ABSTRACT
As they spend most of their time buried in the substrate and are not a fishery resource, heterenchelyids are seldom seen. These eels are characterized by their greatly reduced eyes, which are covered by semi-transparent skin, the absence of a pectoral fin or lateral line, and no pores on the head or body. Pythonichthys sanguineus is a particularly poorly-known species, with only eight scientific records from Cuba, Puerto Rico, Colombia, Venezuela, Guyana, and Suriname. The present study is based on six adult specimens of P. sanguineus captured by vessels of the shrimp trawling fleet along the northern Brazilian coast, in the vicinity of the Amazon Reef. We provide meristic, morphometric and DNA barcoding data. These findings provide insights into the distribution of the species off the northern Brazilian coast and contribute to the discussion about the southern limit of the Greater Caribbean fauna.
KEYWORDS:
fisheries; conservation; Brazilian northern coast; new record; systematics
RESUMO
Heterenchelídeos passam a maior parte do tempo enterrados no substrato e raramente são vistos, não representando recursos pesqueiros importantes. Essas enguias são caracterizadas por seus olhos muito reduzidos e cobertos por pele semi-transparente, ausência de nadadeiras peitorais ou linha lateral, e ausência de poros na cabeça ou no corpo. Pythonichthys sanguineus é uma espécie particularmente pouco conhecida, com apenas oito registros em coleção zoológica para Cuba, Porto Rico, Colômbia, Venezuela, Guiana e Suriname. O presente estudo é baseado em seis espécimes adultos de P. sanguineus capturados pela frota industrial de arrasto de camarão-rosa na costa norte do Brasil, ao largo dos Recifes da Amazônia. Apresentamos dados merísticos, morfométricos e de DNA barcoding dos exemplares examinados. O registro fornece informações importantes sobre a distribuição da espécie na costa norte do Brasil, e contribui para a discussão sobre o limite sul da fauna do Grande Caribe.
PALAVRAS-CHAVE:
pesca; conservação; costa norte do Brasil; novo registro; sistemática
INTRODUCTION
The order Anguilliformes includes 938 species in 19 families and 159 genera (Nelson et al. 2016Nelson, J.S.; Grande, T.C.; Wilson, M.V. 2016. Fishes of the World. John Wiley & Sons, Hoboken, USA. 752p.). The marine mud eels of the family Heterenchelyidae, a sister group to the Myrocongridae and Muraenidae (Inoue et al. 2010Inoue, J.G.; Miya, M.; Miller, M.J.; Sado, T.; Hanel, R.; Hatooka, K.; Aoyama, J.; Minegishi, Y.; Nishida, M.; Tsukamoto, K. 2010. Deep-ocean origin of the freshwater eels. Biology Letters, 6: 363-366. ), are a small group of two genera, Panturichthys Pellegrin 1913 and PythonichthysPoey 1868Poey, F. 1868. Monografia de las Morenas cubanas. Repertorio Fisico-Natural de la Isla de Cuba, 2: 245-268., found in the Mediterranean, tropical Atlantic, and eastern Pacific (Nelson et al. 2016). As they spend most of their time buried in the substrate, heterenchelyids are seldom seen, not representing a fishery resource (Smith 2015Smith, D.G. 2015. Pythonichthys sanguineus. The IUCN Red List of Threatened Species. e.T195661A2398552. ( (https://www.iucnredlist.org/species/195661/2398552
). Accessed on 11 Dec 2017.
https://www.iucnredlist.org/species/1956...
). These eels, which can reach a total length of 1 m, are characterized by their greatly reduced eyes, which are covered by semi-transparent skin, the absence of a pectoral fin or lateral line and no pores on the head or body (Smith 1989Smith, D.G. 1989. Family Heterenchelyidae. In: Böhlke, E.B. (Ed.). Fishes of the Western North Atlantic. v.1. Memoir Sears Foundation for Marine Research. p.48-54.).
At least four species of Pythonichthys are known to exist. Pythonichthys microphthalmus (Regan 1912Regan, C.T. 1912. Description of two new eels from West Africa, belonging to a new genus and family. Annals and Magazine of Natural History, 10: 323-324.) and Pythonichthys macrurus (Regan 1912) are found in the Eastern Atlantic (EA), while Pythonichthys asodesRosenblatt & Rubinoff 1972Rosenblatt, R.H.; Rubinoff, I. 1972. Pythonichthys asodes, a new heterenchelyid eel from the Gulf of Panama. Bulletin of Marine Science, 22: 355-364. occurs in the Eastern Pacific (EP), and Pythonichthys sanguineusPoey 1868Poey, F. 1868. Monografia de las Morenas cubanas. Repertorio Fisico-Natural de la Isla de Cuba, 2: 245-268. is the only species found in the Western Atlantic (WA) (Figure 1a). Smith (1989Smith, D.G. 1989. Family Heterenchelyidae. In: Böhlke, E.B. (Ed.). Fishes of the Western North Atlantic. v.1. Memoir Sears Foundation for Marine Research. p.48-54.) described the history and diversity of the heterenchelyids and redescribed the osteology of P. asodes and P. macrurus.Eagderi and Adriaens (2010Eagderi, S.; Adriaens, D. 2010. Cephalic morphology of Pythonichthys macrurus (Heterenchelyidae: Anguilliformes): specializations for head‐first burrowing. Journal of Morphology, 271: 1053-1065.) also described the osteology of these species, Blache (1977Blache, J. 1977. Leptocephales des poissons anguilliformes dans la zone sud du Golfe de Guinee. ORSTOM, Faune Tropical, 20: 381. ) described the leptocephalus larvae from the Gulf of Guinea. Pythonichthys sanguineus, a senior synonym of Heterenchelys biaggiiBöhlke (1956Böhlke, J.E. 1956. A small collection of new eels from western Puerto Rico. Notulae Naturae, 289: 1-13.) (sensuSmith 1989Smith, D.G. 1989. Family Heterenchelyidae. In: Böhlke, E.B. (Ed.). Fishes of the Western North Atlantic. v.1. Memoir Sears Foundation for Marine Research. p.48-54.; Smith et al. 2012Smith, D.G.; Irmak, E.; Ozen, Ö. 