Abstracts
This study aimed to evaluate the growth characteristics of irrigated Vitória pineapple plants grown in semi-arid conditions and determine its developmental stages based on those characteristics. It was used a randomized block design with four replicates. The experimental treatments were: plant harvest at 270, 330, 390, 450, 510, 570, 690, 750, and 810 days after planting (DAP). The following variables were determined: plant height, stem diameter, D-leaf length, D-leaf fresh and dry mass, biomass production of plants and plant parts (organs), and vegetative biomass. Five phenological stages are proposed based on vegetative biomass production: < 20% biomass production (V1); 21-40% (V2); 41-60% (V3); 61-80% (V4); and > 80% (V5). The maximum growth rate for plant height, D-leaf length, and stem diameter was observed at the end of the phenological stage V1 (390-411 DAP), and at the end of stage V5 these plant traits had average values of 106, 82, and 7 cm, respectively. The maximum biomass accumulation rates were observed at stages V4 and V5, resulting in a final fruit yield and total fresh biomass of 72 t ha-1 and 326 t ha-1, respectively. Finally, we estimated that 80% of the accumulated biomass may remain in the field after fruit and slip harvest, and could be incorporated as plant residue into the soil.
Ananas comosus var. comosus; biomass production; maximum accumulation rate
O estudo avaliou o crescimento do abacaxizeiro 'Vitória' irrigado em condições do semiárido brasileiro e definiu etapas de desenvolvimento por meio de características fenotípicas e morfológicas. O delineamento estatístico utilizado foi em blocos casualizados, com quatro repetições. Os tratamentos foram definidos por nove épocas de amostragem de plantas e seus componentes (270; 330; 390; 450; 510; 570; 690; 750 e 810 dias após o plantio - DAP). Foi avaliada a altura da planta, diâmetro do talo, comprimento e peso fresco e seco de folha 'D', a produção de biomassa total e dos componentes: folha, talo, raiz, fruto, coroa, filhotes e biomassa vegetativa, (folhas, talo e raiz). Por meio da produção relativa de biomassa vegetativa foram propostos cinco estádios fenológicos de crescimento do abacaxizeiro: até 20% de produção de biomassa-V1; 21-40%-V2; 41-60%-V3; 61-80%-V4; acima de 80%-V5. As características altura da planta, comprimento da folha 'D' e diâmetro do talo atingiram taxas de máximo crescimento no final do estágio fenológico V1 (390 e 411 DAP), e a estabilização destas características ocorreu no final do estágio V5, com valores médios de 106 cm, 82 cm e 7 cm, respectivamente. As taxas de máximo acúmulo foram observadas nos estádios V4 e V5, implicando a produção final de 72 t ha-1 de frutos e 326 t ha-1 de biomassa da planta (matéria fresca); desta última, estimou-se que 80% da biomassa acumulada pode permanecer na área de cultivo, após a colheita de frutos e mudas, como fonte de resíduos vegetais para o solo.
Ananas comosus var. comosus; produção de biomassa; taxa de máximo acúmulo
SOILS AND NUTRITION
Growth and production of irrigated vitória pineapple grown in semi-arid conditions1 1 (Trabalho 265-13)
Crescimento e produção do abacaxizeiro 'vitória' irrigado em condições do semiárido
Rodinei Facco PegoraroI; Bruna Aparecida Madureira de SouzaII; Victor Martins MaiaI; Uirá do AmaralIII; Marlon Cristian Toledo PereiraII
IAgronomist, Professor Doctor, Universidade Estadual de Montes Claros (Montes Claros State University). CEP 39440-000. Janaúba-MG, Brazil. Scholarship holder of productivity in research from the FAPEMIG. E-mail: rodinei.pegoraro@unimontes.br; victor.maia@unimontes.br; marlon.pereira@unimontes.br
IIAgronomist, Doctoral student in Agronomy, Universidade Estadual do Sudoeste da Bahia. CEP 45083-900. Vitória da Conquista-BA, Brazil. E-mail: bruna_madureirasouza@yahoo.com.br
IIIAgronomist, Doctoral student in Agronomy, Universidade Federal do Rio de Janeiro. CEP 23890-000. Seropédica-RJ, Brazil. E-mail: uiraagro@gmail.com
ABSTRACT
This study aimed to evaluate the growth characteristics of irrigated Vitória pineapple plants grown in semi-arid conditions and determine its developmental stages based on those characteristics. It was used a randomized block design with four replicates. The experimental treatments were: plant harvest at 270, 330, 390, 450, 510, 570, 690, 750, and 810 days after planting (DAP). The following variables were determined: plant height, stem diameter, D-leaf length, D-leaf fresh and dry mass, biomass production of plants and plant parts (organs), and vegetative biomass. Five phenological stages are proposed based on vegetative biomass production: < 20% biomass production (V1); 21-40% (V2); 41-60% (V3); 61-80% (V4); and > 80% (V5). The maximum growth rate for plant height, D-leaf length, and stem diameter was observed at the end of the phenological stage V1 (390-411 DAP), and at the end of stage V5 these plant traits had average values of 106, 82, and 7 cm, respectively. The maximum biomass accumulation rates were observed at stages V4 and V5, resulting in a final fruit yield and total fresh biomass of 72 t ha-1 and 326 t ha-1, respectively. Finally, we estimated that 80% of the accumulated biomass may remain in the field after fruit and slip harvest, and could be incorporated as plant residue into the soil.
Index terms: Ananas comosus var. comosus, biomass production, maximum accumulation rate.
