Abstracts
The choice for suitable places for female mosquitoes to lay eggs is a key-factor for the survival of immature stages (eggs and larvae). This knowledge stands out in importance concerning the control of disease vectors. The selection of a place for oviposition requires a set of chemical, visual, olfactory and tactile cues that interact with the female before laying eggs, helping the localization of adequate sites for oviposition. The present paper presents a bibliographic revision on the main aspects of semiochemicals in regard to mosquitoes' oviposition, aiding the comprehension of their mechanisms and estimation of their potential as a tool for the monitoring and control of the Culicidae.
Attractancy; repellency; infochemical; mosquito control
A seleção de locais adequados pelas fêmeas de mosquitos para depositarem seus ovos é um fator chave para a sobrevivência de seus imaturos (ovos e larvas). O conhecimento das relações ecológicas implicadas neste processo é de grande importância quando se refere a vetores de agentes patogênicos. A determinação do local de oviposição pelas fêmeas grávidas envolve uma rede de mensagens químicas, visuais, olfativas e táteis que facilitam a localização de lugares adequados para depositarem seus ovos. Neste trabalho é apresentada uma revisão bibliográfica dos principais aspectos relacionados com semioquímicos presentes na oviposição dos mosquitos auxiliando no entendimento dos mecanismos de atuação dos mesmos e potencializando a aplicação destes semioquímicos como uma possível ferramenta de monitoramento e controle de Culicidae.
Ação atraente; ação repelente; infoquímicos; controle de mosquitos
REVIEW
Mario A. Navarro-SilvaI; Francisco A. MarquesIII; Jonny E. Duque LI, II
ILaboratório de Entomologia Médica e Veterinária, Departamento de Zoologia, Universidade Federal do Paraná. Po-box 19020, 81531-980 Curitiba-PR, Brazil. mnavarro@ufpr.br, jonnybiomat@ufpr.br IIBolsista Prodoc/CAPES
IIILaboratório de Ecologia Química e Síntese de Produtos Naturais, Departamento de Química, Universidade Federal do Paraná. tic@quimica.ufpr.br
ABSTRACT
The choice for suitable places for female mosquitoes to lay eggs is a key-factor for the survival of immature stages (eggs and larvae). This knowledge stands out in importance concerning the control of disease vectors. The selection of a place for oviposition requires a set of chemical, visual, olfactory and tactile cues that interact with the female before laying eggs, helping the localization of adequate sites for oviposition. The present paper presents a bibliographic revision on the main aspects of semiochemicals in regard to mosquitoes' oviposition, aiding the comprehension of their mechanisms and estimation of their potential as a tool for the monitoring and control of the Culicidae.
Keywords: Attractancy; repellency; infochemical; mosquito control.
RESUMO
A seleção de locais adequados pelas fêmeas de mosquitos para depositarem seus ovos é um fator chave para a sobrevivência de seus imaturos (ovos e larvas). O conhecimento das relações ecológicas implicadas neste processo é de grande importância quando se refere a vetores de agentes patogênicos. A determinação do local de oviposição pelas fêmeas grávidas envolve uma rede de mensagens químicas, visuais, olfativas e táteis que facilitam a localização de lugares adequados para depositarem seus ovos. Neste trabalho é apresentada uma revisão bibliográfica dos principais aspectos relacionados com semioquímicos presentes na oviposição dos mosquitos auxiliando no entendimento dos mecanismos de atuação dos mesmos e potencializando a aplicação destes semioquímicos como uma possível ferramenta de monitoramento e controle de Culicidae.
Palavras-chave: Ação atraente; ação repelente; infoquímicos; controle de mosquitos.
For the control of the Culicidae there are several methods of chemical and biological control that can be applied with relative success for a rapid decrease in this insect family, which is linked to public health matters. Among them, the ones that provide higher effectivity and reasonable safety for the relationship environment-mankind have the recommendation of the World Health Organization (WHO) for the application in control activities. Then, the most frequently applied methods in the struggle against vectors are the biological insecticides, Bacillus thuringiensis (Bti) and Bacillus sphaericus (Bs), together with chemicals, such as the organophosphorates and pyrethroids (Forattini 2002). However, these approaches confront a great deal of problems that endanger these pests' control, particularly the short effectivity period, the mosquitoes' resistance to the insecticides' active principles, the environments contamination (Stenersen 2004, WHO 1992) and also for not being self-sustainable strategies.
