Abstract
We have been able to label the excretory system of cercariae and all forms of schistosomula, immature and adult worms with the highly fluorescent dye resorufin. We have shown that the accumulation of the resorufin into the excretory tubules and collecting ducts of the male adult worm depends on the presence of extracellular calcium and phosphate ions. In the adult male worms, praziquantel (PZQ) prevents this accumulation in RPMI medium and disperses resorufin from tubules which have been prelabelled. Female worms and all other developmental stages are much less affected either by the presence of calcium and phosphate ions, or the disruption caused by PZQ. The male can inhibit the excretory system in paired female. Fluorescent PZQ localises in the posterior gut (intestine) region of the male adult worm, but not in the excretory system, except for the anionic carboxy fluorescein derivative of PZQ, which may be excreted by this route. All stages of the parasite can recover from damage by PZQ treatment in vitro. The excretory system is highly sensitive to damage to the surface membrane and may be involved in vesicle movement and damage repair processes. In vivo the adult parasite does not recover from PZQ treatment, but what is inhibiting recovery is unknown, but likely to be related to immune effector molecules.
Schistosoma mansoni; praziquantel; excretory system; resorufin; P-glycoproteins; mode of action
The effects of drugs, ions, and poly-l-lysine on the excretory system of Schistosoma mansoni
JR KuselIV,1 1 Corresponding author: johnkusel@hotmail.com ; FA OliveiraI; M ToddIII; F RonkettiII; SF LimaI; ACA MattosI; KT ReisI; PMZ CoelhoI; JA ThornhillIII; F RibeiroI
ICentro de Pesquisas René Rachou-Fiocruz, Av. Augusto de Lima 1715, 39190-002 Belo Horizonte, MG, Brasil
IIQueen Mary College, University of London, London, UK
IIIUniversity of Sydney, Sydney, Australia
IVGlasgow University, Glasgow, Scotland
ABSTRACT
We have been able to label the excretory system of cercariae and all forms of schistosomula, immature and adult worms with the highly fluorescent dye resorufin. We have shown that the accumulation of the resorufin into the excretory tubules and collecting ducts of the male adult worm depends on the presence of extracellular calcium and phosphate ions. In the adult male worms, praziquantel (PZQ) prevents this accumulation in RPMI medium and disperses resorufin from tubules which have been prelabelled. Female worms and all other developmental stages are much less affected either by the presence of calcium and phosphate ions, or the disruption caused by PZQ. The male can inhibit the excretory system in paired female. Fluorescent PZQ localises in the posterior gut (intestine) region of the male adult worm, but not in the excretory system, except for the anionic carboxy fluorescein derivative of PZQ, which may be excreted by this route. All stages of the parasite can recover from damage by PZQ treatment in vitro. The excretory system is highly sensitive to damage to the surface membrane and may be involved in vesicle movement and damage repair processes. In vivo the adult parasite does not recover from PZQ treatment, but what is inhibiting recovery is unknown, but likely to be related to immune effector molecules.
Key words:Schistosoma mansoni - praziquantel - excretory system - resorufin - P-glycoproteins - mode of action
The excretory system of the schistosome consists of numerous distal flame cells connected to tubules leading to connecting ducts which lead to the exterior through a protonephridial pore at the posterior of the worm. The larval stages have a much simplified system, but it is based on this basic form (Wilson & Webster 1972).
The excretory system of the adult male and female Schistosoma mansoni has been well described morphologically by confocal microscopy, using antibodies (Bogers et al. 1994) but its function under different conditions is not understood, although recent publications have indicated the presence in the excretory system of signalling molecules which may be of importance both in the response of the parasite to the host, and to the interactions between male and female worms (Finken et al. 1994). Mecozzi et al. (2000) have demonstrated the localisation of calcineurin to the flame cells of the schistosomula and adult worms and Schechtman et al. (2001) have found 14-3-3 protein in the protonephridial system of the female worm.
