ABSTRACT
Permanent preservation areas (PPAs) adjacent to drainage channels may include forests, savannahs, or grassland Under a former environmental legislation ("Forest Code"), it was required to maintain a strip of native vegetation (at least 30 m wide) at the margin of any drainage channel, to support environmental functions. Under the current native vegetation protection policy, if a riparian margin was degraded prior to 2008, the landholder is required to regenerate a strip of only 5-20 m, in some cases. The present study evaluated and compared the ligneous vegetation structure in 10 preserved PPAs (following the requirements of the old law) and 10 degraded PPAs (conforming to the current environmental legislation), in Mineiros municipality, southwest Goiás. Sixty plots measuring 15 m × 30 m were delineated, and data for all ligneous individuals with a diameter at breast height (DBH) ≥ 5 cm were recorded. A total of 2,662 individuals were documented, distributed among 208 species. Of the total, 1,573 individuals belonging to 167 species were from the preserved PPAs, and 1,089 individuals across 142 species were from the degraded PPAs. According to these results, the preserved PPAs were richer in species composition than in the degraded PPAs (245.5 ± 34.36 and 213.3 ± 34.1, respectively), in addition to being more abundant in terms of individuals. The importance value index (IVI) was well distributed among the species. All the degraded PPAs presented signs of erosion. It was concluded that the reduced width of the PPAs (5-20 m) under the current native vegetation protection policy would result in a loss of the environmental functions sought by the legislation..
Keywords:
Phytosociology; Forest Code; Environmental losses
RESUMO
As Áreas de Preservação Permanente (APPs) de canais de drenagem podem se apresentar através das formações florestais, savânicas e campestres, sendo que a faixa legal de vegetação para manter suas funções ambientais era de, no mínimo 30 metros marginas (Antigo Código Florestal). Atualmente, a política de proteção da vegetação nativa prevê entre cinco a 20 metros marginais para a recomposição de determinadas áreas degradadas até 2008. Este estudo avaliou e comparou a estrutura da vegetação lenhosa de 10 APPs preservadas (considerando-se o antigo Código) e 10 APPs degradadas (exceções da atual legislação ambiental), no município de Mineiros, sudoeste Goiano. Foram definidas 60 parcelas com dimensão de 15x30 m e todos os indivíduos lenhosos com DAP ≥ 5 cm foram amostrados. Foram registrados 2.662 indivíduos distribuídos em 208 espécies, dos quais 1.573 indivíduos pertencentes a 167 espécies são das APPs preservadas e, 1.089 indivíduos distribuídos em 142 espécies são das APPs degradadas. A partir dos resultados, observou-se uma maior riqueza estimada de espécies para as APPs preservadas do que para as degradadas (245,5 ± 34,36 e 213,3 ± 34,1, respectivamente), assim como maior abundância de indivíduos. O IVI foi bem distribuído entre as espécies. Todas as APPs degradadas apresentaram processos erosivos. Conclui-se que a redução da metragem da APPs entre cinco e vinte metros marginais previstas na atual política de proteção da vegetação nativa, acarretará em perda das funções ambientais prevista na legislação.
Palavras-chave:
Fitossociologia; Código Florestal; Perdas Ambientais
1. INTRODUCTION
Currently, there is a consensus in the scientific community about a biodiversity crisis unfolding due to the conflict between conservation and socioeconomic development involving intensive human use of land with farming potential (MYERS, 1996MYERS, N. The biodiversity crisis and the future of evolution. The Environmentalist, v.16, n.1, p.37-47, 1996.). With respect to this, the Cerrado deserves to be highlighted, as it is considered a biodiversity hotspot owing to its great diversity of endemic species as well as a high degree of human interference (MYERS et al., 2000MYERS, N.; MITTERMEIER, R.A.; MITTERMEIER, C.G.; FONSECA, G.A.B.; KENT, J. Biodiversity hotspost for conservation priorities. Nature, v.403, p.853-858, 2000.). Located primarily in the central part of Brazil, the Cerrado originally covered an area of 25 million km2 (SILVA; BATES, 2002SILVA, J.M.C.; BATES, J.M. Biogeographic patterns and conservation in the South American Cerrado: a tropical savanna hotspot. BioScience, v.52, p.225-233, 2002.). It was first colonized by humans at the beginning of the 1930s, but during 1960-1970, it served as a targeted frontier for agricultural expansion, owing to credit concessions and technical modernization that expanded the range of productive areas (TEIXEIRA; HESPANHOL, 2006TEIXEIRA, J.C.; HESPANHOL, A.N. A região centro-oeste no contexto das mudanças agrícolas ocorridas no período pós-1960. Revista Eletrônica da Associação dos Geógrafos Brasileiros, v.1, n.3, p.52-66, 2006.).
Because of this progression of human occupation, the biodiversity conservation efforts in the Cerrado have been observed (DINIZ-FILHO et al., 2006DINIZ FILHO, J.A.F.; BINI, L.M.; PINTO, M.P.; RANGEL, T.F.L.V.B.; CARVALHO, P.; BASTOS, R.P. Anuran species richness, complementarity and conservation conflicts in Brazilian Cerrado. Acta Oecologica, v.27, p.9-15, 2006.; RANGEL et al., 2006RANGEL, T.F.L.V.B.; BINI, L.M.; DINIZ-FILHO, J.A.F.; PINTO, M.P. CARVALHO, P.; BASTOS, R.P. Human development and biodiversity conservation in Brazilian Cerrado. Applied Geography, v.27, p.14-27, 2006.). Increased habitat fragmentation has led to frightening estimates of biodiversity loss in Cerrado, which are not even fully mapped yet (MACHADO et al., 2004MACHADO, R.B.; RAMOS NETO, M.B.; PEREIRA, P.G.P.; CALDAS, E.F.; GONÇALVES, D.A.; SANTOS, N.S.; TABOR, K.; STEININGER, M. Estimativas de perda da área do Cerrado brasileiro. Relatório técnico não publicado. Conservação internacional, Brasília, DF: 2004.). Less than 4% of the Cerrado's area is protected as federal conservation areas (CABRAL; BRITO, 2013CABRAL, R.; BRITO, D. Temporal and spatial investments in the protected area network of a megadiverse country. Zoologia, v.30, n.2, p.177-181, 2013.). Moreover, Law 12.651 of December 2012, which replaced the old Forest Code, might further facilitate the expansion of agricultural frontiers into this biome, to the detriment of biodiversity (MARTINELLI et al., 2010MARTINELLI, L.A.; JOLY, C.A.; NOBRE, C.A.; SPAROVEK, G. A falsa dicotomia entre a preservação da vegetação natural e a produção agropecuária. Biota Neotropica, v.10, n.4, p.323-330, 2010.).
