ABSTRACT
Although tank mixture of pesticides and foliar fertilizers is common practice in agriculture, further clarification and scientific evidence is needed to support regulation. Thus, the objective of this work was to evaluate the effect of tank mixing an organosilicon adjuvant and manganese foliar fertilizer on the insecticide imidacloprid effectiveness against Triozoida limbata in guava trees. The experimental plot consisted of four consecutive trees in the same row subdivided into 4 quadrants. The experiment was arranged in a randomized block design with split plots, with four replications. Treatments were as follows: T1 - Imidacloprid (Imid.); T2 - Imid. + Polyether-polymethyl siloxane copolymer (Sil.); T3 - Imid. + MnSO4; T4 - Imid. + Sil. + MnSO4; and T5 - Control (no application). Physical-chemical characteristics, spray deposition on the leaves and losses to the soil, guava psyllid percentage of infestation and nymph’s number were evaluated. The addition of foliar fertilizer into the mixture decreased the pH and the surface tension and increased the electric conductivity and the viscosity of the insecticide solutions. The silicon adjuvant decreased the surface tension and increased the viscosity and the pH. The tank mixture of organosilicon adjuvant and manganese foliar fertilizer has no influence on the efficacy of the insecticide.
Keywords:
guava psyllid; physical-chemical characteristics; neonicotinoids; foliar fertilizer; organosilicon
INTRODUCTION
Guava (Psidium guajava L.) stands out among the Brazilian tropical fruit species mainly because of its flavor and nutritional value. To improve fruit quality and increase the production, growers must overcome obstacles such as orchard management, problems with fertilization, application technologies, as well as the high incidence of diseases and pests. One of the main problems for guava production is the insect known as guava psyllid (Triozoida limbata - Hemiptera: Triozidae) (Galli et al. 2014Galli JA, Michelotto MD, Soares MBB, Martins ALM & Fischer IH (2014) Damage Caused by Psyllids and Influence of Climatic Factors in Brazilian Accesses of Guava Trees Cultivated in Organic System. American Journal of Plant Sciences, 05:1432.; Barbosa & Lima, 2010Barbosa FR & Lima MF (2010) A cultura da goiaba. 2ª ed. Brasília, Embrapa Informação Tecnológica. 180p.).
The characteristic symptom of guava psyllid attack is the curling of the edges of the leaves caused by feeding of colonies of nymphs. Leaves can fall from this attack, compromising the production (Barbosa et al., 2001Barbosa FR, Souza EA, Siqueira KMM, Moreira WA, Alencar JR & Haji FNP (2001) Eficiência e seletividade de inseticidas no controle de psilídeo (Triozoida sp.) em goiabeira. Pesticidas: Revista de Ecotoxicologia e Meio Ambiente, 11:45-52.; Gallo et al., 2002Gallo D, Nakano O, Silveira Neto S, Carvalho RPL, Batista GC, Berti Filho E, Parra JRP, Zuchi RA, Alves SB, Vendramim JD, Marchini LC, Lopes JRS & Omoto C (2002) Entomologia agrícola. Piracicaba, FEALQ. 920p.). According to Colombi & Galli (2009Colombi CA & Galli JC (2009) Dinâmica populacional e evolução de dano de Triozoida limbata (Hemiptera: Psillydae) em goiabeira, em Jaboticabal, SP. Ciência Agrotecnologia, 33:412-416.), the importance of this psyllid has increased probably because of the adopted production system, with more irrigation and tree pruning, which favors the psyllid population to grow due to the abundant number of new sprouts.
‘Paluma’ is one of the most planted cultivars in Brazilian orchards, mainly because of its capacity to produce fruits for both industry and fresh consumption (Farias et al., 2017Farias WC, Câmara FMM, Silva FSO, Mendonça V, Pereira GA & Leite GA (2017) Qualidade de frutos da goiabeira cv. Paluma submetida a podas de frutificação em diferentes épocas no município de Mossoró-RN. Nativa, 05:05-08.). This cultivar is not resistant to attacks of psyllid, which became one of the main problems for its production (Barbosa & Lima, 2010Barbosa FR & Lima MF (2010) A cultura da goiaba. 2ª ed. Brasília, Embrapa Informação Tecnológica. 180p.).
