecular weight of 7649 daltons, is the primary agent of this family of growth factors. The IGFs stimulate cell proliferation and differentiation, amino acid and glucose uptake, and suppress protein degradation on muscle cells and many other somatic cells. IGF-I is the most potent of the growth factors, and the most potent anabolic agent for skeletal muscle growth (Florini & Magri, 1989; Ewton & Florini, 1980, 1990; Florini et al., 1991).
The fibroblast growth factors are mitogens that induce division of most cultured cells. Fibroblast growth factor exists in two forms, acid FGF (aFGF) and basic FGF (bFGF), both single peptide chains composed of 146 and 140 amino acids, respectively (Thomas, 1987; Yamada et al., 1989; Florini et al., 1991). Fibroblast growth factor is a potent inhibitor of differentiation in many cell types, myoblasts in particular (Florini, 1987; White & Esser, 1989; Florini et al., 1991).
The IGFs, acting alone or in association with other hormones and (or) growth factors, are potent mitogenic and anabolic agents, playing a major role in the processes of proliferation and differentiation of myogenic cells, as well as in the protein turnover on muscle tissue. There is no mechanism yet generally accepted for the actions of FGF on mammalian myoblasts, but it was observed to have a potent mitogenic activity (Florini et al., 1991).
Collodi and Barnes (1990) found that trout embryo extracts may contain a growth factor which stimulates proliferation of established cell lines, representing a first step in understanding the extracellular mechanisms of regulation of piscine cell growth. Further work is required to determine the occurrence of fish-specific versions of peptide growth factors and their biochemical nature, and the present work targets this area of research, investigating the mitogenic activity of IGF-I and FGF on catfish cells, the BB line (Wolf & Quimby, 1969).
MATERIALS AND METHODS
Culture medium supplementation bioassay
The effects of different levels of supplementation of growth medium with fetal bovine serum (FBS; Sigma Chemical Co.), fish fry extract (FE), and a combination FBS/FE on the proliferation of BB cells, a fibroblastic-like cell line isolated from the trunk tissue of 2-year-old brown bullhead catfish Ictalurus nebulosus (Wolf & Quimby, 1969; Hay et al., 1992), were assessed. The initial stock of BB cells was supplied by the Southeastern Cooperative Fish Disease Laboratory, Department of Fisheries and Allied Aquacultures, Alabama Agricultural Experiment Station, Auburn University, AL.
Fish fry extract (FE) was obtained following the recommendations of Coon & Chan (1966) for preparation of chick embryo extract. Seven-day-old channel catfish swim-up fry (Midsouth Aquafarms, Montgomery) were washed once in sterile PBS and once in Hanks balanced salt solution (HBSS), placed in a sterile blender and homogenized with an equal volume of HBSS.
Homogenate was filtered through cheese cloth and centrifuged at 1,000 = g for 10 minutes. The supernatant was collected and centrifuged at 10,000 = g for 20 minutes. The final supernatant was collected and left to precipitate at 20oC overnight, filtered through a 0.45 µm separation filter (Micron Separations, Inc.; Fisher Scientific) and sterile filtered through a 0.22 µm sterilizing Sterivex® filter (Millipore Corp.), and stored in 15 ml aliquots at 20oC. Prior to addition to the treatment media, FE aliquots were thawed in water bath at 36 ± 0.5oC.
Bioassays were set up on Corning® 24-well plates (Fisher Scientific), each plate representing different levels of growth medium supplement-ation, each well representing one replicate. Treatments were: 2.5, 5, 10, and 15% of FBS or FE; and 5/2.5, 5/5, 10/2.5, and 10/5 of FBS/FE combination. Cells were seeded at a density of 1.1 × 106 per well on uncoated surface. Initial growth medium was Eagles minimum essential medium (MEM; Sigma Chemical Co.), HEPES-buffered (HMEM) with a pH of 7.2 ± 0.1, supplemented with 10% FBS. Incubation temperature was 29.5 ± 0.7oC. Unless otherwise noted, culture medium was supplemented with 10 ml/l of Antibiotic-Antimicotic Solution 100X Hybrimax® (ABAM 10,000 units of penicillin, 10 mg streptomycin, and 25 µg of amphotericin B per ml; Sigma Chemical Co.).
