Abstracts
The objective of the present study was to determine the prevalence of certain mycoplasma species, i.e., Mycoplasma hominis, Ureaplasma urealyticum and Mycoplasma penetrans, in urethral swabs from HIV-1 infected patients compared to swabs from a control group. Mycoplasmas were detected by routine culture techniques and by the Polymerase Chain Reaction (PCR) technique, using 16SrRNA generic primers of conserved region and Mycoplasma penetrans specific primers. The positivity rates obtained with the two methods were comparable. Nevertheless, PCR was more sensitive, while the culture techniques allowed the quantification of the isolates. The results showed no significant difference (p < 0.05) in positivity rates between the methods used for mycoplasma detection.
Mycoplasmas; Mycoplasma penetrans; HIV-1; PCR
O objetivo do presente estudo foi determinar a prevalência de determinadas espécies de micoplasmas, tais como, Mycoplasma hominis, Ureaplasma urealyticum e Mycoplasma penetrans, em swabs uretrais de pacientes infectados com HIV-1 comparando com um grupo controle. Micoplasmas foram detectados por técnicas padrão de cultivo e pela reação de polimerase em cadeia para a qual foram utilizados "primers" genéricos obtidos da região conservada 16SrRNA e "primers" específicos para Mycoplasma penetrans. A positividade obtida nos dois métodos foi comparável. Contudo, o PCR mostrou ser mais sensível nas condições empregadas enquanto que o cultivo permitiu a quantificação dos isolados. Os resultados demonstraram não haver diferenças significantes (p < 0,05) nas taxas de positividade entre os métodos empregados para a detecção dos micoplasmas.
Detection of mycoplasmas in urethral swabs from HIV-1 infected patients and control individuals using culture techniques and polymerase chain reaction
Regina Ayr Florio da CUNHA(1 Recebido para publicação em 23/07/1997 Aceito para publicação em 05/01/1998 ), Kioko TAKEI(1 Recebido para publicação em 23/07/1997 Aceito para publicação em 05/01/1998 ), Adelaide José VAZ(1 Recebido para publicação em 23/07/1997 Aceito para publicação em 05/01/1998 ) & Caio ROSENTHAL(2 Recebido para publicação em 23/07/1997 Aceito para publicação em 05/01/1998 )
SUMMARY
The objective of the present study was to determine the prevalence of certain mycoplasma species, i.e., Mycoplasma hominis, Ureaplasma urealyticum and Mycoplasma penetrans, in urethral swabs from HIV-1 infected patients compared to swabs from a control group. Mycoplasmas were detected by routine culture techniques and by the Polymerase Chain Reaction (PCR) technique, using 16SrRNA generic primers of conserved region and Mycoplasma penetrans specific primers. The positivity rates obtained with the two methods were comparable. Nevertheless, PCR was more sensitive, while the culture techniques allowed the quantification of the isolates. The results showed no significant difference (p < 0.05) in positivity rates between the methods used for mycoplasma detection.
Keywords: Mycoplasmas; Mycoplasma penetrans; HIV-1; PCR.
INTRODUCTION
Infection with human immunodeficiency virus type 1 (HIV-1) is characterized by complex physiopathological alterations in which the immunologic system presents a significant decrease in T-helper cell-dependent cellular immune response. Clinical signs and symptoms of AIDS appear after an asymptomatic period ranging from months to years. During this period, only a small number of peripheral blood CD4+ lymphocytes are infected by HIV-1. The reasons for the remarkable variation in the latent period of the virus are unknown. Perhaps there are various factors and/or cofactors to be considered. Some of the widely known factors affecting the period of latency are genetic susceptibility, patient age, viral strain diversity, route of contamination and the characteristics of the inoculum1.
Recent studies have suggested the possibility that certain mycoplasma species may act as cofactors acelerating AIDS progression1. Mycoplasmas are microorganisms that evolved regressively (by genome reduction) from gram-positive bacterial ancestors with a low DNA content of guanine plus cytosine. Many of their properties, such as small genome size, small number of rRNA operons and tRNA genes, lack of a cell wall, fastidious growth and limited metabolic activities, are seen as the result of this evolution. The class Mollicutes comprises approximately 150 distinct species including more than 92 Mycoplasma species found in humans, animals, insects and plants. Among those, 16 species have been isolated from human tissues or fluids, and 15 of them have been isolated and constitute the normal human mollicute flora. While Mycoplasma pneumoniae has been definitively distinguished as a human pathogen, other species have been isolated in significant quantities from patients and healthy individuals. The role of these mycoplasmas in human diseases has intrigued several researchers2, 4.
