Abstracts
Microbial activity and biochemical properties are important indicators of the impact of organic composting on soil. The objective of this study was to evaluate some indicators of soil microbial and biochemical processes after application of compost (household waste). A Typic Acrustox, sampled at a depth of 10 cm under Cerrado biome vegetation, was evaluated in three treatments: control (soil without organic compost amendment) and soil with two doses of domestic organic compost (10 and 20 g kg-1 soil). The following properties were evaluated: released C (C-CO2): microbial respiration 15 days after incubation; microbial biomass C (MBC); total glucose (TG); metabolic quotient (qCO2); and enzyme activity of β-glucosidase and acid and alkaline phosphatase. The application of household compost, at doses of 10 and 20 g kg-1 Typic Acrustox, resulted in significant gains in microbial activity, organic C and C stock, as evidenced by increased MBC and TG levels. On the other hand, qCO2 decreases indicated greater microbial diversity and more efficient energy use. The addition of compost, particularly the 20 g kg-1 dose, strongly influenced the enzyme β-glucosidase and phosphatase (acid and alkaline). The β-glucosidase activity was significantly increased and acid phosphatase activity increased more than the alkaline. The ratio of β-glucosidase to MBC was greater in the control than in the composted treatments which suggests that there were more enzymes in the control than in the substrate or that the addition of compost induced a great MBC increase.
Cerrado; Soil respiration; microbial biomass; glucose; phosphatase; β-glucosidase.
A atividade microbiana e os atributos bioquímicos são indicadores importantes na avaliação dos impactos da adição de compostos orgânicos no solo. Assim, o objetivo deste trabalho foi avaliar alguns indicadores de processos bioquímicos e microbianos de solo, após a aplicação de composto orgânico produzido a partir de resíduos domésticos. As amostras de solo empregadas foram retiradas de um Latossolo Vermelho-Amarelo distrófico, sob vegetação de Cerrado, a 10 cm de profundidade. Foram utilizados três tratamentos: controle (solo sem adição de composto orgânico) e solo com adição de duas doses de composto orgânico doméstico (10 e 20 g kg-1 de solo seco). Os tratamentos foram avaliados quanto ao C liberado (C-CO2), à atividade respiratória microbiana aos 15 dias de incubação, ao C da biomassa microbiana (CBM); a glicose total (GT), ao quociente metabólico (qCO2) e à atividade das enzimas β-glicosidase e fosfatases ácida e alcalina. Verificou-se que as duas doses de adição de composto orgânico doméstico resultaram em aumento significativo da atividade microbiana e, também, em incremento nas frações de C orgânico e no estoque de C do solo, caracterizados pela elevação nos teores de CBM e GT. Entretanto, as duas doses de composto causaram decréscimo no qCO2, o que pode indicar maior diversidade na comunidade microbiana após o acréscimo do composto e maior eficiência no uso da energia. As enzimas β-glicosidase e as fosfatases (ácida e alcalina) foram fortemente influenciadas pela adição de composto orgânico doméstico, sobretudo pela dose de 20 g kg-1 do produto. A atividade da enzima β-glicosidase aumentou significativamente em resposta à adição do composto, e a atividade da fosfatase ácida foi superior à da fosfatase alcalina. A relação β-glicosidase: CBM foi maior no controle do que nos tratamentos com adição de composto, o que sugere que no controle havia mais enzimas do que no substrato ou que houve grande incremento no CBM com a adição do composto ao solo.
Cerrado; respiração do solo; biomassa microbiana; glicose; fosfatase; β-glicosidase.
