Abstract
This study investigated rickettsial infection in animals, humans, ticks, and fleas collected in five areas of the state of São Paulo. Eight flea species (Adoratopsylla antiquorum antiquorum, Ctenocephalides felis felis, Polygenis atopus, Polygenis rimatus, Polygenis roberti roberti, Polygenis tripus, Rhopalopsyllus lugubris, and Rhopalopsyllus lutzi lutzi), and five tick species (Amblyomma aureolatum, Amblyomma cajennense, Amblyomma dubitatum, Ixodes loricatus, and Rhipicephalus sanguineus) were collected from dogs, cats, and opossums. Rickettsia felis was the only rickettsia found infecting fleas, whereas Rickettsia bellii was the only agent infecting ticks, but no animal or human blood was shown to contain rickettsial DNA. Testing animal and human sera by indirect immunofluorescence assay against four rickettsia antigens (R. rickettsii, R. parkeri, R. felis, and R. bellii), some opossum, dog, horse, and human sera reacted to R. rickettsii with titers at least four-fold higher than to the other three rickettsial antigens. These sera were considered to have a predominant antibody response to R. rickettsii. Using the same criteria, opossum, dog, and horse sera showed predominant antibody response to R. parkeri or a very closely related genotype. Our serological results suggest that both R. rickettsii and R. parkeri infected animals and/or humans in the studied areas.
Rickettsia; spotted fever; fleas; ticks; opossum; domestic animals
ARTICLES
Rickettsia infection in five areas of the state of São Paulo, Brazil
Maurício C HortaI, + + Corresponding author: maurivet@yahoo.com ; Marcelo B LabrunaI; Adriano PinterII; Pedro M LinardiIII; Teresinha T S SchumakerIV
IDepartamento de Medicina Veterinária Preventiva e Saúde Animal, Faculdade de Medicina Veterinária e Zootecnia, Universidade de São Paulo, Av. Prof. Orlando Marques de Paiva 87, 05508-270 São Paulo, SP, Brasil
IISuperintendência de Controle de Endemias, São Paulo, SP, Brasil
IIIDepartamento de Parasitologia, Instituto de Ciências Biológicas, Universidade Federal de Minas Gerais, Belo Horizonte, MG, Brasil
IVDepartamento de Parasitologia, Instituto de Ciências Biomédicas, Universidade de São Paulo, SP, Brasil
ABSTRACT
This study investigated rickettsial infection in animals, humans, ticks, and fleas collected in five areas of the state of São Paulo. Eight flea species (Adoratopsylla antiquorum antiquorum, Ctenocephalides felis felis, Polygenis atopus, Polygenis rimatus, Polygenis roberti roberti, Polygenis tripus, Rhopalopsyllus lugubris, and Rhopalopsyllus lutzi lutzi), and five tick species (Amblyomma aureolatum, Amblyomma cajennense, Amblyomma dubitatum, Ixodes loricatus, and Rhipicephalus sanguineus) were collected from dogs, cats, and opossums. Rickettsia felis was the only rickettsia found infecting fleas, whereas Rickettsia bellii was the only agent infecting ticks, but no animal or human blood was shown to contain rickettsial DNA. Testing animal and human sera by indirect immunofluorescence assay against four rickettsia antigens (R. rickettsii, R. parkeri, R. felis, and R. bellii), some opossum, dog, horse, and human sera reacted to R. rickettsii with titers at least four-fold higher than to the other three rickettsial antigens. These sera were considered to have a predominant antibody response to R. rickettsii. Using the same criteria, opossum, dog, and horse sera showed predominant antibody response to R. parkeri or a very closely related genotype. Our serological results suggest that both R. rickettsii and R. parkeri infected animals and/or humans in the studied areas.
Key words:Rickettsia - spotted fever - fleas - ticks - opossum - domestic animals
Rickettsiae are obligate intracellular bacteria that have been classically divided into two groups: the typhus group (TG), composed of Rickettsia prowazekii and Rickettsia typhi, which are associated with lice and fleas, respectively; and the spotted fever group (SFG), which includes more than 20 valid species, mostly associated with ticks (e.g., Rickettsia rickettsii, Rickettsia parkeri), and at least one species associated with fleas (Rickettsia felis). Other species, such as Rickettsia bellii and Rickettsia canadensis, both associated with ticks, have been considered a distinct group sharing antigenic, genetic and ecological characters with both TG and SFG rickettsiae (Parola et al. 2005b). Since all SFG Rickettsia share common outer membrane antigens, conventional serological assays employing a single Rickettsia antigen (e.g., R. rickettsii) allow detecting a SFG infection, but they do not identify the Rickettsia species responsible for the infection, even in the presence of high titers (La Scola & Raoult 1997). For a more specific serological diagnosis, it is recommended techniques such as testing serum against all known Rickettsia species present in a given area, or serum cross-absorption tests (La Scola & Raoult 1997, Parola et al. 2005b).
For many years, SFG rickettsiosis in the Americas was thought to be caused only by R. rickettsii, transmitted by ticks (Weiss & Moulder 1984). This rickettsia has been reported in several countries from North, Central, and South America causing an acute, highly lethal febrile disease receiving different names according to the country [e.g., Rocky Mountain spotted fever in the Unites States, Brazilian spotted fever (BSF) in Brazil] (Guedes et al. 2005). More recently, human spotted fever rickettsiosis due to R. parkeri have been reported in the United States (Paddock et al. 2004, Whitman et al. 2007). R. parkeri is also the most probably rickettsial agent of the Uruguayan spotted fever, a disease that has been reported in this country since early 1990s (Conti-Diaz 2003, Pacheco et al. 2006). While human infection by R. parkeri is yet to be documented in Brazil, two isolates of R. parkeri have been reported infecting Amblyomma ticks in the state of São Paulo (Labruna et al. 2004b, Silveira et al. 2007). In addition, serological evidence of canine infection due to R. parkeri has been reported in São Paulo and in northern Brazil (Horta et al. 2004a, Labruna et al. 2007a). R. felis is another human-pathogenic spotted fever Rickettsia reported in the Americas, including Brazil (Raoult et al. 2001). Unlike the above two Rickettsia species, which are vectored by ticks, R. felis is possibly carried by fleas especially Ctenocephalides spp. (Parola et al. 2005a).
There have been two reports of isolation of R. rickettsii from opossums (Didelphis spp.) from the states of São Paulo and Minas Gerais, Brazil (Moreira & Magalhães 1935, Travassos 1937). Interestingly, R. rickettsii has also been isolated from the North American opossum Didelphis virginiana (Bozeman et al. 1967). These findings led to several authors to suspect that opossums could participate in the natural cycle of R. rickettsii, serving as amplifier host for the agent (Dias & Martins 1939, Bozeman et al. 1967). More recently, R. felis-infected opossums (D. virginiana) were reported by serology in the USA, suggesting that opossums participate in the natural cycle of R. felis (Boostrom et al. 2002).
The present study investigates the presence of antibodies reactive to four Rickettsia species that have been recently described in the State of São Paulo (R. rickettsii, R. parkeri, R. felis, and R. bellii) among opossums, dogs, cats, horses, and humans from different areas of the state with or without history of recent cases of BSF. In addition, direct detection of Rickettsia DNA was attempted by molecular analyses of animal blood samples and from ticks and fleas collected on opossums, dogs and cats.
