Abstract
Introduction
Neurotoxoplasmosis (NT) sometimes manifests unusual characteristics.
Methods
We analyzed 85 patients with NT and AIDS according to clinical, cerebrospinal fluid, cranial magnetic resonance, and polymerase chain reaction (PCR) characteristics.
Results
In 8.5%, focal neurological deficits were absent and 16.4% had single cerebral lesions. Increased sensitivity of PCR for Toxoplasma gondii DNA in the central nervous system was associated with pleocytosis and presence of >4 encephalic lesions.
Conclusions
Patients with NT may present without focal neurological deficit and NT may occur with presence of a single cerebral lesion. Greater numbers of lesions and greater cellularity in cerebrospinal fluid improve the sensitivity of PCR to T gondii.
AIDS; Cerebral toxoplasmosis; Diagnosis
Neurotoxoplasmosis (NT) is the most frequent opportunistic infection of the central nervous system (CNS) among individuals with the acquired immunodeficiency syndrome (AIDS). It indicates severe immunodeficiency and, if it remains untreated, it may lead to death11. Nissapatorn V, Lee C, Quek KF, Leong CL, Mahmud R, Abdullah KA. Toxoplasmosis in HIV/AIDS patients: a current situation. J Infect Dis Jpn 2004; 57:160-165..
The number of cases of NT in Brazil and around the world has declined since 1996, the year highly active antiretroviral therapy (HAART) was introduced. Nonetheless, it is still the most common opportunistic infection of the CNS in Brazil and the third most common AIDS-defining disease in São Paulo22. Camara VD, Tavares W, Ribeiro M, Dumas M. Manifestações neurológicas de toxoplasmose em AIDS. J Bras Doenças Sex Transm 2003; 15:46-50..
In countries with a high seroprevalence of toxoplasmosis, the incidence of NT has been estimated to be between 30% and 40% among AIDS patients who are not receiving prophylaxis33. Antinori A, Larussa D, Cingolani A, Lorenzini P, Bossolasco S, Finazzi MG, et al. Prevalence, associated factors, and prognostic determinants of AIDS-related toxoplasmic encephalitis in the era of advanced highly active antiretroviral therapy. Clin Infect Dis 2004; 39:1681-1691.. Even though NT is frequent neurological manifestation, pleomorphic presentations of the infection are not uncommon, and differential diagnoses must include other neurological infections and tumor diseases.
The aim of the present study was to describe important features of NT that might contribute to recognition of the disease, thereby avoiding delayed diagnoses. The descriptive cross-sectional study was conducted between February 2006 and December 2008, at 2 referral centers for neurological diseases in the City of Recife, State of Pernambuco, Brazil, the Hospital da Restauração and the Oswaldo Cruz University Hospital.
Eighty-five AIDS patients were included in the study. They all also had a diagnosis of NT that had been determined using the criteria of the Centers for Disease Control and Prevention (CDC)44. Centers for Disease Control (CDC). 1993 revised classification system for HIV infection and expanded surveillance case definition for AIDS among adolescents and adults. MMWR Recomm Rep 1992; 41(RR-17):1-19.. These criteria include: I) presence of a recent neurological abnormality consistent with intracranial disease; II) evidence of a cerebral lesion with a mass effect viewed using cranial tomography or magnetic resonance imaging (MRI); III) positive serological findings of Toxoplasma gondii; and IV) a therapeutic response to toxoplasmosis treatment. Aliquots of cerebrospinal fluid (CSF) were subjected to real-time and PCR analysis for detection of the b1 gene of T. gondii.
The patients were characterized by variables relating to gender, age, and presence of focal neurological deficits.
We defined the therapeutic response as good in cases in which there was a clinical and radiologic improvement with regression of neurological signs and symptoms following institution of therapy, which consisted of sulfadiazine in association with pyrimethamine or clindamycin, both administered together with folinic acid.
Patient follow up was maintained for an average of 90 days.
This study was approved by the Research Ethics Committee for studies involving human beings, and was given the protocol number 00430.102.172/05.
There were 43 men and 42 women evaluated, with a mean age of 35.8 ± 0.98 years. The time for neurological symptoms to become established ranged from 1 to 90 days, with a median of 14 days. At the time of diagnosis of cerebral toxoplasmosis, 65.8% of the patients were known to have been HIV-positive.
Among the most frequent manifestations, we observed hemiparesis in 75 (88.2.%) cases, headache in 76 (89.4%), and fever in 46 (54.1%).
