Abstract
In Brazil, malaria is an important public health problem first reported in 1560. Historically, fluctuations in malaria cases in Brazil are attributed to waves of economic development; construction of railroads, highways, and hydroelectric dams; and population displacement and land occupation policies. Vector control measures have been widely used with an important role in reducing malaria cases. In this review article, we reviewed the vector control measures established in the Brazilian territory and aspects associated with such measures for malaria. Although some vector control measures are routinely used in Brazil, many entomological and effectiveness information still need better evidence in endemic areas where Plasmodium vivax predominates. Herein, we outlined some of the needs and priorities for future research: a) update of the cartography of malaria vectors in Brazil, adding molecular techniques for the correct identification of species and complexes of species; b) evaluation of vector competence of anophelines in Brazil; c) strengthening of local entomology teams to perform vector control measures and interpret results; d) evaluation of vector control measures, especially use of insecticide-treated nets and long-lasting insecticidal nets, estimating their effectiveness, cost-benefit, and population acceptance; e) establishment of colonies of malaria vectors in Brazil, i.e., Anopheles darlingi, to understand parasite-vector interactions better; f) study of new vector control strategies with impacts on non-endophilic vectors; g) estimation of the impact of insecticide resistance in different geographical areas; and h) identification of the relative contribution of natural and artificial breeding sites in different epidemiological contexts for transmission.
Keywords:
Vector control; Anopheles; Malaria
INTRODUCTION
Malaria is an acute febrile infectious disease transmitted through the bites of infected female Anopheles mosquitoes. In 2017, approximately 219 million cases of malaria were reported in 91 countries, compared with 237 and 211 million cases in 2010 and 2015, respectively. The change in the endemicity pattern is attributed to the large-scale implementation of malaria control measures, such as adequate and early treatment of patients, use of insecticide-treated nets (ITNs), and insecticide indoor residual spraying (IRS). Despite such global reductions, the case incidence substantially increased in the Region of the Americas between 2014 and 201611. World Health Organization (WHO). World Malaria Report 2017. Geneva: WHO; 2017, 196p..
Historically, fluctuations in malaria cases in Brazil are attributed to waves of economic development; construction of railroads, highways, and other large edifice-like hydroelectric power plants (HPPs); and population displacement and land occupation policies, especially in the Northern region22. Sampaio VS, Siqueira AM, Alecrim MGC, Mourão MPG, Marchesini PB, Albuquerque BC, et al. Malaria in the State of Amazonas: a typical Brazilian tropical disease influenced by waves of economic development. Rev Soc Bras Med Trop. 2015;48(Suppl I):4-11.
3. Coura JR, Suárez-Mutis M, Ladeia-Andrade S. A new challenge for malaria control in Brazil: asymptomatic Plasmodium infection - a review. Mem Inst Oswaldo Cruz. 2006;101(3):229-37.
4. Griffing SM, Tauil PL, Udhayakumar V, Silva-Flannery L. A historical perspective on malaria control in Brazil. Mem Inst Oswaldo Cruz . 2015;110(6):701-18.-55. Gadelha P. Conforming strategies of public health campaigns to disease specificity and national contexts: Rockefeller Foundation’s early campaigns against hookworm and malaria in Brazil. Parassitologia. 1998;40(1-2):159-75.. Consequently, vector control measures were widely used with an important role in reducing the epidemiology of malaria. Indeed, the success in the extinction of An. gambiae in the Brazilian territory is a notable outcome of such a process66. Parmakelis A, Russello MA, Caccone A, Marcondes CB, Costa J, Forattini OP, et al. Historical analysis of a near disaster: Anopheles gambiae in Brazil. Am J Trop Med Hyg. 2008;78(1):176-8.
7. Pinto C. Disseminação da malaria pela aviação: biologia do Anopheles gambiae e outros anofelíneos do Brasil. Mem Inst Oswaldo Cruz . 1939;34:293-430.-88. Packard RM, Gadelha P. A land filled with mosquitoes: Fred L Soper. The Rockefeller Foundation and the Anopheles gambiae invasion of Brazil. Med Anthropol. 1997;17(3):215-38.. In this article, we reviewed the vector control measures established in the Brazilian territory.
MALARIA AND VECTOR CONTROL IN BRAZIL: A BRIEF HISTORICAL OVERVIEW
After the colonial period, there was a substantial increase in malaria cases in the Amazon region, mainly owing to the great displacement of immunologically incompetent Northeastern migrants led by rubber extraction in the Amazon (1879-1912) and the expansion and construction of the Madeira-Mamoré Railroad (1907-1912)33. Coura JR, Suárez-Mutis M, Ladeia-Andrade S. A new challenge for malaria control in Brazil: asymptomatic Plasmodium infection - a review. Mem Inst Oswaldo Cruz. 2006;101(3):229-37.,99. Camargo EP. Malária, maleita, paludismo. Cienc Cult. 2003;55(1):26-9.
10. Stepan NL. Picturing Tropical Nature. Ithaca: Cornell University Press: 2001. 283p.
11. Stepan NL. The only serious terror in these regions: malaria control in the Brazilian Amazon. Armus D, editor. Disease in the History of Modern Latin America: from Malaria to AIDS. Durham: Duke University Press: 2003. p. 25-50.
12. Deane LM. Malaria studies and control in Brazil. Am J Trop Med Hyg . 1988;38(2):223-30.-1313. Ferreira MU, Castro MC. Challenges for malaria elimination in Brazil. Malar J. 2016;15(1):284..
During the late 1930s, the Northeast Region of Brazil was invaded by An. gambiae, an important vector of malaria in sub-Saharan Africa later identified as An. arabiensis66. Parmakelis A, Russello MA, Caccone A, Marcondes CB, Costa J, Forattini OP, et al. Historical analysis of a near disaster: Anopheles gambiae in Brazil. Am J Trop Med Hyg. 2008;78(1):176-8.. This vector was introduced accidentally in Natal, Rio Grande do Norte from Dakar (Senegal) and immediately spread to the immediate vicinity of the railway and canals near the Potengi river77. Pinto C. Disseminação da malaria pela aviação: biologia do Anopheles gambiae e outros anofelíneos do Brasil. Mem Inst Oswaldo Cruz . 1939;34:293-430.,99. Camargo EP. Malária, maleita, paludismo. Cienc Cult. 2003;55(1):26-9.,1313. Ferreira MU, Castro MC. Challenges for malaria elimination in Brazil. Malar J. 2016;15(1):284.,1414. The Rockefeller Foundation. Annual Report, 1931. New York: The Rockefeller Foundation; 1931. 420p.. In 1931, 344 deaths were reported in Natal, and in 1938, it spread silently inwards, reaching less arid valleys of the Jaguaribe river in the state of Ceará99. Camargo EP. Malária, maleita, paludismo. Cienc Cult. 2003;55(1):26-9.,1212. Deane LM. Malaria studies and control in Brazil. Am J Trop Med Hyg . 1988;38(2):223-30.,1515. Tauil P, Deane LM, Sabroza P, Ribeiro C. Malaria in Brazil. Cad Saude Publ. 1985;1(1):71-111.,1616. Killeen GF, Fillinger U, Kiche I, Gouagna LC, Knols BGJ. Eradication of Anopheles gambiae from Brazil: lessons for malaria control in Africa? Lancet Infect Dis. 2002;2(10):618-27.. This uncontrolled spread led to 150,000 cases of malaria and 14,000 deaths in 8 months in both states99. Camargo EP. Malária, maleita, paludismo. Cienc Cult. 2003;55(1):26-9.,1717. Packard RM. The making of a Tropical Disease: a Short History of Malaria. Baltimore: Johns Hopkins University Press; 2007. 320p.. The Northeast Malaria Service was created in the following year, exclusively to fight against the malaria African vector1818. The Rockefeller Foundation. Annual Report, 1939. The Rockefeller Foundation; New York: 1939. 517p.. Larvicides incorporated with Paris green (i.e., copper acetoarsenite, an inorganic compound usually used as a rodenticide and an insecticide) were deployed, and all houses were fumed with pyrethrum99. Camargo EP. Malária, maleita, paludismo. Cienc Cult. 2003;55(1):26-9.,1919. Soper FL. Paris Green in the Eradication of Anopheles gambiae: Brazil, 1940; Egypt, 1945. Mosquito News. 1966;26(4):470-6.. In 1940, the malaria African vector was eradicated in Brazil1313. Ferreira MU, Castro MC. Challenges for malaria elimination in Brazil. Malar J. 2016;15(1):284..
In view of the large number of cases reported in the 1940s, the National Malaria Service was created, but was quickly changed to the Malaria Eradication Campaign (CEM), which adopted the Global Malaria Eradication Program strategies of the World Health Organization (WHO)1212. Deane LM. Malaria studies and control in Brazil. Am J Trop Med Hyg . 1988;38(2):223-30.,2020. Silveira AC, de Rezende DF. Avaliação da estratégia global de controle integrado da malária no Brasil. Brasília: Organização Pan-Americana da Saúde; 2001. 120p.,2121. Alves MJCP, Mayo RC, Donalisio MR. História, epidemiologia e controle da malária na região de Campinas, Estado de São Paulo, Brasil, 1980 a 2000. Rev Soc Bras Med Trop . 2004;37(1):41-5.. During this period, there was a significant and important impact on malaria transmission owing to the indoor application of dichlorodiphenyltrichloroethane (DDT) and the use of antimalarial drugs1212. Deane LM. Malaria studies and control in Brazil. Am J Trop Med Hyg . 1988;38(2):223-30.,2222. Payne D. Did medicated salt hasten the spread of chloroquine resistance in Plasmodium falciparum? Parasitol Today. 1988;4(4):112-5.,2323. Gusmão HH. Fighting disease-bearing mosquitoes through relentless field leadership. Am J Trop Med Hyg . 1982;31(4):705-10..
The CEM was suspended in 1970 owing to the reduction of cases, criticism of the public health administrative model, and concerns regarding DDT use44. Griffing SM, Tauil PL, Udhayakumar V, Silva-Flannery L. A historical perspective on malaria control in Brazil. Mem Inst Oswaldo Cruz . 2015;110(6):701-18.. Malaria control activities are coordinated by the Superintendence of Public Health Campaigns, also responsible for the provision of antimalarial drugs for appropriate use and mass treatment, technical supervision, and use of outdoor ultra-low volume nebulization impregnated curtains and new insecticides33. Coura JR, Suárez-Mutis M, Ladeia-Andrade S. A new challenge for malaria control in Brazil: asymptomatic Plasmodium infection - a review. Mem Inst Oswaldo Cruz. 2006;101(3):229-37.,44. Griffing SM, Tauil PL, Udhayakumar V, Silva-Flannery L. A historical perspective on malaria control in Brazil. Mem Inst Oswaldo Cruz . 2015;110(6):701-18.,2121. Alves MJCP, Mayo RC, Donalisio MR. História, epidemiologia e controle da malária na região de Campinas, Estado de São Paulo, Brasil, 1980 a 2000. Rev Soc Bras Med Trop . 2004;37(1):41-5.
