Abstract
A species' mating system depends on its spatial distribution and temporal availability of mating opportunities, as well as on the resources that create these opportunities. In addition, for many species, courtship is driven by specific behaviors that precede and follow copulation. Although Sphex ingens is a taxonomically well known species of digger wasp, its ecology and behavior remain poorly known. Hence, we analyzed patterns and trends of sexual behavior, in order to understand whether courtship can persist in a polygamous mating system. We monitored by video wasp populations in Ilha Grande, southeastern Brazil. Based on the observed behaviors, we calculated stochastic probabilities with a Markov chain to infer on behavioral trends. We recorded four behavioral phases based on 19,196 behavioral acts observed in 224 copulation attempts. There were no significant differences in common behavioral acts between males and females. The copulation patterns, conflicts, and trends observed in S. ingens clearly show the influence of sexual selection in its promiscuous mating system.
Copulation; Coercive copulation; Breeding season; Praia do Aventureiro; Solitary wasp
Introduction
In several animal species, mating systems are affected by food availability and distribution, breeding sites, and potential partners. The courtship and copulation phases are observed in most mating events. Currently, there is consensus on the conflicts of interest between sexes during mating, and sexual selection is an important mechanism in the choice of an ideal partner.
Among hymenopterans, the spheciform complex (Ampulicidae, Crabronidae, and Sphecidae sensu Melo, 1999) comprises wasps that are usually solitary and have a broad behavioral repertoire, especially in terms of reproductive and nesting behaviors ( Evans and O'Neill, 1978Evans, H.E., O'Neill, K.M., 1978. Alternative mating strategies in digger wasp Philan- thus zebratus Cresson. Proc. Natl. Acad. Sci. U. S. A. 75, 1901-1903., Amarante, 1999Amarante, S.T.P. 1999. Sphecidae, in: Brandão, C.R.F., Cancello, E.M. (Eds.). Inver- tebrados Terrestres. Biodiversidade do Estado de São Paulo. Síntese do conhecimento ao final do século XX, in: Joly, C.A., Bicudo, C.E.M. (orgs.). FAPESP, São Paulo, 5, pp. 183-192., Melo, 2000Melo, G.A.R., 2000. Comportamento social em vespas da família Sphecidae (Hymenoptera, Apoidea). In: Martins, R.P., Lewinsohn, T.M., Barbeiros, M.S. (Eds.), Ecologia e Comportamento de Insetos. Oecologia Brasiliensis, vol. VIII. PPGE-UFRJ, Rio de Janeiro, pp. 85-130. and Alcock and Kemp, 2005Alcock, J., Kemp, D.J., 2005. The scramble competition mating system of the sphecid wasp Palmodes praestans (Kohl). J. Nat. Hist. 39, 2809-2814.). Males increase their reproductive success through adaptive strategies or specialization ( Evans, 1966Evans, H.E., 1966. The Comparative Ethology and Evolution of the Sand Wasps. Harvard University Press, Cambridge., Alcock, 1975Alcock, J., 1975. Territorial behavior by males of Philantus multimaculatus (Hymenoptera: Sphecidae) with a review of territoriality in male Sphecids. Anim. Behav. 23, 889-895. and Kroiss et al., 2010Kroiss, J., Lechner, K., Strohm, E., 2010. Male territoriality and mating system in the European beewolf Philanthus triangulum F (Hymenoptera: Crabronidae): evidence for a "hotspot" lek polygyny. J. Ethol. 28, 295-304.), whereas females increase their reproductive success through multiple mating ( Melo, 2000Melo, G.A.R., 2000. Comportamento social em vespas da família Sphecidae (Hymenoptera, Apoidea). In: Martins, R.P., Lewinsohn, T.M., Barbeiros, M.S. (Eds.), Ecologia e Comportamento de Insetos. Oecologia Brasiliensis, vol. VIII. PPGE-UFRJ, Rio de Janeiro, pp. 85-130.) or by changing and sharing nests after the choice of a partner with better attributes or abilities.
The digger wasp Sphex ingens Smith 1856 is a large sphecid, which is usually black and whose gaster and legs vary from partially to completely red ( Bohart and Menke, 1976Bohart, R.M., Menke, A.S., 1976. Sphecid Wasps of the World. University of California Press, Berkeley.). It has been recorded in the Brazilian states of Bahia, Espírito Santo, Rio de Janeiro, and São Paulo ( Amarante, 2002Amarante, S.T.P., 2002. A synonymic catalog of the Neotropical Crabronidae and Sphecidae (Hymenoptera: Apoidea). Arq. Zool. 37, 1-139.).
