ABSTRACT
Triorla Parks has been reported from the Nearctic and Neotropical regions with 10 described species: T. ambigua (Macquart), T. argyrogaster (Macquart), T. demifasciata (Macquart), T. interrupta (Macquart), T. nervosa (Macquart), T. parastriola Pamplona & Aires, T. rubidiventris (Macquart), T. spinosa Tomasovic, T. striola (Fabricius), and T. trichina Tomasovic. Two species, T. striola and T. parastriola, have been recorded from Brazil. This paper describes four new species of Triorla from Brazil: T. beckeri sp. nov., T. milineae sp. nov., T. paraensis sp. nov., and T. spatulata sp. nov., and includes comments on the morphology of T. parastriola. An identification key to the species of Triorla is provided.
Keywords:
Nearctic; Neotropical; Taxonomy
Introduction
Asilinae Latreille, 1802 is the most diverse subfamily of Asilidae, including 179 extant genera, distributed in all biogeographic regions, except Antarctica (Geller-Grimm, 2004Geller-Grimm, F., 2004. A world catalogue of the genera of the family Asilidae (Diptera). Studia Dipterologica 10, 473-526.; Londt, 2005Londt, J.G.H., 2005. An annotated key to the genera of Afrotropical Apocleinae, with descriptions of six new genera (Diptera: Asilidae). Tijdschrift voor Entomologie 148, 39-62.; Vieira, 2012Vieira, R., 2012. New distribution records of six species of Asilinae (Diptera: Asilidae) Latreille, 1802. Check List 4, 779-781.; Artigas and Vieira, 2014Artigas, J.N., Vieira, R., 2014. New genus and species of Neotropical robber flies (Diptera, Asilidae, Asilinae). Zootaxa 3774, 282-288.; Vieira and Rafael, 2014Vieira, R., Rafael, J., 2014. Longivena, a new robber-fly genus from Brazil (Diptera, Asilidae, Asilinae). ZooKeys 443, 119-138.). Sixty-eight genera are recognized in the Neotropical region, of which 20 occur in Brazil (Papavero, 2009Papavero, N., 2009. Catalogue of Neotropical Diptera. Asilidae. Neotropical Diptera 17, 1-179.; Vieira, 2012Vieira, R., 2012. New distribution records of six species of Asilinae (Diptera: Asilidae) Latreille, 1802. Check List 4, 779-781.; Artigas and Vieira, 2014Artigas, J.N., Vieira, R., 2014. New genus and species of Neotropical robber flies (Diptera, Asilidae, Asilinae). Zootaxa 3774, 282-288.; Vieira and Rafael, 2014Vieira, R., Rafael, J., 2014. Longivena, a new robber-fly genus from Brazil (Diptera, Asilidae, Asilinae). ZooKeys 443, 119-138.). TriorlaParks, 1968Parks, L., 1968. Synopsis of robberfly genera allied to Efferia and Eicherax, including a new genus. Pan-Pac. Entomol. 44, 171-179. is characterized by vein R5 ending before wing apex, male terminalia parallel with longitudinal axis of the body and tergites 9 and 10 of ovipositor fused together (Parks, 1968Parks, L., 1968. Synopsis of robberfly genera allied to Efferia and Eicherax, including a new genus. Pan-Pac. Entomol. 44, 171-179.). The Neotropical and Nearctic Triorla was proposed based on two species – the type species T. interrupta (Macquart, 1834Macquart, P.J.M., 1834. Histoire naturelle des Insectes. Diptères I. Roret, Paris.), with the type locality U.S.A. (distribution: U.S.A., Mexico, Guatemala, Honduras, Nicaragua, Costa Rica, Panama, and Colombia) and T. striola (Fabricius, 1805Fabricius, J.C., 1805. Systema Antliatorum Secundum Ordines, Genera, Species. Brunsvigae (Brunswick).), with the type locality "South-America" (distribution: Panama, Colombia, Venezuela, Guyana and Surinam, and south of Paraguay) (Papavero, 2009Papavero, N., 2009. Catalogue of Neotropical Diptera. Asilidae. Neotropical Diptera 17, 1-179.).
A third species of the genus, T. parastriolaPamplona and Aires, 1999Pamplona, D.M., Aires, C., 1999. Revision of Triorla Parks and description of a new species emphasizing the systematic value of spermatheca (Diptera, Asilidae). Revista Brasileira de Zoologia 16, 1127-1134. was described from Roraima State in the Brazilian Amazon (Pamplona and Aires, 1999Pamplona, D.M., Aires, C., 1999. Revision of Triorla Parks and description of a new species emphasizing the systematic value of spermatheca (Diptera, Asilidae). Revista Brasileira de Zoologia 16, 1127-1134.). Recently, seven species of Triorla were proposed by Tomasovic (2002)Tomasovic, G., 2002. Étude sur matériaux typiques du complexe génital mâle de spet espèce du genre Erax Scopoli, 1763 (Diptera Asilidae) avec la description de trois espèces nouvelles. Notes faunistiques de Gembloux 46, 27-37. based on the type-material of EraxScopoli, 1763Scopoli, J.A., 1763. Entomologia carniolica exhibens insecta carnioliae indigena etdistributa in ordines, genera, species, varietates, methodo Linneana [Reprint Von Guglia, O. (1972)]. Vindobona., described by Macquart (1838Macquart, P.J.M., 1838. Diptères exotiques nouveaux ou peu connus. Mémoires de La Société royale des Sciences, de l'Agriculture et des Artes 1, 5-207., 1846Macquart, P.J.M., 1846. Dipteres exotiques nouveaux ou peu connus. 1. Supplément. Mémoires de la Société royale des Sciences, de l'Agriculture et des Artes (1845), 133-366., 1850)Macquart, P.J.M., 1850. Dipteres exotiques nouveaux ou peu connus. 4. Supplément. Mémoires de la Société royale des Sciences, de l'Agriculture et des Artes (1849), 61–96, 309–479. and deposited in the French museums of Lille and Paris, including: (1) two new species, T. spinosa and T. trichina; (2) five species that were transferred to Triorla: T. argyrogaster (Macquart, 1846Macquart, P.J.M., 1846. Dipteres exotiques nouveaux ou peu connus. 1. Supplément. Mémoires de la Société royale des Sciences, de l'Agriculture et des Artes (1845), 133-366.), T. demifasciata (Macquart, 1850Macquart, P.J.M., 1850. Dipteres exotiques nouveaux ou peu connus. 4. Supplément. Mémoires de la Société royale des Sciences, de l'Agriculture et des Artes (1849), 61–96, 309–479.), T. nervosa (Macquart, 1838Macquart, P.J.M., 1838. Diptères exotiques nouveaux ou peu connus. Mémoires de La Société royale des Sciences, de l'Agriculture et des Artes 1, 5-207.), T. tibialis (Macquart, 1838Macquart, P.J.M., 1838. Diptères exotiques nouveaux ou peu connus. Mémoires de La Société royale des Sciences, de l'Agriculture et des Artes 1, 5-207.), and T. vicina (Macquart, 1846Macquart, P.J.M., 1846. Dipteres exotiques nouveaux ou peu connus. 1. Supplément. Mémoires de la Société royale des Sciences, de l'Agriculture et des Artes (1845), 133-366.); and (3) three species previously synonymized with T. interrupta that were re-established to specific status: T. ambigua (Macquart, 1846Macquart, P.J.M., 1846. Dipteres exotiques nouveaux ou peu connus. 1. Supplément. Mémoires de la Société royale des Sciences, de l'Agriculture et des Artes (1845), 133-366.) and T. maculatus (Macquart, 1838Macquart, P.J.M., 1838. Diptères exotiques nouveaux ou peu connus. Mémoires de La Société royale des Sciences, de l'Agriculture et des Artes 1, 5-207.) synonymized by Martin and Papavero (1970)Martin, C.H., Papavero, N., 1970. A Catalogue of the Diptera of the Americas South of United States. 35b. Family Asilidae. Museu de Zoologia, Universidade de São Paulo, pp. 1–139., and T. rubidiventris (Macquart, 1850Macquart, P.J.M., 1850. Dipteres exotiques nouveaux ou peu connus. 4. Supplément. Mémoires de la Société royale des Sciences, de l'Agriculture et des Artes (1849), 61–96, 309–479.) synonymized by Artigas and Papavero (1997)Artigas, J.N., Papavero, N., 1997. The American genera of Asilidae (Diptera): keys for identification with an atlas of female spermathecae and other morphological details. IX.2. Subfamily Asilinae Leach – Efferia group, with the proposal of five new genera and a catalogue of the neotropical species. Arquivos de Zoologia do Estado de São Paulo 34, 65-95..
Papavero (2009)Papavero, N., 2009. Catalogue of Neotropical Diptera. Asilidae. Neotropical Diptera 17, 1-179., in the Catalogue of Neotropical Asilidae, allocated T. ambigua and T. rubidiventris in Triorla, but did not include T. vicina and T. tibialis as valid species for the genus. He also proposed T. maculatus as a synonym for T. striola. Hence, at the present time, 10 species of Triorla are recognized in Nearctic and Neotropical regions (Table 1).
Here we describe four new species of Triorla from Brazil, and include morphological comments concerning T. parastriola. An identification key, as well as geographical records and biology data (when available) are provided.