SEÇÃO III - BIOLOGIA DO SOLO
Isabel Cristina Vinhal-FreitasI; Dalcimar Regina Batista WangenII; Adão de Siqueira FerreiraIII; Gilberto Fernandes CorrêaIV; Beno WendlingV
IStudent of Doctorate in Agronomy, Post Graduation Program, Instituto de Ciências Agrárias (ICIAG), Universidade Federal de Uberlândia (UFU). Av. Amazonas, Campus Umuarama, Bloco 2E, CEP 38400-902 Uberlândia (MG). E-mail: isabelvinhal@yahoo.com.br
IIStudent of Doctorate in Agronomy, ICIAG, UFU. E-mail: dbwangen@gmail.com
IIIAdjunct Professor of Soil Microbiology, ICIAG, UFU. E-mail: adaosferreira@yahoo.com.br
IVTitular Professor of Pedology, ICIAG, UFU. E-mail: gfcorrea@ufu.br
VAdjunct Professor of Soil Physics, ICIAG, UFU. E-mail: beno@iciag.ufu.br
ABSTRACT
Microbial activity and biochemical properties are important indicators of the impact of organic composting on soil. The objective of this study was to evaluate some indicators of soil microbial and biochemical processes after application of compost (household waste). A Typic Acrustox, sampled at a depth of 10 cm under Cerrado biome vegetation, was evaluated in three treatments: control (soil without organic compost amendment) and soil with two doses of domestic organic compost (10 and 20 g kg-1 soil). The following properties were evaluated: released C (C-CO2): microbial respiration 15 days after incubation; microbial biomass C (MBC); total glucose (TG); metabolic quotient (qCO2); and enzyme activity of β-glucosidase and acid and alkaline phosphatase. The application of household compost, at doses of 10 and 20 g kg-1 Typic Acrustox, resulted in significant gains in microbial activity, organic C and C stock, as evidenced by increased MBC and TG levels. On the other hand, qCO2 decreases indicated greater microbial diversity and more efficient energy use. The addition of compost, particularly the 20 g kg-1 dose, strongly influenced the enzyme β-glucosidase and phosphatase (acid and alkaline). The β-glucosidase activity was significantly increased and acid phosphatase activity increased more than the alkaline. The ratio of β-glucosidase to MBC was greater in the control than in the composted treatments which suggests that there were more enzymes in the control than in the substrate or that the addition of compost induced a great MBC increase.
Index terms: Cerrado, Soil respiration, microbial biomass, glucose, phosphatase, β-glucosidase.
RESUMO
A atividade microbiana e os atributos bioquímicos são indicadores importantes na avaliação dos impactos da adição de compostos orgânicos no solo. Assim, o objetivo deste trabalho foi avaliar alguns indicadores de processos bioquímicos e microbianos de solo, após a aplicação de composto orgânico produzido a partir de resíduos domésticos. As amostras de solo empregadas foram retiradas de um Latossolo Vermelho-Amarelo distrófico, sob vegetação de Cerrado, a 10 cm de profundidade. Foram utilizados três tratamentos: controle (solo sem adição de composto orgânico) e solo com adição de duas doses de composto orgânico doméstico (10 e 20 g kg-1 de solo seco). Os tratamentos foram avaliados quanto ao C liberado (C-CO2), à atividade respiratória microbiana aos 15 dias de incubação, ao C da biomassa microbiana (CBM); a glicose total (GT), ao quociente metabólico (qCO2) e à atividade das enzimas β-glicosidase e fosfatases ácida e alcalina. Verificou-se que as duas doses de adição de composto orgânico doméstico resultaram em aumento significativo da atividade microbiana e, também, em incremento nas frações de C orgânico e no estoque de C do solo, caracterizados pela elevação nos teores de CBM e GT. Entretanto, as duas doses de composto causaram decréscimo no qCO2, o que pode indicar maior diversidade na comunidade microbiana após o acréscimo do composto e maior eficiência no uso da energia. As enzimas β-glicosidase e as fosfatases (ácida e alcalina) foram fortemente influenciadas pela adição de composto orgânico doméstico, sobretudo pela dose de 20 g kg-1 do produto. A atividade da enzima β-glicosidase aumentou significativamente em resposta à adição do composto, e a atividade da fosfatase ácida foi superior à da fosfatase alcalina. A relação β-glicosidase: CBM foi maior no controle do que nos tratamentos com adição de composto, o que sugere que no controle havia mais enzimas do que no substrato ou que houve grande incremento no CBM com a adição do composto ao solo.
Termos de indexação: Cerrado, respiração do solo, biomassa microbiana, glicose, fosfatase, β-glicosidase.
INTRODUCTION
Microbial activity is of great importance for biological and biochemical soil processes because it directly influences the transformation of nutrients and organic compost. It is also qualitatively and quantitatively associated with the presence of extracellular hydrolytic enzymes which are important in the process of decomposition and mineralization of organic matter (Kiss et al., 1975; Nakas et al., 1987; Martens et al., 1992; Ross & Cairns, 1992; Elliott et al., 1993).
