Abstract
Field tests were carried out during the rainy season of 2001/2002 in São Luís de Montes Belos, Goiás, Brazil, to evaluate the potential of the entomopathogenic fungus, Beauveria bassiana, against peridomestic Triatoma sordida. An oil-water formulation of the isolate CG 14 (Embrapa) was applied in triatomine infested hen houses of four farms at a final concentration of 10(6) conidia/cm². Numbers of T. sordida decreased over the next 25 days, after application of the fungus, and B. bassiana developed on dead insects in one hen house. A high number of B. bassiana colonies was detected in substrates collected in treated hen houses 24 h after application of CG 14. In the following three months the presenceof B. bassiana declined to values found before treatment.
field test; Triatoma sordida; Beauveria bassiana; peridomestic; Central Brazil
CONTROL
Activity of oil-formulated Beauveria bassiana against Triatoma sordida in peridomestic areas in central Brazil
Christian LuzI,1 1 Corresponding author. Fax +55-62-521.1839. E-mail: wolf@iptsp.ufg.br ; Luiz FN RochaI; Gustavo V NeryI; Bonifácio P MagalhãesII; Myrian S TiganoII
IInstituto de Patologia Tropical e Saúde Pública, Caixa Postal 131, 74001-970 Goiânia, GO, Brasil
IIEmbrapa Recursos Genéticos e Biotecnologia, Brasília, DF, Brasil
ABSTRACT
Field tests were carried out during the rainy season of 2001/2002 in São Luís de Montes Belos, Goiás, Brazil, to evaluate the potential of the entomopathogenic fungus, Beauveria bassiana, against peridomestic Triatoma sordida. An oil-water formulation of the isolate CG 14 (Embrapa) was applied in triatomine infested hen houses of four farms at a final concentration of 106 conidia/cm2. Numbers of T. sordida decreased over the next 25 days, after application of the fungus, and B. bassiana developed on dead insects in one hen house. A high number of B. bassiana colonies was detected in substrates collected in treated hen houses 24 h after application of CG 14. In the following three months the presenceof B. bassiana declined to values found before treatment.
Key words: field test - Triatoma sordida - Beauveria bassiana - peridomestic - Central Brazil
Entomopathogenic fungi, such as Beauveria bassiana and Metarhizium anisopliae have been reported to be highly active under laboratory conditions against Triatoma infestans and other triatomine species (Romaña & Fargues 1987, Luz et al. 1998 a, b, Luz & Fargues 1999, Fargues & Luz 2000, Lecuona et al. 2001) and to reproduce on their cadavers (Luz & Fargues 1998, Fargues & Luz 1998). However, there is little information about activity of fungi against Triatominae under field conditions. Preliminary simulated field tests with a B. bassiana isolate in Brazil indicated that activity against T. infestans may be reduced compared to laboratory conditions (Luz et al. 1999). The recent development of effective oil-formulations of entomopathogenic fungi is opening new possibilities for environmentally safe control strategies (Lomer et al. 2001). We report here on field and semi-field tests with oil-formulated B. bassiana in Central Brazil to control peridomestic T. sordida.
MATERIALS AND METHODS
Tests were carried out in four different farms in the rural proximity of São Luis de Montes Belos, Goiás, in Central Brazil during the rainy season of 2001/2002. Farms were selected because of their peridomestic infestation with T. sordida and a comparable structure of the hen houses, which were generally built with loose bricks and roofing tiles and covered by wooden planks and fragments of black plastic bags. All hen houses covered an area of about 5 m2. In previous studies, M. anisopliae and B. bassiana were detected two months before application of B. bassiana in substrates of all tested farms with the exception of the first farm where this fungus was not found (C Luz, LFN Rocha, and GV Nery unpublished observations). In each farm, one hen house was fungus treated and another hen house, 5 to 10 m distant, was used as a control without applying B. bassiana. Temperature and humidity were monitored constantly in one of the farms in a shelter close to the hen house, using a mechanical hygrothermograph.
Detection of fungi in hen houses - Prior to and during the trial, six samples of substrate that consisted of soil, organic material such as animal faeces, and plant litter were taken from each hen house at randomly selected locations inside, where triatomines probably circulate during the night. About 25 g substrate were scraped to a depth to 2-3 cm, transferred to plastic bags and stored in a styrofoam cooler at about 20°C until processing in the laboratory. Two different techniques to detect entomopathogenic fungi were used.
In vivo detection - Newly emerged and unfed third instar nymphs (N3) of T. infestans, were used to detect entomopathogenic Hyphomycetes in substrates and to test survival of conidia applied to filter papers and exposed to field conditions. Nymphs of T. infestans were used because they are easier to mass-rear than nymphs of T. sordida. The T. infestans colony was originally from the state of Paraná, Brazil, and has been maintained in the laboratory since 1981. Insects were fed on chickens every two weeks, and maintained at 25 ± 0.5°C, 75 ± 5% relative humidity (RH) and 12 h photophase (Silva 1985).