2012. A redescription of the eel Panturichthys fowleri (Anguilliformes: Heterenchelyidae), with a synopsis of the Heterenchelyidae. Copeia, 2012: 484-493.) is the least well known species of the genus, and nothing is known of its biology (Smith 1989Smith, D.G. 1989. Family Heterenchelyidae. In: Böhlke, E.B. (Ed.). Fishes of the Western North Atlantic. v.1. Memoir Sears Foundation for Marine Research. p.48-54., 2002Smith, D.G. 2002. Family Heterenchelyidae. In: Carpenter, K.E. (Ed.). The living marine resources of the Western Central Atlantic . v.2. FAO Species Identification Guide for Fishery Purposes and American Society of Ichthyologists and Herpetologists Special Publication, Rome, p.601-1374.; Smith et al. 2012Smith, D.G.; Irmak, E.; Ozen, Ö. 2012. A redescription of the eel Panturichthys fowleri (Anguilliformes: Heterenchelyidae), with a synopsis of the Heterenchelyidae. Copeia, 2012: 484-493.). Only eight records of adult specimens of P. sanguineus are found in zoological collections (in 10 collection lots), representing six different localities. Two specimens are known from Cuba (MCZ 32805 and USNM 111340) and three from Puerto Rico, including the type specimens of Heterenchelys biaggii (FMNH 61775, ANSP 73872 and FMNH 61774). From South America, five specimens are available from Venezuela (four specimens, MBUCV 293 and one specimen from the Fernandez Yepez’s collection), and one each from Colombia (UMML 222150), Guyana (BMNH 1961.8.31.99) and Suriname (NSMT 40060), while the 11 specimens examined by Poey (1868) have been lost (Faloh-Gandarilla et al. 2016Faloh-Gandarilla, I.; Gutiérrez, De R.E.; Orozco, M.V.; Cortés, R.; Alfonso, Y.; Lemus, E.; Álvarez, I.L; Pérez, De R.R. 2016. Catálogo de especímenes tipo de peces cubanos (Osteichthyes, clase: Actinopterygii. ordenes: Onguillifomes, Ateleopiformes, Atheriniformes, Aulopiformes, Beloniformes, Beryciformes, Clupeiformes). Parte I. Poeyana Revista Cubana de Zoología, 502: 27-31. ).
Pythonichthys sanguineus, body and head in lateral view. A. Holotype drawn by Poey (1868Poey, F. 1868. Monografia de las Morenas cubanas. Repertorio Fisico-Natural de la Isla de Cuba, 2: 245-268.); B-C. Cataloged Brazilian specimen (MPEG 35269); D-E. Non- cataloged Brazilian specimen. This figure is in color in the eletronic version.
Considering the general lack of data on the fish diversity of the northern coast of Brazil (Marceniuk et al. 2013Marceniuk, A.P.; Caires, R.; Wosiacki, W.B.; Di Dario, F. 2013. Conhecimento e conservação dos peixes marinhos e estuarinos (Chondrichthyes e Teleostei) da Costa Norte do Brasil. Biota Neotropica,13: 251-259. ; Marceniuk et al. 2017Marceniuk, A.P.; Caires, R.; Rotundo, M.M.; Alcantara, R.A.K.; Wosiacki, W.B. 2017. The icthyofauna (Teleostei) of the Rio Caeté estuary, northeast Pará, Brazil, with a species identification key from northern Brazilian coast. PanamJAS, 12: 31-79.), the Centro de Pesquisa e Gestão de Recursos Pesqueiros do Litoral Norte (CEPNOR), together with taxonomists from the Museu Paraense Emílio Goeldi (MPEG), in Belém, Brazil, has obtained specimens from the industrial shrimp otter trawling fleet based in the Brazilian state of Pará, in a concentrated effort to expand the scientific inventory of the marine fish diversity of the region. This sampling effort has resulted in the collection of six specimens of P. sanguineus. Meristic, morphometric and DNA barcoding data are presented (cataloged specimens), and provide important insights into the distribution and diversity of the genus in the western Atlantic.
MATERIAL AND METHODS
The present report on Pythonichthys sanguineus is based on three adult specimens collected in Amapá state on April 28, 2017 and March 2-3, 2018 (cataloged, Figure1b,c) and other three adult specimens captured on March 5, 2018 (not cataloged, Figure 1d,e), all captured by vessels of the shrimp trawling fleet. The cataloged specimens were deposited in the zoological collections of Museu Paraense Emílio Goeldi (MPEG 35269, 493 mm TL (total length), 2°19’42”N, 48°36’46”W, 50 m), Acervo Zoológico da Universidade Santa Cecília (AZUSC 5314, 650 mm TL, 2°19’39”N, 48°26’35”W, 50 m), and the fish collection of Laboratório de Biologia e Genética de Peixes of Universidade Estadual Paulista Júlio de Mesquita Filho (LBP 26612, 431 mm TL, 1°52’45”N, 48°16’32”W, 44 m) (see Material Examined). The specimens were identified based on the original description and subsequent references (Smith 1989Smith, D.G. 1989. Family Heterenchelyidae. In: Böhlke, E.B. (Ed.). Fishes of the Western North Atlantic. v.1. Memoir Sears Foundation for Marine Research. p.48-54., 2002Smith, D.G. 2002. Family Heterenchelyidae. In: Carpenter, K.E. (Ed.). The living marine resources of the Western Central Atlantic . v.2. FAO Species Identification Guide for Fishery Purposes and American Society of Ichthyologists and Herpetologists Special Publication, Rome, p.601-1374., Smith et al. 2012Smith, D.G.; Irmak, E.; Ozen, Ö. 2012. A redescription of the eel Panturichthys fowleri (Anguilliformes: Heterenchelyidae), with a synopsis of the Heterenchelyidae. Copeia, 2012: 484-493.), and measured according to Smith (1989Smith, D.G. 1989. Family Heterenchelyidae. In: Böhlke, E.B. (Ed.). Fishes of the Western North Atlantic. v.1. Memoir Sears Foundation for Marine Research. p.48-54.). All measurements were taken using either a digital calliper (precision 0.01 mm) or a ruler (precision 1 mm) for dimensions over 200 mm. The data on the occurrence of P. sanguineus were obtained from the zoological collection records available at http://www.fishnet2.net and in Smith (1989).
