Abstract:
INTRODUCTION:
Before 2004, the occurrence of acute Chagas disease (ACD) by oral transmission associated with food was scarcely known or investigated. Originally sporadic and circumstantial, ACD occurrences have now become frequent in the Amazon region, with recently related outbreaks spreading to several Brazilian states. These cases are associated with the consumption of açai juice by waste reservoir animals or insect vectors infected with Trypanosoma cruzi in endemic areas. Although guidelines for processing the fruit to minimize contamination through microorganisms and parasites exist, açai-based products must be assessed for quality, for which the demand for appropriate methodologies must be met.
METHODS:
Dilutions ranging from 5 to 1,000 T. cruzi CL Brener cells were mixed with 2mL of acai juice. Four Extraction of T. cruzi DNA methods were used on the fruit, and the cetyltrimethyl ammonium bromide (CTAB) method was selected according to JRC, 2005.
RESULTS:
DNA extraction by the CTAB method yielded satisfactory results with regard to purity and concentration for use in PCR. Overall, the methods employed proved that not only extraction efficiency but also high sensitivity in amplification was important.
CONCLUSIONS:
The method for T. cruzi detection in food is a powerful tool in the epidemiological investigation of outbreaks as it turns epidemiological evidence into supporting data that serve to confirm T. cruzi infection in the foods. It also facilitates food quality control and assessment of good manufacturing practices involving acai-based products.
Keywords:
Chagas disease; Trypanosoma cruzi; Euterpe (açai); PCR detection; Food analysis
INTRODUCTION
In Brazil, both inside and outside the Amazon region, several cases of acute Chagas disease (ACD) have been recorded as an outbreak characterized by a group of individuals gathered in one place who, by ingesting the same type of food, became sick almost simultaneously with fever and general manifestations of a systemic infection11. Ferreira RTB, Branquinho MR, Cardarelli-Leite P. Transmissão oral da doença de Chagas pelo consumo de açaí: um desafio para a vigilância sanitária. Vigil Sanit Debate 2014; 2:4-11..
Until the year 2004, the occurrence of ACD by oral transmission, as associated with food consumption, constituted a scarcely known or investigated event. For the same reason, what was originally considered a sporadic and circumstantial occurrence has now become frequent in the Amazon region, which is associated with the occurrence of recent outbreaks in several Brazilian states22. Ministério da Saúde. Agência Nacional de Vigilância Sanitária. Informe Técnico nº 35 de 19 de junho de 2008. Gerenciamento do Risco Sanitário na Transmissão de Doença de Chagas Aguda por Alimentos (Internet). Brasília: Ministério da Saúde; 2008 (Cited 2009 November 11). Available at: Available at: http://www.portal.anvisa.gov.br/wps/content/Anvisa+Portal/Anvisa/Inicio/Alimentos/Assuntos+de+Interesse/Informes+Tecnicos/2008-06-19-35
http://www.portal.anvisa.gov.br/wps/cont...
. Recent cases of ACD in Brazil are related to the consumption of açai juice where the contamination of the fruit itself or the pulp occurs through waste reservoir animals or insect vectors infected with Trypanosoma cruzi in endemic areas33. Passos LAC, Guaraldo AMA, Alves DP. Relatório final, convênio 667/ 2008 com Ministério da Saúde: "Análise da interferência da polpa de açaí na transmissão oral de Trypanosoma cruzi, contribuindo para o surgimento de surtos de Doença de Chagas Aguda (DCA) na região Norte do Brasil". Campinas, SP; 2010 2010.) (44. Pereira KS, Schmidt FL, Guaraldo AMA, Franco RMB, Dias VL, Passos LAC. Chagas disease as a food borne illness. J Food Protect 2009; 72:441-446.) (55. Valente SAS, Valente VC, Pinto AYN, César MJB, Santos MP, Miranda COS, et al. Analysis of an acute Chagas disease outbreak in the Brazilian Amazon: human cases, triatomines, reservoir mammals and parasites. Trans R Soc Trop Med Hyg 2009; 103: 291-297..
Açai (Euterpe oleracea Mart.) is a typical and popular berry fruit of the Amazon region in Northern Brazil, which in recent years has gained importance due to the health benefits associated with its phytochemical composition and antioxidant capacity.
