Abstracts
BACKGROUND: Little is known about cardiorespiratory and metabolic response in healthy children during progressive maximal exercise test. OBJECTIVE: To test the hypothesis that children show different responses in cardiorespiratory and metabolic parameters during progressive maximal exercise test when compared with adults. METHODS: Twenty-five healthy children (gender, 15M/10F; age, 10.2 ± 0.2) and 20 healthy adults (gender, 11M/9F; age, 27.5 ± 0.4) underwent a progressive treadmill cardiopulmonary test until exhaustion to determine the maximal aerobic capacity and ventilatory anaerobic threshold (VAT). RESULTS: The peak workload (5.9±0.1 vs 5.6±0.1 mph, respectively; p>0.05), exercise time (9.8±0.4 vs 10.2±0.4 min, respectively; p>0.05), and relative aerobic fitness (VO2peak, 39.4±2.1 vs 39.1±2.0 ml.kg-1.min-1, respectively; p>0.05) were similar in children and adults. At ventilatory anaerobic threshold, the heart rate, VO2 ml.kg-1.min-1, respiratory rate (RR), functional estimate of dead space (VD/VT), ventilatory equivalent for oxygen (VE/VO2) and end-tidal pressure for oxygen (PETO2) were higher in children, while tidal volume (VT), O2 pulse and end-tidal pressure for carbon dioxide (PETCO2) were lower. At peak of exercise, children showed higher RR and VD/VT. However, O2 pulse, VT, pulmonary ventilation, PETCO2 and respiratory exchange ratio were lower in children than adults. CONCLUSION: Cardiorespiratory and metabolic responses during progressive exercise test are different in children as compared to adults. Specifically, these differences suggest that children have lower cardiovascular and ventilatory efficiency. However, children showed higher metabolic efficiency during exercise. In summary, despite the differences observed, children showed similar levels of exercising capacity when compared with adults.
Child; exercise test; ventilation; metabolism; comparative study
FUNDAMENTO: Pouco se sabe sobre a resposta cardiorrespiratória e metabólica em crianças saudáveis durante teste de esforço progressivo máximo. OBJETIVO: Testar a hipótese de que as crianças apresentam respostas diferentes nos parâmetros cardiorrespiratórios e metabólicos durante teste de esforço progressivo máximo em comparação aos adultos. MÉTODOS: Vinte e cinco crianças saudáveis (sexo, 15M/10F; idade, 10,2 ± 0,2) e 20 adultos saudáveis (sexo, 11M/9F; idade, 27,5 ± 0,4) foram submetidos a um teste cardiopulmonar progressivo em esteira ergométrica até a exaustão para determinar a capacidade aeróbia máxima e limiar anaeróbio ventilatório (LAV). RESULTADOS: A carga de pico (5,9 ± 0,1 vs 5,6 ± 0,1 mph, respectivamente; p > 0,05), tempo de exercício (9,8 ± 0,4 vs 10,2 ± 0,4 min, respectivamente, p > 0,05), e aptidão cardiorrespiratória (VO2pico, 39,4 ± 2,1 vs 39,1 ± 2,0 ml.kg-1.min-1, respectivamente, p > 0,05) foram semelhantes em crianças e adultos. No limiar anaeróbio ventilatório, a frequência cardíaca, VO2 ml.kg-1.min-1, a frequência respiratória (FR), o espaço morto funcional estimado (VD/VT), o equivalente ventilatório de oxigênio (VE/VO2) e a pressão expiratória final do oxigênio (PETO2) foram maiores nas crianças, enquanto o volume corrente (VC), pulso de O2 e a pressão expiratória final do dióxido de carbono (PETCO2) foram menores. No pico do exercício, as crianças apresentaram FR e VD/VT superiores. No entanto, o pulso de O2, o VC, a ventilação pulmonar, o PETCO2 e a razão de troca respiratória foram menores nas crianças do que em adultos. CONCLUSÃO: Respostas cardiorrespiratórias e metabólicas durante o teste de esforço progressivo são diferentes em crianças em comparação aos adultos. Especificamente, essas diferenças sugerem que as crianças têm menor eficiência cardiovascular e respiratória. No entanto, as crianças apresentaram maior eficiência metabólica durante o teste de esforço. Em resumo, apesar das diferenças observadas, as crianças mostraram níveis semelhantes de capacidade de esforço, quando comparadas aos adultos.