Six hours after plating, initial culture medium was removed, plates were rinsed once with phosphate buffered saline (PBS; Sigma Chemical Co.), and treatment medium was added. Cells were allowed to proliferate for 24 hours. Treatment growth media were removed and cells fixed with 10% formalin (Plumb & Bowser, 1983), stained with Giemsa stain (Freshney, 1987), and counted with the aid of a Nikon® inverted microscope under a 0.0025 mm2 counting grid mounted over a 20X objective. Ten random counting grid fields were counted per well and the total number of cell per well calculated.
Another bioassay similarly designed was carried out with rat myoblast omega (RMo) cells (Merrill, 1989), using the same levels of growth medium supplementation with FBS, FE and FBS/FE, intending to determine responses of FE supplementation on mammalian cells. Base growth medium was Dulbeccos modified eagles medium (DMEM; Sigma Chemical Co.). The RMo cells were obtained from a routinely grown population of the Growth and Muscle Biology Laboratory, Department of Animal and Dairy Sciences, Auburn University, AL.
The initial cell density was 7.2 x 106 cells per well. The RMo cells bioassay was carried out at 36.5 ± 0.7oC, on a 95% air, 5% CO2 incubator. Plates with RMo cells were fixed with methanol and stained with Giemsa stain (Freshney, 1987). The final number of cells per well was determined as described.
Growth factors bioassay
The effects of insulin-like growth factor I (IGF-I; Collaborative Research, Inc.) and fibroblast growth factor (FGF; Collaborative Research, Inc.) on the proliferation of BB cells were assessed. Treatments were set up in Corning® 24-well plates. Treatments were the addition of 0.1, 1.0, 2.5, 10, 25, and 75 ng/ml of either IGF-I or FGF, to the growth medium supplemented with 10% FBS. Four replicates per treatment were provided, each one represented by one well.
Cells were seeded at a density of 1.1 x 106 per well on uncoated surfaces. Initial growth medium and control treatment was HMEM-10% FBS. Incubation temperature was 29.5 ± 0.7oC (Hay et al., 1992). After a 6-hour attachment and resting period, culture medium was removed, plates were rinsed once with PBS and treatment media were added. Cells were allowed to proliferate for 24 hours, and then fixed, stained and counted as described, and the final number of cells per well was determined.
Isolation of a myogenic subclone of BB cells
Because BB line contains myoblast-like cells (ML), an attempt was made to isolate a myogenic subclone of BB cells through sequential dilutions of the cell suspension, based on findings of Merrill (1989) and Hembree et al. (1991). A dense monolayer of BB cells was detached and resuspended, cell clumps dissolved by gentle agitation, and dilutions of the suspension were seeded in multi-well plates to obtain isolation of single ML cells and their clone cultures.
Dilutions of the cell suspension were carried out using uncoated Corning® 96-wells plates, following recommendations of Hembree et al. (1991). Plate wells 1, 25, 49, and 73 were inoculated with 0.2 ml of cell suspension. Wells 12, 24, 48, and 72 were left empty. Remaining wells received 0.1 ml of fresh HMEM-10% FBS.
Dilutions were obtained by transference of 0.1 ml of the cell suspension sequentially to the next well until an empty well also contained 0.1 ml. After a 6-hour resting and attachment period, wells were checked for the presence of one ML. On the average, 3 wells with single ML cells were obtained per plate.
Initial growth medium was removed and wells were gently washed with fresh, unsupplemented HMEM. Four plates were seeded at each time. Single-cell wells received either fresh HMEM supplemented with 5% or 10% FBS, or conditioned medium, unsupplemented or supplemented with 5% FBS. Cells were allowed to proliferate for 48 hours at 30oC, and checked for the development of clonal cultures of BB line MLs.
Conditioned medium was prepared following recommendations of White & Hauschka (1971). Fresh HMEM-5% FBS was exposed for 24 hours to 1-day-old cultures of BB cells contained in 75 cm2 Corning® culture flasks (15 ml/flask). Prior to use, conditioned medium was passed through 0.45 and 0.22 µm filters. Conditioned medium was stored at 20oC and thawed to 30oC prior to use.
Statistical procedures
General Linear Model procedures for orthogonal polynomial mean comparisons were used to analyze the results of the cells proliferation assays. Tukeys Studentized Range test was used to contrast treatment means (Steel & Torrie, 1980; Gill, 1989; Snedcor & Cochran, 1989).
