Abstract
Maltodextrin was produced from cassava and corn starch by enzymatic hydrolysis with alpha-amylase. The cassava starch hydrolysis rate was higher than that of corn starches in maltodextrin production with shorter dextrose equivalent (DE). DE values do not show directly the nature of the obtained oligosaccharides. Maltodextrin produced from cassava and corn starch was analysed by high performance liquid chromatography (HPLC), and the analysis showed that maltodextrin production differs according to the source of the starch. This is important in defining the application of the maltodextrin, according to its desired function.
maltodextrin; cassava; corn
ARTIGO
Cassava and corn starch in maltodextrin production
Geovana Rocha Plácido MooreI; Luciana Rodrigues do CantoI; Edna Regina Amante* * e-mail: eamante@cca.ufsc.br , I; Valdir SoldiII
IDepartamento de Ciência e Tecnologia de Alimentos, Centro de Ciências Agrárias, Universidade Federal de Santa Catarina, Rod. Admar Gonzaga, 1346, 88034-001 Florianópolis - SC
IIDepartamento de Química, Univesidade Federal de Florianópolis, CP 476, 88040-900 Florianópolis - SC
ABSTRACT
Maltodextrin was produced from cassava and corn starch by enzymatic hydrolysis with a-amylase. The cassava starch hydrolysis rate was higher than that of corn starches in maltodextrin production with shorter dextrose equivalent (DE). DE values do not show directly the nature of the obtained oligosaccharides. Maltodextrin produced from cassava and corn starch was analysed by high performance liquid chromatography (HPLC), and the analysis showed that maltodextrin production differs according to the source of the starch. This is important in defining the application of the maltodextrin, according to its desired function.
Keywords: maltodextrin; cassava; corn.
INTRODUCTION
The starch world production is mainly provided by corn and potato, and also by wheat, sago and sorghum. Efforts have been made to study starch alternative sources as the triticalle, savage rice, cassava and amaranthus1.
The native starch has an industrial restrictive use, due to its low functional properties, adequate to specific applications, such as the tendency to retrogradation during cold storage. Several physical, chemical and enzymatic modifications have improved the functional properties of starch allowing a wide range of application. Starch hydrolysis is an easy way of producing carbohydrates with special functional properties. The recent enzyme development has improved starch processing both economically and technically2. Another important factor in hydrolysed starch production is the need for acquiring a technological expertise with rigorous process control, to adequate starch modification from raw material, process and product control.
Maltodextrin is a starch derivative with a wide range of applications. Corn starch has been extensively used as a raw material in maltodextrin production and the cassava starch is traditionally used in Thailand and other cassava starch producers in maltodextrin production. The differences in structure of maltodextrins from different botanical sources determine their physicochemical properties, the characterisation of maltodextrins becoming important according to several sources to defined applications3.
Besides its wide application in food industry, starch is also extensively used in textile, paper, metallurgy, pharmacy and plastic industries. Despite its utility as natural starch, amylose and amylopectin, or its hydrolyse products (dextrin, maltose and glucose), starch can be modified or derivatised to reach technical needs. These modifications can be physical, chemical or enzymatic. As described in the literature, starch can be chemically modified mainly by acids, phosphates, cross-linked, acetates, dialdehydes, propionates, butirates, caproates, benzoates, methyl and benzyl4.
The enzymatic modification of starch can produce maltodextrin, cyclodextrin and oligossaccharides. Maltodextrin is produced with a-amylase with or without hydrochloridric acid, with different molecular weights, according to the production method and source. Maltodextrins have been studied as a plasticyzer to reduce glass transition temperature in materials5.
At the beginning, the starch for this study was hydrolysed by thermal and acid treatments. Both processes showed low polymerisation degree (PD=42) and undesirable characteristics in maltodextrin, associated to the formation of undesirable products. On the other hand, considering that the starch enzymatic hydrolysis produced sugars and maltodextrins with high yield, this method can be used to this objective1,2. The efficiency of starch hydrolysis to produce sugars and maltooligossaccharides depends on the a-amylase, glucoamylase and pullulanase enzymes action. Several moulds and bacteria produce enzymes that degrade starches with good activity and stability at different temperatures and pH2.