RESUMO
O estudo avaliou o crescimento do abacaxizeiro 'Vitória' irrigado em condições do semiárido brasileiro e definiu etapas de desenvolvimento por meio de características fenotípicas e morfológicas. O delineamento estatístico utilizado foi em blocos casualizados, com quatro repetições. Os tratamentos foram definidos por nove épocas de amostragem de plantas e seus componentes (270; 330; 390; 450; 510; 570; 690; 750 e 810 dias após o plantio - DAP). Foi avaliada a altura da planta, diâmetro do talo, comprimento e peso fresco e seco de folha 'D', a produção de biomassa total e dos componentes: folha, talo, raiz, fruto, coroa, filhotes e biomassa vegetativa, (folhas, talo e raiz). Por meio da produção relativa de biomassa vegetativa foram propostos cinco estádios fenológicos de crescimento do abacaxizeiro: até 20% de produção de biomassa-V1; 21-40%-V2; 41-60%-V3; 61-80%-V4; acima de 80%-V5. As características altura da planta, comprimento da folha 'D' e diâmetro do talo atingiram taxas de máximo crescimento no final do estágio fenológico V1 (390 e 411 DAP), e a estabilização destas características ocorreu no final do estágio V5, com valores médios de 106 cm, 82 cm e 7 cm, respectivamente. As taxas de máximo acúmulo foram observadas nos estádios V4 e V5, implicando a produção final de 72 t ha-1 de frutos e 326 t ha-1 de biomassa da planta (matéria fresca); desta última, estimou-se que 80% da biomassa acumulada pode permanecer na área de cultivo, após a colheita de frutos e mudas, como fonte de resíduos vegetais para o solo.
Termos para indexação: Ananas comosus var. comosus, produção de biomassa, taxa de máximo acúmulo.
INTRODUCTION
Pineapple is one of the most cultivated tropical fruits worldwide, and Brazil is one of the largest global pineapple producers. In Brazil, pineapple finds favorable conditions for growth and production and is cultivated commercially in almost all Brazilian states (IBGE, 2012).
The Vitoria cultivar has agronomic characteristics that are similar or superior to the traditional cvs. Pérola and Smooth Cayenne, but unlike these, is not susceptible to fusariosis (Fusarium guttiforme). In addition, Vitoria plants may reach the same size as Pérola plants. The smooth leaves of Vitoria plants represent another advantage over the other two cultivars, because they facilitate the implementation of cultural practices similar to those used in Pérola and Smooth Cayenne plants (VENTURA et al., 2009).
The growth curves of Vitoria plants and their components (leaves, stems, roots, fruits, slips and suckers) positively reflect the adoption of agronomic practices that enable the adequate use of water and fertilizers to maximize productivity and rational use of pesticides and herbicides. Monitoring pineapple growth is crucial for crop management because it enables the identification of problems during the growth cycle such as water and nutritional deficits, weed competition, and compacted or poorly drained soils; and may also help determine the optimal time for adopting practices to efficiently maximize plant growth and fruit production (OMOTOSO and AKINRINDE 2007; SOUZA et al., 2007; FOUNIER et al., 2010).
Growth analysis has been used in an attempt to explain differences in plant growth between cultivars, which may be caused by genetic factors and/or changes in the environment. Growth analysis also represents an efficient tool for identifying promising cultivars, helping plant breeding studies search for more productive materials (HANAFI et al., 2009; FOURNIER et al., 2010; LIMA et al., 2011). Moreover, plant growth curves can be used to evaluate the contribution of different physiological processes and determine photosynthetic activity, which is indirectly related to plant growth variables such as absolute growth rate, net assimilation rate, and culture growth rate (LIMA et al., 2007; GRACIANO et al., 2011; LIMA et al., 2011).
Knowledge of the growth characteristics of new pineapple cultivars is useful for crop management, especially for irrigated plants. This study aimed to determine the growth behavior of irrigated Vitoria pineapple plants grown in semi-arid conditions.
MATERIAL AND METHODS
The study was conducted under field conditions in Janaúba, in the state of Minas Gerais, Brazil (15º43'47.4" S and 43º19'22.1" W, 516 m a.s.l.), between July 2009 and October 2011. The climate in the region is Aw (hot summers and cold, dry winters) according to the Koppen classification system. The annual average precipitation in the area is approximately 870 mm, the average annual temperature is 24 ºC, whereas the annual average insolation and relative humidity are 2,700 hours and 65%, respectively. Maximum and minimum temperatures and monthly precipitation data during the experimental period are shown in Figure 1 (INMET, 2012). For this study, we used micropropagated cv. Vitória plantlets. Vitória is a new pineapple cultivar produced by Embrapa (Brazilian Agricultural Research Corporation) by crossing Primavera and Smooth Cayenne cultivars and released by Incaper (Capixaba Research, Technical Assistance, and Rural Extension Institute).
It was used a randomized block design with four replicates. The treatments were represented by evaluation dates, which were measured in days after planting (DAP). The treatments studied were: plant harvest at 270, 330, 390, 450, 510, 570, 690, 750, and 810 DAP. The first harvest was late due to the slow initial growth of plantlets produced by tissue culture, which had low initial fresh weight and small size (10-20 cm).
Soil preparation consisted of leveling, grading, and furrowing the area. Twenty simple soil samples from the 0-20 cm of soil layer were bulked into a composite sample to determine the physicochemical characteristics of the soil in the cultivation area (Table 1). The soil had low phosphorus content, according to the Minas Gerais Soil Fertility Committee guidelines (RIBEIRO et al., 1999). Therefore, 16.7 g plant-1 of single superphosphate fertilizer was applied in the field furrow. In addition, we applied 500 kg ha"1 of calcium and magnesium silicate to increase Ca and Mg availability.
