ABSTRACT:
Considering the possibility of a intercropping between exotic and native wood species for Legal Reserve areas restoration, information is needed about the possible interactions (positive or negative) between the involved species. The aim of this study was to determine the allelochemicals classes of aqueous extracts from dried and fresh leaves of torelliodora eucalyptus, as well as to evaluate the allelopathic potential of these extracts in the germination and initial growth of Guazuma ulmifola, a native species from Cerrado. To obtain the aqueous extracts at the concentrations of 25 mg mL-1, 50 mg mL-1, 100 mg mL-1 and 200 mg mL-1, the fresh and dried leaves of the eucalyptus were separately subjected to turbolize, followed by an ultrasound bath for two hours and static maceration, in darkness, for 48 hours in a refrigerator. The extracts were subjected to classical phytochemical analysis and determination of pH, electrical conductivity, soluble solids and allelopathic tests. For the germination and growth biological tests, a completely randomized experimental design with four replications per treatment was performed. The phytochemical analysis indicated that the phenolic compounds and derivatives are the major constituents of the aqueous extract of fresh and dried leaves of the tolleriodora eucalyptus, with predominance of the flavonoid contents on the dried leaves. Dried and fresh leaves extracts of torelliodora eucalyptus influenced G. ulmifolia germination, but this effect was dependent on their concentration. Initial seedling growth of G. ulmifolia was negatively influenced only by the dried leaves extract of the exotic species.
Keywords:
allelochemicals; Legal Reserve restoration; Guazuma ulmifolia; exotic species
RESUMO:
Considerando a possibilidade de consórcio entre espécies exóticas e nativas para a restauração de áreas de Reserva Legal, são necessárias informações sobre as possíveis interações (positivas ou negativas) entre as espécies envolvidas. Assim, neste estudo objetivou-se determinar as classes de aleloquímicos dos extratos aquosos de folhas secas e frescas do eucalipto torelliodora, assim como avaliar o potencial alelopático destes extratos na germinação e crescimento inicial de plântulas de Guazuma ulmifolia, uma espécie nativa do Cerrado. Para obtenção dos extratos aquosos nas concentrações de 25 mg mL-1, 50 mg mL-1, 100 mg mL-1 e 200 mg mL-1, as folhas secas e frescas do eucalipto, separadamente, foram submetidas a turbólise, seguida de banho de ultra-som por duas horas e maceração estática, sem a presença de luz, por 48 horas em geladeira. Os extratos foram submetidos a análise fitoquímica clássica e determinação do pH, condutividade elétrica, sólidos solúveis e testes alelopáticos. Para os ensaios biológicos de germinação e crescimento, foram realizados testes com delineamento experimental completamente randomizado, com quatro repetições por tratamento. A análise fitoquímica indicou que os compostos fenólicos e derivados são os constituintes majoritários do extrato aquosos de folhas frescas e secas do eucalipto tolleriodora, com predominância dos teores de flavonoides nas folhas secas. Os extratos aquosos de folhas secas e frescas do eucalipto tolleriodora influenciaram a germinação de G. ulmifolia, porém esse efeito foi dependente da sua concentração. O crescimento inicial de plântulas de G. ulmifolia foi negativamente influenciado apenas pelo extrato de folhas secas da espécie exótica.
Palavras-chave:
aleloquímicos; restauração de Reserva Legal; Guazuma ulmifolia; espécies exóticas
INTRODUCTION
The areas of Legal Reserve (LR) and Permanent Preservation (APP) represent the most important figures of preservation as defined by the new Brazilian Forest Code (Brasil, 2012Brasil. Lei 12.651, de 25 de maio de 2012. Dispõe sobre a proteção da vegetação nativa; altera as Leis nos 6.938, de 31 de agosto de 1981, 9.393, de 19 de dezembro de 1996, e 11.428, de 22 de dezembro de 2006; revoga as Leis nos 4.771, de 15 de setembro de 1965, e 7.754, de 14 de abril de 1989, e a Medida Provisória no 2.166- 67, de 24 de agosto de 2001; e dá outras providências. Diário Oficial da República Federativa do Brasil, Brasília, DF, 25 maio de 2012. ) and rural properties with environmental liabilities will have a term of 20 years for their regularization after registration in Environmental Rural Registry. Specifically regarding the LR, the new law has created mechanisms to facilitate the environmental adequacy, reducing the area to be protected or restored, allowing the discount of APP in the LR calculation, discharging small and medium properties to restore or compensate for LR and allowing the use of exotic species, in consortium with native trees for their restoration (Brancalion et al., 2016Brancalion PHS, Garcia LC, Loyola R, Rodrigues RR, Pillar VD, Lewinsohn TM. Análise crítica da Lei de Proteção da Vegetação Nativa (2012), que substituiu o antigo Código Florestal: atualizações e ações em curso. Nat Conserv. 2016;14:1-16.).
