Abstracts
Biochar (carbonized biomass for agricultural use) has been used worldwide as soil amendment and is a technology of particular interest for Brazil, since its "inspiration" is from the historical Terra Preta de Índios(Amazon Dark Earth), and also because Brazil is the world's largest charcoal producer, generating enormous residue quantities in form of fine charcoal and due to the availability of different residual biomasses, mainly from agroindustry (e.g., sugar-cane bagasse; wood and paper-mill wastes; residues from biofuel industries; sewage sludge etc), that can be used for biochar production, making Brazil a key actor in the international scenario in terms of biochar research and utilization). In the last decade, numerous studies on biochar have been carried out and now a vast literature, and excellent reviews, are available. The objective of this paper is therefore to deliver a critical review with some highlights on biochar research, rather than an exhaustive bibliographic review. To this end, some key points considered critical and relevant were selected and the pertinent literature "condensed", with a view to guide future research, rather than analyze trends of the past.
soil amendments; carbon sequestration; greenhouse gases
O biocarvão (biomassa carbonizada para uso agrícola) tem sido usado como condicionador do solo em todo o mundo, e essa tecnologia é de especial interesse para o Brasil, uma vez que tanto a "inspiração", que veio das Terras Pretas de Índios da Amazônia, como o fato de o Brasil ser o maior produtor mundial de carvão vegetal, com a geração de importante quantidade de resíduos na forma de finos de carvão e diversas biomassas residuais, principalmente da agroindústria, como bagaço de cana, resíduos das indústrias de madeira, papel e celulose, biocombustíveis, lodo de esgoto etc. Na última década, diversos estudos com biocarvão têm sido realizados e atualmente uma vasta literatura e excelentes revisões estão disponíveis. Objetivou-se aqui não fazer uma revisão bibliográfica exaustiva, mas sim uma revisão crítica para apontar alguns destaques na pesquisa sobre biochar. Para isso, foram selecionados alguns temas-chave considerados críticos e relevantes e fez-se um "condensado" da literatura pertinente, mais para orientar as pesquisas e tendências do que um mero olhar para o passado.
condicionadores de solos; sequestro de carbono; gases do efeito estufa
INTRODUCTION
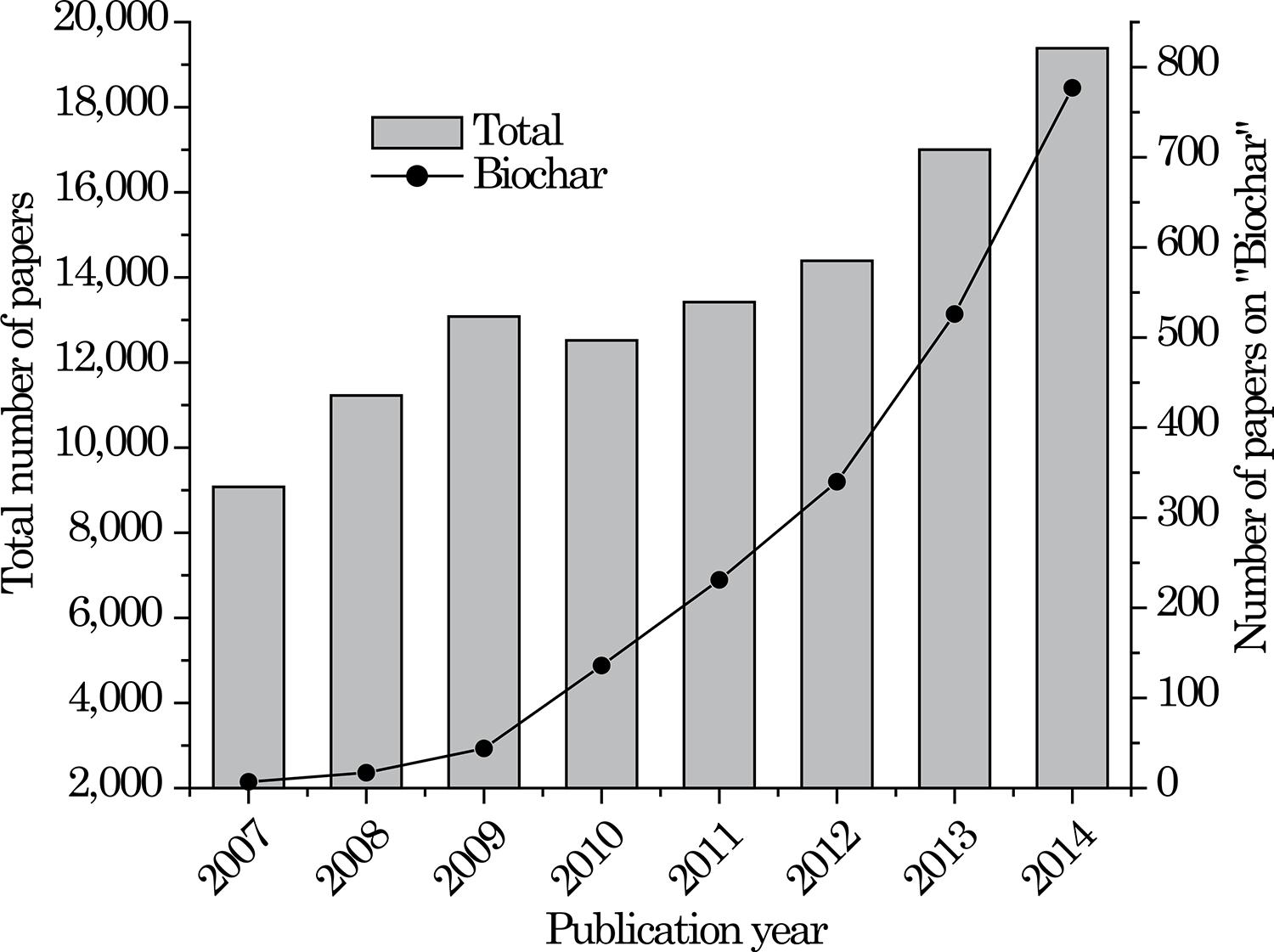
In recent years, the interest in pyrogenic carbon (C) for agricultural use (biochar) has sharply increased (Figure 1). The focus of this growing interest recently shifted from C sequestration and climate change mitigation to: soil fertility improvement and crop growth; water retention and movement in the soil; and soil pollution control, as indicated as a new tendency in a recent article in the journal Nature (Cernansky, 2015Cernansky R. Agriculture: State-of-the-art soil. Nature. 2015;517:258-60.). It is however noteworthy that this discourse change, fromTerra Preta de Índios and climate change to biochar technology, mainly towards "soil fertility improvement" and "improving crop residue management", had already been preconized by the Brazilian Research Network of Biochar since 2006 (Maia et al., 2011Maia CMBF, Madari BE, Novotny EH. Advances in biochar research in Brazil. Dyn Soil Dyn Plant. 2011;5:53-8.; Madari et al., 2012Madari BE, Maia CMBF, Novotny EH. Context and importance of biochar research. Pesq Agropec Bras. 2012;47:i-ii.; Novotny et al., 2012Novotny EH, Auccaise R, Velloso MHR, Corrêa JC, Higarashi MM, Abreu VMN, Rocha JD, Kwapinski W. Characterization of phosphate structures in biochar from swine bones. Pesq Agropec Bras. 2012;47:672-6.; Paiva et al., 2012Paiva DM, Cantarutti RB, Guimarães GGF, Silva IR. Urea coated with oxidized charcoal reduces ammonia volatilization. R Bras Ci Solo. 2012;36:1221-9.; Rittl et al., 2015aRittl TF, Arts B, Kuyper TW. Biochar: an emerging policy arrangement in Brazil? Environ Sci Policy. 2015a;51:45-55.).
Number of scientific articles per year for the queries: "black carbon/black C or pyrogenic carbon/pyrogenic C or charcoal" = Total (columns and left axis) and for the query "biochar" (line and symbol, right axis). Search on April 10, 2015.
However, the term biochar has been used indiscriminately, even in situations where classic and well-defined terms, such as char(coal) (Figure 1), would be more appropriate (see review of Qian et al., 2015Qian K, Kumar A, Zhang H, Bellmer D, Huhnke R. Recent advances in utilization of biochar. Renew Sustain Energy Rev. 2015;42:1055-64.) in which, in our opinion, the term is used erroneously). The term biochar was coined in 2005 by Peter Read, a research fellow at Massey University in New Zealand, to describe finely divided pyrolysed biomass prepared specifically for soil improvement (Read, 2009Read P. This gift of nature is the best way to save us from climate catastrophe. The Guardian. 2009 Mar 27; Commentis free/biochar., emphasis added). When solid pyrogenic C "is produced by thermal decomposition of biomass under limited or absent oxygen (O) and used as soil amendment to increase fertility or sequester atmospheric CO2, it is named "biochar" (Mukherjee et al., 2011Mukherjee A, Zimmerman AR, Harris W. Surface chemistry variations among a series of laboratory-produced biochars. Geoderma. 2011;163:247-55., emphasis added). Distinct from char in general, biochar "is considered to comprise biomass-derived char intended specifically for application to soil, that is, according to its purpose" (Sohi et al., 2010Sohi SP, Krull E, Lopez-Capel E, Bol R. A review of biochar and its use and function in soil. In: Sparks DL, editor. Advances in agronomy. Waltham: Academic Press; 2010. v.105, p.47-82., emphasis added).
In addition to these misuses, concerning the objectives of biochar, of terms dissenting from the definition, the expression "biochar in Terra Preta de Índios (Amazonian Dark Earth)" presupposes that the pre-Columbian Native Americans treated and improved soil intentionally, which is definitely not a consensus in the literature (Novotny et al., 2007Novotny EH, Azevedo ER, Bonagamba TJ, Cunha TJF, Madari BE, Benites VM, Hayes MHB. Studies of the compositions of humic acids from Amazonian Dark Earth soils. Environ Sci Technol. 2007;41:400-5.). The use of the term biochar for the pyrogenic C found in theTerra Preta de Índios must therefore be completely avoided, to prevent the common mistake of assuming that these soils resulted from intentional treatments. The same is true for pyrogenic C in other soils where the intentionality of soil improvement is not confirmed, e.g.: Chernozems (Schmidt et al., 1999Schmidt MWI, Skjemstad JO, Gehrt E, Kögel-Knabner I. Charred organic carbon in German chernozemic soils. Eur J Soil Sci. 1999;50:351-65.; Ponomarenko and Anderson, 2001Ponomarenko EV, Anderson DW. Importance of charred organic matter in Black Chernozem soils of Saskatchewan. Can J Soil Sci. 2001;81:285-97.), Mollisols (Glaser and Amelung, 2003Glaser B, Amelung W. Pyrogenic carbon in native grassland soils along a climosequence in North America. Global Biogeochem Cycle. 2003;17:1064.) and soil charcoal derived from wildfire or anthropic fires (Knicker, 2011Knicker H. Pyrogenic organic matter in soil: Its origin and occurrence, its chemistry and survival in soil environments. Quatern Intern. 2011;243:251-63.;Vleminckx et al., 2014Vleminckx J, Morin-Rivat J, Biwolé AB, Daïnou K, Gillet J-F, Doucet J-L, Drouet T, Hardy OJ. Soil charcoal to assess the impacts of past human disturbances on tropical forests. PLoSONE. 2014;9:108-21.).
As it seems unnecessary and even useless to substitute a new name (biochar) by a well-established term, i.e. charcoal; and striving for scientific rigor and coherence with the definition, in this text, biochar is understood exclusively as pyrolyzed biomass, prepared specifically for the use as soil amendment, i.e. for the purposeful soil application of pyrogenic C, to increase fertility and alleviate anthropic greenhouse gas emissions (e.g. sequester CO2 or decrease N2O emissions).
Biochar production
Biochar is a C-rich product distinct from charcoal and similar materials in that biochar is produced for the purpose of soil application (IBI, 2013International Biochar Initiative - IBI. Frequently asked questions about Biochar International Biochar Initiative [internet]. 2013 [accessed in Mar 13 2014]. Available at: http://www.biochar-international.org/biochar/faqs.
http://www.biochar-international.org/bio...
) as a means to improve its quality; to prevent nutrient leaching; to improve C storage; or to depurate the soil from pollutants. The traditional way to produce biochar is by thermal decomposition of biomass at an oxygen-depleted atmosphere in a wide range of temperatures (300 to 1,000 °C, Zhang et al., 2015a), a process generally known as pyrolysis.
An important aspect of biochar production is the close association to energy production. The choice of energy from biomass is usually due to an environmental need to dispose wastes or to reduce C dioxide emissions from a given system. Therefore, biochar production can be seen as a threefold strategy of integrated environmental benefits, for being associated to clean-energy production, involving waste recycling and allowing soil C storage in the proper biochar.
Chemical composition of feedstock
A wide range of organic materials are suitable as feedstock for thermal processing: from agricultural and wood biomass to any available agricultural, and industrial wastes (bark, husks, straw, seeds, peels, bagasse, nutshells, sawdust, wood shavings, animal beds etc) or municipal wastes.
Plant biomass, henceforth biomass, is defined here as the photosynthetic product resulting from CO2, water and sun energy, whereas animal biomass results from the plant biomass consumed by animals. About 90 % of the biomass consists of C and O, with a typical average elementary composition of ~50 wt.% C and ~40 wt.% O. Other important elements are hydrogen (H, ~5 wt.%) and nitrogen (N, ~1 wt.%) and trace elements such as sulfur and chlorine.
Biomass is composed mainly of cellulose, hemicelluloses and lignin polymers (Zhang, 2010Zhang, Y. Hydrothermal liquefaction to convert biomass into crude oil. In: Blaschek HP, editor. Biofuels from Agriculture Wastes Byproduct. Oxford: Blackwell Publishing; 2010. p.201-32.; Sullivan and Ball, 2012Sullivan AL, Ball R. Thermal decomposition and combustion chemistry of cellulosic biomass. Atmos Environ. 2012;47:133-41.). Other components are minerals, proteins, starches, nucleic acids, oils, and resins (Sullivan and Ball, 2012Sullivan AL, Ball R. Thermal decomposition and combustion chemistry of cellulosic biomass. Atmos Environ. 2012;47:133-41.). Cellulose is the major constituent of most plant-derived biomass, but lignin is also important in woody biomass. In animal biomass, lipids and proteins are relevant constituents (Zhang, 2010Zhang, Y. Hydrothermal liquefaction to convert biomass into crude oil. In: Blaschek HP, editor. Biofuels from Agriculture Wastes Byproduct. Oxford: Blackwell Publishing; 2010. p.201-32.) besides bones (Novotny et al., 2012Novotny EH, Auccaise R, Velloso MHR, Corrêa JC, Higarashi MM, Abreu VMN, Rocha JD, Kwapinski W. Characterization of phosphate structures in biochar from swine bones. Pesq Agropec Bras. 2012;47:672-6.).
Cellulose is a very stable non-branched polysaccharide formed by D-glucose units linked by β-1,4 bonds. The typical amount of cellulose in biomass ranges from 40 to 60 wt% (Zhang et al., 2010a). Closely associated to cellulose are the hemicelluloses, which are heterogeneous polysaccharides structured mainly of hexoses and pentoses, arranged in shorter and branched chains (Zhang, 2010Zhang, Y. Hydrothermal liquefaction to convert biomass into crude oil. In: Blaschek HP, editor. Biofuels from Agriculture Wastes Byproduct. Oxford: Blackwell Publishing; 2010. p.201-32.). Hemicelluloses are less stable than cellulose and form 20 to 40 wt% of the biomass (Zhang et al., 2010aZhang J, Liu J, Liu R. Effects of pyrolysis temperature and heating time on biochar obtained from the pyrolysis of straw and lignosulfonate. Biores Technol. 2015a;176:288-91.). Lignin is a structurally more complex biopolymer than polysaccharides and very resistant to thermal degradation. It is a polyphenol compound formed by apparently random-organized phenyl-propane units, linked by strong covalent bonds (alkyl-aryl ether and C-C bonds). Lignin is the second most abundant component of biomass and accounts for 18 to 40 wt% of the biomass (Tuomela et al., 2000Tuomela M, Vikman M, Hatakka A, It M. Biodegradation of lignin in a compost environment: a review. Biores Technol. 2000;72:169-83.; Amen-Chen et al., 2001Amen-Chen C, Pakdel H, Roy C. Production of monomeric phenols by thermochemical conversion of biomass: A review. Biores Technol. 2001;79:277-99.). Lignin is mainly derived from three p-hydroxycinnamyl alcohols: p-coumaryl, coniferyl and sinapyl. The proportion of these aromatic units in the macromolecular structure depends on the morphological parts and the botanical group of a plant (Thevenot et al., 2010Thevenot M, Dignac M-F, Rumpel C. Fate of lignins in soils: A review. Soil Biol Biochem. 2010;42:1200-11.). Therefore, different species produce their specific types of lignin, where gymnosperms and angiosperms represent distinct groups: the guaiacyl type is found predominantly in softwoods and guaiacyl-syringyl in many hardwoods (Mohan et al., 2006Mohan D, Pittman CU, Steele PH. Pyrolysis of Wood/Biomass for Bio-oil : A critical review. Energy Fuels. 2006;20:848-89.).
For biochar production, it is decisive to know the chemical composition of biomass (cellulose or holocellulose - the total polysaccharide fraction, lignin, ash and extractives) since the thermal degradation dynamics depends on it. Lignin-richer biomass is considered to produce better charcoal (higher calorific value) and Lee et al. (2013)Lee Y, Park J, Ryu C, Gang KS, Yang W, Park YK, Jung J, Hyun S. Comparison of biochar properties from biomass residues produced by slow pyrolysis at 500 °C. Biores Technol. 2013;148:196-201. showed that the higher lignin content, the higher is the biochar yield. Gani and Naruse (2007)Gani A, Naruse I. Effect of cellulose and lignin content on pyrolysis and combustion characteristics for several types of biomass. Renew Energy. 2007;32:649-61. stated that biomass with higher cellulose content pyrolyzes faster than biomass with higher lignin content.
Temperature controls biomass thermo-decomposition
According to Bridgwater (2003)Bridgwater AV. Renewable fuels and chemicals by thermal processing of biomass. Chem Eng J. 2003;91:87-102., biomass can be converted into a more useful energy form through a thermal process and hence, with the strong current trend of declining investment in fossil fuels, thermal conversion of biomass to clean energy has never been studied more thoroughly. Despite the wide variability of biomass compositions, reactors, and experimental conditions and methods, there is a general trend for the distribution of pyrolysis products and its properties as a function of the pyrolysis temperature (Neves et al., 2011Neves D, Thunman H, Matos A, Tarelho L, Gómez-Barea A. Characterization and prediction of biomass pyrolysis products. Prog Energy Combust Sci. 2011;37:611-30.). During pyrolysis, heating is transferred to the biomass particles, increasing temperature locally. The first step of pyrolysis is water evaporation (drying stage) and then, pyrolytic volatiles are progressively released (primary pyrolysis stage) from biomass (Neves et al., 2011Neves D, Thunman H, Matos A, Tarelho L, Gómez-Barea A. Characterization and prediction of biomass pyrolysis products. Prog Energy Combust Sci. 2011;37:611-30.), at temperature-dependent quality and intensity. In a simple explanation, biomass pyrolysis generates a complex combination of products from the individual pyrolysis of cellulose, hemicellulose, lignin and extractives, each with proper kinetic characteristics (Mohan et al., 2006Mohan D, Pittman CU, Steele PH. Pyrolysis of Wood/Biomass for Bio-oil : A critical review. Energy Fuels. 2006;20:848-89.). After the temperature increase, pure hemicellulose decomposes between 200 and 260 °C. During a slow wood pyrolysis, after drying and volatile losses, hemicellulose loss sets on in a range of 130-194 °C, most intensely above 180 °C. However, during a fast pyrolysis, the faster decomposition of hemicellulose than of cellulose is not distinguished since the process occurs in a few seconds at a rapid heating rate (Mohan et al., 2006Mohan D, Pittman CU, Steele PH. Pyrolysis of Wood/Biomass for Bio-oil : A critical review. Energy Fuels. 2006;20:848-89.). Cellulose degradation occurs from 240 to 350 °C and produces anhydro-cellulose and levo-glucosan. Lignin decomposes at 280-500 °C and yields phenols by cleavage of ether and carbon-carbon linkages. The overall primary pyrolysis stage is completed at relatively low temperatures, around 500 °C (Neves et al., 2011Neves D, Thunman H, Matos A, Tarelho L, Gómez-Barea A. Characterization and prediction of biomass pyrolysis products. Prog Energy Combust Sci. 2011;37:611-30.), and yields a C-rich solid: charcoal or biochar, depending on its use. In general, the higher the temperature, the higher will be the pH, C, mineral and ash content, C stability and biochar aromaticity, porosity, and specific surface area, and the lower will be the volatile content and yield (Wu et al., 2012Wu W, Yang M, Feng Q, McGrouther K, Wang H, Lu H, Chen Y. Chemical characterization of rice straw-derived biochar for soil amendment. Biomass Bioenergy. 2012;47:268-76.; Zhao et al., 2013Zhao L, Cao X, Mašek O, Zimmerman A. Heterogeneity of biochar properties as a function of feedstock sources and production temperatures. J Hazard Mater. 2013;256-57:1-9.;Zhang et al., 2015bZhang H, Voroney RP, Price GW. Effects of temperature and processing conditions on biochar chemical properties and their influence on soil C and N transformations. Soil Biol Biochem. 2015b;83:19-28.).
High pyrolysis temperatures also affect sorptive capacity of biochar, similarly to what occurs in the production of activated carbon, i.e.: with increasing temperature the sorptive capacity of coal tends to increase, and consequently its ability to remove organic contaminants from the environment. This feature should be considered carefully when choosing pyrolysis conditions of biochar, since undesirable interactions with certain products (e.g. systemic pesticides) are possible, reducing the effectiveness and requiring dose adjustments, while on the other hand, biochar can be extremely useful to reduce the environmental impact of contact pesticides and for environmental remediation (Spokas et al., 2009Spokas KA, Koskinen WC, Baker JM, Reicosky DC. Impacts of woodchip biochar additions on greenhouse gas production and sorption/degradation of two herbicides in a Minnesota soil. Chemosphere.2009;77:574-81.; Wang et al., 2009Wang H, Lin K, Hou Z, Richardson B, Gan J. Sorption of the herbicide terbuthylazine in two New Zealand forest soils amended with biosolids and biochars. J Soils Sedim. 2009;10:283-9.). Biochar resulting from slow pyrolysis however, can immobilize metals such as Cu2+ and others heavy metals, due its higher carboxyl content (Uchimiya et al., 2010Uchimiya M, Lima IM, Klasson KT, Wartelle LH. Contaminant immobilization and nutrient release by biochar soil amendment: roles of natural organic matter. Chemosphere. 2010;80:935-40.) and the resulting metal complexation capacity.
