Abstract
The acute phase of Trypanosoma cruzi infection is associated with a strong inflammatory reaction in the heart characterised by a massive infiltration of immune cells that is dependent on the T. cruzi strain and the host response. 15d-PGJ2 belongs to a new class of anti-inflammatory compounds with possible clinical applications. We evaluated the effects of 15d-PGJ2 administered during the acute phase of T. cruzi infection in mice. Mice were infected with the Colombian strain of T. cruzi and subsequently treated with 15d-PGJ2 repeatedly for seven days. The inflammatory infiltrate was examined by histologic analysis. Slides were immunohistochemically stained to count the number and the relative size of parasite nests. Infection-induced changes in serum cytokine levels were measured by ELISA. The results demonstrated that treatment with 15d-PGJ2 reduced the inflammatory infiltrate in the skeletal muscle at the site of infection and decreased the number of lymphocytes and neutrophils in the blood. In addition, we found that 15d-PGJ2 led to a decrease in the relative volume density of amastigote nests in cardiac muscle. T. cruzi-infected animals treated with 15d-PGJ2 displayed a statistically significant increase in IL-10 levels with no change in IFN-γ levels. Taken together, we demonstrate that treatment with 15d-PGJ2 in the acute phase of Chagas disease led to a controlled immune response with decreased numbers of amastigote nests, as measured by the volume density.
Chagas disease; Trypanosoma cruzi; 15d-PGJ2; PPAR-gamma
ARTICLES
15d-PGJ2 modulates acute immune responses to Trypanosoma cruzi infection
Wellington F RodriguesI; Camila B MiguelII; Javier E Lazo ChicaII; Marcelo H NapimogaI, + + Corresponding author: marcelo.napimoga@uniube.br
ILaboratório de Biopatologia e Biologia Molecular, Universidade de Uberaba, Av. Nenê Sabino 1801, 38055-500 Uberaba, MG, Brasil
IIDepartamento de Biologia Celular, Universidade do Triângulo Mineiro, Uberaba, MG, Brasil
ABSTRACT
The acute phase of Trypanosoma cruzi infection is associated with a strong inflammatory reaction in the heart characterised by a massive infiltration of immune cells that is dependent on the T. cruzi strain and the host response. 15d-PGJ2 belongs to a new class of anti-inflammatory compounds with possible clinical applications. We evaluated the effects of 15d-PGJ2 administered during the acute phase of T. cruzi infection in mice. Mice were infected with the Colombian strain of T. cruzi and subsequently treated with 15d-PGJ2 repeatedly for seven days. The inflammatory infiltrate was examined by histologic analysis. Slides were immunohistochemically stained to count the number and the relative size of parasite nests. Infection-induced changes in serum cytokine levels were measured by ELISA. The results demonstrated that treatment with 15d-PGJ2 reduced the inflammatory infiltrate in the skeletal muscle at the site of infection and decreased the number of lymphocytes and neutrophils in the blood. In addition, we found that 15d-PGJ2 led to a decrease in the relative volume density of amastigote nests in cardiac muscle. T. cruzi-infected animals treated with 15d-PGJ2 displayed a statistically significant increase in IL-10 levels with no change in IFN-γ levels. Taken together, we demonstrate that treatment with 15d-PGJ2 in the acute phase of Chagas disease led to a controlled immune response with decreased numbers of amastigote nests, as measured by the volume density.
Key words: Chagas disease - Trypanosoma cruzi - 15d-PGJ2 - PPAR-gamma
Chagas disease (CD), or American trypanosomiasis, is a disease caused by Trypanosoma cruzi. T. cruzi is a protozoan belonging to the trypanosomatidae family, which has as an invertebrate host, e.g., insects of the order Hemiptera (Triatominae) and some vertebrate hosts, e.g., humans (Chagas 1909). Infection with T. cruzi results in a strong immune response that controls the parasite load but is unable to completely eliminate the infection. Thus, a persistence of the parasite and a chronic disease, known as CD in humans, results (Kotner & Tarleton 2007).
CD remains a major public health concern for Latin American countries. Although a detailed global incidence or prevalence of the infection and disease by an updated epidemiological and serological inquiry is lacking, Dias (2007) estimates that the number of infected individuals in Latin America ranges from 12-14 million people in 18 endemic countries. Statistics show that the clinical severity of the disease often leads to death, indicating that approximately 21,000 people per year die from this disease (Urbina & Do Campo 2003).
