Abstracts
In order to detect fluctuations in ruminal microbial populations due to forage tannins using 16S ribosomal RNA (rRNA) probes, recovery of intact rRNA is required. The objective of this work was to evaluate the effect of polyethylene glycol (PEG) and polyvinylpirrolidone (PVP) on extraction of bacterial rRNA, in the presence of tannins from tropical legume forages and other sources, that hybridize with oligonucleotide probes. Ruminococcus albus 8 cells were exposed to 8 g/L tannic acid or 1 g/L condensed tannins extracted from Acacia angustissima, banana (Musa sp.) skin, Desmodium ovalifolium, red grape (Vitis vinifera) skin and Inga edulis, or no tannins. Cells were rinsed with Tris buffer pH 7 containing either 8% PEG or 6% PVP prior to cell lysis. Total RNA samples rinsed with either PEG or PVP migrated through denaturing agarose gels. The 16S rRNA bands successfully hybridized with a R. albus species-specific oligonucleotide probe, regardless of tannin source. The effect of rinsing buffers on the density of 16S rRNA bands, as well as on the hybridization signals was compared. There were significant effects (P<0.01) when the controls were compared to either buffer treatments due to tannin type, buffer used and the interaction of tannin type and buffer. The significant interaction indicates the influence of tannin type on the parameters evaluated.
Ruminococcus albus; microbial ecology; animal nutrition
A recuperação de RNA ribossômico (rRNA) intacto é necessária para a detecção de flutuações na população microbiana ruminal decorrentes dos taninos de forrageiras, utilizando-se sondas para 16S rRNA. O objetivo deste trabalho foi avaliar o efeito de polietilenoglicol (PEG) e polivinilpirrolidona (PVP) na extração de rRNA bacteriano, em presença de taninos de leguminosas forrageiras tropicais e de outras fontes, que possa ser hibridizado com sondas de oligonucleotídeos. Culturas de Ruminococcus albus 8 foram expostas ou não a 8 g/L de ácido tânico ou a 1 g/L de taninos condensados, extraídos de Acacia angustissima, casca de banana (Musa sp.), Desmodium ovalifolium, cascas de uvas vermelhas (Vitis vinifera) e Inga edulis. As culturas foram lavadas com tampão Tris pH 7 contendo 8% PEG ou 6% PVP antes do rompimento das células. Amostras de RNA total lavadas com PEG ou PVP migraram em géis de agarose. Bandas de 16S rRNA hibridizaram com uma sonda de oligonucleotídeos espécieespecífica para R. albus, independentemente da fonte de tanino. Comparou-se o efeito dos tampões de lavagem sobre a densidade das bandas de 16S rRNA, assim como sobre a intensidade de hibridização. Ocorreram efeitos significativos para fontes de taninos, tampões e para a interação entre taninos e tampões (P<0.01). A interação significativa indica a influência do tipo de tanino nos parâmetros avaliados.
Ruminococcus albus; ecologia microbiana; nutrição animal
MICROBIOLOGIA
Polyethylene glycol and polyvinylpirrolidone effect on bacterial rRNA extraction and hybridization from cells exposed to tannins1 1 Extracted from Ph.D. dissertation presented by the first author to the Dept. of Animal Science, Cornell University, Ithaca, NY, USA.
Efeito de polietilenoglicol e polivinilpirrolidona na extração e hibridização de rRNA bacteriano de células expostas a taninos
Pedro Braga ArcuriI; Michael Larry ThonneyII; Peter SchofieldII; Alice Nelson PellII
IEmbrapa-Centro Nacional de Pesquisa de Gado de Leite, Rua Eugênio do Nascimento, 610, CEP 36038-330 Juiz de Fora, MG. E-mail: pba1@cnpgl.embrapa.br
IICornell University, Dept. of Animal Sciences, Morrison Hall, Ithaca, NY, 14853-4801 USA. E-mail: ps24@cornell.edu, mlt2@cornell.edu, anp19@cornell.edu
ABSTRACT
In order to detect fluctuations in ruminal microbial populations due to forage tannins using 16S ribosomal RNA (rRNA) probes, recovery of intact rRNA is required. The objective of this work was to evaluate the effect of polyethylene glycol (PEG) and polyvinylpirrolidone (PVP) on extraction of bacterial rRNA, in the presence of tannins from tropical legume forages and other sources, that hybridize with oligonucleotide probes. Ruminococcus albus 8 cells were exposed to 8 g/L tannic acid or 1 g/L condensed tannins extracted from Acacia angustissima, banana (Musa sp.) skin, Desmodium ovalifolium, red grape (Vitis vinifera) skin and Inga edulis, or no tannins. Cells were rinsed with Tris buffer pH 7 containing either 8% PEG or 6% PVP prior to cell lysis. Total RNA samples rinsed with either PEG or PVP migrated through denaturing agarose gels. The 16S rRNA bands successfully hybridized with a R. albus species-specific oligonucleotide probe, regardless of tannin source. The effect of rinsing buffers on the density of 16S rRNA bands, as well as on the hybridization signals was compared. There were significant effects (P<0.01) when the controls were compared to either buffer treatments due to tannin type, buffer used and the interaction of tannin type and buffer. The significant interaction indicates the influence of tannin type on the parameters evaluated.
