Abstracts
OBJECTIVE:
To determine the prevalence of sonographic abnormalities (SA) in the axillary vein of patients with and without post-mastectomy lymphedema.
METHODS:
We studied a sample of 80 women, divided into two equal groups, with and without lymphedema, with B mode ultrasound, color and pulsed Doppler. The primary variable, SA, is defined as change in the venous diameter, parietal thickening, intraluminal images, compressibility, parietal collapse at inspiration and feature of the axillary venous flow on the operated side. Secondary variables were: stage of lymphedema, surgical technique, number of radio and chemotherapy sessions, limb volume, weight and age. The differences between the proportions in the groups were determined using the Chi-square test and / or Fisher's test. For continuous variables, we used the Mann-Whitney Test. To estimate the magnitude of the associations, we used the prevalence rate of SA in both groups as a measure of frequency, and as a measure of association, the prevalence ratio (PR) obtained as a function of relative risk (RR) and estimated by the test Mantel-Haenszel homogeneity test. We adopted the statistical significance level of 5% (p < 0.05).
RESULTS:
only the criterion "parietal thickening" was strongly associated with the lymphedema group (p = 0.001). The prevalence of SA was 55% in patients with lymphedema and 17.5% in the group without it, with difference in prevalence of 37.5%.
CONCLUSION:
the prevalence of SA was higher in patients undergoing mastectomy with lymphedema than in those without lymphedema.
Breast cancer; Axillary Vein; Lymphedema; Ultrasonography
OBJETIVO:
verificar se existe prevalência de alterações ultrassonográficas (AUS) na veia axilar de pacientes portadoras e não portadoras de linfedema do membro superior pós-mastectomia.
MÉTODOS:
uma amostra de 80 mulheres, alocadas em dois grupos iguais, com e sem linfedema foi estudada com ultrassonografia modo B, Doppler colorido e pulsado. A variável primária AUS foi definida como: alteração do diâmetro venoso, espessamento parietal, imagens intraluminares, compressibilidade, colapso parietal à inspiração e característica do fluxo venoso axilar no lado operado. Como variáveis secundárias: estádio do linfedema, técnica operatória, número de sessões de radio e quimioterapia, volume do membro, peso e idade. As diferenças entre as proporções nos grupos foram verificadas com o teste qui-quadrado de Pearson e/ou exato de Fisher. Para variáveis contínuas usamos o teste de Mann-Whitney. Para estimar a magnitude das associações utilizou-se como medida de frequência a prevalência de AUS em ambos os grupos e como medida de associação, a razão de prevalência (RP) obtida em função do risco relativo (RR) e estimada por meio do teste de homogeneidade de Mantel-Haenszel. Adotou-se o nível de significância estatístico de 5% (pd"0,05).
RESULTADOS:
somente o critério "espessamento parietal" se relacionou fortemente com o grupo com linfedema (p=0,001). A prevalência de AUS foi 55% no grupo com linfedema e 17,5% no grupo sem linfedema, com diferença de prevalências de 37,5%.
CONCLUSÃO:
a prevalência de AUS foi maior nas pacientes submetidas à mastectomia e com linfedema do que naquelas sem linfedema.
Neoplasias da mama; Veia Axilar; Linfedema; Ultrassonografia
INTRODUCTION
Many authors believe in the participation of the venous system in the post-mastectomy
lymphedema of the upper limb11 Megret G. Iatrogenic venous pathology and post-radiosurgery swollen arm.
Apropos of 100 cases. Phlebologie. 1985;38(4):609-19.
,
22 Gruffaz J. Venous component in lymphedema in the upper extremity after
radio-surgical therapy of cancer of the breast. Phlebologie.
1986;39(3):517-25., which caused the phlebographic study of venous
hemodynamics in post-mastectomy lymphedema (PML), with finding of 20% obstruction33 McIvor J, O'Connell D. The investigation of postmastectomy oedema of the
arm by lymphography and venography. Clin Radiol. 1978;29(4):457-62.. The flow measurement through the Doppler effect
showed increased venous pressure in the upper lymphedematous limb44 Tarquini A, Arru A, Puliga A, Armeni S, Piga A, Malloci A. Role of
venous injury in the origin of post-mastectomy lymphedema of the arm. Results of 150
measurements of the venous pressure of the homo- and counter-lateral arm in
mastectomy. Minerva Chir. 1987;42(12):1053-9.. The study of the axillary vein with color Doppler ultrasound and
flow speed analysis found between abnormalities in 57% to 70% 55 Svensson WE, Mortimer PS, Tohno E, Cosgrove DO. Colour Doppler
demonstrates venous flow abnormalities in breast cancer patients with chronic arm
swelling. Eur J Cancer. 1994;30A(5):657-60.. This noninvasive method is currently considered the one of choice
to study the venous system, especially in patients with PML55 Svensson WE, Mortimer PS, Tohno E, Cosgrove DO. Colour Doppler
demonstrates venous flow abnormalities in breast cancer patients with chronic arm
swelling. Eur J Cancer. 1994;30A(5):657-60.
6 Svensson WE, Mortimer PS, Tohno E, Cosgrove DO, Badger C, al Murrani B.
The use of colour Doppler to define venous abnormalities in the swollen arm following
therapy for breast carcinoma. Clin Radiol. 1991;44(4):249-52.
7 Szuba A, Razavi M, Rockson S. Diagnosis and treatment of concomitant
venous obstruction in patients with secondary lymphedema. J Vasc Interv Radiol.
2002;13(8):799-803.
-
88 Pain SJ, Vowler S, Purushotham AD. Axillary vein abnormalities
contribute to development of lymphoedema after surgery for breast cancer. Br J Surg.
2005;92(3):311-5..
However, some issues remain unclear, including the reasons for the wide variation in the incidence of PML over the postoperative years99 Erickson VS, Pearson ML, Ganz PA, Adams J, Kahn KL. Arm edema in breast cancer patients. J Natl Cancer Inst. 2001;93(2):96-111.and whether the axillary venous disease relates to the PML. Are there factors absent during surgery that present throughout the postoperative outcome that may influence the onset of post-mastectomy lymphedema? May there be venous factors of late onset? Would the individuals with PML also be suffering from axillary vein post-thrombotic disease? If the cause of venous involvement is associated with the surgical technique and the use of radiation therapy, and if there is a cumulative curve of incidence of the PML, how does the axillary vein behave in the presence of PML over time? Might the changes of the axillary vein have predictive value with respect to the development of PML? Can Ultrasound axillary vein patterns be established that correlate to the different degrees of installed PML?
Finally, is there a difference between the prevalence of changes in the axillary vein or around it detectable bu axillary US in patients undergoing mastectomy with and without upper limb lymphedema?
