ABSTRACT:
Selecting glyphosate-resistant Digitaria insularis may cause changes in the biological cycle of the species. Therefore, the objective of this study was to evaluate the biological cycle of susceptible and glyphosate-resistant D. insularis biotypes in two growth periods (summer/autumn and autumn/winter). Seeds were collected in the municipality of Alagoa Grande (07o02’59" S 35o37’01,5" W), and were considered susceptible (S1), in Cascavel (24o55’27" S 53o27’54.6 ”W) considered as resistant 1 (R1), Entre Rios do Oeste (24o40’44" S 54o17’05" W) considered as resistant 2 (R2) and Marechal Cândido Rondon (24o42’30" S 54o21’10" W) considered as resistant 3 (R3). Evaluations were carried out in the summer/autumn and autumn/winter periods. The collection periods of plants were: 14; 21; 28; 35; 42; 49; 56; 63; 70; 77; 84; 91; 98; 105; 112 and 119 days after emergence (DAE). The selection of glyphosate-resistant D. insularis biotypes in agricultural areas affected the biology of the species, in relation to the original population of the susceptible biotype. In the summer/autumn period (mean temperature 24.6 oC), the S biotype started the emission of tillers and flowering at 14 and 21 DAE on an average, respectively, before the resistant biotypes. However, in the autumn/winter period (mean temperature 19.0 oC) only the R1 biotype was able to develop and complete the biological cycle. The evaluated biotypes presented differences during the development, mainly between the phenological stages, so that the susceptible biotype was the most precocious and with a high reproductive potential. The physiological evaluations showed no differences among the S, R1, R2 and R3 biotypes, as verified in the TCR.
Keywords:
Digitaria insularis; growth analysis; weed; chemical control
RESUMO:
A seleção de Digitaria insularis resistente ao glyphosate pode causar alteração no ciclo biológico da espécie. Portanto, objetivou-se neste estudo avaliar o ciclo biológico de biótipos de D. insularis suscetível e resistentes ao glyphosate em dois períodos de crescimento (verão/outono e outono/inverno). As sementes foram coletadas nos municípios de Alagoa Grande (07o02’59" S 35o37’01,5" W), considerado suscetível (S1), Cascavel (24o55’27" S 53o27’54,6" W), resistente 1 (R1), Entre Rios do Oeste (24o40’44" S 54o17’05" W), resistente 2 (R2), e Marechal Cândido Rondon (24o42’30" S 54o21’10" W), resistente 3 (R3). As avaliações foram realizadas no período de verão/outono e outono/inverno. As épocas de coleta das plantas foram: 14, 21, 28, 35, 42, 49, 56, 63, 70, 77, 84, 91, 98, 105, 112 e 119 dias após a emergência (DAE). A seleção de biótipos de D. insularis resistentes ao glyphosate em áreas agrícolas afetou a biologia da espécie, em relação à população original de biótipo suscetível. No período do verão/outono (temperatura média de 24,6 oC), o biótipo S iniciou a emissão de perfilhos e o florescimento, em média, aos 14 e 21 DAE, respectivamente, antes que os biótipos resistentes. Entretanto, no período outono/inverno (temperatura média de 19,0 oC), apenas o biótipo R1 conseguiu se desenvolver e finalizar o ciclo biológico. Os biótipos avaliados apresentaram diferenças durante o desenvolvimento principalmente entre as fases fenológicas, de modo que o biótipo suscetível foi o mais precoce e com elevado potencial reprodutivo. As avaliações fisiológicas não apresentaram diferenças entre os biótipos S, R1, R2 e R3, conforme verificado na TCR.
Palavras-chave:
Digitaria insularis; análise de crescimento; planta daninha; controle químico
INTRODUCTION
Weeds are any superior plants that interfere with human interests and the environment, causing injuries (Pitelli, 2015Pitelli R.A. O termo planta-daninha. Planta Daninha. 2015;33:1-2.). Thus, in agricultural areas, weeds are reported as important due to the difficulty of controlling them, as well as the fact that they compete for water, light and nutrients with crops (Souza et al., 2005Souza L.S. et al. Eficácia do glifosato potássico em mistura com butafenacil no controle de trapoeraba na cultura do café. Rev Bras Herb. 2005;4:1-8.; Martins et al., 2012Martins D. et al. Manejo químico de espécies de trapoeraba com aplicação isolada e em mistura de diferentes herbicidas. Rev Caatinga. 2012;25:21-8.).