MATERIALS AND METHODS
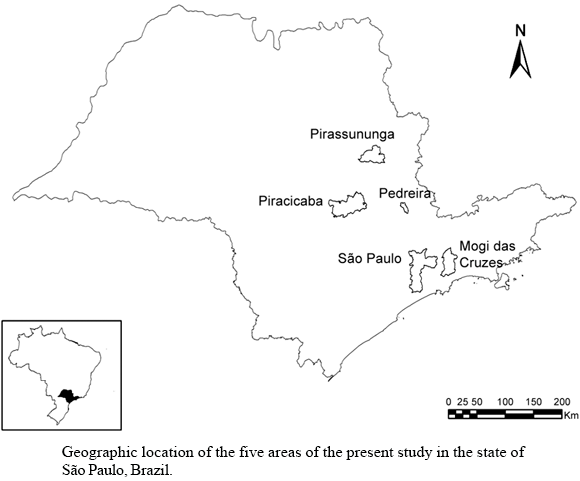
Study areas - This study was conducted in four areas in the state of São Paulo with history of recent confirmed cases of BSF in humans (endemic areas), and a fifth area with no history of BSF cases (nonendemic area) (Figure).
Endemic area 1: Pedreira (22º44'55.7" S, 46º53'55.7" W). 41 confirmed BSF cases of have been reported since 1985 (CVE 2006). The clinical severity (including high lethality), serological analyses, and isolation of Rickettsia from clinical samples of these cases were compatible with R. rickettsii (Lemos et al. 2001, Angerami et al. 2006). The presence of R. bellii and R. parkeri (strain Cooperi) was reported infecting the capybara tick Amblyomma dubitatum (Labruna et al. 2004b), whereas R. felis was reported infecting fleas Ctenocephalides felis felis (Horta et al. 2005). Serological analyses employing different rickettsia antigens and serum cross-absorption tests have indicated infection by R. rickettsii in horses and dogs, and by Rickettsia spp (possibly R. parkeri) in one dog (Horta et al. 2004a). In addition, serological analysis has indicated SFG infection in humans, horses, and dogs (Lemos et al. 1996a); and a SFG rickettsia has been isolated from an A. dubitatum tick (Lemos et al. 1996b). Tick species incriminated as vector of BSF in this area has been Amblyomma cajennense, although A. dubitatum has also been found in high population density. Both tick species are sustained by large populations of free-living capybaras (Hydrochaeris hydrochaeris); but horses are also main hosts for A. cajennense (Labruna et al. 2004b, Sangioni et al. 2005). Samples were collected in the same farms sampled by Horta et al. (2004a) and in other nearby farms.
Endemic area 2: Mogi das Cruzes, district of Taiaçupeba (23º38'34.5" S, 46º11'3.5" W). Several cases of BSF were reported in this municipality from 1957 to 1974 (Tiriba et al. 1968); nine confirmed cases of BSF were reported from 1998 to 2006 (CVE 2006). The clinical severity and high lethality (»80%) of these cases are compatible with R. rickettsii (Fontes et al. 2000). In one case, R. rickettsii was isolated by inoculation of clinical samples in guinea pigs (Melles et al. 1992). Both R. rickettsii and R. bellii have been isolated from ticks, and A. aureolatum was incriminated as the vector of BSF (Pinter & Labruna 2006). In addition, R. felis has been reported in fleas C. felis felis (Horta et al. 2005). Populations of A. aureolatum are sustained mainly by dogs (hosts for adult ticks) and small wild animals (rodents and birds). Capybaras, horses, as well as A. cajennense and A. dubitatum are rarely found in this area (Adriano Pinter et al. unpublished observations). Samples were collected in the Taiaçupeba district, the same area sampled by Pinter and Labruna (2006).
Endemic area 3: Piracicaba (22º42'22.8"S, 47º37'39.4"W). A total of 20 BSF-human cases have been reported in this area since 2002, but only a few of them were laboratory-confirmed (CVE 2006). Some of these cases were lethal, clinically compatible with infection due to R. rickettsii (CVE 2006). The tick species incriminated as vector of BSF is A. cajennense . The capybara tick A. dubitatum occurs in low frequency, and has been found infected by R. bellii (Horta et al. 2004b). Both tick species have been sustained by large populations of free-living capybaras; but horses (less abundant) may also play a role in sustaining A. cajennense populations. Samples were collected in the University of São Paulo campus of Piracicaba.
Endemic area 4: São Paulo (23º35'15.8"S, 46º39'28.1"W): BSF has been reported in different areas of this large municipality since the end of the 1920s (Gomes 1932, Monteiro & Tiriba 1976). During this period, several isolates from humans and two from opossums obtained through guinea pig inoculation showed to be compatible with R. rickettsii (Gomes 1932, Travassos 1937, Tiriba et al. 1968, Monteiro & Tiriba 1976). Recently (2002 to 2006), five laboratory-confirmed cases heve been reported (CVE 2006). A R. rickettsii-infected A. aureolatum tick was reported in this area decades ago (Gomes 1933), which remains as the possible tick vector. Currently, both A. cajennense and A. aureolatum are found in different parts of São Paulo municipality (Vieira et al. 2004). Samples were collected in University of São Paulo campus of São Paulo, Ibirapuera Park, and SESC-Interlagos.
Nonendemic area: Pirassununga (21º56'56.1"S, 47º27'16.9"W). BSF has never been notified or confirmed in this area. Similarly, no Rickettsia-infected tick or flea has been reported. On the other hand, there are very large populations of A. cajennense ticks sustained mainly by horses (Sangioni et al. 2005), and by capybaras in some particular patches. Capybaras also sustains some smaller populations of A. dubitatum. Samples were collected in the University of São Paulo campus of Pirassununga.
Animal and human samples - Capture of free-living opossums was performed from May 2003 to February 2005. In each area, a total of 15-25 work nights were necessary for collection of at least12 opossums per area. The minimum of 12 opossums per area was determined by numerical expectation based on epidemiological studies employing opossum captures (Cáceres & Monteiro Filho 2001). Captured opossums were anesthetized with ketamine 15-30 mg/kg for collection of blood and ectoparasites (fleas and ticks). Each trapped opossum was ear marked before releasing in the capture site, in order to identify possible recapture. Blood samples were collected from all dogs, cats, horses, and humans that used to live or work in the study areas. Serum was individually obtained by centrifugation, and stored at -20ºC for indirect immunofluorescence assay (IFA), and blood clot was stored at -80ºC for molecular analysis. Fleas and ticks were collected from all dogs and cats. This study was submitted and approved by the Ethical Committees of the University of São Paulo (protocol FMVZ 301/2003, ICB 495/CEP/2003), and by the Instituto Brasileiro do Meio Ambiente e dos Recursos Naturais Renováveis - IBAMA (02027.023912/2002).
