Abstract:
INTRODUCTION
Recent studies have shown a high incidence and prevalence of latent tuberculosis infection (LTBI) in indigenous populations around the World. We aimed to estimate the prevalence and annual risk of infection (ARI) as well as to identify factors associated with LTBI in an indigenous population from the Brazilian Amazon.
METHODS
We conducted a cross-sectional study in 2011. We performed tuberculin skin tests (TSTs), smears and cultures of sputum samples, and chest radiographs for individuals who reported cough for two or more weeks. Associations between LTBI (TST ≥5mm) and socio-demographic, clinical, and epidemiological characteristics were investigated using Poisson regression with robust variance. Prevalence ratio (PR) was used as the measure of association.
RESULTS
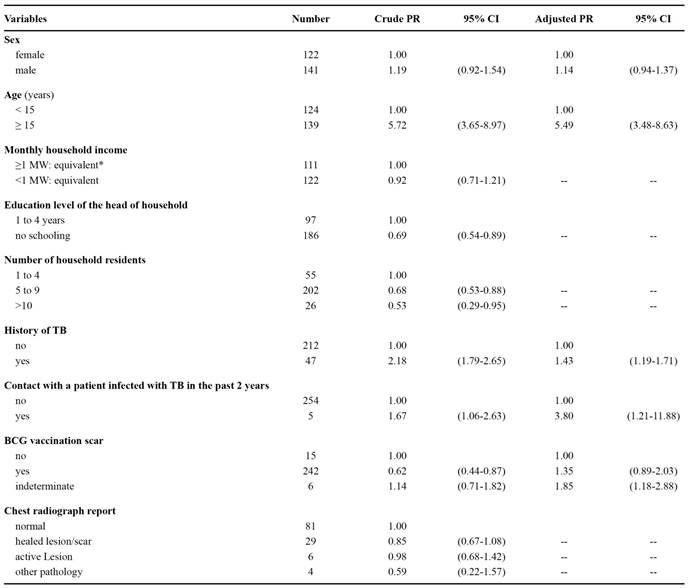
We examined 263 individuals. The prevalence of LTBI was 40.3%, and the ARI was 2.4%. Age ≥15 years [PR=5.5; 95% confidence interval (CI): 3.5-8.6], contact with tuberculosis (TB) patients (PR=3.8; 95% CI: 1.2-11.9), previous TB history (PR=1.4; 95% CI: 1.2-1.7), and presence of Bacillus Calmette-Guérin (BCG) scar (PR=1.9, 95% CI: 1.2-2.9) were associated with LTBI.
CONCLUSIONS
Although some adults may have been infected years prior, the high prevalence of infection and its strong association with age ≥15 years, history of TB, and recent contact with TB patients suggest that the TB transmission risk is high in the study area.
Keywords:
Epidemiology; Health services indigenous; Mycobacterium tuberculosis; Public health surveillance; South American Indians
INTRODUCTION
In recent decades, the National Tuberculosis Control Program of Brazil has employed various interventions to control tuberculosis (TB), including detection of new cases, free distribution of drugs for early treatment of patients, chemoprophylaxis for infected persons, and widespread Bacille Calmette-Guérin (BCG) vaccine distribution. However, the disease remains an important public health problem in Brazil, particularly among populations considered vulnerable, such as homeless persons, prisoners, health professionals, and indigenous populations 11. Ministério da Saúde. Secretaria de Vigilância em Saúde. Departamento de Vigilância Epidemiológica. Manual de recomendações para o controle da tuberculose no Brasil. Brasília: 2011. 288p.) (22. Nava-Aguilera E, Andersson N, Harris E, Mitchell S, Hamel C, Shea B, et al. Risk factors associated with recent transmission of tuberculosis: systematic review and meta-analysis. Int J Tuberc Lung Dis 2009; 13:17-26. .
Recent studies have shown that the incidence of TB among indigenous populations in various parts of the world is higher than that reported for non- indigenous populations in the same countries 33. Tollefson D, Bloss E, Fanning A, Redd JT, Barker K, McCray E. Burden of tuberculosis in indigenous peoples globally: a systematic review. Int J Tuber Lung Dis 2013; 17:1139-1150.) (44. Robertus LM, Konstantinos A, Hayman NE, Paterson DL. Tuberculosis in the Australian Indigenous population: history, current situation and future challenges. Aust N Z J Public Health 2011; 35:6-9.) (55. Culqui DR, Trujillo OV, Cueva N, Aylas R, Salaverry O, Bonilla C. Tuberculosis in the Indigenous population of Peru 2008. Rev Peru Med Exp Salud Publica 2010; 27:8-15.) (66. Canadian Tuberculosis Committee Housing conditions that serve as risk factors for tuberculosis infection and disease. An Advisory Committee Statement (ACS). Can Commun Dis Rep 2007; 33:1-33. . In Brazil, the situation is even more critical, due to higher incidence rates, elevated concentrations of cases along international borders (with a significant proportion of cases involving children and adolescents), drug resistance, prevalence of latent infection with Mycobacterium tuberculosis (MTB) exceeding 40%, and reported patterns of recent and ongoing transmission in the investigated villages 77. Basta PC, Coimbra Jr CEA, Escobar AL, Santos RV, Alves LCC, Fonseca LS. Survey for tuberculosis in an indigenous population of Amazonia: the Suruí of Rondônia, Brazil. Trans R Soc Trop Med Hyg 2006; 100:579-585.) (88. Basta PC, Coimbra Jr CEA, Camacho LAB, Santos RV. Risk of tuberculous infection in an indigenous population from Amazonia, Brazil. Int J Tuber Lung Dis 2006; 10: 1354-1359.) (99. Basta PC, Coimbra Jr CEA, Welch JR, Correia Alves LC, Santos RV, Bastos Camacho LA. Tuberculosis among the Xavante Indians of the Brazilian Amazon: an epidemiological and ethnographic assessment. Ann Hum Biol 2010; 37:643-657.) (1010. Barbosa T, Benigno A, Farre L, Prat JGI; Barral-Netto M, Arruda S. Tuberculosis among Yanomami communities from Alto Rio Negro, State of Amazonas, Brazil, 1990 to 2002. Enf Emerg 2008; 10:138-143.) (1111. Rios DPG, Malacarne J, Alves LCC, Sant'Anna CC, Camacho LAB, Basta PC. Tuberculose em indígenas da Amazônia brasileira: Estudo epidemiológico na região do Alto Rio Negro. Rev Panam Salud Publica 2013; 33:22-29.) (1212. Belo EN, Orellana JDY, Levino A, Basta PC. Tuberculose nos municípios amazonenses da fronteira Brasil-Colômbia-Peru-Venezuela: situação epidemiológica e fatores associados ao abandono. Rev Panam Salud Publica 2013; 34:321-329.) (1313. Cunha EA, Ferrazoli L, Riley LW, Basta PC, Honer MR, Maia R et al. Incidence and transmission patterns of tuberculosis among indigenous populations in Brazil. Mem Inst Oswaldo Cruz 2014; 109:108-113. .
