Abstract
OBJECTIVE: To compare the regression of left ventricular hypertrophy in patients with moderate hypertension treated with enalapril, losartan or a combination of the two drugs at lower doses. METHODS: Patients of both sexes with moderate hypertension confirmed by ambulatory monitoring of arte-rial blood pressure and with left ventricular hypertrophy on echocardiogram were assigned to three groups: enalapril (35 mg/day, n=15), losartan (175 mg/day, n=15) and enalapril+losartan (15 mg+100 mg/day, n=16). The patients received the drugs for 10 months. RESULTS: The three therapeutic regimens were equally effective in reducing blood pressure and left ventricular mass index (LVMI, g/m²): 141±3.9 to 123±3.6 in the enalapril group (p<0.05), from 147±3.8 to 133±2.8 in the losartan group (p<0.05), and from 146±3.0 to 116±4.0 in the enalapril+losartan group (p<0.05). However, the percent reduction of LVMI was significantly greater (p<0.01) in the enalapril+losartan group (20.5±5.0%) than in enalapril (12.4±3.2%) and the losartan (9.1±2.1%) groups. Normalization of LVMI was obtained in 10 out of the 16 patients who received enalapril+ losartan, in 6 out of the 15 patients who received only enalapril and in 4 out of the 15 patients treated with losartan. CONCLUSION: The combination of an angiotensin-converting enzyme inhibitor and an angiotensin II receptor antagonist (AT1 receptor antagonist) in patients produced an additional effect on the reduction of left ventricular hypertrophy. This finding may depend on a more complete inhibition of the cardiac renin-angiotensin.
hypertension; cardiac hypertrophy; angiotensin-converting enzyme; losartan; renin-angiotensin system
Original Article
Reduction in Left Ventricular Hypertrophy in Hypertensive Patients Treated with Enalapril, Losartan or the Combination of Enalapril and Losartan
Antônio Carlos Avanza Jr, Lilian Mameri El Aouar, José Geraldo Mill
Vitória, ES - Brazil
OBJECTIVE - To compare the regression of left ventricular hypertrophy in patients with moderate hypertension treated with enalapril, losartan or a combination of the two drugs at lower doses.
METHODS - Patients of both sexes with moderate hypertension confirmed by ambulatory monitoring of arte-rial blood pressure and with left ventricular hypertrophy on echocardiogram were assigned to three groups: enalapril (35 mg/day, n=15), losartan (175 mg/day, n=15) and enalapril+losartan (15 mg+100 mg/day, n=16). The patients received the drugs for 10 months.
RESULTS - The three therapeutic regimens were equally effective in reducing blood pressure and left ventricular mass index (LVMI, g/m2): 141±3.9 to 123±3.6 in the enalapril group (p<0.05), from 147±3.8 to 133±2.8 in the losartan group (p<0.05), and from 146±3.0 to 116±4.0 in the enalapril+losartan group (p<0.05). However, the percent reduction of LVMI was significantly greater (p<0.01) in the enalapril+losartan group (20.5±5.0%) than in enalapril (12.4±3.2%) and the losartan (9.1±2.1%) groups. Normalization of LVMI was obtained in 10 out of the 16 patients who received enalapril+ losartan, in 6 out of the 15 patients who received only enalapril and in 4 out of the 15 patients treated with losartan.
CONCLUSION - The combination of an angiotensin-converting enzyme inhibitor and an angiotensin II receptor antagonist (AT1 receptor antagonist) in patients produced an additional effect on the reduction of left ventricular hypertrophy. This finding may depend on a more complete inhibition of the cardiac renin-angiotensin.
Key words: hypertension, cardiac hypertrophy, angiotensin-converting enzyme, losartan, renin-angiotensin system
Left ventricular hypertrophy is a common complicating factor in patients with hypertension, representing an independent risk factor to morbidity and mortality from this disorder. Data from Framingham clearly demonstrate that left ventricular hypertrophy is the most important independent risk factor for acute myocardial infarction and heart failure in hypertensive patients 1. This fact explains why the reduction of left ventricular hypertrophy has become, more and more, one of the therapeutic aims to be achieved in treatment of hypertension 2.