According to IBGE (2015Instituto Brasileiro de Geografia e Estatística - IBGE. Produção da extração vegetal e silvicultura. Rio de Janeiro: 2015. 48p.), eucalyptus is the most used essence of exotic forest in commercial plantations in Brasil, totaling 75% of the 9,935,353 hectares occupied by planted forests. Thus, it is plausible to assume that intercropped plantation between native and exotic in the country for restoration of Legal Reserve areas can use these species of this genus. However, several studies show that eucalyptus is an allelopathic species, interfering in the germination, growth and development of species that are established in their vicinity (May and Ash, 1990May FE, Ash JE. An assessment of the Allelopathic Potential of Eucalyptus. Aust J Bot. 1990;38:245-54. ; Goetze and Thomé, 2004Goetze M, Thomé G. Efeito alelopático de extratos de Nicotiana tabacum e Eucalyptus grandis sobre a germinação de três espécies de hortaliças. Rev Bras Agrocienc. 2004;10:43-50.; Bedin et al., 2006Bedin C, Mendes LB, Trecente VC, Silva JMS. Efeito Alelopático de extrato de Eucalyptus citriodora na germinação de sementes de tomate (Lycopersicum esculentum M.). R Cient Eletr Agron. 2006;5:1-7.; Zhang et al., 2010Zhang D, Zhang J, Wu F-Z. Potential allelopathic effect of Eucalyptus grandis across a range of plantation ages. Ecol Res. 2010;25:13-23.; Yamagushi et al., 2011Yamagushi MQ, Gusman GS, Vestena S. Efeito alelopático de extratos aquosos de Eucalyptus globulus Labill. e de Casearia sylvestris Sw. sobre espécies cultivadas. Semina: Cienc Agrar 2011;32(4):1361-74.), adversely affecting the development of native species included in consortium and the establishment of seeds originated from the reproduction of these which are able to establish themselves.
The action of the allelochemicals from different materials of eucalyptus is already described in the literature on different species (Sasikumar et al., 2002Sasikumar K. Allelopathic effects ofeucalyptus on blackgram (Phaseolus mungo L.). Allelopathy J. 2002;9:205-14.; Florentine and Fox, 2003Florentine SK, Fox JED. Allelopathic effects of Eucalyptus victrix L. on Eucalyptus species and grasses. Allelopathy J. 2003;11:77-83.; Javed et al., 2012Javed S, Shoaib A, Mahmood Z, Mushtaq S, Iftikhar S. Analysis of phytochemical constituents of Eucalyptus citriodora L. responsible for antifungal activity against post-harvest fungi. Nat Prod Res. 2012;26(18):1732-6.; Bhagat et al., 2012Bhagat M, Sharma V, Saxena AK. Anti-proliferative effect of leaf extracts of Eucalyptus citriodora against human cancer cells in vitro and in vivo. Indian J Biochem Biophys. 2012;49(6):451-7.; Avello et al., 2016Avello M, Fernández Pola, Fernández M, Schulz B, De Diego M, Sigrid M et al. Efecto pediculicida de una formulación en base a Eucaliptus globulus L. Rev Chil Infectol. 2016;33(4):433-7. ). However, for the torelliodora eucalyptus (hybrid resulting from Corymbia citriodora x Eucalyptus torelliana) no chemical constituents were reported with allelopathic action. This material is interesting for use by producers due to having multiple uses, such as for example, cellulose, paper, sawn timber and beekeeping, among others.