Fleas and adult ticks were immediately preserved in 100% ethanol. Due to the impossibility of a precise identification of subadult ticks, captured larvae and nymphs were reared until the adult stage by feeding on laboratory naïve hosts (rabbits Oryctolagus cuniculus or guinea pigs Cavia porcellus), and subsequently identified to species according to Barros-Battesti et al. (2006). Ticks that died before reaching the adult stage were identified just to the generic level. Fleas were identified according to Linardi and Guimarães (2000)
IFA was used to detect human and animal serum antibodies reactive to Rickettsia spp. Glass slides were prepared according to Horta et al. (2004a), employing four Rickettsia species that are routinely maintained in our laboratory: R. rickettsii strain Taiaçu (Pinter & Labruna 2006), R. parkeri strain São Paulo (Silveira et al. 2007), R. felis strain Pedreira (Horta et al. 2006), and R. bellii strain Mogi (Pinter & Labruna 2006). Each Rickettsia strain was cultivated in Vero cells (or C6/36 cells in case of R. felis), and harvested when nearly 100% of the cells were infected. The infected cells were centrifuged at 4,000 g for 10 min, and pellet was washed in 0.1 M phosphate-buffered saline (PBS), pH 7.4, centrifuged again, and resuspended in PBS containing 1% bovine calf serum and 0.1% sodium azide. Ten microliters of rickettsiae-infected cells were applied onto each of 12 wells on microscopic slides, air-dried, fixed in acetone for 10 min, and stored at -80ºC until used. Human and animal sera were diluted in two-fold increments with PBS starting from a 1:64 dilution. Ten microliters of diluted sera were added to each well of the antigen slides. The slides were incubated at 37ºC for 30 min in a humid chamber. The slides were rinsed once, and then washed twice for 15 min per wash in PBS. The slides were incubated with fluorescein isothiocyanate-labeled sheep anti-opossum IgG (CCZ, São Paulo, Brazil), goat anti-human IgG, goat anti-horse IgG, goat anti-cat IgG, or rabbit anti-dog IgG (Sigma, St Louis, USA), and washed as described earlier. The slides were mounted with buffered glycerin under coverslips. The slides were read using an ultraviolet microscope (Olympus, Tokyo, Japan) at 400x magnification. Serum was considered to contain antibodies against the rickettsiae if it displayed a reaction at the 1:64 dilution. End-point titers against each Rickettsia strain were determined by testing serial two-fold serum dilutions. Reactive sera were tested in two or three replications before determining the endpoint titer. In each slide, a serum previously shown to be non-reactive (negative control) and a known reactive serum (positive control) were tested. Serum showing for a Rickettsia species titer at least four-fold higher than that observed for any other Rickettsia species was considered to elicit a predominant antibody response to the first Rickettsia species or to a very closely related genotype (Horta et al. 2004a, Labruna et al. 2007a).
DNA extraction was performed individually on whole fleas, whole subadult ticks, or on extirpated legs from adult ticks. Each flea or tick or a pool of legs from a single adult tick was ground in 50 µl of TE buffer (10mM Tris HCl; 1 mM ethylene diamine tetracetic acid pH 8.0) in sterile micro tubes. The final suspension was boiled at 100ºC for 20 min and held at -20ºC until tested by polymerase chain reaction (PCR). DNA extraction of animal and human blood samples were performed using Wizard® Genomic DNA Purification Kit (Promega, Madison, WI, USA), starting with 200 ml of blood clot from each individual sample. Fleas and ticks were individually processed by PCR using primers CS-78 (5'-GCAAGTATCG-GTGAGGATGTAAT-3') and CS-323 (5'-GCTTCCTTA-AAATTCAATAAATCAGGAT-3'), which amplify a 401-bp fragment of the citrate sinthase gene (gltA), possibly present in all Rickettsia species (Labruna et al. 2004b). For blood samples, primers CS-5 (5'-GAGAGAAAAT-TATATCCAAATGTTGAT-3') and CS-6 (AGGGTCTT-CGTGCATTTCTT), which amplify a 147-bp fragment of the rickettsial gltA gene were used. These later primers have shown sensitivity down to a single copy of R. rickettsii and 10 copies of R. bellii (Labruna et al. 2004b). For each reaction, 5 µl of the DNA template from each individual sample (flea, tick, animal, or human) were added to 5 µl of PCR buffer (10X), 8 µl of deoxynucleotide triphosphates mixture (1.25 mM), 1.5 µl MgCl2 (50 mM), 25 pmol of each primer, 0.25 µl of Taq polymerase (5,000 U/ml) and bi-distilled water to a final volume of 50 µl. For each reaction, DNA extracted from R. parkeri-infected A. cajennense ticks was used as positive control (Sangioni et al. 2005) and at least three aliquots of bi-distilled water were used as negative controls. PCR thermal conditions were used as previously described (Labruna et al. 2004b). PCR products were stained by ethidium bromide and visualized by electrophoresis in 1.5% agarose gel.
All PCR products of the expected amplicon size were purified, and their forward and reverse nucleotide sequences were determined in a DNA sequencer analyzer (ABI Prism 3100 Genetic pplied Biosystems/Perking Elmer, California, USA). Partial sequences obtained were submitted to BLAST (Basic Local Alignment Search Tool) analysis (Altschl et al. 1990) to determine similarities to other Rickettsia species.
RESULTS
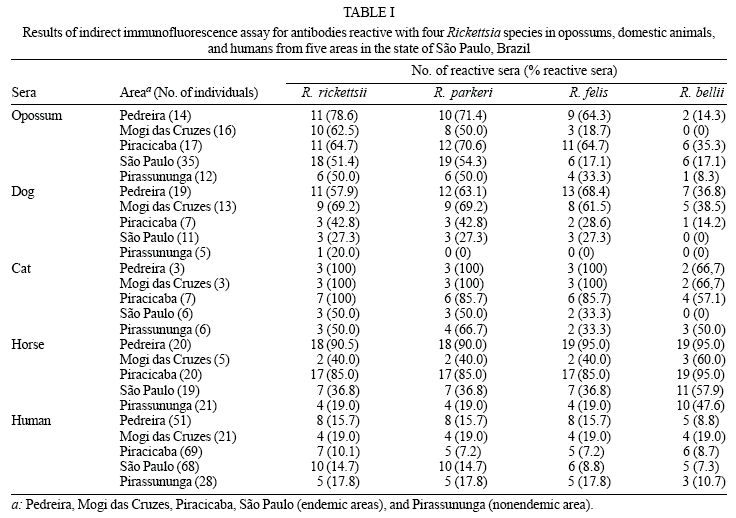
A total of 94 opossums (65 D. aurita and 29 D. albiventris) were trapped in BSF-endemic and nonendemic areas. In Pedreira, Mogi das Cruzes, and São Paulo (endemic areas 1, 2 and 4, respectively), all opossums were D. aurita, whereas D. albiventris comprised all opossums from Piracicaba (endemic area 3) and Pirassununga (non-endemic area). A total of 55 dogs, 25 cats, 85 horses, and 229 humans were sampled. The numbers of animals and humans sampled in each of the five areas are shown in Table I.
IFA - Overall, sera from 64 (68.1%) opossums (43 D. aurita and 21 D. albiventris), 33 dogs (60.0%), 21 cats (84.0%), 62 horses (72.9%), and 34 humans (14.3%) reacted positively (titer > 64) to at least one of the four Rickettsia antigens tested (Table I). In Pedreira, Mogi das Cruzes, Piracicaba, and São Paulo (endemic areas 1, 2, 3, and 4, respectively), at least three, two, four, and one opossum sera (respectively) showed titers to R. rickettsii at least four-fold higher than to any of the other three rickettsial antigens. These serum antibody responses were considered to be produced against to R. rickettsii or a very closely related genotype. Using the same criteria, antibody responses in sera from two dogs from Pedreira and two dogs from Mogi das Cruzes, one horse and one human from Piracicaba, and one human from Pedreira were also considered to be produced to R. rickettsii (Table II). Additionally, one opossum from Mogi das Cruzes, one dog and one horse from Piracicaba, and one opossum from Pedreira showed antibody responses that were considered to be produced to R. parkeri. Regarding Pirassununga (nonendemic area), only a single opossum serum showed antibody response that was considered to be produced to R. parkeri or a very closely related genotype. Despite of several sera also reacted positively to R. felis and R. bellii, none antibody response could be considered to be produced to these agents due to similar or low titers in comparison with titers to R. rickettsii or R. parkeri.
