Abstract
Chagas disease is a neglected chronic condition with a high burden of morbidity and mortality. It has considerable psychological, social, and economic impacts. The disease represents a significant public health issue in Brazil, with different regional patterns. This document presents the evidence that resulted in the Brazilian Consensus on Chagas Disease. The objective was to review and standardize strategies for diagnosis, treatment, prevention, and control of Chagas disease in the country, based on the available scientific evidence. The consensus is based on the articulation and strategic contribution of renowned Brazilian experts with knowledge and experience on various aspects of the disease. It is the result of a close collaboration between the Brazilian Society of Tropical Medicine and the Ministry of Health. It is hoped that this document will strengthen the development of integrated actions against Chagas disease in the country, focusing on epidemiology, management, comprehensive care (including families and communities), communication, information, education, and research .
Keywords:
Chagas disease; Epidemiology; Control; Diagnosis; Treatment; Health care; Consensus; Brazil
INTRODUCTION
General aspects of Chagas disease epidemiology, with specific focus on Brazil
Chagas disease, an infectious condition with an acute and chronic phase, is classified as a neglected disease by the World Health Organization (WHO) 11. World Health Organization. Research priorities for Chagas disease, human African trypanosomiasis and leishmaniasis. WHO: technical report of the TDR Disease Reference Group on Chagas Disease, Human African Trypanosomiasis and Leishmaniasis. Geneva: World Health Organization; 2012. (WHO Technical Report Series, 975).) (22. World Health Organization. Sustaining the drive to overcome the global impact of neglected tropical diseases: second WHO report in neglected tropical diseases. Geneva: World Health Organization; 2013. . As a result of human poverty, it presents high morbidity and mortality load in endemic countries, including Brazil, with focal expression in different epidemiological contexts 11. World Health Organization. Research priorities for Chagas disease, human African trypanosomiasis and leishmaniasis. WHO: technical report of the TDR Disease Reference Group on Chagas Disease, Human African Trypanosomiasis and Leishmaniasis. Geneva: World Health Organization; 2012. (WHO Technical Report Series, 975).) (22. World Health Organization. Sustaining the drive to overcome the global impact of neglected tropical diseases: second WHO report in neglected tropical diseases. Geneva: World Health Organization; 2013.) (33. Dias JC, Silveira AC, Schofield CJ. The impact of Chagas disease control in Latin America: a review. Mem Inst Oswaldo Cruz. 2002 Jul;97(5):603-12.) (44. Coura JR, Dias JC. Epidemiology, control and surveillance of Chagas disease: 100 years after its discovery. Mem Inst Oswaldo Cruz. 2009 Jul;104 Suppl 1:31-40. . The geographic distribution of the disease is limited primarily to the American continent due to the distribution of more than 140 species of insect vectors ( Triatominae, Hemiptera, Reduviidae ); hence, it is also called American trypanosomiasis55. World Health Organization. Chagas disease in Latin America: an epidemiological update based on 2010 estimates. Wkly Epidemiol Rec. 2015 Feb;90(6):33-44. . Gradually, however, the disease has reached non-endemic countries; this as a result of an intense process of international migration and as the mode of disease transmission has changed 66. Moncayo A, Silveira AC. Current epidemiological trends for Chagas disease in Latin America and future challenges in epidemiology, surveillance and health policy. Mem Inst Oswaldo Cruz. 2009 Jul;104 Suppl 1:17-30.) (77. Schmunis GA, Yadon ZE. Chagas disease: a Latin American health problem becoming a world health problem. Acta Trop. 2010 Jul-Aug;115(1-2):14-21.) (88. Centers for Disease Control and Prevention. Center for Global Health. Division of Parasitic Diseases and Malaria. Chagas disease in the Americas: 2013. Atlanta: Centers for Disease Control and Prevention; 2013.) (99. Hotez PJ, Dumonteil E, Betancourt Cravioto M, Bottazzi ME, Tapia-Conyer R, Meymandi S, et al. An unfolding tragedy of Chagas disease in North America. PLoS Negl Trop Dis. 2013 Oct;7(10):e2300.) (1010. Dias JCP. Human chagas disease and migration in the context of globalization: some particular aspects. J Trop Med. 2013:789758.) (1111. Coura JR, Viñas PA, Junqueira AC. Ecoepidemiology, short history and control of Chagas disease in the endemic countries and the new challenge for non-endemic countries. Mem Inst Oswaldo Cruz. 2014 Nov;109(7):856-62.) (1212. European Centre for Disease Prevention and Control. Assessing the burden of key infectious diseases affecting migrant populations in the EU/EEA: technical report. Stockholm: ECDC; 2014.) (1313. Requena-Méndez A, Aldasoro E, Lazzari E, Sicuri E, Brown M, Moore DA, et al. Prevalence of Chagas disease in Latin-American migrants living in Europe: a systematic review and meta-analysis. PLoS Negl Trop Dis. 2015 Feb;9(2):e0003540. .
The World Health Organization (WHO) estimates that approximately 6-7 million people are infected worldwide, mostly in Latin America 1414. World Health Organization. Chagas disease (American trypanosomiasis) [Internet]. Geneva: World Health Organization ; 2015 [citado 2015 Nov 13]. Disponível em: Disponível em: http://www.who.int/mediacentre/factsheets/fs340/en/
http://www.who.int/mediacentre/factsheet...
.Recent estimates for the 21 Latin American countries, based on data from 2010, showed that 5,742,167 people were infected by Trypanosoma cruzi , of which 3,581,423 (62.4%) were living in the countries of the Southern Cone Initiative against Chagas disease, especially Argentina (1,505,235), Brazil (1,156,821), Mexico (876,458), and Bolivia (607,186) 55. World Health Organization. Chagas disease in Latin America: an epidemiological update based on 2010 estimates. Wkly Epidemiol Rec. 2015 Feb;90(6):33-44. . However, these data differ from the estimates of other research groups using other methods to define T. cruzi infection in many countries; this hinders accurate establishment of the prevalence of Chagas disease in the Americas. Nevertheless, the authors agree that the number of infected individuals is still very significant in the health and social context of the continent, requiring priority and attention from the countries 1515. Ramos Júnior AN, Carvalho DM. Chagas' disease: past, present and future. Cad Saude Colet. 2009;17(4):787-94.) (1616. Strasen J, Williams T, Ertl G, Zoller T, Stich A, Ritter O. Epidemiology of Chagas disease in Europe: many calculations, little knowledge. Clin Res Cardiol. 2014 Jan;103(1):1-10. .
Table 1 represents changes in epidemiological parameters specific to Latin America in the recent years 55. World Health Organization. Chagas disease in Latin America: an epidemiological update based on 2010 estimates. Wkly Epidemiol Rec. 2015 Feb;90(6):33-44.) (1717. Pan American Health Organization. Estimación cuantitativa de la enfermedad de Chagas en las Americas. Washington: Pan American Health Organization; 2006. .
Thus, despite advances in vector control and quality assurance of blood transfusions in many of these countries, especially from intergovernmental initiatives started in the 1990s 33. Dias JC, Silveira AC, Schofield CJ. The impact of Chagas disease control in Latin America: a review. Mem Inst Oswaldo Cruz. 2002 Jul;97(5):603-12.) (44. Coura JR, Dias JC. Epidemiology, control and surveillance of Chagas disease: 100 years after its discovery. Mem Inst Oswaldo Cruz. 2009 Jul;104 Suppl 1:31-40.) (1010. Dias JCP. Human chagas disease and migration in the context of globalization: some particular aspects. J Trop Med. 2013:789758.) (1111. Coura JR, Viñas PA, Junqueira AC. Ecoepidemiology, short history and control of Chagas disease in the endemic countries and the new challenge for non-endemic countries. Mem Inst Oswaldo Cruz. 2014 Nov;109(7):856-62.) (1818. Dias JCP. Southern Cone Initiative for the elimination of domestic populations of Triatoma infestans and the interruption of transfusion Chagas disease: historical aspects, present situation, and perspectives. Mem Inst Oswaldo Cruz. 2007 Oct;102 Suppl 1:11-8. , Chagas disease remains a relevant public health problem in Latin America, showing different regional patterns of epidemiological expression. The challenges are magnified by the lack of access to diagnosis and systematic treatment; according to 2015 estimates >80% of individuals affected by Chagas disease globally had no such access. This supports the high impact of morbidity, mortality, and the social cost of this illness.
Uncontrolled human migration, environmental degradation, climate change, high population concentrations in urban areas, and precarious socio-economic conditions (housing, education, sanitation, and income, among others) are determinants and social conditioning factors for the transmission of T. cruzi11. World Health Organization. Research priorities for Chagas disease, human African trypanosomiasis and leishmaniasis. WHO: technical report of the TDR Disease Reference Group on Chagas Disease, Human African Trypanosomiasis and Leishmaniasis. Geneva: World Health Organization; 2012. (WHO Technical Report Series, 975).) (1010. Dias JCP. Human chagas disease and migration in the context of globalization: some particular aspects. J Trop Med. 2013:789758.) (1111. Coura JR, Viñas PA, Junqueira AC. Ecoepidemiology, short history and control of Chagas disease in the endemic countries and the new challenge for non-endemic countries. Mem Inst Oswaldo Cruz. 2014 Nov;109(7):856-62.) (1919. Prata A. Clinical and epidemiological aspects of Chagas disease. Lancet Infect Dis. 2001 Sep;1(2):92-100.) (2020. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Consenso Brasileiro em Doença de Chagas. Rev Soc Bras Med Trop. 2005;38 Supl 3:1-29.) (2121. Organização Pan-americana da Saúde. Guia para vigilância, prevenção, controle e manejo clínico da doença de Chagas aguda transmitida por alimentos. Rio de Janeiro: Organização Pan-Americana da Saúde; 2009. (Série de Manuais Técnicos, 12).) (2222. Silveira AC, Dias JCP. O controle da transmissão vetorial. Rev Soc Bras Med Trop. 2011;44 supl 2:52-63.) (2323. Coura JR. The main sceneries of Chagas disease transmission. The vectors, blood and oral transmissions: a comprehensive review. Mem Inst Oswaldo Cruz. 2015 May;110(3):277-82. . Infected populations are vulnerable to varying degrees of neglect, expressed by overlapping and increased exposure to other diseases, conditions and injuries; lower coverage for preventive interventions; a higher likelihood of illness; less access to a network of health services, to secondary and tertiary health care, and to other services; poor quality of primary care services; lesser likelihood of receiving essential treatments; and greater likelihood of developing severe forms of the disease with increased risk of progression to death 2424. Victora CG, Wagstaff A, Schellenberg JA, Gwatkin D, Claeson M, Habicht JP. Applying an equity lens to child health and mortality: more of the same is not enough. Lancet. 2003 Jul;362(9379):233-41. .
From this perspective, it is essential to obtain greater knowledge concerning the epidemiological scenario of Chagas disease and its transmission dynamics, involving people who are infected or are at risk of infection, different parasite populations, vector species, and T. cruzi reservoirs. This integrated knowledge is central to the search for consistent and sustainable management actions, monitoring, and coherent health and social control programs that are efficient and effective 11. World Health Organization. Research priorities for Chagas disease, human African trypanosomiasis and leishmaniasis. WHO: technical report of the TDR Disease Reference Group on Chagas Disease, Human African Trypanosomiasis and Leishmaniasis. Geneva: World Health Organization; 2012. (WHO Technical Report Series, 975).) (1010. Dias JCP. Human chagas disease and migration in the context of globalization: some particular aspects. J Trop Med. 2013:789758.) (1111. Coura JR, Viñas PA, Junqueira AC. Ecoepidemiology, short history and control of Chagas disease in the endemic countries and the new challenge for non-endemic countries. Mem Inst Oswaldo Cruz. 2014 Nov;109(7):856-62.) (2525. Zingales B, Andrade SG, Briones MR, Campbell DA, Chiari E, Fernandes O, et al. A new consensus for Trypanosoma cruzi intraspecific nomenclature: second revision meeting recommends TcI to TcVI. Mem Inst Oswaldo Cruz. 2009 Nov;104(7):1051-4.) (2626. Zingales B, Miles MA, Moraes CB, Luquetti A, Guhl F, Schijman AG, et al. Drug discovery for Chagas disease should consider Trypanosoma cruzi strain diversity. Mem Inst Oswaldo Cruz. 2014 Aug;109(6):828-33.) (2727. Messenger LA, Miles MA, Bern C. Between a bug and a hard place: Trypanosoma cruzi genetic diversity and the clinical outcomes of Chagas disease. Expert Rev Anti Infect Ther. 2015 Aug;13(8):995-1029. .
Despite a clear need for more evidence, more than 105 years since the discovery of the disease by Carlos Ribeiro Justiniano Chagas (1909) there are still important gaps in technical, scientific, and political fields that must be overcome to effectively face this widely-neglected condition.
Brazil, with its continental dimensions, is undergoing major and rapid demographic, social, and environmental change, but serious socio-economic and regional inequalities persist. Here, diseases associated with social contexts of vulnerability and neglect still afflict a considerable part of the population 2828. Victora CG, Barreto ML, Carmo Leal M, Monteiro CA, Schmidt MI, Paim J, et al. Health conditions and health-policy innovations in Brazil: the way forward. Lancet. 2011 Jun;377(9782):2042-53. . Gradually, however, a consistent improvement in the overall health status and life expectancy of the Brazilian population has been observed. This can be attributed to changes in the social determinants of health, advances in the control of infectious diseases, and implementation of a more comprehensive national health system with relevant social participation 2828. Victora CG, Barreto ML, Carmo Leal M, Monteiro CA, Schmidt MI, Paim J, et al. Health conditions and health-policy innovations in Brazil: the way forward. Lancet. 2011 Jun;377(9782):2042-53.) (2929. Barreto ML, Teixeira MG, Bastos FI, Ximenes RAA, Barata RB, Rodrigues LC. Successes and failures in the control of infectious diseases in Brazil: social and environmental context, policies, interventions, and research needs. Lancet. 2011 May;377(9780):1877-89. .
Despite these social advances, it is recognized that the persistence of large social inequalities, that result in individual, programmatic and social vulnerabilities, is strongly related to Chagas disease. New health problems emerge, however, as the result of an intense urbanization process and changes in the social and environmental field; others tend to persist and coexist 2828. Victora CG, Barreto ML, Carmo Leal M, Monteiro CA, Schmidt MI, Paim J, et al. Health conditions and health-policy innovations in Brazil: the way forward. Lancet. 2011 Jun;377(9782):2042-53. , requiring new healthcare response networks 3030. Mendes EV. O cuidado das condições crônicas na atenção primária à saúde: o imperativo da consolidação da estratégia da saúde da família. Brasília: Organização Pan-Americana da Saúde; 2012. .
These demand specific actions, adjusted to new realities, focused on an integrated approach to the different components involved in the natural history of the disease: humans, vectors, reservoirs, and T. cruzi33. Dias JC, Silveira AC, Schofield CJ. The impact of Chagas disease control in Latin America: a review. Mem Inst Oswaldo Cruz. 2002 Jul;97(5):603-12.) (66. Moncayo A, Silveira AC. Current epidemiological trends for Chagas disease in Latin America and future challenges in epidemiology, surveillance and health policy. Mem Inst Oswaldo Cruz. 2009 Jul;104 Suppl 1:17-30.) (1515. Ramos Júnior AN, Carvalho DM. Chagas' disease: past, present and future. Cad Saude Colet. 2009;17(4):787-94. . The great extent and territorial diversity, with the specific ecologic, demographic, social, and economic dynamics of the regions, implies multiple epidemiological and operational clinical scenarios 33. Dias JC, Silveira AC, Schofield CJ. The impact of Chagas disease control in Latin America: a review. Mem Inst Oswaldo Cruz. 2002 Jul;97(5):603-12.) (44. Coura JR, Dias JC. Epidemiology, control and surveillance of Chagas disease: 100 years after its discovery. Mem Inst Oswaldo Cruz. 2009 Jul;104 Suppl 1:31-40.) (1515. Ramos Júnior AN, Carvalho DM. Chagas' disease: past, present and future. Cad Saude Colet. 2009;17(4):787-94. .
In addition to the challenges, the coming years bring opportunities for shared, more concrete action in fighting Chagas disease. In 2015 the 17 new Sustainable Development Goals (SDGs) were released; these are based on the eight Millennium Development Goals (MDGs). The agenda, which was agreed to and consolidated in the framework document Transforming Our World: The 2030 Agenda for Sustainable Development , represents a historic and unprecedented opportunity for countries to seek new global paths 3131. Nações Unidas. Transformando nosso mundo: a agenda 2030 para o desenvolvimento sustentável [Internet]. [Rio de Janeiro]: Nações Unidas no Brasil; 2015 [citado 2015 out 4]. Disponível em: http://nacoesunidas.org/pos2015/agenda2030/ .
http://nacoesunidas.org/pos2015/agenda20...
. In a broader sense, in order to promote prosperity and well-being for all, to protect the environment and cope with climate changes 3131. Nações Unidas. Transformando nosso mundo: a agenda 2030 para o desenvolvimento sustentável [Internet]. [Rio de Janeiro]: Nações Unidas no Brasil; 2015 [citado 2015 out 4]. Disponível em: http://nacoesunidas.org/pos2015/agenda2030/ .
http://nacoesunidas.org/pos2015/agenda20...
, these decisions will determine the global course of action to confront and eliminate poverty.
Chagas disease is part of the third objective of this document, which aims to ensure a healthy life and promote well-being for all, at all ages . The goal is to control the acquired immunodeficiency syndrome (AIDS) epidemic, to eradicate tuberculosis, malaria, and neglected tropical diseases and to combat hepatitis and waterborne and other transmitted diseases by 2030 3131. Nações Unidas. Transformando nosso mundo: a agenda 2030 para o desenvolvimento sustentável [Internet]. [Rio de Janeiro]: Nações Unidas no Brasil; 2015 [citado 2015 out 4]. Disponível em: http://nacoesunidas.org/pos2015/agenda2030/ .
http://nacoesunidas.org/pos2015/agenda20...
. The progress in specific indicator composition to monitor neglected tropical diseases in the world is highlighted, from the recognition of the number of people requiring interventions against this group of diseases. In addition, Chagas disease and other neglected tropical diseases also interface with the second (zero hunger and sustainable agriculture), fifth (gender equality), and sixth (drinking water and sanitation) objectives 3131. Nações Unidas. Transformando nosso mundo: a agenda 2030 para o desenvolvimento sustentável [Internet]. [Rio de Janeiro]: Nações Unidas no Brasil; 2015 [citado 2015 out 4]. Disponível em: http://nacoesunidas.org/pos2015/agenda2030/ .
http://nacoesunidas.org/pos2015/agenda20...
.
MORBIDITY AND MORTALITY
Trypanosoma cruzi infection
In Brazil, few population-based systematic studies have been conducted. This hinders accurate estimation of the magnitude of the impact Chagas disease throughout history.
Prior to the 1950s, Chagas disease was recognized as an eminently rural endemic disease, transmitted predominantly through vectors, occurring in areas of high social vulnerability. With industrialization, the disease became modeled in a new, urban epidemiological context, increased by internal migration within the country from rural to urban areas with consequent growth of cities 44. Coura JR, Dias JC. Epidemiology, control and surveillance of Chagas disease: 100 years after its discovery. Mem Inst Oswaldo Cruz. 2009 Jul;104 Suppl 1:31-40. .
Between 1975 and 1980, a national serological survey in the rural population of Brazil (except São Paulo), estimated the prevalence to be 4.2%, corresponding to 6.5 million people infected by T. cruzi2020. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Consenso Brasileiro em Doença de Chagas. Rev Soc Bras Med Trop. 2005;38 Supl 3:1-29.) (3232. Camargo ME, Silva GR, Castilho EA, Silveira AC. Serological survey of the prevalence of Chagas' infection in Brazil, 1975/1980. Rev Inst Med Trop São Paulo. 1984 Jul-Aug;26(4):192-204.) (3333. Silveira AC, Silva GR, Prata A. O Inquérito de soroprevalência da infecção chagásica humana (1975-1980). Rev Soc Bras Med Trop. 2011;44 supl 2:33-9. . In 1996, it was estimated that the prevalence of T. cruzi infection in Brazil was at 3.1% in 1978 and 1.3% in 1995, amounting to an estimated 1.96 million people infected 3434. Akhavan D. Análise de custo-efectividade do programa de controle da doença de Chagas no Brasil: relatório final. Brasília: Organização Pan-Americana de Saúde; 1998. . Subsequently, the Pan American Health Organization (PAHO) estimated that 21.8 million people in endemic areas in Brazil were at risk, given the estimated 1.9 million (1.019% of the population) infected individuals 1717. Pan American Health Organization. Estimación cuantitativa de la enfermedad de Chagas en las Americas. Washington: Pan American Health Organization; 2006. .
In 2014, the first systematic review and meta-analysis to estimate the prevalence of Chagas disease in Brazil was published. It included publications from the period between 1980 and 2012, and estimated the pooled prevalence of Chagas disease at 4.2% [95% confidence interval (95% CI): 3.1-5.7], ranging from 4.4% (95% CI: 2.3-8.3) in the 1980s to 2.4% (95% CI: 1.5-3.8) after 2000 3535. Martins-Melo FR, Ramos Júnior AN, Alencar CH, Heukelbach J. Prevalence of Chagas disease in Brazil: a systematic review and meta-analysis. Acta Trop. 2014 Feb;130:167-74. . In that study, the highest prevalence was found in women (4.2%; 95% CI: 2.6-6.8), individuals over 60 years old (17.7%; 95% CI: 11.4-26.5), residents in the Northeastern (5.0%; 95% CI: 3.1-8.1) and Southeastern regions (5.0%; 95% CI: 2.4-9.9), and in mixed urban/rural areas (6.4%; 95% CI: 4.2-9.4). It was estimated that 4.6 million people (95% CI: 2.9-7.2 million) would be infected by T. cruzi in Brazil. Emphasizing the limitation of the findings in the literature, the authors highlight the need for new studies, with a view to obtaining truer estimates 3535. Martins-Melo FR, Ramos Júnior AN, Alencar CH, Heukelbach J. Prevalence of Chagas disease in Brazil: a systematic review and meta-analysis. Acta Trop. 2014 Feb;130:167-74. .
From this and other studies, the most recent estimates in Brazil regarding the number of people infected by T. cruzi vary from 1.9 million to 4.6 million people 3535. Martins-Melo FR, Ramos Júnior AN, Alencar CH, Heukelbach J. Prevalence of Chagas disease in Brazil: a systematic review and meta-analysis. Acta Trop. 2014 Feb;130:167-74.) (3636. Hotez PJ, Fujiwara RT. Brazil's neglected tropical diseases: an overview and a report card. Microbes Infect. 2014 Aug;16(8):601-6.) (3737. Bern C, Kjos S, Yabsley MJ. Montgomery SP. Trypanosoma cruzi and Chagas disease in the United States. Clin Microbiol Rev. 2011 Oct;24(4):655-81. , probably closer now to 1.0-2.4% of the population.
This epidemiological scenario brings challenges in the coming decades for the country to support control actions and establish a consistent plan in the Unified Health System [ Sistema Único de Saúde (SUS)] for diagnosis, treatment, and comprehensive care of millions of people 3838. Dias JC, Coura JR, Yasuda MAS. The present situation, challenges, and perspectives regarding the production and utilization of effective drugs against human Chagas disease. Rev Soc Bras Med Trop. 2014 Jan-Feb;47(1):123-5. , combined with epidemiological surveillance adjusted to this reality 1515. Ramos Júnior AN, Carvalho DM. Chagas' disease: past, present and future. Cad Saude Colet. 2009;17(4):787-94.) (2222. Silveira AC, Dias JCP. O controle da transmissão vetorial. Rev Soc Bras Med Trop. 2011;44 supl 2:52-63. . It is presumed that up to 30% of chronically infected people are likely to develop cardiac alterations and up to 10% may experience digestive, neurologic, or mixed alterations. Given this, there is a heightened need to structure a comprehensive network of timely health care for Chagas disease in the country 3939. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Guia de Vigilância em Saúde. Brasília: Ministério da Saúde; 2014. .
In order to establish estimates for the number of people infected by T. cruzi , the publication Projeção da População do Brasil por Sexo e Idade para o Período 2000/2060 e Projeção da População das Unidades da Federação por Sexo e Idade para o período 2000/2030 (Projection of Brazil's Population by Sex and Age for the Period 2000/2060 and Projection of the Federative Unit's Population by Sex and Age for the Period 2000/2030) by the Brazilian Institute of Geography and Statistics [ Instituto Brasileiro de Geografia e Estatística (IGBE)] was taken as a population-based reference. According to the IBGE the Brazilian population will continue to grow until 2042, when it will reach 228.4 million people, mostly concentrated in urban centers. From there, it will gradually decrease and it will account for approximately 218.2 million individuals in 2060 4040. Instituto Brasileiro de Geografia e Estatística. Projeção da população do Brasil por sexo e idade para o período 2000/2060 e projeção da população das Unidades da Federação por sexo e idade para o período 2000/2030. Brasília: Instituto Brasileiro de Geografia e Estatística; 2013. .
Table 2 shows the relative projection to the estimates of the number of infected people by T. cruzi and the number of Chagas disease cases in the chronic phase with the cardiac and digestive form of the disease in Brazil, between 2015 and 2055, according to five-year periods. In 2015, for example, taking as a basis an estimated population of 204,450,649, it is estimated that between 1,426,994 and 3,357,633 of Brazilian individuals would be infected with T. cruzi . Of these, 142,699 to 335,763 would potentially have the digestive form and 428,098 to 1,007,290 would have the cardiac form of the disease. Moreover, an estimated 856,197 to 2,014,580 individuals would potentially present with T. cruzi infection in the indeterminate form.
In addition to internal migration in Brazil, with the urbanization of Chagas disease its occurrence in migrants from endemic areas in countries not traditionally endemic has brought about an expanded discussion on the contexts of risk and vulnerability, with a challenge to develop control actions for these countries 33. Dias JC, Silveira AC, Schofield CJ. The impact of Chagas disease control in Latin America: a review. Mem Inst Oswaldo Cruz. 2002 Jul;97(5):603-12.) (66. Moncayo A, Silveira AC. Current epidemiological trends for Chagas disease in Latin America and future challenges in epidemiology, surveillance and health policy. Mem Inst Oswaldo Cruz. 2009 Jul;104 Suppl 1:17-30.) (77. Schmunis GA, Yadon ZE. Chagas disease: a Latin American health problem becoming a world health problem. Acta Trop. 2010 Jul-Aug;115(1-2):14-21.) (1010. Dias JCP. Human chagas disease and migration in the context of globalization: some particular aspects. J Trop Med. 2013:789758.) (1111. Coura JR, Viñas PA, Junqueira AC. Ecoepidemiology, short history and control of Chagas disease in the endemic countries and the new challenge for non-endemic countries. Mem Inst Oswaldo Cruz. 2014 Nov;109(7):856-62.) (1212. European Centre for Disease Prevention and Control. Assessing the burden of key infectious diseases affecting migrant populations in the EU/EEA: technical report. Stockholm: ECDC; 2014.) (1313. Requena-Méndez A, Aldasoro E, Lazzari E, Sicuri E, Brown M, Moore DA, et al. Prevalence of Chagas disease in Latin-American migrants living in Europe: a systematic review and meta-analysis. PLoS Negl Trop Dis. 2015 Feb;9(2):e0003540.) (1616. Strasen J, Williams T, Ertl G, Zoller T, Stich A, Ritter O. Epidemiology of Chagas disease in Europe: many calculations, little knowledge. Clin Res Cardiol. 2014 Jan;103(1):1-10.) (1818. Dias JCP. Southern Cone Initiative for the elimination of domestic populations of Triatoma infestans and the interruption of transfusion Chagas disease: historical aspects, present situation, and perspectives. Mem Inst Oswaldo Cruz. 2007 Oct;102 Suppl 1:11-8.) (3737. Bern C, Kjos S, Yabsley MJ. Montgomery SP. Trypanosoma cruzi and Chagas disease in the United States. Clin Microbiol Rev. 2011 Oct;24(4):655-81.) (4141. Bern C, Verastegui M, Gilman RH, Lafuente C, Galdos-Cardenas G, Calderon M, et al. Congenital Trypanosoma cruzi transmission in Santa Cruz, Bolivia. Clin Infect Dis. 2009 Dec 1;49(11):1667-74.) (4242. Rassi Júnior A, Rassi A, Marin-Neto JA. Chagas disease. Lancet. 2010 Apr;375(9723):1388-402. . Multiple bio-ecological, socio-cultural, and political factors have been implicated in this process 1010. Dias JCP. Human chagas disease and migration in the context of globalization: some particular aspects. J Trop Med. 2013:789758. , demanding an increased need for human and social sciences research concerning the disease 4343. Ventura-Garcia L, Roura M, Pell C, Posada E, Gascón J, Aldasoro E, et al. Socio-cultural aspects of Chagas disease: a systematic review of qualitative research. PLoS Negl Trop Dis. 2013 Sep 12;7(9):e2410. .
In 2005, it was estimated that there were a total of 501,036 Brazilians in the United States, with a prevalence of infection by T. cruzi of 1.02% 1717. Pan American Health Organization. Estimación cuantitativa de la enfermedad de Chagas en las Americas. Washington: Pan American Health Organization; 2006. , or 5,106 cases 4444. Bern C, Montgomery SP. An estimate of the burden of Chagas disease in the United States. Clin Infect Dis. 2009 Sep 1;49(5):e52-4. . In a broader sense, it is estimated that the total of people infected by T. cruzi in that country could range from 300 thousand to just over 1.0 million 88. Centers for Disease Control and Prevention. Center for Global Health. Division of Parasitic Diseases and Malaria. Chagas disease in the Americas: 2013. Atlanta: Centers for Disease Control and Prevention; 2013.) (99. Hotez PJ, Dumonteil E, Betancourt Cravioto M, Bottazzi ME, Tapia-Conyer R, Meymandi S, et al. An unfolding tragedy of Chagas disease in North America. PLoS Negl Trop Dis. 2013 Oct;7(10):e2300. , with different impacts on the health system. It is worth noting that this is a non-endemic area 99. Hotez PJ, Dumonteil E, Betancourt Cravioto M, Bottazzi ME, Tapia-Conyer R, Meymandi S, et al. An unfolding tragedy of Chagas disease in North America. PLoS Negl Trop Dis. 2013 Oct;7(10):e2300.) (4545. Buekens P, Almendares O, Carlier Y, Dumonteil E, Eberhard M, Gamboa-Leon R, et al. Mother-to-child transmission of Chagas' disease in North America: why don't we do more? Matern Child Health J. 2008 May;12(3):283-6. .
It is estimated that approximately 72,000 people infected by T. cruzi live in Europe 1212. European Centre for Disease Prevention and Control. Assessing the burden of key infectious diseases affecting migrant populations in the EU/EEA: technical report. Stockholm: ECDC; 2014. . From analysis of the aggregated data of a systematic review and meta-analysis investigating the prevalence of Chagas disease in Latin American people living in Europe (Spain, France, Switzerland, Italy and Germany), it is estimated that the prevalence of this infection in Brazilian migrants is 0.6% (from 0.16 to 1.12%) or 4/954 1313. Requena-Méndez A, Aldasoro E, Lazzari E, Sicuri E, Brown M, Moore DA, et al. Prevalence of Chagas disease in Latin-American migrants living in Europe: a systematic review and meta-analysis. PLoS Negl Trop Dis. 2015 Feb;9(2):e0003540. . Migrants from Bolivia had the highest prevalence of Chagas disease (18.1%), followed by Paraguayan migrants (5.5%). The prevalence among migrants from Argentina was 2.2%; there were no cases of Chagas disease detected among migrants from Uruguay, Venezuela, Panama, Guatemala, and Mexico 1313. Requena-Méndez A, Aldasoro E, Lazzari E, Sicuri E, Brown M, Moore DA, et al. Prevalence of Chagas disease in Latin-American migrants living in Europe: a systematic review and meta-analysis. PLoS Negl Trop Dis. 2015 Feb;9(2):e0003540. .
The occurrence of other important international migration waves within Latin America, such as from Bolivia and Paraguay to Argentina and Brazil or from Colombia to Venezuela, is recognized 1010. Dias JCP. Human chagas disease and migration in the context of globalization: some particular aspects. J Trop Med. 2013:789758. . On the other hand, from the second half of the twentieth century, it is especially emphasized the intensification of the movement of thousands of citizens from Latin America to countries in North America (mainly the United States, but also Canada), Europe (particularly Spain, among other nations), Asia (Japan), and Oceania (Australia), increasing the number of people with Chagas disease living in non-endemic countries 22. World Health Organization. Sustaining the drive to overcome the global impact of neglected tropical diseases: second WHO report in neglected tropical diseases. Geneva: World Health Organization; 2013.) (77. Schmunis GA, Yadon ZE. Chagas disease: a Latin American health problem becoming a world health problem. Acta Trop. 2010 Jul-Aug;115(1-2):14-21.) (1010. Dias JCP. Human chagas disease and migration in the context of globalization: some particular aspects. J Trop Med. 2013:789758.) (1212. European Centre for Disease Prevention and Control. Assessing the burden of key infectious diseases affecting migrant populations in the EU/EEA: technical report. Stockholm: ECDC; 2014.) (4242. Rassi Júnior A, Rassi A, Marin-Neto JA. Chagas disease. Lancet. 2010 Apr;375(9723):1388-402. . Overall, this migration process is associated with the search for improved living conditions for people from endemic countries living in conditions of serious social vulnerability 1010. Dias JCP. Human chagas disease and migration in the context of globalization: some particular aspects. J Trop Med. 2013:789758. .
In addition to Brazilians potentially infected by T. cruzi living abroad, a considerable number of people have migrated to Brazil from other endemic countries such as Bolivia; an estimated 80,000 to 200,000 Bolivian migrants live in the State of Sao Paulo 4646. Silveira C, Carneiro Junior N, Ribeiro MC, Barata RC. Living conditions and access to health services by Bolivian immigrants in the city of São Paulo, Brazil. Cad Saude Publica. 2013 Oct;29(10):2017-27. . This fact has prompted discussion, within the country, about public health policies consistent with the need for greater attention to this migrant population, including viewing the precarious life and work conditions to which they are subjected 4646. Silveira C, Carneiro Junior N, Ribeiro MC, Barata RC. Living conditions and access to health services by Bolivian immigrants in the city of São Paulo, Brazil. Cad Saude Publica. 2013 Oct;29(10):2017-27.) (4747. Martes ACB, Faleiros SM. Acesso dos imigrantes bolivianos aos serviços públicos de saúde na cidade de São Paulo. Saude Soc. 2013 abr-jun;22(2):351-64. .
In addition to the migration context, there is a clear shift in the prevalence of infection by T. cruzi to older age groups, which brings new challenges for the SUS. With the prevalence of chronic phase Chagas disease resulting largely from infection by vectors in the past, the probability of infectious and non-infectious comorbidities has expanded. These comorbidities consist mostly of chronic degenerative conditions (especially diabetes mellitus, systemic arterial hypertension, and other heart diseases), human immunodeficiency virus (HIV) co-infection with T. cruzi , and other immunosuppressive conditions that are acquired or induced 22. World Health Organization. Sustaining the drive to overcome the global impact of neglected tropical diseases: second WHO report in neglected tropical diseases. Geneva: World Health Organization; 2013.) (4848. Almeida EA, Lima JN, Lages-Silva E, Guariento ME, Aoki FH, Torres-Morales AE, et al. Chagas' disease and HIV co-infection in patients without effective antiretroviral therapy: prevalence, clinical presentation and natural history. Trans R Soc Trop Med Hyg. 2010 Jul;104(7):447-52.) (4949. Almeida EA, Ramos Júnior AN, Correia D, Shikanai-Yasuda MA. Co-infection Trypanosoma cruzi /HIV: systematic review (1980-2010). Rev Soc Bras Med Trop. 2011 Nov-Dec;44(6):762-70.) (5050. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Departamento de DST, Aids e Hepatites Virais. Bol Epidemiol. 2014;3(1):1-80) (5151. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Departamento de DST, Aids e Hepatites Virais. Protocolo clínico e diretrizes terapêuticas para manejo da infecção pelo HIV em adultos. Brasília: Ministério da Saúde; 2014. . The risks are amplified by longer survival combined with migration and the urbanization that had occurred over the past five decades 4949. Almeida EA, Ramos Júnior AN, Correia D, Shikanai-Yasuda MA. Co-infection Trypanosoma cruzi /HIV: systematic review (1980-2010). Rev Soc Bras Med Trop. 2011 Nov-Dec;44(6):762-70. .
As for other infectious diseases, T. cruzi can behave as an opportunistic microorganism in individuals with imunosupression 22. World Health Organization. Sustaining the drive to overcome the global impact of neglected tropical diseases: second WHO report in neglected tropical diseases. Geneva: World Health Organization; 2013.) (4949. Almeida EA, Ramos Júnior AN, Correia D, Shikanai-Yasuda MA. Co-infection Trypanosoma cruzi /HIV: systematic review (1980-2010). Rev Soc Bras Med Trop. 2011 Nov-Dec;44(6):762-70.) (5050. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Departamento de DST, Aids e Hepatites Virais. Bol Epidemiol. 2014;3(1):1-80) (5151. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Departamento de DST, Aids e Hepatites Virais. Protocolo clínico e diretrizes terapêuticas para manejo da infecção pelo HIV em adultos. Brasília: Ministério da Saúde; 2014.) (5252. Ministério da Saúde (BR). Recomendações para diagnóstico, tratamento e acompanhamento da co-infecção Trypanosoma cruzi: vírus da imunodeficiência humana. Rev Soc Bras Med Trop. 2006 jul-ago;39(4):392-415. . Although the first case of T. cruzi and HIV co-infection was reported in the 1980s, the frequency of this co-infection and of specific reactivation; the clinical and laboratory features associated with co-infection; specific, adequate treatments for T. cruzi infection in the context of co-infection with HIV; and outcomes (survival and death) of co-infected individuals, persist as knowledge gaps at a global level 4949. Almeida EA, Ramos Júnior AN, Correia D, Shikanai-Yasuda MA. Co-infection Trypanosoma cruzi /HIV: systematic review (1980-2010). Rev Soc Bras Med Trop. 2011 Nov-Dec;44(6):762-70.) (5252. Ministério da Saúde (BR). Recomendações para diagnóstico, tratamento e acompanhamento da co-infecção Trypanosoma cruzi: vírus da imunodeficiência humana. Rev Soc Bras Med Trop. 2006 jul-ago;39(4):392-415.) (5353. Martins-Melo FR, Ramos Júnior AN, Alencar CH, Heukelbach J. Mortality related to Chagas disease and HIV/AIDS coinfection in Brazil. J Trop Med. 2012;2012:534649. .
In Brazil, we estimated the prevalence of co-infection to be 1.3% 4848. Almeida EA, Lima JN, Lages-Silva E, Guariento ME, Aoki FH, Torres-Morales AE, et al. Chagas' disease and HIV co-infection in patients without effective antiretroviral therapy: prevalence, clinical presentation and natural history. Trans R Soc Trop Med Hyg. 2010 Jul;104(7):447-52.) (4949. Almeida EA, Ramos Júnior AN, Correia D, Shikanai-Yasuda MA. Co-infection Trypanosoma cruzi /HIV: systematic review (1980-2010). Rev Soc Bras Med Trop. 2011 Nov-Dec;44(6):762-70.) (5252. Ministério da Saúde (BR). Recomendações para diagnóstico, tratamento e acompanhamento da co-infecção Trypanosoma cruzi: vírus da imunodeficiência humana. Rev Soc Bras Med Trop. 2006 jul-ago;39(4):392-415. . Considering that in 2014 there were approximately 734,000 people living with HIV (without AIDS) in Brazil, corresponding to a prevalence in the general population of 0.4%, 5151. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Departamento de DST, Aids e Hepatites Virais. Protocolo clínico e diretrizes terapêuticas para manejo da infecção pelo HIV em adultos. Brasília: Ministério da Saúde; 2014.) there would be an estimated 9,542 cases of T. cruzi /HIV co-infection. On the other hand, since the beginning of the AIDS epidemic in Brazil to June 2014, total of 757,042 AIDS cases were registered in the Brazilian Notifiable Diseases Information System [Sistema de Informação de Agravos de Notificação] (SINAN) (5050. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Departamento de DST, Aids e Hepatites Virais. Bol Epidemiol. 2014;3(1):1-80 ; it is estimated that more than 9,842 of these would be co-infected. Therefore, it is estimated that there are 19,384 cases of T. cruzi /HIV-AIDS co-infection in Brazil, based on data up to June 2014, stressing that many of these cases probably died, are undiagnosed, or unrecognized as having co-infection. In Brazil, anti- T. cruzi antibodies testing is recommended for HIV-infected patients, taking into account their epidemiological risks 5151. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Departamento de DST, Aids e Hepatites Virais. Protocolo clínico e diretrizes terapêuticas para manejo da infecção pelo HIV em adultos. Brasília: Ministério da Saúde; 2014. . It is worth noting that, for surveillance purposes, since 2004 reactivation of Chagas disease has been added to the list of AIDS-defining illnesses, and has been an important marker, from the definitive diagnosis of Chagas myocarditis and meningoencephalitis 3939. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Guia de Vigilância em Saúde. Brasília: Ministério da Saúde; 2014.) (4949. Almeida EA, Ramos Júnior AN, Correia D, Shikanai-Yasuda MA. Co-infection Trypanosoma cruzi /HIV: systematic review (1980-2010). Rev Soc Bras Med Trop. 2011 Nov-Dec;44(6):762-70.) (5252. Ministério da Saúde (BR). Recomendações para diagnóstico, tratamento e acompanhamento da co-infecção Trypanosoma cruzi: vírus da imunodeficiência humana. Rev Soc Bras Med Trop. 2006 jul-ago;39(4):392-415.) (5454. Ramos Júnior AN. Inclusão da reativação da doença de Chagas como uma condição definidora de AIDS para fins de vigilância epidemiológica no Brasil. Rev Soc Bras Med Trop. 2004 mar-abr;37(2):192-3. . Generally, such HIV co-infection occurs through sexual transmission in individuals previously infected with T. cruzi . A progressive decrease in the number of co-infection cases is expected in Brazil, since Chagas is gradually becoming restricted to higher age groups.
In addition to the above co-infection, generally, despite the high morbidity and mortality of Chagas disease in Brazil, data relating to the specific epidemiological surveillance of human cases of the disease does not allow estimation of its magnitude, since it is only mandatory to report cases in the acute phase to SINAN. Furthermore, it is estimated that only 10-20% of acute Chagas disease cases are actually reported.
In the period 2000-2013 (data updated in May 2014), 1,570 cases of acute Chagas disease were reported ̶ an average of 112 cases per year. This figure was derived from records from most Brazilian states, with the exception of Mato Grosso do Sul and Federal District in the Central-West region and Paraná in the South. Most of these cases (1,430; 91.1%) were concentrated in the North, followed by the Northeast (73; 4.7%), South (28; 0.2%), Central-West (27; 1.8%) and Southeast (12; 0.8%). It is noteworthy that the State of Pará was responsible for 75% of all cases in the country and 82% of cases in the Northern region 5555. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Doença de Chagas aguda no Brasil: série histórica de 2000 a 2013. Bol Epidemiol. 2015;46(21):1-9. .
With regard to the municipality of residence, there were 163 reported cases of acute Chagas disease: 97 (60%) from the Northern region, 37 (23%) from the Northeast, 14 (9%) from the Central-West and, in smaller proportions, 9 (6%) and 6 (4%) from municipalities of the Southern and Southeastern regions, respectively 5555. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Doença de Chagas aguda no Brasil: série histórica de 2000 a 2013. Bol Epidemiol. 2015;46(21):1-9. . This notification pattern is quite distinct from that of the 1990s 44. Coura JR, Dias JC. Epidemiology, control and surveillance of Chagas disease: 100 years after its discovery. Mem Inst Oswaldo Cruz. 2009 Jul;104 Suppl 1:31-40.) (1818. Dias JCP. Southern Cone Initiative for the elimination of domestic populations of Triatoma infestans and the interruption of transfusion Chagas disease: historical aspects, present situation, and perspectives. Mem Inst Oswaldo Cruz. 2007 Oct;102 Suppl 1:11-8.) (1919. Prata A. Clinical and epidemiological aspects of Chagas disease. Lancet Infect Dis. 2001 Sep;1(2):92-100. , but still demarcates operational contexts of epidemiological silence in some areas.
Mortality
The burden of mortality related to Chagas disease in Brazil remains high, despite the introduction of control measures 33. Dias JC, Silveira AC, Schofield CJ. The impact of Chagas disease control in Latin America: a review. Mem Inst Oswaldo Cruz. 2002 Jul;97(5):603-12.) (2929. Barreto ML, Teixeira MG, Bastos FI, Ximenes RAA, Barata RB, Rodrigues LC. Successes and failures in the control of infectious diseases in Brazil: social and environmental context, policies, interventions, and research needs. Lancet. 2011 May;377(9780):1877-89.) (3535. Martins-Melo FR, Ramos Júnior AN, Alencar CH, Heukelbach J. Prevalence of Chagas disease in Brazil: a systematic review and meta-analysis. Acta Trop. 2014 Feb;130:167-74.) (5656. Martins-Melo FR, Ramos Júnior AN, Alencar CH, Heukelbach J. Multiple causes of death related to Chagas' disease in Brazil, 1999 to 2007. Rev Soc Bras Med Trop. 2012 Sep-Oct;45(5):591-6.) (5757. Martins-Melo FR, Ramos Júnior AN, Alencar CH, Heukelbach J. Mortality due to Chagas disease in Brazil from 1979 to 2009: trends and regional differences. J Infect Dev Ctries. 2012 Nov;6(11):817-24. . A study conducted to quantify the mortality pattern of Chagas disease in the period 1981-1998 found that out of 68,936 deaths in Brazilian individuals with a known birthplace, 32,369 (32%) deaths occurred in states other than the deceased's birthplace, varying from 0.3% in Rio Grande do Sul to 100% in Roraima and Amapá. Most (67%) of these deaths in migrants occurred in individuals of Minas Gerais (51%) and Bahia (16%). Mortality rates of residents within the period showed a consistent decline in the Southeast, South, and Central-West, but not in the Northern and Northeastern regions, where the median age at death was comparatively low 5858. Drumond JAG, Marcopito LF. Internal migration and distribution of Chagas disease mortality, Brazil, 1981-1998. Cad Saude Publica. 2006 Oct;22(10):2131-40. .
Later, from 1999 to 2007, Chagas disease was recorded as the cause of death on 53,924 (0.6%) of approximately 9.0 million death certificates from the Brazilian Mortality Information System [ Sistema de Informações sobre Mortalidade (SIM)]: for 44,537 (82.6%) it was recorded as the underlying cause and for 9,387 (17.4%) as an associated cause 5959. Martins-Melo FR, Alencar CH, Ramos Júnior AN, Heukelbach J. Epidemiology of mortality related to Chagas' disease in Brazil, 1999-2007. PLoS Negl Trop Dis. 2012 Feb;6(2):e1508. . Acute Chagas disease was recorded in the statement in 2.8% of deaths. The average number of deaths per year was 5,992, with an average standardized mortality rate of 3.36/100,000 inhabitants/year, verifying a gradual reduction in the specific mortality rate of 26.4%. It is emphasized the decreasing tendency of a standardized mortality rate, especially in the Central-West region (-37.9%). The proportional mortality (for numerous reasons) was 0.6%, highest in the Central-West region (2.17%) where there was a reduction of 19.5%, with a significant decreasing tendency. However, in this study, there was a considerable increase of 38.5% in the Northeast region, with an increasing tendency. Risk factors identified in the bivariate analysis were older age groups and residence in the Central-West region. In the multivariate analysis, however, age over 30 years and residence in the States of Minas Gerais and Goiás and in the Federal District were identified as risk factors 5959. Martins-Melo FR, Alencar CH, Ramos Júnior AN, Heukelbach J. Epidemiology of mortality related to Chagas' disease in Brazil, 1999-2007. PLoS Negl Trop Dis. 2012 Feb;6(2):e1508. . It should be noted that more than 40% of expected deaths associated with Chagas disease in Latin America occurred in Brazil 5656. Martins-Melo FR, Ramos Júnior AN, Alencar CH, Heukelbach J. Multiple causes of death related to Chagas' disease in Brazil, 1999 to 2007. Rev Soc Bras Med Trop. 2012 Sep-Oct;45(5):591-6.) (5959. Martins-Melo FR, Alencar CH, Ramos Júnior AN, Heukelbach J. Epidemiology of mortality related to Chagas' disease in Brazil, 1999-2007. PLoS Negl Trop Dis. 2012 Feb;6(2):e1508. .
Another study of the same Brazilian population base examined 53,930 deaths, recorded between 1999 and 2007, where Chagas disease was mentioned as a cause of death. When analyzed as the underlying cause, Chagas disease was the fourth leading cause of death (10.8%) among all infectious diseases and parasites 5656. Martins-Melo FR, Ramos Júnior AN, Alencar CH, Heukelbach J. Multiple causes of death related to Chagas' disease in Brazil, 1999 to 2007. Rev Soc Bras Med Trop. 2012 Sep-Oct;45(5):591-6. . Of the 53,930 recorded deaths, 37,800 (84.9%) were related to cardiac forms and 4,208 (9.4%) to digestive forms of Chagas disease. Acute cardiac involvement was noted in 1,097 (2.5%) deaths; other organ involvement in 1,157 (2.6%); and finally, other clinical presentations of the disease (central nervous system involvement and no cardiac involvement in the acute phase) in 281 (0.6%). The associated causes of death with Chagas disease as the underlying cause included diseases of the digestive, circulatory, and respiratory systems. Direct complications of heart disease, especially conduction disturbances/arrhythmias and heart failure were identified in more than 35% of deaths, followed by shock (15%). Among the basic causes of death in which Chagas disease was mentioned as an associated cause, the most common diseases were of the circulatory and respiratory systems (59%), followed by cancer (11.6%) and digestive system diseases (10.6%). Cerebrovascular disease, ischemic heart disease, and hypertensive diseases were the main underlying causes of death in cases where Chagas disease had been identified as an associated cause. Chronic lung diseases and, in particular, chronic obstructive pulmonary disease also represented particularly important causes of death 5656. Martins-Melo FR, Ramos Júnior AN, Alencar CH, Heukelbach J. Multiple causes of death related to Chagas' disease in Brazil, 1999 to 2007. Rev Soc Bras Med Trop. 2012 Sep-Oct;45(5):591-6. .
In an additional study from this same database, using different spatial analytical approaches, we identified a cluster of high mortality risk for Chagas disease involving nine States of Brazil's Central-West region: Almost all municipalities from the States of Goiás and Minas Gerais, Federal District and some municipalities from the States of São Paulo, Mato Grosso, Mato Grosso do Sul, Tocantins, Piauí and Bahia. This area was indicated by the authors as a priority for integrated planning of actions to strengthen the network of attention focused on Chagas disease 6060. Martins-Melo FR, Ramos Júnior AN, Alencar CH, Lange W, Heukelbach J. Mortality of Chagas' disease in Brazil: spatial patterns and definition of high-risk areas. Trop Med Int Health. 2012 Sep;17(9):1066-75. .
A study examining regional patterns and tendencies in Brazil over a longer time period (1979-2009), found that of 27,560,043 deaths analyzed, Chagas disease was the underlying cause in 172,066 deaths 5757. Martins-Melo FR, Ramos Júnior AN, Alencar CH, Heukelbach J. Mortality due to Chagas disease in Brazil from 1979 to 2009: trends and regional differences. J Infect Dev Ctries. 2012 Nov;6(11):817-24. . The proportional mortality of Chagas disease was 0.62% with a crude specific death rate of 3.61 deaths/100,000 inhabitants/year and a specific mortality rate adjusted for age of 5.19 deaths/100,000 inhabitants/year. Nationally, there was significant reduction over the period, but with important inter-regional differences; there was significant reduction in the Southeast, South and Central-West regions, but significant growth in the Northeast and Northern regions 5757. Martins-Melo FR, Ramos Júnior AN, Alencar CH, Heukelbach J. Mortality due to Chagas disease in Brazil from 1979 to 2009: trends and regional differences. J Infect Dev Ctries. 2012 Nov;6(11):817-24. .
Another historical series, from 1980 to 2008, found that the proportion of deaths attributed to Chagas disease as the underlying cause remained stable in the period 2929. Barreto ML, Teixeira MG, Bastos FI, Ximenes RAA, Barata RB, Rodrigues LC. Successes and failures in the control of infectious diseases in Brazil: social and environmental context, policies, interventions, and research needs. Lancet. 2011 May;377(9780):1877-89. .
In the State of São Paulo from 1985 to 2006, there were 40,002 Chagas disease-related deaths. Chagas disease was an underlying cause in 34,917 (87.3%) and an associated cause in 5,085 (12.7%). The mortality rate, according to the underlying cause, declined by 56.1%, but remained stable in terms of associated cause of death. There were 44.5% more deaths in men than in women, and 83.5% of deaths occurred in people over 45 years old. The main causes associated with those who had Chagas disease as underlying cause of death were: direct complications of cardiac impairment (conduction disorders, arrhythmias and heart failure). For Chagas disease as an associated cause, the most important underlying causes were ischemic heart disease, cerebrovascular disease, and neoplasms 6161. Santo AH. Tendência da mortalidade relacionada à doença de Chagas, Estado de São Paulo, Brasil, 1985 a 2006: estudo usando causas múltiplas de morte. Rev Panam Salud Publica. 2009 Oct;26(4):299-309. .
On the other hand, from the national database on mortality related to Chagas disease as the underlying cause, from 2000 to 2010, it was found that most deaths (85.9%) occurred in males older than 60 years. These deaths were mainly caused by cardiac involvement, and the mortality rate of this clinical form of the disease decreased in all regions except in the North, where there was an increase of 1.6%. The Northeastern region had the lowest reduction, while the Central-West had the largest. It is noteworthy that the mortality rate related to the digestive form increased in all regions 6262. Nóbrega AA, Araújo WN, Vasconcelos AM. Mortality due to Chagas disease in Brazil according to a specific cause. Am J Trop Med Hyg. 2014 Sep;91(3):528-33. .
A pattern of aging and an expansion of ages at highest age ranges for the population affected by Chagas disease has been verified, not only by previous studies of mortality, but also by other population-based studies based on cases of cohorts followed through long periods of time 6363. Alves RM, Thomaz RP, Almeida EA, Wanderley JS, Guariento ME. Chagas' disease and ageing: the coexistence of other chronic diseases with Chagas' disease in elderly patients. Rev Soc Bras Med Trop. 2009 Nov-Dec;42(6):622-8.) (6464. Lima-Costa MF, Peixoto SV, Ribeiro AL. Chagas disease and mortality in old age as an emerging issue: 10 year follow-up of the Bambuí population-based cohort study (Brazil). Int J Cardiol. 2010 Nov;145(2):362-3. .
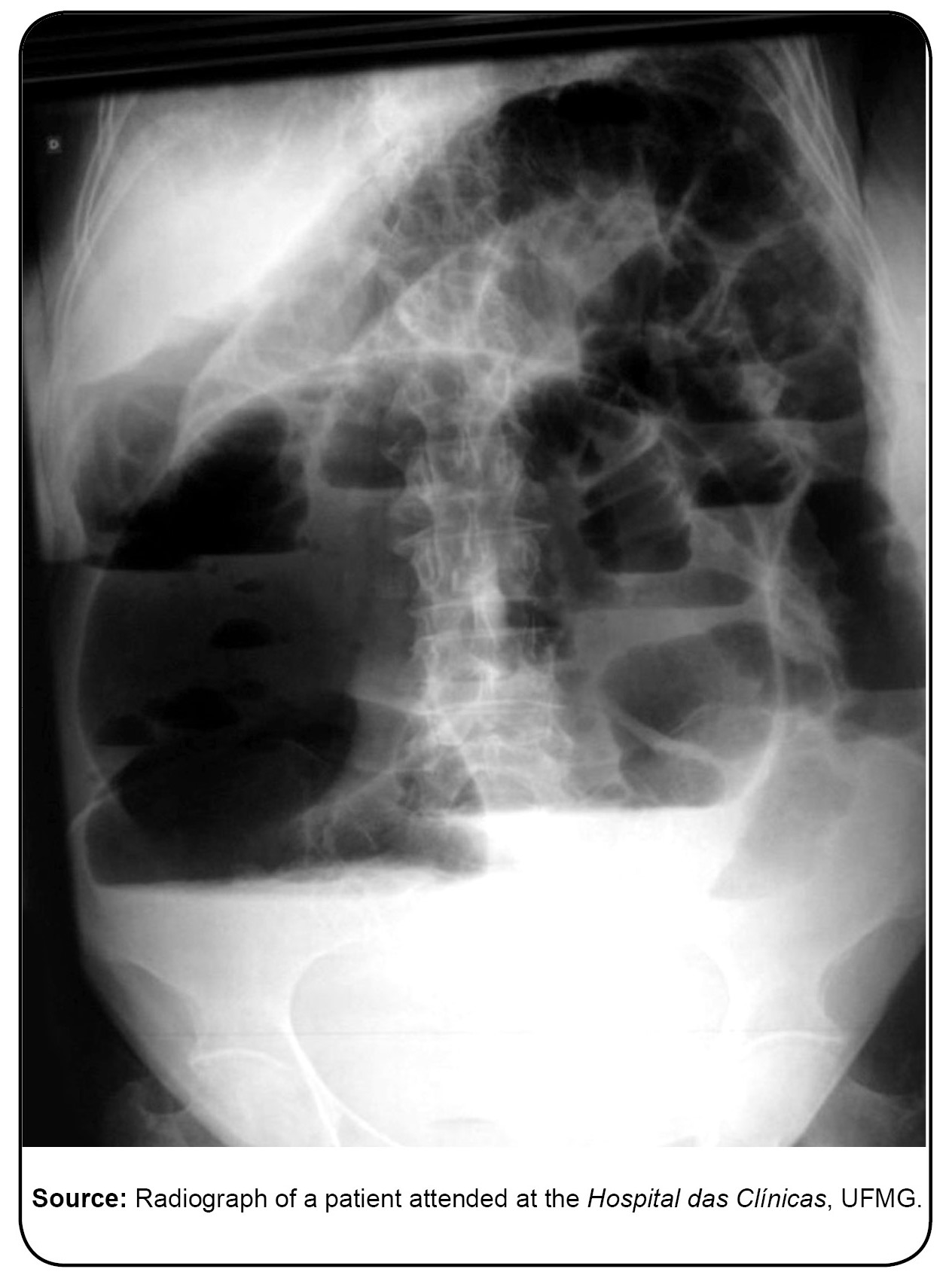
Data from the Ministry of Health on deaths from acute Chagas disease indicate that in the period 2005-2013, the national average annual mortality rate over the 14 years was 2.7% (37.9/14). In 2005, a high (20%) mortality rate was recorded. This coincided with an acute Chagas outbreak by oral transmission in Santa Catarina, at which time little was known about the etiology and management of the disease by this form of transmission. In 2006, the mortality remained high (5.9%), with subsequent reduction that has remained relatively constant (average of 1.7% from 2007 to 2013), ranging from 0.5% in 2009 to 4.7% in 2011. It is worth noting that the lack of disease suspicion and the delay in establishing the diagnosis may worsen the clinical picture and the evolution of the disease, and may affect the prognosis. For example, deaths occurred due to lack of timely intervention 5555. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Doença de Chagas aguda no Brasil: série histórica de 2000 a 2013. Bol Epidemiol. 2015;46(21):1-9. , particularly in terms of the cardiovascular forms of the disease.
In terms of mortality associated with co-infection of T. cruzi /HIV, approximately 9 million deaths were studied in the period 1999-2007. Seventy-four deaths were identified as related to T. cruzi /HIV co-infection; in 57 (77%) the underlying cause was AIDS, and in 13 (17.6%) the underlying cause was Chagas disease 5353. Martins-Melo FR, Ramos Júnior AN, Alencar CH, Heukelbach J. Mortality related to Chagas disease and HIV/AIDS coinfection in Brazil. J Trop Med. 2012;2012:534649. . Co-infection was associated with male sex (51.4%), Caucasian ethnicity (50%), age between 40 and 49 years (29.7%), and place of residence in the Southeastern region (75.7%). The average age at death was significantly lower among co-infected (47.1 years) individuals than among those without co-infection (64.1 years) 5353. Martins-Melo FR, Ramos Júnior AN, Alencar CH, Heukelbach J. Mortality related to Chagas disease and HIV/AIDS coinfection in Brazil. J Trop Med. 2012;2012:534649. . These data probably indicate a underestimation the real impact of co-infection on mortality.
Epidemiological profile according to transmission modes
In more recent years (SINAN, 2000-2013), with the operational limitations as previously indicated, it was found that oral transmission was the most frequent mode of transmission every year (1,081; 68.9%), followed by vectorial transmission (100; 6.4%) 5555. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Doença de Chagas aguda no Brasil: série histórica de 2000 a 2013. Bol Epidemiol. 2015;46(21):1-9. . The persistence of the latter has been observed since 2006, the year that Brazil obtained certification for interrupting transmission of T. cruzi by Triatoma infestans. Transmission still occurs, albeit to a lesser extent, probably due to reduced visibility of these cases. Comparatively, WHO estimates, based on data from 2010, indicated that a total of 46 cases of Chagas disease per year in Brazil originated from vectorial transmission 55. World Health Organization. Chagas disease in Latin America: an epidemiological update based on 2010 estimates. Wkly Epidemiol Rec. 2015 Feb;90(6):33-44. . Together, these data reinforce the likely underestimation of cases relating to this mode of transmission, both nationally and internationally. The mode of transmission was not identified in 372 (23.7%) reported cases; 93% (306 cases) of these were from the State of Pará. In 11 (0.7%) cases other modes of transmission (transfusion or accidental) were identified, and there were 6 (0.4%) cases of vertical transmission 5555. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Doença de Chagas aguda no Brasil: série histórica de 2000 a 2013. Bol Epidemiol. 2015;46(21):1-9. . Underreporting is also clear in the later mode of transmission.
This change in the epidemiological pattern of transmission of Chagas disease is the combined result of the control measures undertaken over the last four decades and major environmental, demographic, economic, and social changes, and the concentration of the population in urban areas 44. Coura JR, Dias JC. Epidemiology, control and surveillance of Chagas disease: 100 years after its discovery. Mem Inst Oswaldo Cruz. 2009 Jul;104 Suppl 1:31-40.) (1515. Ramos Júnior AN, Carvalho DM. Chagas' disease: past, present and future. Cad Saude Colet. 2009;17(4):787-94.) (3333. Silveira AC, Silva GR, Prata A. O Inquérito de soroprevalência da infecção chagásica humana (1975-1980). Rev Soc Bras Med Trop. 2011;44 supl 2:33-9.) (6565. Silveira AC. Os novos desafios e perspectivas futuras do controle. Rev Soc Bras Med Trop. 2011;44 Supl 2:122-4. . Thus, mechanisms directly related to the enzootic cycle of infection, such as extradomiciliary vector transmission or visitation of wild vectors to households, in addition to oral transmission (also mediated by vectors, in most cases), have to have relevance as main modes of T. cruzi transmission to human populations 2222. Silveira AC, Dias JCP. O controle da transmissão vetorial. Rev Soc Bras Med Trop. 2011;44 supl 2:52-63.) (2727. Messenger LA, Miles MA, Bern C. Between a bug and a hard place: Trypanosoma cruzi genetic diversity and the clinical outcomes of Chagas disease. Expert Rev Anti Infect Ther. 2015 Aug;13(8):995-1029.) (6565. Silveira AC. Os novos desafios e perspectivas futuras do controle. Rev Soc Bras Med Trop. 2011;44 Supl 2:122-4. .
Vector transmission
In 1950 and 1951, the first Prophylaxis Campaign for Chagas disease was developed in Brazil, led by the then National Service for Malaria, reaching 74 municipalities on the border of the States of Minas Gerais and São Paulo with chemical vector control activities. By 1975, control activities were conducted intermittently, with varying territorial scope. The area at risk of vector transmission of Chagas disease in Brazil in the late 1970s included 18 states, with more than 2,200 municipalities, which proved the presence of domiciled triatomine. Notably, the Amazon region was excluded from this risk area 44. Coura JR, Dias JC. Epidemiology, control and surveillance of Chagas disease: 100 years after its discovery. Mem Inst Oswaldo Cruz. 2009 Jul;104 Suppl 1:31-40.) (1919. Prata A. Clinical and epidemiological aspects of Chagas disease. Lancet Infect Dis. 2001 Sep;1(2):92-100.) (2020. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Consenso Brasileiro em Doença de Chagas. Rev Soc Bras Med Trop. 2005;38 Supl 3:1-29.) (2222. Silveira AC, Dias JCP. O controle da transmissão vetorial. Rev Soc Bras Med Trop. 2011;44 supl 2:52-63.) (6666. Passos ADC, Silveira AC. Síntese dos resultados dos inquéritos nacionais. Rev Soc Bras Med Trop. 2011;44 supl 2:47-50. .
Since then, with the intensification of control measures, the vectorial transmission of T. cruzi in Brazil has shown marked and systematic reduction. This was achieved by the development of systemized actions of chemical control of domiciled vector populations, introduced in 1975, with a range of coverage of the endemic area in 1983 2020. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Consenso Brasileiro em Doença de Chagas. Rev Soc Bras Med Trop. 2005;38 Supl 3:1-29.) (2222. Silveira AC, Dias JCP. O controle da transmissão vetorial. Rev Soc Bras Med Trop. 2011;44 supl 2:52-63.) (6666. Passos ADC, Silveira AC. Síntese dos resultados dos inquéritos nacionais. Rev Soc Bras Med Trop. 2011;44 supl 2:47-50. . These actions have taken place on a regular basis since then. However, their reach has progressively declined over the past two decades due to technical and political prioritization of changes, and due to political-institutional reorganization in the country 2020. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Consenso Brasileiro em Doença de Chagas. Rev Soc Bras Med Trop. 2005;38 Supl 3:1-29.) (2222. Silveira AC, Dias JCP. O controle da transmissão vetorial. Rev Soc Bras Med Trop. 2011;44 supl 2:52-63. .
