Abstract
INTRODUCTION:
Emerging diseases are of great interest, especially those associated with high mortality rates such as hantaviruses. We aimed to conduct a seroepidemiological survey to determine the levels of hantavirus infection.
METHODS:
In-house enzyme-linked immunosorbent assay (ELISA) was used to detect specific antibodies.
RESULTS:
Of the 429 samples collected, seropositivity of 3.9% to anti-hantavirus immunoglobulin G (IgG) was observed (CI 95%: 2.3-5.7). Moreover, in three cases, immunoglobulin M (IgM) was detected, of which two were diagnosed as hantavirus cardiopulmonary syndrome (HCPS).
CONCLUSIONS:
Our data indicate the considerable occurrence of previous hantavirus infections, highlighting occurrences from sub-clinical cases to HCPS.
Keywords:
Hantavirus; Zoonosis; Hantavirus cardiopulmonary syndrome
Hantaviruses are an emerging zoonosis transmitted mainly by the inhalation of aerosols from the excreta of infected wild rodents. Following the inhalation, viral particles may invade endothelial cells and platelets through tropism by β-integrin receptors. Subsequently, a heightened immune response can lead to pulmonary edema and cardiogenic shock as observed in hantavirus cardiopulmonary syndrome (HCPS). The evolution to this clinical picture reflects in a high case fatality rate ranging from 35 to 50% in several American countries11. Mackow ER, Gavrilovskaya IN. Cellular receptors and hantavirus pathogenesis. Curr Top Microbiol Immunol. 2001;256:91-115.,22. Hjelle B, Torres-Pérez F. Hantaviruses in the americas and their role as emerging pathogens. Viruses. 2010;2(12):2559-86..
Hantaviruses are enveloped virus particles with a mean diameter of 112nm, having a spherical morphology and a single-stranded ribonucleic acid (RNA) genome, having negative polarity, and being divided into three segments: small (S), medium (M), and large (L)33. Alfadhli A, Love Z, Arvidson B, Seeds J, Willey J, Barklis E. Hantavirus nucleocapsid protein oligomerization. J Virol. 2001;75(4):2019-23.. It is reported that more than 21 hantavirus species have already been found, mainly in wild rodents44. Suzuki A, Bisordi I, Levis S, Garcia J, Pereira LE, Souza RP, et al. Identifying rodent hantavirus reservoirs, Brazil. Emerg Infect Dis. 2004;10(12):2127-34..
In the Americas, Brazil has the highest number of hantavirus-related cases, with 2,048 disease cases diagnosed by 201655. Ministério da Saúde. Casos confirmados de Hantavirose: Brasil, Grandes Regiões e Unidades Federadas de 1993-2016 (Internet). Brasília: Secretaria de Vigilância em Saúde. Available from: http://portalsaude.saude.gov.br/index.php/o-ministerio/principal/leia-mais-o-ministerio/708-secretaria-svs/vigilancia-de-a-a-z/hantavirose/11304-situacao-epidemiologica-dados.2017.
http://portalsaude.saude.gov.br/index.ph...
. This fact classifies HCPS as a public health problem in the country. Thus, we aimed to conduct the first seroepidemiological survey for hantavirus in the Goiás State to determine the levels of previous hantavirus infections and to evaluate the knowledge of the population about the disease in the Jataí county-Goiás.