The increase in afterload constitutes the main factor for development of left ventricular hypertrophy in hypertension. However, little is known about molecular mechanisms of transduction between the mechanical signal (increase in wall stress) and growth of cardiac mass. A number of studies have shown that several factors, in addition to pressure stress, may independently influence the growth of cardiac mass 3. Experimental and clinical studies have clearly demonstrated participation of the renin-angiotensin system in the development of cardiac hypertrophy in several pathophysiological states, including hypertension 3-6. Inhibition of the renin-angiotensin system has been shown to be an effective way of reducing increased cardiac mass in primary hypertension, states of catecholamine excess and hyperthyroidism 3,6,7. Angiotensin II, through the AT1 receptor, represents an important trophic factor not only for cardiac myocytes but also for the components of extracellular matrix in the heart 8,9, increasing cardiac mass. Cardiac hypertrophy induced by growth effects of angiotensin II is not exclusively dependent on hemodynamic factors, because stimulation of AT1 receptors of cardiac myocytes in culture promotes a significant increase in protein synthesis and induces expression of the proto-oncogenes c-fos, c-junc and c-myc, associated with both myocyte growth and stimulation to deposition of cardiac extracellular matrix elements 10,11.
The renin-angiotensin system can be inhibited by two classes of drugs: angiotensin-converting enzyme inhibitors and AT1 receptor antagonists. There are a great number of AT1 receptors in the heart. These two classes of drugs have similar antihypertensive effects 12, but they differ in their sites of action. Inhibiting angiotensin-converting enzymes not only decreases angiotensin II generation but also reduces the inactivation rate of bradykinin and substance P. Some studies suggest that the rise in the mean life of circulating bradykinin may contribute to both the antihypertensive and antihypertrophic effects of angiotensin-converting enzyme inhibitors 13,14. At the same time, these drugs promote an important increase in synthesis and secretion of renin and a high elevation in plasma angiotensin I levels, due to the lack of negative feedback of angiotensin II on the juxtaglomerular apparatus of the kidneys 15. It has been shown that even maximum doses of angiotensin-converting enzyme inhibitors do not completely suppress angiotensin II generation 16,17, since it is known that there are several alternative pathways to generate this peptide from angiotensin I 18. This fact might explain the loss of antihypertensive and antihypertrophic effects during long-term use of angiotensin-converting enzyme inhibitors in hypertensive patients 17. Due to these observations, some authors have suggested combining angiotensin-converting enzyme inhibitors and AT1 receptor antagonists in order to achieve a more complete inhibition of the renin-angiotensin system, preserving, at the same time, the possible beneficial effect caused by the increase in mean life of kinins generated in the heart and blood vessels 12,19. Considering the importance of the renin-angiotensin system in the development of cardiac hypertrophy, a higher inhibition of this system might be a more efficient way of reducing hypertrophied myocardium in hypertensive patients. Therefore, the purpose of this study was to compare the reduction in left ventricular hypertrophy caused by monotherapy with enalapril or losartan or the combination of these two drugs, in patients with moderate hypertension. Drug doses were adjusted to produce a similar reduction in blood pressure in the three groups of patients.
Methods
The study comprised hypertensive patients who were screened at the Clínica Vilacor, Vila Velha, Espírito Santo, or at the Clínica de Investigação Cardiovascular, Centro Biomédico, Universidade Federal do Espírito Santo. Patients of both sexes, aged between 40 and 60 years, with moderate hypertension diagnosed by casual blood pressure readings, were referred to one of the cited clinics, with or without previous treatment, and interviewed by one of the authors. Patient consent was obtained from all participants. After a washout period, when any medication that could alter blood pressure levels was discontinued for 12 days, those patients included in the study were screened again, when blood pressure was measured and blood was withdrawn for biochemical analyses (glucose, blood urea nitrogen, creatinine, sodium and potassium). Blood pressure was measured by a mercury sphygmomanometer after a 10-minute period of rest in the sitting position. The first and the fifth Korotkoff sounds were considered indicative of systolic and diastolic blood pressures, respectively. Patients who met criteria for moderate hypertension, according to the V Joint National Committee 20, remained in the study. Patients who had any event in the washout period, including blood pressure levels beyond the limits of moderate hypertension, were excluded from the study and immediately medicated. Ambulatory monitoring of blood pressure and echocardiogram were performed in those patients who remained in the study.