Maltodextrin (C6H10O5)n.H2 O is a polymer of saccharides; nutritive, not sweet that consists of glucose units primarily linked by a-1,4 glucosidic bounds, with DE (dextrose equivalent) values lower than 20. The DE is expressed as a percentage of glucosidic bound hydrolysis, showing its reducing power. The dextrose used as standard in this study is starch (DE=0) and glucose (DE=100)6,7,8. Several physical and functional properties, such as sweetness, compressibility and viscosity vary according to the extent of starch hydrolysis, which is characterised by DE determination8.
Due to the wide range of application of maltodextrins, several chemical and biological properties are required, the dextrose equivalent of a maltodextrin being inadequate to predict products performance in various applications. Maltodextrin with the same DE can even have different properties in various applications that reflect differences in their molecular composition, linearity and branching which must be considered6.
Corn starch has been used in maltodextrin production, but recently some attention has been called to other starches such as: cassava, potato, rice, wheat and others1. Maltodextrins and corn solid syrups are generally prepared from corn starch or waxy corn starch by enzymatic controlled hydrolysis, named liquefaction. The starch gel is adjusted to a desired solid concentration (30 to 40% of solids, dry base weight), the pH is adjusted with acid to 6.5, the enzyme (B. licheniformis a-amylase) and the calcium ion are added. The starch paste is bombed to the cooker machine, where the temperature is increased to 140 ºC, during 5 to 10 min. Then, the gelatinised starch goes to the liquefaction reactor. At this point the starch polymeric material is dispersed and is easily hydrolysed by enzymes. The gelatinised starch is taken to the liquefaction temperature (90-95 ºC) and a new addition of a-amylase is necessary due to enzyme degradation by the high temperature treatment1.
As an ingredient in food, maltodextrin offers consistency, viscosity, mild texture and stability. The increase of soluble solids in foods inhibits crystallisation and controls the freezing point9.
Maltodextrin with low DE has similar properties to those of native starch and can be used as a fat replacer9. As described by Guzmán-Maldonado and López1, new products are available in the market as fat replacers, reducing the risk of cardiac problems by reducing of cholesterol blood level. This characteristic of the new products including maltodextrin has been of interest the general public and to the food industry, mainly.
Cassava and corn starches are abundant raw material in Brazil. The purpose of this work was to study the differences between corn and cassava starch granules, considering that cassava starch would be an important raw material, which can be modified by enzymatic hydrolysis to produce maltodextrin. With the aim of justifying cassava starch as a raw material for maltodextrin production, the amylose content, swelling power and viscosity curves were evaluated for both starches studied.
For both starches, maltodextrins were characterised in terms of the dextrose equivalent, in order to achieve the ideal molecular weight for different applications. The yield of maltodextrin was determined as soluble solids in both systems. On the other hand, Maltooligossaccharides were characterised by high performance liquid chromatography (HPLC).
Maltodextrin has been chosen due to its wide range in market application, its relatively simple production and control processes and, consequently, the possibility of its diffusion among agroindustries. According to the Food and Agricultural Organisation of the United Nations, the properties of tropical starches must be studied in order to help the country development for an efficient competition in the international trade of starch.
EXPERIMENTAL PART
Cornstarch samples were supplied by the National Starch Company (Trombudo Central, SC, Brazil) and by the local market (Florianópolis, SC, Brazil). Cassava starch samples were supplied by Cassava Agro-industrial (Rio do Sul, SC, Brazil) and Indian Chemical Industries (Treze de Maio, SC, Brazil).
Raw material characterisation
Ash and moisture contents were determined by the 923.10 and 923.03 AOAC Methods, respectively10. Each sample was analysed five times and average values were considered.