The substrates of each sample were homogenized and approximately 3 g were transferred to Petri dishes (90 x 15 mm). Ten N3 were set on the substrate and exposed during 15 days at 25°C and relative humidity close to saturation (RH > 98%). Mortality was monitored daily. Dead insects were dipped in 93% alcohol, their surface disinfected in 2.5% sodium hypochlorite for 3 min and then washed three times for 1 min in sterile water. Cadavers were then incubated during 10 days at 25°C and RH > 98%. Fungal development on the cadavers was evaluated daily and sporulating fungi inoculated onto complete medium (CM) amended with chloramphenicol (1 g/1000 ml): 0.001 g FeSO4, 0.5 g KCl, 1.5 g KH2PO4, 0.5 g MgSO4.7 H2O, 6.0 g NaNO3, 0.001 g ZnSO4, 1.5 g hydrolysed caseine, 0.5 g yeast extract, 10 g glucose, 2 g peptone, 20 g agar, and 1000 ml distilled water.
In vitro detection - Hyphomycetes were isolated from the substrates using modified Chase medium (MCM): oatmeal infusion (2%), 20 g agar, 0.3 g dodine (N-dode-cylguanidine monoacetate, Cyprex 65 WP), 5 mg chlortetracycline, 0.4 g penicillin, 1 g streptomycin, 10 mg crystal violet, and 1000 ml distilled water (Chase et al. 1986). Samples of 1 g substrate were suspended in 10 ml of sterile 0.1% Tween 80 and vortexed for 3 min. Each suspension was then diluted 1/100 in distilled sterile water, spread onto MCM and incubated for 20 days at 25°C. Developing CFU (colony forming unit) were examined daily and inoculated separately on CM after formation of conidia. Cultures of B. bassiana and M. anisopliae were identified by microscopic examination.
Field tests - The monosporal B. bassiana strain CG 14 (Embrapa Genetic Resources and Biotechnology, Brasília, Brazil) was used in the tests. This strain was obtained in 1988 in Londrina, Paraná, Brazil from a hemipteran Podisus sp. (Pentatomidae) and reported to be virulent against T. infestans under laboratory conditions (Luz et al. 1998a). The fungus was cultivated in culture bags with 500 g parboiled rice and 300 ml distilled water in each bag for 10 days at 25°C and 12 h photophase. Conidia were harvested directly by sieving and aspiration. They were then dried over silica gel during two weeks and stored at 4°C. Just before application conidia were resuspended in 10% aqueous emulsifier added with vegetable oil (Veget'Oil, Oxiquimica Agrociência Ltda). The final concentration was adjusted to give 106 conidia/cm2 of treated surface. Viability of formulated conidia was tested inoculating 100 µl of the suspension on CM and evaluating quantitative germination within 24 h after inoculation at 25°C. Before application of conidia the approximate number of T. sordida and their instars were checked during 10 min in the hen houses. For this, hen houses were carefully dismantled and rearranged without removing the triatomines or altering the structure of the hen house. Four hen houses were then treated with 5 l of the formulation each and four control hen houses only with 5 l of the emulsion using a manual high pressure 5 l sprayer (Brudden Equipment Ltd). Covering materials were removed before application and the formulation sprayed on the bricks and the roofing tiles. Hen houses were then covered again. All hen houses were checked during 25 days every five days during 10 min for live and dead triatomines as mentioned before. Dead insects were transferred to Petri dishes and kept in the same location. Fungal development on cadavers was evaluated at five-day intervals during at least 10 days. Living insects detected 25 days after spraying were captured and transferred to laboratory at 25°C and RH > 98%.
Tests on survival of conidia in the field - A strip of filter paper (20 x 3 cm) and a 9 cm diameter circular filter paper were treated with CG 14 using the manual sprayer as mentioned above and fixed on the inner wall of a shelter close to the hen house in each farm. Viability of conidia was tested 24 h after application and their activity against T. infestans during three months. A piece of 2 cm was cut from the strip, vortexed for 5 min in 5 ml 0.1% Tween 80, and 200 µl plated on CM amended with chloramphenicol or MCM and incubated at 25°C. Germinating conidia were examined at 12 and 24 h after inoculation, counting 100 conidia for every strip. Formation of CFU on MCM was evaluated during 20 days. Activity of CG 14 was tested monthly by exposing 10 T.infestans N3 on the circular filter paper under laboratory conditions. Insects were incubated at 25°C, 75% RH and RH > 98%, respectively. Relative humidities of 75% and > 98% inside the test chambers were regulated with saturated solutions of NaCl and K2SO4, respectively (Winston & Bates 1960). Mortality and fungal development on cadavers were evaluated as mentioned for the laboratory tests with T. sordida.