Thermochemical treatment of biomass
There are many different treatment processes of biomass involving heating. The target product of most of them is energy, but some are used in fine chemistry and biorefineries. At large, all processes involve temperatures above 50 ºC during a variable residence time. These processes can be performed with dry or a wet biomass whose moisture content is crucial to define the best process for a given feedstock. Whatever the process, the main product of a thermal biomass treatment can be liquid (tar, biofuel, various hydrocarbons), solid (charcoal -or biochar- and ash) or gas (H, C oxides, N, light hydrocarbons), but some solid amount is always present that can be considered for use as biochar.
According to the dominant thermochemical reaction (combustion or pyrolysis), thermal processes can be roughly classified in three main groups: pyrolysis, gasification and combustion (Bridgwater, 2003Bridgwater AV. Renewable fuels and chemicals by thermal processing of biomass. Chem Eng J. 2003;91:87-102.). Liquefaction is another term used in the literature to describe processes of bio-oil production through pyrolytic or hydrothermal treatments (Stevens, 1986Stevens DJ. An overview of biomass thermochemical liquefaction research sponsored by the US Department of Energy. In: Proceedings of the 1985 Biomass Thermochemical Conversion Contractors' Meeting; 1985. Washington, DC: United States Department of Environment; 1986. Available at: https://web.anl.gov/PCS/acsfuel/preprintarchive/Files/Merge/Vol-32_2-0007.pdf .
https://web.anl.gov/PCS/acsfuel/preprint...
). An additional group apart from pyrolysis is thermal processing at milder temperatures, e.g., roasting (torrefaction) and hydrothermal treatments. Figure 2 provides an overview of the main thermochemical processes of biomass production, considering the temperature range.
Thermochemical processes of biomass production, according to the temperature range and predominant biochar characteristics (*Bridgwater, 2003Bridgwater AV. Renewable fuels and chemicals by thermal processing of biomass. Chem Eng J. 2003;91:87-102.).
Mild temperature processes
Here we describe the processes that do not involve pyrolysis in thestricto sensu, but are able to significantly transform the biomass composition, for occurring at temperatures as high as 300 ºC or under high pressure and steam. Two important groups of processes can be described in this category: hydrothermal and dry procedures. In the first, conventional or microwave heating is used and the process involves biomass with high moisture content or even water as reaction medium. On the other hand, torrefaction, or roasting, is a process that usually involves dry feedstock.
a) Hydrothermal processes
The water content in the feedstock can be a challenge for thermal processing, since drying may be economically unfeasible. Moisture contents in tropical grasses, for example, can be as high as 80-85 % (Akhtar and Amin, 2011Akhtar J, Amin NAS. A review on process conditions for optimum bio-oil yield in hydrothermal liquefaction of biomass. Renew Sustain Energy Rev. 2011;15:1615-24.). In this case, the best way to treat biomass is to expose it to a hydrothermal treatment. Hydrothermal pyrolysis, hydrothermal carbonization or hydrothermal liquefaction are the terms used to describe the thermal treatment of wet biomass under high pressure (Akhtar and Amin, 2011Akhtar J, Amin NAS. A review on process conditions for optimum bio-oil yield in hydrothermal liquefaction of biomass. Renew Sustain Energy Rev. 2011;15:1615-24.; Libra et al., 2011Libra JA, Ro KS, Kammann C, Funke A, Berge ND, Neubauer Y, Titirici M-M, Fühner C, Bens O, Kern J, Emmerich K-H. Hydrothermal carbonization of biomass residuals: a comparative review of the chemistry, processes and applications of wet and dry pyrolysis. Biofuels. 2011;2:71-106.; Toor et al., 2011Toor SS, Rosendahl L, Rudolf A. Hydrothermal liquefaction of biomass: A review of subcritical water technologies. Energy. 2011;36:2328-42.). Hydrothermal processes occur at temperatures (>100 °C) and pressure conditions under which water becomes a subcritical fluid. As such, water has a completely different behavior, switching from a polar to a relative nonpolar molecule, because the shared electron between oxygen and hydrogen atoms tends to circulate more evenly, reducing the oxygen electronegativity (Zhang, 2010Zhang, Y. Hydrothermal liquefaction to convert biomass into crude oil. In: Blaschek HP, editor. Biofuels from Agriculture Wastes Byproduct. Oxford: Blackwell Publishing; 2010. p.201-32.). Simultaneously, under hydrothermal conditions, water dissociation increases dramatically (Kw ~10-11) as temperature increases, creating a highly favorable environment for hydrolysis (Pinto and Lanças, 2010Pinto JSDS, Lanças FM. Hidrólise do óleo de Azadirachta indica em água subcrítica e determinação da composição dos triacilglicerídeos e ácidos graxos por cromatografia gasosa dealta resolução a alta temperatura e cromatografia gasosa de alta resolução acoplada à espectrometria. Quim Nova. 2010;33:394-7.; Zhang, 2010)Zhang, Y. Hydrothermal liquefaction to convert biomass into crude oil. In: Blaschek HP, editor. Biofuels from Agriculture Wastes Byproduct. Oxford: Blackwell Publishing; 2010. p.201-32..
Hydrothermal processing can be grouped into three different classes:
-
hydrothermal carbonization occurs at temperatures below 247 °C, and the main product is hydrochar;
-
hydrothermal liquefaction is performed at intermediate temperature ranges (between 247 and 374 °C), resulting in a liquid fuel known as biocrude (a petroleum-like oil);
-
in hydrothermal gasification, performed at temperatures above 374 °C, gasification prevails resulting in syn-gas (Elliott et al., 2014Elliott DC, Biller P, Ross AB, Schmidt AJ, Jones SB. Hydrothermal liquefaction of biomass: Developments from batch to continuous process. Biores Technol. 2014;178:147-56.).
Therefore, not only the solid yield but also its chemical characteristics can be very different, according to the temperature of the process, different biochars will result from these hydrothermal processes.
A particular technology of hydrothermal treatment is microwave heating (Guiotoku et al., 2012Guiotoku M, Hansel FA, Novotny EH, Maia CMBDF. Molecular and morphological characterization of hydrochar produced by microwave-assisted hydrothermal carbonization of cellulose. Pesq Agropec Bras. 2012;47:687-92.). Mašek et al. (2013a)Mašek O, Budarin VLV, Gronnow MJ, Crombie KN, Brownsort P, Fitzpatrick E, Hurst PR, Mase O, Budarin VLV, Gronnow MJ, Crombie KN, Brownsort P, Fitzpatrick E, Hurst PR. Microwave and slow pyrolysis biochar - Comparison of physical and functional properties. J Anal Appl Pyrolysis. 2013a;100:41-8., mention as advantages of microwave heating over conventional heating the controllability and energy and cost efficiency of the process, since microwave heating at 200 °C can induce similar or even stronger chemical alterations than slow pyrolysis at 350 °C. They compared slow (SP) and microwave pyrolysis (MW) of straw and woody biomass and found a comparable stability and similar levels of conversion between biochars but significantly lower char yields from MW pyrolysis than SP. These authors stated that the lower C sequestration potential of MW biochar could be compensated by its higher potential for production of renewable energy, for requiring less heat input. Sun et al. (2014)Sun Y, Gao B, Yao Y, Fang J, Zhang M, Zhou Y, Chen H, Yang L. Effects of feedstock type, production method, and pyrolysis temperature on biochar and hydrochar properties. Chem Eng J. 2014;240:574-8. compared hydrochars to dry-pyrolysis biochars derived from the same feedstock, and found hydrochars more acidic and lower in C contents.
b) Torrefaction
Torrefaction is carried out in an inert atmosphere, e.g., nitrogen (N2) gas. Basically, it reduces moisture, removes low weight organic compounds and, from certain temperature, also leads to depolymerization of long polysaccharide chains (Bridgeman et al., 2008Bridgeman TG, Jones JM, Shield I, Williams PT. Torrefaction of reed canary grass, wheat straw and willow to enhance solid fuel qualities and combustion properties. Fuel. 2008;87:844-56.). The result is a biomass with higher energy density and grindability. More recently, torrefaction has been considered as a pre-treatment to boost biomass for energy, since the process improves its bulk and moisture properties, easing aspects as transport and storage. Torrefaction is also a traditional food technology used for nuts or coffee grains. As for biochar, both food or energy torrefaction wastes are common material that could be used as such.
Pyrolysis
Pyrolysis can be described as the direct thermal decomposition of an organic matrix that yields solid, liquid and gas products (Canabarro et al., 2013Canabarro N, Soares JF, Anchieta CG, Kelling CS, Mazutti MA. Thermochemical processes for biofuels production from biomass. Sustain Chem Proc. 2013;1:1-10.). Yield of products resulting from biomass pyrolysis can be maximized as follows: charcoal (a low-temperature, low heating rate process, long residence times), liquid products (low or moderate temperature, high heating rate, short gas residence time), and fuel gas (a high-temperature, low heating rate process, long gas residence time) (Bridgwater, 2012Bridgwater AV. Review of fast pyrolysis of biomass and product upgrading. Biomass Bioenergy. 2012;38:68-94.; Canabarro et al., 2013Canabarro N, Soares JF, Anchieta CG, Kelling CS, Mazutti MA. Thermochemical processes for biofuels production from biomass. Sustain Chem Proc. 2013;1:1-10.). This facility in combining temperature and residence times makes pyrolysis a very versatile process, which allowed the development of many different technological possibilities. Overall, pyrolysis processes differ among each other in how fast heat is transferred to feedstock particles, the maximum temperature and residence time. They are usually classified according to these reaction conditions and product yield (Libra et al., 2011Libra JA, Ro KS, Kammann C, Funke A, Berge ND, Neubauer Y, Titirici M-M, Fühner C, Bens O, Kern J, Emmerich K-H. Hydrothermal carbonization of biomass residuals: a comparative review of the chemistry, processes and applications of wet and dry pyrolysis. Biofuels. 2011;2:71-106.). The main pyrolysis processes are described below.
a) Slow pyrolysis
This method is the most traditional form of charcoal production (carbonization) all over the world and the main process used in Brazil, the largest world charcoal producer. It is probably the most common way to produce biochar at slow heating rates, relatively low temperature (from 300 to 600 °C) and long residence time (hours to days). Slow pyrolysis is recommended for solids production (20 - 40% of charcoal or biochar), once the operational conditions reduce other fractions such as gas and oil. Typically, one third of each fraction is produced when feedstock is slowly pyrolyzed and the higher the temperature, the lower is the charcoal yield. At low temperatures, lignin is partially degraded and some residual cellulose can still be present. Short residence time can lead to the same result, which is incomplete carbonization and also tar residue impregnates the resulting charcoal. For energy, the quality of slow pyrolysis charcoal is low, but for biochar, this depends on the desired function of the final product. In general, low temperature and residence time lead to a highly functionalized biochar, since the incomplete degradation and insufficient tar draining preserve part of the original structure of carbohydrates and phenols, maintaining a high content of hydroxyl and carboxyl groups. These characteristics are desirable when the main intended function of biochar is to enhance the soil cationic exchange capacity (CEC).
b) Fast pyrolysis
Fast pyrolysis is the main process used for bio-oil production. The process involves faster heating rates and much shorter residence times (seconds) than slow pyrolysis. Under the right conditions, the liquid yield can increase as much as 75 %. A good homogenization of the feedstock (Libra et al., 2011Libra JA, Ro KS, Kammann C, Funke A, Berge ND, Neubauer Y, Titirici M-M, Fühner C, Bens O, Kern J, Emmerich K-H. Hydrothermal carbonization of biomass residuals: a comparative review of the chemistry, processes and applications of wet and dry pyrolysis. Biofuels. 2011;2:71-106.), ground to around 2 mm, and drying to less than 10 % moisture (Bridgwater, 2003Bridgwater AV. Renewable fuels and chemicals by thermal processing of biomass. Chem Eng J. 2003;91:87-102.), are required to reach high yields. In spite of its particular importance for bio-oil, fast pyrolysis produces 10 to 15 % of solids, which can be used as biochar, raw or pelleted charcoal. Since fast pyrolysis is usually performed at high temperatures (above 500 °C), the aromaticity of the resulting biochar is generally higher (Brewer et al., 2009Brewer CE, Schmidt-Rohr K, Satrio JA, Brown RC. Characterization of biochar from fast pyrolysis and gasification systems. Environ Prog Sustain Energy. 2009;28:386-96.; Kim et al., 2012Kim KH; Kim JY, Cho TS, Choi JW. Influence of pyrolysis temperature on physicochemical properties of biochar obtained from the fast pyrolysis of pitch pine (Pinus rigida). Biores Technol. 2012;118:158-62.) and O/C atomic ratio lower than by slow pyrolysis for the same residence time. The O/C ratio is an important indicator of stability and functionality of biochar and can range from 0.2 (highly stable) to 0.6 (highly functional) (Spokas, 2010Spokas KA. Review of the stability of biochar in soils: predictability of O:C molar ratios. Carbon Manage. 2010;1:289-303.).
c) Flash pyrolysis
Flash pyrolysis is performed at an even shorter reaction time (only a few seconds) than fast pyrolysis and so the heating rate is very high. The process requires a special reactor configuration (parallel screw reactor, for example) and a heat conductor (as fluidized bed) must be present to receive the feedstock. The fine particles of feedstock are burned by flash fires. This process is used mainly to produce bio-oil. Temperatures ranging from 400 to 950 °C promote rapid depolymerization and feedstock cracking, resulting in bio-oil with viscosity comparable to diesel oil (Canabarro et al., 2013Canabarro N, Soares JF, Anchieta CG, Kelling CS, Mazutti MA. Thermochemical processes for biofuels production from biomass. Sustain Chem Proc. 2013;1:1-10.). Examples of this process are flash hydro-pyrolysis, which involves hydrogen atmosphere at pressures up to 20 MPa and vacuum flash pyrolysis, which occurs under negative pressure to remove condensable products from the hot reaction zone (Canabarro et al., 2013Canabarro N, Soares JF, Anchieta CG, Kelling CS, Mazutti MA. Thermochemical processes for biofuels production from biomass. Sustain Chem Proc. 2013;1:1-10.).
Gasification
Gasification represents the large group of different technologies that transform biomass into fuel-gas. Kirubakaran et al. (2009)Kirubakaran V, Sivaramakrishnan V, Nalini R, Sekar T, Premalatha M, Subramanian P. A review on gasification of biomass. Renew Sustain Energy Rev. 2009;13:179-86. describe gasification by the three predominant reactions on biomass degradation: drying, devolatilization (pyrolysis) and gasification. The first step of gasification is drying followed by pyrolysis. The process produces gas, vaporized tars and oils and a char residue, consisting of typically 5 to 10 % of the original feedstock mass (Brewer et al., 2009Brewer CE, Schmidt-Rohr K, Satrio JA, Brown RC. Characterization of biochar from fast pyrolysis and gasification systems. Environ Prog Sustain Energy. 2009;28:386-96.).
Gasification can be a partial oxidation or a pyrolytic process (Bridgwater, 2003Bridgwater AV. Renewable fuels and chemicals by thermal processing of biomass. Chem Eng J. 2003;91:87-102.) at high temperatures, typically around 800-900 °C (McKendry, 2002aMcKendry P. Energy production from biomass (part 2): conversion technologies. Biores Technol. 2002a;83:47-54.). In the first case, the oxidizing agent can be pure oxygen or air. At large, the gases produced are hydrogen (H2), carbon monoxide (CO), carbon dioxide (CO2), but nitrogen (N2) and methane (CH4) can also be present (McKendry, 2002aMcKendry P. Energy production from biomass (part 2): conversion technologies. Biores Technol. 2002a;83:47-54.; Bridgwater, 2003Bridgwater AV. Renewable fuels and chemicals by thermal processing of biomass. Chem Eng J. 2003;91:87-102.).
There are basically two gasifier types: the fixed-bed and fluidised-bed, with variations of each (McKendry, 2002bMcKendry P. Energy production from biomass (part 3): Gasification technologies. Biores Technol. 2002b;83:55-63.). The first type consists of the traditional gasification technology and is run at temperatures as high as 1,000 °C. Fixed-bed gasifiers have a simpler design and produce a low calorific value (CV) gas (syngas) with a high tar content (McKendry, 2002bMcKendry P. Energy production from biomass (part 3): Gasification technologies. Biores Technol. 2002b;83:55-63.). Typically, the composition of syngas is 40-50 % N2, 15-20 % H2, 10-15 % CO, 10-15 % CO2 and 3-5 % CH4, with a net CV of 4-6 MJ N-1m-3 (McKendry, 2002bMcKendry P. Energy production from biomass (part 3): Gasification technologies. Biores Technol. 2002b;83:55-63.).
Two groups of gasification, with a different number of steps, can be described: direct biomass gasification and gasification after biomass pyrolysis. The latter group has recently attracted attention as a way to reduce costs (transport and storage) and improve efficiency, since the feedstock consists of high energy-density material (charcoal or bio-oil).
Brewer et al., (2011)Brewer CE, Unger R, Schmidt-Rohr K, Brown RC. Criteria to select biochars for field studies based on biochar chemical properties. BioEnergy Res. 2011;4:312-23. stated that biochar from gasification can produce a suitable material in terms of C stability but recommends paying attention to some concerns, e.g., the ash content. As the process can involve such high temperatures, gasification is used to produce biochars with a high concentration of aromatic structures. Brewer et al. (2009)Brewer CE, Schmidt-Rohr K, Satrio JA, Brown RC. Characterization of biochar from fast pyrolysis and gasification systems. Environ Prog Sustain Energy. 2009;28:386-96., comparing chemical properties of chars produced by different processes, showed that gasification char had similar aromaticity to slow-pyrolysis char, but a larger cluster size of the fused ring systems. According to these authors, aromatic cluster size seems to be more controlled by the reaction temperature than by duration. These features of biochar from gasification enhance the concern with regard to the polycyclic aromatic hydrocarbons (PAH) content, so the presence of aromatic contaminants must be monitored. One last aspect about biochars produced at high temperature, as by gasification, is the specific surface area and porosity, which are usually higher under these conditions than in slow-pyrolysis biochar.
Combustion
When biomass is burnt in an oxygen atmosphere, the main reaction is combustion and the final products will be CO2, water, and ashes. Combustion can reach temperature as high as 800 to 950 °C (Neves et al., 2011Neves D, Thunman H, Matos A, Tarelho L, Gómez-Barea A. Characterization and prediction of biomass pyrolysis products. Prog Energy Combust Sci. 2011;37:611-30.). An inefficient or incomplete combustion process can result in a charcoal-rich material with high ash content. This material, when used as biochar, has to be specifically analyzed for the liming effect, since biomass ashes contain relevant amounts of Ca, Mg, K, and Na oxides. Biochars derived from wood and other lignocellulosic materials usually have smaller acid-neutralizing capacity than biochar from animal wastes (Kookana et al., 2011Kookana RS, Sarmah AK, van Zwieten L, Krull E, Singh B. Biochar application to soil: agronomic and environmental benefits and unintended consequences. Adv Agron. 2011;112:103-43.).
Designer biochar
Enough knowledge is already available about biomass thermo-treatments to direct the pyrolysis conditions to obtain specific properties. Instead of analyzing which biochar results from certain production conditions, it is already possible to define which biochar is desired and then fit the production conditions to obtain it (Mašek and Brownsort, 2010Mašek O, Brownsort P. Research on Production of Bespoke Biochar [internet]; 2010 [accessed in Aug 12 2014]. Available at: http://www.biochar.ac.uk/abstract.php?id=32.
http://www.biochar.ac.uk/abstract.php?id...
; Singh et al., 2010Singh B, Singh BP, Cowie AL. Characterisation and evaluation of biochars for their application as a soil amendment. Aust J Soil Res. 2010;48:516-25.; Kim et al., 2012Kim KH; Kim JY, Cho TS, Choi JW. Influence of pyrolysis temperature on physicochemical properties of biochar obtained from the fast pyrolysis of pitch pine (Pinus rigida). Biores Technol. 2012;118:158-62.; Novak et al., 2014Novak JM, Cantrell KB, Watts DW, Busscher WJ, Johnson MG. Designing relevant biochars as soil amendments using lignocellulosic-based and manure-based feedstocks. J Soils Sedim. 2014;14:330-43.).
Quantification of pyrogenic C
The quantification of pyrogenic C is of paramount importance to evaluate its role in soil C stocks and its fate in the environment, but is a great methodological challenge (Derenne and Largeau, 2001Derenne S, Largeau C. A review of some important families of refractory macromolecules: composition, origin, and fate in soils and sediments. Soil Sci. 2001;166:833-47.; Masiello, 2004Masiello CA. New directions in black carbon organic geochemistry. Mar Chem. 2004;92:201-13.; Simpson and Hatcher, 2004Simpson MJ, Hatcher PG. Overestimates of black carbon in soils and sediments. Naturwissenschaften. 2004;91:436-40.; Novotny et al., 2006Novotny EH, Hayes MHB, Azevedo ER, Bonagamba TJ. Characterisation of black carbon rich samples by 13C solid state nuclear magnetic resonance. Naturwissenshaften. 2006;93:447-50., 2007Novotny EH, Azevedo ER, Bonagamba TJ, Cunha TJF, Madari BE, Benites VM, Hayes MHB. Studies of the compositions of humic acids from Amazonian Dark Earth soils. Environ Sci Technol. 2007;41:400-5.;Hammes et al., 2007Hammes K, Schmidt MWI, Smernik RJ, Currie LA, Ball WP, Nguyen TH, Louchouarn P, Houel S, Elmquist M, Cornelissen G, Skjemstad JO, Masiello CA, Song J, Peng P, Mitra S, Dunn JC, Hatcher PG, Hockaday WC, Smith DM, Hartkopf-Fröder C, Böhmer A, Lüer B, Huebert BJ, Amelung W, Brodowski S, Huang L, Zhang W, Gschwend PM, Flores-Cervantes DX, Largeau C, Rouzaud J-N, Rumpel C, Guggenberger G, Kaiser K, Rodionov A, Gonzalez-Vila FJ, Gonzalez-Perez JA, Rosa JM, Manning DAC, López-Capél E, Ding L. Comparison of quantification methods to measure fire-derived (black/elemental) carbon in soils and sediments using reference materials from soil, water, sediment and the atmosphere. Global Biogeochem Cycle. 2007;21:GB3016.). The reason is that, first of all, the pyrogenic C pool must be considered in a continuum model of carbonization (Figure 3) (Masiello, 2004Masiello CA. New directions in black carbon organic geochemistry. Mar Chem. 2004;92:201-13.), which is very complex and comprises different products, ranging from slightly charred, degradable biomass to highly condensed, refractory soot, with gradual changes in properties and structures, in which the co-existence of several of the continuum products is common.