Amylose in corn and cassava starch samples was determined by Williams et al.11 Method. The standard amylose was extracted from cassava and starch samples according to McCready & Hassid12 method.
The Brabender viscosity curves were obtained from 6% starch suspension on the National Starch Company's Brabender amylograph, Trombudo Central, SC, Brazil, according to the procedure recommended by Mazurs et al.13.
The swelling power was determined following the procedure of Leach et al.14. Water suspensions of starch at 3% (DBW) were submitted to gelatinisation at temperatures of 60, 65, 70, 75, 85, 90 and 95 ºC, with occasional stirring. The water used in gelatinisation was evaluated gravimetrically after centrifugation of the gel at 3000 rpm.
Maltodextrin production
For starch hydrolysis a-amylase TERMAMYL 120 L15, from Bacillus liqueniformis with activity of 120 KNU/g. 1KNU (one Kilogram of a-amylase NOVO) has been used. It was defined as the amount of enzyme necessary to decompose 5.26 g of starch per hour in standard conditions.
Starch liquefaction
The suspension of starch was prepared with 75 g of starch to 175 mL of distilled water (corresponding to 30% starch suspension w/w). 0.222 g chloride calcium was added (80 ppm). pH was adjusted to 6.5 with sodium hydroxide 0.1 N. 40 µL of a-amylase thermamyl 120 L (equivalent to 0.6 Kg of enzyme by ton of starch) was added. Suspensions were submitted to water bath at 100 ºC under stirring.
At each 15 minutes, one fraction was removed from the water bath completing 120 minutes of hydrolysis. Corn and cassava starches were submitted to this treatment in five repetitions at each step of the treatment. Each starch, corn and cassava yielded 40 maltodextrins.
Enzyme inactivation was obtained by addition of hydrochloridric acid 0.1 N, up to pH 4.0. The final volume of analysis was adjusted to 250 mL in volumetric flask with distilled water, homogenised, transferred to a centrifuge flask and centrifuged at 4 ºC, 8000 rpm by 20 minutes, according to Griffin & Brooks16. The liquid containing maltodextrin was frozen in a plate freezer and stored until the time of analysis.
Dextrose equivalent determination
Polymerisation degree during starch hydrolysis was determined by reducing sugar evaluation in the final product. In this method, 3,5-dinitrosalisilic acid (3,5-DNS) in the presence of sugar and sodium hydroxide is reduced to 3 amino-5 nitrosalisilic acid17.
Intrinsic viscosity
Leach14 method was used to determine the intrinsic viscosity of cornstarch after 15 minutes of hydrolysis at 0.0025, 0.0033, 0.005 and 0.01 g/mL concentrations. The measurements were performed in an Ostwald viscometer at 24.2 ºC using pure water as a reference to the flow (To). The relative viscosity was obtained from the ratio T/To, where T is the time of flux in seconds on test concentration. Specific viscosity (SV) was T/To 1, and the relative viscosity as SV/Concentration. The extrapolation from hydrolisate concentration zero and the natural logarithm of viscosity at several hydrolisate concentrations, results on the intrinsic viscosity.
High Performance Liquid Chromatography
The samples for the chromatographic determination of maltooligosaccharides were selected according to the dextrose equivalent (DE). The maltooligosaccharides with dextrose equivalent more commonly commercialised: DE= 18 to 201 were identified. Commercial maltodextrin with this level of DE occurred at 15 minutes. Then, maltodextrin produced in 15 minutes from cassava and corn starches was evaluated.
The carbohydrate profile from selected samples was determined by use a SUPERCOGEL C-610 H column, 30 cm X 7.8 mm ID (Sigma Aldrich Co., 1995). Solution of 0.1% phosphoric acid was used as mobile phase. Samples were solubilized in the mobile phase at 400 µL/mL. The volume injection was 10 µL, at rate flow of 0.5 mL/minute at 32 ºC. The oligosaccharide profile was identified by comparing eluted volume with the retention time supplied by Sigma Aldrich Co.
Soluble solids
The yield of hydrolysis was evaluated by soluble solids determination (ºBRIX) in ABBE Refractometer.