Molecular analysis
Molecular identification of the specimens was done according to the DNA barcoding protocol (Hebert et al. 2003Hebert, P.D.N.; Cywinska, A.; Ball, S.L.; De Waard, J.R. 2003. Biological identifications through DNA barcodes. Proceedings of the Royal Society of London B, 270: 313-322. ) and deposited in the BOLD system (Ratnasingham and Hebert 2007Ratnasingham, S.; Hebert, P.D.N. 2007. BOLD: The Barcode of Life Data System (www.barcodinglife.org). Molecular Ecology Notes, 7: 355-364. ) and in Genbank (https://www.ncbi.nlm.nih.gov/genbank). A partial COI sequence (578 bps) was obtained from a single P. sanguineus specimen deposited at Museu Paraense Emílio Goeldi (MPEG 35269), and deposited in the Genbank database (accession number: MG833002) and BOLD (RMRBR001-18). For comparison, COI sequences of additional 57 anguilliforms were obtained from NCBI (see Supplementary Material, Appendix S1). These sequences were derived from a phylogenetic study that analyzed the mitochondrial genomes of all 19 anguilliform families (Inoue et al. 2010Inoue, J.G.; Miya, M.; Miller, M.J.; Sado, T.; Hanel, R.; Hatooka, K.; Aoyama, J.; Minegishi, Y.; Nishida, M.; Tsukamoto, K. 2010. Deep-ocean origin of the freshwater eels. Biology Letters, 6: 363-366. ) and represent an almost complete sample of Anguilliformes at genus level.
The molecular assays were conducted at the Laboratory of Fish Biology and Genetics at Universidade Estadual Paulista Júlio de Mesquita Filho in Botucatu, São Paulo, Brazil. Total genomic DNA was isolated from muscular tissue using an automated glass fiber protocol (Ivanova et al. 2008Ivanova, N.V.; Fazekas, A.J.; Hebert, P.D.N. 2008. Semi-automated, membrane-based protocol for DNA isolation from plants. Plant Molecular Biology, 26: 186-198.). The fragment at the 5’ end of the mitochondrial COI gene was amplified by PCR using the primers FishF1 and FishR1 (Ward et al. 2005Ward, R.D.; Zemlak, T.S.; Innes, B.H.; Last, P.R.; Hebert, P.D. 2005. DNA barcoding Australia’s fish species. Philosophical Transactions of the Royal Society B, 360: 1847-1857.). The reactions were carried out in a 12.5 µL reaction volume containing 1.25 µL of 10 × PCR buffer, 0.375 µL MgCl2 (50 mM), 0.5 µL of the dNTPs (2 mM), 0.25 µL of each primer (5 µM), 0.2 µL of 1.25 U Taq platinum DNA polymerase, and 1 µL of the DNA template (200 ng), and 8.675 µL of ultrapure water. The PCR cycle was as follows: 95°C for 3 min, followed by 35 cycles of 95°C for 30 s, 54°C for 45 s, 72°C for 1 min, and then 72°C for 5 min. The PCR products were visualized in a 1% agarose gel and purified using ExoSAP-IT (USB Europe GmbH, Staufen, Germany) at 37°C for 60 min followed by 15 min at 80°C. The sequencing reactions were conducted using a BigDye Terminator Cycle Sequencing kit (Applied Biosystems, California, USA) according to the manufacturer’s instructions. The sequences were read in both the forward and reverse directions, and the primers used for sequencing were the same as those used for the PCR reaction. Finally, the labeled sequences were assessed using an ABI PRISM 3130 Genetic Analyzer (Applied Biosystems, California, USA).
Bidirectional sequences were assembled using Geneious v.5.6 (Kearse et al. 2012Kearse, M.; Moir, R.; Wilson, A.; Stones-Havas, S.; Cheung, M.; Sturrock, S.; et al. 2012. Geneious Basic: An integrated and extendable desktop software platform for the organization and analysis of sequence data. Bioinformatics. 28: 1647-1649. ) to obtain consensus sequences and to check for indels or stop codons. Indels or stop codons, which may indicate the amplification of nuclear mithocondrial DNA segment (NUMTs), were absent from all sequences. Genetic distances between and within taxonomic groups were calculated using the Kimura 2-parameter (K2P) substitution model (Kimura 1980Kimura, M. 1980. A simple method for estimating evolutionary rate of base substitutions through comparative studies of nucleotide sequences. Journal of Molecular Biology, 16: 111-120. ) in MEGA 7 (Kumar et al. 2016Kumar, S.; Stecher, G.; Tamura, K. 2016. MEGA7: Molecular Evolutionary Genetics Analysis version 7.0 for bigger datasets. Molecular Biology and Evolution, 33: 1870-1874.). The best model of nucleotide substitution was GTR+G+I as calculated in MEGA 7 (Kumar et al. 2016). The maximum likelihood tree was obtained in MEGA 7 (Kumar et al. 2016) using the GTR+G+I model, with node supports estimated by 1000 nonparametric bootstrap replicates (Felsenstein 1985Felsenstein, J. 1985. Confidence limits on phylogenies: an approach using the bootstrap. Evolution, 39: 783-791.).
RESULTS
Pythonichthys sanguineus Poey 1868Poey, F. 1868. Monografia de las Morenas cubanas. Repertorio Fisico-Natural de la Isla de Cuba, 2: 245-268.