This process, in an integrated manner, culminated with the Certification of Interruption of Chagas Disease Transmission of the main domiciled vector, T. infestans , granted in 2006 by Pan American Health Organization/World Health Organization (PAHO/WHO) to countries of the Southern Cone Initiative 1818. Dias JCP. Southern Cone Initiative for the elimination of domestic populations of Triatoma infestans and the interruption of transfusion Chagas disease: historical aspects, present situation, and perspectives. Mem Inst Oswaldo Cruz. 2007 Oct;102 Suppl 1:11-8.) (2222. Silveira AC, Dias JCP. O controle da transmissão vetorial. Rev Soc Bras Med Trop. 2011;44 supl 2:52-63.) (3939. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Guia de Vigilância em Saúde. Brasília: Ministério da Saúde; 2014. . The control of vector-borne transmission had considerable impact in relation to congenital and transfusion transmissions 44. Coura JR, Dias JC. Epidemiology, control and surveillance of Chagas disease: 100 years after its discovery. Mem Inst Oswaldo Cruz. 2009 Jul;104 Suppl 1:31-40.) (1010. Dias JCP. Human chagas disease and migration in the context of globalization: some particular aspects. J Trop Med. 2013:789758.) (1818. Dias JCP. Southern Cone Initiative for the elimination of domestic populations of Triatoma infestans and the interruption of transfusion Chagas disease: historical aspects, present situation, and perspectives. Mem Inst Oswaldo Cruz. 2007 Oct;102 Suppl 1:11-8.) (2323. Coura JR. The main sceneries of Chagas disease transmission. The vectors, blood and oral transmissions: a comprehensive review. Mem Inst Oswaldo Cruz. 2015 May;110(3):277-82.) (2929. Barreto ML, Teixeira MG, Bastos FI, Ximenes RAA, Barata RB, Rodrigues LC. Successes and failures in the control of infectious diseases in Brazil: social and environmental context, policies, interventions, and research needs. Lancet. 2011 May;377(9780):1877-89. . Despite the advances, the risk of vectorial transmission of Chagas disease persists. This has been evaluated from different perspectives 2222. Silveira AC, Dias JCP. O controle da transmissão vetorial. Rev Soc Bras Med Trop. 2011;44 supl 2:52-63. , including the existence of indigenous species of triatomine with high potential for colonization; the presence of T. cruzi reservoirs and increasing proximity of human populations to these environments; and the persistence of residual foci of T. infestans , still existing in some municipalities of the States of Bahia and Rio Grande do Sul 2222. Silveira AC, Dias JCP. O controle da transmissão vetorial. Rev Soc Bras Med Trop. 2011;44 supl 2:52-63.) (2323. Coura JR. The main sceneries of Chagas disease transmission. The vectors, blood and oral transmissions: a comprehensive review. Mem Inst Oswaldo Cruz. 2015 May;110(3):277-82.) (3939. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Guia de Vigilância em Saúde. Brasília: Ministério da Saúde; 2014.) (6767. Vinhaes MC, Oliveira SV, Reis PO, Lacerda Sousa AC, Silva RA, Obara MT, et al. Assessing the vulnerability of Brazilian municipalities to the vectorial transmission of Trypanosoma cruzi using multi-criteria decision analysis. Acta Trop. 2014 Sep;137:105-10. . The impacts of the certification process achieved by Brazil in 2006 in relation to T. infestans generated concern about the demobilization of society in general and the imprecise way information was obtained and disclosed to society 6868. Ramos Júnior AN, Carvalho DM. Os diferentes significados da certificação conferida ao Brasil como estando livre da doença de Chagas. Cad Saude Publica. 2001 nov-dez;17(6):1403-12. .
Furthermore, additional studies and analyses have sought to evaluate further the operational processes involved in different strategies to control/eliminate of indigenous vector transmission, including the certification model and potential alternatives 6969. Abad-Franch F, Diotaiuti L, Gurgel-Gonçalves R, Gürtler RE. Certifying the interruption of Chagas disease transmission by native vectors: cui bono? Mem Inst Oswaldo Cruz. 2013 Apr;108(2):251-4.) (7070. Valença-Barbosa C, Lima MM, Sarquis O, Bezerra CM, Abad-Franch F. Modeling disease vector occurrence when detection is imperfect II: drivers of site-occupancy by synanthropic Triatoma brasiliensis in the Brazilian northeast. PLoS Negl Trop Dis. 2014 May;8(5):e2861.) (7171. Abad-Franch F, Valença-Barbosa C, Sarquis O, Lima MM. All that glisters is not gold: sampling-process uncertainty in disease-vector surveys with false-negative and false-positive detections. PLoS Negl Trop Dis. 2014 Sep;8(9):e3187.) (7272. Salvatella R, Irabedra P, Castellanos LG. Interruption of vector transmission by native vectors and "the art of the possible". Mem Inst Oswaldo Cruz. 2014 Feb;109(1):122-30. . Data from the Ministry of Health on entomological surveillance (passive or active) specific to the period 2007-2011 ( Epidemiological Bulletin of Chagas Disease 2015 ) indicate a record catch of >770,000 triatomine in local homes and peridomestic contexts. Like human cases, considering the low coverage of entomological surveillance actions, this record probably comprises less than 10% of triatomine in or around residences. Also, according to data from the Ministry of Health, among the 62 species distributed in intradomicile and peridomicile spaces in Brazil, the following stand out as species of epidemiological relevance: Panstrongilus geniculatus, Panstrongilus lutzi, Panstrongilus megistus, Rhodnius nasutus, Rhodnius neglectus, Rhodnius robustus, Rhodnius pictipes, Triatoma infestans, Triatoma brasiliensis, Triatoma maculata, Triatoma pseudomaculata, Triatoma rubrovaria, Triatoma rubrofasciata, Triatoma sordida and Triatoma vitticeps.
According to the data from the Epidemiological Bulletin of the Ministry of Health on Chagas Disease , 591,360 (76.8%) of the captured triatomine were examined for infection with T. cruzi ; the natural infection rate was an estimated 2.7% (15,967 triatomine). The Triatoma vitticeps (52%), R hodnius robustus (33.3%), and Panstrongylus lutzi (29.4%) species had the highest rates of natural infection. According to the data from the Ministry of Health, T. vitticeps appears more frequently in the Southeastern States (Minas Gerais and Espírito Santo), R. robustus in the Northern region (Tocantins, Amazonas, Acre, and Rondônia), and P. lutzi in the Northeastern region (Piauí, Bahia, Sergipe, Alagoas, Paraiba, Rio Grande do Norte, Ceará and Pernambuco) 5555. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Doença de Chagas aguda no Brasil: série histórica de 2000 a 2013. Bol Epidemiol. 2015;46(21):1-9. .
In 2012, the investigation of two cases of acute Chagas disease recorded by vector transmission showed the involvement of wild species with high rates of natural infection in municipalities that were considered non-endemic for Chagas disease. One case occurred in the city of Ibitirama (Espírito Santo), where infected T. vitticeps adults are often found by local residents. The other case occurred in Mangaratiba (Rio de Janeiro), involving Triatoma tibiamaculata found to be infected with T. cruzi . These episodes reflect a nationwide reality; that parasite transmission cycles exist in wild environments close to human dwellings, which favors transmission 5555. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Doença de Chagas aguda no Brasil: série histórica de 2000 a 2013. Bol Epidemiol. 2015;46(21):1-9. .
For the period 2007-2011, the persistence of Triatoma infestans foci was identified in four municipalities of the State of Bahia (Itaguaçu da Bahia, Ibipeba, Novo Horizonte, and Tremedal) and 12 municipalities of Rio Grande do Sul (Ajuricaba Alegria, Coronel Barros, Doutor Mauricio Cardoso, Giruá, Humaitá, Ijuí, Independência, Porto Mauá, Salvador das Missões, Santo Cristo, and São José do Inhacorá) 5555. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Doença de Chagas aguda no Brasil: série histórica de 2000 a 2013. Bol Epidemiol. 2015;46(21):1-9. . Integrated actions of vector control mobilizing Federal, State, and Municipal governments have advanced towards the control of this triatomine.
Currently, the risk related to the transmission of T. cruzi in Brazil depends mainly on: I) the persistence of residual foci of T. infestans , with episodes found in some states, such as Bahia and Rio Grande do Sul; II) the existence of large number of species that were confirmed as indigenous or acting as potential vectors in the country, even though in some cases resident populations have been greatly reduced; III) the emergence of new species at risk of domiciliation ( T. rubrovaria, P. lutzi ); IV) the existence of endemic transmission in the Amazon Region, with still poorly characterized mechanisms of transmission such as domiciliary vectorial without colonization, extradomiciliary vectorial, and oral; and V) the occurrence of outbreaks and micro-epidemics of orally transmitted of T. cruzi infection 2020. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Consenso Brasileiro em Doença de Chagas. Rev Soc Bras Med Trop. 2005;38 Supl 3:1-29.) (3333. Silveira AC, Silva GR, Prata A. O Inquérito de soroprevalência da infecção chagásica humana (1975-1980). Rev Soc Bras Med Trop. 2011;44 supl 2:33-9.) (6565. Silveira AC. Os novos desafios e perspectivas futuras do controle. Rev Soc Bras Med Trop. 2011;44 Supl 2:122-4.) (6767. Vinhaes MC, Oliveira SV, Reis PO, Lacerda Sousa AC, Silva RA, Obara MT, et al. Assessing the vulnerability of Brazilian municipalities to the vectorial transmission of Trypanosoma cruzi using multi-criteria decision analysis. Acta Trop. 2014 Sep;137:105-10. .
The major change in the epidemiological situation of the disease in the country has generated the need for a dynamic review of the control strategies adopted 44. Coura JR, Dias JC. Epidemiology, control and surveillance of Chagas disease: 100 years after its discovery. Mem Inst Oswaldo Cruz. 2009 Jul;104 Suppl 1:31-40.) (2020. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Consenso Brasileiro em Doença de Chagas. Rev Soc Bras Med Trop. 2005;38 Supl 3:1-29.) (2323. Coura JR. The main sceneries of Chagas disease transmission. The vectors, blood and oral transmissions: a comprehensive review. Mem Inst Oswaldo Cruz. 2015 May;110(3):277-82.) (3333. Silveira AC, Silva GR, Prata A. O Inquérito de soroprevalência da infecção chagásica humana (1975-1980). Rev Soc Bras Med Trop. 2011;44 supl 2:33-9.) (6565. Silveira AC. Os novos desafios e perspectivas futuras do controle. Rev Soc Bras Med Trop. 2011;44 Supl 2:122-4.) (6767. Vinhaes MC, Oliveira SV, Reis PO, Lacerda Sousa AC, Silva RA, Obara MT, et al. Assessing the vulnerability of Brazilian municipalities to the vectorial transmission of Trypanosoma cruzi using multi-criteria decision analysis. Acta Trop. 2014 Sep;137:105-10. . From this perspective, identifying areas with greater vulnerability to the synanthropic occurrence of triatomine has been important tool to reorient prevention, control, and surveillance actions 3939. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Guia de Vigilância em Saúde. Brasília: Ministério da Saúde; 2014.) (6767. Vinhaes MC, Oliveira SV, Reis PO, Lacerda Sousa AC, Silva RA, Obara MT, et al. Assessing the vulnerability of Brazilian municipalities to the vectorial transmission of Trypanosoma cruzi using multi-criteria decision analysis. Acta Trop. 2014 Sep;137:105-10. , and to optimize the use of available program resources 2020. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Consenso Brasileiro em Doença de Chagas. Rev Soc Bras Med Trop. 2005;38 Supl 3:1-29. . On the other hand, risk stratification associated with vector transmission has been proposed as an important strategy for the guidance and support of control measures in the country and has been incorporated, in different ways, by the control programs of states and municipalities 44. Coura JR, Dias JC. Epidemiology, control and surveillance of Chagas disease: 100 years after its discovery. Mem Inst Oswaldo Cruz. 2009 Jul;104 Suppl 1:31-40.) (2020. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Consenso Brasileiro em Doença de Chagas. Rev Soc Bras Med Trop. 2005;38 Supl 3:1-29.) (2222. Silveira AC, Dias JCP. O controle da transmissão vetorial. Rev Soc Bras Med Trop. 2011;44 supl 2:52-63.) (3939. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Guia de Vigilância em Saúde. Brasília: Ministério da Saúde; 2014.) (6565. Silveira AC. Os novos desafios e perspectivas futuras do controle. Rev Soc Bras Med Trop. 2011;44 Supl 2:122-4. . This stratification is based on three different scenarios or eco-epidemiological spaces of vector-borne transmission of T. cruzi2222. Silveira AC, Dias JCP. O controle da transmissão vetorial. Rev Soc Bras Med Trop. 2011;44 supl 2:52-63.:
-
an area first considered endemic, with registered domiciliary vectorial transmission, which was submitted for an extended period to intensive chemical control operations, resulting in the almost complete elimination of the main vector ( T. infestans ), and the risk being currently limited to a more or less focal transmission, mainly by native species. This scenario includes the States of Alagoas, Bahia, Ceará, Goiás, Mato Grosso do Sul, Minas Gerais, Paraíba, Pernambuco, Paraná, Piauí, Rio Grande do Norte, Rio Grande do Sul, São Paulo and Sergipe, as well as the Federal District;
-
an area previously considered harmless for human Chagas disease, in which the disease is now transmitted through mechanisms that before were considered extraordinary or improbable and are still poorly characterized but able to maintain endemic transmission of relatively low intensity to sustain endemic transmission (oral, extradomiciliary and domiciliary without vector colonization). This scenario includes the region covering the States of Acre, Amazonas, Amapá, Rondônia, Roraima and Pará; and a transition area between the two previous scenarios, in which both coexist. This scenario includes the States of Maranhão, Mato Grosso, and Tocantins.
As previously discussed, the States of Santa Catarina, Rio de Janeiro, and Espírito Santo, although not covered by the proposal of the previous risk stratification, given the historical patterns observed from the entomological surveillance of the states, have registered autochthonous cases of Chagas disease 3939. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Guia de Vigilância em Saúde. Brasília: Ministério da Saúde; 2014.) (5555. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Doença de Chagas aguda no Brasil: série histórica de 2000 a 2013. Bol Epidemiol. 2015;46(21):1-9.) (7373. Maegawa FA, Damerau EF, Beltrame-Botelho IT, Lopes A, Emmanuelle-Machado P, Steindel M, et al. Autochthonous Chagas' disease in Santa Catarina State, Brazil: report of the first case of digestive tract involvement. Rev Soc Bras Med Trop. 2003 Sep-Oct;36(5):609-12.) (7474. Santos CB, Ferreira AL, Leite GR, Ferreira GE, Rodrigues AA, Falqueto A. Peridomiciliary colonies of Triatoma vitticeps (Stal, 1859) (Hemiptera, Reduviidae, Triatominae) infected with Trypanosoma cruzi in rural areas of the state of Espírito Santo, Brazil. Mem Inst Oswaldo Cruz. 2005 Aug;100(5):471-3.) (7575. Lorosa ES, Santos CM, Jurberg J. Foco de doença de Chagas em São Fidélis, no estado do Rio de Janeiro. Rev Soc Bras Med Trop. 2008 jul-ago;41(4):419-20.) (7676. Shikanai-Yasuda MA, Carvalho NB. Oral transmission of Chagas disease. Clin Infect Dis. 2012 Mar;54(6):845-52.) (7777. Sangenis LH, Saraiva RM, Georg I, Castro L, Santos Lima V, Roque AL, et al. Autochthonous transmission of Chagas disease in Rio de Janeiro State, Brazil: a clinical and eco-epidemiological study. BMC Infect Dis. 2015 Jan;15:4. .
The evaluation and systematic monitoring of this process should incorporate the possibility of colonization of the Amazon and other agricultural fronts (extensive cattle ranching and soybean cultivation, among others) by domiciliated vectors, with epidemiological importance in the areas of human migration 2222. Silveira AC, Dias JCP. O controle da transmissão vetorial. Rev Soc Bras Med Trop. 2011;44 supl 2:52-63.) (6565. Silveira AC. Os novos desafios e perspectivas futuras do controle. Rev Soc Bras Med Trop. 2011;44 Supl 2:122-4. .
The great diversity of vector-borne transmission of T. cruzi inherent to the aforementioned scenarios or eco-epidemiological spaces include 2222. Silveira AC, Dias JCP. O controle da transmissão vetorial. Rev Soc Bras Med Trop. 2011;44 supl 2:52-63.) (6565. Silveira AC. Os novos desafios e perspectivas futuras do controle. Rev Soc Bras Med Trop. 2011;44 Supl 2:122-4.: I) the remote risk of reestablishment by introduced species, such as Triatoma infestans ; II) focal transmission by native species known as vectors, such as Panstrongilus megistus, Triatoma brasiliensis and, less likely, Triatoma pseudomaculata and Triatoma sordida ; III) domiciliation of some species typically considered as exclusively wild, and the risk that these then act as vectors in some areas, as in case of Triatoma rubrovaria in the State of Rio Grande do Sul and Panstrongilus lutzi in the States of the Northeastern region; and IV) the possibility of extra-domiciliary transmission or triatomine house visitation (basic form of outbreak generation by oral transmission, at least in rural areas), as already observed with Rhodnius brethesi in the region of Alto Rio Negro, in the State of Amazonas, and with Rhodnius pictipes in the State of Tocantins.
Given the coexistence of areas with different degrees of risk, surveillance and vector control actions should be adjusted and based on established risks 2222. Silveira AC, Dias JCP. O controle da transmissão vetorial. Rev Soc Bras Med Trop. 2011;44 supl 2:52-63.) (6565. Silveira AC. Os novos desafios e perspectivas futuras do controle. Rev Soc Bras Med Trop. 2011;44 Supl 2:122-4. . In addition to considering the operational capacity of municipalities, the stratification of a large traditionally endemic area is based on a set of variables that potentially influence the process of infestation and/or reinfestation, and therefore of transmission or reintroduction of vectorial transmission of Chagas disease in the house environment 2222. Silveira AC, Dias JCP. O controle da transmissão vetorial. Rev Soc Bras Med Trop. 2011;44 supl 2:52-63. .
It is recognized that the process of simplification of fauna is the result of unplanned occupation of the environment, having the following potential consequences: I) lower diversity of food supply for triatomines in different habitats; II) a greater tendency of displacement of triatomines in search of food (hematophagous activity) from their original habitat, to feed on more eclectic mammals species that remained and expanded numerically; III) the populations of the parasite will also expand from these species of mammals with this environmental eclecticism, in general large reservoirs of T. cruzi2121. Organização Pan-americana da Saúde. Guia para vigilância, prevenção, controle e manejo clínico da doença de Chagas aguda transmitida por alimentos. Rio de Janeiro: Organização Pan-Americana da Saúde; 2009. (Série de Manuais Técnicos, 12).)7878. Xavier SC, Roque AL, Lima VS, Monteiro KJ, Otaviano JC, Ferreira da Silva LF, et al. Lower richness of small wild mammal species and Chagas disease risk. PLoS Negl Trop Dis. 2012;6(5):e1647.(7979. Xavier SC, Roque AL, Bilac D, Araújo VA, Costa Neto SF, Lorosa ES, et al. Distantiae transmission of Trypanosoma cruzi: a new epidemiological feature of acute Chagas disease in Brazil. PLoS Negl Trop Dis. 2014 May;8(5):e2878.) (8080. Roque AL, Xavier SC, Gerhardt M, Silva MF, Lima VS, D'Andrea PS, et al. Trypanosoma cruzi among wild and domestic mammals in different areas of the Abaetetuba municipality (Pará State, Brazil), an endemic Chagas disease transmission area. Vet Parasitol. 2013 Mar;193(1-3):71-7. . As these animals (such as marsupials and rodents) have a high degree of synanthropy, the epidemiological risk becomes clear 7878. Xavier SC, Roque AL, Lima VS, Monteiro KJ, Otaviano JC, Ferreira da Silva LF, et al. Lower richness of small wild mammal species and Chagas disease risk. PLoS Negl Trop Dis. 2012;6(5):e1647.) (7979. Xavier SC, Roque AL, Bilac D, Araújo VA, Costa Neto SF, Lorosa ES, et al. Distantiae transmission of Trypanosoma cruzi: a new epidemiological feature of acute Chagas disease in Brazil. PLoS Negl Trop Dis. 2014 May;8(5):e2878.) (8080. Roque AL, Xavier SC, Gerhardt M, Silva MF, Lima VS, D'Andrea PS, et al. Trypanosoma cruzi among wild and domestic mammals in different areas of the Abaetetuba municipality (Pará State, Brazil), an endemic Chagas disease transmission area. Vet Parasitol. 2013 Mar;193(1-3):71-7. .
T. cruzi is a multi-host flagellate parasite capable of infecting tens of species of wild and domestic mammals distributed in all phytogeographic regions in Brazil; it is found in the most diverse ecological niches, contributing, in each type of ecotope, to form singular modalities of natural transmission foci 2121. Organização Pan-americana da Saúde. Guia para vigilância, prevenção, controle e manejo clínico da doença de Chagas aguda transmitida por alimentos. Rio de Janeiro: Organização Pan-Americana da Saúde; 2009. (Série de Manuais Técnicos, 12).) (2323. Coura JR. The main sceneries of Chagas disease transmission. The vectors, blood and oral transmissions: a comprehensive review. Mem Inst Oswaldo Cruz. 2015 May;110(3):277-82.) (2727. Messenger LA, Miles MA, Bern C. Between a bug and a hard place: Trypanosoma cruzi genetic diversity and the clinical outcomes of Chagas disease. Expert Rev Anti Infect Ther. 2015 Aug;13(8):995-1029.) (3939. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Guia de Vigilância em Saúde. Brasília: Ministério da Saúde; 2014.) (7878. Xavier SC, Roque AL, Lima VS, Monteiro KJ, Otaviano JC, Ferreira da Silva LF, et al. Lower richness of small wild mammal species and Chagas disease risk. PLoS Negl Trop Dis. 2012;6(5):e1647.) (7979. Xavier SC, Roque AL, Bilac D, Araújo VA, Costa Neto SF, Lorosa ES, et al. Distantiae transmission of Trypanosoma cruzi: a new epidemiological feature of acute Chagas disease in Brazil. PLoS Negl Trop Dis. 2014 May;8(5):e2878.) (8080. Roque AL, Xavier SC, Gerhardt M, Silva MF, Lima VS, D'Andrea PS, et al. Trypanosoma cruzi among wild and domestic mammals in different areas of the Abaetetuba municipality (Pará State, Brazil), an endemic Chagas disease transmission area. Vet Parasitol. 2013 Mar;193(1-3):71-7. .
The integration of the reservoirs in the routine monitoring process should be taken into account given that domestic animals are not confined and can also act as a link between wild and domestic transmission routes 8080. Roque AL, Xavier SC, Gerhardt M, Silva MF, Lima VS, D'Andrea PS, et al. Trypanosoma cruzi among wild and domestic mammals in different areas of the Abaetetuba municipality (Pará State, Brazil), an endemic Chagas disease transmission area. Vet Parasitol. 2013 Mar;193(1-3):71-7.) (8181. Roque AL, Xavier SC, Rocha MG, Duarte AC, D'Andrea PS, Jansen AM. Trypanosoma cruzi transmission cycle among wild and domestic mammals in three areas of orally transmitted Chagas disease outbreaks. Am J Trop Med Hyg. 2008 Nov;79(5):742-9. . As with wild mammals, the importance of domestic animals (for example, dogs and cats) as reservoirs of T. cruzi varies in different places, and they can be used as sentinel populations of transmission of T. cruzi in a given area 2121. Organização Pan-americana da Saúde. Guia para vigilância, prevenção, controle e manejo clínico da doença de Chagas aguda transmitida por alimentos. Rio de Janeiro: Organização Pan-Americana da Saúde; 2009. (Série de Manuais Técnicos, 12).) (8181. Roque AL, Xavier SC, Rocha MG, Duarte AC, D'Andrea PS, Jansen AM. Trypanosoma cruzi transmission cycle among wild and domestic mammals in three areas of orally transmitted Chagas disease outbreaks. Am J Trop Med Hyg. 2008 Nov;79(5):742-9. . In general, they are always exposed and their infection usually precedes human infection 7878. Xavier SC, Roque AL, Lima VS, Monteiro KJ, Otaviano JC, Ferreira da Silva LF, et al. Lower richness of small wild mammal species and Chagas disease risk. PLoS Negl Trop Dis. 2012;6(5):e1647. . Therefore, the presence of domestic animals infected with T. cruzi in a given area indicates that transmission is occurring in areas where these animals circulate, which reinforces the need to widen control actions 8181. Roque AL, Xavier SC, Rocha MG, Duarte AC, D'Andrea PS, Jansen AM. Trypanosoma cruzi transmission cycle among wild and domestic mammals in three areas of orally transmitted Chagas disease outbreaks. Am J Trop Med Hyg. 2008 Nov;79(5):742-9. .
The research of entomological and reservoirs must be associated with sustained environmental surveillance actions 3939. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Guia de Vigilância em Saúde. Brasília: Ministério da Saúde; 2014. . To this end, the understanding of the processes of habitat selection by triatomines is fundamental for the construction of epidemiological evidence, in order to plan and develop local systems of epidemiological surveillance and control, thus reinforcing the role of surveillance 2222. Silveira AC, Dias JCP. O controle da transmissão vetorial. Rev Soc Bras Med Trop. 2011;44 supl 2:52-63.) (3939. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Guia de Vigilância em Saúde. Brasília: Ministério da Saúde; 2014.) (8282. Abad-Franch F, Ferraz G, Campos C, Palomeque FS, Grijalva MJ, Aguilar HM, et al. Modeling disease vector occurrence when detection is imperfect: infestation of Amazonian palm trees by triatomine bugs at three spatial scales. PLoS Negl Trop Dis. 2010 Mar;4(3):e620. . However, operational failures identified in the detection of vectors can generate critical classification errors and, therefore, prevent the scope of the actions, hampering the composition of scenarios and dynamics of these insects in their habitat 7070. Valença-Barbosa C, Lima MM, Sarquis O, Bezerra CM, Abad-Franch F. Modeling disease vector occurrence when detection is imperfect II: drivers of site-occupancy by synanthropic Triatoma brasiliensis in the Brazilian northeast. PLoS Negl Trop Dis. 2014 May;8(5):e2861.) (7171. Abad-Franch F, Valença-Barbosa C, Sarquis O, Lima MM. All that glisters is not gold: sampling-process uncertainty in disease-vector surveys with false-negative and false-positive detections. PLoS Negl Trop Dis. 2014 Sep;8(9):e3187.) (8282. Abad-Franch F, Ferraz G, Campos C, Palomeque FS, Grijalva MJ, Aguilar HM, et al. Modeling disease vector occurrence when detection is imperfect: infestation of Amazonian palm trees by triatomine bugs at three spatial scales. PLoS Negl Trop Dis. 2010 Mar;4(3):e620. .
Assuming that no sampling technique is perfect , some studies in Brazil have applied methods that explicitly incorporate the failures in the detection of infestation foci in the research of the ecology and the surveillance of vectors of Chagas disease in different environments, including the Amazon and Northeast region 7070. Valença-Barbosa C, Lima MM, Sarquis O, Bezerra CM, Abad-Franch F. Modeling disease vector occurrence when detection is imperfect II: drivers of site-occupancy by synanthropic Triatoma brasiliensis in the Brazilian northeast. PLoS Negl Trop Dis. 2014 May;8(5):e2861.) (7171. Abad-Franch F, Valença-Barbosa C, Sarquis O, Lima MM. All that glisters is not gold: sampling-process uncertainty in disease-vector surveys with false-negative and false-positive detections. PLoS Negl Trop Dis. 2014 Sep;8(9):e3187.) (8282. Abad-Franch F, Ferraz G, Campos C, Palomeque FS, Grijalva MJ, Aguilar HM, et al. Modeling disease vector occurrence when detection is imperfect: infestation of Amazonian palm trees by triatomine bugs at three spatial scales. PLoS Negl Trop Dis. 2010 Mar;4(3):e620. . This approach has and will increasingly enable future improvement of the estimates of eco-epidemiological indicators and may significantly strengthen the strategies for integrated monitoring and control of vectors 7070. Valença-Barbosa C, Lima MM, Sarquis O, Bezerra CM, Abad-Franch F. Modeling disease vector occurrence when detection is imperfect II: drivers of site-occupancy by synanthropic Triatoma brasiliensis in the Brazilian northeast. PLoS Negl Trop Dis. 2014 May;8(5):e2861.) (7171. Abad-Franch F, Valença-Barbosa C, Sarquis O, Lima MM. All that glisters is not gold: sampling-process uncertainty in disease-vector surveys with false-negative and false-positive detections. PLoS Negl Trop Dis. 2014 Sep;8(9):e3187.) (8383. Abad-Franch F, Santos WS, Schofield CJ. Research needs for Chagas disease prevention. Acta Trop. 2010 Jul-Aug;115(1-2):44-54.) (8484. Padilla-Torres SD, Ferraz G, Luz SL, Zamora-Perea E, Abad-Franch F. Modeling dengue vector dynamics under imperfect detection: three years of site-occupancy by Aedes aegypti and Aedes albopictus in urban Amazonia. PLoS One. 2013;8(3):e58420. . The failures were observed in areas with well-established triatomine infestation, where active searches by trained and motivated agents usually detect approximately 40-60% of intradomiciliary and extradomiciliary infestation outbreaks, while undetected foci are not eliminated 7070. Valença-Barbosa C, Lima MM, Sarquis O, Bezerra CM, Abad-Franch F. Modeling disease vector occurrence when detection is imperfect II: drivers of site-occupancy by synanthropic Triatoma brasiliensis in the Brazilian northeast. PLoS Negl Trop Dis. 2014 May;8(5):e2861. . In addition, from the analysis of the monitoring process by means of active searches conducted by control agents, its sensitivity is estimated at approximately 20% for locations with low intensity infestation (few outbreaks, with few triatominae) and at approximately 40% in locations with more intense infestation 7171. Abad-Franch F, Valença-Barbosa C, Sarquis O, Lima MM. All that glisters is not gold: sampling-process uncertainty in disease-vector surveys with false-negative and false-positive detections. PLoS Negl Trop Dis. 2014 Sep;8(9):e3187. .
In addition to this approach, the integrated use of spatial analysis techniques to extend the analytical capability, with insertion of spatial and temporal dimensions of the transmission of Chagas disease, has shown great potential, in particular by the significant development of portable electronic equipment and software for data processing and analysis 7070. Valença-Barbosa C, Lima MM, Sarquis O, Bezerra CM, Abad-Franch F. Modeling disease vector occurrence when detection is imperfect II: drivers of site-occupancy by synanthropic Triatoma brasiliensis in the Brazilian northeast. PLoS Negl Trop Dis. 2014 May;8(5):e2861.) (8585. Abad-Franch F, Vega MC, Rolón MS, Santos WS, Rojas de Arias A. Community participation in Chagas disease vector surveillance: systematic review. PLoS Negl Trop Dis. 2011 Jun;5(6):e1207.) (8686. Roux E, Fátima Venâncio A, Girres JF, Romaña CA. Spatial patterns and eco-epidemiological systems--part II: characterising spatial patterns of the occurrence of the insect vectors of Chagas disease based on remote sensing and field data. Geospat Health. 2011 Nov;6(1):53-64.) (8787. Roux E, Fátima Venâncio A, Girres JF, Romaña CA. Spatial patterns and eco-epidemiological systems--part I: multi-scale spatial modelling of the occurrence of Chagas disease insect vectors. Geospat Health. 2011 Nov;6(1):41-51. .
In the past two decades, several studies have been published describing the different degrees of resistance of triatomine populations to insecticides 8888. Mougabure-Cueto G, Picollo MI. Insecticide resistance in vector Chagas disease: evolution, mechanisms and management. Acta Trop. 2015 Sep;149:70-85. . However, the actual factors behind the emergence of these phenotypes are unknown and the impact of this on the development of standardized strategies for vector control are unclear 8989. Pessoa GCA, Pinheiro LC, Ferraz ML, Mello BV, Diotaiuti L. Standardization of laboratory bioassays for the study of Triatoma sordida susceptibility to pyrethroid insecticides. Parasit Vectors. 2015 Feb;8:109.) (9090. Pessoa GCA, Trevizani NAB, Dias LS, Bezerra CM, Melo BV, Diotaiut L. Toxicological profile of deltamethrin in Triatoma brasiliensis (Hemiptera: Reduviidae) in State of Ceará, Northeastern Brazil. Rev Soc Bras Med Trop. 2015 Jan-Feb;48(1):39-43. . This new context will require endemic countries to build networks of cooperation between laboratories for the analysis of resistance in triatomine populations in a standardized manner, integrating this activity in the process of epidemiological surveillance 8888. Mougabure-Cueto G, Picollo MI. Insecticide resistance in vector Chagas disease: evolution, mechanisms and management. Acta Trop. 2015 Sep;149:70-85. .
Oral transmission
In Brazil, vectorial transmission of Chagas disease by its principal vector, T. infestans , is under control. However, oral transmission of T. cruzi has become increasingly epidemiologically relevant, particularly in the Amazon Region 1111. Coura JR, Viñas PA, Junqueira AC. Ecoepidemiology, short history and control of Chagas disease in the endemic countries and the new challenge for non-endemic countries. Mem Inst Oswaldo Cruz. 2014 Nov;109(7):856-62.) (2323. Coura JR. The main sceneries of Chagas disease transmission. The vectors, blood and oral transmissions: a comprehensive review. Mem Inst Oswaldo Cruz. 2015 May;110(3):277-82.) (3939. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Guia de Vigilância em Saúde. Brasília: Ministério da Saúde; 2014.) (7676. Shikanai-Yasuda MA, Carvalho NB. Oral transmission of Chagas disease. Clin Infect Dis. 2012 Mar;54(6):845-52.) (9191. Pinto AYN, Valente SA, Valente VC, Ferreira Júnior AG, Coura JR. Fase aguda da doença de Chagas na Amazônia brasileira: estudo de 233 casos do Pará, Amapá e Maranhão observados entre 1988 e 2005. Rev Soc Bras Med Trop. 2008 nov-dec;41(6):602-14.) (9292. Nóbrega AA, Garcia MH, Tatto E, Obara MT, Costa E, Sobel J, et al. Oral transmission of Chagas disease by consumption of açaí palm fruit, Brazil. Emerg Infect Dis. 2009 Apr;15(4):653-5. . Transmission by the oral route is considered a primary mechanism, in particular in the wild cycle 9393. Dias JCP, Amato Neto V. Prevenção referente às modalidades alternativas de transmissão do Trypanosoma cruzi no Brasil. Rev Soc Bras Med Trop. 2011;44 supl 2:68-72. ; it will occur regardless of control actions undertaken 2121. Organização Pan-americana da Saúde. Guia para vigilância, prevenção, controle e manejo clínico da doença de Chagas aguda transmitida por alimentos. Rio de Janeiro: Organização Pan-Americana da Saúde; 2009. (Série de Manuais Técnicos, 12).) (3939. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Guia de Vigilância em Saúde. Brasília: Ministério da Saúde; 2014. . It presents a common feature in the early enzootic cycle of this parasite: ingestion of vectors and reservoirs of infection by susceptible mammals 2121. Organização Pan-americana da Saúde. Guia para vigilância, prevenção, controle e manejo clínico da doença de Chagas aguda transmitida por alimentos. Rio de Janeiro: Organização Pan-Americana da Saúde; 2009. (Série de Manuais Técnicos, 12). . In the case of humans, this transmission occurs in a sporadic and circumstantial manner, through food contaminated with the parasite, mainly from triatomines or their feces, with records since the 1960s 9393. Dias JCP, Amato Neto V. Prevenção referente às modalidades alternativas de transmissão do Trypanosoma cruzi no Brasil. Rev Soc Bras Med Trop. 2011;44 supl 2:68-72. . Therefore, the analysis of this epidemiological context refers indirectly to vectors.
In Brazil, most cases of orally transmitted Chagas disease have been reported in the Amazon as outbreaks in families or multifamily contexts 1111. Coura JR, Viñas PA, Junqueira AC. Ecoepidemiology, short history and control of Chagas disease in the endemic countries and the new challenge for non-endemic countries. Mem Inst Oswaldo Cruz. 2014 Nov;109(7):856-62.) (2323. Coura JR. The main sceneries of Chagas disease transmission. The vectors, blood and oral transmissions: a comprehensive review. Mem Inst Oswaldo Cruz. 2015 May;110(3):277-82.) (9191. Pinto AYN, Valente SA, Valente VC, Ferreira Júnior AG, Coura JR. Fase aguda da doença de Chagas na Amazônia brasileira: estudo de 233 casos do Pará, Amapá e Maranhão observados entre 1988 e 2005. Rev Soc Bras Med Trop. 2008 nov-dec;41(6):602-14.) (9292. Nóbrega AA, Garcia MH, Tatto E, Obara MT, Costa E, Sobel J, et al. Oral transmission of Chagas disease by consumption of açaí palm fruit, Brazil. Emerg Infect Dis. 2009 Apr;15(4):653-5. . Outside the Amazon region, few events have been investigated; most were related to sugar cane juice, the likely food carrier in the oral transmission of these cases 7676. Shikanai-Yasuda MA, Carvalho NB. Oral transmission of Chagas disease. Clin Infect Dis. 2012 Mar;54(6):845-52.) (9393. Dias JCP, Amato Neto V. Prevenção referente às modalidades alternativas de transmissão do Trypanosoma cruzi no Brasil. Rev Soc Bras Med Trop. 2011;44 supl 2:68-72. . A recent review of the subject identified records of outbreaks or micro-outbreaks in Brazil, in the States of Rio Grande do Sul, Pará, Paraíba, Santa Catarina, Bahia, and Ceará. Other countries with records included Venezuela and Colombia 7676. Shikanai-Yasuda MA, Carvalho NB. Oral transmission of Chagas disease. Clin Infect Dis. 2012 Mar;54(6):845-52. .
The Ministry of Health reported 112 outbreaks nationally between 2005 and 2013, involving all 35 municipalities in the Amazon Region. The probable source of infection was food contaminated with T. cruzi , including: açaí, bacaba, jaci ( Syagrus ), sugar cane juice, and palm hearts of babaçu nuts. Most of the outbreaks occurred in the States of Pará, 75.9% (85 outbreaks) and Amapá, 12.5% (14 outbreaks) and, to a lesser extent, in the States of Amazonas, 4.5% (5 outbreaks), Tocantins, 1.8% (2 outbreaks), and Bahia, 1.8% (2 outbreaks) 5555. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Doença de Chagas aguda no Brasil: série histórica de 2000 a 2013. Bol Epidemiol. 2015;46(21):1-9. .
The process of surveillance of Chagas disease and acute oral transmission was potentiated by the increased sensitivity of the surveillance system. The first officially investigated outbreak of orally transmitted Chagas disease in Brazil occurred in Santa Catarina in 2005, probably linked to the intake of sugar cane juice contaminated with T. cruzi5555. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Doença de Chagas aguda no Brasil: série histórica de 2000 a 2013. Bol Epidemiol. 2015;46(21):1-9. . Between 2007 and 2013, it should be emphasized that more than 50% of orally transmitted Chagas disease cases presented with symptoms between August and November, a period that coincides with the açaí harvest in Pará 5555. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Doença de Chagas aguda no Brasil: série histórica de 2000 a 2013. Bol Epidemiol. 2015;46(21):1-9. .
Available experimental evidence suggests that oral transmission may occur from trypomastigote, epimastigotes, and probably amastigotes forms of T. cruzi and cell masses, originating from mammals or contaminated vectors, as well as, accidentally, through artificial cultures of the parasite 2121. Organização Pan-americana da Saúde. Guia para vigilância, prevenção, controle e manejo clínico da doença de Chagas aguda transmitida por alimentos. Rio de Janeiro: Organização Pan-Americana da Saúde; 2009. (Série de Manuais Técnicos, 12).) (2323. Coura JR. The main sceneries of Chagas disease transmission. The vectors, blood and oral transmissions: a comprehensive review. Mem Inst Oswaldo Cruz. 2015 May;110(3):277-82.) (9494. Dias JCP. Notas sobre o Trypanosoma cruzi e suas características bio-ecológicas, como agente de enfermidades transmitidas por alimentos. Rev Soc Bras Med Trop. 2006 jul-ago;39(4):370-5. . Depending on the predominance of a large group or lineage of T. cruzi transmitted, as well as of the inoculum in question, there is a diversity in pathogenicity, histiotropism, morbidity, and mortality 2121. Organização Pan-americana da Saúde. Guia para vigilância, prevenção, controle e manejo clínico da doença de Chagas aguda transmitida por alimentos. Rio de Janeiro: Organização Pan-Americana da Saúde; 2009. (Série de Manuais Técnicos, 12). . In relation to the mechanism of oral transmission of T. cruzi , the following possible scenarios have been identified 2121. Organização Pan-americana da Saúde. Guia para vigilância, prevenção, controle e manejo clínico da doença de Chagas aguda transmitida por alimentos. Rio de Janeiro: Organização Pan-Americana da Saúde; 2009. (Série de Manuais Técnicos, 12).) (3939. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Guia de Vigilância em Saúde. Brasília: Ministério da Saúde; 2014.) (9393. Dias JCP, Amato Neto V. Prevenção referente às modalidades alternativas de transmissão do Trypanosoma cruzi no Brasil. Rev Soc Bras Med Trop. 2011;44 supl 2:68-72.) (9494. Dias JCP. Notas sobre o Trypanosoma cruzi e suas características bio-ecológicas, como agente de enfermidades transmitidas por alimentos. Rev Soc Bras Med Trop. 2006 jul-ago;39(4):370-5.:
-
ingestion of triatomine feces or urine of or even infected triatomines, on the assumption that these are processed or carried along with food, as verified in episodes investigated in which the infection was assigned to the consumption of acai fruit, typical of the Brazilian Amazon Region 9292. Nóbrega AA, Garcia MH, Tatto E, Obara MT, Costa E, Sobel J, et al. Oral transmission of Chagas disease by consumption of açaí palm fruit, Brazil. Emerg Infect Dis. 2009 Apr;15(4):653-5. ;
-
intake of food or drinks contaminated with metacyclic trypomastigote forms in the secretion of the anal gland or in the urine of infected marsupials of the genus Didelphis ;
-
intake of suspensions of T. cruzi in pipettes in the context of research or diagnostic laboratories;
-
intake of breast milk from mothers diagnosed with: acute Chagas disease, T. cruzi -HIV co-infection in the AIDS stage, documented reactivation of Chagas disease, or bleeding from fissures of the nipples.
Other speculative situations of exposure include 2121. Organização Pan-americana da Saúde. Guia para vigilância, prevenção, controle e manejo clínico da doença de Chagas aguda transmitida por alimentos. Rio de Janeiro: Organização Pan-Americana da Saúde; 2009. (Série de Manuais Técnicos, 12).) (3939. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Guia de Vigilância em Saúde. Brasília: Ministério da Saúde; 2014.) (9393. Dias JCP, Amato Neto V. Prevenção referente às modalidades alternativas de transmissão do Trypanosoma cruzi no Brasil. Rev Soc Bras Med Trop. 2011;44 supl 2:68-72.) (9494. Dias JCP. Notas sobre o Trypanosoma cruzi e suas características bio-ecológicas, como agente de enfermidades transmitidas por alimentos. Rev Soc Bras Med Trop. 2006 jul-ago;39(4):370-5.:
-
ingestion of raw or poorly cooked meat or blood from infected mammals, especially wild animals;
-
consumption of the blood of infected animals for therapeutic purposes, as performed by some indigenous groups in the Amazon. This has been reported in some regions of Colombia, where the intake of blood of armadillos and skunks has been observed;
-
contamination of equipment used in the handling of carcasses of infected mammals;
-
ingestion of triatomines by primitive or exotic habits.
It is noteworthy that the feces of infected triatomines can retain infectious potential for a few hours in environments with high humidity. Therefore, they can potentially contaminate food directly or indirectly, by the feet and oral cavity of secondary carriers, like flies and cockroaches. Experimental studies have shown that the parasite can remain viable for ≥24 hours in foods such as milk or sugar cane juice if stored at room temperature. Although gastric juice is able to destroy a considerable number of parasites, some are able to escape this through chemical mechanisms of external protection, allowing the parasites to penetrate the intestinal mucosa 2121. Organização Pan-americana da Saúde. Guia para vigilância, prevenção, controle e manejo clínico da doença de Chagas aguda transmitida por alimentos. Rio de Janeiro: Organização Pan-Americana da Saúde; 2009. (Série de Manuais Técnicos, 12).) (9292. Nóbrega AA, Garcia MH, Tatto E, Obara MT, Costa E, Sobel J, et al. Oral transmission of Chagas disease by consumption of açaí palm fruit, Brazil. Emerg Infect Dis. 2009 Apr;15(4):653-5. .
Although oral transmission is generally associated with outbreaks, it can also occur in isolated cases 2121. Organização Pan-americana da Saúde. Guia para vigilância, prevenção, controle e manejo clínico da doença de Chagas aguda transmitida por alimentos. Rio de Janeiro: Organização Pan-Americana da Saúde; 2009. (Série de Manuais Técnicos, 12).) (3939. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Guia de Vigilância em Saúde. Brasília: Ministério da Saúde; 2014. . In fact, we can identify two main profiles of outbreaks, according to the place of occurrence: urban and rural. Urban outbreak derives from the consumption of semi-industrialized and commercialized açaí, reflecting an issue of sanitary surveillance, associated with food safety and good food handling practices. In rural outbreaks, there is the participation of various types of food prepared locally by hand, generally by family or people of the social network/community of a given family.
For research and surveillance purposes, food recall is a useful tool to identify the possibility of suspect food intake. It allows one to identify the source, place of preparation and consumption of suspect food, and to list all the people who may have ingested the suspect food 2121. Organização Pan-americana da Saúde. Guia para vigilância, prevenção, controle e manejo clínico da doença de Chagas aguda transmitida por alimentos. Rio de Janeiro: Organização Pan-Americana da Saúde; 2009. (Série de Manuais Técnicos, 12).) (3939. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Guia de Vigilância em Saúde. Brasília: Ministério da Saúde; 2014.) (9292. Nóbrega AA, Garcia MH, Tatto E, Obara MT, Costa E, Sobel J, et al. Oral transmission of Chagas disease by consumption of açaí palm fruit, Brazil. Emerg Infect Dis. 2009 Apr;15(4):653-5. . The focus of surveillance actions and control should therefore include the productive chain, based on good food handling practices 2121. Organização Pan-americana da Saúde. Guia para vigilância, prevenção, controle e manejo clínico da doença de Chagas aguda transmitida por alimentos. Rio de Janeiro: Organização Pan-Americana da Saúde; 2009. (Série de Manuais Técnicos, 12).) (3939. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Guia de Vigilância em Saúde. Brasília: Ministério da Saúde; 2014. .
Transmission by blood transfusion and transplantation of tissues/organs
The transmission of T. cruzi in blood transfusions was amplified by the urbanization of Chagas disease in Brazil and in other endemic countries in Latin America 33. Dias JC, Silveira AC, Schofield CJ. The impact of Chagas disease control in Latin America: a review. Mem Inst Oswaldo Cruz. 2002 Jul;97(5):603-12.) (44. Coura JR, Dias JC. Epidemiology, control and surveillance of Chagas disease: 100 years after its discovery. Mem Inst Oswaldo Cruz. 2009 Jul;104 Suppl 1:31-40.) (1010. Dias JCP. Human chagas disease and migration in the context of globalization: some particular aspects. J Trop Med. 2013:789758.) (9595. Dias JCP, Schofield CJ. Controle da transmissão transfusional da doença de Chagas na Iniciativa do Cone Sul. Rev Soc Bras Med Trop. 1998 jul-ago;31(4):373-83.) (9696. Moraes-Souza H, Ferreira-Silva MM. Controle da transmissão transfusional. Rev Soc Bras Med Trop. 2011;44 supl 2:64-7. . The risk of transmission by blood transfusion is dependent on various factors: I) the presence of the parasite in the blood or blood component transfused; II) type and number of infected blood products transfused; III) immune status of the recipient; IV) quality of the clinical-epidemiological screening; V) level of coverage of the serologic screening of donors; and VI) sensitivity of serologic tests used to screen blood donation candidates 44. Coura JR, Dias JC. Epidemiology, control and surveillance of Chagas disease: 100 years after its discovery. Mem Inst Oswaldo Cruz. 2009 Jul;104 Suppl 1:31-40.) (2323. Coura JR. The main sceneries of Chagas disease transmission. The vectors, blood and oral transmissions: a comprehensive review. Mem Inst Oswaldo Cruz. 2015 May;110(3):277-82.) (9595. Dias JCP, Schofield CJ. Controle da transmissão transfusional da doença de Chagas na Iniciativa do Cone Sul. Rev Soc Bras Med Trop. 1998 jul-ago;31(4):373-83.) (9696. Moraes-Souza H, Ferreira-Silva MM. Controle da transmissão transfusional. Rev Soc Bras Med Trop. 2011;44 supl 2:64-7. .
Given that the majority of potential blood donors are aged 18-35 years-the age group with the lowest number of candidates potentially infected with T. cruzi- and that, in recent years, the obligation of clinical, epidemiological, and serologic status screening has been established in most endemic countries, the risk of transfusion transmission of Chagas disease has been largely reduced in Latin America as a whole 44. Coura JR, Dias JC. Epidemiology, control and surveillance of Chagas disease: 100 years after its discovery. Mem Inst Oswaldo Cruz. 2009 Jul;104 Suppl 1:31-40.) (2323. Coura JR. The main sceneries of Chagas disease transmission. The vectors, blood and oral transmissions: a comprehensive review. Mem Inst Oswaldo Cruz. 2015 May;110(3):277-82.) (9595. Dias JCP, Schofield CJ. Controle da transmissão transfusional da doença de Chagas na Iniciativa do Cone Sul. Rev Soc Bras Med Trop. 1998 jul-ago;31(4):373-83.) (9696. Moraes-Souza H, Ferreira-Silva MM. Controle da transmissão transfusional. Rev Soc Bras Med Trop. 2011;44 supl 2:64-7. . In Brazil in the 1950s, the estimated average prevalence of positive serology for T. cruzi among potential blood donors was 8.3%. This decreased to 6.9% in the 1960s and 1970s, dropping to 3.2% in the late 1980s and early 1990s 9696. Moraes-Souza H, Ferreira-Silva MM. Controle da transmissão transfusional. Rev Soc Bras Med Trop. 2011;44 supl 2:64-7. . A Health ministry report from January to June 1994 stated an estimated prevalence of Chagas disease through blood transfusion, by the public blood donation network to different geographic regions of the country, of 0.75% 9595. Dias JCP, Schofield CJ. Controle da transmissão transfusional da doença de Chagas na Iniciativa do Cone Sul. Rev Soc Bras Med Trop. 1998 jul-ago;31(4):373-83. . In 2006, this estimate was 0.21%, a reduction of more than 95% compared with previous data 1717. Pan American Health Organization. Estimación cuantitativa de la enfermedad de Chagas en las Americas. Washington: Pan American Health Organization; 2006. . Most recent WHO data from 2010, estimated this prevalence to be 0.18% in Brazil 55. World Health Organization. Chagas disease in Latin America: an epidemiological update based on 2010 estimates. Wkly Epidemiol Rec. 2015 Feb;90(6):33-44. .
In Brazil, not only the safety of blood transfusion has improved, but also the process of hemovigilance, with the establishment of a national system integrating blood banks and epidemiological and sanitary surveillance 9797. Ministério da Saúde (BR). Agência Nacional de Vigilância Sanitária. Manual técnico para a investigação da transmissão de doenças pelo sangue. Brasília: Ministério da Saúde; 2004. (Série A. Normas e manuais técnicos).) (9898. Ministério da Saúde (BR). Agência Nacional de Vigilância Sanitária. Manual técnico de hemovigilância. Brasília: Ministério da Saúde; 2004. (Série A. Normas e manuais técnicos). . With this, one can classify the rare, but possible cases of Chagas disease transmitted by blood, associated with the residual blood transfusion risk and any failures within the process. The same applies for situations associated with screening of candidates for donation of tissues or organs. For both, sanitary inspection in hematology and transplantation services (hospitals or blood banks) are instituted, as is communication and integrated action with haemovigilance 9797. Ministério da Saúde (BR). Agência Nacional de Vigilância Sanitária. Manual técnico para a investigação da transmissão de doenças pelo sangue. Brasília: Ministério da Saúde; 2004. (Série A. Normas e manuais técnicos).) (9898. Ministério da Saúde (BR). Agência Nacional de Vigilância Sanitária. Manual técnico de hemovigilância. Brasília: Ministério da Saúde; 2004. (Série A. Normas e manuais técnicos). , recalling that the transfusion transmission can only be determined if the infected person received blood or some other component within the 120 days before the onset of symptoms 9797. Ministério da Saúde (BR). Agência Nacional de Vigilância Sanitária. Manual técnico para a investigação da transmissão de doenças pelo sangue. Brasília: Ministério da Saúde; 2004. (Série A. Normas e manuais técnicos). .
A similar trend in the reduction of the risk of transmission from blood transfusions has been observed in other endemic countries, to a greater or lesser degree. For non-endemic countries, in the last decade several strategies have been adopted to prevent and control transfusion transmission of Chagas disease. Nations like the United States, Canada, Spain, France, the United Kingdom, Switzerland and Australia, for example, have already introduced strategies of universal or selective serologic screening of blood donation candidates 9999. Angheben A, Boix L, Buonfrate D, Gobbi F, Bisoffi Z, Pupella S, et al. Chagas disease and transfusion medicine: a perspective from non-endemic countries. Blood Transfus. 2015 Oct;13(4):540-50. .
In case of organ transplants, endemicity patterns of areas of origin and residence should be considered, both for donors and recipients, in order to outline the best approach possible for each case in terms of screening and management of possible infection by T. cruzi100100. Pinazo MJ, Miranda B, Rodríguez-Villar C, Altclas J, Brunet Serra M, García-Otero EC, et al. Recommendations for management of Chagas disease in organ and hematopoietic tissue transplantation programs in nonendemic areas. Transplant Rev (Orlando). 2011 Jul;25(3):91-101. .
Vertical transmission (congenital)
Vertical transmission of Chagas disease is still relatively important in Brazil, despite the absence of systematic preventive measures against this type of transmission in the country. To assess its impact and also the impact of vector control, between 2001 and 2008 a new national serologic survey was conducted, now having as a reference population 105,000 children aged 0-5 years 101101. Ostermayer AL, Passos ADC, Silveira AC, Ferreira AW, Macedo V, Prata AR. O inquérito nacional de soroprevalência de avaliação do controle da doença de Chagas no Brasil (2001-2008). Rev Soc Bras Med Trop. 2011;44 supl 2:108-21. . The estimated prevalence of children infected with T. cruzi was 0.03% (32 cases). Of these, 20 (0.02%) had concomitant maternal positivity, suggesting congenital transmission [coming from the States of Rio Grande do Sul (12), Minas Gerais (3), Paraná (1), Pernambuco (1), Alagoas (1), Bahia (1), and Acre (1)]. In the other 11 (0.01%) cases, only the child tested positive, indicating probable vector transmission [derived from the State of Piauí (2), Ceará (2), Paraíba (2), Alagoas (2), Rio Grande do Norte (1), Amazon (1) and Paraná (1)]. This study was the first to identify regional differences in the congenital transmission of Chagas disease in Brazil, possibly related to the existence of T. cruzi TcV and TcVI 101101. Ostermayer AL, Passos ADC, Silveira AC, Ferreira AW, Macedo V, Prata AR. O inquérito nacional de soroprevalência de avaliação do controle da doença de Chagas no Brasil (2001-2008). Rev Soc Bras Med Trop. 2011;44 supl 2:108-21. . Moreover, it points to the absence of Chagas disease transmission by a sustained domiciliary vector in the country 3939. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Guia de Vigilância em Saúde. Brasília: Ministério da Saúde; 2014.) (101101. Ostermayer AL, Passos ADC, Silveira AC, Ferreira AW, Macedo V, Prata AR. O inquérito nacional de soroprevalência de avaliação do controle da doença de Chagas no Brasil (2001-2008). Rev Soc Bras Med Trop. 2011;44 supl 2:108-21. .
Official data from the Ministry of Health concerning epidemiological surveillance by reporting cases of acute Chagas disease indicate consistency with the findings obtained in the aforementioned survey: 50% of notifications of this form of transmission were from the State of Rio Grande do Sul 5555. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Doença de Chagas aguda no Brasil: série histórica de 2000 a 2013. Bol Epidemiol. 2015;46(21):1-9. .
The overall estimate of the prevalence of T. cruzi infection in pregnant women ranges from 1 to 40% 102102. Torrico F, Alonso-Vega C, Suarez E, Rodriguez P, Torrico MC, Dramaix M, et al. Maternal Trypanosoma cruzi infection, pregnancy outcome, morbidity, and mortality of congenitally infected and non-infected newborns in Bolivia. Am J Trop Med Hyg. 2004 Feb;70(2):201-9.) (103103. Salas NA, Cot M, Schneider D, Mendoza B, Santalla JA, Postigo J, et al. Risk factors and consequences of congenital Chagas disease in Yacuiba, South Bolivia. Trop Med Int Health. 2007 Dec;12(12):1498-505.) (104104. Carlier Y, Torrico F, Sosa-Estani S, Russomando G, Luquetti A, Freilij H, et al. Congenital Chagas disease: recommendations for diagnosis, treatment and control of newborns, siblings and pregnant women. PLoS Negl Trop Dis. 2011 Oct;5(10):e1250. , and approximately 1.8 million women of childbearing age in Latin America are infected 1717. Pan American Health Organization. Estimación cuantitativa de la enfermedad de Chagas en las Americas. Washington: Pan American Health Organization; 2006. . The latest data from WHO estimate that 1,124,930 women aged 15-44 years are infected with T. cruzi55. World Health Organization. Chagas disease in Latin America: an epidemiological update based on 2010 estimates. Wkly Epidemiol Rec. 2015 Feb;90(6):33-44. . A recent meta-analysis estimated the global risk for congenital T. cruzi infection in children born to infected mothers as being 5% (range: 0-28.6%) 101101. Ostermayer AL, Passos ADC, Silveira AC, Ferreira AW, Macedo V, Prata AR. O inquérito nacional de soroprevalência de avaliação do controle da doença de Chagas no Brasil (2001-2008). Rev Soc Bras Med Trop. 2011;44 supl 2:108-21. . In Latin America, more than 15,000 cases per year of congenital Chagas disease are estimated to occur 1717. Pan American Health Organization. Estimación cuantitativa de la enfermedad de Chagas en las Americas. Washington: Pan American Health Organization; 2006. . More recent studies estimate that 8,668 children are vertically infected 55. World Health Organization. Chagas disease in Latin America: an epidemiological update based on 2010 estimates. Wkly Epidemiol Rec. 2015 Feb;90(6):33-44. . On the other hand, in non-endemic areas, the availability of evidence to estimate incidence is even more limited 55. World Health Organization. Chagas disease in Latin America: an epidemiological update based on 2010 estimates. Wkly Epidemiol Rec. 2015 Feb;90(6):33-44.) (1717. Pan American Health Organization. Estimación cuantitativa de la enfermedad de Chagas en las Americas. Washington: Pan American Health Organization; 2006.) (104104. Carlier Y, Torrico F, Sosa-Estani S, Russomando G, Luquetti A, Freilij H, et al. Congenital Chagas disease: recommendations for diagnosis, treatment and control of newborns, siblings and pregnant women. PLoS Negl Trop Dis. 2011 Oct;5(10):e1250. . In North America, for example, the estimate is of 2,000 cases of congenital Chagas disease per year 4545. Buekens P, Almendares O, Carlier Y, Dumonteil E, Eberhard M, Gamboa-Leon R, et al. Mother-to-child transmission of Chagas' disease in North America: why don't we do more? Matern Child Health J. 2008 May;12(3):283-6. .
A systematic review with meta-analysis of studies conducted in Brazil estimated the prevalence of infection in pregnant women to be 1.1% with a vertical transmission rate of 1.7% 106106. Martins-Melo FR, Lima MS, Ramos Júnior AN, Alencar CH, Heukelbach J. Prevalence of Chagas disease in pregnant women and congenital transmission of Trypanosoma cruzi in Brazil: a systematic review and meta-analysis. Trop Med Int Health. 2014 Aug;19(8):943-57. . According to data from the Brazilian Live Birth Information System [ Sistema de Informações sobre Nascidos Vivos (SINASC)], 2,861,868 live births were recorded in the country in 2010. From these data it was estimated that 34,629 pregnant women would be infected with T. cruzi this year; the number of congenitally infected children would thus range from 312 to 1,073 (average: 589 cases) 106106. Martins-Melo FR, Lima MS, Ramos Júnior AN, Alencar CH, Heukelbach J. Prevalence of Chagas disease in pregnant women and congenital transmission of Trypanosoma cruzi in Brazil: a systematic review and meta-analysis. Trop Med Int Health. 2014 Aug;19(8):943-57. . These data are consistent with those of an epidemiological survey conducted in the State of Minas Gerais in 1998, extrapolated to the Brazilian population 107107. Gontijo ED, Andrade GMQ, Santos SE, Galvão LMC, Moreira EF, Pinto FS, et al. Triagem neonatal da infecção pelo Trypanosoma cruzi em Minas Gerais, Brasil: transmissão congênita e mapeamento das áreas endêmicas. Epidemiol Serv Saude. 2009 jul-set;18(3):243-54. , and with the global estimate provided by the WHO of 571 Brazilian children infected by vertical transmission in 2010 55. World Health Organization. Chagas disease in Latin America: an epidemiological update based on 2010 estimates. Wkly Epidemiol Rec. 2015 Feb;90(6):33-44. . In 2006, PAHO estimated that 5,000 Brazilian children would be vertically infected 1717. Pan American Health Organization. Estimación cuantitativa de la enfermedad de Chagas en las Americas. Washington: Pan American Health Organization; 2006. . Considering the gradual reduction in fertility rates in Brazil, it is estimated that over the next few decades, vertical transmission will be substantially reduced, with possible elimination as public health problem for the country in the next 10 to 20 years.
A recent retrospective study from a referral hospital of the Federal University of Goiás [ Universidade Federal de Goiás (UFG)] analyzed 1,211 individuals born to mothers known to be infected by T. cruzi and identified a rate of vertical transmission of 2%, consistent with previous estimates. This suggests that the presence of TcII in the study area may be associated with lower rates of transmission than TcV, which predominates in the southern regions of Brazil and other Southern Cone countries, where the vertical transmission rates are generally known 108108. Luquetti AO, Tavares SBN, Siriano LR, Oliveira RA, Campos DE, Morais CA, et al. Congenital transmission of Trypanosoma cruzi in central Brazil. A study of 1,211 individuals born to infected mothers. Mem Inst Oswaldo Cruz. 2015 May;110(3):369-76. . Despite existing scientific evidence, important gaps remain in terms of the role of genetic diversity of T. cruzi in relation to the evolution of the chronic phase of the disease, the risk of congenital transmission or reactivation, and the occurrence of oral outbreaks transmission 2727. Messenger LA, Miles MA, Bern C. Between a bug and a hard place: Trypanosoma cruzi genetic diversity and the clinical outcomes of Chagas disease. Expert Rev Anti Infect Ther. 2015 Aug;13(8):995-1029. .
In Brazil, congenital Chagas disease is considered acute and therefore its registration is mandatory 2020. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Consenso Brasileiro em Doença de Chagas. Rev Soc Bras Med Trop. 2005;38 Supl 3:1-29.) (3939. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Guia de Vigilância em Saúde. Brasília: Ministério da Saúde; 2014. . However, surveillance for Chagas disease specifically in pregnant women or exposed/infected children has not yet been established in the country. On the other hand, it is recognized that provision of anti- T. cruzi treatment to non-pregnant infected women of childbearing age may be an effective strategy to reduce vertical transmission in future pregnancies 4141. Bern C, Verastegui M, Gilman RH, Lafuente C, Galdos-Cardenas G, Calderon M, et al. Congenital Trypanosoma cruzi transmission in Santa Cruz, Bolivia. Clin Infect Dis. 2009 Dec 1;49(11):1667-74.) (109109. Sosa-Estani S, Cura E, Velazquez E, Yampotis C, Segura EL. Etiological treatment of young women infected with Trypanosoma cruzi , and prevention of congenital transmission. Rev Soc Bras Med Trop. 2009 Sep-Oct;42(5):484-7. . It should also be noted that vertical transmission may be repeated in every pregnancy 104104. Carlier Y, Torrico F, Sosa-Estani S, Russomando G, Luquetti A, Freilij H, et al. Congenital Chagas disease: recommendations for diagnosis, treatment and control of newborns, siblings and pregnant women. PLoS Negl Trop Dis. 2011 Oct;5(10):e1250. .
Accidental transmission
Accidental transmission has been recorded in different contexts: triatomine laboratories, vector capture actions in endemic areas, experimental work with infected mammals and cultures, aerosols of infected materials, surgical infection and infection during collection of blood samples from people with acute infection. In these cases, safety deficiencies are evident in the transportation of contaminated materials, among other factors 9393. Dias JCP, Amato Neto V. Prevenção referente às modalidades alternativas de transmissão do Trypanosoma cruzi no Brasil. Rev Soc Bras Med Trop. 2011;44 supl 2:68-72. . Risk factors include ignorance, inattention, lack or misuse of personal protective equipment, inadequate facilities and equipment, poor lighting, lack of training, non-compliance with standard precautionary measures, and non-adoption of routine technical protocols 9393. Dias JCP, Amato Neto V. Prevenção referente às modalidades alternativas de transmissão do Trypanosoma cruzi no Brasil. Rev Soc Bras Med Trop. 2011;44 supl 2:68-72.) (110110. Herwaldt BL. Laboratory-acquired parasitic infections from accidental exposures. Clin Microbiol Rev. 2001 Oct;14(4):659-88. .
The process of monitoring and control must include the development of continuing education actions in health and supervision, to check the proper use of personal protective equipment and, if applied, the communication of work accidents within the demarcated processes for the health surveillance of workers 3939. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Guia de Vigilância em Saúde. Brasília: Ministério da Saúde; 2014.) (9393. Dias JCP, Amato Neto V. Prevenção referente às modalidades alternativas de transmissão do Trypanosoma cruzi no Brasil. Rev Soc Bras Med Trop. 2011;44 supl 2:68-72.) (110110. Herwaldt BL. Laboratory-acquired parasitic infections from accidental exposures. Clin Microbiol Rev. 2001 Oct;14(4):659-88. .