Fleas and Ticks - A total of 312 fleas were collected from opossums. Overall, the fleas C. felis felis, Polygenis (Neopolygenis) atopus, and Rhopalopsyllus lutzi lutzi comprised 94.5% of the collected fleas. Other five species [Polygenis (Polygenis) roberti roberti, P. (Polygenis) tripus, P. (Polygenis) rimatus, Rhopalopsyllus lugubris, and Adoratopsylla (Adoratopsylla) antiquorum antiquorum)] comprised 5.5% of the collected fleas. C. felis felis was the most common flea species infesting opossums in the four endemic areas. However, it was absent from Pirassununga (nonendemic area), where P. atopus and R. lutzi lutzi were the most prevalent flea species. A total of 278 fleas were collected from dogs (n = 212 fleas) and cats (n = 66). All fleas from dogs were identified as C. felis felis, whereas fleas from cats were identified as C. felis felis (n = 59) and R. lutzi lutzi (n = 7). No flea was collected from horses or humans.
R. felis was the only Rickettsia species found infecting fleas in all five areas. Infection rates varied from 33.3 to 72.7% among C. felis felis fleas from opossums (Table III), from 13.5 to 52.7% among C. felis felis fleas from dogs, and from 29.4 to 83.3% among C. felis felis fleas from cats (Table IV). In addition, 3.3 and 8.0% of the P. atopus fleas collected on opossums from Mogi das Cruzes and Pirassununga, respectively, were also infected by R. felis. Additional flea species collected from opossums and that did not contain rickettsial DNA were R. lutzi lutzi (Pedreira, n = 16; Piracicaba, 12; Pirassununga, 24), P. roberti roberti (Mogi das Cruzes, 7; Pirassununga, 1), P. tripus (Mogi das Cruzes, 2; Piracicaba, 1; Pirassununga, 1), R. lugubris (Pedreira, 3), A. antiquorum antiquorum (Mogi das Cruzes, 1), and P. rimatus (Mogi das Cruzes, 1).
A total of 709 tick specimens were collected from opossums, whereas 115 and 10 ticks were collected from dogs and cats, respectively. Ticks were found infesting animals from all studied areas, except for dogs, and cats from Pirassununga, and cats from São Paulo. Three tick species were found infesting opossums (A. cajennense in Pedreira and Piracicaba; A. dubitatum in Mogi das Cruzes, Piracicaba and Pirassununga; and Ixodes loricatus in Mogi das Cruzes, São Paulo and Pirassununga), whereas three tick species were found infesting dogs and cats (Rhipicephalus sanguineus and A. cajennense in Pedreira and Piracicaba; and A. aureolatum in Mogi das Cruzes and São Paulo). Ticks were not collected on horses, although infestations by both A. cajennense and Anocentor nitens ticks were visually detected in most of the horses from Pedreira, Piracicaba, and Pirassununga. No tick was collected from humans, although humans complained about suffering from frequent tick infestation in all areas, except for Mogi das Cruzes and São Paulo, where tick infestation was occasionally noticed.
All ticks collected on opossums, dogs and cats were tested by PCR. No Rickettsia was detected in ticks from dogs and cats, whereas R. bellii was the only species detected in ticks (I. loricatus, A. dubitatum, and Amblyomma sp) from opossums (Table III). No rickettsial DNA was detected in animal or human blood samples.
DISCUSSION
BSF is thought to be caused by R. rickettsii, however the vast majority of the laboratory-confirmed cases in the state of São Paulo have been diagnosed solely by serology employing a single antigen (R. rickettsii) or/and isolation of the agent in cell culture without further species identification, and/or PCR without DNA sequencing (Nascimento & Schumaker 2004). Technically, these diagnostic procedures have not allowed a precise identification of the rickettsial agent; at best, defining SFG rickettsiosis, since the three employed techniques could be efficient for any spotted fever group Rickettsia species. During the last six years, four SFG species other than R. rickettsii were reported in Brazil, two of them recognized as agents of spotted fever rickettsiosis in humans (R. parkeri and R. felis) (Raoult et al. 2001, Labruna et al. 2004a, b, Horta et al. 2005, Labruna et al. 2007c, Silveira et al. 2007). Thus, it is possible that some of the BSF cases diagnosed in the state of São Paulo could be caused by other SFG rickettsia, such as R. parkeri or R. felis.
The strong serological reactivity with R. rickettsii is an evidence that this species might be inducing an infection in humans and/or animals from the four endemic areas, whereas no convincing evidence of infection due to this agent was found in Pirassununga, the nonendemic area (Table II). On the other hand, serological reactivity for R. parkeri was detected in animals from four out of the five studied areas, including the nonendemic area. Since R. parkeri spotted fever is a newly identified disease, which is less virulent (no lethality has been reported), and less widely recognized than the infection caused by R. rickettsii, it is quite possible that human-R. parkeri spotted fever has been occurring without a proper diagnosis in Pedreira, Mogi das Cruzes, Piracicaba, and Pirassununga. A potential tick vector of R. parkeri in these areas is the capybara tick A. dubitatum, which has been recently found infected by R. parkeri (strain Cooperi) in Pedreira, exactly in the same site sampled in the present study (Labruna et al. 2004b), and where a R. parkeri infection was reported in one dog by both IFA and cross-absorption technique (Horta et al. 2004a). In addition, successful experimental infection of R. parkeri has been achieved in A. cajennense (Sangioni et al. 2005), which should also be considered a potential vector.
R. felis is a spotted fever group rickettsia that has been proved to be pathogenic for humans in several countries, including Brazil (Raoult et al. 2001). However, we found no clear serological reactivity of animal or human-R. felis infection in the present study. This finding was surprising, since R. felis was detected in high frequencies among the most common flea species from opossums, dogs and cats in the five studied areas. In the USA, Boostrom et al. (2002) reported R. felis-infected opossums (D. virginiana) by means of serology. However, these authors did not find correlation between seropositive opossums and R. felis-infected fleas collected on these opossums. After its first report infecting C. felis felis fleas in the USA (Adams et al. 1990), R. felis has turned to be a cosmopolitan species infecting mostly C. felis fleas (Parola et al. 2005a). The contrast of few reported human cases of R. felis-spotted fever with the cosmopolitan distribution of R. felis-infected fleas deserves further investigations.
No clear reactivity of R. bellii infection in humans or animals was found in the present study. R. bellii has been the most frequent rickettsia species infecting ticks in Brazil, being reported in at least nine tick species (Labruna et al. 2007c). In contrast to the present study, there has been substantial serological evidence that R. bellii produces infection in capybaras, which are often parasitized by A. dubitatum (Pacheco et al. 2007). This tick species has been often found infected by R. bellii in the state of São Paulo (Labruna et al. 2004b, Horta et al. 2004b).