Pythonichthys sanguineusPoey 1868Poey, F. 1868. Monografia de las Morenas cubanas. Repertorio Fisico-Natural de la Isla de Cuba, 2: 245-268.: 265 (original description); Uyeno and Sasaki in Uyeno et al. 1983Uyeno, T.; Sasaki, T. 1983. Scapanorhynchidae. In: Uyeno, T.; Matsuura, K.; Fujii, E. (Ed.). Fishes trawled off Suriname and French Guiana. Japan Marine Fishery Resource Research Center, Tokyo, p.48. :101 (Suriname); Smith 1989Smith, D.G. 1989. Family Heterenchelyidae. In: Böhlke, E.B. (Ed.). Fishes of the Western North Atlantic. v.1. Memoir Sears Foundation for Marine Research. p.48-54.:52 (taxonomic considerations); Cervigón 1991Cervigón, F. 1991. Los peces marinos de Venezuela. Fundación Científica Los Roques, Caracas, 295p.:29 (Venezuela); Aguilera 1998Aguilera, O. 1998. Los Peces Marinos del Occidente de Venezuela. Acta Biologica Venezuelica,18: 43-57.:46 (Venezuela); Smith 2002Smith, D.G. 2002. Family Heterenchelyidae. In: Carpenter, K.E. (Ed.). The living marine resources of the Western Central Atlantic . v.2. FAO Species Identification Guide for Fishery Purposes and American Society of Ichthyologists and Herpetologists Special Publication, Rome, p.601-1374.:694 (Western Central Atlantic); Smith et al. 2012Smith, D.G.; Irmak, E.; Ozen, Ö. 2012. A redescription of the eel Panturichthys fowleri (Anguilliformes: Heterenchelyidae), with a synopsis of the Heterenchelyidae. Copeia, 2012: 484-493.:492 (Caribbean); Faloh-Gandarilla et al. 2016Faloh-Gandarilla, I.; Gutiérrez, De R.E.; Orozco, M.V.; Cortés, R.; Alfonso, Y.; Lemus, E.; Álvarez, I.L; Pérez, De R.R. 2016. Catálogo de especímenes tipo de peces cubanos (Osteichthyes, clase: Actinopterygii. ordenes: Onguillifomes, Ateleopiformes, Atheriniformes, Aulopiformes, Beloniformes, Beryciformes, Clupeiformes). Parte I. Poeyana Revista Cubana de Zoología, 502: 27-31. :28 (Cuba).
Heterenchelys biaggiiBöhlke 1956Böhlke, J.E. 1956. A small collection of new eels from western Puerto Rico. Notulae Naturae, 289: 1-13.:8, Fig. 2A (original description, Puerto Rico).
Diagnosis
Some authors have expressed doubts about the distinction between the genera Pythonichthys and Panturichthys (Rosenblatt and Rubinoff 1972Rosenblatt, R.H.; Rubinoff, I. 1972. Pythonichthys asodes, a new heterenchelyid eel from the Gulf of Panama. Bulletin of Marine Science, 22: 355-364.). The identification of the specimens analyzed here as Pythonichthys was supported by the diagnostic characters proposed by Blache (1968Blache, J. 1968. Contribution à la connaissance des poisons angulliformes de la cote occidentale d’Afrique, neuvième note: lês Heterenchelyidae. Bulletin de I’I.F.A.N., 30:c1540-1581.): the absence of a longitudinal cutaneous ridge on the upper surface of the head (vs. present in Panturichthys), the absence of the fleshy fold located posteriorly on the inner edge of the maxilla (vs. present in Panturichthys), the presence of a gap between the vomerine and ethmoid dentition (vs. absent in Panturichthys), the inner row of maxillary teeth is approximately equal in length to the outer row (vs. shorter in Panturichthys), the pectoral girdle originates dorsally (vs. on or slightly anterior to the pectoral waist in Panturichthys), head and trunk 22-36% TL (vs. 12-20% TL in Panturichthys, Table 1), and 109-134 vertebrae (vs. 141-227 in Panturichthys, Table 1).
While counts of vertebrae and proportional measurements can be used to distinguish P. sanguineus from P. macrurus (EA) and P. asodes (EP), these parameters are highly similar to those observed in P. microphthalmus (EA). Pythonichthys sanguineus can nevertheless be distinguished from P. microphthalmus by the number of vomerine tooth rows (2-3), in contrast with a single row in P. microphthalmus, and 3-5 maxillary and mandibular tooth rows (vs. 2 rows in P. microphthalmus). The two species are also differentiated clearly by the molecular data (see below).
Morphometric and meristic data of three specimens of Pythonichthys sanguineus captured in the vicinity of the Amazon Reef and other geographic areas (Smith, 1989Smith, D.G. 1989. Family Heterenchelyidae. In: Böhlke, E.B. (Ed.). Fishes of the Western North Atlantic. v.1. Memoir Sears Foundation for Marine Research. p.48-54.), expressed in millimeters (mm), percent of total length (TL%) or percent of standard length (SL%).
Molecular analysis
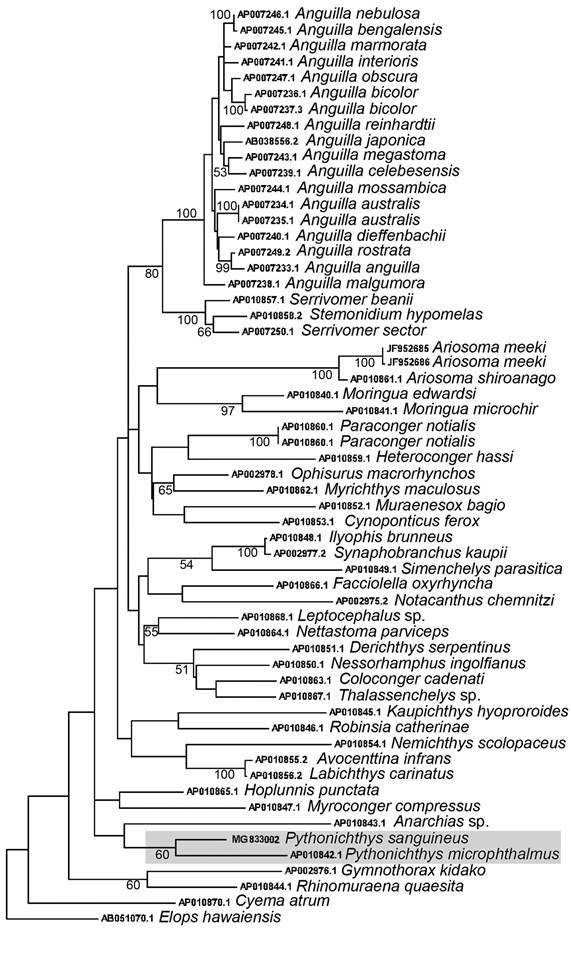
Maximum likelihood analysis showed that the specimen analyzed here clusters with Pythonichthys microphthalmus, with a bootstrap support of 60% (Figure 2), corroborating the assignment of our specimens to the genus Pythonichthys. The K2P genetic distance between P. sanguineus and P. microphthalmus was 25.0 ± 1.9%, with 89/578 positions being different between species, corroborating our hypotheses that these are in fact different species.