It is estimated that over the long term, such an environmental policy could significantly harm the environment, as the native vegetation areas would be converted into farmland. Under Law 12.651/2012, the rural properties that have between one and four "fiscal modules," and were deforested prior to 2008, would satisfy the permanent preservation area (PPA) requirement if they have a 5-15 m wide strip of native forest adjacent to the watercourses. Thus, legally protected areas along the margins of drainage channels are considerably reduced in size. Klink and Machado (2005)KLINK, C.A.; MACHADO, R.B. Conservation of Brazilian Cerrado. Conservation Biology, v.19, n.3, p.707-713, 2005. have emphasized the importance of conserving Cerrado; however, in the light of these recent changes, the task has become more urgent.
Currently, 7.000 species of vascular plants are found in the Cerrado (MENDONÇA et al., 1998MENDONÇA, R.C.; FELFILI, J.M.; WALTER, B.M.T. Flora vascular do Cerrado. In: SANO, S.M.; ALMEIDA, S.P. Cerrado: ambiente e flora. Planaltina: Embrapa/CPAC, 1998. p.289-556.). However, despite the high number of species in this biome, forest and phytosociological studies have started only recently, especially in the southeast region of the Goias state (CABACINHA; CASTRO, 2009CABACINHA, C.D.; CASTRO, S.S. Relationships between floristic diversity and vegetation index, forest structure and landscape metrics of fragments and species diversity in fragments of Gallery forest in Brazilian Cerrado. Forest Ecology and Management, v.257, n.10, p.2157-2165, 2009.; CARNEIRO et al., 2011CARNEIRO, G.T.; CABACINHA, C.D.; FARIA, K.M.S.; SIQUEIRA, M.N.; LIMA, J.C.S. Cobertura Florestal do Município de Rio Verde, GO: estrutura e composição da paisagem entre 2005 e 2008. Geografia, v.36, p.335-357, 2011.). Considering the recent changes to the Brazilian Forest Code, studies that document the ecological parameters of plant communities for comparing the scenario under the old forest code (with the minimum requirement of a 30-m strip along the margin) might be useful for exploring the possibilities of conservation in the biome under the current policy (with the minimum requirement of 5-20 m strip for certain areas that are already altered).
PPAs adjacent to water resources present diverse phytophysiognomies. Ribeiro and Walter (2008)RIBEIRO, J.F.; WALTER, B.M.T. As principais fitofisionomias do bioma cerrado. In: SANO, S.M.; ALMEIDA, S.P.; RIBEIRO, J.F. Cerrado: ecologia e flora. Brasília: EMBRAPA Cerrados, 2008. p.151-212. describe gallery forests, riparian forests, and wetlands as phytophysiognomies associated with drainage channels. It is worth highlighting the fact that when natural habitats are close to human influence, there might be many physical, biotic, and ecological alterations that adversely affect the biota (MURCIA, 1995MURCIA, C. Edge effects in fragmented forest: implication for conservation. Trend in Ecology and Evolution, v.10, n.2, p.58-62, 1995.). However, there is a scarcity of studies that show the alterations to ligneous vegetation in PPAs, especially ones that are less than 30 m wide, which are described here as degraded.
Cabacinha and Castro (2009)CABACINHA, C.D.; CASTRO, S.S. Relationships between floristic diversity and vegetation index, forest structure and landscape metrics of fragments and species diversity in fragments of Gallery forest in Brazilian Cerrado. Forest Ecology and Management, v.257, n.10, p.2157-2165, 2009. and Cabacinha et al. (2010)CABACINHA, C.D.; CASTRO, S.S.; GONÇALVES, D.A. Análise da estrutura da paisagem da alta bacia do Rio Araguaia na savana brasileira. Floresta, v.40, n.4, p.675-690, 2010. carried out spatial and ecological studies of forest vegetation around drainage channels in mid-2007 and 2008 near the sources of the Araguaia River, in the city of Mineiros, Goiás. This was before the new law (12.651/2012) came into existence. The authors observed that a great part of the vegetation was in a good state from a conservation viewpoint, and some fragmented environments could still be restored.
According to the new legislation (Law 12.651/2012), PPAs can be defined as follows: "protected area covered or not by native vegetation, with the environmental function of preserving water resources, landscape, geological stability, and biodiversity, to facilitate the genetic flux of flora and fauna, protect the soil, and ensure the well-being of human populations." In this context, the present study had the objective of evaluating whether there are ecological differences between the drainage channel PPAs with different degrees of preservation, in Mineiros, Goiás. Specifically, this study investigated whether the abundance, diversity, and species richness in ligneous vegetation differed between PPAs in which a 30 m margin of native vegetation was in place (in a good state of preservation) and PPAs where human interference was evident within the 30 m vegetation margin (also in a bad state of preservation).
2. MATERIALS AND METHODS
The study region is in the city of Mineiros, located in southwest Goiás, Brazil. The region is ecologically important, as it is home to the sources of the Araguaia and Paranaíba rivers, the most important hydrographic basins in the region. The municipality has an approximate area of 900,000 ha and borders the municipalities of Jataí, Santa Rita do Araguaia, Serranópolis, Portelândia, Chapadão do Céu, and Caiapônia in Goiás, the municipalities of Alto Taquari, Alto Araguaia, and Ponte Branca of the state of Mato Grosso, and the municipality of Costa Rica in the state of Mato Grosso do Sul (IBGE, 2010INSTITUTO BRASILEIRO DE GEOGRAFIA E ESTATÍSTICA - IBGE. Cidades. 2010. Disponível em: http://www.ibge.gov.br/cidadesat/painel/painel.phpAcesso em: 02 maio. 2013.
http://www.ibge.gov.br/cidadesat/painel/...