Epidemiological surveillance
Epidemiological surveillance is a set of strategic actions that provide knowledge, detection, or prevention of any change in determinants and conditioning factors of individual or collective health, in order to recommend and adopt measures for the prevention and control of diseases and disorders 3939. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Guia de Vigilância em Saúde. Brasília: Ministério da Saúde; 2014. . In the case of Chagas disease, it includes necessarily integrated actions involving human cases, vectors, and reservoirs, interfacing closely with the health care network, with emphasis on primary care or primary health 44. Coura JR, Dias JC. Epidemiology, control and surveillance of Chagas disease: 100 years after its discovery. Mem Inst Oswaldo Cruz. 2009 Jul;104 Suppl 1:31-40.) (1515. Ramos Júnior AN, Carvalho DM. Chagas' disease: past, present and future. Cad Saude Colet. 2009;17(4):787-94. .
Systematized below are the main elements in Brazil for the development of surveillance processes for human cases and for entomological surveillance. Several questions related to this process have already been presented and previously discussed.
Chagas disease cases surveillance
Epidemiological surveillance actions for Chagas disease in Brazil have the following main objectives 2020. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Consenso Brasileiro em Doença de Chagas. Rev Soc Bras Med Trop. 2005;38 Supl 3:1-29.) (3939. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Guia de Vigilância em Saúde. Brasília: Ministério da Saúde; 2014.:
-
to detect early cases of acute Chagas disease, with the aim to treat newly diagnosed cases timeously and to apply preventive measures of occurrence to new cases;
-
to conduct epidemiologic investigation of all acute cases, aiming to identify the form of transmission and, consequently, to institute appropriate control measures;
-
to monitor T. cruzi infection in humans, through periodic serologic surveys in key populations, and by means of adopting a national screening process of blood donation candidates in blood donation centers;
-
to monitor the profile of morbidity and mortality of Chagas disease in the country, outlining scenarios for strengthening the healthcare network for infected people;
-
to maintain the elimination of vector transmission by T. infestans and to monitor/control other important species involved in transmission to humans;
-
to integrate health surveillance and environmental actions (regarding vectors and reservoirs) with epidemiological surveillance activities.
The change in the epidemiological patterns of the disease in the country required structuring of Chagas Epidemiological Surveillance in the Northern region due to oral transmission. There is a clear need to closely interfacing with the Health Surveillance, with the aim to improve prevention and control measures, besides the definition of reference flows for the diagnosis, treatment and monitoring of disease complications. The high percentage of cases in the Health Ministry database with an undetermined mode of transmission suggests a weakness of the surveillance process and indicates the need for improvement in the opportunity of surveillance activities regarding the detection and investigation of suspect cases 5555. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Doença de Chagas aguda no Brasil: série histórica de 2000 a 2013. Bol Epidemiol. 2015;46(21):1-9. . It also reinforces the need for quality health surveillance actions in the SUS, potentiated through continuing education activities and monitoring and evaluation by the health teams.
The occurrence of suspected cases of acute Chagas disease in Brazil requires immediate notification (within 24 hours after suspicion) using a specific instrument, the Acute Chagas Disease Research Sheet, which is standardized throughout the national territory. For epidemiologic surveillance purposes, the following case definitions have been established 3939. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Guia de Vigilância em Saúde. Brasília: Ministério da Saúde; 2014.:
1. Suspected case of acute Chagas disease
-
Person with persistent fever (more than 7 days) with one or more of the following clinical manifestations: swelling of the face or limbs, rash, lymphadenopathy, hepatomegaly, splenomegaly, acute cardiomyopathy (tachycardia and signs of heart failure), hemorrhagic manifestations, jaundice, Romaña's sign, inoculation chagoma, or:
-
He/she has had a direct contact with triatomines or their excreta; or
-
He/she has received blood/blood products or transplantation of cells/tissues/organs contaminated with T. cruzi ; or
-
He/she has ingested food suspected of being contaminated with T. cruzi ; or
-
He/she is a newborn, from an infected mother.
2. Confirmed case of acute Chagas disease
Laboratory criteria
-
Parasitologic: T. cruzi circulating in the peripheral blood identified by direct parasitologic examination.
-
Serologic: suspected case with serology reactive with anti- T. cruzi IgM antibodies by the indirect immunofluorescent antibody test (IFA); or serology reactive with anti- T. cruzi immunoglobulin G (IgG) antibodies by indirect IFA, with change in IgG concentration of at least two titers after a minimum interval of 21 days in preferably paired samples; or seroconversion by any of the methods [enzyme-linked immunosorbent assay (ELISA), indirect hemagglutination (IHA) or indirect IFA].
Clinical-epidemiological criteria
Cases of acute Chagas disease should always be confirmed by laboratory diagnosis. Only in specific situations can clinical-epidemiological criteria be used for suspected cases, such as during an outbreak of the disease in the acute phase by oral transmission. Such cases have initial negative or non-reactive serologic and parasitologic test results but possess an epidemiological link to confirmed cases of acute Chagas disease by laboratory criteria.
3. Discarded case of acute Chagas disease
This refers to a suspected case with negative or non-reactive laboratory test results, or in whom another illness is diagnosed. Due to the limited clinical expression presented in many cases in the acute phase and the long, silent course of the disease, the reported case can be discarded as being a chronic case or a case of disease reactivation.
4. Definition of a case according possible forms of transmission in Brazil
Reserved for confirmation of cases of acute Chagas disease characterized according to the probable form of transmission.
Confirmed case of Chagas disease by oral transmission
Case in which other routes of transmission are excluded, with epidemiological evidence of a food as a common source of transmission, and usually the simultaneous occurrence of more than one case with an epidemiological link (origin, customs, and cultural elements).
Confirmed case of Chagas disease by vector transmission
Case in which other routes of transmission are excluded, with clinical evidence (Romaña's sign or inoculation chagoma) and/or epidemiological evidence of triatomine occurrence at the site of the infection. Typically, this is observed as an isolated case.
Confirmed case of Chagas disease by laboratory accident
Case that had contact with T. cruzi cultures, exposure to contaminated triatomine feces or blood (human or animal cases) containing infective forms of the parasite. Usually, this occurs in laboratory scientists, health professionals, or researchers.
Confirmed case of Chagas disease by vertical transmission
Newborn from mother with positive parasitologic examination or reactive serology with T. cruzi and presenting:
-
Positive parasitologic examination from birth; or
-
Serologic examination reactive from 9 months of age (before that, maternal antibodies may still be present in the child) and no evidence of infection by other forms of exposure to T. cruzi .
In addition to the surveillance of current outbreaks for the specific surveillance of Chagas disease in Brazil, with the revision of the case definition for epidemiological surveillance of AIDS in the country, from January 2004 throughout the national territory, reactivation Chagas disease (myocarditis and/or meningoencephalitis) has been introduced as indicative of AIDS for SUS 4949. Almeida EA, Ramos Júnior AN, Correia D, Shikanai-Yasuda MA. Co-infection Trypanosoma cruzi /HIV: systematic review (1980-2010). Rev Soc Bras Med Trop. 2011 Nov-Dec;44(6):762-70.) (5454. Ramos Júnior AN. Inclusão da reativação da doença de Chagas como uma condição definidora de AIDS para fins de vigilância epidemiológica no Brasil. Rev Soc Bras Med Trop. 2004 mar-abr;37(2):192-3. .
Although so far, these are not the subject of epidemiologic surveillance in Brazil 5454. Ramos Júnior AN. Inclusão da reativação da doença de Chagas como uma condição definidora de AIDS para fins de vigilância epidemiológica no Brasil. Rev Soc Bras Med Trop. 2004 mar-abr;37(2):192-3. , individuals with the chronic form of Chagas disease should be confirmed, considering the evaluation of people with no indicative picture of febrile illness in the last 60 days and the presence of one of the following tests 3939. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Guia de Vigilância em Saúde. Brasília: Ministério da Saúde; 2014.:
-
Serology: Anti- T. cruzi IgG reactive by two methods based on different principles [ELISA, indirect fluorescent antibody technique (IFAT) or IHA]; or
-
Xenodiagnosis (artificial or indirect): positive for T. cruzi ; or
-
Positive blood culture for T. cruzi in blood samples or cerebrospinal fluid; or, even from postmortem samples.
It is noteworthy that the current epidemiological context in Brazil brings to debate the expansion of the focus and approaches of epidemiological surveillance in the human population for Chagas disease in its chronic phase, considering, among other factors, the possibility of increased sensitivity of epidemiological research actions of other cases in the family, social network and communities (in the acute phase or chronic), as well as the design of spatial contexts of transmission (past or recent, active or not) in a manner integrated with vectors and reservoirs. At the same time, consideration should be given to expanding the criteria for the specific treatment of Chagas disease, considering that millions of infected Brazilian citizens would clearly benefit 1515. Ramos Júnior AN, Carvalho DM. Chagas' disease: past, present and future. Cad Saude Colet. 2009;17(4):787-94.) (5959. Martins-Melo FR, Alencar CH, Ramos Júnior AN, Heukelbach J. Epidemiology of mortality related to Chagas' disease in Brazil, 1999-2007. PLoS Negl Trop Dis. 2012 Feb;6(2):e1508. . Therefore, these questions bring the need to overcome operational problems of the health care network, which should be prepared for longitudinal management of this chronic condition. There is also a need to guarantee sustainable production of medicines, given the potential increase in the demand for specific treatment, and international agreements 3838. Dias JC, Coura JR, Yasuda MAS. The present situation, challenges, and perspectives regarding the production and utilization of effective drugs against human Chagas disease. Rev Soc Bras Med Trop. 2014 Jan-Feb;47(1):123-5. . Extensive national discussion is therefore ongoing, to assess the inclusion of surveillance through mandatory reporting of human cases in the chronic phase in order to qualify the actions of both epidemiology and control, and health care.
Entomological surveillance
Entomological surveillance for Chagas disease should be implemented throughout the country, supported mainly on two pillars: passive surveillance, with the participation of the population in the notification of triatomine; and active surveillance, carried out by entomological teams of the municipalities in partnership with regional health states, without necessarily being based on prior notification by the resident 3939. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Guia de Vigilância em Saúde. Brasília: Ministério da Saúde; 2014. .
The entomological surveillance has been enhanced with a community-based support, responsible for the network of Triatomine Information Posts (TIPs), which is being slowly consolidated in the country 2222. Silveira AC, Dias JCP. O controle da transmissão vetorial. Rev Soc Bras Med Trop. 2011;44 supl 2:52-63. . The home research by the institutional technical team has been recommended in a more systematic and comprehensive form, depending on the existing risk. However, few municipalities have adopted this approach, and those that developed it, generally, have low sensitivity in active search, not detecting at least half of infestation foci. Systematic review addressing impacts of community participation in the entomological surveillance process found that the development of strategies to ensure such participation should be incorporated as a component of entomological surveillance process. This study emphasizes that only the standardized chemical control seems to be consistently effective in eliminating foci of infection 8585. Abad-Franch F, Vega MC, Rolón MS, Santos WS, Rojas de Arias A. Community participation in Chagas disease vector surveillance: systematic review. PLoS Negl Trop Dis. 2011 Jun;5(6):e1207. . It was strengthened, therefore, the necessary participation of individuals, families and communities of endemic areas in all stages of this process, from the planning to actions evaluation, considering the social dimensions in which they fall 1818. Dias JCP. Southern Cone Initiative for the elimination of domestic populations of Triatoma infestans and the interruption of transfusion Chagas disease: historical aspects, present situation, and perspectives. Mem Inst Oswaldo Cruz. 2007 Oct;102 Suppl 1:11-8.) (4343. Ventura-Garcia L, Roura M, Pell C, Posada E, Gascón J, Aldasoro E, et al. Socio-cultural aspects of Chagas disease: a systematic review of qualitative research. PLoS Negl Trop Dis. 2013 Sep 12;7(9):e2410.) (8585. Abad-Franch F, Vega MC, Rolón MS, Santos WS, Rojas de Arias A. Community participation in Chagas disease vector surveillance: systematic review. PLoS Negl Trop Dis. 2011 Jun;5(6):e1207. , and recognizing the need for empowerment and participation of these populations.
The strategies adopted must be appropriate to the reality of each location, since passive surveillance with the participation of the population is a priority in Brazil and is suitable for the vast majority of scenarios, safeguarding the necessary and prompt response by the services to the demands of the population 8585. Abad-Franch F, Vega MC, Rolón MS, Santos WS, Rojas de Arias A. Community participation in Chagas disease vector surveillance: systematic review. PLoS Negl Trop Dis. 2011 Jun;5(6):e1207. . On the other hand, active surveillance should be carried out obligatorily in municipalities with residual foci of T. infestans2323. Coura JR. The main sceneries of Chagas disease transmission. The vectors, blood and oral transmissions: a comprehensive review. Mem Inst Oswaldo Cruz. 2015 May;110(3):277-82.) (3939. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Guia de Vigilância em Saúde. Brasília: Ministério da Saúde; 2014. .
Amplification of T. cruzi transmission, registered in areas previously considered harmless or with no risk for transmission of Chagas disease, such as the Amazon region, required greater attention to the epidemiologic surveillance teams 7676. Shikanai-Yasuda MA, Carvalho NB. Oral transmission of Chagas disease. Clin Infect Dis. 2012 Mar;54(6):845-52.) (9191. Pinto AYN, Valente SA, Valente VC, Ferreira Júnior AG, Coura JR. Fase aguda da doença de Chagas na Amazônia brasileira: estudo de 233 casos do Pará, Amapá e Maranhão observados entre 1988 e 2005. Rev Soc Bras Med Trop. 2008 nov-dec;41(6):602-14.) (9292. Nóbrega AA, Garcia MH, Tatto E, Obara MT, Costa E, Sobel J, et al. Oral transmission of Chagas disease by consumption of açaí palm fruit, Brazil. Emerg Infect Dis. 2009 Apr;15(4):653-5. . In several Brazilian regions, environmental changes favoring the adaptation of vectors to artificial environments, thus establishing new spaces for the appearance of disease, have been observed 2222. Silveira AC, Dias JCP. O controle da transmissão vetorial. Rev Soc Bras Med Trop. 2011;44 supl 2:52-63.) (2323. Coura JR. The main sceneries of Chagas disease transmission. The vectors, blood and oral transmissions: a comprehensive review. Mem Inst Oswaldo Cruz. 2015 May;110(3):277-82.) (3939. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Guia de Vigilância em Saúde. Brasília: Ministério da Saúde; 2014.) (6565. Silveira AC. Os novos desafios e perspectivas futuras do controle. Rev Soc Bras Med Trop. 2011;44 Supl 2:122-4.) (6666. Passos ADC, Silveira AC. Síntese dos resultados dos inquéritos nacionais. Rev Soc Bras Med Trop. 2011;44 supl 2:47-50. . Similarly, it has also been observed the domiciliation of secondary species, even in a much smaller scale to that seen previously to the control of T. infestans2222. Silveira AC, Dias JCP. O controle da transmissão vetorial. Rev Soc Bras Med Trop. 2011;44 supl 2:52-63.) (2323. Coura JR. The main sceneries of Chagas disease transmission. The vectors, blood and oral transmissions: a comprehensive review. Mem Inst Oswaldo Cruz. 2015 May;110(3):277-82.) (3939. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Guia de Vigilância em Saúde. Brasília: Ministério da Saúde; 2014.) (6666. Passos ADC, Silveira AC. Síntese dos resultados dos inquéritos nacionais. Rev Soc Bras Med Trop. 2011;44 supl 2:47-50. .
The emergence or re-emergence of Chagas disease in Brazil characterizes a new epidemiological profile that is independent of the intradomiciliary transmission by T. infestans . Thus, the control of transmission of T. cruzi , in the current scenario, should be examined under a new perspective. Surveillance attention is extremely important, especially mainly maintaining the functioning of sensitive entomological surveillance in the municipalities, in addition to health and environmental educational actions.
Challenges for Brazil
In Brazil, the SUS is based on core values and principles within society, such as universality, equity, integrity, social participation and control, which should be the foundation for policies and programs of public interest. The country is one of the main endemic areas of Chagas disease in the world, in various contexts of great complexity for prevention and control. Due to the high morbidity and mortality burden associated with Chagas disease and their relative invisibility in society, the government must ensure its prioritization for public health and mobilize necessary resources and capabilities with other state actors or non-governmental organizations to its confrontation, by strengthening the singular role that primary care plays for the national health system.
National estimates of millions of people infected by T. cruzi indicate the great responsibility of the country in the technical-scientific and political fields, not only for the prevention of new cases, but especially in the implementation of better decisions and benefits for patients with disease that are both acute and chronic. Brazil and the other Latin American countries have a key role in conducting this process and implementing the action commitments internationally agreed under the seals of PAHO and WHO. The emerging social movement should be valued and promoted, in order to seek the sustainability of actions for coping.
The health surveillance activities for Chagas disease, based on primary health care, should have as principles: territorialization, intersectoriality, focus on people and not the disease, formation of multidisciplinary teams, focus on the needs and expectations of the population and finally, the search quality. The qualified and ethical approach of families affected by Chagas disease should be integrated with promotion and prevention actions, health care for diagnosis and timely treatment, but also to physical, psychological and social rehabilitation. Similarly, for transversal epidemiological surveillance activities in these territories. Therefore, new pacts and agendas must be built, by inserting the Chagas disease as a relevant issue, aimed at ensuring access to inputs necessary for the diagnosis and treatment of the disease in the SUS.
Therefore, in addition to the elements presented previously, the country must continually review the current goals and focuses of epidemiological surveillance of Chagas disease, considering the accumulation of scientific evidence and successful experiences. The social and human development must be strongly linked to promote strategic research focusing on overcoming science, market and public health system failures 111111. Mahoney RT, Morel CMA. Global Health Innovation System (GHIS). Innovation Strategy Today. 2006;2(1):1-12. , with a broad popular participation and the emerging social movement in Chagas disease. The establishment of the International Federation of Associations of People Affected by Chagas Disease (FINDECHAGAS), with the important participation of various representations of Brazil and other countries, strengthens the possibility of raising and catalyzing discussions and decisions from a technical and political point of view based on evidence, to reach the true control and prevention.
The Ministry of Health, in partnership with states, Federal District and municipalities, has played a key inducer role and should be guaranteed the strengthening and sustainability of the National Program for Chagas Disease Control, to enable the scientific evidence, many of which are summarized in this Consensus, in fact be applied in the different realities of the SUS. It is worth to reiterate its strategic role to ensure the sustainability of research funding for control and prevention of Chagas disease in Brazil, more adjusted to different situations and contexts in the country 8383. Abad-Franch F, Santos WS, Schofield CJ. Research needs for Chagas disease prevention. Acta Trop. 2010 Jul-Aug;115(1-2):44-54. .
The expansion of epidemiologic surveillance activities, in addition to acute cases of Chagas disease, integrating cases of the disease in the chronic phase, has to be incorporated strategically in this perspective, expanding network access to health care for diagnosis, timely infection treatment and potential complications of the disease. The antiparasitic treatment should be guaranteed to all cases that have indication for its use, and new safe and effective treatment options must be continuously pursued.
The priority areas for the development of monitoring programs should be designed for both current and future epidemiological scenarios, encompassing the redefinition of instruments and tools with innovative character for the development of monitoring procedures, integrating environmental aspects, reservoirs, vectors, parasites and human population. The macroecologic and eco-geographic perspective should be integrated into the planning of actions in the country, in coordination with the other nations of the Southern Cone Initiative against Chagas disease.
Considering the risk contexts outlined for Brazil, it should be defined the role of other forms of surveillance and how these will be integrated into existing processes of surveillance, including: hemovigilance, technovigilance, pharmacovigilance, vertical transmission, accidents with biologic materials, transplants, HIV/AIDS infections, other immunosuppressive conditions, unusual acute events, such as outbreaks and microepidemics, among others.
It should be emphasized that the integration of Chagas disease control program to other communicable disease control programs (vectorial programs or not) is required and non-communicable chronic diseases in the country - all linked mainly to the Secretariat of Health Surveillance [ Secretaria de Vigilância em Saúde (SVS)], as well as with other areas of the Ministry of Health: Secretariat of Science, Technology and Strategic Inputs (SCTIE), Secretariat of Health Care (SAS), Secretariat of Strategic and Participative Management (SGEP), Secretariat of Labor Management and Education Health (SGTES), and Special Secretariat of Indigenous Health (SESA), aiming to strengthen intersectorial actions for the effective control of Chagas disease.
Faced with such prospects, it is justified the construction of this consensus, which, in addition to focusing on the Brazilian reality, could be a reference tool, encouraging and mobilizing all actors involved in the global fight against Chagas disease.
Vertical transmission of Trypanosoma cruzi
Vertical transmission (from mother to child) of T. cruzi remains a reality, even with the favorable impact of vector control actions and the qualification of the transfusion process in several countries, including 104104. Carlier Y, Torrico F, Sosa-Estani S, Russomando G, Luquetti A, Freilij H, et al. Congenital Chagas disease: recommendations for diagnosis, treatment and control of newborns, siblings and pregnant women. PLoS Negl Trop Dis. 2011 Oct;5(10):e1250.) (105105. Howard EJ, Xiong X, Carlier Y, Sosa-Estani S, Buekens P. Frequency of the congenital transmission of Trypanosoma cruzi: a systematic review and meta-analysis. BJOG. 2014 Jan;121(1):22-33.) (112112. Norman FF, López-Vélez R. Chagas disease and breast-feeding. Emerg Infect Dis. 2013 Oct;19(10):1561-6.) (113113. Gebrekristos HT, Buekens P. Mother-to-Child transmission of Trypanosoma cruzi . J Ped Infect Dis Soc. 2014 Sep;3 Suppl 1:S36-40.) included in Brazil 2020. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Consenso Brasileiro em Doença de Chagas. Rev Soc Bras Med Trop. 2005;38 Supl 3:1-29.) (106106. Martins-Melo FR, Lima MS, Ramos Júnior AN, Alencar CH, Heukelbach J. Prevalence of Chagas disease in pregnant women and congenital transmission of Trypanosoma cruzi in Brazil: a systematic review and meta-analysis. Trop Med Int Health. 2014 Aug;19(8):943-57.) (107107. Gontijo ED, Andrade GMQ, Santos SE, Galvão LMC, Moreira EF, Pinto FS, et al. Triagem neonatal da infecção pelo Trypanosoma cruzi em Minas Gerais, Brasil: transmissão congênita e mapeamento das áreas endêmicas. Epidemiol Serv Saude. 2009 jul-set;18(3):243-54. . The existence of infected women in childbearing age, in endemic countries or not, supports this risk, which tends to be reduced over the next two decades.
In Brazil, in 2011, according to SINASC data were collected about 3 million live births. Based on the estimated prevalence of 1% of T. cruzi infection in women between 25 and 44 years of age 1717. Pan American Health Organization. Estimación cuantitativa de la enfermedad de Chagas en las Americas. Washington: Pan American Health Organization; 2006. and a risk of vertical transmission estimated as maximum of 1% for the State of Minas Gerais, it is estimated that up to 300 children were born infected that year 107107. Gontijo ED, Andrade GMQ, Santos SE, Galvão LMC, Moreira EF, Pinto FS, et al. Triagem neonatal da infecção pelo Trypanosoma cruzi em Minas Gerais, Brasil: transmissão congênita e mapeamento das áreas endêmicas. Epidemiol Serv Saude. 2009 jul-set;18(3):243-54. . Detection of vertical transmission is complicated in practice, since the vast majority of congenital cases is totally asymptomatic 44. Coura JR, Dias JC. Epidemiology, control and surveillance of Chagas disease: 100 years after its discovery. Mem Inst Oswaldo Cruz. 2009 Jul;104 Suppl 1:31-40.) (1010. Dias JCP. Human chagas disease and migration in the context of globalization: some particular aspects. J Trop Med. 2013:789758.) (102102. Torrico F, Alonso-Vega C, Suarez E, Rodriguez P, Torrico MC, Dramaix M, et al. Maternal Trypanosoma cruzi infection, pregnancy outcome, morbidity, and mortality of congenitally infected and non-infected newborns in Bolivia. Am J Trop Med Hyg. 2004 Feb;70(2):201-9.) (104104. Carlier Y, Torrico F, Sosa-Estani S, Russomando G, Luquetti A, Freilij H, et al. Congenital Chagas disease: recommendations for diagnosis, treatment and control of newborns, siblings and pregnant women. PLoS Negl Trop Dis. 2011 Oct;5(10):e1250.) (113113. Gebrekristos HT, Buekens P. Mother-to-Child transmission of Trypanosoma cruzi . J Ped Infect Dis Soc. 2014 Sep;3 Suppl 1:S36-40.) (114114. De Rissio AM, Riarte AR, Martin García M, Esteva MI, Quaglino M, Ruiz AM. Congenital Trypanosoma cruzi infection. Efficacy of its monitoring in an Urban Reference Health Center in non-endemic area of Argentina. Am J Trop Med Hyg. 2010 May;82(5):838-45.) (115115. González-Tomé MI, Rivera M, Camaño I, Norman F, Flores-Chávez M, Rodríguez-Gómez L, et al. Recomendaciones para el diagnóstico, seguimiento y tratamiento de la embarazada y del niño con enfermedad de Chagas. Enferm Infecc Microbiol Clin. 2013 Oct;31(8):535-42.) (116116. Carlier Y, Sosa-Estani S, Luquetti AO, Buekens P. Congenital Chagas disease: an update. Mem Inst Oswaldo Cruz. 2015 May;110(3):363-8. .
It is a complex issue, considering the existence of little scientific evidence of population base. In Brazil, as congenital Chagas disease is considered acute, its notification is compulsory within the epidemiological surveillance activities.
Diagnosis and management of Trypanosoma cruzi infection during pregnancy
It is recommended to carry out a serologic assessment (screening) for T. cruzi infection in all pregnant women living in or are from endemic areas, preferably during their first prenatal visit (2020. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Consenso Brasileiro em Doença de Chagas. Rev Soc Bras Med Trop. 2005;38 Supl 3:1-29.) (117117. Russomando G, Almirón M, Candia N, Franco L, Sánchez Z, Guillen I. Implementación y evaluación de un sistema localmente sustentable de diagnóstico prenatal que permite detectar casos de transmisión congénita de la enfermedad de Chagas en zonas endêmicas del Paraguay. Rev Soc Bras Med Trop. 2005;38 Supl 2:49-54. . The Technical Group on Prevention and Control of Congenital Transmission and Case Management of Congenital Infections (IVa) of WHO Programme on Control of Chagas disease also recommends that screening for those women who have a history of having received blood transfusions performed in endemic areas or who were born in these areas 104104. Carlier Y, Torrico F, Sosa-Estani S, Russomando G, Luquetti A, Freilij H, et al. Congenital Chagas disease: recommendations for diagnosis, treatment and control of newborns, siblings and pregnant women. PLoS Negl Trop Dis. 2011 Oct;5(10):e1250. . Although transmission depends fundamentally by maternal parasitemia, there is no clear evidence on which women may actually be transmitting the infection, which emphasizes the importance of prenatal screening 112112. Norman FF, López-Vélez R. Chagas disease and breast-feeding. Emerg Infect Dis. 2013 Oct;19(10):1561-6.) (116116. Carlier Y, Sosa-Estani S, Luquetti AO, Buekens P. Congenital Chagas disease: an update. Mem Inst Oswaldo Cruz. 2015 May;110(3):363-8.) (117117. Russomando G, Almirón M, Candia N, Franco L, Sánchez Z, Guillen I. Implementación y evaluación de un sistema localmente sustentable de diagnóstico prenatal que permite detectar casos de transmisión congénita de la enfermedad de Chagas en zonas endêmicas del Paraguay. Rev Soc Bras Med Trop. 2005;38 Supl 2:49-54. .
The transmission may occur at any time during pregnancy, possibly being greater in the third quarter, with increasing parasitemia 116116. Carlier Y, Sosa-Estani S, Luquetti AO, Buekens P. Congenital Chagas disease: an update. Mem Inst Oswaldo Cruz. 2015 May;110(3):363-8.) (118118. Brutus L, Castillo H, Bernal C, Salas NA, Schneider D, Santalla JA, et al. Detectable Trypanosoma cruzi parasitemia during pregnancy and delivery as a risk factor for congenital Chagas disease. Am J Trop Med Hyg. 2010 Nov;83(5):1044-7. . On the other hand, the timely diagnosis of infection during pregnancy allows a more qualified care to the infected pregnant women, who should be monitored throughout gestation.
Reports of accidental exposure to benznidazole in pregnant women do not indicate adverse effects on the newborn. However, given the evidence of teratogenicity demonstrated in animals, the specific antiparasitic treatment of infection by T. cruzi is contraindicated during pregnancy and not recommended during breastfeeding 2020. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Consenso Brasileiro em Doença de Chagas. Rev Soc Bras Med Trop. 2005;38 Supl 3:1-29.) (115115. González-Tomé MI, Rivera M, Camaño I, Norman F, Flores-Chávez M, Rodríguez-Gómez L, et al. Recomendaciones para el diagnóstico, seguimiento y tratamiento de la embarazada y del niño con enfermedad de Chagas. Enferm Infecc Microbiol Clin. 2013 Oct;31(8):535-42. , and should be given only after these periods 112112. Norman FF, López-Vélez R. Chagas disease and breast-feeding. Emerg Infect Dis. 2013 Oct;19(10):1561-6. . Accidental exposure to antiparasitic drug during pregnancy is not a criterion for interrupting the gestation 115115. González-Tomé MI, Rivera M, Camaño I, Norman F, Flores-Chávez M, Rodríguez-Gómez L, et al. Recomendaciones para el diagnóstico, seguimiento y tratamiento de la embarazada y del niño con enfermedad de Chagas. Enferm Infecc Microbiol Clin. 2013 Oct;31(8):535-42. .
Studies point to the possible benefit of antiparasitic treatment, with better evolution of Chagas disease 116116. Carlier Y, Sosa-Estani S, Luquetti AO, Buekens P. Congenital Chagas disease: an update. Mem Inst Oswaldo Cruz. 2015 May;110(3):363-8.) (119119. Organización Panamericana de la Salud; Organización Mundial de la Salud. Tratamiento etiológico de la enfermidad de Chagas: conclusiones de reunión de especialistas. Rev Patol Trop. 1999;28(2):247-79.) (120120. Fabbro DL, Streiger ML, Arias ED, Bizai ML, Barco M, Amicone NA. Trypanocide treatment among adults with chronic Chagas disease living in Santa Fe City (Argentina), over a mean follow-up of 21 years: parasitological, serological and clinical evolution. Rev Soc Bras Med Trop. 2007 Jan-Feb;40(1):1-10.) (121121. Lana M, Lopes LA, Martins HR, Bahia MT, Machado-de-Assis GF, Wendling AP, et al. Clinical and laboratory status of patients with chronic Chagas disease living in a vector-controlled area in Minas Gerais, Brazil, before and nine years after aetiological treatment. Mem Inst Oswaldo Cruz. 2009 Dec;104(8):1139-47.) (122122. Pérez-Molina JA, Pérez-Ayala A, Moreno S, Fernández-González MC, Zamora J, López-Velez R. Use of benznidazole to treat chronic Chagas' disease: a systematic review with a meta-analysis. J Antimicrob Chemother. 2009 Dec;64(6):1139-47. and reduction of the risk of vertical transmission in future gestations 109109. Sosa-Estani S, Cura E, Velazquez E, Yampotis C, Segura EL. Etiological treatment of young women infected with Trypanosoma cruzi , and prevention of congenital transmission. Rev Soc Bras Med Trop. 2009 Sep-Oct;42(5):484-7.) (116116. Carlier Y, Sosa-Estani S, Luquetti AO, Buekens P. Congenital Chagas disease: an update. Mem Inst Oswaldo Cruz. 2015 May;110(3):363-8. . High maternal parasitemia is associated with an increased risk of vertical transmission and abortion 103103. Salas NA, Cot M, Schneider D, Mendoza B, Santalla JA, Postigo J, et al. Risk factors and consequences of congenital Chagas disease in Yacuiba, South Bolivia. Trop Med Int Health. 2007 Dec;12(12):1498-505.) (115115. González-Tomé MI, Rivera M, Camaño I, Norman F, Flores-Chávez M, Rodríguez-Gómez L, et al. Recomendaciones para el diagnóstico, seguimiento y tratamiento de la embarazada y del niño con enfermedad de Chagas. Enferm Infecc Microbiol Clin. 2013 Oct;31(8):535-42. . Pregnant women who are in the acute phase of Chagas disease should be evaluated case by case, preferably with the participation of experts, to define the risk-benefit of indicating the antiparasitic treatment. The pregnant women with chronic Chagas disease must be accompanied in referral centers for high-risk pregnancy, as they may present the need for high-complexity care.
The evidence of T. cruzi infection does not justify the abdominal delivery indication (Cesarean), 115115. González-Tomé MI, Rivera M, Camaño I, Norman F, Flores-Chávez M, Rodríguez-Gómez L, et al. Recomendaciones para el diagnóstico, seguimiento y tratamiento de la embarazada y del niño con enfermedad de Chagas. Enferm Infecc Microbiol Clin. 2013 Oct;31(8):535-42. since the congenital T. cruzi infection may result in delayed uterine growth, detected in ultrasound, and premature delivery 102102. Torrico F, Alonso-Vega C, Suarez E, Rodriguez P, Torrico MC, Dramaix M, et al. Maternal Trypanosoma cruzi infection, pregnancy outcome, morbidity, and mortality of congenitally infected and non-infected newborns in Bolivia. Am J Trop Med Hyg. 2004 Feb;70(2):201-9.) (115115. González-Tomé MI, Rivera M, Camaño I, Norman F, Flores-Chávez M, Rodríguez-Gómez L, et al. Recomendaciones para el diagnóstico, seguimiento y tratamiento de la embarazada y del niño con enfermedad de Chagas. Enferm Infecc Microbiol Clin. 2013 Oct;31(8):535-42.) (116116. Carlier Y, Sosa-Estani S, Luquetti AO, Buekens P. Congenital Chagas disease: an update. Mem Inst Oswaldo Cruz. 2015 May;110(3):363-8.) (123123. CarlierI Y, Torrico F, organizers. Congenital infection with T. cruzi: from mechanisms of transmission to strategies for diagnosis and control. Rev Soc Bras Med Trop . 2003 Nov-Dec;36(6):767-71. .
The importance of carrying out all the recommended evaluations during prenatal care, including anti-HIV testing is emphasized. The coinfection of T. cruzi and HIV in pregnant women increases the risk for congenital transmission of T. cruzi , by high parasitemia, also implying an increased morbidity and mortality 2020. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Consenso Brasileiro em Doença de Chagas. Rev Soc Bras Med Trop. 2005;38 Supl 3:1-29.) (116116. Carlier Y, Sosa-Estani S, Luquetti AO, Buekens P. Congenital Chagas disease: an update. Mem Inst Oswaldo Cruz. 2015 May;110(3):363-8.) (120120. Fabbro DL, Streiger ML, Arias ED, Bizai ML, Barco M, Amicone NA. Trypanocide treatment among adults with chronic Chagas disease living in Santa Fe City (Argentina), over a mean follow-up of 21 years: parasitological, serological and clinical evolution. Rev Soc Bras Med Trop. 2007 Jan-Feb;40(1):1-10.) (123123. CarlierI Y, Torrico F, organizers. Congenital infection with T. cruzi: from mechanisms of transmission to strategies for diagnosis and control. Rev Soc Bras Med Trop . 2003 Nov-Dec;36(6):767-71.) (124124. Scapellato PG, Bottaro EG, Rodríguez-Brieschke MT. Mother-child transmission of Chagas disease: could infection with human immunodeficiency virus increase the risk? Rev Soc Bras Med Trop . 2009 Mar-Apr;42(2):107-9. .
After the delivery, the woman should be evaluated as to the conduct of Chagas disease, from the clinic of this disease, aiming at an appropriate specific treatment.
Diagnosis and management in exposed children
The risk of vertical transmission of T. cruzi depends on the level of parasitemia, maternal immune status, infecting strain and placental factors, and might occur at any stage of disease 4141. Bern C, Verastegui M, Gilman RH, Lafuente C, Galdos-Cardenas G, Calderon M, et al. Congenital Trypanosoma cruzi transmission in Santa Cruz, Bolivia. Clin Infect Dis. 2009 Dec 1;49(11):1667-74.) (104104. Carlier Y, Torrico F, Sosa-Estani S, Russomando G, Luquetti A, Freilij H, et al. Congenital Chagas disease: recommendations for diagnosis, treatment and control of newborns, siblings and pregnant women. PLoS Negl Trop Dis. 2011 Oct;5(10):e1250.) (113113. Gebrekristos HT, Buekens P. Mother-to-Child transmission of Trypanosoma cruzi . J Ped Infect Dis Soc. 2014 Sep;3 Suppl 1:S36-40.) (115115. González-Tomé MI, Rivera M, Camaño I, Norman F, Flores-Chávez M, Rodríguez-Gómez L, et al. Recomendaciones para el diagnóstico, seguimiento y tratamiento de la embarazada y del niño con enfermedad de Chagas. Enferm Infecc Microbiol Clin. 2013 Oct;31(8):535-42.) (116116. Carlier Y, Sosa-Estani S, Luquetti AO, Buekens P. Congenital Chagas disease: an update. Mem Inst Oswaldo Cruz. 2015 May;110(3):363-8. .
Although most cases are asymptomatic, in some cases prolonged fever, hepatosplenomegaly, respiratory failure, prematurity, low birth weight and stillbirth may occur. Signs of meningoencephalitis and myocarditis were observed in coinfection with HIV 102102. Torrico F, Alonso-Vega C, Suarez E, Rodriguez P, Torrico MC, Dramaix M, et al. Maternal Trypanosoma cruzi infection, pregnancy outcome, morbidity, and mortality of congenitally infected and non-infected newborns in Bolivia. Am J Trop Med Hyg. 2004 Feb;70(2):201-9.) (104104. Carlier Y, Torrico F, Sosa-Estani S, Russomando G, Luquetti A, Freilij H, et al. Congenital Chagas disease: recommendations for diagnosis, treatment and control of newborns, siblings and pregnant women. PLoS Negl Trop Dis. 2011 Oct;5(10):e1250.) (113113. Gebrekristos HT, Buekens P. Mother-to-Child transmission of Trypanosoma cruzi . J Ped Infect Dis Soc. 2014 Sep;3 Suppl 1:S36-40.) (114114. De Rissio AM, Riarte AR, Martin García M, Esteva MI, Quaglino M, Ruiz AM. Congenital Trypanosoma cruzi infection. Efficacy of its monitoring in an Urban Reference Health Center in non-endemic area of Argentina. Am J Trop Med Hyg. 2010 May;82(5):838-45.) (115115. González-Tomé MI, Rivera M, Camaño I, Norman F, Flores-Chávez M, Rodríguez-Gómez L, et al. Recomendaciones para el diagnóstico, seguimiento y tratamiento de la embarazada y del niño con enfermedad de Chagas. Enferm Infecc Microbiol Clin. 2013 Oct;31(8):535-42.) (123123. CarlierI Y, Torrico F, organizers. Congenital infection with T. cruzi: from mechanisms of transmission to strategies for diagnosis and control. Rev Soc Bras Med Trop . 2003 Nov-Dec;36(6):767-71. . Therefore, they have been recommended as routine assessments in children with clinical signs of congenital Chagas disease: complete blood count, serum biochemistry, urinalysis, chest X-ray, electrocardiogram (ECG), echocardiogram, and cerebral and abdominal ultrasonography 115115. González-Tomé MI, Rivera M, Camaño I, Norman F, Flores-Chávez M, Rodríguez-Gómez L, et al. Recomendaciones para el diagnóstico, seguimiento y tratamiento de la embarazada y del niño con enfermedad de Chagas. Enferm Infecc Microbiol Clin. 2013 Oct;31(8):535-42. .
The strategy employed for the diagnosis of T. cruzi infection depends essentially on the child's age. Children exposed to T. cruzi , by vertical transmission, have circulating maternal IgG antibodies that can be detected by routine serologic testing up to 9 months of age, and their detection in this period, does not necessarily characterize a congenital infection. In turn, the persistence of unaltered titers of anti- T. cruzi antibodies in children from 9 months old, is indicative of congenital infection, and the absence of these antibodies removes the possibility of infection in children 104104. Carlier Y, Torrico F, Sosa-Estani S, Russomando G, Luquetti A, Freilij H, et al. Congenital Chagas disease: recommendations for diagnosis, treatment and control of newborns, siblings and pregnant women. PLoS Negl Trop Dis. 2011 Oct;5(10):e1250.) (123123. CarlierI Y, Torrico F, organizers. Congenital infection with T. cruzi: from mechanisms of transmission to strategies for diagnosis and control. Rev Soc Bras Med Trop . 2003 Nov-Dec;36(6):767-71. .
The diagnosis in suspected cases of Chagas disease by vertical transmission (excluding other forms of transmission), because it is an acute infection, should be confirmed using direct parasitological methods (fresh examination, micro-hematocrit, leukocyte cream and/or Strout method) in cord blood or of the newborn in the first 30 days of age (preferably in the first week of life), with evaluation of two or three samples in the absence of signs and symptoms to expand sensibility 2020. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Consenso Brasileiro em Doença de Chagas. Rev Soc Bras Med Trop. 2005;38 Supl 3:1-29.) (104104. Carlier Y, Torrico F, Sosa-Estani S, Russomando G, Luquetti A, Freilij H, et al. Congenital Chagas disease: recommendations for diagnosis, treatment and control of newborns, siblings and pregnant women. PLoS Negl Trop Dis. 2011 Oct;5(10):e1250.) (123123. CarlierI Y, Torrico F, organizers. Congenital infection with T. cruzi: from mechanisms of transmission to strategies for diagnosis and control. Rev Soc Bras Med Trop . 2003 Nov-Dec;36(6):767-71. .
For symptomatic children or newborn from mother presenting an acute T. cruzi infection or reactivation in the presence of coinfection with HIV/ T. cruzi , these parasitological tests should be performed repeatedly and, if negative, parasitological methods of enrichment and/or molecular (in research laboratories) have been used. However, these are not accessible and standardized for use in the routine of health services 115115. González-Tomé MI, Rivera M, Camaño I, Norman F, Flores-Chávez M, Rodríguez-Gómez L, et al. Recomendaciones para el diagnóstico, seguimiento y tratamiento de la embarazada y del niño con enfermedad de Chagas. Enferm Infecc Microbiol Clin. 2013 Oct;31(8):535-42.) (125125. Bua J, Volta BJ, Velazquez EB, Ruiz AM, Rissio AM, Cardoni RL. Vertical transmission of Trypanosoma cruzi infection: quantification of parasite burden in mothers and their children by parasite DNA amplification. Trans R Soc Trop Med Hyg. 2012 Oct;106(10):623-8. .
In case of negative parasitological/molecular tests in the first months or in the impossibility of using parasitological methods, it is recommended to search for anti- T. cruzi IgG antibodies after the ninth month in two tests 2020. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Consenso Brasileiro em Doença de Chagas. Rev Soc Bras Med Trop. 2005;38 Supl 3:1-29.) (104104. Carlier Y, Torrico F, Sosa-Estani S, Russomando G, Luquetti A, Freilij H, et al. Congenital Chagas disease: recommendations for diagnosis, treatment and control of newborns, siblings and pregnant women. PLoS Negl Trop Dis. 2011 Oct;5(10):e1250.) (123123. CarlierI Y, Torrico F, organizers. Congenital infection with T. cruzi: from mechanisms of transmission to strategies for diagnosis and control. Rev Soc Bras Med Trop . 2003 Nov-Dec;36(6):767-71.) (125125. Bua J, Volta BJ, Velazquez EB, Ruiz AM, Rissio AM, Cardoni RL. Vertical transmission of Trypanosoma cruzi infection: quantification of parasite burden in mothers and their children by parasite DNA amplification. Trans R Soc Trop Med Hyg. 2012 Oct;106(10):623-8.)) . Conventional reagent serology in children during this period is strongly indicative of congenital transmission, especially when excluding possibilities of vector transmission and transfusion 2020. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Consenso Brasileiro em Doença de Chagas. Rev Soc Bras Med Trop. 2005;38 Supl 3:1-29. .
In Brazil, the inclusion of serologic tests searching IgG for T. cruzi in the National Newborn Screening Program ( heel prick blood test ) is recommended, especially in endemic areas for T. cruzi infection, representing an useful and low cost strategy 2020. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Consenso Brasileiro em Doença de Chagas. Rev Soc Bras Med Trop. 2005;38 Supl 3:1-29.) (107107. Gontijo ED, Andrade GMQ, Santos SE, Galvão LMC, Moreira EF, Pinto FS, et al. Triagem neonatal da infecção pelo Trypanosoma cruzi em Minas Gerais, Brasil: transmissão congênita e mapeamento das áreas endêmicas. Epidemiol Serv Saude. 2009 jul-set;18(3):243-54. .
The search for anti- T. cruzi IgM antibodies has a low sensitivity and there are still difficulties to standardize this technique and obtain controls. The use of serological methods employing recombinant antigens, such as the Shed Acute Phase Antigen (SAPA), it may be used if available. There are reports that anti-SAPA maternal antibodies disappear earlier than conventional antibodies in about three months 126126. Mallimaci MC, Sosa-Estani S, Russomando G, Sanchez Z, Sijvarger C, Alvarez IM, et al. Early diagnosis of congenital infection, using shed scute phase antigen, in Ushuaia, Tierra del Fuego, Argentina. Am J Trop Med Hyg. 2010 Jan;82(1):55-9. .
Once the diagnosis of T. cruzi infection has been established, the child must be treated with benznidazole (10-15mg/kg/day in two or three doses for 60 days); this is a well-tolerated regimen 2020. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Consenso Brasileiro em Doença de Chagas. Rev Soc Bras Med Trop. 2005;38 Supl 3:1-29.) (104104. Carlier Y, Torrico F, Sosa-Estani S, Russomando G, Luquetti A, Freilij H, et al. Congenital Chagas disease: recommendations for diagnosis, treatment and control of newborns, siblings and pregnant women. PLoS Negl Trop Dis. 2011 Oct;5(10):e1250.) (115115. González-Tomé MI, Rivera M, Camaño I, Norman F, Flores-Chávez M, Rodríguez-Gómez L, et al. Recomendaciones para el diagnóstico, seguimiento y tratamiento de la embarazada y del niño con enfermedad de Chagas. Enferm Infecc Microbiol Clin. 2013 Oct;31(8):535-42. . There is a clear need to develop formulations in suspension that are more suitable for this treatment, despite the recent advance in providing pediatric tablets (12.5mg) for these clinical situations. Treatment is mandatory in all cases of congenital infection, since it treatment is highly efficacious and safe for the vast majority of children. In addition, studies show a high cure rate (≥ 95%) when treatment is started before 1 year of age, already in the first weeks of life 104104. Carlier Y, Torrico F, Sosa-Estani S, Russomando G, Luquetti A, Freilij H, et al. Congenital Chagas disease: recommendations for diagnosis, treatment and control of newborns, siblings and pregnant women. PLoS Negl Trop Dis. 2011 Oct;5(10):e1250.) (112112. Norman FF, López-Vélez R. Chagas disease and breast-feeding. Emerg Infect Dis. 2013 Oct;19(10):1561-6.) (115115. González-Tomé MI, Rivera M, Camaño I, Norman F, Flores-Chávez M, Rodríguez-Gómez L, et al. Recomendaciones para el diagnóstico, seguimiento y tratamiento de la embarazada y del niño con enfermedad de Chagas. Enferm Infecc Microbiol Clin. 2013 Oct;31(8):535-42.) (127127. Chippaux JP, Clavijo AN, Santalla JA, Postigo JR, Schneider D, Brutus L. Antibody drop in newborns congenitally infected by Trypanosoma cruzi treated with benznidazole. Trop Med Int Health. 2010 Jan;15(1):87-93. .
Clinical and laboratory evaluation (blood count testing) should be performed at the beginning of the treatment and 30, 60, and 90 days after the use of benznidazole. In children with the clinical syndrome of Chagas disease, other complementary assessments should be carried out according to the table presented. The cure control must be performed with serologic tests every six months with titration until the child has two consecutive non-reactive serologic test results. Persistence of positive serology or evidence of positive parasitologic tests may indicate treatment failure and the child must be monitored and evaluated for possible complications of the disease. In refractory cases, benznidazole therapy may be repeated or may be substituted with nifurtimox (up to 15mg/kg/day in two or three doses, for 60 days).
In newborns from infected mothers and asymptomatic, whenever possible, T. cruzi research should be performed. Those with initial negative parasitologic examination or not carried out must be submitted at nine months after birth, for the research with serologic tests of anti- T. cruzi IgG antibodies or conventional serology for infection by T. cruzi107107. Gontijo ED, Andrade GMQ, Santos SE, Galvão LMC, Moreira EF, Pinto FS, et al. Triagem neonatal da infecção pelo Trypanosoma cruzi em Minas Gerais, Brasil: transmissão congênita e mapeamento das áreas endêmicas. Epidemiol Serv Saude. 2009 jul-set;18(3):243-54.) (117117. Russomando G, Almirón M, Candia N, Franco L, Sánchez Z, Guillen I. Implementación y evaluación de un sistema localmente sustentable de diagnóstico prenatal que permite detectar casos de transmisión congénita de la enfermedad de Chagas en zonas endêmicas del Paraguay. Rev Soc Bras Med Trop. 2005;38 Supl 2:49-54.) (127127. Chippaux JP, Clavijo AN, Santalla JA, Postigo JR, Schneider D, Brutus L. Antibody drop in newborns congenitally infected by Trypanosoma cruzi treated with benznidazole. Trop Med Int Health. 2010 Jan;15(1):87-93. . A reactive serologic test after this age necessarily implies the need to initiate antiparasitic treatment. On the other hand, no evidence of positive serology excludes infection.
It is not recommended to suspend breastfeeding of infants whose mothers have Chagas disease in the chronic phase - except in cases where there is bleeding by nipple fissure, when the interruption of breastfeeding in the affected breast would be appropriate - or in situations of high parasitemia, such as in reactivation of Chagas disease or in women in the acute phase of the pathology 112112. Norman FF, López-Vélez R. Chagas disease and breast-feeding. Emerg Infect Dis. 2013 Oct;19(10):1561-6.) (115115. González-Tomé MI, Rivera M, Camaño I, Norman F, Flores-Chávez M, Rodríguez-Gómez L, et al. Recomendaciones para el diagnóstico, seguimiento y tratamiento de la embarazada y del niño con enfermedad de Chagas. Enferm Infecc Microbiol Clin. 2013 Oct;31(8):535-42. . If exposed to breast milk of mother with acute or chronic infection with the presence of nipple cracks, the infant should be monitored for the acquisition of T. cruzi infection during the exposure period, using the same parasitologic and/or serologic criteria described above. In some of these cases, heat treatment of milk prior to administration to infants can be considered 112112. Norman FF, López-Vélez R. Chagas disease and breast-feeding. Emerg Infect Dis. 2013 Oct;19(10):1561-6. .
In T. cruzi /HIV coinfection, it is recommended that all infected mother is advised not to breastfeed, considering that breastfeeding, regardless of the association with Chagas disease, it is associated with an additional 7-22% risk of HIV transmission. Meanwhile, in cases of acute maternal infection by HIV, breastfeeding increases the likelihood of vertical transmission of HIV to 29%. In Brazil, the mother is entitled to receive infant milk formula, at least until your child reaches 6 months of age 128128. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Departamento de DST, Aids e Hepatites Virais. Recomendações para profilaxia da transmissão vertical do HIV e terapia antirretroviral em gestantes. Brasília: Ministério da Saúde; 2010. (Série Manuais, 46). . Figure 1 shows the general flow diagram for the approach of T. cruzi infection in the mother/child pair.
LABORATORY DIAGNOSIS OF TRYPANOSOMA CRUZI INFECTION
In Brazil, an attempt to establish the etiologic diagnosis of Chagas disease should be made in all suspected cases, both in the acute phase and in the chronic phase 2020. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Consenso Brasileiro em Doença de Chagas. Rev Soc Bras Med Trop. 2005;38 Supl 3:1-29.) (3939. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Guia de Vigilância em Saúde. Brasília: Ministério da Saúde; 2014.) (129129. Luquetti AO, Rassi A. Diagnóstico laboratorial da infecção pelo Trypanosoma cruzi . In: Brener Z, Andrade Z, Barral-Netto M, editores. Trypanosoma cruzi e doença de Chagas. Rio de Janeiro: Guanabara-Koogan; 2000. p. 344-78. . Therefore, it is essential to integrate epidemiologic, clinical, and laboratory evidence, in order to increase the degree of predictive and diagnostic accuracy. The complementary diagnosis of infection by T. cruzi through various laboratory techniques should follow defined criteria, depending on the stage of the disease. The following recommendations for diagnosis include the current Brazilian regulations.
Laboratory criteria to define the diagnosis of Chagas disease
Acute phase
Parasitologic criterion
Parasitologic examination is the most appropriate at this stage. This criterion is defined by the presence of T. cruzi trypomastigotes, identified by direct examination of the peripheral blood (with or without centrifugation) using microscopy (with or without staining) 3939. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Guia de Vigilância em Saúde. Brasília: Ministério da Saúde; 2014.) (129129. Luquetti AO, Rassi A. Diagnóstico laboratorial da infecção pelo Trypanosoma cruzi . In: Brener Z, Andrade Z, Barral-Netto M, editores. Trypanosoma cruzi e doença de Chagas. Rio de Janeiro: Guanabara-Koogan; 2000. p. 344-78.) (130130. Chiari E. Parasitological diagnosis. In: Wendel S, Brener Z, Camargo ME, Rassi A, editores. Chagas disease (American Trypanosomiasis): its impact on transfusion and clinical medicine. São Paulo: Editora Sociedade Brasileira de Hematologia e Hemoterapia; 1992. p. 153-64.) (131131. Luquetti AO, Castro AM. Diagnóstico sorológico da doença de Chagas. In: Dias JCP, Coura JR, editores. Clínica e terapêutica da doença de Chagas: uma abordagem prática para o clínico geral. Rio de Janeiro: Editora Fiocruz; 1997. p. 99-113.) (132132. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Programa Nacional de Controle de Chagas. Doença de Chagas aguda: aspectos epidemiológicos, diagnóstico e tratamento: guia de consulta rápida para profissionais de saúde. Rev Patol Trop. 2007 Set-Dez;36(3):1-32 (anexo). .
Simultaneous performance of different modalities of direct parasitological examinations is recommended - fresh examination of trypanosomatids, concentration methods, or thick smear or smear stained slide. When the results of the fresh examination and concentration methods are negative in the first collection, new collections must be carried until the case definition and/or the disappearance of the symptoms of the acute phase, or until the confirmation of another diagnostic hypothesis 2020. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Consenso Brasileiro em Doença de Chagas. Rev Soc Bras Med Trop. 2005;38 Supl 3:1-29.) (3939. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Guia de Vigilância em Saúde. Brasília: Ministério da Saúde; 2014.) (133133. Ministério da Saúde (BR). Recomendações sobre o diagnóstico parasitológico, sorológico e molecular para confirmação da doença de Chagas aguda e crônica. Rev Patol Trop. 2013 out-dez;42(4):475-8. .
Serologic criterion
The serologic criterion is based on indirect methods for diagnosis that may be performed when parasitologic tests are negative but clinical suspicion persists. Such methods have complementary usefulness, and should always be performed in suspected or confirmed cases of acute Chagas disease 3939. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Guia de Vigilância em Saúde. Brasília: Ministério da Saúde; 2014.) (134134. Luquetti AO, Schmuñis GA. Diagnosis of Trypanosoma cruzi infection. In: Telleria J, Tibayrenc M, editors. American Trypanosomiasis Chagas disease: one hundred years of research. Amsterdam: Elsevier; 2010. p. 743-92. .
When the parasite is not identified by direct observation, verifying the presence of anti- T. cruzi IgM antibodies in the peripheral blood is considered suggestive of the acute stage, particularly when combined with the patient's epidemiologic context and clinical events 3939. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Guia de Vigilância em Saúde. Brasília: Ministério da Saúde; 2014.) (135135. Camargo ME, Amato Neto V. Anti- Trypanosoma cruzi IgM antibodies as serological evidence of recent infection. Rev Inst Med Trop São Paulo. 1974 Jul-Aug;16(4):200-2. . This represents a more complex technique and is most suitable in the late acute phase, when repeated direct examinations are negative 129129. Luquetti AO, Rassi A. Diagnóstico laboratorial da infecção pelo Trypanosoma cruzi . In: Brener Z, Andrade Z, Barral-Netto M, editores. Trypanosoma cruzi e doença de Chagas. Rio de Janeiro: Guanabara-Koogan; 2000. p. 344-78.) (133133. Ministério da Saúde (BR). Recomendações sobre o diagnóstico parasitológico, sorológico e molecular para confirmação da doença de Chagas aguda e crônica. Rev Patol Trop. 2013 out-dez;42(4):475-8.) (134134. Luquetti AO, Schmuñis GA. Diagnosis of Trypanosoma cruzi infection. In: Telleria J, Tibayrenc M, editors. American Trypanosomiasis Chagas disease: one hundred years of research. Amsterdam: Elsevier; 2010. p. 743-92. .
Seroconversion to T. cruzi infection is defined by an initial non-reactive serum sample for anti- T. cruzi IgG antibodies, followed by a reactive sample collected 2-4 weeks later, based on an assay that includes both samples simultaneously 133133. Ministério da Saúde (BR). Recomendações sobre o diagnóstico parasitológico, sorológico e molecular para confirmação da doença de Chagas aguda e crônica. Rev Patol Trop. 2013 out-dez;42(4):475-8.) (134134. Luquetti AO, Schmuñis GA. Diagnosis of Trypanosoma cruzi infection. In: Telleria J, Tibayrenc M, editors. American Trypanosomiasis Chagas disease: one hundred years of research. Amsterdam: Elsevier; 2010. p. 743-92. .
Alternately, an increase of at least two titers between two reactive samples taken 2-4 weeks apart, in a clinical and epidemiologic context suggestive of acute Chagas disease, may also be considered diagnostic of acute Chagas disease 3939. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Guia de Vigilância em Saúde. Brasília: Ministério da Saúde; 2014.) (132132. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Programa Nacional de Controle de Chagas. Doença de Chagas aguda: aspectos epidemiológicos, diagnóstico e tratamento: guia de consulta rápida para profissionais de saúde. Rev Patol Trop. 2007 Set-Dez;36(3):1-32 (anexo).) (133133. Ministério da Saúde (BR). Recomendações sobre o diagnóstico parasitológico, sorológico e molecular para confirmação da doença de Chagas aguda e crônica. Rev Patol Trop. 2013 out-dez;42(4):475-8.) (134134. Luquetti AO, Schmuñis GA. Diagnosis of Trypanosoma cruzi infection. In: Telleria J, Tibayrenc M, editors. American Trypanosomiasis Chagas disease: one hundred years of research. Amsterdam: Elsevier; 2010. p. 743-92. .
In situations where vertical transmission is possible, the strategy used for the diagnosis of infection by T. cruzi would depend on the child's age; this is discussed in a specific part of this document. It is noteworthy that every case of congenital transmission means an acute case of Chagas disease and, as such, should be reported and managed 104104. Carlier Y, Torrico F, Sosa-Estani S, Russomando G, Luquetti A, Freilij H, et al. Congenital Chagas disease: recommendations for diagnosis, treatment and control of newborns, siblings and pregnant women. PLoS Negl Trop Dis. 2011 Oct;5(10):e1250. .
Chronic phase
Parasitologic criterion
Due to the low parasite burden in the blood in the chronic phase of Chagas disease, parasitologic methods of enrichment/multiplication, blood culture 136136. Chiari E, Dias JC, Lana M, Chiari CA. Hemocultures for the parasitological diagnosis of human chronic Chagas disease. Rev Soc Bras Med Trop. 1989 Jan-Mar;22(1):19-23.) (137137. Chiari E, Galvão LMC. Diagnóstico parasitológico da doença de Chagas. In: Dias JCP, Coura JR, editores. Clínica e terapêutica da doença de Chagas: uma abordagem prática para o clínico geral. Rio de Janeiro: Fiocruz; 1997. p. 85-97. and xenodiagnosis 138138. Cerisola JA, Rohwedder R, Segura EL, Del Prado CE, Alvarez M, Martini GJW. El xenodiagnóstico. Buenos Aires: Imp Inst Nac Invest Cardiovasc; 1974. have demonstrated low sensitivity, which implies the absence of diagnostic value when the result is negative 2020. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Consenso Brasileiro em Doença de Chagas. Rev Soc Bras Med Trop. 2005;38 Supl 3:1-29.) (133133. Ministério da Saúde (BR). Recomendações sobre o diagnóstico parasitológico, sorológico e molecular para confirmação da doença de Chagas aguda e crônica. Rev Patol Trop. 2013 out-dez;42(4):475-8.) (139139. Castro AM, Luquetti AO, Rassi A, Rassi GG, Chiari E, Galvão LMC. Blood culture and polymerase chain reaction for the diagnosis of the chronic phase of human infection with Trypanosoma cruzi . Parasitol Res. 2002 Oct;88(10):894-900. . When positive, the results are useful, mainly to monitor the specific treatment, or in unusual cases where serology presented inconclusive results 129129. Luquetti AO, Rassi A. Diagnóstico laboratorial da infecção pelo Trypanosoma cruzi . In: Brener Z, Andrade Z, Barral-Netto M, editores. Trypanosoma cruzi e doença de Chagas. Rio de Janeiro: Guanabara-Koogan; 2000. p. 344-78.) (134134. Luquetti AO, Schmuñis GA. Diagnosis of Trypanosoma cruzi infection. In: Telleria J, Tibayrenc M, editors. American Trypanosomiasis Chagas disease: one hundred years of research. Amsterdam: Elsevier; 2010. p. 743-92. .
Serologic criterion
In the chronic phase, the diagnosis is essentially serologic, and must be performed using a test with high sensitivity in conjunction with another having high specificity 2020. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Consenso Brasileiro em Doença de Chagas. Rev Soc Bras Med Trop. 2005;38 Supl 3:1-29.) (3939. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Guia de Vigilância em Saúde. Brasília: Ministério da Saúde; 2014.) (132132. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Programa Nacional de Controle de Chagas. Doença de Chagas aguda: aspectos epidemiológicos, diagnóstico e tratamento: guia de consulta rápida para profissionais de saúde. Rev Patol Trop. 2007 Set-Dez;36(3):1-32 (anexo).) (133133. Ministério da Saúde (BR). Recomendações sobre o diagnóstico parasitológico, sorológico e molecular para confirmação da doença de Chagas aguda e crônica. Rev Patol Trop. 2013 out-dez;42(4):475-8.) (134134. Luquetti AO, Schmuñis GA. Diagnosis of Trypanosoma cruzi infection. In: Telleria J, Tibayrenc M, editors. American Trypanosomiasis Chagas disease: one hundred years of research. Amsterdam: Elsevier; 2010. p. 743-92. .
An individual is considered in the chronic phase of infection if anti- T. cruzi IgG antibodies are detected by means of two serologic tests with different principles/methods having different antigenic preparations. Other diseases (for example, visceral leishmaniasis, virchowian (lepromatous) hanseniasis, and autoimmune diseases) should be considered in the differential diagnosis 140140. Camargo ME. Serological diagnosis: an appraisal of Chagas disease serodiagnosis. In: Wendel S, Brener Z, Camargo ME, Rassi A, editors. Chagas Disease (American Trypanosomiasis), its impact on Transfusion and Clinical Medicine. São Paulo: Sociedade Brasileira de Hematologia e Hemoterapia; 1992. p. 165-78.) (141141. Ferreira AW, Ávila SLM. Doença de Chagas. In.: Ferreira AW, Ávila SLM. Diagnóstico laboratorial das principais doenças infecciosas e auto-imunes. 2 ed. Rio de Janeiro: Guanabara Koogan; 2001. p. 241-9.) (142142. World Health Organization. Control of Chagas disease. Geneva: World Health Organization ; 2002. (Technical Report Series, 905). .
Principles and Guidelines for the Laboratory Diagnosis of Trypanosoma cruzi Infection
Parasitologic diagnosis in the acute phase
The parasitologic diagnosis of Chagas disease at this stage is mainly based on the identification of the parasite, and its sensitivity depends on the level of parasitemia. In the acute phase, the number of parasites in the peripheral blood is high. It is recommended that, if presented with a suspected case of Chagas disease in the acute phase, different methods of direct parasitologic examination be used for immediate and repeated reading, in order to clarify the diagnosis 2020. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Consenso Brasileiro em Doença de Chagas. Rev Soc Bras Med Trop. 2005;38 Supl 3:1-29.) (3939. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Guia de Vigilância em Saúde. Brasília: Ministério da Saúde; 2014.) (104104. Carlier Y, Torrico F, Sosa-Estani S, Russomando G, Luquetti A, Freilij H, et al. Congenital Chagas disease: recommendations for diagnosis, treatment and control of newborns, siblings and pregnant women. PLoS Negl Trop Dis. 2011 Oct;5(10):e1250.) (132132. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Programa Nacional de Controle de Chagas. Doença de Chagas aguda: aspectos epidemiológicos, diagnóstico e tratamento: guia de consulta rápida para profissionais de saúde. Rev Patol Trop. 2007 Set-Dez;36(3):1-32 (anexo).) (133133. Ministério da Saúde (BR). Recomendações sobre o diagnóstico parasitológico, sorológico e molecular para confirmação da doença de Chagas aguda e crônica. Rev Patol Trop. 2013 out-dez;42(4):475-8.) (134134. Luquetti AO, Schmuñis GA. Diagnosis of Trypanosoma cruzi infection. In: Telleria J, Tibayrenc M, editors. American Trypanosomiasis Chagas disease: one hundred years of research. Amsterdam: Elsevier; 2010. p. 743-92. .
The fresh search for trypanosomatids is quick and easy to perform, and is more sensitive than staining the smear. The ideal situation is to collect them from a febrile patient within 30 days of symptoms onset 134134. Luquetti AO, Schmuñis GA. Diagnosis of Trypanosoma cruzi infection. In: Telleria J, Tibayrenc M, editors. American Trypanosomiasis Chagas disease: one hundred years of research. Amsterdam: Elsevier; 2010. p. 743-92. . The examination can be performed directly under the microscope with a drop of blood between the slide and cover slip. Blood collection should be carried out simultaneously for techniques of blood concentration.