Criança; teste de esforço; ventilação; metabolismo; estudo comparativo
Fundamento: Poco se sabe acerca de la respuesta cardiorrespiratoria y metabólica en niños saludables durante prueba de esfuerzo progresivo máximo. OBJETIVO: Probar la hipótesis de que los niños presentan respuestas diferentes en los parámetros cardiorrespiratorios y metabólicos durante la prueba de esfuerzo progresivo máximo en comparación con los adultos. MÉTODOS: Veinticinco niños saludables (sexo, 15M/10F; edad, 10,2 ± 0,2) y 20 adultos saludables (sexo, 11M/9F; edad, 27,5 ± 0,4) fueron sometidos a una prueba cardiopulmonar progresiva en cinta ergométrica hasta el agotamiento para determinar la capacidad aeróbica máxima y el umbral anaeróbico ventilatorio (Uan). RESULTADOS: La carga de pico (5,9 ± 0,1 vs 5,6 ± 0,1 mph, respectivamente; P > 0,05), tiempo de ejercicio (9,8 ± 0,4 vs 10,2 ± 0,4 min, respectivamente, P > 0,05), y aptitud cardiorrespiratoria (VO2pico, 39,4 ± 2,1 vs 39,1 ± 2,0 ml.kg-1.min-1, respectivamente, P > 0,05) fueron similares en niños y adultos. En el umbral anaeróbico ventilatorio, la frecuencia cardíaca, VO2 ml.kg-1.min-1, la frecuencia respiratoria (FR), el espacio muerto funcional estimado (VD/VT), el equivalente ventilatorio para el oxígeno (VE/VO2) y presión espiratoria final de oxígeno (PETO2) fueron mayores en los niños, mientras que el volumen corriente (VC), pulso de O2 y la presión espiratoria final de dióxido de carbono (PETCO2) fueron menores. En el pico del ejercicio, los niños presentaron FR y VD/VT superiores. Con todo, el pulso de O2, el VC, la ventilación pulmonar, el PETCO2 y la cociente de intercambio respiratorio fueron menores en niños que en adultos. CONCLUSIÓN: Respuestas cardiorrespiratorias y metabólicas durante la prueba de esfuerzo progresivo son diferentes en niños en comparación con los adultos. Específicamente, estas diferencias sugieren que los niños tienen menor eficiencia cardiovascular y respiratoria. Con todo, los niños presentaron mayor eficiencia metabólica durante la prueba de esfuerzo. En resumen, a pesar de las diferencias observadas, los niños mostraron niveles similares de capacidad de esfuerzo, al compararlos con los adultos.
Niños; prueba de esfuerzo; ventilación; metabolismo; estudio comparativo
ORIGINAL ARTICLE
Cardiorespiratory responses during progressive maximal exercise test in healthy children
Danilo Marcelo Leite do Prado; Ana Maria F. Wanderley Braga; Maria Urbana Pinto Rondon; Luciene Ferreira Azevedo; Luciana D. N. J. Matos; Carlos Eduardo Negrão; Ivani Credidio Trombetta
Instituto do Coração da Faculdade de Medicina da Universidade de São Paulo - InCor-HCFMUSP, São Paulo, SP - Brazil
Mailing address
ABSTRACT
BACKGROUND: Little is known about cardiorespiratory and metabolic response in healthy children during progressive maximal exercise test.
OBJECTIVE: To test the hypothesis that children show different responses in cardiorespiratory and metabolic parameters during progressive maximal exercise test when compared with adults.
METHODS: Twenty-five healthy children (gender, 15M/10F; age, 10.2 ± 0.2) and 20 healthy adults (gender, 11M/9F; age, 27.5 ± 0.4) underwent a progressive treadmill cardiopulmonary test until exhaustion to determine the maximal aerobic capacity and ventilatory anaerobic threshold (VAT).