Model of carbonization continuum and associated key properties and functions. Formation Temp.: peak heat treatment temperature; H/C and O/C: atomic ratios; Org. comp. sorption: sorption capacity for polar and nonpolar compounds; CEC: cation exchange capacity; Soft Lewis acid ads.: adsorption capacity for soft Lewis acids (e.g. some heavy metals such as Hg2+, Hg2+2 , Cd2+); CP/MAS 13C NMR: cross polarization by carbon-13 nuclear magnetic resonance with magic angle sample spinning, DP: direct polarization MAS 13C NMR, the quantification range of DP covers a broader range than CP; BPCA: benzene polycarboxilic acids (carbonization molecular markers) analysis. Source: Adapted from Hedges et al. (2000), Masiello (2004), Hammes et al. (2007), Bird and Ascough (2012). Bottom figure (a and b), source: Keiluweit et al. (2010) Reprinted. Copyright© (2010) American Chemical Society.
According to Masiello (2004)Masiello CA. New directions in black carbon organic geochemistry. Mar Chem. 2004;92:201-13., pyrogenic C measurement techniques could be grouped into six general classes: microscopic; optical; thermal; chemical; spectroscopic; molecular marker; and also combinations of these six techniques. Microscopic measurements are based on the identification and counting of charcoal pieces visible under an optical microscope; optical techniques measure the mass absorption coefficient (light absorption) at a specific visible wavelength (usually 500-650 nm) of atmospheric aerosols collected in filters (Clarke et al., 1987Clarke AD, Noone KJ, Heintzenberg J, Warren SG, Covert DS. Aerosol light absorption measurement techniques: Analysis and intercomparisons. Atmos Environ. 1987;21:1455-65.) or in solid residues after oxidation of non-pyrogenic material by Thermal/Optical Transmittance and Reflectance (TOT/R) (Hammes et al., 2007Hammes K, Schmidt MWI, Smernik RJ, Currie LA, Ball WP, Nguyen TH, Louchouarn P, Houel S, Elmquist M, Cornelissen G, Skjemstad JO, Masiello CA, Song J, Peng P, Mitra S, Dunn JC, Hatcher PG, Hockaday WC, Smith DM, Hartkopf-Fröder C, Böhmer A, Lüer B, Huebert BJ, Amelung W, Brodowski S, Huang L, Zhang W, Gschwend PM, Flores-Cervantes DX, Largeau C, Rouzaud J-N, Rumpel C, Guggenberger G, Kaiser K, Rodionov A, Gonzalez-Vila FJ, Gonzalez-Perez JA, Rosa JM, Manning DAC, López-Capél E, Ding L. Comparison of quantification methods to measure fire-derived (black/elemental) carbon in soils and sediments using reference materials from soil, water, sediment and the atmosphere. Global Biogeochem Cycle. 2007;21:GB3016.); thermal and chemical methods quantify pyrogenic C remaining after oxidation by heating and chemical oxidation, respectively; spectroscopic techniques identify signals of specific functional groups (i.e. polycondensed aromatic rings) associated with carbonized products, usually after oxidative (chemical-; thermal- or photo-oxidation) removal of operationally defined non-pyrogenic material, however the use of multivariate mathematical tools in spectroscopic data, e.g. Multivariate Curve Resolution, can also provide interesting results about C speciation - pyrogenic and non-pyrogenic C (Novotny et al., 2009Novotny EH, Hayes MHB, Madari BE, Bonagamba TJ, Azevedo ER, Souza AA, Song G, Nogueira CM, Mangrich AS. Lessons from the Terra Preta de Índio of the Amazon region for the utilization of charcoal for soil amendment. J Braz Chem Soc. 2009;20:1003-10.) without requiring the removal of non-pyrogenic material. Molecular marker techniques, in turn, quantify specific compounds produced during biomass carbonization, e.g. levoglucosan (Elias et al., 2001Elias VO, Simoneit BRT, Cordeiro RC, Turcq B. Evaluating Levoglucan as an indicator of biomass burning in Carajás, Amazônia: a comparison to the charcoal record. Geochim Cosmochim Acta. 2001;65:267-72.) or benzene polycarboxilic acids (BPCA), generated by chemical oxidation of polycyclic aromatic structures (Glaser et al., 1998Glaser B, Haumaier L, Guggenberger G, Zech W. Black carbon in soils: the use of benzenecarboxylic acids as specific markers. Org Geochem. 1998;29:811-9.). In this way, the pyrogenic C content can be estimated by extrapolation, assuming a constant BPCA/pyrogenic C mass ratio (Glaser et al., 1998Glaser B, Haumaier L, Guggenberger G, Zech W. Black carbon in soils: the use of benzenecarboxylic acids as specific markers. Org Geochem. 1998;29:811-9.).
Other authors (Schmidt and Noack, 2000Schmidt MWI, Noack AG. Black carbon in soils and sediments: Analysis, distribution, implications, and current challenges. Global Biogeochem Cycle. 2000;14:777-94.) suggested the grouping of these analytical approaches in four main categories: thermal, chemical, optical, and indirect evidence (i.e. molecular markers and magnetic susceptibility).
Except for the microscopic and molecular marker techniques, all other methods involve the removal of non-pyrogenic material by chemical, thermal or photo-oxidation and quantification of the remaining C in the residue. However, aside from pyrogenic C, the residue can contain recalcitrant biopolymers, such as acid-insoluble components of plant waxes and lipids resistant to chemical oxidation (Knicker et al., 2008aKnicker H, Wiesmeier M, Dick DP. A simplified method for the quantification of pyrogenic organic matter in grassland soils via chemical oxidation. Geoderma. 2008a;147:69-74.), and therefore, this quantification of residual C alone could lead to an overestimation of pyrogenic C. To overcome this limitation, the characterization of the residues by spectroscopic methods, such as13C Nuclear Magnetic Resonance (Simpson and Hatcher, 2004Simpson MJ, Hatcher PG. Overestimates of black carbon in soils and sediments. Naturwissenschaften. 2004;91:436-40.; Knicker et al., 2008aKnicker H, Wiesmeier M, Dick DP. A simplified method for the quantification of pyrogenic organic matter in grassland soils via chemical oxidation. Geoderma. 2008a;147:69-74.) and infrared spectroscopy (Smith et al., 1975Smith DM, Griffin JJ, Goldberg ED. Spectrometric method for the quantitative determination of elemental carbon. Anal Chem. 1975;47:233-8.) can provide reliable results.
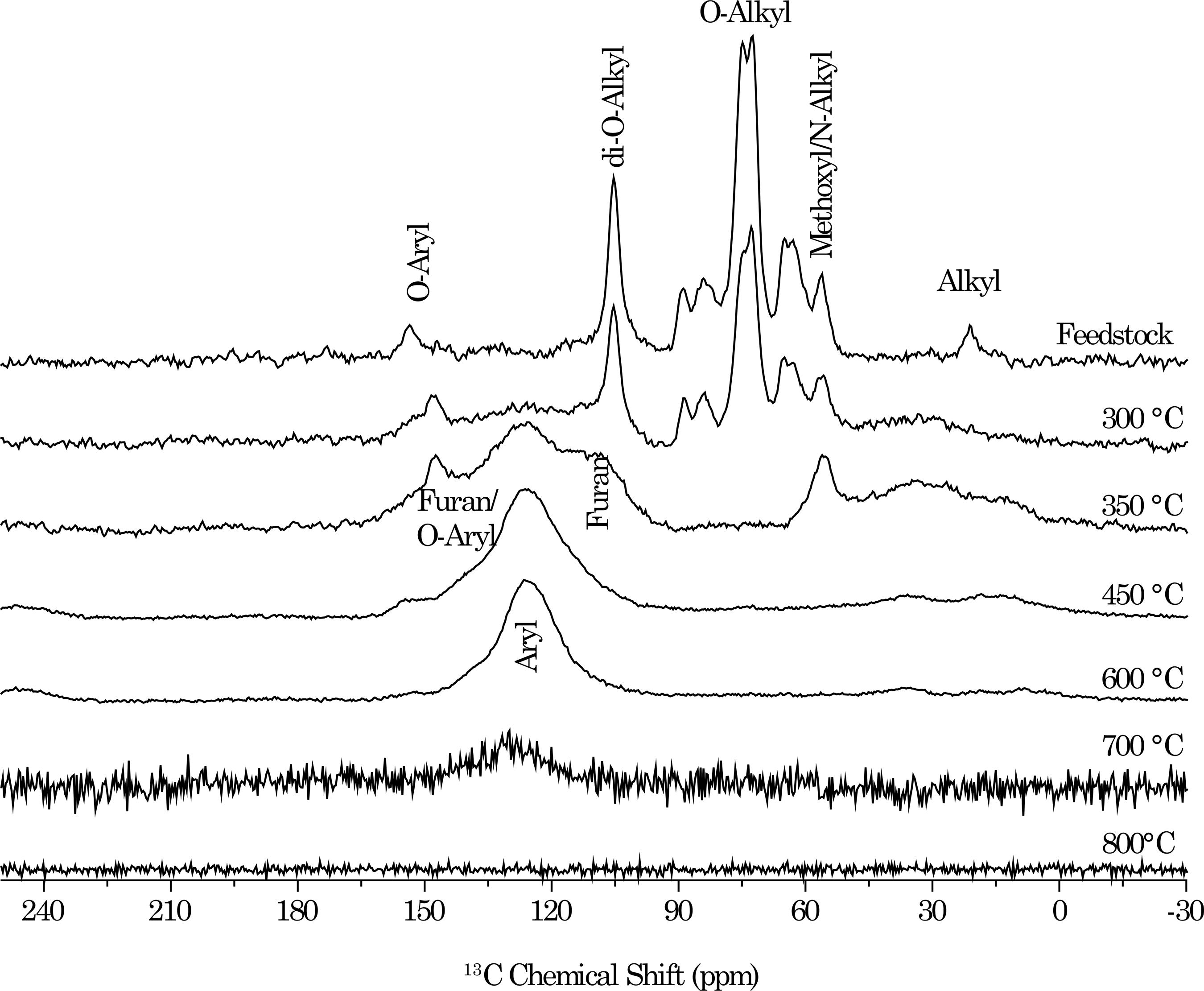
In spite of what was stated in the literature (Masiello, 2004Masiello CA. New directions in black carbon organic geochemistry. Mar Chem. 2004;92:201-13.), even the 13C Nuclear Magnetic Resonance spectroscopy under "quantitative" conditions (i.e. direct polarization), when performed with high magnetic field equipment (>1.76 T), as is usual, underestimates highly condensed aromatic structures (soot and graphitic structures). This is a result of the high heterogeneity in local magnetic susceptibility and/or chemical shift anisotropy, which is not completely averaged out at the usual rates of magic angle sample spinning (Freitas et al., 1999Freitas JCC, Bonagamba TJ, Emmerich FG. 13C high resolution solid-state NMR study of peat carbonization. Energy Fuels. 1999;13:53-9., 2001Freitas JCC, Bonagamba TJ, Emmerich FG. Investigation of biomass- and polymer-based carbon materials using 13C high-resolution solid-state NMR. Carbon. 2001;39:535-45.; Novotny et al., 2006)Novotny EH, Hayes MHB, Azevedo ER, Bonagamba TJ. Characterisation of black carbon rich samples by 13C solid state nuclear magnetic resonance. Naturwissenshaften. 2006;93:447-50.. The underestimation is even worse for the cross-polarization technique (Figure 4), due to the inefficient H-C cross polarization (very low H content).
Cross polarization with magic angle sample spinning 13C nuclear magnetic resonance of Eucalyptus grandis wood feedstock and its biochar at different highest heat temperature treatments (1 h carbonization). Spectra from 600 to 800 °C represent acid-insoluble coffee husk residues (Rambo et al., 2014).
On the other hand, methods of molecular markers are non-stoichiometric for all species of pyrogenic C, since the method of Elias takes only levoglucans into account, or C loss due to the breaking of the polycondensed aromatic structure, which is tentatively offset by a correction factor (Glaser et al., 1998Glaser B, Haumaier L, Guggenberger G, Zech W. Black carbon in soils: the use of benzenecarboxylic acids as specific markers. Org Geochem. 1998;29:811-9.). This correction however assumes that the mass ratio of BPCA/pyrogenic C is constant, which was not properly evaluated for a broad spectrum of charred materials or weathered coal, which is already partially oxidized to aromatic carboxylic acids (Glaser et al., 2002Glaser B, Lehmann J, Zech W. Ameliorating physical and chemical properties of highly weathered soils in the tropics with charcoal - a review. Biol Fertil Soils. 2002;35:219-30.; Kramer et al., 2004Kramer RW, Kujawinski EB, Hatcher PG. Identification of black carbon derived structures in a volcanic ash soil humic acid by fourier transform ion cyclotron resonance mass spectrometry. Environ Sci Technol. 2004;38:3387-95.; Novotny et al., 2007Novotny EH, Azevedo ER, Bonagamba TJ, Cunha TJF, Madari BE, Benites VM, Hayes MHB. Studies of the compositions of humic acids from Amazonian Dark Earth soils. Environ Sci Technol. 2007;41:400-5., 2009Novotny EH, Hayes MHB, Madari BE, Bonagamba TJ, Azevedo ER, Souza AA, Song G, Nogueira CM, Mangrich AS. Lessons from the Terra Preta de Índio of the Amazon region for the utilization of charcoal for soil amendment. J Braz Chem Soc. 2009;20:1003-10.; Maia et al., 2011Maia CMBF, Madari BE, Novotny EH. Advances in biochar research in Brazil. Dyn Soil Dyn Plant. 2011;5:53-8.; Linhares et al., 2012Linhares CR, Lemke J, Auccaise R, Duó DA, Ziolli RL, Kwapinski W, Novotny EH. Reproducing the organic matter model of anthropogenic dark earth of Amazonia and testing the ecotoxicity of functionalized charcoal compounds. Pesq Agropec Bras. 2012;47:693-8.;Araujo et al., 2014Araujo JR, Archanjo BS, Souza KR, Kwapinski W, Falcão NPS, Novotny EH, Achete CA. Selective extraction of humic acids from an anthropogenic Amazonian dark earth and from a chemically oxidized charcoal. Biol Fertil Soils. 2014;50:1223-32.).
Aside from these methods, a very interesting technique, not usually cited in quantification studies and reviews (Glaser et al., 1998Glaser B, Haumaier L, Guggenberger G, Zech W. Black carbon in soils: the use of benzenecarboxylic acids as specific markers. Org Geochem. 1998;29:811-9.; Schmidt et al., 2001Schmidt MWI, Skjemstad JO, Czimczik CI, Glaser B, Prentice KM, Gelinas Y, Kuhlbusch TAJ. Comparative analysis of black carbon in soils. Global Biogeochem Cycle. 2001;15:163-7.; Masiello, 2004Masiello CA. New directions in black carbon organic geochemistry. Mar Chem. 2004;92:201-13.; Simpson and Hatcher, 2004Simpson MJ, Hatcher PG. Overestimates of black carbon in soils and sediments. Naturwissenschaften. 2004;91:436-40.; Brodowski et al., 2005Brodowski S, Rodionov A, Haumaier L, Glaser B, Amelung W. Revised black carbon assessment using benzene polycarboxylic acids. Org Geochem. 2005;36:1299-310.; Hammes et al., 2007Hammes K, Schmidt MWI, Smernik RJ, Currie LA, Ball WP, Nguyen TH, Louchouarn P, Houel S, Elmquist M, Cornelissen G, Skjemstad JO, Masiello CA, Song J, Peng P, Mitra S, Dunn JC, Hatcher PG, Hockaday WC, Smith DM, Hartkopf-Fröder C, Böhmer A, Lüer B, Huebert BJ, Amelung W, Brodowski S, Huang L, Zhang W, Gschwend PM, Flores-Cervantes DX, Largeau C, Rouzaud J-N, Rumpel C, Guggenberger G, Kaiser K, Rodionov A, Gonzalez-Vila FJ, Gonzalez-Perez JA, Rosa JM, Manning DAC, López-Capél E, Ding L. Comparison of quantification methods to measure fire-derived (black/elemental) carbon in soils and sediments using reference materials from soil, water, sediment and the atmosphere. Global Biogeochem Cycle. 2007;21:GB3016.; Koide et al., 2011Koide RT, Petprakob K, Peoples M. Quantitative analysis of biochar in field soil. Soil Biol Biochem. 2011;43:1563-8.; Wiedemeier et al., 2015Wiedemeier DB, Abiven S, Hockaday WC, Keiluweit M, Kleber M, Masiello CA, McBeath AV, Nico PS, Pyle LA, Schneider MPW, Smernik RJ, Wiesenberg GLB, Schmidt MWI. Aromaticity and degree of aromatic condensation of char. Org Geochem. 2015;78:135-43.), is the C stable isotopic ratio (δ13C) (Bird and Ascough, 2012Bird MI, Ascough PL. Isotopes in pyrogenic carbon: A review. Org Geochem. 2012;42:1529-39.), which is widely used in studies on biochar mineralization and priming (Hamer et al., 2004Hamer U, Marschner B, Brodowski S, Amelung W. Interactive priming of black carbon and glucose mineralisation. Org Geochem. 2004;35:823-30.; Cross and Sohi, 2011Cross A, Sohi SP. The priming potential of biochar products in relation to labile carbon contents and soil organic matter status. Soil Biol Biochem. 2011;43:2127-34.; Keith et al., 2011Keith A, Singh B, Singh BP. Interactive priming of biochar and labile organic matter mineralization in a smectite-rich soil. Environ Sci Technol. 2011;45:9611-8.; Luo et al., 2011Luo Y, Durenkamp M, De Nobili M, Lin Q, Brookes PC. Short term soil priming effects and the mineralisation of biochar following its incorporation to soils of different pH. Soil Biol Biochem. 2011;43:2304-14.; Fang et al., 2015Fang Y, Singh B, Singh BP. Effect of temperature on biochar priming effects and its stability in soils. Soil Biol Biochem. 2015;80:136-45.;Rittl et al., 2015bRittl, TF, Novotny EH, Balieiro FC, Hoffland E, Alves BJR, Kuyper TW. Negative priming on mineralization of native soil organic carbon by oilseed biochars of contrasting quality. Eur J Soil Sci. 2015b(in press).), but still uncommon for pyrogenic C quantification. The application of biochar with isotopically distinct δ13C values of the native soil organic matter (distinct photosynthetic pathway) would provide a powerful tool, aside from tracing the fate of pyrogenic C in the environment, as reference method to study quantification and stability (e.g. biochar half-life). This technique would be particularly interesting if the emitted CO2 is quantified and its source determined by δ13C measurement of the emitted CO2 (Rittl et al., 2015bRittl, TF, Novotny EH, Balieiro FC, Hoffland E, Alves BJR, Kuyper TW. Negative priming on mineralization of native soil organic carbon by oilseed biochars of contrasting quality. Eur J Soil Sci. 2015b(in press).), thus allowing to estimate the real mineralization of the applied biochar, since important losses of pyrogenic C occur via dissolution and transport to rivers and sea (Jaffé et al., 2013Jaffé R, Ding Y, Niggemann J, Vähätalo AV, Stubbins A, Spencer RGM, Campbell J, Dittmar T. Global charcoal mobilization from soils via dissolution and riverine transport to the oceans. Science. 2013;340:345-7.), aside from losses by runoff (Major et al., 2010aMajor J, Lehmann J, Rondon M, Goodale C. Fate of soil-applied black carbon: downward migration, leaching and soil respiration. Global Change Biol. 2010a;16:1366-79.) and bioturbation (Elmer et al., 2015Elmer WH, Lattao CV, Pignatello JJ. Active removal of biochar by earthworms (Lumbricus terrestris). Pedobiologia. 2015;58:1-6.).
And, last but not least, it is evident that each of these analytical approaches measures a different region of carbonization continuum (Figure 3), which can partly explain the discrepant results (by up to two orders of magnitude) of pyrogenic C content obtained by different methods for the same samples (Schmidt et al., 2001Schmidt MWI, Skjemstad JO, Czimczik CI, Glaser B, Prentice KM, Gelinas Y, Kuhlbusch TAJ. Comparative analysis of black carbon in soils. Global Biogeochem Cycle. 2001;15:163-7.). For example, microscopic counting techniques only take relatively large pieces of C into account, excluding any colloidal C, soot and coal degradation products; on the other hand, the methods that quantify the residue after removal by thermal oxidation, chemical and photochemical oxidation of non-pyrogenic C depend on the oxidation intensity, which is overall lowest in the case of photo-oxidation (Schmidt et al., 2001Schmidt MWI, Skjemstad JO, Czimczik CI, Glaser B, Prentice KM, Gelinas Y, Kuhlbusch TAJ. Comparative analysis of black carbon in soils. Global Biogeochem Cycle. 2001;15:163-7.). A fraction of pyrogenic C, especially at the beginning of the carbonization continuum, may be oxidized and therefore underestimated (Masiello, 2004Masiello CA. New directions in black carbon organic geochemistry. Mar Chem. 2004;92:201-13.; Hammes et al., 2007Hammes K, Schmidt MWI, Smernik RJ, Currie LA, Ball WP, Nguyen TH, Louchouarn P, Houel S, Elmquist M, Cornelissen G, Skjemstad JO, Masiello CA, Song J, Peng P, Mitra S, Dunn JC, Hatcher PG, Hockaday WC, Smith DM, Hartkopf-Fröder C, Böhmer A, Lüer B, Huebert BJ, Amelung W, Brodowski S, Huang L, Zhang W, Gschwend PM, Flores-Cervantes DX, Largeau C, Rouzaud J-N, Rumpel C, Guggenberger G, Kaiser K, Rodionov A, Gonzalez-Vila FJ, Gonzalez-Perez JA, Rosa JM, Manning DAC, López-Capél E, Ding L. Comparison of quantification methods to measure fire-derived (black/elemental) carbon in soils and sediments using reference materials from soil, water, sediment and the atmosphere. Global Biogeochem Cycle. 2007;21:GB3016.; Knicker et al., 2007Knicker H, Müller P, Hilscher A. How useful is chemical oxidation with dichromate for the determination of "Black Carbon" in fire-affected soils? Geoderma. 2007;142:178-96.); while spectroscopic techniques of molecular markers, in turn, focus on the chemical signature of carbonization, and thus fulfill the expectation of detecting the broadest spectrum of carbonization continuum (Masiello, 2004Masiello CA. New directions in black carbon organic geochemistry. Mar Chem. 2004;92:201-13.), although the limitations of these techniques have already been pointed out above. However, Hammes et al. (2007)Hammes K, Schmidt MWI, Smernik RJ, Currie LA, Ball WP, Nguyen TH, Louchouarn P, Houel S, Elmquist M, Cornelissen G, Skjemstad JO, Masiello CA, Song J, Peng P, Mitra S, Dunn JC, Hatcher PG, Hockaday WC, Smith DM, Hartkopf-Fröder C, Böhmer A, Lüer B, Huebert BJ, Amelung W, Brodowski S, Huang L, Zhang W, Gschwend PM, Flores-Cervantes DX, Largeau C, Rouzaud J-N, Rumpel C, Guggenberger G, Kaiser K, Rodionov A, Gonzalez-Vila FJ, Gonzalez-Perez JA, Rosa JM, Manning DAC, López-Capél E, Ding L. Comparison of quantification methods to measure fire-derived (black/elemental) carbon in soils and sediments using reference materials from soil, water, sediment and the atmosphere. Global Biogeochem Cycle. 2007;21:GB3016. can be cited as follows: "The ultimate value of the various BC [Black C, pyrogenic C] quantification methods is not how they compare to one another, but whether they provide useful information for the application for which they are used. BC quantification is not an end in itself, but rather a means to an end".
