Abstract
In a large Phase III trial conducted in 10 Latin American countries, the safety and efficacy of the live attenuated monovalent rotavirus vaccine RIX4414 was evaluated in 15,183 healthy infants followed up during the first two years of life. Belém was the only site in Brazil included in this multicentre trial. The study in Belém included a subset of 653 infants who were followed up until 24 months of age for protection against severe rotavirus gastroenteritis. These subjects were randomly assigned in a 1:1 ratio to receive two doses of vaccine (n = 328) or two doses of placebo (n = 325) at approximately two and four months of age. Of the 653 enrolled infants, 23 dropped out during the study period. For the combined two-year period, the efficacy of RIX4414 was 72.3% [95% confidence interval (CI) 37.5-89.1%] against severe rotavirus-related gastroenteritis, reaching a protection rate of 81.8% (95% CI 36.4-96.6%) against circulating wild-type G9 rotavirus strains. It is concluded that two doses of RIX4414 are highly efficacious against severe rotavirus gastroenteritis in Belém during the first two years of life and provide high protection against the worldwide emergence and spread of G9P[8] strains.
rotavirus; gastroenteritis; vaccine efficacy; G9P[8] strains
ARTICLES
Oral live attenuated human rotavirus vaccine (RotarixTM) offers sustained high protection against severe G9P[8] rotavirus gastroenteritis during the first two years of life in Brazilian children
Maria Cleonice A JustinoI; Eliete C AraújoII; Leen-Jan van DoornIII; Consuelo S OliveiraI; Yvone B GabbayI; Joana D'Arc P MascarenhasI; Yllen S MirandaI; Sylvia de Fátima S GuerraI; Veronilce B da SilvaI; Alexandre C LinharesI, + + Corresponding author: alexandrelinhares@iec.pa.gov.br
IInstituto Evandro Chagas, Secretaria de Vigilância em Saúde, Ministério da Saúde, Belém, PA, Brasil
IIUniversidade Federal do Pará, Belém, PA, Brasil
IIIDDL Diagnostic Laboratory, Voorburg, The Netherlands
ABSTRACT
In a large Phase III trial conducted in 10 Latin American countries, the safety and efficacy of the live attenuated monovalent rotavirus vaccine RIX4414 was evaluated in 15,183 healthy infants followed up during the first two years of life. Belém was the only site in Brazil included in this multicentre trial. The study in Belém included a subset of 653 infants who were followed up until 24 months of age for protection against severe rotavirus gastroenteritis. These subjects were randomly assigned in a 1:1 ratio to receive two doses of vaccine (n = 328) or two doses of placebo (n = 325) at approximately two and four months of age. Of the 653 enrolled infants, 23 dropped out during the study period. For the combined two-year period, the efficacy of RIX4414 was 72.3% [95% confidence interval (CI) 37.5-89.1%] against severe rotavirus-related gastroenteritis, reaching a protection rate of 81.8% (95% CI 36.4-96.6%) against circulating wild-type G9 rotavirus strains. It is concluded that two doses of RIX4414 are highly efficacious against severe rotavirus gastroenteritis in Belém during the first two years of life and provide high protection against the worldwide emergence and spread of G9P[8] strains.
Key words: rotavirus - gastroenteritis - vaccine efficacy - G9P[8] strains
Rotavirus infection remains a major cause of severe diarrhoeal disease in young children worldwide and accounts for approximately 2.4 million hospitalisations and > 450,000 deaths each year (Parashar et al. 2009, WHO 2009, Tate et al. 2012). It is estimated that 85% of these deaths occur in developing countries. The highest mortality rates are in low-income areas of Asia and sub-Saharan Africa, where access to medical care is very limited and risk factors for rotavirus infection are significant. According to a recent study in 14 Latin American countries, the burden of disease attributable to rotavirus, in the absence of rotavirus vaccination, has been estimated at 229,656 hospitalisations and 6,302 deaths annually among children younger than five years of age (Desai et al. 2012).
Rotavirus strains are mainly classified by a dual nomenclature system according to two surface proteins, namely VP4 (a protease-sensitive protein) and VP7 (a glycoprotein), defining the P and G protein types, respectively. Currently, 35 P-types and 27 G-types have been reported in mammalian and avian species. Of these, 15 P-types and 12 G-types have been identified in human infections (Matthijnssens et al. 2011). Rotavirus strains bearing G1P[8], G2P[4], G3P[8], G4P[8] and G9P[8] type specificities account for approximately 90% of the cases of rotavirus gastroenteritis worldwide (Santos & Hoshino 2005). Significantly, strains that have G9 combined with either P[8] or P[6] VP4 specificity are currently recognised as one of the globally emerging rotavirus types.
