Abstracts
Intra-puparial development of the females of Chrysomya albiceps (Wiedemann) (Diptera, Calliphoridae). The chronology and morphological changes that take place during intra-puparial development of Chrysomya albiceps is described based on 254 specimens reared in the laboratory. Larvae were obtained from the eggs laid by a single female. The pre-pupae were separated according to the reduction of larval length and the degree of pigmentation and sclerotization of the cuticle. After pupation, 10 individuals were fixed in Carnoy's solution and preserved in 70% ethanol, 10 individuals were fixed every 3 hours up to complete the first 24 hours (n = 80), the remaining individuals were fixed every six hours up to the 90th hour (n = 110) when 54 females emerged. The pupae were immersed in 5% formic acid for 48 hours and maintained in 70% ethanol, and then dissected and analyzed. C. albiceps shows four intra-puparial stages, each of which were described and compared with those described for Musca domestica, Calliphora erythrocephala, Sarcophaga bullata, Cuterebra tenebrosa, Oestrus ovis and Dermatobia hominis. Four developmental stages may be described: (1) the larva-pupa apolysis, after three hours; (2) the criptocephalic pupa, after six hours; (3) the phanerocephalic pupa, after nine hours; (4) the pharate pupa, after nine hours. The pharate adult is completely formed after 81 hours.
Forensic entomology; metamorphosis; morphology; Oestroidea; taxonomy
Desenvolvimento intra-pupal de fêmeas de Chrysomya albiceps (Wiedemann) (Diptera, Calliphoridae) A cronologia e as mudanças morfológicas que ocorrem durante o desenvolvimento intra-pupal de Chrysomya albiceps são descritos com base em 254 espécimes criados em laboratório. As larvas foram obtidas a partir os ovos postos por uma única fêmea. As pré-pupas foram separadas de acordo com a redução do comprimento larval, o grau de pigmentação e esclerotização da cutícula, depois da formação das pupas, 10 indivíduos foram fixados em solução de Carnoy e conservados em etanol 70%, e 10 indivíduos foram fixados a cada 3 horas, até completar as primeiras 24 horas (n = 80), os demais indivíduos foram fixados a cada seis horas até a hora 90 (n = 110), quando 54 fêmeas haviam emergido. As pupas foram imersas em de ácido fórmico a 5% por 48 horas e mantidos em etanol 70%, e então dissecados e analisados. C. albiceps mostrou quatro estágios intra-pupais, que foram descritos e comparados com aqueles descritos para Musca domestica, Calliphora erythrocephala, Sarcophaga bullata, Cuterebra tenebrosa, Oestrus ovis e Dermatobia hominis. Quatro estágios de desenvolvimento podem ser descritos: (1) apólise da larva-pupa, depois de três horas; (2) a pupa criptocefálica, depois de seis horas, (3) a pupa fanerocefálica, depois de nove horas, e a pupa farada após nove horas. O adulto farado está completamente formado após 81 horas.
Entomologia forense; metamorfose; morfologia; Oestroidea; taxonomia
Intra-puparial development of the females of Chrysomya albiceps (Wiedemann) (Diptera, Calliphoridae)
Desenvolvimento intra-pupal de fêmeas de Chrysomya albiceps (Wiedemann) (Diptera, Calliphoridae)
José Roberto Pujol-LuzI; Karine Brenda Barros-CordeiroI
INúcleo de Entomologia Forense, Departamento de Zoologia, Instituto de Ciências Biológicas, Universidade de Brasília, 70910_900 Brasília-DF, Brasil. jrpujol@unb.br, kbbc22@yahoo.com.br
ABSTRACT
Intra-puparial development of the females of Chrysomya albiceps (Wiedemann) (Diptera, Calliphoridae). The chronology and morphological changes that take place during intra-puparial development of Chrysomya albiceps is described based on 254 specimens reared in the laboratory. Larvae were obtained from the eggs laid by a single female. The pre-pupae were separated according to the reduction of larval length and the degree of pigmentation and sclerotization of the cuticle. After pupation, 10 individuals were fixed in Carnoy's solution and preserved in 70% ethanol, 10 individuals were fixed every 3 hours up to complete the first 24 hours (n = 80), the remaining individuals were fixed every six hours up to the 90th hour (n = 110) when 54 females emerged. The pupae were immersed in 5% formic acid for 48 hours and maintained in 70% ethanol, and then dissected and analyzed. C. albiceps shows four intra-puparial stages, each of which were described and compared with those described for Musca domestica, Calliphora erythrocephala, Sarcophaga bullata, Cuterebra tenebrosa, Oestrus ovis and Dermatobia hominis. Four developmental stages may be described: (1) the larva-pupa apolysis, after three hours; (2) the criptocephalic pupa, after six hours; (3) the phanerocephalic pupa, after nine hours; (4) the pharate pupa, after nine hours. The pharate adult is completely formed after 81 hours.
