ABSTRACT
Introduction:
Human T-lymphotropic virus type 1 and 2 (HTLV-1/2) is endemic in Brazil, but few studies have investigated the seroprevalence of HTLV and its subtypes among blood donors in the capital city Manaus, Amazonas State, Brazil.
Aim:
To estimate the seroprevalence of HTLV-1/2 and to identify circulating subtypes among blood donors in Manaus.
Materials and Methods:
Blood donors (2001-2003) were screened for HTLV-1/2 antibodies by ELISA. Positive results were confirmed and subtyped by Western blot assays. Prevalence rates were calculated and compared with demographic data.
Results:
Among the 87,402 individuals screened, 116 (0.13%) were seropositive for HTLV-1/2. A second sample (76/116) was collected and retested by HTLV-1/2 ELISA, of which only 41/76 were positive. Western blot confirmed HTLV infection in 24/41 retested blood donors [HTLV-1 (n=16), HTLV-2 (n=5) and HTLV-untypable (n=3)].
Discussion:
HTLV-1 and HTLV-2 are prevalent among blood donors in Manaus. However, additional studies are needed to comprehend the epidemiology of HTLV-1/2 in Amazonas not only to understand the pathophysiology of the disease providing adequate medical assistance, but also to reduce or block virus transmission.
KEYWORDS:
HTLV; Blood donors; HTLV subtyping; HTLV1; HTLV2; T-lymphotropic virus type 1 and 2; Epidemiology of HTLV infection; Prevalence of HTLV
INTRODUCTION
Human T-lymphotropic virus 1 and 2 (HTLV-1 and HTLV-2) are retroviruses discovered in the 1980s11. Poiesz BJ, Ruscetti FW, Gazdar AF, Bunn PA, Minna JD, Gallo RC. Detection and isolation of type C retrovirus particles from fresh and cultured lymphocytes of a patient with cutaneous T-cell lymphoma. Proc Natl Acad Sci USA. 1980;77:7415-9.,22. Kalyanaraman VS, Sarngadharan MG, Robert-Guroff M, Miyoshi I, Golde D, Gallo RC. A new subtype of human T-cell leukemia virus (HTLV-II) associated with a T-cell variant of hairy cell leukemia. Science. 1982;218:571-3.. It is estimated that at least 10 million people worldwide are infected with HTLV-1, with endemic foci in Japan, the Caribbean, South America, and Central Africa33. Gessain A, Cassar O. Epidemiological aspects and world distribution of HTLV-1 infection. Front Microbiol. 2012;3:388.. HTLV transmission occurs primarily via the following three routes: 1) vertically from mother to child, predominantly through breastfeeding44. Takahashi K, Takezaki T, Oki T, Kawakami K, Yashiki S, Fujiyoshi T, et al. Inhibitory effect of maternal antibody on mother-to-child transmission of human T-lymphotropic virus type I. The Mother-to-Child Transmission Study Group. Int J Cancer. 1991;49:673-7.,55. Percher F, Jeannin P, Martin-Latil S, Gessain A, Afonso P, Vidy-Roche A, et al. Mother-to-child transmission of HTLV-1 epidemiological aspects, mechanisms and determinants of mother-to-child transmission. Viruses 2016;8:40.; 2) between sexual partners through unprotected intercourse66. Paiva A, Casseb J. Sexual transmission of human T-cell lymphotropic virus type 1. Rev Soc Bras Med Trop. 2014;47:265-74.,77. Stuver SO, Tachibana N, Okayama A, Shioiri S, Tsunetoshi Y, Tsuda K, et al. Heterosexual transmission of human T cell leukemia/lymphoma virus type I among married couples in southwestern Japan: an initial report from the Miyazaki Cohort Study. J Infect Dis. 1993;167:57-65.; and 3) blood transfusion from a HTLV positive donor or sharing or re-use of needles and syringes to inject drugs88. Okochi K, Sato H, Hinuma Y. A Retrospective study on transmission of adult T cell leukemia virus by blood transfusion: seroconversion in recipients. Vox Sang. 1984;46:245-53.,99. Feigal E, Murphy E, Vranizan K, Bacchetti P, Chaisson R, Drummond JE, et al. Human T cell lymphotropic virus types I and II in intravenous drug users in San Francisco: risk factors associated with seropositivity. J Infect Dis. 1991;164:36-42.. A number of diseases have been associated with HTLV-1, including adult T-cell leukemia–lymphoma (ATLL), myelopathy/tropical spastic paraparesis (HAM/TSP)1010. Yoshida M, Miyoshi I, Hinuma Y. Isolation and characterization of retrovirus from cell lines of human adult T-cell leukemia and its implication in the disease. Proc Natl Acad Sci U S A. 1982;79:2031-5.
