ABSTRACT:
Studies have demonstrated sympathetic cardiac denervation in the MPTP mouse model. MPTP toxicity causes sympathetic nerve damage and depletion of heart norepinephrine. Previous evaluations of impairments in heart innervation have been based on imaging, electrophysiological and biochemical methods. However, these studies lacked information that can be obtained from morphoquantitative analyses. Thus, this study aimed to apply a design-based stereological method for evaluating the morphoquantitative alterations of myocardium following treatment with the neurotoxin MPTP in the C57/BL mouse. Our results showed that MPTP reduced the number of cardiomyocytes in the left ventricle.
INDEX TERMS:
Methyl; phenyl; tetrahydropyridine; MPTP; mice; myocardial; morphology; heart; rodent; stereology
RESUMO:
Estudos têm demonstrado a desnervação simpática cardíaca no modelo da administração do MPTP em camundongo. A toxicidade do MPTP causa lesão ao nervo simpático e depleção da norepinefrina. As avaliações dos danos na inervação do coração são baseadas em métodos de imagem, eletrofisiológico e bioquímico. Contudo, estes estudos carecem de informações provenientes de análises morfoquantitativas. Assim, objetivou-se aplicar métodos estereológicos para avaliar as alterações morfoquantitativas do miocárdio após o tratamento com a neurotoxina MPTP no camundongo C57/BL. Nossos resultados mostraram que o MPTP causa redução no número de cardiomiócitos no ventrículo esquerdo.
TERMOS DE INDEXAÇÃO:
Metil; fenil; tetrahidropiridina; MPTP; morfologia; miocárdio; camundongo; coração; roedor; estereologia
Introduction
Several studies have demonstrated sympathetic cardiac dysfunction in MPTP-induced animal models, especially in C57/BL mice (Goldstein et al. 2000Goldstein D.S., Holmes C., Li S.T., Bruce S., Metman L.V. & Cannon 3rd R.O. 2000. Cardiac sympathetic denervation in Parkinson disease. Ann. Intern. Med. 133(5):338-347. <http://dx.doi.org/10.7326/0003-4819-133-5-200009050-00009> <PMid:10979878>
https://doi.org/10.7326/0003-4819-133-5-...
, Li et al. 2002Li S.T., Dendi R., Holmes C. & Goldstein D. 2002. Progressive loss of cardiac sympathetic innervation in Parkinson’s disease. Ann. Neurol. 52(2):220-223. <http://dx.doi.org/10.1002/ana.10236> <PMid:12210793>
https://doi.org/10.1002/ana.10236...
, Ren et al. 2004Ren J., Porter J.E., Wold L.E., Aberle N.S., Muralikrishnan D. & Haselton J.R. 2004. Depressed contractile function and adrenergic responsiveness of cardiac myocytes in an experimental model of Parkinson disease, the MPTP-treated mouse. Neurobiol. Aging 25(1):131-138. <http://dx.doi.org/10.1016/S0197-4580(03)00035-6> <PMid:14675739>
https://doi.org/10.1016/S0197-4580(03)00...
, Haensch et al. 2009Haensch C.A., Lerch H., Jörg J.A. & Isenmann S. 2009. Cardiac denervation occurs independent of orthostatic hypotension and impaired heart rate variability in Parkinson’s disease. Parkinsonism Relat. Disord. 15(2):134-137. <http://dx.doi.org/10.1016/j.parkreldis.2008.04.031> <PMid:18515170>
https://doi.org/10.1016/j.parkreldis.200...
). Animal models of cardiac dysautonomia provide an opportunity to evaluate and understand the neurodegenerative process and develop unbiased methods of evaluation (Joers & Emborg 2014Joers V. & Emborg M.E. 2014. Modeling and imaging cardiac sympathetic neurodegeneration in Parkinson’s disease. Am. J. Nucl. Med. Mol. Imaging 4(2):125-159. <PMid:24753981>). One of the most commonly used chemicals in such animal models is 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP), which is considered the gold standard model cardiac denervation in neurodegenerative disease as Parkinson’s disease (Jackson-Lewis et al. 2012Jackson-Lewis V., Blesa J. & Przedborski S. 2012. Animal models of Parkinson’s disease. Parkinsonism Relat. Disord. 18(Suppl.1):183-185. <http://dx.doi.org/10.1016/S1353-8020(11)70057-8> <PMid:22166429>
https://doi.org/10.1016/S1353-8020(11)70...
).
The findings of cardiac denervation are based on radiolabelling imaging techniques, electrophysiological studies, pharmacological dosage or immunohistochemistry in either humans or animal models (Takatsu et al. 2000Takatsu H., Nishida H., Matsuo H., Watanabe S., Nagashima K., Wada H., Noda T., Nishigaki K. & Fujiwara H. 2000. Cardiac sympathetic denervation from the early stage of Parkinson’s disease: clinical and experimental studies with radiolabeled MIBG. J. Nucl. Med. 41(1):71-77. <PMid:10647607>, Ren et al. 2004Ren J., Porter J.E., Wold L.E., Aberle N.S., Muralikrishnan D. & Haselton J.R. 2004. Depressed contractile function and adrenergic responsiveness of cardiac myocytes in an experimental model of Parkinson disease, the MPTP-treated mouse. Neurobiol. Aging 25(1):131-138. <http://dx.doi.org/10.1016/S0197-4580(03)00035-6> <PMid:14675739>
https://doi.org/10.1016/S0197-4580(03)00...
, Amino et al. 2005Amino T., Orimo S., Itoh Y., Takahashi A., Uchihara T. & Mizusawa H. 2005. Profound cardiac sympathetic denervation occurs in Parkinson disease. Brain Pathol. 15(1):29-34. <http://dx.doi.org/10.1111/j.1750-3639.2005.tb00097.x> <PMid:15779234>
https://doi.org/10.1111/j.1750-3639.2005...
