Abstracts
The objective of this study was to describe a pseudo-outbreak of C. difficile in a hospital, following a change in the method used to detect the toxin. In February 2002, there were two cases of CDAD and in March 7 occurred, coinciding with a change of the test (from detection of toxin A to toxin A/B). An outbreak was suspected. Active surveillance and education of staff were started. A CDAD case was defined as a patient with acute onset of diarrhea (³ three episodes of liquid stools) and a positive stool test. They were classified as hospital or community-acquired. Stool samples were also collected for C. difficile culture and isolates were typed using AP-PCR. From March 2002 through December 2003 there were 138 cases of CDAD: 70% were hospital-acquired and among the 30% with CDAD present on admission, most (81%) came directly from the community (50% had no history of hospitalization). Fifty-two percent of hospital-acquired CDAD and 94% of cases on admission had already used antibiotics. The incidence of CDAD in hospitalized patients during surveillance was 3.3 per 1000 patient-admissions. The incidence of CDAD present on admission was 6.1/1000 patients. Sixteen isolates were typed and presented 13 different profiles. In conclusion, the CDAD increase in our study occurred due to change in diagnostic methods and not due to an outbreak, as suspected initially. The incidence in hospitalized patients was much lower than in reported outbreaks. There were 13 molecular types suggesting that an outbreak did not occur. CDAD was largely community-acquired.
Clostridium difficile; pseudo-outbreak; molecular typing; community-acquired
O objetivo deste estudo foi descrever um pseudo-surto de C. difficile em um hospital após a troca do método de detecção de toxina. Em fevereiro de 2002 houve dois casos de DACD e em março ocorreram sete casos, que coincidiram com a mudança de teste (que detectava apenas toxina A e passou a detectar toxinas A e B). Foi suspeitado que houvesse um surto e vigilância ativa e reforço educacional para os profissionais de saúde foi implantado. Um caso de DACD foi definido como um paciente com início abrupto de diarréia (> 3 episódios de fezes líquidas) e um teste positivo. Os casos foram classificados como de aquisição comunitária ou hospitalar. Foram colhidas fezes para cultura para C. difficile e os isolados foram tipados por AP-PCR. De março de 2002 a dezembro de 2003 houve 138 casos de DACD: 70% foram hospitalares e, entre os 30% de casos comunitários, a maioria (81%) foi de pacientes provenientes diretamente da comunidade (50% não tinham histórico de internação). Cinquenta e dois por cento dos casos de DCAD hospitalar e 94% de casos na admissão haviam utilizado antimicrobianos. A incidência de DCAD em pacientes internados foi de 3,3/100 pacientes e na admissão foi 6,1/1000 pacientes. Dezesseis isolados foram tipados e apresentaram 13 perfis diferentes. Em conclusão, o aumento de DACD no nosso estudo ocorreu por uma mudança de método diagnóstico e não devido a um surto como foi suspeitado inicialmente. A incidência em pacientes internados foi muito inferior ao que já foi relatado em surtos. Houve 13 perfis moleculares sugerindo que não ocorreu um surto. DACD foi, em grande parte, de aquisição comunitária.
BACTERIOLOGY
Pseudo-outbreak of Clostridium difficile associated diarrhea (CDAD) in a tertiary-care hospital
Pseudo-surto de diarréia associada a Clostridium difficile (DACD) em hospital terciário
M. Beatriz Souza DiasI,II; Juliana YamashiroII; Vera L. BorrascaI; Valeska A. StempliukIII; Maria Rita E. AraújoIV; Silvia F. CostaV; Anna S. LevinII,V
IInfection Control Unit, Hospital Sírio Libanês, São Paulo, SP, Brazil
IIDepartment of Infection Control, Hospital das Clínicas, University of São Paulo, São Paulo, SP, Brazil
IIIInstituto Sírio-Libanês de Ensino e Pesquisa, São Paulo, SP, Brazil
IVLaboratory of Microbiology, Hospital da Beneficência Portuguesa, São Paulo, SP, Brazil
VDepartment of Infectious Diseases and LIM-54, University of São Paulo, São Paulo, SP, Brazil
Correspondence to Correspondence to: Anna S. Levin Rua Banibas 618, 05460-010 São Paulo, SP, Brasil Phone/fax: +55.11.3069 7066 E-mail: gcih@hcnet.usp.br
SUMMARY
The objective of this study was to describe a pseudo-outbreak of C. difficile in a hospital, following a change in the method used to detect the toxin.
