Abstract
INTRODUCTION:
We investigated the occurrence of relapsing fever (RF) causing Borrelia genus spirochetes in synanthropic bats from the municipality of Maringá, Paraná, South of Brazil.
METHODS:
Tissue samples from the wings of bats were collected monthly from April 2013 to February 2014 and extracted DNA was used to evaluate the presence of RF causing Borrelia spp.
RESULTS:
All bat tissues tested negative for RF causing Borrelia spp.
CONCLUSIONS:
Borrelia spp. do not occur in chiropterans from Maringá.
Keywords:
Urban bats; Chiropterans; Relapsing fever; Borrelia spp
Numerous bat species currently live in urban environments; these animals, thus, participate in the epidemiological chain of several zoonoses, such as those caused by viruses, bacteria, fungi, protozoa, and ectoparasites11. Kotait I, Aguiar EAC, Carrieri ML, Harmani NMS. Manejo de quirópteros em áreas urbanas. Manual Técnico nº 7. São Paulo: Instituto Pasteur; 2003. 45 p..
Among the diseases caused by bacteria, borreliosis is an infectious illness caused by spirochetes from the genus Borrelia, transmitted by ticks to animals and/or humans22. Díaz P, Remesar S, Venzal JM, Vásquez-López ME, Fernández G, López C, et al. Occurrence of Borrelia and Borreliella species in Ixodes ricinus collected from roe deer in northwestern Spain. Med Vet Entomol. 2019.. Relapsing fever (RF), a type of borreliosis, is characterized by a severe disease that progresses rapidly with recurring cycles of fever. RF occurs worldwide and is endemic in 118 countries33. Gideon Informatics. Relapsing fever in the United States [Internet]. Berger SA: Relapsing fever in the United States; 2011[update August 23, 2011; quoted 2016 February 6]. Available at: Available at: https://www.gideononline.com/2011/08/23/relapsing-fever-in-the-united-states-2/
https://www.gideononline.com/2011/08/23/...
.
Studies about Borrelia spp. are still scarce in Brazil and are limited to the Lyme disease Borrelia group. There is also a lack of Brazilian studies on bats, which live in great proximity to humans44. Pacheco SM, Sodré M, Gama AR, Bredt A, Cavallini-Sanches EM, Marques RV, et al. Morcegos urbanos: status do conhecimento e plano de ação para a conservação no Brasil. Chiropt Neotrop. 2010; 16(1):630-47. and have already been described as RF reservoirs in the United States, United Kingdom, and France55. Gill JS, Ullmann AJ, Loftis AD, Schwan TG, Raffel SJ, Schrumpf ME, et al. Novel relapsing fever spirochete in bat tick. J Emerg Infect Dis. 2008;14(3):522-3.-66. Socolovschi C, Mediannikov O, Sokhna C, Tall A, Diatta G, Bassene H, et al. Rickettsia felis-associated uneruptive fever, Senegal. J Emerg Infect Dis. 2010; 16(7):1140-2.. Therefore, the present study investigated the occurrence of spirochetes of different RF-causing Borrelia ssp. in the DNA from bat wing tissue of synanthropic bats from the municipality of Maringá, State of Paraná, South of Brazil, using PCR employing specific primers.
