Abstracts
The involvement of the gastrointestinal tract in the co-infection of HIV and Leishmania is rarely reported. We report the case of an HIV-infected adult man co-infected with a disseminated form of leishmaniasis involving the liver, lymph nodes, spleen and, as a feature reported for the first time in the English literature, the pancreas. Light microscopy showed amastigote forms of Leishmania in pancreatic macrophages and immunohistochemical staining revealed antigens for Leishmania and also for HIV p24. Microscopic and ultrastructural analysis revealed severe acinar atrophy, decreased zymogen granules in the acinar cytoplasm and also nuclear abnormalities such as pyknosis, hyperchromatism and thickened chromatin. These findings might correspond to the histologic pattern of protein-energy malnutrition in the pancreas as shown in our previous study in pancreas with AIDS and no Leishmania. In this particular case, the protein-energy malnutrition may be due to cirrhosis, or, Leishmania or HIV infection or all mixed. We believe that this case represents the morphologic substratum of the protein energy malnutrition in pancreas induced by the HIV infection. Further studies are needed to elucidate these issues.
Visceral Leishmaniasis; Pancreas; AIDS; Opportunistic infection; Malnutrition
O comprometimento do trato gastrointestinal é uma manifestação incomum da co-infecção HIV-Leishmaniose Visceral (LV). Descreveremos o primeiro caso de comprometimento pancreático por LV disseminada na AIDS, como primeira manifestação oportunista (além do comprometimento do figado, baço, linfonodos e rins). A necropsia revelou pâncreas normal ao exame macroscópico. A microscopia ótica do pâncreas revelou: atrofia pancreática severa, diminuição dos grânulos de zimogênio, anormalidades nucleares discretas e presença de formas amastigotas de Leishmania em macrófagos intersticiais, além do antígeno da LV e HIV-p24 (imunohistoquímica-IH). À microscopia eletrônica: pâncreas com grânulos de zimogênio diminuídos, proliferação do retículo granuloso, mitocôndrias aumentadas, evidenciando o aspecto já observado por nós em pâncreas de pacientes com AIDS sem LV. Presença de formas amastigotas da Leishmania no fígado, baço, linfonodos além do antígeno da LV no rim (IH). As alterações histológicas pancreáticas são sugestivas de desnutrição protéico calórica, que pode estar representada neste caso pela desnutrição induzida pela cirrose, pela leishmaniose ou pela AIDS. Acreditamos ser esse último o principal responsável pelas alterações histológicas encontradas. No entanto estudos futuros são necessários para elucidar esses achados.
CASE REPORT
PANCREATIC INVOLVEMENT IN CO-INFECTION VISCERAL LEISHMANIASIS AND HIV: HISTOLOGICAL AND ULTRASTRUCTURAL ASPECTS
Ethel Zimberg CHEHTER(1(1) Disciplin of Clinic Gastroenterology, Department of Gastroenterology, Medical School of the São Paulo University, São Paulo, Brazil. ) Disciplin of Clinic Gastroenterology, Department of Gastroenterology, Medical School of the São Paulo University, São Paulo, Brazil. (2) Laboratory of Pathology of the Transmissible Diseases, Department of Pathology, Medical School of the São Paulo University, São Paulo, Brazil.), Maria Antonieta LONGO(2(1) Disciplin of Clinic Gastroenterology, Department of Gastroenterology, Medical School of the São Paulo University, São Paulo, Brazil. ) Disciplin of Clinic Gastroenterology, Department of Gastroenterology, Medical School of the São Paulo University, São Paulo, Brazil. (2) Laboratory of Pathology of the Transmissible Diseases, Department of Pathology, Medical School of the São Paulo University, São Paulo, Brazil.), Antonio Atílio LAUDANNA(1(1) Disciplin of Clinic Gastroenterology, Department of Gastroenterology, Medical School of the São Paulo University, São Paulo, Brazil. ) Disciplin of Clinic Gastroenterology, Department of Gastroenterology, Medical School of the São Paulo University, São Paulo, Brazil. (2) Laboratory of Pathology of the Transmissible Diseases, Department of Pathology, Medical School of the São Paulo University, São Paulo, Brazil.) & Maria Irma Seixas DUARTE(2(1) Disciplin of Clinic Gastroenterology, Department of Gastroenterology, Medical School of the São Paulo University, São Paulo, Brazil. ) Disciplin of Clinic Gastroenterology, Department of Gastroenterology, Medical School of the São Paulo University, São Paulo, Brazil. (2) Laboratory of Pathology of the Transmissible Diseases, Department of Pathology, Medical School of the São Paulo University, São Paulo, Brazil.).