The ecology and the behavior of S. ingens in Brazil were studied by Piliackas et al. (2007)Piliackas, J.M., Meira, F.V., Suhogusoff, V.G., Rossi, P.G., Badio, A.A., 2007. Estudo sobre a capacidade reprodutiva da população de Sphex ingens Smith (Hymenoptera, Sphecidae) em período reprodutivo no Parque Estadual da Ilha Anchieta, Ubatuba, SP, Brasil. Publicações Avulsas do Instituto Pau Brasil de História Nat- ural 10, 73-80. and Buys (2009)Buys, S.C., 2009. Sphecidae (Hymenoptera: Apoidea) of Rio de Janeiro State (South- eastern Brazil): inventory of species and notes on biology and distribution. Arq. Mus. Nac. Rio de Janeiro 67, 275-282., respectively. Although these studies comprise short-term preliminary assessments and casual observations, they are still considered pioneering in Brazil, as few scientists have tried to understand the natural history of S. ingens.
The greatest challenges of a detailed study on the ecology and behavior of a wild population of S. ingens are the seasonality of its breeding and its restricted distribution. However, the poor information available on the ecology and behavior of S. ingens, in particular in terms of its sexual behavior deserves more attention, for instance, on the type of mating, phases and acts of mating repertoire, sexual behavior trends, influence of the courtship in the process of mate selection and selection criteria of females.
Preliminary observations indicate that the S. ingens mating system is polygamous, and the selection of partners does not occur through the courtship behavior. For this reason, we asked: how extensively does courtship behavior coexist in a polygamous mating system? Hence, the present study aimed at describing and analyzing the mating patterns and trends of the digger wasp S. ingens, aiming at advancing the knowledge of its nesting dynamics.
Materials and methods
Study area and sampling design. Ilha Grande, a coastal island, is
located in the municipality of Angra dos Reis, state of Rio de Janeiro, southeastern
Brazil. It is a fragment of the coastal massif with a rugged relief, and has
approximately 19,300 ha. Its predominant vegetation type is montane and submontane
forest ( Veloso et al., 1991Veloso, H.P., Rangel Filho, A.L.R., Lima, J.C.A., 1991. Classificação da
Vegetação Brasileira: Adaptada a um Sistema Universal. IBGE, Departamento de Recursos
Naturais e Estudos Ambientais, Rio de Janeiro.). The Praia do Sul
State Biological Reserve (RBEPS) covers 18.6% (3.600 ha: 23° 10' S-44° 17' W) of Ilha
Grande's area, and harbors in nearly all its area a continuous complex of Atlantic
Forest, which comprises forest environments (sensu stricto),
restinga forests, mangroves, lagoons, shrubby
restingas, rocky shores, and the beaches Meros, Aventureiro, Demo,
Sul, and Leste ( FEEMA, 2008FEEMA, 2008. Fundação Estadual de Engenharia e Meio Ambiente, Available
at: http://www.inea.rj.gov.br/ (accessed 14.03.15).
http://www.inea.rj.gov.br/...
).
We delimited transects above the high tide line and in foredune areas on Aventureiro Beach to find nesting sites. When we found a nesting site we delimited the area. In each nesting site, we captured males and females, marked their mesoscutum with colored non-toxic paint (Martins, 1993Martins, R.P., 1993. The biology of Editha magnifica (Perty, 1834) (Hymenoptera Sphecidae). Trop. Zool. 6, 109-123.), and released them for individual identification and behavioral analysis.
We daily monitored in copulo pairs formed randomly with exchange of individuals, using a digital camcorder and a chronometer. The sampling effort totaled 131 h of observation, comprising the first fortnight of January 2010 and the first and second fortnights of January 2011. However, only 50 h were selected for analysis, i.e., 45 h of observation including all recorded copulations plus 5 h of observation used to interpret the mating behavior of the species.
At first, in order to interpret the behaviors we used the ad libitum method ( Altmann, 1974Altmann, J., 1974. Observational study of behavior: sampling methods. Behaviour 49, 227-267.), which is based on the behaviors that precede, trigger, and end copulation attempts, whereas for quantitative observations we used the sequence sampling method ( Altmann, 1974Altmann, J., 1974. Observational study of behavior: sampling methods. Behaviour 49, 227-267.). These procedures allowed the construction of a flow chart with the observed behaviors, in order to support the analysis and the construction of another flow chart on the conditional probabilities of occurrence and formation of stochastic processes ( Gottman and Roy, 1990Gottman, J.M., Roy, A.K., 1990. Sequential Analysis: A Guide for Behavioral Researchers. Cambridge University Press, New York.).