RESULTS
Starches characterisation
Before the evaluation of maltodextrin production, corn and cassava starches were characterised in terms of moisture, amylose and amylopectin content, swelling power and viscoamylographic properties. Considering that the raw material can affect maltodextrin production, it is important to know the main properties of both starches. For example, moisture was corrected to 11.13 and 12.40% for cassava and corn starch, respectively, in order to achieve the best condition for hydrolysis.
Another important property, is ash content, which in starches is considered as a purity parameter. In this study, ash content average values of 0.11 and 0.10 % of were determined for corn and cassava starch, respectively. These values are lower than the maximum amount defined by Brazilian Food Standard18, which is 0.20, and 0.25 % for corn and cassava starch, respectively. This result shows that both starches have a good quality of mineral content to be used as raw material in maltodextrin production, and statistically no differences were observed between cassava and corn starch ash content.
The amylose contents determined for corn and cassava starches were 24.26 and 16.67 %, respectively. These values are in agreement to those determined by Williams et al.12. In general, amylose content can explain different rheologic properties in starch gels and the generation of characteristic oligosaccharides.
Viscoamylographic properties of the cassava and corn starch
Generally, several aspects related to granule structure are associated to rheologic properties. Average values of the paste initial temperature, temperature at the maximum viscosity peak, maximal Brabender viscosity and viscosity after 20 minutes at 90 °C and on cooling at 50 °C, are shown in Table 1. The paste initial temperature, higher in corn than in cassava starch, can be explained by the swelling power of the latter as discussed in the following section, swelling depends on the starch granule compaction. As described by Whistler et al.19 the water entrance in corn starch is more difficult than in cassava starch due to its more compact structure. The lower value of the paste initial temperature for the cassava starch reflects this behaviour. The higher value of the Maximal Brabender Viscosity in cassava starch is apparently also associated to the water entrance in the granule.
The significant differences in terms of the Maximal Brabender viscosity observed between cassava (950 B.U.; occurring at 64.5 °C) and corn starch (280 B. U.; occurring at 90 °C) can be explained also by the swelling power and amylose content. The internal arrangement of the granule depends on the amylose and amylopectin distribution, which affects consequently the viscosity behaviour. The high maximal viscosity achieved by cassava starch is indicative of the facility of water entrance in the cassava starch granule and, in consequence, a lower viscosity in the cooling. The high maximal viscosity achieved by cassava starch can be explained by its amylose contents.
The paste formation occurs only with the entrance of water in the granule, which is naturally more difficult in higher amylose content granules. After the swelling, the starch becomes more susceptible to enzymatic hydrolysis.
Swelling power
The swelling power of the starch was analysed in order to obtain information about the structural differences between different botanical origin and molecular arrangement of the granules. The molecular arrangement, which depends on the present amount of amylose and amylopectin, allows an estimation of the kind of organisation occurring in the interior of the granule. The behaviour of the swelling power for corn and cassava starches is shown in Figure 1. The results indicated a higher degree of swelling for cassava starch. At ca. 65 °C the cassava swelling power is 23 %, remaining practically constant up to 95 °C, whereas for corn starch it is 10 % at 75 °C increasing to 20% at 95 °C. The higher facility for water entrance in cassava starch is due to amylopectin high content and the low molecular density.
On the other hand, resistance to water entrance in corn starch is due to intramolecular hydrogen bonding in the amylose, what increases the crystallinity regions in the starch granule.
In comparison to cassava starch, the corn starch swelling power is lower at every evaluated temperature. This fact reveals that the corn starch granule, besides offering more resistance to gelatinisation, doesn't have a granular structure for water retention, which is a characteristic of starches with low content of amylopectin.
Maltodextrin characterisation
Cassava and corn starches are different on amylose and amylopectin content, swelling power and viscoamylographic properties. The differences are apparently associated to the origin of the granule, climatic conditions and several other factors. At the same time maltodextrin with different functional properties can be produced from different starches, due to the different behaviour of the starch when submitted to hydrolysis by a-amylase.