RESULTS: The peak workload (5.9±0.1 vs 5.6±0.1 mph, respectively; p>0.05), exercise time (9.8±0.4 vs 10.2±0.4 min, respectively; p>0.05), and relative aerobic fitness (VO2peak, 39.4±2.1 vs 39.1±2.0 ml.kg-1.min-1, respectively; p>0.05) were similar in children and adults. At ventilatory anaerobic threshold, the heart rate, VO2 ml.kg-1.min-1, respiratory rate (RR), functional estimate of dead space (VD/VT), ventilatory equivalent for oxygen (VE/VO2) and end-tidal pressure for oxygen (PETO2) were higher in children, while tidal volume (VT), O2 pulse and end-tidal pressure for carbon dioxide (PETCO2) were lower. At peak of exercise, children showed higher RR and VD/VT. However, O2 pulse, VT, pulmonary ventilation, PETCO2 and respiratory exchange ratio were lower in children than adults.
CONCLUSION: Cardiorespiratory and metabolic responses during progressive exercise test are different in children as compared to adults. Specifically, these differences suggest that children have lower cardiovascular and ventilatory efficiency. However, children showed higher metabolic efficiency during exercise. In summary, despite the differences observed, children showed similar levels of exercising capacity when compared with adults. (Arq Bras Cardiol 2010; 94(4):464-470)
Key words: Child; exercise test; ventilation; metabolism; comparative study.
Introduction
Children are not adults in miniature; they grow up, and as such, physiologic and metabolic responses to exercise vary accordingly as they progress through childhood and adolescence1-5. In relation to the bioenergetic profile, a large number of studies1,3,6-8 suggested that children have lower anaerobic or glycolytic capacity to produce adenosine 5-triphosphate (ATP) during exercise. According to Eriksson et al9, this immature glycolytic metabolism may be explained by lower activities of anaerobic enzymes such as lactate dehydrogenase and phosphofructokinase-1. Nevertheless, other investigations10,11 showed that children can better adapt to aerobic exercise than adults because their energy expenditure appears to rely more on oxidative metabolism. In addition, some studies9,12 reported higher levels of oxidative enzymes such as succinate dehydrogenase and isocitrate dehydrogenase in children than adults.
Furthermore, several reports have showed differences on ventilation and pulmonary gas exchange during exercise in children. For instance, previous studies4,13,14 have observed differences in the respiratory control between children and adults. These differences were due to the fact that arterial PCO2 is regulated at lower levels in children4,13,15, and has a higher central ventilatory neural drive16 resulting in higher ventilatory rates for a given metabolic demand. In addition, it has been reported17 that children have higher values of heart rate and lower stroke volume than adults at a given submaximal workload.
Therefore, it is evident that the responses of certain physiologic variables which include cardiovascular, ventilatory and metabolic parameters depend on maturity, making the evaluation more complex. The use of a computer system with ventilatory direct measurements allows bringing on an evaluation of physiologic and metabolic variables in the noninvasive way. As a result, it became an important tool that provides information of cardiorespiratory responses during incremental exercise in pediatric population.
Thus, the purpose of this study was to test the hypothesis that the cardiorespiratory and metabolic responses at different phases of the maximal progressive exercise test will be different in children comparing to adults.
Methods
Study population and design
This study used a sample of subjects who entered into cardiopulmonary evaluation database from a referral hospital, collected from 2003 to 2006, in a retrospective, cross-sectional design. Eight hundred eighty subjects meeting the following inclusion/exclusion criteria were offered participation in the study: 1) age between 8 to 12 years for children and 25 to 30 years for adults; 2) no overweight or obesity18; 3) untrained (i.e., those reporting no regular physical activity); 4) no medication; 5) no evidence of metabolic, hormonal, respiratory, and cardiovascular disease; 6) no smoking; and 7) achievement of a true maximal effort during graded exercise test. Subjects were considered to have reached a maximal effort if they achieved at least two of the following criteria:
For children19: 1) subjective evidence of exhaustion (unsteady gait, facial flushing, hyperpnea); 2) peak heart rate > 190 beats/min; and 3) maximal respiratory exchange ratio > 1.00. For adults20,21: 1) peak heart rate > 95% to the age-predicted maximal; 2) maximal respiratory exchange ratio > 1.10; and 3) achievement of a VO2 plateau. All subjects were instructed to refrain from eating 2 h before the maximal graded exercise test, and to abstain from caffeine and alcohol. In addition, the subjects were instructed to avoid strenuous physical activity during the day before the test. The maturation status was not assessed in this investigation. The study protocol was approved by the Human Subject Protection Committees of the Heart Institute, School of Medicine, University of São Paulo.