Thirty-nine (45.9%) patients presented with convulsive crises. Analysis of the presence of seizures in patients with up to 4 encephalic lesions in relation to a group with more than 4 lesions did not show any statistically significant difference (p = 0.89). However, analysis of whether the seizures depended on lesion location showed that patients with cortical lesions had a greater rate of seizures than did a group with deep lesions, and this difference was statistically significant (p = 0.02).
Ten (8.5%) patients did not present with any focal neurological signs, and among this group, the initial manifestation consisted predominantly of onset of convulsive crisis.
With regard to level of consciousness, 71 (83.5%) patients were alert. However, among this group, 47% presented with states of confusion on admission to hospital.
Deficits of cranial nerve pairs were observed in 12 (14.1%) patients, with CN III most (66.6%) frequently affected.
CD4 evaluations revealed a mean count of 66.6 cells/mm33. Antinori A, Larussa D, Cingolani A, Lorenzini P, Bossolasco S, Finazzi MG, et al. Prevalence, associated factors, and prognostic determinants of AIDS-related toxoplasmic encephalitis in the era of advanced highly active antiretroviral therapy. Clin Infect Dis 2004; 39:1681-1691., with 93%-94% of the cases serologically positive for anti-Toxoplasma IgG.
Eighty-three samples of cerebrospinal fluid were evaluated, and pleocytosis was observed in 52 (62.6%) cases, with a mean cell density of 27.1 cells/mm33. Antinori A, Larussa D, Cingolani A, Lorenzini P, Bossolasco S, Finazzi MG, et al. Prevalence, associated factors, and prognostic determinants of AIDS-related toxoplasmic encephalitis in the era of advanced highly active antiretroviral therapy. Clin Infect Dis 2004; 39:1681-1691.. The predominant pattern was lymphomononuclear. High levels of cerebrospinal fluid proteins were also present in 85.3% of the cases, with a mean protein concentration of 104.4mg/dL.
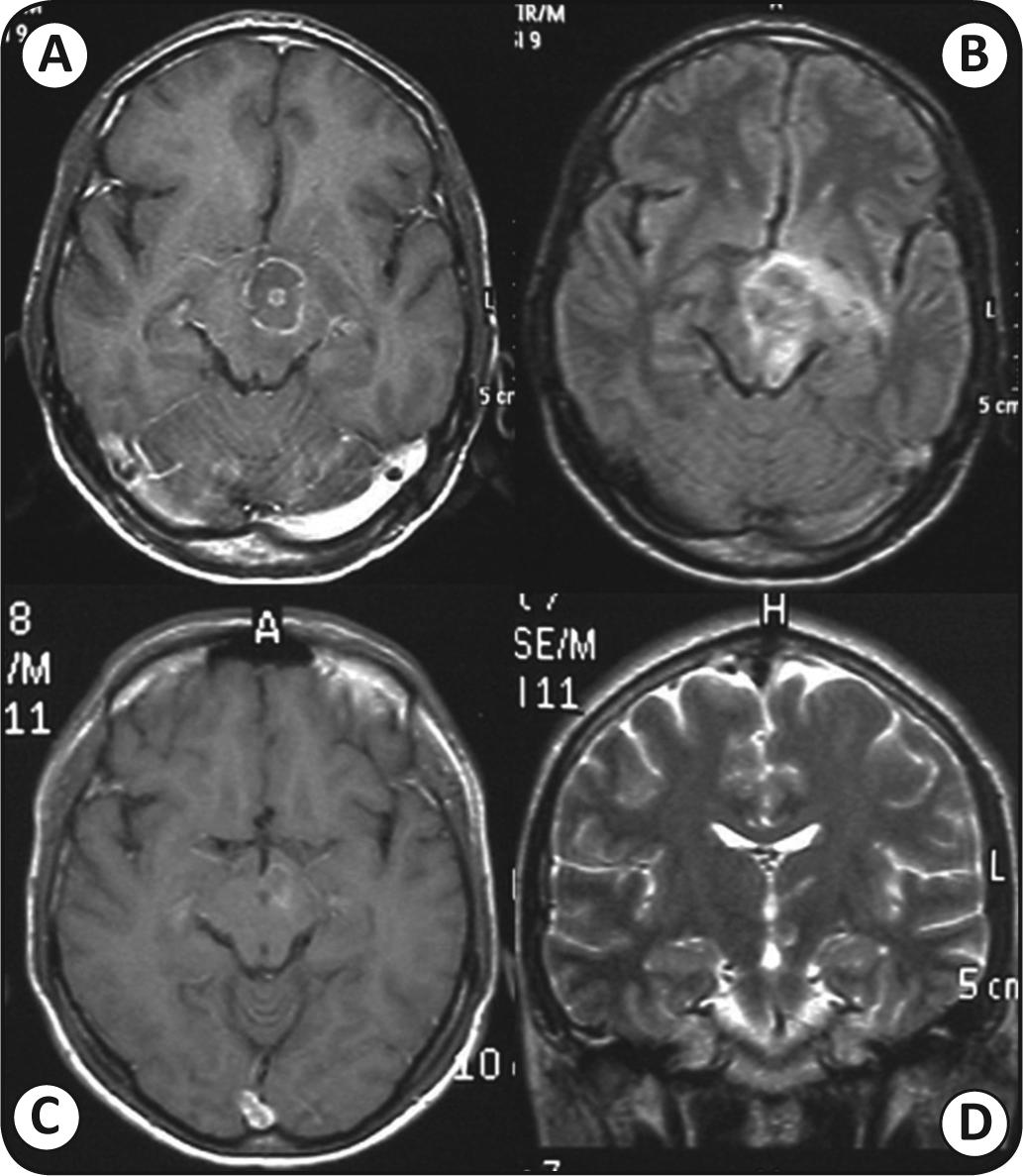
Cranial magnetic resonance imaging (MRI) was performed in 69 cases. Cases with 4 or more encephalic lesions predominated, accounting for 55.1% of all cases. There were 14 (16.4%) patients with single cerebral lesions (Figures 1 and 2).
Mesencephalic lesion with a central target. (A) and (B), sequences before treatment; (C) and (D), sequences after treatment. Note reduction of the lesion and the cerebral edema. Images: author's photos.
Parietal lesion, next to the meningeal plane. (A) and (B), sequences before treatment; (C) and (D), sequences after treatment. Note complete disappearance of the lesion and edema. Images: author's photos.