Brazil is the world's largest producer, consumer, and exporter of açai. This fruit is marketed and consumed by the local people in the producing areas of Pará, Maranhão, Amapá, Rondônia, and Acre, which together comprise a distinct region considered endemic for the disease66. Homma AKO, Nogueira OL, Menezes AJEA, Carvalho JEU, Nicoli CML, Matos GB. Açaí: Novos desafios e tendências. Amazônia: Ci & Desen 2006; 1:7-23.) (77. Menezes EMS, Torres AT, SabaaSrur AU. Valor nutricional da polpa de açaí (Euterpe oleracea Mart.) liofilizada. Acta Amaz 2008; 38:311-316.. With the increasing growth in commerce, not only has the consumption of the product spread across larger Brazilian cities, but it is now also being exported to America, Japan, China, and some European countries77. Menezes EMS, Torres AT, SabaaSrur AU. Valor nutricional da polpa de açaí (Euterpe oleracea Mart.) liofilizada. Acta Amaz 2008; 38:311-316..
The Ministry of Agriculture and Supply, through Normative Instruction number 1 of January 07, 2000, issued the technical regulation for setting the standards of identity and quality for fruit pulp, which includes acai pulp88. Ministério da Agricultura e do Abastecimento. Instrução Normativa no1, de 7 de janeiro de 2000. Regulamentação técnica para fixação dos padrões de Identidade e Qualidade para polpas de frutas. Diário Oficial da União, 10 jan. 2000. Brasília: Ministério da Agricultura e do Abastecimento; 2000.. Pertinently, the pulp should be obtained from fruit that is fresh, sound, mature, and free of soil, dirt, parasites, and microorganisms that can render the product unfit for consumption88. Ministério da Agricultura e do Abastecimento. Instrução Normativa no1, de 7 de janeiro de 2000. Regulamentação técnica para fixação dos padrões de Identidade e Qualidade para polpas de frutas. Diário Oficial da União, 10 jan. 2000. Brasília: Ministério da Agricultura e do Abastecimento; 2000..
Although the guidelines for the processing of the fruit have been established with a view to minimize contamination through microorganisms and parasites, it is necessary to assess the quality of acai-based products, for which appropriate methodologies must be adopted99. Empresa Brasileira de Pesquisa Agropecuária (EMBRAPA). Sistema de produção do açaí. (Internet). Brasília, DF: 2008 (Cited 2013 November 1). Available at: Available at: http://www.sistemasdeproducao.cnptia.embrapa.br/FontesHtML/Acai/SistemaProducao Acai_2ed/paginas/apresentacao.htm.
http://www.sistemasdeproducao.cnptia.emb...
.
Several recommendations and incentives on research methodologies for the detection of T. cruzi in food exist22. Ministério da Saúde. Agência Nacional de Vigilância Sanitária. Informe Técnico nº 35 de 19 de junho de 2008. Gerenciamento do Risco Sanitário na Transmissão de Doença de Chagas Aguda por Alimentos (Internet). Brasília: Ministério da Saúde; 2008 (Cited 2009 November 11). Available at: Available at: http://www.portal.anvisa.gov.br/wps/content/Anvisa+Portal/Anvisa/Inicio/Alimentos/Assuntos+de+Interesse/Informes+Tecnicos/2008-06-19-35
http://www.portal.anvisa.gov.br/wps/cont...
) (1010. Perez-Gutierrez E, Salvatella R, Figueroa R (orgs). Consulta técnica em epidemiologia, prevenção e manejo da transmissão da doença de Chagas como doença transmitida por alimentos. Relatório Técnico. PANAFTOSA; Rio de Janeiro, 2006. Rev Soc Bras Med Trop 2006; 39:512-514.; however, fully developed methods aimed at the detection of parasites in food are still lacking not only for furnishing a response to epidemiological investigations in cases of outbreaks, but also for controlling the quality of commercialized products.
Molecular methods for the quality control of foods are a reality, and as such, deserve special attention with regard to their availability for the regulation of products subject to sanitary surveillance.
Polymerase chain reaction (PCR) is widely used for the detection of food pathogens. Due to its high sensitivity and specificity, PCR-based methods have the advantage of providing rapid and precise identification of the pathogen and can therefore serve as a powerful tool in the hygienic and sanitary assessment of açai-based foods.
Polymerase chain reaction requires a high-quality amplifiable deoxyribonucleic acid (DNA). Therefore, the choice of the method for DNA extraction is of great importance and constitutes a pre-requisite for molecular analysis. DNA amplification by PCR is influenced by the structural integrity of the DNA and the presence of matrix co-purification inhibitors or extraction reagents, which may reduce PCR efficiency.