All procedures of the present study complied with the guidelines of the permit for scientific research #002/2011 issued by the Directorate of Biodiversity and Protected Areas (INEA-RJ). The captured specimens were deposited as vouchers in the Natural History Collection of the Federal University of Piauí (CHNUFPI: Hymenoptera Collection), campus Amilcar Ferreira Sobral: CHNUFPI(HYM)0002, CHNUFPI(HYM)0003, CHNUFPI(HYM)0004, and CHNUFPI(HYM)0005.
Statistical analysis
We transformed the absolute frequencies of the behavioral acts that compose the behavioral states into relative frequencies. We submitted the frequency of behavioral acts (state) common to males and females to a chi-squared test and logarithmized them to reduce the variation of frequencies whenever necessary. We modeled the relative frequencies of behavioral states with a Poisson distribution and a Markov chain, in order to estimate conditional probabilities and formation of stochastic processes. We ran the analysis using an add-in for Microsoft Excel(r) developed by the Operation Research Group of the University of Texas. We submitted the comparison between the Poisson distribution and the Markov chain to a Kolmogorov-Smirnov test (p < 0.01). All statistical analyses were made using free software.
Results
We analyzed 224 copulation attempts in 137 randomly formed pairs with interchange of males and females. The behavioral repertoire was divided in four phases: identification, pre-copulation, copulation, and post-copulation, based on a total of 19,196 behavioral acts distributed in 24 sexual behaviors (Table 1).
Sexual behavior recorded for male and female digger wasps Sphex ingens (Smith 1856) (Hymenoptera, Sphecidae).
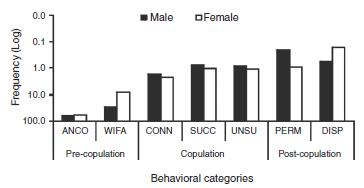
Female mating behavior was composed of nine exclusive behaviors (2319 behavioral acts, 12.0%), whereas males showed eight exclusive behaviors (940 behavioral acts, 4.8%). Seven behaviors were common to both sexes (15,937 behavioral acts, 83.0%), but there was no difference between sexes (x 2 = 16.622, df = n - 1, p < 0.01) ( Fig. 1). The most frequent mating phases in both sexes were pre-copulation and copulation, and behavioral acts related to communication were significant and recurrent in these phases.
Fig. 1. Comparative fluctuation between the logarithmized and non-significant frequency (x2 = 16.622, df = n - 1, p < 0.01) of the common behavioral acts of Sphex ingens Smith 1856 (Hymenoptera, Sphecidae). The common behavioral acts were: antennation (ANCO), wing fanning (WIFA), connubium/strict copulation (CONN), genital clasping/successful copulation (SUCC), no genital clasping/unsuccessful copulation (UNSU), permanence of males in the nesting arena (PERM), and dispersion/males leave the nesting arena (DISP).
Description of the sexual behavior based on the observed behavior
The identification phase consisted of visual recognition and male assault against females (n = 155), which were engaged in activities in their nests or were seized while flying over the nesting site. All approaches recorded were aggressive, preceding the attempt of immobilization, corroborating the existence of coercive copulation. The success in immobilization of females was 80.0% (n = 124) among all approach attempts ( Fig. 2). In 18.7% of the cases the males could not immobilize the females or were rejected before any attempt, as the females were always aggressive.
Fig. 2. Sequence of the phase of identification and pre-copulation: (A) immobilization attempts by males while the female is supplying its nest with prey, (B) efficient immobilization of the female and mount in the nesting area, (C) aedeagus extrusion. Female shows a mixed behavior of receptiveness (last pairs of appendages up) and aggressiveness (partially opened mandibles). Photo by: Jonas Pederassi.
The beginning of pre-copulation is characterized by males mounting females, followed by intensive communication, culminating in the extrusion of the aedeagus before its intromission in the female's genital opening. In some cases males rubbed the aedeagus laterally on the females' gaster before the intromission, whereas females hindered the copulation by curving their gaster.
In the copulation phase, receptive females positioned their gaster parallel to the substrate for aedeagus intromission. Contraction and bending movements directed the genital clasp to assure reproductive success. Females showed self-cleaning behavior during genital clasping (Fig. 3), and then were stimulated to adopt a disengagement behavior.
Fig. 3. Sequence of the copulation phase: (A) beginning of aedeagus intromission. Copulation during nest digging - female carrying a clod of sand. (B) Connubium, gaster's contraction movements. (C) Connubium, gaster's bending movements. (D) Successful copulation - clasping of clasping organs. Male with the body vertically inclined and female self-cleaning. Photo by: Jonas Pederassi.
