ABSTRACT:
Adult learning, or andragogy, provides a novel way of appreciating using food analogies as an effective learning tool in veterinary pathology. Facilitation of adult learning requires that new concepts be presented in a way that draws on the learner’s experience. Because veterinary students will have had considerable experience with a plethora of food items prior to enrolling in a pathology course, food analogies can provide an easy conduit for incorporating key learning concepts regarding veterinary pathology. In this paper, many of these analogies are presented, along with the mechanisms responsible for each of the characteristic lesions, in the hopes that their usefulness in the classroom can be highlighted to create a more engaging and facilitated learning environment.
INDEX TERMS:
Pathology; veterinary pathology; food analogies
RESUMO:
A aprendizagem de adultos, ou andragogia, é uma nova maneira de apreciar o uso de analogias de alimentos como uma ferramenta eficaz no aprendizado em patologia veterinária. A facilitação da aprendizagem de adultos requer que novos conceitos sejam apresentados de uma forma que se baseie na experiência do aluno. Como os estudantes de medicina veterinária já terão sido expostos a um número considerável de tipos de alimentos antes de se matricularem na disciplina de patologia, as analogias de alimentos podem fornecer um ótimo conduto para incorporar os conceitos-chave na aprendizagem da patologia veterinária. Neste artigo, muitos dessas analogias são apresentadas juntamente com os mecanismos responsáveis por cada uma das lesões características, na esperança de que sua utilidade na sala de aula possa ser destacada para criar um ambiente de aprendizado mais envolvente e favorável.
TERMOS DE INDEXAÇÃO:
patologia veterinária; analogias de alimentos
Introduction
Pathology is a central element in the teaching/learning of medicine. It is the bridge that helps students understand underlying mechanisms of disease and make the connection between organ dysfunction and clinical syndromes. If students can appreciate mechanisms of disease through judging the morphologic changes in an organ, they can better craft differential diagnoses and propose more relevant therapies. Unfortunately, the teaching of pathology often involves rote learning regarding lesions, making it difficult for students to remember each lesion and/or to grasp the significance of the morphologic changes.
Andragogy, or the science of adult learning, has received increasing attention in recent years. It is now established that adults, who includes all veterinary students globally, learn in ways that are different from children (pedagogy). One of the tenets of andragogy is that adults bring a wide range of experience into the learning environment. If adults can take a new concept and compare it to something with which they are already familiar, the filing into long-term memory is greatly facilitated (Taylor & Marienau 2016Taylor K. & Marienau C. 2016. The learning, changing adult brain: experiences, memory and emotions, p.33-58. In: Ibid. (Eds), Facilitating Learning with the Adult Brain in Mind: a conceptual and practical guide. Jossey-Bass, San Francisco.).
Although there are many who disparage the use of “kitchen pathology” as a means of learning, food analogies are widely used in human medicine (Mark et al. 2015Mark N.M., Lessing J.N., Buckley S.A. & Tierney Junior J.R.L.M. 2015. Diagnostic utility of food terminology: culinary clues for the astute diagnostician. Am. J. Med. 128(9):933-934. <http://dx.doi.org/10.1016/j.amjmed.2015.03.018> <PMid:25863151>
https://doi.org/10.1016/j.amjmed.2015.03...
, Vimal & Nishanthi 2017Vimal M. & Nishanthi A. 2017. Complex pa’thological terminologies simplified with food eponyms. J. Clin. Diagn. Res. 11:EE01-EE06., Kobayashi & Iwasaki 2019Kobayashi A. & Iwasaki H. 2019. Doughnut lesions. N. Engl. J. Med. 380(10):10. <http://dx.doi.org/10.1056/NEJMicm1810556> <PMid:30855746>
https://doi.org/10.1056/NEJMicm1810556...
, Sacks 2019Sacks C.A. 2019. Doughnut lesions. N. Engl. J. Med 380(10):10. <PMid:30855746>). Indeed, when “kitchen pathology” terms are viewed in light of the science of adult learning, they make great sense and can greatly facilitate the learning of lesions. It is the purpose of this paper to review some of the prominent lesions that resemble food, to give the reasons for the appearance, and to hope that they can be used successfully to allow the field of pathology to be “easier to digest”.
MACROSCOPIC FINDINGS
Body cavities
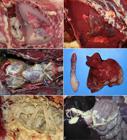
Milky effusion. Chylous pleural effusion, also known as chylothorax, is defined by the presence of chyle in the pleural space. At the macroscopic level, the diagnosis of chylothorax is pursued when the appearance of the fluid is “milky” (in Portuguese: “leitoso”) (Fig.1A). However, biochemical analysis is imperative to determine the type of effusion. When a chylous effusion occurs in the peritoneal cavity, it is called chyleabdomen. Chyle is a non-inflammatory, alkaline, and bacteriostatic fluid of the lymphatic system composed mainly of fat, cholesterol, electrolytes, proteins, glucose, and abundant lymphocytes, mostly T lymphocytes (Tutor 2014Tutor J.D. 2014. Chylothorax in infants and children. Pediatrics 133(4):722-733. <http://dx.doi.org/10.1542/peds.2013-2072> <PMid:24685960>
https://doi.org/10.1542/peds.2013-2072...
).
(A) Chylothorax, thorax, cat. Food analogy: milky fluid. Approximately 80mL of white, opaque fluid fills the thoracic cavity. The pressure of the accumulated fluid resulted in pulmonary atelectasis. (B) Marked, diffuse, chronic, pyogranulomatous pleuritis with secondary pulmonary atelectasis, thoracic cavity, dog. Food analogy: tomato soup. A thick red exudate partially fills the thoracic cavity. Note the atelectasis of the lungs. Mesothelial hyperplasia and hypertrophy is on the caudoventral area of the thoracic cavity, which it is typical in chronic inflammation of serosal surfaces. (C) Marked, diffuse, urate deposits; coelomic cavity; parrot; visceral gout. Food analogy: frosting or sugar icing. The heart and liver are diffusely covered by a thin layer of white, opaque, gritty material (urates). (D) Mild to moderate, multifocal to coalescing, chronic, fibrinous and granulomatous splenic and hepatic capsulitis, cat; feline infectious peritonitis. Food analogy: frosting or sugar icing. The splenic and hepatic capsular surfaces are multifocally covered by fibrin. (E) Fibrinous pleuritis, thoracic cavity, horse. Food analogy: scrambled eggs. Large accumulations of yellow, friable exudate (fibrin) loosely adhered to the parietal and visceral pleurae. (F) Moderate, multifocal to coalescing, acute, fibrinous peritonitis; peritoneum; pig; Glasser’s disease. Food analogy: scrambled eggs. Multifocal strands of fibrin are on the surface of the liver and spiral colon.
In domestic animals, mainly dogs and cats, chylothorax occurs in cases of rupture of the thoracic duct (trauma) or, more commonly, in association with obstruction of impaired flow in the thoracic duct (López & Martinson 2017López A. & Martinson S.A. 2017. Respiratory system, mediastinum, and pleurae, p.471-558. In: Zachary J.F. (Ed.), Pathologic Basis of Veterinary Disease. 6th ed. Elsevier, St Louis. <http://dx.doi.org/10.1016/B978-0-323-35775-3.00009-6>
https://doi.org/10.1016/B978-0-323-35775...
) due to regional space-occupying lesions or cardiac disease. In many cases, the cause of the chylous effusion is not found at necropsy.
Tomato soup effusion. In dogs and cats, red-brown exudate within the thoracic cavity that resembles tomato soup (in Portuguese: “sopa de tomate”) (Fig.1B) is characteristic of chronic pyogranulomatous pleuritis caused by the following bacteria: Nocardia, Actinomyces, or Bacteroides. Diffuse red velvety to granular thickening of pleura reflects the fibrovascular tissue; the leakage of erythrocytes from newly formed blood vessels adds the red color to the exudate (Lopez & Martinson 2017López A. & Martinson S.A. 2017. Respiratory system, mediastinum, and pleurae, p.471-558. In: Zachary J.F. (Ed.), Pathologic Basis of Veterinary Disease. 6th ed. Elsevier, St Louis. <http://dx.doi.org/10.1016/B978-0-323-35775-3.00009-6>
https://doi.org/10.1016/B978-0-323-35775...
). This exudate may contain yellowish soft granules called “sulfur granules”. In chronic cases, the exudate turns brown, similar to chocolate milkshake. In dogs and cats, the disease usually occurs from inoculation of organisms through puncture wounds or foreign bodies, bites or scratches (Edwards 2006Edwards D.F. 2006. Actinomycosis and nocardiosis, p.451-461. In: Greene C.E. (Ed.), Infectious Diseases of the Dog and Cat. 3rd ed. Elsevier, St Louis.).
Sugar icing or frosting. Gout is a common metabolic disorder that results in hyperuricemia and abnormal deposition of white-chalky material on the surface of many visceral organs as well as joints of avian, reptilian and mammalian species (Fig.1C) (Lierz 2003Lierz M. 2003. Avian renal disease: pathogenesis, diagnosis, and therapy. Vet. Clin. N. Am., Exotic Anim. Pract. 6(1):29-55. <PMid:12616833>, Fink et al. 2018Fink D., Drumond L., Basílio M.I., Sartori C.M., Andrade T.R., Santos N.Z. & Cremer M.J. 2018. Gota úrica visceral em bobo-pequeno (Puffinus puffinus) no sul do Brasil. Arq. Bras. Med. Vet. Zootec. 70(2):486-490. <http://dx.doi.org/10.1590/1678-4162-9916>
https://doi.org/10.1590/1678-4162-9916...
). The urate aggregates have a similar appearance to frosting (in Portuguese: “glacê/cobertura de bolo”). When plasma concentration of uric acid exceeds solubility, it precipitates and accumulates on the surfaces of pericardial sac, kidney, liver, lungs, subcutaneous tissue (visceral gout) and joints (articular gout) (Lierz 2003Lierz M. 2003. Avian renal disease: pathogenesis, diagnosis, and therapy. Vet. Clin. N. Am., Exotic Anim. Pract. 6(1):29-55. <PMid:12616833>).