In the last decades, in search for new methodologies that supply the deficiency of the traditional methods, emphasis has been given to methods supported by the theoretical foundations of chemical ecology, especially concerning the semiochemicals. 'Semiochemical' include infochemicals, toxins and nutrients (Dicke & Sabelis 1988). Infochemicals are substances that, in their natural context, carry information or chemical cues for a given interaction between organisms, triggering a behavior or a physiological response in the receiving individual. The infochemicals are subdivided into allelochemicals, related to interspecific communications, and pheromones, in intraspecific communications (Vilela & Della Lucia 2001).
The allelochemicals are subdivided into allomones, whose information's exchange favors the emitting species; kairomones favor the receiving species; synomones favor both species. Also considered allelochemicals are the antimones; substances produced or acquired by an organism that, when in encounter to another individual of a different species in the natural environment, activate in the receiving individual a repellent response to the emitting and receiving individuals. The apneumones are chemical substances emitted by non-living material that evokes a behavioral or physiological reaction that is adaptively favorable to a receiving organism (Vilela & Della Lucia 2001).
Pheromones are defined as substances secreted by an individual for the environment, which are received by a second individual of the same species provoking a specific reaction or defined physiological process. These may include different kinds of behavior, such as sexual, aggregation, dispersion, alarm, territoriality, trail, oviposition and others (Mordue 2003).
The aim of the present work is to provide a bibliographical revision concerning the main aspects related to semiochemicals, especially pheromones, allelochemicals and chemical compounds, that act on the chemical communication and play an important role in the choice of oviposition sites. This should allow a better understanding of the mechanisms involved in the attractancy and repellency of females regarding the oviposition sites, allied to the great potential that the use of semiochemicals provide as monitoring and vector control tools.
Behavior and selection of oviposition sites. The selection of sites for oviposition is a critical factor for the survival and population dynamics of the species. It initiates with the reception of environmental (visual, tactile, and olfactory) stimuli, which may either attract or repel, limiting the possibilities of finding oviposition sites. The cues include color and optical density water, texture and moisture, temperature and reflectance of the oviposition substrate (Bates 1940, Beckel 1955, Fay & Perry 1965, Hazard et al. 1967, Snow 1971, Benzon & Apperson 1988, Bentley & Day 1989, Davis & Bowen 1994, Kline 1994, McCall & Cameron 1995, Bandano & Regidor 2002). Dethier et al. (1960) provided a more accurate description of this behavior. Those authors showed that an attractant ensures to the insect the direction towards a suitable place, inducing oviposition. A repellent is a stimulus that unleashes movements towards an oviposition site, restricting egg-laying.
This behavior occurs because the insect's sensorial system is a complex composed by chemoreceptors, mechanoreceptors, higroreceptors and thermoreceptors. This system can detect a wide range of volatile compounds in the environment that inform qualitative aspects, such as food source, presence of mating partners or suitable oviposition sites (Mordue 2003). These receptors are connected to neurons by specialized setae known as olfactory and gustatory sensillae. The olfactory sensillae occur in pairs and may be observed on the head, antennae and maxillary palpus, including internal and external buccal parts, wing margin and female ovipositors (Romoser & Stoffolano 1998, Dahanukar et al. 2005, Hallem et al. 2006).