Among the main weeds that are difficult to control over the world, there are Digitaria insularis (sourgrass), which has the potential to reduce maize productivity by more than 35% (Gemelli et al., 2013Gemelli A. et al. Strategies to control of sourgrass (Digitaria insularis) glyphosate resistant in the out-of-season corn crop. Rev Bras Herb. 2013;12:162-70.). Therefore, it is necessary to control this species; the chemical method is the most used.
Based on the grain area sown in the State of Paraná in the 2015/16 crop, which was 9,752.3 million ha-1 (Conab, 2016Companhia Nacional de Abastecimento - Conab. Acompanhamento da safra brasileira de grãos. Nono levatamento. Brasília: 2016. 174p. [acessado em: 03 de jul. de 2016]. Disponível em: Disponível em: http://www.conab.gov.br/OlalaCMS/uploads/arquivos/16_06_09_16_49_15_boletim_graos_junho__2016_-_final.pdf
.
http://www.conab.gov.br/OlalaCMS/uploads...
), it is estimated that only about 24.2 thousand tons of the acid equivalent of glyphosate (average of 2.5 L ha-1) were used for desiccation management. Thus, with the increase of areas sown with transgenic crops (soybean and corn) and with the winter crop management, the number of applications can reach a minimum of five sprayings, further increasing the amount of herbicides that is used.
The intensive use of herbicides in crop management can cause selection of resistant weed biotypes (Beckie and Reboud, 2009Beckie H.J., Reboud X. Selecting for weed resistance: herbicide rotation and mixture. Weed Technol. 2009;23:363-70.; Vencill et al., 2012Vencill W.K. et al. Herbicide resistance: toward an understanding of resistance development and the impact of herbicide-resistant crops. Weed Sci. 2012;60:2-30.), as already verified by Carvalho et al. (2011Carvalho L.B. et al. Detection of sourgrass ( Digitaria insularis ) biotypes resistant to glyphosate in Brazil. Weed Sci. 2011;59:171-6.) in populations of D. insularis. Carvalho et al. (2012Carvalho L.B. et al. Pool of resistance mechanisms to glyphosate in Digitaria insularis. J Agric Food Chem. 2012;60:615-22.) found mutations in glyphosate-resistant plants - double mutations in the amino acid sequence that translate the protein - at positions 182 and 310 of the enzyme EPSPS (enzyme 5-enolpyruvylsyquimate 3-phosphate synthase). Thus, these authors concluded that the expression of the EPSPS enzyme could be influenced by these mutations or by the expression of these different alleles.
In Paraná, the occurrence of D. insularis biotypes that resistant to EPSPS inhibiting glyphosate was reported in the agricultural areas of four municipalities; likewise, the occurrence of differentiated sensitivity to clethodim (ACCase inhibitor) was observed in these biotypes. In the areas of Santa Mariana and Cascavel, clethodim doses to control 80% of the biotypes were 179.81 and 191.95 gha-1 respectively, while to obtain the same control of the biotypes in the area of Maringá and Palotina, it was necessary to apply 82.15 and 96.54 g ha-1 of clethodim (Licorini et al., 2015Licorini L.R. et al. Identificação e controle de biótipos resistentes de Digitaria insularis (L.) Fedde ao glyphosate. Rev Bras Herb. 2015;14:141-7.).
One of the alternatives to avoid the impact caused by herbicides may be the use of the integrated weed management (IWM). This practice is based on the integration of control methods, reducing the selection pressure provided by herbicides (Nunes et al., 2010Nunes A.L. et al. Manejo integrado de plantas daninhas na cultura do milho. Bragantia. 2010;69:299-304.). Thus, the bioecological study of weeds is important, since knowing the behavior of these plants provides subsidies to devise management strategies (Vargas et al., 2005Vargas L. et al. Alteração das características biológicas dos biótipos de azevém (Lolium multiflorum) ocasionada pela resistência ao herbicida glyphosate. Planta Daninha. 2005;23:153-60. ).