Studies conducted over the last decade in the State of Rondônia also illustrate the gravity of the situation in our study area 77. Basta PC, Coimbra Jr CEA, Escobar AL, Santos RV, Alves LCC, Fonseca LS. Survey for tuberculosis in an indigenous population of Amazonia: the Suruí of Rondônia, Brazil. Trans R Soc Trop Med Hyg 2006; 100:579-585.) (88. Basta PC, Coimbra Jr CEA, Camacho LAB, Santos RV. Risk of tuberculous infection in an indigenous population from Amazonia, Brazil. Int J Tuber Lung Dis 2006; 10: 1354-1359.) (1414. Escobar AL, Coimbra Jr CEA, Camacho LA, Portela MC. Tuberculose em populações indígenas de Rondônia, Amazônia, Brasil. Cad Saude Publica 2001; 17:285-298.) (1515. Escobar AL, Coimbra Jr CEA, Camacho LA, Santos V. Tuberculin reactivity and tuberculosis epidemiology in the Paakanova (Wari') Indians of Rondônia, Southwestern Brazilian Amazon. Int J Tuberc Lung Dis 2004; 8:45-51.) (1616. Basta PC, Oelemann MAC, Oelemann WMR, Fonseca LS; Coimbra Jr CEA. Detection of Mycobacterium tuberculosis in sputum from Suruí Indian subjects, Brazilian Amazon. Mem Inst Oswaldo Cruz 2006; 101:581-584. . The occurrence of TB in indigenous territories (IT) was recently documented in a study of the geographic distribution of TB cases reported in the Information System for Notifiable Diseases [ Sistema de Informação de Agravos de Notificação (SINAN)] 1717. Melo TEMP, Resendes APC, Souza-Santos R, Basta PC. Distribuição espacial e temporal da tuberculose em indígenas e não indígenas de Rondônia, Amazônia Ocidental, Brasil. Cad Saude Publica 2012; 28:267-280. . The authors of that study showed that in many of Wari's indigenous territories, the incidence rates were surprisingly high, exceeding 240/100,000 inhabitants from 1997 to 2006.
According to Basta and Camacho 1818. Basta PC, Camacho LAB. Teste tuberculínico na estimativa da prevalência de infecção por Mycobacterium tuberculosis em populações indígenas do continente americano: uma revisão da literatura. Cad Saude Publica 2006; 22:245-254. , incidence rate is not the best indicator of the dynamic transmission trends of TB, particularly in small populations like indigenous populations. Young and Mirdad 1919. Young TK, Mirdad S. Determinants of tuberculin sensitivity in a child population covered by mass BCG vaccination. Tuber Lung Dis 1992; 73:94-100. found that using incidence rate to assess the levels of transmission of MTB might be limited by the fact that most individuals who are infected with MTB do not develop the disease, and those who become ill might have been infected for months or years. Therefore, the incidence rate depends on the case report system, which in many places might not reflect the true disease burden due to operational difficulties. Additionally, confirmation of TB diagnosis requires isolation of the bacillus, and this procedure is less accessible in some regions of Brazil, especially in areas with indigenous people (9 ) (1111. Rios DPG, Malacarne J, Alves LCC, Sant'Anna CC, Camacho LAB, Basta PC. Tuberculose em indígenas da Amazônia brasileira: Estudo epidemiológico na região do Alto Rio Negro. Rev Panam Salud Publica 2013; 33:22-29.) (1212. Belo EN, Orellana JDY, Levino A, Basta PC. Tuberculose nos municípios amazonenses da fronteira Brasil-Colômbia-Peru-Venezuela: situação epidemiológica e fatores associados ao abandono. Rev Panam Salud Publica 2013; 34:321-329.) (1414. Escobar AL, Coimbra Jr CEA, Camacho LA, Portela MC. Tuberculose em populações indígenas de Rondônia, Amazônia, Brasil. Cad Saude Publica 2001; 17:285-298.) (2020. Orellana JDY, Gonçalves MJF, Basta PC. Sociodemographic features and operating indicators of tuberculosis control between indigenous and non-indigenous people of Rondônia, Western Amazon, Brazil. Rev Bras Epidemiol 2012; 15:714-724. . Considering these limitations, some authors 1919. Young TK, Mirdad S. Determinants of tuberculin sensitivity in a child population covered by mass BCG vaccination. Tuber Lung Dis 1992; 73:94-100.) (2121. Arnadottir T, Rieder HL, Trébucq A, Waaler HT. Guidelines for conducting tuberculin skin test surveys in high prevalence countries. Tuber Lung Dis 1996; 77 (suppl 1):1-19.) (2222. American Thoracic Society. Diagnostic Standards and Classification of Tuberculosis in Adults and Children. Am J Respir Crit Care Med 2000; 161:1376-1395. have argued that the most appropriate indicator of TB transmission in a particular location is the prevalence of MTB-specific infection.
This study aimed to screen for active TB, to estimate the prevalence and annual risk of infection (ARI) of MTB, and to identify factors associated with latent tuberculosis infection (LTBI) among indigenous Wari' (Pakaanóva) populations from Rondônia (Brazilian Amazon region) in order to provide additional relevant data necessary to reassess control activities targeting indigenous peoples.
METHODS
Study population and area. The State of Rondônia is located in Northern Brazil and is part of the Amazon river basin. According to the latest national census (2010), 1,562,409 inhabitants live in Rondônia, of whom 13,076 (0.8%) declared themselves as indigenous, representing approximately 30 different ethnic groups that live in 20 indigenous territories ( Figure 1 ).
Map of the State of Rondônia, including municipalities and Indian Reservations (indigenous territories).