The most important general indicators of soil microbial activity are microbial biomass C and soil respiration, while specific indicators are related to the activity of extracellular hydrolytic enzymes such as phosphatase and β-glucosidase, involved in nutrient cycling (Gil-Sotres et al., 2005). The evaluation of biological and biochemical soil properties and β-glucosidase activity has been suggested because of their relationship to the soil C cycle and the sensitivity of these indicators to detect changes resulting from agricultural management practices (Nannipieri et al., 1990; Dick & Tabatabai, 1993; Gil-Sotres et al., 2005; Lagomarsino et al., 2009). The activity of phosphomonoesterase enzymes, such as acid and alkaline phosphatases, has been widely studied because of its importance in organic P mineralization, releasing orthophosphates that are readily assimilated by plants and soil microorganisms (Sylvia et al., 1999).
Microbial communities in the soil are enhanced and stimulated by the addition of organic waste, especially due to the presence of readily available nutrients and C compounds. In general, organic waste has high levels of macronutrients such as N, P, K, Ca (Aita et al., 2007; Giacomini et al., 2009), and micronutrients such as B, Zn and Mn. Since the application of organic waste can change biological and biochemical indicators, studies are needed to measure the effect of this practice on soil (Martens, 2000; Ros et al., 2003; Tejada et al., 2006).
Although several studies have shown that compost can improve soil by promoting appropriate biological activity and improving nutrient availability and soil structure (soil particle aggregation) (Pascual et al., 1999a; Ros et al., 2003; Crecchio et al., 2004), few studies show the effect of household compost application on soil microbial and enzymatic activity.
The objective of this study was to evaluate microbial and enzymatic (phosphatases and β-glucosidase) activity in Cerrado soil samples after applying organic compost from household residues.
METHODS AND MATERIALS
The compost used in this study was produced by aerobic composting of solid organic household residues; i.e. food residues (fruit and vegetable stalks and peels, egg shells, ground coffee) and yard waste from trees, shrubs and grass, generated and collected from households in Uberlândia, MG. The waste was placed in a composter with holes for air circulation and slurry drainage during composting. Approximately 50 % of the composter volume was filled with residues. The composting material was turned and the temperature measured daily. After 120 days, the compost temperatures had dropped from approximately 50-60 °C to about 30-35 °C, indicating the end of the biostabilization phase (Teixeira et al., 2004). A sample of the compost was taken for chemical analysis and a moisture content of 60 % was detected. It was stated that all levels (C, total nitrogen, C/N ratio and pH) (Table 1) except moisture (Table 2) were within the limits set by Brasil (2009). According to Brasil, this type of organic fertilizer is classified as "C" because this product of domestic residues is safe for agricultural use (Brasil, 2009).
In the study, samples of a Typic Acrustox (Embrapa, 2006) were used, which were taken from an area of Cerrado vegetation on the farm Floresta do Lobo, in Uberlândia, state of Minas Gerais. Soil samples were collected from a depth of 0 to 10 cm during a rainy period in December 2008. Each sample consisted of four subsamples mixed to form a composite sample.
The composite soil sample was sieved through a 3.35 mm mesh and samples of 200 g soil were air-dried, crushed in a mortar and tested to determine the chemical properties (Table 1) according to Tedesco et al. (1995). Total organic C was determined by the method described by Yeomans & Bremmer (1989) (Table 1). To determine soil respiratory activity and total glucose, bioassays were performed with undisturbed soil on the sampling day. The remaining soil was stored at 4 ºC for the enzymatic activity experiments.
There were three treatments: a control (SCN, soil without organic compost amendment) and soil with two application rates of domestic organic compost (10 and 20 g kg-1 of dry soil) which corresponded to 10 and 20 Mg ha-1 respectively. Soils of all treatments were analyzed to determine the respiratory activity of soil microorganisms, microbial biomass C, total glucose, metabolic quotient and β-glucosidase and phosphatase enzyme activity.
The microbial respiratory activity in the soil was estimated from the amount of C-CO2 released in 15 days of incubation, as proposed by Stotzky (1965). For this purpose, the two compost doses were mixed with 40 g of SCN and placed in airtight glass jars (500 mL) for incubation. Then, 40 mL plastic cups containing 5 mL of NaOH (1 mol L-1) were placed in each jar containing soil (control) or soil + compost (other treatments) to capture CO2 released by microbial respiration after 2, 7 and 15 days. The jars were sealed and maintained at room temperature (average of 23 ºC). After each incubation period, 2.5 mL of BaCl2.2H2O (1 mol L-1) and 3 drops of phenolphthalein indicator were added to the NaOH in the plastic cups and titrated with HCl (0.25 mol L-1). New cups with new NaOH solutions were placed in the jars for each subsequent incubation period. The amount of C released was estimated in mg kg-1 of C-CO2 in dry soil.