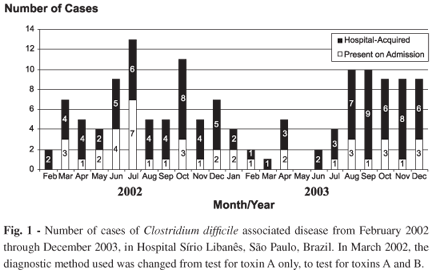
In February 2002, there were two cases of CDAD and in March 7 occurred, coinciding with a change of the test (from detection of toxin A to toxin A/B). An outbreak was suspected. Active surveillance and education of staff were started. A CDAD case was defined as a patient with acute onset of diarrhea (³ three episodes of liquid stools) and a positive stool test. They were classified as hospital or community-acquired. Stool samples were also collected for C. difficile culture and isolates were typed using AP-PCR.
From March 2002 through December 2003 there were 138 cases of CDAD: 70% were hospital-acquired and among the 30% with CDAD present on admission, most (81%) came directly from the community (50% had no history of hospitalization). Fifty-two percent of hospital-acquired CDAD and 94% of cases on admission had already used antibiotics. The incidence of CDAD in hospitalized patients during surveillance was 3.3 per 1000 patient-admissions. The incidence of CDAD present on admission was 6.1/1000 patients. Sixteen isolates were typed and presented 13 different profiles.
In conclusion, the CDAD increase in our study occurred due to change in diagnostic methods and not due to an outbreak, as suspected initially. The incidence in hospitalized patients was much lower than in reported outbreaks. There were 13 molecular types suggesting that an outbreak did not occur. CDAD was largely community-acquired.
Keywords:Clostridium difficile; pseudo-outbreak; molecular typing; community-acquired.
RESUMO
O objetivo deste estudo foi descrever um pseudo-surto de C. difficile em um hospital após a troca do método de detecção de toxina.
Em fevereiro de 2002 houve dois casos de DACD e em março ocorreram sete casos, que coincidiram com a mudança de teste (que detectava apenas toxina A e passou a detectar toxinas A e B). Foi suspeitado que houvesse um surto e vigilância ativa e reforço educacional para os profissionais de saúde foi implantado. Um caso de DACD foi definido como um paciente com início abrupto de diarréia (> 3 episódios de fezes líquidas) e um teste positivo. Os casos foram classificados como de aquisição comunitária ou hospitalar. Foram colhidas fezes para cultura para C. difficile e os isolados foram tipados por AP-PCR.
De março de 2002 a dezembro de 2003 houve 138 casos de DACD: 70% foram hospitalares e, entre os 30% de casos comunitários, a maioria (81%) foi de pacientes provenientes diretamente da comunidade (50% não tinham histórico de internação). Cinquenta e dois por cento dos casos de DCAD hospitalar e 94% de casos na admissão haviam utilizado antimicrobianos. A incidência de DCAD em pacientes internados foi de 3,3/100 pacientes e na admissão foi 6,1/1000 pacientes. Dezesseis isolados foram tipados e apresentaram 13 perfis diferentes.
Em conclusão, o aumento de DACD no nosso estudo ocorreu por uma mudança de método diagnóstico e não devido a um surto como foi suspeitado inicialmente. A incidência em pacientes internados foi muito inferior ao que já foi relatado em surtos. Houve 13 perfis moleculares sugerindo que não ocorreu um surto. DACD foi, em grande parte, de aquisição comunitária.
INTRODUCTION
Clostridium difficile is the most frequent cause of hospital-acquired diarrhea18,20. More recently C. difficile associated disease has become more severe leading to more frequent complications such as toxic megacolon and is more refractory to treatment14. Since the early 2000s, a new hyper virulent strain, NAP1/BI/027, has been responsible for outbreaks in North America and Europe20,27,31. Its epidemiology in Brazil is unknown and there have been no reports of outbreaks with hyper virulent strains in this country3,30.
The objective of this study is to describe a pseudo-outbreak of C. difficile in a hospital, following a change in the method used to detect the toxin.
METHODS
Hospital Sírio Libanês is a private hospital in the city of São Paulo, Brazil. In 2002-3 it had 250 beds, divided between surgical and medical wards, two intensive care units (ICU), a semi-critical unit and an oncology unit. There was also a day-hospital, an emergency department and an outpatient clinic. Surgical and oncological patients were the majority.
