Abstract
INTRODUCTION
Mutations in the propeller domain of the Plasmodium falciparum kelch13 (k13) gene are associated with artemisinin resistance.
METHODS:
We developed a PCR protocol to sequence the pfk13 gene and determined its sequence in a batch of 50 samples collected from 2003 to 2016 in Brazil.
RESULTS:
We identified 1 K189T substitution located outside the propeller domain of the PfK13 protein in 36% of samples.
CONCLUSIONS:
Although the sample size is relatively small, these results suggest that P. falciparum artemisinin-resistant mutants do not exist in Brazil, thereby supporting the continuation of current treatment programs based on artemisinin-based combination therapy.
Keywords:
Malaria; Plasmodium falciparum; Artemisinin Resistance; Kelch 13
Malaria is a devastating vector-borne disease that claims nearly half a million human lives every year 11. WHO - World Malaria Report [homepage of internet]. 2017. [cited 10 May 2018]. Available from: Available from: http://www.who.int/malaria/publications/world-malaria-report-2017/report/en/
.
http://www.who.int/malaria/publications/...
. A great majority of fatalities is caused by Plasmodium falciparum, the most lethal of all human malarial parasite species. Using artemisinin-based combination therapy (ACT) i.e., the introduction of artemisinin derivatives in combination with chemically distinct drugs, to manage P. falciparum infections has proved to be extremely effective in reducing the number of malaria-related cases and deaths over the last 15 years 11. WHO - World Malaria Report [homepage of internet]. 2017. [cited 10 May 2018]. Available from: Available from: http://www.who.int/malaria/publications/world-malaria-report-2017/report/en/
.
http://www.who.int/malaria/publications/...
. However, natural P. falciparum parasite populations in Southeast Asia have already evolved resistance towards artemisinin derivatives, endangering the future of all malaria treatment-based control campaigns due to the absence of effective alternative treatments 22. Miotto O, Amato R, Ashley EA, MacInnis B, Almagro-Garcia J, Amaratunga C, et al. Genetic architecture of artemisinin-resistant Plasmodium falciparum. Nat Genet. 2015;47:226-34..
Although ACTs remain largely effective in endemic areas other than Asia, there are fears that resistance to artemisinin derivatives may spread from Asia to Africa and potentially, to South America. However, it is also possible that resistant parasite mutants may evolve independently in these areas. Consequently, close monitoring of parasite populations for the timely detection of potential ACT resistance foci is considered top priority in terms of public health malaria surveillance policies. Since the genetic basis of resistance to artemisinin derivatives in P. falciparum has been unraveled 33. Ariey F, Witkowski B, Amaratunga C, Beghain J, Langlois AC , Khim N, et al. A molecular marker of artemisinin-resistant Plasmodium falciparum malaria. Nature. 2014; 505(7481):50-5.
4. Witkowski B, Amaratunga C, Khim N, Sreng S, Chim P, Kim S, et al. Novel phenotypic assays for the detection of artemisinin-resistant Plasmodium falciparum malaria in Cambodia: in-vitro and ex-vivo drug-response studies. Lancet Infect Dis. 2013; 13(12):1043-9.-55. Amaratunga C1, Witkowski B2, Khim N2, Menard D2, Fairhurst RM. Artemisinin resistance in Plasmodium falciparum. Lancet Infect Dis . 2014;14(6):449-50., there are molecular markers that can be used to monitor resistance in the field. Resistance to these drugs is associated with mutations in the Kelch13 (K13) propeller domain, with 8 of these mutations (P441L, F446I, S449A, N458Y, P553L, V568G, P574L, and L675V) being associated with delayed parasite clearance and 5 (Y493H, R539T, I543T, R561H, and C580Y) with in vitro and/or in vivo resistance 22. Miotto O, Amato R, Ashley EA, MacInnis B, Almagro-Garcia J, Amaratunga C, et al. Genetic architecture of artemisinin-resistant Plasmodium falciparum. Nat Genet. 2015;47:226-34.. In addition to drug clinical trial data, the WHO recommends using these mutations as molecular markers to detect the presence of artemisinin resistance in countries where P. falciparum malaria is endemic.