11. Gessain A, Barin F, Vernant JC, Gout O, Maurs L, Calender A, et al. Antibodies to human T-lymphotropic virus type-I in patients with tropical spastic paraparesis. Lancet. 1985;2:407-10.
12. Osame M, Usuku K, Izumo S, Ijichi N, Amitani H, Igata A, et al. HTLV-I associated myelopathy, a new clinical entity. Lancet. 1986;1:1031-2.-1313. Takatsuki K, Matsuoka M, Yamaguchi K. Adult T-cell leukemia in Japan. J Acquir Immune Defic Syndr Hum Retrovirol. 1996;13 Suppl 1:S15-9., uveitis, infective dermatitis, and other inflammatory disorders1414. Martin F, Taylor GP, Jacobson S. Inflammatory manifestations of HTLV-1 and their therapeutic options. Expert Rev Clin Immunol. 2014;10:1531-46., although most virus carriers remain asymptomatic throughout their lives. Brazil is one of the largest endemic areas for HTLV-1 and -2 infections33. Gessain A, Cassar O. Epidemiological aspects and world distribution of HTLV-1 infection. Front Microbiol. 2012;3:388.. Seroprevalence rates of HTLV-1/2 in Brazilian blood donors range from 0.04% to 1.0% depending on the geographic region1515. Catalan-Soares B, Carneiro-Proietti AB, Proietti FA. Heterogeneous geographic distribution of human T-cell lymphotropic viruses I and II (HTLV-I/II): serological screening prevalence rates in blood donors from large urban areas in Brazil. Cad Saude Publica. 2005;21:926-31.. In Manaus capital of the Amazonas State, the prevalence of HTLV-1 in first-time blood donors was reported as 0.14% (2008-2009)1616. Passos LN, Moraes MP, Tamegão-Lopes B, Lemos JA, Machado PR, Mira MT, et al. Absence of HTLV-1/2 infection and dermatological diseases in Manaus, State of Amazonas, Brazil. Rev Soc Bras Med Trop. 2014;47:507-9.. However, few studies have tried to identify HTLV subtypes in blood donors and the prevalence of HTLV in this population. The aim of this study was to estimate the seroprevalence of HTLV and to identify the subtypes among blood donors in Manaus.
MATERIALS AND METHODS
Ethical considerations
This study was approved by the Research Ethics Committee of Fundação Hospitalar de Hematologia e Hemoterapia do Amazonas (FHEMOAM), in accordance with the Brazilian law, which complied with the Declaration of Helsinki. All the study participants signed an informed consent prior to enrolment.
Patient information
Individuals donating blood at FHEMOAM, Manaus, Amazonas, were recruited for the study between August 2001 and August 2003. A questionnaire was used to collect individual information (age, gender, marital status, educational level, first time or regular donor).
Serological analysis
Blood donors were screened for antibodies against HTLV-1/2 by using an enzyme-linked immunosorbent assay (ELISA-Murex HTLV 1, GE 80/81, + 2, Murex Diagnostics). Positive samples were then confirmed by Western blot (HTLV BLOT 2.4, Genelabs Diagnostics®, Singapore), following the manufacturer's instructions.
