Abstract
Methanolic extracts of the leaves of Atlantia monophylla (Rutaceae) were evaluated for mosquitocidal activity against immature stages of three mosquito species, Culex quinquefasciatus, Anopheles stephensi, and Aedes aegypti in the laboratory.Larvae of Cx. quinquefasciatus and pupae of An. stephensi were found more susceptible, with LC50 values of 0.14 mg/l and 0.05 mg/l, respectively. Insect growth regulating activity of this extract was more pronounced against Ae. aegypti, with EI50 value 0.002 mg/l. The extract was found safe to aquatic mosquito predators Gambusia affinis, Poecilia reticulata, and Diplonychus indicus, with the respective LC50 values of 23.4, 21.3, and 5.7 mg/l. The results indicate that the mosquitocidal effects of the extract of this plant were comparable to neem extract and certain synthetic chemical larvicides like fenthion, methoprene, etc.
Atlantia monophylla; plant extract; mosquitocidal activities
CONTROL
Laboratory evaluation of methanolic extract of Atlantia monophylla (Family: Rutaceae) against immature stages of mosquitoes and non-target organisms
N Sivagnaname1 1 Corresponding author. Fax: +91-413-227.2041. E-mail: nsivagnaname@yahoo.com ; M Kalyanasundaram
Vector Control Research Centre, Indian Council of Medical Research, Indira Nagar, Pondicherry, 605 006, India
ABSTRACT
Methanolic extracts of the leaves of Atlantia monophylla (Rutaceae) were evaluated for mosquitocidal activity against immature stages of three mosquito species, Culex quinquefasciatus, Anopheles stephensi, and Aedes aegypti in the laboratory.Larvae of Cx. quinquefasciatus and pupae of An. stephensi were found more susceptible, with LC50 values of 0.14 mg/l and 0.05 mg/l, respectively. Insect growth regulating activity of this extract was more pronounced against Ae. aegypti, with EI50 value 0.002 mg/l. The extract was found safe to aquatic mosquito predators Gambusia affinis, Poecilia reticulata, and Diplonychus indicus, with the respective LC50 values of 23.4, 21.3, and 5.7 mg/l. The results indicate that the mosquitocidal effects of the extract of this plant were comparable to neem extract and certain synthetic chemical larvicides like fenthion, methoprene, etc.
Key words:Atlantia monophylla - plant extract - mosquitocidal activities
The problems of high cost and development of resistance in many vector mosquito species to several of the synthetic insecticides have revived interest in exploiting the pest control potential of plants (Grainge & Ahamed 1988). In addition to application as general toxicants against mosquito immature, phytochemicals may also have potential uses as growth and reproduction inhibitors, repellents, and oviposition deterrents. Plants belonging to six families: Asteraceae, Cladophoraceae, Labiatae, Meliaceae, Oocystaceae, and Rutaceae appear to have potential for providing future mosquito control agents (Sukumar et al. 1991). Atlantia monophylla belonging to family Rutaceae has been earlier reported for insecticidal activity against pests of agricultural importance (Grainge & Ahamed 1988, Sukumar et al. 1991), but there has been no report on its activity against public health pests. The plant is available year round in Pondicherry, India. A study was therefore undertaken to test the extract of this plant for its efficacy against immature stages of selected mosquito species.
MATERIALS AND METHODS
Preparation of stock solution of plant extract - Fully developed leaves of the plant A. monophylla were collected during the flowering season of the plant, shade-dried and finely ground. The finely ground plant material was extracted with petroleum ether (boiling point range 60-80ºC) and methanol by standard method of extraction (Sujatha et al. 1988). The residue was then made into a 1% stock solution with acetone. The stock solutions for various test concentrations were prepared and one ml of the stock solution was added to 249 ml of tap water in a 500 ml enamel bowl.