Index terms:Ruminococcus albus, microbial ecology, animal nutrition.
RESUMO
A recuperação de RNA ribossômico (rRNA) intacto é necessária para a detecção de flutuações na população microbiana ruminal decorrentes dos taninos de forrageiras, utilizando-se sondas para 16S rRNA. O objetivo deste trabalho foi avaliar o efeito de polietilenoglicol (PEG) e polivinilpirrolidona (PVP) na extração de rRNA bacteriano, em presença de taninos de leguminosas forrageiras tropicais e de outras fontes, que possa ser hibridizado com sondas de oligonucleotídeos. Culturas de Ruminococcus albus 8 foram expostas ou não a 8 g/L de ácido tânico ou a 1 g/L de taninos condensados, extraídos de Acacia angustissima, casca de banana (Musa sp.), Desmodium ovalifolium, cascas de uvas vermelhas (Vitis vinifera) e Inga edulis. As culturas foram lavadas com tampão Tris pH 7 contendo 8% PEG ou 6% PVP antes do rompimento das células. Amostras de RNA total lavadas com PEG ou PVP migraram em géis de agarose. Bandas de 16S rRNA hibridizaram com uma sonda de oligonucleotídeos espécieespecífica para R. albus, independentemente da fonte de tanino. Comparou-se o efeito dos tampões de lavagem sobre a densidade das bandas de 16S rRNA, assim como sobre a intensidade de hibridização. Ocorreram efeitos significativos para fontes de taninos, tampões e para a interação entre taninos e tampões (P<0.01). A interação significativa indica a influência do tipo de tanino nos parâmetros avaliados.
Termos para indexação:Ruminococcus albus, ecologia microbiana, nutrição animal.
Introduction
Tannins are polyphenolic polymers with high affinity for proteins and other biopolymers. Tannins provide important plant defense mechanisms against animals, insects and microbes. Bacteria may be inhibited by tannins that bind to extracellular polymers, causing substrate and ion deprivation and by inhibiting enzymes (Krause et al., 2001). Mechanisms used by different bacteria to tolerate tannins appear to differ, but these mechanisms are poorly understood (Jones et al., 1994; Nelson et al., 1997).
The study of ruminal microbial ecology of animals fed on tannin-rich diets is of fundamental importance for ruminants grazing tropical legumes. Tropical legumes contain more protein than grasses, but tannins and other secondary compounds restrict their use as forages. Tannin-containing forages may alter the efficiency of use and main site of digestion of proteins (Barry, 1989). The study of microbial ecology has been greatly enhanced by the development of molecular techniques. However, the presence of tannins or other polyphenols creates problems for nucleic acid extraction (Alm et al., 2000; Krause et al., 2001). Although there are several methods for isolating nucleic acids in the presence of polyphenols from soils containing humic acids (Moré et al., 1994; Ogram, 1998), most of the methods are related to extraction from plant material (Pich & Schubert, 1993; MacKenzie et al., 1997). Krause et al. (2001) looked at extraction methods using polyethylene glycol (PEG) and condensed tannin from commercial Quebracho (barks from Quercus sp.) at up to 8% of DM. Without PEG, the authors could only amplify nucleic acid sequences using the polymerase chain reaction (PCR) at 0.5% and 1% tannin, but with PEG they could amplify at all concentrations. They also did some tests of their extraction protocol on Calliandra sp., Acacia angustissima, A. boliviana, A. villosa, Leucaena pallida, L. diversifolia and L. leucocephala tannins, with similar results. However, most of these methods were developed for microbial detection, not enumeration, so that little emphasis was placed on the quantitative recovery of the nucleic acids. A common approach to extraction of nucleic acids from environments containing polyphenols is to add compounds with high affinity for the polyphenols to reduce formation of nucleic acid-polyphenol complexes (Loomis, 1974; Krause et al., 2001). Polyvinylpyrrolidone (PVP) (Molyneux, 1983) and PEG (Molyneux, 1984) are among the chemicals identified as potentially useful as tannin precipitants due to their high hydrogen bonding affinity, therefore their high affinity for polyphenols (Loomis, 1974). In addition, their low toxicity, water solubility and low cost make them preferred compounds for complexing polyphenols.