METHODS
This was a prevalence study conducted in 80 patients who underwent mastectomy and radiation therapy, who were attended sequentially and allocated into two groups: with lymphedema (n = 40) and without lymphedema (n = 40). Patients with the following conditions were excluded: bilateral mastectomy or non-neoplastic causes, lack of post-mastectomy radiotherapy treatment, history of central venous puncture, edema of upper limbs prior to mastectomy of venous or lymphatic origin, acute infection of the affected limb, psychiatric disorders, age lower than 18 years, indigenous people, pregnant women, prisoners and men. The study was approved by the Ethics in Research Committee of the Hospital Aristides Maltez (HAM) on 31/08/2009, Protocol 23/09, and all participants signed a free and informed consent.
We used ultrasound equipment model Sonoace X8 or SA 8000EX Prime, with a 5-12 MHz
multifrequency linear transducer, both from Medison Co. Ltd., 1003 Daechi-dong,
Gangnam-gu, Seoul 135-280 Korea. We searched the following sonographic abnormalities
(SA) in the transverse and longitudinal directions: increase in venous diameter,
parietalt hickness, echogenic material inside the vein, compressibility, decrease or
loss of collapsing capacity of venous walls upon deep breathing, and loss of phasic
blood flow rate55 Svensson WE, Mortimer PS, Tohno E, Cosgrove DO. Colour Doppler
demonstrates venous flow abnormalities in breast cancer patients with chronic arm
swelling. Eur J Cancer. 1994;30A(5):657-60.
,
1010 Gooding GA, Woodruff A. Color Doppler imaging in the subclavian-axillary
region and upper extremity. Clin Imaging. 1994;18(3):165-72.
11 Nazarian GK, Foshager MC. Color Doppler sonography of the thoracic inlet
veins. Radiographics. 1995;15(6):1357-71.
-
1212 Patel MC, Berman LH, Moss HA, McPherson SJ. Subclavian and internal
jugular veins at Doppler US: abnormal cardiac pulsatility and respiratory phasicity
as a predictor of complete central occlusion. Radiology.
1999;211(2):579-83..
As secondary variables, we studied the stage of lymphedema, surgical technique, number of radio and chemotherapy sessions, limb volume, weight and age. Lymphedema stadium was clinically defined by inspection and palpation, and later revised to verify that it agreed with the findings of volume and classified according to the standard adopted by the International Society of Linfology1313 International Society of Lymphology. The diagnosis and treatment of peripheral lymphedema. 2009 Concensus Document of the International Society of Lymphology. Lymphology. 2009;42(2):51-60..
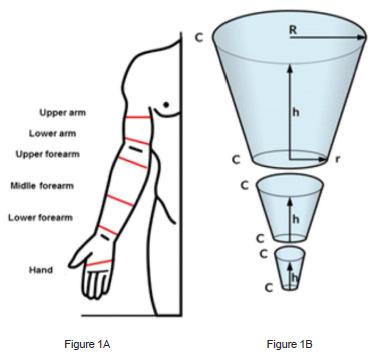
The volume of the limb was indirectly obtained by calculation of the cone section volume1414 Sander AP, Hajer NM, Hemenway K, Miller AC. Upper-extremity volume measurements in women with lymphedema: a comparison of measurements obtained via water displacement with geometrically determined volume. Phys Ther. 2002;82(12):1201-12.. To obtain the volume of each of the upper limbs, we represented each one as a succession of cone sections (Figure 1A), exemplified on a right upper member1515 Taylor R. Jayasinghe UW, Koelmeyer L, Ung O, Boyages J. Reliability and validity of arm volume measurements for assessment of lymphedema. Phys Ther. 2006;86(2):205-14.. The calculation of the volume of each cone section was obtained with the measures of the radii of the circles at the bottom and at the top of the section and their heights, which correspond to the length of the represented limb segments, defined as the distance between two circumferences on the selected points. We measured the circumference of the hand, lower third of the forearm 2 cm above the wrist, middle third midway between the wrist and the elbow, upper third of the forearm, 2 cm below the elbow fold, lower third of the arm and to 65% of distance from the elbow to the shoulder. Additionally, we measured the total distance between the lower (hand) and top (upper arm) measurements by proportionality dividing this total value by the lengths of the segments, obtaining with the h values (Figure 1B). As it was not possible to obtain the radius of the circle directly in the studied member, we used the measurement of the circumference, and with this value we calculated the radius:R=C/2?, where C is the circumference.
A- Notable places or local measurements of the circumference to obtain the perimetry (c); b- cone sections and measures C,h, R and r to calculate the volumes: Where: h = height of the cone seciton, corresponding to the length of a given limb section, defined between two landmark points; R = Radius calculated on the cone base, which correspond to the calculated value of the circularity taken in one of two landmark points that define the limb's section; CR = cone base circumference, which corresponds to the value calculated for R; r = radius calculated at the top of the cone, which corresponds to the calculated value of the circularity in the other two landmark points defining the limb section, opposite to where R is calculated; Cr = the cone top circumference, which corresponds to the value calculated for r.
Results were imputed to the mathematical formula for calculating the volume of the cone sections, and the sum of the volumes obtained for each segment provided the measure of the total volume member. For calculating the volume we used the following formula:
where V is the volume, h is the height of the truncated cone, R is the radius of the larger base and r is the radius of the smaller base.
From the height and weight, we calculated the participants' body mass index (BMI).
The surgical technique and the operated side were searched by direct vision of the surgical area and by consulting the records. In the complete absence of the breast, the operation was classified as a total mastectomy and the presence of a nipple, classified as partial. The postoperative time, age, number of sessions of radiotherapy and chemotherapy were obtained by direct research in the medical records.
Statistical method
The sample was calculated with the estimate of the presence of venous disease in people with lymphedema in the proportion of 20%, which was the lowest mean prevalence observed 33 McIvor J, O'Connell D. The investigation of postmastectomy oedema of the arm by lymphography and venography. Clin Radiol. 1978;29(4):457-62., and in the population without lymphedema, 1%; we set the power at 80%, significance level of 5 % and two-tailed test. We obtained a convenience, non-probabilistic sample, composed of the first 80 patients who underwent mastectomy and consecutively referred to the ultrasound service that met the inclusion criteria. Descriptive and exploratory analyzes aimed to characterize the groups and verify the goals of the study through descriptive measures (mean, standard deviation and median), distributions of bivariate frequencies and measures of association. The differences between the proportions were verified by using the chi-square test of Pearson and Fisher's exact test (when necessary). To verify differences between continuous variables we used the Mann-Whitney test. To estimate the magnitude of associations we used, as a frequency measure, the prevalence of ultrasound changes (USC) in both groups and, as association measure, the prevalence ratio (PR) obtained as a function of the relative risk (RR) and estimated by the Mantel-Haenszel Uniformity test. In all analyzes, we adopted the level of statistical significance of 5% (p?0,05).
