Abstract
Integrons play a role in horizontal acquisition and expression of genes, as well as gene reservoir, contributing for the resistance phenotype, particularly relevant to bacteria of clinical importance. We aimed to determine the composition and the organization of the class 1 integron variable region present in Pseudomonas aeruginosa clinical isolates from Brazil. Strains carrying class 1 integrons were resistant to the majority of antibiotics tested, except to imipenem and ceftazidime. Sequence analysis of the integron variable region revealed the presence of the blaCARB-4 gene into two distinct cassette arrays: aacA4-dhfrXVb-blaCARB-4 and aadB-aacA4-blaCARB-4 . dhfrXVb gene cassette, which is rare in Brazil and in P. aeruginosa species, was found in one isolate. PFGE analysis showed the spread of blaCARB-4 among P. aeruginosa clones. The occurrence of blaCARB-4 and dhfrXVb in Brazil may contribute for developing resistance to clinically important antibiotics, and shows a diversified scenarium of these elements occurring in Amazon clinical settings, where no study about integron dinamycs was performed to date.
dhfr gene cassette; blaCARB-4; class 1 integron; Pseudomonas aeruginosa; Brazilian clinical setting
ARTICLES
Emergence of dhfrXVb and blaCARB-4 gene cassettes in class 1 integrons from clinical Pseudomonas aeruginosa isolated in Amazon region
Érica L FonsecaI, 1 1 Corresponding author. E-mail: ericafon@ioc.fiocruz.br ; Verônica V VieiraII; Rosângela CiprianoIII; Ana CP VicenteI
IDepartamento de Genética, Instituto Oswaldo Cruz-Fiocruz, Av. Brasil 4365, 21040-900 Rio de Janeiro, RJ, Brasil
IIDepartamento de Microbiologia, INCQS-Fiocruz, Rio de Janeiro, RJ, Brasil
IIIUFMA, São Luís, MA, Brasil
ABSTRACT
Integrons play a role in horizontal acquisition and expression of genes, as well as gene reservoir, contributing for the resistance phenotype, particularly relevant to bacteria of clinical importance. We aimed to determine the composition and the organization of the class 1 integron variable region present in Pseudomonas aeruginosa clinical isolates from Brazil. Strains carrying class 1 integrons were resistant to the majority of antibiotics tested, except to imipenem and ceftazidime. Sequence analysis of the integron variable region revealed the presence of the blaCARB-4 gene into two distinct cassette arrays: aacA4-dhfrXVb-blaCARB-4 and aadB-aacA4-blaCARB-4 . dhfrXVb gene cassette, which is rare in Brazil and in P. aeruginosa species, was found in one isolate. PFGE analysis showed the spread of blaCARB-4 among P. aeruginosa clones. The occurrence of blaCARB-4 and dhfrXVb in Brazil may contribute for developing resistance to clinically important antibiotics, and shows a diversified scenarium of these elements occurring in Amazon clinical settings, where no study about integron dinamycs was performed to date.
Key words:dhfr gene cassette - blaCARB-4 - class 1 integron - Pseudomonas aeruginosa - Brazilian clinical setting
Integrons are genetic elements able to capture, excise, and rearrange genes, specially resistance gene cassettes, by a site-specific recombination mechanism, and express them under control of promoter Pant (Stokes & Hall 1989). Therefore, integrons are considered important tools for disseminating resistance among bacteria, acting as gene reservoirs.
The carbenicillin-hydrolysing b-lactamase CARB-4 gene encodes the sulbactam- and clavulanic acid-inhibited enzyme that belongs to Ambler class A b-lactamases, and is able to readily hydrolyse carbenicillin at high levels. A unique report showed the presence of the blaCARB-4 gene in a transposon found in the plasmid pUD12 from the clinical strain of Pseudomonas aeruginosa P83372 isolated in France in the 1980s (Philippon et al. 1986, Sanschagrin et al. 1998). Descriptions of CARB-type genes in South America (Argentina) were restricted to blaCARB-7 and blaCARB-9, both from Vibrio cholerae super-integron (Melano et al. 2002, Petroni et al. 2004).