Semi-field tests - Bricks (19 x 19 x 9 cm) with eight openings were treated at a dosage of 106 conidia/cm2 surface by submersion into a formulation of conidia prepared as mentioned above. After drying, bricks were transferred to cloth bags (33 x 40 x 22 cm) with 10 field collected T. sordida of randomly selected nymphal instars (N2 - N5) and adults on the bricks. The insects had been captured the day before treatment in other farms of the same region. No first instar nymphs were used due to their vulnerability. The number of N2 and N3 in the bags varied between zero and one specimen and each bag contained one N4, five N5 and three adults. Bags were carefully closed and fixed at about 2 m height in a shelter close to the hen houses. In each farm four bricks treated with the fungal formulation and two bricks with the formulation without conidia which served as controls were tested separately. Mortality was evaluated during 25 days every five days. Dead insects were transferred to Petri dishes and exposed in the bags. Fungal development was evaluated as mentioned before.
Laboratory tests - Field-collected nymphs of different instars and adults of T. sordida were brought to laboratory. Ten randomly selected nymphs and adults were set on filter paper (9 cm diameter) and treated topically at a final dose of 106 conidia/cm2 surface using a Potter sprayer (Burckard, Hertfordshire, UK). After drying, insects were transferred to untreated filter paper in a Petri dish and incubated during 15 days at 25°C, 75% RH and RH > 98%, respectively. Mortality was examined daily and fungal development on cadavers evaluated in a humid chamber as mentioned above. Four replicates were assayed for both humidities.
Characterization of B. bassiana isolates - A total of 17 B. bassiana isolates detected before and after application of the isolate CG 14 were analyzed by RAPD and compared with CG 14 and an outgroup isolate, CG 478, from Anthonomus grandis (Coleoptera: Curculionidae), Campinas, São Paulo, Brazil in 1983. One isolate from the second and third farm and two isolates from the fourth farm originated from substrates which had been collected before application of CG 14. Another 13 isolates detected 24 h, 25 days or one month after application of CG 14 in substrates, or on T.sordida were analyzed. Mycelium used for RAPD analysis was obtained from a submersed culture of conidia in CM without agar shaking at 150 rpm at 25°C for three days. Mycelium was harvested by filtration through filter paper (Whatman No. 1), lyophylized and stored at - 80°C. Genomic DNA was extracted using a simple method described by Al-Samarrai and Schmid (2000). The polymerase chain reaction (PCR) reactions were performed in 30 µl volume, with 15 ng of each template, using the PTC-100 programmable thermal controller (MJ Research), and a temperature profile described by Tigano et al. (1995). The amplifications were done using the following reaction mix: 1 unit of Taq polymerase (Life Technologies), 1 x Taq polymerase reaction buffer, 4 mM of MgCl2, 200 µM of each deoxynucleotides triphosphate (Pharmacia Biotec), and 1 µM of 10-mer primer (Operon Technologies). Twenty primers were used: OPA-09, OPA-13, OPA-14, OPA-16, OPB-05, OPB-06, OPB-10, OPB-17, OPE-01, OPE-02, OPE-03, OPE-04, OPE-07, OPE-14, OPE-15, OPE-16, OPE-19, OPE-20, OPR-08, and OPR-14. Amplified products were electrophored in 2% agarose gel dissolved in 0.5 x Tris-borato-EDTA (TBE) buffer. After electrophoresis, gels were stained with ethidium bromide (Sambrook et al. 1989) and photographed under UV light. DNA fingerprints were scored directly from the photographs. The presence or absence of each fragment was considered as an independent character.
Data analysis -Insect mortality and number of CFU were analyzed by ANOVA (F) or Kruskal-Wallis analysis (H) and the multiple Student-Newman-Keuls range test of comparison of means. Means were considered significantly different at P < 0.05. Values of recovery of live T. sordida in the hen houses were analyzed with the T-test (t) or Mann-Whitney-Rank-Sum test (T) at a < 5% (SAS Institute Inc. 2000). RAPD markers were analyzed using NTSYS-pc V2.1 (Exeter Software, Setauket, NY). A similarity matrix was calculated using Jaccard similarity coefficients. Clustering was done by UPGMA.
RESULTS
All hen houses were found infested with T. sordida before application of CG 14. No other triatomine species was identified among captured specimens. Highest rates (up to 97 specimens) were observed in the hen houses of the first farm. The number of captured insects in the other farms was between 15 and 29 specimens. The number of different instars of T. sordida found in all farms is presented in Table I. Temperature and relative humidity monitored in the shelter during 25 days after treatment varied between 17°C and 31°C and 37% and > 98%, respectively. Highest temperatures with lowest humidities were registered between 12 and 4 p. m. and lowest temperatures with highest humidities during the night between 10 p. m. and 6 a. m. Periods of constant relative humidity > 98% in this shelter did not exceed 10 h.
Field tests - Germination of formulated conidia tested on CM either directly without application in the field or after application on filter paper and exposure under field conditions during one day was > 98%, 24 h after inoculation. After suspending one month old filter paper in 0.1% Tween and inoculation of 100 ml of the suspension on CM a high number of bacteria and fungi, but no B. bassiana was found. However, inoculating the same suspension on MCM, B. bassiana colonies developed. No B. bassiana was observed when testing two or three months old filter papers independently of the medium utilized.