RESULTS
The sample
The groups were proportionally similar in age, number of radiotherapy and chemotherapy sessions (p> 0.005) and differed with respect to BMI (pd"0,005) and postoperative time (Table 1).
The primary variable
We found that out of all the sonographic features used, the one statistically different in the two groups was the thickening of the vein wall (p = 0.001), although the collapse at inspiration has been present in greater proportion in those with lymphedema (57.5%), as well as the wall thick aspect (55%). Venous diameter showed little variation between groups. In both groups there was a predominance of phasic blood flow, compressible vein and absence of intraluminal images (Table 2).
Using only the feature "venous wall thickening" as a criterion for defining ultrasound changes due to its significance, there was high risk of such alteration in the group with lymphedema. The difference in prevalence was 37.5%. To estimate the association between the groups and the occurrence of ultrasound changes, we identified a statistically significant association and the increased risk of ultrasound changes of occurrences among those with lymphedema, which was 3.14 times higher compared with the group without lymphedema (Table 3).
We evaluated the gross association between the groups with and without lymphedema and the presence of ultrasound changes (USC) when all defined USC were used. Based on these characteristics, we set up the indicator "presence of ultrasound changes", and we found that the groups did not differ proportionally (p = 0.762). We observed a high prevalence of USC in both groups, 85.0% in the group with lymphedema and 82.5% in the group without it (Table 3).
We have not identified a statistically significant association between the groups and the presence of ultrasound changes, nor regarding the increase in the chance of occurrences among those with lymphedema (PR = 1.03; 95% CI: 0.85 to 1.25).
In the case of the ultrasound finding "venous wall thickening" (Figure 2), there is the lower thickness of the skin and normal subcutaneous aspect on the right axilla. The muscle has normal striated image and venous walls are thin and well defined. On the left side, which corresponds to the operated side, the skin is thicker and subcutaneous has a messy aspect, the muscle displays longitudinal lines that are more coarse and refringent, and the venous walls, proximal and distal to the transducer, are thickened with loss of the precise design of the wall.
Secondary variables
By analyzing the relationship between the groups with and without lymphedema and the presence of ultrasound changes (USC) and adjusting for clinical characteristics that define the secondary variables, we decided once again to use for the indicator "presence of USC" only the criterion "venous wall thickening ". Regarding the groups with and without lymphedema, there was a 3.14-fold increased risk in the group with lymphedema compared with the group without lymphedema. By observing the prevalence of lymphedema by age group, there is a positive and statistically significant association between the occurrence of venous wall thickening and the clinical occurrence "aged between 47-57 years," after adjusting for age group, with increased risk of 3.82 times compared with other age groups.
With respect to BMI, there was a statistically significant association and increased risk of thickening of the vessel wall occurrences among people with overweight 3.49 fold higher when compared with morbidly obese patients (Table 4).
We also identified a positive and statistically significant association between the clinical occurrence "operated side", interchangeably, and the presence of thickening of the vessel wall. There was an increase in the risk of the independent occurrence "operated side", the increased risk being 3.11 times on the left, while on the right, 3.18 (Table 4).
Total mastectomy also displayed an increased risk of 3.17 times compared with partial mastectomy, with a statistically significant association (95% CI: 1.46 to 6.84).
The number of radiotherapy sessions proved to be positively associated with the presence of thickening of the vessel wall. We identified an increased risk of 9.33 among those with 20 to 25 sessions when compared with other categories, this association being statistically significant (95% CI: 1.36 to 64.15).
The number of chemotherapy sessions was positively associated with the presence of thickening of the vessel wall. There was increased risk of 3.31 times among those with five to nine sessions when compared with other categories, and this association was also statistically significant (95% CI: 1.39 to 7.86).
The lymphedema characteristics showed proportionally similar in age, but diffeedr proportionally in relation to the evolutionary stage, predominantly in grade 2 (p <0.005).
The volumetric study of the limbs revealed the average volume of 915 (± 746) ml with a median 660ml in limbs with lymphedema and 84 (± 56) ml with a median of 73ml in limbs without lymphedema.
With respect to BMI, we observed that the proportion of obese was twice among those with lymphedema (45%) compared with the group without lymphedema (20%). Therefore, there was a predominance of women with lymphedema in the obesity range, in a statistically significant manner (p = 0.017).
As for post-operative time, in the group with lymphedema the postoperative period was longer than six years in 65% of cases, while in the group without lymphedema this time was less than six years in 67% cases.
The other characteristics were similar. The surgical technique and operated side were proportionally similar in both groups, as well as the number of radiotherapy and chemotherapy sessions (p>0.005).
DISCUSSION
Discussion of the method
Ultrasonography is the diagnostic method of choice in suspected venous thrombosis of any kind in the upper limbs1616 Grassi CJ, Polak JF. Axillary and subclavian venous thrombosis: follow-up evaluation with color Doppler flow US and venography. Radiology. 1990;175(3):651-4.. Compared to venography it showed 82% sensitivity and specificity1717 Baarslag HJ, van Beek EJ, Koopman MM, Reekers JA. Prospective study of color duplex ultrasography compared with contrast venography in patients suspected of having deep venous thrombosis of the upper extremities. Ann Intern Med. 2002;136(12):865-72. Erratum in: Ann Intern Med. 2003;138(5):438.. It allows the vision of venous walls and its surroundings, the study of its flow and the effect of physiological maneuvers over it1616 Grassi CJ, Polak JF. Axillary and subclavian venous thrombosis: follow-up evaluation with color Doppler flow US and venography. Radiology. 1990;175(3):651-4., plus the ability to identify tissue structures around the studied vein1010 Gooding GA, Woodruff A. Color Doppler imaging in the subclavian-axillary region and upper extremity. Clin Imaging. 1994;18(3):165-72., which is currently facilitated by the technical feature of the second harmonic, available in our equipment and not reported in previous studies. One can observe axillary vein stenosis by thrombus or post-thrombotic scar, extrinsic compression by adjacent mass and flow acceleration or turbulence. The indirect signs of disease are: the attenuation of the flow wave, reduction of the speed and loss of pulsatile characteristic transmitted by the respiratory and cardiac movement. All these findings should be compared with the contralateral side. Some errors are to be avoided, such as excessive compression with the transducer, which can change the shape and spectrum of blood flow from the vein, temporarily narrowing of the vein by deep breath, and reduced flow to between 3 and 5 cm / s, which can produce absence of color and simulate lack of flow and cause confusion between a great collateral and the axillary vein itself1111 Nazarian GK, Foshager MC. Color Doppler sonography of the thoracic inlet veins. Radiographics. 1995;15(6):1357-71.. In our study there was no lack of vein identification or presence of collateral, and the maneuver to produce the collapse of venous walls was not reliable because obese participants had difficulty getting a deep breath and there was variation in the execution of the maneuver between the various subjects examined. With the surgical dissection and local irradiation, there is the possibility of development of fibrosis in the region, which may be reduce the effectiveness of the maneuver1818 Segerström K, Bjerle P, Henriksson TG, Nyström A. Decompression of the axillary vein for oedema of the arm caused by axillary dissection and irradiation. Scand J Plast Reconstr Surg Hand Surg. 1991;25(3):245-8.. The use of ultrasound to study the axillary vein specifically in patients with lymphedema arose from the need to study the possibility of a venous component in the post-mastectomy lymphedema (PML), since we already knew the thrombogenic effects of postoperative radiotherapy on the subclavian and axillary vessels1919 Wilson CB, Lambert HE, Scott RD. Subclavian and axillary vein thrombosis following radiotherapy for carcinoma of the breast. Clin Radiol. 1987;38(1):95-6..