, Goldstein et al. 2005Goldstein D.S., Eldadah B.A., Holmes C., Pechnik S., Moak J., Saleem A. & Sharabi Y. 2005. Neurocirculatory abnormalities in Parkinson Disease with orthostatic hypotension independence from levodopa treatment. Hypertension 46(6):1333-1339. <http://dx.doi.org/10.1161/01.HYP.0000188052.69549.e4> <PMid:16216982>
https://doi.org/10.1161/01.HYP.000018805...
, Haensch et al. 2009Haensch C.A., Lerch H., Jörg J.A. & Isenmann S. 2009. Cardiac denervation occurs independent of orthostatic hypotension and impaired heart rate variability in Parkinson’s disease. Parkinsonism Relat. Disord. 15(2):134-137. <http://dx.doi.org/10.1016/j.parkreldis.2008.04.031> <PMid:18515170>
https://doi.org/10.1016/j.parkreldis.200...
, Ziemssen & Reichmann 2010Ziemssen T. & Reichmann H. 2010. Cardiovascular autonomic dysfunction in Parkinson’s disease. J. Neurol. Sci. 289(1-2):74-80. <http://dx.doi.org/10.1016/j.jns.2009.08.031> <PMid:19740484>
https://doi.org/10.1016/j.jns.2009.08.03...
, Jain & Goldstein 2012Jain S. & Goldstein D.S. 2012. Cardiovascular dysautonomia in Parkinson disease: from pathophysiology to pathogenesis. Neurobiol. Dis. 46(3):572-580. <http://dx.doi.org/10.1016/j.nbd.2011.10.025> <PMid:22094370>
https://doi.org/10.1016/j.nbd.2011.10.02...
), but little attention has been given to the morphological aspects of the heart, especially in studies using efficient 3-D quantitative methods in MPTP models. Assuming that sympathetic cardiac dysfunction and cardiac dysautonomia have negative consequences on cardiac muscle, this study aimed to describe a design-based stereological method for evaluating the morphoquantitative alterations of myocardium following treatment with the neurotoxin MPTP in the C57/BL mouse model. The present results may contribute to a better understanding of the pathophysiological mechanisms underlying sympathetic cardiac denervation and the extent to which cardiac dysautonomia affects the myocardium.
Materials and Methods
In this study were used ten two-month-old male C57/BL mice with a mean body weight of 20g. These animals were obtained from the Centre of Experimental Model Development (Cedeme) at Federal University of São Paulo (Unifesp). This study was approved by the Animal Care Commission of the College of Veterinary Medicine at the University of São Paulo (Application number 1068/2007) in accordance with the directives of the Brazilian College of Experimentation (COBEA) and with the Code of Ethics of the World Medical Association. The animals were assigned to two groups. Group I included MPTP-induced animals (5 animals), whereas Group II was the control and included sham animals (5 animals).
The neurotoxin 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP Sigma?) was diluted in 100ml of saline solution to obtain 1% solution. The animals received two doses of 50mg/kg with a 16h interval between doses. This dose was previously reported to induce an almost complete depletion of dopaminergic cells (Sonsalla & Heikkila 1986Sonsalla P.K. & Heikkila R.E. 1986. The influence of dose and dosing interval on MPTP- induced dopaminergic neurotoxity in mice. Eur. J. Pharmacol. 129(3):339-345. <http://dx.doi.org/10.1016/0014-2999(86)90444-9> <PMid:3490988>
https://doi.org/10.1016/0014-2999(86)904...
, Sundström et al. 1987Sundström E., Strömberg I., Tsutsumi T., Olson L. & Jonsson G. 1987. Studies on the effect of 1-methyl-4-phenyl-1,2,3,6-tetrahydroxypyridine (MPTP) on central catecholamine neurons in C57 BL/6 mice: comparison with three other strains of mice. Brain Res. 405(1):26-38. <http://dx.doi.org/10.1016/0006-8993(87)90986-3> <PMid:2882814>
https://doi.org/10.1016/0006-8993(87)909...
) and was sufficient to induce cardiac alterations (Ren et al. 2004Ren J., Porter J.E., Wold L.E., Aberle N.S., Muralikrishnan D. & Haselton J.R. 2004. Depressed contractile function and adrenergic responsiveness of cardiac myocytes in an experimental model of Parkinson disease, the MPTP-treated mouse. Neurobiol. Aging 25(1):131-138. <http://dx.doi.org/10.1016/S0197-4580(03)00035-6> <PMid:14675739>
https://doi.org/10.1016/S0197-4580(03)00...
, Amino et al. 2008Amino T., Uchihara T., Tsunekawa H., Takahata K., Shimazu S., Mizusawa H. & Orimo S. 2008. Myocardial nerve fibers are preserved in MPTP-treated mice, despite cardiac sympathetic dysfunction. Neurosci. Res. 60(3):314-318. <http://dx.doi.org/10.1016/j.neures.2007.11.011> <PMid:18242750>
https://doi.org/10.1016/j.neures.2007.11...
). The animals in Group II received saline solution (0.9%) with the same administration schedule as Group I. The administration of the MPTP and saline solutions was intraperitoneal by using an insulin syringe.
The animals were euthanised with an overdose of sodium pentobarbital (120mg/kg/i.p) seven days after the last administration of MPTP. The animals were perfused through the ascending aorta with phosphate buffered saline (PBS; Sigma?; 0.1 M, pH 7.4) containing 2% of heparin (Roche?), followed by fixation with 4% paraformaldehyde in phosphate buffered saline (PBS; Sigma?; 0.1 M, pH 7.4). Then, the hearts were removed and immersed in the same fixative solution and stored at 4°C for 24 hours.