In Brazil, there are approximately 600 indigenous territories, which are demarcated Indian reserves set up by the federal government (http://www.funai.gov.br/index.php/indios-no-brasil/terras-indigenas, accessed 16 may 2016). The majority of these territories are located in the Amazon and in remote areas, distant from urban centers. In general, there are natural resources (food and clean water) in these territories, but the population faces some difficulties when attempting to reach municipalities to access the health facilities.
The Wari' (Pakaanova) are a Txapakura-speaking indigenous society located in the municipalities of Guajará-Mirim and Nova Mamoré within Rondônia in the southwest Brazilian Amazonia (approximately 10-11° S, 65-66° W). At the time of fieldwork, there were 2,721 Wari' people who lived in a total of seven indigenous territories ( Igarapé Lage, Igarapé Ribeirão, Pacaás Novas, Rio Guaporé, Sagarana, Rio Negro, and Rio Negro Ocaia ) at the margins of the Mamoré and Pakaanova rivers and in lands covered by interfluvial tropical rainforest on the border with Bolivia. Different sub-groups of Wari' who maintain the same cultural habits and have strong family ties live in each indigenous territory (https://pib.socioambiental.org/pt/povo/wari, accessed 16 May 2016).
Traditionally, subsistence was based on hunting and agriculture, especially of maize. Nowadays, timber and Brazil nuts have become central to the Wari' economy. As with other indigenous groups who live in Rondônia, the Wari' society was strongly affected by massive migration of settlers to Amazonia in the 1960-1970s 77. Basta PC, Coimbra Jr CEA, Escobar AL, Santos RV, Alves LCC, Fonseca LS. Survey for tuberculosis in an indigenous population of Amazonia: the Suruí of Rondônia, Brazil. Trans R Soc Trop Med Hyg 2006; 100:579-585.) (88. Basta PC, Coimbra Jr CEA, Camacho LAB, Santos RV. Risk of tuberculous infection in an indigenous population from Amazonia, Brazil. Int J Tuber Lung Dis 2006; 10: 1354-1359. . Permanent contact between the Wari' and Brazilian national society took place in 1956 and was followed by several epidemics of infectious diseases, including malaria, flu, measles, and TB. Ever since, TB has become an important cause of morbidity and mortality in this population.
We selected one of the Wari´ territories (Igarapé Ribeirão), where TB incidence is six times higher than that reported in the non-indigenous populations and higher than that in other indigenous territories in Rondônia during the last decade 1717. Melo TEMP, Resendes APC, Souza-Santos R, Basta PC. Distribuição espacial e temporal da tuberculose em indígenas e não indígenas de Rondônia, Amazônia Ocidental, Brasil. Cad Saude Publica 2012; 28:267-280. .
Study design. A cross-sectional epidemiological study was conducted from January 31 st to February 14 th , 2011, in Igarapé Ribeirão (S 10º 17' 50.1''; W 65° 08' 18.4''), located in the municipality of Nova Mamoré.
In this indigenous territory, our team visited all houses of the heads of household who agreed to be interviewed. They were asked about cough lasting for two or more weeks, weight loss, night sweats, weakness, and other symptoms related to active TB occurring among household members during the time of fieldwork.
Active tuberculosis screening. Physical examinations were performed by three of the authors (JL, DPGR, and PCB), who were accompanied by an indigenous health agent who helped explain the research plan and acted as interpreter when needed, diminishing cultural and linguistic barriers between the researchers and the Wari' (Pakaanova).
The criteria for suspicion of TB were cough lasting for two or more weeks, fever and/or weight loss, and/or weakness 11. Ministério da Saúde. Secretaria de Vigilância em Saúde. Departamento de Vigilância Epidemiológica. Manual de recomendações para o controle da tuberculose no Brasil. Brasília: 2011. 288p.) (2222. American Thoracic Society. Diagnostic Standards and Classification of Tuberculosis in Adults and Children. Am J Respir Crit Care Med 2000; 161:1376-1395. . Adults and children under 10 years who were suspected of being infected with underwent sputum examinations. Whenever possible, two sputum samples were collected from the subjects who reported cough for two or more weeks: one at the time of the interview and another the next morning, before breakfast ( Figure 2 ). In accordance with standard techniques, the samples were fixed and stained with Ziehl Neelsen stain for sputum smear microscopy and inoculated into Ogawa-Kudoh medium. These procedures were performed at the study site, after which the samples were sent to the National Institute of Amazon Research [ Instituto Nacional de Pesquisas da Amazônia (INPA)] laboratory and incubated at 98.6°F for 60 days.
Flowchart of the screening stages for participants in Igarapé Ribeirão, Rondônia, Brazilian Amazon in 2011. TB: tuberculosis; MOTT: Nontuberculous Mycobacteria .
Chest radiographs were performed using the posterior-anterior view with portable X-ray equipment adapted to the difficult conditions of the village. Individuals were selected based on the presence of any symptoms suggestive of active TB, previous history of TB, or contact with sick infectious individuals. Abnormalities identified on the posterior-anterior images were further investigated using lateral views. Standardized reports were issued by an experienced pulmonologist in accordance with the recommendations proposed by Den Boon et al 2323. Den Boon S, Bateman ED, Enarson DA, Borgdorff MW, Verver S, Lombard CJ, et al. Development and evaluation of a new chest radiograph reading and recording system for epidemiological surveys of tuberculosis and lung disease. Int J Tuberc Lung Dis 2005; 9:1088-1096. .
Latent tuberculosis screening. To estimate the prevalence of latent TB infection (LTBI) and the annual risk of TB infection, we administered tuberculin skin tests (TSTs) to all residents regardless of immunization history. We excluded pregnant women, children younger than 2 years, and those with medical conditions that could impair cellular immune response, such as cancer, acquired immunodeficiency syndrome (AIDS), and renal failure ( Figure 2 ). These medical conditions were determined during the questionnaire interview. TSTs involved intradermal injection of 0.1mL [2 tuberculin units (TU)] of tuberculin Purified Protein Derivative (PPD) RT23 (Statens Serum Institute, Copenhagen, Denmark) in the middle third of the left forearm, according to the guidelines proposed by Arnadottir et al. 21 The evaluation of the TST reactions was performed within 72 hours after inoculation by three different readers who were trained to use a standardized method.