Mean recovery rates of live insects decreased in most fungus-treated and control hen houses compared to the number of individuals found directly before application of B. bassiana. Higher rates of live T. sordida were found in the control hen houses, especially of the first farm (Fig. 1) compared to the rates found in the fungus-treated hen houses. In the other farms with lower initial insect infestation, percentages of live individuals in the treated hen houses varied from 0% up to 130% related to the number of insects counted before treatment. A highly significant difference between values of recovery of live insects in the fungus-treated and control hen house during 25 days of observation was found for the hen houses of the first farm with the highest initial number of insects (t = 3.7, P = 0.006). The composition of T. sordida instars which were detected live in all hen houses after application of the fungus, related to the total number found before the treatment is shown in Table I. Twenty five days after application, nine T. sordida nymphs and adults were captured in the fungus-treated hen houses and 15 in the control areas and transferred to the laboratory. Mortality was 33.3% for the insects originating from fungus-treated areas and 6.7% from the control areas, 15 days after incubation at RH > 98%. On all cadavers, including one dead adult from a control hen house, development of B. bassiana was observed, 10 days after exposure in a humid chamber.
Fig. 1: recovery of live Triatoma sordida after application of Beauveria bassiana, CG 14, in a triatomine-infested farm compared to the number of insects found before treatment.
Dead insects were found only in the fungus-treated hen houses during the 10 first days after treatment, with highest rates in the hen house of the first farm. The recovery of live insects, as compared with the initial population before fungal application was as follows. At five days after application, 18.8% adults and 22.2% N5 were recovered; at 10 days after application, 21.9% adults, 38.9% N5, and 18.2% N4 were recovered. Most of the cadavers in the first farm were detected close to a colony of ants at the bottom of the hen house and ants were observed carrying entire dead individuals or fragments away into their colony. No cadavers were found between 15 and 25 days after treatment in this hen house. Distinct development of B. bassiana five days after transfer to Petri dishes and exposure in the fungus-treated hen house of the first farm was observed on one dead N4, three N5 and four adults. However, B.bassiana was not more visible on those cadavers, 25 days after treatment and could not be isolated with MCM. Other saprophytic fungi were observed on the cadavers. In the second and fourth farm, in the fungus-treated hen houses, one dead adult was detected five and ten days after treatment, respectively. No fungi had developed on these cadavers after exposure in Petri dishes in the hen houses up to 25 days after treatment.
Detection of B. bassiana and M. anisopliaein substrates, after application of CG 14 - Mortality of T. infestans N3 after exposure to substrates collected 24 h after fungal application in the four hen houses varied between 8% in the third hen house and 94% in the second hen house, 15 days after incubation under laboratory conditions. Mean values of mortality and fungi detected on cadavers are presented in Table II. B. bassiana was detected on 60.6% of the insects which had died after exposure to substrates originating from all hen houses. The highest number (80%) of mummified cadavers with only B. bassiana were found testing substrates from the hen house of the first farm. For the hen house of the third farm only one dead individual was found with B. bassiana. In addition, M. anisopliae was found on cadavers in the first (20%), third (50%) and fourth (4.8%) farms. One dead nymph showed a mixed development of both, B. bassiana and M. anisopliae on the integument. High amounts of not identified saprophytic fungi were detected on dead insects after exposure of N3 to substrates from the second (44.7%) and fourth (38.1%) farms.
Mortality of T. infestans N3, 15 days after exposure to substrates, which had been collected and tested one month after application of CG 14 in treated hen houses of the farms, varied between 0% in the first farm and 52% in the fourth farm. Both, B. bassiana and M. anisopliae were isolated from cadavers (Table II). Mortality of N3 decreased when insects were exposed to substrates collected in the farms two or three months after treatment and varied between 0% and 8%, two months, and between 0 and 10%, three months after application. M. anisopliae, but no B. bassiana was detected on cadavers after exposure of N3 on two and three months old substrates.
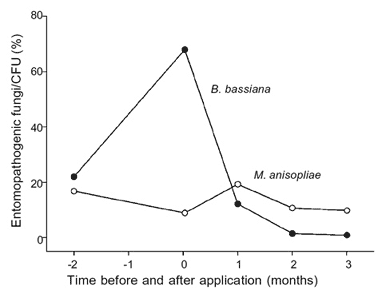
Percent of CFU identified as B. bassiana originating from substrates collected after application of CG 14 in the hen houses was highest, 24 h after treatment, with 67.7 (standard error of the mean 15.8%) and decreased significantly from numbers found one month, 7.4 (5.6%) two months, 1.5 (1.1%), and three months, 0.9 (0.5%) (H = 8.6, P = 0.036, DF = 15; 3) (Fig. 2). No difference in the number of CFU identified as M. anisopliae, which varied between 8.9 (7.7%), 24 h after application and 19.3 (8.8%), one month after treatment, was observed up to three months after application of CG 14 (H = 1.6, P = 0.65, DF = 15; 3) (Table II).