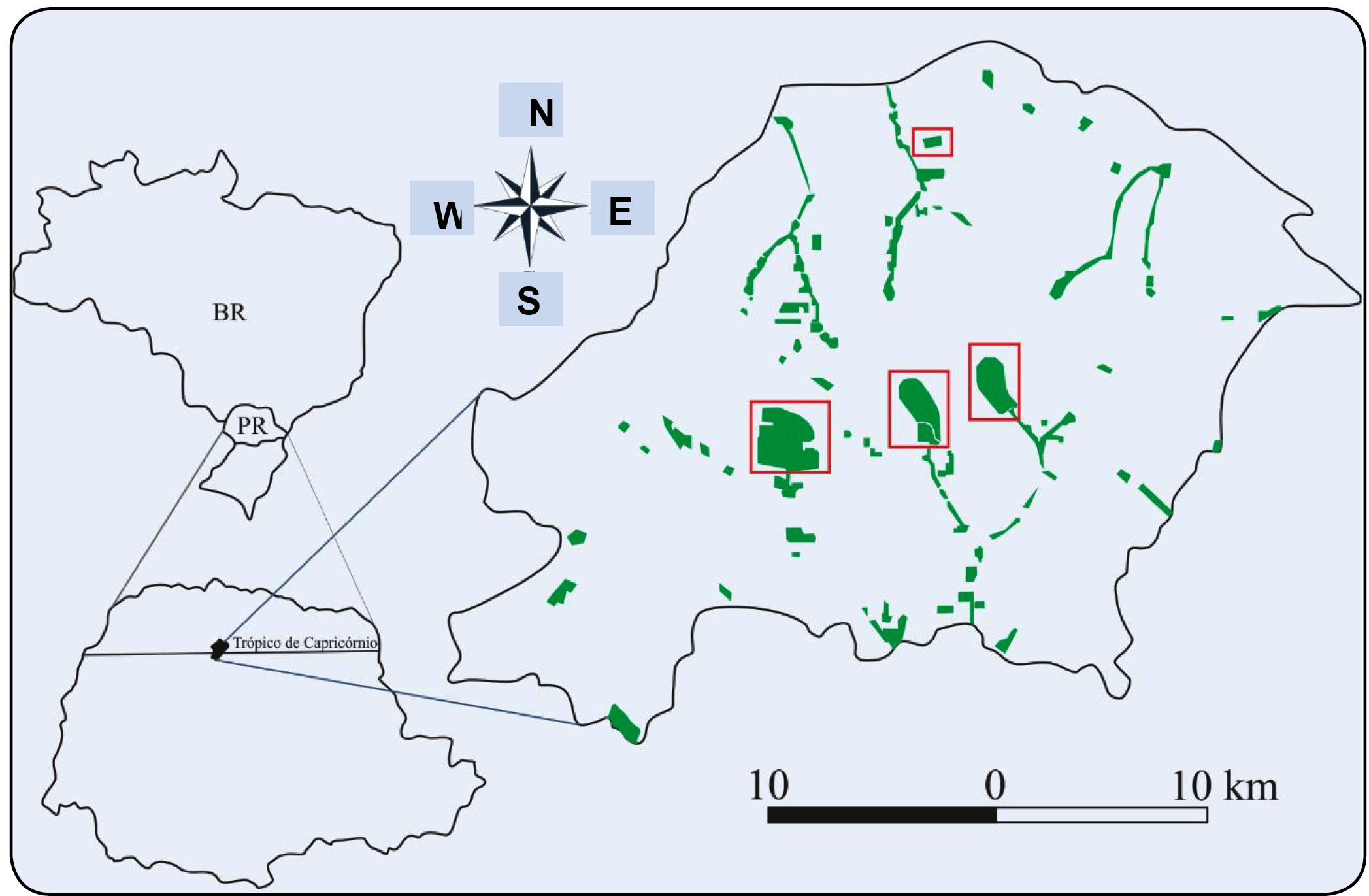
The study was conducted in urban Maringá (location: 23° 25′ 30″ S, 51° 56′ 20″ W) from April 2013 to February 2014. Sampling was performed on four nights in each month, two in residential homes and two in areas of the native forest (Figure 1). Sampling on private properties was performed through voluntary scheduling through the Bats Project, run by the State University of Maringá. The method of sampling within homes varied depending on the structure of the house and involved nylon nets, fishing nets, and the direct capture of bats by entering the ceiling.
Area of study. The green areas represent the Municipality of Maringá, and the red boxes denote the parks sampled in the present research.
The forest areas in which sampling was performed included the Municipal Park of Ingá, Forest Garden Doutor Luiz Teixeira Mendes, and the Municipal Forest Park from the Palmeiras and Forest Park Pioneiros. Sampling within the forest started at sunset using eight nylon nets of 9 m length and 3 m height and was performed for 12 h per night.