List of species of Triorla in Nearctic and Neotropical regions, followed by author, year, and distribution. The list includes the new species here described.
Material and Methods
Specimens from four Brazilian insect collections were examined. The abbreviations of the collections referred to in this paper are: INPA – Coleção de Invertebrados do Instituto Nacional de Pesquisa da Amazônia, Manaus, Amazonas, Brazil; MPEG – Coleção de Invertebrados do Museu Paraense Emilio Goeldi, Belém, Pará, Brazil; MZFS – Coleção Entomológica Prof. Johann Becker do Museu de Zoologia da Universidade Estadual de Feira de Santana, Feira de Santana, Bahia, Brazil; MZUSP – Museu de Zoologia da Universidade de São Paulo, São Paulo, São Paulo, Brazil. Dissected terminalia were placed in a 10% KOH solution at 40 °C for 8 h, washed in water, treated with 10% acetic acid, and subsequently examined in concavity slides in glycerin. After examination and illustration, the detached parts were placed in microvials with glycerin and pinned with their respective specimens. The female terminalia were mounted on permanent slides using Canada balsam. The terminology used in the morphological descriptions follows Cumming and Wood (2009)Cumming, J.M., Wood, D.M., 2009. Adult morphology and terminology. In: Brown, B.V., Borkent, A., Cumming, J.M., Wood, D.M., Woodley, N.E., Zumbado, M.A. (Eds.), Manual of Central American Diptera, vol. 1. National Research Council Research Press, Ottawa, pp. 9–50..
Taxonomy
Triorla Parks
TriorlaParks, 1968Parks, L., 1968. Synopsis of robberfly genera allied to Efferia and Eicherax, including a new genus. Pan-Pac. Entomol. 44, 171-179. – Parks (1968: 175, 176)Parks, L., 1968. Synopsis of robberfly genera allied to Efferia and Eicherax, including a new genus. Pan-Pac. Entomol. 44, 171-179..
Type-species: Asilus interruptusMacquart, 1834Macquart, P.J.M., 1834. Histoire naturelle des Insectes. Diptères I. Roret, Paris. – Macquart (1834: 310)Macquart, P.J.M., 1834. Histoire naturelle des Insectes. Diptères I. Roret, Paris..
Diagnosis based on Parks (1968)Parks, L., 1968. Synopsis of robberfly genera allied to Efferia and Eicherax, including a new genus. Pan-Pac. Entomol. 44, 171-179. and Pamplona and Aires (1999)Pamplona, D.M., Aires, C., 1999. Revision of Triorla Parks and description of a new species emphasizing the systematic value of spermatheca (Diptera, Asilidae). Revista Brasileira de Zoologia 16, 1127-1134.. Ocellar tubercle with few macrosetae or setae; stylus without microsegment; R5 ending before wing apex; short stump vein at base of R4 not reaching base of R2+3; male terminalia parallel with longitudinal axis of the body; gonostylus situated on proximal portion of gonocoxite; hypandrium well-developed, approximately 0.5× the length of epandrium; apex of aedeagus with 3 thin prongs, approximately as long as the flagellum (postpedicel sensu Stuckenberg, 1999Stuckenberg, B.R., 1999. Antennal evolution in the Brachycera (Diptera), with a reassessment of terminology relating to the flagellum. Studia Dipterologica 6, 33-48.), with dilations near apex; prongs tapered after dilations; female terminalia with tergite VII pruinescence, except on shiny posterior margin; segment VIII shiny; sternite VIII basally platelike and hypogynial valves forming keel distally; ovipositor conical, not laterally compressed, not longer than abdominal segments VI and VII together; ovipositor without apical spur.
Remarks. The grammatical genus of Triorla is feminine. This affirmation is based on the specific epithet of the type-species, T. interrupta. Parks (1968)Parks, L., 1968. Synopsis of robberfly genera allied to Efferia and Eicherax, including a new genus. Pan-Pac. Entomol. 44, 171-179. did not address this point in the original description of the genus.
Two species, previously transferred to Triorla by Tomasovic (2002)Tomasovic, G., 2002. Étude sur matériaux typiques du complexe génital mâle de spet espèce du genre Erax Scopoli, 1763 (Diptera Asilidae) avec la description de trois espèces nouvelles. Notes faunistiques de Gembloux 46, 27-37., Erax tibialis Macquart and Erax vicinus Macquart, are not included in the list of recognized species in the Catalogue of Neotropical Asilidae prepared by Papavero (2009)Papavero, N., 2009. Catalogue of Neotropical Diptera. Asilidae. Neotropical Diptera 17, 1-179.. We agree with this view as Tomasovic's illustrations of their male terminalia make it unlikely that they are Triorla species.
Key to the males of Triorla Parks from Nearctic and Neotropical regions
Head in frontal view. 1. Triorla beckeri sp. nov.; 2. Triorla milineae sp. nov.; 3. Triorla spatulata sp. nov.; 4. Triorla paraensis sp. nov.; 5. Triorla parastriolaPamplona and Aires, 1999Pamplona, D.M., Aires, C., 1999. Revision of Triorla Parks and description of a new species emphasizing the systematic value of spermatheca (Diptera, Asilidae). Revista Brasileira de Zoologia 16, 1127-1134..
Head in lateral view. 6. Triorla beckeri sp. nov.; 7. Triorla milineae sp. nov.; 8. Triorla spatulata sp. nov.; 9. Triorla paraensis sp. nov.; 10. Triorla parastriolaPamplona and Aires, 1999Pamplona, D.M., Aires, C., 1999. Revision of Triorla Parks and description of a new species emphasizing the systematic value of spermatheca (Diptera, Asilidae). Revista Brasileira de Zoologia 16, 1127-1134..
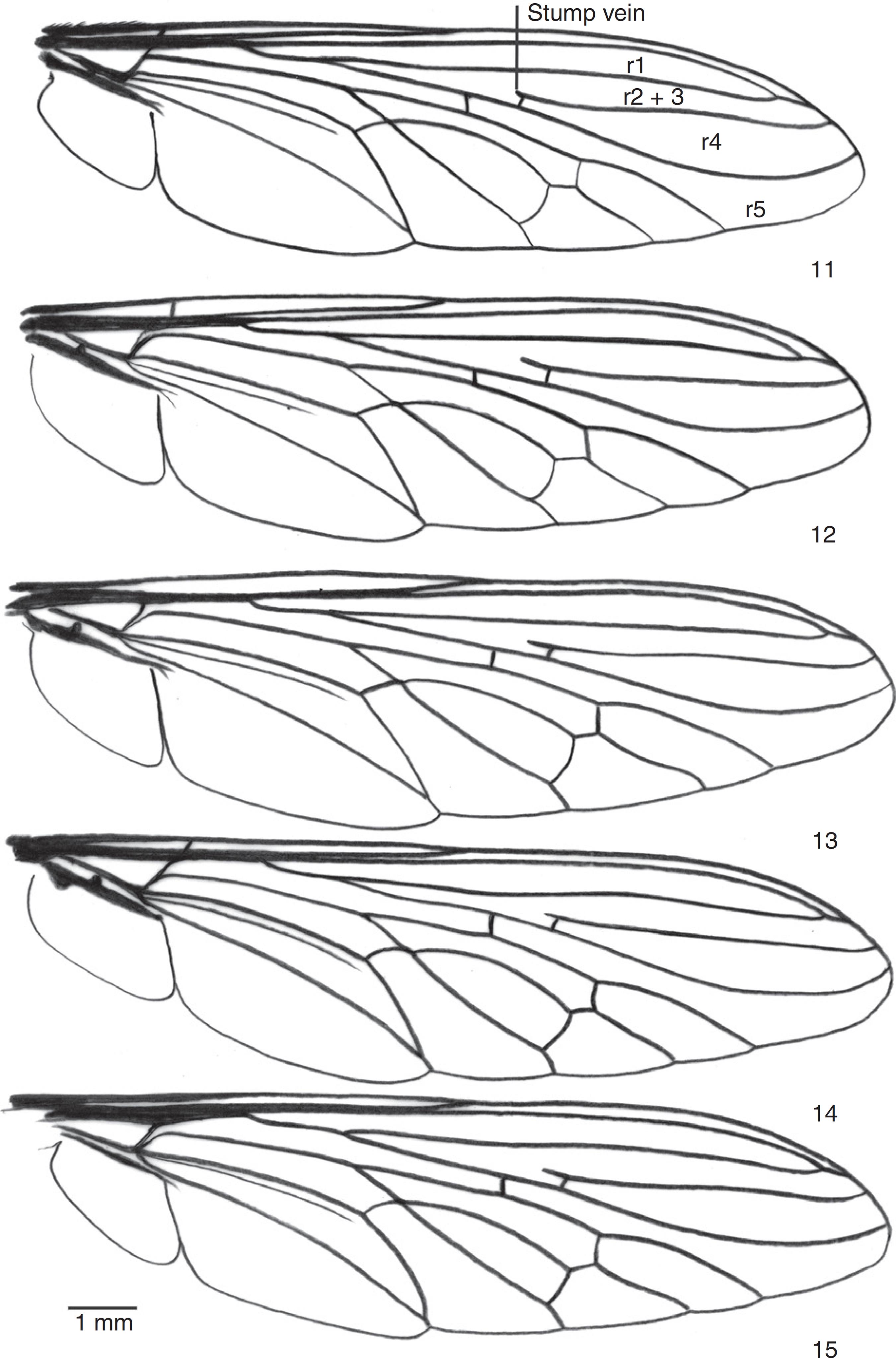
Wing. 11. Triorla beckeri sp. nov.; 12. Triorla milineae sp. nov.; 13. Triorla paraensis sp. nov.; 14. Triorla parastriolaPamplona and Aires, 1999Pamplona, D.M., Aires, C., 1999. Revision of Triorla Parks and description of a new species emphasizing the systematic value of spermatheca (Diptera, Asilidae). Revista Brasileira de Zoologia 16, 1127-1134.; 15. Triorla spatulata sp. nov.