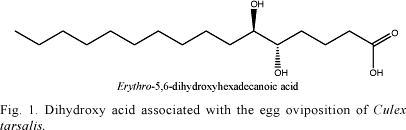
Identification of semiochemicals involved in the selection of oviposition sites. At first several studies performed different experiments trying to evaluate the influence of physic-chemical factors in the oviposition, such as light reflection, odor, temperature, humidity, substrate texture and other breeding sites' features (Gjullim 1961, Gjullim et al. 1965, Fay & Perry 1965, Perry & Fay 1967). Yet, the first scientists to erect a hypothesis concerning the existence of a pheromone that should stimulate oviposition in mosquitoes were Hudson & Mclintock (1967). Later, Osgood (1971) verified this hypothesis studying the behavior of gravid females of Culex tarsalis Coquillet which displayed a preference to lay eggs in water with conspecific larvae, instead of distilled water. With the use of gas-liquid chromatography, Starratt & Osgood (1972) detected the presence of a mixture of 1,3-diglycerides in the active fraction associated with egg oviposition of the mosquito Cx. tarsalis. Acid-catalyzed methanolysis of the mixture yielded methyl esters of mono- and dihydroxy fatty acids being the erythro-5,6-dihydroxyhexadecanoic acid the major component among the dihydroxy ones (Fig. 1).
Bentley et al. (1979) identified p-cresol through gas chromatography coupled with mass spectrometry (GC-MS) in wood infusions that showed to be attractant to females of Aedes triseriatus (Say) (Fig. 2).
Hwang et al. (1980) proved the repellency of the carboxylic acids isobutyric, butyric, isovaleric and hexanoic in the oviposition of Culex quinquefasciatus (Say) (Fig. 3).
Hwang et al. (1982) assessed the repellency and attractancy of a series of carboxylic acids, from pentanoic to tridecanoic, in several concentrations, in Cx. quinquefasciatus, Cx. tarsalis Coquillett and Aedes aegypti (Linnaeus).
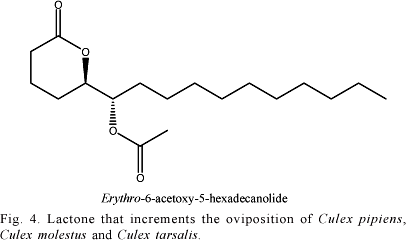
Bruno & Laurence (1979) inferred that the increment observed in the oviposition of Cx. pipiens could occur due to droplets present on the egg's apex, though without specificity, as they were equally attractant to females of Cx. moslestus and Cx. tarsalis. Afterwards, Laurence & Pickett (1982), through gas chromatography-mass spectrometry, proved that the active compound in the droplets observed by Bruno & Laurence (1979) was erythro-6-acetoxy-5-hexadecanolide, supporting the existence of a mosquito oviposition pheromone (MOP) (Fig. 4).
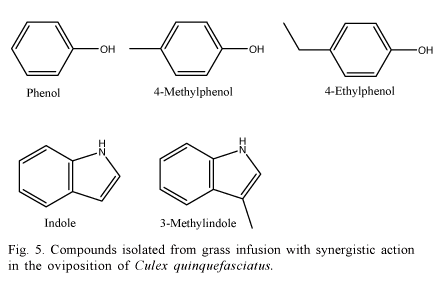
Millar et al. (1992) identified five compounds in an infusion prepared with grass: phenol, 4-methylphenol, 4-ethylphenol, indole and 3-metylindole, indicating a synergistic action in the oviposition stimuli in Cx. quinquefasciatus (Fig. 5).
The 3-metylindole presented higher effectivity to attract mosquitoes when used in concentrations between 1 and 10 ng/L. Millar et al. (1994) also evaluated the effects of the 3-metylindole added to the synthetically derived oviposition pheromone 6-acetoxy-5-hexadecanolide, using combinations of the two products. A mixture of these compounds significantly increased the oviposition of Cx. quinquefasciatus between 0.01 and 0.1 mg; higher to this level, a repellent trend was observed. When separately applied, under this same concentration range, the compounds presented distinct attractancy, thus, determining an additive behavior, not synergistic. Barbosa et al. (2007) demonstrated that high concentrations of synthetic oviposition pheromone (SOP) act as a repellent for oviposition in Cx. quinquefasciatus in laboratory.