Fig. 2: relative number of colony forming units (CFU) identified as Beauveria bassiana and Metarhizium anisopliae, and detected within two months before and three months after application of B. bassiana, CG 14, in substrates from fungus-treated areas. compared to the number of insects found before treatment.
Characterization ofB. bassianaisolates - Analysis by RAPD showed high similarity (> 97%) of most tested isolates (82.4%) with CG 14, including all isolates detected in the four farms in substrates or T. sordida after application of CG 14. One isolate detected in substrate in the third farm before fungal treatment was 100% similar to CG 14. Another isolate in the second and two isolates in the fourth farm detected in substrates collected before treatment, differed at > 82%, > 85% and 39%, respectively from CG 14 (Fig. 3). These isolates were not observed within the tested group of B. bassiana isolated from substrates or T. sordida after treatment of hen houses.
Fig. 3: dendrogram constructed from RAPD data, indicating the relationships among Beauveria bassiana, CG 14, and B. bassiana isolates detected in substrates or dead Triatoma sordida adults (Ad) or fifth instar nymph (N5) originating from hen houses located in four farms in Central Brazil within two months before and one month after application of CG 14 and an outgroup isolate, CG 478. A similarity matrix was calculated using the Jaccard coefficient, and the tree was generated from this matrix by unweighted pair group method, arithmetic mean (UPGMA).
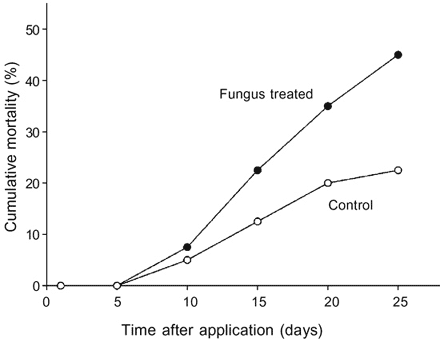
Semi-field test - Initial mortality was observed during 10 days after exposure of T. sordida to fungus-treated bricks inside the bags which had been installed in each farm (Fig. 4). Median cumulative mortality of insects, 25 days after treatment, reached 45 (2.9%) compared to 22.5 (6.3%) for the control insects with no significant difference between values obtained for the test and control mortalities (t = 25.0, P = 0.057). No fungal development was observed on cadavers which had been exposed in the bags under field conditions. Twenty five days after exposure of insects on the bricks, 80 live T. sordida nymphs (N1 - N5) and adults found on fungus-treated bricks and 56 individuals from the control bricks were transferred to the laboratory. Mortality of insects, 10 and 25 days after incubation at RH > 98% was 33.9 (7.8%) and 61.5 (12%), respectively. Control mortality 25 days after incubation was 12 (6%). Except one N5 from fungus-treated bricks of the fourth farm and dead control insects, all cadavers showed development of B.bassiana after exposure in a humid chamber.
Fig. 4: percent cumulative mortality of Triatoma sordida after ex-posure to bricks containing conidia of Beauveria bassiana, CG 14, in peridomestic areas in Central Brazil.
Laboratory assays - Cumulative mortality of T. sordida was > 95%, 15 days after direct spraying of conidia on the insects and exposure at 75% RH and RH > 98%. No significant difference was found between mortalities obtained at different humidities tested, 5 (t = 1.4, P = 0.21), 10 (t = 0.8, P = 0.48) and 15 days (t = 0.7, P = 0.54) after treatment. However, no mortality was observed after exposure of T. sordida on a fungus-treated brick and exposure at 75% RH during 15 days. At relative humidity close to saturation, 90% of insects had died, 10 days and all insects were found dead, 15 days after exposure on the brick. B. bassiana developed on all cadavers in a humid chamber, independently of the application method and humidity during the infection tested.
All T.infestans N3 survived after exposure on fungus-treated filter papers from different farms and incubation at 75% RH and 25°C during 15 days, independently of the period of exposure of the filter papers under field conditions. When insects were incubated on filter papers which had been transferred 24 h after fungal application to the laboratory at RH > 98%, 52.5 (16%) of N3 had died within 10 days and total mortality was observed 15 days after incubation. Mortality reached 12.5 (12.5%), 15 days after exposure, when tested on one month old filter papers and no more mortality was observed, testing filter papers which had been exposed two and three months under field conditions. Colored spots and growth of saprophytic fungi were observed on the filter paper, one month after treatment and exposure in the farms. B. bassiana developed on all dead insects after transfer to a humid chamber.