The presence of Mycoplasma fermentans, Mycoplasma pirum and Mycoplasma penetrans in patients with AIDS and its association with the progression of HIV infection is drawing attention to the possible mechanisms by which mycoplasmas may influence the pathogenesis of HIV-1. This pathogenesis may be due to the activation of the immune system by the mycoplasma through the production of one or more superantigens, contributing to the oxidative "stress" observed in HIV-1-infected patients6, 8, 11.
The main purpose of the continuous reactivation of the immune system is to keep HIV replication. Considering that mycoplasmas present a series of immunomodulatory effects, the additional activation of immunocompetent cells by these microorganisms would assure not only the maintenance but also the increase of viral replication.
During HIV-1 infection, one or more superantigens may be involved in activation of the immune system. The role of superantigens in the development of human diseases is still an open question. HIV may code for this superantigen, but the latter may also be expressed by various microorganisms acting as cofactors, among them the mycoplasmas3, 13.
Recent in vitro studies have established the capacity of Mycoplasma penetrans to activate peripheral T-lymphocytes both from healthy and HIV positive individuals and to cause oxidative stress and apoptosis in peripheral blood mononuclear cells15. Considering the high seroprevalence found for this mycoplasma in HIV-positive individuals, the objective of the present study was to establish the prevalence of Mycoplasma hominis, Ureaplasma urealyticum and Mycoplasma penetrans in a group of HIV infected patients, and to compare the results with those obtained for a control group11.
MATERIAL AND METHODS
Patients
A group of 24 male patients was selected among those who sought the Infectious Diseases Service of Hospital Servidor Público Estadual of São Paulo, all of them presenting signs and symptoms of HIV-1 (AIDS) infection and the following characteristics:
Epidemiological and clinical history compatible with HIV (AIDS) infection, in
stages 3 to 6 of the Walter Reed Army Institute classification.
Positive serological tests in two samples collected on different days.
CD4 counts below 500 mm3.
No antibiotic therapy during the previous 10 days.
Control group
The control group consisted of 24 male volunteers presenting the following characteristics:
Negative serological tests in two samples collected on different days.
Absence of signs and symptoms of sexually transmissible diseases (STD).
No antibiotic therapy during the previous 10 days.
Serological tests employed for anti-HIV antibody detection
Gelatinous Particle Agglutination Test (PA) sensitized with HIV-1 antigen
obtained from a purified viral culture lysate (Fujirebio Inc., Japan).
Immunoenzymatic Solid Phase Test (ELISA) with recombined HIV-1 antigen
(Abbott Laboratories Inc., USA).
Western Blot Test (Dupont Co., USA) with clinical interpretation according to criteria recommended by the Food and Drug Administration (USA).
Determination of lymphocyte subpopulations
CD4 counts by the direct immunofluorescence method using monoclonal antibody
(Ortho Diag. Sy. Inc., USA).
Samples
Urethral swabs
The samples were collected with the aid of neutral alginate swabs delicately introduced into the urethra of each patient. The swabs were placed in 1 ml of saline solution and homogenized.
Clinical sample culture
The identification system used for the detection of Mycoplasma hominis and Ureaplasma urealyticum was the Mycofast "All in" from International Mycoplasma (France) using standard mycoplasma culture methods for comparative technology (Shepard mod.)16, 17. The microorganisms were quantitated in the samples by microtitration12. Mycoplasma penetrans was detected using SP-4 medium19. Parts of the collected samples were submitted to mycoplasma culture and part was treated with proteinase K for use in the PCR technique.
Sample treatment with proteinase K for use in the PCR technique
Three hundred µl of the sample and 700 µl of the washing solution (10 mM Tris-HCl, pH 8.3; 50 mM KCl; and 1.5 mM MgCl2 filtered through a 22 µm Millipore membrane) were transferred to an Eppendorf tube and the mixture was centrifuged for 15 min at 13,500 rpm at 4°C. The supernatant was discarded and the sediment was added to: 25 µl of buffer solution A (10 mM Tris-HCl, pH 8.3; 100 mM KCl; 2.5 mM MgCl2) plus 25 µl of the mixture: 500 µl of buffer solution B (10 mM Tris-HCl, pH 8.3; 2.5 mM MgCl2; 1% Tween 20; 1% Triton X-100) plus 3 µl of K proteinase (120 µg/ml). The mixture was incubated at 60°C for 1 hour, and then at 100°C for 10 minutes.