Since 2006, the following two human, live-attenuated oral vaccines have been licensed and are commercially available: a human-bovine pentavalent reassortant rotavirus vaccine (RV) (RotaTeqTM; Merck & Co) and a monovalent vaccine composed of a human G1P[8] rotavirus strain (RotarixTM; GlaxoSmithKline Biologicals). Data from large, pivotal clinical trials that included both vaccines and were conducted in Latin America, the United States and Europe yielded results demonstrating a highly protective efficacy against severe rotavirus gastroenteritis. The results also demonstrated a safety profile that indicated a lower risk of intussusception than that previously reported for RotashieldTM (O'Ryan & Linhares 2009). Of particular importance were the results from two Phase III trials recently completed. In a study performed in South Africa and Malawi (RotarixTM) and another study performed in Kenya, Ghana, Mali, Bangladesh and Vietnam (RotateqTM), an overall lower, though significant, performance was noted compared with that in the pivotal studies (Armah et al. 2010, Madhi et al. 2010, Zaman et al. 2010). A two-dose schedule of RotarixTM, for instance, yielded protective efficacy rates of 72% [95% confidence interval (CI) 40-88%] and 49% (95% CI 11-72%) in South Africa and Malawi, respectively (Madhi et al. 2010). In light of data from the clinical trials conducted to date, rotavirus vaccination is largely recognised as the most effective public health strategy for the prevention of rotavirus disease and vaccination is believed to have a significant impact on the incidence of severe rotavirus gastroenteritis (Bernstein 2009). The World Health Organization (WHO) currently recognises that rotavirus vaccines are crucial in achieving the United Nations' Millennium Development Goal 4 of reducing by two-thirds the mortality rate among children younger than five years of age (United Nations Millennium Development Goals Report 2010) (un.org/millenniumgoals). Furthermore, the WHO has recently recommended the inclusion of rotavirus vaccination of infants in all national immunisation programs (WHO 2009).
During the last decade, large, randomised, double-blind Phase II and III placebo-controlled trials with RotarixTM were conducted in Latin America and Europe, involving over 73,000 healthy infants (Salinas et al. 2005, Ruiz-Palacios et al. 2006, O'Ryan & Linhares 2009). Overall, RotarixTM yielded protection rates of up to 85% (95% CI: 71-93%) and 96% (95% CI: 90-99%) in Latin America and Europe, respectively, in preventing severe rotavirus gastroenteritis graded as ≥ 11 according to the Vesikari score. Of note, in an efficacy subset cohort of 15,183 infants recruited from 10 Latin American countries, a sustained 80% (95% CI: 71.3-87.1%) protective efficacy was achieved by RotarixTM in children younger than two years of age (Linhares et al. 2008). In addition to providing protection against individual G1, G3, G4 and G9 types, efficacy was also significant against pooled non-G1 (G3, G4 and G9) rotavirus serotypes [80% (95% CI: 67.9-88.8%)]. A trend for protection against G2P[4] strains was also observed.
Since 2006, 12 countries in Latin America have incorporated RotarixTM into their national immunisation programs and its positive efficacy under "real conditions" has been demonstrated in recent efficacy studies conducted in Brazil, El Salvador and Panama (Gurgel et al. 2009, Correia et al. 2010, de Palma et al. 2010, Justino et al. 2011, Molto et al. 2011). In addition, vaccination impact studies have demonstrated that RotarixTM is likely to have had a major influence in the decrease in rates of morbidity and mortality in Brazil and Mexico during the post-introduction period (Lanzieri et al. 2010, 2011, Richardson et al. 2010, do Carmo et al. 2011).
Although pivotal clinical trials and post-licensure studies with currently available rotavirus vaccines have clearly documented their effectiveness in protecting against severe rotavirus gastroenteritis, it seems crucial to continue the surveillance of rotavirus disease burden and circulating strains. This surveillance is particularly important in developing countries in which the great diversity of co-circulating rotavirus serotypes, with possible emergence of either reassortant or even potential zoonotic strains, may be potentially challenging for current vaccine strategies (Santos & Hoshino 2005, Matthijnssens et al. 2009).
This paper focuses on the efficacy of RotarixTM in Belém, northern Brazil, one of the 11 settings in Latin America where a major Phase III study was conducted between 2003-2005 to evaluate both the safety and efficacy of this vaccine (Ruiz-Palacios et al. 2006, Linhares et al. 2008). Because G9 rotavirus was the predominant serotype in Belém throughout the study period, we were able to provide further evidence of significant, sustained protection during the first two years of life against severe rotavirus gastroenteritis caused by this globally emergent strain and the VP-7 serotypically unrelated strain.