Statistics
Statistical analysis used the Chi-square or the Fisher exact test if the expected frequencies were less than five. A p-value of less than 0.05 was considered statistically significant. Analyses were performed using the Epi info 6 software.
RESULTS
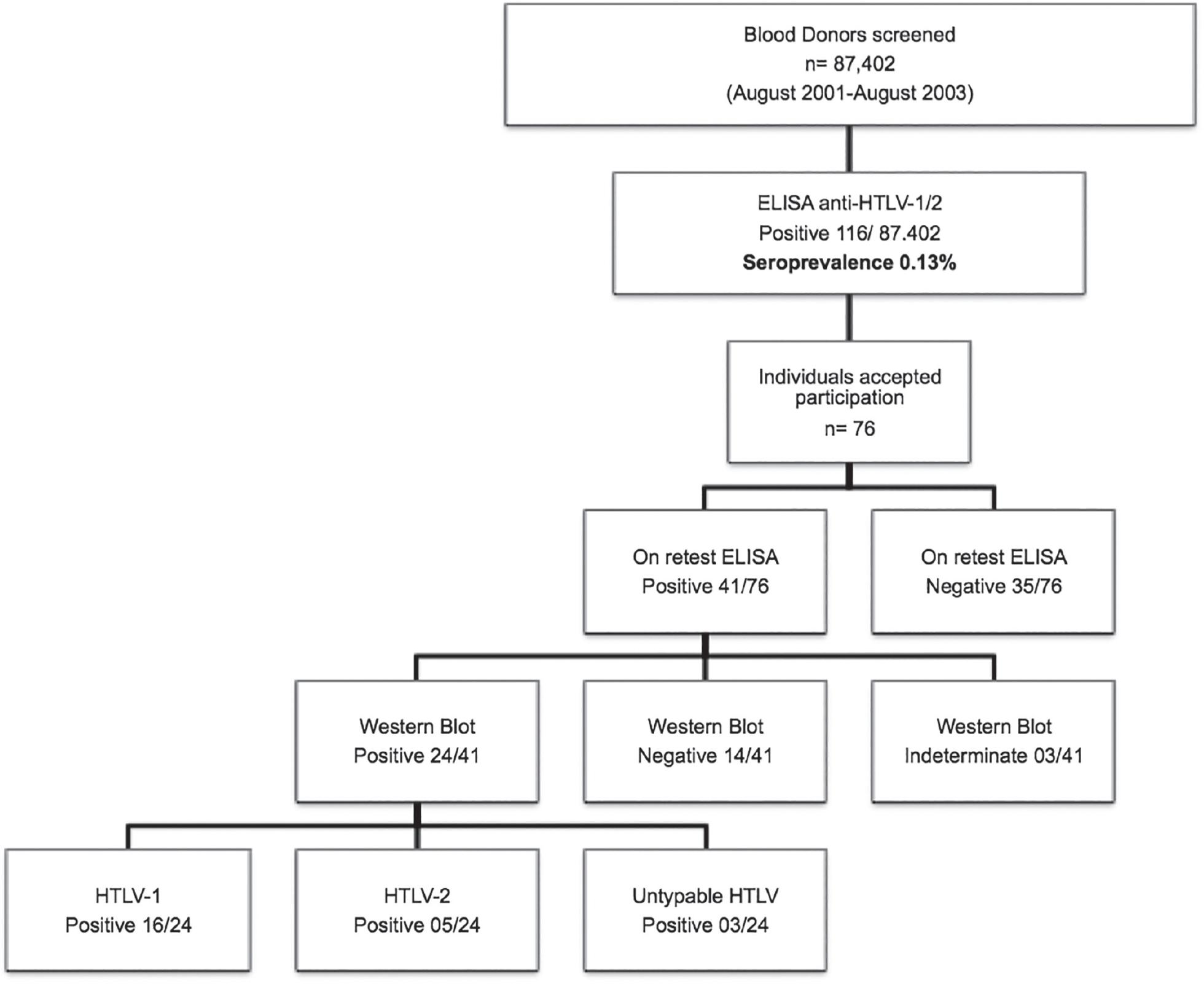
A total of 87,402 blood donors were screened by HTLV-1/2 ELISA between August 2001 and August 2003. A total of 116 individuals were tested positive with a seroprevalence of 0.13% in the primary screening (Figure 1). However, only 76/116 (65.5%) volunteered to participate in the confirmatory test. On retesting, only 41/76 individuals were found to be positive by HTLV-1/2 ELISA, positive samples were then tested by Western blot (WB) to confirm diagnoses and to identify the HTLV subtype. According to the WB assay, three samples were indeterminate; 14 samples were negative and 24 were positive for HTLV. Among the 24 positive samples, 16 were HTLV-1, five were HTLV-2 positive and three samples were HTLV positive, but untypable (Figure 1).