In foods, the problems most often linked to DNA extraction include contamination by phenols and polysaccharides, among others. Açai is composed of lipids, carbohydrates, fiber, vitamin E, proteins, minerals (Mn, Fe, Zn, Cu, and Cu), and antioxidant compounds such as anthocyanins, and phenolic compounds1111. Portinho JA, Zimmermann LM, Bruck MR. Beneficial effects of açaí. Int J Nutrol 2012; 5:15-20., which pose a big challenge for DNA extraction from the parasites in such a complex food matrix.
In this context, the aim of this study was to compare the quantity and quality of T. cruzi DNA isolated from açai artificially contaminated with T. cruzi cells, whereby four different DNA extraction methods were applied, and spectrophotometric absorbance readings were used to confirm the quality of the extracted DNA by PCR amplification of the gp85 gene superfamily/sialidase.
METHODS
Staining
The stains used in the present work were for the identification of 1) Leishmania (Leishmania (V.) braziliensis MHOM/BR/1975/M2903 - IOC-L 0566, Leishmania (L.) amazonensis IFLA/BR/1967/PH8 - IOC-L 575), Leishmania Collection (CLIOC/FIOCRUZ), Instituto Oswaldo Cruz; 2) Trypanosoma cruzi CL Brener, Instituto Nacional de Infectologia Evandro Chagas, Fundação Oswaldo Cruz (INI/FIOCRUZ); and 3) Trypanosoma rangeli COLPROT 273, Protozoa Collection (COLPROT/FIOCRUZ), Fundação Oswaldo Cruz.
Artificial contamination of açai
Labeled açai medium, one kilogram of frozen fruit pulp was purchased from a supermarket in Rio de Janeiro, Brazil and thawed at 4°C. The pulp was then homogenized in a Seward Stomacher(r)400 Laboratory Blender (Seward, UK) and distributed in 50-mL polypropylene vials. For each extraction method, five 30-mL glass bottle vials received 2mL of the homogenate of açai pulp and T. cruzi CL Brener with a cell suspension rate of 1,000 (Dilution 1), 100 (Dilution 2), 50 (Dilution 3), 10 (Dilution 4), and 5 (Dilution 5) parasite cells. This procedure was repeated three times, resulting in 15 glass bottles used per method. The homogenate of acai pulp and T. cruzi was subjected to a freeze-drying for about 20 hours for reducing the volume of the mixture and for obtaining a lyophilisate to allow efficient extraction.
DNA extraction
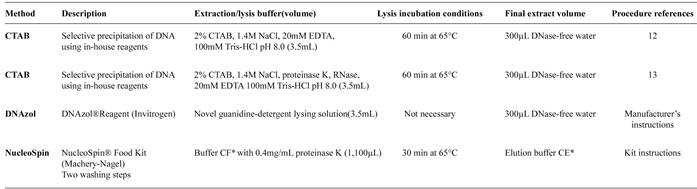
Four different methods for genomic DNA extraction were tested as displayed in Table 1. Two of these were performed using cetyltrimethyl ammonium bromide (CTAB), as described by Cardarelli et al. 20051212. Cardarelli-Leite P, Branquinho MR, Ferreira RTB. Detection of GMO in food products in Brasil: the INCQS experience. Food control 2005; 16:859-866. and the Joint Research Centre 20051313. Join Research Centre (JRC). Event-specific method for the quantification of maize line NK603 using real-time PCR protocol. (Internet). 2005 (Cited 2015 October 1). Available at: Available at: http://www.gmo-crl.jrc.ec.europa.eu/statusofdoss.htm.
http://www.gmo-crl.jrc.ec.europa.eu/stat...
, and the other two were performed using commercially available agents, namely DNAzol(r) (Invitrogen) and Nucleo Spin(r) (MACHEREY-NAGEL), strictly in accordance with the manufacturer's recommendations. The extractions were performed in triplicate for each method as indicated in Table 1.
Assessment of concentration and purity of the extracted DNA
Deoxyribonucleic acid concentration was assessed by spectrophotometric measurement of the optical density (OD) at 260 nm using the GeneQuant TM equipment pro ribonucleic acid/deoxyribonucleic acid (RNA/DNA) calculator (Amersham Biosciences, UK). Ratios of the absorbance at A260/280 and A260/230 were determined to verify the purity of the preparations, which yielded samples that were found to be of acceptable quality and integrity.