Considering the possibility of a consortium between torelliodora and eucalyptus and native forest species, such as the Guazuma ulmifolia Lam., a common tree species in Cerrado and widely used for the restoration of degraded areas in the Biome, information is needed about the possible interactions (positive or negative) between the involved species. Although Oliveira et al. (2016Oliveira APP, Pereira SR, Cândido ACS, Laura VA Peres MTLP. Can allelopathic grasses limit seed germination and seedling growth of Mutambo? A test with two species of Brachiaria grasses. Planta Daninha. 2016;34(4):639-48.) have demonstrated that G. ulmifolia is susceptible to the allelopathic effects of two species of Urochloa, an exotic grass, the result of the interaction between the native species and species of eucalyptus has still not been investigated, as well as the allelochemicals of torelliodora eucalyptus. Therefore, this study had as objective determine the classes of allelochemicals of aqueous extracts of dried and fresh leaves, as well as to evaluate the allelopathic potential of torelliodora eucalyptus on germination and initial seedlings growth of G. ulmifola.
MATERIAL AND METHODS
Collection of botanical material
The collection of torelliodora eucalyptus leaves was performed manually, with the aid of a trimmer, on 08/24/2016 between 7:00 and 9:00 in the morning period, observing the premise of absence of rain in three days prior and on the day of collection. Young leaves were collected from eight trees with three years of age (100 grams per tree), cut at a height of approximately 6 meters from the ground, in the experimental field of Anhanguera-Uniderp University, Campo Grande-MS (20o26’2" S 54o32’27" W). The samples were packed in plastic bags and taken to the laboratory. Part of the botanical material was used for the extracts preparation at concentrations of 25 mg mL-1, 50.0 mg mL-1, 100 mg mL-1 and 200 mg mL-1 (20.0%). In parallel, the other part was dried at room temperature (25 ± 2 oC), for two days, and in sequence crushed in a blender and the extracts were obtained in different concentrations (25 mg mL-1, 50 mg mL-1, 100 mg mL-1 and 200 mg mL-1), and part of the dried material was stored in polyethylene bottle.
The fruits of Guazuma ulmifolia were collected from 13 arrays located in Parque das Nações Indígenas in Campo Grande - MSS (20o27’23.0" S 54o34’47.5" W), brought to the Seed Laboratory of the University, where the manual extraction of the seeds was later carried out.
Preparation of the extracts
To obtain the aqueous extracts at the concentrations of 25 mg mL-1, 50 mg mL-1, 100 mg mL-1 and 200 mg mL-1, the fresh and dried leaves of the eucalyptus were separately subjected to turbolize, followed by an ultrasound bath for two hours and static maceration, in darkness, for 48 hours in a refrigerator, following the adapted methodology of Oliveira et al. (2011aOliveira AKM, Ribeiro JWF, Matias R, Gusmão DDH, Pereira KCL. Potencial alelopático de folhas frescas de bacupari (Rheedia brasiliensis (Mart.) Planch. & Triana) na germinação de alface. Rev Bras Biocienc. 2011a;4(9):550-3. ). The extracts were filtered (funnel glass and cotton) in 100 mL volumetric flask (±0.12 mL) and denominated FreshExtH2O (Aqueous fresh leaves extract) DriedExtH2O (Aqueous dried leaves extract).