Characterization of Biochar
Roughly speaking, all analytical techniques employed in studies on environmental organic matter and charcoal can be useful for biochar characterization as well. However, since the objective of this critical review is not an exhaustive bibliographic review but to present some highlights of biochar research, the determination of only two key parameters is detailed here: aromaticity and degree of aromatic condensation.
The basic chemical structure of carbonized biomass consists of the polycondensed aromatic units. The aromaticity and degree of aromatic condensation is governed mainly by the highest heat treatment temperature - HTT (Keiluweit et al., 2010Keiluweit M, Nico PS, Johnson MG, Kleber M. Dynamic molecular structure of plant-derived black carbon (biochar). Environ Sci Technol. 2010;44:1247-53.; Zimmerman, 2010Zimmerman A. Abiotic and microbial oxidation of laboratory-produced black carbon (biochar). Environ Sci Technol. 2010;44:1295-301.; McBeath et al., 2011McBeath A, Smernik R, Plant E. Determination of the aromaticity and the degree of aromatic condensation of a thermosequence of wood charcoal using NMR. Org Geochem. 2011;42:1194-202.), although other pyrolysis parameters, such as: residence time (Knicker et al., 2005Knicker H, Totsche KU, Almendros G, González-Vila FJ. Condensation degree of burnt peat and plant residues and the reliability of solid-state VACP MAS 13C NMR spectra obtained from pyrogenic humic material. Org Geochem. 2005;36:1359-77.; Melligan et al., 2012Melligan F, Dussan K, Auccaise R, Novotny EH, Leahy JJ, Hayes MHB, Kwapinski W. Characterisation of the products from pyrolysis of residues after acid hydrolysis of Miscanthus. Biores Technol. 2012;108:258-63.; Rutherford et al., 2012Rutherford DW, Wershaw RL, Rostad CE, Kelly CN. Effect of formation conditions on biochars: Compositional and structural properties of cellulose, lignin, and pine biochars. Biomass Bioenergy. 2012;46:693-701.); O2 availability (Ascough et al., 2008Ascough PL, Bird MI, Wormald P, Snape CE, Apperley D. Influence of production variables and starting material on charcoal stable isotopic and molecular characteristics. Geochim Cosmochim Acta. 2008;72:6090-102.); and pressure (Melligan et al., 2011Melligan F, Auccaise R, Novotny EH, Leahy JJ, Hayes MHB, Kwapinski W. Pressurised pyrolysis of Miscanthus using a fixed bed reactor. Biores Technol. 2011;102:3466-70.), as well as the precursor biomass (Wiedemeier et al., 2015Wiedemeier DB, Abiven S, Hockaday WC, Keiluweit M, Kleber M, Masiello CA, McBeath AV, Nico PS, Pyle LA, Schneider MPW, Smernik RJ, Wiesenberg GLB, Schmidt MWI. Aromaticity and degree of aromatic condensation of char. Org Geochem. 2015;78:135-43.), can also significantly affect these fundamental chemical properties of biochar. These properties can be assessed by several analytical methods, described and evaluated by Wiedemeier et al. (2015)Wiedemeier DB, Abiven S, Hockaday WC, Keiluweit M, Kleber M, Masiello CA, McBeath AV, Nico PS, Pyle LA, Schneider MPW, Smernik RJ, Wiesenberg GLB, Schmidt MWI. Aromaticity and degree of aromatic condensation of char. Org Geochem. 2015;78:135-43.: chemical (elemental; BPCA; and lipid analyses); spectroscopic (Infrared; NEXAFS; and13C NMR spectroscopies); and physical (helium pycnometry: skeletal density, which is the sample mass divided by its skeletal volume, where skeletal volume is the volume occupied by the solid sample and any pores not accessible to the gas analysis). The only techniques that provide, concomitantly, the aromaticity and degree of aromatic condensation, are BPCA analysis and 13C NMR spectroscopy (Figure 3 in Wiedemeier et al., 2015Wiedemeier DB, Abiven S, Hockaday WC, Keiluweit M, Kleber M, Masiello CA, McBeath AV, Nico PS, Pyle LA, Schneider MPW, Smernik RJ, Wiesenberg GLB, Schmidt MWI. Aromaticity and degree of aromatic condensation of char. Org Geochem. 2015;78:135-43.). For the others, the combination of two techniques is required, for example: elemental analysis (aromaticity) and helium picnometry (degree of aromatic condensation) (Wiedemeier et al., 2015Wiedemeier DB, Abiven S, Hockaday WC, Keiluweit M, Kleber M, Masiello CA, McBeath AV, Nico PS, Pyle LA, Schneider MPW, Smernik RJ, Wiesenberg GLB, Schmidt MWI. Aromaticity and degree of aromatic condensation of char. Org Geochem. 2015;78:135-43.), although both are indirect measurements.
Apart from the extensively studied changes in the chemical structure of biomass by carbonization (Figure 4), major physical changes occur beyond these, and associated to changes in porosity and surface area. In this sense, Keiluweit et al. (2010)Keiluweit M, Nico PS, Johnson MG, Kleber M. Dynamic molecular structure of plant-derived black carbon (biochar). Environ Sci Technol. 2010;44:1247-53.proposed a physical multi-phase model of carbonization continuum, with four char categories (Figures 3a and 3b). In this model, gradual changes occur with increasing carbonization intensity: from transition chars, that partially preserve the crystalline character of the precursor materials (e.g. crystalline/amorphous cellulose); past a region dominated by amorphous chars, where heat-altered biomacromolecules and incipient small clusters of aromatic polycondensates (fewer than six aromatic rings) are randomly mixed (Knicker et al., 2005Knicker H, Totsche KU, Almendros G, González-Vila FJ. Condensation degree of burnt peat and plant residues and the reliability of solid-state VACP MAS 13C NMR spectra obtained from pyrogenic humic material. Org Geochem. 2005;36:1359-77., 2008bKnicker H, Hilscher A, González-Vila FJ, Almendros G. A new conceptual model for the structural properties of char produced during vegetation fires. Org Geochem. 2008b;39:935-9.); to a region in which amorphous and turbostratic chars coexist (composite chars); ending in turbostratic chars, which are dominated by disordered graphitic crystallites, mainly between planes disorders.
This model shows important environmental implications concerning biochar persistence in the environment as well as its performance as sorbent (Keiluweit et al., 2010Keiluweit M, Nico PS, Johnson MG, Kleber M. Dynamic molecular structure of plant-derived black carbon (biochar). Environ Sci Technol. 2010;44:1247-53.).
The multicomponent character of biochar, with a labile fraction that mineralizes as fast as ordinary soil organic matter, is well-documented (Hilscher et al., 2009Hilscher A, Heister K, Siewert C, Knicker H. Mineralisation and structural changes during the initial phase of microbial degradation of pyrogenic plant residues in soil. Org Geochem. 2009;40:332-42.; Smith et al., 2010Smith JL, Collins HP, Bailey VL. The effect of young biochar on soil respiration. Soil Biol Biochem. 2010;42:2345-7.; Bruun et al., 2011Bruun EW, Hauggaard-Nielsen H, Ibrahim N, Egsgaard H, Ambus P, Jensen PA, Dam-Johansen K. Influence of fast pyrolysis temperature on biochar labile fraction and short-term carbon loss in a loamy soil. Biomass Bioenergy. 2011;35:1182-9.;Calvelo Pereira et al., 2011Calvelo Pereira R, Kaal J, Camps Arbestain M, Pardo Lorenzo R, Aitkenhead W, Hedley M, Macías F, Hindmarsh J, Maciá-Agulló JA. Contribution to characterisation of biochar to estimate the labile fraction of carbon. Org Geochem. 2011;42:1331-42.; Zimmerman et al., 2011Zimmerman A, Gao B, Ahn MY. Positive and negative carbon mineralization priming effects among a variety of biochar-amended soils. Soil Biol Biochem. 2011;43:1169-79.; Rittl et al., 2015bRittl, TF, Novotny EH, Balieiro FC, Hoffland E, Alves BJR, Kuyper TW. Negative priming on mineralization of native soil organic carbon by oilseed biochars of contrasting quality. Eur J Soil Sci. 2015b(in press).). This labile fraction is mainly residual cellulose (O-alkyl and di-O-Alkyl groups; see figure 4) from incomplete carbonization (Hilscher et al., 2009Hilscher A, Heister K, Siewert C, Knicker H. Mineralisation and structural changes during the initial phase of microbial degradation of pyrogenic plant residues in soil. Org Geochem. 2009;40:332-42.; Bruun et al., 2011Bruun EW, Hauggaard-Nielsen H, Ibrahim N, Egsgaard H, Ambus P, Jensen PA, Dam-Johansen K. Influence of fast pyrolysis temperature on biochar labile fraction and short-term carbon loss in a loamy soil. Biomass Bioenergy. 2011;35:1182-9.) and alkyl groups (Hilscher et al., 2009Hilscher A, Heister K, Siewert C, Knicker H. Mineralisation and structural changes during the initial phase of microbial degradation of pyrogenic plant residues in soil. Org Geochem. 2009;40:332-42.; Rittl et al., 2015bRittl, TF, Novotny EH, Balieiro FC, Hoffland E, Alves BJR, Kuyper TW. Negative priming on mineralization of native soil organic carbon by oilseed biochars of contrasting quality. Eur J Soil Sci. 2015b(in press).), that correspond to the transition chars in the multi-phase model. However, amorphous and composite chars, which are also part of the aromatic structure, are prone to oxidation in the first days of incubation (Hilscher et al., 2009Hilscher A, Heister K, Siewert C, Knicker H. Mineralisation and structural changes during the initial phase of microbial degradation of pyrogenic plant residues in soil. Org Geochem. 2009;40:332-42.). Finally, some condensates adsorbed to the biochar (amorphous char) can also decompose quickly (Smith et al., 2010Smith JL, Collins HP, Bailey VL. The effect of young biochar on soil respiration. Soil Biol Biochem. 2010;42:2345-7.).
In general, a selective degradation of part of the biochar "blend" is observed: if the carbonization process was mild, with residual plant biomacromolecules such as cellulose and lignin (Figure 4), these structures will be mineralized first; in case of biomass rich in aliphatic structures, such as fatty acids and waxes (Rittl et al., 2015bRittl, TF, Novotny EH, Balieiro FC, Hoffland E, Alves BJR, Kuyper TW. Negative priming on mineralization of native soil organic carbon by oilseed biochars of contrasting quality. Eur J Soil Sci. 2015b(in press).) or peptides (Hilscher et al., 2009Hilscher A, Heister K, Siewert C, Knicker H. Mineralisation and structural changes during the initial phase of microbial degradation of pyrogenic plant residues in soil. Org Geochem. 2009;40:332-42.), and if the carbonization is moderate, with partial thermal decomposition of cellulose and lignin, the microbial community is able to decompose these aliphatic structures as well. In other words, similarly to ordinary soil organic matter, according to the input, the microbial community will first deplete the most labile fraction. If no very labile compounds (e.g. cellulose) are available, the ecosystem will adapt (ecological succession) to consume the available organic material (Maia et al., 2013), which may even ultimately be very recalcitrant C forms.
But in general, despite the fast decomposition of these labile fractions, the net result is C sequestration (Hilscher et al., 2009Hilscher A, Heister K, Siewert C, Knicker H. Mineralisation and structural changes during the initial phase of microbial degradation of pyrogenic plant residues in soil. Org Geochem. 2009;40:332-42.; Smith et al., 2010Smith JL, Collins HP, Bailey VL. The effect of young biochar on soil respiration. Soil Biol Biochem. 2010;42:2345-7.; Bruun et al., 2011Bruun EW, Hauggaard-Nielsen H, Ibrahim N, Egsgaard H, Ambus P, Jensen PA, Dam-Johansen K. Influence of fast pyrolysis temperature on biochar labile fraction and short-term carbon loss in a loamy soil. Biomass Bioenergy. 2011;35:1182-9.; Zimmerman et al., 2011Zimmerman A, Gao B, Ahn MY. Positive and negative carbon mineralization priming effects among a variety of biochar-amended soils. Soil Biol Biochem. 2011;43:1169-79.; Rittl et al., 2015bRittl, TF, Novotny EH, Balieiro FC, Hoffland E, Alves BJR, Kuyper TW. Negative priming on mineralization of native soil organic carbon by oilseed biochars of contrasting quality. Eur J Soil Sci. 2015b(in press).), probably due to the expected long residence time of the turbostratic chars, even when positive priming occurs in the mineralization of native soil organic matter (Jones et al., 2011Jones DL, Murphy DV, Khalid M, Ahmad W, Edwards-Jones G, DeLuca TH. Short-term biochar-induced increase in soil CO2 release is both biotically and abiotically mediated. Soil Biol Biochem. 2011;43:1723-31.). Besides, the interaction between soil type and biochar structure and composition, allows to "design" the biochar (Novak et al., 2014Novak JM, Cantrell KB, Watts DW, Busscher WJ, Johnson MG. Designing relevant biochars as soil amendments using lignocellulosic-based and manure-based feedstocks. J Soils Sedim. 2014;14:330-43.) even for C sequestration aims (Steinbeiss et al., 2009Steinbeiss S, Gleixner G, Antonietti M. Effect of biochar amendment on soil carbon balance and soil microbial activity. Soil Biol Biochem. 2009;41:1301-10.).
Alternatively, the labile fraction of biochar can also be determined by simple and indirect methods such as thermal decomposition (Benites et al., 2005Benites VM, Mendonça ES, Schaefer CEGR, Novotny EH, Reis EL, Ker JC. Properties of black soil humic acids from high altitude rocky complexes in Brazil. Geoderma. 2005;127:104-13.; Calvelo Pereira et al., 2011Calvelo Pereira R, Kaal J, Camps Arbestain M, Pardo Lorenzo R, Aitkenhead W, Hedley M, Macías F, Hindmarsh J, Maciá-Agulló JA. Contribution to characterisation of biochar to estimate the labile fraction of carbon. Org Geochem. 2011;42:1331-42.) and chemical oxidation (Calvelo Pereira et al., 2011Calvelo Pereira R, Kaal J, Camps Arbestain M, Pardo Lorenzo R, Aitkenhead W, Hedley M, Macías F, Hindmarsh J, Maciá-Agulló JA. Contribution to characterisation of biochar to estimate the labile fraction of carbon. Org Geochem. 2011;42:1331-42.) or a combination of both (Mašek et al., 2013bMašek O, Brownsort P, Cross A, Sohi S. Influence of production conditions on the yield and environmental stability of biochar. Fuel. 2013b;103:151-5.).
Concerning sorption, the multi-phase model of Keiluweit and cols fits very well to the results and discussion, for example, of the excellent paper of Chen et al. (2008)Chen B, Zhou D, Zhu L. Transitional adsorption and partition of nonpolar and polar aromatic contaminants by biochars of pine needles with different pyrolytic temperatures. Environ Sci Technol. 2008;42:5137-43.. Similar to humic substances, that have a different sorption (by partition) capacity, correlated with the content of amorphous poly-(methylene) structures, but poorly correlated with polarity and aromaticity indices (Mao et al., 2002Mao JD, Hundal LS, Thompson ML, Schmidt-Rohr K. Correlation of poly(methylene)-rich amorphous aliphatic domains in humic substances with sorption of a nonpolar organic contaminant, phenanthrene. Environ Sci Technol. 2002;36:929-36.), the sorption of polar and nonpolar aromatic contaminants by biochar occurs by different mechanisms (partition and adsorption), each with specific transitions (Chen et al., 2008Chen B, Zhou D, Zhu L. Transitional adsorption and partition of nonpolar and polar aromatic contaminants by biochars of pine needles with different pyrolytic temperatures. Environ Sci Technol. 2008;42:5137-43.).
According to Chen et al. (2008)Chen B, Zhou D, Zhu L. Transitional adsorption and partition of nonpolar and polar aromatic contaminants by biochars of pine needles with different pyrolytic temperatures. Environ Sci Technol. 2008;42:5137-43., the partition mechanism evolves from partition to an amorphous aliphatic domain, in biochar produced at low temperature (up to 300 °C - transition/amorphous chars in the multi-phase model of Keiluweit and cols), to a condensed aromatic core with higher pyrolytic temperature (400-600 °C - composite/turbostratic chars). Simultaneously, the adsorption contribution produces a transition from a surface polarity-selective effect (biochar produced at 200-400 °C - transition/amorphous chars) to a porosity-selective effect (pyrolysis temperature of 400-600 °C - composite/turbostratic chars). These results corroborate with Chun et al. (2004)Chun Y, Sheng G, Chiou CT, Xing B. Compositions and sorptive properties of crop residue-derived chars. Environ Sci Technol. 2004;38:4649-55., who described adsorption on surfaces for highly carbonized chars and concomitant adsorption and partition into residual biomacromolecule phases in low temperature chars.
Again, the importance of mechanistic in addition to empirical studies becomes evident, with a view to develop designed biochars, to address specific functions in the soil (Chen et al., 2008Chen B, Zhou D, Zhu L. Transitional adsorption and partition of nonpolar and polar aromatic contaminants by biochars of pine needles with different pyrolytic temperatures. Environ Sci Technol. 2008;42:5137-43.; Novak et al., 2014Novak JM, Cantrell KB, Watts DW, Busscher WJ, Johnson MG. Designing relevant biochars as soil amendments using lignocellulosic-based and manure-based feedstocks. J Soils Sedim. 2014;14:330-43.).
Standard methods to qualify biochar
Biochar as a soil amendment has to be analyzed as for its agronomic and environmental properties. A series of methods and procedures are available for the physical chemical characterization of biochar. In addition to other methods of particular interest, American and European scientific communities have already established guidelines to standardize methods and allow safe comparisons of biochar samples (EBC 2012European Biochar Certificate - EBC. Guidelines for a sustainable production of biochar. Arbaz. [Switzerland]:European Biochar Foundation (EBC); 2012 [accessed April 2 2015]. Available at: www.european-biochar.org/en/download.
www.european-biochar.org/en/download...
, 2014European Biochar Certificate - EBC. Comparison of European Biochar Certificate Version 4.8 and IBI Biochar Standards Version 2.0; 2014 [accessed April 2 2015]. Available at: http://www.european-biochar.org/biochar/media/doc/IBI-EBC.pdf.
http://www.european-biochar.org/biochar/...
; IBI, 2014International Biochar Initiative - IBI. Standardized product definition and product testing guidelines for biochar that is used in soil [internet]. 2014. Ithaca: [s.n.]. Available at: http://www.biochar-international.org/sites/default/files/IBI_Biochar_Standards_V20_final_2014.pdf .
http://www.biochar-international.org/sit...
). The chemical and physical characterization of biochar required both by IBI and EBC (EBC, 2014European Biochar Certificate - EBC. Comparison of European Biochar Certificate Version 4.8 and IBI Biochar Standards Version 2.0; 2014 [accessed April 2 2015]. Available at: http://www.european-biochar.org/biochar/media/doc/IBI-EBC.pdf.
http://www.european-biochar.org/biochar/...
) includes:
-
Proximate analysis: gravimetric analysis to determine moisture, ash and volatiles;
-
CHNS elemental analysis by dry-combustion: the recommended method to determine pyrogenic C due to its high stability; sulfur is especially important for feedstock where this element is expected to be high, as for example in kraft-paper mill residues. Oxigen is estimated by the difference. With this analysis, aside from total N and C, it is also possible to calculate H/C and O/C ratios, which indicate biochar stability, the latter is required only by EBC;
-
Inorganic carbon: necessary when carbonates are present in considerable amounts. Recommended method is the CO2 evolved determination after HCl attack. Organic C can be then calculated by difference from Total C;
-
Total P, K, Ca and Mg: important to evaluate the potential for save fertilizers, an optional determination for IBI;
-
pH and electro-conductivity: different methods are recommended by IBI (H2O) and EBC (CaCl2);
-
Liming equivalence: if pH is higher than 7, liming effect of biochar must be estimated and considered especially if apply in massive amounts (t ha-1). The parameter is not required by EBC;
-
Bulk density: required only by EBC;
-
Particle size distribution: not required by EBC;
-
Surface area: the recommended method is N2 adsorption (BET method,Brunauer et al., 1938Brunauer S, Emmett PH, Teller E. Adsorption of gases in multimolecular layers. J Am Chem Soc. 1938;60:309-19.); this determination is optional for IBI;
-
Heavy metals determination: Pb, Cd, Cu, Ni, Hg, Zn and Cr are required by both IBI and EBC; IBI also requires other elements as Co, Mo, B, As, Se, Cl and Na;
-
Organic pollutants: polyaromatic hydrocarbons (PAH) and polyaromatic chlorides (PCB), polychlorinated dibenzodioxins (PCDDs) and polychlorinated dibenzofurans (PCDFs) after Soxhlet extraction with toluene;
-
Germination inhibition: not required by EBC.
Apart from these recommended tests, the determination of carboxyl and phenolic acidity is also useful to understand or forecast biochar behavior in the soil and the Boehm method is the most commonly used for this purpose in biochar studies (Fidel et al., 2013Fidel RB, Laird DA, Thompson ML. Evaluation of modified Boehm titration methods for use with biochars. J Environ Qual. 2013;42:1771-8.). However attention should be given to materials with high ash and/or soluble C contents, since the assumption that the Boehm's reactants only interact with organic surface functional groups is violated (Cheng et al., 2006Cheng C-H, Lehmann J, Thies JE, Burton SD, Engelhard M. Oxidation of black carbon by biotic and abiotic processes. Org Geochem. 2006;37:1477-88.). Cationic exchangeable capacity can also be estimated by using the biochar contents of extractable Ca2+, Mg2+, Na+ and K+, after removing the ashes. Ash-rich biochars, either from partial combustion processes or from ash-rich feedstock can be applied, particularly when liming effect is desired, although the ash quality must be analyzed and its base content (mainly of Ca and Mg) well-determined. Some biochars with high ash content sometimes are silica-rich (Si, as in some grass) or Al or Fe-rich (if the feedstock was contaminated by soil or sediments). Ash content of biochar is very variable and depends not only on the feedstock but also on the process temperature.