Serological screening and confirmation of HTLV-1 and HTLV-2 positive blood donors - Flowchart depicts an approach for detection and confirmation of HTLV infection in blood donors
Demographic and epidemiologic data of HTLVconfirmed samples are shown in Table 1. Average age of positive blood donors was 35.3 ± 11.5 years; 41.6% (n=10) of positive individuals were women. Moreover, 83.33% of positive individuals were first-time blood donors and 62.5% identified themselves as married. Among the HTLVconfirmed individuals, 16.6% (n=4) reported transfusion history and 54.2% described having had multiple sex partners along their entire lives (data not shown).
DISCUSSION
This is the first cross-sectional study to demonstrate the presence of HTLV-1 and HTLV-2 among blood donors in Manaus, Amazonas. Previous studies have only detected HTLV-1 in blood donors from Manaus1616. Passos LN, Moraes MP, Tamegão-Lopes B, Lemos JA, Machado PR, Mira MT, et al. Absence of HTLV-1/2 infection and dermatological diseases in Manaus, State of Amazonas, Brazil. Rev Soc Bras Med Trop. 2014;47:507-9.,1717. Galvão-Castro B, Loures L, Rodriques LG, Sereno A, Ferreira Júnior OC, Franco LG, et al. Distribution of human T-lymphotropic virus type I among blood donors: a nationwide Brazilian study. Transfusion. 1997;37:242-3.. In this study, the overall seroprevalence of HTLV 1/2 was 0.13% (2001-2003). In 1993, HTLV-1 prevalence in 1,200 blood donors from Manaus was 0.08%1717. Galvão-Castro B, Loures L, Rodriques LG, Sereno A, Ferreira Júnior OC, Franco LG, et al. Distribution of human T-lymphotropic virus type I among blood donors: a nationwide Brazilian study. Transfusion. 1997;37:242-3.. In addition, Catalan-Soares et al.1515. Catalan-Soares B, Carneiro-Proietti AB, Proietti FA. Heterogeneous geographic distribution of human T-cell lymphotropic viruses I and II (HTLV-I/II): serological screening prevalence rates in blood donors from large urban areas in Brazil. Cad Saude Publica. 2005;21:926-31. reported a HTLV 1/2 seroprevalence of 0.53% between 1995 and 2000, whereas, prevalence of HTLV-1 was estimated to be 0.14% in first-time blood donors (2008 to 2009)1616. Passos LN, Moraes MP, Tamegão-Lopes B, Lemos JA, Machado PR, Mira MT, et al. Absence of HTLV-1/2 infection and dermatological diseases in Manaus, State of Amazonas, Brazil. Rev Soc Bras Med Trop. 2014;47:507-9.. On other hand, HTLV infection was absent in pregnant women (n=674)1818. Machado Filho AC, Sardinha JF, Ponte RL, Costa EP, Silva SS, Martinez-Espinosa FE. Prevalência de infecção por HIV, HTLV, VHB e de sífilis e clamídia em gestantes numa unidade de saúde terciária na Amazônia ocidental brasileira. Rev Bras Ginecol Obstet. 2010;32:176-83. and individuals with dermatological disease (n=1,200)1616. Passos LN, Moraes MP, Tamegão-Lopes B, Lemos JA, Machado PR, Mira MT, et al. Absence of HTLV-1/2 infection and dermatological diseases in Manaus, State of Amazonas, Brazil. Rev Soc Bras Med Trop. 2014;47:507-9. in Manaus. Our results are in accordance with the above studies as we found a low HTLV-1/2 seroprevalence among blood donors in Manaus.