Ambulatory monitoring of blood pressure was performed in a SpaceLabs 90207 device and readings were recorded every 20 minutes, during a 24- to 26-hour period. The equipment was always installed early in the afternoon (around 2:00 p.m.) for better detection of morning elevation of blood pressure. The same investigator, who knew no diagnoses or possible therapies, conducted all echocardiographic examinations. M-mode and pulsed Doppler recordings were performed with GERT 6800 equipment. Anatomical and functional parameters were measured to evaluate left ventricular mass and cardiac performance, respectively. Left ventricular mass was calculated with the Devereux-Reichek algorithm 21, which has the greatest sensitivity and accuracy when left ventricular mass obtained by echocardiographic methods is compared with that seen on necropsy of hypertensive patients 22. Left ventricular mass index was calculated by the ratio of left ventricular mass (g), obtained by echocardiogram, to body surface [(weight (kg) + height (cm) - 60)/100].
After these examinations, only those subjects who met criteria for moderate hypertension based on ambulatory monitoring of arterial blood pressure, according to the Brazilian Consensus for the Use of Ambulatory Blood Pressure Monitoring 23, and those with left ventricular mass index greater than 110 g/m2 (for women) or 130 g/m2 (for men), remained in the study. Among 90 patients who underwent ambulatory blood pressure monitoring, 29 (32%) did not meet criteria to classify them as patients with moderate hypertension 23. These subjects were excluded from the study.
Sixty-one patients met the two main inclusion criteria and were assigned to three groups: the enalapril group, treated with enalapril maleate (35mg/day), 20mg at 8:00 a.m. and 15mg at 8:00 p.m.; the enalapril+losartan group, treated with enalapril maleate 15 mg/day at 8:00 a.m. and losartan potassium 100mg/day at 8:00 p.m.; and the losartan group, treated with losartan potassium (175mg/day), 100mg at 8:00 a.m. and 75mg at 8:00 p.m.
The first two groups, which included therapy with enalapril, were previously established. The patients were sequentially allocated to either the enalapril or the enalapril+losartan group, which comprised 22 and 23 subjects, respectively. The 17 patients in the losartan group were recruited after the other two groups had been initiated.
Fifty milligrams of losartan potassium was considered equivalent to 10 mg of enalapril maleate, because it was shown that a similar fall in blood pressure occurs when this equivalence ratio is used in healthy subjects 19. Both enalapril and losartan were administered through commercially available formulations. This therapeutic regimen was prolonged for 10 months, and patients were clinically reevaluated 1, 3, 5, 7 and 10 months after commencing medication. These reevaluations allowed the authors to check adherence to treatment, verify the antihypertensive effect of medication and detect adverse reactions. Only two patients did not show a satisfactory reduction in blood pressure after one month of medication (reduction in systolic blood pressure equal to or less than 5mmHg). These patients needed to receive other drugs and were excluded from the study. Blood biochemical analyses and echocardiogram, to obtain the degree of left ventricular hypertrophy regression, were performed again at the last evaluation (month 10).
Exclusion criteria were the following: black subjects, obesity (body mass index > 30kg/m2), diabetes, valvular heart disease, secondary hypertension, complications of hypertension (myocardial infarction or heart failure) and long-term use of drugs that could interfere with the effect of enalapril or losartan, such as corticosteroids, neuroleptics and antidepressants.
Data are shown as mean ± standard deviation. Comparisons among means observed in the three groups were done by one-way analysis of variance (ANOVA), followed by the post hoc Tukey test. Comparisons between two means for the same variable, before and after treatment, in the same group, were done by the Student's t test for paired samples. Comparisons among several means in the same group were done by one-way ANOVA, followed by the post hoc Bonferroni test, for comparison of a variable before and during the therapy, or by the Tukey test, for comparison among different groups. All differences of the mean values with p<0.05 were considered significant.