To date, no phenotypic evidence demonstrates the evolution of artemisinin derivative resistance in P. falciparum malaria in South America, particularly in the countries comprising of the so-called International Amazon region, where the disease is prevalent 66. World Health Organization. Status report on artemisinin and ACT resistance. World Health Organization, Geneva, Switzerland. 2015.
7. Chenet SM, Akinyi Okoth S, Huber CS, Chandrabose J, Lucchi NW, Talundzic E, et al. Independent Emergence of the Plasmodium falciparum Kelch Propeller Domain Mutant Allele C580Y in Guyana. J Infect Dis. 2016; 213(9):1472-5.
8. Ladeia-Andrade S, de Melo GN, de Souza-Lima Rde C, Salla LC, Bastos MS, Rodrigues PT, et al. No Clinical or Molecular Evidence of Plasmodium falciparum Resistance to Artesunate-Mefloquine in Northwestern Brazil. Am J Trop Med Hyg. 2016; 95 (1):148-54.-99. Rahman R, Martin MJ, Persaud S, Ceron N, Kellman D, Musset L, et al. Continued Sensitivity of Plasmodium falciparum to Artemisinin in Guyana, With Absence of Kelch Propeller Domain Mutant Alleles. Open Forum Infect Dis. 2016; 3(3):ofw185.. Nevertheless, a study in Guyana consisting of samples collected in 2010 found 5 isolates that carried the C580Y mutant allele in the PfK13 propeller domain with flanking microsatellites different from those observed in Southeast Asia, thereby suggesting that these mutants were of regionally-independent origin 77. Chenet SM, Akinyi Okoth S, Huber CS, Chandrabose J, Lucchi NW, Talundzic E, et al. Independent Emergence of the Plasmodium falciparum Kelch Propeller Domain Mutant Allele C580Y in Guyana. J Infect Dis. 2016; 213(9):1472-5.. However, subsequent studies in French Guiana, Suriname, and Brazil neither found evidence of phenotypic resistance to artemisinin derivatives, nor any of the mutations previously associated with resistance in Southeast Asia 77. Chenet SM, Akinyi Okoth S, Huber CS, Chandrabose J, Lucchi NW, Talundzic E, et al. Independent Emergence of the Plasmodium falciparum Kelch Propeller Domain Mutant Allele C580Y in Guyana. J Infect Dis. 2016; 213(9):1472-5.
8. Ladeia-Andrade S, de Melo GN, de Souza-Lima Rde C, Salla LC, Bastos MS, Rodrigues PT, et al. No Clinical or Molecular Evidence of Plasmodium falciparum Resistance to Artesunate-Mefloquine in Northwestern Brazil. Am J Trop Med Hyg. 2016; 95 (1):148-54.-99. Rahman R, Martin MJ, Persaud S, Ceron N, Kellman D, Musset L, et al. Continued Sensitivity of Plasmodium falciparum to Artemisinin in Guyana, With Absence of Kelch Propeller Domain Mutant Alleles. Open Forum Infect Dis. 2016; 3(3):ofw185..
In Brazil (where this study was carried out), malaria is still endemic in the northern states, which are part of the Amazon region, despite a significant reduction in P. falciparum cases since 2010 11. WHO - World Malaria Report [homepage of internet]. 2017. [cited 10 May 2018]. Available from: Available from: http://www.who.int/malaria/publications/world-malaria-report-2017/report/en/
.
http://www.who.int/malaria/publications/...
. Therefore, close monitoring of the resident parasite population is top priority in order to identify potential foci of resistance, since P. falciparum malaria treatment programs are fully dependent on ACTs. In this regard, the sequence of the P. falciparum k13 gene was determined from the samples of infected patients collected during this study in Brazil, thereby aiming to garner molecular data that may be helpful in drawing up P. falciparum malaria treatment policies.