The seedlings were planted in furrows using the double row system and spaced 0.90 x 0.40 x 0.30 m, resulting in a density of 51,280 plants ha-1. Each plot was composed of three double rows with 10 plants per row, totaling 60 plants and 20 useful plants per plot. Side-dressing was done six times, in January, May, July, September, and November 2010, and February 2011, before the onset of flower induction (April). Fifteen g of N and K2O were applied per plant, using urea as an N source and potassium chloride as a K source. Foliar fertilization was also applied, using boric acid (0.07%), zinc sulfate (0.1%), and copper sulfate (0.1%) solutions during the same period as N and K fertilization.
Weed control was done by hand during the first twelve months. After that period, we used chemical weed control until the flower induction stage, using Gesapax 500-Ametrina (20 mL L-1 of water) and Verdict R-Triflurometil (5 mL L-1 of water) to control broad and narrow leaf weeds, respectively. After the flower induction stage, weed control was done using the hand-weeding technique. In addition, we used Abamectin Nortox (3 mL L-1 of water) for mite control.
We used conventional sprinkler irrigation with fixed Agropolo sprinklers (model NY 25) with a nominal flow rate of 500 L h-1. Sprinklers were spaced 12 m apart along the lateral transect line, and lines were spaced 12 m apart. Irrigation was performed three times per week, with intervals between irrigations depending on precipitation levels and water demand at different growth phases.
Flower induction was done 630 days after planting using 50 mL of 1% Ethrel® solution plus calcium hydroxide (lime) in a dose of 0.35 g L-1 of water (REINHARDT et al., 2001). Irrigation was suspended 24 h before flower induction to achieve greater efficiency and uniform blooming. Fruits were protected using newspaper 60 days after induction to reduce burning by solar radiation. Fruits were harvested when at least 50% of the skin was yellow.
The plants collected at each sampling time were taken to the Soil Laboratory at Montes Claros State University (Unimontes). Plants were cleaned to remove soil debris and separated into roots, stems, leaves, the highest plant leaf (D-leaf), fruit, crown, and seedlings. Next, plant parts were weighed to determine fresh matter and oven dried under forced ventilation (65 ºC) for approximately 72 h until reaching a constant mass. The dry biomass of plant components and the total biomass were calculated and the fresh and dry matter of each plant compartment was converted to t ha-1. We also calculated the vegetative biomass, which corresponds to the average fresh and dry matter of leaves, roots, and stems.
The following plant characteristics were evaluated at each sampling time: plant height, stem diameter, D-leaf length, fresh and dry matter mass of plant components (leaves, stems, roots, fruit, crown, and slips), vegetative biomass (leaves, stems, and roots), total biomass (sum of all plant components), and fruit production (t ha-1).
The data were submitted to an analysis of variance (ANOVA). The effect of the number of days after planting on plant growth was analyzed using regression analysis when the source of variation was significant (p < 0.05), and the proper model was selected to represent the variables. All analyses were performed using the Sisvar® software (FERREIRA, 2000).
Model selection was based on goodnessof-fit (percentage of explained variance, R2) and best representation of the biological phenomenon. Sigmoid, non-linear regression models with three parameters were used to represent growth characteristics (plant height, stem diameter, D-leaf length, vegetative biomass, and total biomass) and pineapple components (leaves, stem, and D-leaf), according to the equation below:
where: a = the maximum accumulation value;
x0 = the x value (DAP) used to determine the maximum nutrient accumulation; and
b = the range of x values (in DAP) between the inflection point and the maximum point.
Thus, the inflection point (IP) of the curve could be accurately determined from the adjusted model, as follows:
IP = xo - b
Mathematically, the IP corresponds to the x value at which the slope of the adjusted model changes its sign; in practical terms, it corresponds to the x value (in DAP) at which the daily accumulation rate, even if positive, starts decreasing.
A Gaussian model with three parameters was selected to represent root biomass production, according to the equation below:
where: a = the maximum accumulation value;
x0 = the x value (in DAP) at which the curve inflection begins (inflection point, at which the maximum accumulation rate of nutrient dry matter is observed); and
b = the range of x values (in DAP) at which the highest variation in y (growth or fresh and dry matter accumulation) occurs.
The adjusted models were used to estimate the maximum accumulation rate (TMA) in kg ha-1 day-1, which corresponded to the beginning of the inflection curve of biomass production in the plant components in relation to DAP. The Pearson's correlation coefficient was used to determine the relationship between plant growth characteristics and fresh and dry biomass accumulation with all pineapple components.
RESULTS AND DISCUSSION
The variables plant height, D-leaf length, D-leaf mass, stem diameter, fresh and dry matter of plant components (leaves, stems, roots, fruit, crown, and slips), and total biomass were significantly affected by cultivation time (ANOVA, p < 0.05).
A sigmoid, non-linear regression model was fitted to most pineapple growth characteristics, which best represented the different growth stages. According to Reinhardt et al. (2000), the pineapple growth cycle is divided into vegetative, reproductive, and propagative stages. At the beginning of the growth cycle, plants are in the vegetative stage, which extends until flower induction; plants are small and growth is slow. A few days after planting, metabolic activity increases with the increasing number of leaves and larger stem diameter, resulting in higher plant growth, which stabilizes after the flower induction phase at the beginning of the reproductive stage. Fruit formation occurs at this stage and nutritional demand increases, with higher amounts of nutrients allocated to the fruit, crown, and slips until fruit harvest. At the propagative stage, the nutritional requirement for seedling production is minimal. However, the growth cycle may start again with the development of ratoon suckers and the propagative stage (slip and sucker formation) may occur simultaneously with the reproductive stage.