A preliminary study in four patients shows that the method can be used in patients with PML with advantages over venography, and describes a investigation sequence 66 Svensson WE, Mortimer PS, Tohno E, Cosgrove DO, Badger C, al Murrani B. The use of colour Doppler to define venous abnormalities in the swollen arm following therapy for breast carcinoma. Clin Radiol. 1991;44(4):249-52., making room for a larger study with 81 PML patients and 28 without edema in the control group, which analyzed direct flow obstruction signs, such as the following: the presence of thrombus, no flow at Doppler, no identification of the vein, stenosis or absence of venous walls collapse with inspiration, and flow cessation with limb lifting or upper rotation. Indirect signals were the presence of collaterals, continuous flow pattern and loss of normal phasic movement of venous walls55 Svensson WE, Mortimer PS, Tohno E, Cosgrove DO. Colour Doppler demonstrates venous flow abnormalities in breast cancer patients with chronic arm swelling. Eur J Cancer. 1994;30A(5):657-60., these parameters being analogous to those of this study.
The diagnosis of lymphedema is clinical. However, to quantify its volume is useful to assess the degree of lymphatic dysfunction, to classify it in evolutionary stages (Table 1) and to assess the results of provided treatment 1313 International Society of Lymphology. The diagnosis and treatment of peripheral lymphedema. 2009 Concensus Document of the International Society of Lymphology. Lymphology. 2009;42(2):51-60..
The measure of the lymphedematous limb volume by the water displacement technique device is reliable and accurate, but operationally cumbersome, and unhygienic. Moreover, due to requiring advance preparation, being less available and difficult to transport, it may be underutilized, which helps to reduce the number of diagnoses and periodic measurements during treatment, with negative implications for the health of affected individuals. Thus, alternative ways of measuring lymphedema volume are searched and studied. A water displacement model for connecting pipes that skirted the issue of water overflow has been developed. However, we had no opportunity to use it, since this study was already completed2020 Carvalho RM, Perez MCJ, Miranda Jr F. Volúmetro com câmaras comunicantes: apresentação de um protótipo de dimensões reduzidas desenvolvido para a avaliação objetiva do volume de mão. J vasc bras. 2010;9(2):76-80..
Comparative studies between the water displacement method and the cylinder volume and cone section showed a strong relationship between the volume measured by water displacement and geometric measurements in the affected limb, especially with the truncated cone volume, the cylinder volume and even rectangular and trapezoidal solids. Although there is the possibility of variation in the measurement of the circumference with tape measure, the findings showed that the method is reliable, economical and, above all, practical1414 Sander AP, Hajer NM, Hemenway K, Miller AC. Upper-extremity volume measurements in women with lymphedema: a comparison of measurements obtained via water displacement with geometrically determined volume. Phys Ther. 2002;82(12):1201-12..
There are, however, various other ways to take these measurements. Two different ways were carried out, comparing the results of both. Taylor started from the end of the third chirodactyl up intervals of 30, 40 and 50 cm and 65% of the distance between the elbow and the shoulder, therefore obtaining four cone sections, whose combined volumes represent the limb volume. He also measured the distance from the fist to the half of the forearm, from the half of the forearm to the elbow, from the elbow to the half of the arm and then to 65% of the distance between the elbow and the shoulder, obtaining also four cone sections. He correlated the findings with the measured volume of water displacement and concluded that the circumference measures with the anatomical repair points were more accurate than those obtained at regular intervals from the digital end1515 Taylor R. Jayasinghe UW, Koelmeyer L, Ung O, Boyages J. Reliability and validity of arm volume measurements for assessment of lymphedema. Phys Ther. 2006;86(2):205-14.. The method of obtaining limb volume through circumference measures ended up being the most widely used due to its simplicity and low cost, the one based on the anatomical repair points having better accuracy1515 Taylor R. Jayasinghe UW, Koelmeyer L, Ung O, Boyages J. Reliability and validity of arm volume measurements for assessment of lymphedema. Phys Ther. 2006;86(2):205-14..
In the present study, this method was used with little variation, since there were no measurements in the joint line of the wrist or elbow. For anatomical reasons, these two regions vary in circumference less the rest of the limb, thus not representing the additions and decreases or fluid observed in lymphedematous limbs. Hence they are called skin folds. Therefore, we amended the measures at the points of anatomical repair to 2cm above the crease of the wrist and we named this measure as "lower forearm," 2 cm below and 2 cm above the elbow crease and labeled "higher forearm" and " lower arm", respectively, where the circumference was less affected by cutaneous and subcutaneous folds (Figure 1).
Other methods for the diagnosis of lymphedema such as lymphoscintigraphy and MRI were not used in this study, since clinical diagnosis and the measured volume sufficed to select the participants of the two groups.