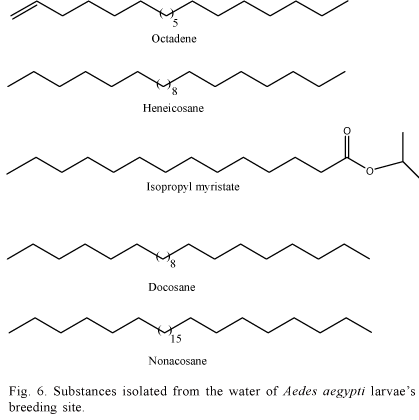
Mendki et al. (2000) identified other five compounds linked to Ae. aegypti, analyzing the water in their larvae's breeding site. The compounds are octadene, isopropyl myristrate, heneicosane, docosane and nonacosane, being the heneicosane the most attractant to oviposition (Fig. 6).
Torres-Estrada et al. (2005) noted that in Anopheles albimanus Wiedemman the oviposition is mediated by the effect of some plants, such as Cynodonton dactylon, Jouvea straminea, Fimbristylis spadicea, Ceratophyllum demersum and Brachiaria mutica. In that study no significant statistical differences in the mosquitoes' attraction to the plant's extracts was observed, nevertheless, a repellent effect was evident with the extracts in high concentrations. As a result, guaiacol, phenol, isoeugenol, longifolene, caryophyllene, phenylethyl alcohol and pcresol were identified, which did not have their biological activities separately determined (Fig. 7).
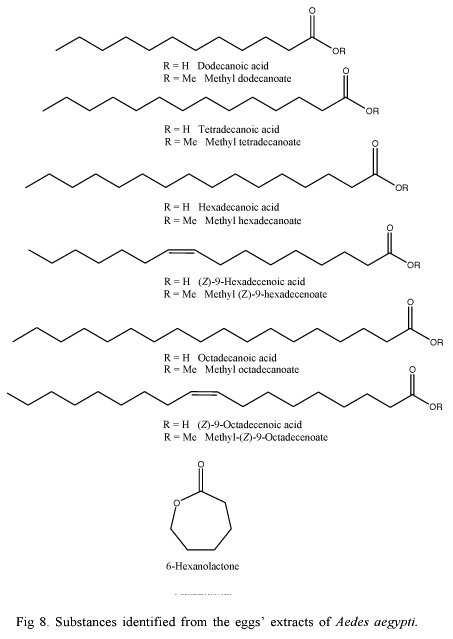
Ganesan et al. (2006), using Ae. aegypti eggs' extracts, identified the dodecanoic, tetradecanoic, hexadecanoic, (Z)-9-hexadecenoic, octadecanoic and (Z)-9-octadecenoic acids, the esters methyl dodecanoate, methyl tetradecanoate, methyl hexadecanoate, methyl octadecanoate, methyl (Z)-9-hexadecenoate, methyl-(Z)-9-octadecenoate and 6-hexanolactone. In the experimental tests, the dodecanoic and (Z)-9-hexadecenoic acids showed positive response to oviposition, whereas the esters showed repellent ovipositional response (Fig. 8).
Sharma et al. (2008) evaluated the oviposition responses of Ae. aegypti and Ae. alpopictus to several C21 fatty acid esters. They observed that hexadecyl pentanoate, tetradecyl heptanoate and tridecyl octanoate presented significant oviposition repellent activity against the two mosquito species, while propyl octadecanoate was found to attract Ae. aegypti to oviposition substrates (Fig. 9).
Ponnusamy et al. (2008) showed that Ae. aegypti females direct most of their eggs to bamboo (Arundinaria gigantean) leaf infusions, due to the oviposition-stimulating kairomones produced by microorganisms. The methanol extract obtained from lyophilized bacteria revealed the presence of a mixture of carboxylic acids from nonanoic to octadecanoid and carboxylic acids methyl esters. Most fatty acids and esters were ineffective, however, others, namely nonanoic acid, tetradecanoic acid and methyl tetradecanoate, were highly effective at inducing egg laying but at extremely narrow dosage ranges (Fig. 10).