The samples were then frozen at 20°C until the time for PCR.
Amplification of sample DNA by PCR
All the samples were submitted to amplification using the MGSO and GPO1 primers obtained from the conserved region 16SrRNA. Forty µM of each primer were added to each reaction tube mixed in 50 mM KCl, 10 mM Tris-HCl, pH 8.3; 1.5 mM MgCl2, 0.1% gelatin 0.1 (Difco), 2.5 × 105 trimethyl ammonia chloride (Aldrich Milwaukee, USA), 0.4 µl mM dNTP; 1 unit of Taq polymerase and two drops of mineral oil. Two µl of each sample were added through the oil, to a final volume of 50 µl per reaction tube. The samples were heated at 95°C for 15 minutes, followed by 30 cycles at 95°C for 30 seconds/58°C for 1 minute and 30 seconds/72°C for 1 minute and 30 seconds and a final cycle of 10 minutes at 72°C, and then incubated at 4°C until the time for use14.
Analysis of amplified DNA
Twenty µl of the amplified products were submitted to electrophoresis on 0.8% agarose gel, into which 3 µl of ethidium bromide were incorporated to develop the bands. The positive control was DNA from the Mycoplasma penetrans species, and the negative control was the reaction mixture. The DNA from l phage digested with the enzyme HindIII was used as molecular weight marker.
Those samples that were positive to the PCR with generic primers were submitted to a new PCR using the oligonucleotides Mp1 and Mp2 specific for the Mycoplasma penetrans species as primers. The specificity of the observed bands was tested by treatment of the amplified product with Ava II restriction enzyme.
Statistical Analysis
Data were analyzed statistically by the X2 test of Pearson, with the level of significance set at a = 5%18.
RESULTS
The results obtained by culture technique and by PCR using generic primers were as follows. Of the 24 samples from patients with AIDS, 9 (37%) were positive for mycoplasmas by the culture technique and 13 (54%) by PCR. For the control samples, the results were 7 (29%) and 9 (37%), respectively.
In both groups all samples found to be positive by the culture technique were also found to be positive by PCR, while four samples from the study group and two from the control group were found to be positive only by PCR.
Since the culture technique is the standard, the relative sensitivity of PCR compared with culture was 100% and its specificity was 88% (15/17). The comparative analysis of the positive results obtained by culture for both groups indicated that, using a Yates correction index for a = 5%, X2obtained < X2critic, from which we conclude that the differences were not statistically significant. For PCR, the difference in positivity between the samples from patients with AIDS and the control samples were not statistically significant.
The PCR technique proved to be more sensitive than the standard culture technique. The comparison was made using the quadruple table to determine X2 for a = 5%, leading to X2obtained > X2critic, which indicated significant differences in positivity between the two techniques, with higher values obtained by PCR.
Among the samples from the infected patients, 25% (6/24) contained Ureaplasma urealyticum and 8.33% (2/24) contained the association Ureaplasma urealyticum-Mycoplasma hominis. The results for the control group were 16.6% (4/24) and 12.5% (3/24), respectively. Mycoplasma penetrans was not detected by the culture methods, but was detected by PCR in four samples, two of them (8.33%) from the infected group and two (8.33%) from the control group. Among these four samples, one sample from the infected group indicated an association with Ureaplasma urealyticum, and one sample from the control group indicated the presence of the Ureaplasma urealyticum-Mycoplasma hominis association. The values obtained are shown in Figure 1.
The cases considered to be positive were those with titers (= concentration) of 104 CCU/ml or higher, as determined by microtitration and by the "Mycofast all in" kit, which presented identical results. The positivity rate was 8/24 (29.1%) for the infected group and 7/24 (10.8%) for the control group.
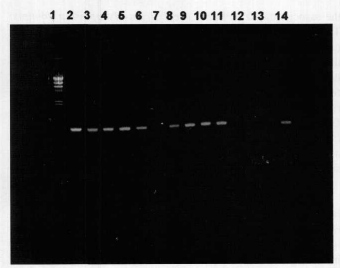
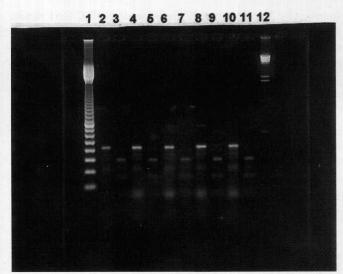
The results obtained by PCR are presented in Figures 2 and 3.