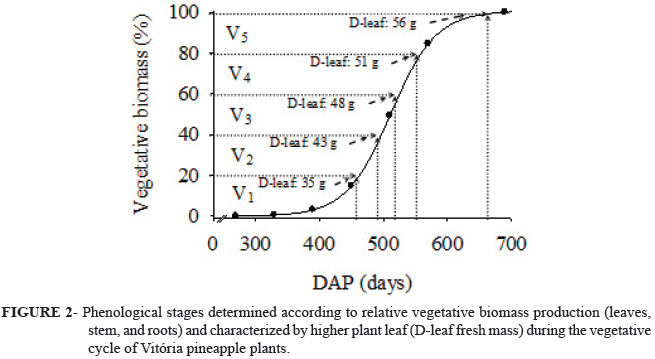
We propose that the vegetative stage be divided into five phenological stages characterized by D-leaf growth (D-leaf fresh mass) and relative vegetative biomass production for efficient water and fertilizer use (Fig. 2). The first vegetative stage (V1), at the beginning of cultivation, is characterized by up to 35 g of D-leaf fresh mass, which represents an estimated 20% of the vegetative biomass. Stage V1 was long, lasting 463 days from planting to production of 20% vegetative biomass. Thus, crop management should be based on plant growth rather than cultivation time because of variations in seedling type and size, cultivar, and cultivation region.
The second vegetative phenological stage (V2) occurred between 463 and 493 DAP (V2) (Fig. 2). Plant growth was high at this stage (V2), with an increase in vegetative biomass production of 21 to 40% and in D-leaf mass of 35.1 to 43 g. At the third phenological stage (V3), between 493 and 526 DAP, vegetative biomass increased from 41 to 60% and D-leaf mass ranged from 43.1 to 48 g. At the fourth phenological stage (V4), between 526 and 560 DAP, vegetative biomass increased from 61 to 80% and D-leaf mass ranged from 48.1 to 51 g, whereas at the fifth stage (V5), between 560 and 691 DAP, fresh biomass production and D-leaf mass were greater than 80% and 51 g, respectively. Pineapple flower induction occurred at the end of stage V5, simultaneously with the onset of the reproductive stage. It should be noted that determining the proper time for pineapple flower induction depends on phytomass production during the vegetative stage, because of the direct relationship between plant mass and fruit mass. In this study, a D-leaf fresh matter of approximately 60 g resulted in an average fruit mass of 1.3-1.4 kg (data not shown).
Plant growth was slow during the initial cultivation stage (V1) (Fig. 3), possibly due to the small size of the seedlings used in the study. After the initial growth period, plant growth characteristics (plant height, stem diameter, D-leaf length and mass) and fresh and dry biomass production of different plant components (leaves, stem, root) increased significantly, followed by the flower induction process at the end of stage V5 (approximately 630 DAP), with a subsequent stabilization in vegetative growth. These results are in accordance with the findings by Marques et al. (2011), who reported that the vegetative growth of pineapple plants is drastically reduced after flowering/fructification when the plant's metabolism is directed to fruit development. These observations are common knowledge and serve as a basis for determining the phenological stages of pineapple plants (FOURNIER et al., 2010; RODRIGUES et al., 2010; SAMPAIO et al., 2011).
The maximum daily growth rate for plant height was 2.4 mm day-1 and was observed at vegetative stage V1 (388 DAP) (Table 2). However, maximum plant height (106 cm) was observed only at the end of stage V5. According to Souza et al. (2007), an adult plant growing under ideal climate conditions and no water deficit reaches a height of approximately 100-120 cm, whereas a maximum height of 133 and 100 cm has been observed for Pérola and Smooth Cayenne cultivars, respectively, 12 months after planting (Rodrigues et al., 2010).
The maximum D-leaf growth rate (0.16 cm day-1) and maximum D-leaf length (82 cm) were observed at the end of stages V1 (342 DAP) and V5 (630 DAP), respectively (Fig. 3). A similar result for D-leaf length (81.4 cm) of Vitória plants grown with 600 kg ha-1 of N was observed 420 DAP by Silva et al. (2012) in the state of Paraíba, Brazil. However, a shorter D-leaf length was observed by Cabral and Matos (2009) and Ramos et al. (2013) (68 and 66 cm, respectively) for the Imperial cultivar, and by Guarçoni and Ventura (2011), who harvested heavier fruits when D-leaf reached a minimum length of 75.5 cm at flower induction.
The maximum D-leaf fresh mass (55.77 g) was also recorded at the end of stage V5 (600 DAP) (Fig. 3 and Table 2). This figure is lower than the values recorded for Gold, Jupi, Smooth Cayenne, Imperial, and Gomo de Mel cultivars, which reached 75.75, 93.75, 70.25, 63.25, and 58.75 g, respectively (SAMPAIO et al., 2011).
D-leaf length and mass are important parameters used to estimate crop yield before natural induction, enabling greater control of induction time and resulting in heavier fruits. According to Py et al. (1987), D-leaf must reach a minimum length of 80 cm and 70 g of fresh mass before flower induction for production of 1.5 kg of Smooth Cayenne pineapple. However, a D-leaf mass of 78, 75, and 70 g for MD-2, Smooth Cayenne, and Flhoran 41 cultivars, respectively, resulted in 2-kg fruits in the Ivory Coast (FOURNIER et al., 2010). Thus, the growth indicator (D-leaf mass) commonly used by growers to determine floral induction time varies among cultivars.
The lower D-leaf mass of Vitória plants at the time of flower induction may be explained by the slow initial growth of seedlings produced by tissue culture, resulting in a longer cultivation cycle. Regardless of planting material origin, vegetative and reproductive performance of large slips is higher than that of small slips (REINHARDT et al., 2003), and large slips and suckers have higher yields (OMOTOSO and AKINRINDE, 2007). Thus, the larger leaf area and greater nutritional storage volume of large plantlets result in greater biological production and fresh and dry mass accumulation compared to small slips or plantlets (REINHARDT et al., 2003).