Phytochemistry
The phytochemical analysis of the extracts FreshExtH2O and DriedExtH2O were performed according to Matos (2009Matos JFA. Introdução a fitoquímica experimental. 2ª. ed. Fortaleza: UFC.; 2009. 150p.), in triplicates, through wet route, through reactions of precipitation and/or change of color. The results were classified as: in negative (-), Partial(±), a weak positive(+), moderately moderate(++), moderate (++±) and high intensity(+++), with a frequency of 0, 10, 25, 50, 75 and 100%, respectively, as described by Fontoura et al. (2015Fontoura FM, Matias R, Ludwig J, Oliveira AKM, Bono JAMaior, Martins PFRB, et al. Seasonal effects and antifungal activity from bark chemical constituents of Sterculia apetala (Malvaceae) at Pantanal of Miranda, Mato Grosso do Sul, Brazil. Acta Amaz. 2015;45(3):283-92. ), with exception to the saponins analysis that was determined using one g of dried leaves, to estimate the rate of saponins (afrosymmetric Rate), the same procedure occurred with the fresh leaves (Schenkel et al., 2010Schenkel EP, Gosmann G, Athayde ML. Saponinas. In: Simões CMO, Schenkel EP, Mello JCP, Mentz LA, Petrovick PR. editores. Farmacognosia: da planta ao medicamento. Florianopolis: UFSC; 2010. p.711-34. ).
Confirmation of the major constituents in the extracts occurred by microscopy in UV-Visible spectrum (FEMTO®, 800XI), a region of 190 to 750 nm in quartz cuvettes. The analyzes were carried out with three repetitions and the absorption bands, compared with the literature (Mabry et al., 1970Mabry TJ, Markham KR, Thomas MB. The systematic identification of Flavonoids. Austin: University of Texas; 1970.; Ugaz, 1994Ugaz O. Investigación fitoquímica: Métodos en el estudio de produtos naturales. Lima: Fondo Editorial de la Pontifícia de la Universidad Católica del Perú; 1994. p.23-42.; Kumar, 2006Kumar S. Organic chemistry: Spectroscopy of organic compounds. Amritsar: Department of Chemistry, Guru Nanak Dev University; 2006. 36p.; Silverstein, 2007Silverstein RM, Webster FX, Kiemle DJ. Identificação espectrométrica de compostos orgânicos. 7ª. ed. Rio de Janeiro: LTC; 2007. 490p.; Zuanazzi et al., 2017Zuanazzi JAS, Montanha JA, Zucolotto SM. Flavonoides. In: Simões CMO, Schenkel EP, Mello JCP, Mentz LA, Petrovick PR, editores. Farmacognosia: do produto natural ao medicamento. Porto Alegre: Artmed; 2017. p.209-33.).
Determination of the content of phenolic compounds and flavonoids
For the determination of the content of phenolic compounds in the extracts FreshExtH2O and DriedExtH2O, the Folin-Ciocalteau method was employed , as standard gallic acid was used (10 to 350 mg mL-1) for the construction of the calibration curve (; r² = 0.9959) (Sousa et al., 2007Sousa CMM, Silva HR, Vieira-Jr GM, Ayres MCC, Costa CLS, Araújo DS, et al. Fenóis totais e atividade antioxidante de cinco plantas medicinais. Quím Nova. 2007;30:351-5.). The method employed for determination of flavonoids, in the extracts in the present study, was the aluminum chloride and for the construction of the calibration curve quercetin was used as standard (6.0, 8.0, 10.0, 12.0, 16.0 and 20.0 µg mL-1) ( R² = 0.9949) (Peixoto Sobrinho et al., 2008Peixoto Sobrinho TJS, Silva CHTP, Nascimento JE, Monteiro JM, Albuquerque UP, Amorim ELC. Validação de metodologia espectrofotométrica para quantificação dos flavonóides de Bauhinia cheilantha (Bongard) Steudel. Rev Bras Cienc Farm. 2008;44(4):683-9.). For the two measurements the experimental design was three replicates for each concentration and the calculation of averages was accompanied by the standard deviation.
Determination of pH, electrical conductivity and soluble solids
The extracts were also subjected to the analysis of pH (Digimed DM-20), electrical conductivity (Digimed DM3) and concentration of soluble solids, determined using a refractometer (Digital Refractometer, RTD-45 model), with results expressed in degrees Brix corrected to 20 oC.