Triorla parastriolaPamplona and Aires, 1999Pamplona, D.M., Aires, C., 1999. Revision of Triorla Parks and description of a new species emphasizing the systematic value of spermatheca (Diptera, Asilidae). Revista Brasileira de Zoologia 16, 1127-1134.. 16. Aedeagal prong; 17. Gonocoxite; 18. Hypandrium; 19. Gonostylus in lateral view; 20. Aedeagus and ejaculatory apodeme in lateral view; 21. Male terminalia in lateral view. Abbreviations: cerc: cercus; ej apod: ejaculatory apodeme; epand: epandrium; goncx: gonocoxite; gonst: gonostylus; hypd: hypandrium; aed: aedeagus; pr: prong.
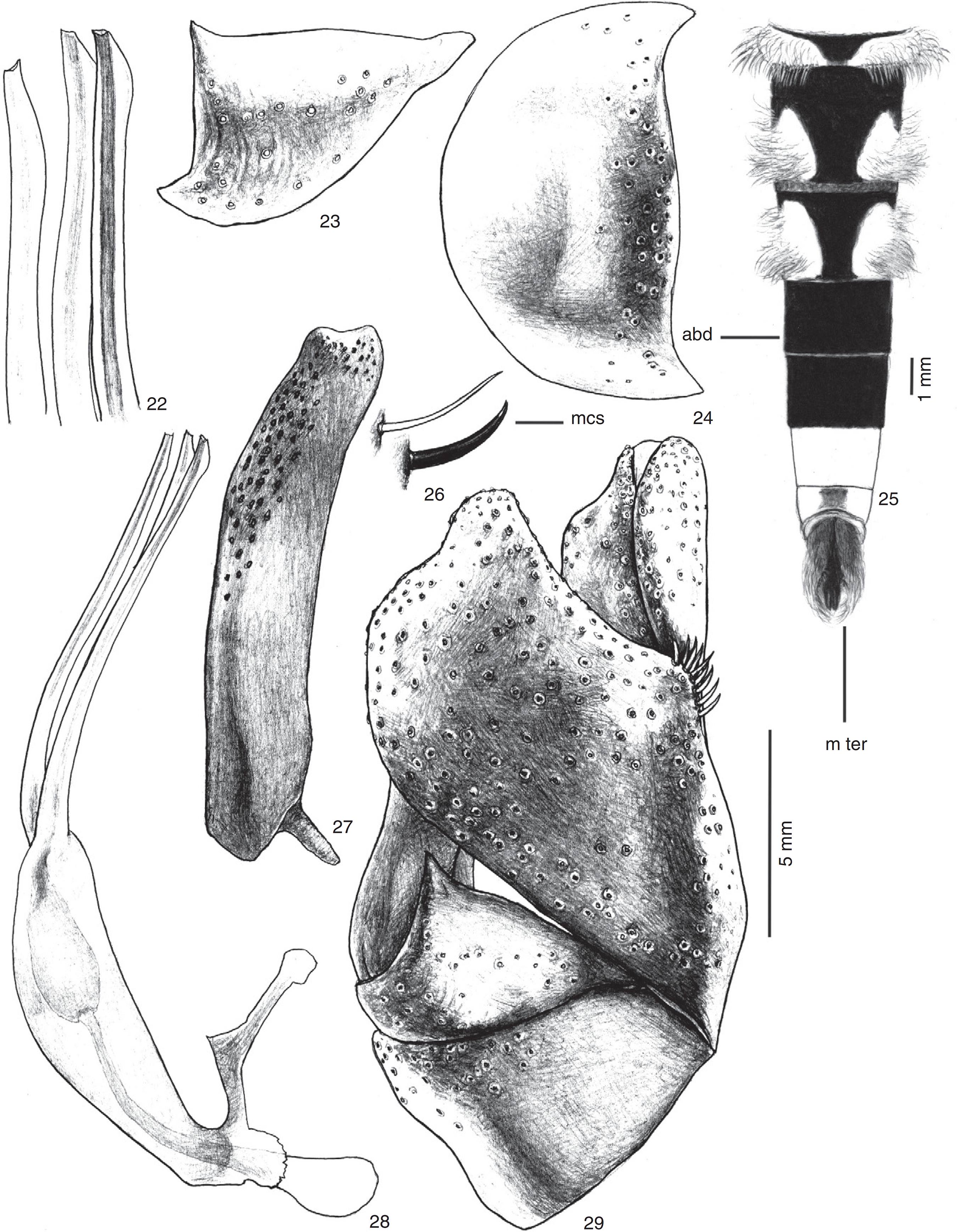
Triorla beckerisp. nov. 22. Apex of aedeagal prongs; 23. Gonocoxite; 24. Hypandrium; 25. Male abdomen; 26. Macrosetae of epandrium; 27. Gonostylus in lateral view; 28. Aedeagus and ejaculatory apodeme in lateral view; 29. Male terminalia in lateral view. Abbreviations: abd: abdomen; mcs: macrosetae; m ter: male terminalia.
Triorla milineaesp. nov. 30. Male habitus in lateral view; 31. Male habitus in frontal view; 32. Male habitus in dorsal view; 33. Female habitus in dorsal view.
Triorla milineaesp. nov. 34. Male terminalia in dorsal view; 35. Male terminalia in ventral view; 36. Male terminalia in lateral view; 37. Ovopositor in ventral view; 38. Ovopositor in lateral view; 39. Genital fork and spermathecae. Abbreviations: cerc: cercus; ej apod: ejaculatory apodeme; epand: epandrium; f apod: furcal apodeme; goncx apod: gonocoxal apodeme; gen fk: genital fork; goncx: gonocoxite; gonst: gonostylus; hypd: hypandrium; ind duc: individual duct; spmth: spermathecae; st VIII: sternite VIII; t IX+X: tergite IX+X.
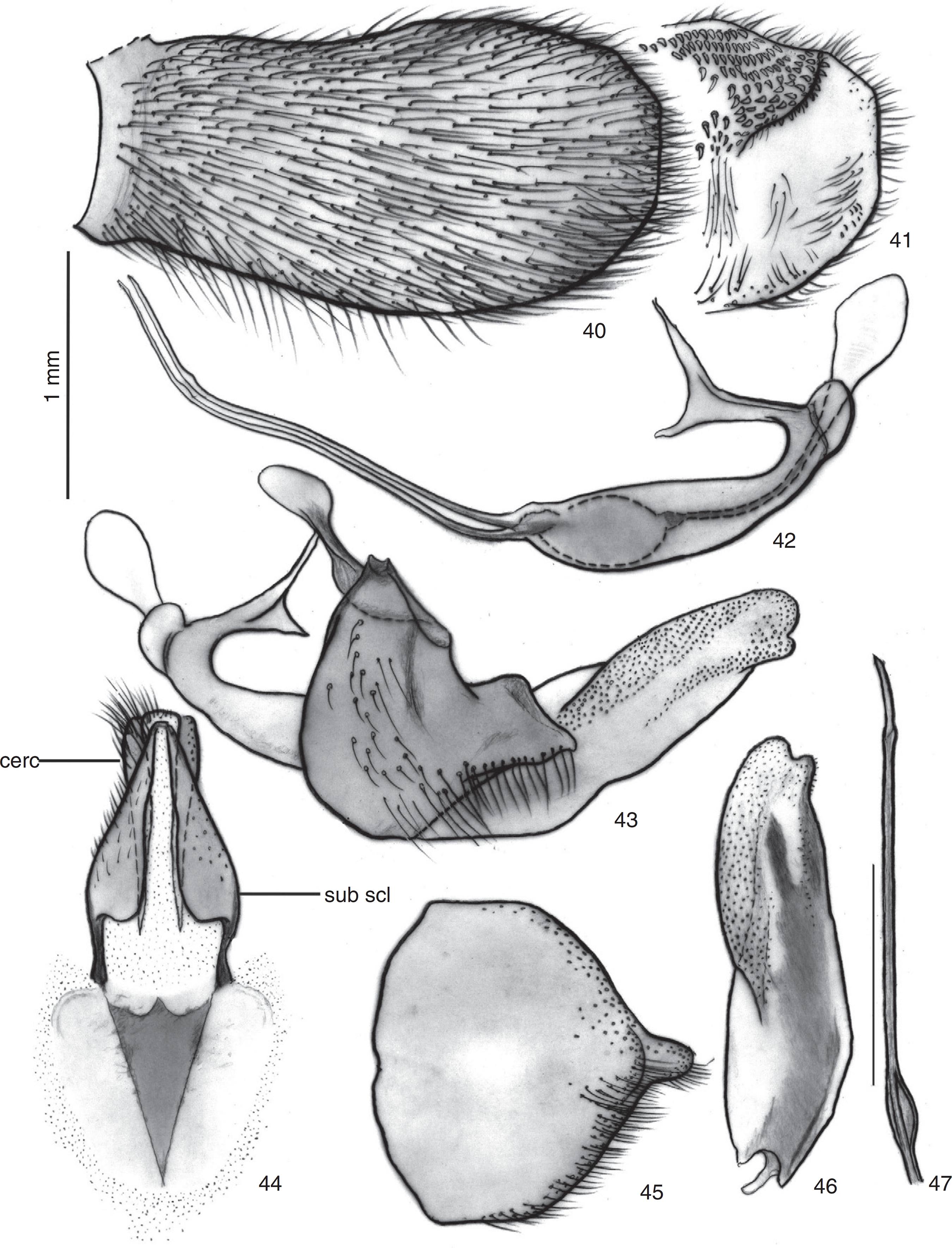
Triorla milineaesp. nov. 40. Epandrium in lateral view; 41. Apex of inner margin of epandrium; 42. Aedeagus and ejaculatory apodeme in lateral view; 43. Aedeagus and ejaculatory apodeme in lateral view, gonocoxite and gonostylus; 44. subepandrial sclerite; 45. Hypandrium; 46. Gonostylus in lateral view; 47. Aedeagal prong. Abbreviations: cerc: cercus; sub scl: subepandrial sclerite.