Visceral gout may occur for two main reasons: reduced excretion of uric acid or increased production of uric acid (Pollock 2006Pollock C. 2006. Diagnosis and treatment of avian renal disease. Vet. Clin. N. Am., Exotic Anim. Pract. 9(1):107-128. <PMid:16407082>). Gout is rather a clinical manifestation resulting of severe renal injury than a disease, and might be caused by toxic, infectious, nutritional or a combination of these factors. Dehydration due to water deprivation is a common factor associated with the onset of visceral gout. Nephrotoxic agents, such as mycotoxins (e.g. ochratoxin A), heavy metals (e.g. lead), anti-inflammatory drugs (e.g. diclofenac) and antibiotics (e.g. gentamycin), may also interfere with normal kidney function and consequently cause visceral gout (Shultz et al. 2004Shultz S., Baral H.S., Charman S., Cunningham A.A., Das D., Ghalsasi G.R., Goudar M.S., Green R.E., Jones A., Nighot P., Pain D.J. & Prakash V. 2004. Diclofenac poisoning is widespread in declining vulture populations across the Indian subcontinent. Proc. Biol. Sci. 271(Suppl.6):S458-S460. <http://dx.doi.org/10.1098/rsbl.2004.0223> <PMid:15801603>
https://doi.org/10.1098/rsbl.2004.0223...
, Herbert et al. 2011Herbert J.D., Coulson J.O. & Coulson T.D. 2011. Quantification of tissue uric acid levels in a Harris’s hawk with visceral gout. BioOne 55(3):513-515. <PMid:22017058>). Bacterial (e.g. Pasteurella multocida) and viral (e.g. astrovirus, haemorrhagic polyomavirus) causes have also been implicated as potential etiologies for visceral gout (Rahimi et al. 2015Rahimi M., Minoosh Z. & Haghighi S. 2015. Visceral urate deposition in a little bittern (Ixobrychus minutus). Vet. Res. Forum, Int. Q. J. 6(2):177-180., Zhang et al. 2018Zhang Q., Cao Y., Wang J., Fu G., Sun M., Zhang L., Meng L., Cui G., Huang Y., Hu X. & Su J. 2018. Isolation and characterization of an astrovirus causing fatal visceral gout in domestic goslings. Emerg. Microbes Infect. 7(1):71. <http://dx.doi.org/10.1038/s41426-018-0074-5> <PMid:29674726>
https://doi.org/10.1038/s41426-018-0074-...
). On histology, urate tophi are comprised of radiating array of colorless to eosinophilic, acicular, crystalline deposits that are birefringent under polarized light.
Frosting of the surface of the abdominal organs, mainly on the surface of the liver and spleen, is observed in the wet or effusive form of feline infectious peritonitis (FIP) in cats. The disease is caused by a virulent feline coronavirus, called feline infectious peritonitis virus (FIPV), a RNA virus of the family Coronaviridae. FIPV infects and replicates in monocytes/macrophages and can gain access to the blood and induce the disease. The host determines the severity of FIP lesions. When there is no cell-mediated immune response from the host, the wet form develops. In such cases, the abdomen usually contains gelatinous, straw-colored fluid with fibrin strands both within the fluid and adhered to all serosal surfaces (fibrinous peritonitis) (Fig.1D). This arises secondary to phlebitis, mediated and dominated by activated virus-infected monocytes (Kipar & Meli 2014Kipar A. & Meli M.L. 2014. Feline infectious peritonitis: still an enigma? Vet. Pathol. 51(2):505-526. <http://dx.doi.org/10.1177/0300985814522077> <PMid:24569616>
https://doi.org/10.1177/0300985814522077...
).
Scrambled eggs. Scrambled eggs is a dish made from eggs mixed stirred or beaten together with milk in a pan while being gently heated. Florid fibrinous exudate on the serosal surfaces, mainly in the thoracic and peritoneal cavities, resembles scrambled eggs (in Portuguese: “ovos mexidos”) due to its thick, stringy, light or bright yellow appearance (Fig.1E) (Peek & McGuirk 2008Peek S.F. & McGuirk S.M. 2008. Pericarditis, p.56-58. In: Divers T.J. & Peek D.F. (Eds), Rebhun’s Diseases of Dairy Cattle. 2nd ed. W.B. Saunders, Philadelphia). Fibrin originates from leakage of fibrinogen from capillaries and postcapillary venules secondary to severe endothelial cell injury during the acute inflammatory response.
Fibrinous exudate occurs secondarily to many bacterial infections, mainly in horses, ruminants and pigs. A few classical examples in veterinary medicine include: polyserositis in Glasser’s disease caused by Glaesserella (Haemophilus) parasuis in pigs (Fig.1F), pleuropneumonia due to Streptococcus equi subsp. zooepidemicus in horses, traumatic reticulopericarditis (hardware disease) in cattle or any bacterial infection secondary to failure of passive immunity transfer in neonatal foals, ruminants and pigs.
Several organs
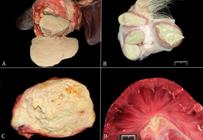
Sweetened condensed milk and cheese. The content of an abscess is usually tan to yellow, but it can widely vary in consistency. By definition, an abscess is a collection of pus (dead neutrophils) surrounded by a fibrous capsule. Neutrophils release many enzymes from their cytoplasmic granules (e.g. myeloperoxidase) to kill pyogenic bacteria such as Staphylococcus sp. or Streptococcus sp. among others. As the lesion progresses to a chronic stage, it walls off the inciting agent by producing a fibrous capsule around it (Ackermann 2017Ackermann M.R. 2017. Inflammation and healing, p.73-131. In: Zachary J.F. (Ed.), Pathologic Basis of Veterinary Disease. 6th ed. Elsevier, St Louis. <http://dx.doi.org/10.1016/B978-0-323-35775-3.00003-5>.
https://doi.org/10.1016/B978-0-323-35775...
). The content of the abscesses is formed by liquefactive necrosis, called pus, and resembles sweetened condensed milk (in Portuguese: “leite condensado”) (Fig.2A) or slightly creamy with a consistency of cream cheese (in Portuguese: “requeijão”) (Fig.2B). In chronic abscesses or in the center of granulomas or pyogranulomas, the contents are caseous (Latin word for cheese) and become curdled with an appearance of cottage cheese (Fig.2C). Caseous necrosis occurs in the center of chronic abscesses of caseous lymphadenitis in sheep and goats and in the center of tuberculoid granulomas (Ackermann 2017Ackermann M.R. 2017. Inflammation and healing, p.73-131. In: Zachary J.F. (Ed.), Pathologic Basis of Veterinary Disease. 6th ed. Elsevier, St Louis. <http://dx.doi.org/10.1016/B978-0-323-35775-3.00003-5>.
https://doi.org/10.1016/B978-0-323-35775...
).
(A) Focal, chronic, hepatic abscess; liver; horse. Food analogy for the abscess content: sweetened condensed milk. On cut section, the contents are slightly yellow and liquefied (pus) surrounded by a thick fibrous capsule. (B) Multifocal, subcutaneous, chronic abscesses; haired skin; goat; caseous lymphadenitis. Food analogy for the abscess content: cream cheese. The contents of these abscesses are slightly yellow and creamy (pus) and are surrounded by a thin fibrous capsule. (C) Marked, diffuse, chronic, pyogranulomatous lymphadenitis; mesenteric lymph node; foal; Rhodococcus equi infection. Food analogy for the content: caseous. The mesenteric lymph node is markedly enlarged and replaced by abundant caseous material. (D) Moderate, multifocal, acute, suppurative embolic nephritis or glomerulitis; kidney; foal; septicemic actinobacillosis. Food analogy for the distribution of the lesions: miliary. Miliary, 0.1-0.3cm in diameter, pale tan foci are randomly distributed through the renal cortex.
Millet seeds. Millets are a group of highly variable small-seeded grasses, widely grown around the world as cereal crops or grains for fodder and human food. Accordingly, in descriptive pathology, the term miliary (which stems from miliarius, the Latin word for millet seeds; in Portuguese: “miliar”) is used to describe a specific type of multifocal distribution (Stromberg 2015Stromberg P. 2015. Fundamental principles of descriptive anatomic pathology. Available at <Available at https://www.coursehero.com/file/37810733/DESCRIPCION-MACROSCOPICApdf/
> Accessed on Apr. 12, 2019.
https://www.coursehero.com/file/37810733...
) in which there are numerous tiny foci randomly distributed (Fig.2D).
Alimentary system
Rice grains. Developmental stages of several parasites such as Sarcocystis gigantea; Sarcocystis rileyi and Dipylidium caninum resemble rice grains (in Portuguese: “grãos de arroz”) on macroscopic examination. Sarcocystis gigantea is an Apicomplexan protozoan in the esophagus, and less commonly in the tongue, larynx and diaphragm of sheep (Fig.3A) (Dubey et al. 2015Dubey J.P., Calero-Bernal R., Rosenthal B.M., Speer C.A. & Fayer R. 2015. Sarcocystosis of Animals and Humans. CRC Press, Boca Raton, p.1-481. <http://dx.doi.org/10.1201/b19184>
https://doi.org/10.1201/b19184...
). Macrocysts remain quiescent and do not cause an inflammatory reaction to the surrounding musculature. Macrocysts grow for several years and bradyzoites are not highly infectious for cats, which are the definitive hosts. Although S. gigantea is mildly pathogenic for sheep and is not zoonotic, carcasses with cysts are still condemned during meat inspection at the slaughterhouse (Dubey et al. 2015Dubey J.P., Calero-Bernal R., Rosenthal B.M., Speer C.A. & Fayer R. 2015. Sarcocystosis of Animals and Humans. CRC Press, Boca Raton, p.1-481. <http://dx.doi.org/10.1201/b19184>
https://doi.org/10.1201/b19184...