After invading a mammalian host, T. cruzi replicates within the cytoplasm of multiple cell types, including macrophages, fibroblasts, skeletal muscle, heart muscle, neuronal cells and epithelial cells. In CD, one major controversial question has been the specificity of the immune response within the myocardium. Two main hypotheses for this ongoing inflammation have been proposed: parasite persistence or autoimmunity (Tarleton & Zhang 1999). Gutierrez et al. (2009) have suggested that both these phenomena co-exist in CD. Thus, the immune evasion strategies of the parasite and defects in homeostasis of the host's immune system, which are largely genetically determined, are both important for the development of clinical symptoms during CD.
The current treatment of CD is controversial because the two available drugs, nifurtimox and benznidazole, are toxic, may be carcinogenic and have poor efficacy against the chronic stage of the disease (Coura & De Castro 2002, Wilkinson & Kelly 2009). On the other hand, it has been demonstrated that inhibitors of the COX-2 enzyme nearly abolish parasitemia (Freire-de-Lima et al. 2000), enhance the release of IL-2 by splenocytes from T. cruzi-infected animals and prolong the survival of infected mice. Furthermore, the release of TNF-α, IFN-γ, IL-4 and IL-10 by Con-A-stimulated splenocytes obtained from infected mice was significantly inhibited by treatment of the animals with salicylate or meloxicam (Michelin et al. 2005). Interestingly, indomethacin binds and activates the peroxisome proliferator-activated receptor γ (PPARγ), a ligand-activated transcription factor known to play a pivotal role in adipogenesis (Lehmann et al. 1997) in addition to the inflammatory process (Di Paola & Cuzzocrea 2007, Napimoga et al. 2008).
PPARs belong to the nuclear hormone receptor superfamily and have been found to regulate inflammation, immunity and metabolism (Bassaganya-Riera et al. 2005, Guri et al. 2006). Functionally, PPARγ downregulates the expression of proinflammatory cytokines by antagonising the activities of transcription factors such as AP-1 and NF-κB (Guri et al. 2006). As a consequence of the important roles, PPARs play in controlling metabolic homeostasis and inflammatory processes, they are often the therapeutic molecular targets for metabolic diseases such as diabetes (Chinetti et al. 2000) and in the treatment of inflammatory disorders (Napimoga et al. 2008, Hassumi et al. 2009, Klotz et al. 2009, Park et al. 2009). In the present paper, we have investigated the effects of the 15d-PGJ2, an agonist of PPAR-γ, on disease outcome and immune function in T. cruzi-infected C57BL/6 mice.
MATERIALS AND METHODS
Animals - Adult male C57BL/6 mice weighing 20-25 g were housed in temperature-controlled rooms (22-25°C) with access to water and food ad libitum. All experiments were conducted in accordance with the National Health guidelines for the welfare of experimental animals and with the approval of the Ethical Committee of the University of Uberaba (# 55/09). None of the animals were used in more than one experimental group.
Parasite strain and mouse infections - To produce an acute inflammatory response of CD, we used the Colombian strain of T. cruzi [strain MHOM/CO/00/Colombian (T. cruzi l)], which was provided by the University of São Paulo (Brazil) and maintained in the department of Cell Biology at the Federal University of Triangulo Mineiro. The mice were infected by subcutaneous injection of 3 x 103 blood-derived trypomastigotes.
Treatment of the infected mice - After three days of infection, a group of eight animals received a subcutaneous administration of 1 mg/kg 15d-PGJ2 (Calbiochem, San Diego, CA, USA) dissolved in DMSO and repeated doses of 15d-PGJ2 were given every 12 h for seven days. This dose of 15d-PGJ2 was chosen based on a previous study showing that it was effective as an anti-inflammatory agent (Napimoga et al. 2008). The control group of infected animals (n = 8) was treated with DMSO only. A second control group received 15d-PGJ2 according to the same schedule but was not infected with T. cruzi (n = 5). After the experimental phase, the animals were euthanised for further analysis.
Parasitemia - Infection with T. cruzi was monitored by visualising the presence of the trypomastigotes in samples of heparin-treated blood from the orbital plexus.