PATIENTS, MATERIALS AND METHODS
The safety and efficacy of RotarixTM were originally assessed in a large, randomised, double-blind Phase III trial in 11 Latin American countries between August 2003-October 2005. The trial included over 63,000 healthy infants who were followed until they were one year old (Ruiz-Palacios et al. 2006). Of these infants, an efficacy subset cohort of approximately 15,000 infants from 10 Latin American countries was followed up to the age of two years. This follow-up study included the area of Belém (Linhares et al. 2008) where 653 healthy infants were recruited to assess vaccine efficacy up to the second year of life. Inclusion and exclusion criteria were described in detail in a previous publication (Ruiz-Palacios et al. 2006).
Study approvals were granted by the Institutional Ethical Review Committee and the National Ethical Review Board. The trial was carried out in compliance with the October 1996 version of the Helsinki Declaration and the Good Clinical Practices guidelines. Written informed consent was obtained from parents or legal guardians prior to the enrolment of the 653 infants in Belém. Most of the study participants were from low-income families living in the outskirts of Belém, where generally poor sanitation conditions exist. The eligible subjects were healthy full-term infants between six-13 weeks of age. They were randomly assigned in a 1:1 ratio to receive two doses of either RotarixTM (RIX4414 strain) or placebo. Vaccine and placebo compositions are provided in detail elsewhere (Ruiz-Palacios et al. 2006, Linhares et al. 2008). Of note, treatment allocation was concealed from the investigators and the parents/legal guardians of participating infants throughout the entire study period. Two oral doses of RotarixTM or placebo were administered to 328 and 325 healthy infants in Belém, respectively, at approximately two and four months of age. All enrolled infants received other routine childhood immunisations in compliance with the official national schedule. Oral poliovirus vaccine was given at least two weeks before or after the administration of RotarixTM or placebo.
A hospital-based surveillance for severe gastroenteritis of any cause was conducted in Belém during the follow-up period, as described previously (Ruiz-Palacios et al. 2006). In Belém, trained field workers made daily visits to 24 hospitals and 26 official health care units across the city to identify cases of severe gastroenteritis among the enrolled subjects. Cases of severe gastroenteritis were defined as an episode of diarrhoea (the passage of 3 or more looser-than-normal or watery stools within a 24-h period), with or without vomiting, requiring an overnight hospitalisation or rehydration treatment according to the WHO's plan B (oral rehydration therapy) or plan C (intravenous fluid therapy). The severity of gastroenteritis was assessed using the 20-point Vesikari scoring system, where episodes graded ≥ 11 were considered to be severe (Ruuska & Vesikari 1990).
The stool samples were collected from children within 48 h after being hospitalised or at any health unit where they received rehydration therapy. Specimens were stored at 2-8ºC before daily transfer to the Evandro Chagas Institute, Belém, a Ministry of Health's National Rotavirus Reference Laboratory, and frozen at -20ºC until processing. Stool samples were analysed locally (and further retested at the laboratories of GlaxoSmithKline Biologicals in Rixensart, Belgium) to detect rotavirus antigen by means of an enzyme-linked immunobosorbent assay (Rotaclone, Meridian Bioscience, Cincinatti, OH, USA). Rotavirus-positive stool samples were tested using reverse transcriptase-polymerase chain reaction of the VP7 and VP4 genes, followed by reverse hybridisation and (optional) sequencing at DDL Diagnostic Laboratory (Voorburg, The Netherlands) to identify G and P genotypes and to differentiate between the presence of wild-type G1 rotavirus and the vaccine strain virus. The data analysis was performed at GlaxoSmithKline Biologicals, Rixensart, Belgium, using SAS software (version 8.2, SAS Institute Inc, Cary, NC, US) and Proc StatXact 5 on Windows NT (version 4.0) and was described in detail previously (Ruiz-Palacios et al. 2006, Linhares et al. 2008). In brief, the Belém cohort consisted of subjects who completed the full two-dose vaccination course and were followed up from two weeks after dose 2 until approximately 24 months of age. An original sample size [10 Latin American countries (Linhares et al. 2008)] of 5,600 children per group (vaccine and placebo) was estimated (similar numbers for each country were planned). This estimate was determined by assuming a 60% efficacy in the second year efficacy follow-up and an incidence rate of 1% for severe rotavirus gastroenteritis to achieve at least 90% power for the study and a 95% CI for the vaccine efficacy that would be above 0%. In Belém, the overall assumed incidence of rotavirus-related severe gastroenteritis in the placebo group is comparable to that reported elsewhere in Brazil. The vaccine and placebo groups were compared in terms of incidence rate with a two-sided Fisher's exact test (α = 0.05) and vaccine efficacy was calculated with the associated 95% CI computed using the formula (1-relative risk) x 100.