Validation of semiochemicals under field conditions. The activity observed under laboratory conditions allowed the advancement towards field work. Beehler et al. (1994) were the first to perform a study in real field conditions using traps with semiochemicals in California, USA. That study confirmed the validity of the laboratory results with skatole (3-methylindole) as an attractant to females of Cx. quinquefasciatus, Cx. stigmatosoma Dyar, and Cx. tarsalis. In Tanzania, Mboera et al. (1999) also evaluated the skatole's residual time for Cx. quinquefasciatus, Cx. tigripes Grandpré & de Chamosy and Cx. cinereus Theobald, besides its effectivity. This study determined that the pheromone is active for up to nine days with a decrease in its activity after this period. It demonstrated, for the first time, the attraction of Cx. cinereus Theobald to the skatole. Moreover, Mboera et al. (2000a) evaluated the ovipositional behavior using skatole and the synthetic oviposition pheromone (SOP) (5R,6S)-6-acetoxy-5-hexadecanolide in field conditions in Tanzania, concluding that both intervene in the selection of sites for oviposition under natural conditions. Mboera et al. (2000b) included the synthetic oviposition pheromone in traps designed by Reiter (1983) for the capture of adult mosquitoes and composed of grass infusion, confirming this pheromone's effectivity as an attractant for gravid females of Cx. quinquefasciatus. This demonstrated its potential to monitor the mosquito. As for other species, such as Ae. albopictus, the results did not yield significant statistical differences either in field or laboratory for the synthetic compounds indole, 3-metylindole and 4-metylindole, suggesting that these compounds are highly specific attractants for Culex (Trexler et al. 2003) (Fig. 11).
New perspectives that can be explored with ovipositional semiochemicals. As a large amount of the documentation found refers to semiochemicals that act in the oviposition of Culex, a study with other culicids is required for a better understanding on the semiochemicals' role in the selection of oviposition sites.
The determination of kairomones in the predator-prey systems may be a vast area to explore new and promising compounds, since females may detect a predator and search for another place. It could work as a females' remover which are ready to lay their eggs in risky areas with the presence of arboviruses like dengue, or other culicid vectors. An example that illustrates this line of investigation is the capacity of Culex spp. to detect predators, such as Notonecta irrorata Fabricius (Hemiptera: Notonectidae) and Culiceta longiareolata Macquart (Diptera; Culicidae) (Blaustein et al. 2004, Blaustein et al. 2005). Studies that could lead to the identification of synomones, antimones and apneumones would be necessary, due to the lack of records on the semiochemicals and their respective mechanisms of activity (Eiras 2001). The development of methodologies to detect and synthesize new compounds should be a priority, most importantly if they are meant to be employed as a large scale tool for monitoring and control (Fuganti et al. 1982, Olagbemiro et al. 1999, Olagbemiro et al. 2004, Michaelakis et al. 2005).
Integrated systems for pest management, which include strategies of attraction towards predefined places for the capture of mosquitoes ("Push Pull strategy") might be the best way of sustainable control of mosquitoes in the future. Under this perspective, compounds with repellent effect would act pushing away vectors from places close to their hosts and attractant compounds would guide them to specific traps for their capture. Such control strategies would require little insecticide or they could even become unnecessary (Cook et al. 2007).
Final considerations. Chemical cues undoubtedly play a crucial role in the selection of oviposition sites and, when adequately applied, may provide promising results in the control and monitoring of mosquitoes' populations. Notwithstanding, the utilization of semiochemicals must be done cautiously in order to avoid undesired repellent effects to the mosquitoes. This could be an unwanted consequence as such populations could disperse to new places, carrying with them aetiological agents that cause diseases.
The semiochemicals may be perceived as a tool that ought to be integrated to other control methodologies with advantages, such as faster detection of circulation sites of arboviruses and a higher selectivity in the monitoring and capture of targeted species. Further, with the utilization of traps for adults, the number of egg-laying females can be inferred, as well as it employs a dynamic control of these populations.
Acknowledgments. We wish to thank our friend Dr. Gustavo Sene Silva for the revision of the English version of our manuscript.
Received 29/04/2008; accepted 23/10/2008
- Badano, E. I. & H. A. Regidor. 2002. Selección de hábitat de oviposición en Aedes aegypti (Diptera: Culicidae) mediante estímulos físicos. Ecología Austral 12: 129-134.