The maximum daily accumulation rate for D-leaf fresh and dry mass was 0.28 g day-1 (437 DAP) and 0.04 g day-1 (451 DAP), respectively (Table 2). The cultivation time (in DAP) to achieve the maximum accumulation rate indicates the phase of accelerated daily biomass accumulation by plant components, suggesting a high demand for natural resources such as water and nutrients by the plants. In the Vitoria plants analyzed in this study, this period seems to have occurred between the end of phenological stage V1 up to V4 for most vegetative components (leaves, stem, and roots) (Fig. 3 and Table 2).
Stem diameter growth was low at the early cultivation stages, increased at stages V3 and V4, and decreased at stage V5. This pattern suggests that, as a result of the increased number of leaves, photosynthetic metabolites accumulate in the stem as the plant grows until vegetative growth decreases (Fig. 3). Maximum stem diameter was 7.0 cm at 411 DAP and maximum daily growth rate was 0.21 mm day-1 (Table 2). A similar stem diameter (7.2 cm) at the flower induction stage was observed by Sampaio et al. (2011) for the Gold cultivar.
The maximum accumulation of total fresh(291.46 t ha-1) and dry biomass (91.56 t ha-1) occurred at stages V4 (558 DAP) and V5 (694 DAP), respectively, and the maximum biomass accumulation rate was 1,069.15 and 183.69 kg ha-1 day-1 (Table 2). Total biomass production is a result of plant growth, because as the plant metabolic rate increases, vegetative growth also increases, resulting in greater biomass production.
In general, fresh and dry leaf matter production was low until stage V2 (450 DAP), increased until stage V3, and remained constant after the flower induction phase, at the end of stage V5 (Fig. 3). The maximum fresh and dry matter accumulation was 116.35 and 22.42 t ha-1, respectively, at stage V5. The maximum daily accumulation rate of fresh and dry leaf matter was 972.66 and 145.21 kg ha-1 day-1 at 502 and 514 DAP (stage V3), respectively (Table 2), suggesting that nutritional demand for the production of leaves was high at this stage. Leaf dry matter production in this study was higher than in the study by Melo et al. (2006) for irrigated Pérola cultivar (16.99 t ha-1) and similar to the study by Souza et al. (2007) for the Pérola cultivar grown with supplemental irrigation (158.5 and 20.55 t ha-1 of fresh and dry leaf matter, respectively) at the vegetative growth stage.
Stem growth and fresh and dry matter accumulation were low until the start of stage V2, at approximately 460 DAP (Fig. 3). As leaf production and metabolic activity increased, stem growth and biomass accumulation also increased: at the onset of the reproductive stage, fresh stem matter was 45.93 t ha-1 (656 DAP) and dry stem matter was 24.11 t ha-1 (679 DAP).
Root growth, represented by fresh and dry matter production, was estimated by a non-linear regression model (Gaussian function with three parameters, Fig. 3 and Table 2). Root production was low at early cultivation stages (V1 and V2) and reached its maximum after stage V5 (686-700 DAP): 2.33 and 0.63 t ha-1 of fresh and dry matter, respectively. However, the inflection point (IP) corresponding to the maximum daily accumulation rate occurred at stage V4, with an average fresh and dry matter production of 9.47 and 2.33 kg ha-1 day-1, respectively. The reduction in fresh and dry matter accumulation in roots after stage V5 may be associated with lower photoassimilate flow and greater root death as a result of the higher demand for photoassimilates by reproductive organs at the reproductive stage.
Fresh and dry matter production of fruits (fruit with crown) and seedlings were determined at the end of the cultivation cycle (Table 3). The average production of fresh fruits with crown was estimated at 72 t ha-1, which corresponds to an average fruit mass of 1.4 kg and 22% of total fresh biomass per fruit. This productivity is similar to the values recorded for cvs. Gold and Jupi (SAMPAIO et al., 2011), whereas Guarçoni and Ventura (2011) reported a maximum estimated production of 68.6 t ha-1 and an average fruit with crown weight of 1.34 kg for the MD-2 cultivar grown with 620.15 and 570.13 kg ha-1 of N and K, respectively. Conversely, Melo et al. (2006) recorded a higher yield (77.2 t ha-1), with a population of 55,555 plants ha-1, for Vitória plants in an irrigated area, whereas Souza et al. (2007) reported lower crop yields of 57.0 t ha-1 and 61.5 t ha-1 for Pérola and Smooth Cayenne cultivars.
At the end of the pineapple cycle, the average total biomass accumulation was estimated at 326 and 68 t ha-1 of fresh and dry matter, respectively (Table 3). Of the plant parts evaluated, vegetative biomass accounted for an average total fresh and dry matter accumulation of 67.5% (222 t ha-1) and 80% (55 t ha-1), respectively (Table 3); whereas leaves alone accumulated 54 and 47% of total fresh and dry biomass. Plant parts associated with reproductive organs (fruits and seedlings), which may be exported from the cultivation area for commercial use, accumulated an average 20% of the pineapple's dry biomass. These results suggest that vegetative biomass greatly contributes to organic matter deposition and soil carbon storage and may be available in future cultivations. Significant dry matter accumulation was also observed in leaves (45.0%), stems (16.0%), and roots (5.0%) of Josapine plants grown in sandy tin tailings (HANAFI and HALIMAH, 2004).