Results
Of the 61 patients who were initially enrolled, 15 (24%) were excluded from the study during the treatment period. Seven of them had been included in the enalapril group (4 were excluded because of persistent cough, 2 due to noncompliance with the treatment and one due to acute myocardial infarction at month 4). Two excluded patients had been allocated to the losartan group (both stopped taking the study medication). Finally, 6 out of the 15 excluded patients had been assigned to the enalapril+losartan group (2 were excluded due to unsatisfactory reduction in blood pressure in the first month, leading to the addition of other drugs to the therapeutic regimen, 2 stopped taking the study medication and 2 altered the medication schedule prescribed for their own account). Therefore, the following data represent the results obtained from the 46 patients who remained in the study until the end of the previously established period (10 months). No patient had been previously medicated with AT1 receptor antagonist, while 9 had already been treated with angiotensin-converting enzyme inhibitors for different periods of time, for treatment of hypertension. Most of them, however, used this medication without regularity.
The features of the patients who concluded the study protocol, in the three groups, are shown in Table I. All the patients were white or mulatto. Baseline levels of systolic and diastolic blood pressure in the sitting position, mean age, weight and body mass index were all similar in the three groups.
Figure 1 shows systolic and diastolic blood pressure levels in the three groups throughout the 10-month treatment period. Reduction in blood pressure levels was significant and similar in the three groups until month 7, when systolic blood pressure was 146±1.9mmHg, 146±2.1 mmHg and 143±1.9mmHg in the enalapril group, losartan group and enalapril+losartan group, respectively (p>0.05). At the end of the treatment period, blood pressure levels in the losartan group were slightly higher than those in the other two groups. At the end of month 10, almost all the patients exhibited blood pressure levels in the normal range, i.e., systolic blood pressure lower than 140mmHg and diastolic blood pressure lower than 90mmHg. Five patients in the losartan group showed systolic blood pressure between 140 and 150mmHg at the end of the treatment period. One of them had diastolic blood pressure of 95mmHg at this time. Mean systolic and diastolic blood pressure levels during the 10 months of treatment, calculated from the levels obtained at each consultation (area under the curve), were similar in the three groups. Mean systolic blood pressure during the 10 months was 145±8mmHg in the enalapril group, 148±7mmHg in the losartan group and 144±9mmHg in the enalapril+losartan group (p>0.05). Mean diastolic blood pressure was not significantly different among the three groups, either. Therefore, considering a longer period of time, all three therapeutic regimens can be considered equally effective in reducing blood pressure, according to these data.
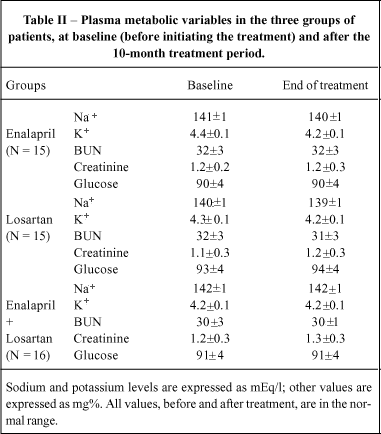
Plasma glucose, blood urea nitrogen, creatinine, sodium and potassium concentrations at baseline and month 10 were in the normal range in all patients. These data are shown in Table II.
Mean left ventricular mass index values for each group, before and after treatment, are found in Table III. Figure 2 shows these values for each patient, at the two conditions. At baseline, there was no statistically significant difference among left ventricular mass index values in the three groups. All the three regimens significantly reduce this index. However, left ventricular mass index at the end of the study was significantly lower (p=0.011) in the group treated with the enalapril-losartan combination (116±4.0g/m2) than in the enalapril group (123±2.8g/m2) or in the losartan group (133±2.8g/m2). These data suggest that the combination of enalapril and losartan may promote a greater reduction in left ventricular hypertrophy than those seen when the two drugs are used in isolation. This finding can also be seen in Figure 3, which shows the percent reduction in left ventricular mass index in the three groups. The reduction of 20.5±5.0% in the left ventricular mass index observed in the enalapril-losartan group was significantly greater (p<0.01) than the reduction obtained in the enalapril group (12.4±3.2%) and the losartan group (9.1±2.1%). From a statistical point of view, the reduction in left ventricular mass index was lower (p<0.05) in the losartan group than in the enalapril group.
An individual analysis of these data shows that reduction in left ventricular hypertrophy was achieved in all patients (Fig. 2). Nevertheless, a better response was seen in the enalapril-losartan group. Normalization of left ventricular mass index was noted in 10 out of the 16 patients treated with both drugs, in 6 out of the 15 patients treated with enalapril alone and in only 4 out of 15 patients medicated only with losartan. We did not note a significant correlation between reduction in blood pressure and decrease in left ventricular mass index in any of the three groups.