On analysis of results of real-time PCR seeking T. gondii DNA in the CSF, with regard to cell density in the fluid, 89.7% of samples with more than 4 cells/mm33. Antinori A, Larussa D, Cingolani A, Lorenzini P, Bossolasco S, Finazzi MG, et al. Prevalence, associated factors, and prognostic determinants of AIDS-related toxoplasmic encephalitis in the era of advanced highly active antiretroviral therapy. Clin Infect Dis 2004; 39:1681-1691. also had positive PCR results, whereas 35.8% of specimens with up to 3 cells/mm33. Antinori A, Larussa D, Cingolani A, Lorenzini P, Bossolasco S, Finazzi MG, et al. Prevalence, associated factors, and prognostic determinants of AIDS-related toxoplasmic encephalitis in the era of advanced highly active antiretroviral therapy. Clin Infect Dis 2004; 39:1681-1691. had negative PCR results. This difference was statistically significant (p = 0.013).
Analysis of the CSF PCR results in relation to the number of encephalic lesions (as identified by MRI) showed that 54.3% of patients with a maximum of 3 lesions had negative PCR, whereas 73.9% of the group with 4 or more intracranial lesions had positive results from PCR. This difference was also statistically significant (p = 0.026) (Table 1).
- Cellularity of cerebrospinal fluid and number of encephalic lesions with PCR results from cerebrospinal fluid.
Clinical reassessments of 78 patients were performed, and 64 (82.1%) had improvement defined as full regression or recovery from the signs and symptoms. There were 8 (10.3%) deaths, and the PCR results from the cerebrospinal fluid were positive in all 8 cases. Necropsies were only performed in 2 of these cases, and parasitic pseudocysts of T. gondii were observed in both. It was not possible to obtain data on the evolution of the other cases.
With regard to demographic characteristics, we did not find any predominance of the disease according to gender. Some studies have indicated that NT occurs predominantly among males55. Raffi F, Aboulker JP, Michelet C, Reliquet V, Pelloux H, Huart, et al. A prospective study of criteria for the diagnosis of toxoplasmic encephalitis in 186 AIDS patients. AIDS 1997; 11:177-184.,66. Hernández-González E, Zamora F, Barnès J, Bender-del Busto J, Rodríguez Delgado F, Millan-Marcelo JC. Manifestaciones clínicas de la toxoplasmosis cerebral en pacientes cubanos con Sida. Rev Neurol 2002; 34:618-621., but without demonstrating statistical significance for such an association77. Richards FO, Kovacs JA, Luft BJ. Preventing toxoplasmic encephalitis in persons infected with human immunodeficiency virus. Clin Infec Dis 1995; 21(supl I):49-56.,88. Belanger F, Derouin F, Grangeot-Keros L, Meyer L. Incidence and risk factors for toxoplasmosis in a cohort of human immunodeficiency vírus-infected patients. Clin Infec Dis 1999; 28:575-581.. The mean age of patients in our study was similar to that of other studies11. Nissapatorn V, Lee C, Quek KF, Leong CL, Mahmud R, Abdullah KA. Toxoplasmosis in HIV/AIDS patients: a current situation. J Infect Dis Jpn 2004; 57:160-165.,66. Hernández-González E, Zamora F, Barnès J, Bender-del Busto J, Rodríguez Delgado F, Millan-Marcelo JC. Manifestaciones clínicas de la toxoplasmosis cerebral en pacientes cubanos con Sida. Rev Neurol 2002; 34:618-621..