As for the axillary vein, this is classically defined as starting on the lower edge of the teres major muscle and extending to the lower edge of the first rib2121 Warwick R, Williams PL. Gray Anatomia. 35a Ed. Rio de Janeiro: Guanabara Koogan; 1979. Capítulo 6, Angiologia; p. 558-706.. A reported biometric study in Brazil concluded that the axillary starts at the union of medial brachial and Basilica veins in 72.5% of cases and in another 20% at a single stem formed by the union of the medial and lateral brachial veins and the basilica, which is near the lower edge of the pectoralis major muscle. It stretches over 13,81cm on average, to cross the lower edge of the subclavian muscle, where it is then called subclavian vein. In the reported study, the found diameters were 0.69cm on its infrapeitoral segment, 0.96cm on the retropeitoral segment and 0.99cm on the suprapeitoral segment2222 Rivello T, Messière B. Estudo biométrico de veia axilar em seres humanos. Rev Angiol Cir Vasc. 2002;11(2):80-4.. Even if one takes into consideration such anatomic limits, this and other morphological studies have their basis in cadavers, whereas this study assessed living subjects and used ultrasound. For this reason, it was important to know the morphological study of the axillary vein in living and the use of ultrasound, which found minimum and maximum diameters of 0.35cm and 2.53 cm, respectively, with an average of 1.3 cm2323 Galloway S, Bodenham A. Ultrasound imaging of the axillary vein-anatomical basis for central venous access. Br J Anaesth. 2003;90(5):589-95.. Thus, studied veins were measured with ultrasound equipment and the best normality parameter would be the contralateral side and not absolute measures in diameter. Use of the contralateral side as parameter is a common rule in routine ultrasound studies1111 Nazarian GK, Foshager MC. Color Doppler sonography of the thoracic inlet veins. Radiographics. 1995;15(6):1357-71.. Therefore, we excluded from the study patients who underwent bilateral mastectomy or had been subjected to central venous catheterization, thus unfit for such comparisons at the reasonable expectation that the contralateral side did not remain unscathed. In our study, the best image definition venous contour occurred at approximately 4-2 cm outside the midclavicular line in the retropeitoral and infrapeitoral segments.
The exclusion of patients who had not undergone radiotherapy was due to the correlate lymphedema expectation of the upper limb with the damage to the vein wall that can be triggered by radiation, with the non-irradiated contralateral side as control. We also excluded patients with edema of upper limbs prior to mastectomy due to the possibility of introducing a confounding variable. We did not include patients with acute infection for ethical reasons.
For the calculation of our sample we used the hypothesis that ultrasound changes (USC) are 20% higher in women with post-mastectomy lymphedema (PML) than in individuals without it. Studies similar to ours reported 70% of global USC and 57% of obstructions55 Svensson WE, Mortimer PS, Tohno E, Cosgrove DO. Colour Doppler demonstrates venous flow abnormalities in breast cancer patients with chronic arm swelling. Eur J Cancer. 1994;30A(5):657-60.. Keeping the parameters used, if we used the estimated prevalence of 70%, we would get the total of six research participants for each group in the sample, and if we used 57%, we would get only nine for each group. Studies with the use of equipment for flow measurement with CW Doppler plus venous occlusion reopletismography conducted in carriers of PML in sitting and lying positions shown that 31% of them had changes in two positions and 20%, changes in only one of the positions22 Gruffaz J. Venous component in lymphedema in the upper extremity after radio-surgical therapy of cancer of the breast. Phlebologie. 1986;39(3):517-25.. Wen calculating an estimated 31%, the total survey participants found was 22, and with 20%, we obtained a total of 40 participants in each group with and without lymphedema. A study with venography33 McIvor J, O'Connell D. The investigation of postmastectomy oedema of the arm by lymphography and venography. Clin Radiol. 1978;29(4):457-62. also found 20% of venous obstructions and as we found no other anatomical study that addresses prevalence of venous changes in PML, we used the lowest observed prevalence, which was 20% despite coming from studies with methods different from ours, only with the aim of ensuring an ideal sample to obtain reliable results.
Discussion of results
The initial finding of overall prevalence of 83.8% of the axillary vein ultrasound of women with post-mastectomy lymphedema (PML) is higher than that found by other authors. We found 85% prevalence in the group with lymphedema and 82.5% in the group without lymphedema. A study previous to ours, with 81 patients with post-mastectomy lymphedema assessed by color Doppler ultrasonography (CDUs), found no evidence of venous obstruction in 57%, plus signs of venous congestion in 14%, totaling 71% of venous abnormalities. In that study, the author considered as lymphedema the increase in volume of the largest limb to 200ml calculated from multiple circumference measurements. He used, as criteria of abnormality, changes of the venous flow phasic characteristics, presence of thrombus, failure to observe the vein, reduction in its diameter or immobility of its walls during inspiration and presence of collaterals, criteria similar to those used in our present study55 Svensson WE, Mortimer PS, Tohno E, Cosgrove DO. Colour Doppler demonstrates venous flow abnormalities in breast cancer patients with chronic arm swelling. Eur J Cancer. 1994;30A(5):657-60..
The highest prevalence rate in our study may be related to improving the quality of ultrasound images obtained by the current equipment, the use of second harmonic being an example.
In the present study, however, we observed, that the criterion "change in venous diameter" proved to be very similar in both groups, producing sample homogenization and contributing to the higher results. With the removal of this criterion and new analysis of the data, we found a decrease in prevalence to 77.5% in the group with lymphedema and 62.5% in the group without lymphedema, close to the results found by another author55 Svensson WE, Mortimer PS, Tohno E, Cosgrove DO. Colour Doppler demonstrates venous flow abnormalities in breast cancer patients with chronic arm swelling. Eur J Cancer. 1994;30A(5):657-60..
With these data, the feature axillary "venous wall thickening" was significantly higher in patients with lymphedema compared with the group without lymphedema (p = 0.001). Discarding the other sonographic features due their low statistical significance and solely relating the characteristic "venous wall thickening " with the two groups, we found its prevalence to be 55% in the group with lymphedema and only 17.5% in the group without it (PR = 3.14; 95% CI: 1.51 to 6.51). The cause of this thickening is not clear, and we can attribute it to the axillary vein thrombosis or to factors external to it such as surgery and radiotherapy.
The similarity of prevalence in both groups of other sonographic features, namely, change of diameter, presence of intraluminal images, compressibility, flow easiness and collapsing walls at inspiration, gave greater statistical value the unique feature that proved dominant in a given group, the one with lymphedema.
In reviewing the other sonographic features, we found that the flow was phasic in all sample cases, not being able to separate interest groups. In addition, the collapsing findings of the axillary vein similar to inspiration in both groups, and even slightly higher in patients with lymphedema, the venous diameter similar in both groups, and also the finding of just one intraluminal image in the whole sample, made us consider the possibility that the occurrence of thrombosis of the axillary vein can be infrequent, non-occlusive or quick resolution, or that the resulting impairment of venous function is of minor significance.
Furthermore, we observed that venous compression maneuver for venous thrombus was not as reliable, as some veins showed initially incompressible, though with normal flow demonstrable by Doppler. In most cases, venous occlusion obtained with the maneuver required more compression than on the contralateral control side. The compression caused by the equipment transducer promoted the movement of all the tissue block surrounding the vein, keeping it with its normal diameter or with small deformation, precisely in an area where sometimes the transducer's own weight is sufficient to occlude the vein. In patients submitted to total mastectomy, there is anatomical change in the chest wall at this location, which, associated with postoperative fibrosis, contributes to alter from convexity and typical softness to concavity and reduction of elasticity and thickening of the overlying skin. Subsequent radiotherapy may contribute to exacerbate this situation and this set of tissue changes makes the compressive maneuver difficult2424 Kocak Z, Overgaard J. Risk factors of arm lymphedema in breast cancer patients. Acta Oncol. 2000;39(3):389-92..