Although there are some studies on the biology and morphology and the effect of temperature on D. insularis plants (Machado et al., 2006Machado A.F.L. et al. Análise de crescimento de Digitaria insularis. Planta Daninha. 2006;24:641-7.; Marques et al., 2014Marques B.S. et al. Growth and development of sourgass based on days or thermal units. Planta Daninha. 2014;32:483-90.), there are still few studies on growth periods, biological cycle and development, with susceptible and resistant glyphosate biotypes.
Therefore, the hypotheses of this study are based on the fact that the selection of glyphosate-resistant D. insularis can cause changes in the biological cycle of the species, in relation to the population of susceptible biotypes, depending on the plant development period.
In this context, the objective of this study was to evaluate the biological cycle of glyphosate-resistant D. insularis biotypes in the western region of Paraná, in experiments conducted in two growth periods.
MATERIAL AND METHODS
Two experiments were conducted: the first between the summer/autumn period and the second in the autumn/winter period of 2015, conducted in pots, which were placed under field conditions (protection-free area).
Seeds from the susceptible Digitaria insularis biotype were collected in populations from geographically distant agricultural areas, belonging to producers of the Brejo Paraibano, in the municipality of Alagoa Grande (S: 07o02’59" S 35o37’01,5" W), in a pasture area, with no history of herbicide use. Seeds from the resistant biotypes were collected in the western region of Paraná, in the municipalities of: Cascavel (R1: 24o55’27" S; 53o27’54,6" W), Entre Rios do Oeste R2: 24o40’44" S; 54o17’05" W), and Marechal Cândido Rondon (R3: 24o42’30" S; 54o21’10" W), in maize and soybean areas, with a history of resistance to glyphosate.
Choosing the biotype from Paraíba was due to the difficulty to obtain seeds of susceptible plants in Paraná. As for the resistant biotypes, the regional ones were chosen, because they underwent great selection pressure from the herbicide glyphosate.
For the collection of seeds, 50 plants were sampled with panicles initiating seed dispersal. For this procedure, the inflorescence was placed inside a bag and agitated until the seeds were detached.
Preliminarily, a test of resistance demonstration of the biotypes was conducted, using an adaptation of the methodology proposed by Reinert et al. (2013Reinert C.S. et al. Curvas de dose-resposta comparativas entre os biótipos resistente e suscetível de capim-amargoso (Digitaria insularis) ao herbicida glyphosate. Rev Bras Herb. 2013;12:260-7.). The biotypes from Cascavel (R1), Entre Rios do Oeste (R2) and Marechal Cândido Rondon (R3) presented a Resistance Factor (FR50) of 2.7, 3.4 and 7.7, respectively.
For the growth analysis of biotypes, the experimental design was completely randomized with four replications. Treatments consisted in plant collection periods, performed at regular seven-day intervals, which occurred 14, 21, 28, 35, 42, 49, 56, 63, 70, 77, 84, 91, 98, 105 , 112 and 119 days after emergence (DAE), corresponding to 16 collections.
In the evaluations of the growth curves of both experiments the same methodologies were used. The first experiment was conducted between January 21st and May 20th, 2015, during the summer/autumn period, with a mean temperature of 24.6 oC; and the second was conducted between May 11th and September 8th, 2015, in the autumn/winter period, with an average temperature of 19 oC.
The meteorological data observed in each experimental period are described in Figure 1.
Seeds from the studied biotypes were placed to germinate in plastic trays containing Plant Max® organic substrate. Germination started seven days after sowing. Irrigations were carried out daily, maintaining the substrate near 80% of the field capacity.
Seedlings were transplanted 14 days after emergence (DAE) into pots with a capacity of 12 dm-3, containing clayey soil classified as Eutroferric Red Latosol, previously fertilized with a recommendation of basic fertilization for maize (Poaceae), using 380 kg ha-1 of the NPK-11-19-14 formula (Raij et al., 1997Raij B.van et al. Recomendações de adubação e calagem para o Estado de São Paulo. 2ªed. Campinas: IAC, 1997. 285p. (Boletim técnico, 100).).
Such formula was used because plants belong to the same family and present an excellent development when present in agricultural areas.
Four top-dressings were also carried out during the growth of plants, at 35, 56, 77 and 98 DAE. Doses were equivalent to 0.015 g of N, 0.005 g of P2O5 and 0.010 g of K2O per pot, in each fertilization, according to Machado et al. (2006Machado A.F.L. et al. Análise de crescimento de Digitaria insularis. Planta Daninha. 2006;24:641-7.).