). The Mineiros municipality has a population of approximately 53,000 people (IBGE, 2010INSTITUTO BRASILEIRO DE GEOGRAFIA E ESTATÍSTICA - IBGE. Cidades. 2010. Disponível em: http://www.ibge.gov.br/cidadesat/painel/painel.phpAcesso em: 02 maio. 2013.
http://www.ibge.gov.br/cidadesat/painel/...
). The natural habitats in this region have been transformed for human use, primarily farming (CABACINHA and CASTRO, 2009CABACINHA, C.D.; CASTRO, S.S. Relationships between floristic diversity and vegetation index, forest structure and landscape metrics of fragments and species diversity in fragments of Gallery forest in Brazilian Cerrado. Forest Ecology and Management, v.257, n.10, p.2157-2165, 2009.; CABACINHA et al., 2010CABACINHA, C.D.; CASTRO, S.S.; GONÇALVES, D.A. Análise da estrutura da paisagem da alta bacia do Rio Araguaia na savana brasileira. Floresta, v.40, n.4, p.675-690, 2010.; PRADO et al., 2012PRADO, L.A.; MIZIARA, F.E.; FERREIRA, M.E. Expansão da fronteira agrícola e mudanças no uso do solo na região sul de Goiás: ação antrópica e características naturais do espaço. Boletim Goiano de Geografia, v.32, n.1, p.151-165, 2012.).
The climate in the study region is rainy tropical, type Aw (hot and humid with a wet summer and dry winter), with temperatures between 18-32 °C and an average annual rainfall of 1700 mm (CARNEIRO et al., 2009CARNEIRO, M.A.C.; SOUZA, E.D.; REIS, E.F.; PEREIRA, H.S.; AZEVEDO, W.R. Atributos físicos, químicos e biológicos do solo do Cerrado sob diferentes sistemas de uso e manejo. Revista Brasileira de Ciência do Solo, v.33, n.1, p.147-157, 2009.). The topography of the region varies from flat to slightly hilly, with extensive areas of Latosol (40.27% of the municipality) and Neosol soils (31.91%) (RADAMBRASIL, 1982RADAMBRASIL. Levantamentos dos Recursos Naturais. Ministério das Minas e Energia, Secretária Geral. Projeto RADAMBRASIL - Folha SD 21 Cuiabá. V. 26. 540 p, 1982.), allowing easy use of farm machinery.
The sampling of ligneous vegetation took place in July 2013, and during this period, phytosociological surveys were performed in the PPAs following the random methodology proposed by Felfili et al. (2011)FELFILI, J.M.; ROITAM, I.; MEDEIROS, M.M.; SANCHEZ, M. Procedimentos e métodos de amostragem de vegetação. In: FELFILI, J.M.; EISENLOHR, P.V.; MELO, M.M.R.F.; ANDRADE, L.A.; MEIRA NETO, J.A.A. Fitossociologia no Brasil: métodos e estudos de casos. Viçosa, MG: UFV, 2011.. The sampling design took in 20 PPAs in the study area, 10 in a good state of preservation (having 30 m of native vegetation at the margins of streams that are up to 10 m wide) and 10 degraded (having human interference within 10-25 m of streams that are up to 10 m wide). This analysis allows a comparison of the plant community structure within the PPAs that are in accord with the old and the current legislation regarding the protection of native vegetation (Law 4.771/1965 and Law 12.651/2012, respectively).
The current law permits the continuation of human activities within the 30-m margin of watercourses, such as agrosilvopastoral activities, ecotourism, and rural tourism, if already converted for that use by July 22, 2008, and the property has between one and four fiscal modules (or more - see point 4 below). Such areas must restore a strip along the watercourse to the following widths to make up the PPA: 1) if the property is one fiscal module, only 5 m need to be restored, 2) if the property has greater than 1 (one) module and less than 2 (two) modules, 8 m need to be restored, 3) if the property has more than 2 (two) modules and less than 4 (four) modules, 15 m need to be restored, 4) if the property has more than 4 (four) modules, a minimum of 20 m and a maximum of 100 m need to be restored (Law 12.651/2012).
Given that the cited exceptions in relation to PPAs deforested or converted to human use up to 2008 might continue that use, this study considers PPAs as degraded if they currently maintain some agrosilvopastoral, ecotourism, or rural tourism activity within a 30-m margin of a drainage channel that is up to 10 m wide, independent of the year in which the occupation/deforestation first occurred and independent of the area of the property in terms of fiscal modules. PPAs with a maintained 30 m wide margin were considered as preserved. By using this standard, it was possible to test the hypothesis that there may be differences between the PPAs with a preserved 30 m margin and those with some other use of the soil within this margin. For the survey of arboreal vegetation, the method of plots proposed by Felfili et al. (2011)FELFILI, J.M.; ROITAM, I.; MEDEIROS, M.M.; SANCHEZ, M. Procedimentos e métodos de amostragem de vegetação. In: FELFILI, J.M.; EISENLOHR, P.V.; MELO, M.M.R.F.; ANDRADE, L.A.; MEIRA NETO, J.A.A. Fitossociologia no Brasil: métodos e estudos de casos. Viçosa, MG: UFV, 2011. was used - delineating three plots in each preserved PPA and another three in each degraded PPA. Overall, 60 plots were used, 30 in preserved PPAs and 30 in degraded PPAs. The PPAs were chosen randomly within the municipality of Mineiros. Each plot measured 30 m × 15 m, totaling 2.7 ha of sample area.
In each plot, all individual trees with a diameter at breast height (DBH) greater than or equal to 5 cm were assessed in accordance with the recommendations of Scolforo (1998)SCOLFORO, J.R.S. Manejo florestal. Lavras: UFLA/FAEPE, 1998. 438p.. Characterization of each plot was done, recording the location of the first sample plot in the PPA, the approximate width of the vegetation strip, the matrix where the PPA is situated, as well as the presence or absence of erosive processes (Table 1), given that the other two plots within each PPA were random in relation to the first.
Characterization of the 20 permanent preservation areas (PPAs) investigated (10 preserved and 10 degraded) in the Mineiros municipality, Goiás, Brazil. (DN: degraded north; DS: degraded south; PN: preserved north; PS: preserved south; MG: mata de galeria (gallery forest); MC: mata ciliar (riparian forest); MGI: mata de galeria inundável (flooded gallery forest); C: cerradão (humid cerrado).