In post-copulation, males cleaned their genitals after they were disengaged. We observed both dispersal and permanence behavior in nesting arenas after disengagement in the post-copulation phase in males and females. Most females remained in the nesting arena (84.8%) and only a few dispersed (15.1%), whereas males showed the opposite behavior with higher dispersal (73.2%) than permanence (26.7%) in nesting arenas. We observed among males that remained in the nesting arena after effective copulation a decrease in motor activities reflected in low antenna mobility and wing fanning, characterizing a recovery period of 124.10 ± 39.30 s before a new copulation attempt.
Sexual behavior: behavioral trends
The conditional probabilities of transition differed significantly between the Poisson distribution model and the Markov chain (D max = 0.240, df = n - 1, p < 0.01: Kolmogorov-Smirnov), so that the vectors of limiting probabilities were built according to the Markov chain. The predictions derived from the Markov chain corroborated the observation that the behaviors exhibited during mating differed between males and females in terms of observed transition of behavior. However, sexual behaviors recorded in loco (observed behavior) in females, such as eye and antenna cleaning with their first pair of ambulatorial legs during genital clasp, dragging on the ground, and the attempt of flight with clasped males, were considered irrelevant and, hence, excluded from the analysis.
The results expressed by the diagram of the Markov chain highlighted that the behavioral transitions in communication including the antennation (7.9%) and wing fanning (21.4%) behaviors during pre-copulation and copulation have weak influence on the mating behavior of S. ingens.
The transition of the agonistic behaviors displayed by females during the identification (aggressive display 1: 10.3%, confrontation: 33.3%, and rejection: 33.0%), and copulation phases (aggressive display 2: 24.5%, swaying body movements: 31.1%, drumming: 64.3%, and rejection: 45.9%) indicate a strong trend to select males. Another trend pointed out by the analysis was the attempt of males to remain mounted on females (47.9%) when copulation attempts were unsuccessful (Fig. 4).
Fig. 4. Diagram representing the Markov chain for the sexual behavior repertoire of the wasp Sphex ingens Smith 1856 (Hymenoptera, Sphecidae). Symbols represent sexes. Symbol size and line thickness represent the frequency of occurrence of each behavior. Arrowheads represent the transition from one behavior to the next and arrow thickness represents the frequency class.
Discussion
In the digger wasp S. ingens, even though males and females showed common behavioral acts during mating, differences in the frequency and diversity of these acts indicate different energy investment between sexes. Historically, the description of mating events in hymenopterans is characterized by different efforts and methods, which hinder comparisons.
However, although a behavioral similarity among species between copulation and mating phases has been described for some hymenopteran species, such as Mormoniella vitripennis Walker ( Barrass, 1960Barrass, R., 1960. The courtship behavior of Mormoniella vitripennis (Walker) (Hymenoptera: Pteromalidae). Behaviour 15, 185-209. and Barrass, 1961Barrass, R., 1961. A quantitative study of the behavior of the male Mormoniella vitripennis (Walker) (Hymenoptera: Pteromalidae) - towards two constant stim- ulus situations. Behaviour 18, 288-311.), Campoletis sonorensis Cameron ( Vinson, 1976Vinson, S.B., 1976. Host selection by insect parasitoids. Ann. Rev. Entomol. 21, 109-133.) Brachymeria intermedia Nees ( Leonard and Ringo, 1978Leonard, S.H., Ringo, J.M., 1978. Analysis of male courtship patterns and mating behavior of Brachymeria intermedia. Ann. Entomol. Soc. Amer. 71, 817-826.), Apanteles glomeratus Linnaeus ( Tagawa et al., 1985Tagawa, J., Asano, S., Ohtsubo, T., Kamomae, M., Gotoh, T., 1985. Influence of age on the mating behaviour of the braconid wasp Apanteles glomeratus L. Appl. Ent. Zool. 20, 227-230.), Ascogaster reticulatus Watanabe ( Kainoh, 1986), Editha magnifica Perty ( Martins, 1993Martins, R.P., 1993. The biology of Editha magnifica (Perty, 1834) (Hymenoptera Sphecidae). Trop. Zool. 6, 109-123.), Nasonia vitripennis Walker ( Assem and Werren, 1994Assem, J. van den, Werren, J.H., 1994. A comparison of the courtship and mating behavior of tree species of Nasonia (Hymenoptera: Pteromalidae). J. Insect Behav. 7, 53-66.), and Rubrica nasuta Christ ( Pimenta and Martins, 1999Pimenta, H.R., Martins, R.P., 1999. The natural history of the neotropical sand wasp Rubrica nasuta (Christ 1791) (Hymenoptera Sphecidae) in Brazil. Trop. Zool. 12, 273-288.), the details of the behavior of S. ingens, as well as the variation in behavioral acts, their sequence, time, and frequency differ from those of other wasp species.