The concentration methods are quick and inexpensive (Strout's method, micro-hematocrit and buffy coat). They are recommended as the first choice of diagnostic test for symptomatic cases with more than 30 days of evolution, due to the decline in parasitemia with time. Blood samples should be examined within 24 hours due to possible lysis of the parasites 129129. Luquetti AO, Rassi A. Diagnóstico laboratorial da infecção pelo Trypanosoma cruzi . In: Brener Z, Andrade Z, Barral-Netto M, editores. Trypanosoma cruzi e doença de Chagas. Rio de Janeiro: Guanabara-Koogan; 2000. p. 344-78.) (134134. Luquetti AO, Schmuñis GA. Diagnosis of Trypanosoma cruzi infection. In: Telleria J, Tibayrenc M, editors. American Trypanosomiasis Chagas disease: one hundred years of research. Amsterdam: Elsevier; 2010. p. 743-92. .
Direct examination of a stained thick blood drop or blood smear can be used, but this has lower sensitivity than the methods described above. However, in the Northern region of Brazil, belonging to Legal Amazon, this method is widely used; it is convenient and able to be integrated into malaria diagnostic actions 2020. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Consenso Brasileiro em Doença de Chagas. Rev Soc Bras Med Trop. 2005;38 Supl 3:1-29.) (3939. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Guia de Vigilância em Saúde. Brasília: Ministério da Saúde; 2014. . It also represents an important method for verification and morphologic characterization, especially in geographic areas where the infection by T. rangeli can coexist with T. cruzi .
It is emphasized that in cases with high parasitemia ̶ as in the acute phase of the disease, but also in transfusion transmission and in immunocompromised individuals ̶ parasites can be incidentally found on blood smear when performing a differential leukocyte count 2020. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Consenso Brasileiro em Doença de Chagas. Rev Soc Bras Med Trop. 2005;38 Supl 3:1-29.) (3939. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Guia de Vigilância em Saúde. Brasília: Ministério da Saúde; 2014.) (4949. Almeida EA, Ramos Júnior AN, Correia D, Shikanai-Yasuda MA. Co-infection Trypanosoma cruzi /HIV: systematic review (1980-2010). Rev Soc Bras Med Trop. 2011 Nov-Dec;44(6):762-70.) (5252. Ministério da Saúde (BR). Recomendações para diagnóstico, tratamento e acompanhamento da co-infecção Trypanosoma cruzi: vírus da imunodeficiência humana. Rev Soc Bras Med Trop. 2006 jul-ago;39(4):392-415.) (100100. Pinazo MJ, Miranda B, Rodríguez-Villar C, Altclas J, Brunet Serra M, García-Otero EC, et al. Recommendations for management of Chagas disease in organ and hematopoietic tissue transplantation programs in nonendemic areas. Transplant Rev (Orlando). 2011 Jul;25(3):91-101. .
In cases where a strong clinical and epidemiological suspicion of acute phase T. cruzi infection exists but direct parasitologic examinations are negative, molecular diagnosis using polymerase chain reaction (PCR) methods with hybridization has shown promising results. However, these are in-house tests, performed by only a few research/reference centers. The methods, protocols, and operating procedures should follow the recent recommendations for the standardization for the PCR use 143143. Hijar G, Padilla C, Balbuena J, Bisio MMC, Bailon H, Vega S, et al. Estandarización y validación del uso clínico de la Reacción en Cadena de la Polimerasa para la detección de infección por Trypanosoma cruzi. In: Anales 9º Congreso Peruano de Enfermedades Infecciosas y Tropicales "Jose Neyra Ramirez"; 2009 nov 18-21; Lima. Lima: Sociedad Peruana de Enfermedades Infecciosas y Tropicales; 2009.) (144144. Schijman AG, Bisio M, Orellana L, Sued M, Duffy T, Mejia Jaramillo AM, et al. International study to evaluate PCR methods for detection of Trypanosoma cruzi DNA in blood samples from Chagas disease patients. PLoS Negl Trop Dis. 2011 Jan;5(1):e931. . In Brazil, due to the absence of defined protocols and standard operating procedures, as well as of commercial kits to be routinely used in health surveillance, PCR cannot be considered an isolated diagnostic method to confirm or discard cases of acute or chronic Chagas disease 3939. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Guia de Vigilância em Saúde. Brasília: Ministério da Saúde; 2014. .
Serologic diagnosis in the acute phase
In Brazil, there are currently difficulties in performing serologic tests in patients in the acute phase, due to the lack of registered commercial kits approved by the Brazilian National Health Surveillance Agency (ANVISA) and difficulty in obtaining positive controls for IgM 2020. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Consenso Brasileiro em Doença de Chagas. Rev Soc Bras Med Trop. 2005;38 Supl 3:1-29.) (3939. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Guia de Vigilância em Saúde. Brasília: Ministério da Saúde; 2014.) (133133. Ministério da Saúde (BR). Recomendações sobre o diagnóstico parasitológico, sorológico e molecular para confirmação da doença de Chagas aguda e crônica. Rev Patol Trop. 2013 out-dez;42(4):475-8. . Therefore, it has been traditionally recommended that indirect IFA methods are performed in reference laboratories to detect IgM 145145. Camargo ME. Fluorescent antibody test for the diagnosis of American trypanosomiasis. Technical modification employing preserved culture forms of Trypanosoma cruzi in a slide test. Rev Inst Med Trop São Paulo. 1966 Sep-Oct;8(5):227-35. in addition to the conventional techniques already in use, such as indirect IFA to detect IgG, IHA 146146. Cerisola JA, Chaben MF, Lazari JO. Test de hemaglutinación para el diagnóstico de la enfermedad de Chagas. Prensa Méd Argent. 1962;49:1761-67. , and ELISA 147147. Voller A, Draper C, Bidwell DE, Bartlett A. Microplate enzyme-linked immunosorbent assay for Chagas disease. Lancet. 1975 Feb;305(7904):426-8. .
Parasitologic diagnosis in the chronic phase
Indirect conventional methods for the isolation and identification of T. cruzi (xenodiagnosis and blood culture) have low sensitivity; sensitivity can be increased through repetition 2020. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Consenso Brasileiro em Doença de Chagas. Rev Soc Bras Med Trop. 2005;38 Supl 3:1-29.) (129129. Luquetti AO, Rassi A. Diagnóstico laboratorial da infecção pelo Trypanosoma cruzi . In: Brener Z, Andrade Z, Barral-Netto M, editores. Trypanosoma cruzi e doença de Chagas. Rio de Janeiro: Guanabara-Koogan; 2000. p. 344-78.) (133133. Ministério da Saúde (BR). Recomendações sobre o diagnóstico parasitológico, sorológico e molecular para confirmação da doença de Chagas aguda e crônica. Rev Patol Trop. 2013 out-dez;42(4):475-8.) (148148. Castro CN, Alves MT, Macedo VO. Importância da repetição do xenodiagnóstico para avaliação da parasitemia na fase crônica da doença de Chagas. Rev Soc Bras Med Trop. 1983 abr-jun;16(2):98-103.) (149149. Junqueira AC, Chiari E, Wincker P. Comparison of the polymerase chain reaction with two classical parasitological methods for diagnosis of Chagas disease patients in a north-eastern endemic region of north-eastern Brazil. Trans R Soc Trop Med Hyg. 1996 Mar-Apr;90(2):129-32. . A negative test does not rule out the possibility of infection, but a positive test has an absolute diagnostic value. The PCR at this stage, despite being limited by the lack of standardized protocols, is indicated when serologic tests present an indeterminate result or to determine cure after antiparasitic treatment. PCR should be performed by laboratories with recognized competence, carried out by experts in the field 150150. Britto CC. Usefulness of PCR-based assays to assess drug efficacy in Chagas disease chemotherapy: value and limitations. Mem Inst Oswaldo Cruz. 2009 Jul;104 Suppl 1:122-35. .
Serologic diagnosis in the chronic phase
Diagnosis in the chronic phase is essentially serologic, and should be performed using a test with high sensitivity (ELISA with total antigen or indirect IFA) in conjunction with another method with high specificity (IHA) 129129. Luquetti AO, Rassi A. Diagnóstico laboratorial da infecção pelo Trypanosoma cruzi . In: Brener Z, Andrade Z, Barral-Netto M, editores. Trypanosoma cruzi e doença de Chagas. Rio de Janeiro: Guanabara-Koogan; 2000. p. 344-78.) (133133. Ministério da Saúde (BR). Recomendações sobre o diagnóstico parasitológico, sorológico e molecular para confirmação da doença de Chagas aguda e crônica. Rev Patol Trop. 2013 out-dez;42(4):475-8.) (134134. Luquetti AO, Schmuñis GA. Diagnosis of Trypanosoma cruzi infection. In: Telleria J, Tibayrenc M, editors. American Trypanosomiasis Chagas disease: one hundred years of research. Amsterdam: Elsevier; 2010. p. 743-92. . Known conventional tests (IHA, indirect IF, and ELISA) 131131. Luquetti AO, Castro AM. Diagnóstico sorológico da doença de Chagas. In: Dias JCP, Coura JR, editores. Clínica e terapêutica da doença de Chagas: uma abordagem prática para o clínico geral. Rio de Janeiro: Editora Fiocruz; 1997. p. 99-113. can determine the diagnosis in almost 100% of cases. Unconventional tests (with recombinant antigens, for example) may preferably be used in parallel with another conventional test 151151. Silveira JF, Umezawa ES, Luquetti AO. Chagas disease: recombinant Trypanosoma cruzi antigens for serological diagnosis. Trends Parasitol. 2001 Jun;17(6):286-91. .
Performing reactions in eluates from collected blood on filter paper is not recommended to diagnose the infection, although these are usually used in the screening steps of epidemiological investigations 152152. Zicker F, Smith PG, Luquetti AO, Oliveira OS. Mass screening for Trypanosoma cruzi infections using the immunofluorescence, ELISA and haemagglutination tests on serum samples and on blood eluates from filter-paper. Bull World Health Organ. 1990;68(4):465-71. . The Guerreiro and Machado reaction (or complement fixation) for Chagas disease does not meet the standards currently required. Since it is not commercially available, it is also not indicated 2020. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Consenso Brasileiro em Doença de Chagas. Rev Soc Bras Med Trop. 2005;38 Supl 3:1-29.) (129129. Luquetti AO, Rassi A. Diagnóstico laboratorial da infecção pelo Trypanosoma cruzi . In: Brener Z, Andrade Z, Barral-Netto M, editores. Trypanosoma cruzi e doença de Chagas. Rio de Janeiro: Guanabara-Koogan; 2000. p. 344-78. .
The chemiluminescence test also allows the identification of IgG antibodies. While some kits are available on the market, the technique is still not recommended by the Ministry of Health of Brazil 133133. Ministério da Saúde (BR). Recomendações sobre o diagnóstico parasitológico, sorológico e molecular para confirmação da doença de Chagas aguda e crônica. Rev Patol Trop. 2013 out-dez;42(4):475-8. .
The recent evaluation of 11 rapid test kits used for serologic diagnosis of Chagas disease indicates the potential of these methods. However, further studies should be conducted in the laboratory and in the field to confirm these data, in particular assessing their reproducibility in the context of limited resources or the use of whole blood in the real contexts of endemic and non-endemic areas 153153. Sánchez-Camargo CL, Albajar-Viñas P, Wilkins PP, Nieto J, Leiby DA, Paris L, et al. Comparative evaluation of 11 commercialized rapid diagnostic tests for detecting Trypanosoma cruzi antibodies in serum banks in areas of endemicity and nonendemicity. J Clin Microbiol. 2014 Jul;52(7):2506-12. .
The following flowchart ( Figure 2 ) summarizes the steps for the laboratory diagnosis of T. cruzi infection in the chronic phase of the disease.
Flowchart of steps for the laboratory diagnosis of T. cruzi infection in suspected cases of chronic Chagas
ACUTE PHASE OF CHAGAS DISEASE
Acute Chagas disease is becoming a less frequent event in endemic countries, including Brazil, where control of vector (especially by T. infestans ) and transfusion transmission has altered the epidemiological scenario 2020. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Consenso Brasileiro em Doença de Chagas. Rev Soc Bras Med Trop. 2005;38 Supl 3:1-29.) (3939. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Guia de Vigilância em Saúde. Brasília: Ministério da Saúde; 2014. . On the other hand, the occurrence of cases and outbreaks by oral transmission, transmission by domiciliary vectors without colonization and by extradomiciliary vectors, especially in Amazônia Legal, has gained epidemiological importance 2020. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Consenso Brasileiro em Doença de Chagas. Rev Soc Bras Med Trop. 2005;38 Supl 3:1-29.) (2121. Organização Pan-americana da Saúde. Guia para vigilância, prevenção, controle e manejo clínico da doença de Chagas aguda transmitida por alimentos. Rio de Janeiro: Organização Pan-Americana da Saúde; 2009. (Série de Manuais Técnicos, 12).) (3939. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Guia de Vigilância em Saúde. Brasília: Ministério da Saúde; 2014.) (7676. Shikanai-Yasuda MA, Carvalho NB. Oral transmission of Chagas disease. Clin Infect Dis. 2012 Mar;54(6):845-52.) (154154. Pinto AYN, Valente SAS, Valente VC. Emerging acute Chagas disease in Amazonian Brazil: case reports with serious cardiac involvement. Braz J Infect Dis. 2004 Dec;8(6):454-60.) (155155. Dias JP, Bastos C, Araújo E, Mascarenhas AV, Martins Netto E, Grassi F, et al. Acute Chagas disease outbreak associated with oral transmission. Rev Soc Bras Med Trop. 2008 May-Jun;41(3):296-300.) (156156. Pinto AY, Valente VC, Coura JR, Valente SAS, Junqueira AC, Santos LC, et al. Clinical follow-up of responses to treatment with benznidazol in Amazon: a cohort study of acute Chagas disease. Plos One. 2013 May;8(5):e64450. .
Urbanization and globalization of Chagas disease due to large migrations, both in endemic and non-endemic countries, has contributed to making the epidemiological scenarios even more complex. In addition, reactivation episodes of Chagas disease associated with immunodeficiencies have become emerging challenges for health systems 2020. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Consenso Brasileiro em Doença de Chagas. Rev Soc Bras Med Trop. 2005;38 Supl 3:1-29.) (3939. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Guia de Vigilância em Saúde. Brasília: Ministério da Saúde; 2014.) (4949. Almeida EA, Ramos Júnior AN, Correia D, Shikanai-Yasuda MA. Co-infection Trypanosoma cruzi /HIV: systematic review (1980-2010). Rev Soc Bras Med Trop. 2011 Nov-Dec;44(6):762-70.) (157157. Centers for Disease Control and Prevention. Chagas disease after organ transplantation: United States, 2001. MMWR Morb Mortal Wkly Rep. 2002 Mar;51(10):210-2.) (158158. Centers for Disease Control and Prevention. Chagas disease after organ transplantation: Los Angeles, California, 2006. MMWR Morb Mortal Wkly Rep. 2006 Jul;55(29):798-800.) (159159. Rocha A, Ramos Júnior AN, Sartori AM, Correia D, Gontijo ED, Tatto E, et al. Recomendações para diagnóstico, tratamento e acompanhamento da co-infecção Trypanosoma cruzi: vírus da imunodeficiência humana. Rev Soc Bras Med Trop. 2006 jul-ago;39(4):392-415. . In this sense, the epidemiological investigation of the mode of transmission of Chagas disease is of particular importance, not only in order to determine actions to be implemented, but also because the different routes potentially lead to specificity in the clinical expression of the acute disease 2020. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Consenso Brasileiro em Doença de Chagas. Rev Soc Bras Med Trop. 2005;38 Supl 3:1-29.) (3939. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Guia de Vigilância em Saúde. Brasília: Ministério da Saúde; 2014.) (7676. Shikanai-Yasuda MA, Carvalho NB. Oral transmission of Chagas disease. Clin Infect Dis. 2012 Mar;54(6):845-52.) (132132. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Programa Nacional de Controle de Chagas. Doença de Chagas aguda: aspectos epidemiológicos, diagnóstico e tratamento: guia de consulta rápida para profissionais de saúde. Rev Patol Trop. 2007 Set-Dez;36(3):1-32 (anexo). .
Clinical and complementary examination aspects
Acute phase of Chagas disease by vectorial transmission
The clinical picture is characterized by the appearance of a set of manifestations of variable intensity after an incubation period that is inversely proportional to the load of the inoculum and the inoculation route. There may be a lesion at the inoculation site 2020. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Consenso Brasileiro em Doença de Chagas. Rev Soc Bras Med Trop. 2005;38 Supl 3:1-29.) (2121. Organização Pan-americana da Saúde. Guia para vigilância, prevenção, controle e manejo clínico da doença de Chagas aguda transmitida por alimentos. Rio de Janeiro: Organização Pan-Americana da Saúde; 2009. (Série de Manuais Técnicos, 12).) (3939. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Guia de Vigilância em Saúde. Brasília: Ministério da Saúde; 2014.) (7676. Shikanai-Yasuda MA, Carvalho NB. Oral transmission of Chagas disease. Clin Infect Dis. 2012 Mar;54(6):845-52.) (9191. Pinto AYN, Valente SA, Valente VC, Ferreira Júnior AG, Coura JR. Fase aguda da doença de Chagas na Amazônia brasileira: estudo de 233 casos do Pará, Amapá e Maranhão observados entre 1988 e 2005. Rev Soc Bras Med Trop. 2008 nov-dec;41(6):602-14.) (155155. Dias JP, Bastos C, Araújo E, Mascarenhas AV, Martins Netto E, Grassi F, et al. Acute Chagas disease outbreak associated with oral transmission. Rev Soc Bras Med Trop. 2008 May-Jun;41(3):296-300.) (160160. Rassi A, Rassi Júnior A, Rassi GG. Fase aguda. In: Brener Z, Andrade Z, Barral-Netto M (orgs). Trypanosoma cruzi e Doença de Chagas. 2 ed. Rio de Janeiro: Guanabara Koogan; 2000. p.231-45. . The clinical picture of acute illness in cases of vectorial transmission is polymorphic, ranging from the classical description ̶ a sign of infection at the inoculation site, fever, subcutaneous edema, lymphadenopathy, hepatomegaly, splenomegaly, and evidence of myocarditis and meningoencephalitis ̶ to asymptomatic and oligosymptomatic situations 2020. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Consenso Brasileiro em Doença de Chagas. Rev Soc Bras Med Trop. 2005;38 Supl 3:1-29.) (7676. Shikanai-Yasuda MA, Carvalho NB. Oral transmission of Chagas disease. Clin Infect Dis. 2012 Mar;54(6):845-52.) (9191. Pinto AYN, Valente SA, Valente VC, Ferreira Júnior AG, Coura JR. Fase aguda da doença de Chagas na Amazônia brasileira: estudo de 233 casos do Pará, Amapá e Maranhão observados entre 1988 e 2005. Rev Soc Bras Med Trop. 2008 nov-dec;41(6):602-14.) (160160. Rassi A, Rassi Júnior A, Rassi GG. Fase aguda. In: Brener Z, Andrade Z, Barral-Netto M (orgs). Trypanosoma cruzi e Doença de Chagas. 2 ed. Rio de Janeiro: Guanabara Koogan; 2000. p.231-45. . The incubation period may vary from 4 to 15 days.
The portal of entry is the ocular conjunctiva (Romaña sign) in 50% of cases and the skin (inoculation chagoma) in 25% of cases; it is unknown in the remaining 25% of cases 160160. Rassi A, Rassi Júnior A, Rassi GG. Fase aguda. In: Brener Z, Andrade Z, Barral-Netto M (orgs). Trypanosoma cruzi e Doença de Chagas. 2 ed. Rio de Janeiro: Guanabara Koogan; 2000. p.231-45. . However, it is assumed that the ocular route is the most frequently diagnosed as it is easy to recognize, both by the affected individual and the healthcare team. The Romaña sign is essentially characterized by unilateral, painless, elastic edema of the eyelids with satellite lymph node reaction (mainly pre-auricular). Often, the swelling spreads to the ipsilateral side of the face; its degree is variable and can be severe enough to cause total occlusion of the palpebral fissure. The presence of the Romaña sign is an excellent marker for the diagnosis of the acute phase, allowing recognition of many cases 2020. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Consenso Brasileiro em Doença de Chagas. Rev Soc Bras Med Trop. 2005;38 Supl 3:1-29.) (160160. Rassi A, Rassi Júnior A, Rassi GG. Fase aguda. In: Brener Z, Andrade Z, Barral-Netto M (orgs). Trypanosoma cruzi e Doença de Chagas. 2 ed. Rio de Janeiro: Guanabara Koogan; 2000. p.231-45.) (161161. Romaña C. Acerca de un síntoma inicial de valor para el diagnóstico de la forma aguda de la enfermedad de Chagas: la conjuntivitis schizotripanosómica unilateral: hipótesis sobre la puerta de entrada conjuntival de la enfermedad. Mision de Estudios de Patologia Regional Argentina (MEPRA)1935;22:16-25. . In terms of the skin, the inoculation chagoma presents as a hard, erythematous, slightly painful nodule surrounded by elastic edema. It is also accompanied by satellite lymph node reaction and, sometimes, ulceration. It can be found on any region of the body, especially exposed parts 2020. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Consenso Brasileiro em Doença de Chagas. Rev Soc Bras Med Trop. 2005;38 Supl 3:1-29.) (160160. Rassi A, Rassi Júnior A, Rassi GG. Fase aguda. In: Brener Z, Andrade Z, Barral-Netto M (orgs). Trypanosoma cruzi e Doença de Chagas. 2 ed. Rio de Janeiro: Guanabara Koogan; 2000. p.231-45. .
Along with these signs of entry, general symptoms such as fever, malaise, headache, asthenia, and pyrexia appear. Fever is an initial symptom is almost all cases; it usually does not exceed 39°C; although it can be higher, especially in children. The thermal curve is not characteristic; the temperature record may show a continuous or relapsing and intermittent pattern, with significant increases in the late afternoon 160160. Rassi A, Rassi Júnior A, Rassi GG. Fase aguda. In: Brener Z, Andrade Z, Barral-Netto M (orgs). Trypanosoma cruzi e Doença de Chagas. 2 ed. Rio de Janeiro: Guanabara Koogan; 2000. p.231-45.) (162162. Lugones HS. Enfermedad de Chagas. Diagnóstico de su faz aguda. Santiago del Estero (Argentina): Universidad Católica de Santiago del Estero; 2001. .
Around the second week of the disease, generalized or localized edema may appear (on the face or lower limbs), regardless of the inoculation site or presence/absence of cardiac failure 160160. Rassi A, Rassi Júnior A, Rassi GG. Fase aguda. In: Brener Z, Andrade Z, Barral-Netto M (orgs). Trypanosoma cruzi e Doença de Chagas. 2 ed. Rio de Janeiro: Guanabara Koogan; 2000. p.231-45. . Its consistency can be elastic or soft; the elastic is observed only in young children. The pathogenesis of this edema is unclear; several hypotheses have been suggested.
Usually, lymph nodes are slightly to moderately increased in size, isolated, mobile, smooth, painless, and firm. Of those accessible to palpation, lymphadenopathy is most commonly found in the cervical, axillary, and inguinal regions; this manifestation occurs early in disease 160160. Rassi A, Rassi Júnior A, Rassi GG. Fase aguda. In: Brener Z, Andrade Z, Barral-Netto M (orgs). Trypanosoma cruzi e Doença de Chagas. 2 ed. Rio de Janeiro: Guanabara Koogan; 2000. p.231-45. .
Hepatomegaly and splenomegaly, alone or in combination, are often observed. In general, the increase in liver and spleen volume is small or moderate, the consistency does not change, the edge and the surface are smooth and, sometimes, there is pain on palpation of the liver. These signs are an early part of the clinical framework 160160. Rassi A, Rassi Júnior A, Rassi GG. Fase aguda. In: Brener Z, Andrade Z, Barral-Netto M (orgs). Trypanosoma cruzi e Doença de Chagas. 2 ed. Rio de Janeiro: Guanabara Koogan; 2000. p.231-45.) (163163. Ferreira HO, Pucci H. Estudo evolutivo da doença de Chagas com fase aguda conhecida. Rev Soc Bras Med Trop . 1982 jan-dez;15(1):125-30. .
Although less common, other features (neurologic and cutaneous) may be present. Neurologic manifestations refer to signs of meningoencephalitis (vomiting, agitation, convulsions, opisthotonos, neck stiffness, etc.), which is mainly observed in young children. Meningoencephalitis may present with abnormal cerebrospinal fluid (CSF) findings. The characteristic examination findings are clear CSF, increased cellularity with mild lymphocytosis (<100 cells/mL), low glucose and slightly increased protein levels; it is possible to see trypomastigote forms of T. cruzi after centrifugation of CSF, using specific staining methods 2020. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Consenso Brasileiro em Doença de Chagas. Rev Soc Bras Med Trop. 2005;38 Supl 3:1-29.) (160160. Rassi A, Rassi Júnior A, Rassi GG. Fase aguda. In: Brener Z, Andrade Z, Barral-Netto M (orgs). Trypanosoma cruzi e Doença de Chagas. 2 ed. Rio de Janeiro: Guanabara Koogan; 2000. p.231-45. .
Cutaneous manifestations (rare in Brazil and relatively frequent in Argentina) include rashes (morbilliform, urticariform, and macular) called esquizotripanides and hematogenous chagomas (usually flat formations). These affect skin and subcutaneous tissues without changing their color; are non-adherent to the deep planes; usually painless; and of variable size, from that of a coin to large plaques 160160. Rassi A, Rassi Júnior A, Rassi GG. Fase aguda. In: Brener Z, Andrade Z, Barral-Netto M (orgs). Trypanosoma cruzi e Doença de Chagas. 2 ed. Rio de Janeiro: Guanabara Koogan; 2000. p.231-45. and, according to Lugones (2001), they are more palpable than visible 162162. Lugones HS. Enfermedad de Chagas. Diagnóstico de su faz aguda. Santiago del Estero (Argentina): Universidad Católica de Santiago del Estero; 2001. .
The heart presents changes of greater or lesser severity; with a moderate frequency. However, the clinical, radiologic, or ECG manifestations involved are not significant, are not in keeping with the histologic findings, and may even be absent. On the other hand, it is necessary to bear in mind that serial radiologic and electrocardiographic examinations are required in the short-term, seeking signs of cardiac impairment, given the transience of some of the manifestations 2020. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Consenso Brasileiro em Doença de Chagas. Rev Soc Bras Med Trop. 2005;38 Supl 3:1-29.) (2121. Organização Pan-americana da Saúde. Guia para vigilância, prevenção, controle e manejo clínico da doença de Chagas aguda transmitida por alimentos. Rio de Janeiro: Organização Pan-Americana da Saúde; 2009. (Série de Manuais Técnicos, 12).) (160160. Rassi A, Rassi Júnior A, Rassi GG. Fase aguda. In: Brener Z, Andrade Z, Barral-Netto M (orgs). Trypanosoma cruzi e Doença de Chagas. 2 ed. Rio de Janeiro: Guanabara Koogan; 2000. p.231-45.) (164164. Ferreira HO, Miziara JL. Aspectos clínicos da cardiopatia chagásica aguda. In: Cançado JR, Chuster M (editores). Cardiopatia Chagásica. Belo Horizonte: Imprensa Oficial; 1985. .
The symptoms of acute Chagas myocarditis are essentially identical to those of acute myocarditis of other etiologies, sometimes being masked by other clinical manifestations. Tachycardia is often reported and, generally, not dependent on the degree of temperature increase; in most cases this is observed early. A systolic murmur with functional blow characteristics can be detected in the mitral area. Excitability arrhythmias are only occasionally found. Heart failure, when present, is global; its symptomatology is no different from that related to other etiologies. There may be a degree of arterial hypotension 160160. Rassi A, Rassi Júnior A, Rassi GG. Fase aguda. In: Brener Z, Andrade Z, Barral-Netto M (orgs). Trypanosoma cruzi e Doença de Chagas. 2 ed. Rio de Janeiro: Guanabara Koogan; 2000. p.231-45.) (164164. Ferreira HO, Miziara JL. Aspectos clínicos da cardiopatia chagásica aguda. In: Cançado JR, Chuster M (editores). Cardiopatia Chagásica. Belo Horizonte: Imprensa Oficial; 1985. .
Already in the first weeks of infection, radiologic and/or electrocardiographic changes, of greater or lesser significance, may be observed. These tests are not always simultaneously abnormal. Practically, if one test result is abnormal it has the same diagnostic value as both being abnormal, as both, individually, reveal disorders with the same frequency. When performed in combination and serially, however, these allow more frequent demonstration of impairment 160160. Rassi A, Rassi Júnior A, Rassi GG. Fase aguda. In: Brener Z, Andrade Z, Barral-Netto M (orgs). Trypanosoma cruzi e Doença de Chagas. 2 ed. Rio de Janeiro: Guanabara Koogan; 2000. p.231-45.) (164164. Ferreira HO, Miziara JL. Aspectos clínicos da cardiopatia chagásica aguda. In: Cançado JR, Chuster M (editores). Cardiopatia Chagásica. Belo Horizonte: Imprensa Oficial; 1985. . Therefore, the study of cardiac function by echocardiography is indicated in cases with signs and symptoms of myocarditis.
Radiologically, the heart size can be normal or may be mildly, moderately, or severely increased. Cardiomegaly is global, predominantly as a result of enlargement of the ventricles, but in some cases, due to a dilation of only the left ventricle. The exudate in the pericardial cavity contributes to increase the heart shadow 160160. Rassi A, Rassi Júnior A, Rassi GG. Fase aguda. In: Brener Z, Andrade Z, Barral-Netto M (orgs). Trypanosoma cruzi e Doença de Chagas. 2 ed. Rio de Janeiro: Guanabara Koogan; 2000. p.231-45.) (164164. Ferreira HO, Miziara JL. Aspectos clínicos da cardiopatia chagásica aguda. In: Cançado JR, Chuster M (editores). Cardiopatia Chagásica. Belo Horizonte: Imprensa Oficial; 1985. .
The electrocardiographic changes most frequently encountered are sinus tachycardia, decreased QRS complex voltage, first-degree atrioventricular block (AVB), primary alteration of ventricular repolarization, and increased electrical systole. Arrhythmias, except for first-degree AVB, are only occasionally observed 160160. Rassi A, Rassi Júnior A, Rassi GG. Fase aguda. In: Brener Z, Andrade Z, Barral-Netto M (orgs). Trypanosoma cruzi e Doença de Chagas. 2 ed. Rio de Janeiro: Guanabara Koogan; 2000. p.231-45.) (164164. Ferreira HO, Miziara JL. Aspectos clínicos da cardiopatia chagásica aguda. In: Cançado JR, Chuster M (editores). Cardiopatia Chagásica. Belo Horizonte: Imprensa Oficial; 1985. .
The chronologic asynchrony between the appearance of echocardiography findings and the occurrence of acute phase is not a reason not to perform this examination. In a study conducted in Venezuela that followed 58 patients in the acute phase, the ECG findings proved to be abnormal in 27 (52%); the most important finding was pericardial effusion, present in 42% of the cases, ranging from mild to moderate intensity in 17 patients and severe in 5; in 11 (21%) patients earlier and/or apical dyskinesia was demonstrated, and in 3 (6%) patients, left ventricular dilation was observed 165165. Parada H, Carrasco HA, Añez N, Fuenmayor C, Inglessis I. Cardiac involvement is a constant finding in acute Chagas' disease: a clinical, parasitological and histopathological study. Int J Cardiol. 1997 Jun;60(1):49-54. . A study of 158 patients with acute Chagas disease in the Amazon (most cases due to oral transmission) revealed the presence of 108 changes, with more than one alteration present in the same individual 9191. Pinto AYN, Valente SA, Valente VC, Ferreira Júnior AG, Coura JR. Fase aguda da doença de Chagas na Amazônia brasileira: estudo de 233 casos do Pará, Amapá e Maranhão observados entre 1988 e 2005. Rev Soc Bras Med Trop. 2008 nov-dec;41(6):602-14. . The main abnormal findings were pericardial effusion (from small to large volume), mitral or tricuspid valve regurgitation, and symmetric hypertrophy of the left ventricle. Occasionally, accentuation of sinus tachycardia was observed, in contrast to the reduction in intensity or disappearance of acute manifestations, including fever; this was previously reported by Chagas 7676. Shikanai-Yasuda MA, Carvalho NB. Oral transmission of Chagas disease. Clin Infect Dis. 2012 Mar;54(6):845-52.) (9191. Pinto AYN, Valente SA, Valente VC, Ferreira Júnior AG, Coura JR. Fase aguda da doença de Chagas na Amazônia brasileira: estudo de 233 casos do Pará, Amapá e Maranhão observados entre 1988 e 2005. Rev Soc Bras Med Trop. 2008 nov-dec;41(6):602-14.) (160160. Rassi A, Rassi Júnior A, Rassi GG. Fase aguda. In: Brener Z, Andrade Z, Barral-Netto M (orgs). Trypanosoma cruzi e Doença de Chagas. 2 ed. Rio de Janeiro: Guanabara Koogan; 2000. p.231-45. .
The main nonspecific laboratory abnormalities found are, in order of frequency: anemia, leukopenia, relative lymphocytosis, and an increase, from mild to moderate, of aminotransferases. Also described are thrombocytopenia and, more rarely, thrombocytosis and atypical lymphocytosis 2121. Organização Pan-americana da Saúde. Guia para vigilância, prevenção, controle e manejo clínico da doença de Chagas aguda transmitida por alimentos. Rio de Janeiro: Organização Pan-Americana da Saúde; 2009. (Série de Manuais Técnicos, 12).) (7676. Shikanai-Yasuda MA, Carvalho NB. Oral transmission of Chagas disease. Clin Infect Dis. 2012 Mar;54(6):845-52. .
The complete blood count reveals that the total leukocyte count is generally increased, although it may be normal or slightly decreased, with intense lymphocytosis, plasmacytosis, and related neutropenia. A high percentage of atypical lymphocytes and leukocytoid cells appear after the second week of the disease. As the disease evolves to chronicity, these changes disappear, and eosinophilia emerges 2020. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Consenso Brasileiro em Doença de Chagas. Rev Soc Bras Med Trop. 2005;38 Supl 3:1-29.) (2121. Organização Pan-americana da Saúde. Guia para vigilância, prevenção, controle e manejo clínico da doença de Chagas aguda transmitida por alimentos. Rio de Janeiro: Organização Pan-Americana da Saúde; 2009. (Série de Manuais Técnicos, 12).) (3939. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Guia de Vigilância em Saúde. Brasília: Ministério da Saúde; 2014.) (160160. Rassi A, Rassi Júnior A, Rassi GG. Fase aguda. In: Brener Z, Andrade Z, Barral-Netto M (orgs). Trypanosoma cruzi e Doença de Chagas. 2 ed. Rio de Janeiro: Guanabara Koogan; 2000. p.231-45. .
Acute phase of Chagas disease by blood transmission
In the acute phase of transfusion transmitted Chagas disease, the clinical syndrome is practically identical to that seen when transmitted by triatomines, except for the absence of an inoculation chagoma. Similarly, it must be assessed with complementary examinations. The incubation period can range from 30 to 112 days, being a little longer than that observed in vector-borne transmission, although much shorter periods (such as 8 days) or longer periods (such as 120 days) have been observed 160160. Rassi A, Rassi Júnior A, Rassi GG. Fase aguda. In: Brener Z, Andrade Z, Barral-Netto M (orgs). Trypanosoma cruzi e Doença de Chagas. 2 ed. Rio de Janeiro: Guanabara Koogan; 2000. p.231-45.) (166166. Wendell S. Doença de Chagas transfusional. In: Dias JCP, Coura JR, editores. Clínica e terapêutica da doença de Chagas: uma abordagem prática para o clínico geral. Rio de Janeiro: Fiocruz ; 1997. p. 411-27. . Chagas disease must always be considered in cases presenting with a fever of unknown origin, particularly if the patient under investigation has received a transfusion in a remote or hyperendemic region 166166. Wendell S. Doença de Chagas transfusional. In: Dias JCP, Coura JR, editores. Clínica e terapêutica da doença de Chagas: uma abordagem prática para o clínico geral. Rio de Janeiro: Fiocruz ; 1997. p. 411-27. .
Fever is the most common symptom, affecting 80-100% of cases. It is often the only symptom found. Lymphadenopathy and splenomegaly are also frequently observed, while anemia/pallor, perimalleolar and periorbital edema, skin rash, and hepatomegaly appear in <50% of cases 166166. Wendell S. Doença de Chagas transfusional. In: Dias JCP, Coura JR, editores. Clínica e terapêutica da doença de Chagas: uma abordagem prática para o clínico geral. Rio de Janeiro: Fiocruz ; 1997. p. 411-27. .
Acute phase of Chagas disease by oral transmission
Oral transmission of Chagas disease has been reported in the Amazon and extra-Amazon regions 7676. Shikanai-Yasuda MA, Carvalho NB. Oral transmission of Chagas disease. Clin Infect Dis. 2012 Mar;54(6):845-52.) (9191. Pinto AYN, Valente SA, Valente VC, Ferreira Júnior AG, Coura JR. Fase aguda da doença de Chagas na Amazônia brasileira: estudo de 233 casos do Pará, Amapá e Maranhão observados entre 1988 e 2005. Rev Soc Bras Med Trop. 2008 nov-dec;41(6):602-14.) (9494. Dias JCP. Notas sobre o Trypanosoma cruzi e suas características bio-ecológicas, como agente de enfermidades transmitidas por alimentos. Rev Soc Bras Med Trop. 2006 jul-ago;39(4):370-5.) (167167. Noya BA, Diaz- Bello Z, Colmenares C, Ruiz-Guevara R, Mauriello L, Zavala-Jaspe R, et al. Large urban outbreak of orally acquired acute Chagas disease at a school in Caracas, Venezuela. J Infect Dis. 2010 May;201(9):1308-15. with peculiar clinical characteristics when compared to vectorial transmission, and sometimes with regional differences 2121. Organização Pan-americana da Saúde. Guia para vigilância, prevenção, controle e manejo clínico da doença de Chagas aguda transmitida por alimentos. Rio de Janeiro: Organização Pan-Americana da Saúde; 2009. (Série de Manuais Técnicos, 12). . The major clinical differences between the descriptions of past endemic areas and the Amazon region (in which oral transmission prevails) is the clinical presentation of high morbidity, highlighting the regional epidemiological characteristics regarding the occurrence of a more efficient mode of transmission than the vectorial route 7676. Shikanai-Yasuda MA, Carvalho NB. Oral transmission of Chagas disease. Clin Infect Dis. 2012 Mar;54(6):845-52.) (9494. Dias JCP. Notas sobre o Trypanosoma cruzi e suas características bio-ecológicas, como agente de enfermidades transmitidas por alimentos. Rev Soc Bras Med Trop. 2006 jul-ago;39(4):370-5. . Evaluation by non-specific complementary tests is generally similar to that previously seen.
Evidence suggestive of inoculation (Romaña sign and inoculation chagoma) ̶ typical of the disease induced by vector transmission ̶ is rarely described in cases of oral transmission, emphasizing the clinical differential diagnosis between the two forms of transmission and the low frequency of contact between humans and non-domiciled vectors in this region 9191. Pinto AYN, Valente SA, Valente VC, Ferreira Júnior AG, Coura JR. Fase aguda da doença de Chagas na Amazônia brasileira: estudo de 233 casos do Pará, Amapá e Maranhão observados entre 1988 e 2005. Rev Soc Bras Med Trop. 2008 nov-dec;41(6):602-14.) (9494. Dias JCP. Notas sobre o Trypanosoma cruzi e suas características bio-ecológicas, como agente de enfermidades transmitidas por alimentos. Rev Soc Bras Med Trop. 2006 jul-ago;39(4):370-5.) (160160. Rassi A, Rassi Júnior A, Rassi GG. Fase aguda. In: Brener Z, Andrade Z, Barral-Netto M (orgs). Trypanosoma cruzi e Doença de Chagas. 2 ed. Rio de Janeiro: Guanabara Koogan; 2000. p.231-45. .
The incubation period ranges from 3 to 22 days. The main signs and symptoms recorded in acute Chagas disease are, in order of frequency: prolonged fever, headache, pallor, myalgia, skin rash, and edema of the face and lower limbs 2121. Organização Pan-americana da Saúde. Guia para vigilância, prevenção, controle e manejo clínico da doença de Chagas aguda transmitida por alimentos. Rio de Janeiro: Organização Pan-Americana da Saúde; 2009. (Série de Manuais Técnicos, 12).) (3939. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Guia de Vigilância em Saúde. Brasília: Ministério da Saúde; 2014. . The characteristic signs and symptoms of cardiac impairment include tachycardia without fever, palpitations, dyspnea, and chest pain 7676. Shikanai-Yasuda MA, Carvalho NB. Oral transmission of Chagas disease. Clin Infect Dis. 2012 Mar;54(6):845-52. .
Fever is the predominant manifestation in almost all cases. In general, it is characterized by daily high temperatures, from the morning until the late afternoon. This continues for an average of 18 days (range: 3-25 days). In individuals manifesting prolonged fever, the fever is initially high (38.7-39.0ºC) and is accompanied by chills and generalized pain. After approximately 12-15 days, it begins to settle; the temperatures range between 37.0ºC and 37.8ºC, occur daily, and the fever usually disappears in the afternoon 7676. Shikanai-Yasuda MA, Carvalho NB. Oral transmission of Chagas disease. Clin Infect Dis. 2012 Mar;54(6):845-52.) (9191. Pinto AYN, Valente SA, Valente VC, Ferreira Júnior AG, Coura JR. Fase aguda da doença de Chagas na Amazônia brasileira: estudo de 233 casos do Pará, Amapá e Maranhão observados entre 1988 e 2005. Rev Soc Bras Med Trop. 2008 nov-dec;41(6):602-14. .
Pallor (anemia) occurs from the beginning of the febrile syndrome, in particular on days 3-5, and lasts 20-25 days. Myalgia can be intense; it is often described as an acute stabbing pain, or similar to that experienced in dengue fever (generalized and confused with polyarthralgia) 9191. Pinto AYN, Valente SA, Valente VC, Ferreira Júnior AG, Coura JR. Fase aguda da doença de Chagas na Amazônia brasileira: estudo de 233 casos do Pará, Amapá e Maranhão observados entre 1988 e 2005. Rev Soc Bras Med Trop. 2008 nov-dec;41(6):602-14.) (168168. Shaw J, Lainson R, Fraiha H. Considerações sobre a epidemiologia dos primeiros casos autóctones de doença de Chagas registrados em Belém, Pará, Brasil. Rev Saude Publica. 1969;3(2):153-7. .
When Chagas disease is induced by oral transmission, skin rash appears as an important sign and is associated with abundant parasitemia. It occurs more frequently than when Chagas disease is caused by vectorial transmission. In general, it presents as a macular rash that is not itchy or painful. It occurs on around days 4-8 of disease, usually affecting the chest, back, lower limbs, and neck, and sparing the face and palmoplantar regions. It is relatively transient and most often goes unnoticed by the patient, found only on physical examination 9191. Pinto AYN, Valente SA, Valente VC, Ferreira Júnior AG, Coura JR. Fase aguda da doença de Chagas na Amazônia brasileira: estudo de 233 casos do Pará, Amapá e Maranhão observados entre 1988 e 2005. Rev Soc Bras Med Trop. 2008 nov-dec;41(6):602-14.) (169169. Pinto AYN, Ferreira Júnior AG, Valente VC, Harada GS, Valente SAS. Urban outbreak of acute Chagas disease in Amazon region of Brazil: four-year follow-up after treatment with benznidazole. Rev Panam Salud Publica. 2009 Jan;25(1):77-83.) (170170. Shikanai-Yasuda MA, Marcondes CB, Guedes LA, Siqueira GS, Barone AA, Dias JC, et al. Possible oral transmission of acute Chagas disease in Brasil. Rev Inst Med Trop Sao Paulo. 1991 Sep-Oct;33(5):351-7. .
The edema of the lower limbs generally appears after approximately 12 days of illness and lasts until day 20, coinciding with the disappearance of fever. Invariably, it has the characteristics of inflammatory edema, inelastic and slightly painful. It affects the malleolar region or the entire lower limb. Facial edema can occur in the same period; it has no specific features 7676. Shikanai-Yasuda MA, Carvalho NB. Oral transmission of Chagas disease. Clin Infect Dis. 2012 Mar;54(6):845-52.) (9191. Pinto AYN, Valente SA, Valente VC, Ferreira Júnior AG, Coura JR. Fase aguda da doença de Chagas na Amazônia brasileira: estudo de 233 casos do Pará, Amapá e Maranhão observados entre 1988 e 2005. Rev Soc Bras Med Trop. 2008 nov-dec;41(6):602-14. .
In most cases, acute myocarditis may start slightly before the disappearance of fever, on average from day 15 to 20 of the disease. The main signs and symptoms are dyspnea, palpitations, tachycardia (without fever) and, possibly, precordial chest pain simulating myocardial infarction. Myocarditis is one of the most common complications among patients in the acute phase. ECG and echocardiographic examinations should be performed immediately after diagnosis 7676. Shikanai-Yasuda MA, Carvalho NB. Oral transmission of Chagas disease. Clin Infect Dis. 2012 Mar;54(6):845-52.) (160160. Rassi A, Rassi Júnior A, Rassi GG. Fase aguda. In: Brener Z, Andrade Z, Barral-Netto M (orgs). Trypanosoma cruzi e Doença de Chagas. 2 ed. Rio de Janeiro: Guanabara Koogan; 2000. p.231-45.) (164164. Ferreira HO, Miziara JL. Aspectos clínicos da cardiopatia chagásica aguda. In: Cançado JR, Chuster M (editores). Cardiopatia Chagásica. Belo Horizonte: Imprensa Oficial; 1985. .
Among patients evaluated in the Amazon, 52.3% had altered ECG findings, consisting of diffuse changes with ventricular repolarization abnormalities (VRA), low voltage of the QRS complex, deviation of the electrical axis to the left, and sinus tachycardia, demonstrating some involvement of milder conduction system of the heart and, more often, diffuse signs of inflammation. Cardiac electrical conduction disorders are more evident in adults. Myopericarditis, evidenced by the presence of VRA and pericardial effusion (seen on ECG and echocardiography, respectively) is important in both adults and children 154154. Pinto AYN, Valente SAS, Valente VC. Emerging acute Chagas disease in Amazonian Brazil: case reports with serious cardiac involvement. Braz J Infect Dis. 2004 Dec;8(6):454-60.) (160160. Rassi A, Rassi Júnior A, Rassi GG. Fase aguda. In: Brener Z, Andrade Z, Barral-Netto M (orgs). Trypanosoma cruzi e Doença de Chagas. 2 ed. Rio de Janeiro: Guanabara Koogan; 2000. p.231-45.) (164164. Ferreira HO, Miziara JL. Aspectos clínicos da cardiopatia chagásica aguda. In: Cançado JR, Chuster M (editores). Cardiopatia Chagásica. Belo Horizonte: Imprensa Oficial; 1985. . In outbreaks outside the Amazon Region, a high frequency of pericardial effusion was observed 155155. Dias JP, Bastos C, Araújo E, Mascarenhas AV, Martins Netto E, Grassi F, et al. Acute Chagas disease outbreak associated with oral transmission. Rev Soc Bras Med Trop. 2008 May-Jun;41(3):296-300. , emphasizing the need for early diagnosis in view of severe evolution if not promptly diagnosed and treated.
Pericardial and/or pleural effusion may occur in more than 50% of cases infected via the oral transmission route, suggesting that pericarditis may be more important than involvement of the cardiac electrical conduction system during the acute phase 154154. Pinto AYN, Valente SAS, Valente VC. Emerging acute Chagas disease in Amazonian Brazil: case reports with serious cardiac involvement. Braz J Infect Dis. 2004 Dec;8(6):454-60.) (160160. Rassi A, Rassi Júnior A, Rassi GG. Fase aguda. In: Brener Z, Andrade Z, Barral-Netto M (orgs). Trypanosoma cruzi e Doença de Chagas. 2 ed. Rio de Janeiro: Guanabara Koogan; 2000. p.231-45.) (164164. Ferreira HO, Miziara JL. Aspectos clínicos da cardiopatia chagásica aguda. In: Cançado JR, Chuster M (editores). Cardiopatia Chagásica. Belo Horizonte: Imprensa Oficial; 1985. .
Painful nodules of lower limbs may be associated with edema, and have been invariably recorded in lower limbs of women. The presence of erythema nodosum has been frequently observed in cases of oral transmission. Similar descriptions in children were also recorded in an Argentinian case series, in which some skin and subcutaneous lesions were labeled as hematogenous or metastatic chagomas 169169. Pinto AYN, Ferreira Júnior AG, Valente VC, Harada GS, Valente SAS. Urban outbreak of acute Chagas disease in Amazon region of Brazil: four-year follow-up after treatment with benznidazole. Rev Panam Salud Publica. 2009 Jan;25(1):77-83.) (171171. Jörg ME, Freire RS. Lipochagomata genii or lipochagomata of the cheeks, characteristic lesions of acute trypanosomiasis cruzi in children under three years of age. Acta Trop. 1961;18:318-50. .
Other manifestations associated with this form of transmission can be present, such as epigastric pain, jaundice of the skin and mucous membranes, hepatomegaly, and lymphadenopathy. Lymphadenopathy is common in children, affecting especially cervical lymph nodes chains; it is not generalized. Splenomegaly and diarrhea are rare events 9191. Pinto AYN, Valente SA, Valente VC, Ferreira Júnior AG, Coura JR. Fase aguda da doença de Chagas na Amazônia brasileira: estudo de 233 casos do Pará, Amapá e Maranhão observados entre 1988 e 2005. Rev Soc Bras Med Trop. 2008 nov-dec;41(6):602-14.) (9494. Dias JCP. Notas sobre o Trypanosoma cruzi e suas características bio-ecológicas, como agente de enfermidades transmitidas por alimentos. Rev Soc Bras Med Trop. 2006 jul-ago;39(4):370-5. .
It has been reported that 13.3% of cases are severe. Severity is almost always related to manifestations of acute myocarditis and severe gastrointestinal bleeding. The presence of gastrointestinal bleeding might represent the portal of entry 9494. Dias JCP. Notas sobre o Trypanosoma cruzi e suas características bio-ecológicas, como agente de enfermidades transmitidas por alimentos. Rev Soc Bras Med Trop. 2006 jul-ago;39(4):370-5. , with the inflammatory infiltrate containing amastigotes. There are rare reports of acute meningoencephalitis 172172. Medeiros MB, Guerra JAO, Lacerda MVG. Meningoencephalitis in a patient with acute Chagas disease in the Brazilian Amazon. Rev Soc Bras Med Trop. 2008 Sep-Oct; 41(5):520-1. . Hemorrhage represents a critical scenario of the epidemic that occurred in the City of Navegantes, in the State of Santa Catarina (Southern region of Brazil), providing differential diagnosis for severe diseases that presented a jaundice-hemorrhagic condition, such as leptospirosis, dengue and hantavirosis 7676. Shikanai-Yasuda MA, Carvalho NB. Oral transmission of Chagas disease. Clin Infect Dis. 2012 Mar;54(6):845-52. . The mortality rate was significant among adults aged over 50 years (5.6%).
In just over half (54.1%) of the cases in the Amazon, the diagnosis was confirmed by parasitologic examination 154154. Pinto AYN, Valente SAS, Valente VC. Emerging acute Chagas disease in Amazonian Brazil: case reports with serious cardiac involvement. Braz J Infect Dis. 2004 Dec;8(6):454-60.) (156156. Pinto AY, Valente VC, Coura JR, Valente SAS, Junqueira AC, Santos LC, et al. Clinical follow-up of responses to treatment with benznidazol in Amazon: a cohort study of acute Chagas disease. Plos One. 2013 May;8(5):e64450.) (169169. Pinto AYN, Ferreira Júnior AG, Valente VC, Harada GS, Valente SAS. Urban outbreak of acute Chagas disease in Amazon region of Brazil: four-year follow-up after treatment with benznidazole. Rev Panam Salud Publica. 2009 Jan;25(1):77-83. . Therefore, in cases with negative parasitologic tests, but with strong clinical suspicion, concomitant serologic testing is recommended, considering in particular their higher sensitivity after the first month of clinical history.
Specific treatment
Specific and immediate treatment is indicated in the acute phase, with long-term follow-up to identify the treatment response serologically 7676. Shikanai-Yasuda MA, Carvalho NB. Oral transmission of Chagas disease. Clin Infect Dis. 2012 Mar;54(6):845-52.) (156156. Pinto AY, Valente VC, Coura JR, Valente SAS, Junqueira AC, Santos LC, et al. Clinical follow-up of responses to treatment with benznidazol in Amazon: a cohort study of acute Chagas disease. Plos One. 2013 May;8(5):e64450.) (169169. Pinto AYN, Ferreira Júnior AG, Valente VC, Harada GS, Valente SAS. Urban outbreak of acute Chagas disease in Amazon region of Brazil: four-year follow-up after treatment with benznidazole. Rev Panam Salud Publica. 2009 Jan;25(1):77-83. . For more details, see the specific treatment part of this Consensus.
A case series was reported of 179 patients treated with benznidazole in the acute phase of Chagas disease induced by oral transmission who were followed-up under a research protocol for an average period of 5.6 years. A serologic response to treatment was found in 26.3% of patients, most evident four years after treatment; 2.7% developed a chronic mild-to-moderate cardiomyopathy; and 73.7% had persistently reactive serology, although with a significant decrease in the levels of antibodies 156156. Pinto AY, Valente VC, Coura JR, Valente SAS, Junqueira AC, Santos LC, et al. Clinical follow-up of responses to treatment with benznidazol in Amazon: a cohort study of acute Chagas disease. Plos One. 2013 May;8(5):e64450. .
Reactivation of Chagas disease
The management of cases with reactivation of Chagas disease, in the context of immunosuppression, is an important and emerging public health problem in all countries where people with chronic Chagas disease reside. In particular, the high congenital transmission rate (>50%) in T. cruzi/ HIV coinfected pregnant women is highlighted in the literature. It is advised, therefore, that careful follow-up with clinical assessment and periodic direct search for the parasite be conducted in these women 2020. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Consenso Brasileiro em Doença de Chagas. Rev Soc Bras Med Trop. 2005;38 Supl 3:1-29.) (173173. Freilij H, Altcheh J, Muchinik G. Perinatal human immunodeficiency virus infection and congenital Chagas' disease. Pediatr Infect Dis J. 1995 Feb;14(2):161-2.) (174174. Nisida IV, Amato Neto V, Braz LM, Duarte MI, Umezawa ES. A survey of congenital Chagas' disease, carried out at three health institutions in São Paulo City, Brazil. Rev Inst Med Trop Sao Paulo. 1999 Sep-Oct;41(5):305-11.) (175175. Sartori AM, Ibrahim KY, Nunes Westphalen EV, Braz LM, Oliveira Junior OC, Gakiya E, et al. Manifestations of Chagas disease (American trypanosomiasis ) in patients with HIV/AIDS. Ann Trop Med Parasitol. 2007 Jan;101(1):31-50. .
Reactivation of chronic Chagas disease in previously infected cases is defined as positivity of the following tests, regardless of the presence of other signs and symptoms 159159. Rocha A, Ramos Júnior AN, Sartori AM, Correia D, Gontijo ED, Tatto E, et al. Recomendações para diagnóstico, tratamento e acompanhamento da co-infecção Trypanosoma cruzi: vírus da imunodeficiência humana. Rev Soc Bras Med Trop. 2006 jul-ago;39(4):392-415.:
-
Parasite detected by direct microscopic examination of blood or biological secretions: cerebrospinal fluid, pleura, pericardium, ascitic fluid, among others. In terms of sensitivity, parasite search in buffy coat or micro-hematocrit specimens or sediment from other materials is more sensitive than fresh direct search in biological material or smear. The thick blood smear is more difficult to perform and interpret, and can only be carried out by a trained technician.
-
Histopathologic examination of tissue lesions (panniculitis, myocarditis, encephalitis, enteritis, colpitis, among others), with parasite nests within the acute inflammatory infiltrate.
Reactivation of Chagas disease was first reported in patients immunocompromised by hematologic neoplasms 176176. França LCM, Lemos S, Fleury RN, Melaragno Filho R, Ramos Júnior HA, Pasternak J. Moléstia de Chagas crônica associada à leucemia linfática: ocorrência de encefalite aguda como alteração do estado imunitário. Arq Neuro-Psiquiatr. 1969 mar;27(1):59-66. . From the late 1980s and early 1990s, cases of reactivation of Chagas disease began to be reported in HIV-infected individuals 159159. Rocha A, Ramos Júnior AN, Sartori AM, Correia D, Gontijo ED, Tatto E, et al. Recomendações para diagnóstico, tratamento e acompanhamento da co-infecção Trypanosoma cruzi: vírus da imunodeficiência humana. Rev Soc Bras Med Trop. 2006 jul-ago;39(4):392-415.) (175175. Sartori AM, Ibrahim KY, Nunes Westphalen EV, Braz LM, Oliveira Junior OC, Gakiya E, et al. Manifestations of Chagas disease (American trypanosomiasis ) in patients with HIV/AIDS. Ann Trop Med Parasitol. 2007 Jan;101(1):31-50.) (177177. Spina-França A, Livramento JA, Machado LR, Yasuda N. Anticorpos a Trypanosoma cruzi no líquido cefalorraqueano: pesquisa pelas reações de fixação do complemento e de imunofluorescência. Arq Neuro-Psiquiatr. 1988 dez;46(4):374-8.) (178178. Livramento JA, Machado LR, Spina-França A. Anormalidades do líquido cefalorraqueano em 170 casos de AIDS. Arq Neuro-Psiquiatr. 1989 set;47(3):326-31.) (179179. Del Castilho M, Mendoza G, Oviedo J, Perez Bianco PR, Anselmo AE, Silva M. AIDS and Chagas' disease with central nervous system tumor-like lesion. Am J Med. 1990 Jun;88(6):693-4.) (180180. Ferreira MS, Nishioka SA, Rocha A, Silva AM, Ferreira RG, Olivier W, et al. Acute fatal Trypanosoma cruzi meningoencephalitis in a human immunodeficiency virus-positive hemophiliac patient. Am J Trop Med Hyg. 1991 Dec;45(6):723-7. . In the largest prospective study of T. cruzi/ HIV coinfection ( n = 53 patients), the rate of reactivation was 20.8% 175175. Sartori AM, Ibrahim KY, Nunes Westphalen EV, Braz LM, Oliveira Junior OC, Gakiya E, et al. Manifestations of Chagas disease (American trypanosomiasis ) in patients with HIV/AIDS. Ann Trop Med Parasitol. 2007 Jan;101(1):31-50. . Considering only followed-up patients and excluding hospitalized patients diagnosed with reactivation, this value was approximately 10% 175175. Sartori AM, Ibrahim KY, Nunes Westphalen EV, Braz LM, Oliveira Junior OC, Gakiya E, et al. Manifestations of Chagas disease (American trypanosomiasis ) in patients with HIV/AIDS. Ann Trop Med Parasitol. 2007 Jan;101(1):31-50. .
In kidney transplant recipients, the rates of transmission (from infected donors) and reactivation (in chronically infected patients submitted to transplantation) are 18.7% and 21.7%, respectively 181181. Riarte A, Luna C, Sabatiello R, Sinagra A, Schiavelli R, De Rissio A, et al. Chagas' disease in patients with kidney transplants: 7 years of experience 1989-1996. Clin Infect Dis. 1999 Sep;29(3):561-7. . In the context of heart transplant recipients, a reactivation rate of 26.4-40% was found 182182. Campos SV, Strabelli TM, Amato Neto V, Silva CP, Bacal F, Bocchi EA, et al. Risk factors for Chagas' disease reactivation after heart transplantation. J Heart Lung Transplant. 2008 Jun;27(6):597-602. and, although use of infected donor hearts is prohibited, this has been reported in the absence of donor screening in non-endemic countries 157157. Centers for Disease Control and Prevention. Chagas disease after organ transplantation: United States, 2001. MMWR Morb Mortal Wkly Rep. 2002 Mar;51(10):210-2.) (158158. Centers for Disease Control and Prevention. Chagas disease after organ transplantation: Los Angeles, California, 2006. MMWR Morb Mortal Wkly Rep. 2006 Jul;55(29):798-800. .
In Brazil, in addition to prohibiting the use of infected donor heart and intestine in transplants, it is recommended not to use other transplant organs from donors with infection; exceptional situations should be discussed on a case-by-case basis with specialized teams 183183. Ministério da Saúde (BR). Portaria nº 2.600, de 21 de outubro de 2009. Aprova o Regulamento Técnico do Sistema Nacional de Transplantes. Diário Oficial da República Federativa do Brasil, Brasília (DF), 2009 out 21;Seção 1:77. .
The rate of reactivation in recipients of allogeneic hematopoietic stem cell 184184. Altclas J, Sinagra A, Dictar M, Luna C, Verón MT, De Rissio AM, et al. Chagas disease in bone marrow transplantation: an approach to preemptive therapy. Bone Marrow Transplant. 2005 Jul;36(2):123-9. and liver 185185. Barcán L, Luna C, Clara L, Sinagra A, Valledor A, De Rissio AM, et al. Transmission of T. cruzi infection via liver transplantation to a nonreactive recipient for Chagas' disease. Liver Transpl. 2005 Sep;11(9):1112-6. transplants is 27.3%, and 18.7%, respectively. The use of corticosteroids in association with increased parasitemia and a possible effect with immunosuppressive drugs to treat mesenchymal diseases have been recorded, without a well-defined causal relationship 186186. Rassi A, Amato Neto V, Siqueira AF, Ferriolli Filho F, Amato VS, Rassi Júnior A. Protective effect of benznidazole against parasite reactivation in patients chronically infected with Trypanosoma cruzi and treated with corticoids for associated diseases. Rev Soc Bras Med Trop. 1999 Sep-Oct;32(5):475-82. .
Diagnosing reactivation of Chagas disease
Clinically, the most common manifestations of reactivation are fever, panniculitis (subcutaneous nodules), myocarditis, meningoencephalitis, stroke, and symptoms such as anorexia, myalgia, malaise, and diarrhea. Case series studies suggest that reactivation associated with HIV infection has a higher morbidity and mortality than other causes of immunosuppression, such as in transplant recipients, where the monitoring of organ recipients is suggested, with high success rate for some groups 181181. Riarte A, Luna C, Sabatiello R, Sinagra A, Schiavelli R, De Rissio A, et al. Chagas' disease in patients with kidney transplants: 7 years of experience 1989-1996. Clin Infect Dis. 1999 Sep;29(3):561-7.) (184184. Altclas J, Sinagra A, Dictar M, Luna C, Verón MT, De Rissio AM, et al. Chagas disease in bone marrow transplantation: an approach to preemptive therapy. Bone Marrow Transplant. 2005 Jul;36(2):123-9. .
In reactivation associated with HIV infection, meningoencephalitis (74% of the cases), myocarditis (17.1%), and both occurrences (7.9%) have been reported 4848. Almeida EA, Lima JN, Lages-Silva E, Guariento ME, Aoki FH, Torres-Morales AE, et al. Chagas' disease and HIV co-infection in patients without effective antiretroviral therapy: prevalence, clinical presentation and natural history. Trans R Soc Trop Med Hyg. 2010 Jul;104(7):447-52.) (4949. Almeida EA, Ramos Júnior AN, Correia D, Shikanai-Yasuda MA. Co-infection Trypanosoma cruzi /HIV: systematic review (1980-2010). Rev Soc Bras Med Trop. 2011 Nov-Dec;44(6):762-70.) (159159. Rocha A, Ramos Júnior AN, Sartori AM, Correia D, Gontijo ED, Tatto E, et al. Recomendações para diagnóstico, tratamento e acompanhamento da co-infecção Trypanosoma cruzi: vírus da imunodeficiência humana. Rev Soc Bras Med Trop. 2006 jul-ago;39(4):392-415.) (175175. Sartori AM, Ibrahim KY, Nunes Westphalen EV, Braz LM, Oliveira Junior OC, Gakiya E, et al. Manifestations of Chagas disease (American trypanosomiasis ) in patients with HIV/AIDS. Ann Trop Med Parasitol. 2007 Jan;101(1):31-50.) (187187. Cordova E, Boschi A, Ambrosioni J, Cudos C, Corti M. Reactivation of Chagas disease with central nervous system involvement in HIV-infected patients in Argentina, 1992-2007. Int J Infect Dis. 2008 Nov;12(6):587-92. . Oligosymptomatic forms were diagnosed in mothers who gave birth to children with severe Chagas disease and in patients with prolonged fever in long-term follow-up, with good therapeutic responses to benznidazole. Other rare manifestations of reactivation in immunosuppressed patients have been observed, such as erythema nodosum, myelitis, peritonitis, and colpitis 4949. Almeida EA, Ramos Júnior AN, Correia D, Shikanai-Yasuda MA. Co-infection Trypanosoma cruzi /HIV: systematic review (1980-2010). Rev Soc Bras Med Trop. 2011 Nov-Dec;44(6):762-70.) (159159. Rocha A, Ramos Júnior AN, Sartori AM, Correia D, Gontijo ED, Tatto E, et al. Recomendações para diagnóstico, tratamento e acompanhamento da co-infecção Trypanosoma cruzi: vírus da imunodeficiência humana. Rev Soc Bras Med Trop. 2006 jul-ago;39(4):392-415. .
Complementary examinations, such as computed tomography and/or magnetic resonance imaging of the brain, are fundamental to localize lesions; to differentiate other diagnostic possibilities, such as neurotoxoplasmosis and primary lymphoma of the central nervous system; and to assess whether it is possible to perform a lumbar puncture to obtain CSF for examination to confirm the diagnosis, by recognizing the presence of the parasite (and subsequent cultivation). These tests are more sensitive when performed on CSF than on peripheral blood. CSF examination reveals discrete pleocytosis (with predominance of lymphocytes and monocytes), average or slightly reduced glucose level, and a mild-to-moderately raised protein level 4848. Almeida EA, Lima JN, Lages-Silva E, Guariento ME, Aoki FH, Torres-Morales AE, et al. Chagas' disease and HIV co-infection in patients without effective antiretroviral therapy: prevalence, clinical presentation and natural history. Trans R Soc Trop Med Hyg. 2010 Jul;104(7):447-52.) (4949. Almeida EA, Ramos Júnior AN, Correia D, Shikanai-Yasuda MA. Co-infection Trypanosoma cruzi /HIV: systematic review (1980-2010). Rev Soc Bras Med Trop. 2011 Nov-Dec;44(6):762-70.) (159159. Rocha A, Ramos Júnior AN, Sartori AM, Correia D, Gontijo ED, Tatto E, et al. Recomendações para diagnóstico, tratamento e acompanhamento da co-infecção Trypanosoma cruzi: vírus da imunodeficiência humana. Rev Soc Bras Med Trop. 2006 jul-ago;39(4):392-415. .