Before each destructive evaluation, the characterization of the phenological stages was carried out, following a model adapted from Bleiholder et al. (1991Bleiholder H. et al. Codificação unificada dos estádios fenológicos de culturas e ervas daninhas. Pesq Agropec Bras.1991;26:1423-9.). The leaf area was obtained by the Licor Instruments area meter (model LI 3100), which measures the area of each leaf in cm-2.
All parts of the plants were then dried in a forced air ventilation oven at 65 oC, until constant weight. The determination of the total dry matter of plants was obtained by the sum of the shoot and root matter.
With the leaf area and dry matter value it was possible to determine, for each evaluation period, the Net Assimilation Rate (NAR), the Leaf Area Ratio (LAR) and the Relative Growth Rate (RGR), according to the methodology suggested by Benincasa (2003Benincasa M.M.P. Análise de crescimento de plantas; noções básicas. Jaboticabal: FUNEP, 2003. 41p.).
Data about number of tillers, inflorescence number and total dry matter, in addition to growth analysis data, were submitted to regression analysis, and the model was chosen taking into account the logic of the biological phenomenon, the normality, the regression significance and the high determination coefficient.
RESULTS AND DISCUSSION
Comparing the phenological stage differences of the Digitaria insularis biotypes in the summer/autumn experiment, the beginning of tillering in the S biotype was observed 28 days after emergence (DAE), in R1 and R3 at 42 DAE, and in R2 at 49 DAE (Table 1). The full flowering was verified at 56 DAE for the S biotype, in the R1 biotype at 77 DAE, while in the R2 and R3 biotypes only at 84 DAE.
Comparison and characterization of of the phenological stages of susceptible (S) and glyphosate resistant (R1), (R2), and (R3) biotypes in the summer/autumn and autumn/winter
Thus, when observing the beginning of the vegetative and reproductive phase development of plants in the summer/autumn period, it was verified that the S biotype was the most precocious, R1 and R3 had an intermediate development, and R2 was the latest.
In the autumn/winter period, only the R1 biotype was able to develop and end the cycle, while S, R2, and R3 showed paralyzed growth (they did not die, but did not develop either).
In the vegetative stage of biotype R1, in the autumn/winter period, the development was similar to the one observed in the summer/autumn period, but in the reproductive stage the cycle was prolonged (Figure 1). The behavior shown by biotype R1 may be a physiological response of the plant to compensate for the energy output observed in the off-season development, since a low temperature does not favor the growth and development of C4 plants.
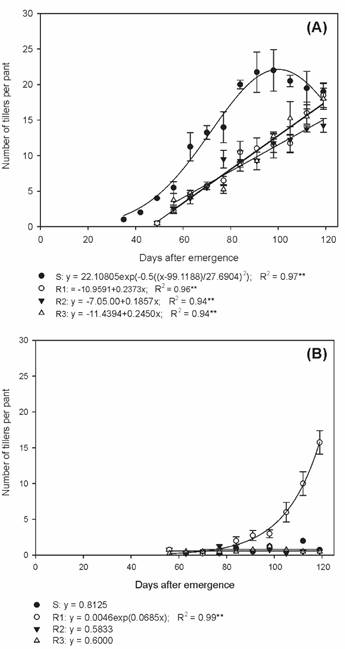
Biotype S presented the highest number of tillers in all evaluations during the summer/autumn period, in relation to the others (Figure 2A). At the end of the cycle, biotypes S, R1, R2 and R3 presented an average of 19.0, 18.5, 14.2 and 18.0 tillers per plant, respectively. In the autumn/winter period, the R1 biotype presented an average of 15.7 tillers per plant (Figure 2B), being approximately 86.2% lower than the number of tillers per plant observed during the summer/autumn period (Figure 2A).
Number of tillers of susceptible and glyphosate-resistant Digitaria insularis biotypes in summer/autumn (A) and autumn/winter (B).