- Barbosa, R. M. R.; S. Souto.; A. E. Eiras & L. Regis. 2007. Laboratory and field evaluation of an oviposition trap for Culex quinquedasciatus (Diptera:Culicidae). Memórias do Instituto Oswaldo Cruz 102: 523-529.
- Bates, M. 1940. Oviposition experiments with Anopheline mosquitoes. American Journal of Tropical Medicine 20: 569-583.
- Beckel, W. E. 1955. Oviposition site preference of Aedes mosquitoes (Culicidae) in the laboratory. Mosquito News 15: 225-228.
- Beehler, J. W.; J. G. Millar & M. S. Mulla. 1994. Field evaluation of synthetic compounds mediating oviposition in Culex mosquitoes (Diptera: Culicidae). Journal of Chemical Ecology 20: 281-291.
- Bentley, M. D. & J. F. Day. 1989. Chemical ecology and behavioral aspects of mosquito oviposition. Annual Review of Entomology 34: 401-421.
- Bentley, M. D.; I. N. Mcdaniel; M. Yatagai; H. P. Lee & R. Maynard. 1979. pCresol: an oviposition attractant of Aedes triseriatus Envirom Entomol 8: 206209.
- Benzon, G. L & C. S Apperson. 1988. Reexamination of chemically mediated oviposition behavior in Aedes aegypti (L.) (Diptera: Culicidae). Journal Medical of Entomology 25: 158-164.
- Blaustein, L.; J. Blaustein & J. Chase. 2005. Chemical detection of the predator Notonecta irrorata by ovipositing Culex mosquitoes. Journal of Vector Ecology 30: 299-301.
- Blaustein, L.; K. Moshe; A. Eitam; M. Mangel & J. E. Cohen. 2004. Oviposition habitat selection in response to risk of predation in temporary pools: mode of detection an consistency across experimental venue. Oecologia 138: 300-305.
- Bruno, D. W & B. R. Laurence. 1979. The influence of the apical droplet of Culex egg rafts on oviposition of Culex pipiens fatigans (Diptera:Culicidae). Journal Medical of Entomology 16: 300-305.
- Cook, S. M.; Z. R. Khan & J. A. Pickett. 2007. The use of Push-pull strategies in integrated pest management. Annual Review of Entomology 52: 375-400.
- Dahanukar, A.; E. A. Hallen & J. R. Carlson. 2005. Insect chemoreception. Current Opinion in Neurobiology 15: 423-430.
- Davis, E. E & M. F. Bowen. 1994. Sensory physiological basis for attraction in mosquitoes. Journal of the American Mosquito Control Association 10: 316-325.
- Dethier, V. G.; L. B. Browne & C. N. Smith. 1960. The designation of chemicals in terms of the responses they elicit from insects. Journal of Economic Entomology 53: 134-36.
- Dicke, M. & M. W. Sabelis. 1988. Infochemical terminology: should it be based on cost-benefit analysis rather than origin of compound? Functional Ecology 2: 131-139.
- Eiras, A. E. 2001. Mediadores químicos entre hospedeiros e insetos vetores de doenças médico veterinárias, cap. 12. p. 99-122. In: Vilela, E. F. & M. T. D. Lúcia (eds.) Ferômonios de insetos: biologia, química e emprego no manejo de pragas Editora Holos, 206 p.
- Fay, R. W. & A. S. Perry. 1965. Laboratory studies of ovipositional preferences of Aedes aegypti Mosquito News 25: 276-281.
- Forattini, O. P. 2002. Culicidologia Médica Editora da Universidade de São Paulo, 860 p.
- Fuganti, C.; P. Grasselli & S. Servi. 1982. Synthesis of the two enantiomeric forms of erythro-6-acetoxy-5-hexadecanolide, the major component of a mosquito oviposition attractant pheromone. Journal of the Chemical Society, Chemical Communications 22: 1285-1286.
- Ganesan, K.; M. J. Mendki; M. V. S. Suryanarayana; S. Prakash & R. C. Malhotra. 2006. Studies of Aedes aegypti (Diptera: Culicidae) ovipositional responses to newly identified semiochemicals from conspecific eggs. Australian Journal of Entomology 45: 75-80.