In reactivation disease, myocarditis is manifested as heart failure induced by arrhythmia, cardiogenic shock, and low response to symptomatic drugs for congestive heart failure. Echocardiography and other complementary examinations to assess cardiac function are indicated 4949. Almeida EA, Ramos Júnior AN, Correia D, Shikanai-Yasuda MA. Co-infection Trypanosoma cruzi /HIV: systematic review (1980-2010). Rev Soc Bras Med Trop. 2011 Nov-Dec;44(6):762-70.) (159159. Rocha A, Ramos Júnior AN, Sartori AM, Correia D, Gontijo ED, Tatto E, et al. Recomendações para diagnóstico, tratamento e acompanhamento da co-infecção Trypanosoma cruzi: vírus da imunodeficiência humana. Rev Soc Bras Med Trop. 2006 jul-ago;39(4):392-415.) (175175. Sartori AM, Ibrahim KY, Nunes Westphalen EV, Braz LM, Oliveira Junior OC, Gakiya E, et al. Manifestations of Chagas disease (American trypanosomiasis ) in patients with HIV/AIDS. Ann Trop Med Parasitol. 2007 Jan;101(1):31-50. . Diagnosis is facilitated when there is a concomitant increase in parasitemia in peripheral blood; however, reactivation can occur in the tissue without systemic repercussions 4949. Almeida EA, Ramos Júnior AN, Correia D, Shikanai-Yasuda MA. Co-infection Trypanosoma cruzi /HIV: systematic review (1980-2010). Rev Soc Bras Med Trop. 2011 Nov-Dec;44(6):762-70.) (159159. Rocha A, Ramos Júnior AN, Sartori AM, Correia D, Gontijo ED, Tatto E, et al. Recomendações para diagnóstico, tratamento e acompanhamento da co-infecção Trypanosoma cruzi: vírus da imunodeficiência humana. Rev Soc Bras Med Trop. 2006 jul-ago;39(4):392-415. .
The reactivated form of Chagas disease, in its manifestations of meningoencephalitis and/or myocarditis, is officially considered in Brazil as indicative of severe immunodeficiency in individuals over 13 years, and have been included as AIDS-defining conditions since January 2004 5454. Ramos Júnior AN. Inclusão da reativação da doença de Chagas como uma condição definidora de AIDS para fins de vigilância epidemiológica no Brasil. Rev Soc Bras Med Trop. 2004 mar-abr;37(2):192-3. .
For epidemiological surveillance purposes, cases of reactivation of Chagas disease are those that present a definitive diagnosis of T. cruzi infection by parasitologic diagnosis through direct investigation of blood or body fluids (CSF, pericardial, or peritoneal fluid), associated with:
-
Meningoencephalitis: Brain injury image with mass effect (magnetic resonance imaging or computed tomography, with or without injection of contrast medium, showing ring enhancement); and/or
-
Acute myocarditis: Arrhythmias and/or heart failure diagnosed by electrocardiography and echocardiography.
Factors associated with the reactivation of Chagas disease
A CD4 + T lymphocyte count <200 cells/mm 3 is observed in >80% of cases of reactivation 4949. Almeida EA, Ramos Júnior AN, Correia D, Shikanai-Yasuda MA. Co-infection Trypanosoma cruzi /HIV: systematic review (1980-2010). Rev Soc Bras Med Trop. 2011 Nov-Dec;44(6):762-70.) (159159. Rocha A, Ramos Júnior AN, Sartori AM, Correia D, Gontijo ED, Tatto E, et al. Recomendações para diagnóstico, tratamento e acompanhamento da co-infecção Trypanosoma cruzi: vírus da imunodeficiência humana. Rev Soc Bras Med Trop. 2006 jul-ago;39(4):392-415.) (175175. Sartori AM, Ibrahim KY, Nunes Westphalen EV, Braz LM, Oliveira Junior OC, Gakiya E, et al. Manifestations of Chagas disease (American trypanosomiasis ) in patients with HIV/AIDS. Ann Trop Med Parasitol. 2007 Jan;101(1):31-50. . Reactivation is rarely seen in patients with a CD4 + T lymphocyte count >350 cells per mm 3(4848. Almeida EA, Lima JN, Lages-Silva E, Guariento ME, Aoki FH, Torres-Morales AE, et al. Chagas' disease and HIV co-infection in patients without effective antiretroviral therapy: prevalence, clinical presentation and natural history. Trans R Soc Trop Med Hyg. 2010 Jul;104(7):447-52.) (4949. Almeida EA, Ramos Júnior AN, Correia D, Shikanai-Yasuda MA. Co-infection Trypanosoma cruzi /HIV: systematic review (1980-2010). Rev Soc Bras Med Trop. 2011 Nov-Dec;44(6):762-70. . Available data do not suggest the importance of HIV viral load as prognostic factor for reactivation or as factor related to the severity of the cases. On the other hand, HIV viral load increases have been observed in the context of reactivation 4949. Almeida EA, Ramos Júnior AN, Correia D, Shikanai-Yasuda MA. Co-infection Trypanosoma cruzi /HIV: systematic review (1980-2010). Rev Soc Bras Med Trop. 2011 Nov-Dec;44(6):762-70.) (159159. Rocha A, Ramos Júnior AN, Sartori AM, Correia D, Gontijo ED, Tatto E, et al. Recomendações para diagnóstico, tratamento e acompanhamento da co-infecção Trypanosoma cruzi: vírus da imunodeficiência humana. Rev Soc Bras Med Trop. 2006 jul-ago;39(4):392-415. .
Regarding the role of the protozoan, prospective studies have shown the importance of high parasitemia (observed by quantitative methods) as a predictive factor of reactivation (50% of cases). In addition, the participation of different subpopulations of T. cruzi in the genesis of clinical manifestations of reactivation is discussed; this remains controversial in the literature 4848. Almeida EA, Lima JN, Lages-Silva E, Guariento ME, Aoki FH, Torres-Morales AE, et al. Chagas' disease and HIV co-infection in patients without effective antiretroviral therapy: prevalence, clinical presentation and natural history. Trans R Soc Trop Med Hyg. 2010 Jul;104(7):447-52.) (4949. Almeida EA, Ramos Júnior AN, Correia D, Shikanai-Yasuda MA. Co-infection Trypanosoma cruzi /HIV: systematic review (1980-2010). Rev Soc Bras Med Trop. 2011 Nov-Dec;44(6):762-70.) (159159. Rocha A, Ramos Júnior AN, Sartori AM, Correia D, Gontijo ED, Tatto E, et al. Recomendações para diagnóstico, tratamento e acompanhamento da co-infecção Trypanosoma cruzi: vírus da imunodeficiência humana. Rev Soc Bras Med Trop. 2006 jul-ago;39(4):392-415. .
In heart transplant situations, factors associated with reactivation are the number of rejection episodes, the presence of neoplasms, and tangentially, the use of mycophenolate mofetil 182182. Campos SV, Strabelli TM, Amato Neto V, Silva CP, Bacal F, Bocchi EA, et al. Risk factors for Chagas' disease reactivation after heart transplantation. J Heart Lung Transplant. 2008 Jun;27(6):597-602. .
Evolution and prognosis of reactivated Chagas disease
If early treatment is not provided, most patients with AIDS and reactivation of Chagas disease die before or shortly after diagnosis 159159. Rocha A, Ramos Júnior AN, Sartori AM, Correia D, Gontijo ED, Tatto E, et al. Recomendações para diagnóstico, tratamento e acompanhamento da co-infecção Trypanosoma cruzi: vírus da imunodeficiência humana. Rev Soc Bras Med Trop. 2006 jul-ago;39(4):392-415.) (175175. Sartori AM, Ibrahim KY, Nunes Westphalen EV, Braz LM, Oliveira Junior OC, Gakiya E, et al. Manifestations of Chagas disease (American trypanosomiasis ) in patients with HIV/AIDS. Ann Trop Med Parasitol. 2007 Jan;101(1):31-50. . Of patients who complete at least 30 days of treatment, about 80% survive 159159. Rocha A, Ramos Júnior AN, Sartori AM, Correia D, Gontijo ED, Tatto E, et al. Recomendações para diagnóstico, tratamento e acompanhamento da co-infecção Trypanosoma cruzi: vírus da imunodeficiência humana. Rev Soc Bras Med Trop. 2006 jul-ago;39(4):392-415. . The prognosis in cases of HIV infection and reactivation of Chagas disease was reserved before the advent of highly active antiretroviral therapy (HAART), with an estimated average survival of 10 days 2020. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Consenso Brasileiro em Doença de Chagas. Rev Soc Bras Med Trop. 2005;38 Supl 3:1-29. . In immunocompromised patients, the severity depends on the degree of immunosuppression, the parasite load and the sensitivity of the isolated parasite to antiparasitic treatment. The outcomes recorded in kidney transplant recipients were favorable, due to monitoring, diagnosis, and early treatment. The clinical picture is quite severe in newborn infants with congenital infection from born to T. cruzi /HIV coinfected mothers; the morbidity and mortality is high 173173. Freilij H, Altcheh J, Muchinik G. Perinatal human immunodeficiency virus infection and congenital Chagas' disease. Pediatr Infect Dis J. 1995 Feb;14(2):161-2.) (174174. Nisida IV, Amato Neto V, Braz LM, Duarte MI, Umezawa ES. A survey of congenital Chagas' disease, carried out at three health institutions in São Paulo City, Brazil. Rev Inst Med Trop Sao Paulo. 1999 Sep-Oct;41(5):305-11.) (175175. Sartori AM, Ibrahim KY, Nunes Westphalen EV, Braz LM, Oliveira Junior OC, Gakiya E, et al. Manifestations of Chagas disease (American trypanosomiasis ) in patients with HIV/AIDS. Ann Trop Med Parasitol. 2007 Jan;101(1):31-50. .
CHRONIC INDETERMINATE FORM OF CHAGAS DISEASE
The indeterminate chronic form (ICF) of Chagas disease is particularly relevant as it is the most prevalent clinical form the disease. It has a benign nature and low evolutionary potential in the short and medium term 1919. Prata A. Clinical and epidemiological aspects of Chagas disease. Lancet Infect Dis. 2001 Sep;1(2):92-100.) (2020. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Consenso Brasileiro em Doença de Chagas. Rev Soc Bras Med Trop. 2005;38 Supl 3:1-29.) (3939. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Guia de Vigilância em Saúde. Brasília: Ministério da Saúde; 2014.) (188188. Validade do conceito de forma indeterminada de doença de Chagas. Rev Soc Bras Med Trop. 1985 jan-mar;18(1):46.) (189189. Dias JC. The indeterminate form of human chronic Chagas' disease: a clinical epidemiological review. Rev Soc Bras Med Trop. 1989 Jul-Sep;22(3):147-56.) (190190. Macêdo VO. Forma indeterminada da doença de Chagas. In: Dias JCP, Coura JR, editores. Clínica e Terapêutica da doença de Chagas: uma abordagem prática para o clínico geral. Rio de Janeiro: Editora Fiocruz ; 1997. p.135-51.) (191191. Macêdo VO. Indeterminate form of Chagas disease. Mem Inst Oswaldo Cruz. 1999 Sep;94 Suppl 1:311-6. . With the reduced incidence of acute Chagas disease and improved clinical management, the duration of survival of patients with chronic Chagas disease has increased, reflecting a tendency to proportional reduction of ICF with advanced age 1919. Prata A. Clinical and epidemiological aspects of Chagas disease. Lancet Infect Dis. 2001 Sep;1(2):92-100.) (189189. Dias JC. The indeterminate form of human chronic Chagas' disease: a clinical epidemiological review. Rev Soc Bras Med Trop. 1989 Jul-Sep;22(3):147-56.) (192192. Ianni BM, Arteaga E, Frimm CC, Pereira Barretto AC, Mady C. Chagas' heart disease: evolutive evaluation of electrocardiographic and echocardiographic parameters in patients with the indeterminate form. Arq Bras Cardiol. 2001 Jul;77(1):59-62.) (193193. Dias JCP, Machado EMM, Borges ÉC, Moreira EF, Gontijo C, Azeredo BVM. Doença de Chagas em Lassance, MG. Reavaliação clínico-epidemiológica 90 anos após a descoberta de Carlos Chagas. Rev Soc Bras Med Trop. 2002 mar-apr;35(2):167-76.) (194194. Almeida EA, Barbosa Neto RM, Guariento ME, Wanderley JS, Souza ML. Apresentação clínica da doença de Chagas crônica em indivíduos idosos. Rev Soc Bras Med Trop. 2007 mai-jun;40(3):311-5. . On the other hand, since the last Brazilian Consensus in 2005 2020. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Consenso Brasileiro em Doença de Chagas. Rev Soc Bras Med Trop. 2005;38 Supl 3:1-29. , no applicable strategy of clinical or complementary evaluation to the classic concept of ICF has been proposed.
Diagnostic criteria
Patients are considered to have ICF if in a chronic phase of Chagas disease with positive serology and/or positive parasitologic examination for T. cruzi188188. Validade do conceito de forma indeterminada de doença de Chagas. Rev Soc Bras Med Trop. 1985 jan-mar;18(1):46.) (189189. Dias JC. The indeterminate form of human chronic Chagas' disease: a clinical epidemiological review. Rev Soc Bras Med Trop. 1989 Jul-Sep;22(3):147-56. but no typical clinical syndrome specific of the disease; conventional ECG results; and normal radiologic examination of the chest, esophagus, and colon 188188. Validade do conceito de forma indeterminada de doença de Chagas. Rev Soc Bras Med Trop. 1985 jan-mar;18(1):46. . No other complementary examinations in addition to those previously listed, are required to define ICF. The expression indetermined chronic form was first used by Carlos Chagas and stablished by its use 195195. Villela E. Moléstia de Chagas: descrição clínica, 5ª parte. Folha Médica. 1923; 4:65-6.) (196196. Dias E, Laranja FS, Miranda A, Nóbrega G. Chagas' disease: a clinical, epidemiologic and pathologic study. Circulation. 1956 Dec;14(6):1035-60.) (197197. Prata A, Macedo V. Morbidity of Chagas' heart disease. Mem Inst Oswaldo Cruz. 1984;79 Suppl:93-6.) (198198. Organización Mundial de la Salud; Organización Panamericana de la Salud. Aspectos clínicos de la enfermedad de Chagas. Informe de una reunión conjunta OMS/OPAS de investigadores. Bol Oficina Sanit Panam. 1974;76:141-58. . The reference of this term, mainly in the context of scientific research, was consolidated by Brazilian researchers in 1984 2020. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Consenso Brasileiro em Doença de Chagas. Rev Soc Bras Med Trop. 2005;38 Supl 3:1-29.) (142142. World Health Organization. Control of Chagas disease. Geneva: World Health Organization ; 2002. (Technical Report Series, 905).) (188188. Validade do conceito de forma indeterminada de doença de Chagas. Rev Soc Bras Med Trop. 1985 jan-mar;18(1):46.) (199199. Conselho Nacional de Pesquisa. Epidemiologia da doença de Chagas: objetivos e metodologia dos estudos longitudinais. Rio de Janeiro: Conselho Nacional de Pesquisa; 1974. 46 p. Relatório Técnico nº 1. .
In field and operational studies, for asymptomatic cases with normal physical examination, conventional ECG, and chest and esophagus radiography findings, the term ICF was used without performing radiologic investigation of the colon 1919. Prata A. Clinical and epidemiological aspects of Chagas disease. Lancet Infect Dis. 2001 Sep;1(2):92-100.) (189 (190190. Macêdo VO. Forma indeterminada da doença de Chagas. In: Dias JCP, Coura JR, editores. Clínica e Terapêutica da doença de Chagas: uma abordagem prática para o clínico geral. Rio de Janeiro: Editora Fiocruz ; 1997. p.135-51.) (191191. Macêdo VO. Indeterminate form of Chagas disease. Mem Inst Oswaldo Cruz. 1999 Sep;94 Suppl 1:311-6. . This conduct is justified, taking into account the low availability and feasibility of these complementary tests in the real context of the national health systems in endemic regions.
Treatment and clinical follow-up
Specific antiparasitic treatment is indicated for all cases with ICF (see specific section on antiparasitic treatment in this Consensus) 2020. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Consenso Brasileiro em Doença de Chagas. Rev Soc Bras Med Trop. 2005;38 Supl 3:1-29.) (3939. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Guia de Vigilância em Saúde. Brasília: Ministério da Saúde; 2014. .
While the ECG finding is normal, the prognosis of cases with ICF of Chagas disease is similar to the general population, since serial repetition of these examinations can detect evolution to a cardiac form of Chagas disease 2020. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Consenso Brasileiro em Doença de Chagas. Rev Soc Bras Med Trop. 2005;38 Supl 3:1-29.) (189189. Dias JC. The indeterminate form of human chronic Chagas' disease: a clinical epidemiological review. Rev Soc Bras Med Trop. 1989 Jul-Sep;22(3):147-56.) (192192. Ianni BM, Arteaga E, Frimm CC, Pereira Barretto AC, Mady C. Chagas' heart disease: evolutive evaluation of electrocardiographic and echocardiographic parameters in patients with the indeterminate form. Arq Bras Cardiol. 2001 Jul;77(1):59-62.) (197197. Prata A, Macedo V. Morbidity of Chagas' heart disease. Mem Inst Oswaldo Cruz. 1984;79 Suppl:93-6. . Based on this key concept, is not recommended performing other routine complementary tests while the ECG finding is normal 1919. Prata A. Clinical and epidemiological aspects of Chagas disease. Lancet Infect Dis. 2001 Sep;1(2):92-100.) (2020. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Consenso Brasileiro em Doença de Chagas. Rev Soc Bras Med Trop. 2005;38 Supl 3:1-29.) (189189. Dias JC. The indeterminate form of human chronic Chagas' disease: a clinical epidemiological review. Rev Soc Bras Med Trop. 1989 Jul-Sep;22(3):147-56.) (190190. Macêdo VO. Forma indeterminada da doença de Chagas. In: Dias JCP, Coura JR, editores. Clínica e Terapêutica da doença de Chagas: uma abordagem prática para o clínico geral. Rio de Janeiro: Editora Fiocruz ; 1997. p.135-51.) (191191. Macêdo VO. Indeterminate form of Chagas disease. Mem Inst Oswaldo Cruz. 1999 Sep;94 Suppl 1:311-6. .
Cases with ICF should be advised not to donate blood, and in principle, tissues or organs 2020. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Consenso Brasileiro em Doença de Chagas. Rev Soc Bras Med Trop. 2005;38 Supl 3:1-29.) (3939. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Guia de Vigilância em Saúde. Brasília: Ministério da Saúde; 2014. . Due to the benign nature of ICF, the (still common) practice of requesting specific serologic exams for Chagas disease in pre-employment assessments for labor purposes and periodic examinations conducted by institutions and/or public or private companies, is not justified 192192. Ianni BM, Arteaga E, Frimm CC, Pereira Barretto AC, Mady C. Chagas' heart disease: evolutive evaluation of electrocardiographic and echocardiographic parameters in patients with the indeterminate form. Arq Bras Cardiol. 2001 Jul;77(1):59-62.) (200200. Oliveira Júnior W. Forma indeterminada da doença de Chagas: implicações médico-trabalhistas. Arq Bras Cardiol. 1990 fev;54(2):89-91.) (201201. Guariento ME, Camilo MVF, Camargo AMA. Working conditions of Chagas' disease patients in a large Brazilian city. Cad Saude Publica. 1999 Apr-Jun;15(2):381-6.) (202202. Dias JCP, Siqueira IOC, Dias RB. Doença de Chagas: diagnóstico e manejo na atenção primária à saúde. In: Gusso G, Lopes JMC. Tratado de Medicina de Família e Comunidade: 2 Volumes: Princípios, Formação e Prática. Porto Alegre: Artmed; 2012. p. 2067-80. . As for other complementary tests, these may be ordered according to the specific labor activities that the individual will perform. Similarly, in these cases, temporary or permanent removal from work activity is not recommended 192192. Ianni BM, Arteaga E, Frimm CC, Pereira Barretto AC, Mady C. Chagas' heart disease: evolutive evaluation of electrocardiographic and echocardiographic parameters in patients with the indeterminate form. Arq Bras Cardiol. 2001 Jul;77(1):59-62.) (200200. Oliveira Júnior W. Forma indeterminada da doença de Chagas: implicações médico-trabalhistas. Arq Bras Cardiol. 1990 fev;54(2):89-91.) (201201. Guariento ME, Camilo MVF, Camargo AMA. Working conditions of Chagas' disease patients in a large Brazilian city. Cad Saude Publica. 1999 Apr-Jun;15(2):381-6.) (202202. Dias JCP, Siqueira IOC, Dias RB. Doença de Chagas: diagnóstico e manejo na atenção primária à saúde. In: Gusso G, Lopes JMC. Tratado de Medicina de Família e Comunidade: 2 Volumes: Princípios, Formação e Prática. Porto Alegre: Artmed; 2012. p. 2067-80.) (203203. Castro I, Andrade JP, Paola AAV, Vilas-Boas F, Oliveira GMM, Marin-Neto JA, et al. Sociedade Brasileira de Cardiologia. I Diretriz Latino-Americana para o diagnóstico e tratamento da cardiopatia chagásica. Arq Bras Cardiol. 2011 Aug;97(2 Suppl 3):1-48. .
Engaging in usual, regular physical activity is not contraindicated in patients with ICF. For professional activities involving individual and collective risk that may require high demand of physical effort and/or psychological stress, the adoption of a complementary specific cardiologic evaluation is recommended. There is no restriction of sexual activity for patients with ICF 192192. Ianni BM, Arteaga E, Frimm CC, Pereira Barretto AC, Mady C. Chagas' heart disease: evolutive evaluation of electrocardiographic and echocardiographic parameters in patients with the indeterminate form. Arq Bras Cardiol. 2001 Jul;77(1):59-62.) (200200. Oliveira Júnior W. Forma indeterminada da doença de Chagas: implicações médico-trabalhistas. Arq Bras Cardiol. 1990 fev;54(2):89-91.) (201201. Guariento ME, Camilo MVF, Camargo AMA. Working conditions of Chagas' disease patients in a large Brazilian city. Cad Saude Publica. 1999 Apr-Jun;15(2):381-6.) (202202. Dias JCP, Siqueira IOC, Dias RB. Doença de Chagas: diagnóstico e manejo na atenção primária à saúde. In: Gusso G, Lopes JMC. Tratado de Medicina de Família e Comunidade: 2 Volumes: Princípios, Formação e Prática. Porto Alegre: Artmed; 2012. p. 2067-80. .
ICF carriers must be assessed annually, preferably in the primary healthcare services, by means of medical evaluation and conventional ECG (2020. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Consenso Brasileiro em Doença de Chagas. Rev Soc Bras Med Trop. 2005;38 Supl 3:1-29.) (202202. Dias JCP, Siqueira IOC, Dias RB. Doença de Chagas: diagnóstico e manejo na atenção primária à saúde. In: Gusso G, Lopes JMC. Tratado de Medicina de Família e Comunidade: 2 Volumes: Princípios, Formação e Prática. Porto Alegre: Artmed; 2012. p. 2067-80. . If disease evolution is observed, these cases should be referred to more specialized healthcare services with the aim to provide the patient with a more precise direction (to define the appropriate investigation and treatment interventions) for the integrated management with primary health care teams (up- and down-referral).
Indeterminate chronic form does not interfere with the management of associated diseases and does not justify neglecting the follow-up and treatment of comorbidities that may possibly be present 4949. Almeida EA, Ramos Júnior AN, Correia D, Shikanai-Yasuda MA. Co-infection Trypanosoma cruzi /HIV: systematic review (1980-2010). Rev Soc Bras Med Trop. 2011 Nov-Dec;44(6):762-70.) (197197. Prata A, Macedo V. Morbidity of Chagas' heart disease. Mem Inst Oswaldo Cruz. 1984;79 Suppl:93-6.) (203203. Castro I, Andrade JP, Paola AAV, Vilas-Boas F, Oliveira GMM, Marin-Neto JA, et al. Sociedade Brasileira de Cardiologia. I Diretriz Latino-Americana para o diagnóstico e tratamento da cardiopatia chagásica. Arq Bras Cardiol. 2011 Aug;97(2 Suppl 3):1-48.) (204204. Ozaki Y, Guariento ME, Almeida EA. Quality of life and depressive symptoms in Chagas disease patients. Qual Life Res. 2011 Feb;20(1):133-8. . If immunodeficiency (acquired or induced) occurs in patients with ICF, special attention should be given to possible reactivation of Chagas disease (see specific section on reactivation of Chagas disease) 2020. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Consenso Brasileiro em Doença de Chagas. Rev Soc Bras Med Trop. 2005;38 Supl 3:1-29.) (4949. Almeida EA, Ramos Júnior AN, Correia D, Shikanai-Yasuda MA. Co-infection Trypanosoma cruzi /HIV: systematic review (1980-2010). Rev Soc Bras Med Trop. 2011 Nov-Dec;44(6):762-70.) (159159. Rocha A, Ramos Júnior AN, Sartori AM, Correia D, Gontijo ED, Tatto E, et al. Recomendações para diagnóstico, tratamento e acompanhamento da co-infecção Trypanosoma cruzi: vírus da imunodeficiência humana. Rev Soc Bras Med Trop. 2006 jul-ago;39(4):392-415. . The surgical risk for the patient with ICF does not differ from that observed in the general, uninfected population 2020. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Consenso Brasileiro em Doença de Chagas. Rev Soc Bras Med Trop. 2005;38 Supl 3:1-29.) (194194. Almeida EA, Barbosa Neto RM, Guariento ME, Wanderley JS, Souza ML. Apresentação clínica da doença de Chagas crônica em indivíduos idosos. Rev Soc Bras Med Trop. 2007 mai-jun;40(3):311-5.) (200200. Oliveira Júnior W. Forma indeterminada da doença de Chagas: implicações médico-trabalhistas. Arq Bras Cardiol. 1990 fev;54(2):89-91.) (202202. Dias JCP, Siqueira IOC, Dias RB. Doença de Chagas: diagnóstico e manejo na atenção primária à saúde. In: Gusso G, Lopes JMC. Tratado de Medicina de Família e Comunidade: 2 Volumes: Princípios, Formação e Prática. Porto Alegre: Artmed; 2012. p. 2067-80.) (203203. Castro I, Andrade JP, Paola AAV, Vilas-Boas F, Oliveira GMM, Marin-Neto JA, et al. Sociedade Brasileira de Cardiologia. I Diretriz Latino-Americana para o diagnóstico e tratamento da cardiopatia chagásica. Arq Bras Cardiol. 2011 Aug;97(2 Suppl 3):1-48. .
Regarding pregnant women infected with T. cruzi with this classification, attention should be paid to the possibility of vertical transmission, and opportunities should be created for adequate assessment of the newborn infant (see specific section on vertical transmission) 2020. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Consenso Brasileiro em Doença de Chagas. Rev Soc Bras Med Trop. 2005;38 Supl 3:1-29.) (3939. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Guia de Vigilância em Saúde. Brasília: Ministério da Saúde; 2014.) (202202. Dias JCP, Siqueira IOC, Dias RB. Doença de Chagas: diagnóstico e manejo na atenção primária à saúde. In: Gusso G, Lopes JMC. Tratado de Medicina de Família e Comunidade: 2 Volumes: Princípios, Formação e Prática. Porto Alegre: Artmed; 2012. p. 2067-80. . Women with ICF should not restrict breastfeeding, except in the presence of fissures and/or bleeding of the nipple/areola 2020. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Consenso Brasileiro em Doença de Chagas. Rev Soc Bras Med Trop. 2005;38 Supl 3:1-29.) (202202. Dias JCP, Siqueira IOC, Dias RB. Doença de Chagas: diagnóstico e manejo na atenção primária à saúde. In: Gusso G, Lopes JMC. Tratado de Medicina de Família e Comunidade: 2 Volumes: Princípios, Formação e Prática. Porto Alegre: Artmed; 2012. p. 2067-80. .
It is emphasized that health professionals should avoid the adoption of any practice that may induce stigma or prejudice. Health services should provide space and resources for advice, clarification, and orientation of the population regarding the characteristics of this form of disease. It is strongly recommended that this approach is led by a multidisciplinary team 2020. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Consenso Brasileiro em Doença de Chagas. Rev Soc Bras Med Trop. 2005;38 Supl 3:1-29.) (189189. Dias JC. The indeterminate form of human chronic Chagas' disease: a clinical epidemiological review. Rev Soc Bras Med Trop. 1989 Jul-Sep;22(3):147-56.) (190190. Macêdo VO. Forma indeterminada da doença de Chagas. In: Dias JCP, Coura JR, editores. Clínica e Terapêutica da doença de Chagas: uma abordagem prática para o clínico geral. Rio de Janeiro: Editora Fiocruz ; 1997. p.135-51.) (191191. Macêdo VO. Indeterminate form of Chagas disease. Mem Inst Oswaldo Cruz. 1999 Sep;94 Suppl 1:311-6.) (193193. Dias JCP, Machado EMM, Borges ÉC, Moreira EF, Gontijo C, Azeredo BVM. Doença de Chagas em Lassance, MG. Reavaliação clínico-epidemiológica 90 anos após a descoberta de Carlos Chagas. Rev Soc Bras Med Trop. 2002 mar-apr;35(2):167-76.) (202202. Dias JCP, Siqueira IOC, Dias RB. Doença de Chagas: diagnóstico e manejo na atenção primária à saúde. In: Gusso G, Lopes JMC. Tratado de Medicina de Família e Comunidade: 2 Volumes: Princípios, Formação e Prática. Porto Alegre: Artmed; 2012. p. 2067-80. .
CARDIAC FORMS OF CHAGAS DISEASE
Chronic Chagas heart disease is the most prevalent symptomatic clinical form, responsible for the high burden of morbidity and mortality, with great impact on social and medical systems 44. Coura JR, Dias JC. Epidemiology, control and surveillance of Chagas disease: 100 years after its discovery. Mem Inst Oswaldo Cruz. 2009 Jul;104 Suppl 1:31-40.) (1919. Prata A. Clinical and epidemiological aspects of Chagas disease. Lancet Infect Dis. 2001 Sep;1(2):92-100.) (2020. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Consenso Brasileiro em Doença de Chagas. Rev Soc Bras Med Trop. 2005;38 Supl 3:1-29. . Recently, the notion has been considered that the pathogenesis of myocardial injury in the chronic phase of Chagas disease depends mainly on parasite persistence and the unfavorable immune system response to this incessant infectious stimulus 205205. Higuchi ML, Benvenuti LA, Reis MR, Metzger M. Pathophysiology of the heart in Chagas' disease: current status and new developments. Cardiovasc Res. 2003 Oct;60(1):96-107.) (206206. Higuchi ML. Chagas disease: importance of the parasite in the pathogenesis of the cardiac chronic disease. Arq Bras Cardiol. 1995 Mar;64(3):251-4.) (207207. Higuchi ML. Chronic chagasic cardiopathy: the product of a turbulent host-parasite relationship. Rev Inst Med Trop Sao Paulo. 1997 Jan-Feb;39(1):53-60.) (208208. Marin-Neto JA, Cunha-Neto E, Maciel BC, Simões MV. Pathogenesis of chronic Chagas heart disease. Circulation. 2007 Mar;115(9):1109-23. . Among the most peculiar characteristics of chronic Chagas heart disease, the following are highlighted: Its inflammatory and intensely fibrosing nature, presence of complex ventricular arrhythmias in association with disturbances in atrioventricular and intraventricular formation and conduction of the electrical stimulus, high incidence of sudden death and thromboembolic events, and right ventricular dysfunction and ventricular aneurysms 209209. Chagas C, Villela E. Forma cardíaca da Trypanosomíase americana. Mem Inst Oswaldo Cruz. 1922;14(1):5-61.) (210210. Coura JR, Abreu LL, Pereira JB, Willcox HP. Morbidity in Chagas' disease. IV. longitudinal study of 10 years in Pains and Iguatama, Minas Gerais, Brazil. Mem Inst Oswaldo Cruz. 1985 Jan-Mar;80(1):73-80.) (211211. Pereira Barretto AC, Mady C, Arteaga-Fernandez E, Stolf N, Lopes EA, Higuchi ML, et al. Right ventricular endomyocardial biopsy in chronic Chagas' disease. Am Heart J. 1986 Feb;111(2):307-12.) (212212. Prata A, Lopes ER, Chapadeiro E. Characteristics of unexpected sudden death in Chagas disease. Rev Soc Bras Med Trop. 1986 Jan-Mar;19(1):9-12.) (213213. Dias JC, Kloetzel K. The prognostic value of the electrocardiographic features of chronic Chagas' disease. Rev Inst Med Trop Sao Paulo. 1968 May-Jun;10(3):158-62.) (214214. Bestetti RB, Muccillo G. Clinical course of Chagas' heart disease: a comparison with dilated cardiomyopathy. Int J Cardiol. 1997 Jul;60(2):187-93. .
Acute Chagas disease and consequently, acute Chagas myocarditis, was previously restricted to vectorial transmission of Chagas disease, now rare in Brazil. However, their epidemiological importance in Brazil and other countries, endemic and non-endemic, has expanded due to reactivation of the disease in immunocompromised individuals, favored by the spread and chronicity of HIV infection, as well as by the increased access to organ transplantation. In addition to this scenario, there is the challenge posed by Chagas disease in the Amazon, attributed to oral transmission. As a result of changing the epidemiological profile of disease transmission, cases of acute Chagas heart disease, which currently present clinical and specific epidemiological aspects, has again increased 7676. Shikanai-Yasuda MA, Carvalho NB. Oral transmission of Chagas disease. Clin Infect Dis. 2012 Mar;54(6):845-52.) (9191. Pinto AYN, Valente SA, Valente VC, Ferreira Júnior AG, Coura JR. Fase aguda da doença de Chagas na Amazônia brasileira: estudo de 233 casos do Pará, Amapá e Maranhão observados entre 1988 e 2005. Rev Soc Bras Med Trop. 2008 nov-dec;41(6):602-14.) (9292. Nóbrega AA, Garcia MH, Tatto E, Obara MT, Costa E, Sobel J, et al. Oral transmission of Chagas disease by consumption of açaí palm fruit, Brazil. Emerg Infect Dis. 2009 Apr;15(4):653-5.) (169169. Pinto AYN, Ferreira Júnior AG, Valente VC, Harada GS, Valente SAS. Urban outbreak of acute Chagas disease in Amazon region of Brazil: four-year follow-up after treatment with benznidazole. Rev Panam Salud Publica. 2009 Jan;25(1):77-83.) (170170. Shikanai-Yasuda MA, Marcondes CB, Guedes LA, Siqueira GS, Barone AA, Dias JC, et al. Possible oral transmission of acute Chagas disease in Brasil. Rev Inst Med Trop Sao Paulo. 1991 Sep-Oct;33(5):351-7. .
The importance of the composition of a resolute healthcare network for people with Chagas disease, in particular the role of primary care as the first contact and provider of secondary prevention of the disease, is emphasized. From the recognition of the context of clinical cases, this primary health care network should be integrated into the matrix/reference network for more complex situations.
Acute Chagas heart disease
1. Acute Chagas myocarditis associated with oral transmission
In Amazônia Legal, especially, a systematic record exists of cases with the acute form of Chagas disease, occurring in sporadically, in outbreaks, or in micro epidemics, where the main form of transmission is oral, involving food contaminated with triatomine feces 2121. Organização Pan-americana da Saúde. Guia para vigilância, prevenção, controle e manejo clínico da doença de Chagas aguda transmitida por alimentos. Rio de Janeiro: Organização Pan-Americana da Saúde; 2009. (Série de Manuais Técnicos, 12).) (7676. Shikanai-Yasuda MA, Carvalho NB. Oral transmission of Chagas disease. Clin Infect Dis. 2012 Mar;54(6):845-52. . The clinical presentation differs from classical acute Chagas myocarditis (vectorial) mainly by the absence of evidence of a portal of entry (inoculation chagoma), the involvement of community groups or families in outbreaks, and having no identified predilection for a specific age group or for disease severity 9191. Pinto AYN, Valente SA, Valente VC, Ferreira Júnior AG, Coura JR. Fase aguda da doença de Chagas na Amazônia brasileira: estudo de 233 casos do Pará, Amapá e Maranhão observados entre 1988 e 2005. Rev Soc Bras Med Trop. 2008 nov-dec;41(6):602-14.) (169169. Pinto AYN, Ferreira Júnior AG, Valente VC, Harada GS, Valente SAS. Urban outbreak of acute Chagas disease in Amazon region of Brazil: four-year follow-up after treatment with benznidazole. Rev Panam Salud Publica. 2009 Jan;25(1):77-83. .
Clinical manifestations of acute disease associated with oral transmission are variable, ranging from asymptomatic cases up to those developing severe heart failure, cardiogenic shock, and even death. It can also manifest as a nonspecific infectious syndrome, with prolonged fever (usually for more than three weeks). The manifestations of acute myocarditis are described in Figure 3 215215. Pinto AY, Harada GS, Valente VD, Abud JE, Gomes FD, Souza GC, et al. Cardiac attacks in patients with acute Chagas disease in a family micro-outbreak, in Abaetetuba, Brazilian Amazon. Rev Soc Bras Med Trop. 2001 Sep-Oct;34(5):413-9. .
Laboratory and radiologic examinations
Chest radiography may be normal or may show varying degrees of cardiomegaly and pleural effusion. The ECG readings are altered in most acute cases. The main ECG findings are non-specific changes in ventricular repolarization, followed by prolonged QTc interval, left atrial overload, low voltage of QRS complex, AVB, bundle branch block, sinus tachycardia, and atrial fibrillation 216216. Souza DSM, Almeida AJB, Costa FA, Costa EG, Figueiredo MTS, Póvoa RMS. O eletrocardiograma na fase aguda da Doença de Chagas por transmissão oral. Rev Bras Cardiol. 2013 mar-abr;26(2):127-30. . ECG can detect pericardial effusion, atrioventricular valve regurgitation, increase in the size of the heart chambers, presence of thrombi, and changes in left ventricular systolic function.
Treatment
Pharmacological management of acute Chagas myocarditis is even recommended for the treatment of heart failure in patients with acute myocarditis of other etiologies: Routine use of a combination of three types of drugs [diuretics, angiotensin converting enzyme inhibitors (ACEI) or angiotensin II AT1 receptor blockers (ARBs), and beta-blockers] is always combined with specific treatment of T. cruzi infection (using benznidazole or nifurtimox) 203203. Castro I, Andrade JP, Paola AAV, Vilas-Boas F, Oliveira GMM, Marin-Neto JA, et al. Sociedade Brasileira de Cardiologia. I Diretriz Latino-Americana para o diagnóstico e tratamento da cardiopatia chagásica. Arq Bras Cardiol. 2011 Aug;97(2 Suppl 3):1-48. .
The natural history of the acute phase of Chagas myocarditis, induced by oral transmission of Chagas disease, is still not fully known. The most severe cases, with ventricular dysfunction, pericardial effusion, and atrial fibrillation, tend to have worse outcomes in the acute phase, requiring treatment in an intensive care environment.
2. Acute Chagas myocarditis in immunocompromised individuals
Individuals infected with T. cruzi , when exposed to immunosuppressive agents or who have other concomitant diseases such as cancer and other infections, particularly HIV infection, may experience reactivation of Chagas disease. The frequency of this reactivation is not fully known. However, in a prospective study in which cases were evaluated pre- and post-antiretroviral therapy, the occurrence of reactivation was observed in 20% of the cases 4848. Almeida EA, Lima JN, Lages-Silva E, Guariento ME, Aoki FH, Torres-Morales AE, et al. Chagas' disease and HIV co-infection in patients without effective antiretroviral therapy: prevalence, clinical presentation and natural history. Trans R Soc Trop Med Hyg. 2010 Jul;104(7):447-52. .
The heart appears to be involved in approximately 30-40% of cases of reactivation of Chagas disease in individuals coinfected with HIV. However, the incidence of myocarditis alone does not seem to be common. Cardiac involvement is usually characterized by acute myocarditis, with diffuse or focal cardiac involvement. Clinically, it is characterized by signs and symptoms of heart failure (tachycardia, edema, hepatomegaly) or severe arrhythmias. In some cases, only electrocardiographic changes are observed; in others, myocarditis is confirmed only by histopathologic examination of endomyocardial biopsy material. Anatomopathological examination reveals acute myocarditis with intense inflammatory infiltrate, cardiac fiber injury with focal necrosis, and large numbers of amastigote forms of the parasite 217217. Nishioka SA, Ferreira MS, Rocha A, Burgarelli MK, Silva AM, Duarte MI, et al. Reactivation of Chagas' disease successfully treated with benznidazole in a patient with acquired immunodeficiency syndrome. Mem Inst Oswaldo Cruz. 1993 Jul-Sep;88(3):493-6 . In cases where there was previous cardiac involvement (for chronic Chagas heart disease), there may be overlapping reactivation Chagas disease cardiomyopathy and decompensation of preexisting Chagas heart disease. Determining if the clinical picture is solely due to reactivation of Chagas disease, myocarditis associated with HIV itself, or the overlap of the two conditions, is complex 218218. Almeida EA, Silva EL, Guariento ME, Souza ML, Aoki FH, Pedro RJ. Fatal evolution of Chagas' disease/Aids co-infection: diagnostic difficulties between myocarditis reactivation and chronic chagasic myocardiopathy. Rev Soc Bras Med Trop. 2009 Mar-Apr;42(2):199-202. . Acute myocarditis due to the reactivation of T. cruzi infection might be also confused with the natural progression and worsening of chronic Chagas heart disease. The differential diagnosis between these two conditions is important in terms of deciding whether or not to administer the specific treatment of T. cruzi infection, as many patients do not tolerate the medication. High levels of parasitemia are indicative of reactivation. However, there is no record of patients who developed low parasitemia 219219. Perez-Ramirez L, Barnabé C, Sartori AM, Ferreira MS, Tolezano JE, Nunes EV, et al. Clinical analysis and parasite genetic diversity in human immunodeficiency virus/Chagas' disease coinfections in Brazil. Am J Trop Med Hyg. 1999 Aug;61(2):198-206. .
Diagnosis
Confirmation of reactivated Chagas disease in patients with immunosuppression is performed by parasite detection in peripheral blood and other body fluids (cerebrospinal fluid, cavity effusions, etc.), by direct observation methods, or in sites of organ damage (heart, skin, etc.) in individuals with co-infection 4848. Almeida EA, Lima JN, Lages-Silva E, Guariento ME, Aoki FH, Torres-Morales AE, et al. Chagas' disease and HIV co-infection in patients without effective antiretroviral therapy: prevalence, clinical presentation and natural history. Trans R Soc Trop Med Hyg. 2010 Jul;104(7):447-52. .
Treatment
There are no specific treatment measures for this group. Treatment should follow evidence-based recommendations developed for the treatment of heart failure in general. Acute decompensated heart failure, resulting from the hemodynamic effects of heart failure, is associated with neurohumoral activation and symptoms of congestion and/or low cardiac output. Early recognition of this condition and the implementation of appropriate therapeutic measures for heart failure, associated with the specific treatment for T. cruzi infection, can reduce the high rate of mortality observed in cases of myocarditis induced by reactivation of Chagas disease in immunocompromised patients.
Chronic Chagas cardiomyopathy
Definition
Chronic Chagas cardiomyopathy (CCC) is defined as the presence of ECG changes suggestive of cardiac involvement typical of Chagas disease in symptomatic and asymptomatic individuals. Since ECG examination is easily accessible and has high sensitivity and specificity (around 90%), the modified conventional ECG is used, a priori , to diagnose CCC in people chronically infected with T. cruzi2020. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Consenso Brasileiro em Doença de Chagas. Rev Soc Bras Med Trop. 2005;38 Supl 3:1-29. . Cardiac involvement in the chronic phase of Chagas disease includes a wide spectrum of manifestations, ranging from the presence of clinically unapparent abnormalities to severe forms, such as end-stage heart failure, thromboembolic complications, refractory ventricular arrhythmias, and sudden death.
Taking as a reference only conventional 12-lead ECG, it is possible to establish, in a simplified way, the degree of cardiac involvement in routine evaluation of the patient with chronic Chagas disease, as shown in the following flowchart ( Figure 4 ) 2020. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Consenso Brasileiro em Doença de Chagas. Rev Soc Bras Med Trop. 2005;38 Supl 3:1-29. . Cases with nonspecific ECG changes would not be diagnosed with CCC, but they should be re-evaluated according to the concomitant symptoms and changes found in additional tests.
Chronic Chagas cardiomyopathy staging
Presence of severe heart failure [functional class III and IV of the New York Heart Association (NYHA)] and left ventricular global systolic dysfunction are the most important prognostic factors in CCC 220220. Bestetti RB, Dalbo CM, Freitas OC, Teno LA, Castilho OT, Oliveira JS. Noninvasive predictors of mortality for patients with Chagas' heart disease: a multivariate stepwise logistic regression study. Cardiology. 1994;84(4-5):261-7.) (221221. Mady C, Cardoso RH, Barretto AC, Luz PL, Bellotti G, Pileggi F. Survival and predictors of survival in patients with congestive heart failure due to Chagas' cardiomyopathy. Circulation. 1994 Dec;90(6):3098-102.) (222222. Rassi Júnior A, Rassi A, Little WC, Xavier SS, Rassi SG, Rassi AG, et al. Development and validation of a risk score for predicting mortality in Chagas' heart disease. N Engl J Med. 2006 Aug 24;355(8):799-808.) (223223. Xavier SS, Sousa AS, Hasslocher-Moreno A. Application of the new classification of cardiac insufficiency (ACC/AHA) in chronic Chagas cardiopathy: a critical analysis of the survival curves. Rev SOCERJ. 2005 mai-jun;18(3):227-32. .
Echocardiography allows evaluation of both the overall and segmental myocardial function. It can also identify important markers for staging the disease, such as size of chambers, alterations in segmental mobility, and presence of aneurysms and mural thrombosis. For these reasons, and because it is a noninvasive and inexpensive examination, echocardiography is of great value for the initial staging of CCC, in association with the functional NYHA classification. From a prognostic and therapeutic point of view, it is possible to identify five different subgroups of CCC, as shown in Figure 5 223223. Xavier SS, Sousa AS, Hasslocher-Moreno A. Application of the new classification of cardiac insufficiency (ACC/AHA) in chronic Chagas cardiopathy: a critical analysis of the survival curves. Rev SOCERJ. 2005 mai-jun;18(3):227-32.) (224224. Rodriguez-Salas LA, Klein E, Acquatella H, Catalioti F, Davalos V, Gomez-Mancebo JR, et al. Echocardiographic and clinical predictors of mortality in chronic Chagas' disease. Echocardiography. 1998 Apr;15(3):271-278.) (225225. Viotti RJ, Vigliano C, Laucella S, Lococo B, Petti M, Bertocchi G, et al. Value of echocardiography for diagnosis and prognosis of chronic Chagas' disease cardiomyopathy without heart failure. Heart. 2004 Jun;90(6):655-60.) (226226. Acquatella H, Schiller NB, Puigbó JJ, Giordano H, Suárez JA, Casal H, et al. M-mode and two-dimensional echocardiography in chronic Chagas' heart disease: a clinical and pathologic study. Circulation. 1980 Oct;62(4):787-99. .
In addition to the impairment of myocardial function, rhythm disturbances and electrical impulse conduction abnormalities are significant changes in CCC; in some cases, arrhythmias may occur alone, without global ventricular dysfunction, or only with small regional dyskinesias 227227. Dias JCP. História natural da cardiopatia chagásica. In: Cançado JR, Chuster M. Cardiopatia chagásica. Belo Horizonte: Fundação Carlos Chagas; 1985. p. 99-110.) (228228. Carrasco HA, Parada H, Guerrero L, Duque M, Durán D, Molina C. Prognostic implications of clinical, electrocardiographic and hemodynamic findings in chronic Chagas' disease. Int J Cardiol. 1994 Jan;43(1):27-38.) (229229. Garzon SA, Lorga AM, Nicolau JC. Electrocardiography in Chagas' heart disease. Sao Paulo Med J. 1995 Mar-Apr;113(2):802-13.) (230230. Marin-Neto JA, Simões MV, Sarabanda AV. Chagas' heart disease. Arq Bras Cardiol. 1999;72(3):247-80.) (231231. Almeida-Filho OC, Maciel BC, Schmidt A, Pazin-Filho A, Marin-Neto JA. Minor segmental dyssynergy reflects extensive myocardial damage and global left ventricle dysfunction in chronic Chagas disease. J Am Soc Echocardiogr. 2002 Jun;15(6):610-6.) (232232. Pazin-Filho A, Romano MM, Gomes Furtado R, Almeida Filho OC, Schmidt A, Marin-Neto JA, et al. Left ventricular global performance and diastolic function in indeterminate and cardiac forms of Chagas' disease. J Am Soc Echocardiogr. 2007 Dec;20(12):1338-43. .
If possible, it is obligatory to perform a 24 hour-electrocardiogram (Holter) in all cases with suspected arrhythmia. Less frequently, an ergometric test may be used instead of a Holter, to show ventricular arrhythmias during standardized physical effort. When possible, both tests should be performed, as they are complementary in the evaluation of these cases.
There is also a group of cases in the chronic phase of Chagas disease (not included in the classification above) that shows normal ECG findings, but presents changes ̶ usually discrete and not associated with an increased risk of death ̶ in other cardiac tests, such as echocardiography, myocardial scintigraphy, and magnetic resonance imaging. However, since the risk of progression of these cases to the cardiac form, classically defined, appears to be higher than that of cases of Chagas disease without these changes, they should receive regular clinical follow-up 231231. Almeida-Filho OC, Maciel BC, Schmidt A, Pazin-Filho A, Marin-Neto JA. Minor segmental dyssynergy reflects extensive myocardial damage and global left ventricle dysfunction in chronic Chagas disease. J Am Soc Echocardiogr. 2002 Jun;15(6):610-6.) (232232. Pazin-Filho A, Romano MM, Gomes Furtado R, Almeida Filho OC, Schmidt A, Marin-Neto JA, et al. Left ventricular global performance and diastolic function in indeterminate and cardiac forms of Chagas' disease. J Am Soc Echocardiogr. 2007 Dec;20(12):1338-43. .
Chronic Chagas cardiomyopathy prognosis
In a systematic review of studies that used multivariate analysis to evaluate the prognosis of patients with CCC 233233. Rassi A Júnior, Rassi A, Rassi SG. Predictors of mortality in chronic Chagas disease: a systematic review of observational studies. Circulation . 2007 Mar;115(9):1101-8 , four independent prognostic variables were identified: functional class III/IV of NYHA, cardiomegaly on chest radiograph, left ventricular systolic dysfunction, and non-sustained ventricular tachycardia (NSVT) on Holter examination. By using these four variables in an integrated manner, it was possible to develop an algorithm capable of stratifying the prognosis of cases with CCC ( Figure 6 ).
It is important to highlight that the presence of functional class III or IV NYHA, per se , identifies high-risk cases, since virtually all these cases have ventricular systolic dysfunction (evident on echocardiography) and NSVT (detected by Holter). The combination of ventricular dysfunction with NSVT (regardless of functional class) already identifies a group with a risk about 15 times higher than cases without these two variables 222222. Rassi Júnior A, Rassi A, Little WC, Xavier SS, Rassi SG, Rassi AG, et al. Development and validation of a risk score for predicting mortality in Chagas' heart disease. N Engl J Med. 2006 Aug 24;355(8):799-808.) (233233. Rassi A Júnior, Rassi A, Rassi SG. Predictors of mortality in chronic Chagas disease: a systematic review of observational studies. Circulation . 2007 Mar;115(9):1101-8) (234234. Xavier SS, Sousa AS, Brasil PEAA, Gabriel FG, Holanda MT, Hasslocher-Moreno A, et al. Incidência e preditores de morte súbita na cardiopatia chagásica crônica com função sistólica preservada. Rev SOCERJ. 2005 set-out;18(5):457-63. .
Clinical manifestations of chronic Chagas cardiomyopathy
Clinical manifestations of CCC can be grouped into three syndromes: arrhythmic, heart failure, and thromboembolic. These syndromes may occur alone or in combination in the same case, and may be associated with megaesophagus and/or megacolon 235235. Cunha GP. Síndromes clínicas na cardiomiopatia chagásica crônica. Hospital. 1960;57:71-95.) (236236. Rassi Jr A, Marin-Neto JA. Estado da Arte. Cardiopatia chagásica crônica. Rev Soc Cardiol Est São Paulo. 2000;10(4):6-12. .
I - Arrhythmic syndrome
Ventricular arrhythmias
Ventricular arrhythmias are common in Chagas disease and are of various types, including ventricular extrasystoles, isolated and in pairs; NSVT; sustained ventricular tachycardia (SVT); and ventricular fibrillation 237237. Rassi Júnior A, Rassi AG, Rassi SG, Rassi Júnior L, Rassi A. Arritmias ventriculares na doença de Chagas. Particularidades diagnósticas, prognósticas e terapêuticas. Arq Bras Cardiol. 1995 out;65(4):377-87. . These commonly present in combination. Clinically, they present as palpitations, lipothymia, syncope, and sudden death 237237. Rassi Júnior A, Rassi AG, Rassi SG, Rassi Júnior L, Rassi A. Arritmias ventriculares na doença de Chagas. Particularidades diagnósticas, prognósticas e terapêuticas. Arq Bras Cardiol. 1995 out;65(4):377-87.) (238238. Rassi Júnior A, Rassi SG, Rassi A. Morte súbita na Doença de Chagas. Arq Bras Cardiol. 2001 jan;76(1):75-85. . Syncope and other symptoms of low output in cases with CCC should be quickly investigated, due to the risk of complex ventricular arrhythmias and sudden death.
Ventricular extrasystole is the most common arrhythmia. Initially uncommon, monomorphic, and isolated, it becomes polymorphic and repetitive with the evolution of the disease. Holter and ergometric tests are the methods of choice for the detection of ventricular arrhythmias and, when possible, should be performed in all cases with CCC, regardless of the presence of symptoms 4242. Rassi Júnior A, Rassi A, Marin-Neto JA. Chagas disease. Lancet. 2010 Apr;375(9723):1388-402. . Ventricular tachycardia may be sustained (duration ≥30 sec or shorter than this when interrupted electrically or pharmacologically) or non-sustained (3 heartbeats or more and a duration <30 sec).
NSVT relates to the degree of ventricular dysfunction, occurring in about 40% of cases with CCC associated with regional changes in ventricular contractility, and in 90% of cases with left ventricular global systolic dysfunction associated with heart failure 222222. Rassi Júnior A, Rassi A, Little WC, Xavier SS, Rassi SG, Rassi AG, et al. Development and validation of a risk score for predicting mortality in Chagas' heart disease. N Engl J Med. 2006 Aug 24;355(8):799-808.) (237237. Rassi Júnior A, Rassi AG, Rassi SG, Rassi Júnior L, Rassi A. Arritmias ventriculares na doença de Chagas. Particularidades diagnósticas, prognósticas e terapêuticas. Arq Bras Cardiol. 1995 out;65(4):377-87. . This may occur in cases with normal ventricular function 234234. Xavier SS, Sousa AS, Brasil PEAA, Gabriel FG, Holanda MT, Hasslocher-Moreno A, et al. Incidência e preditores de morte súbita na cardiopatia chagásica crônica com função sistólica preservada. Rev SOCERJ. 2005 set-out;18(5):457-63. . There is evidence that the frequency of ventricular arrhythmias is increased in heart failure due to Chagas disease, compared with other etiologies 239239. Rassi Júnior A, Rassi AG, Rassi SG, Rassi Júnior L, Las Casas AA, Rassi A. Arritmia ventricular na insuficiência cardíaca de etiologia chagásica e não chagásica. Arq Bras Cardiol. 1993;61 supl II:11-9. . SVT can occur spontaneously or be reproduced by electrophysiological study, which is indicated for cases whose history suggests this hypothetical diagnosis but for whom non-invasive methods have failed to confirm the diagnosis 240240. Scanavacca M, Sosa E. Electrophysiologic study in chronic Chagas' heart disease. Sao Paulo Med J. 1995 Mar-Apr;113(2):841-50. . Ventricular fibrillation is the leading cause of sudden death in CCC. It occurs more frequently in patients who have had previous episodes of ventricular tachycardia, although it may also be the first manifestation of the disease or its terminal event, especially in cases with severe ventricular dysfunction and chronic cardiac insufficiency (CCI ) (238238. Rassi Júnior A, Rassi SG, Rassi A. Morte súbita na Doença de Chagas. Arq Bras Cardiol. 2001 jan;76(1):75-85. .
Treatment of ventricular arrhythmias
Treatment of the arrhythmia has two main objectives: to control symptoms and to prevent sudden death 238238. Rassi Júnior A, Rassi SG, Rassi A. Morte súbita na Doença de Chagas. Arq Bras Cardiol. 2001 jan;76(1):75-85. . Simple ventricular arrhythmias (isolated and monomorphous ventricular extrasystoles) are not associated with an increased risk of death. Hence, they should be treated only if causing symptoms of functional impairment. If needed, suitable treatment consists of normal doses of amiodarone, sotalol, or beta blockers.
In cases of life-threatening arrhythmias (NSVT, SVT, and ventricular fibrillation), the most effective antiarrhythmic, and the safer to use, is amiodarone 241241. Chiale PA, Halpern MS, Nau GJ, Tambussi AM, Przybylski J, Lázzari JO, et al. Efficacy of amiodarone during long-term treatment of malignant ventricular arrhythmias in patients with chronic chagasic myocarditis. Am Heart J. 1984 Apr;107(4):656-65. . It is necessary to pay attention to the side effects of this drug - that are generally related to the cumulative dose - which can trigger serious bradyarrhythmias. In such cases, evaluation of the need for permanent pacemaker implantation is required. Extra-cardiac toxicity, thyroid dysfunction, and skin abnormalities are not uncommon, while severe pulmonary toxicity is rare 242242. Silva CP, Bacal F, Pires PV, Drager LF, Souza GEC, Fajardo GM, et al. Importance of amiodarone pulmonary toxicity in the differential diagnosis of a patient with dyspnea awaiting a heart transplant. Arq Bras Cardiol. 2006 Sep;86(3):e4-7. . Periodic assessment of the thyroid function is recommended in patients treated with amiodarone.
In turn, cases with SVT (a relatively common condition) and those who recover from cardiorespiratory arrest in an out-of-hospital environment (a much rarer condition) are at high risk of death and deserve a rigorous evaluation. For these cases, there are not only antiarrhythmic drugs, but also arrhythmic focus catheter ablation techniques (or, rarely, surgical techniques) and, especially, implantable cardioverter-defibrillator (ICD) devices 237237. Rassi Júnior A, Rassi AG, Rassi SG, Rassi Júnior L, Rassi A. Arritmias ventriculares na doença de Chagas. Particularidades diagnósticas, prognósticas e terapêuticas. Arq Bras Cardiol. 1995 out;65(4):377-87. .
The treatment of ventricular arrhythmias in CCC is described briefly in Figure 7 . Drug treatment and ICD recommendations are described in Figure 8 and Figure 9 , respectively.
Recommendations and levels of evidence for the use of antiarrhythmic drugs in the treatment of ventricular arrhythmias in chronic Chagas cardiomyopathy.
Class of recommendation and levels of evidence for the use of CDI in the treatment of ventricular arrhythmias in chronic Chagas cardiomyopathy.
Supraventricular arrhythmias
Atrial fibrillation is the sustained supraventricular arrhythmia more frequently observed in CCC, found in 4-12% of cases 229229. Garzon SA, Lorga AM, Nicolau JC. Electrocardiography in Chagas' heart disease. Sao Paulo Med J. 1995 Mar-Apr;113(2):802-13. . It tends to manifests later on and is often associated with marked cardiomegaly. Treatment consists in controlling the ventricular rate, which can be obtained through the use of drugs prolonging the refractory period of the atrioventricular node. If there is an associated heart failure, preference is given to digitalis and beta-blockers (metoprolol succinate, carvedilol, or bisoprolol). If ventricular function is normal, the use of conventional beta-blockers (propranolol and atenolol) or calcium channel blockers (verapamil and diltiazem) is recommended for the initial control of the heart rate, with subsequent evaluation of the possible need for electrical or pharmacologic cardioversion. Anticoagulation is indicated when atrial fibrillation is associated with heart failure and cardiomegaly, CHA 2 DS 2 VASc score ≥2, or when there is evidence of intracavitary thrombosis or previous embolic episodes. The drug of choice is warfarin, in a dose sufficient to maintain the international normalization factor (INR) between 2 and 3 243243. Zimerman LI, Fenelon G, Martinelli Filho M, Grupi C, Atié J, Lorga Filho A, et al; Sociedade Brasileira de Cardiologia. Diretrizes brasileiras de fibrilação atrial. Arq Bras Cardiol. 2009;92(6 supl 1):1-39. .
Bradyarrhythmias
The treatment of bradyarrhythmias in CCC does not differ from that recommended for other types of cardiomyopathies. It consists of the implantation of a permanent cardiac pacemaker in symptomatic or high-risk blockage cases. These recommendations are well defined in the Brazilian Guidelines for Implantable Cardiac Electronic Devices, published in 2007 244244. Martinelli Filho M, Zimerman LI, Lorga AM, Vasconcelos JTM, Rassi Júnior A; Sociedade Brasileira de Cardiologia. Diretrizes brasileiras de dispositivos eletrônicos implantáveis (DCEI). Arq Bras Cardiol. 2007; 89(6):e210-e238. .
The association between disorders of the heart conduction system and frequent and complex arrhythmias is common in CCC. In these cases, the effective pharmacologic antiarrhythmic drug therapy may require permanent pacemaker implantation in order to prevent a possible AVB or high-risk bradyarrhythmia induced by antiarrhythmic. The choice of the pacing mode is, to date, the subject of controversy in the literature.
II - Heart failure syndrome
In the acute phase of Chagas disease, heart failure (HF) can occur due to severe myocarditis. Although the outcome may be death, in most cases, it is reversible, progressing to the indeterminate form of the disease. Approximately 30-40% of cases with the indeterminate form will progress to the heart form of the disease, usually after decades of evolution 4242. Rassi Júnior A, Rassi A, Marin-Neto JA. Chagas disease. Lancet. 2010 Apr;375(9723):1388-402.) (236236. Rassi Jr A, Marin-Neto JA. Estado da Arte. Cardiopatia chagásica crônica. Rev Soc Cardiol Est São Paulo. 2000;10(4):6-12. .
The chronic heart form, characterized by the gradual appearance of electrocardiographic changes, is caused by the slow but steady destruction of myocardial fibers, caused by a continuous chronic inflammatory process, with intense reparative fibrosis and progressive ventricular remodeling 208208. Marin-Neto JA, Cunha-Neto E, Maciel BC, Simões MV. Pathogenesis of chronic Chagas heart disease. Circulation. 2007 Mar;115(9):1109-23. .
In the early chronic stage of the cardiomyopathy, it is not uncommon that only the ECG findings are changed; individuals remain asymptomatic, capable of performing unrestricted, sometimes even extreme, physical activity. Therefore, they are included into NYHA class I 227227. Dias JCP. História natural da cardiopatia chagásica. In: Cançado JR, Chuster M. Cardiopatia chagásica. Belo Horizonte: Fundação Carlos Chagas; 1985. p. 99-110.) (230230. Marin-Neto JA, Simões MV, Sarabanda AV. Chagas' heart disease. Arq Bras Cardiol. 1999;72(3):247-80.) (239239. Rassi Júnior A, Rassi AG, Rassi SG, Rassi Júnior L, Las Casas AA, Rassi A. Arritmia ventricular na insuficiência cardíaca de etiologia chagásica e não chagásica. Arq Bras Cardiol. 1993;61 supl II:11-9. . Rarely, in this group of cases, the first and only manifestation of CCC is sudden death 245245. Baroldi G, Oliveira SJ, Silver MD. Sudden and unexpected death in clinically 'silent' Chagas' disease. A hypothesis. Int J Cardiol. 1997 Feb;58(3):263-8. . In most cases, a progressive reduction in the ability to perform physical activities is observed, followed by deterioration of contractile function of the left ventricle, initially in the form of regional dyskinesia and diastolic dysfunction, followed by a drop in the global systolic function of the chamber, due to several associated factors, such as the progressive destruction of cardiomyocytes, microvascular changes, the breakdown of the muscle structure and fibrosis 208208. Marin-Neto JA, Cunha-Neto E, Maciel BC, Simões MV. Pathogenesis of chronic Chagas heart disease. Circulation. 2007 Mar;115(9):1109-23.) (236236. Rassi Jr A, Marin-Neto JA. Estado da Arte. Cardiopatia chagásica crônica. Rev Soc Cardiol Est São Paulo. 2000;10(4):6-12.) (247247. Simões MV, Almeida Filho OC, Pazin Filho A, Castro RBP, Schmidt A, Maciel BC, et al. Insuficiência cardíaca na doença de Chagas. Rev Soc Cardiol Est São Paulo. 2000 Jan-Fev;10(1):50-64. . It is not common for cases with Chagas disease in the chronic phase to present with acute pulmonary edema. There are also many cases with CCC in whom the initial manifestations of heart failure are fatigue and edema, with minimal dyspnea 246246. Marin-Neto JA, Simões MV, Maciel BC. Aspectos peculiares da fisiopatologia da insuficiência cardíaca na doença de Chagas. Rev Soc Cardiol Est São Paulo. 1998;8(2):243-52.) (247247. Simões MV, Almeida Filho OC, Pazin Filho A, Castro RBP, Schmidt A, Maciel BC, et al. Insuficiência cardíaca na doença de Chagas. Rev Soc Cardiol Est São Paulo. 2000 Jan-Fev;10(1):50-64. . In these cases there is the early involvement of the right ventricle, with impaired systolic function. In the more advanced stages of the disease, when heart failure is completely manifested, predominant symptoms and signs of systemic congestion may arise, but with little significant pulmonary congestion. Chest teleradiography shows a marked cardiomegaly, with little engorgement of the pulmonary vasculature. It is important to identify early signs of left ventricular systolic failure, since the treatment at this early stage could, in theory, delay the deterioration of contractile cardiac function 4242. Rassi Júnior A, Rassi A, Marin-Neto JA. Chagas disease. Lancet. 2010 Apr;375(9723):1388-402.) (227227. Dias JCP. História natural da cardiopatia chagásica. In: Cançado JR, Chuster M. Cardiopatia chagásica. Belo Horizonte: Fundação Carlos Chagas; 1985. p. 99-110. . In addition, left ventricular systolic dysfunction is also the most important indicator of risk in the chronic phase of Chagas disease-induced cardiomyopathy 222222. Rassi Júnior A, Rassi A, Little WC, Xavier SS, Rassi SG, Rassi AG, et al. Development and validation of a risk score for predicting mortality in Chagas' heart disease. N Engl J Med. 2006 Aug 24;355(8):799-808.) (224224. Rodriguez-Salas LA, Klein E, Acquatella H, Catalioti F, Davalos V, Gomez-Mancebo JR, et al. Echocardiographic and clinical predictors of mortality in chronic Chagas' disease. Echocardiography. 1998 Apr;15(3):271-278.) (225225. Viotti RJ, Vigliano C, Laucella S, Lococo B, Petti M, Bertocchi G, et al. Value of echocardiography for diagnosis and prognosis of chronic Chagas' disease cardiomyopathy without heart failure. Heart. 2004 Jun;90(6):655-60. .