The microbial biomass C (MBC) after compost amendment and 15 days of incubation was determined by fumigation-extraction as described by Vance et al. (1987), using 20 g of SCN in a non-fumigated sample and fumigated with chloroform for 24 h (25 ºC). Carbon was extracted with 40 mL of K2SO4 (0.5 mol L-1), and MBC calculated by the equation: CBM = Cext x 2.66 (Vance et al., 1987), where Cext is the difference between the C extracted from the fumigated and non-fumigated samples. The metabolic quotient (qCO2) was calculated as the ratio of basal respiration to C from the microbial biomass (Anderson & Domsch, 1990). Total Glucose (TG) was determined according to the procedure described by Ferreira et al. (2008).
β-glucosidase and phosphatase (acid and alkaline) activity was analyzed by methods proposed by Eivazi & Tabatabai (1988) and Tabatabai & Bremner (1969), respectively. Synthetic substrates were added to 1 g of soil: ρ-nitrophenyl-β-D-glucopyranoside for β-glucosidase and ρ-nitrophenyl phosphate for phosphatase using adjusted pH buffers in each assessment (acid phosphatase: pH 4.0 and phosphatase alkaline: pH 9.0) and incubated at 37 ºC for 1 h. The reaction of β-glucosidase activity was stopped with Tris pH 12.0 (0.02 mol L-1). For phosphatases, the reaction was stopped with 0.5 mol L-1 CaCl2: and 0.5 mol L-1 NaOH . Substrate cleavage produced ρ-nitrophenol glucoside (β-glucosidase) and ρ-nitrophenol phosphate (phosphatase) which were identified by spectrophotometry at absorbance values of 464 and 505 nm, respectively.
The experiment consisted of four replications with confidence intervals of 5 %.
RESULTS AND DISCUSSION
Compost application had a positive effect on C mineralization and influenced accumulated soil respiration after 15 days of incubation. The highest application rate of compost (20 g kg-1) resulted in significant increases in microbial activity (Figure 1). For the compost-amended treatments, daily C-CO2 release rates were 1.7 and 3.0 times greater than of the control. The microbial activity was probably increased because of the contribution of organic C and nutrients available to soil microorganisms (Emmerling et al., 2000). Other studies reported significant increases in microbial activity resulting from organic compost amendments (Pascual et al., 1999b; Crecchio et al., 2001). These short-term results observed in these studies were due to an increase in both the specific activity of the microbial community and possible effects of a change in microbial community structure. However, the possible increase in community size only partially explains the increase in respiratory activity (Saison et al., 2006).
Microbial biomass C (MBC) reflects the size of the microbial community (Nannipieri et al., 1990). Compared to the control, MBC was 2.14 times higher for the treatments with the low compost dose and 3.88 times higher for the treatments with the high dose (Table 3) which induced basal respiration increases, as commonly found after adding organic matter to the soil (Leita et al., 1995; Sastre et al., 1996; Garcia-Gil et al., 2000; Albiach et al., 2000). Saison et al. (2006) also found that when compost is mixed with soil, microbial biomass increases rapidly for 10 days and reaches a maximum after 22 days.
It was also observed that the total glucose (TG) stock increased with compost application (Table 3). TG increased from 27.9 µg kg-1 of C in the control to 39.5 and 59.6 µg kg-1 of C with the addition of 10 and 20 g kg-1 of compost to the soil, respectively. These increases of 41.6 and 113.6 % may be related to the great increase in soil microbial activity, especially in the treatment with the highest compost dose.
The results indicate that compost application increased TG levels, which suggests that sugar increases could be related to greater soil microbial activity. Moreover, changes in microbial activity may be subject to nutrient input from compost application (Table 1). However, the reason for the changes in microbial activity in this experiment was probably that the organic compost represents a source of C and energy. Reports from Dilly & Munch (1998), Dilly (2001) and Cleveland et al. (2007) showed that the increased response in soil microbial activity was associated with readily available C sources in the soil.