We have shown that the excretory system of S. mansoni functions with the aid of P-glycoproteins (PgP) and multiresistance proteins (MRP) to pump out certain fluorescent substrates (Sato et al. 2002, 2004). These groups of proteins are often overexpressed in drug resistant tumour cells and function to reduce the concentration of cytotoxic drugs in the cytoplasm of these cells. These proteins belong to a large family of ABC transporters which have numerous functions in normal eukaryotic and prokaryotic cells. They can transport inorganic and organic ions, xenobiotics, a variety of hydrophobic compounds, peptides, lipids, and proteins (Dahl et al. 2004, Stavrovskaya 2005). The PgP in eukaryotes are 170 kDa membrane proteins, having 12 transmembrane domains and a cytoplasmic nucleotide binding domains in the molecule (Dahl et al. 2004). PgP can be regulated in their amount and activity by a variety of environmental agents but the response depends on the cell type or tissue. For example, reactive oxygen species can up or down regulate P glycoprotein activity depending on the tissue and context (Sukai & Piquette-Miller 2000, Stavrovskaya 2005). The MRP have a molecular weight of 190 kDa, and can pump organic anions, cations, and compounds complexed with or co-transported with glutathione, glucuronic acid, and other adducts. PgP have been detected in schistosomes (Bosch & Shoemaker 1994).
In this paper we describe the responses of the excretory system to a number of compounds in an attempt to understand the regulation of the excretory system, and the role it might play in the biology of the parasite.
MATERIALS AND METHODS
Reagents - Resorufin, Hoechst 33258, poly-l-lysine (Molecular weight 84kDa), rhodamine labelled Glycine max lectin and beta methyl cyclodextrin were obtained from Sigma Chemical Co. St. Louis, MO, US. Praziquantel + enantiomer was a kind gift from Dr Achim Harder, Sodium penthobarbital "Hypnol" (Merck, Sharp and Dohme, Inc.) was used as an anaesthetic for both mice and parasite. Texas red bovine serum albumin (Texas Red BSA) was obtained from Molecular Probes, Eugene, Oregon, US.
Life cycle of parasite and infection of animals - Swiss mice were infected with 100 cercariae of S. mansoni by the method of Smithers and Terry (1965). Cercariae were of the LE strain (Belo Horizonte) supplied by the Malacology Division of the CPqRR. Schistosomula were produced from cercariae by the method of Colley and Wikel (1974). Lung stage and immature worms were obtained as follows: Swiss mice were infected with 500 cercariae and 5 days after infection the animals were perfused to remove blood from the lungs, which were excised and chopped into pieces with a scalpel, after which they were incubated in RPMI for 2 h to allow the lung worms to migrate from the lung chop. Similar infections (500 cercariae per mouse) yielded 3 week or 4 week immature parasites, which were obtained by perfusion at these times after infection.
Labelling of excretory system of adult worms with resorufin and incubation with various effectors - Adult worms were perfused by the method of Smithers and Terry (1965) using RPMI and added heparin as the perfusing fluid. Worms were washed in RPMI and 5% bovine foetal calf serum (FCS), and divided into Petri dishes (5 cm diameter) in 2 ml RPMI medium and 5% bovine serum albumin. Additions of drugs or effector compounds were made to the Petri dish after the addition of the worms (5 pairs per Petri dish). After incubation of the worms in the Petri dishes, 25 µg in 10 µl of resorufin in methanol was added and incubated for a further 30 min to label the excretory system. Worms were then washed gently five times with 2 ml RPMI and placed on a microscope slide within a vaseline surround. To prevent movement of the worms when photography was required, a solution of Hypnol (MSD) (10% in RPMI) was added to the slide (50 µl RPMI, and an equal volume of Hypnol solution). The slides were then examined under the rhodamine filter of a Leitz fluorescence microscope. In some experiments the worms were labelled first and then treated with PZQ and other compounds.
Growth medium without calcium ions and phosphate ions - To examine the effect of calcium and phosphate ions on excretory activity of immature and mature male and female worms, a medium was used which allowed maintenance of the parasites in the absence of these ions. The medium composition was: 116 mM NaCl, 5 mM KCl, 5.5 mM glucose, 20 mM sodium bicarbonate, 0.8 mM MgSO4. No calcium or phosphate ions but add 2 mM EGTA.