Discussion
Several studies confirm that angiotensin-converting enzyme inhibitors and AT1 receptor antagonists, used in adequate doses, are equally effective in reducing blood pressure in patients with primary hypertension 12,24,25. Data shown in the present study are in agreement with these findings, because enalapril (35mg/day) and losartan (175mg/day) promoted similar reduction of both systolic and diastolic pressures in patients with moderate hypertension. These drugs, in the same proportion of doses (1:5), caused similar decrease in blood pressure in normotensive subjects 19,26. To create the enalapril+losartan group from the enalapril group, 20 mg of enalapril was replaced by 100mg of losartan, because this last drug can cause an additional antihypertensive effect, even when maximum doses of angiotensin-converting enzyme inhibitors are used 27. The results confirmed that the three medication schedules were equally effective in reducing both systolic and diastolic blood pressures, normalizing blood pressure in almost all the patients. A small differentiation occurred in the losartan group, i.e., about one third of the patients remained slightly hypertensive at the end of the 10-month treatment period. This difference, however, was mainly due to the last readings, at month 10. Previous measurements of both mean systolic and mean diastolic pressures were not statistically different among the groups, even though they were always slightly higher in the group treated with losartan. We do not have a reasonable explanation for this finding at the last clinical examination, but the occurrence of an blood pressure run off due to long-term use of the drug might be considered. Analysis of whole data, however, allows the conclusion that all the three therapeutic regimens caused a very similar reduction in blood pressure in moderate hypertension. Otherwise, different effects on the reduction of left ventricular hypertrophy were observed among the groups. Therefore, the absence of a positive correlation between the reduction in systolic and diastolic blood pressures and the reduction in left ventricular mass index may suggest that the antihypertrophic effect of these drugs may be primarily dependent on their direct actions on the myocardium, and not secondary to afterload reduction 3,4,7. Combining angiotensin-converting enzyme inhibitors and AT1 receptor antagonists produces a more potent direct effect on the myocardium, by reasons still unknown. However, this conclusion is impaired by the fact that the drugs were used at very near maximum doses and, in most patients, blood pressure was already at its lower plateau after month 6. As all the patients had similar blood pressure values at baseline, the interval of variation of the independent variable (fall in blood pressure during the treatment) was very short, making difficult the finding of a possible correlation with reduction in left ventricular mass index. In spite of these methodological difficulties, mean values in the three groups clearly show a higher reduction in left ventricular hypertrophy when the two drugs are combined.
From a physiological point of view, the general mechanism of action of both angiotensin-converting inhibitor and AT1 receptor antagonist is the same, i.e., inhibition of the renin-angiotensin system. However, their sites of action are different, and therefore differences exist in their intrinsic mechanisms of action. Inhibition of angiotensin-converting enzyme reduces angiotensin II generation and, at the same time, increases mean life of bradykinin and other peptides metabolized by angiotensin-converting enzyme or kininase II 14. As kinins have a very short mean life, global effects of angiotensin-converting enzyme inhibitor and AT1 receptor antagonist (blood pressure levels, for instance) tend to be similar. Otherwise, their effects on specific organs (hypertrophy and hyperplasia) could be distinct one from another, because local effects of both angiotensin II and bradykinin might be more dependent on their local generation, particularly of bradykinin. Another factor to be considered is the conversion of angiotensin I into angiotensin II, catalyzed not only by angiotensin-converting enzyme but also by other peptidases, such as chymase, tonin and cathepsin G 18. This explains why relatively high plasma angiotensin II levels are found in patients with hypertension or congestive heart failure treated with maximum doses of angiotensin-converting enzyme inhibitor 27. One fact that produces obstacles to our knowledge about the endocrine and paracrine effects of the renin-angiotensin system is that it has not been precisely established what amount of angiotensin I is converted into angiotensin II, in each organ, through angiotensin-converting enzyme or through the so-called alternative pathways, from which the chymase pathway has deserved major concern, particularly in patients with congestive heart failure 18.