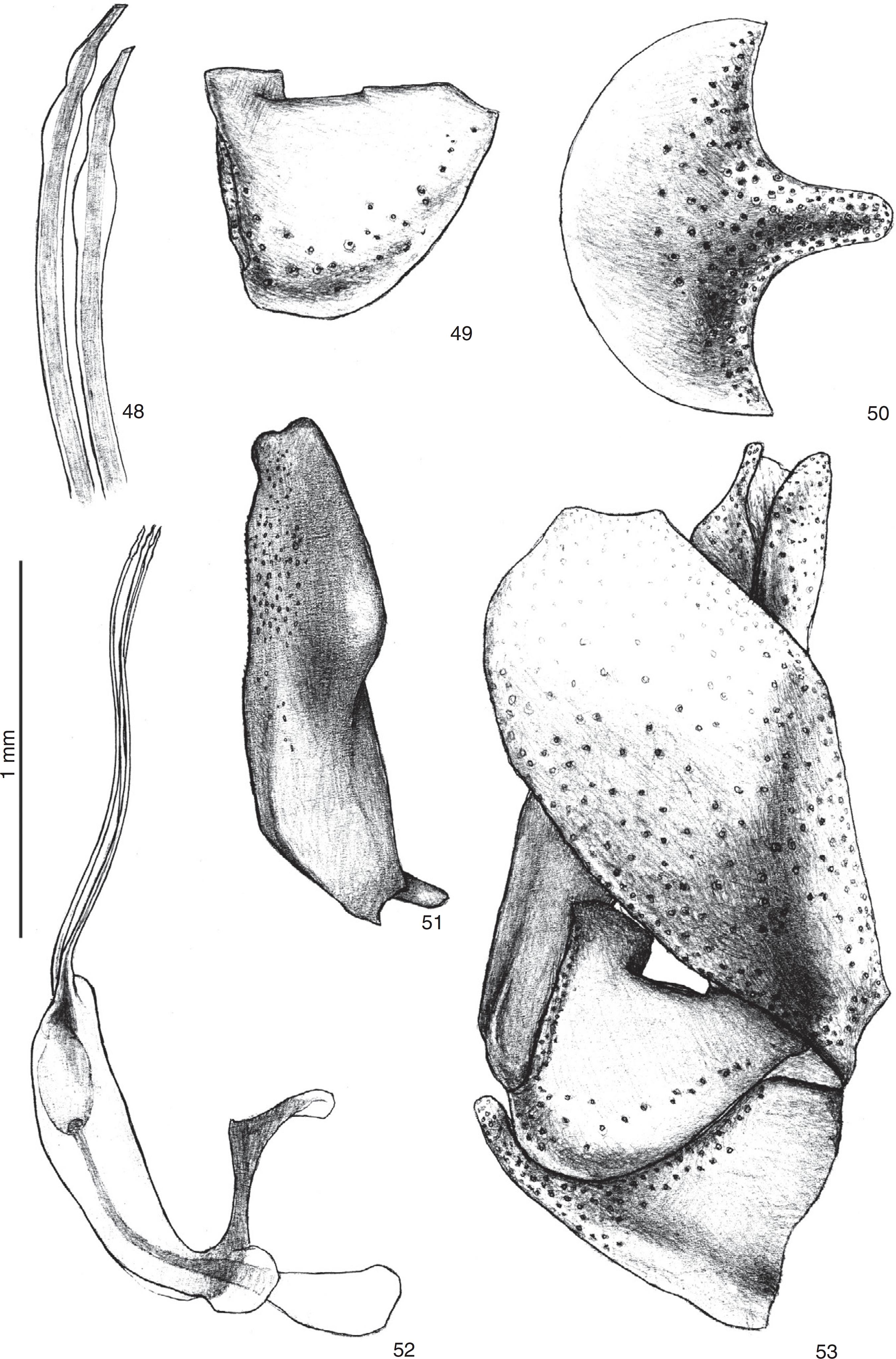
Triorla paraensissp. nov. 48. Apex of aedeagal prongs; 49. Gonocoxite; 50. Hypandrium; 51. Gonostylus in lateral view; 52. Aedeagus and ejaculatory apodeme in lateral view; 53. Male terminalia in lateral view.
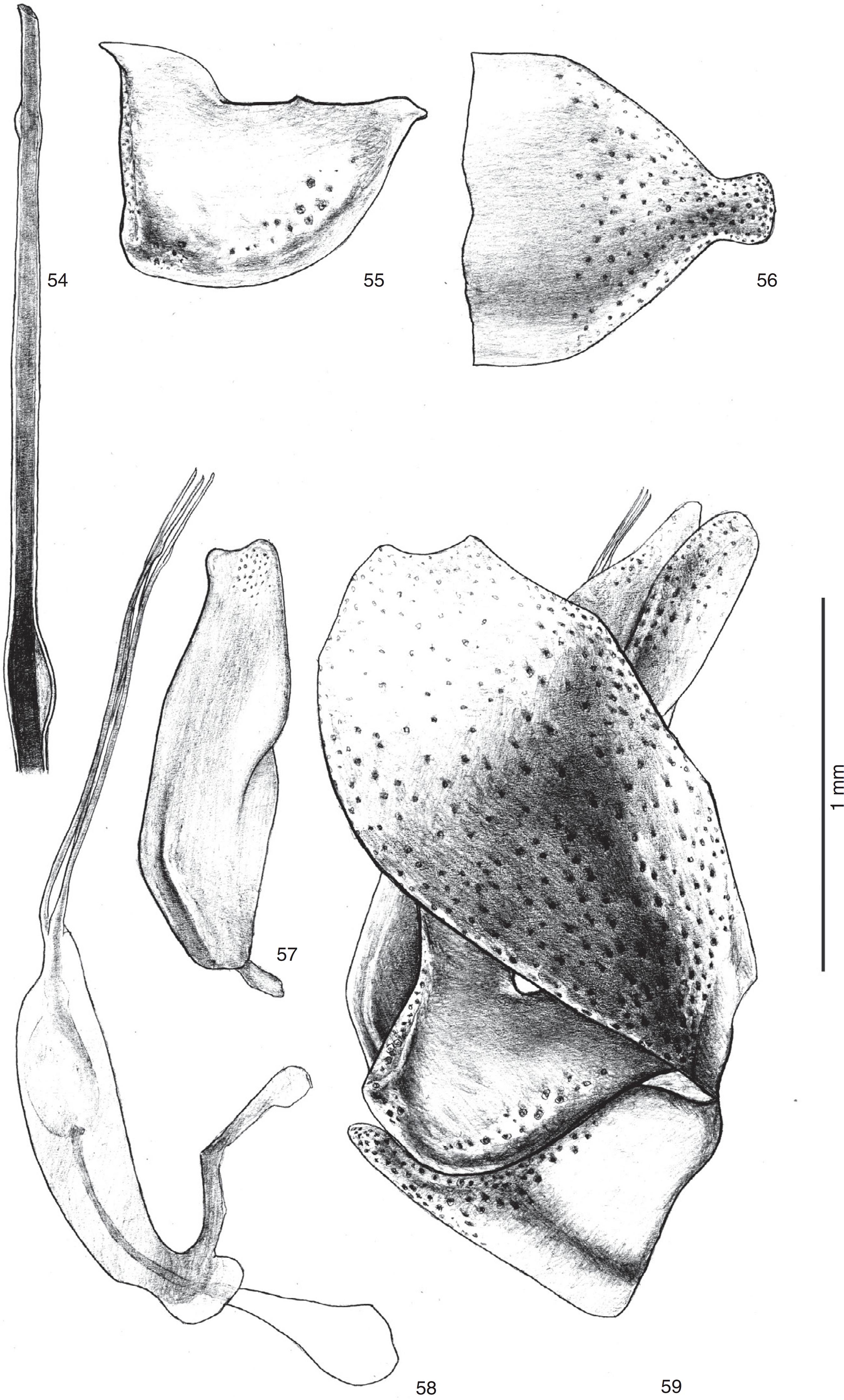
Triorla spatulatasp. nov. 54. Apex of aedeagal prongs; 55. Gonocoxite; 56. Hypandrium; 57. Gonostylus in lateral view; 58. Aedeagus and ejaculatory apodeme in lateral view; 59. Male terminalia in lateral view.