Besides, seroprevalence of HTLV-1/2 in blood donors has been among the highest in other Northern States of Brazil like Amapá (0.71%) and Pará (0.91%) compared to Amazonas1515. Catalan-Soares B, Carneiro-Proietti AB, Proietti FA. Heterogeneous geographic distribution of human T-cell lymphotropic viruses I and II (HTLV-I/II): serological screening prevalence rates in blood donors from large urban areas in Brazil. Cad Saude Publica. 2005;21:926-31., which might be explained by the phenomenon of clustering of HTLV infection in some populations33. Gessain A, Cassar O. Epidemiological aspects and world distribution of HTLV-1 infection. Front Microbiol. 2012;3:388.,1919. Mueller N. The epidemiology of HTLV-I infection. Cancer Causes Control. 1991;2:37-52..
Presence of a considerable number of HTLV-2 positive individuals in our study can be explained by the fact that a significant number of HTLV positive individuals were indigenous descendants. Several researchers have shown that HTLV-2 is predominant among Brazilian indigenous groups, with an area of high endemicity in the Amazon region2020. Ishak R, Harrington WJ Jr, Azevedo VN, Eiraku N, Ishak MO, Guerreiro JF, et al. Identification of human T cell lymphotropic virus type IIa infection in the Kayapo, an indigenous population of Brazil. AIDS Res Hum Retroviruses. 1995;11:813-21.,2121. Paiva A, Casseb J. Origin and prevalence of human T-lymphotropic virus type 1 (HTLV-1) and type 2 (HTLV-2) among indigenous populations in the Americas. Rev Inst Med Trop Sao Paulo. 2015;57:1-13.. Moreover, HTLV-2 infections in non-indigenous populations have been documented among blood donors in urban areas of the Amazon region of Brazil2222. Ishak R, Ishak MO, Azevedo VN, Santos DE, Vallinoto AC, Saraiva JC, et al. Detection of HTLV-IIa blood donors in an urban area of the Amazon Region of Brazil (Belém, PA). Rev Soc Bras Med Trop. 1998;31:193-7.,2323. Santos EL, Tamegão-Lopes B, Machado LF, Ishak MO, Ishak R, Lemos JA, et al. Caracterização molecular do HTLV-1/2 em doadores de sangue em Belém, Estado do Pará: primeira descrição do subtipo HTLV-2b na região Amazônica. Rev Soc Bras Med Trop. 2009;42:271-6..
Furthermore, in this study, the prevalence of HTLV infection in married individuals was higher (62.5%) compared to single individuals (37.5%), which needs to be confirmed in a larger population. For transmission of HTLV by sexual route, a higher frequency of exposure has been described, which may be a reason for the higher transmission rates between married individuals77. Stuver SO, Tachibana N, Okayama A, Shioiri S, Tsunetoshi Y, Tsuda K, et al. Heterosexual transmission of human T cell leukemia/lymphoma virus type I among married couples in southwestern Japan: an initial report from the Miyazaki Cohort Study. J Infect Dis. 1993;167:57-65.. In addition, first-time blood donors had a higher prevalence of HTLV infection compared to regular blood donors. This confirms that regular blood donors are perceived to be less risky or safer than first-time blood donors. A higher prevalence of HTLV was observed in women, however, we did not observe any statistically significant association between age and sex in HTLV1/2 infection. Nevertheless, in most endemic areas, HTLV-1 prevalence has been shown to increase with age and to be higher in females2424. Murphy EL, Figueroa JP, Gibbs WN, Brathwaite A, Holding-Cobham M, Waters D, et al. Sexual transmission of human T-lymphotropic virus type I (HTLV-I). Ann Intern Med. 1989;111:555-60.. Besides, absence of a molecular test to identify and confirm the circulating strains of HTLV-1 and HTLV-2 has been one of the greater limitations of this study due to unavailability of samples. Nevertheless, using serological tools, we conclusively demonstrated circulation of HTLV-1 and HTLV-2 in blood donors in Amazonas.