Bats were captured and handled using leather gloves for data was collectioned such as sex, reproductive status and species. The animals were released after data collection. Bat tissue samples were collected through the non-lethal method described by Worthington Wilmer & Barratt77. Wilmer JW, Barrat E. A non-lethal method of tissue sampling for genetic studies of chiropterans. Bat Res News. 1996; 37:1-3.. After local asepsis, a piece of the bat-wing membrane (patagium) was removed with the aid of a disposable biopsy punch, being careful to not strike any blood vessels. The biopsy punch diameter used in this research was 1 mm (Disposable Biopsy Punch - Miltex®, Austin, EUA). At the end of the procedure, the bats were marked with an animal tattoer and released. The tissue samples were stored in numbered containers with 1 ml ethanol and analyzed at the Lyme Disease Lab at the Texas A&M University.
For DNA extraction from the tissue samples (1-mm skin biopsies), 50.0 µL of collagenase (Sigma®) and 50.0 µL of proteinase K (concentration of 200 µg/mL; Sigma®) were added to each sample-containing tube. Samples were incubated for 4 h in a water bath set at 57oC with manual shaking every 30 min to aid tissue digestion. The digested samples were transferred to clean PCR tubes, where they were incubated with equal volumes of the extraction buffer provided in the ForensicGen®Tissue DNA extraction kit (ZyGEM®, Charlottesville, EUA ) for 30 min at 75oC, followed by a 5 min incubation at 95oC to inactivate enzymes. This protocol was performed in an Eppendorf™ thermocycler. DNA quantification and quality were evaluated through spectrophotometric measurement (NanoDrop®). This protocol was standardized to ensure the extraction of a sufficient amount of good quality DNA from small tissue biopsies. In this study, we considered a DNA quantity of 5 ng/µl satisfactory, to verify the occurrence of Borrelia ssp., considering the small size of the samples. Thirty nanograms of DNA per sample was used to carry out the PCR reaction.
PCR was performed using specific primers for detecting spirochetes of Borrelia spp., causing RF (5'ATGCTAGAAACTGCATGA-3' and 5'TCGTCTGAGTCCCCATCT-3') and amplifying a fragment of the 16S rRNA. These primers have been previously used successfully to detect the presence of DNA from Borrelia crocidurae, Borrelia duttonii, Borrelia recurrentis, Borrelia hispanica, Borrelia coriaceae, Borrelia lonestari, Borrelia miyamotoi, Borrelia parkeri, Borrelia turicatae, Borrelia hermsii, and Borrelia anserine88. Ras NM, Lascola B, Postic D, Cutler SJ, Rodhain F, Baranton G, et al. Phylogenesis of relapsing fever Borrelia spp. Int J Syst Bacteriol. 1996; 46(4):859-65.. As a positive control, we used DNA from a B. turicatae culture maintained in the Lyme Disease laboratory at Texas A&M University. The PCR products were separated via 1% agarose gel electrophoresis (Biolin® Scientific, Linthicum Heights, EUA) in TAE buffer, with the addition of 20 µl of ethidium bromide (10 mg/mL; Invitrogen®, Carlsbad, EUA) and using a molecular marker of 1 Kb (New England Biolabs, Rowley, EUA).
Licenses included the Permanent License to Collect Zoological Material from the Chico Mendes Institute of Biodiversity Conservation (ICMBio) number: 17869-2 (date of emission: 05/02/2009), certification of the Committee of Ethical Conduct in the Use of Animals in Experiments (CEAE) from the Internal Commission of Biosafety (CIBIO) of the State University of Maringá, and the Institutional Biosafety Committee permit 2013-039 from Texas A&M University. All sampling and laboratory procedures were performed in accordance with the ethical principles of the consulted entities.