The Pearson's correlation coefficient was used to determine the relationship between stem diameter, plant height, and D-leaf length with fresh vegetative biomass of plant parts (total number of leaves, D-leaf, stem, and root) (Table 4). The positive and significant correlations observed for all characteristics analyzed, suggest that D-leaf fresh mass, D-leaf length, stem diameter, and plant height may be used as estimators of total biomass production and fruit yield. In fact, plants with higher biomass production produce proportionally heavier fruits (HANAFI and HALIMAH, 2004; HANAFI et al., 2009; RODRIGUES et al., 2010; GUARCONI and VENTURA, 2011; RAZZAQUE and HANAFI, 2011).
CONCLUSIONS
The indication of vegetative stages through relative biomass production is appropriate instrument for evaluating the growth of Vitoria pineapple.
Plant growth was higher between vegetative stages V2 and V4 (463-560 days after planting, DAP), with a 60% increase in total biomass. Thus, nutrient and water use require special attention during these stages.
The maximum growth rate for plant height, D-leaf length, and stem diameter was observed at the end of the phenological stage V1 (390-411 DAP), and at the end of stage V5 these plant traits had average values of 106 cm, 82 cm, and 7 cm, respectively.
The maximum fresh and dry vegetative biomass accumulation occurred between stages V4 and V5. Leaves accounted for the highest biomass accumulation during the vegetative and reproductive stages, with a 54% and 47% fresh and dry biomass accumulation at the end of the cultivation period.
Fruit yield at the reproductive stage was 72 t ha-1 (1.4 kg/fruit) and fresh biomass production was 326 t ha-1 of plant biomass (fresh matter); between 70% and 80% of the accumulated biomass remains in the field following fruit and slip harvest, and can be incorporated as plant residue into soil.
ACKNOWLEDGMENTS
The authors thanks CNPq and FAPEMIG for their financial support.
REFERENCES
CABRAL, J.R.S.; MATOS, A.P. Imperial, a new pineapple cultivar resistant to fusariosis. Acta Horticulturae, Leuven, n.822, p.47-50, 2009.
FERREIRA, D.F. Sistema de análises de variância para dados balanceados (SISVAR 4.1). Lavras: Universidade Federal de Lavras, 2000.
FOURNIER, P.; DUBOIS, C.; BENNEVEAU, A.; SOLER, A. Growth Indicators for Different Pineapple Cultivars Compared with the Current Standard 'Smooth Cayenne' in West Africa and Reunion Island: A First Step toward Modeling Growth. Agronomy Journal, Madison, v.102, n.6, p.1572-1577, 2010.
GRACIANO, E. S. A.; NOGUEIRA, R. J. M. C.; LIMA, D.R.M.; PACHECO, C. M.; SANTOS, R. Crescimento e capacidade fotossintética da cultivar de amendoim BR1 sob condições de salinidade. Revista Brasileira de Engenharia Agrícola e Ambiental, Campo Grande, v.15, n. 8, p.794-800, 2011.
GUARÇONI, A. M.; VENTURA, J. A. Adubação N-P-K e o desenvolvimento, produtividade e qualidade dos frutos de abacaxi 'gold' (MD-2). Revista Brasileira de Ciência do Solo, Viçosa, MG, v.35, n.4, p.1367-1376, 2011.
HANAFI, M.M.; SELAMAT, M.M.; HUSNI, M.H.A.; ADZEMI, M.A. Dry matter and nutrient partitioning of selected pineapple cultivars grown on mineral and tropical peat soils. Communications in Soil Science and Plant Analysis, New York, v.40, n.21-22, p.3263-3280, 2009.
HANAFI, M.M.; HALIMAH, A. Nutrient supply and dry-matter partitioning of pineapple cv. Josapine on sandy tin tailings. Fruits, Cambridge, v.59, n.5, p.359-366, 2004.
IBGE- Instituto Brasileiro de Geografia e Estatística. 2009. Levantamento sistemático da produção agrícola Municipal. Disponível em: <http://www.ibge.gov.br/home/estatºstica/indicadores/agropecuaria/lspa/lspa_201209.pd>. Citado em: 10 set.2012.
INMET- Instituto Nacional de Meteorologia. 2012. Banco de dados meteorológicos para ensino e pesquisa. Disponível em: <http://www.inmet.gov.br/portal/index.php?r=bdmep/bdmep>. Acesso em: 29 set. 2012.
LIMA, J.F.; PEIXOTO, C.P.; LEDO, A.S. Índices fisiológicos e crescimento inicial do mamoeiro (Caryca papaya L.) em casa de vegetação. Ciência Agrotécnica, Lavras, v.31, n.5, p.1358-1363, 2007.
LIMA, J.F.; PEIXOTO, C.P.; SILVA, A.L.L.; BORGES, V.P. Crescimento e alocação de fitomassa em plantas de mamoeiro em casa de vegetação. Magistra, Cruz das Almas, v.23, n.4, p.186-192, 2011.
MARQUES, L.S.; ANDREOTTI, M.; BUZETTI, S.; ISEPON, J.S. Produtividade e qualidade de abacaxizeiro cv. Smooth Cayenne, cultivado com aplicação de doses e parcelamentos do nitrogênio, em Guaraçi - SP. Revista Brasileira de Fruticultura, Jaboticabal, v.33, n.3, p.1004-1014, 2011.
MELO, A. S.; AGUIAR NETO, A. O.; DANTAS NETO, J.; BRITO, M. E. B.; VIÉGAS, P. R. A.; MAGALHÃES, L. T. S.; FERNANDES, P. D. Desenvolvimento vegetativo, rendimento da fruta e otimização do abacaxizeiro cv. Pérola em diferentes níveis de irrigação. Ciência Rural, Santa Maria, v.36, n. 1, p.93-98, 2006.