Starch hydrolysis control, such as the stopping of the ±-amylase action in the due moment to achieve specific maltodextrins molecular weight was followed by dextrose equivalent (DE) evaluation.
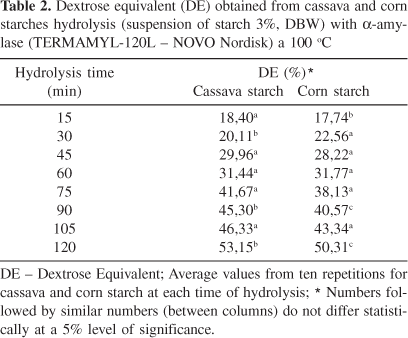
The hydrolysis rate, or the tendency of a-amylase to hydrolyse the corn and cassava starches, was followed each 15 minute during 120 minutes, at 100 ºC, in ideal conditions of the a-amylase TERMAMYL-120 L (Table 2).
These results reveal that, statistically, the action of the a-amylase on cassava starch is different from the action of this same enzyme on corn starch in the first fifteen minutes of hydrolysis. Due to its lower amylose content, cassava starch presents lower paste initial temperature and higher swelling point, which are favourable to the enzyme penetration via amorphous regions that are higher in the cassava starch granule.
Several publications described that the enzymatic susceptibility is higher in granules with low amylose content. This fact shows that hydrolysis occurs in the branched region of the granule (amylopectin)20.
The hydrolysis rate was statistically similar most of the time, but the nature of the maltodextrin produced depended on the remaining units not achieved by the enzyme, in both starches. The constant rate of (K) for starch hydrolysis was 0.39 and 0.33 for cassava and corn starch, respectively. This value suggests that the cassava starch was more easily hydrolysed.
In addition to the kinetic for maltodextrin production from cassava and corn starch, it is important to consider the nature of the maltodextrin produced: functional properties due to molecular weight, polymerisation degree, glucose number and the point of glucosidic bonds. Several maltodextrins with similar molecular weight are different in terms of their functional properties, starch nature and hydrolysis way. For example, the predominance of maltodextrin with branched structure affects the retrogradation and increases the maltodextrin solution stability21.
Another aspect to be considered is related to the tendency of precipitation during storage, which is affected by the time or enzyme concentration in the hydrolysis. A high concentration of linear olygomers in maltodextrin increases the tendency of precipitation. The use of a-amylase and pullulanase, the control in reaction time can both contribute to the olygosaccharides composition and stability on storage21.
Through High Performance Liquid Chromatography the kind of maltodextrin produced can be identified. The presence of high molecular weight oligosaccharides in maltodextrin is higher in cassava starch than in corn starch, while maltotriose content is higher in maltodextrin from corn starch. In general, maltodextrins from both starches are poorer in glucose, maltose and maltotriose. Studies showed that the hydrolysis at high temperature with a-amylase from Bacillus licheniformis produce species with a low heterogeneous molecular weight distribution during the initial hydrolysis phase due to the increase of transglicosilation6. As described by Griffin & Brooks16, differences on the profile of maltooligosaccharides affect maltodextrin yield with different physico-chemical characteristics. For example, saccharides with high molecular weight affect the solubility and solution stability, while those with low molecular weight are recognised to affect properties like fermentability, viscosity, sweet power, humectancy and crystallisation.
While DE can inform about the amylose and amylopectin hydrolysis degree, soluble solids determination (ºBRIX) can express the starch hydrolysis yield, informing about the amount of soluble starch.
Soluble solids suspensions of cassava and cornstarch (30% DBW) were evaluated during the enzyme action. Desirable DE was achieved after 15' of hydrolysis. In this time, 40.46 % of the cassava starch and 43.00% the corn starch were used to produce maltodextrin (Table 3).