Anthropometric measurements - Body weight was measured using a mechanic scale (Filizola model 31-São Paulo, Brazil) to the nearest 0.1kg, with subjects dressed in a light T- shirt and shorts. The standing height was measured using the Filizola stadiometer (model 31, São Paulo, Brazil) to the nearest 0.1cm.
Cardiorespiratory evaluation - A maximal graded exercise test was carried out on a programmable treadmill (Quinton Q65, model 645, Quinton Instruments Co, Washington, USA). Gas exchange and ventilatory variables were measured continuously during the gas exchange tests breath by breath, using an open-circuit spirometry procedure on a based exercise system (SensorMedics - model Vmax 229 Pulmonary Function/Cardiopulmonary Exercise Testing Instrument, Yorba Linda, CA, USA). The following variables were obtained breath by breath and expressed as 30-s averages: pulmonary oxygen uptake (VO2 l.min-1 and ml.kg-1.min-1 STPD); pulmonary carbon dioxide output (VCO2 l.min-1 and ml.kg-1.min-1 STPD); respiratory exchange ratio (RER); pulmonary ventilation (VE l.min-1 BTPS); tidal volume (VT l.min-1 BTPS); respiratory rate (RR breath/min); functional estimate of dead space (VD/VT); ventilatory equivalents for oxygenand carbon dioxide(VE/VO2 and VE/VCO2) and end-tidal pressures for oxygenand carbon dioxide(PETO2 and PETCO2 mmHg). Before each test, the gas analyzers were calibrated using gases of known concentration of carbon dioxide and oxygen balanced with nitrogen, and the flow meter was calibrated using a 3-L syringe. Heart rate was continuously recorded at rest, during the graded exercise testing and recovery period using a 12-lead ECG (Marquette Medical Systems, InC. CardioSoft, Wisconsin, USA). All tests in this study were performed in the same laboratory at the same condition.
Exercise protocol - The subjects performed a ramp-like progressive exercise test until exhaustion on treadmill. The exercise workload (speed and/or slope) was increased every 60s with completion of the incremental part of exercise test between approximately 8 to 12 minutes.
Ventilatory anaerobic threshold - The ventilatory anaerobic threshold (VAT) was determined to occur at the break-point between the increase in the carbon dioxide output and VO2 (V-Slope) or the point at which the ventilatory equivalent for oxygen reached the minimum value and began to rise without a concomitant rise in ventilatory equivalent for carbon dioxide22,23.
Oxygen peak consumption - The VO2peak was defined as the maximal attained VO2 at the end of the exercise period in which the subject refers exhaustion (analog scale of the perceived exertion to Borg scale).
Experimental protocol - Initially, the subjects underwent resting ECG. Thereafter, they performed on the progressive treadmill exercise test, in following sequence: 1) Equipment adaptation was observed through ventilatory parameters during few minutes; 2) After this, the cardiorespiratory parameters were collected for 2 minutes at rest and 2 minutes in warm- up; 3) Therefore, incremental exercise test was started; and 4) It was closed with six minutes of monitored recovery.
Statistical analysis
The data are presented as mean ± SE. Unpaired Student t tests was used to examine physical characteristics, workload, time of exercise and the relative difference (percentage) for the cardiorespiratory variables. The chi-square (x2) test was used to assess the gender differences between children and adults. The cardiorespiratory responses during graded exercise test were analyzed between two groups at rest, VAT and peak of exercise using two-way analysis of variance (ANOVA) with repeated measures to test possible differences between groups. When significance was found, Scheffés' post-hoc comparison was performed. Probability values of < 0.05 were considered statistically significant.