These data have led some authors to suggest that combining angiotensin-converting enzyme inhibitors and AT1 receptor antagonists might represent a therapeutic advantage when a more complete inhibition of renin-angiotensin-aldosterone is desirable 26. The need to promote faster drops in blood pressure or greater reduction in left ventricular hypertrophy is a possibility of concern. 26. Our results only partially support this consideration; the three therapeutic regimens caused similar decrease in blood pressure but different effects on the reduction of left ventricular hypertrophy. As this was a basic point of the study, we enrolled only patients who had already had a clear development of left ventricular hypertrophy on echocardiogram, i.e., left ventricular mass index greater than 130g/m2 for men and greater than 110g/m2 for women. Echocardiographic examinations were always performed by the same investigator, who knew neither the diagnosis nor the drugs the patient was receiving. The duration of hypertension of each patient was not systematically assessed because of difficulties in establishing the exact beginning of the disease. Most patients, however, reported knowing the diagnosis of hypertension for more than 2 years.
Our results showed that the combination of enalapril and losartan was clearly more efficient in reducing left ventricular hypertrophy than enalapril or losartan alone, which is in agreement with a recent finding in rats with spontaneous hypertension (SHR strain). However, doses of enalapril and losartan were simply added in this study 28 and the greater reduction in left ventricular hypertrophy noted when the two drugs were combined was attributed to a greater decrease in blood pressure, compared with that achieved with monotherapies. Our results did not confirm this explanation. When we combined losartan and enalapril, we reduced their doses in order to obtain a uniform antihypertensive effect, allowing us to compare the reduction in left ventricular hypertrophy at similar conditions of afterload reduction. This aim was fully achieved in groups I (enalapril) and II (enalapril and losartan). In these groups, while the reduction in blood pressure was almost identical, the reduction in left ventricular mass index was different, suggesting that the presence of AT1 receptor antagonist may be important if a greater reduction in left ventricular hypertrophy is considered an additional objective of treatment in hypertension. Explanations for these different effects on left ventricular hypertrophy must be searched for. Differences in regulation of blood pressure during dose intervals and in organ-specific effects of these drugs (on kidneys, heart and blood vessels) must be taken into account.
The combination of enalapril (10mg) and losartan (50mg) in healthy volunteers caused similar reductions in blood pressure compared with enalapril 20mg. However, the area under the arterial blood pressure curve, which better evaluates the duration of the antihypertensive effect, was greater when the two drugs were combined 26. This finding may be due to the fact that losartan, despite its relatively short half-life, generates a metabolite with much longer half-life and high capacity for blocking AT1 receptors 29. A similar effect might have occurred in the present study, i.e., the combination of the two drugs might have promoted a more stable reduction in blood pressure levels during the 24 hours, determining a greater reduction in left ventricular hypertrophy, which is more related to the mean of blood pressure throughout the day than to casual measurements of blood pressure 30. However, it seems that this possibility did not occur in our study, because ambulatory blood pressure monitoring at the end of the treatment period showed a similar pattern of blood pressure in the three groups.
Another hypothesis to explain our results relates to specific effects of the drugs on the heart. Rise in gene expression of angiotensin-converting enzyme and angiotensinogen has been observed in both the myocardium and vascular wall of hypertensive laboratory animals 31. Under these conditions, both angiotensin II generation and bradykinin inactivation would be enhanced in the heart. Experimental studies show that the antihypertrophic effect of low doses of angiotensin-converting enzyme inhibitor on myocardium and vascular smooth muscle in rats can be partially reverted by concomitant use of bradykinin receptor antagonist 14. Another fact to be considered is that angiotensin-converting enzyme inhibitor increases renin synthesis and circulating angiotensin II levels. Under these conditions, angiotensin II generation through alternative pathways probably increases due to a rise in its substrate angiotensinogen, allowing an run off the antihypertensive and possibly the antihypertrophic effects of the angiotensin-converting enzyme inhibitor 17. The combination of angiotensin-converting enzyme inhibitor and AT1 receptor antagonist could prevent effects of angiotensin receptor stimulation and, at the same time, preserve the antihypertrophic effect of angiotensin-converting enzyme inhibitor related to an increase in bradykinin levels in the heart.