Nucleic acids can be extracted from fresh samples using a variety of protocols (Ausubel et al., 1996), most of which are based on the phenol-chloroform-alcohol (Stahl et al., 1988) or the guanidinium thiocyanate (Chomczynski & Sacchi, 1987) methods.
The objective of this work was to evaluate the effect of PEG and PVP on extraction of bacterial rRNA, in the presence of tannins from tropical legume forages and other sources, that could successfully hybridize with oligonucleotide probes.
Material and Methods
Culture and medium
Cultures of Ruminococcus albus 8 initially obtained from the University of Illinois, Urbana-Champaign culture collection, were grown in anaerobically prepared basal medium (Nelson et al., 1997), without the addition of resazurin. Basal medium contained ammonium as the only N source. The energy source used was 0.4% cellobiose (w/v). This medium was chosen to prevent protein precipitation due to tannins. Butyl rubber-stoppered serum bottles (150 mL) or 150x18 mm Balch tubes were used to grow 100 mL or 10 mL cultures at 39oC. After cultures had grown to the desired optical density, they were exposed to tannins. Microscopic observations to assess lysis and culture purity were performed from fresh mounts in a Nikon Labphoto optical microscope with a contrast phase objective.
Optical densities were measured at 600 nm in a Spectronic 601 spectrophotometer from 1 mL aliquots in quartz cuvettes (1cm length path) or through the 150x18 mm anaerobic culture tubes. Pure culture stocks were maintained in Rum 10 medium (Latham et al., 1978) where the energy source was approximately 0.2 g of acids-wollen cellulose.
Tannin solutions
Condensed tannins were added to yield 1 g/L culture medium (Nelson et al., 1997). Tannic acid was added at concentrations of 8 g/L culture medium. This was the highest concentration that a tannin-tolerant Streptococcus sp. could grow (Nelson et al., 1997). All tannin solutions were added into the medium using sterile syringes, from stock solutions prepared immediately before use.
Isolation of condensed tannins from plant samples
Samples of young leaves were collected from A. angustissima Mill. (Leguminosae) and Inga edulis Mart. (Leguminosae) from plants grown in a greenhouse at Cornell University, Ithaca, NY. Other samples were mature leaves of Desmodium ovalifolium Desv. (Leguminosae) harvested and freeze dried at the Escuela Agricola Panamericana (El Zamorano, Honduras), the skins of ripe red seedless grapes (Vitis vinifera L., Vitaceae) and the skins of unripe bananas (Musa paradisiaca L., Musaceae). Except for the D. ovalifolium sample, all other samples were frozen in liquid nitrogen and ground in a pre-chilled ceramic mortar and pestle. The fruit skins were peeled off and immediately placed in liquid nitrogen. All samples were lyophilized and stored in the dark at 4oC.
Tannins were extracted from plant samples using the procedure described by Hagerman (1988). Variable amounts of plant material (10 to 25 g) were suspended in 10 mL of 80% (v/v) aqueous ethanol per 1.0 g of sample dry matter, stirred for 30 min and stored overnight at 4oC. The suspension was filtered through Whatman filter paper no. 40 using slight vacuum. Condensed tannins were separated from low molecular weight tannins, other phenolics and pigments using 100 g Sephadex LH-20 dextran resin in 80% aqueous ethanol. After elution with 50% acetone, two extractions with ethyl acetate and lyophilization, the purified condensed tannin powders were stored at 4oC in the dark.