In a recent study with horses, dogs, and humans in Pedreira, Sangioni et al. (2005) recommended surveys of horse sera (and dogs in a lesser extent) as a useful method for BSF surveillance in areas where humans are exposed to A. cajennense ticks. This recommendation is based on the fact that horses are primary hosts for all parasitic stages of A. cajennense, a tick that also attacks dogs and other medium-sized mammals. Our results showed that horses were in fact excellent sentinels for BSF in Pedreira and Piracicaba, two areas where A. cajennense is incriminated as the main vector of R. rickettsii. In these two areas, 85 to 90% of the horses were seropositive for R. rickettsii. On the other hand, much lower frequencies (19 - 40%) of seropositive horses for R. rickettsii were detected in Mogi das Cruzes, São Paulo, and Pirassununga. In Mogi das Cruzes, A. aureolatum is the primary vector of R. rickettsii, corroborating our finding of 69.2% of seropositive dogs, which are primary hosts for the adult stage of A. aureolatum. In São Paulo and Pirassununga, neither horses nor dogs were demonstrated to be good sentinels for BSF or R. parkeri infection; in these two areas, cats and opossums showed the highest seropositivities for R. rickettsii and R. parkeri. In addition, cats and opossums were also excellent sentinels for these two agents in Pedreira, Mogi das Cruzes, and Piracicaba. These results indicate that surveys of cats and opossums sera would be more efficient than horse or dog for BSF surveillance. This superiority of cats and opossums is possibly related to the fact that they are more exposed to ticks than the other animals. Opossums are wild animals with synantropic habits. Rural cats are domestic animals with wild habits; thus they also live in both wild and domestic environments. In contrast, horses are usually restricted to pastures while rural dog habits are high variable, ranging from completely domestic to those with some wild habits.
Comparing to other animals, human sera demonstrated the lowest frequencies of seropositivity for Rickettsia spp. This result was expected since humans are much less exposed to ticks than other animals. Only two human sera were considered homologous to R. rickettsii. Several other human sera reacted positively to Rickettsia spp., but it was not possible to determine the Rickettsia species involved in a predominant antibody response. Based on the results obtained with animals, it is possible that some of these humans were infected by R. rickettsii or R. parkeri; also, infection by R. felis could not be ruled out, since sera showed similar titers (< 4 fold difference) for these three agents (data not shown).
Our molecular analysis showed that 13.5 - 83.3% of the C. felis felis fleas collected from the five areas of the present study were infected with R. felis. In addition, 3.3 and 8.1% of the P. atopus fleas collected on opossums from Mogi das Cruzes and Pirassununga, respectively, were also infected by R. felis. This is the first report of this rickettsia infecting this wild flea species, indigenous from the Neotropical region. Interestingly, in Pirassununga, there was no C. felis felis infesting opossums. This result suggests that P. atopus is capable of maintaining the infection by R. felis without the presence of C. felis felis.
It is noteworthy our findings of A. dubitatum nymphs parasitizing opossums for the first time. This tick species uses capybaras as primary hosts for all parasitic stages. However, larvae and nymphs seems to have a much broader host range, including humans (Labruna et al. 2007b). Five tick species collected from opossums, dogs, and cats were tested for rickettsial infection. No rickettsia was found infecting ticks from dogs and cats, while only R. bellii was detected in two species of ticks from opossums (A. dubitatum and I. loricatus). Previously, R. bellii was reported infecting these two tick species in the state of São Paulo (Labruna et al. 2004b, Horta et al. 2004b). Previous studies in the state of São Paulo reported R. rickettsii in A. aureolatum (Gomes 1933, Pinter & Labruna 2006) and A. cajennense (Vallejo-Freire 1946) ticks, and R. parkeri (strain COOPERI) in A. dubitatum ticks (Labruna et al. 2004b). However, none of these agents were found infecting ticks in the present study. The reasons for these negative results are possibly the small number of tested ticks, and the low infection rates of R. rickettsii and R. parkeri that are usually reported for A. aureolatum, A. cajennense, or A. dubitatum (Labruna et al. 2004b, Sangioni et al. 2005, Pinter & Labruna 2006)
We did not detect rickettsial DNA in any human and animal blood samples in the present study. Detection of rickettsia in vertebrate blood is usually a rare event, since once infected, vertebrates display a ricketsemia for only a few days or weeks, and thereafter no rickettsia is found in the blood (Burgdorfer et al. 1988). In addition, Rickettsia spp. are known to infect endothelial cells in vertebrates, thus its concentration in the blood is very low, incapable of be detected by molecular analysis (La Scola & Raoult 1997).
Previously, R. rickettsii was identified infecting humans from Pedreira, Mogi das Cruzes, and São Paulo. The present study presents serological reactivity of R. rickettsii infection in humans from Piracicaba. Previous serological diagnosis of BSF in this area did not allow a precise identification of the agent, although it was clearly a spotted fever disease clinically and epidemiologically compatible with R. rickettsii infection. Despite of the presence of at least four Rickettsia species in most of the studied areas, there was serological evidence of rickettsial infection in humans and/or animals by only two species: R. rickettsii and R. parkeri. These findings strongly suggest that some of the BSF cases occurring in BSF-endemic areas of the state of São Paulo could be caused by R. parkeri. In addition, our results showed that R. parkeri could also be present in areas considered non-endemic for BSF. Thus, there is a possibility that the distribution area of R. parkeri is much broader in the state of São Paulo, since one of its main tick hosts (the capybara tick, A. dubitatum) is widely distributed (Vieira et al. 2004). Finally, since R. rickettsii has been the sole antigen regularly employed for serological diagnosis of BSF, it is likely that human spotted fever cases due to R. parkeri or other spotted fever group rickettsiae are being misidentified as BSF in Brazil.
ACKNOWLEDGEMENTS
To Maria Rita N Rodrigues, Maria C Bataglioli, Prof. Dr. Gilberto José de Morais, Prefeitura do Campus da Escola Superior de Agricultura Luiz de Queiroz (ESALQ/USP), Luiza Shigueno, Prefeitura do Campus Administrativo da USP Pirassununga, Maria Eugênia L Summa, Vilma C Geraldi, Francis W Rossi, and Adriana M Joppert from the Departamento de Parques e Áreas Verdes de São Paulo (DEPAVE), Lucia EO Yai from Centro de Controle de Zoonoses (CCZ) of São Paulo city, Daniela P Chiebao, Marcello S Nardi, Patrícia B Bertola, Patrícia M Ferreira and Renata Paixão from the University of São Paulo (USP) for technical support.
REFERENCES
Adams JR, Schmidtmann ET, Azad AF 1990. Infection of colonized cat fleas, Ctenocephalides felis (Bouché), with a rickettsia-like microorganism. Am J Trop Med Hyg 43: 400-409.
Altschl SF, Gish W, Miller W, Myers EW, Lipman DJ 1990. Basic local alignment search tool. J Mol Biol 215: 403-410.
Angerami RN, Resende MR, Feltrin AF, Katz G, Nascimento EM, Stucchi RS, Silva LJ 2006. Brazilian spotted fever: a case series from an endemic area in southeastern Brazil: epidemiological aspects. Ann N Y Acad Sci 1078: 170-172
Barros-Battesti DM, Arzua M, Bechara GH 2006. Carrapatos de importância médico-veterinária da região Neotropical. Um guia ilustrado para identificação de espécies, Vox, ICTTD-3, Butantan, São Paulo 223pp.
Boostrom A, Beier MS, Macaluso JA, Macaluso KR, Sprenger D, Hayes J, Radulovic S, Azad AF 2002. Geographic association of Rickettsia felis-infected opossums with human murine typhus, Texas. Emerg Infect Dis 8: 549-554.
Bozeman FM, Shirai A, Humphries JW, Fuller HS 1967. Ecology of Rocky Mountain spotted fever II. Natural infection of wild mammals and birds in Virginia and Maryland. Am J Trop Med Hyg 16: 48-59.