The major cause of trimethoprim resistance in Gram-negative bacteria is plasmid-born dfr genes, which are usually found in association with integrons (Grape et al. 2003). These genes are dispersed in various enterobacteria species from different geographic areas, mainly from Asian countries (Adrian et al. 1998, Gibreel & Sköld 1998, Poirel et al. 2000, Lee et al. 2001, Thungapathra et al. 2002, Navia et al. 2004, Yu et al. 2004), however, they are rare among P. aeruginosa isolates. The dhfrXVb (dfrA15b) gene cassette only found in a class 1 integron from a clinical Klebsiella pneumoniae isolated in French Guiana (Poirel et al. 2000), was recently identified in a Salmonella enterica subsp. enterica serovar Agona in Brazil (Michael et al. 2005), where dfr alleles have never been found before.
MATERIALS AND METHODS
Bacterial strains and antimicrobial susceptibility - P. aeruginosa isolates were recovered from different specimens of inpatients proceeding from clinical sources between February and October 2001. These strains were obtained from six hospitals in São Luís city, Maranhão, Brazil, and were screened for antibiotic resistance according to NCCLS recommendations (NCCLS 2003).
The MICs of the antibiotics were performed using the VITEK automated system (BioMerieux, Hazelhood, MO), and the MIC of imipenem were determined by the E-test method (AB Biodisk, Solna, Sweden) (Table I).
PCR amplifications and sequencing analysis - PCR reactions were performed using primers targeting the class 1 integrase, the integron cassette region, and the 3' conserved segment of class 1 integrons (Fonseca et al. 2005). The amplicons corresponding to the entire integron cassette regions were directly sequenced on both strands by the primer walking sequence strategy using the BigDye Terminator Cycle Sequencing Kit on an ABI Prism 377 Automated DNA Sequencer.
PFGE genome analysis - Genomic DNA of the four isolates carrying class 1 integrons was digested with XbaI and SpeI restriction enzymes. PFGE was performed using a Gene Navigator Pulsed Field System (Amersham). The l DNA-PFGE marker (Amersham) was used as size marker. PFGE banding patterns were analyzed and compared visually. Isolates were considered to be clonal when the macrorestriction DNA patterns differed by fewer than three bands (Tenover et al. 1995).
Nucleotide sequence accession numbers - The GenBank accession numbers of the class 1 integrons from isolates 83, 182, and 262, and from isolate 158 are AY913771 and AY913772, respectively.
RESULTS
The strains positive for the presence of class 1 integrons were resistant to all antibiotics tested, except for ceftazidime. PS 83, 158, and 182 were also susceptible to imipenem (MIC, 0,75 and 1 µg/ml, respectively), while PS 262 showed resistance to this antibiotic (MIC, > 32 µg/ml) (Table I).
Sequence analysis of the integron variable regions revealed that PS 83, 182, and 262 harboured a class 1 integron with the same cassette composition and arrangement: aadB, aacA4 and blaCARB-4 (Fig. 1A), while PS 158 presented a class 1 integron carrying aacA4, dhfrXVb, and blaCARB-4 (Fig. 1B). dhfrXVb was identical to that previously reported in a integron found in a K. pneumoniae strain from French Guiana (Poirel et al. 2000), including the non-coding regions, but presented an amino acid substitution compared with the dfrA15b identified in a Brazilian S. Agona isolate (Michael et al. 2005). All signatures of class 1 integrons, which include the integrase gene (intI1), the recombination site (attI), the promoter region (Pant) and the qacED1 and sulI genes of 3' CS, were found in the four integrons analyzed. The promoter Pant seemed to be identical to a weak version described previously (Lévesque et al. 1994, Collis & Hall 1995) with a single point mutation in regions 35 and 10 (Fig. 1A).
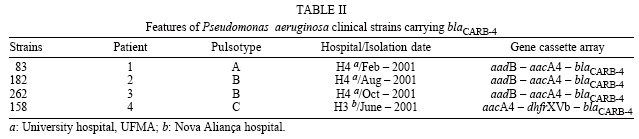
Despite the identical integron composition and similar susceptibility profiles, the PFGE analysis revealed that isolate 83 is distinct from 182 and 262, which belong to a unique clone. Isolate 158 also presented a unique and distinct PFGE profile (Fig. 2, Table II).