RESULTS AND DISCUSSION
Effects of FBS and FE on the proliferation of BB and RMo cells.
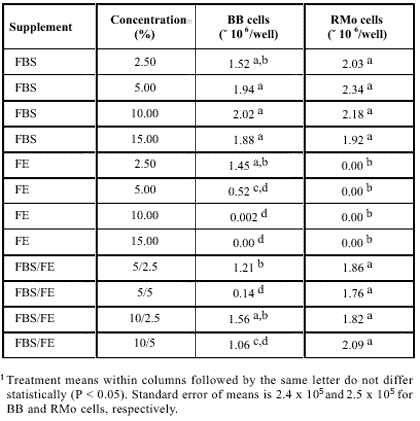
The results of the bioassays on the effects of the culture medium supplementation with FBS, FE or combination FBS/FE on the proliferation of BB and RMo cells are summarized on Table 1. The orthogonal polynomial analysis of the results show a positive effect (P < 0.05) of the increasing levels of FBS on the proliferation of BB cells. This effect can be represented by the equation Y = 1.1810 x 106 + 1.7945 × 106 X 8.9070 × 103 X2. Treatment means did not differ significantly (P > 0.05). The 10% FBS supplement level yielded the highest final number of cells per plate.
Effects of growth medium supplementation with fetal bovine serum (FBS), fish fry extract (FE) and FBS/FE combination on the proliferation of brown bullhead (BB) and rat myoblast omega (RMo) cell lines.1
The proliferative activity of RMo cells was positively affected by increasing levels of FBS (P < 0.05). The polynomial analysis show that increased levels of FBS exerts a quadratic effect on the proliferation of RMo cells (P < 0.05) which can be expressed by the equation: Y = 1.8470 × 106 1.1547 × 106 X 7.460 × 104 X2. Treatment means did not differ significantly (P > 0.05).
Results were predictable to a certain degree, since FBS is the choice growth media supplement for many cultured mammalian and (or) fish cells (Wolf & Quimby, 1969; Freshney, 1987, 1989; Allen, 1987; Konigsberg, 1979; Bowser & Plumb, 1980a, b; Hay et al., 1992; Dayton & Allen, 1987). Recently Lee et al. (1988) showed that the ideal level of supplementing growth medium for culture of RTG-2 cells is 10% FBS, and Gopalakrishnan (1991) showed a positive response of rat myogenic cells to increasing levels of horse serum up to 10%. Apparently mammalian-derived serum supports growth of both mammalian and fish cells.
The supplementation of growth medium with increasing levels of FE show a negative quadratic effect on the proliferation of BB cells (Y = 2.3110 × 106 4.1791 × 106 X + 1.7720 × 104 X2). The mean of the 2.50% FE treatment did not differ significantly from the means of the FBS-supplemented treatments (P > 0.05), but differed significantly from the means of the other FE supplementation treatments (P < 0.05). Supplementing with any level of FE inhibited the proliferation of RMo cells.
The proliferation of both BB and RMo cells was negatively affected by increasing levels of FE in the FBS/FE combination (P < 0.05), but those effects were minimized by increasing levels of FBS in the combination (P < 0.05). Apparently, fish fry extract did not support the in vitro growth of either mammalian or fish cells, or inhibits the proliferation of the cells in vitro.
Collodi & Barnes (1990), prepared an extract of Shasta strain rainbow trout embryos, excluding the yolk sac, and applied the extract as a supplement to growth medium in cultures of established fish cell lines: epithelial papilloma of carp (EPC), goldfish fin (CAR), rainbow trout gonad (RTG-2) and hepatoma (RTH-149), steelhead trout embryo (STE-137) and chinook salmon embryo (CHSE-214). Cells were plated at 105 cells per well in 6-well tissue culture plates and grown for 14 days in 3:1 DMEM/Hams F-12 medium supplemented with 10% FBS. The embryo extract was mitogenic for the EPC, CAR and RTG-2, but was not mitogenic for RTH-149. The embryo extract allowed long term maintenance of serum-free CHSE-214 cells (20 passages, 50 population doublings). Increasing concentrations of the embryo extracts protein of 2.5, 10, 25, 100, and 250 µg/ml on growth medium stimulated proliferation of CHSE-214 cells up to 100 µg/ml, at which point it apparently became toxic to the cells.