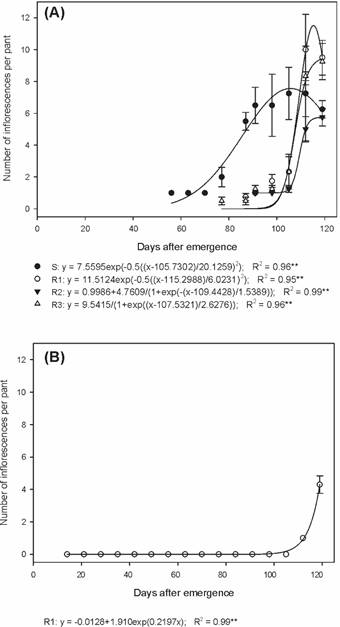
In the summer/autumn period, the S biotype produced 42.7, 20.3 and 47.1% more inflorescences per plant than the R1, R2 and R3 biotypes, respectively (Figure 3A). The emission of inflorescences of the R1 biotype during the autumn/winter was only verified in the last two evaluations, with an average of 4.3 inflorescences per plant at 119 DAE (Figure 3B).
Number of inflorescences of susceptible and glyphosate-resistant Digitaria insularis biotypes, conducted in the summer/autumn (A) and autumn/winter (B).
Despite the differences observed among the biotypes at the two stages (vegetative and reproductive) during the summer/autumn period, it was verified that the maximum accumulation of total dry matter was similar for all biotypes. However, the S biotype presented 37.3 g per plant at 91 DAE, while the maximum total dry matter accumulation for R1 was 37.8 g per plant at 112 DAE; for R2, it was 31.8 g per plant at 119 DAE; and for R3, it was 33.9 g per plant at 112 DAE (Figure 4A). R1 plants in the autumn/winter period had a total dry matter 74.0% lower than the one observed in the summer/autumn period (Figure 4B).
Total dry matter of susceptible and glyphosate-resistant Digitaria insularis biotypes in summer/autumn (A) and autumn/winter (B).
Machado et al. (2006Machado A.F.L. et al. Análise de crescimento de Digitaria insularis. Planta Daninha. 2006;24:641-7.) observed, in D. insularis plants, a slow growth up to 45 DAE and the maximum total dry matter accumulation (30.7 g per plant) at 105 DAE.
Generally speaking, these results confirm that the S biotype showed a more precocious development than the resistant biotypes, as well as the fact that resistant biotypes can also develop even under unfavorable growth conditions.
Therefore, the adaptation of D. insularis to periods with low temperatures can lead to an increase in the population of resistant biotypes and make it difficult to manage the species at any time of the year (Gemelli et al., 2012Gemelli A. et al. Aspectos da biologia de Digitaria insularis resistente ao glyphosate e implicações para o seu controle. Rev Bras Herb. 2012;11:231-40.).
Similar results were also found by Vargas et al. (2005Vargas L. et al. Alteração das características biológicas dos biótipos de azevém (Lolium multiflorum) ocasionada pela resistência ao herbicida glyphosate. Planta Daninha. 2005;23:153-60. ), who found, in glyphosate-resistant Lolium multiflorum biotypes, a lower number of tillers and later flowering than the susceptible ones.
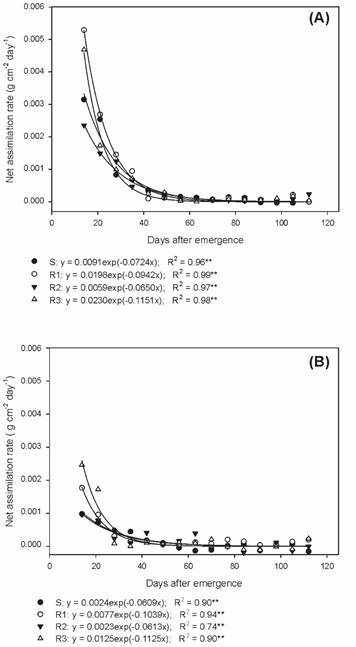
In the physiological studies of the biotypes, it was observed that the net assimilation rate (NAR) decreased throughout the development in all biotypes, in a similar manner, up to 42 DAE in the summer/autumn period, and from that date, there was stabilization until the end of the cycle (Figure 5A). Likewise, the decrease in NAR was up to 28 DAE during autumn/winter, with later stabilization until the end of the experiment (Figure 5B).
Net assimilation rate of susceptible and glyphosate-resistant Digitaria insularis biotypes in summer/autumn.
Thus, it is evident that when plants increase their leaf area, there is self-shading and, consequently, a decrease of the liquid photosynthesis, reflecting directly in the NAR; the lower the value of NAR, the greater the competitiveness of the species (Gomide et al., 2003Gomide C.A.M. et al. Índices morfogênicos e de crescimento durante o estabelecimento e a rebrotação do capim-mombaça (Panicum maximum Jacq.). Rev Bras Zoot. 2003;32:795-803.).