Burgdorfer W 1988. Ecological and epidemiological considerations of Rock Mountain spotted fever and scrub typhus. In DH Walker, Biology of Rickettsial Diseases, Vol.1, CRC Inc, Boca Raton, p. 33-50.
Cáceres NC, Monteiro Filho ELA 2001. Food habits, home range and activity of Didelphis aurita (Mammalia, Marsupialia) in a forest fragment of southern Brazil. Stud Neotrop Fauna Environ 36: 85-92.
Conti-Diaz IA 2003. Rickettsioses caused by Rickettsia conorii in Uruguay. Ann N Y Acad Sci 990: 264-266.
CVE - Centro de Vigilância Epidemiológica 2006. Governo do Estado de São Paulo. [cited 31 December 2006] Available from http://www.cve.saude.sp.gov.br/htm/cve_fmb.html.
Dias E, Martins AV 1939. Spotted fever in Brazil. Am J Trop Med 19: 103-108.
Fontes LR, Gomes SMA, Anjos ED, Andrade JCR 2000. Brazilian Spotted Fever transmitted by Amblyomma aureolatum (Acari) in Mogi das Cruzes, Brazil: Reported of four human cases and environmental control measures, XXI International Congress of Entomology, Brazil, Abstract Book, p.749.
Gomes LS 1932. Estudo experimental do typho exanthematico de São Paulo. In JT Piza, GR Meyer, LS Gomes, Typho exanthematico de São Paulo, Sociedade Impressora Paulista, São Paulo, p. 138-156.
Gomes LS 1933. Thypho exanthematico de São Paulo. Brasil-Médico 17: 919-921.
Guedes E, Leite RC, Prata MCA, Pacheco RC, Walker DH, Labruna MB 2005. Detection of Rickettsia rickettsii in the tick Amblyomma cajennense in a new Brazilian spotted fever-endemic area in the state of Minas Gerais. Mem Inst Oswaldo Cruz 100: 841-845.
Horta MC, Labruna MB, Durigon EL, Schumaker TTS 2006. Isolation and cultivation of Rickettsia felis in the mosquito cell line C6/36. Appl Environ Microbiol 72: 1705-1707.
Horta MC, Labruna MB, Sangioni LA, Vianna MCB, Gennari SM, Galvão MAM, Mafra CL, Vidotto O, Schumaker TTS, Walker DH 2004a. Prevalence of antibodies to Spotted Fever Group Rickettsiae in humans and domestic animals in a Brazilian Spotted Fever-Endemic area in the State of São Paulo, Brazil: Serologic evidence for infection by Rickettsia rickettsii and another spotted fever group Rickettsia. Am J Trop Med Hyg 71: 93-97.
Horta MC, Pinter A, Cortez A, Soares RM, Gennari SM, Schumaker TTS, Labruna MB 2005. Rickettsia felis (Rickettsiales:Rickettsiaceae) in Ctenocephalides felis felis (Siphonaptera: Pulicidae) in the State of São Paulo, Brazil. Arq Bras Med Vet Zootec 57: 321-325.
Horta MC, Pinter A, Souza CE, Neto EJR, Souza SSAL, Soares RM, Silva LJ, Labruna MB 2004b. Ocorrência de Rickettsia bellii em carrapatos colhidos nos Municípios de Piracicaba, Pedreira, Campinas, Itu e Cordeirópolis, Estado de São Paulo. Rev Bras Parasitol Vet 13 (Suppl.): 364.
Labruna MB, Horta MC, Aguiar DM, Cavalcante GT, Pinter A, Gennari SM, Camargo LMA 2007a. Prevalence of Rickettsia infection in dogs from the urban and rural areas of Monte Negro Municipality, western Amazon, Brazil. Vector Borne Zoonotic Dis 7: 249-255.
Labruna MB, Pacheco RC, Ataliba AC, Szabó MPJ 2007b. Human parasitism by the capibara tick, Amblyomma dubitatum. Entomol News 118: 77-80.
Labruna MB, Pacheco RC, Richtzenhain LJ, Szabo MP 2007c. Isolation of Rickettsia rhipicephali and Rickettsia bellii from ticks Haemaphysalis juxtakochi in the state of São Paulo, Brazil. Appl Environ Microbiol 73: 869-873.
Labruna MB, Teixeira RH, Walker DH 2004a. Rickettsia bellii infectando carrapatos Ixodes loricatus em São Paulo. Rev Bras Parasitol Vet 13 (Suppl.): 362.
Labruna MB, Whitworth T, Horta MC, Bouyer DH, McBride J, Pinter A, Popov V, Gennari SM, Walker DH 2004b. Rickettsia species infecting Amblyomma cooperi ticks from an area in the State of São Paulo, Brazil, where Brazilian spotted fever is endemic. J Clin Microbiol 42: 90-98.
La Scola B, Raoult D 1997. Laboratory diagnosis of Rickettsioses: current approaches to diagnosis of old and new Rickettsial diseases. J Clin Microbiol 35: 2715-2727.
Lemos ERS, Alvarenga FB, Cintra ML, Ramos MC, Paddock CD, Ferebee TL, Zaki SR, Ferreira FC, Ravagnani RC, Machado RD, Guimarães MA, Coura JR 2001. Spotted fever in Brazil: a seroepidemiological study and description of clinical cases in an endemic area in the state of São Paulo. Am J Trop Med Hyg 65: 329-334.
Lemos ERS, Machado JR, Coura JR, Guimarães MAAM, Chagas N 1996a. Epidemiological aspects of the Brazilian spotted fever: Serological survey of dogs and horses in an endemic area in the state of São Paulo, Brazil. Rev Inst Med Trop São Paulo 38: 427-430.
Lemos ERS, Melles HHB, Colombo S, Machado RD, Coura JR, Guimarães MAA, Sanseverino SR, Moura A. 1996b. Primary isolation of spotted Fever Group Rickettsiae from Amblyomma cooperi collected from Hydrochaeris hydrochaeris in Brazil. Mem Inst Oswaldo Cruz 91: 273-275.
Linardi PM, Guimarães LR 2000. Sifonápteros do Brasil, Museu de Zoologia USP/FAPESP, São Paulo, 291p.
Melles HHB, Colombo S, Silva MV 1992. Febre maculosa: isolamento de Rickettsia em amostra de biopsia de pele. Rev Inst Med Trop São Paulo 34: 37-41.
Monteiro EVL, Tiriba AC 1976. Rickettsioses. In R Veronesi (ed.), Doenças Infecciosas e Parasitárias, Guanabara Koogan, São Paulo, p. 236-249.
Moreira JA, Magalhães O 1935. Thypho exanthematico em Minas Gerais. Brasil-Médico 44: 465-470.
Nascimento EM, Schumaker TTS 2004. Isolamento e identificação de riquétsias no Brasil. Rev Bras Parasitol Vet 13 (Suppl.): 193-196.
Pacheco RC, Horta MC, Moraes-Filho J, Ataliba AC, Pinter A, Labruna MB. 2007. Rickettsial infection in capybaras (Hydrochaeris hydrochoerus) from São Paulo, Brazil: serological evidence for infection by Rickettsia bellii and Rickettsia parkeri. Biomedica 27: 364-371.
Pacheco RC, Venzal JM, Richtzenhain LJ, Labruna MB 2006. Rickettsia parkeri in Uruguay. Emerg Infec Dis 12: 1804-1805.