In response to the epidemiological situation, one of the major recommendations by the Global Virus Network's Task Force on HTLV is an improved understanding of HTLV epidemiology in diverse populations, which not only will stimulate basic research in identifying disease biomarkers, but will also unravel mechanisms of viral infectivity, persistence, replication and pathogenesis to open insights into novel treatments2525. Willems L, Hasegawa H, Accolla R, Bangham C, Bazarbachi A, Bertazzoni U, et al. Reducing the global burden of HTLV-1 infection: An agenda for research and action. Antiviral Res. 2017;137:41-8.. In this context, further studies are needed to understand the epidemiology of HTLV-1/2 in Amazonas not only to estimate the disease burden, but also to create a mechanism for continued followup and to reduce or block intra-familial transmission.
REFERENCES
-
1Poiesz BJ, Ruscetti FW, Gazdar AF, Bunn PA, Minna JD, Gallo RC. Detection and isolation of type C retrovirus particles from fresh and cultured lymphocytes of a patient with cutaneous T-cell lymphoma. Proc Natl Acad Sci USA. 1980;77:7415-9.
-
2Kalyanaraman VS, Sarngadharan MG, Robert-Guroff M, Miyoshi I, Golde D, Gallo RC. A new subtype of human T-cell leukemia virus (HTLV-II) associated with a T-cell variant of hairy cell leukemia. Science. 1982;218:571-3.
-
3Gessain A, Cassar O. Epidemiological aspects and world distribution of HTLV-1 infection. Front Microbiol. 2012;3:388.
-
4Takahashi K, Takezaki T, Oki T, Kawakami K, Yashiki S, Fujiyoshi T, et al. Inhibitory effect of maternal antibody on mother-to-child transmission of human T-lymphotropic virus type I. The Mother-to-Child Transmission Study Group. Int J Cancer. 1991;49:673-7.
-
5Percher F, Jeannin P, Martin-Latil S, Gessain A, Afonso P, Vidy-Roche A, et al. Mother-to-child transmission of HTLV-1 epidemiological aspects, mechanisms and determinants of mother-to-child transmission. Viruses 2016;8:40.
-
6Paiva A, Casseb J. Sexual transmission of human T-cell lymphotropic virus type 1. Rev Soc Bras Med Trop. 2014;47:265-74.
-
7Stuver SO, Tachibana N, Okayama A, Shioiri S, Tsunetoshi Y, Tsuda K, et al. Heterosexual transmission of human T cell leukemia/lymphoma virus type I among married couples in southwestern Japan: an initial report from the Miyazaki Cohort Study. J Infect Dis. 1993;167:57-65.
-
8Okochi K, Sato H, Hinuma Y. A Retrospective study on transmission of adult T cell leukemia virus by blood transfusion: seroconversion in recipients. Vox Sang. 1984;46:245-53.
-
9Feigal E, Murphy E, Vranizan K, Bacchetti P, Chaisson R, Drummond JE, et al. Human T cell lymphotropic virus types I and II in intravenous drug users in San Francisco: risk factors associated with seropositivity. J Infect Dis. 1991;164:36-42.
-
10Yoshida M, Miyoshi I, Hinuma Y. Isolation and characterization of retrovirus from cell lines of human adult T-cell leukemia and its implication in the disease. Proc Natl Acad Sci U S A. 1982;79:2031-5.