Cardiac morphoquantitative study
Left ventricle volume V (LV). The V(LV) was estimated according to the Cavalieri principle. The left ventricle was sectioned in 1mm thick slices. The following formulae were applied in the analysis:
Σpw is the number of points hitting the wall and Σpl is the number of points hitting the lumen. The area associated with each test point (area per point) was 0.518mm for the wall and 0.038 for the lumen. The thickness (t) was 1mm (Fig.1). The V(LV) was calculated as the sum of V (W) + V (L). The error variance of the Cavalieri principle was estimated according to Gundersen et al. (1999)Gundersen H.J.G., Jensen E.B.V., Kiêu K. & Jensen J. 1999. The efficiency of systematic sampling in stereology reconsidered. J Microsc. 193(Pt 3):199-211. <http://dx.doi.org/10.1046/j.1365-2818.1999.00457.x> <PMid:10348656>
https://doi.org/10.1046/j.1365-2818.1999...
.
Estimation of the left ventricle (LV) volume by Cavalieri principle. (A) Points hitting the left ventricle wall, (B) points hitting the lumen. Bar = 2mm.
Sampling of the left ventricle. The left ventricle (LV) sampling was based on the Smooth Fractionator (Gundersen 2002Gundersen H.J.G. 2002. The smooth fractionator. J. Microsc. 207(Pt 3):191-210. http://dx.doi.org/10.1046/j.1365-2818.2002.01054.x.
https://doi.org/10.1046/j.1365-2818.2002...
). Following the LV volume by Cavalieri principle, the LV was sectioned in slices, bars and in fragments with different sizes. These fragments were organised in ascending order and the LV fragments were systematic randomly sampled (SURS) (Gundersen et al. 1999Gundersen H.J.G., Jensen E.B.V., Kiêu K. & Jensen J. 1999. The efficiency of systematic sampling in stereology reconsidered. J Microsc. 193(Pt 3):199-211. <http://dx.doi.org/10.1046/j.1365-2818.1999.00457.x> <PMid:10348656>
https://doi.org/10.1046/j.1365-2818.1999...
) (Fig.2). They were then embedded in agar in rubber moulds to generate small spheres (Isectors) (Nyengaard & Gundersen 1992Nyengaard J.R. & Gundersen H.J.G. 1992. The isector: a simple and direct method for generating isotropic, uniform random sections from small specimens. J. Microsc. 165(3):427-431. <http://dx.doi.org/10.1111/j.1365-2818.1992.tb01497.x>
https://doi.org/10.1111/j.1365-2818.1992...
) (Fig.3). To create isotropic orientation, the isectors were rolled on a surface before embedding in agar or in paraffin. The sections were stained by cresyl violet.
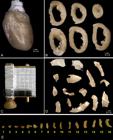
Left ventricle sampling. (A) Heart, (B) left ventricle slices, (C) tissue slicer, (D) left ventricle (LV) fragments with different size, (E) LV fragments arranged in ascending order.
Rubber mould to generate small spheres (Isectors). (B) Left ventricle fragment embedded in agar.
Heart microstructure quantification
Cardiac muscle fibre (CMF) density volume and cardiac interstitial (CI) density volume (Vv). In each section sampled, a point system, which was generated with the stereological software NewCASTTM (3.4.1.0 version) (Hoersholm, Denmark), was randomly superimposed on the image sections. The points hitting the CFM and CI were counted. The Vv (CMF) was estimated by dividing the number of CMF points counted by the total points hitting the entire ventricle wall. The same calculations were performed for CI: the total number of CI points divided by the total points hitting the entire ventricle wall. The error variance was calculated according to previous reports (Gundersen et al. 1999Gundersen H.J.G., Jensen E.B.V., Kiêu K. & Jensen J. 1999. The efficiency of systematic sampling in stereology reconsidered. J Microsc. 193(Pt 3):199-211. <http://dx.doi.org/10.1046/j.1365-2818.1999.00457.x> <PMid:10348656>
https://doi.org/10.1046/j.1365-2818.1999...
, Nyengaard 1999Nyengaard J.R. 1999. Stereologic methods and their application in kidney research. J. Am. Soc. Nephrol. 10(5):1100-1123. <PMid:10232698>). The error variance was 0.09 for control and MPTP groups and 0.11 for CMF and CI.
Cardiac muscle fibre total volume (V(CMF) and cardiac interstitial total volume (V(CI). Total volume of CMF was estimated by multiplying the Vv (CMF) by the left ventricle wall volume (VW). Following the same principle, the total volume of CI was estimated by multiplying the Vv (CI) by the left ventricle wall volume (VW).
Total number of cardiomyocyte nuclei. The total number of cardiomyocyte nuclei was estimated using the optical fractionator principle (West et al. 1991West M.J., Slomianka L. & Gundersen H.J. 1991. Unbiased stereological estimation of the total number of neurons in the subdivisions of the rat hippocampus using the optical fractionator. Anat. Rec. 231(4):482-497. <http://dx.doi.org/10.1002/ar.1092310411> <PMid:1793176>
https://doi.org/10.1002/ar.1092310411...
). The nuclei were counted using the stereological software NewCASTTM and sampled following the SURS principle (Gundersen et al. 1999Gundersen H.J.G., Jensen E.B.V., Kiêu K. & Jensen J. 1999. The efficiency of systematic sampling in stereology reconsidered. J Microsc. 193(Pt 3):199-211. <http://dx.doi.org/10.1046/j.1365-2818.1999.00457.x> <PMid:10348656>
https://doi.org/10.1046/j.1365-2818.1999...