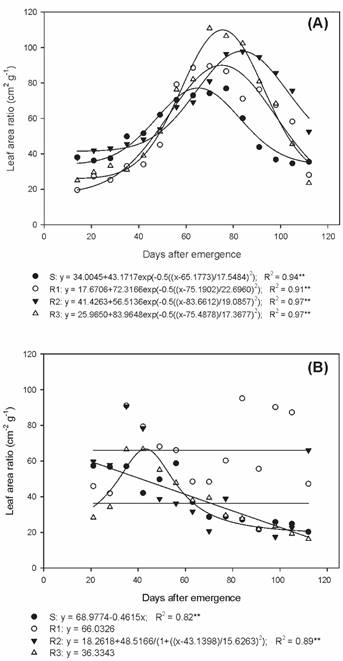
As for the leaf area ratio (LAR) during summer/autumn, distinct peaks were observed for the studied biotypes (Figure 6A). The S biotype reached the LAR peak at 77 DAE, with 76.9 cm2 g-1; biotypes R1 and R3 presented a peak at 70 DAE, with 89.5 and 110.8 cm2 g-1, respectively; and R2 showed the peak at 84 DAE with 97.7 cm2 g-1. In autumn/winter, the R1 biotype presented a LAR with a mean of 66.0 cm2 g-1 (Figure 6B).
Leaf area ratio of susceptible and glyphosate-resistant Digitaria insularis biotypes in autumn/winter.
The LAR values of the resistant biotypes were higher than those of the S biotype in the summer/autumn period, mainly at the reproductive stage. This fact reveals the occurrence of morpho-physiological changes between resistant and susceptible biotypes, since glyphosate-resistant biotypes required a larger leaf area that is photosynthetically useful to produce 1 g of dry matter, in relation to the S biotype.
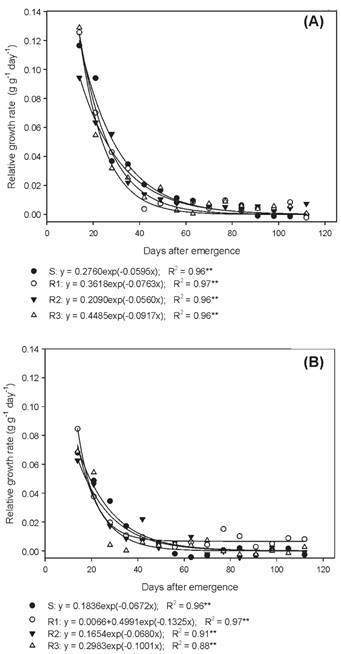
The relative growth rate (RGR) estimates the increase in the dry matter of plants, per unit of initial weight, over a given time interval (Benincasa, 2003Benincasa M.M.P. Análise de crescimento de plantas; noções básicas. Jaboticabal: FUNEP, 2003. 41p.). The RGR values observed among the biotypes in the summer/autumn and autumn/winter showed a similar behavior, corroborating NAR data (Figure 7A, B).
Relative growth rate of susceptible and glyphosate-resistant Digitaria insularis biotypes in autumn/winter.
Generally speaking, it was found that the best growth season of D. insularis was the summer/autumn period. Biotype S presented precocity in the emission of tillers and inflorescences, as well as a greater efficiency in accumulating dry matter (<LAR). It is worth highlighting that the greater reproductive capacity of the susceptible biotype constitutes an innate evolutionary characteristic of aggressiveness to dominate the environment and to perpetuate the species and that, consequently, it suppresses the development of slower growing biotypes.
Therefore, the selection of glyphosate-resistant D. insularis biotypes has negative impacts on the growth and developmental ability of plants; the distribution in the environment of these biotypes is favored by the absence of competition with the susceptible (more adapted) biotype, caused by the intensive use of chemical management (Vargas et al., 2005Vargas L. et al. Alteração das características biológicas dos biótipos de azevém (Lolium multiflorum) ocasionada pela resistência ao herbicida glyphosate. Planta Daninha. 2005;23:153-60. ). However, some resistant biotypes can develop under unfavorable conditions (autumn/winter) and cause interference in winter crops.