For both groups, the relative difference was calculated for the following cardiorespiratory variables: absolute oxygen consumption (VO2 l.min-1), respiratory rate (RR), pulmonary ventilation (VE), carbon dioxide production (VCO2 l.min-1), tidal volume (VT) and respiratory exchange ratio (RER) for the intervals between rest to VAT, VAT to peak of exercise, and rest to peak of exercise.
Results
Baseline measurements - 25 children and 20 adults meeting the inclusion/exclusion criteria were considered for this study. The physical characteristics of the subjects are displayed in table 1. As expected, we found differences in age, height, weight, body surface area (BSA) and body mass index (BMI). However, we did not observe differences in gender (table 1).
Exercise measurements - At VAT, the children showed a significantly higher workload (speed and slope) and minutes of exercise than adults. However, at peak of exercise these variables are similar in both groups (table 2).
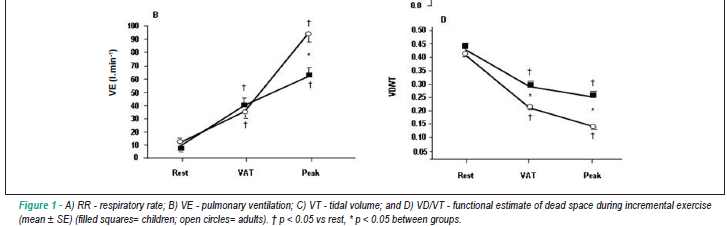
Cardiorespiratory measurements - The cardiorespiratory measurements at rest were similar between groups (table 3, figure 1 and 2). However, during exercise, responses at ventilatory anaerobic threshold (VAT) were significantly greater for HR, VO2 ml.kg-1.min-1, RR, VCO2 ml.kg-1.min-1, VD/VT, VE/VO2, VE/VCO2 and PETO2, while VT, O2 pulse and PETCO2 have a slighter increase in children than adults. At peak of exercise, differences significantly higher in children for the variables RR, VD/VT and VE/VCO2 were observed. However, for the variables O2 pulse, VO2 l.min-1, VT, VE, VCO2 l.min-1, VCO2 ml.kg-1.min-1, RER and PETCO2 children exhibited values significantly lower than adults.
Furthermore, the subsequent analysis in cardiorespiratory responses during graded exercise test to group by time interaction showed significant differences between the two groups for all variables included in this study (table 3, figure 1 and 2).
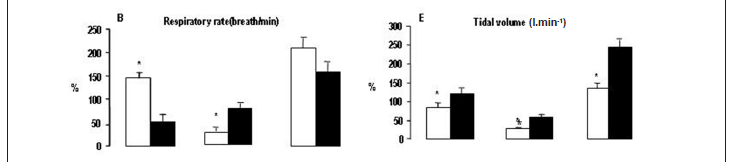
Relative differences in cardiorespiratory variables - Changes in cardiorespiratory variables between rest to VAT, VAT to peak of exercise, and rest to peak of exercise are shown in figure 3. Statistical analysis showed that the percentage in VE and RR between rest to VAT was significantly higher in children compared to adults. However, for variable VT, children exhibited values significantly lower. For the interval between VAT to peak of exercise, the percentage in VO2 l.min-1, RR, VE, VCO2 l.min-1, VT and RER was significantly lower in children. Statistical analysis also showed that the percentage in VCO2 l.min-1, VT and RER between rest to peak of exercise was significantly lower in children.