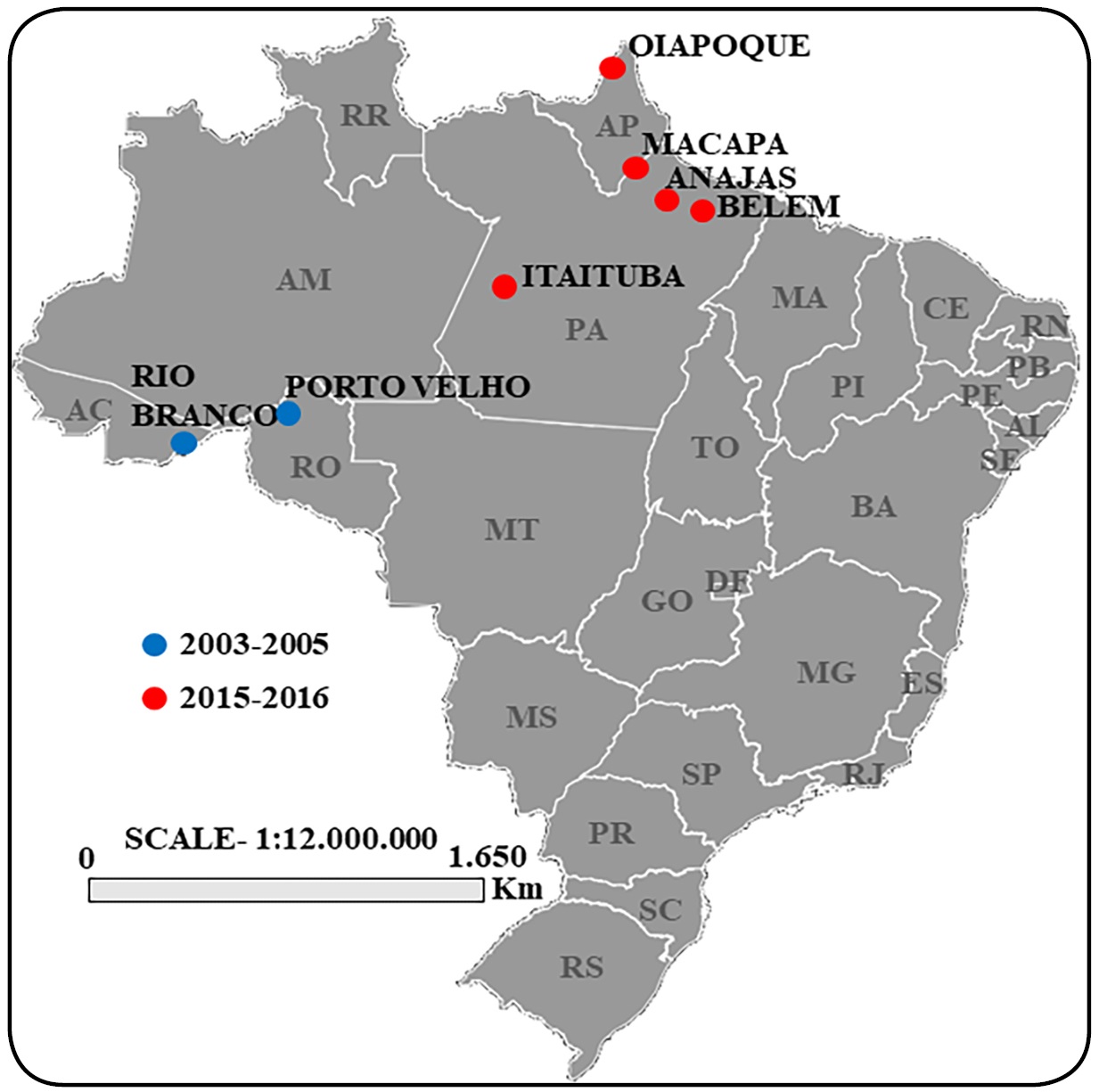
After obtaining written informed consent, whole blood samples were collected via venipuncture from patients with confirmed symptoms of P. falciparum malaria who were recruited while seeking medical treatment at health facilities in the following 7 regions of the Brazilian Amazon: Anajás (ANJ), Belém (BEL), and Itaituba (ITB) in the state of Pará; Macapá (MCP) and Oiapoque (OPQ) in the state of Amapá; Porto Velho (PVL) in the state of Rondônia; and Rio Branco (RBR) in the state of Acre (Figure 1). The following inclusion criteria were set for participation in the study: adult male or female; not pregnant; signs of clinical symptoms of malaria; thick smear positive for P. falciparum monoinfection, and not undergoing antimalarial treatment. After confirmation of P. falciparum monoinfection, samples were collected and stored at 4 ºC until DNA extraction. This study was approved by the Research Ethics Committee of the Evandro Chagas Institute under process no. 004/2003/CEP/IEC/SVS/MS and the Ethics Committee for 2015-2016 samples (CAAE - 001.219.346-15), which was certified by the Research Ethics Committee of the Federal University of Amapá (CAAE 18740413.7.0000.0003).