The use of pesticides is frequent during the guava cycle, mostly as tank mixtures, a common practice in Brazilian farms to ensure productivity and reduce application costs. The main problem is the unexpected effects that different product combinations can cause to the application (Gazziero, 2015Gazziero DLP (2015) Mistura de agrotóxicos em tanque nas propriedades agrícolas do Brasil. Planta Daninha , 33:83-92.).
Neonicotinoid insecticides are remarkably effective against sucking insect pests and have been shown to effectively control the guava-psyllid (Barbosa et al., 2001Barbosa FR, Souza EA, Siqueira KMM, Moreira WA, Alencar JR & Haji FNP (2001) Eficiência e seletividade de inseticidas no controle de psilídeo (Triozoida sp.) em goiabeira. Pesticidas: Revista de Ecotoxicologia e Meio Ambiente, 11:45-52.). However, Lima & Gravina (2009Lima JOGD & Gravina GDA (2009) Failure of imidacloprid and thiacloprid to control the guava-psyllid, Triozoida limbata (Enderlein) (Hemiptera: Psyllidae). Ciência e Agrotecnologia, 33:1888-1891.) described a different effect on this pest, as they found that this product was inefficient on a high density of infestation after some time. Besides the decrease in the efficiency of the products, the association of different compounds within the spray tank can change their effectiveness (Khaliq et al., 2012Khaliq A, Matloob A, Mahmood S, Abbas RN & Khan MB (2012) Seeding density and herbicide tank mixtures furnish better weed control and improve growth, yield and quality of direct seeded fine rice. International Journal of Agriculture and Biology, 14:499-508.).
Tank mixing has been a very common practice in farming, especially with the addition of adjuvants and foliar fertilizers. However, the effects of these blends have not been well understood. Physicochemical properties are altered with these mixes and may influence the efficacy of plant protection products. The adjuvant composition and formulation can also affect physicochemical characteristics of the spray, mainly pH, surface tension, and viscosity (Cunha & Alves, 2009Cunha JPAR & Alves GS (2009) Características físico-químicas de soluções aquosas com adjuvantes de uso agrícola. Interciência, 34:655-659.), which justifies the need for further studies on the behavior of these properties in relation to certain mixtures and the possible biological effects.
The penetration and physiological effect of leaf-applied nutrient sprays involves a series of intricate mechanisms ranging from the mode of application to the physicochemical characteristics of the solution, the prevailing environmental conditions, or the target plant species. There are many processes involved, which make it difficult to develop new strategies to optimize the efficiency of foliar sprays under different conditions during the growing of the crops (Fernandez & Eichart, 2009Fernández V & Eichert T (2009) Uptake of hydrophilic solutes through plant leaves: current state of knowledge and perspectives of foliar fertilization. Critical Review in Plant Science, 28:36-68.).
Adjuvants are added to the mixture to enhance the efficiency of the solution in different ways. They can improve greately the spreading of the droplet and the wetting of the spray on the target (Cunha et al. 2010ACunha JPAR, Bueno MR & Ferreira MC (2010A) Espectro de gotas de pontas de pulverização com adjuvantes de uso agrícola. Planta Daninha, 28:1153-1158.), as well as influence the penetration through the cuticle (Wang & Liu, 2007Wang CJ & Liu ZQ (2007) Foliar uptake of pesticides - present status and future challenge. Pesticide Biochemistry and Physiology, 87:01-08.). However, despite of the advantages of adjuvants, more information is needed about their association with foliar fertilizers and the effects on the efficiency of biological molecules such as pesticides.
Therefore, the objectives of this work were to evaluate the effect of tank mixing an organosilicon adjuvant with a manganese foliar fertilizer on effectiveness of the insecticide imidacloprid in the control of T. limbata in guava trees.
MATERIAL AND METHODS
Experimental site
The present work was carried out in a guava orchard (P. guajava L.), cultivar ‘Paluma’, at “Água Limpa” experimental farm (19°6'16,49"S and 48°20'54,38"W; 795 m altitude), belonging to the Universidade Federal de Uberlândia (UFU), Uberlândia - MG - Brazil. According to Köppen classification, the area is characterized as Aw (tropical, hot humid area with cold and dry winter).