Active surveillance for nosocomial infections was continuous in the ICU and oncology unit and was performed during four months, every year, in the other hospital areas (February, May, August and November). Until February 2002 the Infection Control Department did not perform active surveillance for C. difficile associated disease (CDAD); when CDAD was suspected by the attending physician, a stool EIA test for toxin A was requested. From March 2002 on, the hospital started to perform an EIA test for toxins A and/or B (Premier Toxin A and B, Meridian Bioscience, Cincinnati, USA).
In February 2002, there were two cases of CDAD, and in March seven cases were detected, which coincided with the new test. However, a potential outbreak was suspected and active surveillance was started in all units of the hospital, allied with education of the staff on prevention measures.
A case of CDAD was defined as a patient who presented an acute onset of diarrhea (three or more episodes of liquid stools) and a positive stool test for C. difficile toxin. Only the first episode was considered for each patient. CDAD cases were classified as:
- Hospital-acquired: if they occurred after 48 hours of hospitalization.
- Present on admission: if CDAD was present at hospital admission or within the first 48 hours of hospitalization, even if the patient had been hospitalized elsewhere or had received healthcare on an outpatient basis.
Stool cultures for C. difficile: Stool samples positive for toxin were frozen at -20 ºC to be submitted later to culturing for C. difficile. One mL of the sample was added to 2 mL of 70% alcohol and incubated for one hour at room temperature. The mixture was inoculated in 10 mL of thioglycolate broth and maintained at 37 ºC for seven days. After this period, the sample was plated on blood agar and maintained under anaerobic conditions for 48h. If there was a pure culture, it was plated on Cycloserine Cefoxitin Fructose Agar (CCFA) under anaerobic conditions. Culturing in blood agar was also performed10. If growth on this selective medium occurred, one colony was inoculated in 10 mL of BHI for DNA extraction. If there was a mixed culture on the blood agar, colonies were re-inoculated in thioglycolate broth and the procedure was repeated. Antimicrobial susceptibility tests were not performed.
DNA extraction and molecular typing (genotyping) by arbitrary primed polymerase chain reaction (AP-PCR)
One colony from each culture was inoculated in 10 mL of BHI and grown for 48 h in anaerobic conditions. Then two mL were frozen at -70 ºC and DNA extraction was carried out with the remaining 8 mL. Extractions of DNA were performed as described elsewhere9. For typing AP-PCR was used with the arbitrary primer oligonucleotide T7 (GTAATACGACTCACTATAG)4,8,11,24,25. The PCRs were prepared in PCR buffer (50 mM KCl, 20 mM Tris-HCl, 2.5 mM MgCl2, 100 mg of bovine serum albumin per mL [pH 8.4]) containing 40 pmol of T-7, 1 mM of each deoxynucleoside triphosphate, 2 U Taq of DNA polymerase and one mg of DNA. The PCR profile was one step at 95 ºC for five min, followed by twice at 95 ºC for one min, 26 ºC for one min and two min at 72 ºC. After these two low stringency cycles, the arbitrary primer was heated at 95 ºC for one min, 50 ºC for one min, and 72 ºC for two min in the subsequent 55 cycles.
Identification of C. difficile was confirmed using primers for gene 16 S rDNA11. Products of amplification were analyzed using electrophoresis in agarose gel and staining with ethidium bromide. Genotypes were defined on the basis of DNA banding patterns. Isolates with identical patterns were considered genotypically "indistinguishable", while those that differed by one to four bands were subtypes of the same type and differing by five or more bands were considered distinct types26.
Outbreak control: All healthcare workers in the hospital were re-trained in control measures, with emphasis on contact precautions and hand hygiene with chlorhexidine 2% soap and water. Alcohol based hand disinfection was discouraged in C. difficile patient rooms. Routine concurrent cleaning of the environment with organic chlorine (500 ppm) was initiated in case patient rooms, all intensive care units and operating rooms. Post discharge disinfection of all patient rooms was also chlorine based. No cohorting was instituted since all the patients were in individual rooms.
RESULTS
From March 2002 through December 2003 there were 138 cases of CDAD. Among the 138 patients, 53% were male and the mean age was 64 years (range: 1-95); 70% of the cases were hospital-acquired, and among the 30% with CDAD present on admission, most (81%) had come directly from the community and 50% had no history of previous hospitalization. Fifty-two percent of the patients with hospital-acquired CDAD and 94% of cases present on admission had used antibiotics: 19% had used third-generation cephalosporins, 13% intravenous vancomycin, 6% a carbapenem, and 35% had used other antibiotics. Twenty-seven percent had used more than one antibiotic simultaneously. No patients received oral vancomycin prior to the diagnosis of CDAD.