Tabela 1
Caracterização das 20 APPs investigadas (10 preservadas e 10 degradadas) no município de Mineiros, Estado de Goiás, Brasil. (DN: degradada norte; DS: degradada sul; PN: preservada norte; PS: preservada sul; MG: mata de galeria; MC: mata ciliar; MGI: mata de galeria inundável; C: cerradão).
In addition, a comparative study was carried out between the preserved PPAs and those having some type of human interference through phytosociological data, as recommended by Mueller-Dombois and Ellenberg (1974)MUELLER-DOMBOIS, D.; ELLENBERG, H. Aims and methods of vegetation ecology. New York: Willey & Sons, 1974. 557p.. The parameters considered were absolute density, absolute frequency, absolute dominance expressed in basal area, relative density, relative frequency, relative dominance, and importance value. Also calculated were the Shannon biodiversity index (H2 ) and the Pielou evenness index (J2 ), in accordance with Brower and Zar (1984)BROWER, J.E.; ZAR, J.H. Field and laboratory methods for general ecology. Dubuque: W.M.C. Brow, 1984. 226p.. To verify if there was a difference in the total abundance of individuals and the Shannon diversity index between the two categories of PPA, Student's t-test was used with a 5% significance level, following Zar (1996)ZAR, J.H. Biostatistical analysis. Englewood Cliffs: Prentice-Hall, 1996..
To estimate the species richness in the sampled PPAs, the methodology proposed by Coddington et al. (1991)CODDINGTON, J.A.; GRISWOLD, C.E.; SILVA, D.; PEQARANDA, E.; LARCHER, S.F. Designing and testing sampling protocols to estimate biodiversity in tropical ecosystems. In: DUDLEY, E.C. The unity of evolutionary biology. In: INTERNATIONAL CONGRESS OF SYSTEMATIC AND EVOLUTIONARY BIOLOGY, 4., 1991, Portland. Proceedings... Portland: Dioscorides Press, 1991. p.44-60. and Colwell and Coddington (1994)COLWELL, R.K.; CODDINGTON, J.A. Estimating terrestrial biodiversity through extrapolation. Philosophical Transactions of the Royal Society of London. Series B, Biological Sciences, v.345, p.101-118, 1994. was used, in which the non-parametric first order Jackknife estimator was considered, using 100 randomizations. This estimate extrapolates the observed richness through the frequency of rare species ("unique") present in the samples, and produces an accurate estimate of the richness of the community (KREBS, 1999KREBS, C.J. Ecological methodology. Menlo Park: Addison Wesley Longman, 1999. 620p.). In addition, such a procedure provides a confidence interval, allowing statistical comparisons between two or more sample locations. Finally, the estimated species richness was tested for the preserved and degraded PPAs, and the difference was calculated using the confidence intervals.
3. RESULTS
Data for 2,662 ligneous individuals belonging to 208 plant species were recorded. In the preserved PPAs, 1,573 individuals from 167 species were documented, while in the degraded PPAs, 1,089 individuals from 142 species were documented. A total of 150 dead individuals were also surveyed, 77 of which were in preserved PPAs (4.66% of the total) and 73 were in degraded PPAs (6.28% of the total). According to the Jackknife 1 estimator, species richness for the preserved and degraded PPAs was 245.5 ± 34.36 and 213.3 ± 34.1, respectively (Figure 1).
A) Species accumulation curve (observed and estimated richness—Jackknife 1) of ligneous plants in the preserved permanent preservation areas (PPAs), Mineiros municipality, Goiás, Brazil. B) Species accumulation curve (observed and estimated richness—Jackknife 1) of ligneous plants in the degraded PPAs, Mineiros municipality, Goiás, Brazil.
Figura 1
A) Curva de acumulação de espécies (riqueza observada e estimada – Jackknife 1) de plantas lenhosas nas APPs preservadas, Município de Mineiros, estado de Goiás, Brasil. B) Curva de acumulação de espécies (riqueza observada e estimada – Jackknife 1) de plantas lenhosas nas APPs degradadas, Município de Mineiros, estado de Goiás, Brasil.
Twenty species from the preserved and degraded environments, which had the highest values of importance, are listed in Table 02. In the preserved PPAs, the 10 species with the highest values of importance were Protium spruceanum (19.33), Tapirira guianensis (15.88), Attalea phalerata (15.69), Mauritia flexuosa (7.71), Xylopia emarginata (7.63), Hieronyma alchorneoides (6.94), Copaifera langsdorffii (6.26), Ilex affinis (6.62), Hirtella gracilipes (6.03), and Ormosia paraensis (5.23); these make up 32.29% of the importance value of these PPAs. On the other hand, in the degraded PPAs, the 10 species with the greatest values of importance constituted 33.34% of the total value, namely, Tapirira guianensis (20.40), Hirtella gracilipes (16.64), Tachigali vulgaris (16.56), Copaifera langsdorffii (10.59), Simarouba amara (8.18), Calophyllum brasiliensis (7.13), Protium spruceanum (6.89), Coussarea hydrangeifolia (6.74), Licania sp. (6.49), and Vochysia pyramidalis (6.38) (Table 02).
Phytosociological values of 20 species with the greatest importance values in the preserved and the degraded permanent preservation areas (PPAs), in decreasing order of IVI (N.I.: number of individuals; N.P.: number of plots; DR: relative density (%); DoR: dominância relative (relative dominance) (%); and IVI: importance value index).
Tabela 2
Valores fitossociológicos das 20 espécies que apresentaram maior valor de importância nas APPs preservadas em ordem decrescente de IVI e nas APPs degradadas (N.I.: número de indivíduos; N.P.: número de parcelas; DR: densidade relativa (%); DoR: dominância relativa (%) e IVI: índice de valor de importância).