Triorla parastriola Pamplona and Aires, 1999Pamplona, D.M., Aires, C., 1999. Revision of Triorla Parks and description of a new species emphasizing the systematic value of spermatheca (Diptera, Asilidae). Revista Brasileira de Zoologia 16, 1127-1134.
Triorla parastriolaPamplona & Aires, 1999Pamplona, D.M., Aires, C., 1999. Revision of Triorla Parks and description of a new species emphasizing the systematic value of spermatheca (Diptera, Asilidae). Revista Brasileira de Zoologia 16, 1127-1134.: 1128, 1130–1131, Figs. 1, 2, 7, 8, 11, 14; Papavero, 2009Papavero, N., 2009. Catalogue of Neotropical Diptera. Asilidae. Neotropical Diptera 17, 1-179.: 24 (catalogue).
Comments: T. parastriola differs from the others species of the genus by the characters presented in the key. Head as shown (Figs. 5, 10). T. parastriola has a predominantly black abdomen, except for the lateral silver areas; tergite I laterally with 2–4 black macrosetae and yellow setae; apex of epandrium curved distally (Fig. 21); inner margin of epandrium with spines on distal 2/3; hypandrium pentagonal with apex of median-apical rounded projection (Fig. 18); ejaculatory apodeme without spines (Fig. 20). Head, wing and other anatomical characteristics of male terminalia as shown (Figs. 5, 10, 14, 16–21).
Type material examined. Paratypes: BRASIL, Roraima: Surumu, IX.1966, M. Alvarenga & F.M. Oliveira leg. (7♂, 3♀ MZUSP).
Triorla beckeri Castro, Bravo and Vieira sp. nov.
Diagnosis: four ocellar setae; face and front yellow pruinose; dorsal margin of anepisternum white setose; 8 pronotal macrosetae; tergite I with 3–4 black and yellow macrosetae; epandrium with apical concavity in lateral view, macrosetae in mediodorsal region (Fig. 29); gonocoxite triangular, ending in an acute, short projection in posteroventral region (Fig. 23); gonostylus 5.5× longer than wide, bilobed at apex (Fig. 27); distal region of hypandrium without median projection (Fig. 24); ejaculatory apodeme without spines (Fig. 28); aedeagal prongs with 1 subapical dilation (Figs. 22, 28).
Description. Male. Head (Figs. 1, 6). Ocellar tubercle with 4 yellow setae at apex, 4–6 brown setae laterally, 17–19 yellow postocular macrosetae, 4–7 black and yellow lateral orbital macrosetae. Mystax: black macrosetae dorsally, yellow macrosetae laterally; median and inferior region with 12 macrosetae and setae, the inferior macrosetae transversally distributed; 3–4 pairs of subvibrissal macrosetae. Inferior occipital setae white and branched, and 12–13 occipital macrosetae. Basal proboscial white setae. Mentum with yellow and apically undulated setae. Palpus brown setose on distal half and yellow to pale on proximal half. Face and front yellow pruinose; face wide in frontal view, face at antennal level 1.2× as wide as eyes. Front with brown transversal vitae; gena and epistomal margin brownish-white pruinose. Scape 2.0× the length of pedicel, black setae dorsally and yellow setae ventrally. Facial gibbosity developed. Lateral width of gena 0.25× the lateral length of compound eye.
Thorax. Eight pronotal macrosetae. Postpronotal lobe white setose; 2 pairs of black notopleural macrosetae; 2 pairs of black supraalar macrosetae; 2 pairs black postalar macrosetae; 6 black scutellar macrosetae apically; scutellar disc yellow, setose yellow basally, black apically, undulated at apex; posterior acrostichal setae replaced by black apically undulated setae; 4pairs dorsocentral black macrosetae. Mesonotum mainly brownish. Anepisternum with white setae on dorsal margin; basalar sclerite black setose; katepisternum with few white setae; anepimeron with some white setae; katepimeron glabrous.
Wing. (Fig. 11); membrane yellowish. Halters brownish.
Abdomen (Fig. 25). Tergite I with 3–4 black and yellow macrosetae; posterior margin fringed with yellow macrosetae, remaining surfaces yellow setose. Lateral margins of tergite II yellow setose; dorsal surface yellow setose. Lateral margins of tergite III with short yellow setae. Tergites IV–V dark brown to black with transversal posterior band light-brown. Tergite VI white, with a median and triangular black spot. Tergite VII black, with lateral white margins.
Terminalia (Figs. 22–24, 26–29). Epandrium in lateral view, with mediodorsal region bearing differentiated macrosetae (Figs. 26, 29); cercus elliptic, ending at same level as alate projections of subepandrial sclerite. Hypandrium with rounded distal margin, without median projection (Fig. 24). Gonocoxite triangular, ending in acute, short projection in posteroventral region (Fig. 23). Gonostylus 5.5× longer than wide and 1.5× the length of gonocoxite, bilobed at apex (Fig. 27). Aedeagal prongs with subapical dilation (Figs. 22, 28); aedeagal sheath as long as prongs (Fig. 28), dilation of median prong situated more anteriorly than same on lateral prongs (Fig. 22).
Size. Body length 15 mm.
Female. Unknown.
Etymology. This species is named for Johann Becker, a Brazilian entomologist.
Geographical records. Brazil: Bahia State.
Type material. Holotype: BRASIL, Bahia, Camaçari, Dunas de Jauá [12°46'10" S, 38°10'21" W], xii.1993. J. Becker col., MZFS#29005 (♂ MZFS).
Triorla mileneae Castro, Bravo & Vieira sp. nov.
Diagnosis: seven black ocellar setae; face and front covered with yellow and brown pruinescence; anepisternum with white setae on dorsal surface; seven to nine pairs of yellow pronotal macrosetae; tergite I with four to six black macrosetae; distal margin of gonocoxite pointed, ventral margin curved (Fig. 43); gonostylus 2.0× longer than wide (Figs. 43, 46); epandrial apex without posteroventral acute projection and without rounded median projection (Figs. 36, 40); ejaculatory apodeme without concentric rows of diminute spines (Fig. 42); aedeagal prongs with two subapical dilations (Figs. 42, 47).
Description. Male, habitus lateral (Fig. 30) and frontal (Fig. 31). Head (Figs. 2, 7). Seven black ocellar setae, some of them undulated at apex; seven to eight black and brown postocular macrosetae; five to seven orbital macrosetae; dorsal portion of mystax with five to six macrosetae; median and ventral portion of mystax with 12 brown macrosetae; three to five black subvibrissal macrosetae; inferior occipital setae white and branched; five to seven occipital macrosetae, yellow or white; proboscial setae white and branched basally, apically covered with white setae undulated at apex; palpus with black setae. Face and front covered with yellow and brown pruinescence; face wide at antennal level, as wide as eye in lateral view; front with a dark brown transversal spot; gena with brown pruinescence. Scape 2.0× the length of pedicel, with white setae on ventral surface; postpedicel 1.6–1.7× the length of style. Gena covered with dark brown pruinescence.
Thorax. Seven to nine pairs of yellow pronotal macrosetae; postpronotal lobe with white setae; two black notopleural macrosetae; two black supraalar macrosetae; one to two pairs of postalar black macrosetae; nine to ten black apical scutellar macrosetae; discal setae black, undulated at apex; posterior acrostichal setae missing, replaced by undulated setae; two dorsocentral macrosetae. Anepisternum with white setae on dorsal surface; basalar sclerite with black setae; katepisternum, anepimeron, and katepimeron glabrous.
Wing (Fig. 12); membrane yellowish. Halter brown.
Abdomen (Figs. 30, 32, 33). Tergite I with four to six macrosetae, black; posterior margin fringed with white macrosetae, remaining surfaces covered with white setae, some of them undulated at apex. Lateral margins of tergite II with white setae; dorsal margin with short setae. Lateral margins of tergite III with white setae, dorsal margin with black setae. Tergite VI white, with black median spot. Tergite VII white, with median trapezoidal spot. Tergite VIII white.
Terminalia (Figs. 34–36, 40–47). Lateral view (Fig. 36). Reddish to brown; cercus elliptic, ending at same level as alate projections of subepandrial sclerite. Hypandrium with distal margin bearing a terminal projection with basal bulbosity (Fig. 45). Distal margin of gonocoxite pointed, ventral margin curved (Fig. 43). Gonostylus 2× longer than wide (Fig. 46). Aedeagal prongs with two subapical dilations (Figs. 42, 47); aedeagal sheath as long as length of prongs (Fig. 42). Dilation of median prong situated more anteriorly than lateral prongs (Figs. 42, 47). Ejaculatory apodeme without concentric rows of diminute spines (Figs. 42, 43).