Tests of germination and growth
The germination bioassays were conducted in the gerbox-type plastic boxes containing two sheets of blotter paper moistened with 14 mL of solutions of FreshExtH20 and DriedExtH2O of eucalyptus plantations in concentrations of 25 mg mL-1, 50 mg mL-1, 100 mg mL-1 and 200 mg mL-1. As a control, similar procedure was performed, using, however, 14 mL of distilled water. For dormancy break of Guazuma ulmifolia seeds were immersed in water at 90 oC, removing the heat source and keeping them in the same water for an hour (Brasil, 2013Brasil. Ministério da Agricultura, Pecuária e Abastecimento. Instruções para análise de sementes florestais. Brasília: 2013. 97p.). Then 100 seeds were sown per gerbox, using four replicates for each treatment of both extracts (fresh leaves and dried leaves). The boxes containing the seeds were maintained at constant temperature of 25 ºC, with a photoperiod of 12 h (Brasil, 2013Brasil. Ministério da Agricultura, Pecuária e Abastecimento. Instruções para análise de sementes florestais. Brasília: 2013. 97p.). The Average Germination Time (AGT) was evaluated, as estimated by the equation AGT = ∑niti) / ∑ni, being: ni = the number of germinated seeds per day and ti = time of incubation in days (Krzyzanowski et al., 1999Krzyzanowski FC, Vieira RD, França Neto JB. Vigor de sementes: conceitos e testes. Londrina: ABRATES; 1999. 218p.). For this parameter daily counts were performed, considering germinated the seeds that presented root protrusion with at least 2.0 mm in length (Juntila, 1976Juntila O. Seed and embryo germination in S. vulgaris and S. reflexa as affected by temperature during seed development. Physiol Plant. 1976;29:264-8.). It was also evaluated the final percentage of germination, considering germinated the seeds that produced seedlings classified as normal (Brasil, 2013). The experiment was considered completed when no germination occurred for three consecutive days (Ferreira and Aquila, 2000Ferreira AG, Aquila MEA. Alelopatia: uma área emergente da ecofisiologia. Rev Bras Fisiol Veg. 2000;12:175-204.).
In bioassays of growth the methodology described by Macias et al. (2000Macias FA, Galindo JC, Castellano D, Velasco RF Sesquiterpene lactones with potencial useas natural herbicides models. 2. guaianolides. J Agric Food Chem. 2000;48(11):5288-96.) was used. After germination in distilled water, having as a criterion the radicle protrusion of at least 2.0 mm in length, 80 seedlings were selected (four replicates of 20 seedlings), for each treatment, being then transferred to gerbox boxes containing the treatment solutions. After five days of installation of the assays, the length of the root and hypocotyl/coleoptile was measured, using a digital pachymeter.
Data Analysis
The data of germination percentage, average germination time and size of seedlings were subjected to analysis of variance, and when significant to polynomial regression analysis with 5% level of significance. The data of germination percentage were transformed into arcsine ?x/100 (Santana and Ranal, 2004Santana DG, Ranal MA. Análise da germinação: um enfoque estatístico. Brasília: UnB; 2004. 248p.).
RESULTS AND DISCUSSION
The phytochemical analysis indicated that the aqueous extracts from fresh leaves (FreshExtH2O) and dried (DriedExtH2O) of torelliodora eucalyptus have the same nine classes of phytochemicals, and as major constituents (100%) for both extracts the phenolic compounds, followed by (75%) anthraquinones. The steroids, triterpenes, cardiotonic heterosides, saponins and reducing sugars were higher in fresh leaves in relation to the dried leaves, except for the flavonoids and tannins (Figure 1).
Class and intensity of secondary metabolites of aqueous extract of fresh leaves (FreshExtH2O) and dried (DriedExtH2O) of torelliodora eucalyptus cultivated in Campo Grande - MS.