A production area was selected with 80 plants (nine years old) spaced 5.0 m between rows and 3.0 m between plants. The total experimental area was about 1000 m2. The experimental plot consisted of four consecutive trees in the same row with each tree subdivided into 4 quadrants (Q1, Q2, Q3 and Q4). The net plot consisted of the two inner trees and the borders were formed by the first and the fourth trees. Each block was divided in 5 plots arranged linearly.
The treatments consisted of applications of 600 L ha−1 of the mixture at 0.46 km h−1, in duplicate (two times of application). Q1 and Q3 were allocated along the row and Q2 and Q4 were across the row (Figure 1). During the treatments, a plastic protection was used to avoid product drift to adjacent plots in the direction of application.
of the experimental plot used for the application of the treatments and the direction of application used in the subplots.
The first period (1st application) was conducted in the 2017/2018 harvest, in December 14th 2017, a period of high infestation of guava psyllid. The second period was in April 13th 2018 (2nd application), after harvesting the fruits, the experiment was repeated in the same area, following the same methodology.
Treatments
Treatment solutions were prepared with a manganese salt (Manganese sulfate - MnSO4), an adjuvant (polyether-polymethyl siloxane copolymer), and an insecticide (Imidacloprid), at the Agricultural Mechanization Laboratory (LAMEC), of the Federal University of Uberlândia (UFU). The product specifications are presented in Table 1.
Treatments were as follows: T1 - Imidacloprid (Imid.); T2 - Imid. + Polyether-polymethyl siloxane copolymer (Sil.); T3 - Imid. + MnSO4; T4 - Imid. + Sil. + MnSO4; and T5 - Control (no application).
Experimental conduction
The applications were performed using a pneumatic backpack sprayer (Stihl® SR450, 14 L tank with a 2900W engine). Droplets are formed by action of the wind and as a function of the setting of the orifice, which is the output of the spray (not using a hydraulic nozzle). The sprayer was run at half engine speed to reduce drift and overload, with a flow rate of 1.45 L min-1.
The treatments were applied at right angles to the line of rows (Q2 and Q4 received direct applications) on both sides of the tree (Figure 1), and at a distance of approximately 1.5 m between the sprayer and the tree. To avoid contamination of plots, a plastic canvas was used during the applications to cover the adjacent area. After the applications, the samples were collected (leaves and petri dishes) and analyzed at LAMEC.
During the experiments, the environmental conditions were monitored. Data on the first period were: temperature (°C) between 23.7 - 27.5; humidity (%) between 60.5 - 70.0; and wind speed 4.5 - 11.7 (km h-1). Data on the second period were: temperature (°C) between 23.9 - 29.1; humidity (%) between 55.6 - 75.6; and wind speed 0.1 - 5.6 (km h-1).
Evaluations
Physicochemical evaluations
Different tank mixes were prepared with the pesticide to evaluate the following physical-chemical characteristics: density, pH, electric conductivity (EC), viscosity (Visc.), and surface tension (ST). As a way of comparison, distilled water was used as negative control. The procedures of the evaluations are described elsewhere (Cunha et al., 2010BCunha JPAR, Alves GS & Reis EF (2010B) Efeito da temperatura nas características físico-químicas de soluções aquosas com adjuvantes de uso agrícola. Planta Daninha , 28:665-672.). The evaluations of physical-chemical properties were carried out at LAMEC.
Application technology evaluation
To evaluate the application technology, each plot was formed by four trees and each tree was subdivided into four quadrants.
The net plot consisted of the two inner trees, from which we collected two leaves per quadrant, resulting in 16 leaves per plot. The leaves were collected immediately after the applications on the middle third of the trees, in the middle of the crown.
The evaluations of leaf deposition and the losses to the soil were described elsewhere (Tavares et al., 2017Tavares RM, Cunha JPAR, Alves TC, Bueno MR, Silva SM & Zandonadi CHS (2017) Electrostatic spraying in the chemical control of Triozoida limbata (Enderlein) (Hemiptera: Triozidae) in guava trees (Psidium guajava L.). Pest management science, 73:1148-1153.).