Treatments with PZQ - To enable us to quantify the immediate response to these compounds, adult worms were perfused by a modification of the method of Smithers and Terry (1965). We found that when worms left the mouse during perfusion they could be damaged if allowed to be collected on a plastic gauze, as is routine in this method. To avoid this damage the worms were perfused into a Petri dish containing 60 mg/ml bovine serum albumin. These worms were then transferred to another Petri dish with a plastic tipped pipette and were incubated in medium (1 h at 37ºC) with the addition of 1.5 µg/ml resorufin and 10 µl of 1mg/ml Hoechst 33258 to assess damage to worms (Lima et al. 1994). This lower concentration of resorufin yielded a less pronounced labelling of tubules but allowed any activation or inhibition to be assessed more readily. Worms were washed very gently and placed singly within a vaseline circle on a microscope slide, four rings per slide. This arrangement allowed us to observe the fluorescent labelling of the worms and to record any damage before adding the effector molecules. Worms observed to be damaged were not used in the subsequent experiment. The reagent in growth medium was added to the worms in the vaseline circles, but without the addition of a coverslip or Hypnol. This obviated the development of anaerobiosis around the worms and allowed a more prolonged observation of the resorufin distribution (reduced resorufin is colourless). The activity of the excretory system was observed over a 30 min period and was scored by the method of Sato et al. (2002).
Recording and quantification of labelling of excretory system - The tubules and branches of the excretory system were counted under the ´ 10 objective in the head, mid-body region and tail at different times after mounting the worms, and a system identical to that described in Sato et al. (2002) was used to evaluate the significance of the differences in excretory activity. Photographic records were also taken with a digital camera (Cannon Rebel, model EOS 300D).
Detection of surface membrane damage by fluorescent probes - Detection of damage: methods for detecting damage have been described by Lima et al. (1994). Briefly, after treatment with the relevant compound, and subsequent washing, worms were incubated for 1 h with 10 µ of Alexa fluor phalloidin (50 µg/ml), rhodamine Soy bean (Glycine max) lectin (5 µg/ml) and Hoechst 33258. Damage was assessed by fluorescence microscopy after washing the worms to remove the probes. Recovery from damage: to investigate the repair of surface membranes damaged by PZQ, adult worm pairs, treated with PZQ for 15 min at 37ºC were divided into two portions, one portion was stained with the fluorescent probes and the other portion incubated for 2 h at 37ºC and then stained with the fluorescent probes. Incubated worms exhibited minimal staining.
Alternatively, individual worm pairs, whose damage was detected by fluorescent Hoechst 33258 patches on the body, and head were shown to be permeable in these regions to Texas-red BSA (10 µg/ml) incubated with the damaged parasite (Wippersteg et al. 2003). To detect repair of these regions worm pairs were incubated for 30 min subsequent to the 2 h at 37ºC with the Texas-red BSA. Repair was indicated by a lack of permeability to Texas-red BSA in the Hoechst stained region.
Treatment of adult worms with PZQ - In vitro: PZQ (isomer plus or commercial mixture (+/) in concentrations ranging from 0.1 to 5 µg/ ml, were added to Petri dishes containing 5 pairs of adult worms. In vivo: infected mice were treated through stomach tube with 400 mg/ kg PZQ in distilled water. Mice were perfused 2 h after treatment so as to compare with the in vitro treatment.
Synthesis of fluorescent PZQ - Three compounds were used in this study: rhodamine labelled PZQ (Rh-PZQ), fluorescein PZQ (F-PZQ), and carboxy fluorescein PZQ (CF-PZQ). These were synthesised using the following method: Fluorescently-tagged PZQ was synthesised from commercial PZQ according to the scheme bellow. Amino PZQ (3) was conjugated with fluorescein, carboxyfluo-rescein and rhodamine B under standard amide bond forming conditions. The nitration method was from Schofield (1980).
RESULTS
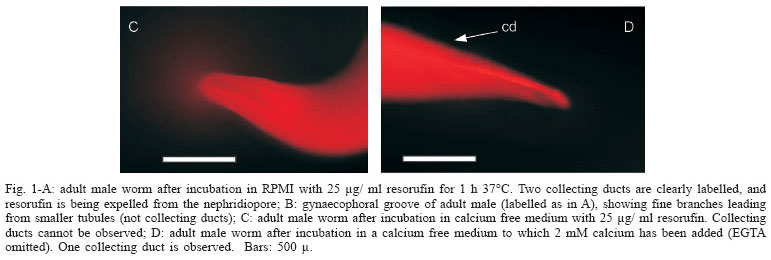
Incubation of parasites in RPMI medium - Adult (8 weeks old), immature forms (fresh schistosomula to 4 week worms) and cercariae were incubated for 1 h 37ºC with 25 µg/ ml resorufin. The results for adult worms, after extensive washing showed strong labelling of the collective ducts, and strong labelling of the very numerous small branches attached to the collecting ducts (Fig. 1A, B). As described in Oliveira et al. (2006), the paired female worms are rarely labelled in their excretory system, while unpaired females are very heavily labelled in the collecting ducts. Expulsion of resorufin was readily observed from the nephridiopore during observation of the labelled parasites.