Effects on target species - Larvicidal and pupicidal activities of the methanolic extracts of A. monophylla were determined by following the standard procedure (WHO 1975). Twenty early fourth instar larvae and 20 pupae of three vector mosquito species, Culex quinquefasciatus, Anopheles stephensi, and Aedes aegypti (obtained from the cyclic colony that has been maintained for the past 24 years at the Centre) were transferred to 249 ml of tap water taken in 500 ml bowls. Five replicates were set up for each test concentration and the plant extract was tested at concentrations ranging from 0.001 to 5 mg/l with two replicates of control with addition of 1 ml of acetone alone to 249 ml of tap water. Bioassay was conducted at room temperature 27 ± 3ºC with 85% relative humidity. In the case of experiment for determining pupicidal activity, the mouth of each bowl containing pupae was covered with muslin cloth to prevent the escape of any emerged adult mosquitoes. Mortality in larvae/pupae was recorded 24 h post-treatment.
Insect growth regulating (IGR) activity -Methanolic extract of A. monophylla was also tested for IGR activity against the three mosquito species. Twenty early third instar larvae of the three species were transferred into 500 ml enamel bowls containing 249 ml of tap water. The plant extract was tested at concentrations ranging from 0.001 to 2 mg/l. Each test concentration was replicated five times. Two replicates of control were also maintained. Mortality of the larvae and pupae was recorded at 24 h intervals. Observation was continued in treated and control bowls until the last immature pupated. The treated and untreated bowls containing pupae were kept separately in one-foot cages for adult emergence. Morphological abnormalities in pupae and adults and partially emerged adults were recorded as dead. Percentage of emergence inhibition (% EI) in the treated and control was monitored so that the EI50 and EI90 values can be calculated by probit analysis (Busvine 1971).
Effect on non-target organisms -The effect of the methanolic extract of A. monophylla was tested against non-target mosquito predators, Toxorhynchites splendens (mosquito predator), Gambusia affinis, Poecilia reticulata (predatory fishes), Diplonychus indicus (predatory water-bug), and Anisops bouvieri (Notonectasp.). T. splendens larvae were obtained from the cyclic colony that has been maintained for the past 23 years at the Centre, whereas other predator species were collected from field and maintained in cement tanks (85 cm diameter and 30 cm depth) containing field collected water at 27 ± 3ºC and relative humidity 85%. One predator was released into 500 ml disposable bowl containing 250 ml tap water. Only one predator was used so as to avoid cannibalism. The predators were exposed to test concentrations ranging from 1 to 50 mg/l. Ten replicates were performed for each test concentration along with four replicates of untreated controls. The predators were observed for mortality and other abnormalities such as sluggishness and reduced swimming activity after 24 h exposure. The exposed predators were also observed continuously for ten days to understand the post treatment effect of this extract on survival and swimming activity. LC50 and LC90 values were obtained by probit analysis. Suitability index (SI) or Predator safety factor (PSF) was calculated for each species of predator using the following formula (Deo et al. 1988).
RESULTS
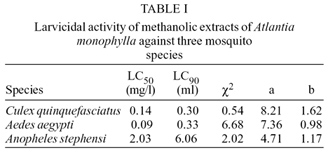
Larvicidal and pupicidal activities -Petroleum ether extract of the leaves of A. monophylla did not show any larvicidal activity against the three species of mosquito tested at concentrations ranging from 10 mg/l to 100 mg/l. Methanolic extract of the leaves was found to be effective against early fourth instar larvae of Cx. quinquefasciatus and Ae. aegypti and pupae of all the three mosquito species. Though the extract was less effective against the larvae of An. stephensi, it was more effective against the pupae of An. stephensi compared to other species (Table I, II). Ae. aegypti larvae were 1.5 and 22.4 times more susceptible than larvae of Cx. quinquefasciatus and An. stephensi respectively. Cx. quinquefasciatus and Ae. aegypti pupae were equally susceptible to this extract but pupae of An.stephensi were 1.4 times more susceptible than the other two vector species (Table II).