). Sarcocystis rileyi occurs in the pectoral muscle of ducks and the presence of numerous macrocysts is known by the hunters as “rice breast” disease. S. rileyi infection in ducks occurs only in North America, which is probably related to the geographic range of the sole definitive host, the striped skunk (Mephitis mephitis) (Dubey et al. 2010Dubey J.P., Rosenthal B.M. & Felix T.A. 2010. Morphologic and molecular characterization of the sarcocysts of Sarcocystis rileyi (Apicomplexa: Sarcocystidae) from the mallard duck (Anas platyrhynchos). J. Parasitol. 96(4):765-770. <http://dx.doi.org/10.1645/GE-2413.1> <PMid:20496959>
https://doi.org/10.1645/GE-2413.1...
).
(A) Esophageal sarcocystosis; esophagus; sheep. Food analogy: rice grains. Scattered throughout the muscular layer of the esophagus are many 3-7mm long, white macrocysts of Sarcocystis gigantea. (B) Intestinal dipylidiasis; small intestine; dog. Food analogy: rice grains or cucumber seeds. The lumen of the small intestine contains numerous gravid segments of Dipylidium caninum. (C) Moderate, multifocal to coalescing, subacute, gastric ulcers; stomach, dog. Food analogy of the stomach contents: coffee ground. Multiple, linear ulcers are in the mucosa and cover by brown to black fluid (digested blood). (D) Marked, multifocal, chronic, granulomatous and eosinophilic colitis with mucosal and submucosal edema and cyathostomin larvae; colon, horse. Food analogy: salt and pepper colon. The colon contains numerous, 1- 5 mm long, red, coiled nematode larvae encysted in the mucosa and submucosa.
Dipylidium caninum is a common tapeworm found in the small intestine of dogs. Cysticercoids develop in fleas (Ctenocephalides canis) and the dog acquires them by their ingestion (Bowman 2009Bowman D.D. 2009. Helminths, p.149. In: Ibid. (Ed), Giorgi’s Parasitology for Veterinarians. 9th ed. Elsevier, St Louis.). The flat, segmented proglottids seen in the small intestine resemble rice grains or cucumber seeds (Fig.3B).
Coffee grounds. In any species, digested blood in the stomach has a similar appearance of coffee grounds (in Portuguese: “borra de café”) (Fig.3C). In domestic animals, red-brown gastric contents are usually secondary to gastric or abomasal ulcers due to a variety of causes. In dogs, causes of gastric ulcers include: (i) non-steroidal inflammatory drugs administration leading to decreased protective effects of prostaglandin E2 due to inhibition of cyclooxygenase, (ii) uremia leading to arteriolar fibrinoid necrosis and thrombosis with secondary mucosal infarction and coagulative necrosis of the gastric mucosa, (iii) paraneoplastic syndromes secondary to mast cell tumors due to increased histamine levels and gastrinomas due to increased stimulation by gastrin and elevated gastric secretion (Zollinger-Ellison syndrome). In cattle, abomasal ulcers are usually linear and can be result of stress, bovine viral diarrhea virus and malignant catarrhal fever virus or overlying the abomasal wall infiltrated by lymphoma. In calves, copper and selenium deficiency has been associated with abomasal ulcers. In pigs, the predilection site for gastric ulcers is the pars esophagea (Uzal et al. 2016Uzal F.A., Plattner B.L. & Hostetter J.M. 2016. Alimentary System: equine strongylosis, p.216-217. In: Maxie M.G. (Ed.), Jubb, Kennedy and Palmer’s Pathology of Domestic Animals. Vol.2. 6th ed. Elsevier, St Louis. <http://dx.doi.org/10.1016/B978-0-7020-5318-4.00007-3>
https://doi.org/10.1016/B978-0-7020-5318...
). Nevertheless, copious nasal bleeding can be swallowed and digested blood may be present in the gastric contents. Therefore, if no gastric ulcers are noted during necropsy, the nasal cavity should be examined for potential source of bleeding.
Salt and pepper. Numerous, pinpoint, dark red foci with encysted coiled larval nematodes of small strongyles in the cecum and large colon of horses are called “salt and pepper” colon (in Portuguese: “sal e pimenta do reino”) (Fig.3D). Small strongyles (Nematoda, Strongylida) are also called cyathostomes, or cyathostomins are a group of more than 50 species. They have a direct life cycle (Love et al. 1999Love S., Murphy D. & Mellor D. 1999. Pathogenicity of cyathostome infection. Vet. Parasitol. 85(2/3):113-121. <http://dx.doi.org/10.1016/S0304-4017(99)00092-8> <PMid:10485358>
https://doi.org/10.1016/S0304-4017(99)00...
). The infective stage is third-stage larvae where they can encyst for up to 2 years. Encysted larvae are surrounded by macrophages, lymphocytes, plasma cells and eosinophils. The emergence of arrested third-stage larvae in a short period of time causes edema and rupture of the muscularis mucosa and the development of clinical disease. Cyathostomiasis has been associated with colic and chronic diarrhea more commonly in young horses in late winter and early spring, time when the encysted larvae emerge (Uzal et al. 2016Uzal F.A., Plattner B.L. & Hostetter J.M. 2016. Alimentary System: equine strongylosis, p.216-217. In: Maxie M.G. (Ed.), Jubb, Kennedy and Palmer’s Pathology of Domestic Animals. Vol.2. 6th ed. Elsevier, St Louis. <http://dx.doi.org/10.1016/B978-0-7020-5318-4.00007-3>
https://doi.org/10.1016/B978-0-7020-5318...
).
Hepatobiliary system
Milk spots. A common lesion in the liver of pigs is called milk spot (in Portuguese: “mancha de leite”) indicating the similarity between the lesion and a drop of milk on a mahogany color surface. Milk spots are large white patches of variable and irregular contours distributed throughout all the lobes of the liver (Fig.4A). These white areas of fibrosis represent the scars caused by the migration of ascarid roundworm, Ascaris suum, into the liver. This lesion has no clinical significance, but a large number of organs are condemned in meat inspection; due to the aspect of milk spots the livers are rejected as unfit for human consumption (Fausto et al. 2015Fausto M.C., Oliveira I.C., Fausto G.C., Carvalho L.M., Valente F.L., Campos A.K. & Araújo J.V. 2015. Ascaris suum in pigs of the Zona da Mata, Minas Gerais State, Brazil. Braz. J. Vet. Parasitol. 24(3):375-378. <http://dx.doi.org/10.1590/S1984-29612015047>
https://doi.org/10.1590/S1984-2961201504...
).
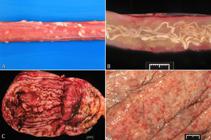
(A) Moderate, multifocal to coalescing, chronic, capsular fibrosis; liver, pig. Food analogy: milk spots. Scattered throughout the Glisson’s capsule and adjacent hepatic parenchyma are multifocal to coalescing white firm areas of fibrosis. (B) Multinodular hepatocellular regeneration and fibrosis; liver, dog. Food analogy: chocolate with crisped rice. Nodules of attempts of hepatocellular regeneration are imprisoned (and therefore not fully functional) by bands of fibrous tissue. This lesion is also called end-stage liver or nodular hepatic cirrhosis. (C) Chronic congestive heart failure; liver, calf. Food analogy: nutmeg liver. Alternate areas of congestion and fibrosis in the liver imparts an aspect of nutmeg liver to the hepatic cut surface. This lesion is caused by right heart failure. (D) Chronic congestive heart failure; liver, calf. Food analogy: nutmeg liver. For comparison, a round fragment of the fixed liver from a dog with chronic congestive heart failure (left) is placed side by side with a piece of nutmeg of similar shape and size. (E) Mucocele; gallbladder; dog. Food analogy: kiwi gallbladder. On cross section, the mucosa of the gallbladder is hyperplastic and the center is filled with inspissated bile and mucus. (F) Multiple renal cysts; kidney, cat; polycystic kidney disease. Food analogy: swiss cheese kidney. The renal cortex and medulla are distorted by variably-sized cysts.
The relationship between A. suum larvae migration and the development of milk spots had been, for long, demonstrated experimentally (Yoshihara et al. 1983Yoshihara S., Nakagawa M., Suda H., Ikeda K. & Hanashiro K. 1983. White spots of the liver in pigs experimentally infected with Ascaris suum. Natl Inst. Anim. Health Q. 23(4):127-137. <PMid:6680775>). In a nutshell, the pathogenesis is as follows: pigs ingest eggs containing third stage larvae (L3) of A. suum which are released in the lumen small intestine. Then, L3 migrate then through the liver parenchyma and capsule, producing necrosis initially with eosinophilic infiltrate (Nakagawa et al. 1983Nakagawa M., Yoshihara S., Suda H. & Ikeda K. 1983. Pathological studies on white spots of the liver in fattening pigs. Natl Inst. Anim. Health Q. 23(4):138-149. <PMid:6680776>), which was, in the past, referred to as interstitial eosinophilic hepatitis. On resolution, the active lesions are gradually replaced by fibrous tissue, which is white and lends itself to be compared with a milk stain. Although the injury is very conspicuous in the Glisson’s capsule, it can also be seen on the cut surface.
Chocolate with crisped rice. A chronic and severe hepatic lesion called end-stage liver or cirrhosis (Brown et al. 2017Brown D.L., Van Vettere A.J. & Cullen J. 2017. End-stqge liver or cirrhosis, p.428-429. In: Zachary J.F. (Ed.), Pathologic Basis of Veterinary Disease. 6th ed. Elsiever, St Louis.) is essentially a scar of the injured liver parenchyma. Typically, a dog with an end-stage liver displays loss of hepatic architecture via loss of large numbers of hepatocytes and associated fibrosis. Regeneration of hepatocytes between bands of fibrous tissue gives rise to multiple nodules of hepatic parenchyma imprisoned by fibrosis (Fig.4B). In some Brazilian veterinary pathology laboratories, this lesion is referred to by the colorful name of a chocolate covered by caramel fudge with crisped rice (in Portuguese: “chocolate caramelizado com flocos crocantes”). The crisp aspect is somewhat similar to the nodules in the liver.