Map of Brazil displaying the locations of the collection sites based on the collection period. Source: the author.
Whole DNA from each sample was isolated using the QIAamp DNA mini kit (Qiagen®) following the manufacturer’s protocol. Each DNA sample was subjected to single-round PCR amplification using 4 primer pairs to generate overlapping fragments that amplified near-full length coding sequence of the pfk13 gene (Table 1). These oligonucleotides were designed de novo using the P. falciparum 3D7 reference sequence as a template (accession no. NC_004331) aided by the OligoCalc tool 1010. Kibbe WA. OligoCalc: an online oligonucleotide properties calculator. Nucleic Acids Res. 2007; W43-W46. to minimize the probability of complementarity and hairpin formation.
One microliter of template DNA was used for each PCR reaction, in which the following reaction mixture was used: 0.5 μM of each primer, 1× PCR buffer (Invitrogen®), 2.5 mM MgCl2 (Invitrogen®), 0.2 mM dNTPs (Invitrogen®), and 0.05 U/μL of Taq DNA polymerase (Invitrogen®). PCR reaction conditions were as follows: 40 cycles at 94 °C for 3 min (initial denaturation), 94 °C for 30’’ (denaturation), 56 °C for 35’’ (oligonucleotide annealing), 72 °C for 45’’ (extension), and 72 °C for 10’’ (final extension).
The quality and concentration of the PCR products generated from each fragment were ascertained via agarose gel electrophoresis and then used as templates for DNA sequencing after purification with a commercial kit (PureLink PCR Purification Kit, Invitrogen®) following the manufacturer’s instructions. DNA sequences were generated from both forward and reverse primers using the BigDye® Terminator v3.1 kit (Life Technologies, USA and aligned with each other and the P. falciparum 3D7 reference sequence to check for genetic polymorphisms after resolving potential ambiguities via chromatogram inspection.
After effectively developing our PCR protocol, a total of 50 samples collected during 2 different time periods (32 samples in 2003-2005 in Porto Velho (RO) and Rio Velho (AC); and 18 samples in 2015-2016 in Anajás (PA), Belém (PA), Itaituba (PA), Macapá (AP), and Oiapoque (AP); (Figure 1) were successfully amplified and sequenced (GenBank accession numbers MH021044 to MH021093). An analysis of the pfk13 gene in these samples revealed that none of them carried mutations in the propeller domain that were previously associated with delayed parasite clearance in in vitro and/or in vivo resistance. Instead, only an A>C nucleotide change at position 566, resulting in a K189T non-synonymous substitution, was observed in 36% of the samples distributed over the two collection periods and different regions surveyed (Table 2).
Prevalence of the PfK13 K189T polymorphism in Brazilian samples based on site and collection period.
This polymorphism is located in a P. falciparum-specific N-terminal domain of the K13 protein, upstream of the BTB/POZ and propeller domains 1111. Mita T, Tachibana SI, Hashimoto M, Hirai M. Plasmodium falciparum kelch 13: a potential molecular marker for tackling artemisinin-resistant malaria parasites. Expert Rev Anti Infect Ther. 2016;14 (1):125-35.. The K189T polymorphism has been shown to be common in African parasite populations and has not been linked with delayed parasite clearance after treatment with artemisinin 22. Miotto O, Amato R, Ashley EA, MacInnis B, Almagro-Garcia J, Amaratunga C, et al. Genetic architecture of artemisinin-resistant Plasmodium falciparum. Nat Genet. 2015;47:226-34.. Interestingly, our results corroborated recent work in which the K189T polymorphism was also found to be the only variation occurring in P. falciparum isolates from Northwestern Brazil 88. Ladeia-Andrade S, de Melo GN, de Souza-Lima Rde C, Salla LC, Bastos MS, Rodrigues PT, et al. No Clinical or Molecular Evidence of Plasmodium falciparum Resistance to Artesunate-Mefloquine in Northwestern Brazil. Am J Trop Med Hyg. 2016; 95 (1):148-54.. Although our sample size may not be representative of the whole parasite population, these findings suggest that artemisinin derivative-resistant P. falciparum mutants do not exist in Brazil, thus supporting the continuation of the current ACT-based malarial treatments.