Treatment of the amplified product (410 bp) with restriction enzyme AvaII yielded two fragments of 269 bp and 141 bp. The results are illustrated in Figure 4.
DISCUSSION
The present study showed the presence of Mycoplasma hominis, Ureaplasma urealyticum and Mycoplasma penetrans in two groups, one consisting of AIDS patients and the other of control subjects. Although the presence and participation of Mycoplasma hominis and Ureaplasma urealyticum in diseases of the genitourinary tract have already been very well established, the objective of our study was to evaluate these findings in immunologically compromised patients.
The results allowed us to conclude that the species Ureaplasma urealyticum, Mycoplasma hominis and Mycoplasma penetrans were isolated at the same frequency from both study groups. These results did not agree with preliminary studies that revealed a significantly higher prevalence of Mycoplasma penetrans species in the urine of AIDS patients compared to controls10, 20.
Our findings suggest that Mycoplasma penetrans may be a common inhabitant of the genitourinary tract, especially in the urethra. The use of urethral swabs and the similar rates of isolation from the two groups indicated that Mycoplasma penetrans may be present only as a colonizer of the genitourinary tract, and its pathogenicity would depend on its concentration, as is the case also for Mycoplasma hominis and Ureaplasma urealyticum.
The evaluation of the techniques employed indicated that both the standard culture and the kit yielded comparable results, allowing the quantification of the isolates. The PCR technique revealed greater sensitivity for the generic detection of mycoplasmas14. The cost of mycoplasma PCR quantification precludes its use for routine diagnosis. Mycoplasma penetrans proved to be difficult to identify by culture, and could only be detected by PCR5.
It is easy to understand that the PCR technique is highly useful for the identification of the species of pathogenic mycoplasma (quantification unnecessary), because the speed of diagnosis would allow a more rapid introduction of therapy. In any case, further studies on the Mycoplasma penetrans species are needed, with the analysis of a larger number of samples.
RESUMO
Detecção de micoplasmas em swabs uretrais de pacientes infectados por HIV-1 e controles utilizando-se técnicas de cultivo e da reação de polimerase em cadeia (PCR)
O objetivo do presente estudo foi determinar a prevalência de determinadas espécies de micoplasmas, tais como, Mycoplasma hominis, Ureaplasma urealyticum e Mycoplasma penetrans, em swabs uretrais de pacientes infectados com HIV-1 comparando com um grupo controle. Micoplasmas foram detectados por técnicas padrão de cultivo e pela reação de polimerase em cadeia para a qual foram utilizados "primers" genéricos obtidos da região conservada 16SrRNA e "primers" específicos para Mycoplasma penetrans. A positividade obtida nos dois métodos foi comparável. Contudo, o PCR mostrou ser mais sensível nas condições empregadas enquanto que o cultivo permitiu a quantificação dos isolados. Os resultados demonstraram não haver diferenças significantes (p < 0,05) nas taxas de positividade entre os métodos empregados para a detecção dos micoplasmas.
ACKNOWLEDGEMENTS
We would like to thank Dr. Alain Blanchard of Institut Pasteur that kindly provided the M. penetrans strain and Ronaldo Milhim and Rosangela Verlengia for excellent technical assistance.
(1) Faculdade de Ciências Farmacêuticas da Universidade de São Paulo, São Paulo, Brazil.
(2) Hospital do Servidor Público Estadual de São Paulo, São Paulo, Brazil.
Correspondence to: Regina Ayr Florio da Cunha. Faculdade de Ciências Farmacêuticas, Universidade de São Paulo, Av. Prof. Lineu Prestes 580, bloco 17, 05508-900 São Paulo, SP, Brasil. Telefone: +5511-818-3662. Fax: +5511-813-2197.
- 1. BLANCHARD, A. & MONTAGNIER, L. AIDS: associated mycoplasmas. Ann. Rev. Microbiol., 48: 687-712, 1994.
- 2. BOVÉ, J. M. Molecular features of Millicutes. Clin. infect. Dis., 17(S1): S10-S31, 1993.
- 3. COLE, B. C. The immunobiology of Mycoplasma arthritidis and its superantigen MAM. Curr. Trop. Microbiol. Immunol., 174: 107-119, 1991.