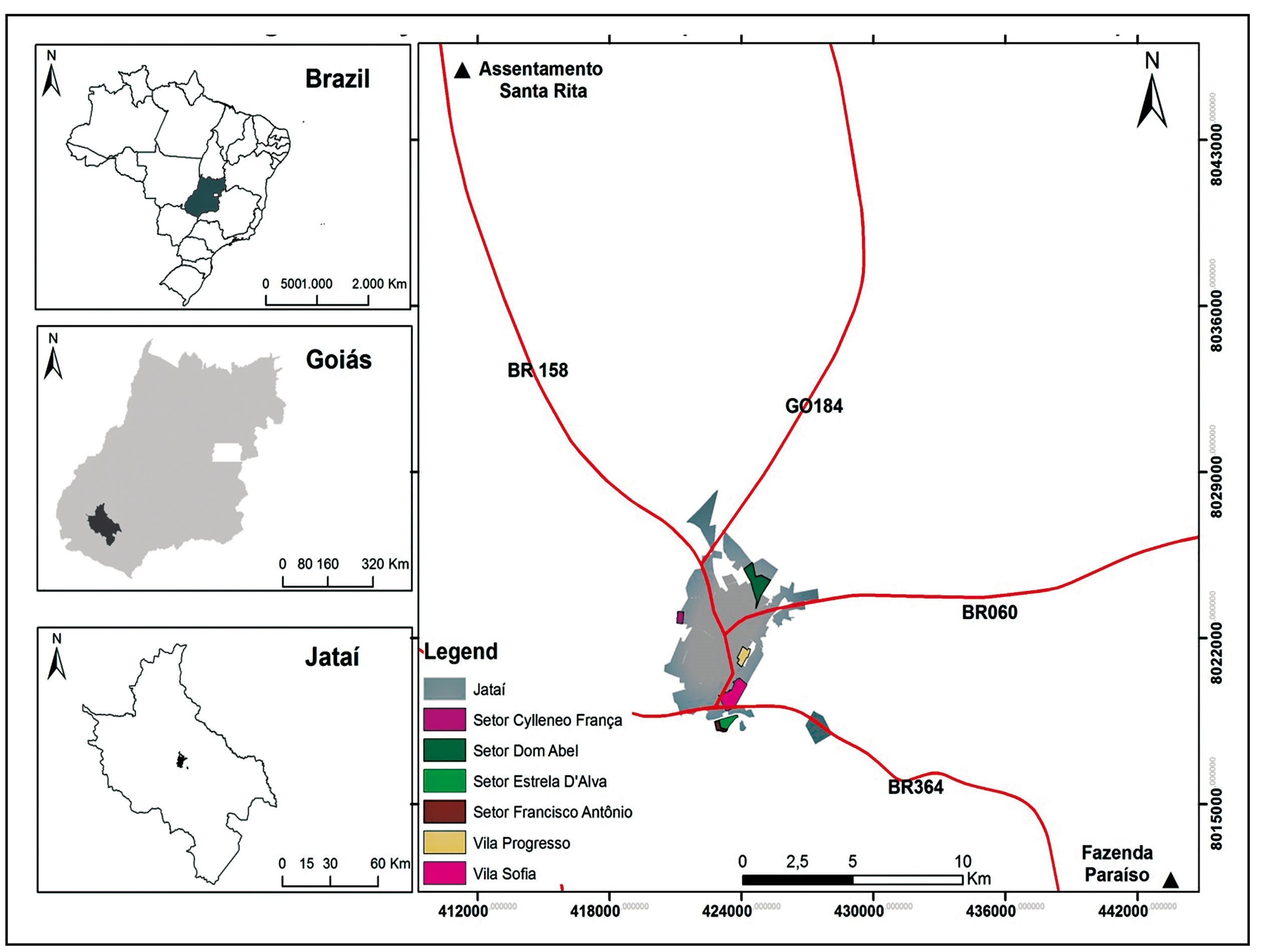
The study site was the municipality of Jataí, located in southwestern part of the state with a distance of 320km from the capital (Goiânia) and a population of 97,077 individuals (Figure 1). Using a seroepidemiological survey design, the levels of anti-hantavirus immunoglobulin G (IgG) and immunoglobulin M (IgM) were investigated and a questionnaire was administered to the participants. Part of the survey involved epidemiological data related to risk factors such as occupational activity, place of residence and recreational activities such as leisure in the rural area.
Serological analysis for hantavirus infection in Jataí-GO. The study region is shown with its geographic location in the country. Organization: Martins, Alécio Perini (2016).
The households drawn to participate in this study, from an urban plan map, were visited from August 2012 to February 2015. Individuals who agreed to participate in the study donated whole blood, collected through a sterile disposable micro lance from the digital pulp and the blood was soaked in paper strips (Nobuto Blood Sampling Paper) of high-quality. After the collection, georeferencing was carried out through the software ArcGis10, for the points where the serological research was carried out (Figure 2).
Sector model used in the epidemiological survey. The black points indicate the homes where samples were collected. Organization: Martins, Alécio Perini (2016).