OMOTOSO, S.O.; AKINRINDE, E.A. Influence of propagule masss and nitrogen fertilizer rates on growth and yield of pineapple (Ananas comosus). Asian Journal of Agricultural Research, Malaysia, v.1, n.3, p.131-136, 2007.
PY, C.; LACOEUILHE, J.J.; TEISON, C. The pineapple: cultivation and uses. Paris: G.P. Maisonneuve et Larose, 1987. 568p.
RAMOS, M.J.M.; MONNERAT, P.H.; PINHO, L.G.R. Leitura SPAD em abacaxizeiro imperial cultivado em deficiência de macronutrientes e de boro. Revista Brasileira de Fruticultura, Jaboticabal, v.35, n.1, p. 277-281, 2013.
RAZZAQUE, A.H.M.; HANAFI, M.M. Effect of potassium on growth, yield and quality of pineapple in tropical peat. Fruits, Paris, v.56, n.1, p.45-49, 2011.
REINHARDT, D.H.; SOUZA, L.F.S.; CABRAL, J.R.S.
Abacaxi. Produção: aspectos técnicos. Brasília: EMBRAPA/Comunicação para Transferência de Tecnologia, 2000. 77p.
REINHARDT, D.H.; SOUZA, L.F.S.; CABRAL, J.R.S. Abacaxi irrigado em condições semiáridas. Cruz das Almas: Embrapa Mandioca e Fruticultura, 2001. 108p.
REINHARDT, D.H.; SOUZA, A.P.M.; CALDAS, R.C.; ALCÂNTARA, J.P.; ALMEIDA, A.A. Management of slips and its effect on growth and production of 'Pérola' pineapple plants. Revista Brasileira de Fruticultura, Jaboticabal, v.25, n.2, p.248-252, 2003.
RIBEIRO A.C.; GUIMARÃES, P.T.G; ALVAREZ, V V.H. Recomendações para o uso de corretivos e fertilizantes em Minas Gerais - 5º Aproximação. Viçosa: Comissão de Fertilidade do Solo do Estado de Minas Gerais, 1999. 359 p.
RODRIGUES, A. A.; MEDONÇA. R. M. N.; SILVA, A. P.; SILVA, S. M.; PEREIRA, W. E. Desenvolvimento vegetativo de abacaxizeiros 'Pérola' e 'Smooth Cayenne' no Estado da Paraíba. Revista Brasileira de Fruticultura, Jaboticabal, v.32, n.1, p.126-134, 2010.
SAMPAIO, A.C.; FUMES, T.F.; LEONEL, L. Crescimento vegetativo e características dos frutos de cinco cultivares de abacaxi na região de Bauru-SP. Revista Brasileira de Fruticultura, Jaboticabal, v.33, n.3, p.816-822, 2011.
SILVA, A.L.P.; SILVA, A.P.; SOUZA, A.P.; SANTOS, D.S.; SILVA, S.M.; SILVA, V.B. Resposta do abacaxizeiro 'Vitória' a doses de nitrogênio em solos de tabuleiros costeiros da Paraíba. Revista Brasileira de Ciência do Solo, Viçosa, MG, v.36, n.2, p.447-456, 2012.
SOUZA, C.B.; SILVA, B.B.; AZEVEDO, P.V. Crescimento e rendimento do abacaxizeiro nas condições climáticas dos Tabuleiros Costeiros do Estado da Paraíba. Revista Brasileira de Engenharia Agrícola e Ambiental, Campina Grande, v.11, n. 2, p.134-141, 2007.
VENTURA, J. A.; COSTA, H.; CAETANO, L. C. Abacaxi Vitória: uma cultivar resistente a fusariose. Revista Brasileira de Fruticultura, Jaboticabal, v.31, n.4, p. 931, 2009.
Recebido em: 05-08-2013.
Aceito para publicação em: 08-07-2014.
- CABRAL, J.R.S.; MATOS, A.P. Imperial, a new pineapple cultivar resistant to fusariosis. Acta Horticulturae, Leuven, n.822, p.47-50, 2009.
- FERREIRA, D.F. Sistema de análises de variância para dados balanceados (SISVAR 4.1). Lavras: Universidade Federal de Lavras, 2000.
- FOURNIER, P.; DUBOIS, C.; BENNEVEAU, A.; SOLER, A. Growth Indicators for Different Pineapple Cultivars Compared with the Current Standard 'Smooth Cayenne' in West Africa and Reunion Island: A First Step toward Modeling Growth. Agronomy Journal, Madison, v.102, n.6, p.1572-1577, 2010.
- GRACIANO, E. S. A.; NOGUEIRA, R. J. M. C.; LIMA, D.R.M.; PACHECO, C. M.; SANTOS, R. Crescimento e capacidade fotossintética da cultivar de amendoim BR1 sob condições de salinidade. Revista Brasileira de Engenharia Agrícola e Ambiental, Campo Grande, v.15, n. 8, p.794-800, 2011.
- GUARÇONI, A. M.; VENTURA, J. A. Adubação N-P-K e o desenvolvimento, produtividade e qualidade dos frutos de abacaxi 'gold' (MD-2). Revista Brasileira de Ciência do Solo, Viçosa, MG, v.35, n.4, p.1367-1376, 2011.
- HANAFI, M.M.; SELAMAT, M.M.; HUSNI, M.H.A.; ADZEMI, M.A. Dry matter and nutrient partitioning of selected pineapple cultivars grown on mineral and tropical peat soils. Communications in Soil Science and Plant Analysis, New York, v.40, n.21-22, p.3263-3280, 2009.
- HANAFI, M.M.; HALIMAH, A. Nutrient supply and dry-matter partitioning of pineapple cv. Josapine on sandy tin tailings. Fruits, Cambridge, v.59, n.5, p.359-366, 2004.