DISCUSSION
Our results clearly showed that peridomestic populations of T. sordida were reduced after application of B. bassiana strain CG 14 in their habitats. Insects were killed and B. bassiana was found recycling on cadavers. However, triatomine populations could not be eliminated permanently in the fungus-treated areas. Investigations on the activity of entomopathogenic fungi on triatomine bugs under field conditions are important in order to evaluate their potential for biological control of those vectors. Frequently, results obtained in the laboratory are not reproduced in field tests, where interaction among pathogens, vectors and environment are complex. In the laboratory, triatomines were highly susceptible to B. bassiana and M. anisopliae (Romaña & Fargues 1987, Luz et al. 1998a, b, Lecuona et al. 2001). In another study, the strain CG 14 induced total mortality of T. infestans N3 in the laboratory at RH > 98% and 85% mortality at 50% RH, 15 days after inoculation (Luz et al. 1998a). Reduced activity of this and other B.bassiana strains against T. infestans found in that study and in preliminary field tests by Luz et al. (1999) may be due to sub-optimal conditions of humidity. Temperature and moisture in the triatomine habitats are not constant and may vary according to daily or seasonal cycles. Simulation of humidity under laboratory conditions showed that constant or minimal periods of 12 h 97% RH induced rapid and total mortality of R. prolixus treated with B. bassiana and permitted recycling of the fungus on cadavers. At lower moistures more insects survived and B. bassiana did not emerge from dead insects (Luz & Fargues 1998, 1999, Fargues & Luz 1998, 2000). Only at 43% RH an increasing susceptibility of R. prolixus and T. infestans to fungal infection was reported by Luz (1990) and Romaña (1992), respectively, and can be expected also for other triatomine species. Many triatomine species, such as T. sordida occur in regions with arid or semi-arid climate and humidity during the dry season may then be unfavorable for external fungal development. It is important to consider microclimatic conditions in triatomine's habitat which may differ significantly from conditions prevailing in the area. A dumping effect of fluctuating temperature and humidity, which delays conditions of temperature and humidity as shown by Lorenzo and Lazzari (1999) and Vazquez-Prokopec et al. (2002) in domestic and peridomestic ecotopes of T. infestans, compared to ambient conditions, can interfere with fungal development on insects. During the experiment moderate precipitations were observed, but RH monitored in one of the shelters did not reach 97% during at least 12 h a day. However, in the fungus-treated hen house of the first farm, where most dead triatomines were detected after application of CG 14, RH was obviously favorable for high rates of infection and development of B. bassiana on cadavers. Similar observations in peridomestic habitats with prolonged periods of elevated moisture were also reported in simulated field trials with B. bassiana and R. prolixus in Colombia (Luz 1994).
Interestingly high mortalities were observed in the laboratory after direct application of the oil-based formulation, independently of humidity tested. This was not observed after indirect treatment by contact to treated bricks under semi-field conditions, where ambient humidity was unfavorable for fungal development due to the permanent ventilation of the bags or in the laboratory at 75% RH. Direct treatment provided a distinct higher dose of conidia on each insect than indirect application after exposure to treated bricks. However, direct treatment of triatomine insects, which are active during the night seems difficult. Formulations and application techniques which increase quantitative inoculation of fungal propagules on target insects will probably enhance fungal activity against triatomines under field conditions.
Quantitative evaluation of the effect of CG 14 on free-living peridomestic triatomine bugs proved to be difficult. T. sordida such as most other species are active at night and hide in their resting places during the day. Triatomine nymphs and adults often remain immobile even after exposure to day light and younger instars are more difficult to detect due to their small size. Moreover, T. sordida as other species (Zeledón et al. 1973) have a distinct camouflaging activity, covering their body with fine particles. The time-limited day capture was considered as the most adequate method to detect routinely live and dead triatomines during a prolonged period in their habitats without affecting live individuals. However, numerous agonizing or dead insects, even adults, were probably not detected and consequently not considered. Infected individuals can modify their behavior and leave their hiding places during the day. Many of these insects were presumably preyed by fowl and other vertebrates or arthropods and numerous dead insects disappeared without delay by the activity of ants. The high heterogeneity of live insects found in fungus-treated and control hen houses throughout the field tests was probably related to the small experimental areas and the initial reduced population size in three of the tested farms. Insects could move freely between treated, control and untreated areas. Behavior of the triatomines in the treated areas may have been altered at the beginning of the experiment and during controls of mortality. Many triatomine species including Triatoma spp. avoid wet habitats. Application of aqueous oil formulation, which caused a temporary moistening of the habitats, induced probably a time limited evasion in both, fungus-treated and control hen houses of a part of the T. sordida population to untreated areas. A part of contaminated individuals remained and died there and consequently disappeared undetected. Wiesinger (1956) did not find an influence of humidity on hiding activity of T. infestans and insects were not repelled by a wet surface. However, other studies showed that the same species preferred to stay in refuges with 20% RH or even at dryer conditions (Roca & Lazzari 1994, Lorenzo & Lazzari 1999). Infected or healthy insects from control or untreated areas moved later to fungus-treated areas, which explains why populations in the treated areas varied and were not completely eliminated. Fungal application in larger areas would certainly reduce the interfering migration and permit a better quantitative evaluation during field tests.