The filter paper strips soaked with the biological samples were stored at a low temperature in the Virology laboratory of Federal University of Goiás [Universidade Federal de Goiás (UFG)] in Jataí. Subsequently, they were diluted in phosphate-buffered saline (PBS) buffer and processed by the in-house enzyme-linked immunosorbent assay (ELISA) test66. Figueiredo LTM, Moreli ML, Borges AA, de Figueiredo GG, Badra SJ, Bisordi I, et al. Evaluation of an enzyme-linked immunosorbent assay based on Araraquara virus recombinant nucleocapsid protein. Am J Trop Med Hyg. 2009;81(2):273-6..
Briefly, a 96-well, flat-bottom ELISA microplate [PolySorp™, Nunc, Chicago, IL, United States of America (USA)] was sensitized at the top (wells A, B, C, and D of rows one to 12) with the recombinant N protein of Araraquara virus (antigen positive). It was also sensitized at the bottom part (wells E, F, G, and H of rows 1 to 12), with Escherichia coli extract (antigen negative), both diluted in 0.05M carbonate buffer at pH 9.6 and a concentration of 2μg/ml. After the dilution and the addition of the antigen (100μl/well), the plates were incubated in a moist chamber inside a refrigerator at 4°C for 18 hours (overnight). Thereafter, the wash solution (PBS 1X with tween (T) −20) was used in all phases, except after the addition of the chromogen in the final phase. The microplate was then blocked with skimmed milk powder (SMP, Molico Milk Nestlé®) and remained incubated in humid chambers for 2 hours at 37°C. Subsequently, the test sample, which was diluted 1: 100 in PBS-T and SMP, was added. The next step included adding the conjugate (goat anti-human IgG peroxidase, diluted 1: 2000 in PBS-T and SMP). Finally, the colorimetric substrate used was tetramethylbenzidine (TMB) (Sigma-Aldrich®, St. Louis, MO, USA) and the Stop solution was 1M hydrochloric acid (HCl). The absorbance was measured on an ELISA reader (Thermoplate) with a primary filter of 450nm and the secondary filter of 630 nm. The cut off was determined by the mean optical densities (OD) of the negative controls plus three standard deviations66. Figueiredo LTM, Moreli ML, Borges AA, de Figueiredo GG, Badra SJ, Bisordi I, et al. Evaluation of an enzyme-linked immunosorbent assay based on Araraquara virus recombinant nucleocapsid protein. Am J Trop Med Hyg. 2009;81(2):273-6..
STATA IC/64 software (version 13.1, College Station, TX) was used for data analysis. Therefore, for the measure of association, prevalence ratio (PR) was reported in the statistical analysis with confidence interval (CI) of 95%. The study was approved by the research ethics committee involving human beings of the Federal University of Goiás (n° 348/2010) and participants provided written informed consent.
A total of 429 samples were screened by the in-house ELISA method. Of these samples, 52% belonged to males, 84% came from the urban area and the average age of participants was 36.2 years. The seropositivity of anti-hantavirus IgG in the Jataí population was 3.9% (17/429; 95% CI: 2.3-5.7), with higher seroprevalence for men (5%, 12/238) than for women (2.6%, 5/191) (Table 1). However, no significant difference was found between the seropositivity by gender (PR: 1.9; 95% CI 0.7-5.0), the participants living in rural/urban areas (PR: 1.1; 95% CI 0.3-3.8) and age of the participants (PR: 0.7; 95% CI 0.3-2).
Despite numerous hantavirosis-related mortalities in the county, the participants vaguely remembered symptoms that are suggestive of HCPS, such as intense fever, respiratory problems, chest pain, and headache22. Hjelle B, Torres-Pérez F. Hantaviruses in the americas and their role as emerging pathogens. Viruses. 2010;2(12):2559-86.,77. Limongi JE, da Costa FC, Pinto RMC, de Oliveira RC, Bragagnolo C, Lemos ERS, et al. Cross-sectional survey of Hantavirus infection, Brazil. Emerg Infect Dis . 2009; 15(12):1981-83.. Hence, according to Figueiredo et al.88. Figueiredo LT, Souza WM, Ferrés M, Enria DA. Hantaviruses and cardiopulmonary syndrome in South America. Virus Res. 2014;187:43-54., the reported confirmed cases of hantavirosis correspond to only a small part of the totality. Most of the infections could have been diagnosed as non-specific acute febrile illnesses such as influenza A (H1N1), leptospirosis and dengue. Therefore, owing to the inability of most participants to recall prior clinical presentations and because they also present with antibodies typical of the chronic phase, it is not possible to confirm the existence of subclinical infections.
