Abstracts
Furosemide (40mg) was administered to 20 street dogs, 10 males and 10 females, in two different pharmaceutical forms: (1) compressed furosemide 40mg formulated at the Federal University of Pernambuco (UFPE-tablet), and (2) a commercial formulation with equal bioequivalence produced by the Laboratory for Pharmaceutical Technology of Pernambuco State (LAFEPE), the LAFEPE-furosemide. The study aimed to evaluate the kinetics of dissolution of the UFPE-tablet in order to analyze the behavior of bioavailability of the best formulation for veterinary use. The plasmatic concentrations of furosemide for the determination of parameters of pharmacological kinetics were analyzed by high-performance liquid chromatographic method (HPLC). The in vitro study accomplished through physiochemical analyses demonstrated that the formulas of the furosemide tablets attained the pharmaceutical requirements in agreement with USP 23 and the Brazilian Pharmacopoeia. The evaluation accomplished in dogs with UFPE-tablets given in only dose demonstrated uniformity in blood levels indicating stability in maintenance of the pharmaceutical formulation and efficiency in absorption of the active compound. These values are not significantly different in relation to the 5% confidence limit. Regarding maximum concentration (Tmax) time and global bioavaibility assessed by AUC means, there were no considerable differences as well. UFPE-furosemide displayed 743.492µg/mL.h as AUC average value whereas LAFEPE-furosemide had an average of 537.284µg/mL.h.
Furosemide; bioavaibility; lyoavailability; dogs
Furosemida (40mg) foi administrada a 20 cães de rua (cães SRD), 10 machos e 10 fêmeas, em duas formas farmacêuicas distintas: (1) furosemida comprimido 40mg formulada na Universidade Federal de Pernambuco (UFPE-comprimido) e (2) uma formulação comercial bioequivalente produzida pelo Laboratório de Tecnologia Farmacêutica do Estado de Pernambuco (LAFEPE-furosemida). O estudo objetivou avaliar a cinética de dissolução do UFPE-comprimido para analisar o comportamento da liodisponibilidade dos comprimidos buscando a melhor formulação para uso veterinário. As concentrações plasmáticas de furosemida para determinação de parâmetros farmacocinéticos, foram analisadas por cromatografia líquida de alto desempenho (HPLC). O estudo in vitro realizado através de análises físico-químicas demonstrou que as fórmulas dos comprimidos de furosemida preencheram os requisitos farmacêuticos de acordo com USP 23 e a Farmacopéia Brasileira. Avaliações realizadas em cães da formulação UFPE-comprimidos administrados em dose única, demonstrou uma uniformidade nos níveis sangüíneos indicando estabilidade na manutenção da forma farmacêutica e eficiência na absorção do princípio ativo. Estes valores não são significativamente diferentes em relação ao limite de confiança de 5%. Em relação a concentração de máximo (Tmax) e ao tempo de biodisponibilidade global avaliados por meios de AUC, não houve nenhuma diferença considerável. O comprimido UFPE-furosemide exibiu AUC de 743.492µg/mL.h e o LAFEPE-furosemide teve uma média de 537.284µg/mL.h.
Furosemida; biodisponibilidade; liodisponibilidade; cães
Technological development of 40mg furosemide tablets: equivalence and bioavaibility study in dogs
Desenvolvimento tecnológico da furosemida comprimido (40mg): estudo da equivalência e biodisponibilidade em cães
Ednaldo Q. LimaI; Miracy M. AlbuquerqueII; Onaldo G. RodriguesI,* * Corresponding author: onaldo@cstr.ufcg.edu.br ; José R.B. AlencarII; Flávia P.M. MedeirosII; Pedro J. Rolim NetoII
IUnidade Acadêmica de Medicina Veterinária (UAMV), Centro de Saúde e Tecnologia Rural (CSTR), Universidade Federal de Campina Grande (UFCG), Av. Acadêmica s/n, Santa Cecília, Cx. Postal 64, Patos, PB 58708110, Brazil
IINúcleo de Desenvolvimento Farmacêutico e Cosmético (NUDFAC), Depto Ciências Farmaceuticas, Universidade Federal de Pernambuco (UFPE), Av. Prof. Moraes Rêgo 1235, Cidade Acadêmica, Recife, PE 50670-901, Brazil
ABSTRACT
Furosemide (40mg) was administered to 20 street dogs, 10 males and 10 females, in two different pharmaceutical forms: (1) compressed furosemide 40mg formulated at the Federal University of Pernambuco (UFPE-tablet), and (2) a commercial formulation with equal bioequivalence produced by the Laboratory for Pharmaceutical Technology of Pernambuco State (LAFEPE), the LAFEPE-furosemide. The study aimed to evaluate the kinetics of dissolution of the UFPE-tablet in order to analyze the behavior of bioavailability of the best formulation for veterinary use. The plasmatic concentrations of furosemide for the determination of parameters of pharmacological kinetics were analyzed by high-performance liquid chromatographic method (HPLC). The in vitro study accomplished through physiochemical analyses demonstrated that the formulas of the furosemide tablets attained the pharmaceutical requirements in agreement with USP 23 and the Brazilian Pharmacopoeia. The evaluation accomplished in dogs with UFPE-tablets given in only dose demonstrated uniformity in blood levels indicating stability in maintenance of the pharmaceutical formulation and efficiency in absorption of the active compound. These values are not significantly different in relation to the 5% confidence limit. Regarding maximum concentration (Tmax) time and global bioavaibility assessed by AUC means, there were no considerable differences as well. UFPE-furosemide displayed 743.492µg/mL.h as AUC average value whereas LAFEPE-furosemide had an average of 537.284µg/mL.h.