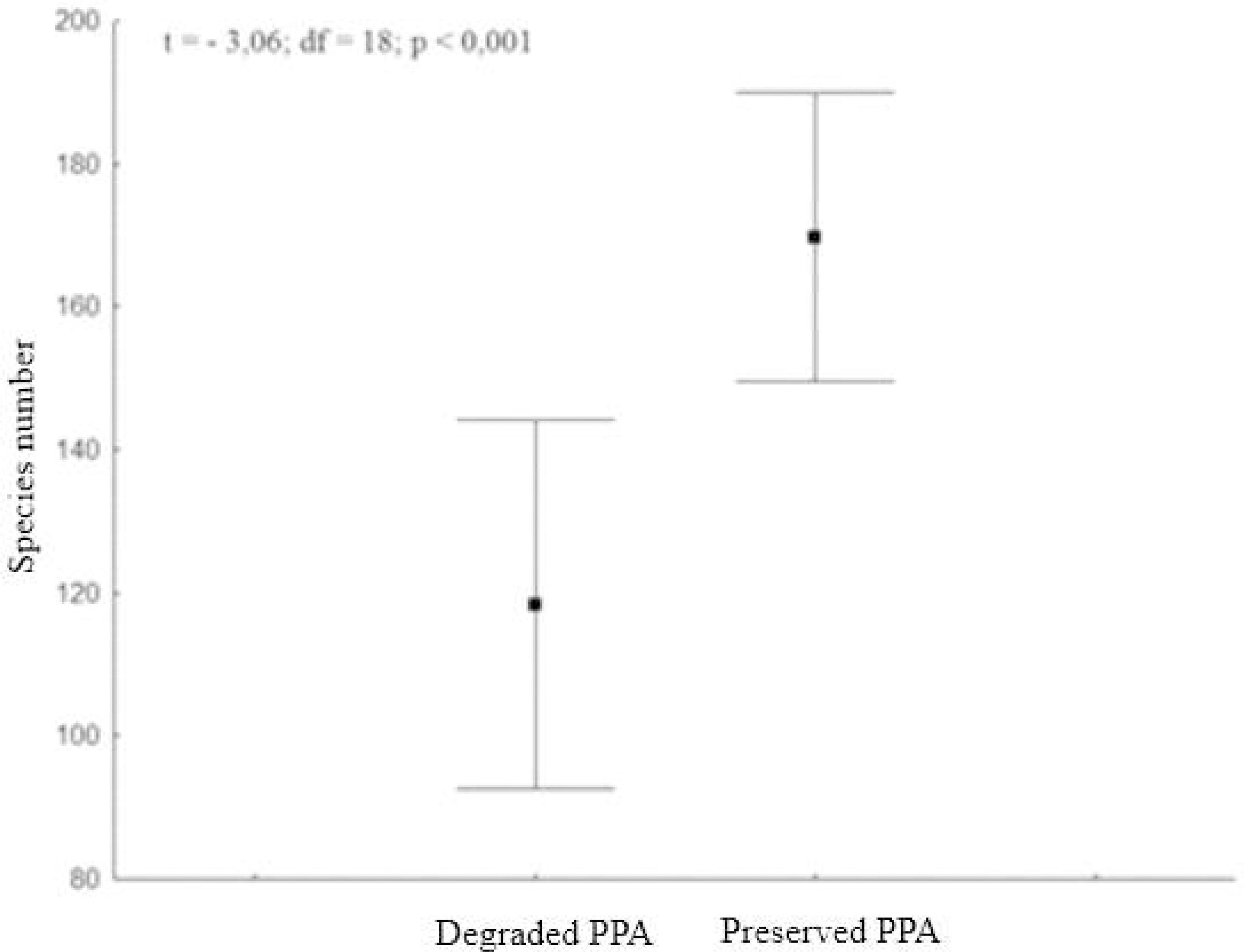
The preserved PPAs had a Shannon diversity index and Pielou evenness index equal to 4.29 nats. ind-1 and 0.839, respectively. On the other hand, the Shannon diversity index and Pielou evenness index in the degraded PPAs were 4.24 nats. ind-1 and 0.857, respectively. It was observed that the diversity index (t = 0.093; df = 58; p = 0.926) did not differ between the preserved and the degraded PPAs. The average number of individuals encountered in the preserved PPAs was greater than that recorded in the degraded PPAs (t = -3.06; df = 58; p < 0.001) (Figure 02), as in the preserved PPAs, there were an average of 50 individuals more than in the degraded PPAs.
Result of Student’s t-test comparing the abundance of individuals in the PPAs preserved and degraded.
Figura 2
Resultado do teste t de Student comparando a abundância de indivíduos das APPs preservadas e degradadas.
4. DISCUSSION
In the present study, both the number of individuals and the estimated species richness were greater in the PPAs with a 30 m margin of vegetation strip than in the PPAs with a strip width of 5-20 m. Therefore, the results are evidence that the reduction in vegetation cover in PPAs correlates negatively with the ecological aspects of the ligneous vegetation in such environments. According to Law 12.651/2012, PPAs are important areas for species maintenance and consequently, various ecosystem services. However, this law may bring alterations in the structure and functioning of the existing PPAs in Brazil, as demonstrated above.
Apart from the environmental losses related to flora, in all the PPAs that had human intrusion into the 30-m margin, there were erosive processes. According to Galindo et al. (2008)GALINDO, I.C.L.; RIBEIRO, M.R.; SANTOS, M.F.A.V.; LIMA, J.F.W.F.; FERREIRA, R.F.A.L. Relações solo-vegetação em áreas sob processo de desertificação no município de Jataúba, PE. Revista Brasileira de Ciências do Solo, v.32, p.1283-1296, 2008., there is a negative relationship between vegetation and erosion, thus, greater the index of vegetation cover, lesser would be the occurrence of erosive processes. In addition, Klink and Machado (2005)KLINK, C.A.; MACHADO, R.B. Conservation of Brazilian Cerrado. Conservation Biology, v.19, n.3, p.707-713, 2005. highlight the fact that erosion is one of the greatest environmental problems caused by changes in the Cerrado, and is one of the major threats to biodiversity. The presence of erosion in the degraded PPAs of the Mineiros municipality reinforces the importance of protecting these soils and in general, the water resources.