Earthworm tests are the most common to evaluate the ecotoxicity of biochar and, for this purpose, aging is an important approach. Seemingly, the main impact caused by biochar on earthworms is due to pH, which can change responses according to the biochar application time (Weyers and Spokas, 2011Weyers SL, Spokas KA. Impact of Biochar on earthworm populations: A review. Appl Environ Soil Sci. 2011:ID 541592:1-12.).
Important Environmental and Agronomic Properties of Biochar
In conclusion, figure 3 shows the relationship of several functions and properties of biochar according to the intensity of carbonization, for example:
-
The stability and C sequestration potential depend mainly on: the chemical structure and composition; aromaticity and degree of aromatic condensation; the content of labile, aliphatic compounds and volatile matter etc (references cited above);
-
The nutrient supply and liming potential depend on: the source composition (Atkinson et al., 2010Atkinson CJ, Fitzgerald JD, Hipps NA. Potential mechanisms for achieving agricultural benefits from biochar application to temperate soils: A review. Plant Soil. 2010;337:1-18.) such as P in bone biochar (Novotny et al., 2012Novotny EH, Auccaise R, Velloso MHR, Corrêa JC, Higarashi MM, Abreu VMN, Rocha JD, Kwapinski W. Characterization of phosphate structures in biochar from swine bones. Pesq Agropec Bras. 2012;47:672-6.); and ash content (DeLuca et al., 2009DeLuca TH, MacKenzie MD, Gundale MJ. Biochar effects on soil nutrient transformations. In: Lehmann J, Joseph S, editors. Biochar for environmental management: Science and Technology. London: Earthscan; 2009. p.251-70.; Gwenzi et al., 2015Gwenzi W, Chaukura N, Mukome FND, Machado S, Nyamasoka B. Biochar production and applications in sub-Saharan Africa: Opportunities, constraints, risks and uncertainties. J Environ Manage. 2015;150:250-61.);
-
The CEC depends mainly on: the content and properties of volatile matter for low carbonized biochar (Mukherjee et al., 2011Mukherjee A, Zimmerman AR, Harris W. Surface chemistry variations among a series of laboratory-produced biochars. Geoderma. 2011;163:247-55.), however due to the lability of this fraction, this effect is probably transient; the cooling of biochar with O2 supply can oxidize it, generating carboxylic functionalities (Spokas et al., 2012Spokas KA, Cantrell KB, Novak JM, Archer DW, Ippolito JA, Collins HP, Boateng AA, Lima IM, Lamb MC, McAloon AJ, Lentz RD, Nichols KA. Biochar: A synthesis of its agronomic impact beyond carbon sequestration. J Environ Qual. 2012;41:973-89.); the aging of biochar also generates these functionalities (Cheng et al., 2006Cheng C-H, Lehmann J, Thies JE, Burton SD, Engelhard M. Oxidation of black carbon by biotic and abiotic processes. Org Geochem. 2006;37:1477-88.; Novotny et al., 2007Novotny EH, Azevedo ER, Bonagamba TJ, Cunha TJF, Madari BE, Benites VM, Hayes MHB. Studies of the compositions of humic acids from Amazonian Dark Earth soils. Environ Sci Technol. 2007;41:400-5., 2009Novotny EH, Hayes MHB, Madari BE, Bonagamba TJ, Azevedo ER, Souza AA, Song G, Nogueira CM, Mangrich AS. Lessons from the Terra Preta de Índio of the Amazon region for the utilization of charcoal for soil amendment. J Braz Chem Soc. 2009;20:1003-10.; Heitkötter and Marschner, 2015Heitkötter J, Marschner B. Interactive effects of biochar aging in soils related to feedstock, pyrolysis temperature, and historic charcoal production. Geoderma. 2015;245-246:56-64.). On the other hand, the anionic exchange capacity decreases with aging (Cheng et al., 2008Cheng C-H, Lehmann J, Engelhard MH. Natural oxidation of black carbon in soils: Changes in molecular form and surface charge along a climosequence. Geochim Cosmochim Acta. 2008;72:1598-610.);
-
Sorption of nutrients and heavy metals (Beesley et al., 2011Beesley L, Moreno-Jiménez E, Gomez-Eyles JL, Harris E, Robinson B, Sizmur T. A review of biochars' potential role in the remediation, revegetation and restoration of contaminated soils. Environ Pollut. 2011;159:3269-82.; Kookana et al., 2011Kookana RS, Sarmah AK, van Zwieten L, Krull E, Singh B. Biochar application to soil: agronomic and environmental benefits and unintended consequences. Adv Agron. 2011;112:103-43.; Melo et al., 2013Melo L, Coscione A, Abreu C, Puga A, Camargo O. Influence of pyrolysis temperature on cadmium and zinc sorption capacity of sugar cane straw-derived biochar. BioResources. 2013;8:4992-5004.): the adsorption of hard Lewis acids (e.g. light alkali ions, and some heavy metals such as Cr3+ and Cr6+) occur predominantly via ion exchange, mainly from carboxylic functionalities (see item c), while the adsorption of soft Lewis acids occurs predominantly via cation-π bonding mechanisms (Harvey et al., 2011Harvey OR, Herbert BE, Rhue RD, Kuo LJ. Metal interactions at the biochar-water interface: Energetics and structure-sorption relationships elucidated by flow adsorption microcalorimetry. Environ Sci Technol. 2011;45:5550-6.), π electron density increases with the aromatic condensation degree and consequently with carbonization intensity;
-
Sorption of pesticides and other organic compounds: the Freundlich sorption coefficient (Kf) for biochar can be several orders of magnitude higher than for humic substances (Kleineidam et al., 1999Kleineidam S, Rugner H, Grathwohl P. Impact of grain scale heterogeneity on slow sorption kinetics. Environ Toxicol Chem. 1999;18:1673-8.) and nonlinear isotherms (Freundlich expoent, n >1); and both Freundlich parameters are directly proportional to carbonization intensity (Chen et al., 2008Chen B, Zhou D, Zhu L. Transitional adsorption and partition of nonpolar and polar aromatic contaminants by biochars of pine needles with different pyrolytic temperatures. Environ Sci Technol. 2008;42:5137-43.), the sorption ability is related to the surface area, aromaticity and porosity (Beesley et al., 2011Beesley L, Moreno-Jiménez E, Gomez-Eyles JL, Harris E, Robinson B, Sizmur T. A review of biochars' potential role in the remediation, revegetation and restoration of contaminated soils. Environ Pollut. 2011;159:3269-82.; Kookana et al., 2011)Kookana RS, Sarmah AK, van Zwieten L, Krull E, Singh B. Biochar application to soil: agronomic and environmental benefits and unintended consequences. Adv Agron. 2011;112:103-43.;
-
The water-holding capacity depends mainly on: porosity and surface area, however the pores can be blocked by recondensation of volatile matter at low-temperature biochar (Atkinson et al., 2010Atkinson CJ, Fitzgerald JD, Hipps NA. Potential mechanisms for achieving agricultural benefits from biochar application to temperate soils: A review. Plant Soil. 2010;337:1-18.; Beesley et al., 2011Beesley L, Moreno-Jiménez E, Gomez-Eyles JL, Harris E, Robinson B, Sizmur T. A review of biochars' potential role in the remediation, revegetation and restoration of contaminated soils. Environ Pollut. 2011;159:3269-82.); and biochar hydrophilicity (Rizhiya et al., 2015Rizhiya EYA, Buchkina NP, Mukhina IM, Belinets AS, Balashov EV. Effect of biochar on the properties of loamy sand Spodosol soil samples with different fertility levels: A laboratory experiment. Eurasian Soil Sci. 2015;48:192-200.), which depends on the pyrolysis temperature (Mohan et al., 2014Mohan D, Sarswat A, Ok YS, Pittman Jr CU. Organic and inorganic contaminants removal from water with biochar, a renewable, low cost and sustainable adsorbent - A critical review. Biores Technol. 2014;160:191-202.) and technique, for example: hydrothermal are more hydrophilic than thermal biochars (Sun et al., 2011Sun K, Ro K, Guo M, Novak J, Mashayekhi H, Xing B. Sorption of bisphenol A, 17α-ethinyl estradiol and phenanthrene on thermally and hydrothermally produced biochars. Biores Technol. 2011;102:5757-63.);
-
Biota interactions depend mainly on: presence of hazardous and beneficial compounds (Sun et al., 2015Sun D, Meng J, Liang H, Yang E, Huang Y, Chen W, Jiang L, Lan Y, Zhang W, Gao J. Effect of volatile organic compounds absorbed to fresh biochar on survival of Bacillus mucilaginosus and structure of soil microbial communities. J Soils Sedim. 2015;15:271-81.); pH; sorption properties; porosity; and surface area (Atkinson et al., 2010Atkinson CJ, Fitzgerald JD, Hipps NA. Potential mechanisms for achieving agricultural benefits from biochar application to temperate soils: A review. Plant Soil. 2010;337:1-18.; Beesley et al., 2011Beesley L, Moreno-Jiménez E, Gomez-Eyles JL, Harris E, Robinson B, Sizmur T. A review of biochars' potential role in the remediation, revegetation and restoration of contaminated soils. Environ Pollut. 2011;159:3269-82.; Lehmann et al., 2011Lehmann J, Rillig MC, Thies J, Masiello CA, Hockaday WC, Crowley D. Biochar effects on soil biota - A review. Soil Biol Biochem. 2011;43:1812-36.).
AGRONOMIC POTENTIAL OF BIOCHAR AS A SOIL AMENDMENT IN CROPPING SYSTEMS
Research on biochar and crop production
According to Lehmann and Joseph (2009)Lehmann J, Joseph S. Biochar for environmental management: Science and technology. London: Earthscan; 2009., biochar can be considered a promising material that can represent a response to a wide range of grave challenges as, for example: soil degradation and food insecurity, climate change, sustainable energy generation, and waste management (Figure 5a). In recent years, research on biochar as a soil amendment to improve crop production has increased (Figure 5b). Two developments have sparked particular interest in using biochar. Firstly, the discovery of the Indian Black Earths (Terra Preta de Índio) suggests that one of the reasons for the extraordinary fertility of these soils is the high C content (up to 15 %), mainly in the form of pyrogenic C, as a result of ancient human activity in the Amazon (Madari et al., 2003Madari BE, Benites VM, Cunha TJF. The effect of management on the fertility of Amazonian Dark earth soils. In: Lehmann J, Kern DC, Glaser B, Woods WI, editors. Amazonian Dark Earths: Origin, properties, management. Dordrecht: Kluwer Academic Publishers; 2003. p.407-26.; Novotny et al., 2009Novotny EH, Hayes MHB, Madari BE, Bonagamba TJ, Azevedo ER, Souza AA, Song G, Nogueira CM, Mangrich AS. Lessons from the Terra Preta de Índio of the Amazon region for the utilization of charcoal for soil amendment. J Braz Chem Soc. 2009;20:1003-10.). A key process in the formation of Terra Preta was pyrolysis, which catalysed the transformation of organic material, "locking" the C in the form of charcoal. Secondly, the production of bioenergy via carbonization of organic residues or biomass has opened the possibility of amplifying the use of biochar, a by-product in this process (Lehmann, 2007Lehmann J. A handful of carbon. Nature. 2007;447:143-44.).
The functionality of biochar (a) and number of scientific articles per year for the query [(TITLE-ABS-KEY(biochar and crop production)] (b). Source: Adapted from Lehmann and Joseph (2009) (a); and Scopus, 157 results, search on 18 June 2014 (b).
The first studies on usefulness of biochar in agriculture/forest soils were conducted in Japan, where intensive agriculture has been practiced since ancient times (Ogawa and Okimori, 2010Ogawa M, Okimori Y. Pioneering works in biochar research, Japan. Aust J Soil Res. 2010;48:489-500.). In Europe, there are reports on the use of peat-charcoal for agricultural purposes by Davy (1856)Davy E. On some experiments made with a view to determine the comparative value of peat and peat-charcoal for agriculture purposes. Lect Chem Carm Scholl Med. 1856;11:172-8., and in USA, Tryon (1948)Tryon EH. Effect of charcoal on certain physical, chemical and biological properties of forest soils. Ecol Soc Am. 1948;18:81-115. reported on the effect of wood biochar on the properties of forest soils. Recent studies suggest that application of biochar as a soil amendment can potentially increase crop productivity on weathered tropical soils (Glaser et al., 2002Glaser B, Lehmann J, Zech W. Ameliorating physical and chemical properties of highly weathered soils in the tropics with charcoal - a review. Biol Fertil Soils. 2002;35:219-30.; Lehmann et al., 2003Lehmann J, Silva Jr JP, Steiner C, Nehls T, Zech W, Glaser B. Nutrient availability and leaching in an archaeological Anthrosol and a Ferralsol of the Central Amazon basin: Fertilizer, manure and charcoal amendments. Plant Soil. 2003;249:343-57.; Steiner et al., 2007Steiner C, Teixeira WG, Lehmann J, Nehls T, Macêdo JLV, Blum WEH, Zech W. Long term effects of manure, charcoal and mineral fertilization on crop production and fertility on a highly weathered Central Amazonian upland soil. Plant Soil. 2007;291:275-90.;Major et al., 2010bMajor J, Lehmann J, Rondon M, Goodale C. Fate of soil-applied black carbon: downward migration, leaching and soil respiration. Global Change Biol. 2010a;16:1366-79.; Petter et al., 2012Petter FA, Madari BE, Silva MAS, Carneiro MAC, Carvalho MTM, Marimon Jr, BH, Pacheco LP. Soil fertility and upland rice yield after biochar application in the Cerrado. Pesq Agropec Bras. 2012;47:699-706.). This is in agreement with a meta-regression analysis by Crane-Droesch et al. (2013)Crane-Droesch A, Abiven S, Jeffery S, Torn MS. Heterogeneous global crop yield response to biochar: a meta-regression analysis. Environ Res Lett. 2013;8:1-8., who found that soil CEC and organic C were strong predictors of yield response in the presence of biochar. Soils with a low CEC on crop yields with different types of biochar amendment and low C were associated with positive crop yield response to biochar application. Moreover, a meta-analysis on the impact of biochar on crop production showed a generally positive effect (~ 10 % increment on crop yields with different types of biochar amendment) (Jeffery et al., 2011Jeffery S, Verheijen FGA, van der Velde M, Bastos AC. A quantitative review of the effects of biochar application to soils on crop productivity using meta-analysis. Agric Ecosyst Environ. 2011;144:175-87.; Biederman and Harpole, 2013Biederman LA, Harpole WS. Biochar and its effects on plant productivity and nutrient cycling: a meta-analysis. Global Change Biol Bioenergy. 2013;5:202-14.; Liu et al., 2013Liu X, Zhang A, Ji C, Joseph S, Bian R, Li L, Pan G, Paz-Ferreiro J. Biochar's effect on crop productivity and the dependence on experimental conditions - a meta-analysis of literature data. Plant Soil. 2013;373:583-94.). However, the extrapolation of these results to field applications is problematic because most studies were conducted over short periods and under environmentally controlled conditions. For example, according to the meta-analysis by Liu et al. (2013)Liu X, Zhang A, Ji C, Joseph S, Bian R, Li L, Pan G, Paz-Ferreiro J. Biochar's effect on crop productivity and the dependence on experimental conditions - a meta-analysis of literature data. Plant Soil. 2013;373:583-94., greater responses were found in pot than in field experiments. In addition, Jeffery et al. (2011)Jeffery S, Verheijen FGA, van der Velde M, Bastos AC. A quantitative review of the effects of biochar application to soils on crop productivity using meta-analysis. Agric Ecosyst Environ. 2011;144:175-87. showed that biochar effects on crop yields could vary with crop species. It is not always clear whether positive effects stem from a direct fertilization effect, an increased water-holding capacity, or a combination of effects. It is also uncertain for how long the effects of biochar application are likely to last in a cropping system. Both Jeffery et al. (2011)Jeffery S, Verheijen FGA, van der Velde M, Bastos AC. A quantitative review of the effects of biochar application to soils on crop productivity using meta-analysis. Agric Ecosyst Environ. 2011;144:175-87. and Liu et al. (2013)Liu X, Zhang A, Ji C, Joseph S, Bian R, Li L, Pan G, Paz-Ferreiro J. Biochar's effect on crop productivity and the dependence on experimental conditions - a meta-analysis of literature data. Plant Soil. 2013;373:583-94. found that crop responses to biochar amendment were greater for dry land crops on acid and sandy-textured soils. Research that addresses the potential of biochar as a soil amendment in crop production systems under field conditions from a multiple year perspective is therefore required.
Determining factors and potential effects of biochar on soil, plant and atmosphere relationships
The International Biochar Intiative (IBI, 2013International Biochar Initiative - IBI. Frequently asked questions about Biochar International Biochar Initiative [internet]. 2013 [accessed in Mar 13 2014]. Available at: http://www.biochar-international.org/biochar/faqs.
http://www.biochar-international.org/bio...
) states that biochar can be used as a product for its own sake or as an ingredient in a blended product, with a range of applications: as agent for soil improvement, improved resource use efficiency, remediation and/or protection against particular environmental pollution, and as a strategy for greenhouse gas emission reduction. However, the large range of biochar types, resulting from different feedstock and pyrolysis processes, diversifies the efficacy of biochars as soil amendment. For example, the higher the temperature of pyrolysis, the greater the surface area, pH and capacity to exchange cations and the lower the percentage of C recovery (Lehmann, 2007Lehmann J. A handful of carbon. Nature. 2007;447:143-44.). Biochar types with high ash contents can reduce soil acidity, increase soil pH and concentration of essential elements such as Ca, Mg and K and decrease Al availability (Deenik et al., 2011Deenik JL, Diarra A, Uehara G, Campbell S, Sumiyoshi Y, Antal Jr MJ. Charcoal ash and volatile matter effects on soil properties and plant growth in an acid Ultisol. Soil Sci. 2011;176:336-45.; Deal et al., 2012Deal C, Brewer CE, Brown RC, Okure MAE, Amoding A. Comparison of kiln-derived and gasifier-derived biochars as soil amendment in the humid tropics. Biomass Bioenergy. 2012;37:161-8.) while high-surface-area biochar can improve soil water retention capacity (Gray et al., 2014Gray M, Johnson MG, Dragila MI, Kleber M. Water uptake in biochars: The roles of porosity and hydrophobicity. Biomass Bioenergy. 2014;61:196-205.). Biochar is mainly composed of C. A major part of this C is recalcitrant (also known as pyrogenic C, char, black C), and a smaller part is labile, which can affect the soil C/N ratio. In periodically burned open Savannah, fire-derived organic matter is an important component of the soil organic C (SOC) (Novotny et al., 2009Novotny EH, Hayes MHB, Madari BE, Bonagamba TJ, Azevedo ER, Souza AA, Song G, Nogueira CM, Mangrich AS. Lessons from the Terra Preta de Índio of the Amazon region for the utilization of charcoal for soil amendment. J Braz Chem Soc. 2009;20:1003-10.). Liming and fertilization effects promoted by ash can persist for at least 2 years in the Brazilian Savannah also known as Cerrado (Pivello et al., 2010Pivello VR, Oliveras I, Miranda HS, Haridasan M, Sato MN, Meirelles ST. Effect of fires on soil nutrient availability in an open savannah in Central Brazil. Plant Soil. 2010;337:111-23.). For instance, in the topsoil of a 23-year-old pasture of the Brazilian Savannah, Roscoe et al. (2001)Roscoe R, Buurman P, Velthorst EJ, Vasconcellos CA. Soil organic matter dynamics in density and particle size fractions as revealed by the13C/12C isotopic ratio in a Cerrado's Oxisol. Geoderma. 2001;104:185-202. observed that 50 % of the total SOC was resilient due to charred material. Jantalia et al. (2007)Jantalia CP, Resck DVS, Alves BJR, Zotarelli L, Urquiaga S, Boddey RM. Tillage effect on C stocks of a clayey Oxisol under a soybean-based crop rotation in the Brazilian Cerrado region. Soil Till Res. 2007;95:97-109.estimated that up to 40 % of total SOC in a Ferralsol under soybean-based crop rotation was pyrogenic. The addition of biochar to Cerradosoils is likely to construct more resilient and fertile cropping systems by improvements and maintenance of SOC, increased soil pH and the capacity to exchange ions and retain water.
Several studies have reported the potential role of biochar in improving soil water retention capacity. Table 1 shows that more studies tested the effect of biochar on the soil water retention capacity of sandy (14) than of clay soils (eigth) and others (two). The majority of these studies were conducted under artificially controlled conditions (16). A variety of types and a wide range of amounts of biochar have been tested, including wood biochar (16). Overall, most studies applied unfeasibly high amounts of biochar (15 studies with rates of ≥2 % w/w) rather than more realistic amounts (nine with rates of ≤2 % w/w). On sandy soils, positive effects were observed in almost all studies, whereas on clay soils neutral (zero) effects were reported more often. The positive effects were generally related to the high specific surface area due to the porous structure of biochar, which behaves as additional capillaries, favoring water retention by the soil. However, only 33 % of the studies in table 1 addressed this characteristic. This brief review points out that positive effects of biochar on water retention capacity will depend not only on the soil type but also on biochar type and rate.
According to Glaser et al. (2002)Glaser B, Lehmann J, Zech W. Ameliorating physical and chemical properties of highly weathered soils in the tropics with charcoal - a review. Biol Fertil Soils. 2002;35:219-30., the physical and chemical composition of slow-pyrolysis wood biochar can increase the soil water-holding capacity and positively affect soil chemical properties. Additionally, its alkaline pH can decrease soil acidity. If soil acidity decreases, the capacity of soil to exchange ions increases. As a result, biochar can improve nutrient use efficiency. Because wood biochar can improve soil chemical and physical properties, higher yields are expected. Apart from the type of biochar, soil type, crop and amount of biochar applied, the time after incorporation of biochar into the soil is a relevant factor for its effect on soil properties and plant production. The residence time of biochar in soils can be significant (McHenry, 2011McHenry MP. Soil organic carbon, biochar, and applicable research results for increasing farm productivity under Australian agricultural conditions. Commun Soil Sci Plant Anal. 2011;42:1187-99.; Schmidt et al., 2011Schmidt MWI, Torn MS, Abiven S, Dittmar T, Guggenberger G, Janssens IA, Kleber M, Kögel-Knabner I, Lehmann J, Manning DA, Nannipieri P, Rasse DP, Weiner S, Trumbore SE. Persistence of soil organic matter as an ecosystem property. Nature. 2011;478:49-56.), which is a desirable characteristic under conditions favorable for mineralization, as in tropical Savannahs. Yet, biochar can interact with the soil matrix and undergo a weathering process that can change its original physical and chemical characteristics, a process known as "aging" (Kookana et al., 2011Kookana RS, Sarmah AK, van Zwieten L, Krull E, Singh B. Biochar application to soil: agronomic and environmental benefits and unintended consequences. Adv Agron. 2011;112:103-43.).