Pest evaluation
To check the psyllid infestation level, the area was sampled on the day before the first application. The evaluations were performed on days 7, 12, and 14 after the application (Daa).
The two inner trees in the plot were sampled for efficacy, as the first and the last tree were considered as borders. The damage threshold of guava psyllids was the point at which 30% of the leaves were damaged, justifying the spraying for pest management in the area. The procedures for sample evaluations were described by Tavares et al. (2017Tavares RM, Cunha JPAR, Alves TC, Bueno MR, Silva SM & Zandonadi CHS (2017) Electrostatic spraying in the chemical control of Triozoida limbata (Enderlein) (Hemiptera: Triozidae) in guava trees (Psidium guajava L.). Pest management science, 73:1148-1153.).
Psyllid nymphs were counted on each leaf, and the mean of the plot was calculated using a digital microscope (Dino-lite pro model: AM - 413ZT) with a 200x magnification,
+Experimental design and statistical analysis
The experiment was arranged in the split-plot randomized block design, with five treatments and four replications. The treatments were assigned to the plots and the quadrants (Q1, Q2, Q3 and Q4) to the subplots. Data were subjected to Shapiro Wilk’s normality test for normal distribution of errors and Levene's test for homogeneity of variances, at 0.01 of significance.
The “F” test was performed to determine levels of significance at 0.05 and 0.01 for the analysis of variance. When the tests detected significance, the means were compared by the Scott-Knott’s test at 0.05 level of probability. When necessary data was transformed by √(x+1). All analyses were performed using the SPSS software.
RESULTS AND DISCUSSION
Physicochemical evaluations
The physicochemical characteristics of the treatments changed as a function of each product added (Table 2). The adjuvant caused no change in EC in the treatment with only insecticide and provided a small increase in pH. On the other hand, the foliar fertilizer reduced the pH and increased EC significantly. The density increased as a function of the addition of the products to the mixture, however, the changes were small. Andrade et al. (2013Andrade DJ, Ferreira MC & Fenólio LG (2013) Compatibilidade entre acaricidas e fertilizantes foliares em função de diferentes águas no controle do ácaro da leprose dos citros Brevipalpus phoenicis. Revista Brasileira de Fruticultura, 35:39-50.) found that some of these characteristics, mainly the pH, were influenced by the addition of foliar fertilizers.
The decrease in the pH of the spray mixture after the addition of MnSO4 is due to the dissociation of this compound, releasing SO4 -2 ions in the solution, which in turn provides H + ions, making the pH acidic. The increase in EC due to the addition of MnSO4 in the two treatments can also be explained by the dissociation of the inorganic salt that provides free H+ ions into the solution and are capable of conducting the electric charge.
For certain herbicide molecules, the decrease in the pH of the spray solution is crucial to maintain product efficacy. During the mixing process, the presence of inorganic salts such as foliar fertilizers cause incompatibility between molecules and loss of efficacy (Bernards et al., 2005Bernards M, Thelen K, Penner D, Muthukumaran R & McCracken J (2005) Glyphosate interaction with manganese in tank mixtures and its effect on glyphosate absorption and translocation. Weed Science, 53:787-794.), which is not yet relevant to other products, like neonicotinoids.
The product mixture reduced the surface tension in relation to water, with emphasis on the foliar fertilizer and the adjuvant, which resulted in the lowest values. The foliar fertilizer had the potential to decrease the surface tension, without using the adjuvant. Again, the dissociation of MnSO4 can explain the reduction in the surface tension. This inorganic compound can reduce the strong intermolecular interactions inside the water molecule during the hydro-dissociation, because it provides free H+ ions to solution.
According to Iost & Raetano (2010Iost CAR & Raetano CG (2010) Tensão superficial dinâmica e ângulo de contato de soluções aquosas com surfatantes em superfícies artificiais e naturais. Engenharia Agrícola, 30:670-680.) silicon adjuvants were more efficient in reducing the surface tension. This reduction was more pronounced due the association with MnSO4, which promotes greater spray droplet spread on the target and, therefore, can favor its absorption.
The viscosity increased with the addition of the fertilizer and the adjuvant to the insecticide. Higher viscosity of the spray solution results in larger droplets. Thus, the addition of ions to the solutions had directly influence on these characteristics.