22. Payne D. Did medicated salt hasten the spread of chloroquine resistance in Plasmodium falciparum? Parasitol Today. 1988;4(4):112-5.-2323. Gusmão HH. Fighting disease-bearing mosquitoes through relentless field leadership. Am J Trop Med Hyg . 1982;31(4):705-10.. At the end of 1998, the Pan American Health Organization released an analysis of the epidemiological situation of malaria in the Americas, in line with the initiative adopted by the WHO known as Roll Back Malaria2424. Loiola CCP, Mangabeira da Silva CJ, Tauil PL. Controle da malária no Brasil: 1965 a 2001. Rev Panam Salud Publica 2002;11(4):235-44.-2525. United Nations Development Programme (UNDP). Multisectoral Action Framework for Malaria. Geneva: Roll Back Malaria Partnership/UNDP; 2013. 68p.. In October 1999, the actions to reduce malaria in Brazil were conducted through the Plan to Intensify Malaria Control in the Legal Amazon (PIACM)2424. Loiola CCP, Mangabeira da Silva CJ, Tauil PL. Controle da malária no Brasil: 1965 a 2001. Rev Panam Salud Publica 2002;11(4):235-44.. However, despite the advances, several problems have not yet been solved, and the incidence of malaria remained high2525. United Nations Development Programme (UNDP). Multisectoral Action Framework for Malaria. Geneva: Roll Back Malaria Partnership/UNDP; 2013. 68p.. In 2003, the PIACM was replaced by the National Malaria Control Plan (NMCP)2626. Ministério da Saúde (MS). Secretaria de Vigilância em Saúde. Diretoria Técnica de Gestão. Guia para Gestão Local do Controle da Malária: Módulo 2: Controle Vetorial. Série B. Textos Básicos de Saúde. Brasília: MS; 2009. 59p. driven by objectives that aimed at reducing the morbidity, mortality, and severity of malaria along with stopping the transmission where possible.
MALARIA VECTORS IN BRAZIL: BIONOMICS AND BEHAVIORAL ASPECTS
The study of anopheline species in a region is important to direct vector control measures especially because the same species at the same site may change their habits over time, mainly as a result of environmental changes2727. Tadei WP, Mascarenhas BM, Podesta MG. Biologia de anofelinos amazônicos. VIII. Conhecimentos sobre a distribuição de espécies de Anopheles na região de Tucuruí-Marabá (Pará). Acta Amaz. 1983;13(1):103-40.
28. Tadei WP, Santos JMM, Costa WLS, Scarpassa VM. Biologia de anofelinos amazônicos. XII. Ocorrência de espécies de Anopheles, dinâmica da transmissão e controle da malária na zona urbana de Ariquemes (Rondônia). Rev Inst Med Trop São Paulo. 1988;30(3):221-51.
29. Osorio Quintero L, Dutary Thatcher B, Tadei WP. Biologia de anofelinos amazônicos. XXI. Ocorrrência de espécies de Anopheles e outros Culicídeos na área de influência da Hidrelétrica de Balbina - cinco anos após o enchimento do reservatório. Acta Amazônica. 1996;26(4):281-95.-3030. World Health Organization (WHO). Malaria vector control: decision making criteria and procedures for judicious use of insecticides/by: JA Najera and M Zaim. Geneva: WHO ; 2003. 106p.. The main vectors in the Americas are widely distributed in diverse environments and present high variability in their behavioral patterns, even including intrapopulation3131. Laporta GZ, Linton YM, Wilkerson RC, Bergo ES, Nagaki SS, Sant’Ana DC, et al. Malaria vectors in South America: current and future scenarios. Parasit Vectors. 2015;8(1):426.. In Brazil, 54 species of Anopheles mosquitoes are known, and the main vectors belong to two Anopheles subgenera: Nyssorhynchus and Kerteszia3232. Deane LM. Malaria vectors in Brazil. Mem Inst Oswaldo Cruz . 1986;81(supl 2):5-14.
33. Tadei WP, Thatcher BD, Santos JM, Scarpassa VM, Rodrigues IB, Rafael MS. Ecologic observations on anopheline vectors of malaria in the Brazilian Amazon. Am J Trop Med Hyg . 1998;59(2):325-35.-3434. Tadei WP, Dutary Thatcher B. Malaria vectors in the Brazilian Amazon: Anopheles of the subgenus Nyssorhynchus. Rev Inst Med Trop S. Paulo. 2000;42(2):87-94.. An. darlingi, An. aquasalis, and members of the An. albitarsis complex are the main transmitting species of malaria in the subgenus Nyssorhynchus3131. Laporta GZ, Linton YM, Wilkerson RC, Bergo ES, Nagaki SS, Sant’Ana DC, et al. Malaria vectors in South America: current and future scenarios. Parasit Vectors. 2015;8(1):426.,3333. Tadei WP, Thatcher BD, Santos JM, Scarpassa VM, Rodrigues IB, Rafael MS. Ecologic observations on anopheline vectors of malaria in the Brazilian Amazon. Am J Trop Med Hyg . 1998;59(2):325-35.,3434. Tadei WP, Dutary Thatcher B. Malaria vectors in the Brazilian Amazon: Anopheles of the subgenus Nyssorhynchus. Rev Inst Med Trop S. Paulo. 2000;42(2):87-94.. Conversely, the subgenus Kerteszia is responsible for the transmission in the Atlantic Forest, notably An. cruzii and An. bellator3535. Forattini OP. Culicidologia Médica. Identificação, Biologia e Epidemiologia. São Paulo: Editora da Universidade de São Paulo. 2002. Vol 2, 860p.,3636. Marrelli MT, Malafronte RS, Sallum MAM, Natal D. Kerteszia subgenus of Anopheles associated with the Brazilian Atlantic rainforest: current knowledge and future challenges. Malar J . 2007;6:127-34..
An. darlingi is considered the primary vector of malaria in Brazil2929. Osorio Quintero L, Dutary Thatcher B, Tadei WP. Biologia de anofelinos amazônicos. XXI. Ocorrrência de espécies de Anopheles e outros Culicídeos na área de influência da Hidrelétrica de Balbina - cinco anos após o enchimento do reservatório. Acta Amazônica. 1996;26(4):281-95.,3232. Deane LM. Malaria vectors in Brazil. Mem Inst Oswaldo Cruz . 1986;81(supl 2):5-14.,3737. Tadei WP, Thatcher BD, Santos JM, Scarpassa VM, Rodrigues IB, Rafael MS. Ecologic observations on anopheline vectors of malaria in the Brazilian Amazon. Am J Trop Med Hyg . 1998;59(2):325-35.. It is collected in greater quantity in peridomiciles3838. Santos JB, Santos F, Macedo V. Variação da densidade anofélica com o uso de mosquiteiros impregnados com deltametrina em uma área endêmica de malária na Amazônia Brasileira. Cad Saúde Pública 1999;15(2):281-92.. The largest period of hematophagic activity occurs during the first 3 hours of the escotophase3939. Gama RA, Santos RLC, Santos F, Silva IM, Resende MC, Eiras AE. Periodicidade de captura de Anopheles darlingi Root (Diptera: Culicidae) em Porto Velho, RO. Neotrop. Entomol. 2009;38(5):677-82., varying from 18:00 to 21:00 or 17:00 to 20:00, depending on the type of locality, density of the Anopheles population, season of the year, distance between residences and forests, and presence of other hosts2929. Osorio Quintero L, Dutary Thatcher B, Tadei WP. Biologia de anofelinos amazônicos. XXI. Ocorrrência de espécies de Anopheles e outros Culicídeos na área de influência da Hidrelétrica de Balbina - cinco anos após o enchimento do reservatório. Acta Amazônica. 1996;26(4):281-95.,3535. Forattini OP. Culicidologia Médica. Identificação, Biologia e Epidemiologia. São Paulo: Editora da Universidade de São Paulo. 2002. Vol 2, 860p.,4040. Voorham J. Intra-population plasticity of Anopheles darlingi's (Diptera, Culicidae) biting activity patterns in the state of Amapá, Brazil. Rev Saúde Pública. 2002;36(1)75-80.,4141. Charlwood JD, Hayes J. Variações geográficas no ciclo de picada do Anopheles darlingi Root no Brasil. Acta Amaz . 1978;8(4): 601-3.. Large reservoirs of water where there are limited current and salinity are used as breeding grounds3535. Forattini OP. Culicidologia Médica. Identificação, Biologia e Epidemiologia. São Paulo: Editora da Universidade de São Paulo. 2002. Vol 2, 860p.,4242. Consoli RAGB, Lourenço-de-Oliveira RL. Principais mosquitos de importância sanitária no Brasil. Rio de Janeiro: Editora da Fundação Oswaldo Cruz; 1994. 225p.. This species still bears uncertainties in relation to its biome and behavior, especially with regard to feeding habits, resting, and periodicity, which may be different according to locality3232. Deane LM. Malaria vectors in Brazil. Mem Inst Oswaldo Cruz . 1986;81(supl 2):5-14.
33. Tadei WP, Thatcher BD, Santos JM, Scarpassa VM, Rodrigues IB, Rafael MS. Ecologic observations on anopheline vectors of malaria in the Brazilian Amazon. Am J Trop Med Hyg . 1998;59(2):325-35.
34. Tadei WP, Dutary Thatcher B. Malaria vectors in the Brazilian Amazon: Anopheles of the subgenus Nyssorhynchus. Rev Inst Med Trop S. Paulo. 2000;42(2):87-94.-3535. Forattini OP. Culicidologia Médica. Identificação, Biologia e Epidemiologia. São Paulo: Editora da Universidade de São Paulo. 2002. Vol 2, 860p.,3737. Tadei WP, Thatcher BD, Santos JM, Scarpassa VM, Rodrigues IB, Rafael MS. Ecologic observations on anopheline vectors of malaria in the Brazilian Amazon. Am J Trop Med Hyg . 1998;59(2):325-35.
38. Santos JB, Santos F, Macedo V. Variação da densidade anofélica com o uso de mosquiteiros impregnados com deltametrina em uma área endêmica de malária na Amazônia Brasileira. Cad Saúde Pública 1999;15(2):281-92.
39. Gama RA, Santos RLC, Santos F, Silva IM, Resende MC, Eiras AE. Periodicidade de captura de Anopheles darlingi Root (Diptera: Culicidae) em Porto Velho, RO. Neotrop. Entomol. 2009;38(5):677-82.
40. Voorham J. Intra-population plasticity of Anopheles darlingi's (Diptera, Culicidae) biting activity patterns in the state of Amapá, Brazil. Rev Saúde Pública. 2002;36(1)75-80.
41. Charlwood JD, Hayes J. Variações geográficas no ciclo de picada do Anopheles darlingi Root no Brasil. Acta Amaz . 1978;8(4): 601-3.-4242. Consoli RAGB, Lourenço-de-Oliveira RL. Principais mosquitos de importância sanitária no Brasil. Rio de Janeiro: Editora da Fundação Oswaldo Cruz; 1994. 225p.. Such differences in behavior can be explained by the high chromosomal polymorphism that allows the species to be able to explore different habitats, especially during the rainy season4343. Tadei WP, Santos JMM, Rabbani MG. Biologia de Anofelinos amazônicos. V. Polimorfismo cromossômico de Anopheles darlingi Root (Diptera, Culicidae). Acta Amaz .1982;12(2):353-69.
44. Tadei WP, Santos JMM. Biologia de Anofelinos amazônicos. VII. Estudo da variação de freqüências das inversões cromossômicas de Anopheles darlingi Root (Diptera, Culicidae). Acta Amaz . 1982;12(4):759-85.-4545. Charlwood JD. Biological variation in Anopheles darlingi Root. Mem Inst Oswaldo Cruz . 1996;91(4):391-8..
An. aquasalis is found along coastal areas, in some localities that are approximately 100 and 200 km from the coast, and in soils rich in sodium chloride, as is the case in some areas of the northeastern backlands or in regions where tidal invasion occurs, such as Belém, Pará4646. Faran ME, Linthicum KJ. A handbook of the Amazonian species of Anopheles (Nyssorhynchus) (Diptera: Culicidae). Mosquito Systematics.1981;13(1):1-81.
47. Galvão ALA, Damasceno RG, Marques AP. Algumas observações sobre a biologia dos anofelinos de importância epidemiológica de Belém Pará. Arq Hig Saúde Púb.1942;12(2):51-110.