The clinical characteristics most frequently found in our study were hemiparesis,
headache, and fever. Subacute forms of presentation were mostly observed. Similar characteristics
have also been reported in other series99. Khan AN, Turnbull I, Al-Okaili R, MacDonald S. Imaging in CNS Toxoplasmosis
[Internet]. Medscape's full drug & disease; [Updated 2011 May, 25; Cited 2008 June 10].
Available from: www.emedicine.com/radio/topic703.htm/.
www.emedicine.com/radio/topic703.htm/...
−1111. Montoya JG. Laboratory diagnosis of Toxoplasma gondii infection and
toxoplasmosis. J Infect Dis 2002; 185 (supl I):73-82., with emphasis not only on focal deficits but also with
attention drawn to headaches as an independent variable contributing towards the diagnosis of NT
among individuals with AIDS55. Raffi F, Aboulker JP, Michelet C, Reliquet V, Pelloux H, Huart, et al. A
prospective study of criteria for the diagnosis of toxoplasmic encephalitis in 186 AIDS patients.
AIDS 1997; 11:177-184..
There was significant rate of convulsive crisis (45.9% of the cases), and this was similar to the findings in most other series1212. Kellinghaus C, Engbring C, Kovac S, Möddel G, Boesebeck F, Fischera M, et al. Frequency of seizure and epilepsy in neurological HIV- infected patients. Seizure 2008; 17:27-33.. The seizures occurred during the acute phase of NT, and it was noteworthy that the number of lesions was not associated with the seizure rate, but the presence of cortical lesions decreased the seizure threshold. Another point to mention is that among the patients who were known to have been HIV-positive at the time of NT diagnosis, 62.5% also had experienced previous symptoms, such as chronic diarrhea, weight loss, or histories of pulmonary tuberculosis, that pointed towards the presence of immunodeficiency, but which were not given their value at the time of initial medical care.
Although the CDC clinical criteria for inferring NT among AIDS patients are not pathognomonic, they are accepted by the scientific community. However, absence of these criteria should not be a factor in ruling out this diagnosis given that in our series there were 10 patients without any focal neurological signs, plus a small percentage of individuals who were serologically negative for anti-Toxoplasma IgG, who could have been judged by these criteria to be presenting other diseases.
From a laboratory point of view, traditional methods generally have not provided defining diagnostic information. Serological detection of anti-Toxoplasma IgG antibodies in immunosuppressed individuals serves as an indicator of chronic infection by the parasite, but it does not help to define active disease1313. Borges AS, Figueiredo JFC. Detecção de imunoglobulinas IgG, IgM e IgA anti-Toxoplasma Gondii no soro, líquor e saliva de pacientes com Síndrome de Imunodeficiência Adquirida e Neurotoplasmose. Arq Neuropsiquiatr 2004; 62:1033-1037.. Studies that have evaluated intrathecal synthesis of IgG have shown low sensitivity and consequently little clinical utility1313. Borges AS, Figueiredo JFC. Detecção de imunoglobulinas IgG, IgM e IgA anti-Toxoplasma Gondii no soro, líquor e saliva de pacientes com Síndrome de Imunodeficiência Adquirida e Neurotoplasmose. Arq Neuropsiquiatr 2004; 62:1033-1037.,1414. Potasman I, Resnick L, Luft BJ, Remington J. Intrathecal production of antibodies against Toxoplasma gondii in patients with toxoplasmic encephalitis and the acquired immunodeficiency syndrome (AIDS). Ann Inter Med 1988; 108:49-51.. In routine cerebrospinal fluid analysis, findings of moderate pleocytosis with elevated protein concentrations are nonspecific1313. Borges AS, Figueiredo JFC. Detecção de imunoglobulinas IgG, IgM e IgA anti-Toxoplasma Gondii no soro, líquor e saliva de pacientes com Síndrome de Imunodeficiência Adquirida e Neurotoplasmose. Arq Neuropsiquiatr 2004; 62:1033-1037., but increased cell counts may contribute to greater sensitivity of PCR results, as we found in a previous study1515. Correia CC, Melo HR, Costa VM. Influence of neurotoxoplasmosis characteristics on real-time PCR sensitivity among AIDS patients in Brazil. Trans R Soc Trop Med Hyg 2010; 104:24-28. in which we reported on clinical variables that influenced and thereby improved the performance of the method.