Stage frequency of triatomine populations in peridomestic habitats varies according to the season and may interfere with the dynamic of the mycosis. Remarkably more elder nymphs and adults than younger instars were observed in the hen houses during the tests performed in January. This corresponds to observations of Forattini et al. (1979) who reported a dispersal activity of T. sordida adults starting between March and April and a clear increase of first instar nymphs between July and August. The low number of individuals of all different stages tested in our study did not allow speculations about a stage-dependent susceptibility of T. sordida to B. bassiana in the field. In the laboratory, susceptibility of R. prolixus to infection with B. bassiana declined with higher nymphal instars (Luz 1994). However, Romaña and Fargues (1992) found an elevated susceptibility of R. prolixus N3 and N5 compared to N1 to B. bassiana.
It is likely that most insects succumbed due to CG 14 inoculation with high doses of conidia and not due to indigenous B. bassiana or M. anisopliae strains. Nevertheless, an interaction between CG 14 and other local strains and their action cannot be excluded, as one isolate found before the treatment showed high similarity to CG 14. Previous tests showed that B. bassiana and M. anisopliae occurred in most of the vector habitats used in this study before application of CG 14 (C Luz, LFN Rocha, and GV Nery unpublished observations).
An elevated number of B. bassiana, all with high similarity to CG 14, when scored by RAPD, was detected 24 h up to one month after fungal application in the substrates or on fungal killed insects. However, not all B. bassiana isolates were analyzed by RAPD. The number of B. bassiana in the substrates collected in the fungus-treated hen houses declined quickly to values found before application. This indicates that persistence of high amounts of CG 14 conidia in the substrates did not exceed one month. Results showed that multiplication of B. bassiana on cadavers in the habitats may occur. However, development and survival of entomopathogenic fungi may be impaired by other saprophytic microorganisms such as fungi and bacteria. B. bassiana showed survival rates of at least three months in dead R. prolixus under laboratory conditions (Luz 1994), and may persist for long periods in cadavers and produce new conidia and propagate fungal infection to healthy insects at favorable conditions. In our study, high fungal inoculum decreased in a few weeks after application to natural levels and must be renewed if necessary. A rapid decrease in conidial populations within 20 days after application of B. bassiana in soils was also reported by Inglis et al. (1997). Integrated control strategies alternating inundative application of fungi in the rainy season and synthetic insecticides in the dry season or a combined application of fungi and insecticides in the rainy season could promote an efficient and permanent reduction of triatomine vectors in peridomestic areas. B. bassiana could contribute to reduce triatomine populations in peridomestic areas and interrupt domestic invasion by secondary vector species in Central Brazil or other regions with prolonged precipitations during the rainy season. Further testing of improved oil-water formulations, application techniques and fungal persistence under field conditions will be useful to consolidate biological control of triatomine vectors.
ACKNOWLEDGEMENTS
To Heloisa Frazão and Nicanor R Silva for the technical support and to the Brazilian National Fund of Health (Funasa) for its collaboration during the field work.
Received 14 August 2003
Accepted 1 March 2004
Financial support: Nacional Council of Scientific and Technological Development (CNPq Centro Oeste). LFNR and GVN were supported by a fellowship from CNPq.
- Al-Samarrai TH, Schmid J 2000. A simple method for extraction of fungal genomic DNA. Letters Appl Microbiol 30: 53-56.
- Chase AR, Osborne LS, Ferguson VM 1986. Selective isolation of the entomopathogenic fungi Beauveria bassiana and Metarhizium anisopliae from an artificial potting medium. Florida Entomol 69: 285-292.
- Fargues J, Luz C 1998. Effects of fluctuating moisture and temperature regimes on sporulation of Beauveria bassiana on cadavers of Rhodnius prolixus Biocontrol Science Technol 8: 323-334.
- Fargues J, Luz C 2000. Effects of fluctuating moisture and temperature regimes on the infection potential of Beauveria bassiana for Rhodnius prolixus J Invertebr Pathol 75: 202-211.
- Forattini OP, Santos JLF, Ferreira AO, Silva EOR, Rabello EX 1979. Aspectos ecológicos da tripanossomíase americana. XVI Dispersão e ciclos anuais de colônias de Triatoma sordida e de Panstrongylus megistus esponataneamente desenvolvidas em ecótopos artificias. Rev Saúde Públ 13: 299-313.
- Inglis GD, Duke GM, Kanagaratnam DL, Johnson DL, Goettel MS 1997. Persistence of Beauveria bassiana in soil following application of conidia through crop canopy. Memoirs Entomol Soc Canada 171: 253-263.
- Lecuona RE, Edelstein JD, Berretta MF, Rossa FR, Argas JA 2001. Evaluation of Beauveria bassiana (Hyphomycetes) strains as potential agents for control of Triatoma infestans (Hemiptera: Reduviidae). J Med Entomol 38: 172-179.