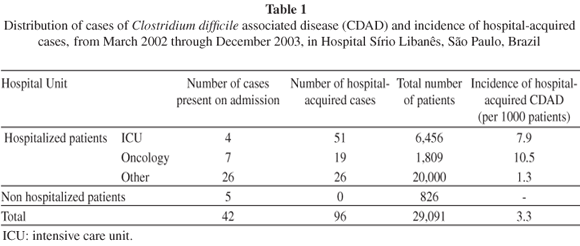
The monthly distribution of CDAD cases can be seen in Figure 1. The incidence of hospital-acquired CDAD during this period was 7.9/1000 patients in ICUs; 10.5/1000 in oncology patients; and 1.3 in the other hospital units (Table 1). The incidence of CDAD present on hospital admission was 6.1/1000 patients.
Stools from all the 138 cases were cultured for C. difficile; however, growth only occurred in 16 samples. AP-PCR identified 13 different clonal profiles (Fig. 2). There were three pairs of isolates that were considered to present similar profiles; however, the isolates in pairs occurred 22 days, 2.3 months and 5.5 months apart. The results of molecular typing were only available in February 2004, when it became clear that this was a pseudo-outbreak.
DISCUSSION
The increase in CDAD observed in our study occurred due to the change in diagnostic methods and not due to an outbreak as suspected initially. There have been no reports of CDAD outbreaks in our country; however, due to reported outbreaks with the particularly virulent strain ribotype 027 (NAP1/BI/027 or CD027) in Europe and North America15,28,29, the increase in cases in a Brazilian hospital was especially worrisome and led to this investigation. In a Brazilian study performed in Rio de Janeiro, with a small number of isolates, the ribotypes identified were 014 and 1061, and the ribotype 027 has not been described in the country.
The incidence of CDAD in hospitalized patients in our study during active surveillance was 3.3 per 1000 patient admissions. This incidence is much lower than that described in other studies, including one in Dublin (21/1000 admissions) during a CD027 outbreak5, but similar to that described in a Turkish hospital (2.2 per 1000 admissions) by active surveillance in a non-outbreak setting6. In Brazil, C. difficile was detected in 5.5% of hospitalized children with acute diarrhea7 and in 4.2% in another study21 that can be considered high for situations that were not described as outbreaks.
The importance of detecting toxin B besides toxin A has been discussed. Initially, toxin A was reported to be responsible for the disease14. However, the role of toxin B has been reviewed and is considered important, or even essential for virulence19. The sudden high incidence of C. difficile diarrhea observed in our study was not due to a monoclonal outbreak, as initially suspected, but to an increase in diagnosis offered by the detection of toxin A and B, thus, it was a pseudo-outbreak.
The low yield of cultures is difficult to explain. It may be due to the long time between freezing of the stool samples and culturing, false positive toxin results, or even the limited experience of our microbiology laboratory with this pathogen.
Many different typing methods have been used for C. difficile. For the early and rapid detection of outbreak situations, methods such as restriction enzyme analysis, arbitrary primed polymerase chain reaction (AP-PCR), and PCR ribotyping are commonly used. For long-term epidemiologic studies, multilocus sequence typing, multilocus variable number of tandem repeats analysis, and amplified fragment length polymorphism are of interest16,17. AP-PCR was compared with immunoblotting and restriction endonuclease analysis, and presented similar results23. Arbitrarily primed PCR, that relies on the T7 oligonucleotide, was used to identify different strains and was sensitive and accurate to type C. difficile2,24. Our use of AP-PCR was sufficiently discriminatory, as there were 13 profiles among 16 isolates, which was enough to conclude that an outbreak did not occur.
Although originally considered a nosocomial-acquired condition, CDAD was largely community-acquired in our investigation (30%). Only half of these community-acquired cases had been hospitalized previously, but most had used antibiotics. A recent study of community-acquired CDAD in the Netherlands demonstrated a prevalence of 1.5% among stool samples sent to reference laboratories by 195 general practitioners. Most patients had not been hospitalized within the previous year, but more than half had used antibiotics within the previous six months2. The cause for this presence in the community has been explained by the previous hospitalization of patients and the use of antibiotics22, but this does still not explain the occurrence in our patients without these risk factors. It is possible that the presence in the community of asymptomatic carriers of C. difficile originating in hospitals has increased the community spread of this agent, especially in patients with underlying conditions.