In conclusion, our results suggest that the combination of angiotensin-converting enzyme inhibitor and AT1 receptor antagonist do not provoke antihypertensive effects greater than those obtained when the two drugs are used in isolation at adequate doses, in patients with moderate hypertension. However, this combination can significantly enhance reduction in left ventricular hypertrophy, providing greater protection to the heart against the overload caused by persistent hypertension. Moreover, the combination of these drugs did not increase the incidence of adverse effects. On the contrary, the possibility of using a lower dose of angiotensin-converting enzyme inhibitor, combined with an AT1 receptor antagonist, might in fact reduce the chance of persistent cough, the main factor limiting the use of angiotensin-converting enzyme inhibitor in hypertension. Therefore, combining angiotensin-converting enzyme inhibitors and AT1 receptor antagonists may be recommended as an alternative therapy when reduction in the angiotensin-converting enzyme inhibitor dosage is needed, preserving its antihypertensive effect, or when reduction in left ventricular hypertrophy constitutes a major aim of antihypertensive treatment.
Acknowledgment
The authors thank Merck Sharp & Dhome for supplying Renitec and Cozaar
and Cozaar , used in this study.
, used in this study.
Universidade Federal do Espírito Santo - Vitória. Supported by Projeto Capes/Nordeste and Funcitec/ES
Mailing address: José Geraldo Mill - Centro Biomédico da UFES - Av. Marechal Campos, 1468 - 29040-090 - Vitória, ES - Brazil
- 1. Levy D, Garrinson RJ, Savage DD, et al. Prognostic implications of echocardiographically determined left ventricular mass in the Framingham Heart Study. N Engl J Med 1990; 322: 1561-6.
- 2. Messerli FH, Aristizabel D, Soria F. Reduction of left ventricular hypertrophy: How beneficial? Am Heart J 1993; 125: 1520-4.
- 3. Morgan HE, Baker KM. Cardiac hypertrophy: mechanical, neural and endocrine dependence. Circulation 1991; 83: 13-25.
- 4. Baker KM, Cherin MI, Wixon SK, Aceto JF. Renin angiotensin system involvement in pressure-overload cardiac hypertrophy in rats. Am J Physiol 1990; 259: H324-H32.
- 5. Schunkert H, Dzau V, Tang SS, Hirsh AT, et al. Increased rat cardiac angiotensin converting enzyme activity and mRNA expression in pressure overload left ventricular hypertrophy: effects on coronary resistance, contractility, and relaxation. J Clin Invest 1990; 86: 1913-20.
- 6. Nagano M, Higaki J, Nakamura F, et al. Role of cardiac angiotensin II in isoproterenol-induced left ventricular hypertrophy. Hypertension 1992; 19: 708-12.
- 7. Lindpaintner K, Wilhelm MJ, Jin M, et al. Tissue renin-angiotensin systems: focus o the heart. J Hypertension 1987; 5(suppl 2): 33-8.
- 8. Pfeffer MA. Left ventricular remodeling after acute myocardial infarction. Annu Rev Med 1995; 46: 455-66.
- 9. Crabos M, Roth M, Hahn AWA, Erne P. Characterization of angiotensin II receptors in cultured adult rat cardiac fibroblasts - coupling to signaling systems and gene expression. J Clin Invest 1994; 93: 2372-8.
- 10. Aceto JF, Baker KM. (Sar)Angiotensin II receptor-mediated stimulation of protein synthesis in chick heart cells. Am J Physiol 1990; 258: H806-H813.
- 11. Sadoshima I, Izumo S. Signal transduction pathways of angiotensin II induced c-fos gene expression in cardiac myocytes in vitro: roles of phospholipid deried second messengers. Circ Res 1993; 73: 424-38.
- 12. Tikkanen I, Omvik P, Jensen HĆ, et al. Comparison of the angiotensin II antagonist losartan with the angiotensin converting enzyme inhibitor enalapril in patients with essential hypertension. J Hypertension 1995; 13: 1343-51.
- 13. Bao G, Gohlke P, Qadri F, et al. Chronic kinin receptor blockade attenuates the antihypertensive effect of ramipril. Hypertension 1992; 20: 74-9.