DISCUSSION
The blaCARB-4 gene sequence had been previously identified as part of a gene cluster present in a plasmid from a French P. aeruginosa clinical strain. In this study, we characterized the blaCARB-4 as a gene cassette widespread in functional class 1 integrons disseminated in hospitals from the Brazilian Amazon region. Although CARB genes have already been observed in association with class 1 integrons, the majority of these genes were not found in resistance gene cassette arrays (Petroni et al. 2004). Here, the blaCARB-4 gene cassette was identified in a class 1 integron genetic environment distinct from that characterized by Sanschagrin et al. (1998) in France. Four versions of promoters exhibiting different strengths were identified in class 1 integrons (Lévesque et al. 1994). Despite of the presence of a weak promoter in the Brazilian integrons, all strains presented high levels of resistance to b-lactams and amikacin (Table I). The lack of b-lactamase inhibitors activity plus the imipenem resistance may indicate that this phenotype is consequence of additional resistance mechanisms present in the analyzed strains, such as overexpression of AmpC b-lactamase, efflux pump systems and/or membrane impermeability (Livermore 2002), and not of the blaCARB-4 expression. This hypothesis was also reinforced by the fact that clonal strains (PS 182 and PS 262) presented differences between their resistance profile. In fact, blaCARB-4 not only hydrolyses carbenicillin, but also other b-lactams like oxacillin and piperacillin, which are used for treating infections caused by Staphylococcus aureus and P. aeruginosa. Therefore, the occurrence of this gene cassette in Brazil may contribute for developing resistance to clinically important b-lactams.
The PFGE results indicated the spread of a clone among patients, as well as the transmission and/or selection of an integron carrying the aadB-aacA4-blaCARB-4 gene cassette arrayamong non-related isolates. Moreover, the identification of blaCARB-4 in class 1 integrons harbouring different cassette array compositions (aacA4-dhfrXVb-blaCARB-4 and aadB-aacA4-blaCARB-4 ) among P. aeruginosa clones is a strong evidence of its mobility and, therefore, of the possibility of its spread to other genera, as has been demonstrated to occur with various gene cassettes.
In this work we also reported the emergence of the dhfrXVb (dfrA15b) allele in a Brazilian P. aeruginosa isolate, which is the first description of a dfr gene cassette in this species in all South America. Despite of the Pant weak configuration, PS 158 displayed high levels of resistance to trimethoprim. In fact, Dubois et al. (2002) have found a P. aeruginosa strain highly resistant to b-lactams, which harboured a class 1 integron containing a weak promoter version and an ESBL gene cassette.
The emergence of blaCARB-4 and dhfrXVb in Brazil and in P. aeruginosa isolates demonstrates, once more, the dynamics and the mobility of gene cassettes. Moreover we showed a considerable gene cassette diversity occurring in Amazon clinical settings, where no previously study focusing this issue had been performed.
ACKNOWLEDGEMENTS
To Anna Beatriz Robottom for the English review.
Received 21 October 2005
Accepted 1 December 2005
Financial support: Capes, Instituto Oswaldo Cruz
- Adrian PV, du Plessis M, Klugman KP, Amys SGB 1998. New trimethoprim-resistance dihydrofolate reductase cassette, dfrXV, inserted in a class 1 integron. Antimicrob Agents Chemother 42: 2221-2224.
- Collis CM, Hall RM 1995. Expression of antibiotic resistance genes in the integrated cassettes of integrons. Antimicrob Agents Chemother 39: 155-162.
- Dubois V, Poirel L, Marie C, Arpin C, Nordmann P, Quentin C 2002. Molecular characterization of a novel class 1 integron containing blaGES-1 and a fused product of aac(3)-Ib/aac(6')-Ib' gene cassettes in Pseudomonas aeruginosa Antimicrob Agents Chemother 46: 638-645.
- Fonseca EL, Vieira VV, Cipriano R, Vicente AC 2005. Class 1 integrons in Pseudomonas aeruginosa isolates from clinical settings in Amazon region, Brazil. FEMS Immunol Med Microbiol 44: 303-309.
- Gibreel A, Sköld O 1998. High-level resistance to trimethoprim in clinical isolates of Campylobacter jejuni by acquisition of foreign genes (dfr1 and dfr9) expressing drug-resistance dihydrofolate reductases. Antimicrob Agents Chemother 42: 3059-3064.
- Grape M, Sundström L, Kronvall G 2003. New dfr2 gene as a single-gene cassette in a class 1 integron from trimethoprim-resistant Escherichia coli isolate. Microb Drug Resist 9: 317-322.