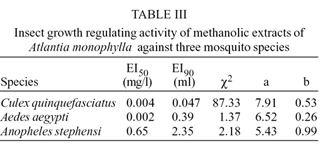
IGR activity -The IGR activity of the methanolic extract of A. monophylla tested against the larvae of three mosquito species is shown in Table III. When exposed to the plant extract, Ae. aegypti and Cx. quinquefasciatus were 325 times and 162.5 times more sensitive than An. stephensi respectively. Dead larval intermediates or pre-pupal stage larvae were found with An. stephensi and Cx. quinquefasciatus exposed to the plant extract. Many dead adults were found with curved tarsi and malformed wings, particularly with An. stephensi. Many "albino" pupae were found white with soft body and without characteristic sclerotization of the pupal cuticle.
Effect on non-target organisms -The effect of methanolic extract of A. monophylla against non-target organisms Tx. splendens, D. indicus, G. affinis,P. reticulata, and A. bouvieri is presented in Table IV. G. affinis and P. reticulata, were the least susceptible, with LC50 values of 23.4 mg/l and 21.3 mg/l, respectively. The extract was found to be highly lethal to A. bouvieri,with a LC50 of 0.15 mg/l. D. indicus was less susceptible to the plant extract than Tx. splendens. SI/PSF indicated that this plant extract is less harmful to predatory fishes than other mosquito predators tested (Table V). Survival and swimming activity of the test species were not altered during the exposure at LC50 and LC90 doses of the plant extract.
DISCUSSION
Vector control is facing a threat due to the emergence of resistance in vector mosquitoes to conventional synthetic insecticides, warranting either countermeasures or development of newer insecticides (Chandre et al. 1998). Botanical insecticides may serve as suitable alternatives to synthetic insecticides in future as they are relatively safe, degradable, and are readily available in many areas of the world. Though several plants from different families have been reported for mosquitocidal activity, only a very few botanicals have moved from the laboratory to field use, like neem based insecticides, which might be due to the light and heat instability of phytochemicals compared to synthetic insecticides (Green et al. 1991).
The methanolic extract of the leaves of the plant A. monophylla has been found to possess various activities, such as larvicidal, pupicidal, and insect growth regulation properties against the three mosquito species tested. The observed biological activity of the plant extract might be due to the alkaloid reported in this plant (Basu & Basa 1972). Minijas and Sarda (1986) reported that crude extract of saponin from the fruit pods of Swartzia madagascariensis produced higher mortality in larvae of An. gambiae than in Ae. aegypti, and no mortality was observed in Cx. quinquefasciatus.
The larvicidal efficacy is comparable to fenthion, a commercially available organophosporus larvicide (Amalraj & Das 1996). Insect growth regulating activity of this plant extract is more pronounced in Ae. aegypti than in other two vectors. The IGR activity of this extract against Cx. quinquefasciatus is 25 times more than methoprene, a juvenile hormone mimicking compound. But it is 7.5 times less effective than diflubenzuron, a chitin synthesis inhibitor (Robert & Olson 1980). The effect of the plant extract is superior to various neem extracts, which are reported to be effective with LC50 values ranging from 55-65 mg/l against mosquito larvae (Ascher & Meisner 1989).
Several plants have been reported to have mimics of insect ecdysones and juvenile hormone activity. As with toxicity, growth inhibition from phytochemicals may also be species-specific (Novak 1985). Sujatha et al. (1988) observed that Acorus calamus extract induced malformations to a greater extent in An. stephensi, and to a lesser extent in Cx. quinquefasciatus and Ae. aegypti. Earlier studies have shown that selected plant extracts were found to exhibit either larvicidal or insect growth regulating activity against mosquito larvae at concentration above 10 mg/l (Deshmukh & Renapurkar 1987, Thangam & Kathiresan 1988). But the LC50 values of the methanolic extract of A. monophylla were less than 0.2 mg/l against Ae. aegypti and Cx. quinquefasciatus.
Results of the effect on non-target organisms have revealed that methanolic extracts of A. monophylla are safe to certain mosquito predators tested. The extract is non-toxic up to a concentration of 5 mg/l to the non-target predatory fishes G. affinis and P. reticulata and aquatic bugs D. indicus. The SI favored predatory fishes more than the other predators exposed to this plant extract, suggesting that this plant extract could be used along with these predatory fishes in Integrated Vector Control (IVM). The LC50 value of this plant extract against A. bouvieri indicates that the usage of this extract should be restricted in mosquito breeding habitats co-inhabited by this predator.