Application technology
The variables analyzed for application technology showed that foliar deposition was significant (P < 0.05) for treatments and quadrants only for the first period of application (Table 3). However, losses to the soil were significant between treatments and quadrants for both applications.
The tracer deposition in the first application was the highest in the treatment with only insecticide, differently from the others, that presented the same deposition pattern (Table 4).
When the products were added to the insecticide, the surface tension was drastically reduced (Table 1). When the leaves were sprayed, the droplets could stay over the leaf, adhered, spread, or even runoff. According to Van Zyl et al. (2010Van Zyl SA, Brink JC, Calitz FJ, Coertze S & Fourie PH (2010) The use of adjuvants to improve spray deposition and Botrytis cinerea control on Chardonnay grapevine leaves. Crop Protection, 29:58-67.), depending of the surfactant concentration, the values of surface tension could become lower and then cause excessive spreading with droplet runoff. This could justify the lower deposition values in the treatments that had more of the other products than the insecticide.
The deposition was higher in Q2 and Q4 as expected (Table 5), mainly because the direction of the application and the leaves of Q1 and Q3 overlapped by leaves of the border trees. Tavares et al. (2017Tavares RM, Cunha JPAR, Alves TC, Bueno MR, Silva SM & Zandonadi CHS (2017) Electrostatic spraying in the chemical control of Triozoida limbata (Enderlein) (Hemiptera: Triozidae) in guava trees (Psidium guajava L.). Pest management science, 73:1148-1153.) found similar results when evaluated electrostatic application in guava trees. The authors observed that the quadrants that received direct application had more deposition than those that did not receive it.
At the first application, the treatments presented almost the same pattern of spray losses to the soil in all quadrants, except the lowest loss in Q2, for the treatment with all products and the treatment Imid + MnSO4 (Table 6). Then as well, at the second application, Imid +Sil and the treatment with all products presented similar losses to the soil in all quadrants. The treatments with only the insecticide had more losses in Q1, but when the adjuvant was added, the losses became higher in Q3 and Q4 (Table 5).
Tavares et al. (2017Tavares RM, Cunha JPAR, Alves TC, Bueno MR, Silva SM & Zandonadi CHS (2017) Electrostatic spraying in the chemical control of Triozoida limbata (Enderlein) (Hemiptera: Triozidae) in guava trees (Psidium guajava L.). Pest management science, 73:1148-1153.) found similar results when evaluating the standard application in guava trees with the same equipment and spray volume.
Pest evaluation
The variables analyzed to evaluate pest attack (nymph number and infestation level) were significant (P < 0.05) as a function of the different evaluation period as Table 7 shows.
At the first application, the percentage of infestation at 7 Daa was higher in Q1 and Q3, whereas at the second application, the percentage of infestation was higher in Q2 and Q4. These results have a relation with the deposition of the tracer in the quadrants, showing that, in this case, the quadrants with more tracer deposition presented a reduction in the percentage of infestation.
In the second period of the experiment, the plants reduced the number of leaves because of the climate and the overlapping of the branches of the neighboring trees in Q1 and Q3, which had not happen in the first period because the size of the trees (Table 8).
Different from the infestation level, the number of nymphs was greater in Q2 and Q4 in the first period of application (Table 9). At the second application, at 0 Daa, nymph distributions were similar in all quadrants. However, at 7 Daa the number of nymphs reduced in Q1 and Q3 and increased in Q2 and Q4 (Table 9).
Marcelino & Barbosa (2016Marcelino MCS & Barbosa JC (2016) Spatial distribution of adults of Triozoida limbata (Enderlein) (Hemiptera: Triozidae) in guava plants. Neotropical entomology, 45:123-128.) found that T. limbata adults showed moderate to high aggregated distribution in all development phases of guava, independent of the average size of the population, which could justify the higher number of nymphs in the quadrants 2 and 4, which had more leaves.
Figure 2 shows that the number of nymphs where similar after the application of the treatments. At 14 Daa, the number of nymphs had increased in the control treatment, differing from the others that had the same averages, showing that the insecticide was effective against the insects up to that time.