48. Rubio-Palis Y, Zimmerman RH. Ecoregional classification of malária vectors in the neotropics. J Med Entomol. 1997;34(5):499-510.-4949. Deane LM, Vernin SC, Damasceno RG. Avaliação das preferências alimentares das fêmeas de Anopheles darlingi e Anopheles aquasalis em Belém, Pará, por provas de precipitina. Rev Serv Esp Saúde Pública. 1949;2(3):793-808.. Galvão et al.4747. Galvão ALA, Damasceno RG, Marques AP. Algumas observações sobre a biologia dos anofelinos de importância epidemiológica de Belém Pará. Arq Hig Saúde Púb.1942;12(2):51-110. pointed to the preferential zoophilic profile of An. aquasalis in Belém, Pará, where the climate is equatorial; conversely, An. aquasalis in the Northeast region, which has a semi-arid climate, presents anthropophilic habits4949. Deane LM, Vernin SC, Damasceno RG. Avaliação das preferências alimentares das fêmeas de Anopheles darlingi e Anopheles aquasalis em Belém, Pará, por provas de precipitina. Rev Serv Esp Saúde Pública. 1949;2(3):793-808.
50. Rachou GR, Moura-Lima M, Barbosa AL. Considerações sobre o An. (N.) tarsimaculatus Goeldi, 1905 (An. (N.) aquasalis, Curry, 1932) no Estado do Ceará, com especial referência ao seu encontro a 52km da orla marítima. Rev Bras Malariol. 1950;2:57-65.
51. Lourenço-de-Oliveira R, Heyden R. Alguns aspectos da ecologia dos mosquitos (Diptera: Culicidae) de uma área de planície (Granjas Calábria), em Jacarepaguá, Rio de Janeiro. IV. Preferências alimentares quanto ao hospedeiro e freqüência domiciliar. Mem Inst Oswaldo Cruz . 1986;81(1):15-27.-5252. Flores-Mendoza C, Cunha RA, Rocha DS, Lourenço-de-Oliveira R. Determinação das fontes alimentares de Anopheles aquasalis (Diptera: Culicidae) no Estado do Rio de Janeiro, Brasil, pelo teste de precipitina. Rev Saúde Pública . 1996;30(2):129-34..
The albitarsis complex brings together the most common Anopheles mosquitoes widely distributed in Brazil, including An. oryzalimnetes, An. deaneorum, An. marajoara, and An. janconnae5353. Wilkerson RC, Gaffigan TV, Lima JB. Identification of species related to Anopheles (Nyssorhynchus) albitarsis by random amplified polymorphic DNA-polymerase chain reaction (Diptera: Culicidae). Mem Inst Oswaldo Cruz . 1995;90(6):721-32.
54. Wilkerson RC, Foster PG, Li C, Sallum MAM. Molecular phylogeny of Neotropical Anopheles (Nyssorhynchus) albitarsis species species complex (Diptera: Culicidae). Ann Ent Soc Amer. 2005;98(6):918-25.-5555. Motoki MT, Wilkerson RC, Sallum MAM. The Anopheles albitarsis complex with the recognition of Anopheles oryzalimnetes Wilkerson and Motoki, n. sp. and Anopheles janconnae Wilkerson and Sallum, n. sp. (Diptera: Culicidae). Mem Inst Oswaldo Cruz . 2009;104(6):823-50.. The cryptic species belonging to this complex cannot be distinguished morphologically in the adult phase; however, several methodologies were used for identification, including allozymes and DNA-based techniques5353. Wilkerson RC, Gaffigan TV, Lima JB. Identification of species related to Anopheles (Nyssorhynchus) albitarsis by random amplified polymorphic DNA-polymerase chain reaction (Diptera: Culicidae). Mem Inst Oswaldo Cruz . 1995;90(6):721-32.
54. Wilkerson RC, Foster PG, Li C, Sallum MAM. Molecular phylogeny of Neotropical Anopheles (Nyssorhynchus) albitarsis species species complex (Diptera: Culicidae). Ann Ent Soc Amer. 2005;98(6):918-25.
55. Motoki MT, Wilkerson RC, Sallum MAM. The Anopheles albitarsis complex with the recognition of Anopheles oryzalimnetes Wilkerson and Motoki, n. sp. and Anopheles janconnae Wilkerson and Sallum, n. sp. (Diptera: Culicidae). Mem Inst Oswaldo Cruz . 2009;104(6):823-50.
56. Wilkerson RC, Hribar LJ, Milstrey EG, Falero GC. The identification of Anopheles (Nyssorhynchus) rondoni (Diptera: Culicidae) in Mato Grosso, Brazil: an analysis of key character variability. Mem Inst Oswaldo Cruz . 1995;90(5):575-82.
57. Wilkerson RC, Parsons TJ, Klein TA, Gaffigan TV, Bergo E, Consolim J. Diagnosis by random amplified polymorphic DNA polymerase chain reaction of four cryptic species related to Anopheles (Nyssorhynchus) albitarsis (Diptera: Culicidae) from Paraguay, Argentina, and Brazil. J Med Entomol . 1995;32(5):697-704.
58. Li C, Wilkerson RC. Identification of Anopheles (Nyssorhynchus) albitarsis complex species (Diptera: Culicidae) using rDNA internal transcribed spacer 2-based polymerase chain reaction primes. Mem Inst Oswaldo Cruz . 2005;100(5):495-500.-5959. Lehr MA, Kilpatrick CW, Wilkerson RC, Conn JE. Cryptic species in the Anopheles (Nyssorhynchus) albitarsis (Diptera: Culicidae) complex: incongruence between random amplified polymorphic DNA-polymerase chain reaction identification and analysis of mitochondrial DNA COI gene sequences. Ann Entomol Soc Am. 2005;98(6):908-17.. The members of the albitarsis complex have large distributions, as well as high behavioral variabilities6060. Li C, Wilkerson RC. Intragenomic rDNA ITS2 variation in the neotropical Anopheles (Nyssorhynchus) albitarsis complex (Diptera: Culicidae). J Hered. 2007;98(1):51-9.. Their preferred breeding sites are clean freshwater reservoirs, preferably in sunny or shaded fields and pastures. In most of the Brazilian territory, they present zoophilic and exophilic habits6060. Li C, Wilkerson RC. Intragenomic rDNA ITS2 variation in the neotropical Anopheles (Nyssorhynchus) albitarsis complex (Diptera: Culicidae). J Hered. 2007;98(1):51-9..
The anophelines An. cruzii and An. bellator breed in bromeliads in the Atlantic Forest and have acrodendrophic habits, a behavioral characteristic of mosquitoes of the subgenus Kerteszia, especially An. cruzii; this refers to the preference of these vectors in practicing hematophagy in the crown of trees, occasionally descending to the ground, which results in the accomplishment of repasts in the two heights of the forest4242. Consoli RAGB, Lourenço-de-Oliveira RL. Principais mosquitos de importância sanitária no Brasil. Rio de Janeiro: Editora da Fundação Oswaldo Cruz; 1994. 225p.. An. cruzii is restricted to the Brazilian coast, where it is a transmitter of the Plasmodium vivax, P. simium, and P. brasilianum, causing human and simian malaria6161. Branquinho MS, Marrelli MT, Curado I, Natal D, Barata JMS, Tubaki R, et al. Infecção do Anopheles (Kerteszia) cruzii por Plasmodium vivax e Plasmodium vivax variante VK247 nos Municípios de São Vicente e Juquitiba, São Paulo. Rev Panam Salud Publica /Pan Am J Public Health. 1997;2(3):189-93.
62. Deane LM, Ferreira-Neto JA, Lima MM. The vertical dispersion of Anopheles (Kerteszia) cruzii in a forest in southern Brazil suggests that human cases of malaria of simian origin might be expected. Mem Inst Oswaldo Cruz . 1984;79(4):461-3.
63. Deane LM. Simian malaria in Brazil. Mem Inst Oswaldo Cruz 1992,87(Suppl 3):1-20.
64. Forattini OP, Gomes AC, Natal D, Santos JLF. Observações sobre atividade de mosquitos Culicidae em mata primitiva da encosta no Vale do Ribeira, São Paulo, Brasil. Rev Saúde Pública . 1986;20(1):1-20.
65. Forattini OP, Gomes AC, Natal D, Santos JLF. Observações sobre atividade de mosquitos Culicidae em matas primitivas da planície e perfis epidemiológicos de vários ambientes no Vale do Ribeira, São Paulo, Brasil. Rev Saúde Pública 1986;20(3):178-203.
66. Forattini OP, Gomes AC, Santos JLF, Kakitani I, Marucci D. 1990. Freqüência ao ambiente humano e dispersão de mosquitos Culicidae em área adjacente à Mata Atlântica primitiva da planície. Rev Saúde Pública . 1990;24 (2):101-7.
67. Forattini OP, Kakitani I, Massad E, Gomes AC. Studies on mosquitoes (Diptera: Culicidae) and anthropic environment. 1- Parity of blood seeking Anopheles (Kerteszia) in South-Eastern Brazil. Rev Saúde Pública .1993;27(1):1-8.
68. Tubaki RM, Carréri-Bruno GC, Moreno Glasser C, Ciaravolo RMC. Biting activity of Anopheles (Kerteszia) cruzii (Diptera, Culicidae) in domiciliary habitats in the Southern Atlantic Forest, Peruibe, State of São Paulo, Brazil. Rev Bras Entomol. 1993;37(3):569-75.-6969. Curado I, Duarte AMRC, Lal AA, Oliveira SG, Kloetzel JK. Antibodies anti bloodstream and circumsporozoite antigens (Plasmodium vivax and Plasmodium malariae/P. brasilianum) in areas of very low malaria endemicity in Brazil. Mem Inst Oswaldo Cruz . 1997; 92(2):235-43.. It is found in peridomiciliary environments, with its 24-hour activity peaking in the crepuscular periods of the day6565. Forattini OP, Gomes AC, Natal D, Santos JLF. Observações sobre atividade de mosquitos Culicidae em matas primitivas da planície e perfis epidemiológicos de vários ambientes no Vale do Ribeira, São Paulo, Brasil. Rev Saúde Pública 1986;20(3):178-203.,6767. Forattini OP, Kakitani I, Massad E, Gomes AC. Studies on mosquitoes (Diptera: Culicidae) and anthropic environment. 1- Parity of blood seeking Anopheles (Kerteszia) in South-Eastern Brazil. Rev Saúde Pública .1993;27(1):1-8.,6868. Tubaki RM, Carréri-Bruno GC, Moreno Glasser C, Ciaravolo RMC. Biting activity of Anopheles (Kerteszia) cruzii (Diptera, Culicidae) in domiciliary habitats in the Southern Atlantic Forest, Peruibe, State of São Paulo, Brazil. Rev Bras Entomol. 1993;37(3):569-75.. An. bellator is found only on the coast and presents eclectic habits regarding hosts. It has preference in breeding in bromeliads exposed to the sun7070. Lorenz C, Marques TC, Sallum MAM, Suesdek L. Morphometrical diagnosis of the malaria vectors Anopheles cruzii, An. homunculus and An. bellator. Parasit Vectors . 2012;5(1):257..