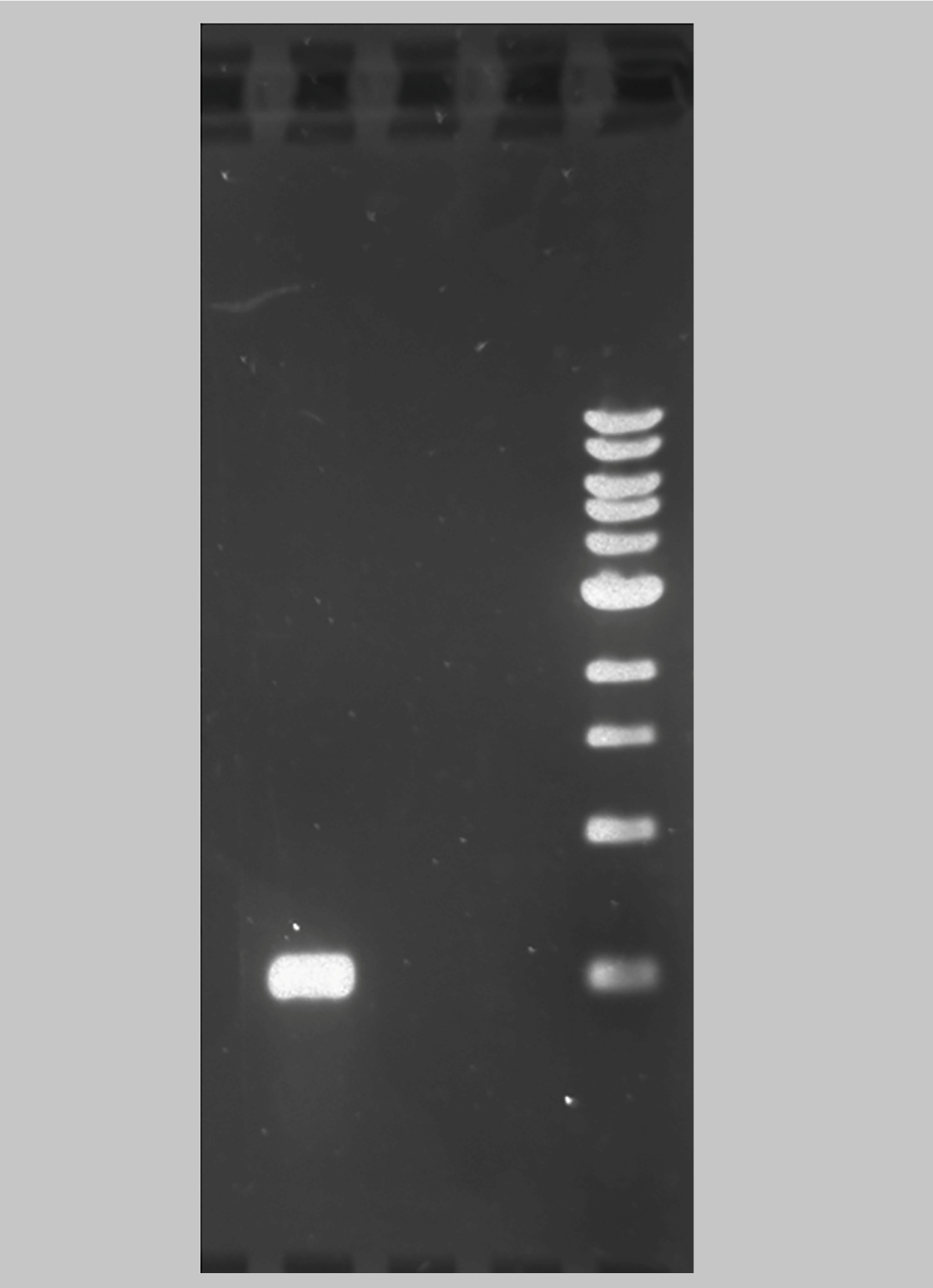
A total of 337 bat-wing tissue samples were analyzed through PCR utilizing primers for the amplification of a fragment corresponding to the 16sRNA gene found in RF caused by bacteria from the Borrelia genus. Seven of these samples were from recaptured bats identified through tattoos, resulting in 330 individual bats of different species (Table 1). All the PCR results were negative for the 16SrRNA RF-causing spirochetes from the Borrelia genus. This indicates that chiropterans from the Maringá region are not carriers of any RF causing Borrelia ssp. described to date. The Borrelia tunicatae positive control showed positivity in all tests (Figure 2).