RESULTS
In Belém, a total of 653 infants were enrolled in the study, 630 of whom completed the two-year follow-up to assess vaccine efficacy. There were 23 infants who dropped out of the study because they were unable to attend the final visit of the follow-up period for the following reasons: 11 moved outside the study area, the parents of seven children withdrew consent and five died of vaccine-unrelated causes (Fig. 1). Vaccine and placebo cohorts were shown to be comparable with respect to demographic characteristics that included mean ages at the time of the first and second doses, gender distribution and race or ethnic background. The number of children who completed the two-year efficacy surveillance period was also similar for both the vaccine (n = 319) and the placebo (n = 311) cohorts (Table I).
Table III presents the vaccine protective efficacy data for severe RGE according to the protocol cohort. The frequency of RGE requiring hospital admission or rehydration therapy (WHO plan B or C) was much lower in the first efficacy follow-up period than in the second efficacy follow-up period [7/617 (1.1%) and 29/609 (4.8%), respectively; p = 0.0001]. Combining the first and second efficacy follow-up periods, eight (2.6%) of the 309 children allocated to the vaccine group vs. 28 (9.3%) of the 300 children assigned to the placebo group developed severe rotavirus gastroenteritis, yielding a vaccine efficacy of 72.3% (95% CI 37.5-89.1; p = 0.0005). The vaccine efficacy against severe rotavirus gastroenteritis was 60.6% (95% CI 140.4-96.3; p = 0.283) in the first-year efficacy period and 74.7% (95% CI 36.0-91.6; p = 0.001) in the second-year efficacy period.
Table IV summarises the protective efficacy of RotarixTM against severe RGE caused by viral serotypes G1P[8] and G9P[8], which were found to be the most prevalent strains circulating during the study period. The overall two-year vaccine efficacy against severe rotavirus gastroenteritis caused by the G9P[8] serotype was 82% (95% CI 38-95; p = 0.002). A moderate protection was achieved against G1P[8] illness (51% CI 28-82; p = 0.104), but this result was not statistically significant. Vaccine protection could not be assessed during the first efficacy period because of the small number of cases. In the second follow-up period, a 92% (95% CI 38-99; p = 0.002) protection was achieved against G9P[8]-related severe gastroenteritis, whereas a trend was observed in preventing episodes associated with G1P[8] [47% (95% CI 41-80)].
DISCUSSION
Our study findings indicated that two oral doses of RotarixTM provided a sustained significant protection against severe rotavirus gastroenteritis among children living in Belém during their first two years of life. Of special importance in this study was the good efficacy against G9 rotavirus, a G type currently recognised as a globally emerging strain reported at remarkably increasing frequency worldwide over the past 10 years (Santos & Hoshino 2005, Linhares et al. 2011). Also of importance is the fact that G9 rotaviruses are representative of non-G1 strains, which do not have the same VP7 type specificity as the vaccine strain.
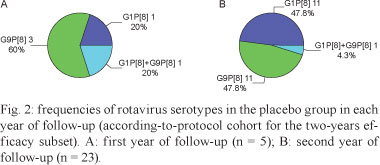
Unlike several other RotarixTM trials, the large predominance of the G9P[8] serotype in Belém during the first (60% of strains) and second (48% of strains) years of follow-up provided a unique opportunity to assess the performance of the vaccine in a setting where a heterotypic rotavirus strain was predominant. In Belém, as in the locations of most RotarixTM efficacy studies all over the world (de Vos et al. 2009), G1P[8] strains were also circulating at high frequencies during the first (20%) of strains and second (48%) years of efficacy surveillance.
Overall, the levels of protection against severe rotavirus gastroenteritis (score ≥ 11 on the Vesikari scoring system) in Belém were similar when comparing the second year of follow-up with the combined two-year follow-up period: 74.7% (95% CI 36.0-91.6; p = 0.001) and 72.3% (95% CI 37.5-89.1; p = 0.0005), respectively. Although vaccination also resulted in an approximately 61% reduction in the incidence of severe rotavirus gastroenteritis during the first year of life, the results were not statistically significant because of the low number of subjects reporting rotavirus illness. The protective efficacy rates in Belém for the second efficacy period and for the two periods combined were similar to those found in a large Phase III trial in Latin America (including Belém as one of the study settings), where the vaccine was 79% (95% CI 66.4-87.4) and 80.5% (95% CI 71.3-87.1) protective in the second efficacy period and the combined two-year efficacy period, respectively (Linhares et al. 2008). Also in accordance with a previously reported vaccine performance in Latin America, the overall level of protection in Belém was lower than that obtained in clinical trials conducted in Europe, where RotarixTM conferred a 90% (95% CI 85-94) protection against severe rotavirus gastroenteritis disease during two consecutive rotavirus seasons (Vesikari et al. 2007, O'Ryan & Linhares 2009). Although statistically significant efficacy data were not available for Belém in the first year of follow-up, due to the small number of cases, it seems likely that the protection rate would be similar to that obtained recently in South Africa [72.2% (95% CI 40.4-88.3)] but higher than that in Malawi [49.2% (95% CI (11.1-71.7)] (Madhi et al. 2010).