Maximum likelihood tree of the Anguilliformes species analyzed, using GTR+G+I genetic model. Bootstrap values greater than 50 are shown. Species of the genus Pythonichthys are highlighted in gray.
Morphological measurements
The morphometric and meristic parameters of the specimens analyzed in here did not exceed the amplitude of values presented in the original description or subsequent studies (Table 1).
Color Pattern
Scharpf and Lazara (2017Scharpf, C.; Lazara, K.J. 2017. The ETYFish Project. ( (http://www.etyfish.org/wp-content/uploads/2013/10/ETYFish_Petromyzontiformes.pdf
). Accessed on 20 Dec 2017.
http://www.etyfish.org/wp-content/upload...
) refer to the coloration of fresh Pythonichthys sanguineus as blood-red. The living specimens (Figure 1d,e) presented the same coloration described by Scharpf and Lazara (2017), while the fresh specimen was light gray, with a brownish anterior dorsal region extending to approximately the anus, and a pale belly (Figure 1b,c). The distal portions of the dorsal and anal fins were blackened posteriorly from the median region of the body.
Distribution
Tropical Western Atlantic Ocean, from Cuba to Brazil (Figure 3). The present study extends the occurrence of the Atlantic mud eels from Suriname to Amapá state in northern Brazil.
Records of Pythonichthys sanguineus in the western Atlantic. Type-locality of Pythonichthys sanguineus = orange; type-locality of Heterenchelys biaggii = green; non-type specimens = light blue; Brazilian specimens = yellow; doubtful records = red; Pirabas Formation (Aguilera et al. 2014Aguilera, O.; Schwarzhans, W.; Moraes-Santos, H.; Nepomuceno, A. 2014. Before the flood: Miocene otoliths from eastern Amazon Pirabas Formation reveal a Caribbean-type fish fauna. Journal of South American Earth Sciences, 56: 422-446.) = white star; Amazon Reef = dark blue. This figure is in color in the eletronic version.
Doubtful records from the Brazilian coast
The occurrence of Pythonichthys sanguineus in Brazilian waters was reported by Miranda-Ribeiro (1919Miranda-Ribeiro, A. 1919. A fauna vertebrada da ilha da Trindade. Arquivos do Museu Nacional do Rio de Janeiro, 22: 171-193.). This record was based on the observation (no specimen collected) of an eel at Trindade Island, off the eastern coast of Brazil, which was identified as Pythonichthys sanguineus based on its intense red coloration. This record was considered doubtful by Simon et al. (2013Simon, T.; Macieira, R.M.; Joyeux, J.C. 2013. The shore fishes of the Trindade-Martin Vaz insular complex: an update. Journal of Fish Biology, 82: 2113-2127.) and was not accepted by Pinheiro et al. (2015Pinheiro, H.T.; Mazzei, E.; Moura, R.L.; Amado-Filho, G.M.; Carvalho-Filho, A.; Braga, A.C.; et al. 2015. Fish biodiversity of the Vitória-Trindade Seamount Chain, Southwestern Atlantic: an updated database. PLoS One, 10: e0118180. ). Hudson Pinheiro (personal information) confirmed that the species does not occur at Trindade Island (Figure 3).
Biology, habitat, depth, and presumed schooling pattern
There is very little information available for this species, that inhabits muddy substrates of reefs and rocky areas (Smith 2015Smith, D.G. 2015. Pythonichthys sanguineus. The IUCN Red List of Threatened Species. e.T195661A2398552. ( (https://www.iucnredlist.org/species/195661/2398552
). Accessed on 11 Dec 2017.
https://www.iucnredlist.org/species/1956...
). Pythonichthys sanguineus has previously been recorded at depths of 12-55 m. The cataloged specimens were captured on a gravel bottom, at a depth of between 44 and 50 m, at 2°19’42”N, 48°36’46”W to 1°52’45”N, 48°16’32”W (Figure 3), and the non cataloged specimens were captured on a gravel bottom, at a depth of 70 m, at 2°26’73”N, 48°41’52”W (Figure 3), both sites in the vicinity of the Amazon Reef (Moura et al. 2016Moura, R.L.; Amado-Filho, G.M.; Moraes, F.C.; Brasileiro, P.S.; Salomon, P.S.; Mahiques, M.M.; et al. 2016. An extensive reef system at the Amazon River mouth. Science Advances, 2: e1501252.; Francini-Filho et al. 2018Francini-Filho, R.B.; Asp, N.E.; Siegle, E.; Hocevar, J.; Lowyck, K.; D’Avila, N.; et al. 2018. Perspectives on the Great Amazon Reef: Extension, Biodiversity, and Threats. Frontiers in Marine Science, 5:142. ).
Fisheries and conservation
Otter trawling is practiced worldwide, and is characterized by the largest discarded catches, causing profound impacts on the environment through the destruction of bottom substrates and the elimination of benthic organisms such as coral reefs (Stobutzki et al. 2001Stobutzki, I.; Miller, M.; Brewer, D. 2001. Sustainability of fishery bycatch: a process for assessing highly diverse and numerous bycatch. Environmental Conservation, 28: 167-181.). In this context, the capture of the mud eel specimens reported here always ocurred after the end of the closure periods determined by the government to protect shrimp stocks. No other specimens were caught after the beginning of the shrimping season, indicating that the fishing activity may have influence on the occurrence of the species in the fishing zone, as this pattern was observed during the two sampling seasons. This question clearly requires further investigation, given the well-known impact of otter trawling on benthic communities.