The evaluated biotypes showed differences during their development, mainly between the phenological stages, and the susceptible biotype was the most precocious and with a high reproductive potential in the summer/autumn period (average temperature of 24.6 oC). However, in the autumn/winter period (average temperature of 19.0 oC) only the R1 biotype was able to develop and complete the biological cycle. In the analysis of growth, similar NAR and RGR values were verified between the biotypes in both evaluation periods, but the LAR values of the resistant biotypes were higher than those of the S biotype in summer/autumn.
REFERENCES
- Beckie H.J., Reboud X. Selecting for weed resistance: herbicide rotation and mixture. Weed Technol. 2009;23:363-70.
- Benincasa M.M.P. Análise de crescimento de plantas; noções básicas. Jaboticabal: FUNEP, 2003. 41p.
- Bleiholder H. et al. Codificação unificada dos estádios fenológicos de culturas e ervas daninhas. Pesq Agropec Bras.1991;26:1423-9.
- Carvalho L.B. et al. Detection of sourgrass ( Digitaria insularis ) biotypes resistant to glyphosate in Brazil. Weed Sci. 2011;59:171-6.
- Carvalho L.B. et al. Pool of resistance mechanisms to glyphosate in Digitaria insularis. J Agric Food Chem. 2012;60:615-22.
- Companhia Nacional de Abastecimento - Conab. Acompanhamento da safra brasileira de grãos. Nono levatamento. Brasília: 2016. 174p. [acessado em: 03 de jul. de 2016]. Disponível em: Disponível em: http://www.conab.gov.br/OlalaCMS/uploads/arquivos/16_06_09_16_49_15_boletim_graos_junho__2016_-_final.pdf
» http://www.conab.gov.br/OlalaCMS/uploads/arquivos/16_06_09_16_49_15_boletim_graos_junho__2016_-_final.pdf - Gemelli A. et al. Aspectos da biologia de Digitaria insularis resistente ao glyphosate e implicações para o seu controle. Rev Bras Herb. 2012;11:231-40.
- Gemelli A. et al. Strategies to control of sourgrass (Digitaria insularis) glyphosate resistant in the out-of-season corn crop. Rev Bras Herb. 2013;12:162-70.
- Gomide C.A.M. et al. Índices morfogênicos e de crescimento durante o estabelecimento e a rebrotação do capim-mombaça (Panicum maximum Jacq.). Rev Bras Zoot. 2003;32:795-803.
- Licorini L.R. et al. Identificação e controle de biótipos resistentes de Digitaria insularis (L.) Fedde ao glyphosate. Rev Bras Herb. 2015;14:141-7.
- Machado A.F.L. et al. Análise de crescimento de Digitaria insularis Planta Daninha. 2006;24:641-7.
- Marques B.S. et al. Growth and development of sourgass based on days or thermal units. Planta Daninha. 2014;32:483-90.
- Martins D. et al. Manejo químico de espécies de trapoeraba com aplicação isolada e em mistura de diferentes herbicidas. Rev Caatinga. 2012;25:21-8.
- Nunes A.L. et al. Manejo integrado de plantas daninhas na cultura do milho. Bragantia. 2010;69:299-304.
- Pitelli R.A. O termo planta-daninha. Planta Daninha. 2015;33:1-2.
- Raij B.van et al. Recomendações de adubação e calagem para o Estado de São Paulo. 2ªed. Campinas: IAC, 1997. 285p. (Boletim técnico, 100).
- Reinert C.S. et al. Curvas de dose-resposta comparativas entre os biótipos resistente e suscetível de capim-amargoso (Digitaria insularis) ao herbicida glyphosate. Rev Bras Herb. 2013;12:260-7.
- Souza L.S. et al. Eficácia do glifosato potássico em mistura com butafenacil no controle de trapoeraba na cultura do café. Rev Bras Herb. 2005;4:1-8.
- Vargas L. et al. Alteração das características biológicas dos biótipos de azevém (Lolium multiflorum) ocasionada pela resistência ao herbicida glyphosate. Planta Daninha. 2005;23:153-60.
- Vencill W.K. et al. Herbicide resistance: toward an understanding of resistance development and the impact of herbicide-resistant crops. Weed Sci. 2012;60:2-30.
Publication Dates
-
Publication in this collection
2018
History
-
Received
15 Jan 2017 -
Accepted
25 Mar 2017