Nutmeg liver. Chronic congestion of the liver is almost invariably associated with congestive right heart failure (Barros 2016aBarros C.S.L. 2016a. Fígado, vias biliares e pâncreas exócrino, p.181-265. In: Santos R.L. & Alessi A.C. (Eds), Patologia Veterinária. 2ª ed. Roca, São Paulo.). In cases of right heart failure, successive characteristic morphological changes accumulate in the liver. Blood is dammed in the center of the lobule as a result of the impediment of venous outflow from the liver usually secondary to congestive heart failure. Central lobular stasis of blood causes anoxia, hepatocellular degeneration, atrophy, and ultimately, loss of hepatocytes from the centrilobular area. Erythrocytes occupy the spaces left by the hepatocytes that went away, forming small lakes of blood. These changes are grossly observed as an accentuation of the lobular pattern, that is, red areas (blood stasis in the centrilobular areas) interspersed with lighter areas of more or less intact periportal hepatocytes. When hepatic congestion becomes chronic, there is proliferation of fibrous connective tissue filling the spaces left by the lost hepatocytes. The liver assumes a reticulated appearance due to the contrast between centrilobular zones of fibrosis, somewhat depressed and firm, alternated with areas of normal color. Over time, centrilobular fibrosis connects the centrilobular veins to each other and to the portal triads accentuating the white cross-linked tracts (Fig.4C). This change is called cardiac fibrosis due to its association with heart failure. This cross-linked pattern, especially when observed on the cut surface of the liver, resembles a nutmeg (Barros 2016aBarros C.S.L. 2016a. Fígado, vias biliares e pâncreas exócrino, p.181-265. In: Santos R.L. & Alessi A.C. (Eds), Patologia Veterinária. 2ª ed. Roca, São Paulo.) (Fig.4D). This liver condition is, throughout the world of pathology known as nutmeg liver (in Portuguese: “fígado de noz-moscada”) and occurs in all species, although it is particularly prominent in ruminants.
Some pathologists also use the term nutmeg to describe livers with centrilobular acute necrosis. However, although there is some similarity between a nutmeg and the surface of a liver with centrilobular acute necrosis, the analogy nutmeg liver should be reserved for the injury resulting from heart failure. In cases of necropsy in which nutmeg liver is observed, other indications of right heart failure such as hydrostatic edema (ascites, hydrothorax, subcutaneous dependent edema) are also seen.
Kiwi gallbladder. Mucocele is a distended gallbladder filled with excessive bile and mucus. On ultrasound examination, the accumulation of mucus in a stellate or finely striated pattern in the absence of gravity dependent bile movement is called “kiwi fruit-like” gallbladder (in Portuguese: “vesícula biliar de quivi”) (Choi et al. 2014Choi J., Kim A., Keh S., Oh J., Kim H. & Yoon J. 2014. Comparison between ultrasonographic and clinical findings in 43 dogs with gallbladder mucoceles. Vet Radiol. Ultrasound 55(2):202-207. <http://dx.doi.org/10.1111/vru.12120> <PMid:24219310>
https://doi.org/10.1111/vru.12120...
). We propose the same terminology in veterinary pathology, since the gallbladder mucocele on cross section is composed of a central area with dark green tenacious mucus and bile surrounded by a hyperplastic mucosa (Fig.4E). Gallbladder mucocele is a common cause of life-threatening biliary disease in several breeds of dogs including the Shetland Sheepdog, American Cocker Spaniel, Chihuahua, Pomeranian, Miniature Schnauzer, and Border Terriers (Allerton et al. 2018Allerton F., Swinbourne F., Barker L., Black V., Kathrani A., Tivers M., Henriques T., Kisielewicz C., Dunning M. & Kent A. 2018. Gall bladder mucoceles in Border terriers. J. Vet. Intern. Med. 32(5):1618-1628. <http://dx.doi.org/10.1111/jvim.15249> <PMid:30079451>
https://doi.org/10.1111/jvim.15249...
). Although the pathogenesis of the gallbladder mucocele in dogs remains unknown, an association between the mucocele and hyperadrenocorticism has been suggested (Mesich et al. 2009Mesich M.L.L., Mayhew P.D., Paek M., Holt D.E. & Brown D.C. 2009. Gall bladder mucoceles and their association with endocrinopathies in dogs: a retrospective case-control study. J. Small Anim. Pract. 50(12):630-635. <http://dx.doi.org/10.1111/j.1748-5827.2009.00811.x> <PMid:19954439>
https://doi.org/10.1111/j.1748-5827.2009...
).
Nervous and urinary system
Swiss cheese. Swiss cheese is riddled by large holes due to fermentation. Swiss cheese appearance (in Portuguese: “queijo suíço”) is characteristic of polycystic kidney disease (PKD) mainly in Persian cats (Fig.4F) (Biller et al. 1990Biller D.S., Chew D.J. & DiBartola S.P. 1990. Polycystic kidney disease in a family of Persian cats. J. Am. Vet. Med. Assoc. 196(8):1288-1290. <PMid:2185204>) and in autolytic change in the brain (Rech et al. 2018Rech R.R., Giaretta P.R., Brown C. & Barros C.S.L. 2018. Gross and histopathological pitfalls found in the examination of 3,338 cattle brains submitted to the BSE surveillance program in Brazil. Pesq. Vet. Bras. 38(11):2099-2116. <http://dx.doi.org/10.1590/1678-5150-pvb-6079>
https://doi.org/10.1590/1678-5150-pvb-60...
). Gas-producing bacteria such as Clostridium spp. form multiple small cavities in many tissues, either as a result of a disease or due to postmortem proliferation.
Cardiovascular system
Bread and butter pericarditis. The non-fastidious feeding behavior of cattle results in the ingestion of foreign bodies such as metallic objects, sometimes sharply pointed, which accumulates in the reticulum. Due to the proximity of the reticulum with the heart, the gastric movements can, sometimes, push these pointed metallic objects cranially in a way that they pass through the diaphragm and penetrate the pericardium frequently puncturing the epicardium and myocardium (Peek & McGuirk 2008Peek S.F. & McGuirk S.M. 2008. Pericarditis, p.56-58. In: Divers T.J. & Peek D.F. (Eds), Rebhun’s Diseases of Dairy Cattle. 2nd ed. W.B. Saunders, Philadelphia). Since the reticular lumen contains microbiota, the foreign body serves as a Trojan horse loaded with bacteria. An acute inflammation then ensues within the pericardial sac. Serous at its onset, the inflammation rapidly evolves to fibrinous or fibrinosuppurative pericarditis involving the external aspect of the visceral membrane (epicardium) and internal aspect of the parietal membrane (visceral pericardium) and accumulates in the pericardial sac. Several layers of fibrin are deposited in a way that when one opens the heart, the two membranes are separated, the fibrinous exudate at both membranes give the vivid impression as if two slices of bread smeared with butter were pulled apart (Fig.5A). The sobriquet bread and butter pericarditis (in Portuguese: “pão com manteiga”) is then applied to describe this lesion colloquially. As time goes by, fibrovascular tissue organizes the exudate closing the space of the pericardial sac. German pathologists used to refer to this scar as Panzer Hertz (armored heart in English and “coração blindado” in Portuguese) in analogy to the armored tanks used in the World War II.
(A) Fibrinous pericarditis, pericardial sac, ox; hardware disease. Food analogy: bread and butter pericarditis. The surface of the epicardium is rough, granular and diffusely covered by fibrin. (B) Multifocal, marked, barbiturate salt crystals deposition; endocardium, right ventricle, dog; Food analogy: salt precipitates. The endocardium has incrusted barbiturate salt crystals that appear as opaque white plaques. (C) Postmortem clots, heart, dog. The right ventricle contains a pale yellow clot (Food analogy: chicken fat) and dark red clots (Food analogy: currant jelly). (D) Diffuse, marked, acute, necrohemorrhagic and emphysematous myositis; skeletal muscle (hindquarter); heifer. Food analogy: aerated chocolate. The skeletal muscle contains multifocal to coalescing areas of hemorrhage and necrosis admixed with many air bubbles cavities (emphysema). Reproduced with permission from Abreu et al. 2017Abreu C.C., Edwards E.E., Edwards J.F., Gibbons P.M., Leal de Araujo J., Rech R.R. & Uzal F.A. 2017. Blackleg in cattle: a case report of fetal infection and a literature review. J. Vet. Diagn. Invest. 29(5):612-621. <http://dx.doi.org/10.1177/1040638717713796> <PMid:28599620>
https://doi.org/10.1177/1040638717713796... .
Mulberry heart disease. Mulberry heart disease (in Portuguese: “doença do coracão de amora”) manifests by sudden death in pigs a few weeks to four months of age when the vitamin E is low. It is characterized by regions of pallor admixed with multifocal areas of hemorrhages throughout the myocardium. Mulberries are red granular elongated fruits from the trees Morus sp. Immature fruits are pale yellow, and turn pink while ripening to dark purple when fully ripe. Pale areas of the heart are of myocardial necrosis and the red areas resemble hemorrhage, similar to a partially ripe mulberry. The areas of hemorrhage are secondary to the fibrinoid necrosis of the arteriolar walls. Additionally, severe hydropericardium with strands of fibrin occurs secondarily to the vascular damage (Boulton 1996Boulton J.G. 1996. The cardiovascular system, p.139-164. In: Sims L.D. & Glastonbury J.R.W. (Eds), Pathology of the Pig: a diagnostic guide. Pig Research and Development Corporation and Agriculture, Victoria.).
Barbiturate “salt” precipitates. An interesting artifact often occurs in animals undergoing euthanasia through barbiturates overdose. When the necropsy is carried out on these animals, the tissues previously exposed to high concentrations of these euthanasia agents, such as the endocardium of the heart and pleura following venous injection have encrusted barbiturate salt crystals (King et al. 2013King J.M., Roth-Johnson L., Dodd D.C. & Newson M.E. 2013. The Necropsy Book. Charles Louis Foundation Publisher, Gurnee, Illinois, 194p., Maxie & Miller 2016Maxie M.G. & Miller M.A. 2016. Introduction to the diagnostic process, p.1-15. In: Maxie M.G. (Ed.), Jubb, Kennedy and Palmer’s Pathology of Domestic Animals. Vol.1. 6th ed. Elsevier, St Louis. <http://dx.doi.org/10.1016/B978-0-7020-5317-7.00001-1>
https://doi.org/10.1016/B978-0-7020-5317...