Rotavirus type G9P[8] was the predominant strain circulating during the first efficacy follow-up period in Belém, accounting for 60% of cases. In the second year of this study, there was a large increase in the proportion of wild-type G1P[8], reaching a prevalence rate (approximately 48%) similar to that of G9P[8]. Of interest, this changing pattern of rotavirus strains during the two-year follow-up period appears to differ from that observed for Latin America as a whole, where a shift in type predominance was noted as follows: G1P[8] wild-type rotavirus was the most common strain (52% of strains) found during the first rotavirus season, whereas G9P[8] gained predominance (54%) during the second year of the efficacy follow-up period (Linhares et al. 2008). These contrasting findings highlight the fact that changes may occur quickly over time and that different rotavirus strains can become predominant in different geographical locations at the same time (O'Ryan & Linhares 2009).
Several pre-licensure studies have demonstrated the efficacy of RotarixTM against severe rotavirus gastroenteritis caused by the common rotavirus, which bears G1, G3, G4, G9 and P[8] type specificities (Salinas et al. 2005, Ruiz-Palacios et al. 2006, Vesikari et al. 2007, O'Ryan & Linhares 2009). This finding was corroborated using an integrated analysis of vaccine efficacy, showing that RotarixTM confers broad protection against gastroenteritis caused by circulating rotavirus strains with and without the VP7 and VP4 type specificities shared with those of the vaccine strain, including the full heterotypic G2P[4] serotype (de Vos et al. 2009). The fact that such a single-strain (G1P[8]) RV can afford cross-protection early in infancy also became evident in recent, post-vaccine introduction studies performed to assess efficacy "under real conditions" (Snelling et al. 2009, Correia et al. 2010, Justino et al. 2011).
The large predominance of the G9P[8] serotype during the first and second years of the efficacy follow-up for period (60% and 48%, respectively) makes our study in Belém unique compared with other RotarixTM trials, in which most of the isolates belonged to the G1 serotype (O'Ryan & Linhares 2009). It is of major importance in this context to highlight that the number of countries reporting G9 strains has increased remarkably during the past 10 years, suggesting that it is a globally emerging serotype (Gentsch et al. 2005, Santos & Hoshino 2005, Linhares et al. 2011). Notably, studies conducted in Belém and elsewhere have suggested that the G9 serotype appears to be associated with a greater disease severity, particularly with regard to the need for rehydration treatment (Cubbit et al. 2000, Iturriza-Gomara et al. 2000, Linhares et al. 2006). Furthermore, in late 2005, rotavirus genotype G9 was associated with an extensive outbreak of acute gastroenteritis in Rio Branco, state of Acre, in the western Amazon area of Brazil. The outbreak affected more than 20% of children younger than five years old (Siqueira et al. 2010) and caused many deaths.
In a previous Phase II trial conducted in Belém, that followed children until one year of age, RotarixTM was found to be highly efficacious [79.8% (95% CI 26.4-96.3)] against G9 strains of rotavirus (Salinas et al. 2005, Araujo et al. 2007). Of importance is the fact that the results of the present study add evidence to support previous findings that high protection against severe rotavirus gastroenteritis, caused by the emerging G9 serotype, is sustained during the first two years of life [82% (95% CI 38-95)], which is when the disease burden is highest in our region (Linhares et al. 1996).
Although the large studies involving 11 Latin American countries (including Belém, Brazil) (Ruiz-Palacios et al. 2006, Linhares et al. 2008) were not originally powered to assess vaccine efficacy for every individual setting, the results from our current post-hoc analysis clearly demonstrate a significant protection against severe rotavirus gastroenteritis caused by G9 strains during the first two years of life. An additional limitation of our analysis is that protection against serotypes G1 and G9 could not be assessed for the first year of life due to the small number of cases.