SUMMARY
The involvement of the gastrointestinal tract in the co-infection of HIV and Leishmania is rarely reported. We report the case of an HIV-infected adult man co-infected with a disseminated form of leishmaniasis involving the liver, lymph nodes, spleen and, as a feature reported for the first time in the English literature, the pancreas. Light microscopy showed amastigote forms of Leishmania in pancreatic macrophages and immunohistochemical staining revealed antigens for Leishmania and also for HIV p24. Microscopic and ultrastructural analysis revealed severe acinar atrophy, decreased zymogen granules in the acinar cytoplasm and also nuclear abnormalities such as pyknosis, hyperchromatism and thickened chromatin. These findings might correspond to the histologic pattern of protein-energy malnutrition in the pancreas as shown in our previous study in pancreas with AIDS and no Leishmania. In this particular case, the protein-energy malnutrition may be due to cirrhosis, or, Leishmania or HIV infection or all mixed. We believe that this case represents the morphologic substratum of the protein energy malnutrition in pancreas induced by the HIV infection. Further studies are needed to elucidate these issues.
KEYWORDS: Visceral Leishmaniasis; Pancreas; AIDS; Opportunistic infection; Malnutrition.
INTRODUCTION
Visceral Leishmaniasis (VL) and HIV overlap in some African, Mediterranean and South American countries including Brazil.
In the North-Eastern region of Brazil, VL is an endemic disease affecting 3,000 cases/year12.Brazil also has high rates of HIV infection, with 170,000 cumulative cases by the end of 19993.
Although HIV-VL co-infection is considered an "emerging disease" (over 850 cases reported1) there are few reported cases of involvement of the gastrointestinal tract. Leishmania amastigotes have been detected in rectal, jejunal, duodenal, gastric and oesophageal biopsy specimens from HIV-infected patients13.
A review of the English literature did not show any report of the histology and/or ultrastructure of the pancreas in leishmaniasis with or without HIV infection, or studies of the effects of malnutrition induced by leishmaniasis infection in the pancreas of patients with and without HIV. This is the first report of the detection of Leishmania in the pancreas of a patient with the disseminated form of VL and HIV co-infection. We report the histologic and ultrastructural findings in the pancreas of this patient and describe the morphological aspects of the effects of malnutrition on the pancreas.
CASE REPORT
The patient was a 44-year-old white male from a VL endemic region of Brazil, an alcohol abuser (180 g/pure ethanol/day/20 years) living in São Paulo for six months. He was admitted to Hospital das Clínicas in October 1995, complaining of fever, ascites, diffuse abdominal pain and intestinal haemorrhage for 6 months. Clinical examination revealed severe wasting (body mass index = 19), ascites and splenomegaly. Laboratory tests disclosed pancytopenia (haemoglobin 8.3 g/dL, white blood cell count 2.0 x 109/L, and platelets 2.3 x 109/L), normal liver function tests, hypergammaglobulinemia (3.7 g/dL) and hypoalbuminemia (1.6 g/dL). The ascitic fluid had low protein levels (0.8 g/dL) and showed no other abnormalities. Abdominal ultrasound revealed cirrhosis of the liver with portal hypertension. Upper gastrointestinal endoscopy revealed a duodenal ulcer. No infection was detected in the lungs, ascitic fluid, blood, stool or urine. Outcome: HIV was diagnosed (serum antibodies detected by ELISA and Western Blot) and the CD4 (+) lymphocyte count was 66 cells/mm3. Since pancytopenia persisted a bone marrow biopsy was done and showed amastigote forms of Leishmania. Treatment with liposomal amphotericin was started. After a cumulative dose of 1.85 g, the patient had a severe intestinal haemorrhage followed by renal failure and death.
The autopsy showed that the pancreas was normal at gross macroscopy.
At light microscopy, the acinar pattern, ductal system and Langerhans islet structures were preserved and no autolysis was observed.
Pancreatic light microscopy at 400X magnification revealed some abnormalities including: a) severe diffuse acinar atrophy (more than 50% in 10 fields), b) reduced zymogen granules in the acinar cytoplasm (25% in 10 fields), and c) abnormal acinar nuclei (25% in 10 fields). The nuclei were pyknotic, hyperchromatic and with thickened chromatin.