Spectrophotometric profile of tannin solutions
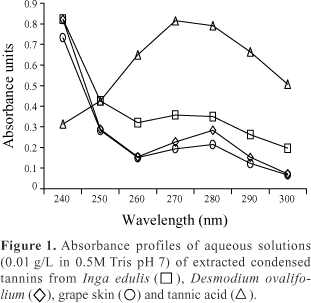
Standardized (1 mg/mL) aqueous solutions of the condensed tannins extracted from D. ovalifolium, grape skin and I. edulis as well as tannic acid were diluted (1:50) in water or 0.5M Tris pH 7.0 and their absorbance profile between 240 and 320 nm was recorded from a Beckman DU 500 spectrophotometer using a 100 mL quartz cuvette. The absorbance profile of the 0.5M Tris buffer was also measured.
Tanninbinding buffers
Polyvinylpyrrolidone-40,000 (PVP-40) and polyethylene glycol (PEG) were used to prepare 6% PVP (Loomis, 1974) and 8% (wt/vol) PEG (Makkar et al., 1995) solutions in 0.5M Tris buffer (pH 7.0).
Experimental procedure
All treatments were tested in triplicate, except for A. angustissima, where two and one replicates were treated with PEG and PVP buffers, respectively.
Tannin exposure
The absorbance (A600) of overnight cultures was measured. Aliquots in 10 mL volume were transferred into 50-mL polypropylene centrifuge flasks. Tannins were added to yield final concentrations to each aliquot of 8.0 g/L tannic acid or 1.0 g/L purified condensed tannin culture medium. The samples were incubated with tannins for 15 min at room temperature. Both positive and negative control were used. The positive control (T+B-) contained tannin and samples rinsed with Tris buffer without PEG or PVP. The negative control (T-B-) contained neither tannin nor PEG or PVP. The negative control was centrifuged once and kept on ice until all other samples were ready for total RNA extraction.
Rinsing with buffers
Tannin-removing buffers containing either 6% PVP or 8% PEG were added to the culture medium at the ratio of 3.5 mL buffer per 1 mL culture medium after the incubation with tannins. The suspension was then incubated for 15 min at room temperature and hand shaken twice. One drop of the suspension was removed for microscopic observation. Subsequently, cells were harvested by centrifugation at 2500 g (Beckman GS-6R bench centrifuge, swinging bucket rotor GH 3.8, for 30 min at room temperature. After the pH was recorded, the supernatant was discarded. A second volume of buffer then was added to the cell pellet in the ratio of 2 mL buffer per 1 mL culture. Pellets were resuspended by scraping the base of the centrifuge tube (back and forth) across the openings of a polypropylene microcentrifuge tube storage rack at least five times (Voo & Jacobsen, 1998). The suspensions were incubated and centrifuged again as described above. The pH of the supernatant was recorded. Cell pellets were then resuspended with repeated pipetting into a 1,000 µL guanidinium thiocyanate solution (lysing/binding solution, RNAqueous kit, Ambion Inc., Cupertino, CA). The suspension was transferred into 2 mL sterilized screwcap microcentrifuge tubes previously filled with 2.1 g of 0.1 mm zirconium beads.
Total RNA extraction
The recommendations for use of the RNAqueousÔ kit (Ambion Inc., Cupertino, CA) were followed. Cells in the presence of beads and lysing/binding solution were beaten for 7 min at maximum speed in a minibead beater 8 (Biospec, Bartlesville, OK) at 4oC (Raskin et al., 1997). Maximum lysate volumes were obtained by inserting the pipette tip deep into the bead layer and pipetting two or three times after the bulk volume had been transferred. The lysate was transferred into a disposable 15x75 mm borosilicate glass culture tube. The elution procedure was repeated three times and the three aliquots were saved altogether.