The confrontation and rejection behaviors recorded in S. ingens were common and present in all mating phases between males and females. According to Cordero and Eberhard (2003)Cordero, C., Eberhard, W.G., 2003. Female choice of sexually antagonistic male adap- tations: a critical review of some current research. J. Evol. Biol. 16, 1-6. pre-copulatory confrontations indicate the existence of conflicts between sexes and sexual selection, which favors manipulative males in coercive copulations. The resistance of females to the antagonistic behavior of males may select individuals according to their capacity of individual manipulation (signaling vigor and fitness) in clasping the female as it tries to get rid of the male ( Allen and Simmons, 1996Allen, G.F., Simmons, L.W., 1996. Coercive mating, fluctuating asymmetry and male mating success in the dung fly Sepsis cynipsea. Anim. Behav. 52, 737-741.). In S. ingens coercive copulation is an alternative strategy of males in response to the constant aggressiveness of females, as the latter use this criterion to select partners.
In S. ingens females, the larger number of copulation attempts in relation to the phase of nest construction (digging the tunnel or supply chamber) reflects a high energy investment, which makes them more evident in their respective arenas and nests, and justifies the trend of remaining in the nest after a copulation attempt, regardless of the result. Evans et al. (1976)Evans, H.E., Matthews, R.W., Alcock, J., Fritz, M.A., 1976. Notes on the nests and prey of two subspecies of Cerceris rufimana Taschenberg (Hymenoptera: Sphecidae: Cercerini). J. Kans. Entomol. Soc. 49, 126-132., Jang et al. (1996)Jang, Y., Wuellner, C.T., Scott, C.S., 1996. Floating and fidelity in nest visitation by Crawfordapis luctuosa (Hymenoptera: Colletidae). J. Insect Behav. 9, 493-504., and Melo (2000)Melo, G.A.R., 2000. Comportamento social em vespas da família Sphecidae (Hymenoptera, Apoidea). In: Martins, R.P., Lewinsohn, T.M., Barbeiros, M.S. (Eds.), Ecologia e Comportamento de Insetos. Oecologia Brasiliensis, vol. VIII. PPGE-UFRJ, Rio de Janeiro, pp. 85-130. consensually affirmed that most time and energy of females of the family Sphecidae is invested in maternal care in the form of construction and provision of nests during their reproductive life.
Male S. ingens were able to copulate repeatedly with females in the nesting site at any phase of nest building. This results in an intensive daily copulation activity that involves a relevant investment of time and in part corroborates the studies by Genaro (1998)Genaro, J.A., 1998. Comportamiento de Sphex jamaicensis durante la nidificación y notas sobre S. mandibulares y S. cubensis (Hymenoptera: Sphecidae). Caribb. J. Sci. 34, 238-242. on the species Sphex jamaicencis Drury. In several insect species the occurrence of multiple copulations suggests the existence of cryptic female choice ( Ebehard, 1996Ebehard, W.G., 1996. Female Control: Sexual Selection by Cryptic Female Choice. Princeton University Press, Princeton.), sperm competition ( Parker, 1970Parker, G.A., 1970. Sperm competition and its evolutionary consequences in the insects. Biol. Rev. 45, 525-567.), reduction in the female's spermatheca or that one or few copulations are not enough for the females to obtain enough sperm for a maximum production of offspring ( Ridley, 1990Ridley, M., 1990. The control and frequency of mating in insects. Funct. Ecol. 4, 75-84.), and even physiological deficiencies in the reproductive system of males ( Martins et al., 2004Martins, G.F., Serrão, J.E., Furieri, K.S., 2004. Notes on the spermatheca of Vespidae and Sphecidae (Hymenoptera). Sociobiology 45, 119-127.).