Acknowledgements
We gratefully acknowledge all the individuals who participated in this study. We are also indebted to Rubens Alex Menezes, Darci Rodrigues, Rogério Brandão, and Sonia Rodrigues for their help during field work.
REFERENCES
-
1WHO - World Malaria Report [homepage of internet]. 2017. [cited 10 May 2018]. Available from: Available from: http://www.who.int/malaria/publications/world-malaria-report-2017/report/en/
» http://www.who.int/malaria/publications/world-malaria-report-2017/report/en/ -
2Miotto O, Amato R, Ashley EA, MacInnis B, Almagro-Garcia J, Amaratunga C, et al. Genetic architecture of artemisinin-resistant Plasmodium falciparum Nat Genet. 2015;47:226-34.
-
3Ariey F, Witkowski B, Amaratunga C, Beghain J, Langlois AC , Khim N, et al. A molecular marker of artemisinin-resistant Plasmodium falciparum malaria. Nature. 2014; 505(7481):50-5.
-
4Witkowski B, Amaratunga C, Khim N, Sreng S, Chim P, Kim S, et al. Novel phenotypic assays for the detection of artemisinin-resistant Plasmodium falciparum malaria in Cambodia: in-vitro and ex-vivo drug-response studies. Lancet Infect Dis. 2013; 13(12):1043-9.
-
5Amaratunga C1, Witkowski B2, Khim N2, Menard D2, Fairhurst RM. Artemisinin resistance in Plasmodium falciparum. Lancet Infect Dis . 2014;14(6):449-50.
-
6World Health Organization. Status report on artemisinin and ACT resistance. World Health Organization, Geneva, Switzerland. 2015.
-
7Chenet SM, Akinyi Okoth S, Huber CS, Chandrabose J, Lucchi NW, Talundzic E, et al. Independent Emergence of the Plasmodium falciparum Kelch Propeller Domain Mutant Allele C580Y in Guyana. J Infect Dis. 2016; 213(9):1472-5.
-
8Ladeia-Andrade S, de Melo GN, de Souza-Lima Rde C, Salla LC, Bastos MS, Rodrigues PT, et al. No Clinical or Molecular Evidence of Plasmodium falciparum Resistance to Artesunate-Mefloquine in Northwestern Brazil. Am J Trop Med Hyg. 2016; 95 (1):148-54.
-
9Rahman R, Martin MJ, Persaud S, Ceron N, Kellman D, Musset L, et al. Continued Sensitivity of Plasmodium falciparum to Artemisinin in Guyana, With Absence of Kelch Propeller Domain Mutant Alleles. Open Forum Infect Dis. 2016; 3(3):ofw185.
-
10Kibbe WA. OligoCalc: an online oligonucleotide properties calculator. Nucleic Acids Res. 2007; W43-W46.
-
11Mita T, Tachibana SI, Hashimoto M, Hirai M. Plasmodium falciparum kelch 13: a potential molecular marker for tackling artemisinin-resistant malaria parasites. Expert Rev Anti Infect Ther. 2016;14 (1):125-35.
-
Financial support: This project received partial federal support from the Evandro Chagas Institute/Brazilian Ministry of Health. This research was also partially financed by FAPESP (São Paulo Research Foundation) through Grant 02/9546-1 and CNPq (National Council for Scientific and Technological Development) through Grant 302353/2003-8 awarded to RLDM. RLDM is also supported by a fellowship from CNPq. AA is supported by CAPES (Federal Agency for the Support and Improvement of Higher Education). PC is supported by a fellowship from FUNADESP (National Foundation for Development of Private Education). These financing agencies played no role in study design, data collection and analysis, decision to publish, or manuscript preparation.
Publication Dates
-
Publication in this collection
21 Feb 2019 -
Date of issue
2019
History
-
Received
08 June 2018 -
Accepted
16 Aug 2018