Discussion
The main findings of the present study are as follows: 1) children have different cardiovascular, ventilatory and metabolic responses during progressive maximal exercise test as compared to adults; and 2) children showed similar levels of both relative aerobic fitness and exercise capacity than adults. Concerning differences in cardiovascular responses, children showed higher chronotropic and lower inotropic response during maximal effort (table 3). In fact, these results are in agreement with previous reports17,24 that showed in children higher heart rate values at the same relative workload as adults. This higher heart rate observed in pediatric population is a compensatory mechanism for their smaller hearts and lower stroke volume17,24. Furthermore, this response may be related to more sensitive peripheral chemoreceptors sensing the build up of metabolites in the exercising muscles15. On the other hand, children demonstrated lower values for O2 pulse during maximal exercise compared with adults. Although O2 pulse is only an indirect estimate of stroke volume, we suggest that this finding may be due to the following factors: 1) smaller heart size; 2) lower stroke volume; and 3) smaller muscle mass which result in an attenuated venous return (preload) observed in children24.
In addition, our study shows alterations in ventilatory responses during progressive exercise, with children demonstrating lower ventilatory efficiency. In fact, we found values significantly higher for the respiratory rate and lower values to tidal volume during exercise in children (figure 1A and 1C). Moreover, children group demonstrate higher values for the ventilatory equivalent for O2 and CO2 to a given energy expenditure suggesting an increase of ventilatory requirements during physical effort (figure 2A and 2C, respectively). These findings emphasized that the ventilatory pattern in pediatric population is dependent on maturity2,25-27. In the same context, Armon et al27 observed in children higher pulmonary ventilation for a given VCO2 as compared to adults. What is the explanation for these findings? There are some evidence that younger children have a lower carbon dioxide set point4,13-15 and a higher central ventilatory neural drive16 resulting in higher ventilatory rates for a given metabolic demand.
Our findings are in agreement with this assumption, since the relative difference (percentage) has not showed differences for the VCO2 at rest to VAT between groups (figure 3D). However, the pulmonary ventilation demonstrated higher increase, suggesting higher ventilatory impulse for a given VCO2 in children (figure 3C). In addition, the children group showed different responses of both end-tidal pressures for oxygen and carbon dioxide during progressive exercise (figure 2B and figure 2D, respectively). Although PETO2 is only an indirect estimate of alveolar and arterial PO228,29,the fact that PETO2 was higher at VAT in children (figure 2B) can be attributed to a greater ventilatory impulse with subsequent rise in alveolar PO2. In fact, the pediatric population may be under relatively alveolar hyperventilation during exercise. In other words, excessive ventilation is observed during physical effort 30,31.
On the other hand, different authors28,29 suggest that PETO2 responses may be influenced by both VE/VO2 and breathing pattern used during exercise. For instance, the decrease or increase in VE/VO2 during exercise conducts a change in the same direction in PETO229. For the variable PETCO2, children have shown values significantly lower during progressive exercise than adults (figure 2D). Although PETCO2 is an indirect estimate of alveolar and arterial PCO228,29, our findings are in accordance with previous report31. Ohuchi et al31 demonstrated that the magnitude and change in end-tidal to arterial carbon dioxide tension difference P(ET- a)DCO2 during graded exercise were smaller, with children showing lower values than adults in both at anaerobic threshold and peak of exercise. We postulate that this finding may be explained by a lower ventilatory efficiency4,13,25-27 and a lower glycolytic capacity during exercise in children than adults1-3,6-9.
Interestingly, other authors29 suggested that higher values of respiratory rate are closely related to the decrease in expiratory time and as a consequence of a premature cessation, a rise in PETCO2 during progressive exercise. In fact, our findings are in accordance with this explanation, since we observed, during progressive exercise, higher respiratory rate in the children group. In addition, this lower ventilatory efficiency observed in children could be reinforced with the lower decline for VD/VT during progressive exercise, suggesting that there is higher ventilation in the anatomic dead space (figure 1D).
Furthermore, in agreement with a previous investigation32, our study also shows differences in metabolic responses during progressive exercise in children. We observed values significantly lower to the respiratory exchange ratio at peak of exercise in the children group (table III). Additionally, for the relative difference (percentage) children showed lower RER increase in both at rest to peak of exercise and at VAT to peak of exercise (figure 3F). These findings suggest that children have lower glycolytic capacity than adults. Several possible reasons are raised in literature for the immaturity of anaerobic metabolism in children during exercise, as follows: 1) lower levels of muscle glycogen5-7,33; 2) reduced activity of phosphofructokinase-1 and lactate dehydrogenase1-3,6-9; and 3) greater proportions of slow twitch fibers1.