According to current guidelines of the Brazilian Ministry of Health for HIV-infected, immunodeficient, and other vulnerable groups, individuals who had TST reactions greater than or equal to 5mm were considered to have LTBI 11. Ministério da Saúde. Secretaria de Vigilância em Saúde. Departamento de Vigilância Epidemiológica. Manual de recomendações para o controle da tuberculose no Brasil. Brasília: 2011. 288p. . In order to assess the reliability and agreement between TST readers, we computed the intra-class correlation coefficient, which was greater than 0.90 for all three readers.
Individuals younger than 15 years who had TST reactions ≥ 5mm, without any signs and symptoms of TB or previous treatment for TB, were referred for treatment of LTBI (chemoprophylaxis) 11. Ministério da Saúde. Secretaria de Vigilância em Saúde. Departamento de Vigilância Epidemiológica. Manual de recomendações para o controle da tuberculose no Brasil. Brasília: 2011. 288p. .
Variables and study instruments. Data on age, sex, education, occupation of the household members, family income (in multiples of the national minimum wage), and number of people per house were collected during home visits. Household contact with patients with active TB in the previous two years was also evaluated. Reports of previous history of TB treatment and BCG vaccination were confirmed by consulting the Information System for Notifiable Diseases database and vaccination cards, respectively.
Data collection instruments for this research were developed from the research group's experience 1111. Rios DPG, Malacarne J, Alves LCC, Sant'Anna CC, Camacho LAB, Basta PC. Tuberculose em indígenas da Amazônia brasileira: Estudo epidemiológico na região do Alto Rio Negro. Rev Panam Salud Publica 2013; 33:22-29. and after consultation and adaptation from (i) The First National Health and Nutrition Survey of Indigenous peoples in Brazil 2424. Coimbra Jr CEA, Santos RV, Welch JR, Cardoso AM, Souza MC, Garnelo L, et al. The First National Survey of Indigenous People's Health and Nutrition in Brazil: rationale, methodology, and overview of results. BMC Public Health 2013; 13:52. and (ii) The Questionnaire for Evaluation of Primary Health Care Performance in TB Control in Brazil 2525. Scatena-Villa TC, Ruffino-Netto A. Questionário para avaliação de desempenho de serviços de atenção básica no controle da TB no Brasil. J Bras Pneumol 2009; 35:610-612. .
Statistical analysis. The following formula was used to estimate the Annual Risk Infection (ARI): ARI = 1 - (1 - p ) 1/b , where p corresponds to the prevalence of infection (TST reactions ≥5mm) obtained from the tuberculin survey and b corresponds to the mean age of individuals who underwent TST 2626. Cauthen GM, Pio A, ten Dam HG. Annual risk of tuberculous infection. 1988 Bull World Health Organ 2002; 80:503-511. .
Data analyses were performed to associate TST results with the following independent variables: sex, age, income, education level of the head of household, number of persons per family, history of TB, contact with TB patients, presence of a BCG scar, and chest radiograph findings.
Prevalence ratio (PR) adjusted for covariates in a Poisson regression model with robust variance was used as the measure of association. After univariate analysis, variables that were statistically significant (p<0.20) were included in the multivariate model using the stepwise forward method. Variables that met the statistical significance threshold of 0.05 or were considered scientifically relevant were retained in the final model.
The analyses were performed with the statistical software R, version 2.13.1 and Statistical Package for Social Science, version 9.0 (IBM, Chicago, USA).
Ethical considerations. This study was performed in accordance with the Declaration of Helsinki. According to the Resolution of the Brazilian National Health Council, which regulates studies involving indigenous populations, the written informed consent was read and explained to volunteers and leaders of the community before beginning fieldwork. Due to the high illiteracy rate, oral consent was often obtained with the health agents as witnesses.
All data were collected and analyzed anonymously, and written informed consents were stored in boxes in the office of the research coordinator. The study protocol was approved by the National Commission for Ethics in Research of the National Health Council and the Research Ethics Committee of the National School of Public Health (#327/2008 and #176/07).
RESULTS
All indigenous individuals present in the village of Igarapé Ribeirão at the time of the team visit were evaluated, totaling 263 subjects in 49 households (approximately 85% of the total resident population). Twenty-six losses due to absence and 3 refusals were reported. In addition, 21 persons were excluded from latent TB screening, including 7 pregnant women, 13 children under two years of age, and one person with chronic renal failure who was undergoing hemodialysis ( Figure 2 ). There were no cases of diabetes, cancer, or human immunodeficiency virus/acquired immunodeficiency syndrome (HIV/AIDS) in the study population.
The mean age was 22.2 years (standard deviation=18.5 years). There was a predominance of men (53.7%) and young people (58.5% were younger than 19 years) in this population. Residents who were absent during fieldwork had similar age and sex distributions (mean age of 19.8 years and 54% men). In most households, the main provider had no schooling, performed informal work, and earned less than one minimum wage equivalent monthly ( Table 1 ). Houses were small and poorly constructed, often with many residents (data not shown). The use of firewood for cooking was a common practice among families.
During the home visits, 49 (18.6%) individuals had a history of TB treatment, and five (1.9%) subjects reported contact with patients infected with TB in the last two years. One 54-year-old woman was in treatment for TB, resulting in a prevalence of active disease of 380.2/100,000 inhabitants. In addition, 24 (9.1%) persons reported respiratory symptoms, and 34 sputum samples were collected for laboratory analysis. All cultures were negative for MTB. Atypical species of the genus Mycobacterium [(Nontuberculous Mycobacteria (MOTT)] were isolated from four subjects, but clinical investigation did not detect active tuberculosis or non-tuberculous mycobacterial disease ( Figure 2 ). BCG scars were apparent in 95.0% of the subjects.
In total, 120 chest radiographs were performed. Of these, 39 demonstrated some abnormality. Twenty-nine (24.1%) were consistent with pulmonary sequelae due to TB, six (5%) were suggestive of active TB, and four showed other pulmonary pathologies. The main radiological findings were calcified nodules (n=17, 43.6%), fibrotic beams (n=11, 28.2%), and infiltration/consolidation (n=11, 28.2%). Notwithstanding, the association with TST response was not statistically significant ( Table 2 ).