- 4. CUNHA, R. A. F.; KOIFFMAN, C. P.; SOUZA, D. H. & TAKEI, K. Clastogenic effects of different Ureaplasma urealyticum serovars on human chromosomes. Braz. J. med. biol. Res., 30: 749-757, 1997.
- 5. DEGUCHI, T.; GILROY, C. B. & TAYLOR-ROBINSON, D. Failure to detect Mycoplasma fermentans, Mycoplasma penetrans, or Mycoplasma pirum in the urethra of patients with acute nongonococcal urethritis. Eur. J. clin. Microbiol. infect. Dis., 15: 169-171, 1996.
- 6. GRAU, O.; KOVACIC, R.; GRIFFAIS, R. & MONTAGNIER, L. Development of a seletive and sensitive polymerase chain reaction assay for the detection of Mycoplasma pirum FEMS Microbiol. Lett., 106: 327-334, 1993.
- 7. GRAU, O.; KOVACIC, R.; GRIFFAIS, R.; LAUNAY, V. & MONTAGNIER, L. Development of PCR based assays for the detection of two human mollicute species, Mycoplasma penetrans and Mycoplasma hominis Molec. cell. Probes, 8: 139-148, 1994.
- 8. GRAU, O.; SLIZEWICZ, B.; TUPPIN, P. et al. Association of Mycoplasma penetrans with human immunodeficiency virus infection. J. infect. Dis., 172: 672-681, 1995.
- 9. KATSENI, V. L.; GILROY, C. B.; RYAIT, B. K. et al. Mycoplasma fermentans in individuals seropositive and seronegative for HIV-1. Lancet, 341: 271-273, 1993.
- 10. LO, S-C; HAYES, M. M.; WANG, R. Y-H et al. Newly discovered mycoplasma isolated from patients infected with HIV. Lancet, 338: 1415-1418, 1991.
- 11. LO, S-C; HAYES, M. M.; KOTANI, H. et al. Adhesion onto and invasion into mammalian cells by Mycoplasma penetrans: a newly isolated mycoplasma from patients with AIDS. Mod. Path., 6: 276-280, 1993.
- 12. MASOVER, G. K.; NAMBA, M. & HAYFLICK, L. Cytotoxic effect of a T-strain Mycoplasma (Ureaplasma urealyticum) on cultured normal human cells (WI-38). Exp. Cell Res., 99: 363-374, 1976.
- 13. RINK, L. & KIRCHNER, H. Mycoplasma arthritidis derived superantigen. Chem. Immunol., 55: 137-145, 1992.
- 14. ROULLAND- DUSSOIX, D.; HENRY, A. & LEMERCIER, B. Detection of mycoplasmas in cell cultures by PCR: a one year study. J. Microbiol. Meth., 19: 127-134, 1994.
- 15. SASAKI, Y.; BLANCHARD, A.; WATSON, H. L. et al. In vitro influence of Mycoplasma penetrans on activation of peripheral T lymphocytes from healthy donors or human immunodeficiency virus-infected individuals. Infect. Immun., 63: 4277-4283, 1995.
- 16. SHEPARD, M. C. & LUNCEFORD, C. D. Differential agar medium (A7) for identification of Ureaplasma urealyticum (human T mycoplasma) in primary cultures of clinical material. J. clin. Microbiol., 3: 625-631, 1976.
- 17. SHEPARD, M. C. Standard fluid medium U10 for cultivation and maintenance of Ureaplasma urealyticum Int. J. system. Bact., 24: 160-171, 1974.
- 18. SOUNIS, E. Introduçăo ŕ teoria da decisăo estatística. In: SOUNIS, E. Bioestatística Săo Paulo, Atheneu, 1985. p. 207-234.
- 19. TULLY, J. G.; TAYLOR-ROBINSON, D.; ROSE, D. L.; COLE, R. M. & BOVÉ, J. M. Mycoplasma genitalium, a new species from human urogenital tract. Int. J. system. Bact., 33: 387-396, 1983.
- 20. WANG, R.Y.-H.; SHIH, J.W.-K.; GRANDINETTI, T. et al. High frequency of antibodies to Mycoplasma penetrans in HIV-infected patients. Lancet, 340: 1312-1316, 1992.
Publication Dates
-
Publication in this collection
16 June 1999 -
Date of issue
Jan 1998
History
-
Received
23 July 1997 -
Accepted
05 Jan 1998