DISCUSSION
Pythonichthys sanguineus is the only species of the genus found in the tropical western Atlantic, and is known from localities in the Caribbean and northern South America, being recognized as part of the typical Greater Caribbean fauna (sensuRobertson and Cramer 2014Robertson, D.R.; Cramer, K.L. 2014. Defining and dividing the greater Caribbean: insights from the biogeography of shorefishes. PloS One, 9: e102918.). The present record of P. sanguineus from the vicinity of the Amazon Reef (Moura et al. 2016Moura, R.L.; Amado-Filho, G.M.; Moraes, F.C.; Brasileiro, P.S.; Salomon, P.S.; Mahiques, M.M.; et al. 2016. An extensive reef system at the Amazon River mouth. Science Advances, 2: e1501252.; Francini-Filho et al. 2018Francini-Filho, R.B.; Asp, N.E.; Siegle, E.; Hocevar, J.; Lowyck, K.; D’Avila, N.; et al. 2018. Perspectives on the Great Amazon Reef: Extension, Biodiversity, and Threats. Frontiers in Marine Science, 5:142. ) is relevant to the discussion on the range of the southern limit of the Greater Caribbean fauna, which has been inferred by different authors to reach the region from the Gulf of Paria in Venezuela to Suriname (Petruch 2013) or specific points on the Brazilian coast (Robins 1971Robins, C.R. 1971. Distributional patterns of fishes from coastal and shelf waters of the tropical western Atlantic. Symposium on Investigations and Resources of the Caribbean Sea and Adjacent Regions. Papers on Fisheries Resources, FAO, Rome. p.249-255. ), an imprecision that stems from the lack of any prominent physical barriers in the region. In this context, the Amazonian-Orinoco plume, formed by the rearrangements of the hydrographic basins of the Amazon (Shepard et al. 2010Shephard, G.E.; Muller, R.D.; Liu, L.; Gumis, M. 2010. Miocene drainage reversal of the Amazon River driven by plate-mantle interaction. Nature Geoscience, 3: 870-875. ) and the Orinoco (Rod 1981Rod, E. 1981. Notes on the shifting course of the ancient Rio Orinoco from late Cretaceous to Oligocene time. GEOS, 26: 54-56.), during the middle to late Miocene (7.2 mya) (Figueiredo et al. 2009Figueiredo, J.; Hoorn, C.V.P.; Soares, E. 2009. Late Miocene onset of the Amazon River and the Amazon deep-sea fan: evidence from the Foz do Amazonas Basin. Geology, 37: 19-622.; Hoorn et al. 2017Hoorn, C.; Bogotá, A.G.R.; Romero-Baez, M.; Lammertsma, E.I.D.; Flantua, S.G.A.; Dantas, E.L.; Dino, R.; Do Carmo, D.A.; Chemale, F. Jr. 2017. The Amazon at sea: Onset and stages of the Amazon River from a marine record, with special reference to Neogene plant turnover in the drainage basin. Global Planet Change. http://dx.doi.org/10.1016/j.gloplacha.2017.02.005.
http://dx.doi.org/10.1016/j.gloplacha.20...
), has been recognized as an important barrier to the dispersal of coral reef fishes of the Caribbean and Brazil (Bernal and Rocha 2011Bernal, M.A.; Rocha, L.A. 2011. Acanthurus tractus Poey, 1860, a valid western Atlantic species of surgeonfish (Teleostei, Acanthuridae), distinct from Acanthurus bahianus Castelnau, 1855. Zootaxa, 2905: 63-68.).
Aguilera et al. (2014Aguilera, O.; Schwarzhans, W.; Moraes-Santos, H.; Nepomuceno, A. 2014. Before the flood: Miocene otoliths from eastern Amazon Pirabas Formation reveal a Caribbean-type fish fauna. Journal of South American Earth Sciences, 56: 422-446.) described Pythonichthys pirabasensis based on a fossil from the Aquitanian period of the early Miocene (20.4-23.0 mya), from the Pirabas Formation of Atalaia Beach (municipality of Salinópolis, in the northern Brazilian state of Pará) and concluded that the current interruption of the shallow marine bioprovince, found off the Amazon delta cannot be perceived in the fish fauna of the Pirabas Formation, which is closely related to that of the Gatunian/proto-Caribbean bioprovince (Petuch 1982Petuch, E.J. 1982. Geographical heterochrony: contemporaneous coexistence of Neogene and recent molluscan faunas in the Americas. Palaeogeography, Palaeoclimatology, Palaeoecology, 37: 277-312., 2013Petuch, E.J. 2013. Biodiversity and Biogeography of Western Atlantic Mollusks. CRC Press, Boca Raton, USA. 380p.; Landau et al. 2008Landau, B.; Vermeij, G.; Da Silva, C.M. 2008. Southern Caribbean Neogene palaeobiogeography revisited. New data from the Pliocene of Cubagua, Venezuela. Palaeogeography, Palaeoclimatology, Palaeoecology, 257: 445-461.). The shift of the mouth of the Amazon River, close to its present day location, has terminated the carbonate cycle of the Pirabas Formation, pushing the Pirabas fish fauna northwards, including Pythonichthys and the congrid genus Paraconger Kanazawa 1961, also captured with the Pythonichthys specimens described herein (A.P. Marceniuk, unpublished data).
Our record of Pythonichthys sanguineus indicates that the shift in the location of the Amazon plume may not have displaced the fauna of the Pirabas Formation further north, but only modified the distribution of these lineages from the coastal zone further offshore, to an area where environmental conditions are more similar to those of the Pirabas Formation of the early Miocene (Aquitanian). This area coincides with the recently-discovered coral reefs off the mouth of the Amazon. The fauna associated with the reefs is still poorly-known, but is possibly closely related to those of the Pirabas Formation and the present-day Caribbean (Robertson and Cramer 2014Robertson, D.R.; Cramer, K.L. 2014. Defining and dividing the greater Caribbean: insights from the biogeography of shorefishes. PloS One, 9: e102918.).