VECTOR CONTROL IN BRAZIL
Vector control is an essential component of malaria prevention7171. World Health Organization (WHO) & Global Partnership to Roll Back Malaria. Malaria vector control and personal protection: report of a WHO study group. Geneva: WHO ; 2006. 62p.. In Brazil, it was initially performed through early management of mosquitoes with the use of Paris green and petroleum derivatives55. Gadelha P. Conforming strategies of public health campaigns to disease specificity and national contexts: Rockefeller Foundation’s early campaigns against hookworm and malaria in Brazil. Parassitologia. 1998;40(1-2):159-75.,77. Pinto C. Disseminação da malaria pela aviação: biologia do Anopheles gambiae e outros anofelíneos do Brasil. Mem Inst Oswaldo Cruz . 1939;34:293-430.,99. Camargo EP. Malária, maleita, paludismo. Cienc Cult. 2003;55(1):26-9.. With the discovery of DDT, the focus of malaria control strategies shifted to managing the adult mosquito population. DDT spraying was officially conducted throughout the Amazon in 19601212. Deane LM. Malaria studies and control in Brazil. Am J Trop Med Hyg . 1988;38(2):223-30.. Indoor spraying of DDT was banned in Brazil in 1999 because of its ecotoxicological risks, and currently, several classes of insecticides have been used, with special emphasis on pyrethroids, etofenprox PM 20% and lambdacyalothrin CE 5%7272. Guimarães RM, Asmus CI, Meyer A. DDT reintroduction for malaria control: the cost-benefit debate for public health. Cad Saude Publ . 2007;23(12):2835-44.,7373. Akhavan D, Musgrove P, Abrantes A, d'A Gusmão R. Cost-effective malaria control in Brazil. Cost-effectiveness of a Malaria Control Program in the Amazon Basin of Brazil, 1988-1996. Soc Sci Med. 1999;49(10):1385-99.. Further, different methods for vector control have been used in the field (Figure 1).
Methods used for malaria vector control. (A) Use of an insecticide-treated bed net; (B) fogging; (C) indoor residual spraying; and (D) larval source management.
INTERVENTIONS BASED ON IRS
Aimed at adult mosquitoes, IRS consists of spraying the interior walls of houses with insecticides7171. World Health Organization (WHO) & Global Partnership to Roll Back Malaria. Malaria vector control and personal protection: report of a WHO study group. Geneva: WHO ; 2006. 62p.,7474. World Health Organization (WHO). Department of Control of Neglected Tropical Diseases. Manual for indoor residual spraying: application of residual sprays for vector control, third edition. Geneva: WHO Pesticide evaluation scheme (WHOPES) ; 2007. 43p.. Once applied, the insecticide will form a film of small crystals on the surface, killing the vector when it rests on the wall before or after a meal of blood7575. World Health Organization (WHO). Malaria entomology and vector control. Guide for Tutors. 2nd edition. Geneva: WHO ; 2013. 50p.. Some of the insecticides used in IRS are also capable of repelling mosquitoes, reducing the number of vectors entering the households. It has been the most widely used method in malaria vector control7575. World Health Organization (WHO). Malaria entomology and vector control. Guide for Tutors. 2nd edition. Geneva: WHO ; 2013. 50p., with some official guidelines available7575. World Health Organization (WHO). Malaria entomology and vector control. Guide for Tutors. 2nd edition. Geneva: WHO ; 2013. 50p.
76. World Health Organization (WHO). Indoor residual spraying. An operational manual for IRS for malaria transmission control and elimination. Second edition. Geneva: WHO ; 2015. 134p.
77. Ministério da Saúde (MS). Secretaria de Vigilância em Saúde. Departamento de Vigilância Epidemiológica. Diretoria Técnica de Gestão. Ações de Controle da Malária: Manual para Profissionais de Saúde na Atenção Básica. Brasília: Editora do Ministério da Saúde; 2006. 52p.-7878. Centro de Controle e Prevenção de Doenças (CDC). Organização Pan-Americana da Saúde/Organização Mundial de Saúde (OPAS/OMS), e RTI. Iniciativa Amazônica contra a Malária/Rede Amazônica de Vigilância da Resistência às Drogas Antimaláricas. Documento de Orientação Estratégica para Vigilância e Controle de Vetores da Malária na América Latina e no Caribe. Apresentado à Agência dos Estados Unidos para o Desenvolvimento Internacional (USAID). 2011. 42p..
Some researchers have evaluated the effectiveness of different IRS insecticides used in Brazil. Roberts and Alecrim7979. Robefcts DR, Alecrim WD. Behavioral response of Anopheles darlingi to DDT-sprayed house walls in Amazonia. Bull Pan Am Health Organ. 1991;25(3):210-7. investigated the behavioral alterations in female An. darlingi mosquitoes after DDT spraying, observing that they stopped entering and leaving houses, with a reduction in the bite rate. However, IRS did not interfere with the persistence of malaria in the study area, probably owing to the sprayed houses presenting incomplete walls. In their field trial, Charlwood et al.8080. Charlwood JD, Alecrim WD, Fe N, Mangabeira J, Martins VJ. A field trial with Lambda-cyhalothrin (ICON) for the intradomiciliary control of malaria transmitted by Anopheles darlingi root in Rondonia, Brazil. Acta Trop. 1995;60(1):3-13. compared the effects of lambdacyalothrin (ICON) and DDT when used as residual sprays on the internal surfaces of houses. ICON killed more mosquitoes than did DDT. From the observed efficacy, prompt acceptance by the local population, and cost-effectiveness of ICON, the authors have suggested its use in antimalaria campaigns. Santos et al.8181. Santos RC, Fayal AS, Aguiar AEF, Vieira DB, Póvoa MM. Evaluation of the residual effect of pyrethroids on Anopheles in the Brazilian Amazon. Rev Saude Publica. 2007;41(2):276-83. evaluated the residual effect of pyrethroids on Anopheles mortality. The insecticides used were a suspension of deltamethrin, pyrethroids, lambda-cyhalothrin, and etofenprox in wettable powder, which were sprayed onto the surface of local dwellings. The authors observed that the insecticides applied to wood and uneven surfaces were more stable and lasted longer. Based on the findings of the study by Santos et al.8181. Santos RC, Fayal AS, Aguiar AEF, Vieira DB, Póvoa MM. Evaluation of the residual effect of pyrethroids on Anopheles in the Brazilian Amazon. Rev Saude Publica. 2007;41(2):276-83. the NMCP changed the insecticide from alpha-cypermethrin to etofenprox in 2013.
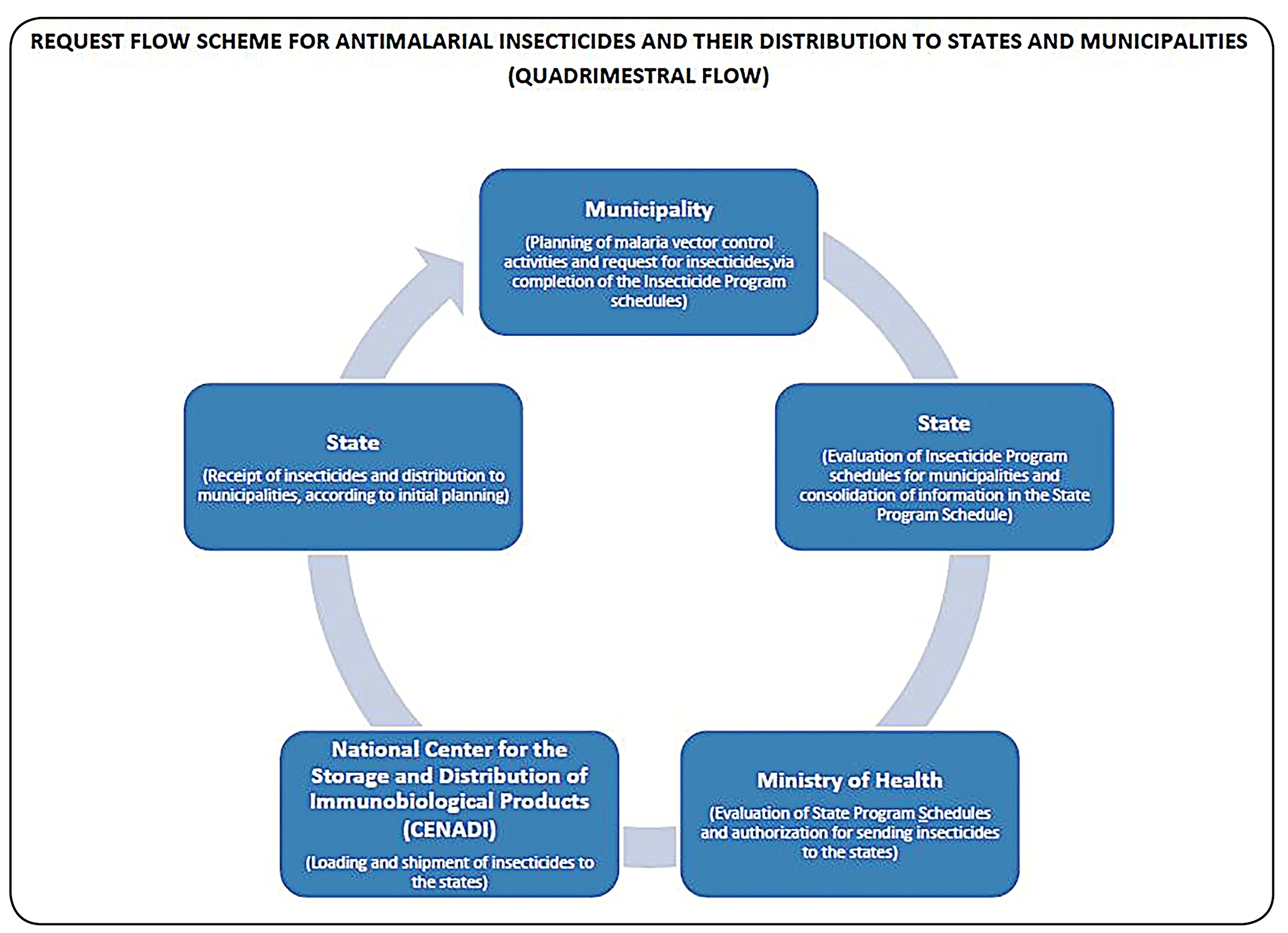
Etofenprox PM 20% is the insecticide used by the Ministry of Health for residual spraying in houses for vector control of malaria. This product has a residual effect for 4 months8181. Santos RC, Fayal AS, Aguiar AEF, Vieira DB, Póvoa MM. Evaluation of the residual effect of pyrethroids on Anopheles in the Brazilian Amazon. Rev Saude Publica. 2007;41(2):276-83.; therefore, three annual application cycles are required. This insecticide is applied by municipalities quarterly by filling out a standardized worksheet with the following information: (i) number of properties to be worked on in each cycle and (ii) number of malaria cases in the same period of the previous year and updated inventory of the insecticide in the municipality (Figure 2). For the Indigenous Special Sanitary Districts (DSEIs), etofenprox PM 20% application is conducted by sending a request with the abovementioned data directly to the Ministry of Health (Figure 3).
Flow chart presenting the distribution of vector control supplies in indigenous Brazilian areas.
Although the work performed by different authors in Amazonian communities showed the role of IRS, it is still necessary to evaluate the effectiveness and costs of this action more specifically, as it is mainly related to the reduction of the morbidity of malaria in the Amazon.
ITNS
Rapid coverage of the population at risk through free and/or subsidized distribution of ITNs has been recommended by the WHO as a primary intervention for malaria control based on evidence of efficacy and effectiveness that this intervention has demonstrated in Africa, Southeast Asia, and South America8282. Lengeler C. Insecticide-treated bed nets and curtains for preventing malaria. Cochrane Database Syst Rev. 2004;(2):CD000363.. Currently, there are long-lasting insecticidal nets (LLINs) that have a shelf life of approximately 2-3 years in the field or should be effective for 20 laboratory washes, with pyrethroid insecticides incorporated in the fabric8383. World Health Organization (WHO). Insecticide-treated mosquito net interventions: A manual for national control programme managers. Roll Back Malaria, editor. Geneve: WHO; 2003. 130p.,8484. World Health Organization (WHO). Guidelines for laboratory and field-testing of long-lasting insecticidal nets. Geneva: WHO ; 2013. 89p..