). The nuclei hitting the green line or inside the counting frame were counted (Fig.4). The area fraction was obtained by dividing the counting frame area (1600μm2) by the step length (375.16μm x 375.16μm). The height fraction was calculated by dividing the dissector height by the overall mean section thickness. The error variance for estimation of the total number of cardiomyocyte nuclei (CE(N)) was estimated according to the procedures of Gundersen et al. (1999)Gundersen H.J.G., Jensen E.B.V., Kiêu K. & Jensen J. 1999. The efficiency of systematic sampling in stereology reconsidered. J Microsc. 193(Pt 3):199-211. <http://dx.doi.org/10.1046/j.1365-2818.1999.00457.x> <PMid:10348656>
https://doi.org/10.1046/j.1365-2818.1999...
.
Left ventricle showing the counting frame. The green line is the acceptance line and the red line is the forbidden line. Counted cardiomyocyte nuclei (A). Cresyl violet stain.
Mean number of cardiomyocyte nuclei per cardiomyocyte. Immunohistochemistry was used to define the cardiomyocyte limits and to estimate the mean number of nuclei per cardiomyocyte. Two antibodies were used for this purpose. Intercalated discs were visualised using the polyclonal rabbit pan-cadherin (1:500) (H-300 Santa Cruz Biotechnology, California, USA) and cardiomyocyte membranes were visualised using the polyclonal rabbit dystrophin (1:200) (Abcam, Cambridge, UK) (Fig.5).
Total number of cardiomyocytes. The total number of cardiomyocytes was estimated by dividing the total number of cardiomyocyte nuclei by the mean number of cardiomyocyte nuclei, which was obtained by sequencing the same cell stained by immunohistochemistry (IHC).
Statistical analysis
The results are presented as the mean of the observed coefficient of variation (CVobs), where CV obs = SD/mean. The data were analysed by the non-parametric Mann-Whitney test using Minitab statistical software version 17. Group differences were considered significant at p<0.05.
Results
Left ventricle volumes V (LV)
The wall volume V(W) was 73.25mm3 (0.05) and 73.87mm3 (0.09) for control and MPTP groups, respectively (p=0.67). The lumen volume V(L) was 13.58mm3 (0.49) for the control group and 12.15mm3 (0.22) for the MPTP group (p=0.52). The volume of the left ventricle V(LV) was 86.83mm3 (0.09) and 86.02mm3 (0.10) for the control and MPTP groups, respectively (p=0.52) (Fig.6A).
(A) Wall volume V (W), lumen volume V (L) and left ventricle volume V (LV) estimated using the Cavalieri principle. (B) Total volumes of cardiac muscle fibre (CMF) and cardiac interstitium (CI). (C) Total number of cardiomyocytes. Triangles indicate individual values; horizontal bars indicate the group means.
Cardiac muscle fibre (CMF) total volume and cardiac interstitial (CI) total volume
The CMF total volume was 43.17mm3 (0.12) and 42.20mm3 (0.12) for control and MPTP groups, respectively (p=0.53). The CI total volume was 30.07mm3 (0.14) and 31.70mm3 (0.26) for control and MPTP groups, respectively (p=0.67) (Fig.6B).
Total number of cardiomyocytes
The total number of cardiomyocytes was 5.7x106 (0.31) for the control group and 3.1x106 (0.44) for the MPTP group (p=0.02) (Fig.6C).
Discussion
We applied three-dimensional and unbiased methods to evaluate the heart in mice. Stereology has been used previously to quantify components of the heart to understand the consequences and progression of heart disease (Gruber et al. 2012Gruber C., Kohlstedt K., Loot A.E., Fleming I., Kummer W. & Mühlfeld C. 2012. Stereological characterization of left ventricular cardiomyocytes, capillaries, and innervation in the nondiabetic, obese mouse. Cardiovasc. Pathol. 21(4):346-354. <http://dx.doi.org/10.1016/j.carpath.2011.11.003> <PMid:22197049>
https://doi.org/10.1016/j.carpath.2011.1...
, Noorafshan et al. 2013Noorafshan A., Khazraei H., Mirkhani H. & Karbalay-Doust S. 2013. Stereological study of the diabetic heart of male rats. Lab. Anim. Res. 29(1):12-18. <http://dx.doi.org/10.5625/lar.2013.29.1.12> <PMid:23573103>
https://doi.org/10.5625/lar.2013.29.1.12...
, Schipke et al. 2014Schipke J., Mayhew T.M. & Mühlfeld C. 2014. Allometry of left ventricular myocardial innervation. J. Anat. 224(4):518-526. <http://dx.doi.org/10.1111/joa.12151> <PMid:24325466>
https://doi.org/10.1111/joa.12151...
, 2016Schipke J., Grimm C., Arnstein G., Kockskämper J., Sedej S. & Mühlfeld C. 2016. Cardiomyocyte loss is not required for the progression of left ventricular hypertrophy induced by pressure overload in female mice. J. Anat. 229(1):75-81. <http://dx.doi.org/10.1111/joa.12463> <PMid:26990078>
https://doi.org/10.1111/joa.12463...
).
Cardiac dysautonomia and sympathetic nervous system impairments of the heart have been reported in neurodegenerative diseases (Goldstein 2014Goldstein D.S. 2014. Dysautonomia in Parkinson’s disease: neurocardiological abnormalities. Compr. Physiol. 4:805-826. <http://dx.doi.org/10.1002/cphy.c130026> <PMid:24715569>
https://doi.org/10.1002/cphy.c130026...