Agarose gel electrophoresis of the positive control. Example of the result obtained in this study. Lines MK: molecular marker of 1 Kb (New England Biolabs), 1: negative control; 2: negative control; 3: positive control Borrelia turicatae grown under laboratory conditions, in the Lyme Disease Laboratory at Texas A&M University, shown in the left.
Borrelia spp. have been described in several animals in Brazil, such as dogs, cattle, and poultry24; however, no study with chiropterans has been conducted in the country. Therefore, the present study represents the first evaluation of the potential occurrence of RF borreliosis in bats in Brazil.
A study performed in Jataizinho, Paraná, a municipality that is only 102 km from Maringá, found the presence of bacteria with 99.9% similarity to the B31 Borrelia burgdorferi sensu stricto in Dermacentor nitens ticks, acting as the first report suggesting the occurrence of these bacteria in Brazil99. Gonçalves DD, Carreira T, Nunes M, Benitez A, Lopes-Mori FMR, Vidotto O, et al. First record of Borrelia burgdorferi B31 strain in Dermacentor nitens ticks in the northern region of Parana (Brazil). Braz J Microbiol. 2013;44(3):883-7.. Montadon et al. in 20041313. Montandon CE, Yoshinari NH, Milagres BS, Mazioli R, Gomes GG, Moreira HN, et al. Evidence of Borrelia in wild and domestic mammals from the state of Minas Gerais, Brazil. Rev Bras Parasitol Vet. 2014;23(2):287-90. found antibodies against B. burgdorferi in marsupials, horses, rodents, and dogs in two small towns in the state of Minas Gerais, providing evidence of infection with Borrelia spp. in both domestic and wild animals.
Studies in the United States and in Europe found RF-causing Borrelia bacteria in bats and their ticks55. Gill JS, Ullmann AJ, Loftis AD, Schwan TG, Raffel SJ, Schrumpf ME, et al. Novel relapsing fever spirochete in bat tick. J Emerg Infect Dis. 2008;14(3):522-3.-66. Socolovschi C, Mediannikov O, Sokhna C, Tall A, Diatta G, Bassene H, et al. Rickettsia felis-associated uneruptive fever, Senegal. J Emerg Infect Dis. 2010; 16(7):1140-2.. Gill et al. in 200855. Gill JS, Ullmann AJ, Loftis AD, Schwan TG, Raffel SJ, Schrumpf ME, et al. Novel relapsing fever spirochete in bat tick. J Emerg Infect Dis. 2008;14(3):522-3. reported a partial molecular characterization of a new RF-causing spirochete found in a Carios kelleyi tick collected from the basement of a house inhabited by bats in Iowa, United States. In addition, Socolovschi et al. in 201066. Socolovschi C, Mediannikov O, Sokhna C, Tall A, Diatta G, Bassene H, et al. Rickettsia felis-associated uneruptive fever, Senegal. J Emerg Infect Dis. 2010; 16(7):1140-2. collected Argas vespertiolionis ticks from a basement where bats used to live that was transformed into a dormitory. The ticks were found to be infected with Borrelia sp. CPB1, the same RF-causing agent found by Evans et al. in 20091414. Evans NJ, Timofte D, Simpson VR, Birtles RJ. Fatal Borreliosis in Bat Caused by Relapsing Fever Spirochete, United Kingdom. J Emerg Infect Dis . 2009; 15(8):1331-3.. In this study, CPB1 was found to be pathogenic for bats too. Moreover, recently, of Borrelia garinii DNA was found in a tick parasitizing a Myotis daubentonii in a cave in Poland1515. Piksa K, Stańczak J, Biernat B, Górz A, Nowak-Chmura M, Siuda K. Detection ofBorrelia burgdorferi sensu latoand spotted fever group rickettsiae in hard ticks (Acari, Ixodidae) parasitizing bats in Poland. Parasitol Res. 2016; 115(4):1727-31..