- Gjullim, C. M. 1961. Oviposition responses of Culex pipiens quinquefasciatus to waters treated with various chemicals. Mosquito News 21: 109-113.
- Gjullin, C. M.; J. O. Johnsen & F. W. Jr. Plapp. 1965. The effect of odors released by various waters on the oviposition sites selected by two species of Culex Mosquito News 25: 268-271.
- Hallem, E. A.; A. Dahanukar & J. R. Carlson. 2006. Insect odor and taste receptors. Annual Review of Entomology 51: 113-135.
- Hazard, E. J.; M. S. Mayer & K. E. Savage. 1967. Attraction and oviposition stimulation of gravid female mosquitoes by bacteria isolated from hay infusions. Mosquito News 27: 133-136.
- Hwang, Y. S.; W. L. Kramer & M. S. Mulla. 1980. Oviposition attractants and repellents of mosquitoes. Journal of Chemical Ecology 6: 71-80.
- Hwang, Y. S.; G. W. Schultz; H. Axelrod; W. L. Kramer & M. S. Mulla. 1982. Ovipositional repellency of fatty acids and their derivatives against Culex and Aedes mosquitoes. Environmental Entomology 11: 223-226.
- Hudson, A & J. Mclintock. 1967. A chemical factor that stimulates oviposition by Culex tarsalis Coquillet (Diptera:Culicidae). Animal Behaviour 15: 336-341.
- Kline, D. L. 1994. Introduction to symposium on attractants for mosquitoes surveillance and control. Journal of the Americam Mosquito Control Association 10: 253-257.
- Laurence, B. R. & J. A. Pickett. 1982. Erythro6Acetoxy5-hexadecanolide, the major component of a mosquito oviposition attractant pheromone. Journal of the Chemical Society, Chemical Communications 1: 59-60.
- McCall, P. J. & M. M. Cameron. 1995. Oviposition pheromones in insect vectors. Parasitology Today 11: 352-355.
- Mendki, M. J.; K. Ganesan; S. Prakash; M. V. S. Suryanarayana; R. C. Malhotra; K. M. Rao & R. Vaidyanathaswamy. 2000. Heneicosane: An oviposition-attractant pheromone of larval origin in Aedes aegypti mosquito. Current Science 78: 1295-1296.
- Mboera, L. E. G.; K. Y. Mdira; F. M. Salud; W. Takken & J. A. Pickett. 1999. Influence of synthetic oviposition pheromone and volatiles from soakage pits and grass infusions upon oviposition site-selection of Culex mosquitoes in Tanzania. Journal of Chemical Ecology 25: 1855-1865.
- Mboera, L. E. G.; W. Takken; K. Y. Mdira & J. A. Pickett. 2000a. Sampling gravid Culex quinquesfasciatus (Diptera:Culicidae) in Tanzania with traps baited with synthetic oviposition pheromone and grass infusions. Journal of Medical Entomology 37: 172-176.
- Mboera, L. E. G.; W. Takken; K. Y. Mdira; G. J. Chuwa & J. A. Pickett. 2000b. Oviposition and behavioral responses of Culex quinquefasciatus to skatole and synthetic oviposition pheromone in Tanzania. Journal of Chemical Ecology 26: 1193-1203.
- Millar, J. G.; J. D. Chaney & M. S. Mulla. 1992. Identification of oviposition attractants for Culex quinquefasciatus from fermented Bermuda grass infusions. Journal of the American Mosquito Control Association 8: 11-17.
- Millar, J. G.; J. D. Chaney; J. W. Beehler & M. S. Mulla. 1994. Interaction of the Culex quinquefasciatus egg raft pheromone whith a natural chemical associated with oviposition sites. Journal of the American Mosquito Control Association 10: 374-379.
- Michaelankis, A.; A. Mihou; E. A. Couladouros; A. K. Zounos & G. Koliopoulos. 2005. Oviposition responses of Culex pipiens to a synthetic racemic Culex quinquefasciatus pheromone oviposition aggregation. Journal of Agricultural and Food Chemistry 53: 5225-5229.