Interestingly, despite these differences in cardiovascular and ventilatory responses during progressive exercise, the children group demonstrated similar levels of exercising capacity as adults (Table 2). We suggest that these results may be due to a greater metabolic efficiency observed in children and as a consequence, smaller muscle fatigue, since they had a minor reliance of the glycolytic metabolism during exercise.
Conclusion
In conclusion, these findings indicate that the cardiovascular, ventilatory and metabolic responses during progressive maximal exercise test are different in children as compared to adults. Specifically, the results found in the present study suggested that children have lower cardiovascular and ventilatory efficiency. On the other hand, children showed higher metabolic efficiency during progressive exercise. In summary, despite the differences observed, children demonstrated similar levels of the exercise capacity as compared to adults.
Limitations
We recognize limitations in the present study. The main limitation is that our patients were selected from a database, in a retrospective cross-sectional study. Information about the relationship between sample and population is limited in this condition of non-probability sampling. Thus, we cannot extrapolate the results of this study to the population.
Potential Conflict of Interest
No potential conflict of interest relevant to this article was reported.
Sources of Funding
There were no external funding sources for this study.
Study Association
This study is not associated with any post-graduation program.
References
- 1. Boisseau N, Delamarche P. Metabolic and hormonal responses to exercise in children and adolescents. Sports Med. 2000; 30 (6): 405-22.
- 2. Malina R, Bouchard C. Growth, maturation, and physical activity. Champaign (IL): Human Kinetics Publishers; 1991.
- 3. Falgairette G, Bedu M, Fellmann N, Van-Praagh E, Coudert J. Bio-energetic profile in 144 boys aged from 6 to 15 years with special reference to sexual maturation. Eur J Appl Physiol. 1991; 62: 151-6.
- 4. Cooper DM, Kaplan MR, Baumgarten L, Weiler-Ravell D, Whipp BJ, Wasserman K. Coupling of ventilation and CO2 production during exercise in children. Pediatr Res. 1987; 21: 568-72.
- 5. Prado DM, Dias RG, Trombetta IC. Cardiovascular, ventilatory, and metabolic parameters during exercise: differences between children and adults. Arq Bras Cardiol. 2006; 87 (4): e149- e155.
- 6. Kuno S, Takahashi H, Fujimoto K, Akima H, Miyamaru M, Nemoto I, et al. Muscle metabolism during exercise using phosphorus- 31 nuclear magnetic resonance spectroscopy in adolescents. Eur J Appl Physiol. 1995; 70: 301-4.
- 7. Inbar O, Bar-Or O. Anaerobic characteristics in male children and adolescents. Med Sci Sports Exerc. 1986; 18 (3): 264-9.
- 8. Zanconato S, Buchthal S, Barstow TJ, Cooper DM. 31P- magnetic resonance spectroscopy of leg muscle metabolism during exercise in children and adults. J Appl Physiol. 1993; 74 (5): 2214-8.
- 9. Eriksson BO, Gollnick PD, Saltin B. Muscle metabolism and enzyme activities after training in boys 11- 13 years old. Acta Physiol Scand. 1973; 87: 485-97.
- 10. Martinez LR, Haymes EM. Substrate utilization during treadmill running in prepubertal girls and women. Med Sci Sports Exerc. 1992; 24 (9): 975-83.
- 11. Rowland TW, Auchinachie JA, Keenan TJ, Green GM Physiologic responses to treadmill running in adult and prepubertal males. Int J Sports Med. 1987; 8 (4): 292-7.
- 12. Haralambie G. Enzyme activities in skeletal muscle of 13-15 years old adolescents. Bull Eur Physiopathol Respir. 1982; 18 (1): 65-74.
- 13. Nagano Y, Baba R, Kuraishi K, Yasuda T, Ikoma M, Nashibata K, et al. Ventilatory control during exercise in normal children. Pediatr Res. 1998; 43: 704-7.