The metabolic quotient (qCO2) value was greatest in the control (Table 3). Compost application resulted in a decrease in qCO2, which could indicate greater microbial community diversity or greater energy use efficiency (Maeder et al., 2002). The qCO2 has been used as a stress indicator and interpreted as microbial efficiency, which is a measure of the energy required to maintain the metabolic activity in relation to the energy required to synthesize biomass (Bardgett & Saggar, 1994). This is visible in the respiration data which ranged from 8.6 in the control to 25.6 mg kg-1 day-1 of C-CO2 in soil with the highest compost dose, and MBC ranging from 111.8 in the control to 433.5 mg kg-1 of C in soil (Table 3). Differences in the effect of compost application rates measured by qCO2 showed that this property is an appropriate measure of soil microbial change.
Biochemical properties detected by enzymatic activity were used to indicate microbial activity in soil amended with compost (Garcia et al., 1994). The β-glucosidase and phosphatase (acid and alkaline) were strongly influenced by compost amendment, and enzyme activity was highest after the application of the 20 g kg-1 compost dose (Figures 2 and 3). Acid phosphatase activity plays a key role in organic P mineralization and, in general, this enzyme is activated when P availability is low (Nannipieri et al., 1979). Phosphatase acid (pH 4.0) activity was higher than that of alkaline (pH 9.0) in all treatments. This result may be related to the low soil pH (Eivazi & Tabatabai, 1977) (Table 1). In general, compost-treated soils had higher phosphatase activity than the control, suggesting greater quantities of available substrates in these soils. This is consistent with many studies about increases in phosphatase activity resulting from organic matter amendments (Jordan et al., 1995, Kremer & Li, 2003). β-glucosidase activity increased significantly in response to the application of organic compost (Figure 3). Increases in β-glucosidase activity after compost application consisting of municipal solid residues were also reported by Marcote et al., 2001 and Ros et al. (2006), indicating the effect of the substrate.
An increased enzyme activity is also an indicator of microbial activity related to the cycling of chemical elements, and significant increases after compost amendment, compared to the control (Figures 2 and 3) may be due to increased microbial biomass, which may have produced these enzymes (Liang et al., 2005; Tejada et al., 2006; Bastida et al., 2008).
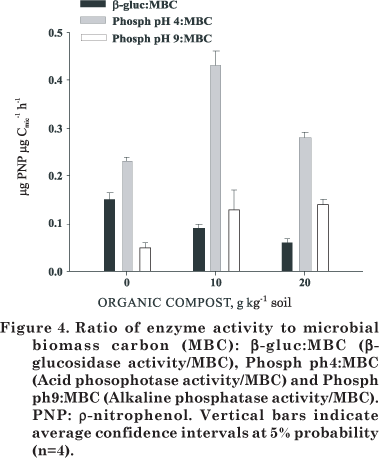
The ratio of enzymatic activity to MBC indicates the enzyme production per unit of microbial biomass (Landi et al., 2000). The β-glucosidase activity to MBC ratio was higher for the control and lower for the treatments with compost (Figure 4), as also verified by Bastida et al. (2008). These results indicate that enzyme activity decreased with the application of organic residues, suggesting that the substrate inhibited the activity of this enzyme. The detection of the β-glucosidase enzyme is related to cellulose decomposition which is synthesized by fungi, bacteria and other soil organisms. However, compost is a stable organic waste (compared to uncomposted residues or sewer sludge) which provides C compounds that are more resistant (Pascual et al., 1997) and more slowly hydrolyzed by enzymes.
The ratio of phosphatase (pH 4.0 and 9.0) to MBC was lower in the control than in the treatments (Figure 4). These changes in enzyme activity of compost-amended soil compared to the control must be mainly associated with the chemical characteristics of the organic material rather than the organic material from the microbial community. In this sense, Saison et al. (2006) showed that compost microbes competed with soil microbes for nutrients. This may happen because of modifications in the microbial community structure caused by compost application. Numerous studies have shown that changes occur in both microbial communities and that soil microbial activity is indirectly attributable to changes in the decomposition of soil organic matter (Innerebner et al., 2006; Saison et al., 2006).
The use of organic matter to restore soils with low organic C content has been very successful. Because compost provides high levels of organic C and nutrients, it effectively improves soil microbial activity, resulting in better soil quality.
CONCLUSIONS
Cerrado vegetation soil (Typic Acrustox) amended with organic compost at (10 and 20 g kg-1 of compost in the soil) resulted in increased microbial respiration, microbial biomass C, and β-glucosidase and phosphatase activity in the soil. There was also a decrease in qCO2, which could indicate greater energy use efficiency by the microbial community.