Size. Body length 15–22 mm (N = 110).
Female (Figs. 33, 37–39). Habitus (Fig. 33). Similar to male, except by: body length17–23 mm (N = 49); tergite VII white with dorsal portion black, with white stripe on lateral margins (Fig. 33); tergite VIII shiny black (Fig. 33). Ovipositor conic (Figs. 33, 37, 38); genital fork with hyaline base (Fig. 39); furcal arms convergent, longer than furcal apodeme (Fig. 39).
Etymology. This species is named after Dra. Milene Castro, the senior author's beloved wife.
Geographical records. BRAZIL: States of Ceará and Bahia.
Type material. Holotype: BRASIL, Bahia, Feira de Santana [12°15'20" S, 38°57'15" W], 26.v.2008. I. Castro col. (MZFS#41892) (♂ MZFS).
Paratypes. MZFS: Bahia state. Barrolândia [15°51'46" S, 38°52'58" W]: 12.iii.1994, J. Santos col. (1♂); 02.xii.1997 (2♂); 2.iii.1998 (1♂); 01.v.1998 (1♂); 22.v.1998 (1♂); 25.ix.1998 (1♂); 18.xii.1998 (1♂); 8.i.1999 (3♂); 5.iii.1999 (1♂); 16.iv.1999 (1♂); 20.ii.2002 (1♂); 12.iii.1994 (1♀); 27.ii.1998 (1♀); 09.x.1998 (2♀ MZFS); 6.xi.1998 (1♀); 11.xii.1998 (1♀); 5.iii.1999 (1♀). Baixios 12°06'S; 37°41'W: 25.iv.2008, P. Rebouças col. (5♂); 14.vi.2008 (1♂); 25.iv.2008 (1♀). Bom Jesus dos Pobres [12°47'60" S, 38°47'0" W]: 20.i.2006, E. Alvim col. (6♂, 2♀). Cachoeira, Fazenda Vila Rial [12°35'20" S, 38°58'8" W]: 14.vi.2007, F. Bravo col. (1♂); 24.v.2004 (6♀ MZFS); 20.vii.2004, R. Vieira col. (2♀). Camaçari, Dunas de Jauá [12°46'10" S 38°10'21" W]: xii. 1993, J. Becker, col. (1♂). Feira de Santana, UEFS [12°15'20" S, 38°57'15" W]: 27.v.2002, I. Castro col. (1♂); 24.ix.2002 (1♂); 29.iv.2003 (1♂); 6.v.2003 (1♂ MZFS); 19.ix.2003 (1♂); 5.vii.2006 (1♂); viii.2005 (1♂); 10.iv.2007 (1♀); 7.x.2002 (1♀); 6.xi.2004 (1♀); 9.vi.2003 (1♀); 19.xi.2003 (1♀); 25.ix.2003 (1♀); 8.x.2003 (1♀); 7.vi.2003, A.P. Assunção col. (1♂); 3.viii.2003 (1♂ MZFS); 3.vii.2008 (1♀) 30.viii.2006, E. Alvim col. (1♂); 14.xii.2006 (1♂); 25.vii.2008, T. Mahlmann col. (1♂); 6.viii.2005, M. Castro col. (1♂). Itabuna [14°47'21" S, 39°16'40" W]: 28.ii.1998, J. Santos col. (2♂); viii.1998 (4♂); 10.x.1998 (2♂); 17.x.1998 (1♂); 30.i.1999 (3♂); 6.ii.1999 (3♂); 5.iv.1999 (2♂); viii.1998 (2♀); 30.i.1999 (3♀); 13.ix.2002 (2♀). Lençóis [12°33'25" S, 41°23'31" W]: 14.xii.2002 (2♂); 5.x.2003, I. Castro col. (2♂); 10.x.2003, D. Lefundes col. (2♂); 11.x.2003, R. Vieira col. (1♀); 10.x.2003 (1♀). Luiz Eduardo Magalhães [12°5'57" S, 45°47'53" W]: 20.vii.2005, E. Alvim col. (1♂ MZFS). Monte Santo [10°26' S, 39°19' W]: 20.vii.2005, E. Alvim col. (3♀). Maracás [13°25'41" S, 40°26'16" W]: 19.xi.2004, I. Castro col. (3♂). Mucugê [12°59'46" S, 41°22'11" W]: 9.xi.2002, F. Bravo (2♂); 19.vii.2003, R. Vieira col. (2♂). Raso da Catarina [10°3'50" S, 38°20'26" W]: 5.ii.2006, D. Souza col. (1♂); 5.ii.2006, P.P. Lopes col. (2♂); (Arredores do alojamento da ESEC, UTM: 05584767/8931611), altitude 700 m, 6.xi.2005, P.P. Lopes col. (1♀). Remanso [9°36'15" S, 42°6'6" W]: 2.vii.2003, I. Castro col. (1♂); 4.vii.2003 (1♀). Santa Terezinha, Serra da Jibóia [12°51' S, 39°28' W]: 30.x.2003 (12♂); 15.viii.2007, F. Bravo col. (1♂). Sobradinho [9°27'33" S, 40°49'31" W]: 5–vii–2003, I. Castro col. (1♂). Ruy Barbosa, Serra do Orobó [12°17' S, 40°29' W]: 8.iv.2005 (2♂). Sauípe [11°51'40" S, 37°35'33" W]: 7.v.2008 (1♂). Una [15°16'10" S, 39°4'9" W]: ESMAI, 7.i.2000, J. Santos col. (1♀). MZUSP: Ceará, Maria Pereira [Mombaça] [5°44'35" S, 39°37'42" W], ix. 1939. D.C. Alves col. (1♂); Bahia, Barrolândia: 01.v.1998, J. Santos col. (1♀); 19.iii.1999 (1♂); 6.xi.1999 (1♂); 9.xi.1999 (1♂); 20.xi.2002 (1♂). Feira de Santana (UEFS): 20.ix.2002, I. Castro col. (1♂); 7.iv.2003 (1♂); 10.x.2006 (1♂); 11.x.2006 (1♂); 20.ix.2002 (1♀); 4.xi.2004, Suzana col. (1♀); 7.x.2003 (1♀); Ituberá (Michelin): 3.iii.2007, E. Alvim col. (1♀). Monte Santo: 11.v.2004, Suzana col. (1♂); Porto Seguro [16°27'4" S, 39°3'52" W]: Veracel, 5.xii.2002, F. Bravo col. (1♂). INPA: Bahia, Barrolândia: EGREB, J. Santos col., 16.iv.1999 (1♂); 10/27.vii.1998 (1♂); 9.x.1998 (1♂); 31.xii.1998 (1♂); 8.ii.1999 (1♂); Dantilândia [15°6'6" S 40°59'58" W]:15.iv.2005, R. Vieira col. (1♂); 24.iv.2004 (1♂ INPA); Feira de Santana (UEFS): 26.ii.2002, I. Castro col. (1♂); 5.ix.2006, F. Bravo col. (1♀); 17.x.2007 (3♀); Senhor do Bonfim [10°27'57" S, 40°10'51" W]: 6.vii.2005, R. Vieira & E. Souza col. (1♂).
Triorla paraensis Castro, Bravo & Vieira sp. nov.
Diagnosis: nine black ocellar setae; face and front covered with yellow to brown pruinescence; anepisternum with a tuff of black and white setae on dorsal margin; 14 yellow pronotal macrosetae; tergite I with black macrosetae and brown undulated setae on apex; gonocoxite with rectangular posteroventral projection (Figs. 49, 53); gonostylus bilobed apically (Fig. 51); proximal margin of hypandrium rounded, distal margin with long median projection (Fig. 50); epandrium in lateral view, with slight concavity (Fig. 53); ejaculatory apodeme without spines (Fig. 52); aedeagal prongs with two adjacent subapical dilations, the more apical one less inflated (Figs. 48, 52).
Description. Male. Head (Figs. 4, 9). Nine ocellar setae, black and undulated at apex; eight to twelve brown setae on lateral surface; six to seven black and brown postocular macrosetae; four to five black and yellow orbital macrosetae; dorsal region of mystax with seven black macrosetae, median and ventral region with 14 brownish macrosetae; three to four black subvibrissal macrosetae; inferior occipital setae white and branched; six occipital brownish macrosetae; proboscial setae white and branched at base, white-yellowish and undulated at apex; palpus with black setae. Face and front covered with yellow to brown pruinescence; face wide at antennal level, the same width as eye in lateral view; front with transversal spot; gena covered with brown pruinescence. Scape 2.0× the length of pedicel; scape with ventral yellow setae and some black setae on dorsal surface; postpedicel 1.5× the lengths of scape and pedicel together, base dark brown, stylus dark brown to black. Gena dark brown, lateral width of gena 0.17× the lateral length of compound eye.
Thorax. 14 yellow pronotal macrosetae; postpronotal lobe covered with black and white seta; two black notopleural macrosetae; two black supraalar macrosetae; one to three black postalar macrosetae; 12–14 apical scutellar macrosetae; discal setae dark brown, undulated at apex; two black acrostichal setae; four dorsocentral macrosetae. Postalar wall with row of yellow setae. Anepisternum with tuff of black and white setae on dorsal margin; basalar sclerite with black setae. Katepisternum with many brown setae. Katepimeron glabrous.