Leucocyte count - Total cells were counted with a Newbauer camera. Differential cell counts (100 cells total) were obtained using blood smear slides stained with Leishman. The results are presented as the percentage of each cell population.
Histological and immunohistochemical studies - The animals were euthanised under deep anaesthesia 11 days after the inoculation with T. cruzi. The heart and skeletal muscle (region of inoculum) were dissected, fixed in 10% buffered neutral formalin for 48 h, briefly washed in running tap water, dehydrated and embedded in paraffin wax. Each sample was sliced into 7-μm sagittal sections. Sections were mounted on glass slides and stained with Giemsa to analyse the inflammatory infiltrate.
Additional sections were mounted on glass slides and used for immunohistochemical analyses. Immunolabelling for T. cruzi antigen was performed using an antibody raised in rabbits (1:250). The slides were pre-treated with 3-aminopropyltriethoxy-silane (Sigma, St. Louis, MO, USA) immersed 3 min in xylene to eliminate the paraffin, dehydrated in absolute alcohol and re-hydrated with Tris-buffered saline (TBS). The sections were rinsed in TBS and immersed in 3% hydrogen peroxide in methanol for 10 min to block the endogenous peroxidase activity followed by 30 min at 90°C to recover the antigen. The slides were then incubated with antibodies directed against T. cruzi for 2 h at 37°C and rinsed three times for 3 min with TBS. After that, the slides were incubated with protein A conjugated to peroxidase (1:500) during 1 h incubation at RT. The slides were washed again and revealed using 3,3-diaminobenzidine tetrahydrochloride (DAB - Sigma) and H2O2. The slides were then counterstained with Mayer's hematoxylin and mounted. Non-specific staining was controlled for by omission of primary antisera.
Histopathologic analysis - Slides stained with Giemsa were subjected to histopathologic analyses in order to determine the level of inflammatory infiltrate in skeletal muscle. Measurements of the inflammatory infiltrate in the skeletal muscle were taken on two slides with four sections each, separated by a distance of 70 μm. Forty random images were analysed using a 10X objective and the results are express as the median ± maximum and minimum values for the total area of inflammatory infiltrate of each animal. The inflammation was obtained through the density of the area which is a parameter that measures the relative occupancy of the tested area (infiltrate) by the total area of the skeletal muscle evaluated (volume density = Vv) (Mandarin-de-Lacerda 2003).
Amastigotes nests - To quantify the number of amastigote nests in heart muscle, 12 randomly selected sections were analysed from both T. cruzi-infected groups (T.cruzi-infected group treated with 15d-PGJ2 and the control, T. cruzi-infected group treated with DMSO). The images were obtained at a final magnification of 40X using a microscope plus camera (Nikon eclipse E200) interfaced with a personal computer using the software Image Pro Plus. Immunolabeled nests were counted and normalised with the total area of heart muscle analysed. The number of the T. cruzi nests were calculated as the ratio of the sum of the nests in the tissue area (mm2) divided by the sum of the heart muscle area examined. Additionally, the relative size of the nests was calculated as the area of the nests normalised to the total heart area analysed (Vv) (Mandarim-de-Lacerda 2003).
Detection of cytokines by ELISA - Serum levels of IFN-γ and IL-10 from infected or control animals were determined by ELISA using protocols supplied by the manufacturer (R&D Systems). The results are expressed in picograms per mL.
Statistical analysis - Statistical analysis was performed using the "Prism" software from GraphPad. The normal distribution and homogeneous variance were tested in all variables. When the distribution was considered normal and the homogeneous variance parametric, ANOVA tests were used to determine statistical significance, followed by Bonferroni test. Values are expressed as the mean and standard deviation. When the adherence proof was done between the groups and the assumption of normality was rejected (p < 0.01), the non-parametric method, the Mann-Whitney test, was used. The values are expressed as the median plus maximum and minimum values. The observed differences were considered significant when p < 0.05.
RESULTS
All of the animals that received an injection of T. cruzi trypomastigotes were positively infected at 11 days post-inoculation, as observed in the microhaematocrit assay. Besides, it was not observed animal mortality during the experimental time.