In the literature, there is a great deal of discussion with respect to the recent changes in the Brazilian Forest Code (AB'SÁBER, 2010AB'SÁBER, A.N. Do Código Florestal para o Código da Biodiversidade. Biota Neotropica, v.10, n.4, p.331-337, 2010.; BRANCALION; RODRIGUES, 2010BRANCALION, P.H.S.; RODRIGUES, R.R. Implicações do cumprimento do Código Florestal vigente na redução de áreas agrícolas: um estudo de caso da produção canavieira no Estado de São Paulo. Biota Neotropica, v.10, n.4, p.63-66, 2010.; CASATTI, 2010CASATTI, L. Alterações no Código Florestal Brasileiro: impactos potenciais sobre a ictiofauna. Biota Neotropica, v.10, n.4, p.31-34, 2010.; DEVELEY; PONGILUPPI, 2010DEVELEY, P.F.; PONGILUPPI, T. Impactos potenciais na avifauna decorrentes das alterações propostas para o Código Florestal Brasileiro. Biota Neotropica, v.10, n.4, p.43-46, 2010.; TOLEDO et al., 2010TOLEDO, L.F.; CARVALHO-E-SILVA, S.P.; SÁNCHEZ, C.; ALMEIDA, M.A.; HADDAD, C.F.B. A revisão do Código Florestal Brasileiro: impactos negativos para a conservação dos Anfíbios. Biota Neotropica, v.10, n.4, p.35-38, 2010.; TUNDISI; MATSUMURA-TUNDISI, 2010TUNDISI, J.G.; MATSUMURA-TUNDISI, T.M. Impactos potenciais das alterações do Código Florestal nos recursos hídricos. Biota Neotropica, v.10, n.4, p.67-76, 2010.), in which a false dichotomy is apparent between the conservation of native vegetation and farming, which according to Martinelli et al. (2010)MARTINELLI, L.A.; JOLY, C.A.; NOBRE, C.A.; SPAROVEK, G. A falsa dicotomia entre a preservação da vegetação natural e a produção agropecuária. Biota Neotropica, v.10, n.4, p.323-330, 2010., affects various ecological communities. With respect to the ligneous vegetation in PPAs, there are few studies discussing the possible alterations resulting from the changes in legislation (PINHEIRO et al., 2015PINHEIRO, M.H.O.; CARVALHO, L.N.; ARRUDA, R.S.; GUILHERME, F.A.G. Consequences of suppressing natural vegetation in drainage areas for freshwater ecosystem conservation: considerations on the new 'Brazilian forest code'. Acta Botanica Brasilica, v.29, n.2, p.262-269, 2015.). The present study promotes such a discussion, given that the results show differences in the ecological parameters between the preserved and degraded PPAs; the observed and estimated species richness and abundance of individuals is greater in the preserved PPAs.
The efficacy of the old Forest Code in protecting arboreal vegetation in gallery forests in the Federal District was evaluated by Silva Júnior (2001)SILVA JÚNIOR, M.C. Comparação entre matas de galeria no distrito federal e a efetividade do código florestal na proteção de sua diversidade arbórea. Acta Botanica Braslica, v.15, n.1, p.139-146. 2001., who concluded that it was inefficient in protecting the diversity of arboreal species in gallery forests. This author showed that the soil humidity is an important factor in defining degrees of clustering of certain species in a gallery forest. Therefore, he found forest communities peculiar to sites with humid, intermediate, and dry soils. Species grouped in the intermediate and dry sites advanced beyond the 30 m margin of the PPA in many cases. In light of this, the data of the Mineiros study support the notion that the new law for native vegetation protection may result in losses of species that are exclusive to areas further from the water course, when using narrower margins based on the fiscal modules of the property.
As discussed above, the estimated species richness (from Jackknife 1 estimator) in the preserved and degraded PPAs was greater than the observed species richness. Therefore, only 68.02% and 66.57% of the possible species present in such PPAs were recorded. Silva et al. (2009)SILVA, A.C.; van den BERG, E.; HIGUCHI, P.; NUNES, M.H. Florística e estrutura da comunidade arbórea em fragmentos de floresta aluvial em São Sebastião da Bela Vista, Minas Gerais, Brasil. Revista Brasileira de Botânica, v.32, n.2, p.283-297, 2009., while studying the structure of arboreal communities in alluvial, human-influenced forests in São Sebastião da Bela Vista (MG), found that 82.4% of the richness was estimated. In the State Park of Araguaia (Parque Estadual do Araguaia) (MT), Barbosa et al. (2011)BARBOSA, D.C.F.; MARIMON, B.S.; LENZA, E.; MARINOM JR, B.H.; OLIVEIRA, E.A.; MARACAHIPES, L. Estrutura da vegetação lenhosa em dois fragmentos naturais de florestas inundáveis (Impucas) no Parque Estadual do Araguaia, Mato Grosso. Revista Árvore, v.35, n.3, p.457-471, 2011. did a comparative analysis of the structure of ligneous vegetation in two fragments of forests subject to flooding (impucas), which were PPAs as well, one preserved and the other one degraded. In keeping with the results of the present study, Barbosa et al. (2011)BARBOSA, D.C.F.; MARIMON, B.S.; LENZA, E.; MARINOM JR, B.H.; OLIVEIRA, E.A.; MARACAHIPES, L. Estrutura da vegetação lenhosa em dois fragmentos naturais de florestas inundáveis (Impucas) no Parque Estadual do Araguaia, Mato Grosso. Revista Árvore, v.35, n.3, p.457-471, 2011. found differences during the ecological analyses between the preserved and the degraded fragments, with greater species richness and individual density in the preserved PPAs, and greater evenness of individual distribution by species in the degraded PPAs. Martins et al. (2008)ARTINS, S.V.; BRITO, E.R.; OLIVEIRA FILHO, A.T.; SILVA, A.F. SILVA, E. Floristic composition of two wetland forests in Araguaian Plain, State of Tocantins, Brazil, and comparison with other areas. Revista Árvore, v.32, n.1, p.129-141, 2008. also analyzed the structure of forest composition in two forests subject to flooding (impucas), one preserved and one degraded, in the lower reaches of the Araguaia River, in Tocantins. They also showed a greater individual density of individuals, numbers of species, as well as Shannon diversity index in the preserved PPAs than in the degraded ones.
With respect to the number of dead individuals in the preserved PPAs (4.66% of the total) and degraded PPAs (6.28% of the total), it is interesting to note that Toniato et al. (1998)TONIATO, M.T.Z.; LEITÃO FILHO, H.F.; RODRIGUES, R.R. Fitossociologia de um remanescente de floresta higrófila (mata de brejo) em Campinas, SP. Revista Brasileira de Botânica, v.21, n.2, p.197-210, 1998. compiled the results of five forest surveys, in which the rate of dead individuals varied between 5-8% of the total of individuals sampled. Murcia (2005)MURCIA, C. Edge effects in fragmented forest: implication for conservation. Trend in Ecology and Evolution, v.10, n.2, p.58-62, 1995. emphasized that high arboreal mortality may point to a process of exclusion, characteristic of fragmented forests, owing to physical and biological alterations created by border effects.