Treatment of heart failure in cases with chronic Chagas cardiomyopathy
Treatment of heart failure aims to relieve the symptoms, slow the progression of ventricular dysfunction, and prolong survival 2020. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Consenso Brasileiro em Doença de Chagas. Rev Soc Bras Med Trop. 2005;38 Supl 3:1-29. . Treatment of heart failure due to Chagas disease, as well as other etiologies, should be comprehensive, starting with general measures, removing the factors that contribute to worsen heart failure, and treating the underlying syndrome 248248. Bocchi EA, Marcondes-Braga FG, Bacal F, Ferraz AS, Albuquerque D, Rodrigues D, et al; Sociedade Brasileira de Cardiologia. Atualização da Diretriz Brasileira de Insuficiência Cardíaca Crônica: 2012. Arq Bras Cardiol. 2012 jan;98(1 supl 1):1-33.) (249249. Rocha MOC, Ribeiro AL, Teixeira MM. Clinical management of chronic Chagas cardiomyopathy. Front Biosci. 2003 Jan;8:e44-54. .
General measures
-
Diet: Obesity should be corrected and the ideal weight should be maintained, avoiding inadequate intake of sodium chloride and, in specific cases, alleviating the symptoms of dysphagia and constipation.
-
Controlling water retention: A balanced intake of salt (3-4g/day of sodium chloride [mild to moderate disease], and ≤2g/day [severe disease]). A simple and reliable way to evaluate water retention is daily assessment of body weight. Variations higher than 1kg/day are indicative of fluid retention. A gain of rapid and constant weight (1kg/day) is an indication that heart failure is worsening. People affected should be encouraged to monitor their weight daily in the morning (after urinating and fasting). In severe heart failure with hypervolemia and/or hyponatremia, fluid restriction may be required.
-
Control of aggravating factors: The intake of alcohol and the use of anti-inflammatories should be avoided. Arterial hypertension, cardiac arrhythmias, anemia, or thyroid function disorders should be controlled. Other comorbidities may contribute to worsening heart failure, such as coronary artery disease and diabetes mellitus.
-
Individualization of the recommendations for rest or physical activity , in accordance with the degree of heart failure and the patient's age.
-
Vaccination against influenza (annually) and pneumococcal 23-valent (a booster after five years for those cases with compromised immune systems, or adults over the age of 60 years who received the first dose before 65 years old) should be provided.
Drug treatment of heart failure caused by Chagas disease
At the time of finalization of this Consensus, the results of the BENEFIT study were published. This was a double-blinded, multicenter, clinical trial that tested the hypothesis that trypanosomicide treatment with benznidazole, compared to placebo, could modify the prognosis of patients with Chagas disease chronic cardiomyopathy 250250. Marin-Neto JA, Rassi A Júnior, Morillo CA, Avezum A, Connolly SJ, Sosa-Estani S, et al; BENEFIT Investigators. BENEFIT Investigators. Rationale and design of a randomized placebo-controlled trial assessing the effects of etiologic treatment in Chagas' cardiomyopathy: the BENznidazole Evaluation For Interrupting Trypanosomiasis (BENEFIT). Am Heart J. 2008 Jul;156(1):37-43.) (251251. Morillo CA, Marin-Neto JA, Avezum A, Sosa-Estani S, Rassi Júnior A, Rosas F, et al; BENEFIT Investigators. Randomized Trial of Benznidazole for Chronic Chagas' Cardiomyopathy. N Engl J Med. 2015 Oct;373(14):1295-306. . Overall, 2,854 patients were evaluated. They were randomly assigned to receive benznidazole or placebo for up to 80 days, with a mean follow-up period of 5.4 years. A reduction in the parasite load was observed with benznidazole, but it had no effect on the progression of heart disease (estimated by complications occurring during clinical follow-up, such as death, ventricular arrhythmias, need for implanted devices, thromboembolic events, heart failure, or heart transplantation): 27.5% of patients in the benznidazole group and 29.1% of patients in the placebo group experienced complications, hazard ratio (HR) = 0.93; 95% CI, 0.81-1.07; p = 0.31) 251251. Morillo CA, Marin-Neto JA, Avezum A, Sosa-Estani S, Rassi Júnior A, Rosas F, et al; BENEFIT Investigators. Randomized Trial of Benznidazole for Chronic Chagas' Cardiomyopathy. N Engl J Med. 2015 Oct;373(14):1295-306. . This result differed from the results of previous observational studies 252252. Viotti R, Vigliano C, Lococo B, Bertocchi G, Petti M, Alvarez MG, et al. Long-term cardiac outcomes of treating chronic Chagas disease with benznidazole versus no treatment: a nonrandomized trial. Ann Intern Med. 2006 May;144(10):724-34.) (253253. Viotti R, Vigliano C, Lococo B, Petti M, Bertocchi G, Alvarez MG, et al. Clinical predictors of chronic chagasic myocarditis progression. Rev Esp Cardiol. 2005 Sep;58(9):1037-44. . These results shift the focus of attention to treatment (of patients with CCC) with conventional anti-failure methods derived from other cardiomyopathies 254254. Marin-Neto JA, Rassi Júnior A, Sousa AS, Dias JCP, Rassi A. Doença de Chagas: moléstia negligenciada. In: Andrade JP, Arnett DK, Pinto FJ (editores). Tratado de Prevenção Cardiovascular. Um Desafio Global. São Paulo: Atheneu; 2014. p. 117-27. .
Long-term treatment of heart failure is usually based on a combination of the following classes of drugs: diuretics; ACE inhibitors or ARBs; adrenergic blockers (BB); and the aldosterone antagonists 254254. Marin-Neto JA, Rassi Júnior A, Sousa AS, Dias JCP, Rassi A. Doença de Chagas: moléstia negligenciada. In: Andrade JP, Arnett DK, Pinto FJ (editores). Tratado de Prevenção Cardiovascular. Um Desafio Global. São Paulo: Atheneu; 2014. p. 117-27. . Positive inotropic drugs (such as catecholamine or milrinone) have limited applicability in intensive care settings, and slightly prolonged cardiac decompensation. Digitalis, however, may be administered chronically in individuals without bradyarrhythmias to alleviate their symptoms and prevent cardiac decompensation and hospital admission, especially in patients with atrial fibrillation with rapid ventricular response 255255. Montera MW, Pereira SB, Colafranceschi AS, Almeida DR, Tinoco EM, Rocha RM, et al. Sumário de Atualização da II Diretriz Brasileira de Insuficiência Cardíaca Aguda: 2009/2011. Arq Bras Cardiol. 2012;98(5):375-83. .
Loop and thiazide diuretics may be used separately or in combination to relieve symptoms and signs of pulmonary and systemic venous congestion. When it is imperative to use high doses of loop diuretics, such as furosemide, one should be aware of the higher likelihood of hypokalemia and/or hyponatremia, which, in turn, can exacerbate or trigger serious arrhythmias 248248. Bocchi EA, Marcondes-Braga FG, Bacal F, Ferraz AS, Albuquerque D, Rodrigues D, et al; Sociedade Brasileira de Cardiologia. Atualização da Diretriz Brasileira de Insuficiência Cardíaca Crônica: 2012. Arq Bras Cardiol. 2012 jan;98(1 supl 1):1-33. .
In CCC with heart failure or asymptomatic systolic dysfunction (ejection fraction <45%), the chronic administration of ACE inhibitors is indicated to reduce the morbidity, mortality or ARB, when patients are intolerant to ACE inhibitors 256256. Roberti RR, Martinez EE, Andrade JL, Araujo VL, Brito FS, Portugal OP, et al. Chagas cardiomyopathy and captopril. Eur Heart J. 1992 Jul;13(7):966-70. . With the same purpose, the use of spironolactone is recommended in cases with heart failure, in NYHA functional classes II-IV 248248. Bocchi EA, Marcondes-Braga FG, Bacal F, Ferraz AS, Albuquerque D, Rodrigues D, et al; Sociedade Brasileira de Cardiologia. Atualização da Diretriz Brasileira de Insuficiência Cardíaca Crônica: 2012. Arq Bras Cardiol. 2012 jan;98(1 supl 1):1-33. . Furthermore, it is thought that aldosterone antagonists can provide additional benefits when used in patients with CCC because of their antifibrotic properties (demonstrated in experimental studies). The combination of hydralazine and nitrates is a recommended alternative to treat cases in functional NYHA class II-III if a contraindication to the use of ACE inhibitors or ARBs (e.g., progressive renal failure or hyperkalemia) exists 248248. Bocchi EA, Marcondes-Braga FG, Bacal F, Ferraz AS, Albuquerque D, Rodrigues D, et al; Sociedade Brasileira de Cardiologia. Atualização da Diretriz Brasileira de Insuficiência Cardíaca Crônica: 2012. Arq Bras Cardiol. 2012 jan;98(1 supl 1):1-33. .
The use of adrenergic beta-blockers in addition to blockade of the renin-angiotensin-aldosterone system (with ACE inhibitors or ARBs) is recommended to reduce the morbidity and mortality associated with the disease. It is assumed that these drugs can help to prevent the worsening of the ventricular remodeling, development of malignant arrhythmias, and sudden death 257257. Davila DF, Angel F, Arata de Bellabarba G, Donis JH. Effects of metoprolol in chagasic patients with severe congestive heart failure. Int J Cardiol. 2002 Oct;85(2-3):255-60.) (258258. Freitas H, Salaltino G, Chizzola P, Costa J, Mansur A, Ramires JF, et al. Betablockers for Chagas' heart disease. J Am Coll Cardiol. 1999;33 Suppl A:184. . However, the use of an optimized dose of adrenergic blockers may be hindered in CCC, given bradyarrhythmias and frequent need of amiodarone administration for the treatment of tachyarrhythmias. To date, there is no consensus among experts about which drug, beta-blockers or amiodarone, should be prioritized in the management of Chagas disease.
Complementary forms of treatment for heart failure caused by Chagas disease
Heart transplantation is the treatment of choice for end-stage heart failure, despite the many limitations of medical and social nature that embargo its broader use in cases of Chagas disease. The results obtained in a selected series of cases, compared with those observed in cases with heart failure due to other etiologies, show an even more favorable evolution in transplanted cases with Chagas disease. Among other possible factors, this is possibly due to the fact that, in this series, cases with Chagas disease were younger and had fewer comorbidities 259259. Bocchi EA, Bellotti G, Uip D, Kalil J, Lourdes Higuchi M, Fiorelli A, et al. Long-term follow-up after heart transplantation in Chagas' disease. Transplant Proc. 1993 Feb;25(1 Pt 2):1329-30.) (260260. Carvalho VB, Sousa EF, Vila JH, Silva JP, Caiado MR, Araujo SR, et al. Heart transplantation in Chagas' disease. 10 Years after the initial experience. Circulation. 1996 Oct;94(8):1815-7. . There is no definitive indication for prophylaxis; however, active control is recommended for possible reactivation the disease, especially in the first year after transplantation, when immunosuppression is more pronounced. Benznidazole, in a dose of 5mg/kg/day, should be promptly started and maintained for 60 days to obtain a good clinical outcome 100100. Pinazo MJ, Miranda B, Rodríguez-Villar C, Altclas J, Brunet Serra M, García-Otero EC, et al. Recommendations for management of Chagas disease in organ and hematopoietic tissue transplantation programs in nonendemic areas. Transplant Rev (Orlando). 2011 Jul;25(3):91-101. .
There is no evidence to support routine indication of cardiac resynchronization therapy (CRT). In addition to the fact that there are no controlled studies exploring in a scientific and valid manner this therapeutic possibility, in CCC the complete block of the right bundle branch is frequent and, in the presence of this condition, evidence of the benefits of CRT, even in other etiologies of heart failure, are even more scarce and less convincing 203203. Castro I, Andrade JP, Paola AAV, Vilas-Boas F, Oliveira GMM, Marin-Neto JA, et al. Sociedade Brasileira de Cardiologia. I Diretriz Latino-Americana para o diagnóstico e tratamento da cardiopatia chagásica. Arq Bras Cardiol. 2011 Aug;97(2 Suppl 3):1-48.) (244244. Martinelli Filho M, Zimerman LI, Lorga AM, Vasconcelos JTM, Rassi Júnior A; Sociedade Brasileira de Cardiologia. Diretrizes brasileiras de dispositivos eletrônicos implantáveis (DCEI). Arq Bras Cardiol. 2007; 89(6):e210-e238. .
Cell implant-based therapies were recently explored in a single study, properly controlled. The results did not show any benefit, even on surrogate outcomes such as the left ventricular ejection fraction 261261. Ribeiro dos Santos R, Rassi S, Feitosa G, Grecco OT, Rassi Júnior A, Cunha AB, et al; Chagas Arm of the MiHeart Study Investigators. Cell therapy in Chagas cardiomyopathy (Chagas arm of the multicenter randomized trial of cell therapy in cardiopathies study): a multicenter randomized trial. Circulation. 2012 May;125(20):2454-61. .
The recommendation classes and levels of evidence for treating heart failure in patients with CCC are summarized in Figure 10 .
Recommendations and levels of evidence for the treatment of heart failure in patients with chronic Chagas cardiomyopathy.
III - Thromboembolic syndrome
Systemic and pulmonary thromboembolic events are common in CCC. Pulmonary thromboembolism is certainly under-diagnosed when considering the results of postmortem studies 262262. Samuel J, Oliveira M, Correa De Araujo RR, Navarro MA, Muccillo G. Cardiac thrombosis and thromboembolism in chronic Chagas' heart disease. Am J Cardiol. 1983 Jul;52(1):147-51. . Embolic events are favored by the combination of several factors. The most important are venous stasis, reduction in cardiac output, and intracardiac mural thrombus, favored by localized ventricular dyskinesias (e.g., apical aneurysm), dilation of the cardiac chambers, and atrial fibrillation in more advanced stages of the cardiomyopathy.
Prevention of thromboembolic complications in chronic Chagas cardiomyopathy
Embolic accidents in the central nervous system are the most severe form of thromboembolism in CCC and contribute to the high morbidity and mortality of the disease 263263. Lopes ER, Marquez JO, Costa Neto B, Menezes AAC, Chapadeiro E. Associação entre acidentes vasculares encefálicos e doença de Chagas. Rev Soc Bras Med Trop. 1991 Apr-Jun;24(2):101-4. . The scientific basis for antithrombotic treatment in CCC is based on findings of a prospective cohort study of 1,043 cases, which assessed the risk and defined prevention strategies for cardioembolic stroke (CES) 264264. Sousa AS, Xavier SS, Freitas GR, Hasslocher-Moreno A. Prevention strategies of cardioembolic ischemic stroke in Chagas' disease. Arq Bras Cardiol. 2008 Nov;91(5):306-10. . In that study, the incidence of CES was 3.0% (or 0.56% per year), and the multivariate statistical analysis allowed the composition of a risk score for CES occurrence: The presence of left ventricular systolic dysfunction contributed 2 points; while presence of apical aneurysm, primary alteration of ventricular repolarization in ECG, and age over 48 years contributed 1 point each. Warfarin may be indicated for cases with 4-5 points (in this subgroup, the incidence of stroke is 4.4% vs. 2.0% of severe bleeding per year). In the subgroup with a score of 3 points, embolism rates and bleeding with oral anticoagulants are equivalent, and acetylsalicylic acid or warfarin may be indicated. In cases with 2 points, with low incidence of CES (1.22% per year), acetylsalicylic acid or no prophylaxis is recommended. Cases with 0-1 point, with an event incidence close to zero, do not require prophylaxis. Obviously, in cases with intracavitary thrombus, atrial fibrillation associated with CHA 2 DS 2 VASc score ≥2, and previous embolic accident, the use of oral anticoagulants is always advisable, maintaining the INR between 2 and 3 264264. Sousa AS, Xavier SS, Freitas GR, Hasslocher-Moreno A. Prevention strategies of cardioembolic ischemic stroke in Chagas' disease. Arq Bras Cardiol. 2008 Nov;91(5):306-10. . Recommendations for oral anticoagulation in CCC are described in Figure 11 .
Pregnancy approach in chronic Chagas cardiomyopathy
Chronic Chagas cardiomyopathy ranks second among the cardiomyopathies present in the pregnancy-puerperal cycle, being second only to rheumatic cardiomyopathy. Most pregnant women with Chagas disease are asymptomatic or oligosymptomatic, being carriers of indeterminate or initial cardiac forms.
The risks of pregnancy in women with Chagas cardiomyopathy depend on the cardiac functional status and the presence and severity of arrhythmias. Pregnancy should be discouraged in cases with heart failure and/or arrhythmias. Pregnant women with these conditions require monitoring and special care, due to the possibility of worsening during pregnancy. In the initial consultation of the pregnant woman with Chagas disease-induced cardiomyopathy, in addition to routine tests, ECG and echocardiography (to assess cardiac dimensions and ventricular function) and a 24-hour Holter (in order to identify conduction disorders and arrhythmias) should be requested 265265. Oliveira Júnior W. O cardiopata chagásico em situações especiais. In: Pinto JC, Rodrigues J (Orgs). Clínica e terapêutica da doença de Chagas. 2 ed. Rio de Janeiro: Fiocruz; 1997. p. 293-322. . The relative and absolute contraindications to the use of drugs acting on the cardiovascular system or with a teratogenic potential should always be observed. Pregnant women with CCC must be attended to at reference centers for high-risk pregnancy, as they may require high-complexity care. It is worth highlighting the importance of carrying out all recommended assessments during the prenatal period, including HIV testing 2020. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Consenso Brasileiro em Doença de Chagas. Rev Soc Bras Med Trop. 2005;38 Supl 3:1-29. .
Surgical risk in patients with chronic Chagas disease
The evaluation of surgical risk in patients with Chagas cardiomyopathy is based on available data related to other heart diseases. However, the unique characteristics of CCC, especially those related to autonomic dysfunction, complex arrhythmias, and intraventricular stimulus conduction disturbances, can cause different responses to surgical trauma 266266. Bestetti RB. Avaliação do risco cirúrgico em pacientes portadores de cardiopatia chagásica crônica em cirurgias não cardíacas. In: Pinto JC, Rodrigues J (Orgs). Clínica e terapêutica da doença de Chagas. 2 ed. Rio de Janeiro: Fiocruz; 1997. p. 281-91. . Cases with more severe myocardial damage (NYHA class IV heart failure or LVEF <30%; moderate systolic dysfunction, but with complex arrhythmia, atrial fibrillation, significant dilation of the left ventricle, total AVB and sinus node dysfunction) are more likely to have perioperative complications.
Asymptomatic sinus bradycardia in patients undergoing surgery under general anesthesia should be monitored by ECG during the procedure, which must be carried out where there is the possibility of performing emergency pacemaker implantation. If bradycardia is symptomatic, preventive implantation of pacemaker should be considered.
Patients with atrial fibrillation with a high ventricular rate (>90 beats/min), regardless of the type of anesthesia, should be operated on under continuous ECG monitoring and prior scanning. In patients with atrial fibrillation with a ventricular rate <60 beats/min, the possibility of a marked decrease in the heart rate due to anesthetic agents must be borne in mind. In these cases, the surgical procedure should be performed under continuous ECG monitoring and in an institution able to perform emergency pacemaker implantation. The same care should be taken in relation to cases with AVB associated with bundle branch block, due to the possibility of developing full AVB during surgery due to the action of anesthetic agents 266266. Bestetti RB. Avaliação do risco cirúrgico em pacientes portadores de cardiopatia chagásica crônica em cirurgias não cardíacas. In: Pinto JC, Rodrigues J (Orgs). Clínica e terapêutica da doença de Chagas. 2 ed. Rio de Janeiro: Fiocruz; 1997. p. 281-91. .
Cases with complex ventricular arrhythmias should be evaluated by Holter and operated on after institution of appropriate antiarrhythmic therapy. The surgical procedure should be performed under continuous ECG monitoring and where there are defibrillators.
Preoperative evaluation of patients with cardiac pacemaker includes knowing the arrhythmia that lead to the implant, as well as generator characteristics. In addition to the clinical history, important data can be obtained from the card that cardiac pacemaker carriers receive and identifies the main characteristics of the device, such as the date of the implant, programmed heart rate, operating mode, and manufacturer.
The main risks associated with the presence of a pacemaker during surgical or diagnostic procedures consist of changing the unit's operating threshold (by the action of drugs, changes in serum potassium levels, and changes in thoracic impedance for pulmonary ventilation), ventricular fibrillation (conduction of the electrical current from the electrocautery by the intracardiac electrode pacemaker) and, finally, damage, inhibition or reprogramming of the system by electrocautery, cardioversion, or magnetic resonance imaging. Whenever possible, biphasic electrocautery (bipolar) should be used, since the electric current remains confined between the region of the surgery and the electrocautery, reducing the risk of leakage and interference with the device 244244. Martinelli Filho M, Zimerman LI, Lorga AM, Vasconcelos JTM, Rassi Júnior A; Sociedade Brasileira de Cardiologia. Diretrizes brasileiras de dispositivos eletrônicos implantáveis (DCEI). Arq Bras Cardiol. 2007; 89(6):e210-e238.) (267267. Ramos G, Ramos Filho J, Rassi Júnior A, Pereira E, Gabriel Neto S, Chaves E. Marcapasso cardíaco artificial: considerações pré e per-operatórias. Rev Bras Anestesiol. 2003 nov-dez;53(6):854-62. .
In any case, the use of electrocautery should be minimized. It should be used in short and irregular intervals, monitoring not only the ECG, but also the pulse by plethysmography or pulse oximeter. In the event of severe bradycardia or tachycardia during the use of the electrocautery, a magnet cover can be used over the pacemaker, which makes it work in its programmed magnetic response. Attention should be paid to the fact that the placement of the magnet should be restricted to the short periods of electrocautery use. The patient should be advised postoperatively to return to the clinic for pacemaker follow-up for the assessment of its operation and eventual reprogramming of its generator 267267. Ramos G, Ramos Filho J, Rassi Júnior A, Pereira E, Gabriel Neto S, Chaves E. Marcapasso cardíaco artificial: considerações pré e per-operatórias. Rev Bras Anestesiol. 2003 nov-dez;53(6):854-62. .
In patients with a CDI, the presence of technician or specialist in the operating room is recommended. The antitachycardia function should be switched off, with staff prepared to treat any possible arrhythmias, including having to perform electrical cardioversion 244244. Martinelli Filho M, Zimerman LI, Lorga AM, Vasconcelos JTM, Rassi Júnior A; Sociedade Brasileira de Cardiologia. Diretrizes brasileiras de dispositivos eletrônicos implantáveis (DCEI). Arq Bras Cardiol. 2007; 89(6):e210-e238.) (267267. Ramos G, Ramos Filho J, Rassi Júnior A, Pereira E, Gabriel Neto S, Chaves E. Marcapasso cardíaco artificial: considerações pré e per-operatórias. Rev Bras Anestesiol. 2003 nov-dez;53(6):854-62. .
If electrical cardioversion is required in patients with pacemakers, the ideal would be to use adhesive pads in the anteroposterior chest position, as remote as possible from the generator, using as little energy as possible for resolution of the arrhythmia. Similarly, the thresholds and the functioning of the pacemaker should ideally be assessed after the procedure 244244. Martinelli Filho M, Zimerman LI, Lorga AM, Vasconcelos JTM, Rassi Júnior A; Sociedade Brasileira de Cardiologia. Diretrizes brasileiras de dispositivos eletrônicos implantáveis (DCEI). Arq Bras Cardiol. 2007; 89(6):e210-e238. .
Occupational health evaluation
Chronic Chagas cardiomyopathy is an important cause of incapacity to work in endemic areas. It should be the subject of surveillance actions for workers' health, included in primary care. Careful clinical examination, with particular attention to the cardiovascular system, is the basic component of functional and occupational health evaluation in CCC 268268. Prata A. Implicações epidemiológicas e sócio-econômicas da doença de Chagas. Brasilia Med. 1973;9:69-71. . These assessments should take into account: the degree of ventricular dysfunction; the presence, magnitude and complexity of conduction and rhythm disorders; and the thromboembolism risk. It is also important to consider the characteristics of the labor activity performed by the worker, especially the need for intense or continued physical effort in the activity, as well as the degree of professional qualification of the worker and his/her ability to enroll in a vocational rehabilitation program. The age of the worker should also be considered, as the possibility of evolution of heart disease is greater for younger workers and vice versa. In general, the worse the LVEF and the greater the ventricular diameter, the worse the prognosis and the greater the inability to work 269269. Porto CC, Rassi A, Faria GHDC. Aspectos sócio-econômicos e médico-trabalhistas. In: Cançado JR, Chuster M (editores). Cardiopatia Chagásica. Belo Horizonte: Fundação Carlos Chagas; 1985. p. 362-6. .
The presence of cardiomegaly and significant ventricular dysfunction implies incapacity to perform activities that require physical effort, and, when vocational rehabilitation is not possible, disability. In all functional assessments of prognosis and working capacity of an individual with Chagas disease, the great clinical variability of these cases should be taken into account, even when in the same stage of any classification of cardiac impairment. Often, but not always, advanced disorders of stimulus conduction are associated with the severity of myocardial damage, leading to the need for a complementary evaluation of the degree of myocardial dysfunction, especially in cases where the work requires intense physical activity and involves personal or social risk (e.g., aviation pilots) 270270. Dias JCP. Aspectos clínicos, sociais e trabalhistas da doença de Chagas em área endêmica sob controle do estado de Minas Gerais, Brasil. Rev Soc Bras Med Trop. 1999 apr-jun;26(2):93-9. .
DIGESTIVE FORM OF CHAGAS DISEASE
The digestive form of Chagas disease can affect all organs in the gastrointestinal tract. However, from a practical point of view, it is manifested as involvement of the esophagus and large intestine, resulting in megaesophagus and megacolon, respectively 2020. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Consenso Brasileiro em Doença de Chagas. Rev Soc Bras Med Trop. 2005;38 Supl 3:1-29. . The association between megaesophagus and megacolon in cases that have an indication for surgery is approximately 92%. The association between megaesophagus, megacolon, and heart disease is 65% 271271. Morais BB, Martins P, Maiolini DD, Silva AL, Melo JRC. Concomitant lesions in Chagas' disease. Eur J Gastroenterol Hepatol. 1991;3(7):523-6. . A specific search for T. cruzi infection should be undertaken in patients with clinical syndromes consistent with those described below, who live in at-risk contexts and/or who have increased vulnerability to Chagas disease 2020. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Consenso Brasileiro em Doença de Chagas. Rev Soc Bras Med Trop. 2005;38 Supl 3:1-29. .
Diagnosis
Digestive manifestations in the acute phase of Chagas disease
Clinical manifestations are usually nonspecific, with practically imperceptible symptoms related to the gastrointestinal tract. However, there are reports describing the occurrence of gastrointestinal bleeding in cases of oral transmission and of dysphagia in rare cases of vectorial transmission 2020. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Consenso Brasileiro em Doença de Chagas. Rev Soc Bras Med Trop. 2005;38 Supl 3:1-29. .
Digestive manifestations in the chronic phase of Chagas disease
Digestive manifestations of Chagas disease are concentrated in the esophagus and colon and are basically dysphagia and constipation, due to chronic alterations that can lead to megaesophagus and/or megacolon 2020. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Consenso Brasileiro em Doença de Chagas. Rev Soc Bras Med Trop. 2005;38 Supl 3:1-29. . However, as the disease causes autonomic nervous system injury throughout the gastrointestinal tract, anatomical and functional abnormalities of the salivary glands, stomach, extra-hepatic bile ducts, duodenum, small intestine, large intestine, and even of organs not belonging to the gastrointestinal tract, such as the ureter, can occur. The prevalence of Helicobacter pylori infection and of characteristic gastric endoscopic and histologic changes were similar in patients with and without Chagas disease. Furthermore, studies have shown that H. pylori is the main cause of gastritis in patients with Chagas disease 272272. Fonseca FM, Etchebehere RM, Queiroz DM, Rocha AM, Junqueira IS, Fonseca DN, et al. Histological and endoscopic features of the stomachs of patients with Chagas disease in the era of Helicobacter pylori . Rev Soc Bras Med Trop. 2014 Nov-Dec;47(6):739-46. . The most common symptoms and signs of the digestive system, in the chronic phase of Chagas disease, are described below.
Esophagus
Clinical diagnosis
Dysphagia is the main symptom of patients with severe impairment of the esophagus. Other dysphagia-related complaints occur in cases of megaesophagus, including ptyalism, hiccups, odynophagia, regurgitation, a sense of nocturnal choking, aspiration pneumonia, and malnutrition 2020. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Consenso Brasileiro em Doença de Chagas. Rev Soc Bras Med Trop. 2005;38 Supl 3:1-29. . In the early stages of the disease, the patient reports a feeling of obstruction at the level of the xiphoid after eating solids and, later, after fluid intake, especially cold fluids. Dysphagia progresses slowly and is well-tolerated for many years. Therefore, patients with achalasia often do not seek care in health services until the progressive dysphagia interferes with their lifestyle. It is observed that the patients themselves spontaneously change their eating habits to alleviate dysphagia. Patients with achalasia feed slowly, ingest large volumes of water to assist the food into the stomach, often bend the back, lift the chin, extend the neck, or walk to help esophageal emptying. Regurgitation of undigested food is common as the disease progresses, with a risk of aspiration, which may lead to pneumonia, lung abscess, bronchiectasis, hemoptysis, and bronchospasm. Great distension of the dilated esophagus may lead to dyspnea, compression of the main stem of the bronchus or lung hilum.
Esophageal involvement occurs to varying degrees, with great variation in morphology and differences of esophageal motor behavior, ranging from minimal changes of esophageal transit to advanced forms of dolicomegaesophagus with an extremely prolonged esophagogastric transit time 2020. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Consenso Brasileiro em Doença de Chagas. Rev Soc Bras Med Trop. 2005;38 Supl 3:1-29. . Considering practical and therapeutic aspects, patients with megaesophagus can be classified into groups, depending on the degree of expansion of the organ, as shown in Figure 12 273273. Rezende JM. Classificação radiológica do megaesôfago. Rev Goiana Med. 1982 jul-dez;28(3/4):187-91. .
Radiologic classification of megaesophagus. (A) Group I: The esophageal diameter is within normal limits, without food stasis, but with an increase in food transit time from mouth to stomach. Functional motor disturbances predominate without corresponding dilation; (B) Group II: Moderate dilation of the esophagus with loss of motor control. Secondary and tertiary waves can be seen in the esophagogram. (C) Group III: More accentuated dilation than in group II, transit time more prolonged, and motor activity reduced. The esophagus behaves like an inert tube. (D) Group IV: Advanced form with massive dilation and stretching of the esophagus.
Most cases of megaesophagus are found in groups II and III. The degree of megaesophagus does not necessarily reflect the duration of the disease. There are cases that rapidly progress to advanced forms, while others remain stable in the early stages. Lower esophageal sphincter dysfunction is greater in cases where the motor abnormalities of the esophagus are more evident. Although there is a correlation between the degree of megaesophagus and the severity of dysphagia, it is common to find patients with bulky megaesophagus complaining about or not even reporting dysphagia 273273. Rezende JM. Classificação radiológica do megaesôfago. Rev Goiana Med. 1982 jul-dez;28(3/4):187-91. . Notably, the incidence of esophageal cancer in patients with idiopathic achalasia is 3.3%, 15 times higher than that of the general population without achalasia 274274. Brücher BL, Stein HJ, Bartels H, Feussner H, Siewert JR. Achalasia and esophageal cancer: incidence, prevalence, and prognosis. World J Surg. 2001 Jun;25(6):745-9.) (275275. Leeuwenburgh I, Scholten P, Alderliesten J, Tilanus HW, Looman CW, Steijerberg EW, et al. Long-term esophageal cancer risk in patients with primary achalasia: a prospective study. Am J Gastroenterol. 2010 Oct;105(10):2144-9. .
Complementary examinations
-
Plain radiography of the thorax (posterior-anterior or lateral views): In cases of megaesophagus grade III or IV, great dilation of the esophagus extending to the posterior mediastinum can be observed ( Figure 13 A and B).
Plain radiography of the thorax. (A) Posterior-anterior view and (B) lateral view, demonstrating the great dilation of the esophagus extending to the posterior mediastinum.
Contrast radiography of the esophagus: This shows not only the degree of esophageal dilation, allowing classification of megaesophagus, but also demonstrates functional changes, such as abnormal or missing peristaltic waves and the emptying time of esophageal contents into the stomach. The radiologic characteristics of megaesophagus in Chagas disease are: increased esophageal diameter and emptying time ( Figure 14 ); a tapered distal third; retention of food debris; esophageal/mediastinal stretching; and reduced gastric volume.
-
Esophageal electromanometry: This is a useful method to distinguish between the differential diagnoses of other diseases that develop due to motor dysfunction that present with dysphagia. In patients without dilatation, or with slight dilation of the esophagus (group I and II), uncoordinated pressure waves can occur in the body of the esophagus and incomplete or atypical relaxation of the lower esophageal sphincter (LES) can be observed. These findings precede radiologically demonstrated abnormalities 276276. Pérez-Ayala A, Pérez-Molina JA, Norman F, Monge-Maillo B, Faro MV, López-Vélez R. Gastro-intestinal Chagas disease in migrants to Spain: prevalence and methods for early diagnosis. Ann Trop Med Parasitol. 2011 Jan;105(1):25-9. .
-
Endoscopy: Although endoscopy is not essential to confirm the diagnosis of megaesophagus, it should be performed to assess the degree of mucosal inflammation and to exclude the presence of neoplastic lesions in patients with dysphagia, varicose veins, and ulcers. This examination is useful in cases requiring forced balloon dilatation of the gastric cardia and in cases requiring botulinum toxin injection.
Contrast study of a patient with esophageal achalasia (group III). Images taken (A) 2 minutes and (B) 6 hours after ingestion of contrast. Note the pacemaker wires implanted in this patient due to AVB secondary to concomitant Chagas disease-associated cardiomyopathy.
Colon
Clinical diagnosis
The main signs and symptoms of megacolon are constipation, paradoxical diarrhea (constipation interspersed by periods of diarrhea), dyschezia, abdominal distension, and fecaloma 2020. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Consenso Brasileiro em Doença de Chagas. Rev Soc Bras Med Trop. 2005;38 Supl 3:1-29. . Anorectal manometry is useful to diagnose internal anal sphincter achalasia. Colonic dilatation located in the rectum and sigmoid is better observed by contrast radiography studies and can be detected in up to 80% of cases. However, dilatation can occur only in the rectum, only in the sigmoid, or in the entire colon 277277. Oliveira RB, Troncon LEA, Dantas RO, Meneghelli UG. Gastrointestinal manifestations of Chagas disease. Am J Gastroenterol. 1998 Jun;93(6):884-9. . It should be emphasized that the clinical picture of severe constipation in patients with idiopathic megacolon who are not infected with T. cruzi is indistinguishable from that induced by Chagas disease 278278. Meier-Ruge WA, Müller-Lobeck H, Stoss F, Bruder E. The pathogenesis of idiopathic megacolon. Eur J Gastroenterol Hepatol. 2006 Nov;18(11):1209-15. .
Complementary tests
-
Plain radiography of the abdomen: Bulky megacolons can be detected on plain radiographic films of the abdomen, especially if a fecaloma is present. Plain radiography is also useful in cases of suspected volvulus, since it can demonstrate rotation of the sigmoid and dilation of small bowel loops caused by obstruction ( Figure 15 ).
-
Contrast radiography of the colon (barium enema): This is the gold standard for the diagnosis of megacolon. It shows dilation and/or stretching of the colon, presence of fecaloma, and anatomic abnormalities of the sigmoid ( Figure 16 ).
-
Computed tomography and magnetic resonance imaging: These more modern imaging tests occupy a prominent place in the evaluation of patients with Chagas disease and are gradually replacing conventional imaging methods. Both methods are more sensitive in evaluating pericolonic and/or perisophageal tissues; they can detect infiltration, not only visible masses and fecalomas detectable by conventional radiography. These modalities also provide greater detail than a barium enema ( Figure 17 A, B and C).
-
Digestive endoscopy: This is useful in cases of megacolon, to assess the degree of mucosal inflammation, detect ulcers, wall necrosis, perforation or bleeding, and to rule out the presence of associated lesions, particularly cancer. It is particularly important in cases of suspected sigmoid volvulus and can be therapeutic, since it might allow laparotomy to be avoided. If it is not possible to reduce the sigmoid volvulus by endoscopic examination, emergency surgery is absolute indicated.
Simple radiograph of the abdomen in a case of sigmoid volvulus. Note the anomalous position of the sigmoid caused by the dilatation and the characteristic image of the small bowel loops secondary to the obstruction caused by the volvulus.
Radiograph of a barium enema examination revealing a hypodense image typical of a fecaloma.
Abdominal computed tomography, showing a megaesophagus with an air-fluid level formed by ingested contrast (A); megacolon with a large dilatation of the air-filled colon with a fecaloma in the rectum (B); and megacolon with a predominant image of a fecaloma in the dilated sigmoid (C).
Megaesophagus
Considering the impossibility of restoring normal physiology due to the irreversible changes caused by denervation, the goal of treatment is to act on the lower esophageal sphincter in a patient with achalasia, by removing the functional barrier to the passage of food into the stomach. The treatment, except if specific, does not cure the disease; it seeks only improve the symptom of dysphagia. 2020. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Consenso Brasileiro em Doença de Chagas. Rev Soc Bras Med Trop. 2005;38 Supl 3:1-29.
Clinical treatment
This is indicated in patients with advanced age, with no history of complications, who are oligosymptomatic, are at high risk for surgical treatment, who refuse to undergo invasive treatments, or those treated in hospitals lacking adequate infrastructure to perform this type of surgery 2020. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Consenso Brasileiro em Doença de Chagas. Rev Soc Bras Med Trop. 2005;38 Supl 3:1-29. . The basis for the treatment is specified below.
-
Counseling and health education related to megaesophagus, in order to improve patient safety.
-
Adequacy of eating habits . It is recommended that the patient chew their food well; eat small amounts at a time; and consume liquid foods, pastes, and solids appropriate to their severity of dysphagia. Irritating foods, such as very spicy or hot foods or ice cream, aggravate dysphagia and should be avoided. Due to the risk of regurgitation and aspiration, food intake should be avoided before bedtime. Balanced enteral nutrition is indicated in patients with megaesophagus in Groups III and IV, malnourished patients, and candidates for surgical treatment. If the passage of a nasoenteric tube is not possible, parenteral nutrition can be administered via a central venous catheter. In both situations, the intervention is temporary.
-
Use of drugs: Medicines that relax smooth muscle fibers of the lower esophageal sphincter can relieve the symptoms of dysphagia. These drugs include:
-
Isosorbide dinitrate, at a dose from 2.5mg to 5mg, administered sublingually, 15 minutes before each meal.
-
Nifedipine, at a dose of 10mg, administered sublingually, 30 minutes before each meal. The use of nifedipine should be performed with caution, considering the significant association of this condition with chronic Chagas cardiopathy; nifedipine may induce hypotension and shock.
-
Botulinum toxin: Botulinum toxin type A, applied locally, blocks the release of acetylcholine from pre-synaptic nerve endings and leads to chemical denervation. The treatment is effective in 70% of patients, and the average duration of symptom remission is 16 months. Patients over 50 years of age respond better to this treatment 279279. Pasricha PJ, Rai R, Ravich WJ, Hendrix TR, Kalloo AN. Botulinum toxin for achalasia: long-term outcome and predictors of response. Gastroenterology. 1996 May;110(5):1410-5.) (280280. Ferguson MK. Achalasia: current evaluation and therapy. Ann Thorac Surg. 1991 Aug;52(2):336-42. . Botulinum toxin treatment of megaesophagus has worse outcomes than those of dilation or surgery (level IIB). Although the effect is temporary, botulinum toxin injection may be indicated in selected cases, presenting the advantage that it can be repeated several times. The dose of botulinum toxin is 20 units injected into each of the lower quadrants of the esophageal sphincter. One of the limitations of this treatment is that the procedure should be guided by endoscopy.
-
Balloon dilatation: Pneumatic dilation is indicated for patients with group I megaesophagus, but can be performed in patients in groups II and III who have a contraindication to surgery. It can also be used preoperatively in patients in groups II and III who are candidates for surgical treatment of megaesophagus, aiming to improve their preoperative nutritional status. In patients in group IV, due to the risk of redundant esophageal perforation, dilatation is not recommended. The procedure is performed with a balloon coupled to gauge introduced endoscopically, which makes it the safest method. The result of megaesophagus treatment by balloon dilation relieves dysphagia in 71% of patients 280280. Ferguson MK. Achalasia: current evaluation and therapy. Ann Thorac Surg. 1991 Aug;52(2):336-42. . However, the effect is transient, worsening over time with successive dilations.
Surgical treatment
-
Conventional surgery (laparotomy): This does not address the underlying cause of the disease, but restores the ability of the patient to swallow. Based on the knowledge gained about the pathogenesis and pathophysiology of megaesophagus, the cardiomyotomy known as Heller surgery 281281. Heller E. Extramukose Cardiaplastik beim chronischen Cardiospasmus mit Dilatation des Oesophagus. Mitt Grenzgeb Med Chir. 1913;27(1):141-49. , associated with some kind of partial fundoplication 282282. Pinotti HW, Habr-Gama A, Cecconello I, Felix VN, Zilbertein B. The surgical treatment of megaesophagus and megacolon. Dig Dis. 1993 Jul-Oct;11(4-5):206-15. , is the most appropriate surgical procedure for cases of megaesophagus grades I to III. The response rate is about 90% 280280. Ferguson MK. Achalasia: current evaluation and therapy. Ann Thorac Surg. 1991 Aug;52(2):336-42. , giving a more long-lasting and effective result than that achieved by pneumatic dilatation. For dolichomegaesophagus, there is still no consensus among surgeons; hence, different surgical techniques are still used.
-
Laparoscopic surgery: This technique was introduced in Brazil in the early 1990s, with simultaneous availability of modern surgical instruments, such as electronic scalpels and staplers. The experience of the surgeons with this technological innovation and its use in the treatment of cases of megaesophagus from achalasia related to Chagas disease, has reduced the morbidity and mortality associated with conventional surgery for megaesophagus induced by Chagas disease (level IA). Laparoscopic cardiomyotomy, associated with some type of antireflux valve, especially those made by partial fundoplication, has become standard surgery; it is virtually the standardized surgical procedure for patients with this condition 283283. Spiess AE, Kahrilas PJ. Treating achalasia: from whalebone to laparoscope. JAMA. 1998 Aug;280(7):638-42. .
-
Robotic surgery: Robotic surgery is probably the most advantageous of all the surgical options. However, the complexity of its implementation and the high associated costs hinder its routine application in practice 2020. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Consenso Brasileiro em Doença de Chagas. Rev Soc Bras Med Trop. 2005;38 Supl 3:1-29. . Megaesophagus treatment by myotomy associated with antireflux surgery carries a risk of esophageal perforation; this complication can be severe. Robotic surgery, by making scheduled and precise movements, will most certainly reduce the rate of this complication.
-
Endoscopic surgery: Technological advances and the increasing expertise of professionals has allowed myotomy to be performed as a totally endoscopic procedure. This surgery is now known by the acronym POEM (per-oral endoscopic myotomy). It uses the Heller technique, performed by transluminal endoscopic surgery through natural orifices [natural orifice transluminal endoscopic surgery (NOTES)]. This procedure was introduced by professionals from the Mayo Clinic Endoscopic division and, after testing on animals, the procedure was widely disseminated, being used in humans since 2012. The POEM technique seems to be a safe method, and is currently an effective alternative for the treatment of achalasia.
-
The procedure is performed with the person in the supine position under general anesthesia, and consists in approaching the esophageal lumen with a high-definition gastroscope with frontal view. Saline stained with indigo carmine is injected to create a safety gap between the mucosa and submucosa of the esophagus. Then, a 2cm longitudinal incision of the mucosa is performed, at the two o'clock position, 10-15cm proximal to the gastroesophageal junction, to expose the submucosal layer. A tunnel is dissected in the submucosa, extending from the point of section to 2-3cm beyond the gastroesophageal junction (GEJ) 284284. NOSCAR POEM White Paper Committee; Stavropoulos SN, Desilets DJ, Fuchs KH, Gostout CJ, Haber G, Inoue H, et al. Per-oral endoscopic myotomy white paper summary. Gastrointest Endosc. 2014 Jul;80(1):1-15. . Myotomy of the lower esophageal sphincter is usually performed starting at 2cm distal to the opening of the mucosa to below the GEJ. As a rule, myotomy should have a minimum length of 6cm (on average, 8-10cm): 2cm in the esophagus, 2-3cm in the lower esophageal sphincter, and 2cm in the cardia. Dissection of the submucosal tunnel is performed in the plane before the muscle of the mucosa, and esophageal muscle section is performed only in the circular layer of the muscle, there is no need for longitudinal muscle layer section.
-
Data from 14 English language publications show that of 804 patients who underwent a POEM procedure, therapeutic success was documented in more than 80% (level A1) 285285. Inoue H, Tianle KM, Ikeda H, Hosoya T, Onimaru M, Yoshida A, et al. Peroral endoscopic myotomy for esophageal achalasia: technique, indication, and outcomes. Thorac Surg Clin. 2011 Nov;21(4):519-25. . The technique is less invasive and the patients do not experience gastroesophageal reflux, as observed in cases submitted to myotomy by conventional laparotomy or laparoscopy surgical technique. The familiarization of the endoscopist with the method enables secure closure of smaller perforations and adequate hemostasis (level B1) 286286. Swanstrom LL, Kurian A, Dunst CM, Sharata A, Bhayani N, Rieder E. Long-term outcomes of an endoscopic myotomy for achalasia: the POEM procedure. Ann Surg. 2012 Oct;256(4):659-67. . If there is a more extensive perforation of the esophagus, or more intense bleeding, the assistance of a surgeon may be necessary.
Megacolon
Clinical treatment
-
Health counseling and education related to megaesophagus, in order to improve patient safety.
-
Adequacy of eating habits: This should focus on a higher degree of self-care. Patients evacuated two to three times a week without reporting a fecaloma should be instructed to drink more fluids, and eat more fruits and vegetables. The use of a diet rich in fiber in these patients is controversial, because there is the fear that excess fiber may favor fecal impaction and the worsening of symptoms. The diet can be the patient's usual diet, with restriction of constipating foods. Abundant water intake is recommended - a volume of at least 2L/day - as well as foods that favor intestinal functioning.
-
Regular defecation: It is necessary to systematically pay attention to the urge to defecate and to create the habit to evacuate at certain hours.
-
Laxatives: These are indicated for patients who do not adequately respond to dietary measures. Osmotic laxatives (20% mannitol or polyethylene glycol) are the most suitable. Mineral oil is also effective.
-
Avoidance of potentially constipating drugs: Opioids, diuretics, antidepressants, antihistamines, anticonvulsants, antiparkinsonians, among others, should only be prescribed under medical monitoring.
-
Removal of fecaloma: Fecalomas, located in the rectum, can be removed manually (under anesthesia) if they do not respond to dietary and drug measures.
-
Intestinal wash-out: Patients who do not respond satisfactorily to drug and dietary treatment, or those with significant fecalomas, should be treated with glycerin or saline enemas. Intestinal wash-out with 500-1,000mL twice a week is recommended for patients resistant to medical treatment. In patients with fecalomas, the procedure may be repeated two to three times a day, taking care not to use more than 3.5L of solution per day. Experience with these treatments shows that, with the removal of the fecalomas and normalization of bowel movements, the diameter of the colon tends to return to normal.
Surgical treatment
-
Elective surgery: Surgical treatment of megacolon is indicated for patients who have large dilatation of the sigmoid, repeated episodes of volvulus, prolonged retention of feces, recurrent fecalomas, and difficulties in applying enemas at home. Similar to the surgical treatment of megaesophagus, the goal of treating the megacolon is to improve or normalize the intestinal rhythm, since the predominant symptom is constipation. Except in cases of emergency caused by volvulus, bleeding, obstruction or perforation, in which emergency procedures must be adopted (colonoscopy or surgery), megacolon surgery is elective and should be well planned. The most commonly used conventional surgical techniques are the aforementioned rectosigmoidectomy and the surgery originally proposed by DuHamel for the treatment of Hirschsprung disease 287287. Duhamel B. Une nouvelle opération pour le mégacolon cologénital: l'abaissement rétro-rectal et transanal du côlon et son application possible au traitement de quelques autres malformations. Nouv Presse Med. 1956; 64(95):2249-50. . With the advent and spread of laparoscopic surgery, the laparoscopic DuHamel procedure has become the gold standard for treatment of these cases 288288. Reis-Neto JA, Pedroso MA, Lupinacci RA, Reis Júnior JA, Ciquini SA, Lupinacci RM, et al. Megacolo adquirido: perspectivas fisiopatológicas para o tratamento laparoscópico. Rev Bras Colo-proctol. 2004 jan-mar;24(1):49-62. .
-
Emergency surgery: The indications for emergency surgery to treat megacolon are:
-
sigmoid volvulus not resolved by endoscopy; and
-
ulceration, necrosis and/or perforation, due to volvulus or endoscopy.
-
The surgery to be performed will depend on the patient's condition and anatomopathological situation. The surgical procedure may vary from a simple colostomy to fixing the part affected by sigmoid resection with primary or delayed anastomosis.
ANTIPARASITIC TREATMENT OF CHAGAS DISEASE
Although there is disagreement about the cure rates of the antiparasitic treatment of Chagas disease, there is consistent evidence showing its usefulness in both acute and chronic phases of the disease and in all clinical forms of chronic infection, as the organic lesions depend exclusively (acute phase) or at least in part (chronic phase) on the presence of the parasite. In addition, there is apparent suppression of the parasitemia with the current antiparasitic treatment 289289. Coura JR, Borges-Pereira J. Chronic phase of Chagas disease: why should it be treated? A comprehensive review. Mem Inst Oswaldo Cruz. 2011 Sep;106(6):641-5.) (290290. Molina I, Gómez i Prat J, Salvador F, Treviño B, Sulleiro E, Serre N, et al. Randomized trial of posaconazole and benznidazole for chronic Chagas' disease. N Engl J Med. 2014 May;370:1899-908. . In turn, the rates of cure and their confirmation depend on several factors, such as the phase and the duration of disease, the age of the patient, the tests used to evaluate therapeutic efficacy, the duration of follow-up after treatment, associated conditions, and the susceptibility of the specific T. cruzi strain to antiparasitic drugs.
Treatment in the acute phase
In the acute phase, treatment should be provided to all cases and as quickly as possible, regardless of the parasite transmission route (Class I, level of evidence B). Studies of a case series followed for over 20 years have shown cure rates above 50% at this stage of the disease 291291. Cançado JR. Terapêutica específica. In: Dias JCP, Coura JR, editores. Clínica e Terapêutica da Doença de Chagas: uma abordagem prática para o clínico geral. Rio de Janeiro: Fiocruz; 1997. p. 323-51. .
Treatment of congenital Chagas disease
Patients diagnosed with congenital Chagas disease should receive antiparasitic treatment (more details on the specific part of vertical transmission) 2020. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Consenso Brasileiro em Doença de Chagas. Rev Soc Bras Med Trop. 2005;38 Supl 3:1-29. , regardless of whether the diagnosis has been made by parasitologic methods in the first weeks of life or by conventional serologic tests at least nine months after birth (Class I, level of evidence B).
Treatment of chronic phase
All children aged less than 12 years with chronic phase Chagas disease should be treated (Class I, level of evidence A). Prospective, randomized, double-blind, controlled studies with placebo, performed in asymptomatic school-aged children, demonstrated therapeutic cure of T. cruzi infection in 58-62% of cases 292292. Sosa Estani S, Segura EL, Ruiz AM, Velazquez E, Porcel BM, Yampotis C. Efficacy of chemotherapy with benznidazole in children in the indeterminate phase of Chagas' disease. Am J Trop Med Hyg. 1988 Oct;59(4):526-9.) (293293. Andrade AL, Zicker F, Oliveira RM, Silva SA, Luquetti A, Travassos LR, et al. Randomised trial of efficacy of benznidazole in treatment of early Trypanosoma cruzi infection. Lancet. 1996 Nov;348(9039):1407-13. . In an uncontrolled study involving Latin American countries 294294. Yun O, Lima MA, Ellman T, Chambi W, Castillo S, Flevaud L, et al. Feasibility, drug safety, and effectiveness of etiological treatment programs for Chagas disease in Honduras, Guatemala, and Bolivia: 10-year experience of Médecins Sans Frontières. PLoS Negl Trop Dis. 2009 Jul;3(7):e488. , large regional differences were observed in treatment response rates: The seroconversion rate in Honduras was 92.7% for children aged 9 months to 12 years and 58.8% in a quarter of the study patients younger than 15 years. There were variations in Bolivia, from 0% in Sucre to 5.4% in Entre Rios. In the latter region this rate was higher in children younger than 5 years (24.4%) than in children aged 5-9 years (4.6%) 294294. Yun O, Lima MA, Ellman T, Chambi W, Castillo S, Flevaud L, et al. Feasibility, drug safety, and effectiveness of etiological treatment programs for Chagas disease in Honduras, Guatemala, and Bolivia: 10-year experience of Médecins Sans Frontières. PLoS Negl Trop Dis. 2009 Jul;3(7):e488. .
For adolescents (aged 13-18 years) and adults with chronic infection, when it can be established that the acute phase occurred up to 12 years before (considered as recent infection) antiparasitic treatment is usually recommended 2020. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Consenso Brasileiro em Doença de Chagas. Rev Soc Bras Med Trop. 2005;38 Supl 3:1-29.) (294294. Yun O, Lima MA, Ellman T, Chambi W, Castillo S, Flevaud L, et al. Feasibility, drug safety, and effectiveness of etiological treatment programs for Chagas disease in Honduras, Guatemala, and Bolivia: 10-year experience of Médecins Sans Frontières. PLoS Negl Trop Dis. 2009 Jul;3(7):e488. , although there is no consistent evidence from randomized studies justifying this (Class IIa, level of evidence C) 295295. Bern C, Montgomery SP, Herwaldt BL, Rassi Júnior A, Marin-Neto JA, Dantas RO, et al. Evaluation and treatment of Chagas disease in the United States: a systematic review. JAMA. 2007 Nov;298(18):2171-81. .
For individuals with Chagas disease aged 19-50 years with no recent documented infection, antiparasitic treatment should be considered on an individual basis, whether in ICF 120120. Fabbro DL, Streiger ML, Arias ED, Bizai ML, Barco M, Amicone NA. Trypanocide treatment among adults with chronic Chagas disease living in Santa Fe City (Argentina), over a mean follow-up of 21 years: parasitological, serological and clinical evolution. Rev Soc Bras Med Trop. 2007 Jan-Feb;40(1):1-10.) (252252. Viotti R, Vigliano C, Lococo B, Bertocchi G, Petti M, Alvarez MG, et al. Long-term cardiac outcomes of treating chronic Chagas disease with benznidazole versus no treatment: a nonrandomized trial. Ann Intern Med. 2006 May;144(10):724-34. (Class IIa, level of evidence B) or in the determined chronic form, without advanced cardiopathy 4242. Rassi Júnior A, Rassi A, Marin-Neto JA. Chagas disease. Lancet. 2010 Apr;375(9723):1388-402.) (120120. Fabbro DL, Streiger ML, Arias ED, Bizai ML, Barco M, Amicone NA. Trypanocide treatment among adults with chronic Chagas disease living in Santa Fe City (Argentina), over a mean follow-up of 21 years: parasitological, serological and clinical evolution. Rev Soc Bras Med Trop. 2007 Jan-Feb;40(1):1-10.) (252252. Viotti R, Vigliano C, Lococo B, Bertocchi G, Petti M, Alvarez MG, et al. Long-term cardiac outcomes of treating chronic Chagas disease with benznidazole versus no treatment: a nonrandomized trial. Ann Intern Med. 2006 May;144(10):724-34.) (295295. Bern C, Montgomery SP, Herwaldt BL, Rassi Júnior A, Marin-Neto JA, Dantas RO, et al. Evaluation and treatment of Chagas disease in the United States: a systematic review. JAMA. 2007 Nov;298(18):2171-81.) (296296. Coura JR, Abreu LL, Willcox HP, Petana W. Comparative controlled study on the use of benznidazole, nifurtimox and placebo, in the chronic form of Chagas' disease, in a field area with interrupted transmission. I. Preliminary evaluation. Rev Soc Bras Med Trop. 1997 Mar-Apr;30(2):139-44.) (297297. Bitto C, Silveira C, Cardoso MA, Marques P, Luquetti A, Macedo V, et al. Parasite persistence in treated chagasic patients revealed by xenodiagnosis and polymerase chain reaction. Mem Inst Oswaldo Cruz. 2001 Aug;96(6):823-6.) (298298. Gallerano RH, Sosa RR. Resultados de un estúdio a largo plazo con drogas antiparasitárias en infectados chagásicos crónicos. Rev Fed Arg Cardiol. 2001;30:289-96. (Class IIb, level of evidence C). Specifically, treatment of chronically infected women of childbearing age, when provided before pregnancy, can reduce congenital transmission 104104. Carlier Y, Torrico F, Sosa-Estani S, Russomando G, Luquetti A, Freilij H, et al. Congenital Chagas disease: recommendations for diagnosis, treatment and control of newborns, siblings and pregnant women. PLoS Negl Trop Dis. 2011 Oct;5(10):e1250.) (109109. Sosa-Estani S, Cura E, Velazquez E, Yampotis C, Segura EL. Etiological treatment of young women infected with Trypanosoma cruzi , and prevention of congenital transmission. Rev Soc Bras Med Trop. 2009 Sep-Oct;42(5):484-7. .
Some observational studies have shown that in this age group, antiparasitic treatment is able to prevent the onset or delay the progression of the disease in a significant proportion of cases 4242. Rassi Júnior A, Rassi A, Marin-Neto JA. Chagas disease. Lancet. 2010 Apr;375(9723):1388-402.) (120120. Fabbro DL, Streiger ML, Arias ED, Bizai ML, Barco M, Amicone NA. Trypanocide treatment among adults with chronic Chagas disease living in Santa Fe City (Argentina), over a mean follow-up of 21 years: parasitological, serological and clinical evolution. Rev Soc Bras Med Trop. 2007 Jan-Feb;40(1):1-10.) (252252. Viotti R, Vigliano C, Lococo B, Bertocchi G, Petti M, Alvarez MG, et al. Long-term cardiac outcomes of treating chronic Chagas disease with benznidazole versus no treatment: a nonrandomized trial. Ann Intern Med. 2006 May;144(10):724-34.) (295295. Bern C, Montgomery SP, Herwaldt BL, Rassi Júnior A, Marin-Neto JA, Dantas RO, et al. Evaluation and treatment of Chagas disease in the United States: a systematic review. JAMA. 2007 Nov;298(18):2171-81.) (298298. Gallerano RH, Sosa RR. Resultados de un estúdio a largo plazo con drogas antiparasitárias en infectados chagásicos crónicos. Rev Fed Arg Cardiol. 2001;30:289-96. . This would lead to a reduction in the complications of the disease, especially those related to heart disease. Classical experimental studies in mice have already documented total or partial regression of early myocardial injuries and the prevention of cardiopathy with antiparasitic treatment 299299. Fragata Filho AA. Tratamento antiparasitário da doença de Chagas. Rev Soc Cardiol Estado de São Paulo. 2009;19:2-5.) (300300. Andrade SG, Magalhães JB, Pontes AL. Terapêutica da fase crônica da infecção experimental pelo Trypanosoma cruzi com o benzonidazol e o nifurtimox. Rev Soc Bras Med Trop. 1989 Jul-Sep;22(3):113-8. . However, regression of inflammatory and fibrotic lesions, observed in experimental studies, has not been verified in the clinical context 2020. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Consenso Brasileiro em Doença de Chagas. Rev Soc Bras Med Trop. 2005;38 Supl 3:1-29. .
A case series demonstrated cure in at least 20% of the cases treated in the late chronic phase 301301. Garcia S, Ramos CO, Senra JFV, Vilas-Boas F, Rodrigues MM, Campos-de-Carvalho AC, et al. Treatment with benznidazole during the chronic phase of experimental Chagas' disease decreases cardiac alterations. Antimicrob Agents Chemother. 2005 Apr;49(4):1521-8. . It should be noted that demonstrating cure depends on various factors, including follow-up of treated cases, which should continue for more than two decades for patients treated in the late phase. Increased seroconversion of cases showing no acute illness and without cardiac failure was also assessed 290290. Molina I, Gómez i Prat J, Salvador F, Treviño B, Sulleiro E, Serre N, et al. Randomized trial of posaconazole and benznidazole for chronic Chagas' disease. N Engl J Med. 2014 May;370:1899-908. .
For individuals older than 50 years, without advanced heart disease, there are no studies justifying antiparasitic treatment. An observational study in elderly subjects infected with T. cruzi demonstrated that the disease is present and active - the percentage of patients with ICF decreases but the percentage with the heart form increases 195195. Villela E. Moléstia de Chagas: descrição clínica, 5ª parte. Folha Médica. 1923; 4:65-6. . Thus, these individuals should not be completely excluded from the etiological treatment plan of Chagas disease; treatment can be individualized (Class IIb, level of evidence C).
Antiparasitic treatment should not be given to patients with chronic phase Chagas disease with severe heart dysfunction, since there is no evidence of clinical benefit 251251. Morillo CA, Marin-Neto JA, Avezum A, Sosa-Estani S, Rassi Júnior A, Rosas F, et al; BENEFIT Investigators. Randomized Trial of Benznidazole for Chronic Chagas' Cardiomyopathy. N Engl J Med. 2015 Oct;373(14):1295-306.) (289289. Coura JR, Borges-Pereira J. Chronic phase of Chagas disease: why should it be treated? A comprehensive review. Mem Inst Oswaldo Cruz. 2011 Sep;106(6):641-5.) (291291. Cançado JR. Terapêutica específica. In: Dias JCP, Coura JR, editores. Clínica e Terapêutica da Doença de Chagas: uma abordagem prática para o clínico geral. Rio de Janeiro: Fiocruz; 1997. p. 323-51.) (295295. Bern C, Montgomery SP, Herwaldt BL, Rassi Júnior A, Marin-Neto JA, Dantas RO, et al. Evaluation and treatment of Chagas disease in the United States: a systematic review. JAMA. 2007 Nov;298(18):2171-81.) (296296. Coura JR, Abreu LL, Willcox HP, Petana W. Comparative controlled study on the use of benznidazole, nifurtimox and placebo, in the chronic form of Chagas' disease, in a field area with interrupted transmission. I. Preliminary evaluation. Rev Soc Bras Med Trop. 1997 Mar-Apr;30(2):139-44.) (302302. Coura JR, Castro SL. A critical review on Chagas disease chemotherapy. Mem Inst Oswaldo Cruz. 2002 Jan;97(1):3-24.) (303303. Maguire JH. Treatment of Chagas' disease: time is running out. N Engl J Med. 2015 Oct;373(14):1369-70. (Class III, level of evidence C).
Recently, a series results from the BENEFIT study - a prospective, multicenter, randomized, double-blind, controlled study with placebo (placebo vs. benznidazole) - was published 251251. Morillo CA, Marin-Neto JA, Avezum A, Sosa-Estani S, Rassi Júnior A, Rosas F, et al; BENEFIT Investigators. Randomized Trial of Benznidazole for Chronic Chagas' Cardiomyopathy. N Engl J Med. 2015 Oct;373(14):1295-306. . This is the first study to evaluate the effect of a specific treatment for up to 80 days in relation to cardiac outcomes and T. cruzi infection. The effects of benznidazole in reducing morbidity and mortality among 2,854 patients with established chronic Chagas cardiopathy were evaluated 250250. Marin-Neto JA, Rassi A Júnior, Morillo CA, Avezum A, Connolly SJ, Sosa-Estani S, et al; BENEFIT Investigators. BENEFIT Investigators. Rationale and design of a randomized placebo-controlled trial assessing the effects of etiologic treatment in Chagas' cardiomyopathy: the BENznidazole Evaluation For Interrupting Trypanosomiasis (BENEFIT). Am Heart J. 2008 Jul;156(1):37-43.) (251251. Morillo CA, Marin-Neto JA, Avezum A, Sosa-Estani S, Rassi Júnior A, Rosas F, et al; BENEFIT Investigators. Randomized Trial of Benznidazole for Chronic Chagas' Cardiomyopathy. N Engl J Med. 2015 Oct;373(14):1295-306. . After 5.4 years of follow-up, the results showed no statistically significant difference between the two groups in relation to the primary composite outcome that included death, sustained ventricular tachycardia, recent heart failure, implantation of a cardioverter-defibrillator or pacemaker, cardiac transplantation, resuscitated cardiac arrest, stroke, or other thromboembolic event 251251. Morillo CA, Marin-Neto JA, Avezum A, Sosa-Estani S, Rassi Júnior A, Rosas F, et al; BENEFIT Investigators. Randomized Trial of Benznidazole for Chronic Chagas' Cardiomyopathy. N Engl J Med. 2015 Oct;373(14):1295-306. . These findings, at first, seem to indicate a divergence of considerably lower rates of parasite detection by PCR in blood in the same study after treatment with benznidazole 303303. Maguire JH. Treatment of Chagas' disease: time is running out. N Engl J Med. 2015 Oct;373(14):1369-70. .