The analyses of Giemsa-stained sections showed a massive inflammatory infiltrate into the skeletal muscles of animals with acute CD (Fig. 1A). The T.cruzi-infected group treated for seven days with 15d-PGJ2 (1 mg/kg) exhibited less inflammatory infiltrate (Fig. 1B). Normalising the infiltrated area to the total area of skeletal muscle analysed (Vv) confirmed a statically significant decrease in the 15d-PGJ2-treated group compared to control-treated mice (Fig. 1C, D).
Next, we evaluated the number and size of the amastigote nests present in heart muscle by immunohistochemistry. It was possible to observe fewer nests in the Chagas-infected group treated with 15d-PGJ2, in comparison to the Chagas-infected mice treated with vehicle (DMSO), although there was no statistical difference (Fig. 2A). On the other hand, the actual size of the nests present in the myocardium, when normalised to the total area of the heart tissue analysed (Vv), was statistically lower in the 15d-PGJ2-treated group compared to the vehicle-treated group (Fig. 2B). A representative immunolabelled amastigote nest is shown in Fig. 2C.
Because the inflammatory infiltrate was lower in the 15d-PGJ2-treated animals, we evaluated which leukocyte population was altered in the blood. We observed a decrease in the number of lymphocytes (Fig. 3A), albeit without statistical significance (p = 0.06) and a statistically significant decrease in the number of neutrophils in the blood of 15d-PGJ2-treated mice (Fig. 3B). Monocyte numbers were slightly increased in 15d-PGJ2-treated animals in comparison to the other two groups (Fig. 3C).
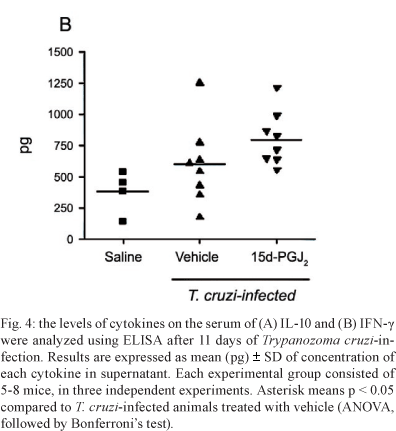
The production of IL-10 in infected mice, treated for seven days with 15d-PGJ2, was significantly increased when compared to IL-10 levels in the serum of either the non-infected group or control-treated T. cruzi-infected mice (Fig. 4A). IFN-γ was increased in the serum of T. cruzi-infected animals that had been treated with 15d-PGJ2, although this difference was not statistically significant when compared to control groups (Fig. 4B).
DISCUSSION
The immune response of the host is an important determinant of the severity and prognosis of CD. However, few therapeutics have been developed that specifically target the deleterious responses of the host to T. cruzi infection. In the present paper, we observed a significant reduction in the level of inflammatory infiltration of skeletal muscles, as well as a decrease in the total area of amastigote nests present in the heart, when normalised to the total area analysed (Vv) in the group treated with 15d-PGJ2.
The major challenge in designing an efficacious treatment for T. cruzi-elicited myocarditis is that any strategy must be able to decrease the intense inflammation, which produces severe tissue damage, without hampering the host's ability to control the parasite load (Marino et al. 2005). The currently available therapeutic options for CD are limited. Most of the therapeutic measures are aimed at treating the consequences of the disease, such as cardiac failure. Although one study showed reduced parasite load, inflammation and conduction disturbances after benznidazole administration to mice chronically infected with T. cruzi (Garcia et al. 2005), there is inconsistent clinical evidence supporting the use of trypanocidal treatment for chronic chagasic cardiomyopathy. Additionally, major economic obstacles impede the development and evaluation of therapeutics intended for diseases that occur predominately in the developing world.