5. CONCLUSIONS
Conserving the water resources, landscape, geological stability, and biodiversity, facilitating the genetic flux within fauna and flora, protecting the soil, and ensuring the well-being of human populations are interdependent functions; compromising any one creates a disequilibrium in others. In this context, the ecological parameters evaluated indicate that the establishment of PPAs in accordance with the current law for protection of native vegetation (Law 12.651/2012) might result in significant losses of species and individuals, as well as facilitate the erosive processes. This suggests that, in the long term, such PPAs will not fulfill the environmental functions foreseen in the legislation. Thus, in the long term, environmental losses with respect to forest diversity, density of individuals, and erosive processes might occur in the PPAs that have human intervention within the 30 m margin.
6. ACKNOWLEDGMENTS
Our sincere thanks to Sebastião de Souza Silva (IBGE/Goiânia staff member) for help in identifying the botanical species in the field, to Dra. Gisele Cristina de Oliveira Menino (IF Goiano-Rio Verde) for help in identifying the collected botanical samples, to LABOGEF/UFG for coordinating the project, and to CIAMB/UFG for promoting scientific investigation. MNS thanks the CNPq (Conselho Nacional de Desenvolvimento Científico e Tecnológico) for her scholarships; SSC thanks the CNPq and FAPEG (Fundação de Amparo à Pesquisa do Estado de Goiás) for her scholarships and financial support to the project (Universal Project no. 479010-9); ARM thanks the CAPES (Coordenação de Aperfeiçoamento de Pessoal de Nível Superior) and FAPEG for the Postdoctoral fellowships. The authors thank to PPGEO/CAPES/FAPEG for financial support.
7. REFERENCES
- AB'SÁBER, A.N. Do Código Florestal para o Código da Biodiversidade. Biota Neotropica, v.10, n.4, p.331-337, 2010.
- BARBOSA, D.C.F.; MARIMON, B.S.; LENZA, E.; MARINOM JR, B.H.; OLIVEIRA, E.A.; MARACAHIPES, L. Estrutura da vegetação lenhosa em dois fragmentos naturais de florestas inundáveis (Impucas) no Parque Estadual do Araguaia, Mato Grosso. Revista Árvore, v.35, n.3, p.457-471, 2011.
- BRANCALION, P.H.S.; RODRIGUES, R.R. Implicações do cumprimento do Código Florestal vigente na redução de áreas agrícolas: um estudo de caso da produção canavieira no Estado de São Paulo. Biota Neotropica, v.10, n.4, p.63-66, 2010.
- BROWER, J.E.; ZAR, J.H. Field and laboratory methods for general ecology Dubuque: W.M.C. Brow, 1984. 226p.
- CABACINHA, C.D.; CASTRO, S.S. Relationships between floristic diversity and vegetation index, forest structure and landscape metrics of fragments and species diversity in fragments of Gallery forest in Brazilian Cerrado. Forest Ecology and Management, v.257, n.10, p.2157-2165, 2009.
- CABACINHA, C.D.; CASTRO, S.S.; GONÇALVES, D.A. Análise da estrutura da paisagem da alta bacia do Rio Araguaia na savana brasileira. Floresta, v.40, n.4, p.675-690, 2010.
- CABRAL, R.; BRITO, D. Temporal and spatial investments in the protected area network of a megadiverse country. Zoologia, v.30, n.2, p.177-181, 2013.
- CARNEIRO, G.T.; CABACINHA, C.D.; FARIA, K.M.S.; SIQUEIRA, M.N.; LIMA, J.C.S. Cobertura Florestal do Município de Rio Verde, GO: estrutura e composição da paisagem entre 2005 e 2008. Geografia, v.36, p.335-357, 2011.
- CARNEIRO, M.A.C.; SOUZA, E.D.; REIS, E.F.; PEREIRA, H.S.; AZEVEDO, W.R. Atributos físicos, químicos e biológicos do solo do Cerrado sob diferentes sistemas de uso e manejo. Revista Brasileira de Ciência do Solo, v.33, n.1, p.147-157, 2009.
- CASATTI, L. Alterações no Código Florestal Brasileiro: impactos potenciais sobre a ictiofauna. Biota Neotropica, v.10, n.4, p.31-34, 2010.
- CODDINGTON, J.A.; GRISWOLD, C.E.; SILVA, D.; PEQARANDA, E.; LARCHER, S.F. Designing and testing sampling protocols to estimate biodiversity in tropical ecosystems. In: DUDLEY, E.C. The unity of evolutionary biology. In: INTERNATIONAL CONGRESS OF SYSTEMATIC AND EVOLUTIONARY BIOLOGY, 4., 1991, Portland. Proceedings.. Portland: Dioscorides Press, 1991. p.44-60.
- COLWELL, R.K.; CODDINGTON, J.A. Estimating terrestrial biodiversity through extrapolation. Philosophical Transactions of the Royal Society of London. Series B, Biological Sciences, v.345, p.101-118, 1994.
- DEVELEY, P.F.; PONGILUPPI, T. Impactos potenciais na avifauna decorrentes das alterações propostas para o Código Florestal Brasileiro. Biota Neotropica, v.10, n.4, p.43-46, 2010.
- DINIZ FILHO, J.A.F.; BINI, L.M.; PINTO, M.P.; RANGEL, T.F.L.V.B.; CARVALHO, P.; BASTOS, R.P. Anuran species richness, complementarity and conservation conflicts in Brazilian Cerrado. Acta Oecologica, v.27, p.9-15, 2006.
- FELFILI, J.M.; ROITAM, I.; MEDEIROS, M.M.; SANCHEZ, M. Procedimentos e métodos de amostragem de vegetação. In: FELFILI, J.M.; EISENLOHR, P.V.; MELO, M.M.R.F.; ANDRADE, L.A.; MEIRA NETO, J.A.A. Fitossociologia no Brasil: métodos e estudos de casos. Viçosa, MG: UFV, 2011.
- GALINDO, I.C.L.; RIBEIRO, M.R.; SANTOS, M.F.A.V.; LIMA, J.F.W.F.; FERREIRA, R.F.A.L. Relações solo-vegetação em áreas sob processo de desertificação no município de Jataúba, PE. Revista Brasileira de Ciências do Solo, v.32, p.1283-1296, 2008.