Quantification and quality assessment of RNA
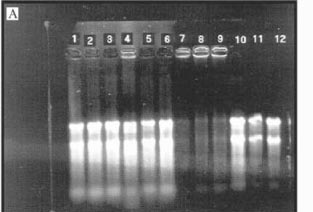
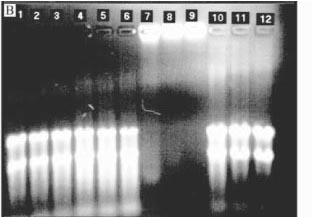
Total RNA concentrations were determined spectrophotometrically at 260 nm, assuming that 1 unit of absorbance at 260 nm = 40 µg RNA/mL (Ausubel et al., 1995). RNA solutions were stored at -70oC. The integrity of rRNA bands was assessed using electrophoresis with agarose denaturing gels (1% agarose (w/v) in formaldehyde, 10 x MOPS buffer), prepared according to Ausubel et al. (1996). Running conditions were 1 x MOPS buffer (pH 7) at 4.8 to 5.0 volts/cm for approximately 2 hours. Gels were photographed over UV light (254 nm) and digitally recorded using the AlphaImagerÔ imaging system with the IS-1000 software version 2.00 (Alpha Innotech Corp., San Leandro, CA).
The density of 16S rRNA bands was measured using the AlphaImager system. The integrated pixel value was obtained using the 1Dmulti toolbox from the IS-1000 software.
Probe hybridization
Oligonucleotide probe S-S-R.al-0196-a-A-18 (5'GTC ATG CGG CTT CGT TAT 3') (Odenyo et al., 1994) was labeled with digoxigenin-11-ddUTP using terminal transferase from the GeniusÔ #6 oligonucleotide tailing kit following the recommendations of the manufacturer. Total RNA samples (either 10 µg, 1 µg or 0.1 µg total RNA) were blotted onto a Magna Charge nylon membrane using a minifold II slot blot vacuum manifold. Membranes were UV-crosslinked in a Spectrolinker XL-1000 UV crosslinker, prehybridized for 2 hours and hybridized overnight at 42oC with 0.5 pmol probe/mL hybridization buffer (5 x sodium-saline-citrate solution, 50% deionized formamide, 0.1% Na-lauroylsarcosine, 0.02% SDS, 2% blocking reagent and 0.1 mg Poly (A)/mL). Stringent washes were performed twice with 2 X SSC solution at room temperature and twice with 0.5 X SSC solution at 42oC. For detection of hybridized 16S rRNA the GeniusÔ #7 luminescent detection kit containing the luminescent substrate disodium 3-(-4methoxyspiro{1,2-dioxetane-3,2'-(5'chloro) tricyclo [3.3.1.13,7]decan}-4-yl) phenyl phosphate (CSPD®) was used and followed the manufacturer's recommendations. Results were photographed onto X-ray film after incubation of membranes with substrate solution at 37oC and 45 minutes of film exposure. Hybridized bands were digitally recorded in the AlphaImagerÔ imaging system and their densities were measured.
Statistical analysis
Analyses of variance of the integrated pixel values obtained from densitometry of 16S rRNA bands and hybridization signals were performed. Factors tested were tannin type, rinsing buffer used and, for the hybridization signals, levels of RNA blotted onto the membranes. The twoway interactions among these factors were also tested. Data were analyzed with ANOVA, the general linear model and SNK test routines of the Minitab software release 11.21 (Minitab Inc. State College, PA).
Results and Discussion
The pH of the suspensions (i.e., culture medium mixed with tannins or rinsing buffers) ranged from 6.7 to 6.9 for all conditions tested. After centrifugation, there were differences in the type of cell pellet formed. Overall, pellets from PEG rinse were smaller but more difficult to resuspend than the pellets rinsed with PVP, when compared within the same tannin type. The linear structure of PEG (Molyneux, 1984) may explain the difference between buffers in the firmness of the pellets formed after rinsing and centrifugation.
Bead beating was performed as recommended by Raskin et al. (1997), except when 2 g of 0.1 mm zirconium beads were used. Microscopic observations performed with the lysate showed that most cells were lysed, although in every field from any slide, 1 or 2 intact cells/field where visible. The RNA extraction method using guanidinium thiocyanate and silica has been used to extract RNA efficiently from several systems, including nucleic acids from fecal virus (Hale et al., 1996).
Average total RNA yields calculated from A260 readings of the negative (T-B-) controls varied from 10.9 to 27.8 µg RNA/mL culture. Because the aqueous condensed tannin solutions absorb light at 270 nm (Waterman & Mole, 1994), the A260:A280 ratio cannot be used to assess RNA purity from cultures with tannins (Figure 1). Total RNA quality expressed as A260:A280 ratios ranged from 2.1 to 2.2 for T-B-. Tannins also interfered with A320 readings, often used to indicate the presence of light-scattering matter (Ausubel et al., 1996).