- 14. Springer C, Barstow TJ, Cooper DM. Effect of hypoxia on ventilatory control during exercise in children and adults. Pediatr Res. 1989; 25: 285-90.
- 15. Springer C, Cooper DM, Wasserman K. Evidence that maturation of the peripheral chemoreceptors is not complete in childhood. Respir Physiol. 1988; 74: 55-64.
- 16. Gaultier C, Perret L, Boule M, Buvrey A, Gerard F. Occlusion pressure and breathing pattern in healthy children. Respir Physiol. 1981; 46: 71-80.
- 17. Vinet A, Nottin S, Lecoq A, Obert P. Cardiovascular responses to progressive cycle exercise in healthy children and adults. Int J Sports Med. 2002; 23: 242-6.
- 18. Cole TJ, Bellizzi MC, Katherine MF, Dietz WH. Establishing a standard definition for child overweight and obesity worldwide: international survey. BMJ. 2000; 320: 1-6.
- 19. Rowland TW. Pediatric laboratory exercise testing: clinical guidelines. Champaign (IL): Human Kinetics Publishers; 1993.
- 20. Maritz JS, Morrison JF, Peter J, Strydom NB, Wyndham CH. A practical method of estimating and individual's maximal oxygen uptake. Ergonomics. 1961; 4: 97-122.
- 21. Howley ET, Bassett DR, Welch HG. Criteria for maximal oxygen uptake: review and commentary. Med Sci Sports Exerc. 1995; 27: 1292-301.
- 22. Wasserman K, Whipp BJ, Koyal SN, Beaver WL. Anaerobic threshold and respiratory gas exchange during exercise. J Appl Physiol. 1973; 33: 236-43.
- 23. Wasserman K. The anaerobic threshold measurement to evaluate exercise performance. Am Rev Respir Dis. 1984; 12: S35-S40.
- 24. Kenneth TR, Wilmore JH. Cardiovascular responses to treadmill and cycle ergometer exercise in children and adults. J Appl Physiol. 1997; 83 (3): 948-57.
- 25. Rowland TW, Cunningham LN. Developmental of ventilatory responses to exercise in normal white children. Chest. 1997; 111: 327-32.
- 26. Prioux J, Ramonatxo M, Mercier J, Granier P, Mercier B, Prefaut C. Changes in maximal exercise ventilation and breathing pattern in boys during growth: a mixed cross-sectional longitudinal study. Acta Physiol Scand. 1997; 161 (4): 447-58.
- 27. Armon Y, Cooper DM, Zanconato S. Maturation of ventilatory responses to 1- minute exercise. Pediatr Res. 1991; 29: 362-8.
- 28. Wasserman K, Hansen JE, Sue DY, Casaburi R, Whipp BJ. Principles of exercise testing and interpretation. Philadelphia: Lippincott Williams & Wilkins; 1999.
- 29. Neder JA, Nery LE. Fisiologia clínica do exercício teoria e prática. São Paulo: Artes Médicas; 2003.
- 30. Gratas-Delamarche A, Mercier J, Ramonatxo M, Dassonville J, Préfaut C. Ventilatory response of prepubertal boys and adults to carbon dioxide at rest and during exercise. Eur J Appl Physiol. 1993; 66: 25-30.
- 31. Ohuchi H, Kato Y, Tasato H, Arakaki Y, Kamiya T. Ventilatory response and arterial blood gases during exercise in children. Pediatr Res. 1999; 45: 389-96.
- 32. Brown JD, Mahon AD, Plank DM. Attainment of maximal exercise criteria in boys and men. J Sports Med Phys Fitness. 2002; 42: 135-40.
- 33. Eriksson BO, Karlsson J, Saltin B. Muscle metabolites during exercise in pubertal boys. Acta Pediatr Scand. 1971; 217: 154-7.
Publication Dates
-
Publication in this collection
05 Mar 2010 -
Date of issue
Apr 2010
History
-
Accepted
18 Aug 2009 -
Reviewed
08 Mar 2009 -
Received
23 Sept 2008