Mean nymph number after the treatment applications (1stperiod).ns;* - non significant; significant at 0.05.; Means followed by the same letter are not significantly different by the Scott Knott’s test (p ≤ 0.05).
At the second application, the number of nymphs fluctuated up to 12 Daa. It was only at 14 Daa that the treatments presented difference from the control. In this application, the number of nymphs had been reduced from the treatments with the insecticide, except when mixing it with the foliar fertilizer, showing a difference between the others and the control as well (Figure 3).
Mean nymph number after the treatment applications (2nd period).ns;* - non significant; significant at 0.05.; Means followed by the same letter are not significantly different by the Scott Knott’s test p ≤ 0.05.
Galli et al. (2014Galli JA, Michelotto MD, Soares MBB, Martins ALM & Fischer IH (2014) Damage Caused by Psyllids and Influence of Climatic Factors in Brazilian Accesses of Guava Trees Cultivated in Organic System. American Journal of Plant Sciences, 05:1432.) found that cultivars “Paluma” and “Rica” were the most attacked by psyllid compared with different commercial accesses and others in test. The percentage of damage were higher than 50% during all the experiment.
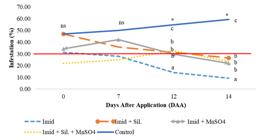
The damage threshold of 30% was reached and the applications were necessary in both periods (Figures 4 and 5). These levels reduced at 12 and 14 Daa, except for the control (Figure 4) in the first period and from day 7 to 14 Daa in the second application (Figure 5). In both periods, the damage threshold was reduced to below the recommended level for another application, which justify only one application in each period.
Leaves infested (%) by psyllid in guava trees (1st period).ns;* - non significant; significant at 0.05.; Means followed by the same letter are not significantly different by the Scott Knott’s test (p ≤ 0.05).
Leaves infested (%) by psyllid in guava trees (2nd period).ns;* - non significant; significant at 0.05.; Means followed by the same letter are not significantly different by the Scott Knott’s test p ≤ 0.05.
The first application reduced the percentage of infestation in all treatments, but the treatment with only insecticide reduced it more than the others (Figure 4). However, in the second application, all the treatments reduced the infestation to the same level, becoming only different from the control (Figure 5). As we can see in Table 4, the foliar deposition of the treatment with only insecticide was greater than the other treatments, which explains the difference between the treatments in the first application.
When the insecticide imidacloprid was applied on high density levels (higher than 50%) and in the interval of 15 days between applications, there was no reduction of the infestation levels above the damage threshold (Lima & Gravina, 2009Lima JOGD & Gravina GDA (2009) Failure of imidacloprid and thiacloprid to control the guava-psyllid, Triozoida limbata (Enderlein) (Hemiptera: Psyllidae). Ciência e Agrotecnologia, 33:1888-1891.).
CONCLUSION
The addition of foliar fertilizer in the mixture reduced the pH and surface tension and increased the electric conductivity and viscosity of the insecticide solutions. The silicon adjuvant reduced the surface tension and increased the viscosity and the pH. The quadrants that received direct application (2 and 4) presented higher spray deposition. All the treatments and quadrants presented almost the same spray losses to the soil.
The number of nymphs as well as the infestation level decreased with the treatment applications. The tank mixture of the adjuvant organosilicon and the manganese foliar fertilizer had no influence on the efficacy level of the insecticide.
ACKNOLEDGEMENTS
The authors thank CNPq, CAPES, and FAPEMIG for the financial support.
REFERENCES
- Andrade DJ, Ferreira MC & Fenólio LG (2013) Compatibilidade entre acaricidas e fertilizantes foliares em função de diferentes águas no controle do ácaro da leprose dos citros Brevipalpus phoenicis. Revista Brasileira de Fruticultura, 35:39-50.
- Barbosa FR & Lima MF (2010) A cultura da goiaba. 2ª ed. Brasília, Embrapa Informação Tecnológica. 180p.
- Barbosa FR, Souza EA, Siqueira KMM, Moreira WA, Alencar JR & Haji FNP (2001) Eficiência e seletividade de inseticidas no controle de psilídeo (Triozoida sp.) em goiabeira. Pesticidas: Revista de Ecotoxicologia e Meio Ambiente, 11:45-52.