ITNs emerged in the 1990s as one of the great hopeful methods of controlling malaria worldwide. In Brazil, the Ministry of Health started to distribute ITNs freely in malaria-endemic areas in 20078585. da Silva-Nunes M, Moreno M, Conn JE, Gamboa D, Abeles S, Vinetz JM, et al. Amazonian malaria: asymptomatic human reservoirs, diagnostic challenges, environmentally driven changes in mosquito vector populations, and the mandate for sustainable control strategies. Acta Trop . 2012;121(3):281-91.. Between 2012 and 2014, there was an increase in the number of protected individuals with this strategy; however, there were discontinuity and subsequent reduction in the following years. LLINs impregnated with deltamethrin have been used in the Amazon3838. Santos JB, Santos F, Macedo V. Variação da densidade anofélica com o uso de mosquiteiros impregnados com deltametrina em uma área endêmica de malária na Amazônia Brasileira. Cad Saúde Pública 1999;15(2):281-92.; however, the results have been contradictory, and there is limited knowledge on the effectiveness of ITNs in affecting the morbidity and mortality of malaria. The biting behavior of An. darlingi mosquitoes can be the main reason why ITBN-based malaria control programs may fail in this region3838. Santos JB, Santos F, Macedo V. Variação da densidade anofélica com o uso de mosquiteiros impregnados com deltametrina em uma área endêmica de malária na Amazônia Brasileira. Cad Saúde Pública 1999;15(2):281-92.,8282. Lengeler C. Insecticide-treated bed nets and curtains for preventing malaria. Cochrane Database Syst Rev. 2004;(2):CD000363.,8686. Xavier PA, Lima JENS. O uso de cortinas impregnadas com deltametrina no controle da malária em garimpos no Território Federal do Amapá. Rev Bras Malariol Doenç Trop.1986;38:137-9.
87. Salgado-Cavalcante ET, Tadei WP, Pinto CT, Xavier PA, Lima IENS. Efeitos da ação residual da deltametrina, em cortinas de ráfia e sarrapilha no controle da malária, em áreas de garimpo, no Estado do Amapá. Rev Soc Bras Med Trop . 1992;25(Supl I):6-7.
88. Galardo CD, Galardo AKR. Relatório técnico sobre o Estudo da Eficácia de Redes Impregnadas com Inseticidas e do uso de de Fendona® em borrifações domiciliares para o Controle de Anopheles sp. em Bioensaios de Campo e Laboratório no Estado do Amapá - Brasil. Departamento de Zoologia-Laboratório de Entomologia Médica, Instituto de Pesquisas Científicas e Tecnolócias do Estado do Amapá, Macapá. 2009. 29p.-8989. Vieira GD, Basano SA, Katsuragawa TH, Camargo LM. Insecticide-treated bed nets in Rondônia, Brazil: evaluation of their impact on malaria control. Rev Inst Med Trop Sao Paulo. 2014;56(6):493-7..
In Brazil, the first work with ITNs was performed by Xavier and Lima8686. Xavier PA, Lima JENS. O uso de cortinas impregnadas com deltametrina no controle da malária em garimpos no Território Federal do Amapá. Rev Bras Malariol Doenç Trop.1986;38:137-9.and Cavalcante et al.8787. Salgado-Cavalcante ET, Tadei WP, Pinto CT, Xavier PA, Lima IENS. Efeitos da ação residual da deltametrina, em cortinas de ráfia e sarrapilha no controle da malária, em áreas de garimpo, no Estado do Amapá. Rev Soc Bras Med Trop . 1992;25(Supl I):6-7. who demonstrated the efficiency of DDT and deltamethrin and the effective repellent effect for up to 120 days, persisting even longer in locations not exposed to sunlight. Santos et al.4040. Voorham J. Intra-population plasticity of Anopheles darlingi's (Diptera, Culicidae) biting activity patterns in the state of Amapá, Brazil. Rev Saúde Pública. 2002;36(1)75-80. showed that ITNs used in Costa Marques, Rondônia did not change the risk of Plasmodium infection and did not reduce the cases of malaria and the average parasitemia associated with infection as observed in other continents. Probably, the protection failure of nets may have been attributed to bites occurring before entering and after leaving the mosquito net, to the more frequent exits during the night, and to the exophilic and endophilic behaviors of the main vector species in the region. These authors also showed that ITNs significantly decreased the number of Anopheles mosquitoes captured; however, impregnated and non-impregnated ITNs showed similar actions on the peridomiciliar density of mosquitoes. Still in Brazil, Galardo and Galardo8888. Galardo CD, Galardo AKR. Relatório técnico sobre o Estudo da Eficácia de Redes Impregnadas com Inseticidas e do uso de de Fendona® em borrifações domiciliares para o Controle de Anopheles sp. em Bioensaios de Campo e Laboratório no Estado do Amapá - Brasil. Departamento de Zoologia-Laboratório de Entomologia Médica, Instituto de Pesquisas Científicas e Tecnolócias do Estado do Amapá, Macapá. 2009. 29p. conducted a study in Mazagão, Amapá, evaluating the residual effect of ITNs impregnated with alpha-cypermethrin and observing an 80% reduction in malaria cases in the municipality. In 2012, approximately 150,000 LLINs were installed in nine municipalities in the state of Rondônia; Vieira et al.8989. Vieira GD, Basano SA, Katsuragawa TH, Camargo LM. Insecticide-treated bed nets in Rondônia, Brazil: evaluation of their impact on malaria control. Rev Inst Med Trop Sao Paulo. 2014;56(6):493-7. evaluated the use of LLINs in nine cities and compared the outcomes with those in cities where LLINs were not used. However, no significant differences were observed in the annual parasite incidence 1 year after LLIN installation.
OTHER VECTOR CONTROL MEASURES
Space and aerial spraying
A spatial spray (fog) is a liquid insecticide dispersed into the air in the form of hundreds of millions of tiny droplets less than 50 µm in diameter9090. World Health Organization (WHO). Space spray application of insecticides for vector and public health pest control: a practitioner's guide. Geneva: WHO ; 2003. 49p.. Currently, neither space spraying nor aerial spraying has a WHO policy recommendation for use in malaria vector control9191. World Health Organization (WHO). Malaria vector control policy recommendations and their applicability to product evaluation. Geneva: WHO ; 2017. 8p.. Fogging might be justified for control of certain exophilic and exophagic vectors and during malaria epidemics, especially in camps for internally displaced individuals, where infective mosquitoes must be eliminated rapidly2626. Ministério da Saúde (MS). Secretaria de Vigilância em Saúde. Diretoria Técnica de Gestão. Guia para Gestão Local do Controle da Malária: Módulo 2: Controle Vetorial. Série B. Textos Básicos de Saúde. Brasília: MS; 2009. 59p.,9292. World Health Organization (WHO). WHO recommended insecticides for space spraying against mosquitoes. Geneva: WHO . Updated 5 February 2016.. This will be important in vector control only in epidemiological emergencies and should never be used as a routine activity9292. World Health Organization (WHO). WHO recommended insecticides for space spraying against mosquitoes. Geneva: WHO . Updated 5 February 2016.. In Brazil, thermal fogging was selectively and exceptionally used in specific areas depending on entomological and epidemiological conditions, including areas of mining or those with lack of vegetation, as was the case in the Belo Monte site9393. Ladislau JLB, Póvoa MM, Sucupira IMC, Galardo AKR, Damasceno OC, Cardoso BS, et al. Malaria control in area of hydroelectric construction on the Amazonian ecosystem can succeed? Rev Pan-Amaz Saude. 2016;7(num esp,):115-22.. Measures, such as thermal fogging, are widely used in the Amazon for malaria control; however, they may be only slightly effective if not performed according to the mosquitoes’ blood feeding activity9494. Santos RC, Padilha A, Costa MDP, Costa EM, Dantas-Filho HC, Povoa MM. Vetores de malária em duas reservas indígenas da Amazônia Brasileira. Rev Saúde Pública . 2009;43(5):859-68.. Further, there is no evidence of the impact of nebulization or spatial spraying of insecticides on malaria transmission.
Lambdacyalothrin CE 5% is the insecticide currently used in Brazil for thermonebulization in malaria control, recommended only in epidemic situations; its application is not a routine activity of the malaria program. The application follows the same flow for municipalities and the Departamento de Saúde Especial Indígenas (DSEIs), but without the use of programming worksheets (Figure 2).
Larval source management (LSM)
LSM targets the immature, aquatic stages of mosquitos (i.e., larvae and pupae), thereby reducing the abundance of adult vectors9595. Silva JJ, Mendes J. Susceptibility of Aedes aegypti (L) to the insect growth regulators diflubenzuron and methoprene in Uberlândia, State of Minas Gerais. Rev Soc Bras Med Trop . 2007;40(6), 612-6.. The use of bacterial entomopathogens, such as Bacillus sphaericus and B. thuringiensis israelensis (Bti), is an alternative to conventional control measures against Anopheles larvae9696. Tadei WP, Rodrigues IB, Rafael MS, Sampaio RTM, Mesquita HG, Pinheiro VCS, et al. Adaptative processes, control measures, genetic background, and resilience of malaria vectors and environmental changes in the Amazon region. Hydrobiologia. 2017;789(1):179-96.,9797. Palma L, Muñoz D, Berry C, Murillo J, Caballero P. Bacillus thuringiensis toxins: an overview of their biocidal activity. Toxins (Basel). 2014;6(12):3296-325.. Bti is a bacterium that produces proteins, such as δ-endotoxins, which are toxic to larvae of several insects9898. Roh JY, Choi JY, Li MS, Jin BR, Je YH. Bacillus thuringiensis as a specific, safe, and effective tool for insect pest control. J Mol Biol. 2007;17(4):547-59.,9999. World Health Organization (WHO). Larval source management: a supplementary measure for malaria vector control. An operational manual. Geneva: WHO ; 2013. 116p.. Rodrigues et al.100100. Rodrigues BI, Tadei WP, Dias JMSD, Lima CAP. Atividade Larvicida de Bacillus sphaericus 2362 contra Anopheles sp. (Diptera, Culicidae) em rios do Amazonas, Brasil. Bio Assay. 2013;8(2):1-7.,101101. Rodrigues IB, Tadei WP, Dias JMCS. Larvicidal activity Bacillus sphaericus 2362 against Anopheles nuneztovari, Anopheles darlingi and Anopheles braziliensis (Diptera, Culicidae). Rev Inst Med Trop S Paulo. 1999;41(2):101-5. analyzed the larvicidal activity of B. sphaericus against larvae of anophelines in Amazonian conditions. They showed that the third larval instar of An. nuneztovari and the second and third larval instars of An. darlingi proved to be the least susceptible, while An. braziliensis was the most susceptible.
Galardo et al.102102. Galardo AKR, Zimmerman R, Galardo CD. Larval control of Anopheles (Nyssorhinchus) darlingiusing granular formulation of Bacillus sphaericus in abandoned gold-miners excavation pools in the Brazilian Amazon rainforest. Rev Soc Bras Med Trop . 2013;46 (2):172-7. evaluated an intervention for malaria vector control using VectoLex(r) CG (granular formulation of B. sphaericus 2362 strain) at a mining site in Calçoene, Amapá. The VectoLex(r) reduced immature An. darlingi infestation levels during the entire study period and reduced adult mosquito populations during the rainy season.
Additionally, there are insect growth regulators, which prevent emergence of adults from the pupal stage. The most common compounds are diflubenzuron, methoprene, novaluron, and pyriproxyfen9595. Silva JJ, Mendes J. Susceptibility of Aedes aegypti (L) to the insect growth regulators diflubenzuron and methoprene in Uberlândia, State of Minas Gerais. Rev Soc Bras Med Trop . 2007;40(6), 612-6..