Cercariae and fresh schistosomula showed little activity in the excretory system. Most of the resorufin accumulated in the pre-and postacetabular glands of cercariae and newly transformed (fresh) schistosomula (Fig. 3) although in some schistosomula the excretory system was well labelled. Lung stage, three and four week parasites showed good accumulation of resorufin in the excretory tubules. The collecting ducts are not well developed in the immature forms, and most of the strong labelling is seen in the tubules observed by ´40 objective. No release of resorufin from labelled fresh, or lung schistosomula, or from 3-4 week immature worms was observed.
Incubation in medium without calcium and phosphate ions - When adult (8 weeks old) worms were incubated in medium without calcium and phosphate ions (25 µg/ml resorufin, 37ºC), most of the male worms showed no accumulation of resorufin, either in the collecting ducts or in the numerous small tubules and branches (Fig. 1C). Female worms paired or unpaired, showed labelling in the collecting ducts and had not been affected as had the male worms. Tubules of immature forms also were labelled in the presence of resorufin in the medium lacking calcium and phosphate.
When male worms were incubated with resorufin in this medium to which 2 mM calcium was added (and no EGTA), but which still lacked phosphate, the collecting ducts were labelled but not to the extent seen in RPMI (Fig. 1D). When phosphate ions (10 mM) were added to the calcium enriched medium, labelling of collecting ducts and small branches were restored to the levels approaching that seen in RPMI.
These observations suggest that for the excretory system of the adult male to function normally, calcium and phosphate ions must be present. A possible mechanism by which the action of the excretory system might be controlled is described in the Discussion.
Activation of the excretory system by damage - Entry of Texas-red BSA into damaged regions of adult worm, and increased resorufin uptake. Oliveira et al. (2006) described the damage to the surface membrane of adult parasites during perfusion, or after poly-l-lysine treatment. General activation of the uptake of resorufin by the excretory system was observed and, at the site of damage, increased numbers of highly labelled small branches could be observed. Ribeiro (unpublished observations) and Wippersteg et al. (2003) has shown an uptake of fluorescent protein (Texas-red BSA) at the site of damage (Fig. 2A,B). This may be due to an increase in endocytosis by the epithelial cells of the excretory system as is shown in schistosomula (Tan et al. 2004) (Fig. 2C).
Damage can also be measured after PZQ treatment, but this is not accompanied by increased endocytosis activity perhaps due to an excess of calcium ions in the region of epithelial cells. PZQ induced damage does not show an increase in excretory activity, probably for the same reason. An excess in calcium ions inhibits the excretory activity.
Effect of PZQ on excretory activity of immature and mature adult male and female worms - PZQ (1 µg/ml) applied for 15 min in RPMI causes total inhibition of the uptake of resorufin in male adult worms. The excretory system of female worms is much less sensitive to PZQ than that of the male. Indeed, the excretory system of the immature worms, are similar to the female adult worm in their insensitivity to PZQ. Adult male worms, after treatment with PZQ, can recover from membrane damage and from inhibition of the excretory system (Oliveira et al. 2006) possibly by reducing the level of free calcium ions in the cells and their associated tegumental cytoplasm.
Schistosomula and cercariae have an uptake of resorufin which is different from the later stages of growth. Resorufin enters the pre-and post-acetabular glands in an energy dependent process, and is transferred to what may be lysosomal vesicles in the region of the nephri-diopore within 2 h (Fig. 3). Little involvement of the excretory system in most schistosomula is observed in these early stages of development in the mammal. PZQ can act to disrupt some of the lysosomal vesicles close to the nephridiopore and delay transfer from the acetabular glands to the lysosomes but no clear effect other than inhibition of the transfer of the dye to lysosomal vesicles within the organism could be ascribed to the effect of PZQ.
Localisation of fluorescent PZQ rhodamine (R-PZQ), fluorescein F-PZQ, and carboxyfluorescein (CF-PZQ) - In adult worms treated with fluorescent PZQ for 15 min in RPMI (1 µg/ml) all forms of fluorescent PZQ stained most regions of the body and had no clear intracellular localisation. The Rh-PZQ became localised in the gut (intestine) as did F-PZQ. Rh-PZQ and F-PZQ never were seen in the excretory system of male or female worms. Fig. 2 shows the manner in which the R-PZQ localises in the gut (intestine). This is very similar to the localisation shown by Texas red bovine serum albumin (Fig. 2). CF-PZQ often appeared in the excretory system, of some worms but this localisation varied from experiment to experiment.