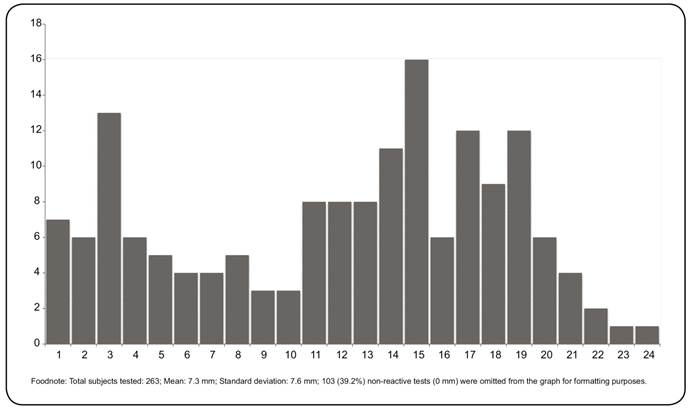
A total of 263 TSTs were performed, with reactions ranging from 0 to 29mm (median=4mm, standard deviation=7.6mm). Reactions greater than 10mm were more prevalent in adults over 20 years old. Of the 103 individuals without TST reactions (0mm), 71 (68.9%) were children under 10 years of age ( Figure 3 ). The prevalence of MTB infection was 40.3%, and the annual risk of infection was 2.4% in the study population.
Distribution of tuberculin test reaction size (mm) in Igarapé Ribeirão, Rondônia, Brazilian Amazon in 2011. There were 263 total subjects tested. The mean (standard deviation) is 7.3 (7.6)mm. One hundred and three (39.2%) non-reactive tests (0mm) were omitted from the graph for formatting purposes.
Using Poisson regression, univariate analysis showed that the following variables were associated with TST responses ≥ 5mm: age, education level of head of household, number of persons per household, previous history of TB, and contact with a patient infected with TB in the previous 2 years. However, in the final model, only age, contact with a patient infected with, previous history of TB, and BCG scar remained associated with TST responses ≥ 5mm ( Table 2 ).
No new cases of active TB were identified in the study population. However, eight children under 15 years old were referred for chemoprophylaxis. In addition, some other diseases were detected, including two cases of chronic obstructive pulmonary disease, one possible case of heart disease, and a lung tumor in one person. Individuals with abnormal radiographic findings, but no bacteriological confirmation, were referred to the local health clinic for follow-up and treatment.
DISCUSSION
As in other studies involving indigenous peoples 2424. Coimbra Jr CEA, Santos RV, Welch JR, Cardoso AM, Souza MC, Garnelo L, et al. The First National Survey of Indigenous People's Health and Nutrition in Brazil: rationale, methodology, and overview of results. BMC Public Health 2013; 13:52.) (2727. Villalba JA, Liu Y, Alvarez MK, Calderon L, Canache M, Cardenas G, et al. Low child survival index in a multi-dimensionally poor Amerindian population in Venezuela. PLoS One 2013; 8:e85638. , the study population was predominantly young and poor with limited access to formal education, and the study participants lived in overcrowded and smoky houses, which are considered favorable for TB transmission 11. Ministério da Saúde. Secretaria de Vigilância em Saúde. Departamento de Vigilância Epidemiológica. Manual de recomendações para o controle da tuberculose no Brasil. Brasília: 2011. 288p.) (66. Canadian Tuberculosis Committee Housing conditions that serve as risk factors for tuberculosis infection and disease. An Advisory Committee Statement (ACS). Can Commun Dis Rep 2007; 33:1-33. .
Similar high levels of ARI and prevalence of MTB infection had been reported among indigenous groups in the Brazilian Amazon region 1717. Melo TEMP, Resendes APC, Souza-Santos R, Basta PC. Distribuição espacial e temporal da tuberculose em indígenas e não indígenas de Rondônia, Amazônia Ocidental, Brasil. Cad Saude Publica 2012; 28:267-280. , in the Midwest region among the Xavante 99. Basta PC, Coimbra Jr CEA, Welch JR, Correia Alves LC, Santos RV, Bastos Camacho LA. Tuberculosis among the Xavante Indians of the Brazilian Amazon: an epidemiological and ethnographic assessment. Ann Hum Biol 2010; 37:643-657. , in indigenous Wari' (Pakaanóva) populations from Lage1515. Escobar AL, Coimbra Jr CEA, Camacho LA, Santos V. Tuberculin reactivity and tuberculosis epidemiology in the Paakanova (Wari') Indians of Rondônia, Southwestern Brazilian Amazon. Int J Tuberc Lung Dis 2004; 8:45-51. , and in the Suruí people of Sete de Setembro88. Basta PC, Coimbra Jr CEA, Camacho LAB, Santos RV. Risk of tuberculous infection in an indigenous population from Amazonia, Brazil. Int J Tuber Lung Dis 2006; 10: 1354-1359. in Rondônia.
The ARI estimate in the present study was comparable to that seen for indigenous/native populations from Canada in the 1970´s, where the ARI was approximately 5% 2828. Gryzbowski S, Styblo K, Dorken E. Tuberculosis in Eskimos. Tubercle 1976; 57 (suppl 4): S1-S58. . The ARI, which does not depend on the reliability of case reporting (!9) , indicated high levels of TB transmission in Rondônia's indigenous territories, consistent with the high frequency of reported previous TB treatment.
The associations between rates of TB infection and age, contact with a patient infected with TB in the last 2 years, and history of TB, were all statistically significant and of substantial magnitude, which is consistent with current knowledge about the transmission of TB and similar to previous studies 99. Basta PC, Coimbra Jr CEA, Welch JR, Correia Alves LC, Santos RV, Bastos Camacho LA. Tuberculosis among the Xavante Indians of the Brazilian Amazon: an epidemiological and ethnographic assessment. Ann Hum Biol 2010; 37:643-657.) (1111. Rios DPG, Malacarne J, Alves LCC, Sant'Anna CC, Camacho LAB, Basta PC. Tuberculose em indígenas da Amazônia brasileira: Estudo epidemiológico na região do Alto Rio Negro. Rev Panam Salud Publica 2013; 33:22-29.) (1515. Escobar AL, Coimbra Jr CEA, Camacho LA, Santos V. Tuberculin reactivity and tuberculosis epidemiology in the Paakanova (Wari') Indians of Rondônia, Southwestern Brazilian Amazon. Int J Tuberc Lung Dis 2004; 8:45-51.) (1616. Basta PC, Oelemann MAC, Oelemann WMR, Fonseca LS; Coimbra Jr CEA. Detection of Mycobacterium tuberculosis in sputum from Suruí Indian subjects, Brazilian Amazon. Mem Inst Oswaldo Cruz 2006; 101:581-584. . In contrast to those studies, which used odds ratios, the present study estimated prevalence ratios, which is considered a more accurate measure of association in settings with a prevalence of infection as high as often seen in indigenous populations 2929. Barros AJ, Hirakata VN. Alternatives for logistic regression in cross-sectional studies: an empirical comparison of models that directly estimate the prevalence ratio. BMC Med Res Methodol 2003; 3:21. .