INSECTICIDE RESISTANCE
Insecticide resistance is the ability of insects to survive exposure to a standard dose of insecticide, owing to physiological or behavioral adaptation103103. Chareonviriyaphap T, Bangs MJ, Suwonkerd W, Kongmee M, Corbel V, Ngoen-Klan R. Review of insecticide resistance and behavioral avoidance of vectors of human diseases in Thailand. Parasit Vectors . 2013;6:280.. The molecular basis of insecticide resistance has been justified by the existence of mutations in insecticide target site genes and alterations in related metabolic pathways, including primarily the detoxification activity104104. World Health Organization (WHO). Test procedures for insecticide resistance monitoring in malaria vectors, bio-efficacy and persistence of insecticide on treated surfaces: report of the WHO informal consultation. Geneva: WHO ; 1998. 43p.. Behavioral resistance results from sub-lethal exposure to the active ingredient of insecticides and involves behavioral changes, resulting in avoidance and reduced contact with lethal doses of an insecticide105105. Sparks TC, Lockwood JA, Byford RL, Graves JB, Leonard BR. The role of behavior in insecticide resistance. Pestic Sci. 1989;26(4):383-99.. Although the mechanisms of resistance to insecticides are known, the impact of resistance on the ability of malaria control interventions to reduce disease transmission is poorly understood106106. Philbert A, Lyantagaye SL, Nkwengulila G. A review of agricultural pesticides use and the selection for resistance to insecticides in malaria vectors. Adv Entomol. 2014;2(3):120-8..
Through the Global Plan for Insecticide Resistance Management in Malaria Vectors created by the WHO107107. World Health Organization (WHO). Global plan for insecticide resistance management in malaria vectors. Geneva: WHO ; 2012. 132p., the basis of any national vector control strategy was constructed, which includes a system based on epidemiological and entomological monitoring and monitoring of resistance to insecticides used, based on bioassays using WHO paper bioassays104104. World Health Organization (WHO). Test procedures for insecticide resistance monitoring in malaria vectors, bio-efficacy and persistence of insecticide on treated surfaces: report of the WHO informal consultation. Geneva: WHO ; 1998. 43p. or CDC bottle bioassays108108. Brogdon WG, Chan A. Diretriz para avaliar a resistência a inseticida em vetores usando o bioensaio com garrafa do CDC. 2018. 2012. 28p. Disponível em: https://www.cdc.gov/malaria/resources/pdf/fsp/ir_manual/ir_cdc_bioassay_pt.pdf
https://www.cdc.gov/malaria/resources/pd...
. Ecological, epidemiological, and susceptibility information will help determine the proper use of insecticides to mitigate or delay the development of resistance.
Galardo et al.109109. Galardo AKR, Póvoa MN, Sucupira IMC, Galardo CD, Santos RC. Anopheles darlingi and Anopheles marajoara (Diptera: Culicidae) susceptibility to pyrethroids in an endemic area of the Brazilian Amazon. Rev Soc Bras Med Trop . 2015;48(6):765-9. evaluated the susceptibility of anophelines from Amapá to pyrethroids used by the NMCP, and no resistance was recorded for An. darlingi; however, An. marajoara requires attention. Silva et al.110110. Silva APB, Alves WS, Martins AJ, Tadei WP, Santos JMM. Adaptação de um bioensaio simplificado para avaliação do status de susceptibilidade em larvas de Anopheles darlingi e Anopheles marajoara ao piretroide deltametrina. BioAssay. 2014;9(4):1-8. evaluated the susceptibility profile of pyrethroid insecticides on An. darlingi and An. marajoara larvae. In this study, loss of susceptibility was observed in the populations of Manaus and Iranduba, possibly owing to the selection effect of the insecticides on the specific resistance alleles. Resistant mosquito populations may also appear owing to the long-term use of a single insecticide for malaria control vectors.
OFFICIAL RECOMMENDATIONS FROM THE BRAZILIAN MALARIA CONTROL PROGRAM
Two guidelines for malaria control are available from the Ministry of Health for professionals7777. Ministério da Saúde (MS). Secretaria de Vigilância em Saúde. Departamento de Vigilância Epidemiológica. Diretoria Técnica de Gestão. Ações de Controle da Malária: Manual para Profissionais de Saúde na Atenção Básica. Brasília: Editora do Ministério da Saúde; 2006. 52p. and local management2626. Ministério da Saúde (MS). Secretaria de Vigilância em Saúde. Diretoria Técnica de Gestão. Guia para Gestão Local do Controle da Malária: Módulo 2: Controle Vetorial. Série B. Textos Básicos de Saúde. Brasília: MS; 2009. 59p.. Through the Guide to Local Management of Malaria Control2626. Ministério da Saúde (MS). Secretaria de Vigilância em Saúde. Diretoria Técnica de Gestão. Guia para Gestão Local do Controle da Malária: Módulo 2: Controle Vetorial. Série B. Textos Básicos de Saúde. Brasília: MS; 2009. 59p., the Ministry of Health establishes important recommendations on the methodologies for controlling anopheline malaria vectors in the country. According to this guide, vector control should follow the principles of selective and integrated control, with community participation and adjusted to the eco-epidemiological situations of each municipality. For this, the central axis of the vector control is the timely use of epidemiological information and the consolidation of a work routine in entomology with robust data on the parameters that must be monitored to support decision-making2626. Ministério da Saúde (MS). Secretaria de Vigilância em Saúde. Diretoria Técnica de Gestão. Guia para Gestão Local do Controle da Malária: Módulo 2: Controle Vetorial. Série B. Textos Básicos de Saúde. Brasília: MS; 2009. 59p..
A careful and systematic analysis of the effect of control actions on malaria behavior depends essentially on the adequate completion and use of the information from the Malaria Epidemiological Surveillance System (Sivep-Malária)2626. Ministério da Saúde (MS). Secretaria de Vigilância em Saúde. Diretoria Técnica de Gestão. Guia para Gestão Local do Controle da Malária: Módulo 2: Controle Vetorial. Série B. Textos Básicos de Saúde. Brasília: MS; 2009. 59p.. After identification of the priority areas, previously collected and updated local characterization information will help select the localities needing IRS and/or use of LLINs and those where control measures for breeding sites are necessary. In addition, adequate training and good maintenance of equipment avoid risks to human and environmental health, as well as economic losses2626. Ministério da Saúde (MS). Secretaria de Vigilância em Saúde. Diretoria Técnica de Gestão. Guia para Gestão Local do Controle da Malária: Módulo 2: Controle Vetorial. Série B. Textos Básicos de Saúde. Brasília: MS; 2009. 59p..
IDENTIFYING RESEARCH PRIORITIES IN VECTOR CONTROL
In the national meeting on malaria research that took place in Recife, Pernambuco, in 2018, some gaps were identified for malaria vector control in Brazil. Considering such, some of the needs and priorities for future research and interventions include as follows:
Update of the cartography of malaria vectors in Brazil, adding molecular techniques for the correct identification of species and complexes of species;
Evaluation of vector competence of anopheline species in Brazil;
-
Strengthening of local entomology teams to perform vector control measures and interpret results;
-
Evaluation of vector control measures, especially the use of ITNs and LLINs, through community trials, estimating their effectiveness, cost-benefit, and population acceptance;
-
Establishment of colonies of malaria-transmitting vectors in Brazil, with emphasis on An. darlingi, to understand parasite-vector interactions better;
-
Study of new vector control strategies with impact on non-endophilic vectors;
-
Estimation of the impact of insecticide resistance in different geographical areas, considering the insecticides indicated by the NMCP;
-
Identification of the relative contribution of natural and artificial breeding sites in different epidemiological contexts for transmission
FINAL REMARKS
Vector control is an essential component of malaria prevention. In summary, incorporating vector control interventions, such as use of ITNs and IRS, is suggested for malaria elimination. Further confirmation from interventional studies is crucial to provide additional evidence for updating the malaria elimination policies in the territory. Vector control surveys are needed to address responses to changes in the NMCP guidelines, especially in the context of changes to eliminate malaria transmission in Brazil.
Acknowledgments
We thank Sheila Victor and Joabi Nascimento for kindly providing the images regarding field vector control measures for the study.
REFERENCES
-
1World Health Organization (WHO). World Malaria Report 2017. Geneva: WHO; 2017, 196p.
-
2Sampaio VS, Siqueira AM, Alecrim MGC, Mourão MPG, Marchesini PB, Albuquerque BC, et al. Malaria in the State of Amazonas: a typical Brazilian tropical disease influenced by waves of economic development. Rev Soc Bras Med Trop. 2015;48(Suppl I):4-11.
-
3Coura JR, Suárez-Mutis M, Ladeia-Andrade S. A new challenge for malaria control in Brazil: asymptomatic Plasmodium infection - a review. Mem Inst Oswaldo Cruz. 2006;101(3):229-37.
-
4Griffing SM, Tauil PL, Udhayakumar V, Silva-Flannery L. A historical perspective on malaria control in Brazil. Mem Inst Oswaldo Cruz . 2015;110(6):701-18.
-
5Gadelha P. Conforming strategies of public health campaigns to disease specificity and national contexts: Rockefeller Foundation’s early campaigns against hookworm and malaria in Brazil. Parassitologia. 1998;40(1-2):159-75.
-
6Parmakelis A, Russello MA, Caccone A, Marcondes CB, Costa J, Forattini OP, et al. Historical analysis of a near disaster: Anopheles gambiae in Brazil. Am J Trop Med Hyg. 2008;78(1):176-8.
-
7Pinto C. Disseminação da malaria pela aviação: biologia do Anopheles gambiae e outros anofelíneos do Brasil. Mem Inst Oswaldo Cruz . 1939;34:293-430.
-
8Packard RM, Gadelha P. A land filled with mosquitoes: Fred L Soper. The Rockefeller Foundation and the Anopheles gambiae invasion of Brazil. Med Anthropol. 1997;17(3):215-38.
-
9Camargo EP. Malária, maleita, paludismo. Cienc Cult. 2003;55(1):26-9.
-
10Stepan NL. Picturing Tropical Nature. Ithaca: Cornell University Press: 2001. 283p.
-
11Stepan NL. The only serious terror in these regions: malaria control in the Brazilian Amazon. Armus D, editor. Disease in the History of Modern Latin America: from Malaria to AIDS. Durham: Duke University Press: 2003. p. 25-50.
-
12Deane LM. Malaria studies and control in Brazil. Am J Trop Med Hyg . 1988;38(2):223-30.
-
13Ferreira MU, Castro MC. Challenges for malaria elimination in Brazil. Malar J. 2016;15(1):284.
-
14The Rockefeller Foundation. Annual Report, 1931. New York: The Rockefeller Foundation; 1931. 420p.
-
15Tauil P, Deane LM, Sabroza P, Ribeiro C. Malaria in Brazil. Cad Saude Publ. 1985;1(1):71-111.
-
16Killeen GF, Fillinger U, Kiche I, Gouagna LC, Knols BGJ. Eradication of Anopheles gambiae from Brazil: lessons for malaria control in Africa? Lancet Infect Dis. 2002;2(10):618-27.
-
17Packard RM. The making of a Tropical Disease: a Short History of Malaria. Baltimore: Johns Hopkins University Press; 2007. 320p.
-
18The Rockefeller Foundation. Annual Report, 1939. The Rockefeller Foundation; New York: 1939. 517p.
-
19Soper FL. Paris Green in the Eradication of Anopheles gambiae: Brazil, 1940; Egypt, 1945. Mosquito News. 1966;26(4):470-6.