These concerns, in addition to the finding of B. burgdorferi in the present study area99. Gonçalves DD, Carreira T, Nunes M, Benitez A, Lopes-Mori FMR, Vidotto O, et al. First record of Borrelia burgdorferi B31 strain in Dermacentor nitens ticks in the northern region of Parana (Brazil). Braz J Microbiol. 2013;44(3):883-7. and the predisposition of bats to participate in several zoonotic cycles because of their lifestyle11. Kotait I, Aguiar EAC, Carrieri ML, Harmani NMS. Manejo de quirópteros em áreas urbanas. Manual Técnico nº 7. São Paulo: Instituto Pasteur; 2003. 45 p., were the reasons motivating our research. We were interested in monitoring the presence of RF Borrelia species in bats to determine whether or not there are favorable conditions for pathogenic RF species.
The negative result for the presence of RF-causing Borrelia among bats found in the city of Maringá, both in the native forest fragments and residences, opposes those of research conducted in Europe and the United States, where these pathogens have been found in bats1414. Evans NJ, Timofte D, Simpson VR, Birtles RJ. Fatal Borreliosis in Bat Caused by Relapsing Fever Spirochete, United Kingdom. J Emerg Infect Dis . 2009; 15(8):1331-3. or their ticks55. Gill JS, Ullmann AJ, Loftis AD, Schwan TG, Raffel SJ, Schrumpf ME, et al. Novel relapsing fever spirochete in bat tick. J Emerg Infect Dis. 2008;14(3):522-3.-66. Socolovschi C, Mediannikov O, Sokhna C, Tall A, Diatta G, Bassene H, et al. Rickettsia felis-associated uneruptive fever, Senegal. J Emerg Infect Dis. 2010; 16(7):1140-2.,1515. Piksa K, Stańczak J, Biernat B, Górz A, Nowak-Chmura M, Siuda K. Detection ofBorrelia burgdorferi sensu latoand spotted fever group rickettsiae in hard ticks (Acari, Ixodidae) parasitizing bats in Poland. Parasitol Res. 2016; 115(4):1727-31.. This can be attributed to the differences in temperature, habitat, and type of ticks that utilize the bats as hosts. Nevertheless, variations occur in Borrelia species, genospecies, and strains as well as in the vectors and vector-pathogen interactions according to the physiogeographic region, resulting in different clinical, epidemiologic, and pathologic disease manifestations1010. Soares CO, Ishikawa MM, Fonseca AH, Yoshinari NH. Borrelioses, agentes e vetores. Pesqui Vet Bras. 2000;20(1):1-19.
11. Fonseca AH, Salles RS, Salles SAN, Madureira RC, Yoshinari NH. Borreliose de Lyme simile: uma doença emergente e relevante para a dermatologia no Brasil. Na Bras Dermatol. 2005;80(2):171-8.-1212. Gouveia EA, Alves MF, Mantovani E, Oyafuso LK, Bonoldi VL, Yoshinari NH. Profile of patients with Baggio-Yoshinari Syndrome admitted at “Instituto de Infectologia Emilio Ribas”. Rev Inst Med Trop Sao Paulo. 2010;52(6):297-303..
In conclusion, the absence of the investigated bacteria in bats in Maringá indicates that these are not infected with RF Borrelia sp. There are no reports of the sampled bat species being infected by Borrelia spp. in other studies, so we cannot conclude that the bacteria or the vector have not been introduced into this area yet, without knowing if the sampled bat species is a competent reservoir. Overall, our findings indicate that endemic RF is not a city health concern at this point. Because of the significant habitat fragmentation of the area of study and the adaptability of bats to manmade constructions, more studies employing other molecular techniques, sampling different habitats from the region studied, evaluating the bats from other areas where Borrelia spp. have been found, and examining ticks that normally feed on chiropterans in this area are recommended.
ACKNOWLEDGMENTS
The authors would like to thank Abha Grover for her support and help while the author was conducting the molecular study in the LymeLab at Texas A&M University and all members of GEEMEA, especially Alexandre Polizel, Mário Dainez, and Carolina Tamura.
REFERENCES
-
1Kotait I, Aguiar EAC, Carrieri ML, Harmani NMS. Manejo de quirópteros em áreas urbanas. Manual Técnico nº 7. São Paulo: Instituto Pasteur; 2003. 45 p.