PCR for the detection of Trypanosoma cruzi
Standardization of the PCR for evaluating its specificity was performed using strains of Leishmania chagasi, L. braziliensis, Trypanosoma rangeli, and T. cruzi, where a pair of T. cruzi-specific primers were employed to amplify a telomeric region of the gp85/sialidase superfamily (Tc189Fw2 - 5'CCAACGCTCCGGGAAAAC-3' and Tc189RV3 - 5'GCGTCTTCTCAGTATGGACTT-3'), as described by Chiurillo et al.1414. Chiurillo MA, Crisante G, Rojas A, Peralta A, Dias M, Guevara, et al. Detection of Trypanosoma cruzi and Trypanosoma rangeli Infection by duplex PCR assay based on telomeric sequences. Clin Diagn Lab Immunol 2003; 10:775-779..
After standardization, the same protocol was used for the detection of T. cruzi in different concentrations of cells mixed with açai. The negative and positive controls used for the PCR were T. rangeli and T. cruzi, respectively. The fragments amplified by PCR were separated by agarose gel electrophoresis (2%w/v) containing 0.3µg/mL ethidium bromide immersed in 1X tris-borato-ethylenediaminetetraacetic acid (TBE) buffer (90mM Tris-borate, 2mM EDTA pH 8, 4), visualized on the "UVP TM-20 Dual-Intensity transilluminator" (Cole-Parmer, US), and recorded in the image analyzer "Image Quant 300 Imager" (Amersham Biosciences, UK).
Validation of the selected DNA extraction method
Eighteen glass bottles containing about 2mL of açai were contaminated with 1,000 T. cruzi CL Brener cells and subjected to lyophilization. DNA was extracted according to the Join Research Centre (JRC) method, 2005, in 6 replicates, repeated for 3 consecutive days. After extraction, the concentration and purity of the extracted DNA were evaluated using the methods described above. The size of the extracted DNA was assessed by electrophoresis on 1% agarose gel. About 10µL of the DNA solution was deposited on an agarose gel in TBE 1X at 80 V for 1 hour.
RESULTS
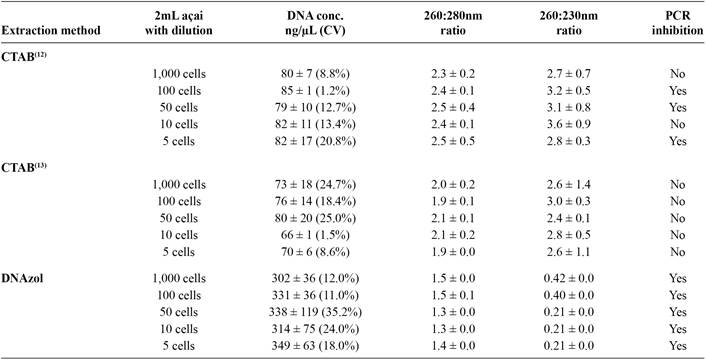
The DNA extracted using the NucleoSpin(r) method showed no reading in the spectrophotometer. The average concentration of the DNA extracted from açai infected with T. cruzi cells using the other three methods is shown in Table 2.
All three methods yielded sufficient quantities of DNA to allow PCR detection. Although the DNA extracted using the DNAzol method produced a four times greater value (about 320ng/µL) than the other two methods using CTAB (about75ng/µL), the eluate with the extracted DNA had a brownish color due to the inefficiency of the method in eliminating the pigments present in açai. This may have led to overestimation of the DNA concentration and/or may have inhibited the PCR.
The purity of the DNA extracted using the three methods after the removal of outliers using the Grubbs test is shown in Table 2.
The A260/A280 ratio is used to test for the presence of proteins and thus helps estimate the purity of nucleic acids. For pure DNA, the value must be between 1.8 and 2.0. The A260/A230 reflects contamination by substances such as carbohydrates, peptides, phenols, and aromatic compounds. In the case of pure samples, the ratio should be above 21515. Corbisier P, Broothaerts W, Gioria S, Schimmel H, Burns M, Baoutina A, et al. Toward metrological traceability for DNA fragment ratios in GM quantification. 1. Effect of DNA extraction methods on the quantitative determination of Bt 176 corn by Real-Time PCR. J Agric Food Chem 2007; 55:3249-3257..
With respect to the A260/A280 ratios obtained, the method using CTAB, RNase, proteinase, and 70% ethanol washes was able to extract DNA with higher purity1313. Join Research Centre (JRC). Event-specific method for the quantification of maize line NK603 using real-time PCR protocol. (Internet). 2005 (Cited 2015 October 1). Available at: Available at: http://www.gmo-crl.jrc.ec.europa.eu/statusofdoss.htm.
http://www.gmo-crl.jrc.ec.europa.eu/stat...
. The DNA extracted using DNAzol proved to exhibit a high protein contamination ratio, with values appreciably below 1.8.