Another relevant factor regarding the use of biochar as a soil amendment is its availability. In developed regions of the world, e.g., in Europe, carbonization of domestic waste and organic residues is already a common practice. Biochar can be the link between biomass/residue management and energy/food production. A network to study the impact of the use of biochar in agriculture systems is already under way, i.e., the European Biochar Research network, running from 2012 to 2016. Programs such as the "Interreg IVB project Biochar: climate saving soils" are launching first final reports of the last four years of extensive research on a number of questions related to biochar production and biochar applications in seven North Sea countries (http://www.biochar-interreg4b.eu/). In Brazil, bioenergy production from biomass is increasing. Production of biodiesel and ethanol from sugarcane plantations is the most widely acknowledged. Further, the area covered with timber plantations specifically for energy production has also increased: in 2010, 35 % of the wood from forest plantations was destined for charcoal production (ABRAF, 2011Associação Brasileira dos Produtores de Florestas Plantadas - ABRAF. Anuário estatístico 2011, ano base 2010 [internet]. Brasília, DF: 2011. Available at: http//www.abraflor.org.br/.
http//www.abraflor.org.br/...
). Wood charcoal, sugarcane straw, crop residues and even organic waste could be used as energy sources in the production of gas, oil and solid by-products, such as biochar. Waste-to-energy plants could deliver both energy and carbonized biomass, which could in turn be used as a soil amendment.
The biochar research in Brazil was initiated about 10-15 years ago (Maia et al., 2011Maia CMBF, Madari BE, Novotny EH. Advances in biochar research in Brazil. Dyn Soil Dyn Plant. 2011;5:53-8.). In an Amazon Ferralsol, Steiner et al. (2007)Steiner C, Teixeira WG, Lehmann J, Nehls T, Macêdo JLV, Blum WEH, Zech W. Long term effects of manure, charcoal and mineral fertilization on crop production and fertility on a highly weathered Central Amazonian upland soil. Plant Soil. 2007;291:275-90. showed a promising positive effect of wood biochar combined with organic fertilization on aerobic rice production. In the Cerrado, the effect of biochar has been studied since 2006 in different soil types (Maia et al., 2011Maia CMBF, Madari BE, Novotny EH. Advances in biochar research in Brazil. Dyn Soil Dyn Plant. 2011;5:53-8.). Initially, pot experiments showed significant effects of wood biochar on decreasing acidity and increasing soil nutrient availability of a clay soil and consequently improving aerobic rice growth and biomass production, as shown by Madari et al. (2006)Madari BE, Costa AR, Castro LM, Santos JLS, Benites VM, Rocha AO, Machado PLOA. Carvão vegetal como condicionador de solo para arroz de terras altas (cultivar Primavera): um estudo prospectivo. Santo Antônio de Goiás: Empresa Brasileira de Pesquisa Agropecuária; 2006. (Comunicado técnico, 125). Accessed in 12 Nov. 2014. Available at: https://www.embrapa.br/busca-de-publicacoes/-/publicacao/215275/carvao-vegetal-como-condicionador-de-solo-para-arroz-de-terras-altas-cultivar-primavera-um-estudo-prospectivo
https://www.embrapa.br/busca-de-publicac...
. Later, Pereira et al. (2012)Pereira RG, Heinemann AB, Madari BE, Carvalho MTM, Kliemann HJ, Santos AP. Transpiration response of upland rice to water deficit changed by different levels of eucalyptus biochar. Pesq Agropec Bras. 2012;47:716-21. demonstrated the significant effect of wood biochar on increasing the capacity of sand to hold water. In a field trial, Petter et al. (2012)Petter FA, Madari BE, Silva MAS, Carneiro MAC, Carvalho MTM, Marimon Jr, BH, Pacheco LP. Soil fertility and upland rice yield after biochar application in the Cerrado. Pesq Agropec Bras. 2012;47:699-706. also reported positive effects of biochar on soil chemical properties and outstanding positive effects on aerobic rice yields on a sandy loam soil immediately and one year after application.
According to Laird et al. (2008)Laird DA. The charcoal vision: A win-win-win scenario for simultaneously producing bioenergy, permanently sequestering carbon, while improving soil and water quality. Agron J. 2008;100:178-81., the so-called "win-win-win" scenario of biochar application refers to the simultaneous use of biochar for bioenergy production, C sequestration, and improvement of soil and water quality. The implementation of this scenario requires the establishment of long-term effects of biochar on soil properties under real farming conditions, which depends, as described above, on a multitude of factors. There is also an urgent need to determine the environmental impact of biochar on cropping systems, such as its effect on N use efficiency. Biochar potentially has influence on the rates of N cycling in soil systems by affecting nitrification rates and adsorption of ammonia, and increasing ammonium storage by enhancing CEC in soils (Clough and Condron, 2010Clough TJ, Condron LM. Biochar and the nitrogen cycle: introduction. J Environ Qual. 2010;39:1218-23.). Its influence on these processes may have further implications in terms of reducing nitrate leaching and gaseous N losses, for example of nitrous oxide (N2O), a potent greenhouse gas. However, excessive applications of biochar and synthetic N in soil could provide energy in the form of labile C and ammonium for nitrate production by nitrifying bacteria. Under intermittent aerobic/anaerobic conditions, denitrifying bacteria can transform nitrate into N2O/N2.
Numerous studies have reported a potential suppressive effect of biochar on N2O emissions (Lehmann et al., 2006Lehmann J, Gaunt J, Rondon M. Biochar sequestration in terrestrial ecosystems - a review. Mitigat Adapt Strat Global Change. 2006;11:403-27.; Spokas et al., 2009Spokas KA, Koskinen WC, Baker JM, Reicosky DC. Impacts of woodchip biochar additions on greenhouse gas production and sorption/degradation of two herbicides in a Minnesota soil. Chemosphere.2009;77:574-81.;Atkinson et al., 2010Atkinson CJ, Fitzgerald JD, Hipps NA. Potential mechanisms for achieving agricultural benefits from biochar application to temperate soils: A review. Plant Soil. 2010;337:1-18.; Cayuela et al., 2010Cayuela ML, Oenema O, Kuikman PJ, Bakker RR, van Groenigen JW. Bioenergy by-products as soil amendments? Implications for carbon sequestration and greenhouse gas emissions. Global Change Biol Bioenergy. 2010;2:201-13.; Sohi et al., 2010Sohi SP, Krull E, Lopez-Capel E, Bol R. A review of biochar and its use and function in soil. In: Sparks DL, editor. Advances in agronomy. Waltham: Academic Press; 2010. v.105, p.47-82.; Zhang et al., 2010bZhang A, Cui L, Pan G, Li L, Hussain Q, Zhang X, Zheng J, Crowley D. Effect of biochar amendment on yield and methane and nitrous oxide emissions from a rice paddy from Tai Lake plain, China. Agric Ecosyst Environ. 2010b;139:469-75.), as well as some evidence of a neutral effect (Karhu et al., 2011Karhu K, Mattila T, Bergström I, Regina K. Biochar addition to agricultural soil increased CH4 uptake and water-holding capacity - Results from a short-term pilot field study. Agric Ecosyst Environ. 2011;140:309-13.; Scheer et al., 2011Scheer C, Grace PR, Rowlings DW, Kimber S, van Zwieten L. Effect of biochar amendment on the soil-atmosphere exchange of greenhouse gases from an intensive subtropical pasture in northern New South Wales, Australia. Plant Soil. 2011;345:47-58.). Reductions in soil bulk density, increased soil aeration and possible immobilization of N are among the causes for reduced N2O fluxes due to biochar application. However, data on the effect of biochar on greenhouse gas emissions are contradictory (Mukherjee and Lal, 2014Mukherjee A, Zimmerman AR, Harris W. Surface chemistry variations among a series of laboratory-produced biochars. Geoderma. 2011;163:247-55.). Conflicting results for the effect of biochar amendment on N2O fluxes seem to be not only related to differences in soil texture and biochar types, but mainly to variations in the soil moisture state. Clearly, the interactions between biochar and synthetic N fertilization and their effect on N2O fluxes in cropping systems need further investigation.
Effects of wood biochar application on soil properties and crop performance in a sandy and a clay soil types of the Cerrado - main results of a field study in Brazil
Knowing that the effect of biochar on soil properties and plant productivity depends on the physical-chemical characteristics and environmental conditions, the main findings of a comprehensive field study carried out with two soil types, a clay Rhodic Ferralsol and a sandy loam Haplic Plintossol, in the Brazilian Cerrado, are presented here. Our research question was whether biochar can be used in an agronomically beneficial and sustainable manner to increase the productivity of cropping systems in a tropical Savannah. The effect of a single application of different rates (0; 8; 16; and 32 Mg ha-1) of wood biochar on soil chemical and physical properties and on crop yields over four to five growing seasons was studied. The wood biochar was a by-product of slow pyrolysis (~450 ºC) of eucalyptus wood, a potentially available resource in the Central West region of Brazil, where the studies were carried out. Pieces of wood charcoal smaller than 8 mm are worthless for domestic or industrial uses and could be applied as soil amendment. Wood charcoal pieces were ground, sieved through 2 mm mesh and incorporated in the 0-15 cm soil layer.
Table 2, adapted from Carvalho (2015)Carvalho MTM. The impact of wood biochar as a soil amendment in aerobic rice systems of the Brazilian Savannah [thesis] Wageningen: Wageningen University; 2015., summarizes the main effects of wood biochar (WB) on key soil properties, crop yield and N2O emissions during the growing seasons. The effect of WB on soil water-holding capacity varied according to the soil type. On sandy soil, WB increased the water retention capacity 2 and 3 yr after its application (Carvalho et al., 2014Carvalho MTM, Maia AHN, Madari BEM, Bastiaans L, van Oort PAJ, Heinemann AB, Da Silva MAS, Petter FA, Meinke H. Biochar increases plant available water in a sandy soil under an aerobic rice cropping system. Solid Earth. 2014;5:939-52.). Conversely, WB amendment decreased the water retention capacity of the clay soil 1.5 and 2.5 yr after its application (Carvalho, 2015Carvalho MTM. The impact of wood biochar as a soil amendment in aerobic rice systems of the Brazilian Savannah [thesis] Wageningen: Wageningen University; 2015.). Many other studies reported an increased water retention capacity of sandy soils after biochar application. However, these findings were mostly results of pot experiments (Table 1). Some field studies also described positive effects of biochar on the water retention capacity of sandy loam soils. Liu et al. (2012a)Liu J, Schulz H, Brandl S, Miehtke H, Huwe B, Glaser B. Short-term effect of biochar and compost on soil fertility and water status of a Dystric Cambisol in NE Germany under field conditions. J Plant Nutr Soil Sci. 2012a;175:698-707., for example, reported a twofold increment in water retention capacity of a sandy soil treated with biochar and compost.
Overview of effects of wood biochar amendment on key soil properties and grain yield throughout four (on sandy soil) and five (on clay soil) main growing seasons in the Brazilian Savannah, adapted from Carvalho (2015)
The negative effect of WB on water retention capacity of the clay soil was probably a consequence of the intrinsic low bulk density of the material and the disaggregation of the clay soil due to biochar incorporation (Carvalho, 2015Carvalho MTM. The impact of wood biochar as a soil amendment in aerobic rice systems of the Brazilian Savannah [thesis] Wageningen: Wageningen University; 2015.). These findings are in agreement with Tryon (1948)Tryon EH. Effect of charcoal on certain physical, chemical and biological properties of forest soils. Ecol Soc Am. 1948;18:81-115.. To our best knowledge, this is the first time such an effect was confirmed in a field study. So far, most reports on effects of biochar on water retention capacity of a clay soil mention the absence of an effect or a positive response (Table 1). The response seems to be strongly related to the amount of biochar added and the conditions under which biochar is applied to the soil, i.e., tillage or no-tillage, pot or field experiment.
In the clay soil, a significant "liming" effect was observed up to 1.5 yr after WB application and in the sandy soil even up to 3 yr after WB application; however, this effect decreased over the seasons and was greater at higher WB rates (Carvalho, 2015Carvalho MTM. The impact of wood biochar as a soil amendment in aerobic rice systems of the Brazilian Savannah [thesis] Wageningen: Wageningen University; 2015.). As liming, the effect of WB application seems to be temporary, requiring repeated applications to ensure an adequate soil pH for crop production. This effect of WB is especially important for tropical Savannah soils, which require pH adjustment to become agronomically productive. The technique of adjusting the soil pH in the Brazilian Savannah was revolutionary, enabling a production of over 40 % of the nation's current total production of major crops in that region (IBGE, 2014Instituto Brasileiro de Geografia e Estatística - IBGE. Indicadores IBGE: Estatística da produção agrícola [internet]. 2014 [accessed in Oct 1st 2014]. Available at: http://www.ibge.gov.br/home/estatistica/indicadores/agropecuaria/lspa/default.shtm.
http://www.ibge.gov.br/home/estatistica/...
). Our findings show that WB can act as a source of K, Ca and Mg, increasing the soil pH, benefitting many crops (Carvalho, 2015Carvalho MTM. The impact of wood biochar as a soil amendment in aerobic rice systems of the Brazilian Savannah [thesis] Wageningen: Wageningen University; 2015.). A comprehensive economical comparison between the use of WB and conventional Ca and Mg sources will show whether biochar is a viable alternative.
The crop response to WB application varied according to the changes observed in soil chemical and physical properties and to the weather conditions throughout the seasons. On the clay soil, WB application rates had no effect on common bean yields, immediately after application. Similarly, WB amendment had no effect on aerobic rice yields, 0.5 yr after biochar application (Carvalho et al., 2013aCarvalho MTM, Madari BE, Leal WGO, Costa AR, Machado PLO, Silveira PM, Moreira JAA, Heinemann AB. Fluxes of nitrogen from irrigated common-bean as affected by mulching and mineral fertilization. Pesq Agropec Bras. 2013b;48:478-86.). One and a half and two and a half years after WB application, rice yields decreased with WB rates and depended on N fertilization: WB increased rice yield only when more than 60 kg ha-1 of N was applied. Three and a half years after WB application, no effect on rice yield was observed. The negative effect of WB on rice yield most probably arises from the negative effects on water retention capacity as well as on soil N availability in the clay soil (Carvalho, 2015Carvalho MTM. The impact of wood biochar as a soil amendment in aerobic rice systems of the Brazilian Savannah [thesis] Wageningen: Wageningen University; 2015.). On the sandy soil, WB had no effect on grain yield 2 and 3 yr after its application, contrary to the very positive effects observed immediately and 1 yr after biochar application. Interestingly, these first two seasons reported by Petter et al. (2012)Petter FA, Madari BE, Silva MAS, Carneiro MAC, Carvalho MTM, Marimon Jr, BH, Pacheco LP. Soil fertility and upland rice yield after biochar application in the Cerrado. Pesq Agropec Bras. 2012;47:699-706. were very dry and the latest two seasons studied by Carvalho et al. (2014)Carvalho MTM, Maia AHN, Madari BEM, Bastiaans L, van Oort PAJ, Heinemann AB, Da Silva MAS, Petter FA, Meinke H. Biochar increases plant available water in a sandy soil under an aerobic rice cropping system. Solid Earth. 2014;5:939-52. were very wet. Due to this unfortunate sequences of dry and wet seasons it was impossible to directly test whether the positive effects observed by Petter et al. (2012)Petter FA, Madari BE, Silva MAS, Carneiro MAC, Carvalho MTM, Marimon Jr, BH, Pacheco LP. Soil fertility and upland rice yield after biochar application in the Cerrado. Pesq Agropec Bras. 2012;47:699-706. were due to fertilization (nutrients added via WB) or to improved water retention, or maybe to both. Therefore, our analysis of the soil water retention curves was valuable for providing the missing link. There was strong indirect evidence that the positive WB effect on rice yields was (at least partially) due to increased water retention. Nevertheless, WB application to sandy loam soils seems to be a strategic option to avoid yield losses and even maintain yield stability in case of dry spells or seasons. Overall, on less fertile sandy soils, biochar can increase crop yields because it represents a source of readily available nutrients (fertilizer), such as K, and enhances the water retention capacity. On a more fertile clay soil, the effect of biochar on crop yields is likely to be related to N availability, particularly in no-tillage and crop rotation systems.
The soil organic matter increased with WB rate, from 2.5 yr after WB application in the clay soil (Carvalho, 2015Carvalho MTM. The impact of wood biochar as a soil amendment in aerobic rice systems of the Brazilian Savannah [thesis] Wageningen: Wageningen University; 2015.). The application of WB, in combination with complementary annual N fertilization and fresh organic matter (added via crop residues) seem to be the key components to build up soil organic matter in the clay soil. Liu et al. (2012a)Liu J, Schulz H, Brandl S, Miehtke H, Huwe B, Glaser B. Short-term effect of biochar and compost on soil fertility and water status of a Dystric Cambisol in NE Germany under field conditions. J Plant Nutr Soil Sci. 2012a;175:698-707. and Schulz et al. (2013)Schulz H, Dunst G, Glaser B. Positive effects of composted biochar on plant growth and soil fertility. Agron Sustain Develop. 2013;33:817-27. reported the importance of combining biochar with organic fertilization or composting biochar to improve soil fertility. Schmidt (2008)Schmidt H-P. Ways of making Terra Preta: Biochar activation [internet]. J Terrier-Wine Biodiversity, ISSN 1663-0521; 2008 [accessed in Oct 17 2014]. Available at: http://www.ithaka-journal.net/wege-zu-terra-preta-aktivierung-von-biokohle?lang=en.
http://www.ithaka-journal.net/wege-zu-te...
argues that biochar is not a fertilizer, but rather a nutrient carrier and a habitat for microorganisms and that it has to be charged first to be biologically active and efficient in soil-enhancing properties.
Concerning the effect of biochar on N2O emissions, no effect of WB amendment (maximum rate of 32 Mg ha-1) on N2O fluxes from clay soil was observed (Carvalho, 2015Carvalho MTM. The impact of wood biochar as a soil amendment in aerobic rice systems of the Brazilian Savannah [thesis] Wageningen: Wageningen University; 2015.). Mineral N fertilization increased N2O emissions, as expected. Gradual changes in soil organic matter and water-filled pore space seem to have a greater impact on the magnitude of N2O fluxes over seasons. The absence of a significant effect of WB amendment on N2O fluxes was probably linked to the physical structure of the clay Ferralsol and the aerobic nature of the cropping system. Under well-drained conditions, there is no accumulation of water on the soil surface and the major part of the mineral N applied as top dressing could be lost by ammonia volatilization (Carvalho et al., 2013b). Verhoeven and Six (2014)Verhoeven E, Six J. Biochar does not mitigate field-scale N2O emissions in a Northern California vineyard: An assessment across two years. Agric Ecosyst Environ. 2014;191:27-38.observed no effect of biochar on N2O fluxes under aerobic conditions either. According to Sanchez-Garcia et al. (2014)Sanchez-Garcia M, Roig A, Sanchez-Monedero MA, Cayuela ML. Biochar increases soil N2O emissions produced by nitrification-mediated pathways. Front Environ Sci. 2014;2:1-10., under aerobic conditions, where most of the N2O fluxes are produced in the process of nitrification, biochar application may fail to reduce N2O fluxes. On the contrary, these could even be intensified by biochar application. Conversely, under anaerobic conditions, such as in flooded rice systems where N2O fluxes are produced mainly due to denitrification, biochar could reduce N2O emissions. Liu et al. (2012b)Liu X, Qu J, Li L, Zhang A, Jufeng Z, Zheng J, Pan G. Can biochar amendment be an ecological engineering technology to depress N2O emission in rice paddies? - A cross site field experiments from South China. Ecol Eng. 2012b;42:168-73. and Zhang et al. (2012)Zhang A, Bian R, Pan G, Cui L, Hussain Q, Li L, Zheng J, Zheng J, Zhang X, Han X, Yu X. Effects of biochar amendment on soil quality, crop yield and greenhouse gas emission in a Chinese rice paddy: a field study of two consecutive rice growing cycles. Field Crops Res. 2012;127:153-60. observed this reduction of emission in rice paddy field experiments. It is known that during the flooding period, when the soil solution reaches highly reductive conditions, the NO3- concentration is reduced (Yu et al., 2007Yu K, Böhme F, Rinklebe J, Neue HU, Delaune RD. Major biogeochemical process in soils-a microcosm incubation from reducing to oxidizing conditions. Soil Sci Soc Am J. 2007;71:1406-17.). Under these conditions, denitrifying bacteria use N2O as N source and reduce it to molecular nitrogen (N2) (Chapuis-Lardy, 2007Chapuis-Lardy L, Wrage N, Metay A, Chotte J-L, Bernoux M. Soils, a sink for N2O? A review. Global Change Biol. 2007;13:1-17.), a process, that may even result in influx (negative flux) of N2O. Biochar could play a role in this process, for example by immobilizing nitrate from the soil solution.
Finally, the effect of WB on reducing rice blast infestation is a promising finding and calls for further investigation (Carvalho et al., 2013aCarvalho MTM, Madari BE, Bastiaans L, van Oort PAJ, Heinemann AB, Silva MAS, Maia AHN, Meinke H. Biochar improves fertility of a clay soil in the Brazilian Savannah: short term effects and impact on rice yield. J Agric Rural Develop Trop Subtrop. 2013a;114:101-7.). Elad et al. (2010)Elad Y, David DR, Harel YM, Borenshtein M, Kalifa HB, Silber A, Graber ER. Induction of systemic resistance in plants by biochar, a soil-applied carbon-sequestering agent. Phytopathology. 2010;100:913-21. and Harel et al. (2012)Harel YM, Elad Y, Rav-David D, Borenstein M, Schulchani R, Lew B, Graber ER. Biochar mediates systemic response of strawberry to foliar fungal pathogens. Plant Soil. 2012;357:245-57.reported on positive effects of biochar on decreasing foliar fungal infections in tomatoes, pepper and strawberry. Improved plant responses to diseases can be yet another benefit of applying biochar to soil (Elad et al., 2011Elad Y, Cytryn E, Meller Harel Y, Lew B, Graber ER. The biochar effect: plant resistance to biotic stresses. Phytopatol Mediter. 2011;50:335-49.).
In conclusion, WB could improve soil fertility by reducing soil acidity and increasing nutrient availability in the weathered soils of the Brazilian Savannah. Obviously, the best opportunities for increases in crop yield were confirmed on sandy soil. Economic evaluations of biochar as a substitute for lime should clarify the prospects of the use of this by-product as a soil amendment by farmers in this region. Our findings show the great importance of longer-term field experimentation addressing effects on both chemical and physical components of soil fertility, with a view to estimate the value of biochar for farming nowadays.