When 20 µg/ml of the embryo extract or FBS protein was added to the cultures, the embryo extract was three times more effective at stimulating proliferation of CHSE-214 cells, but 10% FBS, equivalent to 5 mg protein/ml, was twice more effective than embryo extract at 20 µg/ml. Supplementing with 10% of embryo extract was toxic for CHSE-214 cells.
Effects of IGF-I and FGF on the proliferation of BB cells
Table 2 and summarizes the effects of IGF-I and FGF on the proliferation of BB cells. Neither growth factor had a significant effect (P > 0.05) on the proliferation of BB cells. Treatment means did not differ significantly (P > 0.05). The orthogonal polynomial analyses show that only IGF-I has a positive quadratic effect (P < 0.05) on the proliferation of BB cells, represented by: Y = 2.03 × 106 6.9484 × 103 X + 56.254 X2.
Effects of insulin-like growth factor I (IFG-I) and fibroblast growth factor (FGF) concentrations on the culture medium (MEM) on the proliferation of brown bullhead (BB) cells.
Collodi & Barnes (1990) tested the mitogenic activity of trout embryos and the peptide growth factors, platelet-derived growth factor (PDGF), insulin-like growth factor I and II (IGF-I and II), transforming growth factor-beta (TGF-b), acidic fibroblast growth factor (aFGF) and basic FGF (bFGF), against a control medium supplemented with insulin, transferrin and epidermal growth factor (EGF). Ten ng/ml of PDGF, 100 ng/ml of IGF-I, 1.0 µg/ml of IGF-II, 10 ng/ml of TGF- b, 50 ng/ml of TGF-b, 10 ng/ml of aFGF, and 10 ng/ml of bFGF, did not mimic the effect of 20 µg of protein/ml of trout embryo extract. Transforming growth factor-b, IGF-I and IGF-II were moderately mitogenic for CHSE-214 resulting in 130%, 145 and 150% stimulation of proliferation as percent of the control, as compared to the effectiveness of the embryo extract with 290% of stimulation of proliferation.The trout embryo extract did not mimic the stimulatory effects of the peptide growth factors on the DNA synthesis in a quiescent mouse fibroblast cell line, BALB/c 3T3.
Collodi & Barnes (1990) concluded that the trout embryo extract may contain a fish-specific version of a known mammalian growth factor or a factor not previously described, which stimulates proliferation of established cell lines, representing a first step in understanding the extracellular mechanisms of piscine cells growth regulation. Apparently the same conclusion apply relative to FE in this study.
Several authors have demonstrated that insulin effectively regulates growth of fish. Tashima & Cahill (1968) measured the effects of intravenous injections of 100 IU/kg of insulin on the in vivo and in vitro incorporation of glucose and glycine on the liver and tail muscle of the toadfish, Opsanus tau. They concluded that insulin reduces the levels of blood glucose, stimulates the incorporation of glycine on muscle and liver protein, and stimulates the incorporation of glucose in the liver and muscle lipids and glycogen, showing that insulin presents in fish the same anabolic activity as for mammals.
Ludwig et al. (1977) conducted a preliminary study of insulin participation in the growth regulation of coho salmon. Bi-weekly, intra peritoneal injections of 0.32, 1.0, 3.2, and 10 IU/kg of body weight of bovine insulin stimulated the specific growth rate by 1.601%, 1.477%, 1.567%, and 1.530%/day, respectively, and decreased the food to gain ratios to 1.26, 1.28, 1.28, and 1.33 gram:gram, respectively. However, increase in specific growth rate and decrease in food to gain ratios did not differ significantly from uninjected or solvent-injected fish. No conclusive results were attained, but the authors recommended the use of higher doses of insulin or more frequent treatment, and the use of species-specific insulin in future experiments.
Inui & Ishioka (1983a, b) investigated the in vitro effects of insulin on the incorporation of [14C] glycine into the protein of liver and opercular muscle and on the amino acid transport into the liver and opercular muscle of the eel, Anguilla japonica. The authors incubated slices of liver and opercular muscle of eel in a medium containing 0.1 IU/ml of insulin and 0.1 µCi/ml of [U 14C] glycine. After five hours of incubation, the insulin increased the radioactivity of both trichloroacetic acid (TCA)-soluble fraction and of the opercular muscle protein, indicating that insulin stimulates both the uptake of the amino acid by the tissue and the protein synthesis. An increase on the incorporation of [14C] glycine into the liver protein accompanied by a decrease in the radioactivity of the TCA-soluble fraction was observed for the liver tissue. This indicates stimulation of protein synthesis out of the intracellular amino acid pool, characterizing the regulatory effects of insulin on the protein synthesis in the liver and muscle of eel.