The PPARs, members of the nuclear receptor superfamily, have a newly recognised role in inflammation (Chinetti et al. 1998, 2000). PPARγ is predominantly found at high concentrations in adipocytes, although significant amounts can also be detected in the retina, colonic epithelial cells and cells of the immune system (Bassaganya-Riera et al. 2005). Several studies have demonstrated that its agonist, 15d-PGJ2, may play an important role in regulating inflammation in vivo. Agonists of PPAR are being successfully used with other parasite infections. For instance, a previous study has demonstrated that rosiglitazone improves the outcome of acute malaria infection by increasing macrophage clearance of malaria-parasitised erythrocytes, modulating inflammatory responses to infection and enhancing survival in models of experimental severe and cerebral malaria (Serghides et al. 2009). Recently, a double-blinded randomised placebo-controlled trial study was carried out using rosiglitazone. The authors demonstrated that rosiglitazone was a well-tolerated adjunct to standard therapy for non-severe Plasmodium falciparum malaria. In this case, rosiglitazone enhanced parasite clearance and decreased levels of inflammatory biomarkers that have been associated with adverse malaria outcomes (Boggild et al. 2009). In the present paper, we demonstrate that treatment with 15d-PGJ2 reduces inflammatory infiltration and decreases the size of amastigote nests in the cardiac tissue of T. cruzi-infected animals. A previous study has demonstrated that T. cruzi infection leads to a reduced expression of adiponectin and PPAR-γ and that this reduction perpetuates the inflammatory phenotype found in CD (Nagajyothi et al. 2009). Thus, the use of 15d-PGJ2 to control the inflammatory response associated with CD may be useful.
During the acute phase of T. cruzi infection, an exacerbated immune response is commonly observed in the myocardium of infected individuals. This exaggerated response leads to collateral tissue damage that, in extreme cases, may lead to a systemic inflammatory response and death. Immediately after infection, the recruitment of leucocytes to the infected tissue is triggered. The migration of immune cells depends on the local production of cytokines and chemokines, as well as an increase in the cell surface expression of their associated receptors and adhesion molecules (Gutierrez et al. 2009). Thus, a well-balanced, adaptive immune response plays a critical role in maintaining control of the pathogen, especially during parasitic infections. A major finding of clinical relevance in our study is that administration of 15d-PGJ2 exerted a beneficial effect when given three days after the initial T. cruzi infection. At this timepoint, most of the adverse inflammatory effects of T. cruzi infection have occurred or have started to occur. During experimental CD, the host's resistance is dependent on both innate and acquired immunity and requires the combined effects of a number of different cell types, including NK cells, CD8+ T cells and CD4+ T cells (Medeiros et al. 2009). A predominant Th1 reaction is central to the control of T. cruzi. Treatment of T. cruzi-infected mice with a neutralising mAb against IFN-γ results in exacerbated parasitaemia and mortality (Torrico et al. 1991, Cardillo et al. 1996). Our results demonstrated that, in the acute phase of CD, T. cruzi-infected animals treated with 15d-PGJ2 had a slight increase in the expression of IFN-γ that, while not statistically significant, may explain the observed reduction in the number and size of parasite nests in the tissues.
Furthermore, we show that treatment with 15d-PGJ2 led to increased IL-10 expression, which is in agreement with some studies showing that peripheral mononuclear cells from patients with the indeterminate form of CD produce high levels of IL-10 in addition to IFN-γ and TNF-α. These results are consistent with a modulatory type of immune response (Ribeirao et al. 2000, Abel et al. 2001, Dutra et al. 2009). Importantly, a recent study demonstrated a biased distribution of high IL-10 expression amongst indeterminate chagasic patients. Therefore, the ability to express IL-10 at sufficiently high levels may be genetically determined and may influence the outcome of the disease (Costa et al. 2009). Thus, because IL-10 is consistently associated with the generation of a protective tissue phenotype, the administration of 15d-PGJ2 may be a promising pharmacological tool to treat CD.
In summary, PPAR-γ agonists, such as 15d-PGJ2, represent a novel class of immunomodulatory drugs that may be useful in controlling inflammation during parasitic infections, such as CD.
Received 22 November 2009
Accepted 16 March 2010
Financial support: FAPEMIG (PPM 097/09)
- Abel LC, Rizzo LV, Lanni B, Albuquerque F, Bacal F, Carrara D, Bocchi EA, Teixeira HC, Mady C, Kalil J, Cunha-Neto E 2001. Chronic Chagas' disease cardiomyopathy patients display an increased IFN-gamma response to Trypanosoma cruzi infection. J Autoimmun 17: 99-107.
- Bassaganya-Riera J, Guri A, King J, Hontecillas R 2005. Peroxisome proliferator-activated receptors: the nutritionally controlled molecular networks that integrate inflammation, immunity and metabolism. Curr Nutr Food Sci 1: 179-187.