KEYWORDS. Forensic entomology; metamorphosis; morphology; Oestroidea, taxonomy.
RESUMO
Desenvolvimento intra-pupal de fêmeas de Chrysomya albiceps (Wiedemann) (Diptera, Calliphoridae) A cronologia e as mudanças morfológicas que ocorrem durante o desenvolvimento intra-pupal de Chrysomya albiceps são descritos com base em 254 espécimes criados em laboratório. As larvas foram obtidas a partir os ovos postos por uma única fêmea. As pré-pupas foram separadas de acordo com a redução do comprimento larval, o grau de pigmentação e esclerotização da cutícula, depois da formação das pupas, 10 indivíduos foram fixados em solução de Carnoy e conservados em etanol 70%, e 10 indivíduos foram fixados a cada 3 horas, até completar as primeiras 24 horas (n = 80), os demais indivíduos foram fixados a cada seis horas até a hora 90 (n = 110), quando 54 fêmeas haviam emergido. As pupas foram imersas em de ácido fórmico a 5% por 48 horas e mantidos em etanol 70%, e então dissecados e analisados. C. albiceps mostrou quatro estágios intra-pupais, que foram descritos e comparados com aqueles descritos para Musca domestica, Calliphora erythrocephala, Sarcophaga bullata, Cuterebra tenebrosa, Oestrus ovis e Dermatobia hominis. Quatro estágios de desenvolvimento podem ser descritos: (1) apólise da larva-pupa, depois de três horas; (2) a pupa criptocefálica, depois de seis horas, (3) a pupa fanerocefálica, depois de nove horas, e a pupa farada após nove horas. O adulto farado está completamente formado após 81 horas.
PALAVRAS-CHAVE. Entomologia forense; metamorfose; morfologia; Oestroidea; taxonomia.
The process of molting in holometabolous insects culminates in radical physiological, anatomical, and structural changes. Some aspects and concepts associated to intra-puparial development and metamorphic events, as well as the terminology applied to the stages were discussed and revised in Fraenkel & Bhaskaran (1973) (Diptera) and Costa & Vanin (1985) (Coleoptera, Hymenoptera and Diptera).
In Diptera, the behavior during metamorphosis (Denlinger & Zdárek 1994) and the metamorphic events _ pupariation (the puparium formation) and the puparium (formed from the cuticle of the third instar larva), which provides a casing for the pupa _, were studied by many authors. Most studies on Diptera are concentrated on Muscoidea, and, mainly in Oestroidea (Colwell et al., 2006), e.g., Wolfe, 1954 (Calliphora erythrocephala Macquart, 1834); Bennett 1962 (Cephenemya phobifera (Clark, 1815)); Fraenkel & Bhaskaran 1973 (Musca domestica L., 1758, Sarcophaga bullata (Parker, 1916)); Baird 1972, 1975 (Cuterebra tenebrosa Coquillet, 1898); Scholl 1991 (Cuterebra fontinella Clark, 1827); Lello et al. 1985 (Dermatobia hominis (Linnaeus Jr., 1781)); Scholl & Weintraub 1988 (Hypoderma lineatum (Viller, 1789), Hypoderma bovis (L., 1761)); Cepeda-Palacios & Scholl 2000 (Oestrus ovis L., 1758)).