Several studies reported that the courtship and copulation of many hymenopterans consist of movements of wings, legs, antennae, and mouthparts of males (Barrass, 1960Barrass, R., 1960. The courtship behavior of Mormoniella vitripennis (Walker) (Hymenoptera: Pteromalidae). Behaviour 15, 185-209., Barrass, 1961Barrass, R., 1961. A quantitative study of the behavior of the male Mormoniella vitripennis (Walker) (Hymenoptera: Pteromalidae) - towards two constant stim- ulus situations. Behaviour 18, 288-311., Mathews, 1975Mathews, R.W., 1975. Courtship in parasitic wasps. In: Price, P.W. (Ed.), Evolutionary Strategies of Parasitic Insects and Mites. Plenum Press, New York, pp. 66-86., Vinson, 1976Vinson, S.B., 1976. Host selection by insect parasitoids. Ann. Rev. Entomol. 21, 109-133., Leonard and Ringo, 1978Leonard, S.H., Ringo, J.M., 1978. Analysis of male courtship patterns and mating behavior of Brachymeria intermedia. Ann. Entomol. Soc. Amer. 71, 817-826., Tagawa et al., 1985Tagawa, J., Asano, S., Ohtsubo, T., Kamomae, M., Gotoh, T., 1985. Influence of age on the mating behaviour of the braconid wasp Apanteles glomeratus L. Appl. Ent. Zool. 20, 227-230., Kainoh, 1986Kainoh, Y., 1986. Mating behavior of Ascogaster reticulatus Watanabe (Hymenoptera: Braconidae), an egg-larval parasitoid of the smaller tea tortrix moth, Adoxophyes sp (Lepidoptera: Tortricidae) I. Diel patterns of emergence and mating, and some conditions for mating. Appl. Ent. Zool. 21, 1-7., Martins, 1993Martins, R.P., 1993. The biology of Editha magnifica (Perty, 1834) (Hymenoptera Sphecidae). Trop. Zool. 6, 109-123., Assem and Werren, 1994Assem, J. van den, Werren, J.H., 1994. A comparison of the courtship and mating behavior of tree species of Nasonia (Hymenoptera: Pteromalidae). J. Insect Behav. 7, 53-66. and Pimenta and Martins, 1999Pimenta, H.R., Martins, R.P., 1999. The natural history of the neotropical sand wasp Rubrica nasuta (Christ 1791) (Hymenoptera Sphecidae) in Brazil. Trop. Zool. 12, 273-288.). The sequence of these movements varies accordingly between species and is repeated by a receptive female (Mayr, 1973Mayr, E., 1973. Animal Species and Evolution. Harvard University Press, Cambridge. and Assem, 1986Assem, J. van den, 1986. Mating behavior in parasitc wasps. In: Waage, J., Greathead, D. (Eds.), Insect parasitoids. Academic, New York, pp. 125-157.). Although these studies suggest the existence of visual stimuli that precede mount, we did not observe any pre-copulation gestural behavior in S. ingens. The lack of these behaviors in S. ingens results from the influence of the promiscuous mating system and from the way females select their partners.
Presumably, the energy spent in intersexual communication in S. ingens does not support that this behavior influences courtship, but would represent the exchange of stimuli used in conspecific recognition, as suggested by Mayr (1973)Mayr, E., 1973. Animal Species and Evolution. Harvard University Press, Cambridge., or general excitement signals. However, more specific tests on the performance of antennation and its influence on the successful and unsuccessful mating could clarify the function of this behavior. Furthermore, it is possible that in S. ingens pheromones are the only expressions of female receptivity, as we observed males trying to copulate with prey left by females at the entrance of nests. This behavior was also observed in studies with other species carried out in the laboratory ( Barrass, 1960Barrass, R., 1960. The courtship behavior of Mormoniella vitripennis (Walker) (Hymenoptera: Pteromalidae). Behaviour 15, 185-209., Barrass, 1961Barrass, R., 1961. A quantitative study of the behavior of the male Mormoniella vitripennis (Walker) (Hymenoptera: Pteromalidae) - towards two constant stim- ulus situations. Behaviour 18, 288-311., Mathews, 1975Mathews, R.W., 1975. Courtship in parasitic wasps. In: Price, P.W. (Ed.), Evolutionary Strategies of Parasitic Insects and Mites. Plenum Press, New York, pp. 66-86. and Vinson, 1976Vinson, S.B., 1976. Host selection by insect parasitoids. Ann. Rev. Entomol. 21, 109-133.).
Other aspects of sexual behavior of S. ingens, that would expand the understanding of its reproductive behavior, refer to the adjustments, disputes and strategies adopted by males to increase their reproductive success. Apparently, preliminary tests on the collected specimens suggest that there are size differences between the two sexes, as well between males with territorial and patrol behavior. Therefore, differences in size and behavior could influence individual fitness, with consequences to the results of mating disputes, and in the frequency of the reproductive success.