On the other hand, in contrast to a case-control study that indicated that socioeconomic variables were associated with active TB in an indigenous population from the Midwest region of Brazil 3030. Sacchi FP, Croda MG, Estevan AO, Ko AI, Croda J. Sugar cane manufacturing is associated with tuberculosis in an indigenous population in Brazil. Trans R Soc Trop Med Hyg . 2013; 107:152-157. , in our study, socioeconomic variables, such as income, educational level, and number of persons per household, did not show a significant association to latent tuberculosis infection.
Consequently, we assumed that the factors associated with latent tuberculosis infection are different from those associated with active TB, not only because the study population seems to be homogeneous with regard to the variables related to socioeconomic level, but also because Mycobacterium tuberculosis seems to be spreading over indigenous territories in Brazilian Amazon, regardless of socioeconomic status.
A high prevalence of intestinal parasitosis is another known condition associated with LTBI 3131. Board AR, Suzuki S. The interrelation between intestinal parasites and latent TB infections among newly resettled refugees in Texas. Int Health 2016; 8:67-72. . Some authors have reported high levels of intestinal parasitosis in indigenous groups in Brazil 3232. Assis EM, Oliveira RC, Moreira LE, Pena JL, Rodrigues LC, Machado-Coelho GLL. Prevalência de parasitos intestinais na comunidade indígena Maxakali, Minas Gerais, Brasil, 2009. Cad Saude Publica 2013; 29:681-690. , including in our study area 3333. Palhano-Silva CS, Araújo AJG, Lourenço AEP, Bastos OMP, Santos RV, Coimbra Jr CEA. Intestinal parasitic infection in the Suruí Indians, Brazilian Amazon. Interciencia 2009; 34:259-264. . Additionally, considering that 95% of individuals had a BCG scar, there was a substantial proportion (59.7%) of the population with TST reactions <5mm, of whom 39.2% had no reaction at all (TST equal 0mm). The majority of non-reactors were children under 10 years and individuals who had been vaccinated with BCG. Zembrzuski et al. 3434. Zembrzuski VM, Basta PC, Callegari-Jacques SM, Santos RV, Coimbra CEA, Salzano FM, et al. Cytokine genes are associated with tuberculin skin test response in a native Brazilian population. Tuberculosis (Edinb) 2010; 90: 44-49. observed a 74.4% rate of reactions <5mm despite BCG vaccination coverage (>90%) among the Xavante of Mato Grosso in the Midwest region of Brazil. Similarly, Escobar et al. 1515. Escobar AL, Coimbra Jr CEA, Camacho LA, Santos V. Tuberculin reactivity and tuberculosis epidemiology in the Paakanova (Wari') Indians of Rondônia, Southwestern Brazilian Amazon. Int J Tuberc Lung Dis 2004; 8:45-51. observed reactions <5mm in 58.4% of the Wari' (Pakaanóva) people, whereas in the Suruí people, Basta et al. 88. Basta PC, Coimbra Jr CEA, Camacho LAB, Santos RV. Risk of tuberculous infection in an indigenous population from Amazonia, Brazil. Int J Tuber Lung Dis 2006; 10: 1354-1359. found 27.1% with reactions <5mm, including individuals with recent BCG revaccination. Some TST reactions between 5 and 10mm might have been actually related to infection by atypical Mycobacterium and BCG rather than M. tuberculosis . However, a 10-mm threshold could misclassify true MTB infections, as indicated in the current Brazilian guidelines 11. Ministério da Saúde. Secretaria de Vigilância em Saúde. Departamento de Vigilância Epidemiológica. Manual de recomendações para o controle da tuberculose no Brasil. Brasília: 2011. 288p. .
The reasons for non-reactivity among indigenous subjects with BCG scars remain uncertain. Longhi et al. 3535. Longhi RMP, Zembrzuski VM, Basta PC, Croda J. Genetic polymorphism and immune response to tuberculosis in indigenous populations: a brief review. Braz J Infect Dis 2013; 17:363-368. suggested that non-reactivity to TST might be associated with deficiencies in cellular immune response and genetic susceptibility. The authors argue that there is a predominance of T-helper 2 (Th2) cellular immune responses in indigenous people. This might favor progression to active TB with the manifestation of more severe forms of the disease, exuberant symptoms, and high bacterial loads. Additionally, many polymorphisms have been described in genes associated with the expression of cytokines involved in immune responses. In the case of native populations, these polymorphisms induce a predominantly Th2 immune response, affecting the course of the infection. The authors emphasized the need for studies that analyze the cellular activity of macrophages and lymphocytes in the immune response to MTB and extend current knowledge of polymorphisms related to illness susceptibility, in order to elucidate the involvement of immunological and genetic factors in this process.
Another possible explanation for the significant proportion of non-reactors to TST is that PPD RT23, which was used in the study, could have been improperly refrigerated, causing denaturation and consequent underestimation of the presence of TST reactions >5mm. Given the storage and handling procedures for tuberculin implemented in anticipation of the high risk in field conditions, we consider it highly unlikely that the sensitivity of the PPD RT23 was affected.
This study has some limitations. Misclassification of MTB infection is inherent to TST, which relies on measurements that are difficult to perform and are the basis for a threshold that cannot account for blurred limits of intermediate values (between 5mm and 10mm). As misclassification in this setting is presumably non-differential and the proportion of intermediate reactions was small, the measures of association may have been weakened by a small margin. The high prevalence of respiratory symptoms (9.1%) compared to the expected level of 1% 11. Ministério da Saúde. Secretaria de Vigilância em Saúde. Departamento de Vigilância Epidemiológica. Manual de recomendações para o controle da tuberculose no Brasil. Brasília: 2011. 288p. is another limitation. Additionally, reports obtained in interviews conducted with the aid of interpreters are more susceptible to bias. Communication barriers may have limited the detection of medical conditions, although health workers providing regular health care at the village were able to report severe chronic diseases. However, history of TB treatment was confirmed with data available from the National Information System for Notifiable Diseases.