Wing. (Fig. 13); membrane yellowish. Halters brown.
Abdomen. Tergite I with black macrosetae and brown setae undulated at apex, posterior margin fringed with macrosetae and setae, the remaining surface covered with black and white setae. Lateral margins of tergite II with white setae, dorsal margin covered with short white setae. Lateral margins of tergite III with black setae; dorsal surface covered with short black setae. Tergites IV and V black. Tergite VI white, with median triangular spot. Tergite VII black, with white lateral stripe.
Terminalia (Figs. 48–53). Proximal margin of hypandrium rounded, distal margin with long median projection (Fig. 50). Gonocoxite with rectangular posteroventral projection (Figs. 49, 53). Gonostylus ending with two short lobes (Fig. 51). Aedeagal prongs with two terminal dilations each, proximal dilations wider than apical (Figs. 48, 52). Ejaculatory apodeme without spines (Fig. 52).
Size. Body length 18 mm (N = 02).
Female. Unknown.
Etymology. Name is based on type locality.
Geographical records. BRAZIL: Pará State.
Type material. Holotype: BRASIL, Pará, Primavera, Fazenda Feitoria Quatipuru [00°56'34" S, 47°06'57" W]. 27.xi.1992. Dias, J. col. (♂ INPA).
Paratype: BRASIL, Pará, Primavera, Ilha Arapiranga [00°56'34" S, 47°06'57" W] 23.xi.1992. J. Dias col. (1♂ INPA).
Triorla spatulata Castro, Bravo & Vieira sp. nov.
Diagnosis: eight ocellar setae; face and front covered with yellow and brown pruinescence; anepisternum with tuff of black and white setae on dorsal margin; 12 pronotal macrosetae; tergite I with three to four black macrosetae; hypandrium subtriangular with median-apical spatulate project (Fig. 56); epandrium in lateral view, with slight concavity (Fig. 59); gonostylus bilobed distally (Fig. 57); ejaculatory apodeme without spines (Fig. 58); aedeagal prongs with two subapical dilations, median tube shorter than lateral (Fig. 58).
Description. Male. Head (Figs. 3, 8). Eight ocellar setae on apex; eight to ten brown setae on lateral tubercular, undulated at apex; nine black postocular macrosetae; 12 black orbital macrosetae. Dorsal portion of mystax with 13 black macrosetae, median and ventral region covered with yellowish macrosetae; three to five subvibrissal black macrosetae; inferior occipital setae white and branched; 14–18 orbital macrosetae. Basal half of palpus with white and brown setae; apical half with black setae. Face and front covered with yellow and brown pruinescence; face wide at antennal level, as wide as one eye in frontal view. Front with transverse spot. Scape 2.0× the length of pedicel, with yellow setae on ventral surface, black on dorsal surface; postpedicel 1.5× the lengths of scape and pedicel together. Gena dark brown, lateral width of gena 0.20× the lateral length of compound eye.
Thorax. 12 pronotal macrosetae; postpronotal lobe with black and white setae, undulated at apex; two notopleural macrosetae; two black supraalar macrosetae; one to two pairs of postalar macrosetae; eight apical scutellar macrosetae; discal setae black; two acrostichal setae; two pairs of black dorsocentral macrosetae. Postalar wall with yellow setae. Anepisternum with tuff of black and white setae on dorsal margin; basalar sclerite with one macroseta. Katepisternum with some white setae. Katepimeron glabrous.
Wing. (Fig. 15); membrane yellowish. Halters brown.
Abdomen. Tergite I with three to four black macrosetae; posterior margin without fringe, remaining surfaces covered with black and white setae. Lateral margins of tergite II with brown setae anteriorly and white setae posteriorly; dorsal margin with short black setae. Lateral margins of tergite III with brown setae anteriorly and white and undulated setae posteriorly. Tergite VI white, with triangular median spot. Tergite VII dark brown to black, with lateral white stripe.
Terminalia (Figs. 54–59). Gonocoxite with posteroventral projection with acute extension (Fig. 55). Gonostylus bilobed distally, dorsal lobe longer than ventral (Fig. 57). Gonostylus 3.0× longer than wide (Fig. 57). Aedeagal prongs with two subapical dilations, median tube shorter than lateral (Fig. 58). Ejaculatory apodeme without spines (Fig. 58). Hypandrium subtriangular with median-apical spatulate project (Fig. 56).
Size. Body length 18–22 mm (N = 05).
Female. Unknown.
Etymology. This name refers to the spatulate shape of the hypandrial apex.
Geographical records. BRAZIL: States of Amazonas, Pará, São Paulo, Mato Grosso do Sul.
Type material. Holotype: BRASIL, São Paulo, Araraquara [21°47'38" S, 48°10'27" W], xii.1946. J. Lane col. (♂ MZUSP).
Paratypes: BRASIL, Amazonas, Manaus, Campus da UFAM [Universidade Federal do Amazonas] [3°8'2" S, 60°1'7" W] 30.vi.1979. J.A. Rafael col. (1♂ INPA); [BRASIL], Pará, Gorotire, Xingú [7°50'57" S, 51°50'17" W] 18.iv.1983. W. Overal col. (1♂ MPEG); [BRASIL], Mato Grosso [do Sul], Maracaju [21°37'7" S, 55°10'2" W] v.1937. M.E.S. Bras (1♂ MZUSP); [BRASIL], Mato Grosso do Sul, Corumbá, Fazenda Alegria [19°0'53" S, 57°38'59" W]22.iv.1994. Tadeu & Gorayeb col (1♂ MZUSP).
Discussion
The key presented by Artigas and Papavero (1997)Artigas, J.N., Papavero, N., 1997. The American genera of Asilidae (Diptera): keys for identification with an atlas of female spermathecae and other morphological details. IX.2. Subfamily Asilinae Leach – Efferia group, with the proposal of five new genera and a catalogue of the neotropical species. Arquivos de Zoologia do Estado de São Paulo 34, 65-95. for the genera of the "Efferia group" did not allow for the identification of Triorla, since some of the steps in that key did not correspond to the characters observed in the species of that genus. Therefore, the following discussion covers the steps of the key from Artigas and Papavero (1997)Artigas, J.N., Papavero, N., 1997. The American genera of Asilidae (Diptera): keys for identification with an atlas of female spermathecae and other morphological details. IX.2. Subfamily Asilinae Leach – Efferia group, with the proposal of five new genera and a catalogue of the neotropical species. Arquivos de Zoologia do Estado de São Paulo 34, 65-95. that should lead to the identification of Triorla, plus the novel information found in this study.
-
"Costal section situated between tips of R5 and M1 two or more times longer than costal section situated between tips of R5 and R4; R5 ends at or above wing apex" (step 3, p. 58, Artigas and Papavero, 1997Artigas, J.N., Papavero, N., 1997. The American genera of Asilidae (Diptera): keys for identification with an atlas of female spermathecae and other morphological details. IX.2. Subfamily Asilinae Leach – Efferia group, with the proposal of five new genera and a catalogue of the neotropical species. Arquivos de Zoologia do Estado de São Paulo 34, 65-95.). This character is present in the wings of Triorla (Figs. 11–15).
-
"Hypandrium produced; aedeagus with 3 tubes; female tergite 10 never with spines or spinules; spermathecae with 3 capsules" (step 4', p. 58, Artigas and Papavero, 1997Artigas, J.N., Papavero, N., 1997. The American genera of Asilidae (Diptera): keys for identification with an atlas of female spermathecae and other morphological details. IX.2. Subfamily Asilinae Leach – Efferia group, with the proposal of five new genera and a catalogue of the neotropical species. Arquivos de Zoologia do Estado de São Paulo 34, 65-95.). Regarding the development of the hypandrium, this step is vague and without any indication of its proportion to other terminalia structures. The characters proposed by the authors above are present in all genera of the Efferia group.