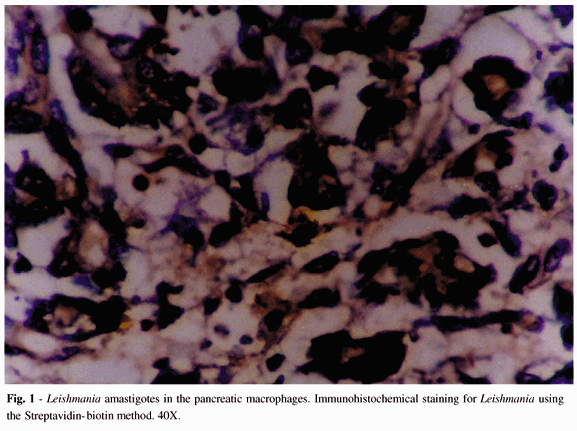
Rare amastigote forms were seen in the cytoplasm of macrophages in the interstitium of pancreatic tissue. Immunohistochemical (IH) staining for Leishmania by the Streptavidin-Biotin method with polyclonal antibodies used for diagnosis in our laboratory 8 showed antigen for Leishmania (Fig. 1). Looking for other opportunistic infections, specific stains and immunohistochemical staining were done. Ziehl-Neelsen, Groccott, Giemsa, Brown-Brenn, and also IH for Cytomegalovirus, Micobacteria, Toxoplasma gondii, Pneumocystis carinii and all of them were negative. The HIV p24 antigen was positive in the acinar cytoplasm macrophages.
Light microscopy of other organs revealed amastigote forms of Leishmania in the liver, lymph nodes and spleen. We also found Leishmania antigens (IH) in the kidney.
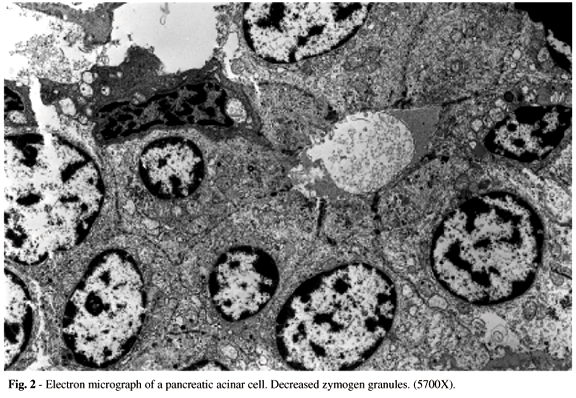
The electron microscopy examination of pancreatic tissue showed severe acinar atrophy and a marked reduction of zymogen granules (Fig. 2). The endoplasmic reticulum was dilated, with an apparent proliferation.
The mitochondria were increased in number and volume with cristae fragmentation and increased dense bodies. Some nuclei of acinar cells were reduced in volume and hyperchromatic, with condensed chromatin near the nuclear membrane, which gave them a peculiar aspect.
Ultrastructural analysis of the liver, spleen and lymph nodes revealed amastigote forms of Leishmania in the macrophage system.
DISCUSSION
The patient was an alcoholic who did not abuse other drugs, coming from an endemic region of Leishmania (probably L. chagasii) and with no previous diagnosis of HIV infection. The first presumed diagnosis was spontaneous bacterial ascites in a cirrhotic patient, but diagnosis was later changed to leishmaniasis and HIV. The leishmaniasis was the first manifestation of HIV infection.
Microscopic and ultrastructural analysis revealed severe acinar atrophy with decreased zymogen granules and nuclear abnormalities. We also observed amastigote forms of Leishmania in the cytoplasm of macrophages and Leishmania antigens demonstrated by immunohistochemistry using a specific polyclonal antibody. These morphological and ultrastructural acinar changes were previously detected in our autopsy studies of the pancreas of patients with AIDS6,7,8. We believe that these morphological alterations may be due to protein-energy malnutrition and represent the morphological pattern of malnutrition in the pancreas of AIDS patients. Some similar alterations have been detected in the pancreas of children who died of Kwashiorkor (protein-energy malnutrition - PEM)2,4.
In addition to these morphological alterations, this patient had physical and laboratory signs of malnutrition.
The malnutrition of this patient may have developed in different manners: a) due to VL, b) due to HIV infection and associated complications, c) due alcoholic cirrhosis of the liver, and d) due to the three conditions combined.
In AIDS, malnutrition/wasting can occur as a consequence of the alteration in oral intake, malabsorption/diarrhoea, metabolic alterations and alterations in energy expenditure11.
In cirrhosis, malnutrition may be due to low calorie ingestion and also to deficient hepatic synthesis of albumin and induces complex alterations in the immune system.
In Leishmaniasis, malnutrition functions as a predisposing factor and play a critical role in the progression to severe disease5. Also, leishmaniasis has a profound effect on nutritional status, resulting in loss of both muscle and fat (catabolic effects - cytokines)10., but the effects of malnutrition in the pancreas of leishmaniasis patients is not known yet.
Although all of the above conditions can contribute to these pancreatic changes, we think that, as shown in a previous study by our group, HIV infection was the main cause of these findings. Future studies are needed to fully elucidate the significance of VL for the pancreas.