- 14. Linz W, Wiemer G, Gohlke P, Unger T, Schoelkens BA. The contribution of bradykinin to the cardiovascular actions of ACE inhibitors. In: Lindpaintner K, Ganten D (Eds) - The Cardiac Renin-Angiotensin System. New York: Futura Publishing Co, 1994; 253-87.
- 15. Mooser V, Nussberger J, Juillerat L, et al. Reactive hyperrreninemia is a major determinant of plasma angiotensin II during ACE inhibition. J Cardiovascular Pharmacol 1990; 15: 276-82.
- 16. Juillerat L, Nussberger J, Ménard J, et al. Determinants of angiotensin II generation during converting enzyme inhibition. Hypertension 1990; 16: 564-72.
- 17. Van Den Meiracker AH, Man in't Veld AJ, Admiraal PJJ, et al. Partial escape of angiotensin converting enzyme inhibition during prolongued ACE inhibitor treatment: does it exist and does it affect the antihypertensive response? J Hypertension 1992; 10: 803-12.
- 18. Urata H, Healy S, Stewart RW, et al. Angiotenin-forming pathways in normal and failing human hearts. Circulation Res 1990; 66: 883-90.
- 19. Azizi M, Guyene TT, Chatellier G, Ménard J. Pharmacological demonstration of the additive effects of angiotensin-converting enzyme inhibition and angiotensin II antagonism in sodium depleted healthy subjects. Clin Exp Hypertension 1997; 19: 937-51.
- 20. Joint National Committee on Detection and Treatment of High Blood Pressure. The Fifth Report. Arch Int Med 1993; 153: 154-83.
- 21. Devereux RB, Lutas EM, Casale PN. Standardization of M-mode echocardiographic left-ventricular measurements. J Am Coll Cardiol 1984; 4: 1222-30.
- 22. Liebson PR, Savage DD. Echocardiography in hypertension: A review. Echocardiography 1986; 3: 181-213.
- 23. Sociedade Brasileira de Cardiologia - Consenso brasileiro para uso de monitorizaçăo ambulatorial da pressăo arterial. Arq Bras Cardiol 1993; 60: 129-34.
- 24. Goldberg AI, Dunlay MC, Sweet CS. Safety and tolerability of losartan potassium, an angiotensin II receptor antagonist, compared with hydrochlorothiazide, atenolol, felodipine ER, and angiotensin converting enzyme inhibitors for the treatment of systemic hypertension. Am J Cardiol 1995; 75: 793-5.
- 25. Ruff D, Gazdick LP, Berman R, Goldberg AI, Sweet CS. Comparative effects of combination drug therapy regimens commencing with either losartan potassium, an angiotensin II receptor antagonist, or enalapril maleate for the treatment of severe hypertension. J Hypertension 1996; 14: 263-70.
- 26. Azizi M, Guyene TT, Chatellier G, Wargon M, Ménard J. Additive effects of losartan and enalapril on blood pressure and plasma active renin. Hypertension 1997; 29: 634-40.
- 27. Hamroff G, Blaufarb I, Mancini D, et al. Angiotensin II-receptor blockade further reduces afterload safely in patients maximally treated with angiotensin-converting enzyme inhibitors for heart failure. J Cardiovasc Pharmacol 1997; 30: 533-6.
- 28. Richer C, Bruneval P, Ménard J, Giudicelli JF. Additive effects of enalapril and losartan in (mREN-2)27 trangenic rats. Hypertension 1998; 31: 692-8.
- 29. Wong PC, Price Jr WA, Chiu AT, et al. Nonpeptide angiotensin II receptor antagonists: Pharmacology of EXP3174: An active metabolite of Dup 753, an orally active antihypertensive agent. J Pharmacol Exp Ther 1990; 255: 211-17.
- 30. Devereux RB, Pickering TG. Relationship between the level, pattern and variability of ambulatory blood pressure and target organ damage in hypertension. J Hypertension 1991; 9(suppl 8); S34-S8.
- 31. Schunkert H, Dzau VJ, Tang SS, et al. Increased rat cardiac angiotensin converting enzyme activity and mRNA expression in pressure overload left ventricular hypertrophy: effects on coronary resistance, contractility, and relaxation. J Clin Invest 1990; 86: 1913-20.
Publication Dates
-
Publication in this collection
05 Sept 2000 -
Date of issue
Feb 2000