Studies on the biology, life cycle and ecology of necrophagous insects in the Neotropics are still wanting. This may be associated to the complexity of these studies, the high cost of setting up laboratory conditions, and delay in obtaining results (Pujol-Luz et al. 2008). Blowflies constitute one of the most important necrophagous insects in the Neotropics. These flies are mechanical vectors of several pathogens and cause several diseases such as myiasis to humans and other animals. Among members of this diverse family of flies, Chrysomya albiceps (Wiedemann, 1819) is one of the most studied blowflies, not only because its importance in human and animal healthy, but also because it also represents an important forensic entomology indicator (Zumpt 1965; Guimarães et al. 1983; Marchenko 1985, 2001; Hall & Wall 1995; Biavatti et al. 2010; Papavero et al. 2010).
In this paper we described the chronology and morfological chances observed in the intra-puparial development of C. albiceps. The results are compared with other muscoid and oestroid-flies.
MATERIAL AND METHODS
The third instar larvae (L3) were originated from the eggs laid by a female (n = 254 females) collected in the city of Santa Maria, State of Goiás, Brazil. All the larvae were kept in a climatized chamber (B.O.D.) at a temperature of 26 ± 1.0°C, humidity 60 ± 10% and photoperiod of 12:12 (L:D) and were monitored as soon as they ceased feeding, in order to observe the pupariation process. The pre-pupae were separated according to the reduction of larval length and the degree of pigmentation and sclerotization of the cuticle. Ten pupae were fixed in Carnoys solution and preserved in 70% ethanol; 10 pupae were fixed every 3 hours until complete the first 24 hours (n = 80); the remainder pupae were fixated every six hours until the 90th hour (n = 110) when the adults emerged. After fixation, the pupae were immersed in 5% formic acid for 48 hours and maintained in 70% ethanol, and then dissected and analysed. The emerged adults (54 females) and puparia were killed by cold (-20°C) and preserved in 70% ethanol. The terminology of the phases of intra-puparial development and definitions employed herein to describe the pupariation and pupation processes and of the puparium's morphology follows Fraenkel & Bhaskaran (1973) and Cepeda-Palacios & Scholl (2000), and are as follows: (i) Pupariation: The period comprehended between the time the larvae ceased feeding to the complete immobilization and the reduction of length of the larvae. A reduction of their mobility and a retraction of the segments gradually occur. The cuticle becomes progressively more opaque, pigmented and sclerotized. (ii) Larva-pupa apolysis: Following the pupariation process, the larva-pupa apolysis takes place, resulting in the formation of the adult epidermis and its separation from the last larval skin which will form the puparium. (iii) Cryptocephalic pupa: A phase also known as hidden head; in this phase it is not possible to see the head nor the thoracic appendages externally; the imaginal discs of the appendages and the cephalic vesicle (cerebral and cephalic ganglia) are located below the thoracic segments. (iv) Phanerocephalic: In this phase occurs the extroversion, external visualization of the cephalic capsule and the thoracic appendages. It also marks the process of apolysis between pupa and adult. (v) Pharate adult: The longest phase of intrapupal development, corresponding to the adult maturation. (vi) Imago: The definite form of the insect after metamorphosis. All the material was deposited in the Coleção Entomológica do Departamento de Zoologia da Universidade de Brasília.
RESULTS
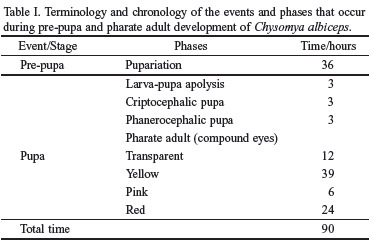
Pupariation. Once they stop feeding, the larvae remained active for several hours on top of the vermiculite; then they begin to burrow themselves into the substrate, with the help of their mouth-hooks. During the process of retraction of the segments, the first segment suffers invagination towards the second segment. The cuticle varies in pigmentation (red and black) and sclerotization. The posterior spiracles collapse and become fused to the anal tubercle, but maintain the shape they had in the preceding stage. The pupae assume a barrel shape, and reduces its size to about 56% of the length of the L3. The process of pupariation takes 36 hours and the average length of the pupae is 9,39 ± 0,29 mm (Table I).
Larva-pupa apolysis. The puparium is formed from the skin of the last larval instar (L3). In C. albiceps this process occurs initially from the middle portion of the puparium towards its extremities, and from the ventral to the dorsal regions. The extremities remain fastened in the anterior region by the maxilla and mandible and in the posterior region by the spiracles. The complete apolysis larva-pupa lasts three hours (Table I).