, Joers & Emborg 2014Joers V. & Emborg M.E. 2014. Modeling and imaging cardiac sympathetic neurodegeneration in Parkinson’s disease. Am. J. Nucl. Med. Mol. Imaging 4(2):125-159. <PMid:24753981>, Piqueras-Flores et al. 2017Piqueras-Flores J., López-García A., Moreno-Reig Á., González-Martínez A., Hernández-González A., Vaamonde-Gamo J. & Jurado-Román A. 2017. Structural and functional alterations of the heart in Parkinson’s disease. Neurol. Res. 23:1-9. <PMid:29057715>). MPTP toxicity studies in mice have suggested a selective vulnerability of monoamine cells. These cells contain transporters that allow MPP+ to move into the cell interior. Therefore, using these transporters with MPP+ could provide access to postganglionic sympathetic nerves and subsequently damage those (Takatsu et al. 2000Takatsu H., Nishida H., Matsuo H., Watanabe S., Nagashima K., Wada H., Noda T., Nishigaki K. & Fujiwara H. 2000. Cardiac sympathetic denervation from the early stage of Parkinson’s disease: clinical and experimental studies with radiolabeled MIBG. J. Nucl. Med. 41(1):71-77. <PMid:10647607>). According to Fuller & Hemrick-Luecke (1986)Fuller R.W. & Hemrick-Luecke S.K. 1986. Depletion of norepinephrine in mouse heart by 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP) mimicked by 1-methyl-4-phenylpyridinium (MPP+) and not blocked by deprenyl. Life Sci. 39(18):1645-1650. <http://dx.doi.org/10.1016/0024-3205(86)90161-X> <PMid:3095601>
https://doi.org/10.1016/0024-3205(86)901...
, the conversion of MPTP to MPP+ is not required to deplete the heart norepinephrine.
The effects of MPTP vary according to mouse strain, sex, age, dose and intervals of application. The most susceptible strain is the C57/BL and male mice (Sonsalla & Heikkila 1986Sonsalla P.K. & Heikkila R.E. 1986. The influence of dose and dosing interval on MPTP- induced dopaminergic neurotoxity in mice. Eur. J. Pharmacol. 129(3):339-345. <http://dx.doi.org/10.1016/0014-2999(86)90444-9> <PMid:3490988>
https://doi.org/10.1016/0014-2999(86)904...
, Przedborski et al. 2001Przedborski S., Jackson-Lewis V., Naini A.B., Jakowec M., Petzinger G., Miller R. & Akram M. 2001. The parkinsonian toxin 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP): a technical review of its utility and safety. J Neurochem. 76(5):1265-1274. <http://dx.doi.org/10.1046/j.1471-4159.2001.00183.x> <PMid:11238711>
https://doi.org/10.1046/j.1471-4159.2001...
, Emborg 2004Emborg M.E. 2004. Evaluation of animal models of Parkinson’s disease for neuroprotective strategies. J. Neurosci. Methods 139(2):121-143. <http://dx.doi.org/10.1016/j.jneumeth.2004.08.004> <PMid:15488225>
https://doi.org/10.1016/j.jneumeth.2004....
, Luchtman et al. 2009Luchtman D.W., Shao D. & Song C. 2009. Behavior, neurotransmitters and inflammation in three regimens of the MPTP mouse model of Parkinson’s disease. Physiol. Behav. 98(1/2):130-138. <http://dx.doi.org/10.1016/j.physbeh.2009.04.021> <PMid:19410592>
https://doi.org/10.1016/j.physbeh.2009.0...
). The MPTP dose used in this study was reported in previous studies (Ren et al. 2004Ren J., Porter J.E., Wold L.E., Aberle N.S., Muralikrishnan D. & Haselton J.R. 2004. Depressed contractile function and adrenergic responsiveness of cardiac myocytes in an experimental model of Parkinson disease, the MPTP-treated mouse. Neurobiol. Aging 25(1):131-138. <http://dx.doi.org/10.1016/S0197-4580(03)00035-6> <PMid:14675739>
https://doi.org/10.1016/S0197-4580(03)00...
, Amino et al. 2008Amino T., Uchihara T., Tsunekawa H., Takahata K., Shimazu S., Mizusawa H. & Orimo S. 2008. Myocardial nerve fibers are preserved in MPTP-treated mice, despite cardiac sympathetic dysfunction. Neurosci. Res. 60(3):314-318. <http://dx.doi.org/10.1016/j.neures.2007.11.011> <PMid:18242750>
https://doi.org/10.1016/j.neures.2007.11...
) to induce sympathetic cardiac dysfunction and contractile function impairments. Three different doses were evaluated by Takatsu et al. (2000)Takatsu H., Nishida H., Matsuo H., Watanabe S., Nagashima K., Wada H., Noda T., Nishigaki K. & Fujiwara H. 2000. Cardiac sympathetic denervation from the early stage of Parkinson’s disease: clinical and experimental studies with radiolabeled MIBG. J. Nucl. Med. 41(1):71-77. <PMid:10647607>: 5mg/kg (one single dose), 5mg/kg (two doses) and 50mg/kg (two doses). The interval between doses was 16h. The later dose was reported to reduce more the 123-I-MIBG, an analogous of norepinephrine, indicating more cardiac damage and almost complete dopaminergic cell depletion. Based on this evidence, and to investigate the possible correlation between the loss of dopaminergic innervation and its consequences in cardiac muscle, this study focused strictly on morphological study of the myocardium at the macroscopic level and no functional or clinical study was done. In addition, the left ventricle volume was examined at the microscopic level and included the cardiac muscle fibre, along with cardiac interstitial volume and the number of cardiomyocytes, the functional heart unit.
The volume of the left ventricle was estimated using the Cavalieri principle (Gundersen et al. 1988Gundersen H.J., Bendtsen T.F., Korbo L., Marcussen N., Moller A., Nielsen K., Nyengaard J.R., Pakkenberg B., Sorensen F.B., Vesterby A. & West M.J. 1988. Some new, simple and efficient stereological methods and their use in pathological research and diagnosis. APMIS 96(5):379-394. <http://dx.doi.org/10.1111/j.1699-0463.1988.tb05320.x> <PMid:3288247>
https://doi.org/10.1111/j.1699-0463.1988...
, Michel & Cruz-Orive 1988Michel R.P. & Cruz-Orive L.M. 1988. Application of the Cavalieri principle and vertical sections method to lung: estimation of volume and pleural surface area. J. Microsc. 150(Pt 2):117-136. <http://dx.doi.org/10.1111/j.1365-2818.1988.tb04603.x> <PMid:3411604>
https://doi.org/10.1111/j.1365-2818.1988...