In the BENEFIT study, the treatment with benznidazole in the protocol used probably has no significant preventive effect on the progression of heart disease in patients with advanced Chagas disease 251251. Morillo CA, Marin-Neto JA, Avezum A, Sosa-Estani S, Rassi Júnior A, Rosas F, et al; BENEFIT Investigators. Randomized Trial of Benznidazole for Chronic Chagas' Cardiomyopathy. N Engl J Med. 2015 Oct;373(14):1295-306. (Class III, level of evidence B). However, the severity of Chagas disease is emphasized in this study, as is the resulting need for diagnosis and timely treatment, based on qualified management, especially of heart disease. In the study, 503 patients died within 5 years (246 the in benznidazole group and 257 in the placebo group), and 63% of patients (in both treatment groups) developed a left ventricular ejection fraction <40%, one of the primary outcomes of the study 251251. Morillo CA, Marin-Neto JA, Avezum A, Sosa-Estani S, Rassi Júnior A, Rosas F, et al; BENEFIT Investigators. Randomized Trial of Benznidazole for Chronic Chagas' Cardiomyopathy. N Engl J Med. 2015 Oct;373(14):1295-306. .
The study highlighted, therefore, the urgent need to structure a network of qualified health care to manage patients with Chagas disease comprehensively, without loss of diagnostic and treatment opportunities. Longitudinal clinical management is needed in terms of the various clinical forms of the disease, especially the advanced cardiopathy form, with protocols adjusted to local realities. In addition, the ability to create specific treatment opportunities for people with therapeutic indications based on more consistent evidence, is required 303303. Maguire JH. Treatment of Chagas' disease: time is running out. N Engl J Med. 2015 Oct;373(14):1369-70. .
Furthermore, the study draws attention to the lower frequency of serious adverse effects to benznidazole compared with previous study findings 251251. Morillo CA, Marin-Neto JA, Avezum A, Sosa-Estani S, Rassi Júnior A, Rosas F, et al; BENEFIT Investigators. Randomized Trial of Benznidazole for Chronic Chagas' Cardiomyopathy. N Engl J Med. 2015 Oct;373(14):1295-306.) (303303. Maguire JH. Treatment of Chagas' disease: time is running out. N Engl J Med. 2015 Oct;373(14):1369-70. . This reinforces the real possibility of including, in a more expanded form, the specific treatment of Chagas disease in the primary healthcare network (primary) not only in Brazil but also in other endemic countries.
The digestive form of Chagas disease is not a contraindication for antiparasitic treatment. Moreover, megaesophagus correction should be performed in order to ensure full transit of the drug and, subsequently, its absorption 2020. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Consenso Brasileiro em Doença de Chagas. Rev Soc Bras Med Trop. 2005;38 Supl 3:1-29.) (302302. Coura JR, Castro SL. A critical review on Chagas disease chemotherapy. Mem Inst Oswaldo Cruz. 2002 Jan;97(1):3-24.) (Class IIa, level of evidence C).
Treatment of immunosuppressed cases
Cases with immunodeficiency, including patients taking immunosuppressive drugs to treat neoplasms, transplants, and other diseases, and patients with T. cruzi /HIV co-infection, can develop reactivation of Chagas disease 181181. Riarte A, Luna C, Sabatiello R, Sinagra A, Schiavelli R, De Rissio A, et al. Chagas' disease in patients with kidney transplants: 7 years of experience 1989-1996. Clin Infect Dis. 1999 Sep;29(3):561-7.) (184184. Altclas J, Sinagra A, Dictar M, Luna C, Verón MT, De Rissio AM, et al. Chagas disease in bone marrow transplantation: an approach to preemptive therapy. Bone Marrow Transplant. 2005 Jul;36(2):123-9.) (304304. D'Albuquerque LA, Gonzalez AM, Filho HL, Copstein JL, Larrea FI, Mansero JM, et al. Liver transplantation from deceased donors serologically positive for Chagas disease. Am J Transplant. 2007 Mar;7(3):680-4. . This is discussed, in themes, below.
Transplantation
In transplants in general, it is necessary to know if the donor or the recipient has Chagas disease, considering the risk of transmission or reactivation of the disease. Thus, there are three possible scenarios:
-
donors with Chagas disease and recipient without Chagas disease;
-
donors without Chagas disease and recipient with Chagas disease;
-
donors and recipient with Chagas disease.
Donor with Chagas disease and recipient without Chagas disease
1. Guidelines for the donor with Chagas disease:
-
The candidate for organ donation who has positive serology for T. cruzi should be treated with antiparasitic drugs for 60 days before the procedure. In special situations requiring transplantation before completing treatment, the transplant can be performed, but if possible, not before 14 days of treatment has been completed (Class I, level of evidence C).
2. Guidelines for the recipient when the donor is treated:
-
Consider etiological treatment soon after transplantation, maintaining treatment for 60 days by means of intense immunosuppression (Class IIa, level of evidence C) 184184. Altclas J, Sinagra A, Dictar M, Luna C, Verón MT, De Rissio AM, et al. Chagas disease in bone marrow transplantation: an approach to preemptive therapy. Bone Marrow Transplant. 2005 Jul;36(2):123-9. .
-
Do not treat, but provide sequential clinical monitoring or parasitologic evaluation as follows: weekly direct search for T. cruzi in the peripheral blood, until 60 days post-transplantation; indirect parasitologic methods (blood and/or xenodiagnosis) of follow-up of the recipient during the period of immunosuppression; and serologic examination 30 and 60 days post-transplant 181181. Riarte A, Luna C, Sabatiello R, Sinagra A, Schiavelli R, De Rissio A, et al. Chagas' disease in patients with kidney transplants: 7 years of experience 1989-1996. Clin Infect Dis. 1999 Sep;29(3):561-7.) (184184. Altclas J, Sinagra A, Dictar M, Luna C, Verón MT, De Rissio AM, et al. Chagas disease in bone marrow transplantation: an approach to preemptive therapy. Bone Marrow Transplant. 2005 Jul;36(2):123-9.) (305305. Altclas JD, Barcan L, Nagel C, Lattes R, Riarte A. Organ transplantation and Chagas disease. JAMA. 2008 Mar;299(10):1134. . In cases where monitoring is not possible, etiologic treatment is recommended. The following clinical and serologic examinations should be performed at 3, 6, 9, and 12 months of follow-up: direct/indirect parasitologic tests (blood culture and/or artificial xenodiagnosis at 3, 6, and 12 months) or PCR. Subsequently, the evaluation is repeated every 6 months, while there is persistent immunosuppression (Class IIa, level of evidence B) 181181. Riarte A, Luna C, Sabatiello R, Sinagra A, Schiavelli R, De Rissio A, et al. Chagas' disease in patients with kidney transplants: 7 years of experience 1989-1996. Clin Infect Dis. 1999 Sep;29(3):561-7.) (184184. Altclas J, Sinagra A, Dictar M, Luna C, Verón MT, De Rissio AM, et al. Chagas disease in bone marrow transplantation: an approach to preemptive therapy. Bone Marrow Transplant. 2005 Jul;36(2):123-9.) (305305. Altclas JD, Barcan L, Nagel C, Lattes R, Riarte A. Organ transplantation and Chagas disease. JAMA. 2008 Mar;299(10):1134. . It is noteworthy that, in some reference centers, PCR, but not indirect parasitologic examination, is performed. At any time, if an acute infection is detected through parasitologic or serologic tests, the conventional etiological treatment should be instituted. Out of the usual controls, any clinical signs suspicious of infection should be investigated through parasitologic examination, and positive cases should receive conventional treatment for the acute phase. Since 2009 in Brazil, organ transplantation laws have restricted the possibility of patients with Chagas disease being accepted as organ donors 183183. Ministério da Saúde (BR). Portaria nº 2.600, de 21 de outubro de 2009. Aprova o Regulamento Técnico do Sistema Nacional de Transplantes. Diário Oficial da República Federativa do Brasil, Brasília (DF), 2009 out 21;Seção 1:77. . In Brazil, Ordinance number 2,600, in 2009, which approved the Technical Regulations of the National Transplant System, states that it is mandatory to perform serologic testing for T. cruzi infection in the following situations 183183. Ministério da Saúde (BR). Portaria nº 2.600, de 21 de outubro de 2009. Aprova o Regulamento Técnico do Sistema Nacional de Transplantes. Diário Oficial da República Federativa do Brasil, Brasília (DF), 2009 out 21;Seção 1:77.: I) for all donations, following the same algorithms used for screening blood donors; II) for registration purposes of potential organ recipients in a Single Technical Registry; III) for all potential deceased donor organs, tissues, cells, or body parts before the allocation of the grafts. It also establishes criteria for the classification of living and deceased donors as well as for potential recipients for the distribution of organs, tissues, cells, and captured parts. It does indicate that the team will decide whether the organ/tissue is acceptable for use or not regarding kidney, kidney/pancreas, pancreas, liver, and lung donations. In the order, the rejection of organs relates only to the heart. For ocular tissues, hematopoietic stem cells, musculoskeletal tissues, skin, and cardiovascular tissue, the issues related to T. cruzi infection are not mentioned 183183. Ministério da Saúde (BR). Portaria nº 2.600, de 21 de outubro de 2009. Aprova o Regulamento Técnico do Sistema Nacional de Transplantes. Diário Oficial da República Federativa do Brasil, Brasília (DF), 2009 out 21;Seção 1:77. .
3. Guidance for the recipient without Chagas disease:
In cases in which the donor with Chagas disease has not been treated or has received incomplete treatment, three possible actions may be taken in relation to the recipient:
-
Start antiparasitic treatment soon after transplantation, for a 60-day period (class IIa, level of evidence C). This is recommended if the sequence monitoring indicated in item c below is not possible.
-
Start treatment immediately after surgery, for an initial 14-day period. Then, proceed to sequential monitoring with clinical, parasitologic, and serologic evaluation (Class IIa, level of evidence C or without evidence). If seroconversion occurs, introduce antiparasitic treatment for the acute phase.
-
Do not treat, but proceed to sequential clinical monitoring and parasitologic evaluation (Class IIa level of evidence B) 181181. Riarte A, Luna C, Sabatiello R, Sinagra A, Schiavelli R, De Rissio A, et al. Chagas' disease in patients with kidney transplants: 7 years of experience 1989-1996. Clin Infect Dis. 1999 Sep;29(3):561-7.) (184184. Altclas J, Sinagra A, Dictar M, Luna C, Verón MT, De Rissio AM, et al. Chagas disease in bone marrow transplantation: an approach to preemptive therapy. Bone Marrow Transplant. 2005 Jul;36(2):123-9.) (305305. Altclas JD, Barcan L, Nagel C, Lattes R, Riarte A. Organ transplantation and Chagas disease. JAMA. 2008 Mar;299(10):1134. . Such evaluation consists of direct search for T. cruzi in the peripheral blood, every week, for up to 60 days, and indirect parasitologic and serologic examinations on days 30 and 60 after transplantation. Thereafter, clinical, serologic, and parasitologic (direct/indirect/PCR) examinations should be performed every two months for up to one year of follow-up; then every six months, for as long as immunosuppression persists (time dependent on the mode and type of transplant). It is worth emphasizing that in some centers PCR, rather than indirect parasitologic examination, is performed.
-
At any time, if an acute infection is detected by parasitologic or serologic tests (IgM or seroconversion), conventional antiparasitic treatment should be started. Outside of the above-stated monitoring intervals, any clinical sign of suspected infectious condition should be investigated through parasitologic examination, and positive cases should receive conventional treatment for acute stage disease 2020. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Consenso Brasileiro em Doença de Chagas. Rev Soc Bras Med Trop. 2005;38 Supl 3:1-29.) (302302. Coura JR, Castro SL. A critical review on Chagas disease chemotherapy. Mem Inst Oswaldo Cruz. 2002 Jan;97(1):3-24. .
-
A few of cases have been reported of kidney transplant recipients without Chagas disease who were treated with antiparasitic prophylaxis for 14 days, as the organ donors had Chagas disease 308308. Freitas VL, Silva SC, Sartori AM, Bezerra RC, Westphalen EV, Molina TD, et al. Real-time PCR in HIV/ Trypanosoma cruzi coinfection with and without Chagas disease reactivation: association with HIV viral load and CD4 level. PLoS Negl Trop Dis. 2011 Aug;5(8):e1277. . In another work, prophylaxis was provided for 60 days to recipients (without Chagas disease) who received livers from donors with Chagas disease 306306. Sousa AA, Lobo MC, Barbosa RA, Bello V. Chagas seropositive donors in kidney transplantation. Transplant Proc. 2004 May;36(4):868-9. as usual strategy; seroconversion was not observed in the surviving recipients, as this may not occur in about 20% of patients in the post-transplant period . Use of antiparasitic drugs in patients without documented infection (no identification of the parasite or evidence of seroconversion) is considered prophylactic use. In this text, even when the drug was administered for the same duration as the duration of treatment (60 days), the term prophylaxis has been used if the drug was used in the absence of a documented infection in the recipient. The works cited are not controlled and, in addition of having been carried out in a small number of cases in kidney and liver transplant recipients, these did not use the systematic monitoring of parasitemia, and the final results were based on the absence of seroconversion in patients who survived.
-
However, since 2009, the possibility of patients with Chagas disease being accepted as organ donors in Brazil has been restricted by transplant regulations 184184. Altclas J, Sinagra A, Dictar M, Luna C, Verón MT, De Rissio AM, et al. Chagas disease in bone marrow transplantation: an approach to preemptive therapy. Bone Marrow Transplant. 2005 Jul;36(2):123-9. , with systematic serologic screening limiting this type of donation in individuals with T. cruzi infection. However, these same organs can function as "marginal organs" in specific situations 184184. Altclas J, Sinagra A, Dictar M, Luna C, Verón MT, De Rissio AM, et al. Chagas disease in bone marrow transplantation: an approach to preemptive therapy. Bone Marrow Transplant. 2005 Jul;36(2):123-9.) (305305. Altclas JD, Barcan L, Nagel C, Lattes R, Riarte A. Organ transplantation and Chagas disease. JAMA. 2008 Mar;299(10):1134.) (307307. Bocchi EA, Belloti G, Mocelin AO, Uip D, Bacal F, Higuchi ML, et al. Heart transplantation for chronic Chagas' disease. Ann Thorac Surg. 1996 Jun;61(6):1727-33. .
Donor without Chagas disease and recipient with Chagas disease
In these cases, the expected complication is the reactivation of Chagas disease in the recipient, due to the use of immunosuppressive drugs to prevent or treat rejections. With time, pre-transplant conventional antiparasitic treatment can be instituted. However, it should not prevent the realization of transplantation, as subsequent actions can be adopted (Class IIa, level of evidence C).
-
Prophylaxis of episodes of reactivation: In heart transplant recipients, antiparasitic treatment may fail 100100. Pinazo MJ, Miranda B, Rodríguez-Villar C, Altclas J, Brunet Serra M, García-Otero EC, et al. Recommendations for management of Chagas disease in organ and hematopoietic tissue transplantation programs in nonendemic areas. Transplant Rev (Orlando). 2011 Jul;25(3):91-101.) (307307. Bocchi EA, Belloti G, Mocelin AO, Uip D, Bacal F, Higuchi ML, et al. Heart transplantation for chronic Chagas' disease. Ann Thorac Surg. 1996 Jun;61(6):1727-33. .
-
Clinical and parasitologic monitoring of the recipient after transplantation should be carried out, as in the case with a donor with Chagas disease and a recipient without Chagas disease, and early conventional treatment of reactivation episodes should be provided 181181. Riarte A, Luna C, Sabatiello R, Sinagra A, Schiavelli R, De Rissio A, et al. Chagas' disease in patients with kidney transplants: 7 years of experience 1989-1996. Clin Infect Dis. 1999 Sep;29(3):561-7.) (184184. Altclas J, Sinagra A, Dictar M, Luna C, Verón MT, De Rissio AM, et al. Chagas disease in bone marrow transplantation: an approach to preemptive therapy. Bone Marrow Transplant. 2005 Jul;36(2):123-9.) (305305. Altclas JD, Barcan L, Nagel C, Lattes R, Riarte A. Organ transplantation and Chagas disease. JAMA. 2008 Mar;299(10):1134. . The monitoring during the post-transplant period in patients receiving cardiac transplantation is mandatory and must include monitoring of blood and tissues, due to the greater sensitivity of the methods in the reactivation site (myocardium).
Donor and recipient with Chagas disease
Both should be considered as carriers of the chronic form of Chagas disease and should be evaluated for antiparasitic treatment, as described for cases with the chronic form of the disease. After transplantation, the recipient should be monitored from a clinical and parasitologic point of view for the diagnosis of potential episodes of reactivation, such as in the case of a donor with Chagas disease and a recipient without Chagas disease. If reactivation occurs, conventional treatment for the acute phase should be provided (class IIa, level of evidence C). Episodes of reactivation can occur more than once and should be treated when documented. Hence, systematic parasitologic monitoring while immunosuppression persists, is recommended.
Coinfection Trypanosoma cruzi /HIV
Observational studies have shown that, in the presence of T. cruzi /HIV coinfection without AIDS, the natural evolution of both diseases occurs, and one should follow the guidelines for antiparasitic treatment of Chagas disease in its acute and chronic forms, or monitoring of parasitemia, quantitatively if possible 4949. Almeida EA, Ramos Júnior AN, Correia D, Shikanai-Yasuda MA. Co-infection Trypanosoma cruzi /HIV: systematic review (1980-2010). Rev Soc Bras Med Trop. 2011 Nov-Dec;44(6):762-70.) (159159. Rocha A, Ramos Júnior AN, Sartori AM, Correia D, Gontijo ED, Tatto E, et al. Recomendações para diagnóstico, tratamento e acompanhamento da co-infecção Trypanosoma cruzi: vírus da imunodeficiência humana. Rev Soc Bras Med Trop. 2006 jul-ago;39(4):392-415.) (175175. Sartori AM, Ibrahim KY, Nunes Westphalen EV, Braz LM, Oliveira Junior OC, Gakiya E, et al. Manifestations of Chagas disease (American trypanosomiasis ) in patients with HIV/AIDS. Ann Trop Med Parasitol. 2007 Jan;101(1):31-50. (Class IIa, level of evidence C) (see specific part of this Consensus). In patients with co-infection and AIDS, reactivation of Chagas disease might occur, and the following measures are recommended: Specific antiparasitic treatment is indicated for reactivation with a clinical syndrome of myocarditis, meningoencephalitis, or other; and parasitologic examination of the blood or cerebrospinal fluid yields positive results by direct microscopy; or characteristic lesions are demonstrated on histologic examination of tissue specimens 2020. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Consenso Brasileiro em Doença de Chagas. Rev Soc Bras Med Trop. 2005;38 Supl 3:1-29.) (4949. Almeida EA, Ramos Júnior AN, Correia D, Shikanai-Yasuda MA. Co-infection Trypanosoma cruzi /HIV: systematic review (1980-2010). Rev Soc Bras Med Trop. 2011 Nov-Dec;44(6):762-70.) (159159. Rocha A, Ramos Júnior AN, Sartori AM, Correia D, Gontijo ED, Tatto E, et al. Recomendações para diagnóstico, tratamento e acompanhamento da co-infecção Trypanosoma cruzi: vírus da imunodeficiência humana. Rev Soc Bras Med Trop. 2006 jul-ago;39(4):392-415. (Class I, level of evidence C). This therapeutic approach should also be adopted in cases without evidence of reactivation but with the associated clinical syndrome and a high level of parasitemia, as defined by Sartori et al. as the presence of more than 20% of positive nymphs in the same xenodiagnostic test (indirect parasitologic examination, quantitative PCR) or sustained parasitemia 4949. Almeida EA, Ramos Júnior AN, Correia D, Shikanai-Yasuda MA. Co-infection Trypanosoma cruzi /HIV: systematic review (1980-2010). Rev Soc Bras Med Trop. 2011 Nov-Dec;44(6):762-70.) (159159. Rocha A, Ramos Júnior AN, Sartori AM, Correia D, Gontijo ED, Tatto E, et al. Recomendações para diagnóstico, tratamento e acompanhamento da co-infecção Trypanosoma cruzi: vírus da imunodeficiência humana. Rev Soc Bras Med Trop. 2006 jul-ago;39(4):392-415.) (175175. Sartori AM, Ibrahim KY, Nunes Westphalen EV, Braz LM, Oliveira Junior OC, Gakiya E, et al. Manifestations of Chagas disease (American trypanosomiasis ) in patients with HIV/AIDS. Ann Trop Med Parasitol. 2007 Jan;101(1):31-50.) (308308. Freitas VL, Silva SC, Sartori AM, Bezerra RC, Westphalen EV, Molina TD, et al. Real-time PCR in HIV/ Trypanosoma cruzi coinfection with and without Chagas disease reactivation: association with HIV viral load and CD4 level. PLoS Negl Trop Dis. 2011 Aug;5(8):e1277. (Class IIa, level of evidence C).
Coinfection and prophylaxis
By analogy with other opportunistic infections in AIDS, secondary prophylaxis can be used for patients with reactivation of Chagas disease, who were treated for and achieved clinical and parasitologic remission, when the levels of CD4 + T lymphocyte were less than 200 cells/mm 3 (IIa class, level of evidence C 2020. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Consenso Brasileiro em Doença de Chagas. Rev Soc Bras Med Trop. 2005;38 Supl 3:1-29.) (4949. Almeida EA, Ramos Júnior AN, Correia D, Shikanai-Yasuda MA. Co-infection Trypanosoma cruzi /HIV: systematic review (1980-2010). Rev Soc Bras Med Trop. 2011 Nov-Dec;44(6):762-70.) (159159. Rocha A, Ramos Júnior AN, Sartori AM, Correia D, Gontijo ED, Tatto E, et al. Recomendações para diagnóstico, tratamento e acompanhamento da co-infecção Trypanosoma cruzi: vírus da imunodeficiência humana. Rev Soc Bras Med Trop. 2006 jul-ago;39(4):392-415.) (175175. Sartori AM, Ibrahim KY, Nunes Westphalen EV, Braz LM, Oliveira Junior OC, Gakiya E, et al. Manifestations of Chagas disease (American trypanosomiasis ) in patients with HIV/AIDS. Ann Trop Med Parasitol. 2007 Jan;101(1):31-50. . However, this recommendation needs to be validated in prospective studies, considering the low frequency of relapse observed in patients taking combined antiretroviral treatment.
Immunosuppression in other diseases associated with Chagas disease
Clinical conditions such as neoplasms and collagen vascular disorders in carriers of Chagas disease might be associated with reactivation. The recommendations are the same as those for reactivation occurring in organ transplantation patients and patients with HIV coinfection 2020. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Consenso Brasileiro em Doença de Chagas. Rev Soc Bras Med Trop. 2005;38 Supl 3:1-29.) (4949. Almeida EA, Ramos Júnior AN, Correia D, Shikanai-Yasuda MA. Co-infection Trypanosoma cruzi /HIV: systematic review (1980-2010). Rev Soc Bras Med Trop. 2011 Nov-Dec;44(6):762-70.) (159159. Rocha A, Ramos Júnior AN, Sartori AM, Correia D, Gontijo ED, Tatto E, et al. Recomendações para diagnóstico, tratamento e acompanhamento da co-infecção Trypanosoma cruzi: vírus da imunodeficiência humana. Rev Soc Bras Med Trop. 2006 jul-ago;39(4):392-415.) (175175. Sartori AM, Ibrahim KY, Nunes Westphalen EV, Braz LM, Oliveira Junior OC, Gakiya E, et al. Manifestations of Chagas disease (American trypanosomiasis ) in patients with HIV/AIDS. Ann Trop Med Parasitol. 2007 Jan;101(1):31-50. .
Accidental transmission
In accidents characterized as high risk for disease transmission, such as percutaneous injury or mucosal contact with biological material containing live parasites (samples for the culture of T. cruzi obtained from of patients with high parasitemia or necropsy material, vectors, and animals that are already infected), primary prophylaxis should be given. Prophylaxis should be initiated immediately after the accident and should be continued for 10 days 309309. Brener Z. Laboratory-acquired Chagas' disease: an endemic disease among parasitologists? In: Morel M, editor. Genes and antigens of parasites: a laboratory manual. 2 ed. Rio de Janeiro: Editora Fiocruz; 1984. p. 3-9.) (310310. Brener Z. Laboratory-acquired Chagas disease: comment. Trans R Soc Trop Med Hyg. 1987;81(3):527.) (311311. Shikanai-Yasuda MA, Umezawa ES, Tolezano JE, Matsubara L. Doença de Chagas aguda após uso profilático de benznidazol (Acidente de laboratório). Rev Soc Bras Med Tropical. 1993;26 supl II:127-8. . (Class IIa, level of evidence C). Serologic tests should be performed before initiating treatment and after 20, 40, and 60 days post-treatment to monitor possible seroconversion. In the case of positive serologic results, conventional antiparasitic treatment should be provided, as described previously for the acute phase.
In minimal risk situations, such as the contact with the blood of a patient with Chagas disease in chronic phase, drug prophylaxis is not indicated. Serologic tests should be performed immediately after the accident, and 20, 40, and 60 days post-infection. If seroconversion occurs, conventional treatment for acute phase of Chagas disease should be given, and post-therapeutic monitoring should be conducted as in the acute phase. If the serology remains positive after treatment, the therapeutic gap should be documented and repeat treatment provided, either with the same drug or with an alternative drug 2020. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Consenso Brasileiro em Doença de Chagas. Rev Soc Bras Med Trop. 2005;38 Supl 3:1-29. .
Treatment options, dosage, and type of administration
The neglected character of Chagas disease is evidenced by the limited therapeutic options; only two antiparasitic drugs with established efficacy are available for specific treatment: benznidazole (a nitroimidazole derivative agent) and nifurtimox (a nitrofuran compound) 1414. World Health Organization. Chagas disease (American trypanosomiasis) [Internet]. Geneva: World Health Organization ; 2015 [citado 2015 Nov 13]. Disponível em: Disponível em: http://www.who.int/mediacentre/factsheets/fs340/en/
http://www.who.int/mediacentre/factsheet...
) (1919. Prata A. Clinical and epidemiological aspects of Chagas disease. Lancet Infect Dis. 2001 Sep;1(2):92-100.) (2020. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Consenso Brasileiro em Doença de Chagas. Rev Soc Bras Med Trop. 2005;38 Supl 3:1-29.) (3838. Dias JC, Coura JR, Yasuda MAS. The present situation, challenges, and perspectives regarding the production and utilization of effective drugs against human Chagas disease. Rev Soc Bras Med Trop. 2014 Jan-Feb;47(1):123-5.) (4242. Rassi Júnior A, Rassi A, Marin-Neto JA. Chagas disease. Lancet. 2010 Apr;375(9723):1388-402.) (293293. Andrade AL, Zicker F, Oliveira RM, Silva SA, Luquetti A, Travassos LR, et al. Randomised trial of efficacy of benznidazole in treatment of early Trypanosoma cruzi infection. Lancet. 1996 Nov;348(9039):1407-13.) (294294. Yun O, Lima MA, Ellman T, Chambi W, Castillo S, Flevaud L, et al. Feasibility, drug safety, and effectiveness of etiological treatment programs for Chagas disease in Honduras, Guatemala, and Bolivia: 10-year experience of Médecins Sans Frontières. PLoS Negl Trop Dis. 2009 Jul;3(7):e488.) (296296. Coura JR, Abreu LL, Willcox HP, Petana W. Comparative controlled study on the use of benznidazole, nifurtimox and placebo, in the chronic form of Chagas' disease, in a field area with interrupted transmission. I. Preliminary evaluation. Rev Soc Bras Med Trop. 1997 Mar-Apr;30(2):139-44. .
The drug of first choice for the antiparasitic treatment of Chagas disease in all the situations previously discussed is benznidazole 2020. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Consenso Brasileiro em Doença de Chagas. Rev Soc Bras Med Trop. 2005;38 Supl 3:1-29.) (3838. Dias JC, Coura JR, Yasuda MAS. The present situation, challenges, and perspectives regarding the production and utilization of effective drugs against human Chagas disease. Rev Soc Bras Med Trop. 2014 Jan-Feb;47(1):123-5.) (100100. Pinazo MJ, Miranda B, Rodríguez-Villar C, Altclas J, Brunet Serra M, García-Otero EC, et al. Recommendations for management of Chagas disease in organ and hematopoietic tissue transplantation programs in nonendemic areas. Transplant Rev (Orlando). 2011 Jul;25(3):91-101.) (294294. Yun O, Lima MA, Ellman T, Chambi W, Castillo S, Flevaud L, et al. Feasibility, drug safety, and effectiveness of etiological treatment programs for Chagas disease in Honduras, Guatemala, and Bolivia: 10-year experience of Médecins Sans Frontières. PLoS Negl Trop Dis. 2009 Jul;3(7):e488. . Currently, this medicine is produced by a Brazilian pharmaceutical company, distributed by request to the Ministry of Health of Brazil, with no distribution to the general pharmaceutical market 2020. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Consenso Brasileiro em Doença de Chagas. Rev Soc Bras Med Trop. 2005;38 Supl 3:1-29.) (3939. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Guia de Vigilância em Saúde. Brasília: Ministério da Saúde; 2014. .
Benznidazole is a well-tolerated drug 2020. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Consenso Brasileiro em Doença de Chagas. Rev Soc Bras Med Trop. 2005;38 Supl 3:1-29.) (3838. Dias JC, Coura JR, Yasuda MAS. The present situation, challenges, and perspectives regarding the production and utilization of effective drugs against human Chagas disease. Rev Soc Bras Med Trop. 2014 Jan-Feb;47(1):123-5.) (251251. Morillo CA, Marin-Neto JA, Avezum A, Sosa-Estani S, Rassi Júnior A, Rosas F, et al; BENEFIT Investigators. Randomized Trial of Benznidazole for Chronic Chagas' Cardiomyopathy. N Engl J Med. 2015 Oct;373(14):1295-306. . Children generally have fewer adverse effects than adults and can tolerate higher doses 104104. Carlier Y, Torrico F, Sosa-Estani S, Russomando G, Luquetti A, Freilij H, et al. Congenital Chagas disease: recommendations for diagnosis, treatment and control of newborns, siblings and pregnant women. PLoS Negl Trop Dis. 2011 Oct;5(10):e1250. . In April 2013, the 19 th Expert Committee on the Selection and Use of Essential Medicines of the WHO discussed and approved the addition of two new formulations of benznidazole in the essential drugs list for children 3838. Dias JC, Coura JR, Yasuda MAS. The present situation, challenges, and perspectives regarding the production and utilization of effective drugs against human Chagas disease. Rev Soc Bras Med Trop. 2014 Jan-Feb;47(1):123-5. . Benznidazole is being increasingly prescribed for the antiparasitic treatment of Chagas disease, not only the expansion of indications based on evidence, but also as a result of increased demand in endemic and non-endemic countries with better structure of their national surveillance and healthcare systems 3838. Dias JC, Coura JR, Yasuda MAS. The present situation, challenges, and perspectives regarding the production and utilization of effective drugs against human Chagas disease. Rev Soc Bras Med Trop. 2014 Jan-Feb;47(1):123-5.) (294294. Yun O, Lima MA, Ellman T, Chambi W, Castillo S, Flevaud L, et al. Feasibility, drug safety, and effectiveness of etiological treatment programs for Chagas disease in Honduras, Guatemala, and Bolivia: 10-year experience of Médecins Sans Frontières. PLoS Negl Trop Dis. 2009 Jul;3(7):e488.) (295295. Bern C, Montgomery SP, Herwaldt BL, Rassi Júnior A, Marin-Neto JA, Dantas RO, et al. Evaluation and treatment of Chagas disease in the United States: a systematic review. JAMA. 2007 Nov;298(18):2171-81.) .
Benznidazole (Class 1, level of evidence b) is produced as tablets of 100mg and 50mg (adults) and 12.5mg and 50mg (children) 2020. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Consenso Brasileiro em Doença de Chagas. Rev Soc Bras Med Trop. 2005;38 Supl 3:1-29.) (4242. Rassi Júnior A, Rassi A, Marin-Neto JA. Chagas disease. Lancet. 2010 Apr;375(9723):1388-402.) (104104. Carlier Y, Torrico F, Sosa-Estani S, Russomando G, Luquetti A, Freilij H, et al. Congenital Chagas disease: recommendations for diagnosis, treatment and control of newborns, siblings and pregnant women. PLoS Negl Trop Dis. 2011 Oct;5(10):e1250.) (109109. Sosa-Estani S, Cura E, Velazquez E, Yampotis C, Segura EL. Etiological treatment of young women infected with Trypanosoma cruzi , and prevention of congenital transmission. Rev Soc Bras Med Trop. 2009 Sep-Oct;42(5):484-7.) (122122. Pérez-Molina JA, Pérez-Ayala A, Moreno S, Fernández-González MC, Zamora J, López-Velez R. Use of benznidazole to treat chronic Chagas' disease: a systematic review with a meta-analysis. J Antimicrob Chemother. 2009 Dec;64(6):1139-47.) (291291. Cançado JR. Terapêutica específica. In: Dias JCP, Coura JR, editores. Clínica e Terapêutica da Doença de Chagas: uma abordagem prática para o clínico geral. Rio de Janeiro: Fiocruz; 1997. p. 323-51.) (296296. Coura JR, Abreu LL, Willcox HP, Petana W. Comparative controlled study on the use of benznidazole, nifurtimox and placebo, in the chronic form of Chagas' disease, in a field area with interrupted transmission. I. Preliminary evaluation. Rev Soc Bras Med Trop. 1997 Mar-Apr;30(2):139-44.) (299299. Fragata Filho AA. Tratamento antiparasitário da doença de Chagas. Rev Soc Cardiol Estado de São Paulo. 2009;19:2-5.) (302302. Coura JR, Castro SL. A critical review on Chagas disease chemotherapy. Mem Inst Oswaldo Cruz. 2002 Jan;97(1):3-24.) (312312. Bern C. Chagas' Disease. N Engl J Med. 2015 Nov;373(19):1881-2.) (313313. Portela-Lindoso AAB, Shikanai-Yasuda MA. Doença de Chagas crônica: do xenodiagnóstico e hemocultura à reação em cadeia da polimerase. Rev Saude Publica. 2003 fev;37(1):107-15. . In Brazil, only the 100mg and 12.5mg tablets are available in the SUS network.
-
Adults: 5mg/kg/day, orally, in two or three daily doses, for 60 days.
-
The maximum dose recommended is 300 mg/day.
The 100mg and 50mg benznidazole tablets are distributed in Latin America by the Argentine Endocrine Laboratory (ELEA, Maprimed) and the PAHO Revolving Fund; in Spain, through the Spanish Agency for Medicines and Health Products; and the rest of the world through the ELEA and the WHO 3838. Dias JC, Coura JR, Yasuda MAS. The present situation, challenges, and perspectives regarding the production and utilization of effective drugs against human Chagas disease. Rev Soc Bras Med Trop. 2014 Jan-Feb;47(1):123-5. . In Brazil, it is acquired by the Ministry of Health, and distributed to State Health Secretariats (SES) upon request in the Information Strategic Procurement System (SIES). Benznidazole distribution flow (100mg) for regional and/or municipalities should be established for each SES, articulating Pharmaceutical Assistance, Epidemiological Surveillance and Primary Care. The distribution of benznidazole in doses of 12.5mg is centralized, considering the small occurrence of cases of the disease in the recommended age for use of the drug. Therefore, to apply for a pediatric formulation, the Chagas Disease Technical Group, Communicable Disease Surveillance Coordination of the Secretariat of Health, Ministry of Health should be contacted 4949. Almeida EA, Ramos Júnior AN, Correia D, Shikanai-Yasuda MA. Co-infection Trypanosoma cruzi /HIV: systematic review (1980-2010). Rev Soc Bras Med Trop. 2011 Nov-Dec;44(6):762-70. .
-
Children: 10mg/kg/day orally in two or three daily doses, for 60 days.
-
In all cases, the dose should not exceed 300mg/day. When the daily dose exceeds 300mg, it is recommended that the treatment duration be extended to achieve the total dose calculated for 60 days.
The main advantage of the 12.5mg tablet is that it can be used to treat infants as young as 2 years of age, allowing proper dosing without the need to fragment and manipulate parts of larger tablets. In addition, the main advantage of 50 mg tablet is that it can be used to treat the rest of the pediatric population, including teenagers and young adults 3838. Dias JC, Coura JR, Yasuda MAS. The present situation, challenges, and perspectives regarding the production and utilization of effective drugs against human Chagas disease. Rev Soc Bras Med Trop. 2014 Jan-Feb;47(1):123-5. .
In addition to the specific treatment for T. cruzi infection, antiparasitic drugs can be used for prophylaxis in specific situations. Secondary prophylaxis should be considered after conventional treatment of reactivation in patients co-infected with HIV, and with evidence of AIDS (2.5 to 5mg/kg/day, orally, three times per week), but there is still no consistent evidence to validate the use of benznidazole prophylactically in this situation 4949. Almeida EA, Ramos Júnior AN, Correia D, Shikanai-Yasuda MA. Co-infection Trypanosoma cruzi /HIV: systematic review (1980-2010). Rev Soc Bras Med Trop. 2011 Nov-Dec;44(6):762-70.) (159159. Rocha A, Ramos Júnior AN, Sartori AM, Correia D, Gontijo ED, Tatto E, et al. Recomendações para diagnóstico, tratamento e acompanhamento da co-infecção Trypanosoma cruzi: vírus da imunodeficiência humana. Rev Soc Bras Med Trop. 2006 jul-ago;39(4):392-415.) (175175. Sartori AM, Ibrahim KY, Nunes Westphalen EV, Braz LM, Oliveira Junior OC, Gakiya E, et al. Manifestations of Chagas disease (American trypanosomiasis ) in patients with HIV/AIDS. Ann Trop Med Parasitol. 2007 Jan;101(1):31-50. .
In turn, primary prophylaxis is indicated in situations where accidental exposure to T. cruzi has occurred. The recommended dose is 5mg/kg/day (adult) or 10mg/kg/day (child), orally, twice a day for 10 days, not exceeding 300mg/day. In cases where the inoculum has a high parasite load (≥10 7 trypomastigotes/mL), one should use conventional treatment for acute phase of Chagas disease for at least 60 days 310310. Brener Z. Laboratory-acquired Chagas disease: comment. Trans R Soc Trop Med Hyg. 1987;81(3):527. (Class IIa, level of evidence C). In the case of children, one should use the pediatric formulation of benznidazole, that is now available.
The adverse effects and toxicity of benznidazole, together with the respective actions to be taken, are shown in Figure 18 . Thus, care should be taken before and during treatment to monitor these events. Before starting treatment, clinical examination and laboratory tests (complete blood count, evaluation of liver enzymes, and renal function tests) must be performed. This must be repeated 30 and 60 days after initiating treatment 2020. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Consenso Brasileiro em Doença de Chagas. Rev Soc Bras Med Trop. 2005;38 Supl 3:1-29. .
In cases of intolerance to benznidazole, nifurtimox is a potential therapeutic option 2020. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Consenso Brasileiro em Doença de Chagas. Rev Soc Bras Med Trop. 2005;38 Supl 3:1-29.) (4242. Rassi Júnior A, Rassi A, Marin-Neto JA. Chagas disease. Lancet. 2010 Apr;375(9723):1388-402.) (119119. Organización Panamericana de la Salud; Organización Mundial de la Salud. Tratamiento etiológico de la enfermidad de Chagas: conclusiones de reunión de especialistas. Rev Patol Trop. 1999;28(2):247-79.) (291291. Cançado JR. Terapêutica específica. In: Dias JCP, Coura JR, editores. Clínica e Terapêutica da Doença de Chagas: uma abordagem prática para o clínico geral. Rio de Janeiro: Fiocruz; 1997. p. 323-51.) (292292. Sosa Estani S, Segura EL, Ruiz AM, Velazquez E, Porcel BM, Yampotis C. Efficacy of chemotherapy with benznidazole in children in the indeterminate phase of Chagas' disease. Am J Trop Med Hyg. 1988 Oct;59(4):526-9.) (296296. Coura JR, Abreu LL, Willcox HP, Petana W. Comparative controlled study on the use of benznidazole, nifurtimox and placebo, in the chronic form of Chagas' disease, in a field area with interrupted transmission. I. Preliminary evaluation. Rev Soc Bras Med Trop. 1997 Mar-Apr;30(2):139-44.) (312312. Bern C. Chagas' Disease. N Engl J Med. 2015 Nov;373(19):1881-2. (Class 1, level of evidence B). Nifurtimox comes as a 120mg tablet (adults) and a 30mg tablet (children).
-
Adults: 10mg/kg/day, orally in three daily doses, for 60 days.
-
Children: 15mg/kg/day, orally in three daily doses, for 60 days.
The adverse effects and toxicity of nifurtimox are similar to those of benznidazole, except for a lower digestive tolerance, reflected in anorexia with weight loss, and psychological disturbances 2020. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Consenso Brasileiro em Doença de Chagas. Rev Soc Bras Med Trop. 2005;38 Supl 3:1-29. . In case of treatment failure with benznidazole, nifurtimox can be used, although cross-resistance between the two drugs has been observed.
It is necessary to remember that nifurtimox is not available in the pharmaceutical market of any country to date. In situations where the use of nifurtimox is essential, the drug must be requested via the PAHO and WHO offices. The Ministry of Health of Brazil, through the SVS Chagas Disease Technical Group, has the availability of nifurtimox to be dispensed under the conditions described above of benznidazole intolerance. Nifurtimox is acquired by the Ministry of Health via PAHO donation and is dispensed directly by the Chagas disease Technical Group of SVS 3939. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Guia de Vigilância em Saúde. Brasília: Ministério da Saúde; 2014. . The product may be requested through a standardized protocol directed to this technical group by the Ministry of Health.
Other drugs, such as allopurinol and azole antifungals (ketoconazole, itraconazole, fluconazole, posaconazole), are recognized as suppressors of T. cruzi parasitemia and may be useful in some specific situations, such as reactivation in immunosuppressed patients, when it is impossible to use benznidazole or nifurtimox 4949. Almeida EA, Ramos Júnior AN, Correia D, Shikanai-Yasuda MA. Co-infection Trypanosoma cruzi /HIV: systematic review (1980-2010). Rev Soc Bras Med Trop. 2011 Nov-Dec;44(6):762-70.) (159159. Rocha A, Ramos Júnior AN, Sartori AM, Correia D, Gontijo ED, Tatto E, et al. Recomendações para diagnóstico, tratamento e acompanhamento da co-infecção Trypanosoma cruzi: vírus da imunodeficiência humana. Rev Soc Bras Med Trop. 2006 jul-ago;39(4):392-415.) (175175. Sartori AM, Ibrahim KY, Nunes Westphalen EV, Braz LM, Oliveira Junior OC, Gakiya E, et al. Manifestations of Chagas disease (American trypanosomiasis ) in patients with HIV/AIDS. Ann Trop Med Parasitol. 2007 Jan;101(1):31-50. (Class IIa, level of evidence C). However, there is undeniable clinical evidence of proven therapeutic efficacy.
The antiparasitic treatment with benznidazole should not be imposed on pregnant women, or women of childbearing age who are not making a regular use of well-known contraceptive methods 2020. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Consenso Brasileiro em Doença de Chagas. Rev Soc Bras Med Trop. 2005;38 Supl 3:1-29. . As discussed in the specific section on vertical transmission of T. cruzi in this Consensus, there is evidence indicating the benefit of the antiparasitic treatment, with better evolution of Chagas disease 116116. Carlier Y, Sosa-Estani S, Luquetti AO, Buekens P. Congenital Chagas disease: an update. Mem Inst Oswaldo Cruz. 2015 May;110(3):363-8.) (119119. Organización Panamericana de la Salud; Organización Mundial de la Salud. Tratamiento etiológico de la enfermidad de Chagas: conclusiones de reunión de especialistas. Rev Patol Trop. 1999;28(2):247-79.) (120120. Fabbro DL, Streiger ML, Arias ED, Bizai ML, Barco M, Amicone NA. Trypanocide treatment among adults with chronic Chagas disease living in Santa Fe City (Argentina), over a mean follow-up of 21 years: parasitological, serological and clinical evolution. Rev Soc Bras Med Trop. 2007 Jan-Feb;40(1):1-10.) (121121. Lana M, Lopes LA, Martins HR, Bahia MT, Machado-de-Assis GF, Wendling AP, et al. Clinical and laboratory status of patients with chronic Chagas disease living in a vector-controlled area in Minas Gerais, Brazil, before and nine years after aetiological treatment. Mem Inst Oswaldo Cruz. 2009 Dec;104(8):1139-47.) (122122. Pérez-Molina JA, Pérez-Ayala A, Moreno S, Fernández-González MC, Zamora J, López-Velez R. Use of benznidazole to treat chronic Chagas' disease: a systematic review with a meta-analysis. J Antimicrob Chemother. 2009 Dec;64(6):1139-47. , reduced risk of vertical transmission in future pregnancies 109109. Sosa-Estani S, Cura E, Velazquez E, Yampotis C, Segura EL. Etiological treatment of young women infected with Trypanosoma cruzi , and prevention of congenital transmission. Rev Soc Bras Med Trop. 2009 Sep-Oct;42(5):484-7.) (116116. Carlier Y, Sosa-Estani S, Luquetti AO, Buekens P. Congenital Chagas disease: an update. Mem Inst Oswaldo Cruz. 2015 May;110(3):363-8. , and lower risk of vertical transmission and evolution to spontaneous abortion by reducing the parasitemia 103103. Salas NA, Cot M, Schneider D, Mendoza B, Santalla JA, Postigo J, et al. Risk factors and consequences of congenital Chagas disease in Yacuiba, South Bolivia. Trop Med Int Health. 2007 Dec;12(12):1498-505.) (115115. González-Tomé MI, Rivera M, Camaño I, Norman F, Flores-Chávez M, Rodríguez-Gómez L, et al. Recomendaciones para el diagnóstico, seguimiento y tratamiento de la embarazada y del niño con enfermedad de Chagas. Enferm Infecc Microbiol Clin. 2013 Oct;31(8):535-42. .
In the case of pregnant women for whom antiparasitic treatment is indicated, the risk-benefit ratio and available treatment options (in particular, acute phase and co-infection) should be considered in each case. The indication in patients with other serious disorders, such as hepatic and renal failure, must be carefully assessed case by case, according to the severity, as well as prior adverse events to drug components that may constitute relative contraindications 2020. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Consenso Brasileiro em Doença de Chagas. Rev Soc Bras Med Trop. 2005;38 Supl 3:1-29.) (3838. Dias JC, Coura JR, Yasuda MAS. The present situation, challenges, and perspectives regarding the production and utilization of effective drugs against human Chagas disease. Rev Soc Bras Med Trop. 2014 Jan-Feb;47(1):123-5.) (104104. Carlier Y, Torrico F, Sosa-Estani S, Russomando G, Luquetti A, Freilij H, et al. Congenital Chagas disease: recommendations for diagnosis, treatment and control of newborns, siblings and pregnant women. PLoS Negl Trop Dis. 2011 Oct;5(10):e1250. .
Evaluation of cure of Chagas disease after antiparasitic treatment
Demonstrating serology becoming negative is considered the only method to determine cure following antiparasitic treatment of Chagas disease. The time required for this to occur is variable and depends on the stage and duration of disease, with one year for congenital infection, 3-5 years for the acute phase, 5-10 years for the recent chronic phase and >10 years (usually 20-25 years) for the chronic phase of long duration. The reduction in serologic test titers occurs gradually. The persistent and progressive decline above three dilutions of the titers of the serologic tests is suggestive that these will be negative. Parasitologic examinations are not mandatory for evidence of cure of Chagas disease, however, at any time, positive parasitologic tests indicate therapeutic failure. PCR is an alternative option to indirect methods (xenodiagnosis and blood culture) in the parasitologic evaluation as a treatment failure criterion after completing antiparasitic treatment 104104. Carlier Y, Torrico F, Sosa-Estani S, Russomando G, Luquetti A, Freilij H, et al. Congenital Chagas disease: recommendations for diagnosis, treatment and control of newborns, siblings and pregnant women. PLoS Negl Trop Dis. 2011 Oct;5(10):e1250.) (313313. Portela-Lindoso AAB, Shikanai-Yasuda MA. Doença de Chagas crônica: do xenodiagnóstico e hemocultura à reação em cadeia da polimerase. Rev Saude Publica. 2003 fev;37(1):107-15. . However, there are still limitations on the availability and standardization of techniques in the health networks of endemic countries 313313. Portela-Lindoso AAB, Shikanai-Yasuda MA. Doença de Chagas crônica: do xenodiagnóstico e hemocultura à reação em cadeia da polimerase. Rev Saude Publica. 2003 fev;37(1):107-15. .
Who and where to treat
Considering the operational specificities of national health systems, use of antiparasitic treatment for Chagas disease by doctors working in primary care who know the peculiarities of the drugs and Chagas disease 2020. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Consenso Brasileiro em Doença de Chagas. Rev Soc Bras Med Trop. 2005;38 Supl 3:1-29.) (303303. Maguire JH. Treatment of Chagas' disease: time is running out. N Engl J Med. 2015 Oct;373(14):1369-70. , is feasible, safe 2020. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Consenso Brasileiro em Doença de Chagas. Rev Soc Bras Med Trop. 2005;38 Supl 3:1-29.) (251251. Morillo CA, Marin-Neto JA, Avezum A, Sosa-Estani S, Rassi Júnior A, Rosas F, et al; BENEFIT Investigators. Randomized Trial of Benznidazole for Chronic Chagas' Cardiomyopathy. N Engl J Med. 2015 Oct;373(14):1295-306.) (294294. Yun O, Lima MA, Ellman T, Chambi W, Castillo S, Flevaud L, et al. Feasibility, drug safety, and effectiveness of etiological treatment programs for Chagas disease in Honduras, Guatemala, and Bolivia: 10-year experience of Médecins Sans Frontières. PLoS Negl Trop Dis. 2009 Jul;3(7):e488.) (303303. Maguire JH. Treatment of Chagas' disease: time is running out. N Engl J Med. 2015 Oct;373(14):1369-70.) (312312. Bern C. Chagas' Disease. N Engl J Med. 2015 Nov;373(19):1881-2. , and operationally possible 294294. Yun O, Lima MA, Ellman T, Chambi W, Castillo S, Flevaud L, et al. Feasibility, drug safety, and effectiveness of etiological treatment programs for Chagas disease in Honduras, Guatemala, and Bolivia: 10-year experience of Médecins Sans Frontières. PLoS Negl Trop Dis. 2009 Jul;3(7):e488. . Depending on the severity of clinical conditions of each case, particularly cases with acute or reactivated and decompensated chronic forms, it may be necessary to refer certain patients to more specialized or reference units that have greater technological capability, or even for hospitalization.
Figure 19 provides an overview of potential situations for specific treatment, taking as reference the recommendation of class and the level of existing evidence. Figure 18 shows the adverse effects of benznidazole and recommended approaches for each given situation.
Tribute
Tribute is paid to members of the group responsible for the Brazilian Consensus on Chagas Disease in 2005 who died in the period between the consensuses: Ademir Rocha, Aluízio Rosa Prata, Antonio Carlos Silveira, Guilherme Rodrigues da Silva, Joffre Marcondes de Rezende, and Vanize de Oliveira Macedo.
Contributions
General coordination of the Consensus and final critical reviewing: Dias JCP and Ramos Jr AN. Thematic coordination, design, writing, critical reviewing and final approval of the version to be published: Dias JCP, Ramos Jr AN, Correia D, Gontijo ED, Ostermayer AL, Shikanai-Yasuda MA, Coura JR, Torres RM, Melo JRC, Almeida EA Oliveira Jr W, Cardoso AV, Costa VM, Lima MM and Alves RV.
Representing the Ministry of Health of Brazil - critical reviewing and final approval of the version to be published: Cardoso AV, Costa VM, Lima MM and Alves RV.
Group of experts who participated in the composition of the Brazilian Consensus on Chagas Disease, 2015 - writing and responsibility for the document in terms of accuracy and integrity: Dias JCP, Ramos Jr AN, Gontijo ED, Ostermayer AL, Shikanai-Yasuda MA, Coura JR, Torres RM, Melo JRC, Almeida EA, Oliveira Jr W, Silveira AC, Rezende JM, Pinto FS, Ferreira AW, Rassi A, Fragata Filho AA, Sousa AS, Correia D, Jansen AM, Andrade GMQ, Britto C, Pinto AYN, Rassi Jr A, Campos DE, Abad-Franch F, Santos SE, Chiari E, Hasslocher-Moreno AM, Moreira EF, Marques DSO, Silva EL, Marin-Neto JA, Galvão LMC, Xavier SS, Valente SAS, Carvalho NB, Cardoso AV, Costa VM, Vivaldini SM, Oliveira SM, Valente VC, Lima MM and Alves RV.
Acknowledgements
We thank Dr. Jarbas Barbosa da Silva Jr., Secretary of Surveillance in Health of the Ministry of Health in the periods 2003-2007 and 2011-2014, for supporting the construction of two issues of the Consensus on Chagas Disease, 2005 and 2015. To Dr. Habib Fraiha Neto and Dr. Ralph Lainson (in memoriam), for the contributions in the section dedicated to oral transmission. To the Brazilian Network of Attention and Studies on Trypanosoma cruzi /HIV Coinfection and other immunosuppressive conditions. We thank Dr. Pedro Albajar-Viñas (World Health Organization - HIV/AIDS, Tuberculosis, Malaria and Neglected Diseases - Control of Neglected Tropical Diseases - Innovative & Intensified Disease Management - Chagas disease) for the support of the translation to the english version of the Brazilian Consensus on Chagas disease.
Referências
-
1World Health Organization. Research priorities for Chagas disease, human African trypanosomiasis and leishmaniasis. WHO: technical report of the TDR Disease Reference Group on Chagas Disease, Human African Trypanosomiasis and Leishmaniasis. Geneva: World Health Organization; 2012. (WHO Technical Report Series, 975).
-
2World Health Organization. Sustaining the drive to overcome the global impact of neglected tropical diseases: second WHO report in neglected tropical diseases. Geneva: World Health Organization; 2013.
-
3Dias JC, Silveira AC, Schofield CJ. The impact of Chagas disease control in Latin America: a review. Mem Inst Oswaldo Cruz. 2002 Jul;97(5):603-12.
-
4Coura JR, Dias JC. Epidemiology, control and surveillance of Chagas disease: 100 years after its discovery. Mem Inst Oswaldo Cruz. 2009 Jul;104 Suppl 1:31-40.
-
5World Health Organization. Chagas disease in Latin America: an epidemiological update based on 2010 estimates. Wkly Epidemiol Rec. 2015 Feb;90(6):33-44.
-
6Moncayo A, Silveira AC. Current epidemiological trends for Chagas disease in Latin America and future challenges in epidemiology, surveillance and health policy. Mem Inst Oswaldo Cruz. 2009 Jul;104 Suppl 1:17-30.
-
7Schmunis GA, Yadon ZE. Chagas disease: a Latin American health problem becoming a world health problem. Acta Trop. 2010 Jul-Aug;115(1-2):14-21.
-
8Centers for Disease Control and Prevention. Center for Global Health. Division of Parasitic Diseases and Malaria. Chagas disease in the Americas: 2013. Atlanta: Centers for Disease Control and Prevention; 2013.
-
9Hotez PJ, Dumonteil E, Betancourt Cravioto M, Bottazzi ME, Tapia-Conyer R, Meymandi S, et al. An unfolding tragedy of Chagas disease in North America. PLoS Negl Trop Dis. 2013 Oct;7(10):e2300.
-
10Dias JCP. Human chagas disease and migration in the context of globalization: some particular aspects. J Trop Med. 2013:789758.
-
11Coura JR, Viñas PA, Junqueira AC. Ecoepidemiology, short history and control of Chagas disease in the endemic countries and the new challenge for non-endemic countries. Mem Inst Oswaldo Cruz. 2014 Nov;109(7):856-62.
-
12European Centre for Disease Prevention and Control. Assessing the burden of key infectious diseases affecting migrant populations in the EU/EEA: technical report. Stockholm: ECDC; 2014.
-
13Requena-Méndez A, Aldasoro E, Lazzari E, Sicuri E, Brown M, Moore DA, et al. Prevalence of Chagas disease in Latin-American migrants living in Europe: a systematic review and meta-analysis. PLoS Negl Trop Dis. 2015 Feb;9(2):e0003540.
-
14World Health Organization. Chagas disease (American trypanosomiasis) [Internet]. Geneva: World Health Organization ; 2015 [citado 2015 Nov 13]. Disponível em: Disponível em: http://www.who.int/mediacentre/factsheets/fs340/en/
» http://www.who.int/mediacentre/factsheets/fs340/en/ -
15Ramos Júnior AN, Carvalho DM. Chagas' disease: past, present and future. Cad Saude Colet. 2009;17(4):787-94.
-
16Strasen J, Williams T, Ertl G, Zoller T, Stich A, Ritter O. Epidemiology of Chagas disease in Europe: many calculations, little knowledge. Clin Res Cardiol. 2014 Jan;103(1):1-10.
-
17Pan American Health Organization. Estimación cuantitativa de la enfermedad de Chagas en las Americas. Washington: Pan American Health Organization; 2006.
-
18Dias JCP. Southern Cone Initiative for the elimination of domestic populations of Triatoma infestans and the interruption of transfusion Chagas disease: historical aspects, present situation, and perspectives. Mem Inst Oswaldo Cruz. 2007 Oct;102 Suppl 1:11-8.
-
19Prata A. Clinical and epidemiological aspects of Chagas disease. Lancet Infect Dis. 2001 Sep;1(2):92-100.
-
20Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Consenso Brasileiro em Doença de Chagas. Rev Soc Bras Med Trop. 2005;38 Supl 3:1-29.
-
21Organização Pan-americana da Saúde. Guia para vigilância, prevenção, controle e manejo clínico da doença de Chagas aguda transmitida por alimentos. Rio de Janeiro: Organização Pan-Americana da Saúde; 2009. (Série de Manuais Técnicos, 12).
-
22Silveira AC, Dias JCP. O controle da transmissão vetorial. Rev Soc Bras Med Trop. 2011;44 supl 2:52-63.
-
23Coura JR. The main sceneries of Chagas disease transmission. The vectors, blood and oral transmissions: a comprehensive review. Mem Inst Oswaldo Cruz. 2015 May;110(3):277-82.
-
24Victora CG, Wagstaff A, Schellenberg JA, Gwatkin D, Claeson M, Habicht JP. Applying an equity lens to child health and mortality: more of the same is not enough. Lancet. 2003 Jul;362(9379):233-41.
-
25Zingales B, Andrade SG, Briones MR, Campbell DA, Chiari E, Fernandes O, et al. A new consensus for Trypanosoma cruzi intraspecific nomenclature: second revision meeting recommends TcI to TcVI. Mem Inst Oswaldo Cruz. 2009 Nov;104(7):1051-4.
-
26Zingales B, Miles MA, Moraes CB, Luquetti A, Guhl F, Schijman AG, et al. Drug discovery for Chagas disease should consider Trypanosoma cruzi strain diversity. Mem Inst Oswaldo Cruz. 2014 Aug;109(6):828-33.
-
27Messenger LA, Miles MA, Bern C. Between a bug and a hard place: Trypanosoma cruzi genetic diversity and the clinical outcomes of Chagas disease. Expert Rev Anti Infect Ther. 2015 Aug;13(8):995-1029.
-
28Victora CG, Barreto ML, Carmo Leal M, Monteiro CA, Schmidt MI, Paim J, et al. Health conditions and health-policy innovations in Brazil: the way forward. Lancet. 2011 Jun;377(9782):2042-53.
-
29Barreto ML, Teixeira MG, Bastos FI, Ximenes RAA, Barata RB, Rodrigues LC. Successes and failures in the control of infectious diseases in Brazil: social and environmental context, policies, interventions, and research needs. Lancet. 2011 May;377(9780):1877-89.
-
30Mendes EV. O cuidado das condições crônicas na atenção primária à saúde: o imperativo da consolidação da estratégia da saúde da família. Brasília: Organização Pan-Americana da Saúde; 2012.
-
31Nações Unidas. Transformando nosso mundo: a agenda 2030 para o desenvolvimento sustentável [Internet]. [Rio de Janeiro]: Nações Unidas no Brasil; 2015 [citado 2015 out 4]. Disponível em: http://nacoesunidas.org/pos2015/agenda2030/ .
» http://nacoesunidas.org/pos2015/agenda2030/ -
32Camargo ME, Silva GR, Castilho EA, Silveira AC. Serological survey of the prevalence of Chagas' infection in Brazil, 1975/1980. Rev Inst Med Trop São Paulo. 1984 Jul-Aug;26(4):192-204.
-
33Silveira AC, Silva GR, Prata A. O Inquérito de soroprevalência da infecção chagásica humana (1975-1980). Rev Soc Bras Med Trop. 2011;44 supl 2:33-9.
-
34Akhavan D. Análise de custo-efectividade do programa de controle da doença de Chagas no Brasil: relatório final. Brasília: Organização Pan-Americana de Saúde; 1998.
-
35Martins-Melo FR, Ramos Júnior AN, Alencar CH, Heukelbach J. Prevalence of Chagas disease in Brazil: a systematic review and meta-analysis. Acta Trop. 2014 Feb;130:167-74.
-
36Hotez PJ, Fujiwara RT. Brazil's neglected tropical diseases: an overview and a report card. Microbes Infect. 2014 Aug;16(8):601-6.
-
37Bern C, Kjos S, Yabsley MJ. Montgomery SP. Trypanosoma cruzi and Chagas disease in the United States. Clin Microbiol Rev. 2011 Oct;24(4):655-81.
-
38Dias JC, Coura JR, Yasuda MAS. The present situation, challenges, and perspectives regarding the production and utilization of effective drugs against human Chagas disease. Rev Soc Bras Med Trop. 2014 Jan-Feb;47(1):123-5.
-
39Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Guia de Vigilância em Saúde. Brasília: Ministério da Saúde; 2014.
-
40Instituto Brasileiro de Geografia e Estatística. Projeção da população do Brasil por sexo e idade para o período 2000/2060 e projeção da população das Unidades da Federação por sexo e idade para o período 2000/2030. Brasília: Instituto Brasileiro de Geografia e Estatística; 2013.
-
41Bern C, Verastegui M, Gilman RH, Lafuente C, Galdos-Cardenas G, Calderon M, et al. Congenital Trypanosoma cruzi transmission in Santa Cruz, Bolivia. Clin Infect Dis. 2009 Dec 1;49(11):1667-74.
-
42Rassi Júnior A, Rassi A, Marin-Neto JA. Chagas disease. Lancet. 2010 Apr;375(9723):1388-402.
-
43Ventura-Garcia L, Roura M, Pell C, Posada E, Gascón J, Aldasoro E, et al. Socio-cultural aspects of Chagas disease: a systematic review of qualitative research. PLoS Negl Trop Dis. 2013 Sep 12;7(9):e2410.
-
44Bern C, Montgomery SP. An estimate of the burden of Chagas disease in the United States. Clin Infect Dis. 2009 Sep 1;49(5):e52-4.
-
45Buekens P, Almendares O, Carlier Y, Dumonteil E, Eberhard M, Gamboa-Leon R, et al. Mother-to-child transmission of Chagas' disease in North America: why don't we do more? Matern Child Health J. 2008 May;12(3):283-6.
-
46Silveira C, Carneiro Junior N, Ribeiro MC, Barata RC. Living conditions and access to health services by Bolivian immigrants in the city of São Paulo, Brazil. Cad Saude Publica. 2013 Oct;29(10):2017-27.
-
47Martes ACB, Faleiros SM. Acesso dos imigrantes bolivianos aos serviços públicos de saúde na cidade de São Paulo. Saude Soc. 2013 abr-jun;22(2):351-64.
-
48Almeida EA, Lima JN, Lages-Silva E, Guariento ME, Aoki FH, Torres-Morales AE, et al. Chagas' disease and HIV co-infection in patients without effective antiretroviral therapy: prevalence, clinical presentation and natural history. Trans R Soc Trop Med Hyg. 2010 Jul;104(7):447-52.
-
49Almeida EA, Ramos Júnior AN, Correia D, Shikanai-Yasuda MA. Co-infection Trypanosoma cruzi /HIV: systematic review (1980-2010). Rev Soc Bras Med Trop. 2011 Nov-Dec;44(6):762-70.
-
50Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Departamento de DST, Aids e Hepatites Virais. Bol Epidemiol. 2014;3(1):1-80
-
51Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Departamento de DST, Aids e Hepatites Virais. Protocolo clínico e diretrizes terapêuticas para manejo da infecção pelo HIV em adultos. Brasília: Ministério da Saúde; 2014.
-
52Ministério da Saúde (BR). Recomendações para diagnóstico, tratamento e acompanhamento da co-infecção Trypanosoma cruzi: vírus da imunodeficiência humana. Rev Soc Bras Med Trop. 2006 jul-ago;39(4):392-415.
-
53Martins-Melo FR, Ramos Júnior AN, Alencar CH, Heukelbach J. Mortality related to Chagas disease and HIV/AIDS coinfection in Brazil. J Trop Med. 2012;2012:534649.
-
54Ramos Júnior AN. Inclusão da reativação da doença de Chagas como uma condição definidora de AIDS para fins de vigilância epidemiológica no Brasil. Rev Soc Bras Med Trop. 2004 mar-abr;37(2):192-3.
-
55Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Doença de Chagas aguda no Brasil: série histórica de 2000 a 2013. Bol Epidemiol. 2015;46(21):1-9.
-
56Martins-Melo FR, Ramos Júnior AN, Alencar CH, Heukelbach J. Multiple causes of death related to Chagas' disease in Brazil, 1999 to 2007. Rev Soc Bras Med Trop. 2012 Sep-Oct;45(5):591-6.
-
57Martins-Melo FR, Ramos Júnior AN, Alencar CH, Heukelbach J. Mortality due to Chagas disease in Brazil from 1979 to 2009: trends and regional differences. J Infect Dev Ctries. 2012 Nov;6(11):817-24.
-
58Drumond JAG, Marcopito LF. Internal migration and distribution of Chagas disease mortality, Brazil, 1981-1998. Cad Saude Publica. 2006 Oct;22(10):2131-40.
-
59Martins-Melo FR, Alencar CH, Ramos Júnior AN, Heukelbach J. Epidemiology of mortality related to Chagas' disease in Brazil, 1999-2007. PLoS Negl Trop Dis. 2012 Feb;6(2):e1508.
-
60Martins-Melo FR, Ramos Júnior AN, Alencar CH, Lange W, Heukelbach J. Mortality of Chagas' disease in Brazil: spatial patterns and definition of high-risk areas. Trop Med Int Health. 2012 Sep;17(9):1066-75.