The RNA in the T+B- lanes of gels with D. ovalifolium (Figure 2a) and grape skin tannin (Figure 2b) did not migrate, unlike RNA from the T+B- treatment for the other tannin sources (Figure 2c). The RNA could not migrate due to complexation with condensed tannins. RNA could not migrate in the presence of D. ovalifolium possibly because this tannin has high biological activity and is a large polymer (Nelson et al., 1997). The complexation is possibly due to the residual negative charge of nucleic acids (Ausubel et al., 1996) and the ability of tannins to bind using hydrogen bonds from hydroxyls of the phenolic residue (Loomis, 1974; Dabo et al., 1993). Most of the tanninprotein complexes are probably denatured by guanidinium thiocyanate, since proteins are rapidly denatured in concentrated (4M) guanidinium solutions (Chomczynski & Sacchi, 1987). The tanninRNA complexes probably were retained in the glass fiber filters and eluted later on.
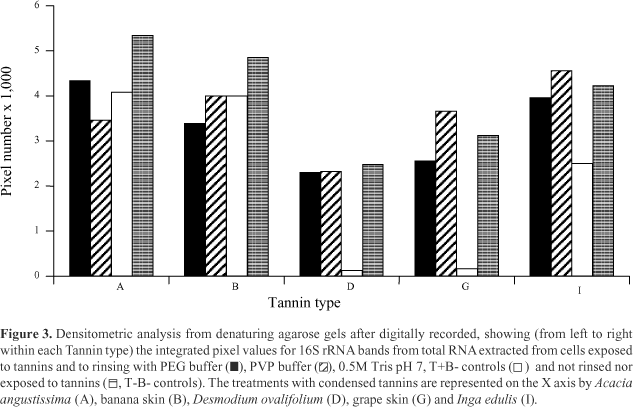
Analysis of variance indicated that band density means were similar for both buffers tested (PVP = 3593.8 pixels; PEG = 3223.5 pixels, P = 0,05). However, band density means differed due to tannin type and rinsing buffer (Table1). In other words, the effect of rinsing buffer on band density was affected by tannin type. For grape skin, the results were similar because its mean for the PVP treatment was close (P = 0.0574) to the significance level. The two buffers had similar effects and yielded less dense bands than the negative control (T+B+ ver- sus T-B-) (Figure 3).
To evaluate if RNA extracted from cells exposed to the different conditions tested could be used in molecular analysis, aliquots of RNA from each of the treatments with the R. albus 8 species-specific RAL 196 probe were hybridized. The species-specific probe hybridized to all samples of RNA from cells exposed to tannins and treated with either PEG or PVP similarly (T+B+). The results showed less intense hybridization signals compared to the negative control (T-B-), as discussed below. Desmodium ovalifolium and I. edulis leaf tannins were the most and the least effective tannins tested (Figure 4). Grape skin results were similar to those of D. ovalifolium leaves, whereas banana skin and A. angustissima leaves treatments hybridized similar to I. eduli leaves. The RNA of cells exposed to either D. ovalifolium leaf or grape skin tannin, but not exposed to PVP or PEG (T+B- controls), did not hybridize with the probe (Figure 4). Three quantities, 0.1, 1 and 10 µg of total RNA per slot, were blotted. The significance of tannin effect over integrated pixel numbers had to be interpreted as confounded with total RNA extraction, membrane and hybridization conditions due to the experimental design adopted (samples exposed to each tannin were blotted and hybridized once). Considering that the membranes used were cut from adjacent regions of the same lot, that the solutions used for hybridization and washing steps were the same as well, and that hybridization and washings were done simultaneously, it is likely that the primary source of variation was the presence of tannin during RNA extraction. The effects of tannin type, buffer and level of total RNA applied were highly significant (P<0.01). Tannin by buffer interaction was significant (P<0.05). The significant tannin by buffer interaction that was evident from the analysis of the hybridized membrane data indicates the appropriateness of using buffers with PVP or PEG to extract RNA in the presence of tannins. There was a higher hybridization signal for the I. edulis leaf and grape skin tannins rinsed with PVP than for the T-B- control (Figure 3). This may be caused by a complex formation among tannins, probe and the proteinlike structure of residual PVP molecules eventually attached to the nylon membranes causing nonspecific binding of the probe (Raskin et al., 1996). The interaction between tannin type and RNA factors (P = 0.038) may indicate that the condensed tannins could be causing differences in integrated pixel values observed across different levels of total RNA applied to the membranes. This effect was probably caused by saturation of the nylon membrane as discussed by Raskin et al. (1996).