). Although this is a time-consuming method, it is an efficient and accurate method (Gundersen et al. 1999Gundersen H.J.G., Jensen E.B.V., Kiêu K. & Jensen J. 1999. The efficiency of systematic sampling in stereology reconsidered. J Microsc. 193(Pt 3):199-211. <http://dx.doi.org/10.1046/j.1365-2818.1999.00457.x> <PMid:10348656>
https://doi.org/10.1046/j.1365-2818.1999...
, Yan et al. 2003Yan X., Polo Carbayo J.J., Weibel E.R. & Hsia C.C. 2003. Variation of lung volume after fixation when measured by immersion of Cavalieri method. Am. J. Physiol. Lung Cell Mol. Physiol. 284(1):L242-L245. <http://dx.doi.org/10.1152/ajplung.00184.2002> <PMid:12388332>
https://doi.org/10.1152/ajplung.00184.20...
, Van Vré et al. 2007Van Vré E.A., Van Beusekom H.M., Vrints C.J., Bosmans J.M., Bult H. & Van Der Giessen W.J. 2007. Stereology: a simplified and more time-efficient method than planimetry for the quantitative analysis of vascular structures in different models of intimal thickening. Cardiovasc. Pathol. 16(1):43-50. <http://dx.doi.org/10.1016/j.carpath.2006.08.001> <PMid:17218214>
https://doi.org/10.1016/j.carpath.2006.0...
), and it is the most appropriate method for estimating the volume of cavity organs. We estimated the left ventricle volume (LV) by summing the lumen volume (L) and the wall volume (W). These two volumes indicate the size of the left ventricle chamber and the thickness of the wall, respectively. An analysis of each of these volumes is useful because they might shed light on how the heart responds to factors such as haemodynamic load, as well as neuroendocrine and chemical stimuli that may cause cardiac tissue remodelling. In our study, the chemical effects of MPTP did not change the left ventricle volume, including the lumen and wall volumes.
At the microscopic level, the cardiac muscle fibre (CMF) and the cardiac interstitial (CI) total volumes can assess cardiac tissue remodelling. Cardiac remodelling is a complex process involving changes in cardiomyocytes and in the interstitium that results in progressive structural and functional alterations, including cardiac dilatation, interstitial fibrosis and reduction in contractility and relaxation (Remme 2003Remme W.J. 2003. Pharmacological modulation of cardiovascular remodeling: A guide to heart failure therapy. Cardiovasc Drugs Ther 17(4):349-360.). The use of MPTP did not change the CMF or the CI total volumes, but the toxic effect of MPTP decreased the total number of cardiomyocyte by 45%. Analysing the cardiac tissue, from the remodelling point of view, this reduction in the number of cardiomyocytes should be accompanied by a reduction in the CMF total volume. In our case, however, this did not happen. Although we did not estimate the cardiomyocyte volume, we suggest that the remaining cardiomyocyte may have increased in size as a compensatory process to maintain cardiac function. Hypertrophy is one of the several mechanisms of cardiac musculature that result from factors (ischaemia, toxic substances, microorganisms etc.) that damage cardiomyocytes (Pangonytė et al. 2008Pangonytė D., Stalioraitytė E., Žiuraitienė R., Kazlauskaitė D., Palubinskienė J. & Balnytė I. 2008. Cardiomyocyte remodeling in ischemic heart disease. Medicina, Kaunas 44(11):848-854. <http://dx.doi.org/10.3390/medicina44110107> <PMid:19124961>
https://doi.org/10.3390/medicina44110107...
).
In summary, this study showed that the effects of the MPTP reduced the number of cardiomyocytes in the mouse left ventricle without changing the ventricular volume. We believe our morphoquantitative results, obtained using a designed stereological method, can be applied in the heart of other animal models to better elucidate the pathophysiological mechanisms of cardiac sympathetic denervation.
Acknowledgements
This study was supported by the São Paulo Research Foundation (FAPESP- grant number 2007/50524-5). The author gratefully acknowledges the Laboratory of Stochastic Stereology and Chemical Anatomy from the University of São Paulo (LSSCA-USP).