- Lee JC, Oh JY, Cho JW, Park JC, Kim JM, Seol SY, Cho DT 2001. The prevalence of trimethoprim-resistance-confering dihydrofolate reductase genes in urinary isolates of Escherichia coli in Korea. J Antimicrob Chemother 47: 599-604.
- Lévesque C, Brassard S, Lapointe J, Roy PH 1994. Diversity and relative strength of tandem promoters for the antibiotic-resistance genes of several integrons. Gene 142: 49-54.
- Livermore DM 2002. Multiple mechanisms of antimicrobial resistance in Pseudomonas aeruginosa: our worst nightmare? Clin Infect Dis 34: 634-640.
- Melano R, Petroni A, Garutti A, Saka HA, Mange L, Pasteran F, Rapoport M, Rossi A, Galas M 2002. New carbenicillin-hydrolysing b-lactamase (CARB-7) from Vibrio cholerae non-O1, non-O139 strains encoded by the VCR region of the V. cholerae genome. Antimicrob Agents Chemother 46: 2162-2168.
- Michael GB, Cardoso M, Schwarz S 2005. Class 1-associated gene cassettes in Salmonella enterica subsp. enterica serovar Agona isolated from pig carcasses in Brazil. J Antimicrob Chemother 55: 776-779.
- Navia MM, Ruiz J, Vila J 2004. Molecular characterization of the integrons in Shigella strains isolated from patients with traveler's diarrhea. Diagn Microbiol Infect Dis 48: 175-179.
- NCCLS-National Committee for Clinical Laboratory Standards 2003. Methods for dilution antimicrobial susceptibility tests for bacteria that grow aerobically, 5th ed., Document M7-A5, Wayne, PA.
- Petroni A, Melano RG, Saka HA, Garutti A, Mange L, Pasteran F, Rapoport M, Miranda M, Faccone D, Rossi A, Hoffman PS, Galas MF 2004. CARB-9, a carbenicillinase encoded in the VCR region of Vibrio cholerae Non-O1, Non-O139 belongs to a family of cassette-encoded b-lactamases. Antimicrob Agents Chemother 48: 4042-4046.
- Philippon AM, Paul GC, Thabaut AP, Jacoby GA 1986. Properties of a novel carbenicillin-hydrolyzing b-lactamase (CARB-4) specified by an IncP-2 plasmid from Pseudomonas aeruginosa Antimicrob Agents Chemother 29: 519-520.
- Poirel L, Le Thomas I, Naas T, Karim A, Nordmann P 2000. Biochemical sequence analyses of GES-1, a novel Class A extended-spectrum b-lactamase, and the class 1 integron In52 from Klebsiella pneumoniae Antimicrob Agents Chemother 44: 622-632.
- Sanschagrin F, Bejaoui N, Levesque RC 1998. Structure of CARB-4 and AER-1 carbenicillin-hydrolyzing b-lactamases. Antimicrob Agents Chemother 42: 1966-1972.
- Stokes HW, Hall RM 1989. A novel family of potentially mobile DNA elements encoding site-specific gene-integration functions: integrons. Mol Microbiol 3: 1669-1683.
- Tenover FC, Arbeit RD, Goering RV, Mickelsen PA, Murray BE, Persing DH, Swaminathan B 1995. Interpreting chromosomal DNA restriction patterns produced by pulsed-field gel electrophoresis: criteria for bacterial strain typing. J Clin Microbiol 33: 2233-2239.
- Thungapathra M, Amita, Sinha KK, Chaudhuri SR, Garg P, Ramamurthy T, Nair GB, Gosh A 2002. Occurrence of antibiotic resistance gene cassettes aac(6')-Ib, dfrA5, dfrA12, and ereA2 in class 1 integrons in non-O1, non-O139 Vibrio cholerae strains in India. Antimicrob Agents Chemother 46: 2948-2955.
- Yu HS, Lee JC, Kang HY, Jeong YS, Lee EY, Choi CH, Tae SH, Lee YC, Seol SY, Cho DT 2004. Prevalence of dfr genes associated with integrons and dissemination of dfrA17 among urinary isolates of Escherichia coli in Korea. J Antimicrob Chemother 53: 445-450.
Publication Dates
-
Publication in this collection
12 May 2006 -
Date of issue
Feb 2006
History
-
Accepted
01 Dec 2005 -
Received
21 Oct 2005