The interaction between buffer and RNA level was not significant (P = 0.44) indicating that the treatment with buffers did not alter the effect of level of total RNA blotted.
Conclusions
1. Tannins from different sources must be removed during RNA extraction for hybridization purposes.
2. Rinsing cells with 0,5M Tris buffers containing either 6% PVP or 8% PEG before RNA extraction significantly reduces tannin effects on gel migration and species-specific oligonucleotide hybridization with total RNA samples.
3. Quantitative protocols are possible using total RNA from cells exposed to different sources of tannin, due to the significant effect of increasing amounts of RNA blotted to hybridization signals.
Accepted for publication on June 20, 2003
- ALM, E. W.; ZHENG, D.; RASKIN, L. The presence of humic substances and DNA in RNA extracts affects hybridization results. Applied and Environmental Microbiology, Washington, v. 66, n. 10, p. 4547-4554, 2000.
- AUSUBEL, F. M.; BRENT, R.; KINGSTON, R. E.; MOORE, D. D.; SEIDMAN, J. G.; SMITH, J. A.; STUHL, K. Current protocols in molecular biology New York: J. Wiley, 1996. 606 p.
- AUSUBEL, F. M.; BRENT, R.; KINGSTON, R. E.; MOORE, D. D.; SEIDMAN, J. G.; SMITH, J. A.; STRUHL, K. (Ed.). Short protocols in molecular biology 3rd ed. New York: J. Wiley, 1995. 729 p.
- BARRY, T. N. Condensed tannins: their role in ruminant protein and carbohydrate digestion and possible effects upon the rumen ecosystem. In: NOLAN, J. V.; LENG, R. A.; DEMEYER, D. I. (Ed.). The roles of protozoa and fungi in ruminant digestion Armidale: Penambul Books, 1989. p. 153-169.
- CHOMCZNSKI, P.; SACCHI, N. Single-step method of RNA isolation by acid guanidinium thiocyanate-phenol-chloroform extraction. Analytical Biochemistry, San Diego, v. 162, p. 156-159, 1987.
- DABO, S. M.; MITCHELL, E. D.; MELCHER, U. A method for the isolation of nuclear DNA from cotton (Gossipium) leaves. Analytical Biochemistry, San Diego, v. 210, n. 1, p. 34-38, 1993.
- HAGERMAN, A. E. Extraction of tannins from fresh and preserved leaves. Journal of Chemical Ecology, New York, v. 14, n. 2, p. 453-461, 1988.
- HALE, A. D.; GREEN, J.; BROWN, D. W. G. Comparison of four RNA extraction methods for the detection of small round structured viruses in faecal specimens. Journal of Virological Methods, Amsterdam, v. 57, n. 2, p. 195-201, 1996.
- JONES, G. A.; MCALLISTER, T. A.; MUIR, A. D.; CHENG, K. -J. Effects of sainfoin (Onobrychis viciifolia Scop.) condensed tannins on growth and proteolysis by four strains of ruminal bacteria. Applied and Environmental Microbiology, Washington, v. 60, n. 4, p. 1374 - 1378, 1994.
- KRAUSE, D. O.; SMITH, W. J.; MCSWEENEY, C. S. Extraction of microbial DNA from rumen contents containing plant tannins. BioTechniques, Natick, v. 31, p. 294-298, 2001.
- LATHAM, M. J.; BROOKER, G. L.; PETTIPHER, G. L.; HARRIS, P. J. Ruminococcus flavefaciens cell coat and adhesion to cotton cellulose and to cell walls in leaves of perennial ryegrass (Lolium perenne). Applied and Environmental Microbiology, Washington, v. 35, p. 156-165, 1978.
- LOOMIS, W. D. Overcoming problems of phenolics and quinones in the isolation of plant enzymes and organelles. Methods in Enzymology, San Diego, v. 31, p. 528-544, 1974.