Inui & Ishioka (1983b) demonstrated that 0.1 IU of insulin per ml of incubation medium stimulates the uptake of the a-[I 14C] aminoisobutyric acid (AIB) by the liver and opercular muscle of the eel. As AIB is a nonmetabolizable amino acid, the findings indicate that the entry of amino acids into the tissues of the eel is stimulated by insulin, independently of the protein synthesis.
Insulin is a major regulator of growth in mammals and fish. Pharmacological levels of insulin were shown to promote proliferation of skeletal muscle satellite cells in a dose-dependent manner, acting through receptors at the cell surface level. This supports the theory that insulin is not a potent proliferative agent, but mimics the effects of peptide growth factors through their receptors in the cell surface, stimulating muscle satellite cell proliferation (Dodson et al., 1985).
A cross effect of IGF-I mimicking the effects of a fish IGF-like compound or fish insulin can be the reason for the slight stimulation of proliferation of BB cells caused by IGF-I. Collodi & Barnes (1990) infer that fish-specific versions of mammalian growth factor or a factor not previously described may exist, which stimulates proliferation of established fish cell lines. The inference of Collodi & Barnes (1990) lend a logical explanation to the findings of this study. Further work is required to determine the occurrence of fish-specific versions of peptide growth factors and their biochemical nature.
Isolation of a myogenic subclone of BB cells
Single myogenic cells isolated from cultures of primary chicken myoblasts (White & Hauschka, 1971), rat MM14D muscle cell line (Merrill, 1989), and swine primary myoblasts (Hembree et al., 1991) proliferate to form clonal colonies. However, single ML cells isolated from BB line, cultured with either HMEM with 5% or 10% FBS, or conditioned medium alone or supplemented 5% FBS did not proliferate to form clonal colonies.
Apparently the paracrine effect of fibroblasts that stimulates the proliferation of ML cells in culture (Quinn et al., 1990) can not be replaced by the use of conditioned medium for ML present in the BB line (White & Hauschka, 1971). The replating of isolated ML was not successful, supporting results of Hay et al. (1992) who reported a 1% plating efficiency for the BB line.
Extending the period of isolated BB cells cultures from 48 to 72, 96 or 120 hours did not result in any proliferative activity. The development of myogenic cell lines and clonal cultures was demonstrated by White & Hauschka (1971) for primary chicken myoblasts, Merrill (1989) for rat MM14D muscle cells, and Hembree et al. (1991) for swine primary myoblasts. Thus, the obtainment of clonal cultures and development of a fish myogenic cell line is a possibility and deserves further studies, perhaps starting from primary cultures of muscle cells.
Acknowledgements Recognition is due to Dr. Steve Jungst, Department of Animal and Dairy Sciences, Auburn University, AL, for the assistance on the statistical analysis of data; to Mr. Ken Hasson and Dr. Yolanda Brady, Department of Fisheries and Allied Aquacultures, Auburn University, AL, for the invaluable orientation on the handling of the BB cells; and to Mr. Ed Forks, Midsouth Aquafarms, for the channel catfish fry used in the obtainment of the fish fry extract.
- ALLEN, R. E., 1987, Muscle cell culture as a tool in animal growth research. Federation Proc., 46: 290-294.
- BEITZ, D. C., 1985, Physiological and metabolic systems important to animal growth: an overview. J. An. Sci., 61(2): 1-20.
- BLACK, J. L., 1988, Animal growth: the whole and the parts. J. An. Sci., 66(3): 1-22.
- BOWSER, P. R. & PLUMB, J. A., 1980a, Fish cell lines: establishment of a line from ovaries of channel catfish.In vitro, 16: 365-368.
- BOWSER, P. R. & PLUMB, J. A., 1980b, Growth rates of a new cell line from channel catfish ovary and channel catfish virus replication at different temperatures. Can. J. Fish. Aquat. Sci., 37: 871-873.