Our analysis corroborates that the mating system and female aggressiveness result in low sexual selectivity and a decrease in courtship effort by males, and, hence, favor the maintenance of coercive copulation. They also suggest that the ability of males to manipulate the females during copulation is the main requirement for a successful copulation. Hence, the decrease in courtship effort by males is an alternative strategy to energy-demanding strategies based on elaborated courtship displays, considering the way that copulations occur, number of copulation attempts observed and constant confrontation with rival males in the present study. The escalation of female aggressiveness is an antagonistic sexual selection criterion different from classic forms found in the literature in terms of assessment of attributes or courtship rites. In addition, although coercive copulation is not an exclusive strategy among insects, it is rare in solitary wasps, in particular sphecids.
Acknowledgments
We thank the INEA-RJ team for the help in the present study. Rogério P. Martins (UFMG) supported our study and provided us with important literature. Sérvio T.P. Amarante identified the first specimens captured. Mr. Waldomiro (camping 7) at Aventureiro Beach was very kind to us and provided us with room to develop the present study. We also thank the reviewers for their suggestions.
References
- Alcock, J., 1975. Territorial behavior by males of Philantus multimaculatus (Hymenoptera: Sphecidae) with a review of territoriality in male Sphecids. Anim. Behav. 23, 889-895.
- Alcock, J., Kemp, D.J., 2005. The scramble competition mating system of the sphecid wasp Palmodes praestans (Kohl). J. Nat. Hist. 39, 2809-2814.
- Allen, G.F., Simmons, L.W., 1996. Coercive mating, fluctuating asymmetry and male mating success in the dung fly Sepsis cynipsea. Anim. Behav. 52, 737-741.
- Altmann, J., 1974. Observational study of behavior: sampling methods. Behaviour 49, 227-267.
- Amarante, S.T.P. 1999. Sphecidae, in: Brandão, C.R.F., Cancello, E.M. (Eds.). Inver- tebrados Terrestres. Biodiversidade do Estado de São Paulo. Síntese do conhecimento ao final do século XX, in: Joly, C.A., Bicudo, C.E.M. (orgs.). FAPESP, São Paulo, 5, pp. 183-192.
- Amarante, S.T.P., 2002. A synonymic catalog of the Neotropical Crabronidae and Sphecidae (Hymenoptera: Apoidea). Arq. Zool. 37, 1-139.
- Assem, J. van den, 1986. Mating behavior in parasitc wasps. In: Waage, J., Greathead, D. (Eds.), Insect parasitoids. Academic, New York, pp. 125-157.
- Assem, J. van den, Werren, J.H., 1994. A comparison of the courtship and mating behavior of tree species of Nasonia (Hymenoptera: Pteromalidae). J. Insect Behav. 7, 53-66.
- Barrass, R., 1960. The courtship behavior of Mormoniella vitripennis (Walker) (Hymenoptera: Pteromalidae). Behaviour 15, 185-209.
- Barrass, R., 1961. A quantitative study of the behavior of the male Mormoniella vitripennis (Walker) (Hymenoptera: Pteromalidae) - towards two constant stim- ulus situations. Behaviour 18, 288-311.
- Bohart, R.M., Menke, A.S., 1976. Sphecid Wasps of the World. University of California Press, Berkeley.
- Buys, S.C., 2009. Sphecidae (Hymenoptera: Apoidea) of Rio de Janeiro State (South- eastern Brazil): inventory of species and notes on biology and distribution. Arq. Mus. Nac. Rio de Janeiro 67, 275-282.
- Cordero, C., Eberhard, W.G., 2003. Female choice of sexually antagonistic male adap- tations: a critical review of some current research. J. Evol. Biol. 16, 1-6.
- Ebehard, W.G., 1996. Female Control: Sexual Selection by Cryptic Female Choice. Princeton University Press, Princeton.
- Evans, H.E., 1966. The Comparative Ethology and Evolution of the Sand Wasps. Harvard University Press, Cambridge.
- Evans, H.E., Matthews, R.W., Alcock, J., Fritz, M.A., 1976. Notes on the nests and prey of two subspecies of Cerceris rufimana Taschenberg (Hymenoptera: Sphecidae: Cercerini). J. Kans. Entomol. Soc. 49, 126-132.
- Evans, H.E., O'Neill, K.M., 1978. Alternative mating strategies in digger wasp Philan- thus zebratus Cresson. Proc. Natl. Acad. Sci. U. S. A. 75, 1901-1903.
- FEEMA, 2008. Fundação Estadual de Engenharia e Meio Ambiente, Available at: http://www.inea.rj.gov.br/ (accessed 14.03.15).