- Boggild AK, Krudsood S, Patel SN, Serghides L, Tangpukdee N, Katz K, Wilairatana P, Liles WC, Looareesuwan S, Kain KC 2009. Use of peroxisome proliferator-activated receptor gamma agonists as adjunctive treatment for Plasmodium falciparum malaria: a randomized, double-blind, placebo-controlled trial. Clin Infect Dis 49: 841-849.
- Cardillo F, Voltarelli JC, Reed SG, Silva JS 1996. Regulation of Trypanosoma cruzi infection in mice by gamma interferon and interleukin 10: role of NK cells. Infect Immun 64: 128-134.
- Chagas CRJ 1909. Nova tripanosomíase humana. Estudos sobre a morphologia e o ciclo evolutivo do Schizotrypanum cruzi n. gen. n. esp., agente da nova entidade mórbida do homem. Mem Inst Oswaldo Cruz 1: 159-218.
- Chinetti G, Fruchart JC, Staels B 2000. Peroxisome proliferator-activated receptors (PPARs): nuclear receptors at the crossroads between lipid metabolism and inflammation. Inflamm Res 49: 497-505.
- Chinetti G, Griglio S, Antonucci M, Torra IP, Delerive P, Majd Z, Fruchart JC, Chapman J, Najib J, Staels B 1998. Activation of proliferator-activated receptors alpha and gamma induces apoptosis of human monocyte-derived macrophages. J Biol Chem 273: 25573-25580.
- Costa GC, da Costa Rocha MO, Moreira PR, Menezes CA, Silva MR, Gollob KJ, Dutra WO 2009. Functional IL-10 gene polymorphism is associated with Chagas disease cardiomyopathy. J Infect Dis 199: 451-454.
- Coura JR, De Castro SL 2002. A critical review on Chagas disease chemotherapy. Mem Inst Oswaldo Cruz 97: 3-24.
- Dias JC 2007. Southern Cone Initiative for the elimination of domestic populations of Triatoma infestans and the interruption of transfusional Chagas disease. Historical aspects, present situation and perspectives. Mem Inst Oswaldo Cruz 102: 11-18.
- Di Paola R, Cuzzocrea S 2007. Peroxisome proliferator-activated receptors ligands and ischemia-reperfusion injury. Naunyn Schmiedebergs Arch Pharmacol 375: 157-17.
- Dutra WO, Menezes CA, Villani FN, da Costa GC, da Silveira AB, Reis D, Gollob KJ 2009. Cellular and genetic mechanisms involved in the generation of protective and pathogenic immune responses in human Chagas disease. Mem Inst Oswaldo Cruz 104: 208-218.
- Freire-de-Lima CG, Nascimento DO, Soares MBP, De Mello FG, Bozza PT, Castro-Faria-Neto HC, Dosreis GA, Lopes MF 2000. Uptake of apoptotic cells drives the growth of a pathogenic trypanosome in macrophages. Nature 403: 199-203.
- Garcia S, Ramos CO, Senra JF, Senra JFV, Vilas-Boas F, Rodrigues MM, Campos-de-Carvalho AC, Ribeiro-dos-Santos R, Soares MBP 2005. Treatment with benznidazole during the chronic phase of experimental Chagas' disease decreases cardiac alterations. Antimicrob Agents Chemother 49: 1521-1528.
- Guri AJ, Hontecillas R, Bassaganya-Riera J 2006. Peroxisome proliferator-activated receptors: bridging metabolic syndrome with molecular nutrition. Clin Nutr 25: 871-885.
- Gutierrez FR, Guedes PM, Gazzinelli RT, Silva JS 2009. The role of parasite persistence in pathogenesis of Chagas heart disease. Parasite Immunol 31: 673-685.
- Hassumi MY, Silva-Filho VJ, Campos-Júnior JC, Vieira SM, Cunha FQ, Alves PM, Alves JB, Kawai T, Gonçalves RB, Napimoga MH 2009. PPAR-γ agonist rosiglitazone prevents inflammatory periodontal bone loss by inhibiting osteoclastogenesis. Int Immunopharmacol 9: 1150-1158.
- Klotz L, Burgdorf S, Dani I, Saijo K, Flossdorf J, Hucke S, Alferink J, Novak N, Beyer M, Mayer G, Langhans B, Klockgether T, Waisman A, Eberl G, Schultze J, Famulok M, Kolanus W, Glass C, Kurts C, Knolle PA 2009. The nuclear receptor PPAR gamma selectively inhibits Th17 differentiation in a T cell-intrinsic fashion and suppresses CNS autoimmunity. J Exp Med 10: 2079-2089.