Among the samples included for the serological investigation were samples belonging to two male participants aged 29 and 32 years with IgM anti-hantavirus antibodies. The infection in the two men evolved into the severe form of the disease (HCPS), with the occurrence of death in the younger patient99. Moreli ML, da Costa VG, Novaes DPS, Flor EC, Silva JF, Vilela KRG, et al. Hantavirus cardiopulmonary syndrome: a report of two cases. J Bras Patol Med Lab. 2013;49(5):312-6.. The third IgM reactive case, another man aged 44 years, presented with a disease similar to dengue, but tested negative for the dengue virus. In this case, the diagnosis occurred retrospectively, thereby reinforcing the concern of underreporting of hantavirosis among suspected dengue cases, since the clinical management of the two diseases differs.
The seroprevalence of anti-hantavirus IgG antibodies detected in this study is higher than that reported in the serological survey conducted by Souza et al.1010. Souza WM, Machado AM, Disner GR, Boff E, Machado ARSR, de Padua M, et al. Antibody levels to hantavirus in inhabitants of western Santa Catarina state, Brazil. Rev Inst Med Trop São Paulo. 2012;54(4):193-6. Despite using similar techniques, they observed 1.97% seroprevalence in the samples from the urban and rural areas of four municipalities of the Santa Catarina State. In another study conducted in cities near the border between Brazil and Argentina, still in the State of Santa Catarina, they found a 3.5% seroprevalence1111. Souza WM, Machado AM, Figueiredo LTM, Boff E. Serosurvey of hantavirus infection in humans in the border region between Brazil and Argentina. Rev Soc Bras Med Trop. 2011;44(2):131-5.. The highest reported level of infection with an index of 4.7% was found in the City of Cássia dos Coqueiros, located in the State of São Paulo, in the Southeast region1212. Badra SJ, Maia FGM, Figueiredo GG, Santos Júnior GS, Campos GM, Figueiredo LTM, et al. A retrospective sorologic survey of hantavirus infections in the county of Cássia dos Coqueiros, State of São Paulo, Brazil. Rev Soc Bras Med Trop . 2012;45(4):468-70.. It is important to point out that most of the HCPS cases occurred in the Southeast of the country, along with the South region.
Recently, in the study conducted by Vieira et al.1313. Vieira CJSP, da Silva DJF, Barreto ES, Siqueira CEH, da Costa VG, Lourenço FJ, et al. Serological evidence of hantavirus infection in an urban area in Mato Grosso State, Brazil. Rev Soc Bras Med Trop . 2016;49(3):348-50., high anti-hantavirus IgG seroprevalence of 13.6% was shown in the City of Sinop in the Mato Grosso State. The study site may justify this in part because the state has the second highest number of HCPS cases nationwide. Vieira et al. also reported higher seropositivity among women from urban areas, differing from our data with a higher seropositivity among men (71%) and from rural areas (4.4%). It is known that hantavirosis is considered primarily a rural disease and, consequently, rural dwellers as well as individuals who are often in contact with the rural area comprise a risk group. Therefore, there are numerous studies from other areas of Brazil demonstrating considerable serological prevalence for hantavirus in men living in rural areas, a fact that seem to link hantavirus infection and field-related activities77. Limongi JE, da Costa FC, Pinto RMC, de Oliveira RC, Bragagnolo C, Lemos ERS, et al. Cross-sectional survey of Hantavirus infection, Brazil. Emerg Infect Dis . 2009; 15(12):1981-83.,1414. Terças ACP, Atanaka dos Santos M, Pignatti MG, Espinosa MM, de Melo Via AVG, Menegatti JA. Hantavirus pulmonary syndrome outbreak, Brazil, December 2009-January 2010. Emerg Infect Dis . 2013;19(11):1824-7..