ACKNOWLEDGEMENT
To the constant encouragement of Dr PK Das, Director, Vector Control Research Centre and also to Dr D Dominic Amalraj, Assistent Director, and Dr Nisha Mathew, Technical Officer who have critically gone through the manuscript and rendered valuable suggestions in improving the quality of this paper. To the technical assistance rendered by Mr S Srinivasan (Chemistry Laboratory) and Mr A Nagavelu (Vector Biology and Control) and to the staff of Insect Rearing and Colonization, Vector Control research Centre, Pondicherry, India. To the help extended by Mrs R Sundarammal, Information Officer, Vector Control Research Centre and Dr T Ganesan, Lecturer, PG Studies, Botany Department, Tagore Arts College, Pondi-cherry, India.
Received 21 September 2003
Accepted 12 January 2004
- Amalraj DD, Das PK 1996. Toxicity of insecticides to Toxorhynchites splendens and three vector mosquitoes and their sublethal effect on biocontrol potential of the predator. South East J Trop Med Pub Health 27: 154-159.
- Ascher KRS, Meisner J 1989. The effects of Neem on insects affecting man and animals. In Martin Jacobson, Focus on Phytochemical Pesticides, Vol. I, The Neem Tree, CRC Press, Inc., Florida, p. 113-131.
- Basu D, Basa SC 1972. N-Methylbicycloatalaphylline, a new alkaloid from Atlantia monophylla Correa. J Org Chem 37: 3035.
- Busvine RJ 1971. A Critical Review of the Techniques for Testing Insecticides, Commonwealth Agricultural Bureau, London, p. 263-288.
- Chandre F, Darriet F, Darder M, Cuany A, Doannio JMC, Pasteur N, Guillet P 1998. Pyrethroid resistance in Culex quinquefasciatus from West Africa. Med Vet Entomol 12: 359-366.
- Deo PG, Hasan SB, Majumdar SK 1988. Toxicity and suitability of some insecticides for household use. Int Pest Control 30: 118-129.
- Deshmukh PB, Renapurkar DM 1987. Insect growth regulatory activity of some indigenous plant extracts. Insect Sci Appli 8: 81-83.
- Grainge M, Ahmed S 1988. Handbook of Plants with Pest Control Properties, John Wiley & Sons, New York, p. 41.
- Green MM, Singer JM, Sutherland DJ, Hibbon CR 1991. Larvicidal activity of Tagetes minuta (Marigold) towards Aedes aegypti J Am Mosq Control Assoc 7: 282-286.
- Minija JN, Sarda RK 1986. Laboratory observations on the toxicity of Swartzia madagscariensis (Leguminosae) extract on mosquito larvae. Trans R Soc Trop Med Hyg 80: 460-461.
- Novak D 1985. Non-chemical approaches to mosquito control in Czechoslavakia. In M Laird, JW Miles (eds), Integrated Mosquito Control Methodologies, Vol. 2, Academic Press, San Diego, p. 185-196.
- Robert LL, Olson JK 1989. Effects of sublethal dosages of insecticides on Culex quinquefasciatus J Am Mosq Control Assoc 5: 239.
- Sujatha CH, Vasuki V, Mariappan T, Kalyanasundaram M, Das PK 1988. Evaluation of plant extracts for biological activity against mosquitoes. Int Pest Control 30: 122-124.
- Sukumar K, Perich MJ, Boobar LR 1991. Botanical derivatives in mosquito control. A review. J Am Mosq Control Assoc 7: 210-217.
- Thangam TS, Kathiresan K 1988. Toxic effects of seaweed extracts on mosquito larvae. Ind J Med Res 88: 35-37.
- WHO-World Health Organization 1975. Instruction for determining the susceptibility or resistance of mosquito larvae to insecticides. WHO/VBC/75.583. Mimeographed document.
Publication Dates
-
Publication in this collection
31 Mar 2004 -
Date of issue
Feb 2004
History
-
Received
21 Sept 2003 -
Accepted
12 Jan 2004