- COLLODI, P. & BARNES, D. W., 1990, Mitogenic activity from trout embryos. Proc. Natl. Acad. Sci. USA, 87: 3498-3502.
- COON, H. S. & CHAN, R. D., 1966, Differentiation in vitro: Effects of Sephadex fractions of chick embryo extract. Science, 153: 1116-1119.
- DAYTON, W. R. & ALLEN, R. E., 1987, Muscle cell culture as a research tool to facilitate studies of muscle growth in meat animals. J. Anim. Sci., 65(2): 1-11.
- DODSON, M. V., ALLEN, R. E. & HOSSNER, K. L., 1985, Ovine somatomedin, multiplication stimulation activity, and insulin promote skeletal muscle satellite cell proliferation in vitro Endocrinology, 117(6): 2357-2363.
- EWTON, D. Z. & FLORINI, J. R., 1980, Relative effects of the somatomedins, multiplication-stimulating activity, and growth hormone on myoblasts and myotubes in culture. Endocrinology, 106: 577-583.
- EWTON, D. Z. & FLORINI, J. R., 1990, Effects of insulin-like growth factors and transforming growth factor-b on the growth and differentiation of muscle cells in culture. PSEBM, 194: 76-80.
- FLORINI, J. R., 1987, Hormonal control of muscle growth. Muscle & Nerve, 10: 577-598.
- FLORINI, J. R., EWTON, D. Z. & MAGRI, K. A., 1991, Hormones, growth factors, and myogenic differentiation. Annu. Rev. Physiol., 53: 201-216.
- FLORINI, J. R. & MAGRI, K. A., 1989, Effects of growth factors on myogenic differentiation. Am. J. Physiol., 256 (Cell Physiol. 25): C701-C711.
- FRESHNEY, R. I., 1987, Culture of Animal Cells: A Manual of Basic Techniques, 2a ed., Alan R. Liss, Inc., New York.
- FRESHNEY, R. I., 1989, Animal Cell Culture: A Practical Approach, 3a ed., IRL Press, Oxford, Washington, DC.
- GILL, J. L., 1989, Design and Analysis of Experiments in the Animal and Medical Sciences, V. I-III. The Iowa State University Press, Ames, IA.
- GOLDBERG, A. L., TISCHLER, M., DeMARTINO, G. & GRIFFIN, G., 1980, Hormonal regulation of protein degradation and synthesis in skeletal muscle. Fed. Proc., 39: 31-36.
- GOPALAKRISHNAN, S. R., 1991, Regulation of the proliferation of rat skeletal muscle satellite cells by genotype and growth factors Auburn University: Auburn, AL. 160p. Master of Science Thesis.
- HAY, R., CAPUTO, J., CHEN, T. R., MACY, M., McCLINTOCK, P. & REID, Y. (eds.), 1992, Catalog of Cell Lines and Hybridomas. 7a ed. American Type Culture Collection, Rockville, MD.
- HEMBREE, J. R., HATHAWAY, M. R. & DAYTON, W. R., 1991, Isolation and culture of fetal porcine myogenic cells and the effect of insulin, IGF-I, and sera on protein turnover in porcine myotube cultures. J. Anim. Sci., 69: 3241-3250.
- INUI, Y. & ISHIOKA, H., 1983a, Effects of insulin and glucagon on the incorporation of [14C] glycine into the protein of the liver and opercular muscle of the eel in vitro Gen Comp. Endocrinol., 51: 208-212.
- INUI, Y. & ISHIOKA, H., 1983b., Effects of insulin and glucagon on amino acid transport into the liver and opercular muscle of the eel in vitro Gen. Comp. Endocrinol., 51: 213-218.
- KONIGSBERG, I., 1979, Skeletal myoblasts in culture. Methods Enzymol., 58: 511-527.
- LEE, L. E. J., MARTINEZ, A. & BOLS, N. C., 1988, Culture conditions for arresting and stimulating the proliferation of a rainbow trout fibroblast cell line, RTG-2. in vitro Cell. & Dev. Biol., 24(8): 795-802.
- LOVELL, R. T., 1986, Weight gain versus protein gain for evaluating fish feeds. Aquaculture Magazine, January-February: 45-47.
- LOVELL, R. T., 1989, Nutrition and Feeding of Fish AVI Books Co., New York.