). These structures are opaque white plaques (Fig.5B) that are referred to as “salt precipitates” (in Portuguese: “depósitos de sal”). Magnesium sulfate, another substance, once used as intravenous agent for euthanasia in animals has a similar effect.
Currant jelly and chicken fat clots. When an animal dies, shiny and smooth postmortem clots form within the right ventricle and atrium as well as in the large vessels. Postmortem clots conform to the shape of the heart chamber (Fig.5C) or vessel in which they form. Red clots are usually at the bottom of the clot and contain sedimented erythrocytes. They are called “currant jelly” (in Portuguese: “geleia de groselha”). The yellow portion represents clotted plasma devoided of red blood cells and is referred to as “chicken fat” (in Portuguese: “gordura de galinha”). Horses more often have pale clots because of a rapid erythrocyte sedimentation rate termed rouleaux formation (Myers et al. 2012Myers R.K., McGavin M.D. & Zachary J.F. 2012. Morphologic appearance of postmortem changes, p.21-23. In: Zachary J.F. & McGavin M.D. (Eds), Pathologic Basis of Veterinary Disease. 5th ed. Elsevier, St Louis.). At necropsy, it is important to differentiate postmortem clots from antemortem thrombi. Thrombi have a dull surface and are adherent to the vessel wall.
Musculoskeletal system
Aerated chocolate. Necrohemorrhagic and emphysematous myositis and myocarditis are characteristic lesions caused by Clostridium chauvoei infection. Due to hemorrhage and necrosis, the affected muscles of the limbs turn dark-red; therefore the disease is called blackleg (Abreu et al. 2017Abreu C.C., Edwards E.E., Edwards J.F., Gibbons P.M., Leal de Araujo J., Rech R.R. & Uzal F.A. 2017. Blackleg in cattle: a case report of fetal infection and a literature review. J. Vet. Diagn. Invest. 29(5):612-621. <http://dx.doi.org/10.1177/1040638717713796> <PMid:28599620>
https://doi.org/10.1177/1040638717713796...
). The emphysematous (gas bubbles) black (necrotic) areas of musclehave the appearance of aerated chocolate (in Portuguese: “chocolate aerado”) on cut surface (Fig.5D).
After death, the process of decomposition of a body, involves autolysis and bacterial putrefaction. As a result of bacterial putrefaction, body cavities and organs become distended by gas bubbles (Brooks 2016Brooks J.W. 2016. Postmortem changes in animal carcasses and estimation of the postmortem interval. Vet. Pathol. 53(5):929-940. <http://dx.doi.org/10.1177/0300985816629720> <PMid:26945004>
https://doi.org/10.1177/0300985816629720...
) which along with the discoloration of the autolyzed tissues, as the liver, also impart the appearance of aerated chocolate (Rech et al. 2013Rech R.R., Silva M.C., Langohr I.M., Marques M.G., Pescador C.A., Silva G.S., Dutra M.C., Brum J.S., Kramer B., Bordin L.C. & Silva V.S. 2013. Nem tudo que parece ser, é lesão: aspectos anatômicos, não lesões, artefatos, lesões sem significado clínico e alterações post mortem encontrados na necropsia de suínos domésticos e selvagens (Sus scrofa). Pesq. Vet Bras. 33(10):1237-1255. <http://dx.doi.org/10.1590/S0100-736X2013001000010>
https://doi.org/10.1590/S0100-736X201300...
).
Hematopoietic
Cola colored urine. Animals with intravascular hemolytic disease or degenerative myopathy present dark urine (Fig.6A), which pathologists refer to as “cola - colored urine” (in Portuguese: “urina cor de refrigerante de cola”) due respectively to the presence of the dark pigment of hemoglobin or myoglobin in the urine. The analogy is with the substance extracted from the nut-cola (the fruit of plants of the genus Cola) that was used to produce “cola” soft drinks and conferred the coffee color characteristic of these beverages. In intravascular hemolysis, the destruction of erythrocytes releases large amounts of hemoglobin in the circulation causing marked hemoglobinemia. The hemoglobin passes through the kidney and is collected within the urinary bladder admixed with urine, imparting the dark-red color.
(A) Hemoglobinuria; urinary bladder, sheep; copper toxicosis. Food analogy: cola colored urine. The urinary bladder contains dark red urine. The adipose tissue is moderately yellow (icterus). Inset: hemoglobinuric nephrosis; kidney, sheep. The renal cortex and medulla are diffusely dark red due to acute tubular necrosis with intratubular hemoglobinuric casts. (B) Marked, multifocal, lymphoid follicular hyperplasia, spleen, dog. Food analogy: sago spleen. Traditionally sago spleen is the term for splenic follicular amyloidosis. However, some pathologists by analogy refer to marked lymphoid hyperplasia as sago spleen. Image courtesy of Dr. Laura Bryan.
Animals with intravascular hemolytic disease present jaundice, dark kidneys (due to hemoglobin impregnation) and hemoglobin imbibition of the serosa of the viscera. The spleen is enlarged with a tense capsule. Examples of this disorder occur in all domestic species. In cattle, it occurs in babesiosis (Barros et al. 2006Barros C.S.L., Driemeier D., Dutra I.S. & Lemos R.A.A. 2006. Babesiose cerebral, p.87-96. In: Ibid (Eds), Doenças do Sistema Nervoso de Bovinos no Brasil, Valée, Montes Claros.), bacillary hemoglobinuria (Barros 2016aBarros C.S.L. 2016a. Fígado, vias biliares e pâncreas exócrino, p.181-265. In: Santos R.L. & Alessi A.C. (Eds), Patologia Veterinária. 2ª ed. Roca, São Paulo.) and leptospirosis in calves up to approximately 30 days old. In sheep, it is observed mainly in chronic copper intoxication (Pilati et al. 1990Pilati C., Barros C.S.L., Giudice J.C. & Bondan E. 1990. Intoxicação crônica por cobre em ovinos. Hora Vet. 54:1-33.). In dogs, it is associated with babesiosis, immune-mediated hemolytic diseases and as a complication of hemolysis resulting from bee sting (Fighera et al. 2007Fighera R.A., Souza T.M. & Barros C.S.L. 2007. Acidente provocado por picada de abelhas como causa de morte de cães. Ciência Rural 37(2):590-593. <http://dx.doi.org/10.1590/S0103-84782007000200050>
https://doi.org/10.1590/S0103-8478200700...
). Horses affected with piroplasmosis, neonatal isoerythrolysis and red maple intoxication (Alward et al. 2006Alward A., Corriher C.A., Barton M.H., Sellon D.C., Blikslager A.T. & Jones S.L. 2006. Red maple (Acer rubrum) leaf toxicosis in horses: a retrospective study of 32 cases. J. Vet. Intern. Med. 20(5):197-201. <http://dx.doi.org/10.1111/j.1939-1676.2006.tb00722.x> <PMid:17063716>
https://doi.org/10.1111/j.1939-1676.2006...
) may present dark red urine. Cola-colored urine is also seen animals with degenerative myopathy. In these diseases, the lysis of the skeletal muscle fiber allows the leakage of myoglobin into the blood with consequent myoglobinemia. The size of the myoglobin molecule will enable it to pass through the renal glomeruli and will be eliminated in the urine, giving it the dark aspect. Cola-colored urine related to degenerative myopathy occurs in diseases such as exercise-associated myopathy of horses, capture myopathy in wild ruminants, poisoning by ionophore antibiotics in various species, poisoning by Senna occidentalis and S. obtusifolia in cattle and horses and nutritional myopathy (vitamin E deficiency in selenium) in several species, but mainly young ruminants (Barros 2016bBarros C.S.L. 2016b. Sistema muscular, p.663-702. In: Santos R.L. & Alessi A.C. (Eds), Patologia Veterinária. Roca, São Paulo.).
Sago spleen. Amyloidosis is the extracellular deposition of amyloid and it is associated with several diseases. Amyloid means starch-like, but is a misnomer for the substance that has a high content of protein. Grossly the amyloid appears as a white, somewhat firm substance (Jones & Hunt 1983Jones T.C. & Hunt R.D. 1983. Veterinary Pathology. Lea and Febiger, Philadelphia, p.43-47.). One predisposing factor for amyloid production is chronic antigenic stimulation. Accordingly, amyloidosis occurs in the spleen of about 60% of the horses used for the production of antitetanic, antivenom and anti-diphtheric serum (Coelho 2002Coelho H.E. 2002. Patologia Veterinária. Manole, São Paulo, p.189.). In the spleen, the deposits either form between the lymphoid follicles or along the sinuses. As the deposits enlarge the lymphoid follicles, they become grossly visible as small nodules of white firm material (Jones & Hunt 1983Jones T.C. & Hunt R.D. 1983. Veterinary Pathology. Lea and Febiger, Philadelphia, p.43-47.). As the white pulp of the spleen is only 4-5% of the volume of the organ (Rooney & Robertson 1996Rooney J.R. & Robertson J.K. 1996. Equine Pathology. Iowa State University Press, Ames, p.350-351.) these white nodules contrast with the more abundant red pulp imparting a suggestion of a spleen sprinkled with sago pellets and hence the appellation, sago spleen (Jones & Hunt 1983Jones T.C. & Hunt R.D. 1983. Veterinary Pathology. Lea and Febiger, Philadelphia, p.43-47.).
Sago (in Portuguese: “sagu”) is a starch extracted from several palm trees (e.g. Metroxylon sagu and Cycas rumphii) and used as staple food in different places in the world. Currently in Western countries, the term is used to refer to small round pellets derived from the cassava root (false sago) from which a dessert is made admixing wine, water, sugar, clover, and cinnamon. When the sago is cooked, the tiny pellets become swollen and translucent mimicking the appearance of amyloid. Sago spleen is an uncommon finding in the necropsy floor. However, some pathologists by analogy refer to marked lymphoid hyperplasia of the white pulp (Fig.6B) as sago spleen.