LITERATURE CITED
Received for publication in February 2009 and approved in February 2010.
- AITA, C.; GIACOMINI, S.J. & HUBNER, A.P. Nitrificação do nitrogênio amoniacal de dejetos líquidos de suínos em solo sob sistema de plantio direto. Pesq. Agropec. Bras., 42:95-102, 2007.
- ALBIACH, R.; CANET, R.; POMARES, F. & INGELMO, F. Microbial biomass content and enzymatic activities after the application of organic amendments to a horticultural soil. Biores. Technol., 75:43-48, 2000.
- ANDERSON, T.H. & DOMSCH, K.H. Application of eco-physiological quotients (qCO2 and Dq) on microbial biomasses from soils of different cropping histories. Soil Biol. Biochem., 22:251-255, 1990.
- BASTIDA, F.; KANDELER, E.; MORENO, J.L.; ROS, M.; GARCÍA, C. & HERNÁNDEZ, T. Application of fresh and composted organic wastes modifies structure, size and activity of soil microbial community under semiarid climate. Applied Soil Ecol., 40:318-329, 2008.
- BARDGETT, G.D. & SAGGAR, S. Effects of heavy metal contamination on the short-term decomposition of labeled (14C) glucose in a pasture soil. Soil Biol. Biochem., 26:727-733, 1994.
- BRASIL - Ministério da Agricultura, Pecuária e Abastecimento. Instrução Normativa SDA nº 25, de 23 de julho de 2009. Anexos I e III. Disponível em: <http://extranet.agricultura.gov.br/sislegis-consulta consultarLegislacao.do?operacao= visualizar&id=20542>Acesso em: 17 ago de 2009.
- CLEVELAND, C.C.; NEMERGUT, D.R.; SCHMIDT, S.K. & TOWNSEND, A.R. Increases in soil respiration following labile carbon additions linked to rapid shifts in soil microbial community composition. Biogeochemistry, 82:229-240, 2007.
- CRECCHIO, C.; CURCI, M.; PIZZIGALLO, M.D.R.; RICCIUTI, P. & RUGGIERO, P. Effect of municipal solid waste compost amendments on soil enzyme activities and bacterial genetic diversity. Soil Biol. Biochem., 36:1595-1605, 2004.
- CRECCHIO, C.; CURCI, M.; RICCIUTI, P. & RUGGIERO, P. Short-term effects of municipal solid waste compost amendments on soil carbon and nitrogen content, some enzyme activities and genetic diversity. Biol. Fert. Soil, 34:311-318, 2001.
- DICK, R.P. Soil enzyme assays as indicators of soil quality. In: DORAN, J.W.; COLEMAN, D.C.; BEZDICEK, D.F. & STEWART, B.A., eds. Defining soil quality for a sustainable environment. Soil Sci. Soc. Am. Special Publication, Madison, 1994. p.107-124.
- DICK, W.A. & TABATABAI, M.A. Significance and potential uses of soil enzymes. In: METTING, F.B. Soil microbial ecology: Application in agricultural and environmental management. New York, Marcel Dekker, 1993. p.95-125.
- DILLY, O. & MUNCH, J.C. Ratios between estimates of microbial biomass content and microbial activity in soils. Biol. Fert. Soils, 27:374-379, 1998.
- DILLY, O. Metabolic and anabolic responses of four arable and forest soils to nutrient addition. Z. Pflanzenernähr Bodenkd., 164:29-34, 2001.
- EIVAZI, F. & TABATABAI, M.A. Phosphatases in soil. Soil Biol. Biochem., 9:167-172, 1977.
- EIVAZI, F. & TABATABAI, M.A. Glucosidases and galactosidases in soils. Soil Biol. Biochem., 20:601-606, 1988.
- ELLIOTT, E.T.; CAMBARDELLA, C.A. & COLE, C.V. Modification of ecosystem processes by management and the mediation of soil organic matter dynamics. Plant Soil, 22:129-138, 1993.
- EMPRESA BRASILEIRA DE PESQUISA AGROPECUÁRIA - EMBRAPA. . Sistema brasileiro de classificação de solos. Brasília, Embrapa Produção de informação. Rio de Janeiro, Embrapa Solos, 2006. 306p.