- Lomer CJ, Bateman RP, Johnson DL, Langewald J, Thomas M 2001. Biological control of locusts and grasshoppers. Ann Rev Entomol 46: 667-702.
- Lorenzo MG, Lazzari CR 1999. Temperature and relative humidity affect the selection of shelters by Triatoma infestans, vector of Chagas disease. Acta Trop 72: 241-249.
- Luz C 1990. Zur Pathogenität von Beauveria bassiana (Fungi Imperfecti) gegenüber mehreren Raubwanzenarten (Reduviidae, Triatominae) und Einfluss der relativen Luftfeuchtigkeit auf die Infektion von Rhodnius prolixus Mitt Dtsch Ges Allg Angew Ent 7: 510-511.
- Luz C 1994. Biologische Bekämpfung der Überträger der Chagaskrankheit (Triatominae). Einfluss von Temperatur und Luftfeuchtigkeit auf die larvale Entwicklung von Rhodnius prolixus sowie die Infektion mit Beauveria bassiana (Deuteromycetes) und Sporulation des Pilzes auf den Kadavern, Ph D Thesis, Eberhard Karls Universität Tübingen, Germany, 175 pp.
- Luz C, Fargues J 1998. Factors affecting conidial production of Beauveria bassiana from fungus-killed cadavers of Rhodnius prolixus J Invertebr Pathol 72: 97-103.
- Luz C, Fargues J 1999. Dependence of the entomopathogenic fungus, Beauveria bassiana, on high humidity for infection of Rhodnius prolixus Mycopathologia 146: 33-41.
- Luz C, Silva IG, Cordeiro CMT, Tigano MS 1998b. Beauveria bassiana (Hyphomycetes) as a possible control agent for the vectors of Chagas disease. J Med Entomol 35: 977-979.
- Luz C, Silva IG, Magalhães BP, Cordeiro CMT, Tigano MS 1999. Control of Triatoma infestans (Reduviidae, Tria-tominae) with Beauveria bassiana: preliminary assays on formulation and application in the field. An Soc Entomol Bras 28: 101-110.
- Luz C, Tigano MS, Silva IG, Cordeiro CMT, Aljanabi SM 1998a. Selection of Beauveria bassiana and Metarhizium anisopliae to control Triatoma infestans Mem Inst Oswaldo Cruz 93: 839-846.
- Roca MJ, Lazzari CR 1994. Effects of relative humidity on the haematophagous bug Triatoma infestans: hygropreference and eclosion success. J Insect Physiol 40: 901-907.
- Romaña CA 1992. Recherches sur les Potentialités des Hypho-mycetes Entomopathogènes (Fungi Imperfecti) dans la Lutte contre les Triatomes (Heteroptera), PhD Thesis, Montpellier, France, 129 pp.
- Romaña CA, Fargues J 1987. Sensibilité des larves de l'hemiptère hématophage, Rhodniusprolixus (Triatominae) aux hyphomycètes entomopathogènes. Entomophaga 32: 167-179.
- Romaña CA, Fargues J 1992. Relative susceptibility of different stages of Rhodnius prolixus to the entomopathogenic hyphomycete Beauveria bassiana Mem Inst Oswaldo Cruz 87: 363-368.
- SAS Institute Inc 2000. What's new in SAS ® Software for Release 8.1, Cary, NC.
- Sambrook J, Fritsch EF, Maniatis T 1989. Molecular Cloning: A Laboratory Manual, 2nd ed., Cold Spring Harbor Laboratory Press, New York, 120 pp.
- Silva IG 1985. Influência da temperatura na biologia de triatomíneos. I. Triatoma rubrovaria (Blanchard, 1834) (Hemiptera, Reduviidae). Rev Goiana Med 34: 29-34.
- Tigano MS, Honeycutt RJ, Lacey LA, Assis R, McClelland M, Sobral BWS 1995. Genetic variability of Paecilomyces fumosoroseus isolates revealed by molecular markers. J Invertebr Pathol 65: 274-282.
- Vazquez-Prokopec GM, Ceballos LA, Cecere MC, Gürtler RE 2002. Seasonal variations of microclimatic conditions in domestic and peridomestic habitats of Triatoma infestans in rural northwest Argentina. Acta Trop 84: 229-238.
- Wiesinger D 1956. Die Bedeutung der Umweltfaktoren für den Saugakt von Triatoma infestans Acta Trop8: 97-141.
- Winston PW, Bates DH 1960. Saturated solutions for the control of humidity in biological research. Ecology 41: 232-237.
- Zeledón R, Valerio CE, Valerio JE 1973. The camouflage phenomenon in several species of Triatominae (Hemiptera: Reduviidae). J Med Entomol 10: 209-211.
Publication Dates
-
Publication in this collection
24 June 2004 -
Date of issue
Mar 2004
History
-
Accepted
01 Mar 2004 -
Received
14 Aug 2003