- LUDWIG, B., HIGGS, D. A., FAGERLUND, U. F. & McBRIDE, J. R., 1977, A preliminary study of insulin participation in the growth regulation of coho salmon (Oncorhynchus kisutch). Can. J. Zool., 55: 1756-1758.
- MacMILLAN, J. R., 1989, Infectious diseases. In: C. S. Tucker (ed.), Channel Catfish Culture. Developments in Aquaculture and Fisheries Science, n. 15. Elsevier Scientific Publishing Co., New York, pp. 405-496.
- MERRILL, G. F., 1989, Clonal derivation of a rat muscle cell strain that forms contraction-competent myotubes. in vitro Cell. and Dev. Biol., 25(5): 471-476.
- MILLWARD, D. J., 1989, The nutritional regulation of muscle growth and protein turnover. Aquaculture, 79: 1-28.
- MULVANEY, D. R., 1991, How growth affects composition. In: R. Kauffman (ed.), Twenty-first Century Concepts of Meat-Animal Evaluation University of Wisconsin Press, Madison, WI., pp. 55-92.
- PEARSON, A. M. & YOUNG, R. B., 1989, Muscle and Meat Biochemistry Academic Press, Inc, New York.
- PLUMB, J. A. & BOWSER, P. R., 1983, Microbial fish disease laboratory manual Department of Fisheries and Allied Aquacultures, Alabama Agricultural Experiment Station, Auburn University, Auburn, AL.
- QUINN, L. S., ONG, L. D. & ROEDER, R. A., 1990, Paracrine control of myoblast proliferation and differentiation by fibroblasts. Dev. Biol., 140: 8-9.
- ROBINSON, E. H. & WILSON, R. P., 1985, Nutrition and feeding. In: C. S. Tucker (ed.), Channel Catfish Culture. Developments in Aquaculture and Fisheries Science n. 15 Elsevier Scientific Publishing Co., New York, pp. 323-404.
- SNEDCOR, G. W. & COCHRAN, W. S., 1989, Statistical Methods, 8a ed., The Iowa State University Press, Ames, IA.
- STEEL, R. G. D. & TORRIE, J. H., 1980, Principles and Procedures of Statistics: A Biometrical Approach McGraw-Hill Book Co., New York.
- STICKNEY, R. R., 1990, A global overview of aquaculture production. Food. Rev. Int., 6: 299-315.
- TASHIMA, L. & CAHILL JR., G. F., 1968, Effects of insulin in the toadfish, Opsanus tau. Gen. Co Endocrinol., 11mp. : 262-271.
- THOMAS, K. A., 1987, Fibroblast growth factors. FASEB J., 1: 434-440.
- VANDENBURGH, H. H., 1983, Cell shape and growth regulation in skeletal muscle: Exogenous versus endogenous factors. J. Cell. Physiol., 116: 363-371.
- WELLBORN, T. L. & TUCKER, C. S., 1985, An overview of commercial catfish culture. In: C. S. Tucker (ed.), Channel Catfish Culture. Developments in aquaculture and fisheries science n. 15 Elsevier Scientific Publishing Co., New York, pp. 1-12.
- WHITE, T. P. & ESSER, K. A., 1989, Satellite cell and growth factor involvement in skeletal muscle growth. Med. Sci. Sports Exerc., 21: S158-S163.
- WHITE, N. K. & HAUSCHKA, S. D., 1971, Muscle development in vitro: A new conditioned medium effect on colony differentiation. Exptl. Cell Res., 67: 479-482.
- WOLF, K. & QUIMBY, M. C., 1969, Fish cell and tissue culture. In: W. S. Hoar & D. J. Randall (eds.), Fish Physiology, v. III: Reproduction and Growth. Bioluminescence, Pigments and Poisons Academic Press, Inc., New York, pp. 253-305.
- YAMADA, S., BUFFINGER, N., DIMARIO, J. & STROHMAN, R. C., 1989, Fibroblast growth factor is stored in fiber extracellular matrix and plays a role in regulating muscle hypertrophy. Med. Sci. Sports Exerc., 21(5): S173-S186.
- YOUNG, V. R., 1987, Genes, molecules and the manipulation of animal growth: A role for biochemistry and physiology. J. An. Sci., 65(2): 107-127.
Publication Dates
-
Publication in this collection
27 Nov 2003 -
Date of issue
Aug 1999
History
-
Received
20 July 1998 -
Accepted
18 Nov 1998