References
- Amino T., Orimo S., Itoh Y., Takahashi A., Uchihara T. & Mizusawa H. 2005. Profound cardiac sympathetic denervation occurs in Parkinson disease. Brain Pathol. 15(1):29-34. <http://dx.doi.org/10.1111/j.1750-3639.2005.tb00097.x> <PMid:15779234>
» https://doi.org/10.1111/j.1750-3639.2005.tb00097.x - Amino T., Uchihara T., Tsunekawa H., Takahata K., Shimazu S., Mizusawa H. & Orimo S. 2008. Myocardial nerve fibers are preserved in MPTP-treated mice, despite cardiac sympathetic dysfunction. Neurosci. Res. 60(3):314-318. <http://dx.doi.org/10.1016/j.neures.2007.11.011> <PMid:18242750>
» https://doi.org/10.1016/j.neures.2007.11.011 - Emborg M.E. 2004. Evaluation of animal models of Parkinson’s disease for neuroprotective strategies. J. Neurosci. Methods 139(2):121-143. <http://dx.doi.org/10.1016/j.jneumeth.2004.08.004> <PMid:15488225>
» https://doi.org/10.1016/j.jneumeth.2004.08.004 - Fuller R.W. & Hemrick-Luecke S.K. 1986. Depletion of norepinephrine in mouse heart by 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP) mimicked by 1-methyl-4-phenylpyridinium (MPP+) and not blocked by deprenyl. Life Sci. 39(18):1645-1650. <http://dx.doi.org/10.1016/0024-3205(86)90161-X> <PMid:3095601>
» https://doi.org/10.1016/0024-3205(86)90161-X - Goldstein D.S., Holmes C., Li S.T., Bruce S., Metman L.V. & Cannon 3rd R.O. 2000. Cardiac sympathetic denervation in Parkinson disease. Ann. Intern. Med. 133(5):338-347. <http://dx.doi.org/10.7326/0003-4819-133-5-200009050-00009> <PMid:10979878>
» https://doi.org/10.7326/0003-4819-133-5-200009050-00009 - Goldstein D.S., Eldadah B.A., Holmes C., Pechnik S., Moak J., Saleem A. & Sharabi Y. 2005. Neurocirculatory abnormalities in Parkinson Disease with orthostatic hypotension independence from levodopa treatment. Hypertension 46(6):1333-1339. <http://dx.doi.org/10.1161/01.HYP.0000188052.69549.e4> <PMid:16216982>
» https://doi.org/10.1161/01.HYP.0000188052.69549.e4 - Goldstein D.S. 2014. Dysautonomia in Parkinson’s disease: neurocardiological abnormalities. Compr. Physiol. 4:805-826. <http://dx.doi.org/10.1002/cphy.c130026> <PMid:24715569>
» https://doi.org/10.1002/cphy.c130026 - Gruber C., Kohlstedt K., Loot A.E., Fleming I., Kummer W. & Mühlfeld C. 2012. Stereological characterization of left ventricular cardiomyocytes, capillaries, and innervation in the nondiabetic, obese mouse. Cardiovasc. Pathol. 21(4):346-354. <http://dx.doi.org/10.1016/j.carpath.2011.11.003> <PMid:22197049>
» https://doi.org/10.1016/j.carpath.2011.11.003 - Gundersen H.J., Bendtsen T.F., Korbo L., Marcussen N., Moller A., Nielsen K., Nyengaard J.R., Pakkenberg B., Sorensen F.B., Vesterby A. & West M.J. 1988. Some new, simple and efficient stereological methods and their use in pathological research and diagnosis. APMIS 96(5):379-394. <http://dx.doi.org/10.1111/j.1699-0463.1988.tb05320.x> <PMid:3288247>
» https://doi.org/10.1111/j.1699-0463.1988.tb05320.x - Gundersen H.J.G., Jensen E.B.V., Kiêu K. & Jensen J. 1999. The efficiency of systematic sampling in stereology reconsidered. J Microsc. 193(Pt 3):199-211. <http://dx.doi.org/10.1046/j.1365-2818.1999.00457.x> <PMid:10348656>
» https://doi.org/10.1046/j.1365-2818.1999.00457.x - Gundersen H.J.G. 2002. The smooth fractionator. J. Microsc. 207(Pt 3):191-210. http://dx.doi.org/10.1046/j.1365-2818.2002.01054.x.
» https://doi.org/10.1046/j.1365-2818.2002.01054.x. - Haensch C.A., Lerch H., Jörg J.A. & Isenmann S. 2009. Cardiac denervation occurs independent of orthostatic hypotension and impaired heart rate variability in Parkinson’s disease. Parkinsonism Relat. Disord. 15(2):134-137. <http://dx.doi.org/10.1016/j.parkreldis.2008.04.031> <PMid:18515170>
» https://doi.org/10.1016/j.parkreldis.2008.04.031 - Jackson-Lewis V., Blesa J. & Przedborski S. 2012. Animal models of Parkinson’s disease. Parkinsonism Relat. Disord. 18(Suppl.1):183-185. <http://dx.doi.org/10.1016/S1353-8020(11)70057-8> <PMid:22166429>
» https://doi.org/10.1016/S1353-8020(11)70057-8 - Jain S. & Goldstein D.S. 2012. Cardiovascular dysautonomia in Parkinson disease: from pathophysiology to pathogenesis. Neurobiol. Dis. 46(3):572-580. <http://dx.doi.org/10.1016/j.nbd.2011.10.025> <PMid:22094370>
» https://doi.org/10.1016/j.nbd.2011.10.025 - Joers V. & Emborg M.E. 2014. Modeling and imaging cardiac sympathetic neurodegeneration in Parkinson’s disease. Am. J. Nucl. Med. Mol. Imaging 4(2):125-159. <PMid:24753981>
- Li S.T., Dendi R., Holmes C. & Goldstein D. 2002. Progressive loss of cardiac sympathetic innervation in Parkinson’s disease. Ann. Neurol. 52(2):220-223. <http://dx.doi.org/10.1002/ana.10236> <PMid:12210793>
» https://doi.org/10.1002/ana.10236 - Luchtman D.W., Shao D. & Song C. 2009. Behavior, neurotransmitters and inflammation in three regimens of the MPTP mouse model of Parkinson’s disease. Physiol. Behav. 98(1/2):130-138. <http://dx.doi.org/10.1016/j.physbeh.2009.04.021> <PMid:19410592>
» https://doi.org/10.1016/j.physbeh.2009.04.021 - Michel R.P. & Cruz-Orive L.M. 1988. Application of the Cavalieri principle and vertical sections method to lung: estimation of volume and pleural surface area. J. Microsc. 150(Pt 2):117-136. <http://dx.doi.org/10.1111/j.1365-2818.1988.tb04603.x> <PMid:3411604>
» https://doi.org/10.1111/j.1365-2818.1988.tb04603.x - Noorafshan A., Khazraei H., Mirkhani H. & Karbalay-Doust S. 2013. Stereological study of the diabetic heart of male rats. Lab. Anim. Res. 29(1):12-18. <http://dx.doi.org/10.5625/lar.2013.29.1.12> <PMid:23573103>
» https://doi.org/10.5625/lar.2013.29.1.12 - Nyengaard J.R. & Gundersen H.J.G. 1992. The isector: a simple and direct method for generating isotropic, uniform random sections from small specimens. J. Microsc. 165(3):427-431. <http://dx.doi.org/10.1111/j.1365-2818.1992.tb01497.x>
» https://doi.org/10.1111/j.1365-2818.1992.tb01497.x - Nyengaard J.R. 1999. Stereologic methods and their application in kidney research. J. Am. Soc. Nephrol. 10(5):1100-1123. <PMid:10232698>
- Pangonytė D., Stalioraitytė E., Žiuraitienė R., Kazlauskaitė D., Palubinskienė J. & Balnytė I. 2008. Cardiomyocyte remodeling in ischemic heart disease. Medicina, Kaunas 44(11):848-854. <http://dx.doi.org/10.3390/medicina44110107> <PMid:19124961>
» https://doi.org/10.3390/medicina44110107 - Piqueras-Flores J., López-García A., Moreno-Reig Á., González-Martínez A., Hernández-González A., Vaamonde-Gamo J. & Jurado-Román A. 2017. Structural and functional alterations of the heart in Parkinson’s disease. Neurol. Res. 23:1-9. <PMid:29057715>
- Przedborski S., Jackson-Lewis V., Naini A.B., Jakowec M., Petzinger G., Miller R. & Akram M. 2001. The parkinsonian toxin 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP): a technical review of its utility and safety. J Neurochem. 76(5):1265-1274. <http://dx.doi.org/10.1046/j.1471-4159.2001.00183.x> <PMid:11238711>
» https://doi.org/10.1046/j.1471-4159.2001.00183.x - Remme W.J. 2003. Pharmacological modulation of cardiovascular remodeling: A guide to heart failure therapy. Cardiovasc Drugs Ther 17(4):349-360.