- Bernards M, Thelen K, Penner D, Muthukumaran R & McCracken J (2005) Glyphosate interaction with manganese in tank mixtures and its effect on glyphosate absorption and translocation. Weed Science, 53:787-794.
- Colombi CA & Galli JC (2009) Dinâmica populacional e evolução de dano de Triozoida limbata (Hemiptera: Psillydae) em goiabeira, em Jaboticabal, SP. Ciência Agrotecnologia, 33:412-416.
- Cunha JPAR & Alves GS (2009) Características físico-químicas de soluções aquosas com adjuvantes de uso agrícola. Interciência, 34:655-659.
- Cunha JPAR, Bueno MR & Ferreira MC (2010A) Espectro de gotas de pontas de pulverização com adjuvantes de uso agrícola. Planta Daninha, 28:1153-1158.
- Cunha JPAR, Alves GS & Reis EF (2010B) Efeito da temperatura nas características físico-químicas de soluções aquosas com adjuvantes de uso agrícola. Planta Daninha , 28:665-672.
- Farias WC, Câmara FMM, Silva FSO, Mendonça V, Pereira GA & Leite GA (2017) Qualidade de frutos da goiabeira cv. Paluma submetida a podas de frutificação em diferentes épocas no município de Mossoró-RN. Nativa, 05:05-08.
- Fernández V & Eichert T (2009) Uptake of hydrophilic solutes through plant leaves: current state of knowledge and perspectives of foliar fertilization. Critical Review in Plant Science, 28:36-68.
- Galli JA, Michelotto MD, Soares MBB, Martins ALM & Fischer IH (2014) Damage Caused by Psyllids and Influence of Climatic Factors in Brazilian Accesses of Guava Trees Cultivated in Organic System. American Journal of Plant Sciences, 05:1432.
- Gallo D, Nakano O, Silveira Neto S, Carvalho RPL, Batista GC, Berti Filho E, Parra JRP, Zuchi RA, Alves SB, Vendramim JD, Marchini LC, Lopes JRS & Omoto C (2002) Entomologia agrícola. Piracicaba, FEALQ. 920p.
- Gazziero DLP (2015) Mistura de agrotóxicos em tanque nas propriedades agrícolas do Brasil. Planta Daninha , 33:83-92.
- Iost CAR & Raetano CG (2010) Tensão superficial dinâmica e ângulo de contato de soluções aquosas com surfatantes em superfícies artificiais e naturais. Engenharia Agrícola, 30:670-680.
- Khaliq A, Matloob A, Mahmood S, Abbas RN & Khan MB (2012) Seeding density and herbicide tank mixtures furnish better weed control and improve growth, yield and quality of direct seeded fine rice. International Journal of Agriculture and Biology, 14:499-508.
- Lima JOGD & Gravina GDA (2009) Failure of imidacloprid and thiacloprid to control the guava-psyllid, Triozoida limbata (Enderlein) (Hemiptera: Psyllidae). Ciência e Agrotecnologia, 33:1888-1891.
- Marcelino MCS & Barbosa JC (2016) Spatial distribution of adults of Triozoida limbata (Enderlein) (Hemiptera: Triozidae) in guava plants. Neotropical entomology, 45:123-128.
- Tavares RM, Cunha JPAR, Alves TC, Bueno MR, Silva SM & Zandonadi CHS (2017) Electrostatic spraying in the chemical control of Triozoida limbata (Enderlein) (Hemiptera: Triozidae) in guava trees (Psidium guajava L.). Pest management science, 73:1148-1153.
- Van Zyl SA, Brink JC, Calitz FJ, Coertze S & Fourie PH (2010) The use of adjuvants to improve spray deposition and Botrytis cinerea control on Chardonnay grapevine leaves. Crop Protection, 29:58-67.
- Wang CJ & Liu ZQ (2007) Foliar uptake of pesticides - present status and future challenge. Pesticide Biochemistry and Physiology, 87:01-08.
Publication Dates
-
Publication in this collection
03 Oct 2019 -
Date of issue
Jul-Aug 2019
History
-
Received
29 Aug 2018 -
Accepted
15 May 2019