- Kotner J, Tarleton R 2007. Endogenous CD25+ regulatory T cells have a limited role in the control of Trypanossoma cruzi infection in mice. Infect Immun 75: 861-869.
- Lehmann JM, Lenhard JM, Oliver BB, Ringold GM, Kliewer SA 1997. Peroxisome proliferator-activated receptors α and γ are activated by indomethacin and other non-steroidal anti-inflammatory drugs. J Biol Chem 272: 3406-3410.
- Mandarim-de-Lacerda CA 2003. Stereological tools in biomedical research. An Acad Bras Cienc 75: 469-486.
- Marino AP, Silva AA, Santos PV, Pinto LM, Gazinelli RT, Teixeira MM, Lannes-Vieira J 2005. CC-chemokine receptors: a potential therapeutic target for Trypanosoma cruzi-elicited myocarditis. Mem Inst Oswaldo Cruz 100: 93-96.
- Medeiros GA, Silvério JC, Marino AP, Roffê E, Vieira V, Kroll-Palhares K, Carvalho CE, Silva AA, Teixeira MM, Lannes-Vieira J 2009. Treatment of chronically Trypanosoma cruzi-infected mice with a CCR1/CCR5 antagonist (Met-RANTES) results in amelioration of cardiac tissue damage. Microbes Infect 11: 264-273.
- Michelin MA, Silva JS, Cunha FQ 2005. Inducible cyclooxygenase released prostaglandin mediates immunosuppression in acute phase of experimental Trypanosoma cruzi infection. Exp Parasitol 111: 71-79.
- Nagajyothi F, Desruisseaux MS, Weiss LM, Chua S, Albanese C, Machado FS, Esper L, Lisanti MP, Teixeira MM, Scherer PE, Tanowitz HB 2009. Chagas disease, adipose tissue and the metabolic syndrome. Mem Inst Oswaldo Cruz 104: 219-225.
- Napimoga MH, Vieira SM, Secco-dal D, Freitas A, Souto FO, Mestriner FL, Filho-Alves JC, Grespan R, Kawai T, Ferreira SH, Cunha FQ 2008. Peroxisome proliferator-activated receptor-γ ligand, 15-deoxy-Δ12,14-prostaglandin J2, reduces neutrophil migration via a nitric oxide pathway. J Immunol 180: 609-617.
- Park SJ, Lee KS, Kim SR, Min KH, Choe YH, Moon H, Chae HJ, Yoo WH, Lee YC 2009. Peroxisome proliferator-activated receptor gamma agonist down-regulates IL-17 expression in a murine model of allergic airway inflammation. J Immunol 5: 3259-3267.
- Ribeirao M, Pereira-Chioccola VL, Renia L, Augusto Fragata Filho A, Schenkman S, Rodrigues MM 2000. Chagasic patients develop a type 1 immune response to Trypanosoma cruzi trans-sialidase. Parasite Immunol 22: 49-53.
- Serghides L, Patel SN, Ayi K, Lu Z, Gowda DC, Liles WC, Kain KC 2009. Rosiglitazone modulates innate immune responses to Plasmodium falciparum and improves outcome in experimental cerebral malaria. J Infect Dis 199: 1536-1545.
- Tarleton RL, Zhang L 1999. Chagas disease etiology: autoimmunity or parasite persistence? Parasitol Today 15: 94-99.
- Torrico F, Heremans H, Rivera MT, Van Marck E, Billiau A, Carlier Y 1991. Endogenous IFN-gamma is required for resistance to acute Trypanosoma cruzi infection in mice. J Immunol 146: 3626-3632.
- Urbina JA, Do Campo R 2003. Specific chemotherapy of Chagas disease: controversies and advances. Trends in Parasitol 19: 495-500.
- Wilkinson SR, Kelly JM 2009. Trypanocidal drugs: mechanisms, resistance and new targets. Expert Rev Mol Med 29: 11-31.
Publication Dates
-
Publication in this collection
26 Apr 2010 -
Date of issue
Mar 2010
History
-
Received
2000 -
Accepted
16 Mar 2010