-
"Wings with a stump vein at the angle of R4, near its junction with R5; aedeagus most characteristically curved, more or less crescent-shaped, formed by a very long common tube which opens at the apex into 3 very short tubes, normally forming a "parrot beak"-like structure; male terminalia slender and elongate, forming an angle (up to 90°) with body axis. Female ovipositor strongly flattened laterally, blade-like, tergite 8, more or less elongate and slender" (step 5, p. 59, Artigas and Papavero, 1997Artigas, J.N., Papavero, N., 1997. The American genera of Asilidae (Diptera): keys for identification with an atlas of female spermathecae and other morphological details. IX.2. Subfamily Asilinae Leach – Efferia group, with the proposal of five new genera and a catalogue of the neotropical species. Arquivos de Zoologia do Estado de São Paulo 34, 65-95.). The wing character proposed by Artigas and Papavero (1997)Artigas, J.N., Papavero, N., 1997. The American genera of Asilidae (Diptera): keys for identification with an atlas of female spermathecae and other morphological details. IX.2. Subfamily Asilinae Leach – Efferia group, with the proposal of five new genera and a catalogue of the neotropical species. Arquivos de Zoologia do Estado de São Paulo 34, 65-95. in step 5 was observed in all Triorla species. The aedeagus of the species of Triorla, on the other hand, always had 3 long tubes (Figs. 20, 28, 42, 52, 58), and is therefore different from that described by Artigas and Papavero (1997)Artigas, J.N., Papavero, N., 1997. The American genera of Asilidae (Diptera): keys for identification with an atlas of female spermathecae and other morphological details. IX.2. Subfamily Asilinae Leach – Efferia group, with the proposal of five new genera and a catalogue of the neotropical species. Arquivos de Zoologia do Estado de São Paulo 34, 65-95. in step 5. The inclination of the male terminalia, in relation to the longitudinal axis of the body, varied among the studied Brazilian specimens of the genus. In the diagnosis of the genus provided by Parks (1968)Parks, L., 1968. Synopsis of robberfly genera allied to Efferia and Eicherax, including a new genus. Pan-Pac. Entomol. 44, 171-179., the male terminalia is aligned with the longitudinal axis of the body. Pamplona and Aires (1999)Pamplona, D.M., Aires, C., 1999. Revision of Triorla Parks and description of a new species emphasizing the systematic value of spermatheca (Diptera, Asilidae). Revista Brasileira de Zoologia 16, 1127-1134. and Tomasovic (2002)Tomasovic, G., 2002. Étude sur matériaux typiques du complexe génital mâle de spet espèce du genre Erax Scopoli, 1763 (Diptera Asilidae) avec la description de trois espèces nouvelles. Notes faunistiques de Gembloux 46, 27-37. did not report this male character in their descriptions of the genus. According to Parks (1968)Parks, L., 1968. Synopsis of robberfly genera allied to Efferia and Eicherax, including a new genus. Pan-Pac. Entomol. 44, 171-179., the ovipositor in Triorla is tapered and shiny black. Pamplona and Aires (1999)Pamplona, D.M., Aires, C., 1999. Revision of Triorla Parks and description of a new species emphasizing the systematic value of spermatheca (Diptera, Asilidae). Revista Brasileira de Zoologia 16, 1127-1134. and Tomasovic (2002)Tomasovic, G., 2002. Étude sur matériaux typiques du complexe génital mâle de spet espèce du genre Erax Scopoli, 1763 (Diptera Asilidae) avec la description de trois espèces nouvelles. Notes faunistiques de Gembloux 46, 27-37. made no reference to the shape and colour of the ovipositor. The ovipositors of the females of the new species currently described are tapered and shiny black, which agrees with Parks (1968)Parks, L., 1968. Synopsis of robberfly genera allied to Efferia and Eicherax, including a new genus. Pan-Pac. Entomol. 44, 171-179..
In spite of the differences between the Artigas and Papavero (1997)Artigas, J.N., Papavero, N., 1997. The American genera of Asilidae (Diptera): keys for identification with an atlas of female spermathecae and other morphological details. IX.2. Subfamily Asilinae Leach – Efferia group, with the proposal of five new genera and a catalogue of the neotropical species. Arquivos de Zoologia do Estado de São Paulo 34, 65-95. identification key and the characters observed herein, some other particularities are also highlighted in this discussion.
Like most of the species of Asilinae, the new species of Triorla currently proposed are very similar to each other, being distinguished by characters of the male and female terminalia – structures that are mentioned in the diagnosis of each species and in the identification key.
Triorla beckerisp. nov., T. milineaesp. nov., Triorla paraensissp. nov. and T. spatulata sp. nov. differ from T. ambigua, T. rubidiventris, T. argyrogaster, and T. nervosa by the epandrium not being V-shaped in lateral view, and by the distal margin of the epandrium with at most two projections (Figs. 26, 36, 53, 59).
The basal margins of the hypandrium of T. spatulata sp. nov. and T. parastriola are more or less straight (Figs. 18, 56), but the apex of the hypandrium of T. spatulata sp. nov. is truncated while in T. parastriola it is rounded. Furthermore, the basal third has parallel lateral margins (Fig. 56) in T. spatulata sp. nov. while in T. parastriola these margins are slightly converging towards the base. (Fig. 18).
Triorla mileneaesp. nov. differs from T. interrupta by the posteroventral margin of the epandrium lacking projections (Fig. 36); differs from T. trichina by the rounded distal margin of the epandrium in lateral view; differs from T. striola by the ejaculatory apodeme lacking rows of spinules; and differs from T. demifasciata by the acute distal margin of the gonocoxite and the curved ventral margin.
Triorla striola can be distinguished from other species of the genus, including the new species proposed in this paper, by the presence of concentric spines on the entire blade of the aedeagal apodeme. Triorla striola has been recorded for South America and Brazil, more specifically for the states of Rio Grande do Norte, Espírito Santo, Minas Gerais, Mato Grosso, Goiás, and Paraná (Pamplona and Aires, 1999Pamplona, D.M., Aires, C., 1999. Revision of Triorla Parks and description of a new species emphasizing the systematic value of spermatheca (Diptera, Asilidae). Revista Brasileira de Zoologia 16, 1127-1134.). Despite its wide distribution, this species has not yet been collected in the state of Bahia.
Acknowledgments
To Augusto Loureiro (INPA), Orlando Tobias (MPEG), Carlos Lamas (MZUSP) for the loan of specimens; to FAPEAM EDITAL N. 022/2013 – FIXAM/AM, # 062.00745/2014; to Roy Funch who revised English manuscript, anonymous reviewers and Sarah Siqueira de Oliveira for helpful comments that improved the manuscript.
References
- Artigas, J.N., Papavero, N., 1997. The American genera of Asilidae (Diptera): keys for identification with an atlas of female spermathecae and other morphological details. IX.2. Subfamily Asilinae Leach – Efferia group, with the proposal of five new genera and a catalogue of the neotropical species. Arquivos de Zoologia do Estado de São Paulo 34, 65-95.
- Artigas, J.N., Vieira, R., 2014. New genus and species of Neotropical robber flies (Diptera, Asilidae, Asilinae). Zootaxa 3774, 282-288.
- Cumming, J.M., Wood, D.M., 2009. Adult morphology and terminology. In: Brown, B.V., Borkent, A., Cumming, J.M., Wood, D.M., Woodley, N.E., Zumbado, M.A. (Eds.), Manual of Central American Diptera, vol. 1. National Research Council Research Press, Ottawa, pp. 9–50.
- Fabricius, J.C., 1805. Systema Antliatorum Secundum Ordines, Genera, Species. Brunsvigae (Brunswick).
- Geller-Grimm, F., 2004. A world catalogue of the genera of the family Asilidae (Diptera). Studia Dipterologica 10, 473-526.
- Londt, J.G.H., 2005. An annotated key to the genera of Afrotropical Apocleinae, with descriptions of six new genera (Diptera: Asilidae). Tijdschrift voor Entomologie 148, 39-62.
- Macquart, P.J.M., 1834. Histoire naturelle des Insectes. Diptères I. Roret, Paris.
- Macquart, P.J.M., 1838. Diptères exotiques nouveaux ou peu connus. Mémoires de La Société royale des Sciences, de l'Agriculture et des Artes 1, 5-207.
- Macquart, P.J.M., 1846. Dipteres exotiques nouveaux ou peu connus. 1. Supplément. Mémoires de la Société royale des Sciences, de l'Agriculture et des Artes (1845), 133-366.
- Macquart, P.J.M., 1850. Dipteres exotiques nouveaux ou peu connus. 4. Supplément. Mémoires de la Société royale des Sciences, de l'Agriculture et des Artes (1849), 61–96, 309–479.
- Martin, C.H., Papavero, N., 1970. A Catalogue of the Diptera of the Americas South of United States. 35b. Family Asilidae. Museu de Zoologia, Universidade de São Paulo, pp. 1–139.
- Parks, L., 1968. Synopsis of robberfly genera allied to Efferia and Eicherax, including a new genus. Pan-Pac. Entomol. 44, 171-179.
- Pamplona, D.M., Aires, C., 1999. Revision of Triorla Parks and description of a new species emphasizing the systematic value of spermatheca (Diptera, Asilidae). Revista Brasileira de Zoologia 16, 1127-1134.
- Papavero, N., 2009. Catalogue of Neotropical Diptera. Asilidae. Neotropical Diptera 17, 1-179.
- Scopoli, J.A., 1763. Entomologia carniolica exhibens insecta carnioliae indigena etdistributa in ordines, genera, species, varietates, methodo Linneana [Reprint Von Guglia, O. (1972)]. Vindobona.
- Stuckenberg, B.R., 1999. Antennal evolution in the Brachycera (Diptera), with a reassessment of terminology relating to the flagellum. Studia Dipterologica 6, 33-48.
- Tomasovic, G., 2002. Étude sur matériaux typiques du complexe génital mâle de spet espèce du genre Erax Scopoli, 1763 (Diptera Asilidae) avec la description de trois espèces nouvelles. Notes faunistiques de Gembloux 46, 27-37.
- Vieira, R., 2012. New distribution records of six species of Asilinae (Diptera: Asilidae) Latreille, 1802. Check List 4, 779-781.
- Vieira, R., Rafael, J., 2014. Longivena, a new robber-fly genus from Brazil (Diptera, Asilidae, Asilinae). ZooKeys 443, 119-138.
Publication Dates
-
Publication in this collection
Apr-Jun 2016
History
-
Received
10 May 2015 -
Accepted
30 Nov 2015