-
20Silveira AC, de Rezende DF. Avaliação da estratégia global de controle integrado da malária no Brasil. Brasília: Organização Pan-Americana da Saúde; 2001. 120p.
-
21Alves MJCP, Mayo RC, Donalisio MR. História, epidemiologia e controle da malária na região de Campinas, Estado de São Paulo, Brasil, 1980 a 2000. Rev Soc Bras Med Trop . 2004;37(1):41-5.
-
22Payne D. Did medicated salt hasten the spread of chloroquine resistance in Plasmodium falciparum? Parasitol Today. 1988;4(4):112-5.
-
23Gusmão HH. Fighting disease-bearing mosquitoes through relentless field leadership. Am J Trop Med Hyg . 1982;31(4):705-10.
-
24Loiola CCP, Mangabeira da Silva CJ, Tauil PL. Controle da malária no Brasil: 1965 a 2001. Rev Panam Salud Publica 2002;11(4):235-44.
-
25United Nations Development Programme (UNDP). Multisectoral Action Framework for Malaria. Geneva: Roll Back Malaria Partnership/UNDP; 2013. 68p.
-
26Ministério da Saúde (MS). Secretaria de Vigilância em Saúde. Diretoria Técnica de Gestão. Guia para Gestão Local do Controle da Malária: Módulo 2: Controle Vetorial. Série B. Textos Básicos de Saúde. Brasília: MS; 2009. 59p.
-
27Tadei WP, Mascarenhas BM, Podesta MG. Biologia de anofelinos amazônicos. VIII. Conhecimentos sobre a distribuição de espécies de Anopheles na região de Tucuruí-Marabá (Pará). Acta Amaz. 1983;13(1):103-40.
-
28Tadei WP, Santos JMM, Costa WLS, Scarpassa VM. Biologia de anofelinos amazônicos. XII. Ocorrência de espécies de Anopheles, dinâmica da transmissão e controle da malária na zona urbana de Ariquemes (Rondônia). Rev Inst Med Trop São Paulo. 1988;30(3):221-51.
-
29Osorio Quintero L, Dutary Thatcher B, Tadei WP. Biologia de anofelinos amazônicos. XXI. Ocorrrência de espécies de Anopheles e outros Culicídeos na área de influência da Hidrelétrica de Balbina - cinco anos após o enchimento do reservatório. Acta Amazônica. 1996;26(4):281-95.
-
30World Health Organization (WHO). Malaria vector control: decision making criteria and procedures for judicious use of insecticides/by: JA Najera and M Zaim. Geneva: WHO ; 2003. 106p.
-
31Laporta GZ, Linton YM, Wilkerson RC, Bergo ES, Nagaki SS, Sant’Ana DC, et al. Malaria vectors in South America: current and future scenarios. Parasit Vectors. 2015;8(1):426.
-
32Deane LM. Malaria vectors in Brazil. Mem Inst Oswaldo Cruz . 1986;81(supl 2):5-14.
-
33Tadei WP, Thatcher BD, Santos JM, Scarpassa VM, Rodrigues IB, Rafael MS. Ecologic observations on anopheline vectors of malaria in the Brazilian Amazon. Am J Trop Med Hyg . 1998;59(2):325-35.
-
34Tadei WP, Dutary Thatcher B. Malaria vectors in the Brazilian Amazon: Anopheles of the subgenus Nyssorhynchus Rev Inst Med Trop S. Paulo. 2000;42(2):87-94.
-
35Forattini OP. Culicidologia Médica. Identificação, Biologia e Epidemiologia. São Paulo: Editora da Universidade de São Paulo. 2002. Vol 2, 860p.
-
36Marrelli MT, Malafronte RS, Sallum MAM, Natal D. Kerteszia subgenus of Anopheles associated with the Brazilian Atlantic rainforest: current knowledge and future challenges. Malar J . 2007;6:127-34.
-
37Tadei WP, Thatcher BD, Santos JM, Scarpassa VM, Rodrigues IB, Rafael MS. Ecologic observations on anopheline vectors of malaria in the Brazilian Amazon. Am J Trop Med Hyg . 1998;59(2):325-35.
-
38Santos JB, Santos F, Macedo V. Variação da densidade anofélica com o uso de mosquiteiros impregnados com deltametrina em uma área endêmica de malária na Amazônia Brasileira. Cad Saúde Pública 1999;15(2):281-92.
-
39Gama RA, Santos RLC, Santos F, Silva IM, Resende MC, Eiras AE. Periodicidade de captura de Anopheles darlingi Root (Diptera: Culicidae) em Porto Velho, RO. Neotrop. Entomol. 2009;38(5):677-82.
-
40Voorham J. Intra-population plasticity of Anopheles darlingi's (Diptera, Culicidae) biting activity patterns in the state of Amapá, Brazil. Rev Saúde Pública. 2002;36(1)75-80.
-
41Charlwood JD, Hayes J. Variações geográficas no ciclo de picada do Anopheles darlingi Root no Brasil. Acta Amaz . 1978;8(4): 601-3.
-
42Consoli RAGB, Lourenço-de-Oliveira RL. Principais mosquitos de importância sanitária no Brasil. Rio de Janeiro: Editora da Fundação Oswaldo Cruz; 1994. 225p.
-
43Tadei WP, Santos JMM, Rabbani MG. Biologia de Anofelinos amazônicos. V. Polimorfismo cromossômico de Anopheles darlingi Root (Diptera, Culicidae). Acta Amaz .1982;12(2):353-69.
-
44Tadei WP, Santos JMM. Biologia de Anofelinos amazônicos. VII. Estudo da variação de freqüências das inversões cromossômicas de Anopheles darlingi Root (Diptera, Culicidae). Acta Amaz . 1982;12(4):759-85.
-
45Charlwood JD. Biological variation in Anopheles darlingi Root. Mem Inst Oswaldo Cruz . 1996;91(4):391-8.
-
46Faran ME, Linthicum KJ. A handbook of the Amazonian species of Anopheles (Nyssorhynchus) (Diptera: Culicidae). Mosquito Systematics.1981;13(1):1-81.
-
47Galvão ALA, Damasceno RG, Marques AP. Algumas observações sobre a biologia dos anofelinos de importância epidemiológica de Belém Pará. Arq Hig Saúde Púb.1942;12(2):51-110.
-
48Rubio-Palis Y, Zimmerman RH. Ecoregional classification of malária vectors in the neotropics. J Med Entomol. 1997;34(5):499-510.
-
49Deane LM, Vernin SC, Damasceno RG. Avaliação das preferências alimentares das fêmeas de Anopheles darlingi e Anopheles aquasalis em Belém, Pará, por provas de precipitina. Rev Serv Esp Saúde Pública. 1949;2(3):793-808.
-
50Rachou GR, Moura-Lima M, Barbosa AL. Considerações sobre o An (N.) tarsimaculatus Goeldi, 1905 (An. (N.) aquasalis, Curry, 1932) no Estado do Ceará, com especial referência ao seu encontro a 52km da orla marítima. Rev Bras Malariol. 1950;2:57-65.
-
51Lourenço-de-Oliveira R, Heyden R. Alguns aspectos da ecologia dos mosquitos (Diptera: Culicidae) de uma área de planície (Granjas Calábria), em Jacarepaguá, Rio de Janeiro. IV. Preferências alimentares quanto ao hospedeiro e freqüência domiciliar. Mem Inst Oswaldo Cruz . 1986;81(1):15-27.
-
52Flores-Mendoza C, Cunha RA, Rocha DS, Lourenço-de-Oliveira R. Determinação das fontes alimentares de Anopheles aquasalis (Diptera: Culicidae) no Estado do Rio de Janeiro, Brasil, pelo teste de precipitina. Rev Saúde Pública . 1996;30(2):129-34.
-
53Wilkerson RC, Gaffigan TV, Lima JB. Identification of species related to Anopheles (Nyssorhynchus) albitarsis by random amplified polymorphic DNA-polymerase chain reaction (Diptera: Culicidae). Mem Inst Oswaldo Cruz . 1995;90(6):721-32.
-
54Wilkerson RC, Foster PG, Li C, Sallum MAM. Molecular phylogeny of Neotropical Anopheles (Nyssorhynchus) albitarsis species species complex (Diptera: Culicidae). Ann Ent Soc Amer. 2005;98(6):918-25.
-
55Motoki MT, Wilkerson RC, Sallum MAM. The Anopheles albitarsis complex with the recognition of Anopheles oryzalimnetes Wilkerson and Motoki, n. sp. and Anopheles janconnae Wilkerson and Sallum, n. sp. (Diptera: Culicidae). Mem Inst Oswaldo Cruz . 2009;104(6):823-50.
-
56Wilkerson RC, Hribar LJ, Milstrey EG, Falero GC. The identification of Anopheles (Nyssorhynchus) rondoni (Diptera: Culicidae) in Mato Grosso, Brazil: an analysis of key character variability. Mem Inst Oswaldo Cruz . 1995;90(5):575-82.
-
57Wilkerson RC, Parsons TJ, Klein TA, Gaffigan TV, Bergo E, Consolim J. Diagnosis by random amplified polymorphic DNA polymerase chain reaction of four cryptic species related to Anopheles (Nyssorhynchus) albitarsis (Diptera: Culicidae) from Paraguay, Argentina, and Brazil. J Med Entomol . 1995;32(5):697-704.
-
58Li C, Wilkerson RC. Identification of Anopheles (Nyssorhynchus) albitarsis complex species (Diptera: Culicidae) using rDNA internal transcribed spacer 2-based polymerase chain reaction primes. Mem Inst Oswaldo Cruz . 2005;100(5):495-500.
-
59Lehr MA, Kilpatrick CW, Wilkerson RC, Conn JE. Cryptic species in the Anopheles (Nyssorhynchus) albitarsis (Diptera: Culicidae) complex: incongruence between random amplified polymorphic DNA-polymerase chain reaction identification and analysis of mitochondrial DNA COI gene sequences. Ann Entomol Soc Am. 2005;98(6):908-17.
-
60Li C, Wilkerson RC. Intragenomic rDNA ITS2 variation in the neotropical Anopheles (Nyssorhynchus) albitarsis complex (Diptera: Culicidae). J Hered. 2007;98(1):51-9.
-
61Branquinho MS, Marrelli MT, Curado I, Natal D, Barata JMS, Tubaki R, et al. Infecção do Anopheles (Kerteszia) cruzii por Plasmodium vivax e Plasmodium vivax variante VK247 nos Municípios de São Vicente e Juquitiba, São Paulo. Rev Panam Salud Publica /Pan Am J Public Health. 1997;2(3):189-93.
-
62Deane LM, Ferreira-Neto JA, Lima MM. The vertical dispersion of Anopheles (Kerteszia) cruzii in a forest in southern Brazil suggests that human cases of malaria of simian origin might be expected. Mem Inst Oswaldo Cruz . 1984;79(4):461-3.
-
63Deane LM. Simian malaria in Brazil. Mem Inst Oswaldo Cruz 1992,87(Suppl 3):1-20.
-
64Forattini OP, Gomes AC, Natal D, Santos JLF. Observações sobre atividade de mosquitos Culicidae em mata primitiva da encosta no Vale do Ribeira, São Paulo, Brasil. Rev Saúde Pública . 1986;20(1):1-20.
-
65Forattini OP, Gomes AC, Natal D, Santos JLF. Observações sobre atividade de mosquitos Culicidae em matas primitivas da planície e perfis epidemiológicos de vários ambientes no Vale do Ribeira, São Paulo, Brasil. Rev Saúde Pública 1986;20(3):178-203.
-
66Forattini OP, Gomes AC, Santos JLF, Kakitani I, Marucci D. 1990. Freqüência ao ambiente humano e dispersão de mosquitos Culicidae em área adjacente à Mata Atlântica primitiva da planície. Rev Saúde Pública . 1990;24 (2):101-7.