The maneuver to obtain venous collapsing at inspiration proved to be very dependent on the examined individual and appears to be hampered in overweight states and in the supine position. Moreover, pleuropulmonary lesions are common in this type of patients and we did not investigate their presence in our sample; their participation in the reported maneuvers is not known.
If we consider the set of signals that we deemed to be ultrasound changes for this work, we find that their occurrence is statistically similar in both groups, with and without lymphedema, except for the vessel wall thickening. We can also conclude that ultrasound changes are not associated with post-mastectomy lymphedema. It is possible that the adopted set of signals is not appropriate to separate the affected from the non-affected as for their local venous circulatory function within the universe of patients exposed to mastectomy plus radiotherapy. However, it is also expected that the venous disease, usually correlated with ultrasound changes, is not an independent factor in the genesis of post-mastectomy lymphedema, but one amongst other contributory factors. We do not know the weight of this participation. Other factors related to the clinical and epidemiological aspects correlated with the presence of post-mastectomy lymphedema.
The groups with and without lymphedema were similar in many aspects, such as age, surgical technique, operated side, and number of radiotherapy and chemotherapy sessions, helping to give confidence in the correlation of the findings related to the risk of USC occurrence in some of the groups.
The age group between 47 and 57 years displayed increased risk of USC occurrence, of 3.82 times, moving far away from the risk seen in younger age groups and maintaining superiority, but closer to the age group above, a finding that suggests USC increased prevalence with increasing age. As the postoperative time did not confer increased risk, age appears to be an independent risk factor2525 Bentzen SM, Dische S. Morbidity related to axillary irradiation in the treatment of breast cancer. Acta Oncol. 2000;39(3):337-47..
Total mastectomy showed 3.17-fold risk of increased USC prevalence compared with
partial mastectomy, which is understandable due to the increased removal of affected
lymph nodes and greater surgical aggression, with sequelae to the corresponding side,
such as local fibrosis and limb mobility reduction, regardless of the operated
side99 Erickson VS, Pearson ML, Ganz PA, Adams J, Kahn KL. Arm edema in breast
cancer patients. J Natl Cancer Inst. 2001;93(2):96-111.. The risk of increased USC prevalence
was also 9.33 times higher when the number of radiotherapy sessions ranged between 20
and 25. This is possibly due to all research subjects in our study underwent both
therapeutic methods, and the association between PML and radiation is reported in the
literature when associated with previous surgery, there being little evidence that it
contributes to PML alone99 Erickson VS, Pearson ML, Ganz PA, Adams J, Kahn KL. Arm edema in breast
cancer patients. J Natl Cancer Inst. 2001;93(2):96-111.
,
2525 Bentzen SM, Dische S. Morbidity related to axillary irradiation in the
treatment of breast cancer. Acta Oncol. 2000;39(3):337-47.. This factor is well accepted in the
literature as capable of aggravating local fibrosis, which can reduce the ability of
spontaneous lymphatic compensation, contributing to the emergence or worsening of the
post-mastectomy lymphedema 2424 Kocak Z, Overgaard J. Risk factors of arm lymphedema in breast cancer
patients. Acta Oncol. 2000;39(3):389-92.
25 Bentzen SM, Dische S. Morbidity related to axillary irradiation in the
treatment of breast cancer. Acta Oncol. 2000;39(3):337-47.
-
2626 Rezende LF, Pedras FV, Ramos CD, Gurgel MSC. Avaliação das compensações
linfáticas no pós-operatório de câncer de mama com dissecção axilar através da
linfocintilografia. J vasc bras. 2008;7(4):370-5..
There were differences between the groups in the aspect lymphedema stadium, which was expected, since in the group without lymphedema would be the stage 0 participants, thus inevitably differing from the other group, where would be individuals with all the other developmental stages.
The aspect BMI also showed differences between groups, with overweight subjectes
predominating in the one without lymphedema, while in the group with lymphedema there
was balance between overweight and obese individuals, suggesting, therefore, that
with the BMI evolution from overweight to obesity, there is migration to the group
with lymphedema. We also observed increased USC prevalence. This correlation between
high BMI and lymphedema has been reported by other authors2525 Bentzen SM, Dische S. Morbidity related to axillary irradiation in the
treatment of breast cancer. Acta Oncol. 2000;39(3):337-47.
26 Rezende LF, Pedras FV, Ramos CD, Gurgel MSC. Avaliação das compensações
linfáticas no pós-operatório de câncer de mama com dissecção axilar através da
linfocintilografia. J vasc bras. 2008;7(4):370-5.
-
2727 Freitas Júnior R, Ribeiro LFJ, Taia L, Kajita D, Fernandes MV, Queiroz
GS. Linfedema em pacientes submetidas à mastectomia radical modificada. Rev Bras
Ginecol Obstet. 2001;23(4):205-8.. We have not found participants with BMI
compatible with ideal weight.
The association between increased USC prevalence and number of chemotherapy sessions was 3.31 times from as early as the fifth session on. There is doubt about the association between chemotherapy and PML, with acceptance, however, of its participation in postoperative painful morbidity 2828 Hack TF, Cohen L, Katz J, Robson LS, Goss P. Physical and psychological morbidity after axillary lymph node dissection for breast cancer. J Clin Oncol. 1999;17(1):143-9..
During participants examination there were no limitations of movements that caused any restrictions, and we observed a wide freedom of movement of the upper limbs, whose amplitude seemed normal.
It was noteworthy the absence of venous collateral circulation in cases of recorded axillary vein thrombosis, which is described in studies involving axillary vein thrombosis due to effort, after catheterization, clavicle fracture, all without radiotherapy. Radiation therapy may have a role in it2424 Kocak Z, Overgaard J. Risk factors of arm lymphedema in breast cancer patients. Acta Oncol. 2000;39(3):389-92..
We recognize some limitations in this study. The lack of information on occupation, income and education did not allow correlating the prevalence of ultrasound changes and the presence of lymphedema with environmental risk.
Implications for clinical practice
This study allowed ultrasound changes to be detected in the axillary vein, showing its involvement with upper limb post-mastectomy lymphedema.
Since ultrasound alterations are easily found in post-thrombotic sequelae, this possibility should be aired in the postoperative period, and measures implemented, including prevention of venous thrombosis and early upper limb motor rehabilitation. Another aspect is that there must be caution with the axillary vein during surgery, plus postoperative care, in order to minimize the resulting scar.