With reference to the A260/A230 ratio, the method that came closest to the expected value was the CTAB method1313. Join Research Centre (JRC). Event-specific method for the quantification of maize line NK603 using real-time PCR protocol. (Internet). 2005 (Cited 2015 October 1). Available at: Available at: http://www.gmo-crl.jrc.ec.europa.eu/statusofdoss.htm.
http://www.gmo-crl.jrc.ec.europa.eu/stat...
. The use of DNAzol resulted in values far below expectation, wherein the DNA extracted showed a high degree of chemical contamination. The specificity of PCR for T. cruzi DNA was established with all four strains belonging to the Trypanosomatidae family (Figure 1).
Specificity of PCR for Trypanosoma cruzi established with four strains belonging to the Trypanosomatidae family. Lanes 1 and 7: contain a 100-bp size ladder (Gibco-BRL); Lane 2: H2O; Lane 3: Leishmania amazonensis; Lane 4: Leishmania braziliensis; Lane 5: Trypanosoma rangeli; Lane 6: Trypanosoma cruzi cL Brener. PCR: polymerase chain reaction.
Five dilutions of the DNA extracted using the CTAB method1313. Join Research Centre (JRC). Event-specific method for the quantification of maize line NK603 using real-time PCR protocol. (Internet). 2005 (Cited 2015 October 1). Available at: Available at: http://www.gmo-crl.jrc.ec.europa.eu/statusofdoss.htm.
http://www.gmo-crl.jrc.ec.europa.eu/stat...
allowed PCR amplification; the results can be observed both in Table 2 and in Figure 2.
PCR amplification of the DNA extracted from the homogenate of açai with Trypanosoma cruzi cL Brener with a cell suspension rate of 1,000 to 5 parasite cells using 3 extraction methods. A) Lanes 1 to 5: DNAzol extraction method (no amplification); Lane 6: 100-bp size ladder (Gibco-BRL); Lanes 7 to 11: CTAB extraction method1212. Cardarelli-Leite P, Branquinho MR, Ferreira RTB. Detection of GMO in food products in Brasil: the INCQS experience. Food control 2005; 16:859-866.; Lane 12: Trypanosoma cruzi cL Brener (positive control). B) Lanes 1 and 9: 100-bp size ladder (Gibco-BRL); Lanes 3 to 7: CTAB extraction method1313. Join Research Centre (JRC). Event-specific method for the quantification of maize line NK603 using real-time PCR protocol. (Internet). 2005 (Cited 2015 October 1). Available at: Available at: http://www.gmo-crl.jrc.ec.europa.eu/statusofdoss.htm.
http://www.gmo-crl.jrc.ec.europa.eu/stat... . PCR: polymerase chain reaction; DNA: deoxyribonucleic acid; CTAB: cetyltrimethyl ammonium bromide.
The result of the validation of the 18 replicas (first day: 69.2, 66.0, 69.4, 72.4, 84.6, 84.4; second day: 78.2, 52.4, 61.0, 75.2, 54.0, 63.6; and third day: 73.4, 66.6, 76.0, 63.0, 87.2, 74.6) bears out an average of 70.6ng/µL, a standard deviation of 9.8ng/µL, and a coefficient of variation of 13.9%. No PCR inhibition was observed in the agarose gel, and amplicons of 100bp were generated in all the 18 replicates (Figure 3).
Agarose gel electrophoresis of all the 18 amplified products obtained from the validation assay with primers Tc189Fw2 and Tc189Rv3. Top - Lane 1: H2O; Lane 2: Trypanosoma rangeli (negative control); Lane 3: 100-bp size ladder (Gibco-BRL); Lanes 4 to 10: homogenate of açai with Trypanosoma cruzi CL Brener with a 100-cell suspension. Bottom - Lanes 1 to 10: homogenate of açai with Trypanosoma cruzi CL Brener with a 100-cell suspension; Lane 11: T. cruzi cL Brener (positive control); Lane 12: 100-bp size ladder.
DISCUSSION
To date, no studies evaluating a methodology for T. cruzi DNA extraction directly from food have been conducted. This study was aimed at identifying an efficient method for the extraction of T. cruzi DNA from T. cruzi-contaminated acai. Although the Nucleo Spin method has been developed for various types of food such as chocolate-based products and fruit concentrates, among others, it was shown to not be adequate for the detection T. cruzi contamination in this study. DNA extraction by the CTAB method yielded satisfactory results with regard to purity and concentration for use in PCR.