-
61Santo AH. Tendência da mortalidade relacionada à doença de Chagas, Estado de São Paulo, Brasil, 1985 a 2006: estudo usando causas múltiplas de morte. Rev Panam Salud Publica. 2009 Oct;26(4):299-309.
-
62Nóbrega AA, Araújo WN, Vasconcelos AM. Mortality due to Chagas disease in Brazil according to a specific cause. Am J Trop Med Hyg. 2014 Sep;91(3):528-33.
-
63Alves RM, Thomaz RP, Almeida EA, Wanderley JS, Guariento ME. Chagas' disease and ageing: the coexistence of other chronic diseases with Chagas' disease in elderly patients. Rev Soc Bras Med Trop. 2009 Nov-Dec;42(6):622-8.
-
64Lima-Costa MF, Peixoto SV, Ribeiro AL. Chagas disease and mortality in old age as an emerging issue: 10 year follow-up of the Bambuí population-based cohort study (Brazil). Int J Cardiol. 2010 Nov;145(2):362-3.
-
65Silveira AC. Os novos desafios e perspectivas futuras do controle. Rev Soc Bras Med Trop. 2011;44 Supl 2:122-4.
-
66Passos ADC, Silveira AC. Síntese dos resultados dos inquéritos nacionais. Rev Soc Bras Med Trop. 2011;44 supl 2:47-50.
-
67Vinhaes MC, Oliveira SV, Reis PO, Lacerda Sousa AC, Silva RA, Obara MT, et al. Assessing the vulnerability of Brazilian municipalities to the vectorial transmission of Trypanosoma cruzi using multi-criteria decision analysis. Acta Trop. 2014 Sep;137:105-10.
-
68Ramos Júnior AN, Carvalho DM. Os diferentes significados da certificação conferida ao Brasil como estando livre da doença de Chagas. Cad Saude Publica. 2001 nov-dez;17(6):1403-12.
-
69Abad-Franch F, Diotaiuti L, Gurgel-Gonçalves R, Gürtler RE. Certifying the interruption of Chagas disease transmission by native vectors: cui bono? Mem Inst Oswaldo Cruz. 2013 Apr;108(2):251-4.
-
70Valença-Barbosa C, Lima MM, Sarquis O, Bezerra CM, Abad-Franch F. Modeling disease vector occurrence when detection is imperfect II: drivers of site-occupancy by synanthropic Triatoma brasiliensis in the Brazilian northeast. PLoS Negl Trop Dis. 2014 May;8(5):e2861.
-
71Abad-Franch F, Valença-Barbosa C, Sarquis O, Lima MM. All that glisters is not gold: sampling-process uncertainty in disease-vector surveys with false-negative and false-positive detections. PLoS Negl Trop Dis. 2014 Sep;8(9):e3187.
-
72Salvatella R, Irabedra P, Castellanos LG. Interruption of vector transmission by native vectors and "the art of the possible". Mem Inst Oswaldo Cruz. 2014 Feb;109(1):122-30.
-
73Maegawa FA, Damerau EF, Beltrame-Botelho IT, Lopes A, Emmanuelle-Machado P, Steindel M, et al. Autochthonous Chagas' disease in Santa Catarina State, Brazil: report of the first case of digestive tract involvement. Rev Soc Bras Med Trop. 2003 Sep-Oct;36(5):609-12.
-
74Santos CB, Ferreira AL, Leite GR, Ferreira GE, Rodrigues AA, Falqueto A. Peridomiciliary colonies of Triatoma vitticeps (Stal, 1859) (Hemiptera, Reduviidae, Triatominae) infected with Trypanosoma cruzi in rural areas of the state of Espírito Santo, Brazil. Mem Inst Oswaldo Cruz. 2005 Aug;100(5):471-3.
-
75Lorosa ES, Santos CM, Jurberg J. Foco de doença de Chagas em São Fidélis, no estado do Rio de Janeiro. Rev Soc Bras Med Trop. 2008 jul-ago;41(4):419-20.
-
76Shikanai-Yasuda MA, Carvalho NB. Oral transmission of Chagas disease. Clin Infect Dis. 2012 Mar;54(6):845-52.
-
77Sangenis LH, Saraiva RM, Georg I, Castro L, Santos Lima V, Roque AL, et al. Autochthonous transmission of Chagas disease in Rio de Janeiro State, Brazil: a clinical and eco-epidemiological study. BMC Infect Dis. 2015 Jan;15:4.
-
78Xavier SC, Roque AL, Lima VS, Monteiro KJ, Otaviano JC, Ferreira da Silva LF, et al. Lower richness of small wild mammal species and Chagas disease risk. PLoS Negl Trop Dis. 2012;6(5):e1647.
-
79Xavier SC, Roque AL, Bilac D, Araújo VA, Costa Neto SF, Lorosa ES, et al. Distantiae transmission of Trypanosoma cruzi: a new epidemiological feature of acute Chagas disease in Brazil. PLoS Negl Trop Dis. 2014 May;8(5):e2878.
-
80Roque AL, Xavier SC, Gerhardt M, Silva MF, Lima VS, D'Andrea PS, et al. Trypanosoma cruzi among wild and domestic mammals in different areas of the Abaetetuba municipality (Pará State, Brazil), an endemic Chagas disease transmission area. Vet Parasitol. 2013 Mar;193(1-3):71-7.
-
81Roque AL, Xavier SC, Rocha MG, Duarte AC, D'Andrea PS, Jansen AM. Trypanosoma cruzi transmission cycle among wild and domestic mammals in three areas of orally transmitted Chagas disease outbreaks. Am J Trop Med Hyg. 2008 Nov;79(5):742-9.
-
82Abad-Franch F, Ferraz G, Campos C, Palomeque FS, Grijalva MJ, Aguilar HM, et al. Modeling disease vector occurrence when detection is imperfect: infestation of Amazonian palm trees by triatomine bugs at three spatial scales. PLoS Negl Trop Dis. 2010 Mar;4(3):e620.
-
83Abad-Franch F, Santos WS, Schofield CJ. Research needs for Chagas disease prevention. Acta Trop. 2010 Jul-Aug;115(1-2):44-54.
-
84Padilla-Torres SD, Ferraz G, Luz SL, Zamora-Perea E, Abad-Franch F. Modeling dengue vector dynamics under imperfect detection: three years of site-occupancy by Aedes aegypti and Aedes albopictus in urban Amazonia. PLoS One. 2013;8(3):e58420.
-
85Abad-Franch F, Vega MC, Rolón MS, Santos WS, Rojas de Arias A. Community participation in Chagas disease vector surveillance: systematic review. PLoS Negl Trop Dis. 2011 Jun;5(6):e1207.
-
86Roux E, Fátima Venâncio A, Girres JF, Romaña CA. Spatial patterns and eco-epidemiological systems--part II: characterising spatial patterns of the occurrence of the insect vectors of Chagas disease based on remote sensing and field data. Geospat Health. 2011 Nov;6(1):53-64.
-
87Roux E, Fátima Venâncio A, Girres JF, Romaña CA. Spatial patterns and eco-epidemiological systems--part I: multi-scale spatial modelling of the occurrence of Chagas disease insect vectors. Geospat Health. 2011 Nov;6(1):41-51.
-
88Mougabure-Cueto G, Picollo MI. Insecticide resistance in vector Chagas disease: evolution, mechanisms and management. Acta Trop. 2015 Sep;149:70-85.
-
89Pessoa GCA, Pinheiro LC, Ferraz ML, Mello BV, Diotaiuti L. Standardization of laboratory bioassays for the study of Triatoma sordida susceptibility to pyrethroid insecticides. Parasit Vectors. 2015 Feb;8:109.
-
90Pessoa GCA, Trevizani NAB, Dias LS, Bezerra CM, Melo BV, Diotaiut L. Toxicological profile of deltamethrin in Triatoma brasiliensis (Hemiptera: Reduviidae) in State of Ceará, Northeastern Brazil. Rev Soc Bras Med Trop. 2015 Jan-Feb;48(1):39-43.
-
91Pinto AYN, Valente SA, Valente VC, Ferreira Júnior AG, Coura JR. Fase aguda da doença de Chagas na Amazônia brasileira: estudo de 233 casos do Pará, Amapá e Maranhão observados entre 1988 e 2005. Rev Soc Bras Med Trop. 2008 nov-dec;41(6):602-14.
-
92Nóbrega AA, Garcia MH, Tatto E, Obara MT, Costa E, Sobel J, et al. Oral transmission of Chagas disease by consumption of açaí palm fruit, Brazil. Emerg Infect Dis. 2009 Apr;15(4):653-5.
-
93Dias JCP, Amato Neto V. Prevenção referente às modalidades alternativas de transmissão do Trypanosoma cruzi no Brasil. Rev Soc Bras Med Trop. 2011;44 supl 2:68-72.
-
94Dias JCP. Notas sobre o Trypanosoma cruzi e suas características bio-ecológicas, como agente de enfermidades transmitidas por alimentos. Rev Soc Bras Med Trop. 2006 jul-ago;39(4):370-5.
-
95Dias JCP, Schofield CJ. Controle da transmissão transfusional da doença de Chagas na Iniciativa do Cone Sul. Rev Soc Bras Med Trop. 1998 jul-ago;31(4):373-83.
-
96Moraes-Souza H, Ferreira-Silva MM. Controle da transmissão transfusional. Rev Soc Bras Med Trop. 2011;44 supl 2:64-7.
-
97Ministério da Saúde (BR). Agência Nacional de Vigilância Sanitária. Manual técnico para a investigação da transmissão de doenças pelo sangue. Brasília: Ministério da Saúde; 2004. (Série A. Normas e manuais técnicos).
-
98Ministério da Saúde (BR). Agência Nacional de Vigilância Sanitária. Manual técnico de hemovigilância. Brasília: Ministério da Saúde; 2004. (Série A. Normas e manuais técnicos).
-
99Angheben A, Boix L, Buonfrate D, Gobbi F, Bisoffi Z, Pupella S, et al. Chagas disease and transfusion medicine: a perspective from non-endemic countries. Blood Transfus. 2015 Oct;13(4):540-50.
-
100Pinazo MJ, Miranda B, Rodríguez-Villar C, Altclas J, Brunet Serra M, García-Otero EC, et al. Recommendations for management of Chagas disease in organ and hematopoietic tissue transplantation programs in nonendemic areas. Transplant Rev (Orlando). 2011 Jul;25(3):91-101.
-
101Ostermayer AL, Passos ADC, Silveira AC, Ferreira AW, Macedo V, Prata AR. O inquérito nacional de soroprevalência de avaliação do controle da doença de Chagas no Brasil (2001-2008). Rev Soc Bras Med Trop. 2011;44 supl 2:108-21.
-
102Torrico F, Alonso-Vega C, Suarez E, Rodriguez P, Torrico MC, Dramaix M, et al. Maternal Trypanosoma cruzi infection, pregnancy outcome, morbidity, and mortality of congenitally infected and non-infected newborns in Bolivia. Am J Trop Med Hyg. 2004 Feb;70(2):201-9.
-
103Salas NA, Cot M, Schneider D, Mendoza B, Santalla JA, Postigo J, et al. Risk factors and consequences of congenital Chagas disease in Yacuiba, South Bolivia. Trop Med Int Health. 2007 Dec;12(12):1498-505.
-
104Carlier Y, Torrico F, Sosa-Estani S, Russomando G, Luquetti A, Freilij H, et al. Congenital Chagas disease: recommendations for diagnosis, treatment and control of newborns, siblings and pregnant women. PLoS Negl Trop Dis. 2011 Oct;5(10):e1250.
-
105Howard EJ, Xiong X, Carlier Y, Sosa-Estani S, Buekens P. Frequency of the congenital transmission of Trypanosoma cruzi: a systematic review and meta-analysis. BJOG. 2014 Jan;121(1):22-33.
-
106Martins-Melo FR, Lima MS, Ramos Júnior AN, Alencar CH, Heukelbach J. Prevalence of Chagas disease in pregnant women and congenital transmission of Trypanosoma cruzi in Brazil: a systematic review and meta-analysis. Trop Med Int Health. 2014 Aug;19(8):943-57.
-
107Gontijo ED, Andrade GMQ, Santos SE, Galvão LMC, Moreira EF, Pinto FS, et al. Triagem neonatal da infecção pelo Trypanosoma cruzi em Minas Gerais, Brasil: transmissão congênita e mapeamento das áreas endêmicas. Epidemiol Serv Saude. 2009 jul-set;18(3):243-54.
-
108Luquetti AO, Tavares SBN, Siriano LR, Oliveira RA, Campos DE, Morais CA, et al. Congenital transmission of Trypanosoma cruzi in central Brazil. A study of 1,211 individuals born to infected mothers. Mem Inst Oswaldo Cruz. 2015 May;110(3):369-76.
-
109Sosa-Estani S, Cura E, Velazquez E, Yampotis C, Segura EL. Etiological treatment of young women infected with Trypanosoma cruzi , and prevention of congenital transmission. Rev Soc Bras Med Trop. 2009 Sep-Oct;42(5):484-7.
-
110Herwaldt BL. Laboratory-acquired parasitic infections from accidental exposures. Clin Microbiol Rev. 2001 Oct;14(4):659-88.
-
111Mahoney RT, Morel CMA. Global Health Innovation System (GHIS). Innovation Strategy Today. 2006;2(1):1-12.
-
112Norman FF, López-Vélez R. Chagas disease and breast-feeding. Emerg Infect Dis. 2013 Oct;19(10):1561-6.
-
113Gebrekristos HT, Buekens P. Mother-to-Child transmission of Trypanosoma cruzi . J Ped Infect Dis Soc. 2014 Sep;3 Suppl 1:S36-40.
-
114De Rissio AM, Riarte AR, Martin García M, Esteva MI, Quaglino M, Ruiz AM. Congenital Trypanosoma cruzi infection. Efficacy of its monitoring in an Urban Reference Health Center in non-endemic area of Argentina. Am J Trop Med Hyg. 2010 May;82(5):838-45.
-
115González-Tomé MI, Rivera M, Camaño I, Norman F, Flores-Chávez M, Rodríguez-Gómez L, et al. Recomendaciones para el diagnóstico, seguimiento y tratamiento de la embarazada y del niño con enfermedad de Chagas. Enferm Infecc Microbiol Clin. 2013 Oct;31(8):535-42.
-
116Carlier Y, Sosa-Estani S, Luquetti AO, Buekens P. Congenital Chagas disease: an update. Mem Inst Oswaldo Cruz. 2015 May;110(3):363-8.
-
117Russomando G, Almirón M, Candia N, Franco L, Sánchez Z, Guillen I. Implementación y evaluación de un sistema localmente sustentable de diagnóstico prenatal que permite detectar casos de transmisión congénita de la enfermedad de Chagas en zonas endêmicas del Paraguay. Rev Soc Bras Med Trop. 2005;38 Supl 2:49-54.
-
118Brutus L, Castillo H, Bernal C, Salas NA, Schneider D, Santalla JA, et al. Detectable Trypanosoma cruzi parasitemia during pregnancy and delivery as a risk factor for congenital Chagas disease. Am J Trop Med Hyg. 2010 Nov;83(5):1044-7.
-
119Organización Panamericana de la Salud; Organización Mundial de la Salud. Tratamiento etiológico de la enfermidad de Chagas: conclusiones de reunión de especialistas. Rev Patol Trop. 1999;28(2):247-79.
-
120Fabbro DL, Streiger ML, Arias ED, Bizai ML, Barco M, Amicone NA. Trypanocide treatment among adults with chronic Chagas disease living in Santa Fe City (Argentina), over a mean follow-up of 21 years: parasitological, serological and clinical evolution. Rev Soc Bras Med Trop. 2007 Jan-Feb;40(1):1-10.
-
121Lana M, Lopes LA, Martins HR, Bahia MT, Machado-de-Assis GF, Wendling AP, et al. Clinical and laboratory status of patients with chronic Chagas disease living in a vector-controlled area in Minas Gerais, Brazil, before and nine years after aetiological treatment. Mem Inst Oswaldo Cruz. 2009 Dec;104(8):1139-47.
-
122Pérez-Molina JA, Pérez-Ayala A, Moreno S, Fernández-González MC, Zamora J, López-Velez R. Use of benznidazole to treat chronic Chagas' disease: a systematic review with a meta-analysis. J Antimicrob Chemother. 2009 Dec;64(6):1139-47.
-
123CarlierI Y, Torrico F, organizers. Congenital infection with T. cruzi: from mechanisms of transmission to strategies for diagnosis and control. Rev Soc Bras Med Trop . 2003 Nov-Dec;36(6):767-71.
-
124Scapellato PG, Bottaro EG, Rodríguez-Brieschke MT. Mother-child transmission of Chagas disease: could infection with human immunodeficiency virus increase the risk? Rev Soc Bras Med Trop . 2009 Mar-Apr;42(2):107-9.
-
125Bua J, Volta BJ, Velazquez EB, Ruiz AM, Rissio AM, Cardoni RL. Vertical transmission of Trypanosoma cruzi infection: quantification of parasite burden in mothers and their children by parasite DNA amplification. Trans R Soc Trop Med Hyg. 2012 Oct;106(10):623-8.
-
126Mallimaci MC, Sosa-Estani S, Russomando G, Sanchez Z, Sijvarger C, Alvarez IM, et al. Early diagnosis of congenital infection, using shed scute phase antigen, in Ushuaia, Tierra del Fuego, Argentina. Am J Trop Med Hyg. 2010 Jan;82(1):55-9.
-
127Chippaux JP, Clavijo AN, Santalla JA, Postigo JR, Schneider D, Brutus L. Antibody drop in newborns congenitally infected by Trypanosoma cruzi treated with benznidazole. Trop Med Int Health. 2010 Jan;15(1):87-93.
-
128Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Departamento de DST, Aids e Hepatites Virais. Recomendações para profilaxia da transmissão vertical do HIV e terapia antirretroviral em gestantes. Brasília: Ministério da Saúde; 2010. (Série Manuais, 46).
-
129Luquetti AO, Rassi A. Diagnóstico laboratorial da infecção pelo Trypanosoma cruzi . In: Brener Z, Andrade Z, Barral-Netto M, editores. Trypanosoma cruzi e doença de Chagas. Rio de Janeiro: Guanabara-Koogan; 2000. p. 344-78.
-
130Chiari E. Parasitological diagnosis. In: Wendel S, Brener Z, Camargo ME, Rassi A, editores. Chagas disease (American Trypanosomiasis): its impact on transfusion and clinical medicine. São Paulo: Editora Sociedade Brasileira de Hematologia e Hemoterapia; 1992. p. 153-64.
-
131Luquetti AO, Castro AM. Diagnóstico sorológico da doença de Chagas. In: Dias JCP, Coura JR, editores. Clínica e terapêutica da doença de Chagas: uma abordagem prática para o clínico geral. Rio de Janeiro: Editora Fiocruz; 1997. p. 99-113.
-
132Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Programa Nacional de Controle de Chagas. Doença de Chagas aguda: aspectos epidemiológicos, diagnóstico e tratamento: guia de consulta rápida para profissionais de saúde. Rev Patol Trop. 2007 Set-Dez;36(3):1-32 (anexo).
-
133Ministério da Saúde (BR). Recomendações sobre o diagnóstico parasitológico, sorológico e molecular para confirmação da doença de Chagas aguda e crônica. Rev Patol Trop. 2013 out-dez;42(4):475-8.
-
134Luquetti AO, Schmuñis GA. Diagnosis of Trypanosoma cruzi infection. In: Telleria J, Tibayrenc M, editors. American Trypanosomiasis Chagas disease: one hundred years of research. Amsterdam: Elsevier; 2010. p. 743-92.
-
135Camargo ME, Amato Neto V. Anti- Trypanosoma cruzi IgM antibodies as serological evidence of recent infection. Rev Inst Med Trop São Paulo. 1974 Jul-Aug;16(4):200-2.
-
136Chiari E, Dias JC, Lana M, Chiari CA. Hemocultures for the parasitological diagnosis of human chronic Chagas disease. Rev Soc Bras Med Trop. 1989 Jan-Mar;22(1):19-23.
-
137Chiari E, Galvão LMC. Diagnóstico parasitológico da doença de Chagas. In: Dias JCP, Coura JR, editores. Clínica e terapêutica da doença de Chagas: uma abordagem prática para o clínico geral. Rio de Janeiro: Fiocruz; 1997. p. 85-97.
-
138Cerisola JA, Rohwedder R, Segura EL, Del Prado CE, Alvarez M, Martini GJW. El xenodiagnóstico. Buenos Aires: Imp Inst Nac Invest Cardiovasc; 1974.
-
139Castro AM, Luquetti AO, Rassi A, Rassi GG, Chiari E, Galvão LMC. Blood culture and polymerase chain reaction for the diagnosis of the chronic phase of human infection with Trypanosoma cruzi . Parasitol Res. 2002 Oct;88(10):894-900.
-
140Camargo ME. Serological diagnosis: an appraisal of Chagas disease serodiagnosis. In: Wendel S, Brener Z, Camargo ME, Rassi A, editors. Chagas Disease (American Trypanosomiasis), its impact on Transfusion and Clinical Medicine. São Paulo: Sociedade Brasileira de Hematologia e Hemoterapia; 1992. p. 165-78.
-
141Ferreira AW, Ávila SLM. Doença de Chagas. In.: Ferreira AW, Ávila SLM. Diagnóstico laboratorial das principais doenças infecciosas e auto-imunes. 2 ed. Rio de Janeiro: Guanabara Koogan; 2001. p. 241-9.
-
142World Health Organization. Control of Chagas disease. Geneva: World Health Organization ; 2002. (Technical Report Series, 905).
-
143Hijar G, Padilla C, Balbuena J, Bisio MMC, Bailon H, Vega S, et al. Estandarización y validación del uso clínico de la Reacción en Cadena de la Polimerasa para la detección de infección por Trypanosoma cruzi. In: Anales 9º Congreso Peruano de Enfermedades Infecciosas y Tropicales "Jose Neyra Ramirez"; 2009 nov 18-21; Lima. Lima: Sociedad Peruana de Enfermedades Infecciosas y Tropicales; 2009.
-
144Schijman AG, Bisio M, Orellana L, Sued M, Duffy T, Mejia Jaramillo AM, et al. International study to evaluate PCR methods for detection of Trypanosoma cruzi DNA in blood samples from Chagas disease patients. PLoS Negl Trop Dis. 2011 Jan;5(1):e931.
-
145Camargo ME. Fluorescent antibody test for the diagnosis of American trypanosomiasis. Technical modification employing preserved culture forms of Trypanosoma cruzi in a slide test. Rev Inst Med Trop São Paulo. 1966 Sep-Oct;8(5):227-35.
-
146Cerisola JA, Chaben MF, Lazari JO. Test de hemaglutinación para el diagnóstico de la enfermedad de Chagas. Prensa Méd Argent. 1962;49:1761-67.
-
147Voller A, Draper C, Bidwell DE, Bartlett A. Microplate enzyme-linked immunosorbent assay for Chagas disease. Lancet. 1975 Feb;305(7904):426-8.
-
148Castro CN, Alves MT, Macedo VO. Importância da repetição do xenodiagnóstico para avaliação da parasitemia na fase crônica da doença de Chagas. Rev Soc Bras Med Trop. 1983 abr-jun;16(2):98-103.
-
149Junqueira AC, Chiari E, Wincker P. Comparison of the polymerase chain reaction with two classical parasitological methods for diagnosis of Chagas disease patients in a north-eastern endemic region of north-eastern Brazil. Trans R Soc Trop Med Hyg. 1996 Mar-Apr;90(2):129-32.
-
150Britto CC. Usefulness of PCR-based assays to assess drug efficacy in Chagas disease chemotherapy: value and limitations. Mem Inst Oswaldo Cruz. 2009 Jul;104 Suppl 1:122-35.
-
151Silveira JF, Umezawa ES, Luquetti AO. Chagas disease: recombinant Trypanosoma cruzi antigens for serological diagnosis. Trends Parasitol. 2001 Jun;17(6):286-91.
-
152Zicker F, Smith PG, Luquetti AO, Oliveira OS. Mass screening for Trypanosoma cruzi infections using the immunofluorescence, ELISA and haemagglutination tests on serum samples and on blood eluates from filter-paper. Bull World Health Organ. 1990;68(4):465-71.
-
153Sánchez-Camargo CL, Albajar-Viñas P, Wilkins PP, Nieto J, Leiby DA, Paris L, et al. Comparative evaluation of 11 commercialized rapid diagnostic tests for detecting Trypanosoma cruzi antibodies in serum banks in areas of endemicity and nonendemicity. J Clin Microbiol. 2014 Jul;52(7):2506-12.
-
154Pinto AYN, Valente SAS, Valente VC. Emerging acute Chagas disease in Amazonian Brazil: case reports with serious cardiac involvement. Braz J Infect Dis. 2004 Dec;8(6):454-60.
-
155Dias JP, Bastos C, Araújo E, Mascarenhas AV, Martins Netto E, Grassi F, et al. Acute Chagas disease outbreak associated with oral transmission. Rev Soc Bras Med Trop. 2008 May-Jun;41(3):296-300.
-
156Pinto AY, Valente VC, Coura JR, Valente SAS, Junqueira AC, Santos LC, et al. Clinical follow-up of responses to treatment with benznidazol in Amazon: a cohort study of acute Chagas disease. Plos One. 2013 May;8(5):e64450.
-
157Centers for Disease Control and Prevention. Chagas disease after organ transplantation: United States, 2001. MMWR Morb Mortal Wkly Rep. 2002 Mar;51(10):210-2.
-
158Centers for Disease Control and Prevention. Chagas disease after organ transplantation: Los Angeles, California, 2006. MMWR Morb Mortal Wkly Rep. 2006 Jul;55(29):798-800.
-
159Rocha A, Ramos Júnior AN, Sartori AM, Correia D, Gontijo ED, Tatto E, et al. Recomendações para diagnóstico, tratamento e acompanhamento da co-infecção Trypanosoma cruzi: vírus da imunodeficiência humana. Rev Soc Bras Med Trop. 2006 jul-ago;39(4):392-415.
-
160Rassi A, Rassi Júnior A, Rassi GG. Fase aguda. In: Brener Z, Andrade Z, Barral-Netto M (orgs). Trypanosoma cruzi e Doença de Chagas. 2 ed. Rio de Janeiro: Guanabara Koogan; 2000. p.231-45.
-
161Romaña C. Acerca de un síntoma inicial de valor para el diagnóstico de la forma aguda de la enfermedad de Chagas: la conjuntivitis schizotripanosómica unilateral: hipótesis sobre la puerta de entrada conjuntival de la enfermedad. Mision de Estudios de Patologia Regional Argentina (MEPRA)1935;22:16-25.
-
162Lugones HS. Enfermedad de Chagas. Diagnóstico de su faz aguda. Santiago del Estero (Argentina): Universidad Católica de Santiago del Estero; 2001.
-
163Ferreira HO, Pucci H. Estudo evolutivo da doença de Chagas com fase aguda conhecida. Rev Soc Bras Med Trop . 1982 jan-dez;15(1):125-30.
-
164Ferreira HO, Miziara JL. Aspectos clínicos da cardiopatia chagásica aguda. In: Cançado JR, Chuster M (editores). Cardiopatia Chagásica. Belo Horizonte: Imprensa Oficial; 1985.
-
165Parada H, Carrasco HA, Añez N, Fuenmayor C, Inglessis I. Cardiac involvement is a constant finding in acute Chagas' disease: a clinical, parasitological and histopathological study. Int J Cardiol. 1997 Jun;60(1):49-54.
-
166Wendell S. Doença de Chagas transfusional. In: Dias JCP, Coura JR, editores. Clínica e terapêutica da doença de Chagas: uma abordagem prática para o clínico geral. Rio de Janeiro: Fiocruz ; 1997. p. 411-27.
-
167Noya BA, Diaz- Bello Z, Colmenares C, Ruiz-Guevara R, Mauriello L, Zavala-Jaspe R, et al. Large urban outbreak of orally acquired acute Chagas disease at a school in Caracas, Venezuela. J Infect Dis. 2010 May;201(9):1308-15.
-
168Shaw J, Lainson R, Fraiha H. Considerações sobre a epidemiologia dos primeiros casos autóctones de doença de Chagas registrados em Belém, Pará, Brasil. Rev Saude Publica. 1969;3(2):153-7.
-
169Pinto AYN, Ferreira Júnior AG, Valente VC, Harada GS, Valente SAS. Urban outbreak of acute Chagas disease in Amazon region of Brazil: four-year follow-up after treatment with benznidazole. Rev Panam Salud Publica. 2009 Jan;25(1):77-83.
-
170Shikanai-Yasuda MA, Marcondes CB, Guedes LA, Siqueira GS, Barone AA, Dias JC, et al. Possible oral transmission of acute Chagas disease in Brasil. Rev Inst Med Trop Sao Paulo. 1991 Sep-Oct;33(5):351-7.
-
171Jörg ME, Freire RS. Lipochagomata genii or lipochagomata of the cheeks, characteristic lesions of acute trypanosomiasis cruzi in children under three years of age. Acta Trop. 1961;18:318-50.
-
172Medeiros MB, Guerra JAO, Lacerda MVG. Meningoencephalitis in a patient with acute Chagas disease in the Brazilian Amazon. Rev Soc Bras Med Trop. 2008 Sep-Oct; 41(5):520-1.
-
173Freilij H, Altcheh J, Muchinik G. Perinatal human immunodeficiency virus infection and congenital Chagas' disease. Pediatr Infect Dis J. 1995 Feb;14(2):161-2.
-
174Nisida IV, Amato Neto V, Braz LM, Duarte MI, Umezawa ES. A survey of congenital Chagas' disease, carried out at three health institutions in São Paulo City, Brazil. Rev Inst Med Trop Sao Paulo. 1999 Sep-Oct;41(5):305-11.
-
175Sartori AM, Ibrahim KY, Nunes Westphalen EV, Braz LM, Oliveira Junior OC, Gakiya E, et al. Manifestations of Chagas disease (American trypanosomiasis ) in patients with HIV/AIDS. Ann Trop Med Parasitol. 2007 Jan;101(1):31-50.
-
176França LCM, Lemos S, Fleury RN, Melaragno Filho R, Ramos Júnior HA, Pasternak J. Moléstia de Chagas crônica associada à leucemia linfática: ocorrência de encefalite aguda como alteração do estado imunitário. Arq Neuro-Psiquiatr. 1969 mar;27(1):59-66.
-
177Spina-França A, Livramento JA, Machado LR, Yasuda N. Anticorpos a Trypanosoma cruzi no líquido cefalorraqueano: pesquisa pelas reações de fixação do complemento e de imunofluorescência. Arq Neuro-Psiquiatr. 1988 dez;46(4):374-8.
-
178Livramento JA, Machado LR, Spina-França A. Anormalidades do líquido cefalorraqueano em 170 casos de AIDS. Arq Neuro-Psiquiatr. 1989 set;47(3):326-31.
-
179Del Castilho M, Mendoza G, Oviedo J, Perez Bianco PR, Anselmo AE, Silva M. AIDS and Chagas' disease with central nervous system tumor-like lesion. Am J Med. 1990 Jun;88(6):693-4.
-
180Ferreira MS, Nishioka SA, Rocha A, Silva AM, Ferreira RG, Olivier W, et al. Acute fatal Trypanosoma cruzi meningoencephalitis in a human immunodeficiency virus-positive hemophiliac patient. Am J Trop Med Hyg. 1991 Dec;45(6):723-7.
-
181Riarte A, Luna C, Sabatiello R, Sinagra A, Schiavelli R, De Rissio A, et al. Chagas' disease in patients with kidney transplants: 7 years of experience 1989-1996. Clin Infect Dis. 1999 Sep;29(3):561-7.
-
182Campos SV, Strabelli TM, Amato Neto V, Silva CP, Bacal F, Bocchi EA, et al. Risk factors for Chagas' disease reactivation after heart transplantation. J Heart Lung Transplant. 2008 Jun;27(6):597-602.
-
183Ministério da Saúde (BR). Portaria nº 2.600, de 21 de outubro de 2009. Aprova o Regulamento Técnico do Sistema Nacional de Transplantes. Diário Oficial da República Federativa do Brasil, Brasília (DF), 2009 out 21;Seção 1:77.
-
184Altclas J, Sinagra A, Dictar M, Luna C, Verón MT, De Rissio AM, et al. Chagas disease in bone marrow transplantation: an approach to preemptive therapy. Bone Marrow Transplant. 2005 Jul;36(2):123-9.
-
185Barcán L, Luna C, Clara L, Sinagra A, Valledor A, De Rissio AM, et al. Transmission of T. cruzi infection via liver transplantation to a nonreactive recipient for Chagas' disease. Liver Transpl. 2005 Sep;11(9):1112-6.
-
186Rassi A, Amato Neto V, Siqueira AF, Ferriolli Filho F, Amato VS, Rassi Júnior A. Protective effect of benznidazole against parasite reactivation in patients chronically infected with Trypanosoma cruzi and treated with corticoids for associated diseases. Rev Soc Bras Med Trop. 1999 Sep-Oct;32(5):475-82.
-
187Cordova E, Boschi A, Ambrosioni J, Cudos C, Corti M. Reactivation of Chagas disease with central nervous system involvement in HIV-infected patients in Argentina, 1992-2007. Int J Infect Dis. 2008 Nov;12(6):587-92.
-
188Validade do conceito de forma indeterminada de doença de Chagas. Rev Soc Bras Med Trop. 1985 jan-mar;18(1):46.
-
189Dias JC. The indeterminate form of human chronic Chagas' disease: a clinical epidemiological review. Rev Soc Bras Med Trop. 1989 Jul-Sep;22(3):147-56.
-
190Macêdo VO. Forma indeterminada da doença de Chagas. In: Dias JCP, Coura JR, editores. Clínica e Terapêutica da doença de Chagas: uma abordagem prática para o clínico geral. Rio de Janeiro: Editora Fiocruz ; 1997. p.135-51.
-
191Macêdo VO. Indeterminate form of Chagas disease. Mem Inst Oswaldo Cruz. 1999 Sep;94 Suppl 1:311-6.
-
192Ianni BM, Arteaga E, Frimm CC, Pereira Barretto AC, Mady C. Chagas' heart disease: evolutive evaluation of electrocardiographic and echocardiographic parameters in patients with the indeterminate form. Arq Bras Cardiol. 2001 Jul;77(1):59-62.
-
193Dias JCP, Machado EMM, Borges ÉC, Moreira EF, Gontijo C, Azeredo BVM. Doença de Chagas em Lassance, MG. Reavaliação clínico-epidemiológica 90 anos após a descoberta de Carlos Chagas. Rev Soc Bras Med Trop. 2002 mar-apr;35(2):167-76.
-
194Almeida EA, Barbosa Neto RM, Guariento ME, Wanderley JS, Souza ML. Apresentação clínica da doença de Chagas crônica em indivíduos idosos. Rev Soc Bras Med Trop. 2007 mai-jun;40(3):311-5.
-
195Villela E. Moléstia de Chagas: descrição clínica, 5ª parte. Folha Médica. 1923; 4:65-6.
-
196Dias E, Laranja FS, Miranda A, Nóbrega G. Chagas' disease: a clinical, epidemiologic and pathologic study. Circulation. 1956 Dec;14(6):1035-60.
-
197Prata A, Macedo V. Morbidity of Chagas' heart disease. Mem Inst Oswaldo Cruz. 1984;79 Suppl:93-6.
-
198Organización Mundial de la Salud; Organización Panamericana de la Salud. Aspectos clínicos de la enfermedad de Chagas. Informe de una reunión conjunta OMS/OPAS de investigadores. Bol Oficina Sanit Panam. 1974;76:141-58.
-
199Conselho Nacional de Pesquisa. Epidemiologia da doença de Chagas: objetivos e metodologia dos estudos longitudinais. Rio de Janeiro: Conselho Nacional de Pesquisa; 1974. 46 p. Relatório Técnico nº 1.
-
200Oliveira Júnior W. Forma indeterminada da doença de Chagas: implicações médico-trabalhistas. Arq Bras Cardiol. 1990 fev;54(2):89-91.
-
201Guariento ME, Camilo MVF, Camargo AMA. Working conditions of Chagas' disease patients in a large Brazilian city. Cad Saude Publica. 1999 Apr-Jun;15(2):381-6.
-
202Dias JCP, Siqueira IOC, Dias RB. Doença de Chagas: diagnóstico e manejo na atenção primária à saúde. In: Gusso G, Lopes JMC. Tratado de Medicina de Família e Comunidade: 2 Volumes: Princípios, Formação e Prática. Porto Alegre: Artmed; 2012. p. 2067-80.
-
203Castro I, Andrade JP, Paola AAV, Vilas-Boas F, Oliveira GMM, Marin-Neto JA, et al. Sociedade Brasileira de Cardiologia. I Diretriz Latino-Americana para o diagnóstico e tratamento da cardiopatia chagásica. Arq Bras Cardiol. 2011 Aug;97(2 Suppl 3):1-48.
-
204Ozaki Y, Guariento ME, Almeida EA. Quality of life and depressive symptoms in Chagas disease patients. Qual Life Res. 2011 Feb;20(1):133-8.
-
205Higuchi ML, Benvenuti LA, Reis MR, Metzger M. Pathophysiology of the heart in Chagas' disease: current status and new developments. Cardiovasc Res. 2003 Oct;60(1):96-107.
-
206Higuchi ML. Chagas disease: importance of the parasite in the pathogenesis of the cardiac chronic disease. Arq Bras Cardiol. 1995 Mar;64(3):251-4.
-
207Higuchi ML. Chronic chagasic cardiopathy: the product of a turbulent host-parasite relationship. Rev Inst Med Trop Sao Paulo. 1997 Jan-Feb;39(1):53-60.
-
208Marin-Neto JA, Cunha-Neto E, Maciel BC, Simões MV. Pathogenesis of chronic Chagas heart disease. Circulation. 2007 Mar;115(9):1109-23.
-
209Chagas C, Villela E. Forma cardíaca da Trypanosomíase americana. Mem Inst Oswaldo Cruz. 1922;14(1):5-61.
-
210Coura JR, Abreu LL, Pereira JB, Willcox HP. Morbidity in Chagas' disease. IV. longitudinal study of 10 years in Pains and Iguatama, Minas Gerais, Brazil. Mem Inst Oswaldo Cruz. 1985 Jan-Mar;80(1):73-80.
-
211Pereira Barretto AC, Mady C, Arteaga-Fernandez E, Stolf N, Lopes EA, Higuchi ML, et al. Right ventricular endomyocardial biopsy in chronic Chagas' disease. Am Heart J. 1986 Feb;111(2):307-12.
-
212Prata A, Lopes ER, Chapadeiro E. Characteristics of unexpected sudden death in Chagas disease. Rev Soc Bras Med Trop. 1986 Jan-Mar;19(1):9-12.
-
213Dias JC, Kloetzel K. The prognostic value of the electrocardiographic features of chronic Chagas' disease. Rev Inst Med Trop Sao Paulo. 1968 May-Jun;10(3):158-62.
-
214Bestetti RB, Muccillo G. Clinical course of Chagas' heart disease: a comparison with dilated cardiomyopathy. Int J Cardiol. 1997 Jul;60(2):187-93.
-
215Pinto AY, Harada GS, Valente VD, Abud JE, Gomes FD, Souza GC, et al. Cardiac attacks in patients with acute Chagas disease in a family micro-outbreak, in Abaetetuba, Brazilian Amazon. Rev Soc Bras Med Trop. 2001 Sep-Oct;34(5):413-9.
-
216Souza DSM, Almeida AJB, Costa FA, Costa EG, Figueiredo MTS, Póvoa RMS. O eletrocardiograma na fase aguda da Doença de Chagas por transmissão oral. Rev Bras Cardiol. 2013 mar-abr;26(2):127-30.
-
217Nishioka SA, Ferreira MS, Rocha A, Burgarelli MK, Silva AM, Duarte MI, et al. Reactivation of Chagas' disease successfully treated with benznidazole in a patient with acquired immunodeficiency syndrome. Mem Inst Oswaldo Cruz. 1993 Jul-Sep;88(3):493-6
-
218Almeida EA, Silva EL, Guariento ME, Souza ML, Aoki FH, Pedro RJ. Fatal evolution of Chagas' disease/Aids co-infection: diagnostic difficulties between myocarditis reactivation and chronic chagasic myocardiopathy. Rev Soc Bras Med Trop. 2009 Mar-Apr;42(2):199-202.
-
219Perez-Ramirez L, Barnabé C, Sartori AM, Ferreira MS, Tolezano JE, Nunes EV, et al. Clinical analysis and parasite genetic diversity in human immunodeficiency virus/Chagas' disease coinfections in Brazil. Am J Trop Med Hyg. 1999 Aug;61(2):198-206.
-
220Bestetti RB, Dalbo CM, Freitas OC, Teno LA, Castilho OT, Oliveira JS. Noninvasive predictors of mortality for patients with Chagas' heart disease: a multivariate stepwise logistic regression study. Cardiology. 1994;84(4-5):261-7.
-
221Mady C, Cardoso RH, Barretto AC, Luz PL, Bellotti G, Pileggi F. Survival and predictors of survival in patients with congestive heart failure due to Chagas' cardiomyopathy. Circulation. 1994 Dec;90(6):3098-102.
-
222Rassi Júnior A, Rassi A, Little WC, Xavier SS, Rassi SG, Rassi AG, et al. Development and validation of a risk score for predicting mortality in Chagas' heart disease. N Engl J Med. 2006 Aug 24;355(8):799-808.
-
223Xavier SS, Sousa AS, Hasslocher-Moreno A. Application of the new classification of cardiac insufficiency (ACC/AHA) in chronic Chagas cardiopathy: a critical analysis of the survival curves. Rev SOCERJ. 2005 mai-jun;18(3):227-32.
-
224Rodriguez-Salas LA, Klein E, Acquatella H, Catalioti F, Davalos V, Gomez-Mancebo JR, et al. Echocardiographic and clinical predictors of mortality in chronic Chagas' disease. Echocardiography. 1998 Apr;15(3):271-278.
-
225Viotti RJ, Vigliano C, Laucella S, Lococo B, Petti M, Bertocchi G, et al. Value of echocardiography for diagnosis and prognosis of chronic Chagas' disease cardiomyopathy without heart failure. Heart. 2004 Jun;90(6):655-60.
-
226Acquatella H, Schiller NB, Puigbó JJ, Giordano H, Suárez JA, Casal H, et al. M-mode and two-dimensional echocardiography in chronic Chagas' heart disease: a clinical and pathologic study. Circulation. 1980 Oct;62(4):787-99.
-
227Dias JCP. História natural da cardiopatia chagásica. In: Cançado JR, Chuster M. Cardiopatia chagásica. Belo Horizonte: Fundação Carlos Chagas; 1985. p. 99-110.
-
228Carrasco HA, Parada H, Guerrero L, Duque M, Durán D, Molina C. Prognostic implications of clinical, electrocardiographic and hemodynamic findings in chronic Chagas' disease. Int J Cardiol. 1994 Jan;43(1):27-38.
-
229Garzon SA, Lorga AM, Nicolau JC. Electrocardiography in Chagas' heart disease. Sao Paulo Med J. 1995 Mar-Apr;113(2):802-13.
-
230Marin-Neto JA, Simões MV, Sarabanda AV. Chagas' heart disease. Arq Bras Cardiol. 1999;72(3):247-80.
-
231Almeida-Filho OC, Maciel BC, Schmidt A, Pazin-Filho A, Marin-Neto JA. Minor segmental dyssynergy reflects extensive myocardial damage and global left ventricle dysfunction in chronic Chagas disease. J Am Soc Echocardiogr. 2002 Jun;15(6):610-6.
-
232Pazin-Filho A, Romano MM, Gomes Furtado R, Almeida Filho OC, Schmidt A, Marin-Neto JA, et al. Left ventricular global performance and diastolic function in indeterminate and cardiac forms of Chagas' disease. J Am Soc Echocardiogr. 2007 Dec;20(12):1338-43.
-
233Rassi A Júnior, Rassi A, Rassi SG. Predictors of mortality in chronic Chagas disease: a systematic review of observational studies. Circulation . 2007 Mar;115(9):1101-8
-
234Xavier SS, Sousa AS, Brasil PEAA, Gabriel FG, Holanda MT, Hasslocher-Moreno A, et al. Incidência e preditores de morte súbita na cardiopatia chagásica crônica com função sistólica preservada. Rev SOCERJ. 2005 set-out;18(5):457-63.
-
235Cunha GP. Síndromes clínicas na cardiomiopatia chagásica crônica. Hospital. 1960;57:71-95.
-
236Rassi Jr A, Marin-Neto JA. Estado da Arte. Cardiopatia chagásica crônica. Rev Soc Cardiol Est São Paulo. 2000;10(4):6-12.
-
237Rassi Júnior A, Rassi AG, Rassi SG, Rassi Júnior L, Rassi A. Arritmias ventriculares na doença de Chagas. Particularidades diagnósticas, prognósticas e terapêuticas. Arq Bras Cardiol. 1995 out;65(4):377-87.
-
238Rassi Júnior A, Rassi SG, Rassi A. Morte súbita na Doença de Chagas. Arq Bras Cardiol. 2001 jan;76(1):75-85.
-
239Rassi Júnior A, Rassi AG, Rassi SG, Rassi Júnior L, Las Casas AA, Rassi A. Arritmia ventricular na insuficiência cardíaca de etiologia chagásica e não chagásica. Arq Bras Cardiol. 1993;61 supl II:11-9.
-
240Scanavacca M, Sosa E. Electrophysiologic study in chronic Chagas' heart disease. Sao Paulo Med J. 1995 Mar-Apr;113(2):841-50.
-
241Chiale PA, Halpern MS, Nau GJ, Tambussi AM, Przybylski J, Lázzari JO, et al. Efficacy of amiodarone during long-term treatment of malignant ventricular arrhythmias in patients with chronic chagasic myocarditis. Am Heart J. 1984 Apr;107(4):656-65.
-
242Silva CP, Bacal F, Pires PV, Drager LF, Souza GEC, Fajardo GM, et al. Importance of amiodarone pulmonary toxicity in the differential diagnosis of a patient with dyspnea awaiting a heart transplant. Arq Bras Cardiol. 2006 Sep;86(3):e4-7.
-
243Zimerman LI, Fenelon G, Martinelli Filho M, Grupi C, Atié J, Lorga Filho A, et al; Sociedade Brasileira de Cardiologia. Diretrizes brasileiras de fibrilação atrial. Arq Bras Cardiol. 2009;92(6 supl 1):1-39.
-
244Martinelli Filho M, Zimerman LI, Lorga AM, Vasconcelos JTM, Rassi Júnior A; Sociedade Brasileira de Cardiologia. Diretrizes brasileiras de dispositivos eletrônicos implantáveis (DCEI). Arq Bras Cardiol. 2007; 89(6):e210-e238.
-
245Baroldi G, Oliveira SJ, Silver MD. Sudden and unexpected death in clinically 'silent' Chagas' disease. A hypothesis. Int J Cardiol. 1997 Feb;58(3):263-8.
-
246Marin-Neto JA, Simões MV, Maciel BC. Aspectos peculiares da fisiopatologia da insuficiência cardíaca na doença de Chagas. Rev Soc Cardiol Est São Paulo. 1998;8(2):243-52.
-
247Simões MV, Almeida Filho OC, Pazin Filho A, Castro RBP, Schmidt A, Maciel BC, et al. Insuficiência cardíaca na doença de Chagas. Rev Soc Cardiol Est São Paulo. 2000 Jan-Fev;10(1):50-64.
-
248Bocchi EA, Marcondes-Braga FG, Bacal F, Ferraz AS, Albuquerque D, Rodrigues D, et al; Sociedade Brasileira de Cardiologia. Atualização da Diretriz Brasileira de Insuficiência Cardíaca Crônica: 2012. Arq Bras Cardiol. 2012 jan;98(1 supl 1):1-33.
-
249Rocha MOC, Ribeiro AL, Teixeira MM. Clinical management of chronic Chagas cardiomyopathy. Front Biosci. 2003 Jan;8:e44-54.
-
250Marin-Neto JA, Rassi A Júnior, Morillo CA, Avezum A, Connolly SJ, Sosa-Estani S, et al; BENEFIT Investigators. BENEFIT Investigators. Rationale and design of a randomized placebo-controlled trial assessing the effects of etiologic treatment in Chagas' cardiomyopathy: the BENznidazole Evaluation For Interrupting Trypanosomiasis (BENEFIT). Am Heart J. 2008 Jul;156(1):37-43.
-
251Morillo CA, Marin-Neto JA, Avezum A, Sosa-Estani S, Rassi Júnior A, Rosas F, et al; BENEFIT Investigators. Randomized Trial of Benznidazole for Chronic Chagas' Cardiomyopathy. N Engl J Med. 2015 Oct;373(14):1295-306.
-
252Viotti R, Vigliano C, Lococo B, Bertocchi G, Petti M, Alvarez MG, et al. Long-term cardiac outcomes of treating chronic Chagas disease with benznidazole versus no treatment: a nonrandomized trial. Ann Intern Med. 2006 May;144(10):724-34.
-
253Viotti R, Vigliano C, Lococo B, Petti M, Bertocchi G, Alvarez MG, et al. Clinical predictors of chronic chagasic myocarditis progression. Rev Esp Cardiol. 2005 Sep;58(9):1037-44.
-
254Marin-Neto JA, Rassi Júnior A, Sousa AS, Dias JCP, Rassi A. Doença de Chagas: moléstia negligenciada. In: Andrade JP, Arnett DK, Pinto FJ (editores). Tratado de Prevenção Cardiovascular. Um Desafio Global. São Paulo: Atheneu; 2014. p. 117-27.
-
255Montera MW, Pereira SB, Colafranceschi AS, Almeida DR, Tinoco EM, Rocha RM, et al. Sumário de Atualização da II Diretriz Brasileira de Insuficiência Cardíaca Aguda: 2009/2011. Arq Bras Cardiol. 2012;98(5):375-83.
-
256Roberti RR, Martinez EE, Andrade JL, Araujo VL, Brito FS, Portugal OP, et al. Chagas cardiomyopathy and captopril. Eur Heart J. 1992 Jul;13(7):966-70.
-
257Davila DF, Angel F, Arata de Bellabarba G, Donis JH. Effects of metoprolol in chagasic patients with severe congestive heart failure. Int J Cardiol. 2002 Oct;85(2-3):255-60.
-
258Freitas H, Salaltino G, Chizzola P, Costa J, Mansur A, Ramires JF, et al. Betablockers for Chagas' heart disease. J Am Coll Cardiol. 1999;33 Suppl A:184.
-
259Bocchi EA, Bellotti G, Uip D, Kalil J, Lourdes Higuchi M, Fiorelli A, et al. Long-term follow-up after heart transplantation in Chagas' disease. Transplant Proc. 1993 Feb;25(1 Pt 2):1329-30.
-
260Carvalho VB, Sousa EF, Vila JH, Silva JP, Caiado MR, Araujo SR, et al. Heart transplantation in Chagas' disease. 10 Years after the initial experience. Circulation. 1996 Oct;94(8):1815-7.
-
261Ribeiro dos Santos R, Rassi S, Feitosa G, Grecco OT, Rassi Júnior A, Cunha AB, et al; Chagas Arm of the MiHeart Study Investigators. Cell therapy in Chagas cardiomyopathy (Chagas arm of the multicenter randomized trial of cell therapy in cardiopathies study): a multicenter randomized trial. Circulation. 2012 May;125(20):2454-61.
-
262Samuel J, Oliveira M, Correa De Araujo RR, Navarro MA, Muccillo G. Cardiac thrombosis and thromboembolism in chronic Chagas' heart disease. Am J Cardiol. 1983 Jul;52(1):147-51.
-
263Lopes ER, Marquez JO, Costa Neto B, Menezes AAC, Chapadeiro E. Associação entre acidentes vasculares encefálicos e doença de Chagas. Rev Soc Bras Med Trop. 1991 Apr-Jun;24(2):101-4.
-
264Sousa AS, Xavier SS, Freitas GR, Hasslocher-Moreno A. Prevention strategies of cardioembolic ischemic stroke in Chagas' disease. Arq Bras Cardiol. 2008 Nov;91(5):306-10.
-
265Oliveira Júnior W. O cardiopata chagásico em situações especiais. In: Pinto JC, Rodrigues J (Orgs). Clínica e terapêutica da doença de Chagas. 2 ed. Rio de Janeiro: Fiocruz; 1997. p. 293-322.
-
266Bestetti RB. Avaliação do risco cirúrgico em pacientes portadores de cardiopatia chagásica crônica em cirurgias não cardíacas. In: Pinto JC, Rodrigues J (Orgs). Clínica e terapêutica da doença de Chagas. 2 ed. Rio de Janeiro: Fiocruz; 1997. p. 281-91.
-
267Ramos G, Ramos Filho J, Rassi Júnior A, Pereira E, Gabriel Neto S, Chaves E. Marcapasso cardíaco artificial: considerações pré e per-operatórias. Rev Bras Anestesiol. 2003 nov-dez;53(6):854-62.
-
268Prata A. Implicações epidemiológicas e sócio-econômicas da doença de Chagas. Brasilia Med. 1973;9:69-71.
-
269Porto CC, Rassi A, Faria GHDC. Aspectos sócio-econômicos e médico-trabalhistas. In: Cançado JR, Chuster M (editores). Cardiopatia Chagásica. Belo Horizonte: Fundação Carlos Chagas; 1985. p. 362-6.
-
270Dias JCP. Aspectos clínicos, sociais e trabalhistas da doença de Chagas em área endêmica sob controle do estado de Minas Gerais, Brasil. Rev Soc Bras Med Trop. 1999 apr-jun;26(2):93-9.
-
271Morais BB, Martins P, Maiolini DD, Silva AL, Melo JRC. Concomitant lesions in Chagas' disease. Eur J Gastroenterol Hepatol. 1991;3(7):523-6.
-
272Fonseca FM, Etchebehere RM, Queiroz DM, Rocha AM, Junqueira IS, Fonseca DN, et al. Histological and endoscopic features of the stomachs of patients with Chagas disease in the era of Helicobacter pylori . Rev Soc Bras Med Trop. 2014 Nov-Dec;47(6):739-46.
-
273Rezende JM. Classificação radiológica do megaesôfago. Rev Goiana Med. 1982 jul-dez;28(3/4):187-91.
-
274Brücher BL, Stein HJ, Bartels H, Feussner H, Siewert JR. Achalasia and esophageal cancer: incidence, prevalence, and prognosis. World J Surg. 2001 Jun;25(6):745-9.
-
275Leeuwenburgh I, Scholten P, Alderliesten J, Tilanus HW, Looman CW, Steijerberg EW, et al. Long-term esophageal cancer risk in patients with primary achalasia: a prospective study. Am J Gastroenterol. 2010 Oct;105(10):2144-9.
-
276Pérez-Ayala A, Pérez-Molina JA, Norman F, Monge-Maillo B, Faro MV, López-Vélez R. Gastro-intestinal Chagas disease in migrants to Spain: prevalence and methods for early diagnosis. Ann Trop Med Parasitol. 2011 Jan;105(1):25-9.
-
277Oliveira RB, Troncon LEA, Dantas RO, Meneghelli UG. Gastrointestinal manifestations of Chagas disease. Am J Gastroenterol. 1998 Jun;93(6):884-9.
-
278Meier-Ruge WA, Müller-Lobeck H, Stoss F, Bruder E. The pathogenesis of idiopathic megacolon. Eur J Gastroenterol Hepatol. 2006 Nov;18(11):1209-15.
-
279Pasricha PJ, Rai R, Ravich WJ, Hendrix TR, Kalloo AN. Botulinum toxin for achalasia: long-term outcome and predictors of response. Gastroenterology. 1996 May;110(5):1410-5.
-
280Ferguson MK. Achalasia: current evaluation and therapy. Ann Thorac Surg. 1991 Aug;52(2):336-42.
-
281Heller E. Extramukose Cardiaplastik beim chronischen Cardiospasmus mit Dilatation des Oesophagus. Mitt Grenzgeb Med Chir. 1913;27(1):141-49.
-
282Pinotti HW, Habr-Gama A, Cecconello I, Felix VN, Zilbertein B. The surgical treatment of megaesophagus and megacolon. Dig Dis. 1993 Jul-Oct;11(4-5):206-15.
-
283Spiess AE, Kahrilas PJ. Treating achalasia: from whalebone to laparoscope. JAMA. 1998 Aug;280(7):638-42.
-
284NOSCAR POEM White Paper Committee; Stavropoulos SN, Desilets DJ, Fuchs KH, Gostout CJ, Haber G, Inoue H, et al. Per-oral endoscopic myotomy white paper summary. Gastrointest Endosc. 2014 Jul;80(1):1-15.
-
285Inoue H, Tianle KM, Ikeda H, Hosoya T, Onimaru M, Yoshida A, et al. Peroral endoscopic myotomy for esophageal achalasia: technique, indication, and outcomes. Thorac Surg Clin. 2011 Nov;21(4):519-25.
-
286Swanstrom LL, Kurian A, Dunst CM, Sharata A, Bhayani N, Rieder E. Long-term outcomes of an endoscopic myotomy for achalasia: the POEM procedure. Ann Surg. 2012 Oct;256(4):659-67.
-
287Duhamel B. Une nouvelle opération pour le mégacolon cologénital: l'abaissement rétro-rectal et transanal du côlon et son application possible au traitement de quelques autres malformations. Nouv Presse Med. 1956; 64(95):2249-50.
-
288Reis-Neto JA, Pedroso MA, Lupinacci RA, Reis Júnior JA, Ciquini SA, Lupinacci RM, et al. Megacolo adquirido: perspectivas fisiopatológicas para o tratamento laparoscópico. Rev Bras Colo-proctol. 2004 jan-mar;24(1):49-62.
-
289Coura JR, Borges-Pereira J. Chronic phase of Chagas disease: why should it be treated? A comprehensive review. Mem Inst Oswaldo Cruz. 2011 Sep;106(6):641-5.
-
290Molina I, Gómez i Prat J, Salvador F, Treviño B, Sulleiro E, Serre N, et al. Randomized trial of posaconazole and benznidazole for chronic Chagas' disease. N Engl J Med. 2014 May;370:1899-908.
-
291Cançado JR. Terapêutica específica. In: Dias JCP, Coura JR, editores. Clínica e Terapêutica da Doença de Chagas: uma abordagem prática para o clínico geral. Rio de Janeiro: Fiocruz; 1997. p. 323-51.
-
292Sosa Estani S, Segura EL, Ruiz AM, Velazquez E, Porcel BM, Yampotis C. Efficacy of chemotherapy with benznidazole in children in the indeterminate phase of Chagas' disease. Am J Trop Med Hyg. 1988 Oct;59(4):526-9.
-
293Andrade AL, Zicker F, Oliveira RM, Silva SA, Luquetti A, Travassos LR, et al. Randomised trial of efficacy of benznidazole in treatment of early Trypanosoma cruzi infection. Lancet. 1996 Nov;348(9039):1407-13.
-
294Yun O, Lima MA, Ellman T, Chambi W, Castillo S, Flevaud L, et al. Feasibility, drug safety, and effectiveness of etiological treatment programs for Chagas disease in Honduras, Guatemala, and Bolivia: 10-year experience of Médecins Sans Frontières. PLoS Negl Trop Dis. 2009 Jul;3(7):e488.
-
295Bern C, Montgomery SP, Herwaldt BL, Rassi Júnior A, Marin-Neto JA, Dantas RO, et al. Evaluation and treatment of Chagas disease in the United States: a systematic review. JAMA. 2007 Nov;298(18):2171-81.
-
296Coura JR, Abreu LL, Willcox HP, Petana W. Comparative controlled study on the use of benznidazole, nifurtimox and placebo, in the chronic form of Chagas' disease, in a field area with interrupted transmission. I. Preliminary evaluation. Rev Soc Bras Med Trop. 1997 Mar-Apr;30(2):139-44.
-
297Bitto C, Silveira C, Cardoso MA, Marques P, Luquetti A, Macedo V, et al. Parasite persistence in treated chagasic patients revealed by xenodiagnosis and polymerase chain reaction. Mem Inst Oswaldo Cruz. 2001 Aug;96(6):823-6.
-
298Gallerano RH, Sosa RR. Resultados de un estúdio a largo plazo con drogas antiparasitárias en infectados chagásicos crónicos. Rev Fed Arg Cardiol. 2001;30:289-96.
-
299Fragata Filho AA. Tratamento antiparasitário da doença de Chagas. Rev Soc Cardiol Estado de São Paulo. 2009;19:2-5.
-
300Andrade SG, Magalhães JB, Pontes AL. Terapêutica da fase crônica da infecção experimental pelo Trypanosoma cruzi com o benzonidazol e o nifurtimox. Rev Soc Bras Med Trop. 1989 Jul-Sep;22(3):113-8.
-
301Garcia S, Ramos CO, Senra JFV, Vilas-Boas F, Rodrigues MM, Campos-de-Carvalho AC, et al. Treatment with benznidazole during the chronic phase of experimental Chagas' disease decreases cardiac alterations. Antimicrob Agents Chemother. 2005 Apr;49(4):1521-8.
-
302Coura JR, Castro SL. A critical review on Chagas disease chemotherapy. Mem Inst Oswaldo Cruz. 2002 Jan;97(1):3-24.
-
303Maguire JH. Treatment of Chagas' disease: time is running out. N Engl J Med. 2015 Oct;373(14):1369-70.
-
304D'Albuquerque LA, Gonzalez AM, Filho HL, Copstein JL, Larrea FI, Mansero JM, et al. Liver transplantation from deceased donors serologically positive for Chagas disease. Am J Transplant. 2007 Mar;7(3):680-4.
-
305Altclas JD, Barcan L, Nagel C, Lattes R, Riarte A. Organ transplantation and Chagas disease. JAMA. 2008 Mar;299(10):1134.
-
306Sousa AA, Lobo MC, Barbosa RA, Bello V. Chagas seropositive donors in kidney transplantation. Transplant Proc. 2004 May;36(4):868-9.
-
307Bocchi EA, Belloti G, Mocelin AO, Uip D, Bacal F, Higuchi ML, et al. Heart transplantation for chronic Chagas' disease. Ann Thorac Surg. 1996 Jun;61(6):1727-33.
-
308Freitas VL, Silva SC, Sartori AM, Bezerra RC, Westphalen EV, Molina TD, et al. Real-time PCR in HIV/ Trypanosoma cruzi coinfection with and without Chagas disease reactivation: association with HIV viral load and CD4 level. PLoS Negl Trop Dis. 2011 Aug;5(8):e1277.
-
309Brener Z. Laboratory-acquired Chagas' disease: an endemic disease among parasitologists? In: Morel M, editor. Genes and antigens of parasites: a laboratory manual. 2 ed. Rio de Janeiro: Editora Fiocruz; 1984. p. 3-9.
-
310Brener Z. Laboratory-acquired Chagas disease: comment. Trans R Soc Trop Med Hyg. 1987;81(3):527.
-
311Shikanai-Yasuda MA, Umezawa ES, Tolezano JE, Matsubara L. Doença de Chagas aguda após uso profilático de benznidazol (Acidente de laboratório). Rev Soc Bras Med Tropical. 1993;26 supl II:127-8.
-
312Bern C. Chagas' Disease. N Engl J Med. 2015 Nov;373(19):1881-2.
-
313Portela-Lindoso AAB, Shikanai-Yasuda MA. Doença de Chagas crônica: do xenodiagnóstico e hemocultura à reação em cadeia da polimerase. Rev Saude Publica. 2003 fev;37(1):107-15.
-
Technical Surveillance Unit of Vector-Borne Diseases, Secretariat of Health Surveillance, Ministry of Health of Brazil, Brasília, Distrito Federal, Brazil. The English version of the original article in Brazilian Portuguese of this Consensus was made possible through a partnership with the World Health Organization.
-
3
*This article was originally published in Portuguese (Brazil), under license of the Creative Commons CC BY NC for non-commercial use, according to the reference: Dias JCP, Ramos Jr. AN, Gontijo ED, Luquetti A, Shikanai-Yasuda MA, Coura JR et al. 2 nd Brazilian Consensus on Chagas Disease, 2015. Epidemiol Serv Saúde 2016; 25 (esp):7-86. Available at: http://scielo.iec.pa.gov.br/scielo.php?script=sci_arttext&pid=S1679-49742016000500007&lng=pt. / http://dx.doi.org/10.5123/S1679-49742016000500002
-
4
1 Based on international publications and particularly endorsed in the First Latin American Guidelines for the Diagnosis and Treatment of Chagas Heart Disease. 203 Evidence levels ranked as B or C cannot be interpreted as weak recommendations. There are many consensus-based recommendations, so with the recommendation of class I, level of evidence C (expert opinions). On the other hand, some indications considered controversial (recommendation grade II) may be grounded in randomized controlled trials (level of evidence A).
ATTACHMENTS
Definitions of grades or classes of recommendations and levels of evidence 1 4 1 Based on international publications and particularly endorsed in the First Latin American Guidelines for the Diagnosis and Treatment of Chagas Heart Disease. 203 Evidence levels ranked as B or C cannot be interpreted as weak recommendations. There are many consensus-based recommendations, so with the recommendation of class I, level of evidence C (expert opinions). On the other hand, some indications considered controversial (recommendation grade II) may be grounded in randomized controlled trials (level of evidence A).
Classes of recommendations
-
Class I: conditions for which there is conclusive evidence, or, in its absence, general agreement that the procedure or treatment is useful and/or effective.
-
Class II: conditions for which there is conflicting evidence and/or divergence of views on the utility/effectiveness and safety of the procedure or treatment:
-
Class IIa: evidence and opinions favor the indication for the procedure or treatment; most professionals involved in specific management approve;
-
Class IIb: utility/effectiveness and safety is less well established by evidence, with opinion divided; procedure or treatment is considered optional.
-
Class III: conditions for which there is conclusive evidence and/or general agreement that the procedure/treatment is not useful and/or effective, and may even be harmful in some circumstances.
Levels of Evidence
-
Level A: data from several consistent and good quality randomized trials or robust meta-analysis of randomized clinical trials.
-
Level B: data obtained from a single randomized clinical trial or from several non-randomized clinical studies, observational (less robust meta-analysis).
-
Level C: data obtained from consensual opinions of specialists on the subject.
Publication Dates
-
Publication in this collection
Dec 2016
History
-
Received
30 Nov 2016 -
Accepted
05 Dec 2016