In Brazil, the stands of different species of eucalyptus have increased to meet the various segments of the industry nationally and internationally and the adoption of silvicultural practices (IBGE, 2015Instituto Brasileiro de Geografia e Estatística - IBGE. Produção da extração vegetal e silvicultura. Rio de Janeiro: 2015. 48p.). Thus, the demand for more productive and adapted genetic materials to different environmental conditions, led to the process of genetic breeding of eucalyptus. For the leaves of C. citriodora, with the exception of alkaloids, the same classes of secondary metabolites were described (terpenes, phenolic compounds, flavonoids, anthraquinones, steroids, cardiotonic heterosides and saponins) (Javed et al., 2012Javed S, Shoaib A, Mahmood Z, Mushtaq S, Iftikhar S. Analysis of phytochemical constituents of Eucalyptus citriodora L. responsible for antifungal activity against post-harvest fungi. Nat Prod Res. 2012;26(18):1732-6.; Bhagat et al., 2012Bhagat M, Sharma V, Saxena AK. Anti-proliferative effect of leaf extracts of Eucalyptus citriodora against human cancer cells in vitro and in vivo. Indian J Biochem Biophys. 2012;49(6):451-7.) of the hybrid in study, which is the result of hybridization of the species Corymbia citriodora and Eucalyptus torelliana (Figure 1). Considering the alkaloids assays with the aqueous extracts and with the dust of the dried plant are conclusive (Matos, 2009Matos JFA. Introdução a fitoquímica experimental. 2ª. ed. Fortaleza: UFC.; 2009. 150p.; Klein Junior and Henriques, 2017Klein Junior LC, Henriques AT. Alcaloides: generalidades e aspectos básicos. In: Simões CMO, Schenkel EP, Mello JCP, Mentz LA, Petrovick PR, editores. Farmacognosia: da planta ao medicamento. Florianopolis: UFSC; 2010. p.223-6.), it can be argued that this class of allelochemical is not present in the hybrid. Whereas for t Eucalyptus torelliana, in general, regardless of the location of cultivation the species is rich in phenolic compounds and derivatives (tannins, flavonoids, anthraquinones) in addition to the saponins, terpenes and cardiotonic heterosides (Adeniyi and Ayepola, 2008Adeniyi BA, Ayepola OO.The phytochemical screening and antimicrobial activity of leaf extracts of Eucalyptus camaldulensis and Eucalyptus torelliana (Myrtaceae). J Med Plants Res. 2008;2(1):34-8.; Adeniyi et al., 2009; Lawal et al., 2012Lawal TO, Adeniyi BA, Adegoke AO, Franzblau SG, Mahady GB. In vitro susceptibility of Mycobacterium tuberculosis to extracts of Eucalyptus camaldulensis and Eucalyptus torelliana and isolated compounds. Pharm Biol. 2012;50:92-8.; Moses et al., 2017Moses DJ, Albert DD, Uwhomagbejo IR. Gas chromatography-mass spectral structural analysis, phytochemical screening and antimicrobial activity of n-hexane leaf extract of Corymbia torelliana. Inter J Adv Chem.2017;5(1):39-53.), classes found in the investigated hybrid.
The extracts FreshExtH2O and DriedExtH2O when analyzed by spectrophotometry in the UV-Visible region (Figure 2), for confirmation of the classes of phytochemicals (Figure 1) showed a similar profile. The absorption bands around 220 and 225 nm (A) are typical of terpenes (mono and sesquiterpenes) and between 415 nm and 420 nm (G) are characteristic of triterpenes and/or triterpenoid saponins. For the terpenoids unsaturated aliphatic bands are common around 230 nm, since the aromatic compounds are typical bands at intervals of 260 to 280 nm, which may correspond to carboline groups. The absorption at around 440 to 480 nm is characteristic of secondary metabolites as pentacyclic triterpenoids and the saponins (Ugaz, 1994Ugaz O. Investigación fitoquímica: Métodos en el estudio de produtos naturales. Lima: Fondo Editorial de la Pontifícia de la Universidad Católica del Perú; 1994. p.23-42.; Silverstein, 2007Silverstein RM, Webster FX, Kiemle DJ. Identificação espectrométrica de compostos orgânicos. 7ª. ed. Rio de Janeiro: LTC; 2007. 490p.), as detected in the two extracts.
Spectrum-photometric Absorption curve of aqueous extract of fresh leaves (FreshExtH2O) and dried (DriedExtH2O) of torelliodora eucalyptus cultivated in Campo Grande - MS.