The results of this Brazilian subset of the Phase III trial in Belém strengthen those results from previous studies performed in Latin America and elsewhere, showing that two oral doses of RotarixTM given early in infancy provide a sustained level of high protection against severe rotavirus gastroenteritis during the first two years of life (O'Ryan & Linhares 2009). This result is of particular importance considering that the local study was conducted in a setting that clearly reflects the low socio-economic conditions and high diarrhoeal disease incidence typical of developing countries. In conclusion, our study findings demonstrate significant heterotypic rotavirus protection against the globally emerging G9 strain. The protection is sustained for up to two years of age, when the disease burden is highest. Nonetheless, with the increasing worldwide adoption of rotavirus vaccines in national immunisation programs, it is crucial to closely monitor long-term trends of viral serotypes. This monitoring would allow for the assessment of any impact of vaccination on circulating rotaviruses and would also allow for the identification of possible newly emerging strains that may evade immunity to rotavirus vaccines.
Received 27 January 2012
Accepted 18 July 2012
Financial support: IEC, SVS, GlaxoSmithKline Biologicals
- Araujo EC, Clemens S-AC, Oliveira CS, Justino MCA, Rubio P, Gabbay YB, da Silva VB, Mascarenhas JDP, Noronha VL, Clemens R, Gusmão RHP, Sanchez N, Monteiro TAF, Linhares AC 2007. Safety, immunogenicity and protective efficacy of two doses of RIX4414 live attenuated human rotavirus vaccine in healthy infants. J Ped 83: 217-224.
- Armah GE, Sow SO, Breiman RF, Dallas MJ, Tapia MD, Feikin DR, Binka FN, Steele AD, Laserson KF, Ansah NA, Levine MM, Lewis K, Coia ML, Attah-Poku M, Ojwando J, Rivers SB, Victor JC, Nyambane G, Hodgson A, Schödel F, Ciarlet M, Neuzil KM 2010. Efficacy of pentavalent rotavirus vaccine against severe rotavirus gastroenteritis in infants in developing countries in sub-Saharan Africa: a randomised, double-blind, placebo-controlled trial. Lancet 376: 606-614.
- Bernstein DI 2009. Rotavirus overview. Pediatr Infect Dis J 28 (Suppl. 3): S50-S53.
- Correia JB, Patel MM, Nakagomi O, Montenegro FMU, Germano EM, Correia NB, Cuevas LE, Parashar UD, Cunliffe NA, Nakagomi T 2010. Effectiveness of monovalent rotavirus vaccine (Rotarix) against severe diarrhea caused by serotypically unrelated G2P[4] strains in Brazil. J Infect Dis 201: 363-369.
- Cubitt WD, Steele AD, Iturriza-Gomara M 2000. Characterisation of rotaviruses from children treated at a London hospital during 1996: emergence of strains G9P2A[6]. J Med Virol 61: 150-154.
- de Palma O, Cruz L, Ramos H, de Baires A, Villatoro N, Pastor D, de Oliveira LH, Kerin T, Bowen M, Gentsch J, Espósito DH, Parashar U, Tate J, Patel M 2010. Effectiveness of rotavirus vaccination against childhood diarrhoea in El Salvador: case-control study. BMJ 340: c2825.
- de Vos B, Han HH, Bouckenooghe A, Debrus S, Gillard P, Ward R, Cheuvart B 2009. Live attenuated human rotavirus vaccine, RIX4414, provides clinical protection in infants against rotavirus strains with and without shared G and p genotypes. Pediatr Infect Dis J 28: 261-266.
- Desai R, Parashar UD, Lopman B, de Oliveira LH, Clark AD, Sanderson CFB, Tate JE, Matus CR, Andrus JK, Patel MM 2012. Potential intussusception risk versus health benefits from rotavirus vaccination in Latin America. Clin Infect Dis 54: 1397-1405.
- do Carmo GMI, Yen C, Cortes J, Siqueira AA, de Oliveira WK, Cortez-Escalante JJ, Lopman B, Flannery B, de Oliveira LH, Carmo EH, Patel M 2011. Decline in diarrhea mortality and admissions after routine childhood rotavirus immunization in Brazil: a time-series analysis. Plos Med 8: e1001024.
- Gentsch J, Laird AR, Bielfelt B, Griffin DD, Banyai K, Ramachandran M, Jain V, Cunliffe NA, Nakagomi O, Kirkwood CD, Fischer TK, Parashar UD, Bresee JS, Jiang B, Glass RI 2005. Serotype diversity and reassortment between human and animal rotavirus strains: implications for rotavirus vaccine programs. J Infect Dis 192 (Suppl.): S146-S159.
- Gurgel RG, Bohland AK, Vieira SC, Oliveira DM, Fontes PB, Barros VF, Ramos MF, Dove W, Nakagomi T, Nakagomi O, Correia JB, Cunliffe N, Cuevas LE 2009. Incidence of rotavirus and all-cause diarrhea in Northeast Brazil following the introduction of a national vaccination program. Gastroenterology 137: 1970-1975.