ACKNOWLEDGEMENTS
The authors are grateful for the Editorial Board of the Revista Brasileira de Ciência do Solo for the kind invitation to publish this review. Etelvino Henrique Novotny thanks the National Council of Technological and Scientific Development (CNPq) for a fellowship. Financial support: National Council of Technological and Scientific Development (CNPq - Projects: 590059/2011-4 and 480602/2007-7) and Embrapa (Projects: 02.09.01.022; 03.08.04.001; and 03.10.01.009).
REFERENCES
- Abel S, Peters A, Trinks S, Schonsky H, Facklam M, Wessolek G. Impact of biochar and hydrochar addition on water retention and water repellency of sandy soil. Geoderma. 2013;202-03:183-91.
- Akhtar J, Amin NAS. A review on process conditions for optimum bio-oil yield in hydrothermal liquefaction of biomass. Renew Sustain Energy Rev. 2011;15:1615-24.
- Amen-Chen C, Pakdel H, Roy C. Production of monomeric phenols by thermochemical conversion of biomass: A review. Biores Technol. 2001;79:277-99.
- Araujo JR, Archanjo BS, Souza KR, Kwapinski W, Falcão NPS, Novotny EH, Achete CA. Selective extraction of humic acids from an anthropogenic Amazonian dark earth and from a chemically oxidized charcoal. Biol Fertil Soils. 2014;50:1223-32.
- Asai H, Samson BK, Stephan HM, Songyikhangsuthor K, Homma K, Kiyono Y, Inoue Y, Shiraiwa T, Horie T. Biochar amendment techniques for upland rice production in Northern Laos 1. Soil physical properties, leaf SPAD and grain yield. Field Crop Res. 2009;111:8-84.
- Ascough PL, Bird MI, Wormald P, Snape CE, Apperley D. Influence of production variables and starting material on charcoal stable isotopic and molecular characteristics. Geochim Cosmochim Acta. 2008;72:6090-102.
- Associação Brasileira dos Produtores de Florestas Plantadas - ABRAF. Anuário estatístico 2011, ano base 2010 [internet]. Brasília, DF: 2011. Available at: http//www.abraflor.org.br/.
» http//www.abraflor.org.br/ - Atkinson CJ, Fitzgerald JD, Hipps NA. Potential mechanisms for achieving agricultural benefits from biochar application to temperate soils: A review. Plant Soil. 2010;337:1-18.
- Basso AS, Miguez FE, Laird DA, Horton R, Westgate M. Assessing potential of biochar for increasing water-holding capacity of sandy soils. Global Change Biol Bioenergy. 2013;5:132-43.
- Beck DA, Johnson GR, Spolek GA. Amending greenroof soil with biochar to affect runoff water quantity and quality. Environ Pollut. 2011;159:2111-18.
- Beesley L, Moreno-Jiménez E, Gomez-Eyles JL, Harris E, Robinson B, Sizmur T. A review of biochars' potential role in the remediation, revegetation and restoration of contaminated soils. Environ Pollut. 2011;159:3269-82.
- Benites VM, Mendonça ES, Schaefer CEGR, Novotny EH, Reis EL, Ker JC. Properties of black soil humic acids from high altitude rocky complexes in Brazil. Geoderma. 2005;127:104-13.
- Biederman LA, Harpole WS. Biochar and its effects on plant productivity and nutrient cycling: a meta-analysis. Global Change Biol Bioenergy. 2013;5:202-14.
- Bird MI, Ascough PL. Isotopes in pyrogenic carbon: A review. Org Geochem. 2012;42:1529-39.
- Brewer CE, Hu Y-Y, Schmidt-Rohr K, Loynachan TE, Laird DA, Brown RC. Extent of pyrolysis impacts on fast pyrolysis biochar properties. J Environ Qual. 2012;41:1115-22.
- Brewer CE, Schmidt-Rohr K, Satrio JA, Brown RC. Characterization of biochar from fast pyrolysis and gasification systems. Environ Prog Sustain Energy. 2009;28:386-96.
- Brewer CE, Unger R, Schmidt-Rohr K, Brown RC. Criteria to select biochars for field studies based on biochar chemical properties. BioEnergy Res. 2011;4:312-23.
- Bridgeman TG, Jones JM, Shield I, Williams PT. Torrefaction of reed canary grass, wheat straw and willow to enhance solid fuel qualities and combustion properties. Fuel. 2008;87:844-56.
- Bridgwater AV. Renewable fuels and chemicals by thermal processing of biomass. Chem Eng J. 2003;91:87-102.
- Bridgwater AV. Review of fast pyrolysis of biomass and product upgrading. Biomass Bioenergy. 2012;38:68-94.
- Brockhoff SR, Christians NE, Killorn RJ, Horton R, Davis DD. Physical and mineral-nutrition properties of sand-based turfgrass root zones amended with biochar. Agron J. 2010;102:1627-31.
- Brodowski S, Rodionov A, Haumaier L, Glaser B, Amelung W. Revised black carbon assessment using benzene polycarboxylic acids. Org Geochem. 2005;36:1299-310.
- Brunauer S, Emmett PH, Teller E. Adsorption of gases in multimolecular layers. J Am Chem Soc. 1938;60:309-19.
- Bruun EW, Hauggaard-Nielsen H, Ibrahim N, Egsgaard H, Ambus P, Jensen PA, Dam-Johansen K. Influence of fast pyrolysis temperature on biochar labile fraction and short-term carbon loss in a loamy soil. Biomass Bioenergy. 2011;35:1182-9.
- Calvelo Pereira R, Kaal J, Camps Arbestain M, Pardo Lorenzo R, Aitkenhead W, Hedley M, Macías F, Hindmarsh J, Maciá-Agulló JA. Contribution to characterisation of biochar to estimate the labile fraction of carbon. Org Geochem. 2011;42:1331-42.
- Canabarro N, Soares JF, Anchieta CG, Kelling CS, Mazutti MA. Thermochemical processes for biofuels production from biomass. Sustain Chem Proc. 2013;1:1-10.
- Carvalho MTM, Madari BE, Bastiaans L, van Oort PAJ, Heinemann AB, Silva MAS, Maia AHN, Meinke H. Biochar improves fertility of a clay soil in the Brazilian Savannah: short term effects and impact on rice yield. J Agric Rural Develop Trop Subtrop. 2013a;114:101-7.
- Carvalho MTM, Madari BE, Leal WGO, Costa AR, Machado PLO, Silveira PM, Moreira JAA, Heinemann AB. Fluxes of nitrogen from irrigated common-bean as affected by mulching and mineral fertilization. Pesq Agropec Bras. 2013b;48:478-86.
- Carvalho MTM, Maia AHN, Madari BEM, Bastiaans L, van Oort PAJ, Heinemann AB, Da Silva MAS, Petter FA, Meinke H. Biochar increases plant available water in a sandy soil under an aerobic rice cropping system. Solid Earth. 2014;5:939-52.
- Carvalho MTM. The impact of wood biochar as a soil amendment in aerobic rice systems of the Brazilian Savannah [thesis] Wageningen: Wageningen University; 2015.
- Cayuela ML, Oenema O, Kuikman PJ, Bakker RR, van Groenigen JW. Bioenergy by-products as soil amendments? Implications for carbon sequestration and greenhouse gas emissions. Global Change Biol Bioenergy. 2010;2:201-13.
- Cernansky R. Agriculture: State-of-the-art soil. Nature. 2015;517:258-60.
- Chapuis-Lardy L, Wrage N, Metay A, Chotte J-L, Bernoux M. Soils, a sink for N2O? A review. Global Change Biol. 2007;13:1-17.
- Chen B, Zhou D, Zhu L. Transitional adsorption and partition of nonpolar and polar aromatic contaminants by biochars of pine needles with different pyrolytic temperatures. Environ Sci Technol. 2008;42:5137-43.
- Chen Y, Shinogi Y, Taira M. Influence of biochar use on sugarcane growth, soil parameters, and groundwater quality. Aust J Soil Res. 2010;48:526-30.
- Cheng C-H, Lehmann J, Engelhard MH. Natural oxidation of black carbon in soils: Changes in molecular form and surface charge along a climosequence. Geochim Cosmochim Acta. 2008;72:1598-610.
- Cheng C-H, Lehmann J, Thies JE, Burton SD, Engelhard M. Oxidation of black carbon by biotic and abiotic processes. Org Geochem. 2006;37:1477-88.
- Chun Y, Sheng G, Chiou CT, Xing B. Compositions and sorptive properties of crop residue-derived chars. Environ Sci Technol. 2004;38:4649-55.
- Clarke AD, Noone KJ, Heintzenberg J, Warren SG, Covert DS. Aerosol light absorption measurement techniques: Analysis and intercomparisons. Atmos Environ. 1987;21:1455-65.
- Clough TJ, Condron LM. Biochar and the nitrogen cycle: introduction. J Environ Qual. 2010;39:1218-23.
- Crane-Droesch A, Abiven S, Jeffery S, Torn MS. Heterogeneous global crop yield response to biochar: a meta-regression analysis. Environ Res Lett. 2013;8:1-8.
- Cross A, Sohi SP. The priming potential of biochar products in relation to labile carbon contents and soil organic matter status. Soil Biol Biochem. 2011;43:2127-34.
- Davy E. On some experiments made with a view to determine the comparative value of peat and peat-charcoal for agriculture purposes. Lect Chem Carm Scholl Med. 1856;11:172-8.
- Deal C, Brewer CE, Brown RC, Okure MAE, Amoding A. Comparison of kiln-derived and gasifier-derived biochars as soil amendment in the humid tropics. Biomass Bioenergy. 2012;37:161-8.
- Deenik JL, Diarra A, Uehara G, Campbell S, Sumiyoshi Y, Antal Jr MJ. Charcoal ash and volatile matter effects on soil properties and plant growth in an acid Ultisol. Soil Sci. 2011;176:336-45.
- DeLuca TH, MacKenzie MD, Gundale MJ. Biochar effects on soil nutrient transformations. In: Lehmann J, Joseph S, editors. Biochar for environmental management: Science and Technology. London: Earthscan; 2009. p.251-70.
- Dempster DN, Jones DL, Murphy DV. Clay and biochar amendments decreased inorganic but not dissolved organic nitrogen leaching in soil. Soil Res. 2012;50:216-21.
- Derenne S, Largeau C. A review of some important families of refractory macromolecules: composition, origin, and fate in soils and sediments. Soil Sci. 2001;166:833-47.
- Devereux RC, Sturrock CJ, Mooney SJ. The effects of biochar on soil physical properties and winter wheat growth. Earth Environ Sci Trans Royal Soc Edinburg. 2012;103:13-18.
- Elad Y, Cytryn E, Meller Harel Y, Lew B, Graber ER. The biochar effect: plant resistance to biotic stresses. Phytopatol Mediter. 2011;50:335-49.
- Elad Y, David DR, Harel YM, Borenshtein M, Kalifa HB, Silber A, Graber ER. Induction of systemic resistance in plants by biochar, a soil-applied carbon-sequestering agent. Phytopathology. 2010;100:913-21.
- Elias VO, Simoneit BRT, Cordeiro RC, Turcq B. Evaluating Levoglucan as an indicator of biomass burning in Carajás, Amazônia: a comparison to the charcoal record. Geochim Cosmochim Acta. 2001;65:267-72.
- Elliott DC, Biller P, Ross AB, Schmidt AJ, Jones SB. Hydrothermal liquefaction of biomass: Developments from batch to continuous process. Biores Technol. 2014;178:147-56.
- Elmer WH, Lattao CV, Pignatello JJ. Active removal of biochar by earthworms (Lumbricus terrestris). Pedobiologia. 2015;58:1-6.
- European Biochar Certificate - EBC. Comparison of European Biochar Certificate Version 4.8 and IBI Biochar Standards Version 2.0; 2014 [accessed April 2 2015]. Available at: http://www.european-biochar.org/biochar/media/doc/IBI-EBC.pdf.
» http://www.european-biochar.org/biochar/media/doc/IBI-EBC.pdf - European Biochar Certificate - EBC. Guidelines for a sustainable production of biochar. Arbaz. [Switzerland]:European Biochar Foundation (EBC); 2012 [accessed April 2 2015]. Available at: www.european-biochar.org/en/download.
» www.european-biochar.org/en/download - Fang Y, Singh B, Singh BP. Effect of temperature on biochar priming effects and its stability in soils. Soil Biol Biochem. 2015;80:136-45.
- Fellet G, Marchiol L, Delle Vedove G, Peressotti A. Application of biochar on mine tailings: Effects and perspectives for land reclamation. Chemosphere. 2011;83:1262-67.
- Fidel RB, Laird DA, Thompson ML. Evaluation of modified Boehm titration methods for use with biochars. J Environ Qual. 2013;42:1771-8.
- Freitas JCC, Bonagamba TJ, Emmerich FG. 13C high resolution solid-state NMR study of peat carbonization. Energy Fuels. 1999;13:53-9.
- Freitas JCC, Bonagamba TJ, Emmerich FG. Investigation of biomass- and polymer-based carbon materials using 13C high-resolution solid-state NMR. Carbon. 2001;39:535-45.
- Gani A, Naruse I. Effect of cellulose and lignin content on pyrolysis and combustion characteristics for several types of biomass. Renew Energy. 2007;32:649-61.
- Glaser B, Amelung W. Pyrogenic carbon in native grassland soils along a climosequence in North America. Global Biogeochem Cycle. 2003;17:1064.
- Glaser B, Haumaier L, Guggenberger G, Zech W. Black carbon in soils: the use of benzenecarboxylic acids as specific markers. Org Geochem. 1998;29:811-9.
- Glaser B, Lehmann J, Zech W. Ameliorating physical and chemical properties of highly weathered soils in the tropics with charcoal - a review. Biol Fertil Soils. 2002;35:219-30.
- Gray M, Johnson MG, Dragila MI, Kleber M. Water uptake in biochars: The roles of porosity and hydrophobicity. Biomass Bioenergy. 2014;61:196-205.
- Guiotoku M, Hansel FA, Novotny EH, Maia CMBDF. Molecular and morphological characterization of hydrochar produced by microwave-assisted hydrothermal carbonization of cellulose. Pesq Agropec Bras. 2012;47:687-92.
- Gwenzi W, Chaukura N, Mukome FND, Machado S, Nyamasoka B. Biochar production and applications in sub-Saharan Africa: Opportunities, constraints, risks and uncertainties. J Environ Manage. 2015;150:250-61.
- Hammes K, Schmidt MWI, Smernik RJ, Currie LA, Ball WP, Nguyen TH, Louchouarn P, Houel S, Elmquist M, Cornelissen G, Skjemstad JO, Masiello CA, Song J, Peng P, Mitra S, Dunn JC, Hatcher PG, Hockaday WC, Smith DM, Hartkopf-Fröder C, Böhmer A, Lüer B, Huebert BJ, Amelung W, Brodowski S, Huang L, Zhang W, Gschwend PM, Flores-Cervantes DX, Largeau C, Rouzaud J-N, Rumpel C, Guggenberger G, Kaiser K, Rodionov A, Gonzalez-Vila FJ, Gonzalez-Perez JA, Rosa JM, Manning DAC, López-Capél E, Ding L. Comparison of quantification methods to measure fire-derived (black/elemental) carbon in soils and sediments using reference materials from soil, water, sediment and the atmosphere. Global Biogeochem Cycle. 2007;21:GB3016.
- Hamer U, Marschner B, Brodowski S, Amelung W. Interactive priming of black carbon and glucose mineralisation. Org Geochem. 2004;35:823-30.
- Harel YM, Elad Y, Rav-David D, Borenstein M, Schulchani R, Lew B, Graber ER. Biochar mediates systemic response of strawberry to foliar fungal pathogens. Plant Soil. 2012;357:245-57.
- Harvey OR, Herbert BE, Rhue RD, Kuo LJ. Metal interactions at the biochar-water interface: Energetics and structure-sorption relationships elucidated by flow adsorption microcalorimetry. Environ Sci Technol. 2011;45:5550-6.
- Hedges JI, Eglinton G, Hatcher PG, Kirchman DL, Arnosti C, Derenne S, Evershed RP, Kögel-Knabner I, DeLeeuw JW, Littke R, Michaelis W, Rullkötter J. The molecularly-uncharacterized component of nonliving organic matter in natural environments. Org Geochem. 2000;31:945-58.
- Heitkötter J, Marschner B. Interactive effects of biochar aging in soils related to feedstock, pyrolysis temperature, and historic charcoal production. Geoderma. 2015;245-246:56-64.
- Hilscher A, Heister K, Siewert C, Knicker H. Mineralisation and structural changes during the initial phase of microbial degradation of pyrogenic plant residues in soil. Org Geochem. 2009;40:332-42.
- Ibrahim HM, Al-Wabel MI, Usman ARA, Al-Omran A. Effect of Conocarpus biochar application on the hydraulic properties of a sandy loam soil. Soil Sci. 2013;178:165-73.
- Instituto Brasileiro de Geografia e Estatística - IBGE. Indicadores IBGE: Estatística da produção agrícola [internet]. 2014 [accessed in Oct 1st 2014]. Available at: http://www.ibge.gov.br/home/estatistica/indicadores/agropecuaria/lspa/default.shtm.
» http://www.ibge.gov.br/home/estatistica/indicadores/agropecuaria/lspa/default.shtm - International Biochar Initiative - IBI. Frequently asked questions about Biochar International Biochar Initiative [internet]. 2013 [accessed in Mar 13 2014]. Available at: http://www.biochar-international.org/biochar/faqs.
» http://www.biochar-international.org/biochar/faqs - International Biochar Initiative - IBI. Standardized product definition and product testing guidelines for biochar that is used in soil [internet]. 2014. Ithaca: [s.n.]. Available at: http://www.biochar-international.org/sites/default/files/IBI_Biochar_Standards_V20_final_2014.pdf .
» http://www.biochar-international.org/sites/default/files/IBI_Biochar_Standards_V20_final_2014.pdf - Jaffé R, Ding Y, Niggemann J, Vähätalo AV, Stubbins A, Spencer RGM, Campbell J, Dittmar T. Global charcoal mobilization from soils via dissolution and riverine transport to the oceans. Science. 2013;340:345-7.
- Jantalia CP, Resck DVS, Alves BJR, Zotarelli L, Urquiaga S, Boddey RM. Tillage effect on C stocks of a clayey Oxisol under a soybean-based crop rotation in the Brazilian Cerrado region. Soil Till Res. 2007;95:97-109.
- Jeffery S, Verheijen FGA, van der Velde M, Bastos AC. A quantitative review of the effects of biochar application to soils on crop productivity using meta-analysis. Agric Ecosyst Environ. 2011;144:175-87.
- Jones BEH, Haynes RJ, Phillips IR. Effect of amendment of bauxite processing sand with organic materials on its chemical, physical and microbial properties. J Environ Manage. 2010;91:2281-88.
- Jones DL, Murphy DV, Khalid M, Ahmad W, Edwards-Jones G, DeLuca TH. Short-term biochar-induced increase in soil CO2 release is both biotically and abiotically mediated. Soil Biol Biochem. 2011;43:1723-31.
- Karhu K, Mattila T, Bergström I, Regina K. Biochar addition to agricultural soil increased CH4 uptake and water-holding capacity - Results from a short-term pilot field study. Agric Ecosyst Environ. 2011;140:309-13.
- Keiluweit M, Nico PS, Johnson MG, Kleber M. Dynamic molecular structure of plant-derived black carbon (biochar). Environ Sci Technol. 2010;44:1247-53.
- Keith A, Singh B, Singh BP. Interactive priming of biochar and labile organic matter mineralization in a smectite-rich soil. Environ Sci Technol. 2011;45:9611-8.
- Kim KH; Kim JY, Cho TS, Choi JW. Influence of pyrolysis temperature on physicochemical properties of biochar obtained from the fast pyrolysis of pitch pine (Pinus rigida). Biores Technol. 2012;118:158-62.
- Kirubakaran V, Sivaramakrishnan V, Nalini R, Sekar T, Premalatha M, Subramanian P. A review on gasification of biomass. Renew Sustain Energy Rev. 2009;13:179-86.
- Kleineidam S, Rugner H, Grathwohl P. Impact of grain scale heterogeneity on slow sorption kinetics. Environ Toxicol Chem. 1999;18:1673-8.
- Knicker H, Hilscher A, González-Vila FJ, Almendros G. A new conceptual model for the structural properties of char produced during vegetation fires. Org Geochem. 2008b;39:935-9.
- Knicker H, Müller P, Hilscher A. How useful is chemical oxidation with dichromate for the determination of "Black Carbon" in fire-affected soils? Geoderma. 2007;142:178-96.
- Knicker H, Totsche KU, Almendros G, González-Vila FJ. Condensation degree of burnt peat and plant residues and the reliability of solid-state VACP MAS 13C NMR spectra obtained from pyrogenic humic material. Org Geochem. 2005;36:1359-77.
- Knicker H, Wiesmeier M, Dick DP. A simplified method for the quantification of pyrogenic organic matter in grassland soils via chemical oxidation. Geoderma. 2008a;147:69-74.
- Knicker H. Pyrogenic organic matter in soil: Its origin and occurrence, its chemistry and survival in soil environments. Quatern Intern. 2011;243:251-63.
- Koide RT, Petprakob K, Peoples M. Quantitative analysis of biochar in field soil. Soil Biol Biochem. 2011;43:1563-8.
- Kookana RS, Sarmah AK, van Zwieten L, Krull E, Singh B. Biochar application to soil: agronomic and environmental benefits and unintended consequences. Adv Agron. 2011;112:103-43.
- Kramer RW, Kujawinski EB, Hatcher PG. Identification of black carbon derived structures in a volcanic ash soil humic acid by fourier transform ion cyclotron resonance mass spectrometry. Environ Sci Technol. 2004;38:3387-95.
- Laird DA, Fleming P, Davis DD, Horton R, Wang B, Karlen DL. Impact of biochar amendments on the quality of a typical Midwestern agricultural soil. Geoderma. 2010;158:443-9.
- Laird DA. The charcoal vision: A win-win-win scenario for simultaneously producing bioenergy, permanently sequestering carbon, while improving soil and water quality. Agron J. 2008;100:178-81.
- Lee Y, Park J, Ryu C, Gang KS, Yang W, Park YK, Jung J, Hyun S. Comparison of biochar properties from biomass residues produced by slow pyrolysis at 500 °C. Biores Technol. 2013;148:196-201.
- Lehmann J, Silva Jr JP, Steiner C, Nehls T, Zech W, Glaser B. Nutrient availability and leaching in an archaeological Anthrosol and a Ferralsol of the Central Amazon basin: Fertilizer, manure and charcoal amendments. Plant Soil. 2003;249:343-57.
- Lehmann J, Gaunt J, Rondon M. Biochar sequestration in terrestrial ecosystems - a review. Mitigat Adapt Strat Global Change. 2006;11:403-27.
- Lehmann J, Joseph S. Biochar for environmental management: Science and technology. London: Earthscan; 2009.