The species of the genus Eucalyptus are rich in terpenoids, followed of steroids, polar compounds (Avello et al., 2016Avello M, Fernández Pola, Fernández M, Schulz B, De Diego M, Sigrid M et al. Efecto pediculicida de una formulación en base a Eucaliptus globulus L. Rev Chil Infectol. 2016;33(4):433-7. ). However other classes of constituents were mentioned for different species, as for example, in the hydroalcoholic extract of dried leaves of E. benthamii Maiden et Cambage were described the triterpenes, steroids, tannins and the flavonic and saponinic glycosides (Döll-Boscardin et al., 2010Döll-Boscardin PM, Farago PV, Nakashima T, Santos PET, Paula JFP. Estudo anatômico e prospecção fitoquímica de folhas de Eucalyptus benthamii Maiden et Cambage. Lat Am J Pharm. 2010;29:94-101.). Triterpenes are extremely diversified and of great interest due to the numerous biological activities presented. For the leaves of E. tereticornis two bioactive triterpenes were isolated , ursolic acid and a lactone of ursolic acid (Maurya and Srivastava, 2012Maurya A, Srivastava SK. Determination of ursolic acid and ursolic acid lactone in the leaves of Eucalyptus tereticornis by HPLC. J Braz Chem Soc. 2012;23(2):468-72.), which corroborates the data obtained in this experiment. The allelopathic activity of triterpenes is known, since they act by inhibiting some stages of mitosis and reduce the chlorophyll formation. The saponins , detected in the two extracts, for being a group of substance with part of their molecule hydrophobic and part hydrophilic, has the capacity to interact with cell membranes, modifying the permeability, because of their activity similar to detergent, besides causing hormonal imbalances and act directly on the photosynthetic process (Macías-Rubalcava et al., 2007Macías-Rubalcava ML, Hernández-Bautista BE, Jiménez-Estrada M, Cruz-Ortega R, Anaya AL. Pentacyclic triterpenes with selective bioactivity from Sebastiania adenophora leaves, Euphorbiaceae. J Chem Ecol. 2007;33(1):147-56.; Rizzi et al., 2016Rizzi ES, Pereira KCL, Abreu CAA, Silva BCFL, Fernandes RM, Oliveira AKM, et al. Allelopathic potential and phytochemistry of cambarazinho (Vochysia haenkeana (Spreng.) Mart.) leaves in the germination and development of lettuce and tomato. Biosci J. 2016;32(1):98-107.).
The bands around 280 nm are indicative of aromatic compounds with auxo chromic replacement (280 nm) as substitutes of hydroxyls, such as the phenolic compounds (Ugaz, 1994Ugaz O. Investigación fitoquímica: Métodos en el estudio de produtos naturales. Lima: Fondo Editorial de la Pontifícia de la Universidad Católica del Perú; 1994. p.23-42.; Silverstein, 2007Silverstein RM, Webster FX, Kiemle DJ. Identificação espectrométrica de compostos orgânicos. 7ª. ed. Rio de Janeiro: LTC; 2007. 490p.). The hydroxylated aromatics by conjugation of the benzene ring can move the band around 203.5 nm effectively (primary band ) and secondary band of 254 nm at a longer wavelength, in some cases, the primary band at 250 nm exceeds the secondary and/or stays together with it (Kumar, 2006Kumar S. Organic chemistry: Spectroscopy of organic compounds. Amritsar: Department of Chemistry, Guru Nanak Dev University; 2006. 36p.), as observed in the bands B (250 - 270 nm) and C (260 - 280 nm) (Figure 2).