Although the risk group for hantavirus is comprised mainly of men in their productive years (aged 20 to 39 years) residing in the rural areas55. Ministério da Saúde. Casos confirmados de Hantavirose: Brasil, Grandes Regiões e Unidades Federadas de 1993-2016 (Internet). Brasília: Secretaria de Vigilância em Saúde. Available from: http://portalsaude.saude.gov.br/index.php/o-ministerio/principal/leia-mais-o-ministerio/708-secretaria-svs/vigilancia-de-a-a-z/hantavirose/11304-situacao-epidemiologica-dados.2017.
http://portalsaude.saude.gov.br/index.ph...
, nevertheless, serological surveys for hantavirus should also be carried out in the urban areas. Residents of urban areas also have links to the field, through work or field-related activities which might increase the risk of exposure to rodent reservoirs1111. Souza WM, Machado AM, Figueiredo LTM, Boff E. Serosurvey of hantavirus infection in humans in the border region between Brazil and Argentina. Rev Soc Bras Med Trop. 2011;44(2):131-5.,1313. Vieira CJSP, da Silva DJF, Barreto ES, Siqueira CEH, da Costa VG, Lourenço FJ, et al. Serological evidence of hantavirus infection in an urban area in Mato Grosso State, Brazil. Rev Soc Bras Med Trop . 2016;49(3):348-50.. Thus, when the questionnaire was administered, a majority of the population expressed having experienced situations considered to enhance the risk for hantavirus infection, including rural tourism (74%) and exposure to house cleaning in the countryside, sheds, and other similarities (71%). Direct and indirect contact with rodents was also common (81%). Hence, it would be interesting to develop a standardized questionnaire, with a score involving risk activities, since the frequency of performing these activities could better correlate with the risks of viral infection.
In epidemiological studies in Brazil, the Araraquara Hantavirus genotype was detected in the rodent Necromys lasiurus, being found in the Cerrado, a predominant biome in the state44. Suzuki A, Bisordi I, Levis S, Garcia J, Pereira LE, Souza RP, et al. Identifying rodent hantavirus reservoirs, Brazil. Emerg Infect Dis. 2004;10(12):2127-34.. In this context, in a study carried out in Ribeirão Preto-São Paulo and Minas Gerais State, near the State of Goiás where sequenced genetic material were collected in patients during the acute phase of infection, showed a high homology with the N gene and partial S segment sequences from the Araraquara virus1515. Moreli ML, Sousa RL, Figueiredo LTM. Detection of Brazilian hantavirus by reverse transcription polymerase chain reaction amplification of N gene in patients with hantavirus cardiopulmonary syndrome. Mem Inst Oswaldo Cruz. 2004;99(6):633-8.,1616. Limongi JE, Oliveira RC, Guterres A, Costa Neto SF, Fernandes J, Vicente LH, et al. Hantavirus pulmonary syndrome and rodent reservoirs in the savanna-like biome of Brazil’s southeastern region. Epidemiol Infect. 2016;144(5):1107-16.. Thus, it is possible that the hantavirus-related morbidities occurring at these sites are caused by this virus, however, it is necessary to carry out viral identification in the future owing to the possibility of other circulating genotypes in this region.
During the course of the study, efforts aimed to raise awareness among the population, through the dissemination of information regarding hantavirosis. Hence, self-explanatory folders on the disease were provided as information regarding symptoms, means of transmission and prevention measures. Our results suggest that most of the participants were unaware of the disease (~ 80%). This reinforces the need for public health measures aimed at raising awareness about the risks of hantavirus. In addition, epidemiological data from the present study provides insights for future studies using molecular biology tools for the identification of hantavirus associated with HCPS and other antigenically related viruses in this region.
Acknowledgments
The authors gratefully acknowledge the medical team and the laboratory staff of Laboratório Elzevir Ferreira Lima, the technical team of Núcleo de Vigilância Epidemiológica e Ambiental em Saúde, Jataí-GO.
REFERENCES
-
1Mackow ER, Gavrilovskaya IN. Cellular receptors and hantavirus pathogenesis. Curr Top Microbiol Immunol. 2001;256:91-115.
-
2Hjelle B, Torres-Pérez F. Hantaviruses in the americas and their role as emerging pathogens. Viruses. 2010;2(12):2559-86.
-
3Alfadhli A, Love Z, Arvidson B, Seeds J, Willey J, Barklis E. Hantavirus nucleocapsid protein oligomerization. J Virol. 2001;75(4):2019-23.