- Lehmann J. A handful of carbon. Nature. 2007;447:143-44.
- Lehmann J, Rillig MC, Thies J, Masiello CA, Hockaday WC, Crowley D. Biochar effects on soil biota - A review. Soil Biol Biochem. 2011;43:1812-36.
- Laird DA, Fleming P, Davis DD, Horton R, Wang B, Karlen DL. Impact of biochar amendments on the quality of a typical Midwestern agricultural soil. Geoderma. 2010;158:443-9.
- Tammeorg P, Simojoki A, Mäkelä P, Stoddard FL, Alakukku L, Helenius J. Biochar application to a fertile sandy clay loam in boreal conditions: effects on soil properties and yield formation of wheat, turnip rape and faba bean. Plant Soil. 2013;374:89-107.
- Libra JA, Ro KS, Kammann C, Funke A, Berge ND, Neubauer Y, Titirici M-M, Fühner C, Bens O, Kern J, Emmerich K-H. Hydrothermal carbonization of biomass residuals: a comparative review of the chemistry, processes and applications of wet and dry pyrolysis. Biofuels. 2011;2:71-106.
- Linhares CR, Lemke J, Auccaise R, Duó DA, Ziolli RL, Kwapinski W, Novotny EH. Reproducing the organic matter model of anthropogenic dark earth of Amazonia and testing the ecotoxicity of functionalized charcoal compounds. Pesq Agropec Bras. 2012;47:693-8.
- Liu J, Schulz H, Brandl S, Miehtke H, Huwe B, Glaser B. Short-term effect of biochar and compost on soil fertility and water status of a Dystric Cambisol in NE Germany under field conditions. J Plant Nutr Soil Sci. 2012a;175:698-707.
- Liu X, Qu J, Li L, Zhang A, Jufeng Z, Zheng J, Pan G. Can biochar amendment be an ecological engineering technology to depress N2O emission in rice paddies? - A cross site field experiments from South China. Ecol Eng. 2012b;42:168-73.
- Liu X, Zhang A, Ji C, Joseph S, Bian R, Li L, Pan G, Paz-Ferreiro J. Biochar's effect on crop productivity and the dependence on experimental conditions - a meta-analysis of literature data. Plant Soil. 2013;373:583-94.
- Luo Y, Durenkamp M, De Nobili M, Lin Q, Brookes PC. Short term soil priming effects and the mineralisation of biochar following its incorporation to soils of different pH. Soil Biol Biochem. 2011;43:2304-14.
- Madari BE, Benites VM, Cunha TJF. The effect of management on the fertility of Amazonian Dark earth soils. In: Lehmann J, Kern DC, Glaser B, Woods WI, editors. Amazonian Dark Earths: Origin, properties, management. Dordrecht: Kluwer Academic Publishers; 2003. p.407-26.
- Madari BE, Costa AR, Castro LM, Santos JLS, Benites VM, Rocha AO, Machado PLOA. Carvão vegetal como condicionador de solo para arroz de terras altas (cultivar Primavera): um estudo prospectivo. Santo Antônio de Goiás: Empresa Brasileira de Pesquisa Agropecuária; 2006. (Comunicado técnico, 125). Accessed in 12 Nov. 2014. Available at: https://www.embrapa.br/busca-de-publicacoes/-/publicacao/215275/carvao-vegetal-como-condicionador-de-solo-para-arroz-de-terras-altas-cultivar-primavera-um-estudo-prospectivo
» https://www.embrapa.br/busca-de-publicacoes/-/publicacao/215275/carvao-vegetal-como-condicionador-de-solo-para-arroz-de-terras-altas-cultivar-primavera-um-estudo-prospectivo - Madari BE, Maia CMBF, Novotny EH. Context and importance of biochar research. Pesq Agropec Bras. 2012;47:i-ii.
- Maia CMBF, Madari BE, Novotny EH. Advances in biochar research in Brazil. Dyn Soil Dyn Plant. 2011;5:53-8.
- Major J, Lehmann J, Rondon M, Goodale C. Fate of soil-applied black carbon: downward migration, leaching and soil respiration. Global Change Biol. 2010a;16:1366-79.
- Major J, Rondon M, Molina D, Riha SJ, Lehmann J. Maize yield and nutrition during 4 years after biochar application to a Colombian savanna Oxisol. Plant Soil. 2010b;333:117-28.
- Mao JD, Hundal LS, Thompson ML, Schmidt-Rohr K. Correlation of poly(methylene)-rich amorphous aliphatic domains in humic substances with sorption of a nonpolar organic contaminant, phenanthrene. Environ Sci Technol. 2002;36:929-36.
- Mašek O, Brownsort P, Cross A, Sohi S. Influence of production conditions on the yield and environmental stability of biochar. Fuel. 2013b;103:151-5.
- Mašek O, Brownsort P. Research on Production of Bespoke Biochar [internet]; 2010 [accessed in Aug 12 2014]. Available at: http://www.biochar.ac.uk/abstract.php?id=32.
» http://www.biochar.ac.uk/abstract.php?id=32 - Mašek O, Budarin VLV, Gronnow MJ, Crombie KN, Brownsort P, Fitzpatrick E, Hurst PR, Mase O, Budarin VLV, Gronnow MJ, Crombie KN, Brownsort P, Fitzpatrick E, Hurst PR. Microwave and slow pyrolysis biochar - Comparison of physical and functional properties. J Anal Appl Pyrolysis. 2013a;100:41-8.
- Masiello CA. New directions in black carbon organic geochemistry. Mar Chem. 2004;92:201-13.
- McBeath A, Smernik R, Plant E. Determination of the aromaticity and the degree of aromatic condensation of a thermosequence of wood charcoal using NMR. Org Geochem. 2011;42:1194-202.
- McHenry MP. Soil organic carbon, biochar, and applicable research results for increasing farm productivity under Australian agricultural conditions. Commun Soil Sci Plant Anal. 2011;42:1187-99.
- McKendry P. Energy production from biomass (part 2): conversion technologies. Biores Technol. 2002a;83:47-54.
- McKendry P. Energy production from biomass (part 3): Gasification technologies. Biores Technol. 2002b;83:55-63.
- Melligan F, Auccaise R, Novotny EH, Leahy JJ, Hayes MHB, Kwapinski W. Pressurised pyrolysis of Miscanthus using a fixed bed reactor. Biores Technol. 2011;102:3466-70.
- Melligan F, Dussan K, Auccaise R, Novotny EH, Leahy JJ, Hayes MHB, Kwapinski W. Characterisation of the products from pyrolysis of residues after acid hydrolysis of Miscanthus Biores Technol. 2012;108:258-63.
- Melo L, Coscione A, Abreu C, Puga A, Camargo O. Influence of pyrolysis temperature on cadmium and zinc sorption capacity of sugar cane straw-derived biochar. BioResources. 2013;8:4992-5004.
- Mohan D, Pittman CU, Steele PH. Pyrolysis of Wood/Biomass for Bio-oil : A critical review. Energy Fuels. 2006;20:848-89.
- Mohan D, Sarswat A, Ok YS, Pittman Jr CU. Organic and inorganic contaminants removal from water with biochar, a renewable, low cost and sustainable adsorbent - A critical review. Biores Technol. 2014;160:191-202.
- Mukherjee A, Lal R, Zimmerman AR. Effects of biochar and other amendments on the physical properties and greenhouse gas emissions of an artificially degraded soil. Sci Total Environ. 2014;487:26-36.
- Mukherjee A, Zimmerman AR, Harris W. Surface chemistry variations among a series of laboratory-produced biochars. Geoderma. 2011;163:247-55.
- Neves D, Thunman H, Matos A, Tarelho L, Gómez-Barea A. Characterization and prediction of biomass pyrolysis products. Prog Energy Combust Sci. 2011;37:611-30.
- Novak JM, Cantrell KB, Watts DW, Busscher WJ, Johnson MG. Designing relevant biochars as soil amendments using lignocellulosic-based and manure-based feedstocks. J Soils Sedim. 2014;14:330-43.
- Novotny EH, Auccaise R, Velloso MHR, Corrêa JC, Higarashi MM, Abreu VMN, Rocha JD, Kwapinski W. Characterization of phosphate structures in biochar from swine bones. Pesq Agropec Bras. 2012;47:672-6.
- Novotny EH, Azevedo ER, Bonagamba TJ, Cunha TJF, Madari BE, Benites VM, Hayes MHB. Studies of the compositions of humic acids from Amazonian Dark Earth soils. Environ Sci Technol. 2007;41:400-5.
- Novotny EH, Hayes MHB, Azevedo ER, Bonagamba TJ. Characterisation of black carbon rich samples by 13C solid state nuclear magnetic resonance. Naturwissenshaften. 2006;93:447-50.
- Novotny EH, Hayes MHB, Madari BE, Bonagamba TJ, Azevedo ER, Souza AA, Song G, Nogueira CM, Mangrich AS. Lessons from the Terra Preta de Índio of the Amazon region for the utilization of charcoal for soil amendment. J Braz Chem Soc. 2009;20:1003-10.
- Ogawa M, Okimori Y. Pioneering works in biochar research, Japan. Aust J Soil Res. 2010;48:489-500.
- Paiva DM, Cantarutti RB, Guimarães GGF, Silva IR. Urea coated with oxidized charcoal reduces ammonia volatilization. R Bras Ci Solo. 2012;36:1221-9.
- Pereira RG, Heinemann AB, Madari BE, Carvalho MTM, Kliemann HJ, Santos AP. Transpiration response of upland rice to water deficit changed by different levels of eucalyptus biochar. Pesq Agropec Bras. 2012;47:716-21.
- Petter FA, Madari BE, Silva MAS, Carneiro MAC, Carvalho MTM, Marimon Jr, BH, Pacheco LP. Soil fertility and upland rice yield after biochar application in the Cerrado Pesq Agropec Bras. 2012;47:699-706.
- Pinto JSDS, Lanças FM. Hidrólise do óleo de Azadirachta indica em água subcrítica e determinação da composição dos triacilglicerídeos e ácidos graxos por cromatografia gasosa dealta resolução a alta temperatura e cromatografia gasosa de alta resolução acoplada à espectrometria. Quim Nova. 2010;33:394-7.
- Pivello VR, Oliveras I, Miranda HS, Haridasan M, Sato MN, Meirelles ST. Effect of fires on soil nutrient availability in an open savannah in Central Brazil. Plant Soil. 2010;337:111-23.
- Ponomarenko EV, Anderson DW. Importance of charred organic matter in Black Chernozem soils of Saskatchewan. Can J Soil Sci. 2001;81:285-97.
- Qian K, Kumar A, Zhang H, Bellmer D, Huhnke R. Recent advances in utilization of biochar. Renew Sustain Energy Rev. 2015;42:1055-64.
- Rambo MKD, Novotny EH, Canellas LP, Aguiar NO, Aucaisse R, Rambo MCD. Pyrolysis of banana and coffee residues after acid hydrolysis. J Chem Eng. 2014;8:960-70.
- Read P. This gift of nature is the best way to save us from climate catastrophe. The Guardian. 2009 Mar 27; Commentis free/biochar.
- Rittl TF, Arts B, Kuyper TW. Biochar: an emerging policy arrangement in Brazil? Environ Sci Policy. 2015a;51:45-55.
- Rittl, TF, Novotny EH, Balieiro FC, Hoffland E, Alves BJR, Kuyper TW. Negative priming on mineralization of native soil organic carbon by oilseed biochars of contrasting quality. Eur J Soil Sci. 2015b(in press).
- Rizhiya EYA, Buchkina NP, Mukhina IM, Belinets AS, Balashov EV. Effect of biochar on the properties of loamy sand Spodosol soil samples with different fertility levels: A laboratory experiment. Eurasian Soil Sci. 2015;48:192-200.
- Roscoe R, Buurman P, Velthorst EJ, Vasconcellos CA. Soil organic matter dynamics in density and particle size fractions as revealed by the13C/12C isotopic ratio in a Cerrado's Oxisol. Geoderma. 2001;104:185-202.
- Rutherford DW, Wershaw RL, Rostad CE, Kelly CN. Effect of formation conditions on biochars: Compositional and structural properties of cellulose, lignin, and pine biochars. Biomass Bioenergy. 2012;46:693-701.
- Sanchez-Garcia M, Roig A, Sanchez-Monedero MA, Cayuela ML. Biochar increases soil N2O emissions produced by nitrification-mediated pathways. Front Environ Sci. 2014;2:1-10.
- Scheer C, Grace PR, Rowlings DW, Kimber S, van Zwieten L. Effect of biochar amendment on the soil-atmosphere exchange of greenhouse gases from an intensive subtropical pasture in northern New South Wales, Australia. Plant Soil. 2011;345:47-58.
- Schmidt H-P. Ways of making Terra Preta: Biochar activation [internet]. J Terrier-Wine Biodiversity, ISSN 1663-0521; 2008 [accessed in Oct 17 2014]. Available at: http://www.ithaka-journal.net/wege-zu-terra-preta-aktivierung-von-biokohle?lang=en.
» http://www.ithaka-journal.net/wege-zu-terra-preta-aktivierung-von-biokohle?lang=en - Schmidt MWI, Noack AG. Black carbon in soils and sediments: Analysis, distribution, implications, and current challenges. Global Biogeochem Cycle. 2000;14:777-94.
- Schmidt MWI, Skjemstad JO, Czimczik CI, Glaser B, Prentice KM, Gelinas Y, Kuhlbusch TAJ. Comparative analysis of black carbon in soils. Global Biogeochem Cycle. 2001;15:163-7.
- Schmidt MWI, Skjemstad JO, Gehrt E, Kögel-Knabner I. Charred organic carbon in German chernozemic soils. Eur J Soil Sci. 1999;50:351-65.
- Schmidt MWI, Torn MS, Abiven S, Dittmar T, Guggenberger G, Janssens IA, Kleber M, Kögel-Knabner I, Lehmann J, Manning DA, Nannipieri P, Rasse DP, Weiner S, Trumbore SE. Persistence of soil organic matter as an ecosystem property. Nature. 2011;478:49-56.
- Schulz H, Dunst G, Glaser B. Positive effects of composted biochar on plant growth and soil fertility. Agron Sustain Develop. 2013;33:817-27.
- Simpson MJ, Hatcher PG. Overestimates of black carbon in soils and sediments. Naturwissenschaften. 2004;91:436-40.
- Singh B, Singh BP, Cowie AL. Characterisation and evaluation of biochars for their application as a soil amendment. Aust J Soil Res. 2010;48:516-25.
- Smith DM, Griffin JJ, Goldberg ED. Spectrometric method for the quantitative determination of elemental carbon. Anal Chem. 1975;47:233-8.
- Smith JL, Collins HP, Bailey VL. The effect of young biochar on soil respiration. Soil Biol Biochem. 2010;42:2345-7.
- Sohi SP, Krull E, Lopez-Capel E, Bol R. A review of biochar and its use and function in soil. In: Sparks DL, editor. Advances in agronomy. Waltham: Academic Press; 2010. v.105, p.47-82.
- Spokas KA, Cantrell KB, Novak JM, Archer DW, Ippolito JA, Collins HP, Boateng AA, Lima IM, Lamb MC, McAloon AJ, Lentz RD, Nichols KA. Biochar: A synthesis of its agronomic impact beyond carbon sequestration. J Environ Qual. 2012;41:973-89.
- Spokas KA, Koskinen WC, Baker JM, Reicosky DC. Impacts of woodchip biochar additions on greenhouse gas production and sorption/degradation of two herbicides in a Minnesota soil. Chemosphere.2009;77:574-81.
- Spokas KA. Review of the stability of biochar in soils: predictability of O:C molar ratios. Carbon Manage. 2010;1:289-303.
- Steinbeiss S, Gleixner G, Antonietti M. Effect of biochar amendment on soil carbon balance and soil microbial activity. Soil Biol Biochem. 2009;41:1301-10.
- Steiner C, Teixeira WG, Lehmann J, Nehls T, Macêdo JLV, Blum WEH, Zech W. Long term effects of manure, charcoal and mineral fertilization on crop production and fertility on a highly weathered Central Amazonian upland soil. Plant Soil. 2007;291:275-90.
- Stevens DJ. An overview of biomass thermochemical liquefaction research sponsored by the US Department of Energy. In: Proceedings of the 1985 Biomass Thermochemical Conversion Contractors' Meeting; 1985. Washington, DC: United States Department of Environment; 1986. Available at: https://web.anl.gov/PCS/acsfuel/preprintarchive/Files/Merge/Vol-32_2-0007.pdf .
» https://web.anl.gov/PCS/acsfuel/preprintarchive/Files/Merge/Vol-32_2-0007.pdf - Sullivan AL, Ball R. Thermal decomposition and combustion chemistry of cellulosic biomass. Atmos Environ. 2012;47:133-41.
- Sun D, Meng J, Liang H, Yang E, Huang Y, Chen W, Jiang L, Lan Y, Zhang W, Gao J. Effect of volatile organic compounds absorbed to fresh biochar on survival of Bacillus mucilaginosus and structure of soil microbial communities. J Soils Sedim. 2015;15:271-81.
- Sun K, Ro K, Guo M, Novak J, Mashayekhi H, Xing B. Sorption of bisphenol A, 17α-ethinyl estradiol and phenanthrene on thermally and hydrothermally produced biochars. Biores Technol. 2011;102:5757-63.
- Sun Y, Gao B, Yao Y, Fang J, Zhang M, Zhou Y, Chen H, Yang L. Effects of feedstock type, production method, and pyrolysis temperature on biochar and hydrochar properties. Chem Eng J. 2014;240:574-8.
- Thevenot M, Dignac M-F, Rumpel C. Fate of lignins in soils: A review. Soil Biol Biochem. 2010;42:1200-11.
- Toor SS, Rosendahl L, Rudolf A. Hydrothermal liquefaction of biomass: A review of subcritical water technologies. Energy. 2011;36:2328-42.
- Tryon EH. Effect of charcoal on certain physical, chemical and biological properties of forest soils. Ecol Soc Am. 1948;18:81-115.
- Tuomela M, Vikman M, Hatakka A, It M. Biodegradation of lignin in a compost environment: a review. Biores Technol. 2000;72:169-83.
- Uchimiya M, Lima IM, Klasson KT, Wartelle LH. Contaminant immobilization and nutrient release by biochar soil amendment: roles of natural organic matter. Chemosphere. 2010;80:935-40.
- Verhoeven E, Six J. Biochar does not mitigate field-scale N2O emissions in a Northern California vineyard: An assessment across two years. Agric Ecosyst Environ. 2014;191:27-38.
- Vleminckx J, Morin-Rivat J, Biwolé AB, Daïnou K, Gillet J-F, Doucet J-L, Drouet T, Hardy OJ. Soil charcoal to assess the impacts of past human disturbances on tropical forests. PLoSONE. 2014;9:108-21.
- Ulyett J, Sakrabani R, Kibblewhite M, Hann M. Impact of biochar addition on water retention, nitrification and carbon dioxide evolution from two sandy loam soils. Eur J Soil Sci. 2014;65:96-104.
- Uzoma KC, Inoue M, Andry H, Fujimaki H, Zahoor A, Ni shihara E. Effect of cow manure biochar on maize productivity under sandy soil condition. Soil Use Manage. 2011;27:205-12.
- Ventura F, Salvatorelli F, Piana S, Pieri L, Pisa PR. The effects of biochar on the physical properties of bare soil. Earth Environ Sci Trans Royal Soc Edinburg. 2012;103:5-11.
- Wang H, Lin K, Hou Z, Richardson B, Gan J. Sorption of the herbicide terbuthylazine in two New Zealand forest soils amended with biosolids and biochars. J Soils Sedim. 2009;10:283-9.
- Weyers SL, Spokas KA. Impact of Biochar on earthworm populations: A review. Appl Environ Soil Sci. 2011:ID 541592:1-12.
- Wiedemeier DB, Abiven S, Hockaday WC, Keiluweit M, Kleber M, Masiello CA, McBeath AV, Nico PS, Pyle LA, Schneider MPW, Smernik RJ, Wiesenberg GLB, Schmidt MWI. Aromaticity and degree of aromatic condensation of char. Org Geochem. 2015;78:135-43.
- Wu W, Yang M, Feng Q, McGrouther K, Wang H, Lu H, Chen Y. Chemical characterization of rice straw-derived biochar for soil amendment. Biomass Bioenergy. 2012;47:268-76.
- Yu K, Böhme F, Rinklebe J, Neue HU, Delaune RD. Major biogeochemical process in soils-a microcosm incubation from reducing to oxidizing conditions. Soil Sci Soc Am J. 2007;71:1406-17.
- Zhang A, Bian R, Pan G, Cui L, Hussain Q, Li L, Zheng J, Zheng J, Zhang X, Han X, Yu X. Effects of biochar amendment on soil quality, crop yield and greenhouse gas emission in a Chinese rice paddy: a field study of two consecutive rice growing cycles. Field Crops Res. 2012;127:153-60.
- Zhang A, Cui L, Pan G, Li L, Hussain Q, Zhang X, Zheng J, Crowley D. Effect of biochar amendment on yield and methane and nitrous oxide emissions from a rice paddy from Tai Lake plain, China. Agric Ecosyst Environ. 2010b;139:469-75.
- Zhang H, Voroney RP, Price GW. Effects of temperature and processing conditions on biochar chemical properties and their influence on soil C and N transformations. Soil Biol Biochem. 2015b;83:19-28.
- Zhang J, Liu J, Liu R. Effects of pyrolysis temperature and heating time on biochar obtained from the pyrolysis of straw and lignosulfonate. Biores Technol. 2015a;176:288-91.
- Zhang L, Xu C, Champagne P. Overview of recent advances in thermo-chemical conversion of biomass. Energy Converson Manage. 2010a;51:969-82.
- Zhang, Y. Hydrothermal liquefaction to convert biomass into crude oil. In: Blaschek HP, editor. Biofuels from Agriculture Wastes Byproduct. Oxford: Blackwell Publishing; 2010. p.201-32.
- Zhao L, Cao X, Mašek O, Zimmerman A. Heterogeneity of biochar properties as a function of feedstock sources and production temperatures. J Hazard Mater. 2013;256-57:1-9.
- Zheng H, Wang Z, Deng X, Herbert S, Xing B. Impacts of adding biochar on nitrogen retention and bioavailability in agricultural soil. Geoderma. 2013;206:32-39.
- Zimmerman A, Gao B, Ahn MY. Positive and negative carbon mineralization priming effects among a variety of biochar-amended soils. Soil Biol Biochem. 2011;43:1169-79.
- Zimmerman A. Abiotic and microbial oxidation of laboratory-produced black carbon (biochar). Environ Sci Technol. 2010;44:1295-301.
Publication Dates
-
Publication in this collection
Mar-Apr 2015
History
-
Received
1 Dec 2014 -
Accepted
30 Mar 2015