- Mordue, A. J. L. 2003. Arthropod semiochemicals: mosquitoes, midges, and sealice. Part 1. Biochemical society transactions. 31: 128-133.
- Osgood, C. E. 1971. An oviposition pheromone associated with the egg rafts of Culex tarsalis Journal of Economic Entomology 64: 10381041.
- Olagbemiro, T. O.; M. A. Birkett; A. J. Mordue & J. A. Pickett. 1999. Production of (5R,6S)-6acetoxy-5-hexadecanolide, the mosquito oviposition pheromone, from the seed oil of the summer Cypress plant, Kochia scoparia (Chenopodiaceae). Journal of Agricultural and Food Chemistry 47: 3411-3415.
- Olagbemiro, T. O.; M. A. Birkett; A. J. Mordue & J. A. Pickett. 2004. Laboratory and field responses of the mosquito, Culex quinquefasciatus, to plant-derived Culex spp. Oviposition cue skatole. Journal of Chemical Ecology 30: 965-976.
- Perry, A. S. & R. W. Fay. 1967. Correlation of chemical constitution and physical properties of fatty acid esters with oviposition response of Aedes aegypti Mosquito News 27: 175-183.
- Ponnusamy, L.; N. Xu; S. Nojima; D.M. Wesson; C. Schal & C. S. Apperson. 2008. Identification of bacteria and bacteria-associated chemical cues that mediate oviposition site preferences by Aedes aegypti The Proceedings of the National Academy of Sciences 105: 9262-9267.
- Reiter, P. 1983. A portable batterypowered trap for collecting gravid Culex mosquitoes. Mosquito News 43: 496-498.
- Romoser, W. S & J. G. Stoffolano. 1998. The science of Entomology Fourth edition. MacGraw-Hill.USA. 605 pp
- Sharma, K. S.; T. Seenivasagan; A. N. Rao.; K. Ganesan.; O. P. Agarwal; R. C. Malhotra & S. Prakash. 2008. Oviposition responses of Aedes aegypti and Aedes albopictus to certain fatty acid esters. Parasitology Research 103: 1065-1073.
- Snow, W. F. 1971. The spectral sensitivity of Aedes aegypti (L.) at oviposition. Bulletin of Entomological Research 60: 683-696.
- Starratt, A. N & C. E. Osgood. 1972. An oviposition pheromone of the mosquito Culex tarsalis: diglyceride composition of the active fraction. Biochimica et Biophysica Acta 280: 187-193.
- Stenersen, J. 2004. Chemical pesticides: mode of action and toxicology CRC Press. 276 pp.
- Torres-Estrada, J. S.; R. M. Meza-Alvarez; J. Cibrían-Tovar; M. H. Rodríguez-López; J. I. Arredondo-Jiménez; L. Cruz-López & J. C. Rojas-Leon. 2005. Vegetation-derived cues for the selection of oviposition substrates by Anopheles albimanus under laboratory conditions. Journal of the American Mosquito Control Association 21: 344-349.
- Trexler, J. D.; C. S. Apperson; C. Gemeno; M. J. Perich; D. Carlson & C. Schal. 2003. Field and laboratory evaluations of potential oviposition attractats for Aedes albopictus (Diptera: Culicidae). Journal of the American Mosquito Control Association 19: 228-234.
- Vilela, E. F. & M. T. Della Lucia. 2001. Feromônios de insetos: biologia, química e emprego no manejo de pragas. 2Ş edição. 206 p.
- World Health Organization. 1992. Vector Resistance to pesticides. Fifteenth Report of The WHO Expert Commitee on Vector Biology and Control. WHO Tecnical Report Series 818: 1-62.
Review of semiochemicals that mediate the oviposition of mosquitoes: a possible sustainable tool for the control and monitoring of Culicidae
Publication Dates
-
Publication in this collection
04 May 2009 -
Date of issue
2009
History
-
Received
29 Apr 2008 -
Accepted
23 Oct 2008