- FERREIRA, A.S.; OLIVEIRA, R.S.; SANTOS, M.A. & BORGES, E.N. Atividade respiratória da microbiota e conteúdo de glicose em resposta à adição de fósforo em solo de cerrado. R. Bras. Ci. Solo, 32:1891-1897, 2008.
- GARCÍA, C.; HERNÁNDEZ, T. & COSTA, F. Microbial activity in soils under Mediterranean environmental conditions. Soil Biol. Biochem., 26:1185-1191, 1994.
- GARCIA-GIL, J.C.; PLAZA, C.; SOLER-ROVIRA, P. & POLO, A. Long-term effects of municipal solid waste compost application on soil enzyme activities and microbial biomass. Soil Biol. Biochem., 32:1907-1913, 2000.
- GIACOMINI, S.J.; AITA, C. & MIOLA, E.C.C. Mineralização do carbono da palha de aveia e dejetos de suínos aplicados na superficie ou incorporados ao solo. R. Bras. Ci. Solo, 32:2661-2668, 2009.
- GIL-SOTRES, F.; TRASAR-CEPEDA, C.; LEIROS, M.C. & SEOANE, S. Different approaches to evaluating soil quality using biochemical properties. Soil Biol. Biochem., 37:877-887, 2005.
- INNEREBNER, G.; KNAPP, B.; VASARA, T.; ROMANTSCHUK, M. & INSAM, H. Traceability of ammonia-oxidizing bacteria in compost-treated soils. Soil Biol. Biochem., 38:1092-1100, 2006.
- JORDAN, D.; KREMER, R.J.; BERGFIELD, W.A.; KIM, K.Y. & CACNIO, V.N. Evaluation of microbial methods as potential indicators of soil quality in historical agricultural fields. Biol. Fert. Soils, 19:297-302, 1995.
- KISS, S.; DRAGAN-BULARDA, M. & RADULESCU, D. Biological significance of enzymes accumulated in soil. Adv. Agron., 27:25-87, 1975.
- KREMER R.J. & LI, J. Developing weed-suppressive soils through improved soil quality management. Soil Till. Res., 72:193-202, 2003.
- LAGOMARSINO, A.; MOSCATELLI, M.C.; DI TIZIO, A.; MANCINELLI, R.; GREGO, S. & MARINARI, S. Soil biochemical indicators as a tool to assess the short-term impact of agricultural management on changes in organic C in a Mediterranean environment. Ecol. Appl., 9:518-527, 2009.
- LANDI, L.; RENELLA, G.; MORENO, J.L.; FALCHINI, L. & NANNIPIERI, P. Influence of cadmium on the metabolic quotient, L-:D-glutamic acid respiration and enzyme activity:microbial biomass ratio under laboratory conditions. Biol. Fert. Soils, 32:8-16, 2000.
- LEITA, L.; DENOBILI, M.; MONDINI, C.; MUHLBACHOURA, G.; MARCHINOL, L.; BRAGATO, G. & CONTIN, M. Influence of inorganic and organic fertilization on soil microbial biomass, metabolic quotient and heavy metal bioavailability. Biol. Fert. Soil, 28:371-376, 1995.
- LIANG, Y.C.; SI, J.; NIKOLIC, M.; PENG, Y.; CHEN, W. & JIANG, Y. Organic manure stimulates biological activity and barley growth in soil subject to secondary salinization. Soil Biol. Biochem., 37:1185-1195, 2005.
- MAEDER, P.; FLIESSBACH, A.; DUBOIS, D.; GUNST, L.; FRIED, P. & NIGGLI, U. Soil fertility and biodiversity in organic farming. Science, 296:1694-1697, 2002.
- MARCOTE, I.; HERNANDEZ, T.; GARCIA, C. & POLO, A. Influence of one or two successive annual applications of organic fertilisers on the enzyme activity of a soil under barley cultivation. Biores. Technol., 79:147-154, 2001.
- MARTENS, D.A. Plant residue biochemistry regulates soil carbon cycling and carbon sequestration. Soil Biol. Biochem., 32:361-369, 2000.
- MARTENS, D.A.; JOHANSON, J.B. & FRANKENBERGER JR, W.T. Production and persistence of soil enzyme with repeated addition of organic residues. Soil Sci., 153:53-61, 1992.
- NAKAS, J.P.; GOULD, W.D. & KLEIN, D.A. Origin and expression of phosphatase activity in semiarid grassland. Soil Biol. Biochem., 19:13-18, 1987.