Cryptocephalic pupa (Figs. 1a-b). Once the process of apolysis stops, the cuticle of the last larval instar forms a hardened and pigmented puparium. The maxilla and the mandible become separated from the rest of the cephalopharyngeal skeleton and remain attached to the puparium; the remaining structures of the skeleton are still observed on the larva, which now has an undefined form and is surrounded by a thin membrane. This stage lasts at least three hours and ends up when the process of extroversion of the head and the thoracic appendages begins (Table I).
Phanerocephalic pupa (Figs. 1c-d): It is a very rapid process. The thoracic appendages begin their invagination before the cephalic capsule does it. During this phase it is already possible to distinguish the head, thorax and abdomen of the imago. In the mesothorax the anterior spiracles of the adult are formed. Each spiracle forms a short, cylindrical, trumpet-like lateral projection in the thorax. This process marks the apolysis pupa-adult (Table I).
Pharate adult (Figs. 2a-b). This phase may be divided in four sub-phases according to the color of the compound eyes (Cepeda-Palacios & Scholl 2000): (1) Transparent eyes: the definition of head, thorax, abdomen, legs and (non-membranous) wings happens during this phase, between the 9th and the 21st hours; (2) Yellow eyes: the thoracic and abdominal sutures may be discerned; the terminalia is also visible. It occurs between the 21st and the 60th hours; (3) Pink eyes: one may observe the antennae, the beginning of the pigmentation of the hairs, bristles, legs and wing veins, as well as the definition of the sex. It occurs between the 60th and the 66th hours; and, (4) Red eyes: the body is completely formed; the antennae, palpi and ocelli are visible; all the hairs and bristles are pigmented; the wings become membranous and the veins are darkened; the external genital organs are visible; the sclerites are delimited and the ptilineal sac is formed. It occurs between the 66th and the 90th hours. According to the eye coloration, the body structures gradually become more sclerotized and pigmented (Table I).
Imago and emergence (Figs. 2c-d). The imagoes were observed in the 84th hour, within the puparium, with the ptilineal sac completely formed and inflated. The adults emerged after the 90th hour (this period is counted since the beginning of the pupal stage).
DISCUSSION
According to Fraenkel & Bhaskaran (1973) the larva-pupa apolysis in S. bullata, at 24°C, lasts 28 hours, corresponding to about 8% of the total time of development. This same phase, in C. tenebrosa, at 21_23°C, lasts 5_6 days, H" 6_11% (Baird 1975); in D. hominis it lasts 3_5 days, H" 8_16% (Lello et al. 1985); in O. ovis, at 16_32°C, lasts 18 hours, H" 3% (Cepeda-Palacios & Scholl 2000). In this study, C. albiceps it lasts 3 hours, H" 3% of the time of intra-puparial development (Table II).
The pharate adult is the period when the insect spends most of its time in the puparium. In S. bullata it begins after the 72nd hour, H" 22% of the pupal period (Fraenkel & Bhaskaran 1973). According to Cepeda-Palacios & Scholl (2000), there are four sub-phases in O. ovis according to the pigmentation of the compound eyes: (i) white eyes occurred between H" 31_63%, (ii) orange eyes between H" 64_77%, (iii) red eyes between H" 81_95% e (iv) brown eyes between H" 95_100% of the time. In C. albiceps we also observed the four phases as pointed out (i) transparent eyes occurred between H" 10_23%, (ii) yellow eyes between H" 22_66%, (iii) pink eyes between H" 67_73% and (iv) red eyes between H" 74_100% (Table II).
The pupal stage of C. albiceps lasted about 90 hours. We recognized four discrete phases in the continuum processes of the intra-puparial formation of the adult: (1) the larva-pupa apolysis, which occurred after three hours; (2) the criptocephalic pupa, wich occurred after six hours, (3) the phanerocephalic pupa, wich occurred after nine hours, and the pharate adult, which begun its maturation after nine hours; and (4) the pharate adult, was totally formed after 81 hours. The intra-puparial development should be related to the feeding habits or with the reproductive strategy. The obligatory parasites flies (e.g. D. hominis; C. tenebrosa and O. ovis) shows a much larger intra-pupal development than the others.