Paddock CD, Sumner JW, Comer JA, Zaki SR, Goldsmith CS, Goddard J, McLellan SL, Tamminga CL, Ohl CA 2004. Rickettsia parkeri: A newly recognized cause of spotted fever rickettsiosis in the United States. Clin Infect Dis 38: 805-811.
Parola P, Davoust B, Raoult D 2005a. Tick- and flea-borne rickettsial emerging zoonoses. Vet Res 36: 469-492.
Parola P, Paddock CD, Raoult D 2005b. Tick-borne rickettsioses around the world: emerging diseases challenging old concepts. Clin Microbiol Rev 18: 719-756.
Pinter A, Labruna MB 2006. Isolation of Rickettsia rickettsii and Rickettsia bellii in cell culture from the tick Amblyomma aureolatum in Brazil. Ann N Y Acad Sci 1078: 523-529.
Raoult D, La Scola B, Enea M, Fournier PE, Roux V, Fenollar F, Galvão MAM, Lamballerie X 2001. A flea-associates Rickettsia pathogenic for humans. Emerg Infec Dis 7: 73-81.
Sangioni LA, Horta MC, Vianna MCB, Gennari SM, Soares RM, Galvão MAM, Schumaker TTS, Ferreira F, Vidotto O, Labruna MB 2005. Rickettsial infection in animals and Brazilian spotted fever endemicity. Emerg Infect Dis 11: 265-270.
Silveira I, Pacheco RC, Szabó MPJ, Ramos HGC, Labruna MB 2007. First report of Rickettsia parkeri in Brazil. Emerg Infect Dis 13: 1111-1113.
Tiriba AC, Godoy CVF, Brito T, Jordão FM, Penna DO, Souza ART 1968. Febre maculosa em São Paulo. Resultados terapêuticos em alguns casos com laurilsulfato de tetraciclina. Rev Inst Med Trop 10: 256-261.
Travassos J 1937. Identification d'un virus semblable a celui du " Typhus exanthématique de Sao Paulo ", isolé de la sarigue marsupiale (Didelphis paraguayensis). Compt Rend Soc Biol 126: 1054-1056.
Vallejo-Freire A 1946. Spotted fever in Mexico. Mem Inst Butantan 19: 159-180.
Vieira AML, Souza CE, Labruna MB, Mayo RC, Souza SSL, Camargo-Neves VLF 2004. Manual de Vigilância Acarológica, Estado de São Paulo, Secretaria de Estado da Saúde, São Paulo, Brasil, 62 pp.
Weiss E, Moulder JW 1984. The Rickettsias and Chlamydias. In NR Kreig, JG Holt (eds.), Bergey's manual of systematic bacteriology, Vol. 1, Williams & Wilkins, Baltimore, MD, p. 687-739.
Whitman TJ, Richards AL, Paddock CD, Tamminga CL, Sniezek PJ, Jiang J, Byers DK, Sanders JW 2007. Rickettsia parkeri infection after tick bite. Virginia Emerg Infect Dis 13: 334-336.
Received 28 February 2007
Accepted 5 November 2007
Finacial support: FAPESP (grants 02/10759-0 to MCH, 03/04728-7 to TTSS, 03/13872-4 to MBL,) and CNPq (research career scholarship to MBL).
- Adams JR, Schmidtmann ET, Azad AF 1990. Infection of colonized cat fleas, Ctenocephalides felis (Bouché), with a rickettsia-like microorganism. Am J Trop Med Hyg 43: 400-409.
- Altschl SF, Gish W, Miller W, Myers EW, Lipman DJ 1990. Basic local alignment search tool. J Mol Biol 215: 403-410.
- Angerami RN, Resende MR, Feltrin AF, Katz G, Nascimento EM, Stucchi RS, Silva LJ 2006. Brazilian spotted fever: a case series from an endemic area in southeastern Brazil: epidemiological aspects. Ann N Y Acad Sci 1078: 170-172
- Barros-Battesti DM, Arzua M, Bechara GH 2006. Carrapatos de importância médico-veterinária da região Neotropical. Um guia ilustrado para identificação de espécies, Vox, ICTTD-3, Butantan, São Paulo 223pp.
- Boostrom A, Beier MS, Macaluso JA, Macaluso KR, Sprenger D, Hayes J, Radulovic S, Azad AF 2002. Geographic association of Rickettsia felis-infected opossums with human murine typhus, Texas. Emerg Infect Dis 8: 549-554.
- Bozeman FM, Shirai A, Humphries JW, Fuller HS 1967. Ecology of Rocky Mountain spotted fever II. Natural infection of wild mammals and birds in Virginia and Maryland. Am J Trop Med Hyg 16: 48-59.
- Burgdorfer W 1988. Ecological and epidemiological considerations of Rock Mountain spotted fever and scrub typhus. In DH Walker, Biology of Rickettsial Diseases, Vol.1, CRC Inc, Boca Raton, p. 33-50.
- Cáceres NC, Monteiro Filho ELA 2001. Food habits, home range and activity of Didelphis aurita (Mammalia, Marsupialia) in a forest fragment of southern Brazil. Stud Neotrop Fauna Environ 36: 85-92.
- Conti-Diaz IA 2003. Rickettsioses caused by Rickettsia conorii in Uruguay. Ann N Y Acad Sci 990: 264-266.
- Dias E, Martins AV 1939. Spotted fever in Brazil. Am J Trop Med 19: 103-108.
- Fontes LR, Gomes SMA, Anjos ED, Andrade JCR 2000. Brazilian Spotted Fever transmitted by Amblyomma aureolatum (Acari) in Mogi das Cruzes, Brazil: Reported of four human cases and environmental control measures, XXI International Congress of Entomology, Brazil, Abstract Book, p.749.
- Gomes LS 1932. Estudo experimental do typho exanthematico de São Paulo. In JT Piza, GR Meyer, LS Gomes, Typho exanthematico de São Paulo, Sociedade Impressora Paulista, São Paulo, p. 138-156.
- Gomes LS 1933. Thypho exanthematico de São Paulo. Brasil-Médico 17: 919-921.
- Guedes E, Leite RC, Prata MCA, Pacheco RC, Walker DH, Labruna MB 2005. Detection of Rickettsia rickettsii in the tick Amblyomma cajennense in a new Brazilian spotted fever-endemic area in the state of Minas Gerais. Mem Inst Oswaldo Cruz 100: 841-845.
- Horta MC, Labruna MB, Durigon EL, Schumaker TTS 2006. Isolation and cultivation of Rickettsia felis in the mosquito cell line C6/36. Appl Environ Microbiol 72: 1705-1707.
- Horta MC, Labruna MB, Sangioni LA, Vianna MCB, Gennari SM, Galvão MAM, Mafra CL, Vidotto O, Schumaker TTS, Walker DH 2004a. Prevalence of antibodies to Spotted Fever Group Rickettsiae in humans and domestic animals in a Brazilian Spotted Fever-Endemic area in the State of São Paulo, Brazil: Serologic evidence for infection by Rickettsia rickettsii and another spotted fever group Rickettsia. Am J Trop Med Hyg 71: 93-97.
- Horta MC, Pinter A, Cortez A, Soares RM, Gennari SM, Schumaker TTS, Labruna MB 2005. Rickettsia felis (Rickettsiales:Rickettsiaceae) in Ctenocephalides felis felis (Siphonaptera: Pulicidae) in the State of São Paulo, Brazil. Arq Bras Med Vet Zootec 57: 321-325.