- NANNIPIERI, P.; PEDRAZZINI, F.; ARCARA, P.G. & PIOVANELLI, C. Changes in amino acids, enzyme activities, and biomass during soil microbial growth. Soil Sci., 127:26-34, 1979.
- NANNIPIERI, P.; GREGO, S. & CECCANTI, B. Ecological significance of the biological activity in soils. In: BOLLAG, J.M. & STOTZKY, G. New York, Marcel Dekker, 1990. p.293-355.
- PASCUAL, J.A.; GARCIA, C. & HERNÁNDEZ, M.T. Comparison of fresh and composted organic waste in their efficacy for the improvement of arid soil quality. Biores. Technol., 68:255-264, 1999b.
- PASCUAL, J.A.; GARCIA, C. & HERNÁNDEZ, T. Lasting microbiological and biochemical effects of the addition of municipal solid waste to an arid soil. Biol. Fert. Soils, 30:1-6, 1999a.
- PASCUAL, J.A.; GARCIA, C.; HERNÁNDEZ, T. & AYUSO, M. Changes in the microbial activity of an arid soil amended with urban organic wastes. Biol. Fert. Soils, 24:429-434, 1997.
- ROS, M.; PASCUAL, J.A.; GARCÍA, C.; HERNÁNDEZ, M.T. & INSAM, H. Hydrolase activities, microbial biomass and bacterial community in a soil alter long-term amendment with different composts. Soil Biol. Biochem., 38:3443-3452, 2006.
- ROS, M.; HERNÁNDEZ, M.T. & GARCÍA, C. Soil microbial activity after restoration of a semiarid soil by organic amendments. Soil Biol. Biochem., 35:463-469, 2003.
- ROSS, D.J. & CAIRNS, A. Effects of earthworms and ryegrass on respiratory and enzyme activities of soil. Soil Biol. Biochem., 14:583-587, 1982.
- SAISON, C.; DEGRANGE, V.; OLIVER, R.; MILLARD, P.; COMMEAUX, C.; MONTANGE, D. & LE ROUX, X. Alteration and resilience of the soil microbial community following compost amendment: Effects of compost level and compost-borne microbial community. Environ. Microbiol., 8:247-257, 2006.
- SARKAR, J.; LEONOWICZ, A. & BOLLAG, J.M. Immobilization of enzymes on clays and soils. Soil. Biol. Biochem., 21:223-230, 1989.
- SASTRE, L.; VICENTE, M.A. & LOBO, M.C. Influence of the application of sewage sludges on soil microbial activity. Biores. Technol., 57:19-23, 1996.
- SYLVIA, D.M.; FUHRMANN, J.J.; HARTEL, P.G. & ZUBERER, D.A. Principles and applications of soil microbiology. New Jersey, Prentice Hall, 1999. 550p.
- STOTZKY, G. Microbial respiration. In: BLACK, C.A., ed. Methods of soil analysis. Madison, American Society of Agronomy, 1965. Part 2. p.1550-1570.
- TEDESCO, M.J.; BOHNEM, H.; GIANELLO, C.; BISSANI, C.A. & VOLKWEISS, S.J. Análise de solo, plantas e outros materiais. 2.ed. Porto Alegre, Universidade Federal do Rio Grande do Sul, 1995. 174p. (Boletim Técnico, 5)
- TEIXEIRA, L.B.; GERMANO, V.L.C.; OLIVEIRA, R.F. & FURLAN JÚNIOR, J. Processo de compostagem, a partir de lixo orgânico urbano, em leira estática com ventilação natural. Belém, Embrapa, 2004. 8p. (Circular Técnica, 33)
- TEJADA, M.; HERNÁNDEZ, M.T. & GARCÍA, C. Application of two organic amendments on soil restoration: Effects on the soil biological properties. Environ. Qual., 35:1010-1017, 2006.
- VANCE, E.D.; BROOKES, P.C. & JENKINSON, D.S. An extraction method for measuring soil microbial biomass-C. Soil Biol. Biochem., 19:703-707, 1987.
- YEOMANS, J. & BREMNER, J.M. A rapid and precise method for routine determination of organic carbon in soil. Comm. Soil. Sci. Plant Anal., 19:1467-1476, 1989.
Microbial and enzymatic activity in soil after organic composting
Publication Dates
-
Publication in this collection
02 Aug 2010 -
Date of issue
June 2010
History
-
Accepted
Feb 2010 -
Received
Feb 2009