The results of this study point to the need to reconsider some ultrasonographic signs found in the axillary vein when one wants to correlate them with the presence of post-mastectomy lymphedema. Changing in the diameter, presence of intraluminal images, compressibility as sign of thrombosis in the region and the absence of venous walls collapse seen at ultrasound as signs of post-thrombotic venous disease showed no reliability in our sample. As the venous wall thickening correlated strongly with the group with post-mastectomy lymphedema, this sonographic characteristic seemed reliable, rendering possible the correlation between the findings and high-risk patients, identifying those candidates for more often clinical follow-up.
Weight gain, with the research subject in the obesity range, correlated with the presence of PML. Efforts should be made in a multidisciplinary way for weight reduction when treating patients with post-mastectomy lymphedema.
The performance of total mastectomy is still a reality and this technique is related to increased risk of PML. There was increased USC prevalence in these cases and effective measures of early diagnosis and treatment should contribute to the decline of such rates. Mastectomy, when held on the right side, does not seem to be a predisposing factor for lymphedema, although most people are right-handed and may put the dominant member to the effort, and consequently to the risk.
Implications for research
Many questions remain open on the subject venous disease and post-mastectomy lymphedema and specifically in its ultrasound assessment.
The presence of vessel wall thickening in 55% of cases with PML compared with only 17.5% in non-carriers correlates vein injury with post-mastectomy lymphedema. Nonetheless, we do not know if such injury precedes or follows the onset of lymphedema. Nor do we know whether such injury is the sequel of venous thrombosis or resulting from radiotherapy. Studies lack before and after mastectomy and before and after radiotherapy, with short, medium and long-term follow-up, to establish the natural history of venous repercussions and their relationship with lymphedema in patients undergoing mastectomy and radiotherapy for breast cancer.
We do not know if the venous walls collapse at inspiration can be influenced by the presence of pleuropulmonary injury secondary to radiotherapy and further research is necessary in this area.
The possibility of spontaneous postoperative lymphatic compensation to ensure the lymphatic outflow in the venous system is well established in the literature in living2929 Földi E, Földi M, Clodius L. The lymphedema chaos: a lancet. Ann Plast Surg. 1989;22(6):505-15. and dead3030 Suami H, Pan WR, Taylor GI. Changes in the lymph structure of the upper limb after axillary dissection: radiographic and anatomical study in a human cadaver. Plast Reconstr Surg. 2007;120(4):982-91., which, of course, may be compromised by tissue aggression subsequent to surgery, such as infections and radiation.
The continuous study of the axillary vein and regional tissue changes in PML carriers by using complementary venous angiotomography or new ultrasound elastography techniques and magnetic resonance imaging can be important to select patients with the disease, since the ultrasound alone fails to diagnose it77 Szuba A, Razavi M, Rockson S. Diagnosis and treatment of concomitant venous obstruction in patients with secondary lymphedema. J Vasc Interv Radiol. 2002;13(8):799-803.. Elastography has emerged as an auxiliary tool for ultrasound diagnosis. Elastograms are images of tissue stiffness and may be colored, in shades of gray, or a combination of the two. Recent advances in elastography include quantification using voltage ratios, the acoustic radiation image, power boost, speed and wave cutting estimate. The first and most common elastography application is for the diagnosis of breast lesions and characterization of the focal masses such as liver cirrhosis of thyroid nodules, evaluation of lymph nodes for metastatic disease, detection of prostate cancer, among others. Promising applications include study of atheroma, assessment of venous and arterial wall and evaluation of characteristics and age of thrombi3131 Garra BS. Elastography: current status, future prospects, and making it work for you. Ultrasound Q. 2011;27(3):177-86..Studies with ultrasound elastography are conducted to select suitable locations for the employment of lymphatic-venous anastomoses3232 Mihara M, Hayashi Y, Murai N, Moriguchi H, Iida T, Hara H, et al. Regional diagnosis of lymphoedema and selection of sites for lymphaticovenular anastomosis using elastography. Clin Radiol. 2011;66(8):715-9. Erratum in: Clin Radiol. 2012;67(11):1128. and determination of the age of the thrombus3333 Karpiouk AB, Aglyamov SR, Mallidi S, Shah J, Scott WG, Rubin JM, et al. Combined ultrasound and photoacoustic imaging to detect and stage deep vein thrombosis: phantom and ex vivo studies. J Biomed Opt. 2008;13(5):054061., while other studies with magnetic resonance elastography, investigate the tissue consequences of disease processes, such as inflammation and fibrosis3434 Mariappan YK, Glaser KJ, Ehman RL. Magnetic resonance elastography: a review. Clin Anat. 2010;23(5):497-511.. the use of these new technologies in a prospective study before and after mastectomy can help to better understand the factors that influence the installation of PML.
More studies are needed to assess the possibility of treatment of axillary venous hypertension with venous decompression by section of the pectoralis major1818 Segerström K, Bjerle P, Henriksson TG, Nyström A. Decompression of the axillary vein for oedema of the arm caused by axillary dissection and irradiation. Scand J Plast Reconstr Surg Hand Surg. 1991;25(3):245-8. or the suitability for venous flow unblocking or correction of stenosis and placement of a stent77 Szuba A, Razavi M, Rockson S. Diagnosis and treatment of concomitant venous obstruction in patients with secondary lymphedema. J Vasc Interv Radiol. 2002;13(8):799-803. if the injuries are appreciable. However, this study is not conclusive in this direction.
Finally, other questions must be answered on the theme post-mastectomy lymphedema and, among those that were addressed in this study, we highlight some, such as the need to deepen the study of the forms of limb volume measurement to lymphedema definition and staging. There is no standard way of measuring a lymphedematous limb and therefore to search correlation between the measured volume obtained by water displacement and the various forms of circumference measurement, which could help to clarify this question.
The prevalence of ultrasound changes was significantly higher in patients undergoing mastectomy and with lymphedema compared with those without lymphedema. These findings correlate with increased risk of developing post-mastectomy lymphedema.
REFERENCES
-
1Megret G. Iatrogenic venous pathology and post-radiosurgery swollen arm. Apropos of 100 cases. Phlebologie. 1985;38(4):609-19.
-
2Gruffaz J. Venous component in lymphedema in the upper extremity after radio-surgical therapy of cancer of the breast. Phlebologie. 1986;39(3):517-25.
-
3McIvor J, O'Connell D. The investigation of postmastectomy oedema of the arm by lymphography and venography. Clin Radiol. 1978;29(4):457-62.