The localisation of the fluorescent PZQ to the gut (intestine) is similar to the situation for BoDipy-verapamil (BD-verapamil, a fluorescent verapamil derivative). Verapamil is a potent inhibitor of PgP. BD-verapamil does not localise in the excretory system, neither does pira-rubicin (Pereira-Maia, pers. commun.), both these being substrates of PgP and might be expected to be excreted by the schistosome excretory system, which is rich in PgP (Sato et al. 2002, 2004). Hence the result with the R-PZQ and F-PZQ suggests that PZQ may be excreted via the gut. The result with CF-PZQ suggests that with an extra negative charge possessed by CF-PZQ, this may determine its selection by the excretory system rather than the gut for excretion.
DISCUSSION
The ability of the highly fluorescent dye resorufin to label the excretory system of the schistosome in RPMI medium is a valuable assay for its activity and is thought to be due to the PgP pumps in the membranes of the excretory epithelia (Sato et al. 2002, 2004). The labelling with resorufin of cercariae, fresh schistosomula, lung schistosomula and 3-4 week immature worms showed accumulation of the dye in all forms, but expulsion of the dye from the nephridiopore was never seen in our experiments. It is possible that the excretory system in all these stages is not able to expel its contents.
Our work with RPMI and medium depleted in calcium and phosphate ions showed (Fig. 1A, B, C, D) that the male adult excretory system is very dependent upon the presence of external calcium and phosphate ions. We have not yet determined the relationship between ion concentration and activity but we can speculate that the surface membrane of the male "senses" the presence of these ions, using signalling mechanisms involving surface calcium and phosphate receptors or pumps rather similar to the extracellular calcium receptor of parathyroid cell membranes which is a GPCR and signals through phospholipase C (Hauache 2001). The extracellular calcium levels may regulate other processes related to the cytoskeleton (Brunig et al. 2004) as well as the activity of PgP (Axiotis et al. 1995).
The crucial role of calcium may explain the dramatic inhibition of the activity of the excretory system of adult male worms, but the lack of effect on female worms and immature stages is not understood. It appears that the latter forms are very insensitive to changes in calcium concentrations. It may be that the female excretory system is under a different kind of regulation, since the male can inhibit the activity of the paired female worm but the unpaired worm is very active (Oliveira et al. 2006).
The localisation of PZQ in the parasite was studied by using fluorescent PZQ. There is a danger in the use of such molecules since the fluorophore is large and may confer certain properties on the molecule not found in PZQ itself. Nevertheless, the fate of these molecules gave some exciting results, in that their passage to the posterior region of the gut (intestine) of the adult male worm (Fig. 2) was also seen with other fluorescent drugs, such as verapamil.
The schistosomula and cercariae were not well labelled, but the compound resorufin showed concentration in the acetabular glands (Fig. 2), and sometimes fluorescent PZQ has been observed to be localised here also. In 3 week and 4 week stage worms no clear localisation of R-PZQ or F-PZQ could be detected but at 4 weeks, CF-PZQ could sometimes be found in the excretory system.
The 3-4 week worms were acted upon by PZQ but the degree of contraction was much less than in mature forms and the excretory tubules were very much less affected. It seems that these immature forms have an excretory system similar in properties to the adult female worm. It is not known what the differences are in enzymic or pump properties or membrane composition between the forms but the differences may give a clue to special metabolic properties of immature and female worms, and confer different susceptibilities and rates of recovery to drugs such as PZQ.
ACKNOWLEDGEMENTS
To Capes for a Bursary to John Robert Kusel in their PVE program and Neusa Araujo, Aureo Almeida, Vera Ribeiro, and Ana Karine Sarvel for help and technical support.
Received 25 May 2006
Accepted 26 June 2006
- Axiotis CA, Bani D, Bianchi S, Pioli P, Tanini A, Brandi ML 1995. P-glycoprotein is expressed in parathyroid epithelium and is regulated by calcium. Calciferous Tissue Int 56: 170-174.