» http://www.inea.rj.gov.br/ - Genaro, J.A., 1998. Comportamiento de Sphex jamaicensis durante la nidificación y notas sobre S. mandibulares y S. cubensis (Hymenoptera: Sphecidae). Caribb. J. Sci. 34, 238-242.
- Gottman, J.M., Roy, A.K., 1990. Sequential Analysis: A Guide for Behavioral Researchers. Cambridge University Press, New York.
- Jang, Y., Wuellner, C.T., Scott, C.S., 1996. Floating and fidelity in nest visitation by Crawfordapis luctuosa (Hymenoptera: Colletidae). J. Insect Behav. 9, 493-504.
- Kainoh, Y., 1986. Mating behavior of Ascogaster reticulatus Watanabe (Hymenoptera: Braconidae), an egg-larval parasitoid of the smaller tea tortrix moth, Adoxophyes sp (Lepidoptera: Tortricidae) I. Diel patterns of emergence and mating, and some conditions for mating. Appl. Ent. Zool. 21, 1-7.
- Kroiss, J., Lechner, K., Strohm, E., 2010. Male territoriality and mating system in the European beewolf Philanthus triangulum F (Hymenoptera: Crabronidae): evidence for a "hotspot" lek polygyny. J. Ethol. 28, 295-304.
- Leonard, S.H., Ringo, J.M., 1978. Analysis of male courtship patterns and mating behavior of Brachymeria intermedia. Ann. Entomol. Soc. Amer. 71, 817-826.
- Martins, G.F., Serrão, J.E., Furieri, K.S., 2004. Notes on the spermatheca of Vespidae and Sphecidae (Hymenoptera). Sociobiology 45, 119-127.
- Martins, R.P., 1993. The biology of Editha magnifica (Perty, 1834) (Hymenoptera Sphecidae). Trop. Zool. 6, 109-123.
- Mathews, R.W., 1975. Courtship in parasitic wasps. In: Price, P.W. (Ed.), Evolutionary Strategies of Parasitic Insects and Mites. Plenum Press, New York, pp. 66-86.
- Mayr, E., 1973. Animal Species and Evolution. Harvard University Press, Cambridge.
- Melo, G.A.R., 1999. Phylogenetic relationships and classification of the major lineages of Apoidea (Hymenoptera), with emphasis on the crabronid waps. Sci. Pap. Nat. Hist. Mus. Univ. Kans. 14, 1-55.
- Melo, G.A.R., 2000. Comportamento social em vespas da família Sphecidae (Hymenoptera, Apoidea). In: Martins, R.P., Lewinsohn, T.M., Barbeiros, M.S. (Eds.), Ecologia e Comportamento de Insetos. Oecologia Brasiliensis, vol. VIII. PPGE-UFRJ, Rio de Janeiro, pp. 85-130.
- Parker, G.A., 1970. Sperm competition and its evolutionary consequences in the insects. Biol. Rev. 45, 525-567.
- Pimenta, H.R., Martins, R.P., 1999. The natural history of the neotropical sand wasp Rubrica nasuta (Christ 1791) (Hymenoptera Sphecidae) in Brazil. Trop. Zool. 12, 273-288.
- Piliackas, J.M., Meira, F.V., Suhogusoff, V.G., Rossi, P.G., Badio, A.A., 2007. Estudo sobre a capacidade reprodutiva da população de Sphex ingens Smith (Hymenoptera, Sphecidae) em período reprodutivo no Parque Estadual da Ilha Anchieta, Ubatuba, SP, Brasil. Publicações Avulsas do Instituto Pau Brasil de História Nat- ural 10, 73-80.
- Ridley, M., 1990. The control and frequency of mating in insects. Funct. Ecol. 4, 75-84.
- Tagawa, J., Asano, S., Ohtsubo, T., Kamomae, M., Gotoh, T., 1985. Influence of age on the mating behaviour of the braconid wasp Apanteles glomeratus L. Appl. Ent. Zool. 20, 227-230.
- Veloso, H.P., Rangel Filho, A.L.R., Lima, J.C.A., 1991. Classificação da Vegetação Brasileira: Adaptada a um Sistema Universal. IBGE, Departamento de Recursos Naturais e Estudos Ambientais, Rio de Janeiro.
- Vinson, S.B., 1976. Host selection by insect parasitoids. Ann. Rev. Entomol. 21, 109-133.
Publication Dates
-
Publication in this collection
Apr-Jun 2015
History
-
Received
17 Sept 2014 -
Reviewed
11 Feb 2015 -
Accepted
09 Apr 2015