In conclusions, our results illustrate the TB epidemic among indigenous people in the state of Rondônia. Inequalities, poverty, and socioeconomic disadvantages create a fertile ground for the spread of the disease, while promoting the transmission of MTB in the region. Considering results from recent studies conducted in Rondônia 77. Basta PC, Coimbra Jr CEA, Escobar AL, Santos RV, Alves LCC, Fonseca LS. Survey for tuberculosis in an indigenous population of Amazonia: the Suruí of Rondônia, Brazil. Trans R Soc Trop Med Hyg 2006; 100:579-585.) (88. Basta PC, Coimbra Jr CEA, Camacho LAB, Santos RV. Risk of tuberculous infection in an indigenous population from Amazonia, Brazil. Int J Tuber Lung Dis 2006; 10: 1354-1359.) (1414. Escobar AL, Coimbra Jr CEA, Camacho LA, Portela MC. Tuberculose em populações indígenas de Rondônia, Amazônia, Brasil. Cad Saude Publica 2001; 17:285-298.) (1515. Escobar AL, Coimbra Jr CEA, Camacho LA, Santos V. Tuberculin reactivity and tuberculosis epidemiology in the Paakanova (Wari') Indians of Rondônia, Southwestern Brazilian Amazon. Int J Tuberc Lung Dis 2004; 8:45-51.) (1616. Basta PC, Oelemann MAC, Oelemann WMR, Fonseca LS; Coimbra Jr CEA. Detection of Mycobacterium tuberculosis in sputum from Suruí Indian subjects, Brazilian Amazon. Mem Inst Oswaldo Cruz 2006; 101:581-584.) (1717. Melo TEMP, Resendes APC, Souza-Santos R, Basta PC. Distribuição espacial e temporal da tuberculose em indígenas e não indígenas de Rondônia, Amazônia Ocidental, Brasil. Cad Saude Publica 2012; 28:267-280.) (2020. Orellana JDY, Gonçalves MJF, Basta PC. Sociodemographic features and operating indicators of tuberculosis control between indigenous and non-indigenous people of Rondônia, Western Amazon, Brazil. Rev Bras Epidemiol 2012; 15:714-724. , the measures employed to control TB in indigenous territories during the last decade do not seem to have impacted TB transmission. The high risk of disease and transmission reported in Igarapé Ribeirão might be representative of other indigenous territories. Therefore, we conclude that within indigenous territories, there is need to apply more stringent control measures, including increased active searches for respiratory symptoms and use of chemoprophylaxis, which has effectively prevented TB in the indigenous Guarani-Kaiowá people from the Midwest region of Brazil 3636. Yuhara LS, Sacchi FPC, Croda J. Impact of latent infection treatment in indigenous populations. PLoS One 2013; 8:e71201. , as well as further rigorously supervised treatment.
Besides that, public investments and inter-sectorial policies to mitigate social inequalities in Brazil are expected to impact control of TB and other endemic diseases related to poverty. In conclusion, we believe the analysis conducted in this manuscript can support policy formulation and improve control strategies for those vulnerable groups.
ACKNOWLEDGMENTS
We acknowledge Dr. Julia Ignez Salem from the National Institute for Amazon Research and Cleoni Alves Mendes Lima for their support in the analysis of laboratory tests. We thank Nilda de Oliveira Barros, coordinator of the state program for tuberculosis control in Rondônia for technical support and Perceverando Ribeiro Machado Neto and Luiz Carlos Corrêa Alves for performing chest radiographs and radiological reports, respectively. We are grateful to the staff members of the Polo Base Guajará-Mirim from Distrito Sanitário Especial Indígena (DSEI) Porto Velho, who supported us in all the stages of the research, including the first contacts with the community, logistical support, and transportation through the last stage when researchers returned to the territories with the lab results and followed up with the research subjects.
References:
-
1Ministério da Saúde. Secretaria de Vigilância em Saúde. Departamento de Vigilância Epidemiológica. Manual de recomendações para o controle da tuberculose no Brasil. Brasília: 2011. 288p.
-
2Nava-Aguilera E, Andersson N, Harris E, Mitchell S, Hamel C, Shea B, et al. Risk factors associated with recent transmission of tuberculosis: systematic review and meta-analysis. Int J Tuberc Lung Dis 2009; 13:17-26.
-
3Tollefson D, Bloss E, Fanning A, Redd JT, Barker K, McCray E. Burden of tuberculosis in indigenous peoples globally: a systematic review. Int J Tuber Lung Dis 2013; 17:1139-1150.
-
4Robertus LM, Konstantinos A, Hayman NE, Paterson DL. Tuberculosis in the Australian Indigenous population: history, current situation and future challenges. Aust N Z J Public Health 2011; 35:6-9.
-
5Culqui DR, Trujillo OV, Cueva N, Aylas R, Salaverry O, Bonilla C. Tuberculosis in the Indigenous population of Peru 2008. Rev Peru Med Exp Salud Publica 2010; 27:8-15.
-
6Canadian Tuberculosis Committee Housing conditions that serve as risk factors for tuberculosis infection and disease. An Advisory Committee Statement (ACS). Can Commun Dis Rep 2007; 33:1-33.
-
7Basta PC, Coimbra Jr CEA, Escobar AL, Santos RV, Alves LCC, Fonseca LS. Survey for tuberculosis in an indigenous population of Amazonia: the Suruí of Rondônia, Brazil. Trans R Soc Trop Med Hyg 2006; 100:579-585.
-
8Basta PC, Coimbra Jr CEA, Camacho LAB, Santos RV. Risk of tuberculous infection in an indigenous population from Amazonia, Brazil. Int J Tuber Lung Dis 2006; 10: 1354-1359.
-
9Basta PC, Coimbra Jr CEA, Welch JR, Correia Alves LC, Santos RV, Bastos Camacho LA. Tuberculosis among the Xavante Indians of the Brazilian Amazon: an epidemiological and ethnographic assessment. Ann Hum Biol 2010; 37:643-657.
-
10Barbosa T, Benigno A, Farre L, Prat JGI; Barral-Netto M, Arruda S. Tuberculosis among Yanomami communities from Alto Rio Negro, State of Amazonas, Brazil, 1990 to 2002. Enf Emerg 2008; 10:138-143.