The method employing the CTAB detergent for cell lysis is widely used for genetically modified organisms (GMO) analysis of raw or processed food; it is also commonly used for plants, especially in combination with other substances such as β-mercaptoethanol1616. Devi KD, Punyarani K, Singh NS, Devi HS. An efficient protocol for total DNA extraction from the members of order Zingiberales-suitable for diverse PCR based downstream applications. Springer Plus 2013; 2:669.) (1717. Minas K, McEwan NR, Newbold CJ, Scott KP. Optimization of a high-throughput CTAB-based protocol for the extraction of qPCR-grade DNA from rumen fluid, plant and bacterial pure cultures. Fems Microbiol Lett 2011; 325:162-169.) (1818. Mondal TK, Ganie SA. Identification and characterization of salt responsive miRNA-SSRmarkers in rice (Oryza sativa). Gene 2014; 535:204-209.. The major difference between the two methods using the CTAB detergent for cell lysis tested in this study was the use of two different enzymes, RNases and Proteinase K, and the 70% ethanol wash1313. Join Research Centre (JRC). Event-specific method for the quantification of maize line NK603 using real-time PCR protocol. (Internet). 2005 (Cited 2015 October 1). Available at: Available at: http://www.gmo-crl.jrc.ec.europa.eu/statusofdoss.htm.
http://www.gmo-crl.jrc.ec.europa.eu/stat...
, which confirms the high sensitivity of the method with amplification of up to 2.5 cells/mL of acai.
Contamination of food by T. cruzi may occur especially when triatomines deposit their droppings on the surface of infected food or food ingredients, or when these triatomines are crushed with foods, mainly during fruit juice processing, such as in the processing of açai juice.
Equally important in the epidemiology of foodborne acute Chagas disease (ACD) is the contamination of equipment and utensils, contamination during transport, and contamination in the food processing environment, where it typically occurs by either urine or anal secretions of marsupials as well as by feces of infected bugs caused due to human invasion of forests55. Valente SAS, Valente VC, Pinto AYN, César MJB, Santos MP, Miranda COS, et al. Analysis of an acute Chagas disease outbreak in the Brazilian Amazon: human cases, triatomines, reservoir mammals and parasites. Trans R Soc Trop Med Hyg 2009; 103: 291-297.) (1010. Perez-Gutierrez E, Salvatella R, Figueroa R (orgs). Consulta técnica em epidemiologia, prevenção e manejo da transmissão da doença de Chagas como doença transmitida por alimentos. Relatório Técnico. PANAFTOSA; Rio de Janeiro, 2006. Rev Soc Bras Med Trop 2006; 39:512-514.) (1919. Organização Panamericana de Saúde, Organização Mundial de Saúde, PANAFTOSA-VP. Guia para vigilância, prevenção, controle e manejo clínico da doença de Chagas aguda transmitida por alimentos. Rio de Janeiro: PANAFTOSA-VP/OPAS/OMS; 2009..
So far, it has not been possible to analyze the type of food involved in the outbreaks of orally transmitted Chagas disease to evidence the presence of the parasite. However, as quoted above, PANAFTOSA1919. Organização Panamericana de Saúde, Organização Mundial de Saúde, PANAFTOSA-VP. Guia para vigilância, prevenção, controle e manejo clínico da doença de Chagas aguda transmitida por alimentos. Rio de Janeiro: PANAFTOSA-VP/OPAS/OMS; 2009. proposes hypotheses that point to an explanation for the several outbreaks that have occurred in recent years in Northern Brazil, which were associated with the intake of possibly T. cruzi-contaminated açai juice.
The recorded cases of ACD in the outbreaks are related to the consumption of handmade acai juice.
In Latin America, around 100 million individuals suffer from Chagas disease acquired by various types of transmission. Additionally, the estimated number of people infected with T. cruzi in non-endemic countries for the disease is >300,000 in the United States; >5,500 in Canada; >80,000 in Europe and Western Pacific Regions; >3,000 in Japan; and >1,500 in Australia2020. Coura JR, Viñas PA. Chagas disease: a new worldwide challenge. Nature 2010; 465:56-57.. Migratory movements and the subsequent transmission by blood transfusion and organ transplantation have been suggested as the cause of the spread of T. cruzi around the World.