- MacKENZIE, D. J.; McLEAN, M. A.; MUKERJI, S.; GREEN, M. Improved RNA extraction from woody plants for the detection of viral pathogens by reverse transcription-polymerase chain reaction. Plant Disease, St. Paul, v. 81, n. 2, p. 222-226, 1997.
- MAKKAR, H. P. S.; BLUMMEL, M.; BECKER, K. Formation of complexes between polyvinyl pyrrolidones or polyethylene glycols and tannins, and their implication in gas production and true digestibility in in vitro techniques. British Journal of Nutrition, Wallingford, v. 73, p. 897-913, 1995.
- MOLYNEUX, P. Nonionic polymers: polyoxides, polyethers, and poly(ethylene Imine) In: Water-soluble synthetic polymers: properties and behavior. Boca Raton: CRC Press, 1984. v. 1, p. 19-74.
- MOLYNEUX, P. Nonionic polymers: the vinyl group. In: Water-soluble synthetic polymers: properties and behavior. Boca Raton: CRC Press, 1983. v. 1, p. 119-193.
- MORÉ, M. I.; HERRICK, J. B.; SILVA, M. C.; GHIORSE, W. C.; MADSEN, E. L. Quantitative cell lysis of indigenous microorganisms and rapid extraction of microbial DNA from sediment. Applied and Environmental Microbiology, Washington, v. 60, n. 5, p. 1572-1580, 1994.
- NELSON, K. E.; PELL, A. N.; DOANE, P. H.; GINER-CHAVEZ, B. I.; SCHOFIELD, P. Chemical and biological assays to evaluate bacterial inhibition by tannins. Journal of Chemical Ecology, New York, v. 23, n. 4, p. 1175-1193, 1997.
- ODENYO, A. A.; MacKIE, R. I.; STAHL, D. A.; WHITE, B. A. The use of 16S rRNA-targeted oligonucleotide probes to study competition between ruminal fibrolytic bacteria: development of probes for Ruminococcus species and evidence for bacteriocin production. Applied and Environmental Microbiology, Washington, v. 60, n. 10, p. 3688-3696, 1994.
- OGRAM, A. Isolation of nucleic acids from environmental samples. In: BURLAGE, R. S.; ATLAS, R.; STAHL, D.; GEESEY, G.; SAYLER, G. (Ed.). Techniques in microbial ecology New York: Oxford University Press, 1998. p. 273-288.
- PICH, U.; SCHUBERT, I. Midiprep method for isolation of DNA from plants with a high content of polyphenolics. Nucleic Acids Research, Oxford, v. 21, n. 14, p. 3328, 1993.
- RASKIN, L.; CAPMAN, W. C.; KANE, M. D.; RITTMANN, B. E.; STAHL, D. A. Critical evaluation of membrane supports for use in quantitative hybridizations. Applied and Environmental Microbiology, Washington, v. 62, n. 1, p. 300-303, 1996.
- RASKIN, L.; CAPMAN, W. C.; SHARP, R.; POULSEN, L. K.; STAHL, D. A. Molecular ecology of gastrointestinal ecosystems. In: MacKIE, R. I.; WHITE, B. A.; ISAACSON, R. E. (Ed.). Gastrointestinal microbiology: gastrointestinal microbes and host interactions. New York: Chapman & Hall / International Thomson, 1997. p. 243-298. (Chapman & Hall Microbiology Series, 2).
- STAHL, D. A.; FLESHER, B.; MANSFIELD, H. R.; MONTGOMERY, L. Use of philogenetically based hybridization probes for studies of ruminal microbial ecology. Applied and Environmental Microbiology, Washington, v. 54, n. 5, p. 1079-1084, 1988.
- VOO, K. S.; JACOBSEN, B. M. Rapid resuspension of pelleted bacterial cells for miniprep plasmid DNA isolation. BioTechniques, Natick, v. 24, p. 240-243, 1998.
- WATERMAN, P. G.; MOLE, S. Analysis of phenolic plant metabolites London: Blackwell Scientific, 1994. 238 p.
Publication Dates
-
Publication in this collection
16 Feb 2004 -
Date of issue
Sept 2003
History
-
Accepted
20 June 2003