- Horta MC, Pinter A, Souza CE, Neto EJR, Souza SSAL, Soares RM, Silva LJ, Labruna MB 2004b. Ocorrência de Rickettsia bellii em carrapatos colhidos nos Municípios de Piracicaba, Pedreira, Campinas, Itu e Cordeirópolis, Estado de São Paulo. Rev Bras Parasitol Vet 13 (Suppl.): 364.
- Labruna MB, Horta MC, Aguiar DM, Cavalcante GT, Pinter A, Gennari SM, Camargo LMA 2007a. Prevalence of Rickettsia infection in dogs from the urban and rural areas of Monte Negro Municipality, western Amazon, Brazil. Vector Borne Zoonotic Dis 7: 249-255.
- Labruna MB, Pacheco RC, Ataliba AC, Szabó MPJ 2007b. Human parasitism by the capibara tick, Amblyomma dubitatum. Entomol News 118: 77-80.
- Labruna MB, Pacheco RC, Richtzenhain LJ, Szabo MP 2007c. Isolation of Rickettsia rhipicephali and Rickettsia bellii from ticks Haemaphysalis juxtakochi in the state of São Paulo, Brazil. Appl Environ Microbiol 73: 869-873.
- Labruna MB, Teixeira RH, Walker DH 2004a. Rickettsia bellii infectando carrapatos Ixodes loricatus em São Paulo. Rev Bras Parasitol Vet 13 (Suppl.): 362.
- Labruna MB, Whitworth T, Horta MC, Bouyer DH, McBride J, Pinter A, Popov V, Gennari SM, Walker DH 2004b. Rickettsia species infecting Amblyomma cooperi ticks from an area in the State of São Paulo, Brazil, where Brazilian spotted fever is endemic. J Clin Microbiol 42: 90-98.
- La Scola B, Raoult D 1997. Laboratory diagnosis of Rickettsioses: current approaches to diagnosis of old and new Rickettsial diseases. J Clin Microbiol 35: 2715-2727.
- Lemos ERS, Alvarenga FB, Cintra ML, Ramos MC, Paddock CD, Ferebee TL, Zaki SR, Ferreira FC, Ravagnani RC, Machado RD, Guimarães MA, Coura JR 2001. Spotted fever in Brazil: a seroepidemiological study and description of clinical cases in an endemic area in the state of São Paulo. Am J Trop Med Hyg 65: 329-334.
- Lemos ERS, Machado JR, Coura JR, Guimarães MAAM, Chagas N 1996a. Epidemiological aspects of the Brazilian spotted fever: Serological survey of dogs and horses in an endemic area in the state of São Paulo, Brazil. Rev Inst Med Trop São Paulo 38: 427-430.
- Lemos ERS, Melles HHB, Colombo S, Machado RD, Coura JR, Guimarães MAA, Sanseverino SR, Moura A. 1996b. Primary isolation of spotted Fever Group Rickettsiae from Amblyomma cooperi collected from Hydrochaeris hydrochaeris in Brazil. Mem Inst Oswaldo Cruz 91: 273-275.
- Linardi PM, Guimarães LR 2000. Sifonápteros do Brasil, Museu de Zoologia USP/FAPESP, São Paulo, 291p.
- Melles HHB, Colombo S, Silva MV 1992. Febre maculosa: isolamento de Rickettsia em amostra de biopsia de pele. Rev Inst Med Trop São Paulo 34: 37-41.
- Monteiro EVL, Tiriba AC 1976. Rickettsioses. In R Veronesi (ed.), Doenças Infecciosas e Parasitárias, Guanabara Koogan, São Paulo, p. 236-249.
- Moreira JA, Magalhães O 1935. Thypho exanthematico em Minas Gerais. Brasil-Médico 44: 465-470.
- Nascimento EM, Schumaker TTS 2004. Isolamento e identificação de riquétsias no Brasil. Rev Bras Parasitol Vet 13 (Suppl.): 193-196.
- Pacheco RC, Horta MC, Moraes-Filho J, Ataliba AC, Pinter A, Labruna MB. 2007. Rickettsial infection in capybaras (Hydrochaeris hydrochoerus) from São Paulo, Brazil: serological evidence for infection by Rickettsia bellii and Rickettsia parkeri Biomedica 27: 364-371.
- Pacheco RC, Venzal JM, Richtzenhain LJ, Labruna MB 2006. Rickettsia parkeri in Uruguay. Emerg Infec Dis 12: 1804-1805.
- Paddock CD, Sumner JW, Comer JA, Zaki SR, Goldsmith CS, Goddard J, McLellan SL, Tamminga CL, Ohl CA 2004. Rickettsia parkeri: A newly recognized cause of spotted fever rickettsiosis in the United States. Clin Infect Dis 38: 805-811.
- Parola P, Davoust B, Raoult D 2005a. Tick- and flea-borne rickettsial emerging zoonoses. Vet Res 36: 469-492.
- Parola P, Paddock CD, Raoult D 2005b. Tick-borne rickettsioses around the world: emerging diseases challenging old concepts. Clin Microbiol Rev 18: 719-756.
- Pinter A, Labruna MB 2006. Isolation of Rickettsia rickettsii and Rickettsia bellii in cell culture from the tick Amblyomma aureolatum in Brazil. Ann N Y Acad Sci 1078: 523-529.
- Raoult D, La Scola B, Enea M, Fournier PE, Roux V, Fenollar F, Galvão MAM, Lamballerie X 2001. A flea-associates Rickettsia pathogenic for humans. Emerg Infec Dis 7: 73-81.
- Sangioni LA, Horta MC, Vianna MCB, Gennari SM, Soares RM, Galvão MAM, Schumaker TTS, Ferreira F, Vidotto O, Labruna MB 2005. Rickettsial infection in animals and Brazilian spotted fever endemicity. Emerg Infect Dis 11: 265-270.
- Silveira I, Pacheco RC, Szabó MPJ, Ramos HGC, Labruna MB 2007. First report of Rickettsia parkeri in Brazil. Emerg Infect Dis 13: 1111-1113.
- Tiriba AC, Godoy CVF, Brito T, Jordão FM, Penna DO, Souza ART 1968. Febre maculosa em São Paulo. Resultados terapêuticos em alguns casos com laurilsulfato de tetraciclina. Rev Inst Med Trop 10: 256-261.
- Travassos J 1937. Identification d'un virus semblable a celui du " Typhus exanthématique de Sao Paulo ", isolé de la sarigue marsupiale (Didelphis paraguayensis). Compt Rend Soc Biol 126: 1054-1056.
- Vallejo-Freire A 1946. Spotted fever in Mexico. Mem Inst Butantan 19: 159-180.
- Vieira AML, Souza CE, Labruna MB, Mayo RC, Souza SSL, Camargo-Neves VLF 2004. Manual de Vigilância Acarológica, Estado de São Paulo, Secretaria de Estado da Saúde, São Paulo, Brasil, 62 pp.
- Weiss E, Moulder JW 1984. The Rickettsias and Chlamydias. In NR Kreig, JG Holt (eds.), Bergey's manual of systematic bacteriology, Vol. 1, Williams & Wilkins, Baltimore, MD, p. 687-739.
- Whitman TJ, Richards AL, Paddock CD, Tamminga CL, Sniezek PJ, Jiang J, Byers DK, Sanders JW 2007. Rickettsia parkeri infection after tick bite. Virginia Emerg Infect Dis 13: 334-336.
Publication Dates
-
Publication in this collection
26 Feb 2008 -
Date of issue
Nov 2007
History
-
Accepted
05 Nov 2007 -
Received
28 Feb 2007