- Iturriza-Gomara M, Green J, Brown DWG, Ramsay M, Desselberger U, Gray J 2000. Molecular epidemiology of human group A rotavirus infections in the United Kingdom between 1995 and 1998. J Clin Microbiol 38: 4394-4401.
- Justino MC, Linhares AC, Lanzieri TM, Miranda Y, Mascarenhas JDP, Abreu E, Guerra SFS, Oliveira ASL, da Silva VB, Sanchez N, Meyer N, Shafi F, Ortega-Barria E, Soriano-Gabarró M, Colindres RE 2011. Effectiveness of the monovalent G1P[8] human rotavirus vaccine against hospitalization for severe G2P[4] rotavirus gastroenteritis in Belém, Brazil. Pediatr Infect Dis J 30: 396-401.
- Lanzieri TM, Costa I, Shafi FA, Cunha MH, Ortega-Barria E, Linhares AC, Colindres RE 2010. Trends in hospitalizations from all-cause gastroenteritis in children younger than 5 years of age in Brazil before and after human rotavirus vaccine introduction, 1998-2007. Pediatr Infect Dis J 29: 673-675.
- Lanzieri TM, Linhares AC, Costa I, Kolhe DA, Cunha MH, Ortega-Barria E, Colindres RE 2011. Impact of rotavirus vaccination on childhood deaths from diarrhea in Brazil. Int J Infect Dis 15: e206-210.
- Linhares AC, Gabbay YB, Mascarenhas JDP, Freitas RB de, Oliveira CS, Bellesi N, Monteiro TAF, Lins-Lainson Z, Ramos FLP 1996. Immunogenicity, safety and efficacy of tetravalent rhesus-human, reassortant rotavirus vaccine in Belém, Brazil. Bull World Health Organ 74: 491-500.
- Linhares AC, Stupka, JA, Ciapponi A, Bardach AE, Glujovsky D, Aruj PK, Mazzoni A, Buendía Rodriguez JA, Rearte A, Lanzieri T, Ortega-Barria E, Colindres RE 2011. Burden and typing of rotavirus group A in Latin America and the Caribbean: systematic review and meta-analysis. Rev Med Virol doi: 10.1002/rmv.682.
- Linhares AC, Velázquez FR, Pérez-Schael I, Sáez-Llorens X, Abate H, Espinoza F, López P, Macías-Parra M, Ortega-Barría E, Rivera-Medina DM, Rivera L, Pavía-Ruz N, Nuñez E, Damaso S, Ruiz-Palacios GM, de Vos B, O'Ryan M, Gillard P, Bouckenooghe A 2008. Efficacy and safety of an oral live attenuated human rotavirus vaccine against rotavirus gastroenteritis during the first 2 years of life in Latin American infants: a randomised, double-blind, placebo-controlled phase III study. Lancet 371: 1181-1189.
- Linhares AC, Verstraeten T, Wolleswinkel-van den Bosch J, Clemens R, Breuer T 2006. Rotavirus serotype G9 is associated with more-severe disease in Latin America. CID 43: 312-314.
- Madhi SA, Cunliffe NA, Steele D, Witte D, Kirsten M, Louw C, Ngwira B, Victor JC, Gillard PH, Cheuvart BB, Han HH, Neuzil KM 2010. Effect of human rotavirus vaccine on severe diarrhea in African infants. N Engl J Med 362: 289-298.
- Matthijnssens J, Bilcke J, Ciarlet M, Martella V, Bányai K, Rahman M, Zeller M, Beutels P, Van Damme P, Van Ranst M 2009. Rotavirus disease and vaccination: impact on genotype diversity. Future Microbiol 4: 1303-1316.
- Matthijnssens J, Ciarlet M, McDonald SM, Attoi H, Bányai K, Brister JR, Buesa J, Esona MD, Estes MK, Gentsch JR, Iturriza-Gómara M, Johne R, Kirkwood CD, Martella V, Mertens PPC, Nakagomi O, Parreño V, Rahman M, Ruggeri FM, Saif LJ, Santos N, Steyer A, Taniguchi K, Patton JT, Desselberger U, Van Ranst M 2011. Uniformity of rotavirus strain nomenclature proposed by the Rotavirus Classification Working Group (RCWG). Arch Virol 156: 1397-1413.
- Molto Y, Cortes JE, de Oliveira LH, Mike A, Solis I, Suman O, Coronado L, Patel MM, Parashar UD, Cortese MM 2011. Reduction of diarrhoea-associated hospitalizations among children aged < 5 years in Panama following the introduction of rotavirus vaccine. Pediatric Infect Dis J 30 (Suppl. 1): S16-S20.