-
2Díaz P, Remesar S, Venzal JM, Vásquez-López ME, Fernández G, López C, et al. Occurrence of Borrelia and Borreliella species in Ixodes ricinus collected from roe deer in northwestern Spain. Med Vet Entomol. 2019.
-
3Gideon Informatics. Relapsing fever in the United States [Internet]. Berger SA: Relapsing fever in the United States; 2011[update August 23, 2011; quoted 2016 February 6]. Available at: Available at: https://www.gideononline.com/2011/08/23/relapsing-fever-in-the-united-states-2/
» https://www.gideononline.com/2011/08/23/relapsing-fever-in-the-united-states-2/ -
4Pacheco SM, Sodré M, Gama AR, Bredt A, Cavallini-Sanches EM, Marques RV, et al. Morcegos urbanos: status do conhecimento e plano de ação para a conservação no Brasil. Chiropt Neotrop. 2010; 16(1):630-47.
-
5Gill JS, Ullmann AJ, Loftis AD, Schwan TG, Raffel SJ, Schrumpf ME, et al. Novel relapsing fever spirochete in bat tick. J Emerg Infect Dis. 2008;14(3):522-3.
-
6Socolovschi C, Mediannikov O, Sokhna C, Tall A, Diatta G, Bassene H, et al. Rickettsia felis-associated uneruptive fever, Senegal. J Emerg Infect Dis. 2010; 16(7):1140-2.
-
7Wilmer JW, Barrat E. A non-lethal method of tissue sampling for genetic studies of chiropterans. Bat Res News. 1996; 37:1-3.
-
8Ras NM, Lascola B, Postic D, Cutler SJ, Rodhain F, Baranton G, et al. Phylogenesis of relapsing fever Borrelia spp Int J Syst Bacteriol. 1996; 46(4):859-65.
-
9Gonçalves DD, Carreira T, Nunes M, Benitez A, Lopes-Mori FMR, Vidotto O, et al. First record of Borrelia burgdorferi B31 strain in Dermacentor nitens ticks in the northern region of Parana (Brazil). Braz J Microbiol. 2013;44(3):883-7.
-
10Soares CO, Ishikawa MM, Fonseca AH, Yoshinari NH. Borrelioses, agentes e vetores. Pesqui Vet Bras. 2000;20(1):1-19.
-
11Fonseca AH, Salles RS, Salles SAN, Madureira RC, Yoshinari NH. Borreliose de Lyme simile: uma doença emergente e relevante para a dermatologia no Brasil. Na Bras Dermatol. 2005;80(2):171-8.
-
12Gouveia EA, Alves MF, Mantovani E, Oyafuso LK, Bonoldi VL, Yoshinari NH. Profile of patients with Baggio-Yoshinari Syndrome admitted at “Instituto de Infectologia Emilio Ribas”. Rev Inst Med Trop Sao Paulo. 2010;52(6):297-303.
-
13Montandon CE, Yoshinari NH, Milagres BS, Mazioli R, Gomes GG, Moreira HN, et al. Evidence of Borrelia in wild and domestic mammals from the state of Minas Gerais, Brazil. Rev Bras Parasitol Vet. 2014;23(2):287-90.
-
14Evans NJ, Timofte D, Simpson VR, Birtles RJ. Fatal Borreliosis in Bat Caused by Relapsing Fever Spirochete, United Kingdom. J Emerg Infect Dis . 2009; 15(8):1331-3.
-
15Piksa K, Stańczak J, Biernat B, Górz A, Nowak-Chmura M, Siuda K. Detection ofBorrelia burgdorferi sensu latoand spotted fever group rickettsiae in hard ticks (Acari, Ixodidae) parasitizing bats in Poland. Parasitol Res. 2016; 115(4):1727-31.
-
Financial Support: We thank the following for their financial support: Texas A&M AgriLife Research: “Molecular ecology of vector-borne zoonosis in the Gulf of Mexico: A One Health approach” and “Improving diagnostic methods for Lyme Disease, and epidemiology of human and animal infections in TX”, project TEXV6579 (I-9524).
Publication Dates
-
Publication in this collection
01 Aug 2019 -
Date of issue
2019
History
-
Received
09 May 2019 -
Accepted
02 July 2019