Blackberry jam. Anthrax, caused by the gram-positive bacterium Bacillus anthracis, is highly pathogenic to herbivorous animals and human beings. The disease has an acute to a peracute course. In affected cattle, the most prominent necropsy finding is a marked enlarged spleen (Valli et al. 2016Valli V.E.O.M., Kiupel M. & Bienzle D. 2016. Anthrax, p.171-174. In: Maxie M.G. (Ed.), Jubb, Kennedy and Palmer’s Pathology of Domestic Animals. Vol.2. 6th ed. Elsevier, St Louis.). The consistency of the spleen is soft, the capsule may rupture, and a pasty floccular splenic pulp extrudes. This pulp is then colloquially referred to as blackberry jam (in Portuguese: “geleia de amora”). Since necropsy of a dead animal from anthrax is not recommended and since anthrax is currently a rare disease in most countries nowadays, this descriptive food analogy is of relatively small value.
Microscopic Findings
Oat cells
In veterinary pathology, necrotic leukocytes with streaming nuclei that occur in the lung of cattle with shipping fever and pigs with contagious pleuropneumonia are called oat cells (in Portuguese: “células em grão de aveia”) (Caswell & Williams 2016Caswell J.L. & Williams K.J. 2016. Respiratory system, p.465-538. In: Maxie M.G. (Ed.), Jubb, Kennedy and Palmer’s Pathology of Domestic Animals. Vol.2. 6th ed. Elsevier, St Louis. <http://dx.doi.org/10.1016/B978-0-7020-5318-4.00011-5>.
https://doi.org/10.1016/B978-0-7020-5318...
). The leukotoxins of Mannheimia haemolytica (Maheswaran et al. 1992Maheswaran S.K., Weiss D.J., Kannan M.S., Townsend E.L., Reddy K.R., Whiteley L.O. & Srikumaran S. 1992. Effects of Pasteurella haemolytica A1 leukotoxin on bovine neutrophils: degranulation and generation of oxygen-derived free radicals. Vet. Immunol. Immunopathol. 33(1/2):51-68. <http://dx.doi.org/10.1016/0165-2427(92)90034-N> <PMid:1321532>
https://doi.org/10.1016/0165-2427(92)900...
) and Actinobacillus pleuropneumoniae (Choi et al. 2001Choi C., Kwon D., Min K. & Chae C. 2001. Detection and localization of apxI, -II, and -III genes of Actinobacillus pleuropneumoniae in natural porcine pleuropneumonia by in situ hybridization. Vet. Pathol. 38(4):390-395. <http://dx.doi.org/10.1354/vp.38-4-390> <PMid:11467472>
https://doi.org/10.1354/vp.38-4-390...
) are believed to cause necrosis of neutrophils and perhaps macrophages with resultant fusiform shape of the nuclei and the streaming of the chromatin, resembling oat grains (Fig.7A).
(A) Necrosuppurative bronchopneumonia, lung, pig; pulmonary actinobacillosis. Food analogy: oat cells. The alveolar lumina are obliterated by numerous elongated degenerated and necrotic leukocytes with nuclear streaming. HE, obj.40x. (B) Mast cell tumor, haired skin, dog. Food analogy: fried egg appearance. Well-differentiated mast cells containing abundant granular cytoplasm and round nucleus are intermixed with eosinophils. HE, obj.40x. (C) Oligodendroglioma, brain, dog. Food analogy: fried egg appearance. Neoplastic cells contain round, uniform, central nuclei surrounded by a halo of clear cytoplasm. The clear halo is an artifact of tissue fixation. HE, obj.20x. (D) Histiocytic interstitial pneumonia, lung, horse. Food analogy: honeycomb material. The alveolar spaces and septa are expanded by foamy, eosinophilic material, macrophages and numerous round, intra and extracellular, inconspicuous fungal bodies. HE, obj.60x. (E) Protozoal cyst, myocardium, dog. Food analogy: onion skin cyst. Encysted stages of Hepatozoon americanum contain a host cell (macrophage) in the center, surrounded by multilamellar, basophilic, amorphous material. HE, obj.20x. (F) Granulomatous bronchointerstitial pneumonia with lymphoid depletion and histiocytic intracytoplasmic botryoid inclusion bodies; lung; pig. Porcine circovirus-associated disease. Food analogy: botryoid inclusion bodies. HE, obj.40x.
Fried egg
Normal and neoplastic mast cells have a centrally located, round nucleus surrounded by abundant cytoplasm that gives a “fried egg” (in Portuguese: “ovo frito”) appearance to the cell (Fig.7B) (Kiupel 2017Kiupel M. 2017. Mast cell tumors, p.179-180. In: Meuten D.J. (Ed.), Tumors of Domestic Animals. 5th ed. Wiley Blackwell, Ames.). A similar pattern is observed in oligodendrogliomas. These are tumors derived from oligodendrocytes within the central nervous system and have a microscopic arrangement of cells with round, uniform, central nuclei surrounded by a halo of clear cytoplasm that is also comparable to the egg white of a fried egg (Fig.7C) (Reifenberger et al. 2000Reifenberger G., Kros J.M., Burger P.C., Louis D.N. & Collins V.P. 2000. Oligodendroglioma, p.56-61. In: Kleihues P. & Cavenee W.K. (Eds), Pathology and Genetics of Tumours of the Nervous System. IARC Press, Lyon.). The clear cytoplasm is actually an artifact of tissue processing from delayed fixation (Reifenberger et al. 2000Reifenberger G., Kros J.M., Burger P.C., Louis D.N. & Collins V.P. 2000. Oligodendroglioma, p.56-61. In: Kleihues P. & Cavenee W.K. (Eds), Pathology and Genetics of Tumours of the Nervous System. IARC Press, Lyon.) and is sometimes referred as honeycomb pattern (Vandevelde et al. 2012Vandevelde M., Higgins R. & Oevermann A. 2012. Veterinary Neuropathology: essentials of theory and practice. Wiley Blackwell, Ames, p.137-139.).
Honeycomb
Pneumocystis spp. are organisms classified as fungi that can cause disease in immunosuppressed animals and people (Skalski et al. 2015Skalski J.H., Kottom T.J. & Limper A.H. 2015. Pathobiology of pneumocystis pneumonia: life cycle, cell wall and cell signal transduction. FEMS Yeast Res. 15(6):1-12. <http://dx.doi.org/10.1093/femsyr/fov046> <PMid:26071598>
https://doi.org/10.1093/femsyr/fov046...
). Pneumonia is the most common clinical presentation. It is characterized histologically by foamy or “honeycomb” (in Portuguese: “favo de mel”) material filling alveoli (Fig.7D) (Caswell & Williams 2016Caswell J.L. & Williams K.J. 2016. Respiratory system, p.465-538. In: Maxie M.G. (Ed.), Jubb, Kennedy and Palmer’s Pathology of Domestic Animals. Vol.2. 6th ed. Elsevier, St Louis. <http://dx.doi.org/10.1016/B978-0-7020-5318-4.00011-5>.
https://doi.org/10.1016/B978-0-7020-5318...
). This aspect is due to numerous intracellular and extracellular, round to crescent-shaped, clear fungal bodies that are about 8 µm in diameter (Caswell & Williams 2016Caswell J.L. & Williams K.J. 2016. Respiratory system, p.465-538. In: Maxie M.G. (Ed.), Jubb, Kennedy and Palmer’s Pathology of Domestic Animals. Vol.2. 6th ed. Elsevier, St Louis. <http://dx.doi.org/10.1016/B978-0-7020-5318-4.00011-5>.
https://doi.org/10.1016/B978-0-7020-5318...
). The organisms are inconspicuous on hematoxylin and eosin stain, but can be highlighted by methenamine silver stain (Caswell & Williams 2016Caswell J.L. & Williams K.J. 2016. Respiratory system, p.465-538. In: Maxie M.G. (Ed.), Jubb, Kennedy and Palmer’s Pathology of Domestic Animals. Vol.2. 6th ed. Elsevier, St Louis. <http://dx.doi.org/10.1016/B978-0-7020-5318-4.00011-5>.
https://doi.org/10.1016/B978-0-7020-5318...
).
Onion skin
Onion skin cysts are characteristic of American canine hepatozoonosis, caused by the protozoan Hepatozoon americanum (Cummings et al. 2005Cummings C.A., Panciera R.J., Kocan K.M., Mathew J.S. & Ewing S.A. 2005. Characterization of stages of Hepatozoon americanum and of parasitized canine host cells. Vet. Pathol. 42(6):788-796. <http://dx.doi.org/10.1354/vp.42-6-788> <PMid:16301575>
https://doi.org/10.1354/vp.42-6-788...
). The onion skin (in Portuguese: “pétalas de cebola”) cyst (Fig.7E) is composed of multiple layers of acid mucopolysaccharide deposits that surround an enlarged macrophage that has been infected by a trophozoite (Cummings et al. 2005Cummings C.A., Panciera R.J., Kocan K.M., Mathew J.S. & Ewing S.A. 2005. Characterization of stages of Hepatozoon americanum and of parasitized canine host cells. Vet. Pathol. 42(6):788-796. <http://dx.doi.org/10.1354/vp.42-6-788> <PMid:16301575>
https://doi.org/10.1354/vp.42-6-788...
). The cysts occur in several tissues, but are more common in striated muscle (Panciera et al. 1998Panciera R.J., Ewing S.A., Mathew J.S., Cummings C.A., Kocan A.A., Breshears M.A. & Fox J.C. 1998. Observations on tissue stages of Hepatozoon americanum in 19 naturally infected dogs. Vet. Parasitol. 78(4):265-276. <http://dx.doi.org/10.1016/S0304-4017(98)00142-3> <PMid:9786627>
https://doi.org/10.1016/S0304-4017(98)00...
).