- O'Ryan, Linhares AC 2009. Update on RotarixTM: an oral human rotavirus vaccine. Expert Rev Vaccines 8: 1627-1641.
- Parashar UD, Burton A, Lanata C, Boschi-Pinto C, Shibuya K, Steele D, Birmingham M, Glass RI 2009. Global mortality associated with rotavirus disease among children in 2004. J Infect Dis 200 (Suppl. 1): S9-S15.
- Richardson V, Hernández-Pichardo J, Quintanar-Soares M, Esparza-Aguilar M, Johnson B, Gomez-Altamirano CM, Parashar U, Patel M 2010. Effect of rotavirus vaccination on death from childhood diarrhea in Mexico. N Engl J Med 362: 299-305.
- Ruiz-Palacios G, Pérez-Schael I, Velázquez RF, Pérez-Schael I, Sáez-Llorens X, Abate H, Espinoza F, López P, Macías-Parra M, Ortega-Barría E, Rivera-Medina DM, Rivera L, Pavía-Ruz N, Nuñez E, Damaso S, Ruiz-Palacios GM, de Vos B, O'Ryan M, Gillard P, Bouckenooghe A, Human Rotavirus Vaccine Study Group 2006. Safety and efficacy of an attenuated vaccine against severe rotavirus gastroenteritis. N Engl J Med 354: 11-22.
- Ruuska T, Vesikari T 1990. Rotavirus disease in Finnish children: use of numerical scores for clinical severity of diarrhoeal episodes. Scand J Infect Dis 22: 259-267.
- Salinas B, Pérez-Schael I, Linhares AC, Ruiz-Palacios GM, Guerrero ML, Yarzábal JP, Cervantes Y, Costa Clemens SA, Damaso S, Hardt K 2005. Evaluation of safety, immunogenicity and efficacy of an attenuated rotavirus vaccine, RIX4414. Pediatr Infect Dis J 24: 807-816.
- Santos N, Hoshino Y 2005. Global distribution of rotavirus serotypes/genotypes and its implication for the development and implementation of an effective rotavirus vaccine. Rev Med Virol 15: 29-26.
- Siqueira AA, Santelli ACFS, Alencar LR, Dantas MP, Dimech CPN, Carmo GMI, Santos DA, Alves RMS, Lucena MBF, Morais M, Assis RMS, Fialho A, Mascarenhas JDP, Costa M, Linhares AC, Leite JPG, Araujo WN, Hatch DL 2010. Outbreak of acute gastroenteritis in young children with death due to rotavirus genotype G9 in Rio Branco, Brazilian Amazon Region, 2005. Int J Infect Dis 14: e898-903.
- Snelling TL, Schultz R, Graham J, Roseby R, Barnes GL, Andrews RM, Carapetis JR 2009. Rotavirus and the indigenous children of the Australian outback: monovalent vaccine effective in a high-burden setting. CID 49: 428-431.
- Tate JE, Burton HA, Boschi-Pinto C, Steele AD, Duque J, Parashar UD, WHO-coordinated Global Rotavirus Surveillance Network 2012. 2008 estimate of worldwide rotavirus associated mortality in children younger than 5 years before the introduction of universal rotavirus vaccination programmes: a systematic review and meta-analysis. Lancet Infect Dis 12: 136-141.
- Vesikari T, Karvonen A, Prymula R, Schuster V, Tejedor JC, Cohen R, Meurice F, Han HH, Damaso S, Bouckenooghe A 2007. Efficacy of human rotavirus vaccine against rotavirus gastroenteritis during the first 2 years of life in European infants: randomised, double-blind, placebo-controlled study. Lancet 370: 1757-1763.
- WHO - World Health Organization 2009. Meeting of the Immunization Strategic Advisory Group of Experts, October 2009 - conclusions and recommendations. Wkly Epidemiol Rec 84: 518.
- Zaman K, Dang DA, Victor JC, Shin S, Yunus M, Dallas MJ, Podder G, Vu DT, Le TP, Luby SP, Le HT, Coia ML, Lewis K, Rivers SB, Sack DA, Schödel F, Steele AD, Neuzil KM, Ciarlet M 2010. Efficacy of pentavalent rotavirus vaccine against severe rotavirus gastroenteritis in infants in developing countries in Asia: a randomised, double-blind, placebo-controlled trial. Lancet 376: 615-623.
Publication Dates
-
Publication in this collection
09 Nov 2012 -
Date of issue
Nov 2012
History
-
Received
27 Jan 2012 -
Accepted
18 July 2012