- Ren J., Porter J.E., Wold L.E., Aberle N.S., Muralikrishnan D. & Haselton J.R. 2004. Depressed contractile function and adrenergic responsiveness of cardiac myocytes in an experimental model of Parkinson disease, the MPTP-treated mouse. Neurobiol. Aging 25(1):131-138. <http://dx.doi.org/10.1016/S0197-4580(03)00035-6> <PMid:14675739>
» https://doi.org/10.1016/S0197-4580(03)00035-6 - Schipke J., Mayhew T.M. & Mühlfeld C. 2014. Allometry of left ventricular myocardial innervation. J. Anat. 224(4):518-526. <http://dx.doi.org/10.1111/joa.12151> <PMid:24325466>
» https://doi.org/10.1111/joa.12151 - Schipke J., Grimm C., Arnstein G., Kockskämper J., Sedej S. & Mühlfeld C. 2016. Cardiomyocyte loss is not required for the progression of left ventricular hypertrophy induced by pressure overload in female mice. J. Anat. 229(1):75-81. <http://dx.doi.org/10.1111/joa.12463> <PMid:26990078>
» https://doi.org/10.1111/joa.12463 - Sonsalla P.K. & Heikkila R.E. 1986. The influence of dose and dosing interval on MPTP- induced dopaminergic neurotoxity in mice. Eur. J. Pharmacol. 129(3):339-345. <http://dx.doi.org/10.1016/0014-2999(86)90444-9> <PMid:3490988>
» https://doi.org/10.1016/0014-2999(86)90444-9 - Sundström E., Strömberg I., Tsutsumi T., Olson L. & Jonsson G. 1987. Studies on the effect of 1-methyl-4-phenyl-1,2,3,6-tetrahydroxypyridine (MPTP) on central catecholamine neurons in C57 BL/6 mice: comparison with three other strains of mice. Brain Res. 405(1):26-38. <http://dx.doi.org/10.1016/0006-8993(87)90986-3> <PMid:2882814>
» https://doi.org/10.1016/0006-8993(87)90986-3 - Takatsu H., Nishida H., Matsuo H., Watanabe S., Nagashima K., Wada H., Noda T., Nishigaki K. & Fujiwara H. 2000. Cardiac sympathetic denervation from the early stage of Parkinson’s disease: clinical and experimental studies with radiolabeled MIBG. J. Nucl. Med. 41(1):71-77. <PMid:10647607>
- Van Vré E.A., Van Beusekom H.M., Vrints C.J., Bosmans J.M., Bult H. & Van Der Giessen W.J. 2007. Stereology: a simplified and more time-efficient method than planimetry for the quantitative analysis of vascular structures in different models of intimal thickening. Cardiovasc. Pathol. 16(1):43-50. <http://dx.doi.org/10.1016/j.carpath.2006.08.001> <PMid:17218214>
» https://doi.org/10.1016/j.carpath.2006.08.001 - West M.J., Slomianka L. & Gundersen H.J. 1991. Unbiased stereological estimation of the total number of neurons in the subdivisions of the rat hippocampus using the optical fractionator. Anat. Rec. 231(4):482-497. <http://dx.doi.org/10.1002/ar.1092310411> <PMid:1793176>
» https://doi.org/10.1002/ar.1092310411 - Yan X., Polo Carbayo J.J., Weibel E.R. & Hsia C.C. 2003. Variation of lung volume after fixation when measured by immersion of Cavalieri method. Am. J. Physiol. Lung Cell Mol. Physiol. 284(1):L242-L245. <http://dx.doi.org/10.1152/ajplung.00184.2002> <PMid:12388332>
» https://doi.org/10.1152/ajplung.00184.2002 - Ziemssen T. & Reichmann H. 2010. Cardiovascular autonomic dysfunction in Parkinson’s disease. J. Neurol. Sci. 289(1-2):74-80. <http://dx.doi.org/10.1016/j.jns.2009.08.031> <PMid:19740484>
» https://doi.org/10.1016/j.jns.2009.08.031
Publication Dates
-
Publication in this collection
01 July 2019 -
Date of issue
May 2019
History
-
Received
24 Aug 2018 -
Accepted
26 Nov 2018