- INSTITUTO BRASILEIRO DE GEOGRAFIA E ESTATÍSTICA - IBGE. Cidades 2010. Disponível em: http://www.ibge.gov.br/cidadesat/painel/painel.phpAcesso em: 02 maio. 2013.
» http://www.ibge.gov.br/cidadesat/painel/painel.php - KLINK, C.A.; MACHADO, R.B. Conservation of Brazilian Cerrado. Conservation Biology, v.19, n.3, p.707-713, 2005.
- KREBS, C.J. Ecological methodology Menlo Park: Addison Wesley Longman, 1999. 620p.
- MACHADO, R.B.; RAMOS NETO, M.B.; PEREIRA, P.G.P.; CALDAS, E.F.; GONÇALVES, D.A.; SANTOS, N.S.; TABOR, K.; STEININGER, M. Estimativas de perda da área do Cerrado brasileiro. Relatório técnico não publicado. Conservação internacional, Brasília, DF: 2004.
- MARTINELLI, L.A.; JOLY, C.A.; NOBRE, C.A.; SPAROVEK, G. A falsa dicotomia entre a preservação da vegetação natural e a produção agropecuária. Biota Neotropica, v.10, n.4, p.323-330, 2010.
- ARTINS, S.V.; BRITO, E.R.; OLIVEIRA FILHO, A.T.; SILVA, A.F. SILVA, E. Floristic composition of two wetland forests in Araguaian Plain, State of Tocantins, Brazil, and comparison with other areas. Revista Árvore, v.32, n.1, p.129-141, 2008.
- MENDONÇA, R.C.; FELFILI, J.M.; WALTER, B.M.T. Flora vascular do Cerrado. In: SANO, S.M.; ALMEIDA, S.P. Cerrado: ambiente e flora. Planaltina: Embrapa/CPAC, 1998. p.289-556.
- MUELLER-DOMBOIS, D.; ELLENBERG, H. Aims and methods of vegetation ecology New York: Willey & Sons, 1974. 557p.
- MURCIA, C. Edge effects in fragmented forest: implication for conservation. Trend in Ecology and Evolution, v.10, n.2, p.58-62, 1995.
- MYERS, N. The biodiversity crisis and the future of evolution. The Environmentalist, v.16, n.1, p.37-47, 1996.
- MYERS, N.; MITTERMEIER, R.A.; MITTERMEIER, C.G.; FONSECA, G.A.B.; KENT, J. Biodiversity hotspost for conservation priorities. Nature, v.403, p.853-858, 2000.
- PINHEIRO, M.H.O.; CARVALHO, L.N.; ARRUDA, R.S.; GUILHERME, F.A.G. Consequences of suppressing natural vegetation in drainage areas for freshwater ecosystem conservation: considerations on the new 'Brazilian forest code'. Acta Botanica Brasilica, v.29, n.2, p.262-269, 2015.
- PRADO, L.A.; MIZIARA, F.E.; FERREIRA, M.E. Expansão da fronteira agrícola e mudanças no uso do solo na região sul de Goiás: ação antrópica e características naturais do espaço. Boletim Goiano de Geografia, v.32, n.1, p.151-165, 2012.
- RADAMBRASIL. Levantamentos dos Recursos Naturais. Ministério das Minas e Energia, Secretária Geral. Projeto RADAMBRASIL - Folha SD 21 Cuiabá. V. 26. 540 p, 1982.
- RANGEL, T.F.L.V.B.; BINI, L.M.; DINIZ-FILHO, J.A.F.; PINTO, M.P. CARVALHO, P.; BASTOS, R.P. Human development and biodiversity conservation in Brazilian Cerrado. Applied Geography, v.27, p.14-27, 2006.
- RIBEIRO, J.F.; WALTER, B.M.T. As principais fitofisionomias do bioma cerrado. In: SANO, S.M.; ALMEIDA, S.P.; RIBEIRO, J.F. Cerrado: ecologia e flora. Brasília: EMBRAPA Cerrados, 2008. p.151-212.
- SCOLFORO, J.R.S. Manejo florestal Lavras: UFLA/FAEPE, 1998. 438p.
- SILVA JÚNIOR, M.C. Comparação entre matas de galeria no distrito federal e a efetividade do código florestal na proteção de sua diversidade arbórea. Acta Botanica Braslica, v.15, n.1, p.139-146. 2001.
- SILVA, A.C.; van den BERG, E.; HIGUCHI, P.; NUNES, M.H. Florística e estrutura da comunidade arbórea em fragmentos de floresta aluvial em São Sebastião da Bela Vista, Minas Gerais, Brasil. Revista Brasileira de Botânica, v.32, n.2, p.283-297, 2009.
- SILVA, J.M.C.; BATES, J.M. Biogeographic patterns and conservation in the South American Cerrado: a tropical savanna hotspot. BioScience, v.52, p.225-233, 2002.
- TEIXEIRA, J.C.; HESPANHOL, A.N. A região centro-oeste no contexto das mudanças agrícolas ocorridas no período pós-1960. Revista Eletrônica da Associação dos Geógrafos Brasileiros, v.1, n.3, p.52-66, 2006.
- TOLEDO, L.F.; CARVALHO-E-SILVA, S.P.; SÁNCHEZ, C.; ALMEIDA, M.A.; HADDAD, C.F.B. A revisão do Código Florestal Brasileiro: impactos negativos para a conservação dos Anfíbios. Biota Neotropica, v.10, n.4, p.35-38, 2010.
- TONIATO, M.T.Z.; LEITÃO FILHO, H.F.; RODRIGUES, R.R. Fitossociologia de um remanescente de floresta higrófila (mata de brejo) em Campinas, SP. Revista Brasileira de Botânica, v.21, n.2, p.197-210, 1998.
- TUNDISI, J.G.; MATSUMURA-TUNDISI, T.M. Impactos potenciais das alterações do Código Florestal nos recursos hídricos. Biota Neotropica, v.10, n.4, p.67-76, 2010.
- ZAR, J.H. Biostatistical analysis Englewood Cliffs: Prentice-Hall, 1996.
Publication Dates
-
Publication in this collection
Jul-Aug 2016
History
-
Received
31 Jan 2014 -
Accepted
20 May 2016