ACKNOWLEDGEMENTS
The authors thanks to Hélio Ricardo da Silva and Carlos Eduardo Guimarães Pinheiro for reviewing the manuscript. To Conselho Nacional de Desenvolvimento Científico e Tecnológico/CNPq and Fundação de Amparo à Pesquisa do Distrito Federal (process FAP/DF 193.000563/2009) (JRPL) for grants.
REFERENCES
Baird, C. R. 1972. Termination of pupal diapauses in Cuterebra tenebrosa (Diptera: Cuterebridae) with injections of ecdysterone. Journal of Medical Entomology 9: 77-80.
Baird, C. R. 1975. Larval development of the rodent botfly, Cuterebra tenebrosa, in bushy-tailed wood rats and its relationship to pupal diapause. Canadian Journal of Zoology 53: 1788-1798.
Bennett, G. F. 1962. On the biology of Cephenemya phobifera (Diptera: Oestridae), the pharyngeal bot of the white-tailed deer, Odocoileus virginianus. Canadian Journal of Zoology 40: 1195-1210.
Biavati, G. M.; F. H. de A. Santana & J. R. Pujol-Luz. 2010. A Checklist of Calliphoridae Blowflies (Insecta, Diptera) Associated with a Pig Carrion in Central Brazil. Journal of Forensic Sciences55: 1603-1606.
Cepeda-Palacios, R. & P. J. Scholl. 2000. Intra-puparial development in Oestrus ovis (Diptera: Oestridae). Journal of Medicine Entomology 37: 239-245.
Colwell, D. D.; M. J. R. Hall & P. J. Scholl. 2006. The Oestrid Flies. Biology, host-parasite relationships, impact and management. London, CABI Publishing, x+359 p.
Costa, C. & S. A. Vanin. 1985. On the concepts of "pre-pupa", with special reference to the Coleoptera. Revista Brasileira de Zoologia 2: 339-345.
Denlinger, D. L. & J. Zdárek. 1994. Metamorphosis behavior of flies. Annual Review Entomology 39: 243-266.
Fraenkel, G. & G. Bhaskaran. 1973. Pupariation and pupation in cyclorraphous flies (Diptera): terminology and interpretation. Annals of the Entomological Society of America 66: 418-422.
Guimarães, J. H.; N. Papavero & A. P. do Prado. 1983. As miíases na região neotropical (identificação, biologia, bibliografia). Revista Brasileira de Zoologia 1: 239-416.
Hall, M. J. R. & R. Wall. 1995. Myiasis of humans and domestic animals. Advances in Parasitology 35: 257-334.
Lello, E. de.; E. A. Gregório & L. A. Toledo. 1985. Desenvolvimento das gônadas de Dermatobia hominis (Diptera: Cuterebridae). Memórias do Instituto Oswaldo Cruz 80: 159-170.
Marchenko, M. I. 1985. Characteristic of development of the fly Chrysomya albiceps (Wd.) (Diptera, Calliphoridae). Entomologicheskoe Obozrenie 64: 79-84.
Marchenko, M. I. 2001. Medicolegal relevance of cadaver entomofauna for the determination of the time of death. Forensic Science International 120: 89-109.
Papavero, N; J. R. Pujol-Luz & M. D. M. Teixeira. 2010. From Homer to Redi. Some historical notes about the problem of necrophagous blowfliesc reprodution. Arquivos de Zoologia 41: 153-170.
Pujol-Luz, J. R.; L. C. Arantes & R. Constantino. 2008. Cem anos da Entomologia Forense no Brasil (1908-2008). Revista Brasileira de Entomologia 52: 485-492.
Scholl, P. J. & J. Weintraub. 1988. Gonotrophic development in Hypoderma lineatum and H. bovis (Diptera: Oestridae), with notes on reproductive capacity. Annals of the Entomological Society of America 81: 318-324.
Scholl, P. J. 1991. Gonotrophic development in the rodent bot fly Cuterebra fontinella (Diptera: Oestridae). Jounal of Medical Entomology 28: 474-476.
Wolfe L. S. 1954. Studies of the development of the imaginal cuticle of Calliphora erythrocephala. Quarterly Journal of Microscopical Science 95: 67-78.
Zumpt, F. 1965. Myiasis in man and animals in the Old World. London, Butterworths Press, xv+267 p.