Grape-like (botryoid) inclusions
The term “botryoid” is used to describe the viral inclusion bodies observed in infection by circoviruses, including porcine circovirus-2 and beak and feather disease virus in psittacine birds (Parrish 2010Parrish C.R. 2010. Circoviridae, p.237-242 In: Maclachlan N. & Dubovi E.J. (Eds), Fenner’s Veterinary Virology. Academic Press, London.). From the Greek, botryoid meaning similar to a cluster of grapes (in Portuguese: “cacho de uva”) (Stedman 2006Stedman T.L. 2006. Stedman’s Medical Dictionary. 28th ed. Lippincott Williams and Wilkins, Philadelphia, 1514p.) that resemble the deeply basophilic, round clusters of viral inclusion bodies that are observed in the cytoplasm of infected cells (Fig.7F) (Parrish 2010Parrish C.R. 2010. Circoviridae, p.237-242 In: Maclachlan N. & Dubovi E.J. (Eds), Fenner’s Veterinary Virology. Academic Press, London.).
Conclusions
We emphasize here we do not recommend the use of macroscopic food analogies as descriptive terminology in the pathology reports. Their use should be applied in the academic setting to solidify technical learning in the long-term memory of the veterinary students since we believe adults must construct knowledge from knowledge they already possess, a principle long advocated by a distinguished Brazilian educator (Freire 2013Freire P. 2013. Pedagogia do Oprimido. Editora Paz e Terra, São Paulo. 256p.).
On the other hand, veterinary pathologists will not hesitate to use terms like oat cells, onion skin cysts, and botryoid inclusion bodies when describing microscopic aspects of lesions or etiologic agents. Many of the analogies mentioned in this article were spontaneously born in the North hemisphere, yet most of them are well-known by Brazilian veterinary professionals. At any event, in this article, we consistently presented, within parenthesis, a translation into Portuguese for each food analogy.
Acknowledgments
We thank Dr. Paula Giaretta and Dr. Richard Ploeg for their valuable contributions.
References
- Abreu C.C., Edwards E.E., Edwards J.F., Gibbons P.M., Leal de Araujo J., Rech R.R. & Uzal F.A. 2017. Blackleg in cattle: a case report of fetal infection and a literature review. J. Vet. Diagn. Invest. 29(5):612-621. <http://dx.doi.org/10.1177/1040638717713796> <PMid:28599620>
» https://doi.org/10.1177/1040638717713796 - Ackermann M.R. 2017. Inflammation and healing, p.73-131. In: Zachary J.F. (Ed.), Pathologic Basis of Veterinary Disease. 6th ed. Elsevier, St Louis. <http://dx.doi.org/10.1016/B978-0-323-35775-3.00003-5>.
» https://doi.org/10.1016/B978-0-323-35775-3.00003-5 - Allerton F., Swinbourne F., Barker L., Black V., Kathrani A., Tivers M., Henriques T., Kisielewicz C., Dunning M. & Kent A. 2018. Gall bladder mucoceles in Border terriers. J. Vet. Intern. Med. 32(5):1618-1628. <http://dx.doi.org/10.1111/jvim.15249> <PMid:30079451>
» https://doi.org/10.1111/jvim.15249 - Alward A., Corriher C.A., Barton M.H., Sellon D.C., Blikslager A.T. & Jones S.L. 2006. Red maple (Acer rubrum) leaf toxicosis in horses: a retrospective study of 32 cases. J. Vet. Intern. Med. 20(5):197-201. <http://dx.doi.org/10.1111/j.1939-1676.2006.tb00722.x> <PMid:17063716>
» https://doi.org/10.1111/j.1939-1676.2006.tb00722.x - Barros C.S.L. 2016a. Fígado, vias biliares e pâncreas exócrino, p.181-265. In: Santos R.L. & Alessi A.C. (Eds), Patologia Veterinária. 2ª ed. Roca, São Paulo.
- Barros C.S.L. 2016b. Sistema muscular, p.663-702. In: Santos R.L. & Alessi A.C. (Eds), Patologia Veterinária. Roca, São Paulo.
- Barros C.S.L., Driemeier D., Dutra I.S. & Lemos R.A.A. 2006. Babesiose cerebral, p.87-96. In: Ibid (Eds), Doenças do Sistema Nervoso de Bovinos no Brasil, Valée, Montes Claros.
- Biller D.S., Chew D.J. & DiBartola S.P. 1990. Polycystic kidney disease in a family of Persian cats. J. Am. Vet. Med. Assoc. 196(8):1288-1290. <PMid:2185204>
- Boulton J.G. 1996. The cardiovascular system, p.139-164. In: Sims L.D. & Glastonbury J.R.W. (Eds), Pathology of the Pig: a diagnostic guide. Pig Research and Development Corporation and Agriculture, Victoria.
- Bowman D.D. 2009. Helminths, p.149. In: Ibid. (Ed), Giorgi’s Parasitology for Veterinarians. 9th ed. Elsevier, St Louis.
- Brooks J.W. 2016. Postmortem changes in animal carcasses and estimation of the postmortem interval. Vet. Pathol. 53(5):929-940. <http://dx.doi.org/10.1177/0300985816629720> <PMid:26945004>
» https://doi.org/10.1177/0300985816629720 - Brown D.L., Van Vettere A.J. & Cullen J. 2017. End-stqge liver or cirrhosis, p.428-429. In: Zachary J.F. (Ed.), Pathologic Basis of Veterinary Disease. 6th ed. Elsiever, St Louis.
- Caswell J.L. & Williams K.J. 2016. Respiratory system, p.465-538. In: Maxie M.G. (Ed.), Jubb, Kennedy and Palmer’s Pathology of Domestic Animals. Vol.2. 6th ed. Elsevier, St Louis. <http://dx.doi.org/10.1016/B978-0-7020-5318-4.00011-5>.
» https://doi.org/10.1016/B978-0-7020-5318-4.00011-5 - Choi C., Kwon D., Min K. & Chae C. 2001. Detection and localization of apxI, -II, and -III genes of Actinobacillus pleuropneumoniae in natural porcine pleuropneumonia by in situ hybridization. Vet. Pathol. 38(4):390-395. <http://dx.doi.org/10.1354/vp.38-4-390> <PMid:11467472>
» https://doi.org/10.1354/vp.38-4-390 - Choi J., Kim A., Keh S., Oh J., Kim H. & Yoon J. 2014. Comparison between ultrasonographic and clinical findings in 43 dogs with gallbladder mucoceles. Vet Radiol. Ultrasound 55(2):202-207. <http://dx.doi.org/10.1111/vru.12120> <PMid:24219310>
» https://doi.org/10.1111/vru.12120 - Coelho H.E. 2002. Patologia Veterinária. Manole, São Paulo, p.189.
- Cummings C.A., Panciera R.J., Kocan K.M., Mathew J.S. & Ewing S.A. 2005. Characterization of stages of Hepatozoon americanum and of parasitized canine host cells. Vet. Pathol. 42(6):788-796. <http://dx.doi.org/10.1354/vp.42-6-788> <PMid:16301575>
» https://doi.org/10.1354/vp.42-6-788 - Dubey J.P., Rosenthal B.M. & Felix T.A. 2010. Morphologic and molecular characterization of the sarcocysts of Sarcocystis rileyi (Apicomplexa: Sarcocystidae) from the mallard duck (Anas platyrhynchos). J. Parasitol. 96(4):765-770. <http://dx.doi.org/10.1645/GE-2413.1> <PMid:20496959>
» https://doi.org/10.1645/GE-2413.1 - Dubey J.P., Calero-Bernal R., Rosenthal B.M., Speer C.A. & Fayer R. 2015. Sarcocystosis of Animals and Humans. CRC Press, Boca Raton, p.1-481. <http://dx.doi.org/10.1201/b19184>
» https://doi.org/10.1201/b19184 - Edwards D.F. 2006. Actinomycosis and nocardiosis, p.451-461. In: Greene C.E. (Ed.), Infectious Diseases of the Dog and Cat. 3rd ed. Elsevier, St Louis.
- Fausto M.C., Oliveira I.C., Fausto G.C., Carvalho L.M., Valente F.L., Campos A.K. & Araújo J.V. 2015. Ascaris suum in pigs of the Zona da Mata, Minas Gerais State, Brazil. Braz. J. Vet. Parasitol. 24(3):375-378. <http://dx.doi.org/10.1590/S1984-29612015047>
» https://doi.org/10.1590/S1984-29612015047 - Fighera R.A., Souza T.M. & Barros C.S.L. 2007. Acidente provocado por picada de abelhas como causa de morte de cães. Ciência Rural 37(2):590-593. <http://dx.doi.org/10.1590/S0103-84782007000200050>
» https://doi.org/10.1590/S0103-84782007000200050 - Fink D., Drumond L., Basílio M.I., Sartori C.M., Andrade T.R., Santos N.Z. & Cremer M.J. 2018. Gota úrica visceral em bobo-pequeno (Puffinus puffinus) no sul do Brasil. Arq. Bras. Med. Vet. Zootec. 70(2):486-490. <http://dx.doi.org/10.1590/1678-4162-9916>
» https://doi.org/10.1590/1678-4162-9916 - Freire P. 2013. Pedagogia do Oprimido. Editora Paz e Terra, São Paulo. 256p.
- Herbert J.D., Coulson J.O. & Coulson T.D. 2011. Quantification of tissue uric acid levels in a Harris’s hawk with visceral gout. BioOne 55(3):513-515. <PMid:22017058>
- Jones T.C. & Hunt R.D. 1983. Veterinary Pathology. Lea and Febiger, Philadelphia, p.43-47.
- King J.M., Roth-Johnson L., Dodd D.C. & Newson M.E. 2013. The Necropsy Book. Charles Louis Foundation Publisher, Gurnee, Illinois, 194p.
- Kipar A. & Meli M.L. 2014. Feline infectious peritonitis: still an enigma? Vet. Pathol. 51(2):505-526. <http://dx.doi.org/10.1177/0300985814522077> <PMid:24569616>
» https://doi.org/10.1177/0300985814522077 - Kiupel M. 2017. Mast cell tumors, p.179-180. In: Meuten D.J. (Ed.), Tumors of Domestic Animals. 5th ed. Wiley Blackwell, Ames.