The present study showed that single lesions on MRI were not uncommon in individuals with NT (16.4%). However, this finding has yet to be extensively explored with regard to its significance as a diagnostic clue.
In conclusion, additional clinical and imaging variables, especially alongside results from real-time PCR, must be identified to improve the diagnosis of cerebral toxoplasmosis.
REFERENCES
-
1Nissapatorn V, Lee C, Quek KF, Leong CL, Mahmud R, Abdullah KA. Toxoplasmosis in HIV/AIDS patients: a current situation. J Infect Dis Jpn 2004; 57:160-165.
-
2Camara VD, Tavares W, Ribeiro M, Dumas M. Manifestações neurológicas de toxoplasmose em AIDS. J Bras Doenças Sex Transm 2003; 15:46-50.
-
3Antinori A, Larussa D, Cingolani A, Lorenzini P, Bossolasco S, Finazzi MG, et al. Prevalence, associated factors, and prognostic determinants of AIDS-related toxoplasmic encephalitis in the era of advanced highly active antiretroviral therapy. Clin Infect Dis 2004; 39:1681-1691.
-
4Centers for Disease Control (CDC). 1993 revised classification system for HIV infection and expanded surveillance case definition for AIDS among adolescents and adults. MMWR Recomm Rep 1992; 41(RR-17):1-19.
-
5Raffi F, Aboulker JP, Michelet C, Reliquet V, Pelloux H, Huart, et al. A prospective study of criteria for the diagnosis of toxoplasmic encephalitis in 186 AIDS patients. AIDS 1997; 11:177-184.
-
6Hernández-González E, Zamora F, Barnès J, Bender-del Busto J, Rodríguez Delgado F, Millan-Marcelo JC. Manifestaciones clínicas de la toxoplasmosis cerebral en pacientes cubanos con Sida. Rev Neurol 2002; 34:618-621.
-
7Richards FO, Kovacs JA, Luft BJ. Preventing toxoplasmic encephalitis in persons infected with human immunodeficiency virus. Clin Infec Dis 1995; 21(supl I):49-56.
-
8Belanger F, Derouin F, Grangeot-Keros L, Meyer L. Incidence and risk factors for toxoplasmosis in a cohort of human immunodeficiency vírus-infected patients. Clin Infec Dis 1999; 28:575-581.
-
9Khan AN, Turnbull I, Al-Okaili R, MacDonald S. Imaging in CNS Toxoplasmosis [Internet]. Medscape's full drug & disease; [Updated 2011 May, 25; Cited 2008 June 10]. Available from: www.emedicine.com/radio/topic703.htm/.
» www.emedicine.com/radio/topic703.htm/ -
10Skiest DJ. Focal neurological disease in patients with acquired immunodeficiency syndrome. Clin Infect Dis 2002; 34:103-115.
-
11Montoya JG. Laboratory diagnosis of Toxoplasma gondii infection and toxoplasmosis. J Infect Dis 2002; 185 (supl I):73-82.
-
12Kellinghaus C, Engbring C, Kovac S, Möddel G, Boesebeck F, Fischera M, et al. Frequency of seizure and epilepsy in neurological HIV- infected patients. Seizure 2008; 17:27-33.
-
13Borges AS, Figueiredo JFC. Detecção de imunoglobulinas IgG, IgM e IgA anti-Toxoplasma Gondii no soro, líquor e saliva de pacientes com Síndrome de Imunodeficiência Adquirida e Neurotoplasmose. Arq Neuropsiquiatr 2004; 62:1033-1037.
-
14Potasman I, Resnick L, Luft BJ, Remington J. Intrathecal production of antibodies against Toxoplasma gondii in patients with toxoplasmic encephalitis and the acquired immunodeficiency syndrome (AIDS). Ann Inter Med 1988; 108:49-51.
-
15Correia CC, Melo HR, Costa VM. Influence of neurotoxoplasmosis characteristics on real-time PCR sensitivity among AIDS patients in Brazil. Trans R Soc Trop Med Hyg 2010; 104:24-28.
Publication Dates
-
Publication in this collection
May-Jun 2013
History
-
Received
13 June 2011 -
Accepted
11 Nov 2011