During the hydrolysis process, the maltodextrin had reduced its molecular weight, after 120 minutes and both cassava and corn starches were not totally soluble. The final yield for cassava and corn starch maltodextrin was 76.26% and 74.83%, respectively. This was expected, considering that several a-1,4 bonds were not accessible to the a-amylase and that the a-1,6 bonds were not hydrolysed.
CONCLUSIONS
Dextrose equivalent evaluation was not enough to make a difference between maltodextrin from cassava or corn starch.
The Dextrose Equivalent to cassava and corn starch at the point of desirable maltodextrin at 15 min was 18.70 and 17.74%, but the hydrolysis general performance was similar in both starches.
The High Performance Liquid Chromatography analysis of maltodextrin shows that the oligosacharides are different according to the origin of the starch, according to the predominance of linear or branched original molecules.
The hydrolysis yield determined from soluble solids (ºBRIX) reveals that the production of desirable maltodextrin can produce secondary products, with possible additional application, according to chemical characterisation.
Recebido em 13/4/04; aceito em 16/12/04; publicado na web em 13/4/05
- 1. Guzmán-Maldonado, G. H.; López, P.; Crit. Rev. Food Sci. Nutr. 1995, 35, 5.
- 2. Bhat, K. M.; Technical Bulletim of the Institute of Food Research, Reading Laboratory, U.K. 1998, 100 (1196b), 372.
- 3. Wang, Y.; Wang, L.; Starch 2000, 52, 8.
- 4. Ciacco, C. F.; Cruz, R.; Série Tecnologia Industrial 1982, 7.
- 5. Lim, -T. S.; Kasemsuwan, T.; Jane, J.; Pure Ap. Chem. 1995, 32, 4.
- 6. Marchal, L. M.; Beeftink, H. H.; Tramper, J.; Food Sci. Technol. 1999, 10, 345.
- 7. Dokic, P.; Jakovljevic, J.; Dokic-Baucal, L.; J. Colloid Surf. A 1998, 141, 435.
- 8. Storz, E.; Steffens, K.; Starch 2004, 56, 1.
- 9. Cônsole, S. F.; Tese de Doutorado, Universidade Estadual de Campinas, Brasil 1998.
- 10. AOAC; Official Methods of Analysis Of Aoac International, 16th ed., USA, 1998.
- 11. Williams, P. C.; Kuzina, D. F.; Hlinka, I.; Cereal Chem. 1970, 47, 197.
- 12. McCready, R. M.; Hassid, Z. W. In: Franco, L. M. C.; Ciacco, F. C.; Starch. 1992, 44, 11.
- 13. Mazurs, E. G.; Schoch, T. J.; Kite, F. E.; Cereal Chem. 1957, 34, 3.
- 14. Leach, H. W.; McCowen, L. D.; Schoch, T. J.; Cereal Chem. 1963, 36, 6.
- 15. NOVO NORDISK Bioindustrial do Brasil; Enzyme Process Division, ficha técnica: AMG, Araucária-PR, 1996.
- 16. Griffin, V. K.; Brooks, J. R.; J. Food Sci. 1989, 54, 1.
- 17. Villela, G. G.; Bacila, M.; Tastaldi, H.; Técnicas e Experimentos de Bioquímica, Ed. Guanabara Koogan, 1973.
- 18. BRASIL - Associação Brasileira das Indústrias da Alimentação. Compêndio da legislação de Alimentos, Consolidação das normas e padrões de alimentos. Resolução da CNNPA.[S.L]:1978, p. 7.12, vol. 2.
- 19. Whistler, R. L; Paschall, E. F.; Starch: Chemistry and Technology, 2th ed., Academic Press: N.Y., 1984, p. 1-718.
- 20. Franco, L. M. C.; Ciacco, F. C.; Starch. 1988, 40, 1.
- 21. Choonakis, S. I.; Crit. Rev. Food Sci. 1998, 38, 7.
Publication Dates
-
Publication in this collection
11 Aug 2005 -
Date of issue
Aug 2005
History
-
Accepted
16 Dec 2004 -
Received
13 Apr 2004