Received 29/11/2011; accepted 27/6/2012
Editor: Marcia Souto Couri
- Baird, C. R. 1972. Termination of pupal diapauses in Cuterebra tenebrosa (Diptera: Cuterebridae) with injections of ecdysterone. Journal of Medical Entomology 9: 77-80.
- Baird, C. R. 1975. Larval development of the rodent botfly, Cuterebra tenebrosa, in bushy-tailed wood rats and its relationship to pupal diapause. Canadian Journal of Zoology 53: 1788-1798.
- Bennett, G. F. 1962. On the biology of Cephenemya phobifera (Diptera: Oestridae), the pharyngeal bot of the white-tailed deer, Odocoileus virginianus. Canadian Journal of Zoology 40: 1195-1210.
- Biavati, G. M.; F. H. de A. Santana & J. R. Pujol-Luz. 2010. A Checklist of Calliphoridae Blowflies (Insecta, Diptera) Associated with a Pig Carrion in Central Brazil. Journal of Forensic Sciences 55: 1603-1606.
- Cepeda-Palacios, R. & P. J. Scholl. 2000. Intra-puparial development in Oestrus ovis (Diptera: Oestridae). Journal of Medicine Entomology 37: 239-245.
- Colwell, D. D.; M. J. R. Hall & P. J. Scholl. 2006. The Oestrid Flies. Biology, host-parasite relationships, impact and management London, CABI Publishing, x+359 p.
- Costa, C. & S. A. Vanin. 1985. On the concepts of "pre-pupa", with special reference to the Coleoptera. Revista Brasileira de Zoologia 2: 339-345.
- Denlinger, D. L. & J. Zdárek. 1994. Metamorphosis behavior of flies. Annual Review Entomology 39: 243-266.
- Fraenkel, G. & G. Bhaskaran. 1973. Pupariation and pupation in cyclorraphous flies (Diptera): terminology and interpretation. Annals of the Entomological Society of America 66: 418-422.
- Guimarães, J. H.; N. Papavero & A. P. do Prado. 1983. As miíases na região neotropical (identificação, biologia, bibliografia). Revista Brasileira de Zoologia 1: 239-416.
- Hall, M. J. R. & R. Wall. 1995. Myiasis of humans and domestic animals. Advances in Parasitology 35: 257-334.
- Lello, E. de.; E. A. Gregório & L. A. Toledo. 1985. Desenvolvimento das gônadas de Dermatobia hominis (Diptera: Cuterebridae). Memórias do Instituto Oswaldo Cruz 80: 159-170.
- Marchenko, M. I. 1985. Characteristic of development of the fly Chrysomya albiceps (Wd.) (Diptera, Calliphoridae). Entomologicheskoe Obozrenie 64: 79-84.
- Marchenko, M. I. 2001. Medicolegal relevance of cadaver entomofauna for the determination of the time of death. Forensic Science International 120: 89-109.
- Papavero, N; J. R. Pujol-Luz & M. D. M. Teixeira. 2010. From Homer to Redi. Some historical notes about the problem of necrophagous blowfliesc reprodution. Arquivos de Zoologia 41: 153-170.
- Pujol-Luz, J. R.; L. C. Arantes & R. Constantino. 2008. Cem anos da Entomologia Forense no Brasil (1908-2008). Revista Brasileira de Entomologia 52: 485-492.
- Scholl, P. J. & J. Weintraub. 1988. Gonotrophic development in Hypoderma lineatum and H. bovis (Diptera: Oestridae), with notes on reproductive capacity. Annals of the Entomological Society of America 81: 318-324.
- Scholl, P. J. 1991. Gonotrophic development in the rodent bot fly Cuterebra fontinella (Diptera: Oestridae). Jounal of Medical Entomology 28: 474-476.
- Wolfe L. S. 1954. Studies of the development of the imaginal cuticle of Calliphora erythrocephala Quarterly Journal of Microscopical Science 95: 67-78.
- Zumpt, F. 1965. Myiasis in man and animals in the Old World London, Butterworths Press, xv+267 p.
Publication Dates
-
Publication in this collection
06 Sept 2012 -
Date of issue
Sept 2012
History
-
Received
29 Nov 2011 -
Reviewed
27 June 2012