Index terms: Furosemide, bioavaibility, lyoavailability, dogs.
RESUMO
Furosemida (40mg) foi administrada a 20 cães de rua (cães SRD), 10 machos e 10 fêmeas, em duas formas farmacêuicas distintas: (1) furosemida comprimido 40mg formulada na Universidade Federal de Pernambuco (UFPE-comprimido) e (2) uma formulação comercial bioequivalente produzida pelo Laboratório de Tecnologia Farmacêutica do Estado de Pernambuco (LAFEPE-furosemida). O estudo objetivou avaliar a cinética de dissolução do UFPE-comprimido para analisar o comportamento da liodisponibilidade dos comprimidos buscando a melhor formulação para uso veterinário. As concentrações plasmáticas de furosemida para determinação de parâmetros farmacocinéticos, foram analisadas por cromatografia líquida de alto desempenho (HPLC). O estudo in vitro realizado através de análises físico-químicas demonstrou que as fórmulas dos comprimidos de furosemida preencheram os requisitos farmacêuticos de acordo com USP 23 e a Farmacopéia Brasileira. Avaliações realizadas em cães da formulação UFPE-comprimidos administrados em dose única, demonstrou uma uniformidade nos níveis sangüíneos indicando estabilidade na manutenção da forma farmacêutica e eficiência na absorção do princípio ativo. Estes valores não são significativamente diferentes em relação ao limite de confiança de 5%. Em relação a concentração de máximo (Tmax) e ao tempo de biodisponibilidade global avaliados por meios de AUC, não houve nenhuma diferença considerável. O comprimido UFPE-furosemide exibiu AUC de 743.492µg/mL.h e o LAFEPE-furosemide teve uma média de 537.284µg/mL.h.
Termos de indexação: Furosemida, biodisponibilidade, liodisponibilidade, cães.
INTRODUCTION
Furosemide is an anthranylic acid derivative commonly used in man as a potent tubule diuretic (Pontc & Schoenwald 1990, Awad et al. 1992). It is frequently used in human edema treatment associated with congestive heart failure, hyper-tension and renal insufficiency. In domestic animals furo-semide is used to treat various forms of edema in " azoturia" , to reduce space-filling lesions and as a prophylactic measure of epistaxis in racing horses (Roberts et al. 1978, Tobin 1978). Furosemide has a protective effect against bronchoconstriction caused by several factors including exercise (Maxson et al. 1995), and has been used illegally to 'dilute out' prohibited drugs (Stevenson et al. 1990, 1994, Sweeney et al. 1990). Therefore, its use in race horses should be monitored (Stevenson et al. 1994).
Nowadays one of the basic tasks of drug formulation is to develop an already existing dosage form in a way that makes the best drug release possible under the given circumstances, in other words, enhancing bioavalailability (Dhanaraju 1998, Sznitowska 2001, Matsuura et al. 2002). Furosemide pharma-cokinetics has been studied previously in several animal species, particularly horses (Roberts et al. 1978, Chay et al. 1983, Singh et al. 1990, Dyke et al. 1996), but also in dogs (Verbeeck et al. 1981, Hirai et al. 1992, Rahman et al. 2001) and rats (Hammarlund & Paalzow 1982).