RESUMO
Comprometimento pancreático na co-infecção Leishmaniose visceral HIV: aspectos histológicos e ultraestruturais
O comprometimento do trato gastrointestinal é uma manifestação incomum da co-infecção HIV-Leishmaniose Visceral (LV). Descreveremos o primeiro caso de comprometimento pancreático por LV disseminada na AIDS, como primeira manifestação oportunista (além do comprometimento do figado, baço, linfonodos e rins). A necropsia revelou pâncreas normal ao exame macroscópico. A microscopia ótica do pâncreas revelou: atrofia pancreática severa, diminuição dos grânulos de zimogênio, anormalidades nucleares discretas e presença de formas amastigotas de Leishmania em macrófagos intersticiais, além do antígeno da LV e HIV-p24 (imunohistoquímica-IH). À microscopia eletrônica: pâncreas com grânulos de zimogênio diminuídos, proliferação do retículo granuloso, mitocôndrias aumentadas, evidenciando o aspecto já observado por nós em pâncreas de pacientes com AIDS sem LV. Presença de formas amastigotas da Leishmania no fígado, baço, linfonodos além do antígeno da LV no rim (IH). As alterações histológicas pancreáticas são sugestivas de desnutrição protéico calórica, que pode estar representada neste caso pela desnutrição induzida pela cirrose, pela leishmaniose ou pela AIDS. Acreditamos ser esse último o principal responsável pelas alterações histológicas encontradas. No entanto estudos futuros são necessários para elucidar esses achados.
Received: 12 June 2000
Accepted: 20 December 2000
Correspondence to: Ethel Zimberg Chehter, Disciplina de Gastroenterologia do Departamento de Gastroenterologia da Faculdade de Medicina da Universidade de São Paulo, Al. Itu, 820/41, 01421-000 São Paulo, SP, Brazil. Fax 5511 257-2057; E-mail: chehter.ops@zaz.com.br
- 1. ALVAR, J.; CANAVATE, C.; GUTIERREZ SOLAR, B. et al - Leishmania and human immunodeficiency virus coinfection: the first 10 years. Clin. Microbiol. Rev., 10: 298-319, 1997.
- 2. BLACKBURN, W.R. & VINIJCHAIKUL, K. - The pancreas in kwashiorkor. An electron microscopic study. Lab. Invest., 20: 305-318, 1969.
- 3. Boletim Epidemiológico - AIDS, 12(2), semana epidemiológica 09 a 21, março a maio, 1999.
- 4. BROOKS, S.E.H. & GOLDEN, M.H. - The exocrine pancreas in kwashiorkor and marasmus. Light and electron microscopy. West Indian med. J., 41: 56-60, 1992.
- 5. CERF, B.J.; JONES, T.C.; BADARÓ, R. et al. - Malnutrition as a risk factor for severe visceral leishmaniasis. J. infect. Dis., 156: 1030-1033, 1987.
- 6. CHEHTER, E.Z.; DUARTE, M.I.S.; LONGO, M.A. & LAUDANNA, A.A. - AIDS and pancreas: a prospective clinical-morphological study of 109 consecutive autopsies in Săo Paulo, Brazil. Gastroenterology, 114: A950-A951, 1998.
- 7. CHEHTER, E.Z.; DUARTE, M.I.S.; LONGO, M.A. & LAUDANNA, A.A. - Pancreas in AIDS: an electron microscopic study of 9 cases. Gut, 45: A307, 1999.
- 8. CHEHTER, E.Z.; LONGO, M.A.; LAUDANNA, A.A & DUARTE, M.I.S. Involvement of the pancreas in AIDS: a prospective study of 109 post-mortems. Aids, 14: 1879-1886, 2000.
- 9. DUARTE, M.I.S.; DA MATTA, V.L.R.; CORBETT, C.E.P. et al - Interstitial pneumonitis in human visceral leishmaniasis. Trans. roy. Soc. trop. Med. Hyg., 83: 73-76,1989.
- 10. HARRISON, L.H.; NAIDU, T.G.; DREW, J.S.; DE ALENCAR, J.E. & PEARSON, R.D. - Reciprocal relationships between undernutrition and the parasitic disease visceral leishmaniasis. Rev. infect. Dis., 8: 447-453, 1986.
- 11. KOTLER, D. - Nutrition and wasting in HIV infection. HIV clin. Manag., 8: 1-33, 1999.
-
12WHO/CTD. Division of Control of Tropical Diseases - Burden and Trends. Leishmaniasis control. WHO/CTD, 1998.
- 13. WILSON, M.E. & STREET, J.A. - Visceral leishmaniasis. Gastroent. Clin. N. Amer., 25: 535-551, 1996.
Publication Dates
-
Publication in this collection
23 Apr 2001 -
Date of issue
Apr 2001
History
-
Received
12 June 2000 -
Accepted
20 Dec 2000