- Kobayashi A. & Iwasaki H. 2019. Doughnut lesions. N. Engl. J. Med. 380(10):10. <http://dx.doi.org/10.1056/NEJMicm1810556> <PMid:30855746>
» https://doi.org/10.1056/NEJMicm1810556 - Lierz M. 2003. Avian renal disease: pathogenesis, diagnosis, and therapy. Vet. Clin. N. Am., Exotic Anim. Pract. 6(1):29-55. <PMid:12616833>
- Love S., Murphy D. & Mellor D. 1999. Pathogenicity of cyathostome infection. Vet. Parasitol. 85(2/3):113-121. <http://dx.doi.org/10.1016/S0304-4017(99)00092-8> <PMid:10485358>
» https://doi.org/10.1016/S0304-4017(99)00092-8 - López A. & Martinson S.A. 2017. Respiratory system, mediastinum, and pleurae, p.471-558. In: Zachary J.F. (Ed.), Pathologic Basis of Veterinary Disease. 6th ed. Elsevier, St Louis. <http://dx.doi.org/10.1016/B978-0-323-35775-3.00009-6>
» https://doi.org/10.1016/B978-0-323-35775-3.00009-6 - Maheswaran S.K., Weiss D.J., Kannan M.S., Townsend E.L., Reddy K.R., Whiteley L.O. & Srikumaran S. 1992. Effects of Pasteurella haemolytica A1 leukotoxin on bovine neutrophils: degranulation and generation of oxygen-derived free radicals. Vet. Immunol. Immunopathol. 33(1/2):51-68. <http://dx.doi.org/10.1016/0165-2427(92)90034-N> <PMid:1321532>
» https://doi.org/10.1016/0165-2427(92)90034-N - Mark N.M., Lessing J.N., Buckley S.A. & Tierney Junior J.R.L.M. 2015. Diagnostic utility of food terminology: culinary clues for the astute diagnostician. Am. J. Med. 128(9):933-934. <http://dx.doi.org/10.1016/j.amjmed.2015.03.018> <PMid:25863151>
» https://doi.org/10.1016/j.amjmed.2015.03.018 - Maxie M.G. & Miller M.A. 2016. Introduction to the diagnostic process, p.1-15. In: Maxie M.G. (Ed.), Jubb, Kennedy and Palmer’s Pathology of Domestic Animals. Vol.1. 6th ed. Elsevier, St Louis. <http://dx.doi.org/10.1016/B978-0-7020-5317-7.00001-1>
» https://doi.org/10.1016/B978-0-7020-5317-7.00001-1 - Mesich M.L.L., Mayhew P.D., Paek M., Holt D.E. & Brown D.C. 2009. Gall bladder mucoceles and their association with endocrinopathies in dogs: a retrospective case-control study. J. Small Anim. Pract. 50(12):630-635. <http://dx.doi.org/10.1111/j.1748-5827.2009.00811.x> <PMid:19954439>
» https://doi.org/10.1111/j.1748-5827.2009.00811.x - Myers R.K., McGavin M.D. & Zachary J.F. 2012. Morphologic appearance of postmortem changes, p.21-23. In: Zachary J.F. & McGavin M.D. (Eds), Pathologic Basis of Veterinary Disease. 5th ed. Elsevier, St Louis.
- Nakagawa M., Yoshihara S., Suda H. & Ikeda K. 1983. Pathological studies on white spots of the liver in fattening pigs. Natl Inst. Anim. Health Q. 23(4):138-149. <PMid:6680776>
- Panciera R.J., Ewing S.A., Mathew J.S., Cummings C.A., Kocan A.A., Breshears M.A. & Fox J.C. 1998. Observations on tissue stages of Hepatozoon americanum in 19 naturally infected dogs. Vet. Parasitol. 78(4):265-276. <http://dx.doi.org/10.1016/S0304-4017(98)00142-3> <PMid:9786627>
» https://doi.org/10.1016/S0304-4017(98)00142-3 - Parrish C.R. 2010. Circoviridae, p.237-242 In: Maclachlan N. & Dubovi E.J. (Eds), Fenner’s Veterinary Virology. Academic Press, London.
- Peek S.F. & McGuirk S.M. 2008. Pericarditis, p.56-58. In: Divers T.J. & Peek D.F. (Eds), Rebhun’s Diseases of Dairy Cattle. 2nd ed. W.B. Saunders, Philadelphia
- Pilati C., Barros C.S.L., Giudice J.C. & Bondan E. 1990. Intoxicação crônica por cobre em ovinos. Hora Vet. 54:1-33.
- Pollock C. 2006. Diagnosis and treatment of avian renal disease. Vet. Clin. N. Am., Exotic Anim. Pract. 9(1):107-128. <PMid:16407082>
- Rahimi M., Minoosh Z. & Haghighi S. 2015. Visceral urate deposition in a little bittern (Ixobrychus minutus). Vet. Res. Forum, Int. Q. J. 6(2):177-180.
- Rech R.R., Silva M.C., Langohr I.M., Marques M.G., Pescador C.A., Silva G.S., Dutra M.C., Brum J.S., Kramer B., Bordin L.C. & Silva V.S. 2013. Nem tudo que parece ser, é lesão: aspectos anatômicos, não lesões, artefatos, lesões sem significado clínico e alterações post mortem encontrados na necropsia de suínos domésticos e selvagens (Sus scrofa). Pesq. Vet Bras. 33(10):1237-1255. <http://dx.doi.org/10.1590/S0100-736X2013001000010>
» https://doi.org/10.1590/S0100-736X2013001000010 - Rech R.R., Giaretta P.R., Brown C. & Barros C.S.L. 2018. Gross and histopathological pitfalls found in the examination of 3,338 cattle brains submitted to the BSE surveillance program in Brazil. Pesq. Vet. Bras. 38(11):2099-2116. <http://dx.doi.org/10.1590/1678-5150-pvb-6079>
» https://doi.org/10.1590/1678-5150-pvb-6079 - Reifenberger G., Kros J.M., Burger P.C., Louis D.N. & Collins V.P. 2000. Oligodendroglioma, p.56-61. In: Kleihues P. & Cavenee W.K. (Eds), Pathology and Genetics of Tumours of the Nervous System. IARC Press, Lyon.
- Rooney J.R. & Robertson J.K. 1996. Equine Pathology. Iowa State University Press, Ames, p.350-351.
- Sacks C.A. 2019. Doughnut lesions. N. Engl. J. Med 380(10):10. <PMid:30855746>
- Skalski J.H., Kottom T.J. & Limper A.H. 2015. Pathobiology of pneumocystis pneumonia: life cycle, cell wall and cell signal transduction. FEMS Yeast Res. 15(6):1-12. <http://dx.doi.org/10.1093/femsyr/fov046> <PMid:26071598>
» https://doi.org/10.1093/femsyr/fov046 - Shultz S., Baral H.S., Charman S., Cunningham A.A., Das D., Ghalsasi G.R., Goudar M.S., Green R.E., Jones A., Nighot P., Pain D.J. & Prakash V. 2004. Diclofenac poisoning is widespread in declining vulture populations across the Indian subcontinent. Proc. Biol. Sci. 271(Suppl.6):S458-S460. <http://dx.doi.org/10.1098/rsbl.2004.0223> <PMid:15801603>
» https://doi.org/10.1098/rsbl.2004.0223 - Stedman T.L. 2006. Stedman’s Medical Dictionary. 28th ed. Lippincott Williams and Wilkins, Philadelphia, 1514p.
- Stromberg P. 2015. Fundamental principles of descriptive anatomic pathology. Available at <Available at https://www.coursehero.com/file/37810733/DESCRIPCION-MACROSCOPICApdf/ > Accessed on Apr. 12, 2019.
» https://www.coursehero.com/file/37810733/DESCRIPCION-MACROSCOPICApdf/ - Taylor K. & Marienau C. 2016. The learning, changing adult brain: experiences, memory and emotions, p.33-58. In: Ibid. (Eds), Facilitating Learning with the Adult Brain in Mind: a conceptual and practical guide. Jossey-Bass, San Francisco.
- Tutor J.D. 2014. Chylothorax in infants and children. Pediatrics 133(4):722-733. <http://dx.doi.org/10.1542/peds.2013-2072> <PMid:24685960>
» https://doi.org/10.1542/peds.2013-2072 - Uzal F.A., Plattner B.L. & Hostetter J.M. 2016. Alimentary System: equine strongylosis, p.216-217. In: Maxie M.G. (Ed.), Jubb, Kennedy and Palmer’s Pathology of Domestic Animals. Vol.2. 6th ed. Elsevier, St Louis. <http://dx.doi.org/10.1016/B978-0-7020-5318-4.00007-3>
» https://doi.org/10.1016/B978-0-7020-5318-4.00007-3 - Valli V.E.O.M., Kiupel M. & Bienzle D. 2016. Anthrax, p.171-174. In: Maxie M.G. (Ed.), Jubb, Kennedy and Palmer’s Pathology of Domestic Animals. Vol.2. 6th ed. Elsevier, St Louis.
- Vandevelde M., Higgins R. & Oevermann A. 2012. Veterinary Neuropathology: essentials of theory and practice. Wiley Blackwell, Ames, p.137-139.
- Vimal M. & Nishanthi A. 2017. Complex pa’thological terminologies simplified with food eponyms. J. Clin. Diagn. Res. 11:EE01-EE06.
- Yoshihara S., Nakagawa M., Suda H., Ikeda K. & Hanashiro K. 1983. White spots of the liver in pigs experimentally infected with Ascaris suum Natl Inst. Anim. Health Q. 23(4):127-137. <PMid:6680775>
- Zhang Q., Cao Y., Wang J., Fu G., Sun M., Zhang L., Meng L., Cui G., Huang Y., Hu X. & Su J. 2018. Isolation and characterization of an astrovirus causing fatal visceral gout in domestic goslings. Emerg. Microbes Infect. 7(1):71. <http://dx.doi.org/10.1038/s41426-018-0074-5> <PMid:29674726>
» https://doi.org/10.1038/s41426-018-0074-5
Publication Dates
-
Publication in this collection
04 Nov 2019 -
Date of issue
Sept 2019
History
-
Received
27 June 2019 -
Accepted
13 July 2019