ACKNOWLEDGMENTS
The authors are grateful to Alex Garcia Cavalleiro de Macedo Klautau, coordinator of the Centro de Pesquisa e Gestão de Recursos Pesqueiros do Litoral Norte (CEPNOR), and Wolmar Benjamin Wosiacki, curator of the fish collection of the Museu Paraense Emílio Goeldi (MPEG), for the opportunity to examine the material collected on the north coast of Brazil. This study was supported by Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) (grants nr. 300462/2016-6 to APM, 306054/2006-0 to CO), the Fundação Amazônia Paraense de Amparo à Pesquisa (FAPESPA) (grant nr. ICAAF 017/2016 to APM), and Fundação de Amparo à Pesquisa do Estado de São Paulo (FAPESP) (grant nr. 2016/09204-6 to CO).
- Aguilera, O. 1998. Los Peces Marinos del Occidente de Venezuela. Acta Biologica Venezuelica,18: 43-57.
- Aguilera, O.; Schwarzhans, W.; Moraes-Santos, H.; Nepomuceno, A. 2014. Before the flood: Miocene otoliths from eastern Amazon Pirabas Formation reveal a Caribbean-type fish fauna. Journal of South American Earth Sciences, 56: 422-446.
- Bernal, M.A.; Rocha, L.A. 2011. Acanthurus tractus Poey, 1860, a valid western Atlantic species of surgeonfish (Teleostei, Acanthuridae), distinct from Acanthurus bahianus Castelnau, 1855. Zootaxa, 2905: 63-68.
- Blache, J. 1968. Contribution à la connaissance des poisons angulliformes de la cote occidentale d’Afrique, neuvième note: lês Heterenchelyidae. Bulletin de I’I.F.A.N., 30:c1540-1581.
- Blache, J. 1977. Leptocephales des poissons anguilliformes dans la zone sud du Golfe de Guinee. ORSTOM, Faune Tropical, 20: 381.
- Böhlke, J.E. 1956. A small collection of new eels from western Puerto Rico. Notulae Naturae, 289: 1-13.
- Cervigón, F. 1991. Los peces marinos de Venezuela Fundación Científica Los Roques, Caracas, 295p.
- Eagderi, S.; Adriaens, D. 2010. Cephalic morphology of Pythonichthys macrurus (Heterenchelyidae: Anguilliformes): specializations for head‐first burrowing. Journal of Morphology, 271: 1053-1065.
- Faloh-Gandarilla, I.; Gutiérrez, De R.E.; Orozco, M.V.; Cortés, R.; Alfonso, Y.; Lemus, E.; Álvarez, I.L; Pérez, De R.R. 2016. Catálogo de especímenes tipo de peces cubanos (Osteichthyes, clase: Actinopterygii. ordenes: Onguillifomes, Ateleopiformes, Atheriniformes, Aulopiformes, Beloniformes, Beryciformes, Clupeiformes). Parte I. Poeyana Revista Cubana de Zoología, 502: 27-31.
- Felsenstein, J. 1985. Confidence limits on phylogenies: an approach using the bootstrap. Evolution, 39: 783-791.
- Figueiredo, J.; Hoorn, C.V.P.; Soares, E. 2009. Late Miocene onset of the Amazon River and the Amazon deep-sea fan: evidence from the Foz do Amazonas Basin. Geology, 37: 19-622.
- Francini-Filho, R.B.; Asp, N.E.; Siegle, E.; Hocevar, J.; Lowyck, K.; D’Avila, N.; et al 2018. Perspectives on the Great Amazon Reef: Extension, Biodiversity, and Threats. Frontiers in Marine Science, 5:142.
- Hall, M.A. 1996. On bycatches. Reviews in Fish Biology and Fisheries, 6: 319-352.
- Hebert, P.D.N.; Cywinska, A.; Ball, S.L.; De Waard, J.R. 2003. Biological identifications through DNA barcodes. Proceedings of the Royal Society of London B, 270: 313-322.
- Hoorn, C.; Bogotá, A.G.R.; Romero-Baez, M.; Lammertsma, E.I.D.; Flantua, S.G.A.; Dantas, E.L.; Dino, R.; Do Carmo, D.A.; Chemale, F. Jr. 2017. The Amazon at sea: Onset and stages of the Amazon River from a marine record, with special reference to Neogene plant turnover in the drainage basin. Global Planet Change http://dx.doi.org/10.1016/j.gloplacha.2017.02.005
» http://dx.doi.org/10.1016/j.gloplacha.2017.02.005 - Inoue, J.G.; Miya, M.; Miller, M.J.; Sado, T.; Hanel, R.; Hatooka, K.; Aoyama, J.; Minegishi, Y.; Nishida, M.; Tsukamoto, K. 2010. Deep-ocean origin of the freshwater eels. Biology Letters, 6: 363-366.
- Ivanova, N.V.; Fazekas, A.J.; Hebert, P.D.N. 2008. Semi-automated, membrane-based protocol for DNA isolation from plants. Plant Molecular Biology, 26: 186-198.
- Kearse, M.; Moir, R.; Wilson, A.; Stones-Havas, S.; Cheung, M.; Sturrock, S.; et al 2012. Geneious Basic: An integrated and extendable desktop software platform for the organization and analysis of sequence data. Bioinformatics 28: 1647-1649.
- Kimura, M. 1980. A simple method for estimating evolutionary rate of base substitutions through comparative studies of nucleotide sequences. Journal of Molecular Biology, 16: 111-120.
- Kumar, S.; Stecher, G.; Tamura, K. 2016. MEGA7: Molecular Evolutionary Genetics Analysis version 7.0 for bigger datasets. Molecular Biology and Evolution, 33: 1870-1874.
- Landau, B.; Vermeij, G.; Da Silva, C.M. 2008. Southern Caribbean Neogene palaeobiogeography revisited. New data from the Pliocene of Cubagua, Venezuela. Palaeogeography, Palaeoclimatology, Palaeoecology, 257: 445-461.
- Marceniuk, A.P.; Caires, R.; Wosiacki, W.B.; Di Dario, F. 2013. Conhecimento e conservação dos peixes marinhos e estuarinos (Chondrichthyes e Teleostei) da Costa Norte do Brasil. Biota Neotropica,13: 251-259.
- Marceniuk, A.P.; Caires, R.; Rotundo, M.M.; Alcantara, R.A.K.; Wosiacki, W.B. 2017. The icthyofauna (Teleostei) of the Rio Caeté estuary, northeast Pará, Brazil, with a species identification key from northern Brazilian coast. PanamJAS, 12: 31-79.