- IBGE- Instituto Brasileiro de Geografia e Estatística. 2009. Levantamento sistemático da produção agrícola Municipal. Disponível em: <http://www.ibge.gov.br/home/estatşstica/indicadores/agropecuaria/lspa/lspa_201209.pd>. Citado em: 10 set.2012.
- INMET- Instituto Nacional de Meteorologia. 2012. Banco de dados meteorológicos para ensino e pesquisa. Disponível em: <http://www.inmet.gov.br/portal/index.php?r=bdmep/bdmep>. Acesso em: 29 set. 2012.
- LIMA, J.F.; PEIXOTO, C.P.; LEDO, A.S. Índices fisiológicos e crescimento inicial do mamoeiro (Caryca papaya L.) em casa de vegetação. Ciência Agrotécnica, Lavras, v.31, n.5, p.1358-1363, 2007.
- LIMA, J.F.; PEIXOTO, C.P.; SILVA, A.L.L.; BORGES, V.P. Crescimento e alocação de fitomassa em plantas de mamoeiro em casa de vegetação. Magistra, Cruz das Almas, v.23, n.4, p.186-192, 2011.
- MARQUES, L.S.; ANDREOTTI, M.; BUZETTI, S.; ISEPON, J.S. Produtividade e qualidade de abacaxizeiro cv. Smooth Cayenne, cultivado com aplicação de doses e parcelamentos do nitrogênio, em Guaraçi - SP. Revista Brasileira de Fruticultura, Jaboticabal, v.33, n.3, p.1004-1014, 2011.
- MELO, A. S.; AGUIAR NETO, A. O.; DANTAS NETO, J.; BRITO, M. E. B.; VIÉGAS, P. R. A.; MAGALHÃES, L. T. S.; FERNANDES, P. D. Desenvolvimento vegetativo, rendimento da fruta e otimização do abacaxizeiro cv. Pérola em diferentes níveis de irrigação. Ciência Rural, Santa Maria, v.36, n. 1, p.93-98, 2006.
- OMOTOSO, S.O.; AKINRINDE, E.A. Influence of propagule masss and nitrogen fertilizer rates on growth and yield of pineapple (Ananas comosus). Asian Journal of Agricultural Research, Malaysia, v.1, n.3, p.131-136, 2007.
- PY, C.; LACOEUILHE, J.J.; TEISON, C. The pineapple: cultivation and uses. Paris: G.P. Maisonneuve et Larose, 1987. 568p.
- RAMOS, M.J.M.; MONNERAT, P.H.; PINHO, L.G.R. Leitura SPAD em abacaxizeiro imperial cultivado em deficiência de macronutrientes e de boro. Revista Brasileira de Fruticultura, Jaboticabal, v.35, n.1, p. 277-281, 2013.
- RAZZAQUE, A.H.M.; HANAFI, M.M. Effect of potassium on growth, yield and quality of pineapple in tropical peat. Fruits, Paris, v.56, n.1, p.45-49, 2011.
- REINHARDT, D.H.; SOUZA, L.F.S.; CABRAL, J.R.S. Abacaxi irrigado em condições semiáridas. Cruz das Almas: Embrapa Mandioca e Fruticultura, 2001. 108p.
- REINHARDT, D.H.; SOUZA, A.P.M.; CALDAS, R.C.; ALCÂNTARA, J.P.; ALMEIDA, A.A. Management of slips and its effect on growth and production of 'Pérola' pineapple plants. Revista Brasileira de Fruticultura, Jaboticabal, v.25, n.2, p.248-252, 2003.
- RIBEIRO A.C.; GUIMARÃES, P.T.G; ALVAREZ, V V.H. Recomendações para o uso de corretivos e fertilizantes em Minas Gerais - 5ş Aproximação. Viçosa: Comissão de Fertilidade do Solo do Estado de Minas Gerais, 1999. 359 p.
- RODRIGUES, A. A.; MEDONÇA. R. M. N.; SILVA, A. P.; SILVA, S. M.; PEREIRA, W. E. Desenvolvimento vegetativo de abacaxizeiros 'Pérola' e 'Smooth Cayenne' no Estado da Paraíba. Revista Brasileira de Fruticultura, Jaboticabal, v.32, n.1, p.126-134, 2010.
- SAMPAIO, A.C.; FUMES, T.F.; LEONEL, L. Crescimento vegetativo e características dos frutos de cinco cultivares de abacaxi na região de Bauru-SP. Revista Brasileira de Fruticultura, Jaboticabal, v.33, n.3, p.816-822, 2011.
- SILVA, A.L.P.; SILVA, A.P.; SOUZA, A.P.; SANTOS, D.S.; SILVA, S.M.; SILVA, V.B. Resposta do abacaxizeiro 'Vitória' a doses de nitrogênio em solos de tabuleiros costeiros da Paraíba. Revista Brasileira de Ciência do Solo, Viçosa, MG, v.36, n.2, p.447-456, 2012.
- SOUZA, C.B.; SILVA, B.B.; AZEVEDO, P.V. Crescimento e rendimento do abacaxizeiro nas condições climáticas dos Tabuleiros Costeiros do Estado da Paraíba. Revista Brasileira de Engenharia Agrícola e Ambiental, Campina Grande, v.11, n. 2, p.134-141, 2007.
- VENTURA, J. A.; COSTA, H.; CAETANO, L. C. Abacaxi Vitória: uma cultivar resistente a fusariose. Revista Brasileira de Fruticultura, Jaboticabal, v.31, n.4, p. 931, 2009.
Publication Dates
-
Publication in this collection
19 Nov 2014 -
Date of issue
Sept 2014
History
-
Received
05 Aug 2013 -
Accepted
08 July 2014