- Bogers JJ, Nibbeling HA, van Marck EA, Deelder AM 1994. Immunofluorescent visualisation of the excretory and gut system of Schistosoma mansoni by confocal laser scanning microscopy. Am J Trop Med Hyg 50: 612-619.
- Borst P, Zelcer N, van Helvoort A 2000. ABC transporters in lipid transport. Bioch Bioph Acta 1486: 128-144.
- Bosch IB, Wang ZX, Tao LF, Shoemaker CB 1994. Two Schistosoma mansoni cDNA's encoding ATP-binding cassette (ABC) family proteins. Mol Biochem Parasitol 65: 351-356.
- Brunig I, Kaech S, Brinkhaus H, Oertner TG, Matus A 2004. Influx of extracellular Calcium regulates actin dependent morphological plasticity in dendritic spines. Neuropharmacology 47: 669-676.
- Dahl SG, Sylte I, Ravna AW 2004. Structures and models of transporter proteins. J Pharm Exp Ther 309: 853-860.
- Finken M, Sobek A, Symmons, P, Kunz W 1994. Characterisation of the complete protein disulfide isomerase gene of Schistosoma mansoni and identification of the tissues of its expression. Mol Biochem Parasitol 64: 135-144.
- Hauache OM 2001. Extracellular sensing calcium receptor: structural and functional features and association with diseases. Braz J Med Biol Res 34: 577-584.
- Lima SF, Vieira LQ, Harder A, Kusel JR 1994. Altered behaviour of carbohydrate-bound molecules and lipids in areas of the tegument of adult Schistosoma mansoni worms damaged by praziquantel. Parasitology 109: 469-477.
- Mecozzi B, Rossi A, Lazzaretti P, Kady M, Kaiser S, Valle C, Cioli D, Klinkert Mo-Q 2000. Molecular cloning of Schistosoma mansoni calcineurin subunits and immuno-localisation to the excretory system. Mol Biochem Parasitol 110: 333-343.
- Oliveira FA, Ribeiro F, Kusel JR, Coelho PMZ 2005. The response of the excretory system of adult Schistosoma mansoni to praziquantel and biomolecules. Parasitology 132: 1-10.
- Sato H, Kusel JR, Thornhill JA 2002. Functional visualisation of the excretory system of adult Schistosoma mansoni by the fluorescent marker resorufin. Parasitology 125: 527-535.
- Sato H, Kusel JR, Thornhill JA 2004. Excretion of fluorescent substrates of mammalian multidrug resistance associated protein (MRP) in the Schistosoma mansoni excretory system. Parasitology 128: 43-55.
- Schechtman D, Winnen R, Tarrab-Hazdai R, Ram D, Schinder V, Grevelding CG, Kunz W, Arnon R 2001. Expression and immunolocalization of the 14-3-3 protein of Schistosoma mansoni Parasitology 123: 573-582.
- Schofield K 1980. Aromatic Nitration, Cambridge University Press, Cambridge, p. 1.
- Skelly PJ, Shoemaker CB 2001. Schistosoma mansoni proteases Sm 31 (cathepsin B) and Sm32 (legumain) are expressed in the caecum and protonephridia of cercariae. J Parasitol 87: 1218-1221.
- Smithers SR, Terry RJ 1964. The infection of laboratory hosts with cercariae of Schistosoma mansoni and the recovery of the adult worms. Parasitology 55: 695-700.
- Stavrovskaya AA 2000. Cellular Mechanisms of multidrug resistance of tumour cells. Biochemistry 65: 95-106.
- Sukai M, Piquette-Miller M 2000. Regulation of multidrug resistance genes by stress signals. J Pharmacol Pharmac 3: 268-280.
- Tan H, Thornhill JA, Al-Adhami BH, Akhkha A, Kusel JR 2003. A study of the effect of surface damage on the uptake of Texas Red BSA by schistosomula of Schistosoma mansoni Parasitology 126: 235-240.
- Wilson RA, Webster LA 1974. Protonephridia. Biol Reviews 49: 127-160.
- Wippersteg V, Ribeiro F, Liedke S, Kusel JR, Grevelding CG 2003.The uptake of Texas Red BSA in the excretory system of schistosomes and its colocalisation with ER60 promoter induced GFP in transiently transformed adult males. Int J Parasitol 33: 1139-1143.
Publication Dates
-
Publication in this collection
12 Feb 2007 -
Date of issue
Oct 2006
History
-
Accepted
26 June 2006 -
Received
25 May 2006