-
11Gessain A, Barin F, Vernant JC, Gout O, Maurs L, Calender A, et al. Antibodies to human T-lymphotropic virus type-I in patients with tropical spastic paraparesis. Lancet. 1985;2:407-10.
-
12Osame M, Usuku K, Izumo S, Ijichi N, Amitani H, Igata A, et al. HTLV-I associated myelopathy, a new clinical entity. Lancet. 1986;1:1031-2.
-
13Takatsuki K, Matsuoka M, Yamaguchi K. Adult T-cell leukemia in Japan. J Acquir Immune Defic Syndr Hum Retrovirol. 1996;13 Suppl 1:S15-9.
-
14Martin F, Taylor GP, Jacobson S. Inflammatory manifestations of HTLV-1 and their therapeutic options. Expert Rev Clin Immunol. 2014;10:1531-46.
-
15Catalan-Soares B, Carneiro-Proietti AB, Proietti FA. Heterogeneous geographic distribution of human T-cell lymphotropic viruses I and II (HTLV-I/II): serological screening prevalence rates in blood donors from large urban areas in Brazil. Cad Saude Publica. 2005;21:926-31.
-
16Passos LN, Moraes MP, Tamegão-Lopes B, Lemos JA, Machado PR, Mira MT, et al. Absence of HTLV-1/2 infection and dermatological diseases in Manaus, State of Amazonas, Brazil. Rev Soc Bras Med Trop. 2014;47:507-9.
-
17Galvão-Castro B, Loures L, Rodriques LG, Sereno A, Ferreira Júnior OC, Franco LG, et al. Distribution of human T-lymphotropic virus type I among blood donors: a nationwide Brazilian study. Transfusion. 1997;37:242-3.
-
18Machado Filho AC, Sardinha JF, Ponte RL, Costa EP, Silva SS, Martinez-Espinosa FE. Prevalência de infecção por HIV, HTLV, VHB e de sífilis e clamídia em gestantes numa unidade de saúde terciária na Amazônia ocidental brasileira. Rev Bras Ginecol Obstet. 2010;32:176-83.
-
19Mueller N. The epidemiology of HTLV-I infection. Cancer Causes Control. 1991;2:37-52.
-
20Ishak R, Harrington WJ Jr, Azevedo VN, Eiraku N, Ishak MO, Guerreiro JF, et al. Identification of human T cell lymphotropic virus type IIa infection in the Kayapo, an indigenous population of Brazil. AIDS Res Hum Retroviruses. 1995;11:813-21.
-
21Paiva A, Casseb J. Origin and prevalence of human T-lymphotropic virus type 1 (HTLV-1) and type 2 (HTLV-2) among indigenous populations in the Americas. Rev Inst Med Trop Sao Paulo. 2015;57:1-13.
-
22Ishak R, Ishak MO, Azevedo VN, Santos DE, Vallinoto AC, Saraiva JC, et al. Detection of HTLV-IIa blood donors in an urban area of the Amazon Region of Brazil (Belém, PA). Rev Soc Bras Med Trop. 1998;31:193-7.
-
23Santos EL, Tamegão-Lopes B, Machado LF, Ishak MO, Ishak R, Lemos JA, et al. Caracterização molecular do HTLV-1/2 em doadores de sangue em Belém, Estado do Pará: primeira descrição do subtipo HTLV-2b na região Amazônica. Rev Soc Bras Med Trop. 2009;42:271-6.
-
24Murphy EL, Figueroa JP, Gibbs WN, Brathwaite A, Holding-Cobham M, Waters D, et al. Sexual transmission of human T-lymphotropic virus type I (HTLV-I). Ann Intern Med. 1989;111:555-60.
-
25Willems L, Hasegawa H, Accolla R, Bangham C, Bazarbachi A, Bertazzoni U, et al. Reducing the global burden of HTLV-1 infection: An agenda for research and action. Antiviral Res. 2017;137:41-8.
Publication Dates
-
Publication in this collection
2017
History
-
Received
08 June 2017 -
Accepted
14 Sept 2017