Furosemide pharmacokinetics demonstrates 11-90% of oral absorption, 0.3-3.4% of half life, 60% of elimination in the unaltered form and 40% in the metabolized form (Hammarlund 1984, Goodman 1996). According to Johnston (1984), furo-semide is rapidly absorbed in the conventional tablet form with high diuretic peaks in dogs after one hour of administration (Chungi 1979, Jitka Huclov 2005). In this kind of study, it is assumed that the success or failure of the pharmaceutical treatment depends largely on certain factors, which are not limited to the usual pharmacodynamics or pharmacokinetics knowledge present in pharmacology compendia used in human medicine.
This study aims to report the technological development of 40mg furosemide tablets produced at Pernambuco's Federal University (UFPE-tablets) in comparison with the ones developed in vitro and in vivo at Pernambuco State Pharmaceutical Laboratory (LAFEPE- furosemide), using street dogs administration model to meet therapeutic needs in Veterinary Medicine practice.
MATERIALS AND METHODS
Experiment place and technological process
This study was carried out in the Laboratory for Pharmaceutical Technology of Pernambuco (LAFEPE) at Federal University of Campina Grande (UFCG) laboratories. The technological development of UFPE-tablets was based on Good Practices of Manufacture (BPF in portuguese). In the pre-formulation study the following materials were used: diluents (starch, calcium phosphate, lactose), agglutinants (arabic gum, PVP K30, carboxymethylcellulose), lubrificants (magnesium stearate, powder), furosemide (active principle) and moisture (hydroholic solution). The compression process by humid via was used in tablets production.
Animals, drug administration and blood sampling
The animal selection was done with a sample of twenty dogs (street-dog), with standard weight and age. Sixteen healthy dogs with ages varying from 3-5 years and weighing between 20 and 30 kilos were selected, 10 males and 6 females. The dogs underwent clinical evaluation during a 30-day-period at the Federal University of Campina Grande (UFCG) Kennel. They were submitted to a twelve-hour fasting after the last meal of the previous day with no water restriction. A single 80mg dose (two 40mg furosemide-tablets 5 per cent w/v, Hoechst Animal Health, Bucks, UK) of the formulations under study (UFPE-tablets and LAFEPE furosemide) was administrated in the morning. The dogs were monitored during the whole study and had the same accompaniment.
Furosemide analysis
6mL of blood samples were collected in heparinized tubes in 0, 20min, 40min, 60min, 90min, 120min, and 180min intervals. The plasma, which was protected from the light until furosemide analysis, was separated by a centrifugation of 3,500 rpm for 10 minutes kept at a -20º C temperature. All the samples of the same animal were analyzed in the same day to avoid variation among analysis. Furosemide plasma concentration was determined by High Pressure Liquid Chromatography (HPLC).
Analytic methodology
The quality of the products under study was evaluated in two stages, according to the 4th edition of the Brazilian Pharmacopeia (Brasil, Ministério da Saúde 1988) and the 23rd edition of the United States Pharmacopeia (USP 1995). At first, physicochemical assay of average weight, hardness, friability and disintegration were fulfilled. Subsequently, essays on assay, content uniformity and liberation kinetics were carried out. These stages determine the in vitro study.
Plasma samples preparation
Furosemide concentration in plasma samples was determined by HPLC, HPLC acetonitrile degree and Merck methanol were used as solvents. The mobile phase was a mixture of water: acetonitrile (70:30) and the pH was adjusted with NaOH 0.2N or glacial acetic acid (1:100) to 6.5. The HPLC system chromatographic conditions consist of a solvent system; a 20 µL Rheodyne valve; an UV detector; an integrator; ODS 25µm spherisorb waters, a 1mL/min air flow and a 235 nm wave length.
Extraction of samples procedure
The following procedure was used in order to determine the furosemide in plasma samples. Firstly, 0.2mL of plasma was taken and 2.4µg/mL of fenacetine in methanol was added. This material was stirred in vortex for 40 seconds and then centrifuged at 3000 rpm for 12 minutes. Secondly, the methanol layer was decanted and centrifuged for 5 minutes. A 1mL portion was transferred to a volumetric balloon concluding the mobile phase. A 20µL volume was injected. A threefold procedure was carried out. The standard curve (n=6, r=0.9899) was used to calculate the unknown furo-semide plasma concentrations. The validation of the analytic methodology employed furosemide methanoic solutions and plasma furosemide. The feasibility of analysis reproduction was assessed through the extraction of six plasma samples with concentrations of 200, 240, 280, 320, 360 and 400µg/mL. The following parameters were compared by means of variance analysis (ANOVA): maximum plasma concentration (Cmax), time for reaching maximum concentration (Tmax) and area under the time of maximum concentration curve (AUC).