-
4Suzuki A, Bisordi I, Levis S, Garcia J, Pereira LE, Souza RP, et al. Identifying rodent hantavirus reservoirs, Brazil. Emerg Infect Dis. 2004;10(12):2127-34.
-
5Ministério da Saúde. Casos confirmados de Hantavirose: Brasil, Grandes Regiões e Unidades Federadas de 1993-2016 (Internet). Brasília: Secretaria de Vigilância em Saúde. Available from: http://portalsaude.saude.gov.br/index.php/o-ministerio/principal/leia-mais-o-ministerio/708-secretaria-svs/vigilancia-de-a-a-z/hantavirose/11304-situacao-epidemiologica-dados2017.
» http://portalsaude.saude.gov.br/index.php/o-ministerio/principal/leia-mais-o-ministerio/708-secretaria-svs/vigilancia-de-a-a-z/hantavirose/11304-situacao-epidemiologica-dados -
6Figueiredo LTM, Moreli ML, Borges AA, de Figueiredo GG, Badra SJ, Bisordi I, et al. Evaluation of an enzyme-linked immunosorbent assay based on Araraquara virus recombinant nucleocapsid protein. Am J Trop Med Hyg. 2009;81(2):273-6.
-
7Limongi JE, da Costa FC, Pinto RMC, de Oliveira RC, Bragagnolo C, Lemos ERS, et al. Cross-sectional survey of Hantavirus infection, Brazil. Emerg Infect Dis . 2009; 15(12):1981-83.
-
8Figueiredo LT, Souza WM, Ferrés M, Enria DA. Hantaviruses and cardiopulmonary syndrome in South America. Virus Res. 2014;187:43-54.
-
9Moreli ML, da Costa VG, Novaes DPS, Flor EC, Silva JF, Vilela KRG, et al. Hantavirus cardiopulmonary syndrome: a report of two cases. J Bras Patol Med Lab. 2013;49(5):312-6.
-
10Souza WM, Machado AM, Disner GR, Boff E, Machado ARSR, de Padua M, et al. Antibody levels to hantavirus in inhabitants of western Santa Catarina state, Brazil. Rev Inst Med Trop São Paulo. 2012;54(4):193-6.
-
11Souza WM, Machado AM, Figueiredo LTM, Boff E. Serosurvey of hantavirus infection in humans in the border region between Brazil and Argentina. Rev Soc Bras Med Trop. 2011;44(2):131-5.
-
12Badra SJ, Maia FGM, Figueiredo GG, Santos Júnior GS, Campos GM, Figueiredo LTM, et al. A retrospective sorologic survey of hantavirus infections in the county of Cássia dos Coqueiros, State of São Paulo, Brazil. Rev Soc Bras Med Trop . 2012;45(4):468-70.
-
13Vieira CJSP, da Silva DJF, Barreto ES, Siqueira CEH, da Costa VG, Lourenço FJ, et al. Serological evidence of hantavirus infection in an urban area in Mato Grosso State, Brazil. Rev Soc Bras Med Trop . 2016;49(3):348-50.
-
14Terças ACP, Atanaka dos Santos M, Pignatti MG, Espinosa MM, de Melo Via AVG, Menegatti JA. Hantavirus pulmonary syndrome outbreak, Brazil, December 2009-January 2010. Emerg Infect Dis . 2013;19(11):1824-7.
-
15Moreli ML, Sousa RL, Figueiredo LTM. Detection of Brazilian hantavirus by reverse transcription polymerase chain reaction amplification of N gene in patients with hantavirus cardiopulmonary syndrome. Mem Inst Oswaldo Cruz. 2004;99(6):633-8.
-
16Limongi JE, Oliveira RC, Guterres A, Costa Neto SF, Fernandes J, Vicente LH, et al. Hantavirus pulmonary syndrome and rodent reservoirs in the savanna-like biome of Brazil’s southeastern region. Epidemiol Infect. 2016;144(5):1107-16.
-
Financial Support: Fundação de Amparo à Pesquisa do Estado de Goiás (FAPEG) research grant call notice PPSUS n° 004/2010.
Publication Dates
-
Publication in this collection
Jul-Aug 2017
History
-
Received
30 Jan 2017 -
Accepted
17 May 2017