This study has limitations. Only isolates from 16 of 138 cases (12%) were typed, but our results showed such a large variety of molecular types that it is highly unlikely that a clonal outbreak was missed. However, this low positive rate was a limitation of the study. We did not compare our isolates directly with the CD027 strain, but the relatively mild clinical presentation of our cases suggests that this was not a problem in our hospital. Although the data are relatively old and therefore may not represent what occurs currently, no changes in the clinical features of C. difficile associated disease have been observed in our hospital since the occurrence of the pseudo-outbreak, which suggests that, if present, very virulent strains such as CD027, or the more recently described CD0789, are not yet an important problem in our setting. However, the environmental control measures instituted during the pseudo-outbreak were kept in place until the present time.
Received: 10 March 2010
Accepted: 6 May 2010
- 1. Balassiano IT, Miranda KR, Boente RF, Pauer H, Oliveira IC, Santos-Filho J, et al. Characterization of Clostridium difficile strains isolated from immunosuppressed inpatients in a hospital in Rio de Janeiro, Brazil. Anaerobe. 2009;15:61-4.
- 2. Bauer MP, Veenendaal D, Verhoef L, Bloembergen P, van Dissel JT, Kuijper EJ. Clinical and microbiological characteristics of community-onset Clostridium difficile infection in The Netherlands. Clin Microbiol Infect. 2009;15:1087-92.
- 3. Carignan A, Allard C, Pépin J, Cossette B, Nault V, Valiquette L. Risk of Clostridium difficile infection after perioperative antibacterial prophylaxis before and during an outbreak of infection due to a hypervirulent strain. Clin Infect Dis. 2008;46:1838-43.
- 4. Clabots CR, Johnson S, Bettin KM, Mathie PA, Mulligan ME, Schaberg DR, et al. Development of a rapid and efficient restriction endonuclease analysis typing system for Clostridium difficile and correlation with other typing systems. J Clin Microbiol. 1993;31:1870-75.
- 5. Drudy D, Harnedy N, Fanning S, Hannan M, Kyne L. Emergence and control of fluoroquinolone-resistant, toxin A-negative, toxin B-positive Clostridium difficile Infect Control Hosp Epidemiol. 2007;28:932-40.
- 6. Ergen EK, Akalın H, Yılmaz E, Sınırtas M, Alver O, Heper Y, et al. Nosocomial diarrhea and Clostridium difficile associated diarrhea in a Turkish University Hospital. Med Mal Infect. 2009;39:382-7.
- 7. Ferreira CE, Nakano V, Durigon EL, Avila-Campos MJ. Prevalence of Clostridium spp. and Clostridium difficile in children with acute diarrhea in São Paulo City, Brazil. Mem Inst Oswaldo Cruz. 2003;98:451-4.
- 8. Giesemann T, Egerer M, Jank T, Aktories K. Processing of Clostridium difficile toxins. J Med Microbiol. 2008;57:690-6.
- 9. Goorhuis A, Bakker D, Corver J, Debast SB, Harmanus C, Notermans DW, et al. Emergence of Clostridium difficile infection due to a new hypervirulent strain, polymerase chain reaction ribotype 078. Clin Infect Dis. 2008;47:1162-70.
- 10. Groeschel DH. Clostridium difficile infection. Crit Rev Clin Lab Sci. 1996;33:203-45.
- 11. Gumerlock PH, Tang YJ, Meyers FJ, Silva J Jr. Use of the polymerase chain reaction for the specific and direct detection of Clostridium difficile in human feces. Rev Infect Dis. 1991;13:1053-60.
- 12. Gumerlock PH, Tang YJ, Weiss JB, Silva J Jr. Specific detection of toxigenic strains of Clostridium difficile in stool specimens. J Clin Microbiol. 1993;31:507-11.
- 13. Hall IC, O'Toole E. Intestinal flora in new-born infants: with a description of a new pathogenic anaerobe, Bacillus difficilis Am J Dis Child. 1935;49:390-402.
- 14. Hookman P, Barkin JS. Clostridium difficile associated infection, diarrhea and colitis. World J Gastroenterol. 2009;15:1554-80.