RESULTS AND DISCUSSION
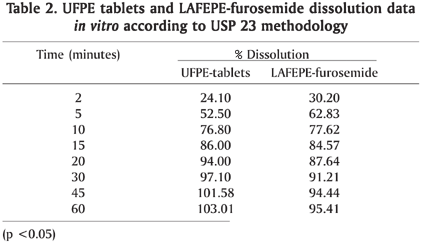
The in vitro study carried out through physicochemical essays showed that the furosemide tablet formulations met the pharmaceutical requirements according to USPC (1995) and Brazilian Pharmacopoeia, 4th edition (Table 1). The data concerning the studied dissolution formulation of Furosemida tablets reached the monography requirements - more than 65% was dissolved after 30min (Table 2). Still, the profiles showed that the products reach 65% of dissolution in vitro in 10 minutes after the beginning of the test and show a similar profile in the active principle liberation (Fig.1).
The furosemide in the dog's plasma was carried out as a flowing stage (Table 3). The calibration line was determined with the results obtained from the standard plasma furose-mide solutions and underwent statistic treatment, with (R = 0.9942) and p <0.0001. The correlation coefficient was R = 0.9899 (Fig.2).
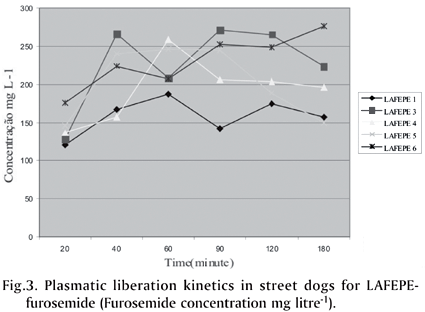
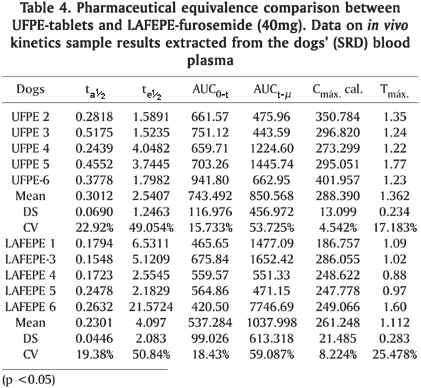
The bioavailability evaluation according to study specification entails the determination of pharmacy absorption quantity in 20, 40, 60, 90, 120 and 180 min intervals, i.e., the velocity in which this process happens (United States Pharmacopoeia 1990, Ritchel 1992) (Table 4). Since bio-equivalence is the bioavailability comparative study between two pharmaceutical forms or products containing the same pharmacy in the equal doses, adequate parameters, which describe the absorption process, must be chosen (Jackson 1994). In the bio-equivalence study, the pharmacokinetic parameters calculus is usually obtained from blood line pharmacy concentration versus time after administration of the formulations under study. In this case, the area under the curve (AUC), the maximum blood concentration (Cmax) and the time in which the Cmax is reached are determined. As observed in Table 4, furosemide displays short plasmatic half time, being liberated in about 60-80% in the blood plasma.
The plasmatic concentration released in the street dogs plasma in both UFPE-tablets and LAFEPE-furosemide (40mg) is not different when concentration versus time (Cmax and tmax) is observed (Fig.3 and 4 ). Table 4 shows some variability among individuals. Nevertheless, it is consistent with what was reported by Straughn et al. (1986) and Wallwe et al. (1982). Following furosemide administration, when comparing UFPE'S and LAFEPE's data, the following blood plasma maximum concentration average values are reached: 288.390µg/mL for tablets furosemide (40mg) the UFPE product and 261.248µg/mL for the LAFEPE product. These values are not significantly different in relation to the 5% confidence limit. Regarding maximum concentration (Tmax) time and global bioavaibility assessed by AUC means, there were no considerable differences as well. UFPE-furosemide displayed 743.492µg/mL.h as AUC average value whereas LAFEPE-furosemide had an average of 537.284µg/mL.h.
CONCLUSION
The liberation half times (t1/2)) for UFPE and LAFEPE furosemide vary between 60 and 90 minutes. The results obtained from this study indicate that following a single oral dose of two 40mg furosemide tablets, the differences shown by the data related to the test and reference products were considered statistically non-relevant within the 5% confidence level that, in turn, demonstrates that the two products are bioavailable.
The bioavailability data in plasma obtained after furosemide oral doses administration assure that it is possible to compare the bioequivalence of the products with absolute security.
Based on the comparison of in vitro data and on bioavailability blood plasma levels it is possible to guarantee the bioequivalence of the products.