-
11Rios DPG, Malacarne J, Alves LCC, Sant'Anna CC, Camacho LAB, Basta PC. Tuberculose em indígenas da Amazônia brasileira: Estudo epidemiológico na região do Alto Rio Negro. Rev Panam Salud Publica 2013; 33:22-29.
-
12Belo EN, Orellana JDY, Levino A, Basta PC. Tuberculose nos municípios amazonenses da fronteira Brasil-Colômbia-Peru-Venezuela: situação epidemiológica e fatores associados ao abandono. Rev Panam Salud Publica 2013; 34:321-329.
-
13Cunha EA, Ferrazoli L, Riley LW, Basta PC, Honer MR, Maia R et al. Incidence and transmission patterns of tuberculosis among indigenous populations in Brazil. Mem Inst Oswaldo Cruz 2014; 109:108-113.
-
14Escobar AL, Coimbra Jr CEA, Camacho LA, Portela MC. Tuberculose em populações indígenas de Rondônia, Amazônia, Brasil. Cad Saude Publica 2001; 17:285-298.
-
15Escobar AL, Coimbra Jr CEA, Camacho LA, Santos V. Tuberculin reactivity and tuberculosis epidemiology in the Paakanova (Wari') Indians of Rondônia, Southwestern Brazilian Amazon. Int J Tuberc Lung Dis 2004; 8:45-51.
-
16Basta PC, Oelemann MAC, Oelemann WMR, Fonseca LS; Coimbra Jr CEA. Detection of Mycobacterium tuberculosis in sputum from Suruí Indian subjects, Brazilian Amazon. Mem Inst Oswaldo Cruz 2006; 101:581-584.
-
17Melo TEMP, Resendes APC, Souza-Santos R, Basta PC. Distribuição espacial e temporal da tuberculose em indígenas e não indígenas de Rondônia, Amazônia Ocidental, Brasil. Cad Saude Publica 2012; 28:267-280.
-
18Basta PC, Camacho LAB. Teste tuberculínico na estimativa da prevalência de infecção por Mycobacterium tuberculosis em populações indígenas do continente americano: uma revisão da literatura. Cad Saude Publica 2006; 22:245-254.
-
19Young TK, Mirdad S. Determinants of tuberculin sensitivity in a child population covered by mass BCG vaccination. Tuber Lung Dis 1992; 73:94-100.
-
20Orellana JDY, Gonçalves MJF, Basta PC. Sociodemographic features and operating indicators of tuberculosis control between indigenous and non-indigenous people of Rondônia, Western Amazon, Brazil. Rev Bras Epidemiol 2012; 15:714-724.
-
21Arnadottir T, Rieder HL, Trébucq A, Waaler HT. Guidelines for conducting tuberculin skin test surveys in high prevalence countries. Tuber Lung Dis 1996; 77 (suppl 1):1-19.
-
22American Thoracic Society. Diagnostic Standards and Classification of Tuberculosis in Adults and Children. Am J Respir Crit Care Med 2000; 161:1376-1395.
-
23Den Boon S, Bateman ED, Enarson DA, Borgdorff MW, Verver S, Lombard CJ, et al. Development and evaluation of a new chest radiograph reading and recording system for epidemiological surveys of tuberculosis and lung disease. Int J Tuberc Lung Dis 2005; 9:1088-1096.
-
24Coimbra Jr CEA, Santos RV, Welch JR, Cardoso AM, Souza MC, Garnelo L, et al. The First National Survey of Indigenous People's Health and Nutrition in Brazil: rationale, methodology, and overview of results. BMC Public Health 2013; 13:52.
-
25Scatena-Villa TC, Ruffino-Netto A. Questionário para avaliação de desempenho de serviços de atenção básica no controle da TB no Brasil. J Bras Pneumol 2009; 35:610-612.
-
26Cauthen GM, Pio A, ten Dam HG. Annual risk of tuberculous infection. 1988 Bull World Health Organ 2002; 80:503-511.
-
27Villalba JA, Liu Y, Alvarez MK, Calderon L, Canache M, Cardenas G, et al. Low child survival index in a multi-dimensionally poor Amerindian population in Venezuela. PLoS One 2013; 8:e85638.
-
28Gryzbowski S, Styblo K, Dorken E. Tuberculosis in Eskimos. Tubercle 1976; 57 (suppl 4): S1-S58.
-
29Barros AJ, Hirakata VN. Alternatives for logistic regression in cross-sectional studies: an empirical comparison of models that directly estimate the prevalence ratio. BMC Med Res Methodol 2003; 3:21.
-
30Sacchi FP, Croda MG, Estevan AO, Ko AI, Croda J. Sugar cane manufacturing is associated with tuberculosis in an indigenous population in Brazil. Trans R Soc Trop Med Hyg . 2013; 107:152-157.
-
31Board AR, Suzuki S. The interrelation between intestinal parasites and latent TB infections among newly resettled refugees in Texas. Int Health 2016; 8:67-72.
-
32Assis EM, Oliveira RC, Moreira LE, Pena JL, Rodrigues LC, Machado-Coelho GLL. Prevalência de parasitos intestinais na comunidade indígena Maxakali, Minas Gerais, Brasil, 2009. Cad Saude Publica 2013; 29:681-690.
-
33Palhano-Silva CS, Araújo AJG, Lourenço AEP, Bastos OMP, Santos RV, Coimbra Jr CEA. Intestinal parasitic infection in the Suruí Indians, Brazilian Amazon. Interciencia 2009; 34:259-264.
-
34Zembrzuski VM, Basta PC, Callegari-Jacques SM, Santos RV, Coimbra CEA, Salzano FM, et al. Cytokine genes are associated with tuberculin skin test response in a native Brazilian population. Tuberculosis (Edinb) 2010; 90: 44-49.
-
35Longhi RMP, Zembrzuski VM, Basta PC, Croda J. Genetic polymorphism and immune response to tuberculosis in indigenous populations: a brief review. Braz J Infect Dis 2013; 17:363-368.
-
36Yuhara LS, Sacchi FPC, Croda J. Impact of latent infection treatment in indigenous populations. PLoS One 2013; 8:e71201.
-
We received support from the Brazilian National Council for Technological Development [Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq)], process number 402505/2008-5.
Publication Dates
-
Publication in this collection
Jul-Aug 2016
History
-
Received
07 June 2016 -
Accepted
08 Aug 2016