- 15. Indra A, Huhulescu S, Fiedler A, Kernbichler S, Blaschitz M, Allerberger F. Outbreak of Clostridium difficile 027 infection in Vienna, Austria 2008-2009. Euro Surveill. 2009;14(17):pii=19186.
- 16. Killgore G, Thompson A, Johnson S, Brazier J, Kuijper E, Pepin J, et al. Comparison of seven techniques for typing international epidemic strains of Clostridium difficile: restriction endonuclease analysis, pulsed-field gel electrophoresis, PCR-ribotyping, multilocus sequence typing, multilocus variable-number tandem-repeat analysis, amplified fragment length polymorphism, and surface layer protein A gene sequence typing. J Clin Microb. 2008;46:431-7.
- 17. Kuijper EJ, van den Berg RJ, Brazier JS. Comparison of molecular typing methods applied to Clostridium difficile Methods Mol Biol. 2009;551:159-71.
- 18. Kuijper EJ, van Dissel JT. Spectrum of Clostridium difficile infections outside health care facilities. CMAJ. 2008;179:747-8.
- 19. Lyras D, O'Connor JR, Howarth PM, Sambol SP, Carter GP, Phumoonna T, et al. Toxin B is essential for virulence of Clostridium difficile Nature. 2009;458:1176-9.
- 20. McMaster-Baxter NL, Musher DM. Clostridium difficile: recent epidemiologic findings and advances in therapy. Pharmacotherapy. 2007;27:1029-39.
- 21. Pinto LJ, Alcides AP, Ferreira EO, Avelar KE, Sabrá A, Domingues RM, et al.. Incidence and importance of Clostridium difficile in paediatric diarrhoea in Brazil. J Med Microbiol. 2003;52:1095-9.
- 22. Pituch H. Clostridium difficile is no longer just a nosocomial infection or an infection of adults. Int J Antimicrob Agents. 2009;33(suppl. 1):S42-5.
- 23. Tang YJ, Houston ST, Gumerlock PH, Mulligan ME, Gerding DN, Johnson S, et al. Comparison of arbitrarily primed PCR with restriction endonuclease and immunoblot analyses for typing Clostridium difficile isolates. J Clin Microbiol. 1995;33:3169-73.
- 24. Titov L, Lebedkova N, Shabanov A, Tang YJ, Cohen SH, Silva S Jr. Isolation and molecular characterization of Clostridium difficile strains from patients and the hospital environment in Belarus. J Clin Microb. 2000;38:1200-2.
- 25. Tyler KD, Wang G, Tyler SD, Johnson WM. Factors affecting reliability and reproducibility of amplification-based DNA fingerprinting of representative bacterial pathogens. J Clin Microbiol. 1997;35:339-46.
- 26. van Belkum A, Tassios PT, Dijkshoorn L, Haeggman S, Cookson B, Fry NK, et al. Guidelines for the validation and application of typing methods for use in bacterial epidemiology. Clin Microbiol Infect. 2007;13(suppl. 3):1-46.
- 27. Vonberg RP, Kuijper EJ, Wilcox MH, Barbut F, Tüll P, Gastmeier P, et al. Infection control measures to limit the spread of Clostridium difficile. Clin Microbiol Infect. 2008;14(suppl. 5):2-20.
- 28. Warny M, Pepin J, Fang A, Killgore G, Thompson A, Brazier J, et al. Toxin production by an emerging strain of Clostridium difficile associated with outbreaks of severe disease in North America and Europe. Lancet. 2005;366:1079-84.
- 29. Weiss B, Kleinkauf N, Eckmanns T, van der Heiden M, Neumann M, Michels H, et al. Risk factors related to a hospital associated cluster of Clostridium difficile PCR ribotype 027 infections in Germany during 2007. Infect Control Hosp Epidemiol. 2009;30:282-4.
- 30. Zilberberg MD, Shorr AF, Kollef MH. Increase in adult Clostridium difficile - related hospitalizations and case-fatality rate, United States, 2000-2005. Emerg Infect Dis. 2008;14:929-31.
- 31. Zumbado-Salas R, Gamboa-Coronado MM, Rodríguez-Cavallini E, Chaves-Olarte E. Clostridium difficile in adult patients with nosocomial diarrhea in a Costa Rican Hospital. Am J Trop Med Hyg. 2008;79:164-5.
Publication Dates
-
Publication in this collection
23 June 2010 -
Date of issue
June 2010
History
-
Received
10 Mar 2010 -
Accepted
06 May 2010