Hence, there is an urgent need for the government to put into practice the current policy for the Brazilian generics which will make the bioequivalence studies a compulsory item by invigilating organs hence, improving security, efficiency and quality of commercialized veterinary products.
Finally, we believe that the bioequivalence study carried out through the product development (pharmaceutical development) compared to the reference product has proved to be a superior gain over two unknown products.
Aknowlegments.- The study was financially supported by a especial research fund of the Pernambuco State Pharmaceutical Laboratory (LAFEPE).
Received for publication on September 20, 2006.
Accepted for publication on October 18, 2007.
- Brasil, Ministério da Saúde 1988. Farmacopéia Brasileira. Pte 1. 4ª ed. Atheneu, São Paulo, p.487-488.
- Dhanaraju M.D., Kumaran K.S., Baskaran T. & Moorthy T.S. 1998. Enhancement of bioavailability of griseofulvin by its complexation with beta-cyclodextrin. Drug Dev. Ind. Pharm. 24:583-587.
- Goodman L.S. & Gilman A. 1996. The Pharmacological basis of therapeutics. 9th ed. Editora McGraw-Hill, New York, p.701.
- Hammarlund M.M., Paalzow L. & Odlind B. 1984. Pharmacokinetics of furosemide in man after intravenous and oral administration. Application of moment analysis. Europ. J. Clin. Pharmacol. 26:197-207.
- Jackson A.J. 1994. Generics and Bioequivalence. CRC, Rockville. 203p.
- Johnston J.T. 1984. The Pharmacokinetics of Furosemide in Dogs. Dissertation, University of Texas, Austin. 112p.
- Jitka Huclov A.A., Dalibor J., Satýnsk Y.A., Maiab T. & Araujo B.A.N. 2005. Sequential injection extraction based on restricted access material for determination of furosemide in serum. J. Chromatogr. 1087:245-251.
- Kaojarern S. & Utiswannakul A. 1990. Bioavailability and pharmacokinetics of furosemide marketed in Thailand. J. Med. Assoc. Thai., p.191-197.
- Matsuura A., Nagayama T. & Kitagawa T. 2002. Automated high-performance liquid chromatographic method for determination of furosemide in dog plasma. Shionogi Res. Lab. Japan 4:5-12.
- Ponto L.L.B. & Schoenwald D. 1990. Furosemide (Frusemide): a pharma-cokinetic/pharmacodynamic review (Part I). Clin. Pharmacokinetics 18:381-471.
- Sznitowska M., Gajewska M., Janicki S., Radwanska A. & Lukowski G. 2001. Bioavailability of diazepam from aqueous-organic solution, submicron emulsion and solid lipid nanoparticles after rectal administration in rabbits. Europ. J. Pharmacy Biopharm. 52:159-163.
- Smal J., Haapala O., Marvola M. & Happonen I. 1995. Prolonged-release hard gelatin capsules of furosemide for treatment of dogs. Europ. J. Pharm. Biopharm. 18:17-23.
- Smal J., Haapala O., Marvola M., Kuosela S. & Happonen I. 1995. Prolonged-release hard gelatin capsules of furosemide for treatment of dogs. J. Vet. Pharmacol. Therap. 19:482-487.
- Smal J., Marvola M., Liljequist C. & Happonen I. 1996. Prolonged-release hydroxypropyl methylcelcellulose matrix tablets of furosemide for administration to dogs. Europ. J. Pharm. Biopharm. 19:482-482.
- Ritschel W.A. 1992. Handbook of Basic Pharmacokinetics. 4th ed. Drug Intelligence, Hamilton, WA, p.588.
- Rahman M., Kimura A., Yoneyama H.B., Kosaka H.B. & Akira Nishiyama A. 2001. Effects of furosemide on the tubular reabsorption of nitrates in anesthetized dogs. Europ. J. Pharmacol. 428:113-119.
- Straughn W.G., Raghow G. & Meyer M. 1986. Bioavaibility of seven furosemide tablets in man. Biopharm Dry Disparities 7:113-122.
- USP 1995. United States Pharmacopoeia. 23rd ed. United States Pharmacopeial Convention, Rockville, MD, p.987-991.
- Waller E., Hamilton S., Massarela J., Shraneyuycb M., Smith R., Yakatan G. & Doluisio J. 1982. Disposition and absolute bioavaibility of furosemide in healthy males. Pharmacy 71:11-05.
Publication Dates
-
Publication in this collection
21 Jan 2008 -
Date of issue
Nov 2007
History
-
Received
20 Sept 2006 -
Accepted
18 Oct 2007