- Miranda-Ribeiro, A. 1919. A fauna vertebrada da ilha da Trindade. Arquivos do Museu Nacional do Rio de Janeiro, 22: 171-193.
- Moura, R.L.; Amado-Filho, G.M.; Moraes, F.C.; Brasileiro, P.S.; Salomon, P.S.; Mahiques, M.M.; et al 2016. An extensive reef system at the Amazon River mouth. Science Advances, 2: e1501252.
- Nelson, J.S.; Grande, T.C.; Wilson, M.V. 2016. Fishes of the World John Wiley & Sons, Hoboken, USA. 752p.
- Ratnasingham, S.; Hebert, P.D.N. 2007. BOLD: The Barcode of Life Data System (www.barcodinglife.org). Molecular Ecology Notes, 7: 355-364.
- Regan, C.T. 1912. Description of two new eels from West Africa, belonging to a new genus and family. Annals and Magazine of Natural History, 10: 323-324.
- Petuch, E.J. 1982. Geographical heterochrony: contemporaneous coexistence of Neogene and recent molluscan faunas in the Americas. Palaeogeography, Palaeoclimatology, Palaeoecology, 37: 277-312.
- Petuch, E.J. 2013. Biodiversity and Biogeography of Western Atlantic Mollusks CRC Press, Boca Raton, USA. 380p.
- Pinheiro, H.T.; Mazzei, E.; Moura, R.L.; Amado-Filho, G.M.; Carvalho-Filho, A.; Braga, A.C.; et al 2015. Fish biodiversity of the Vitória-Trindade Seamount Chain, Southwestern Atlantic: an updated database. PLoS One, 10: e0118180.
- Poey, F. 1868. Monografia de las Morenas cubanas. Repertorio Fisico-Natural de la Isla de Cuba, 2: 245-268.
- Robertson, D.R.; Cramer, K.L. 2014. Defining and dividing the greater Caribbean: insights from the biogeography of shorefishes. PloS One, 9: e102918.
- Robins, C.R. 1971. Distributional patterns of fishes from coastal and shelf waters of the tropical western Atlantic. Symposium on Investigations and Resources of the Caribbean Sea and Adjacent Regions. Papers on Fisheries Resources, FAO, Rome. p.249-255.
- Rod, E. 1981. Notes on the shifting course of the ancient Rio Orinoco from late Cretaceous to Oligocene time. GEOS, 26: 54-56.
- Rosenblatt, R.H.; Rubinoff, I. 1972. Pythonichthys asodes, a new heterenchelyid eel from the Gulf of Panama. Bulletin of Marine Science, 22: 355-364.
- Scharpf, C.; Lazara, K.J. 2017. The ETYFish Project. ( (http://www.etyfish.org/wp-content/uploads/2013/10/ETYFish_Petromyzontiformes.pdf ). Accessed on 20 Dec 2017.
» http://www.etyfish.org/wp-content/uploads/2013/10/ETYFish_Petromyzontiformes.pdf - Shephard, G.E.; Muller, R.D.; Liu, L.; Gumis, M. 2010. Miocene drainage reversal of the Amazon River driven by plate-mantle interaction. Nature Geoscience, 3: 870-875.
- Simon, T.; Macieira, R.M.; Joyeux, J.C. 2013. The shore fishes of the Trindade-Martin Vaz insular complex: an update. Journal of Fish Biology, 82: 2113-2127.
- Smith, D.G. 1989. Family Heterenchelyidae. In: Böhlke, E.B. (Ed.). Fishes of the Western North Atlantic v.1. Memoir Sears Foundation for Marine Research. p.48-54.
- Smith, D.G. 2002. Family Heterenchelyidae. In: Carpenter, K.E. (Ed.). The living marine resources of the Western Central Atlantic . v.2. FAO Species Identification Guide for Fishery Purposes and American Society of Ichthyologists and Herpetologists Special Publication, Rome, p.601-1374.
- Smith, D.G.; Irmak, E.; Ozen, Ö. 2012. A redescription of the eel Panturichthys fowleri (Anguilliformes: Heterenchelyidae), with a synopsis of the Heterenchelyidae. Copeia, 2012: 484-493.
- Smith, D.G. 2015. Pythonichthys sanguineus The IUCN Red List of Threatened Species. e.T195661A2398552. ( (https://www.iucnredlist.org/species/195661/2398552 ). Accessed on 11 Dec 2017.
» https://www.iucnredlist.org/species/195661/2398552 - Stobutzki, I.; Miller, M.; Brewer, D. 2001. Sustainability of fishery bycatch: a process for assessing highly diverse and numerous bycatch. Environmental Conservation, 28: 167-181.
- Uyeno, T.; Sasaki, T. 1983. Scapanorhynchidae In: Uyeno, T.; Matsuura, K.; Fujii, E. (Ed.). Fishes trawled off Suriname and French Guiana Japan Marine Fishery Resource Research Center, Tokyo, p.48.
- Ward, R.D.; Zemlak, T.S.; Innes, B.H.; Last, P.R.; Hebert, P.D. 2005. DNA barcoding Australia’s fish species. Philosophical Transactions of the Royal Society B, 360: 1847-1857.
-
CITE AS:
Rotundo, M.M.; Machado, L.; Oliveira, C.; Santos, W.C.R. dos; Marceniuk, A.P. 2019. Record of the rare Caribbean mud eel, Pythonichthys sanguineus (Heterenchelyidae, Anguilliformes), in the region of the Amazon Reef. Acta Amazonica 49: 131-138.
SUPPLEMENTARY MATERIAL (only available in the electronic version)
ROTUNDO et al.Record of the rare Caribbean mud eel, Pythonichthys sanguineus (Heterenchelyidae, Anguilliformes), in the region of the Amazon Reef
Edited by
ASSOCIATE EDITOR:
Publication Dates
-
Publication in this collection
06 May 2019 -
Date of issue
Apr-Jun 2019
History
-
Received
31 July 2017 -
Accepted
23 Dec 2017