-
4Tarquini A, Arru A, Puliga A, Armeni S, Piga A, Malloci A. Role of venous injury in the origin of post-mastectomy lymphedema of the arm. Results of 150 measurements of the venous pressure of the homo- and counter-lateral arm in mastectomy. Minerva Chir. 1987;42(12):1053-9.
-
5Svensson WE, Mortimer PS, Tohno E, Cosgrove DO. Colour Doppler demonstrates venous flow abnormalities in breast cancer patients with chronic arm swelling. Eur J Cancer. 1994;30A(5):657-60.
-
6Svensson WE, Mortimer PS, Tohno E, Cosgrove DO, Badger C, al Murrani B. The use of colour Doppler to define venous abnormalities in the swollen arm following therapy for breast carcinoma. Clin Radiol. 1991;44(4):249-52.
-
7Szuba A, Razavi M, Rockson S. Diagnosis and treatment of concomitant venous obstruction in patients with secondary lymphedema. J Vasc Interv Radiol. 2002;13(8):799-803.
-
8Pain SJ, Vowler S, Purushotham AD. Axillary vein abnormalities contribute to development of lymphoedema after surgery for breast cancer. Br J Surg. 2005;92(3):311-5.
-
9Erickson VS, Pearson ML, Ganz PA, Adams J, Kahn KL. Arm edema in breast cancer patients. J Natl Cancer Inst. 2001;93(2):96-111.
-
10Gooding GA, Woodruff A. Color Doppler imaging in the subclavian-axillary region and upper extremity. Clin Imaging. 1994;18(3):165-72.
-
11Nazarian GK, Foshager MC. Color Doppler sonography of the thoracic inlet veins. Radiographics. 1995;15(6):1357-71.
-
12Patel MC, Berman LH, Moss HA, McPherson SJ. Subclavian and internal jugular veins at Doppler US: abnormal cardiac pulsatility and respiratory phasicity as a predictor of complete central occlusion. Radiology. 1999;211(2):579-83.
-
13International Society of Lymphology. The diagnosis and treatment of peripheral lymphedema. 2009 Concensus Document of the International Society of Lymphology. Lymphology. 2009;42(2):51-60.
-
14Sander AP, Hajer NM, Hemenway K, Miller AC. Upper-extremity volume measurements in women with lymphedema: a comparison of measurements obtained via water displacement with geometrically determined volume. Phys Ther. 2002;82(12):1201-12.
-
15Taylor R. Jayasinghe UW, Koelmeyer L, Ung O, Boyages J. Reliability and validity of arm volume measurements for assessment of lymphedema. Phys Ther. 2006;86(2):205-14.
-
16Grassi CJ, Polak JF. Axillary and subclavian venous thrombosis: follow-up evaluation with color Doppler flow US and venography. Radiology. 1990;175(3):651-4.
-
17Baarslag HJ, van Beek EJ, Koopman MM, Reekers JA. Prospective study of color duplex ultrasography compared with contrast venography in patients suspected of having deep venous thrombosis of the upper extremities. Ann Intern Med. 2002;136(12):865-72. Erratum in: Ann Intern Med. 2003;138(5):438.
-
18Segerström K, Bjerle P, Henriksson TG, Nyström A. Decompression of the axillary vein for oedema of the arm caused by axillary dissection and irradiation. Scand J Plast Reconstr Surg Hand Surg. 1991;25(3):245-8.
-
19Wilson CB, Lambert HE, Scott RD. Subclavian and axillary vein thrombosis following radiotherapy for carcinoma of the breast. Clin Radiol. 1987;38(1):95-6.
-
20Carvalho RM, Perez MCJ, Miranda Jr F. Volúmetro com câmaras comunicantes: apresentação de um protótipo de dimensões reduzidas desenvolvido para a avaliação objetiva do volume de mão. J vasc bras. 2010;9(2):76-80.
-
21Warwick R, Williams PL. Gray Anatomia. 35a Ed. Rio de Janeiro: Guanabara Koogan; 1979. Capítulo 6, Angiologia; p. 558-706.
-
22Rivello T, Messière B. Estudo biométrico de veia axilar em seres humanos. Rev Angiol Cir Vasc. 2002;11(2):80-4.
-
23Galloway S, Bodenham A. Ultrasound imaging of the axillary vein-anatomical basis for central venous access. Br J Anaesth. 2003;90(5):589-95.
-
24Kocak Z, Overgaard J. Risk factors of arm lymphedema in breast cancer patients. Acta Oncol. 2000;39(3):389-92.
-
25Bentzen SM, Dische S. Morbidity related to axillary irradiation in the treatment of breast cancer. Acta Oncol. 2000;39(3):337-47.
-
26Rezende LF, Pedras FV, Ramos CD, Gurgel MSC. Avaliação das compensações linfáticas no pós-operatório de câncer de mama com dissecção axilar através da linfocintilografia. J vasc bras. 2008;7(4):370-5.
-
27Freitas Júnior R, Ribeiro LFJ, Taia L, Kajita D, Fernandes MV, Queiroz GS. Linfedema em pacientes submetidas à mastectomia radical modificada. Rev Bras Ginecol Obstet. 2001;23(4):205-8.
-
28Hack TF, Cohen L, Katz J, Robson LS, Goss P. Physical and psychological morbidity after axillary lymph node dissection for breast cancer. J Clin Oncol. 1999;17(1):143-9.
-
29Földi E, Földi M, Clodius L. The lymphedema chaos: a lancet. Ann Plast Surg. 1989;22(6):505-15.
-
30Suami H, Pan WR, Taylor GI. Changes in the lymph structure of the upper limb after axillary dissection: radiographic and anatomical study in a human cadaver. Plast Reconstr Surg. 2007;120(4):982-91.
-
31Garra BS. Elastography: current status, future prospects, and making it work for you. Ultrasound Q. 2011;27(3):177-86.
-
32Mihara M, Hayashi Y, Murai N, Moriguchi H, Iida T, Hara H, et al. Regional diagnosis of lymphoedema and selection of sites for lymphaticovenular anastomosis using elastography. Clin Radiol. 2011;66(8):715-9. Erratum in: Clin Radiol. 2012;67(11):1128.
-
33Karpiouk AB, Aglyamov SR, Mallidi S, Shah J, Scott WG, Rubin JM, et al. Combined ultrasound and photoacoustic imaging to detect and stage deep vein thrombosis: phantom and ex vivo studies. J Biomed Opt. 2008;13(5):054061.
-
34Mariappan YK, Glaser KJ, Ehman RL. Magnetic resonance elastography: a review. Clin Anat. 2010;23(5):497-511.
-
Source of funding: none.
Publication Dates
-
Publication in this collection
Mar-Apr 2015
History
-
Received
01 July 2014 -
Accepted
15 Aug 2014