In Brazil, 2 million individuals are infected with T. cruzi2121. World Health Organization. Crompton DWT, Peters P, editors. Working to Overcome the Global Impact of Neglected Tropical Diseases: First WHO Report on Neglected Tropical Diseases. Nonserial Publication Series. WHO report on neglected tropical diseases. volume 1; 2010. 172p.. Considering the high consumption of açai by tourists visiting the country, plus Brazilians themselves traveling to the above-mentioned non-endemic areas of the world, the oral transmission is assumed to be contributing to the increase in overseas cases. This assumption is rooted in the fact that, having acquired the asymptomatic disease, or even the manifestation of symptoms similar to those of other types of illnesses, visitors to Brazil are likely to return to their countries where blood banks may not be equipped with the proper framework required to provide an effective diagnosis of the disease.
Barbosa-Labello et al, in 20102222 ;Barbosa-Labello R. Transmissão oral do Trypanosoma cruzi pela polpa de açaí em camundongos. 2010 [dissertation]. [São Paulo]: Universidade Estadual de Campinas; 2010. 120 p. , used animal models to show that, in addition to surviving in açai pulp by different incubation periods and under different low temperatures treatments, T. cruzi was also able to preserve its virulence in mice. Regarded to be of great epidemiological importance, this finding discards the use of cooling processes and conventional freezing during long periods, which have been previously used as control methods for orally transmitted ACD. This finding also reinforces the need to use bleaching as for the conservation of açai.
In consideration of the above findings, safety measures must be taken at all stages of the commercial or craft supply chain during the processing of açai. The development of large-scale detection and isolation methods for T. cruzi in food products is essential so that epidemiological evidence may serve as supporting data to certify that the food in question is actually contaminated with the parasite. Concurrently, despite the fact that DNA identification in foods itself does not constitute a methodology to assess the viability of the parasite, it can become an assessment tool to ensure proper application of good manufacturing practices.
In the case of açai, these tests will allow the identification of possible contamination during the production chain. This procedure is of great importance since oral transmission aggravates an epidemic of the disease, and disposes off refrigeration and food freezing processes as control methods for the oral transmission of ACD, thus inducing a new reflection, strongly related to underdevelopment33. Passos LAC, Guaraldo AMA, Alves DP. Relatório final, convênio 667/ 2008 com Ministério da Saúde: "Análise da interferência da polpa de açaí na transmissão oral de Trypanosoma cruzi, contribuindo para o surgimento de surtos de Doença de Chagas Aguda (DCA) na região Norte do Brasil". Campinas, SP; 2010 2010..
ACKNOWLEDGMENTS
The authors thank Dra Maria de Fátima Madeira from Instituto Nacional de Infectologia Evandro Chagas/Fundação Oswaldo Cruz (INI/FIOCRUZ) for their support in cultivating strains of trypanosomatídeos.
-
1Ferreira RTB, Branquinho MR, Cardarelli-Leite P. Transmissão oral da doença de Chagas pelo consumo de açaí: um desafio para a vigilância sanitária. Vigil Sanit Debate 2014; 2:4-11.
-
2Ministério da Saúde. Agência Nacional de Vigilância Sanitária. Informe Técnico nº 35 de 19 de junho de 2008. Gerenciamento do Risco Sanitário na Transmissão de Doença de Chagas Aguda por Alimentos (Internet). Brasília: Ministério da Saúde; 2008 (Cited 2009 November 11). Available at: Available at: http://www.portal.anvisa.gov.br/wps/content/Anvisa+Portal/Anvisa/Inicio/Alimentos/Assuntos+de+Interesse/Informes+Tecnicos/2008-06-19-35
» http://www.portal.anvisa.gov.br/wps/content/Anvisa+Portal/Anvisa/Inicio/Alimentos/Assuntos+de+Interesse/Informes+Tecnicos/2008-06-19-35 -
3Passos LAC, Guaraldo AMA, Alves DP. Relatório final, convênio 667/ 2008 com Ministério da Saúde: "Análise da interferência da polpa de açaí na transmissão oral de Trypanosoma cruzi, contribuindo para o surgimento de surtos de Doença de Chagas Aguda (DCA) na região Norte do Brasil". Campinas, SP; 2010 2010.
-
4Pereira KS, Schmidt FL, Guaraldo AMA, Franco RMB, Dias VL, Passos LAC. Chagas disease as a food borne illness. J Food Protect 2009; 72:441-446.
-
5Valente SAS, Valente VC, Pinto AYN, César MJB, Santos MP, Miranda COS, et al. Analysis of an acute Chagas disease outbreak in the Brazilian Amazon: human cases, triatomines, reservoir mammals and parasites. Trans R Soc Trop Med Hyg 2009; 103: 291-297.
-
6Homma AKO, Nogueira OL, Menezes AJEA, Carvalho JEU, Nicoli CML, Matos GB. Açaí: Novos desafios e tendências. Amazônia: Ci & Desen 2006; 1:7-23.