-
67Forattini OP, Kakitani I, Massad E, Gomes AC. Studies on mosquitoes (Diptera: Culicidae) and anthropic environment. 1- Parity of blood seeking Anopheles (Kerteszia) in South-Eastern Brazil. Rev Saúde Pública .1993;27(1):1-8.
-
68Tubaki RM, Carréri-Bruno GC, Moreno Glasser C, Ciaravolo RMC. Biting activity of Anopheles (Kerteszia) cruzii (Diptera, Culicidae) in domiciliary habitats in the Southern Atlantic Forest, Peruibe, State of São Paulo, Brazil. Rev Bras Entomol. 1993;37(3):569-75.
-
69Curado I, Duarte AMRC, Lal AA, Oliveira SG, Kloetzel JK. Antibodies anti bloodstream and circumsporozoite antigens (Plasmodium vivax and Plasmodium malariae/P. brasilianum) in areas of very low malaria endemicity in Brazil. Mem Inst Oswaldo Cruz . 1997; 92(2):235-43.
-
70Lorenz C, Marques TC, Sallum MAM, Suesdek L. Morphometrical diagnosis of the malaria vectors Anopheles cruzii, An. homunculus and An. bellator Parasit Vectors . 2012;5(1):257.
-
71World Health Organization (WHO) & Global Partnership to Roll Back Malaria. Malaria vector control and personal protection: report of a WHO study group. Geneva: WHO ; 2006. 62p.
-
72Guimarães RM, Asmus CI, Meyer A. DDT reintroduction for malaria control: the cost-benefit debate for public health. Cad Saude Publ . 2007;23(12):2835-44.
-
73Akhavan D, Musgrove P, Abrantes A, d'A Gusmão R. Cost-effective malaria control in Brazil. Cost-effectiveness of a Malaria Control Program in the Amazon Basin of Brazil, 1988-1996. Soc Sci Med. 1999;49(10):1385-99.
-
74World Health Organization (WHO). Department of Control of Neglected Tropical Diseases. Manual for indoor residual spraying: application of residual sprays for vector control, third edition. Geneva: WHO Pesticide evaluation scheme (WHOPES) ; 2007. 43p.
-
75World Health Organization (WHO). Malaria entomology and vector control. Guide for Tutors. 2nd edition. Geneva: WHO ; 2013. 50p.
-
76World Health Organization (WHO). Indoor residual spraying. An operational manual for IRS for malaria transmission control and elimination. Second edition. Geneva: WHO ; 2015. 134p.
-
77Ministério da Saúde (MS). Secretaria de Vigilância em Saúde. Departamento de Vigilância Epidemiológica. Diretoria Técnica de Gestão. Ações de Controle da Malária: Manual para Profissionais de Saúde na Atenção Básica. Brasília: Editora do Ministério da Saúde; 2006. 52p.
-
78Centro de Controle e Prevenção de Doenças (CDC). Organização Pan-Americana da Saúde/Organização Mundial de Saúde (OPAS/OMS), e RTI. Iniciativa Amazônica contra a Malária/Rede Amazônica de Vigilância da Resistência às Drogas Antimaláricas. Documento de Orientação Estratégica para Vigilância e Controle de Vetores da Malária na América Latina e no Caribe. Apresentado à Agência dos Estados Unidos para o Desenvolvimento Internacional (USAID). 2011. 42p.
-
79Robefcts DR, Alecrim WD. Behavioral response of Anopheles darlingi to DDT-sprayed house walls in Amazonia. Bull Pan Am Health Organ. 1991;25(3):210-7.
-
80Charlwood JD, Alecrim WD, Fe N, Mangabeira J, Martins VJ. A field trial with Lambda-cyhalothrin (ICON) for the intradomiciliary control of malaria transmitted by Anopheles darlingi root in Rondonia, Brazil. Acta Trop. 1995;60(1):3-13.
-
81Santos RC, Fayal AS, Aguiar AEF, Vieira DB, Póvoa MM. Evaluation of the residual effect of pyrethroids on Anopheles in the Brazilian Amazon. Rev Saude Publica. 2007;41(2):276-83.
-
82Lengeler C. Insecticide-treated bed nets and curtains for preventing malaria. Cochrane Database Syst Rev. 2004;(2):CD000363.
-
83World Health Organization (WHO). Insecticide-treated mosquito net interventions: A manual for national control programme managers. Roll Back Malaria, editor. Geneve: WHO; 2003. 130p.
-
84World Health Organization (WHO). Guidelines for laboratory and field-testing of long-lasting insecticidal nets. Geneva: WHO ; 2013. 89p.
-
85da Silva-Nunes M, Moreno M, Conn JE, Gamboa D, Abeles S, Vinetz JM, et al. Amazonian malaria: asymptomatic human reservoirs, diagnostic challenges, environmentally driven changes in mosquito vector populations, and the mandate for sustainable control strategies. Acta Trop . 2012;121(3):281-91.
-
86Xavier PA, Lima JENS. O uso de cortinas impregnadas com deltametrina no controle da malária em garimpos no Território Federal do Amapá. Rev Bras Malariol Doenç Trop.1986;38:137-9.
-
87Salgado-Cavalcante ET, Tadei WP, Pinto CT, Xavier PA, Lima IENS. Efeitos da ação residual da deltametrina, em cortinas de ráfia e sarrapilha no controle da malária, em áreas de garimpo, no Estado do Amapá. Rev Soc Bras Med Trop . 1992;25(Supl I):6-7.
-
88Galardo CD, Galardo AKR. Relatório técnico sobre o Estudo da Eficácia de Redes Impregnadas com Inseticidas e do uso de de Fendona® em borrifações domiciliares para o Controle de Anopheles sp. em Bioensaios de Campo e Laboratório no Estado do Amapá - Brasil. Departamento de Zoologia-Laboratório de Entomologia Médica, Instituto de Pesquisas Científicas e Tecnolócias do Estado do Amapá, Macapá. 2009. 29p.
-
89Vieira GD, Basano SA, Katsuragawa TH, Camargo LM. Insecticide-treated bed nets in Rondônia, Brazil: evaluation of their impact on malaria control. Rev Inst Med Trop Sao Paulo. 2014;56(6):493-7.
-
90World Health Organization (WHO). Space spray application of insecticides for vector and public health pest control: a practitioner's guide. Geneva: WHO ; 2003. 49p.
-
91World Health Organization (WHO). Malaria vector control policy recommendations and their applicability to product evaluation. Geneva: WHO ; 2017. 8p.
-
92World Health Organization (WHO). WHO recommended insecticides for space spraying against mosquitoes. Geneva: WHO . Updated 5 February 2016.
-
93Ladislau JLB, Póvoa MM, Sucupira IMC, Galardo AKR, Damasceno OC, Cardoso BS, et al. Malaria control in area of hydroelectric construction on the Amazonian ecosystem can succeed? Rev Pan-Amaz Saude. 2016;7(num esp,):115-22.
-
94Santos RC, Padilha A, Costa MDP, Costa EM, Dantas-Filho HC, Povoa MM. Vetores de malária em duas reservas indígenas da Amazônia Brasileira. Rev Saúde Pública . 2009;43(5):859-68.
-
95Silva JJ, Mendes J. Susceptibility of Aedes aegypti (L) to the insect growth regulators diflubenzuron and methoprene in Uberlândia, State of Minas Gerais. Rev Soc Bras Med Trop . 2007;40(6), 612-6.
-
96Tadei WP, Rodrigues IB, Rafael MS, Sampaio RTM, Mesquita HG, Pinheiro VCS, et al. Adaptative processes, control measures, genetic background, and resilience of malaria vectors and environmental changes in the Amazon region. Hydrobiologia. 2017;789(1):179-96.
-
97Palma L, Muñoz D, Berry C, Murillo J, Caballero P. Bacillus thuringiensis toxins: an overview of their biocidal activity. Toxins (Basel). 2014;6(12):3296-325.
-
98Roh JY, Choi JY, Li MS, Jin BR, Je YH. Bacillus thuringiensis as a specific, safe, and effective tool for insect pest control. J Mol Biol. 2007;17(4):547-59.
-
99World Health Organization (WHO). Larval source management: a supplementary measure for malaria vector control. An operational manual. Geneva: WHO ; 2013. 116p.
-
100Rodrigues BI, Tadei WP, Dias JMSD, Lima CAP. Atividade Larvicida de Bacillus sphaericus 2362 contra Anopheles sp. (Diptera, Culicidae) em rios do Amazonas, Brasil. Bio Assay. 2013;8(2):1-7.
-
101Rodrigues IB, Tadei WP, Dias JMCS. Larvicidal activity Bacillus sphaericus 2362 against Anopheles nuneztovari, Anopheles darlingi and Anopheles braziliensis (Diptera, Culicidae). Rev Inst Med Trop S Paulo. 1999;41(2):101-5.
-
102Galardo AKR, Zimmerman R, Galardo CD. Larval control of Anopheles (Nyssorhinchus) darlingiusing granular formulation of Bacillus sphaericus in abandoned gold-miners excavation pools in the Brazilian Amazon rainforest. Rev Soc Bras Med Trop . 2013;46 (2):172-7.
-
103Chareonviriyaphap T, Bangs MJ, Suwonkerd W, Kongmee M, Corbel V, Ngoen-Klan R. Review of insecticide resistance and behavioral avoidance of vectors of human diseases in Thailand. Parasit Vectors . 2013;6:280.
-
104World Health Organization (WHO). Test procedures for insecticide resistance monitoring in malaria vectors, bio-efficacy and persistence of insecticide on treated surfaces: report of the WHO informal consultation. Geneva: WHO ; 1998. 43p.
-
105Sparks TC, Lockwood JA, Byford RL, Graves JB, Leonard BR. The role of behavior in insecticide resistance. Pestic Sci. 1989;26(4):383-99.
-
106Philbert A, Lyantagaye SL, Nkwengulila G. A review of agricultural pesticides use and the selection for resistance to insecticides in malaria vectors. Adv Entomol. 2014;2(3):120-8.
-
107World Health Organization (WHO). Global plan for insecticide resistance management in malaria vectors. Geneva: WHO ; 2012. 132p.
-
108Brogdon WG, Chan A. Diretriz para avaliar a resistência a inseticida em vetores usando o bioensaio com garrafa do CDC. 2018. 2012. 28p. Disponível em: https://www.cdc.gov/malaria/resources/pdf/fsp/ir_manual/ir_cdc_bioassay_pt.pdf
» https://www.cdc.gov/malaria/resources/pdf/fsp/ir_manual/ir_cdc_bioassay_pt.pdf -
109Galardo AKR, Póvoa MN, Sucupira IMC, Galardo CD, Santos RC. Anopheles darlingi and Anopheles marajoara (Diptera: Culicidae) susceptibility to pyrethroids in an endemic area of the Brazilian Amazon. Rev Soc Bras Med Trop . 2015;48(6):765-9.
-
110Silva APB, Alves WS, Martins AJ, Tadei WP, Santos JMM. Adaptação de um bioensaio simplificado para avaliação do status de susceptibilidade em larvas de Anopheles darlingi e Anopheles marajoara ao piretroide deltametrina. BioAssay. 2014;9(4):1-8.
-
Financial support: This study received financial support from Coordenação de Aperfeiçoamento de Pessoal de Nível Superior- Brasil (CAPES) - Finance Code 001. Paulo Pimenta, Marcus Vinícius Guimarães de Lacerda and Wuelton Marcelo Monteiro are CNPq fellows.
Publication Dates
-
Publication in this collection
07 Mar 2019 -
Date of issue
2019
History
-
Received
22 Dec 2018 -
Accepted
20 Feb 2019