-
7Menezes EMS, Torres AT, SabaaSrur AU. Valor nutricional da polpa de açaí (Euterpe oleracea Mart.) liofilizada. Acta Amaz 2008; 38:311-316.
-
8Ministério da Agricultura e do Abastecimento. Instrução Normativa no1, de 7 de janeiro de 2000. Regulamentação técnica para fixação dos padrões de Identidade e Qualidade para polpas de frutas. Diário Oficial da União, 10 jan. 2000. Brasília: Ministério da Agricultura e do Abastecimento; 2000.
-
9Empresa Brasileira de Pesquisa Agropecuária (EMBRAPA). Sistema de produção do açaí. (Internet). Brasília, DF: 2008 (Cited 2013 November 1). Available at: Available at: http://www.sistemasdeproducao.cnptia.embrapa.br/FontesHtML/Acai/SistemaProducao Acai_2ed/paginas/apresentacao.htm.
» http://www.sistemasdeproducao.cnptia.embrapa.br/FontesHtML/Acai/SistemaProducao Acai_2ed/paginas/apresentacao.htm. -
10Perez-Gutierrez E, Salvatella R, Figueroa R (orgs). Consulta técnica em epidemiologia, prevenção e manejo da transmissão da doença de Chagas como doença transmitida por alimentos. Relatório Técnico. PANAFTOSA; Rio de Janeiro, 2006. Rev Soc Bras Med Trop 2006; 39:512-514.
-
11Portinho JA, Zimmermann LM, Bruck MR. Beneficial effects of açaí. Int J Nutrol 2012; 5:15-20.
-
12Cardarelli-Leite P, Branquinho MR, Ferreira RTB. Detection of GMO in food products in Brasil: the INCQS experience. Food control 2005; 16:859-866.
-
13Join Research Centre (JRC). Event-specific method for the quantification of maize line NK603 using real-time PCR protocol. (Internet). 2005 (Cited 2015 October 1). Available at: Available at: http://www.gmo-crl.jrc.ec.europa.eu/statusofdoss.htm.
» http://www.gmo-crl.jrc.ec.europa.eu/statusofdoss.htm. -
14Chiurillo MA, Crisante G, Rojas A, Peralta A, Dias M, Guevara, et al. Detection of Trypanosoma cruzi and Trypanosoma rangeli Infection by duplex PCR assay based on telomeric sequences. Clin Diagn Lab Immunol 2003; 10:775-779.
-
15Corbisier P, Broothaerts W, Gioria S, Schimmel H, Burns M, Baoutina A, et al. Toward metrological traceability for DNA fragment ratios in GM quantification. 1. Effect of DNA extraction methods on the quantitative determination of Bt 176 corn by Real-Time PCR. J Agric Food Chem 2007; 55:3249-3257.
-
16Devi KD, Punyarani K, Singh NS, Devi HS. An efficient protocol for total DNA extraction from the members of order Zingiberales-suitable for diverse PCR based downstream applications. Springer Plus 2013; 2:669.
-
17Minas K, McEwan NR, Newbold CJ, Scott KP. Optimization of a high-throughput CTAB-based protocol for the extraction of qPCR-grade DNA from rumen fluid, plant and bacterial pure cultures. Fems Microbiol Lett 2011; 325:162-169.
-
18Mondal TK, Ganie SA. Identification and characterization of salt responsive miRNA-SSRmarkers in rice (Oryza sativa). Gene 2014; 535:204-209.
-
19Organização Panamericana de Saúde, Organização Mundial de Saúde, PANAFTOSA-VP. Guia para vigilância, prevenção, controle e manejo clínico da doença de Chagas aguda transmitida por alimentos. Rio de Janeiro: PANAFTOSA-VP/OPAS/OMS; 2009.
-
20Coura JR, Viñas PA. Chagas disease: a new worldwide challenge. Nature 2010; 465:56-57.
-
21World Health Organization. Crompton DWT, Peters P, editors. Working to Overcome the Global Impact of Neglected Tropical Diseases: First WHO Report on Neglected Tropical Diseases. Nonserial Publication Series WHO report on neglected tropical diseases. volume 1; 2010. 172p.
-
22;Barbosa-Labello R. Transmissão oral do Trypanosoma cruzi pela polpa de açaí em camundongos. 2010 [dissertation]. [São Paulo]: Universidade Estadual de Campinas; 2010. 120 p.
Publication Dates
-
Publication in this collection
Mar-Apr 2016
History
-
Received
18 Dec 2015 -
Accepted
05 Apr 2016