The phenolic compounds and derivatives, flavonoids and tannins, are also common in species of the genus, specifically in the barks of E. grandis and E. pellita, tannins were found and in leaves of E. consideniana, E. viminalis, E. ovata, besides the tannins the glucoflavonoids (Tuffi Santos et al., 2005Tuffi Santos LD, Ferreira FA, Meira RMSA, Barros NF, Ferreira LR, Machado AFL. Crescimento e morfoanatomia foliar de eucalipto sob efeito de deriva do glyphosate. Planta Daninha. 2005;23(1);133-42.), and on the leaves of E. grandis and E. urophylla GL9, also the phenolic and flavonoid compounds (Chen et al., 2014Chen Y, Wang J, Ou Y, Chen H, Xiao S, Liu G, et al. Cellular antioxidant activities of polyphenols isolated from Eucalyptus leaves (Eucalyptus grandis × Eucalyptus urophylla GL9). J Funct Foods. 2014;7:737-45. ). On mature leaves of E. globulus Labill., the allelopathic activity on mustard (Brassica campestris L.), cabbage (B. oleracea L. cv. capitata), broccoli (B. oleracea L. cv. italica), kale (B. pekinensis L.), lettuce (Lactuca sativa L. cv. grand rapids), tomato (Lycopersicum esculentum Miller), turnip (B. rapa L.), arugula (Eruca sativa L.) and radish (Raphanus sativus L.) was attributed to the phenolic compounds such as Eucalyptol, tannins and the monoterpenes (Yamagushi et al., 2011Yamagushi MQ, Gusman GS, Vestena S. Efeito alelopático de extratos aquosos de Eucalyptus globulus Labill. e de Casearia sylvestris Sw. sobre espécies cultivadas. Semina: Cienc Agrar 2011;32(4):1361-74.). In general, the allelochemicals released by green leaves or when leached of dried leaves, among them the phenolic compounds and derivatives, specifically the simple phenols, such as phenolic acids, act on the membrane permeability, inhibiting the growth of the aerial part and root elongation (Rice, 1984Rice EL. Allelopathy. New York: Academic Press; 1984. 422p.; Ferrarese et al., 2000Ferrarese MLL, Ferrarese-Filho O, Rodrigues JD. Ferulic acid uptake by soybean root in nutrient culture. Acta Physiol Plant. 2000;22(2):121-4.), in addition to blocking the mitochondrial respiration (Weir, 2004Weir TL, Park S-W, Vivanco JM. Biochemical and physiological mechanisms mediated by allelochemicals. Curr Opin Plant Biol. 2004;7(4):472-9.) or still by a combination of these factors (Ferreira and Aquila, 2000Ferreira AG, Aquila MEA. Alelopatia: uma área emergente da ecofisiologia. Rev Bras Fisiol Veg. 2000;12:175-204.).
The absorption bands between 330 nm to 350 nm (D, E and F) are indicative of flavonoids. The flavonoids present in the ultraviolet, two maximum bands of absorption, one between 240-285 nm, band corresponding to ring A (hemicetal), and another between 300-400 nm, referring to t ring B of flavonoids, in general the two absorptions are relative to the ring A of the benzoyl system and the band I to the ring B, cinamoil System (Mabry et al., 1970Mabry TJ, Markham KR, Thomas MB. The systematic identification of Flavonoids. Austin: University of Texas; 1970.; Zuanazzi et al., 2017). Among the most often cited polyphenols as natural herbicides, due to their hydro soluble characteristics and released in natural conditions, there are the flavonoids that may affect directly and indirectly the permeability of the mitochondria membrane and the chloroplast (Moreland and Novitzky, 1987Moreland DE, Novitzky WP. Effects of phenolic acids, coumarins, and flavonoids on isolated chloroplasts and mitochondria. ACS Symp Ser. 1987;330:247-61.). The tannins, another group of polyphenols, act in the process of cell division (Rice, 1984Rice EL. Allelopathy. New York: Academic Press; 1984. 422p.; Ferreira and Aquila, 2000Ferreira AG, Aquila MEA. Alelopatia: uma área emergente da ecofisiologia. Rev Bras Fisiol Veg. 2000;12:175-204.).
The maximum absorptions at 445 nm are indicative to aromatic compounds condensed with conjunction with the carboxyl group and carboxylic related to anthraquinones (Figure 2). The quinones are common, by conjugate pairs present in their structure, absorption in the ultraviolet/visible region (Falkenberg, 2010Falkenberg MB. Quinonas. In: Simões CMO, Schenkel EP, Mello JCP, Mentz LA, Petrovick PR. Farmacognosia: da planta ao medicamento. Florianopolis: UFSC; 2010. p.254-255.), with maximum absorption at 480 nm (Silverstein et al., 2007Silverstein RM, Webster FX, Kiemle DJ. Identificação espectrométrica de compostos orgânicos. 7ª. ed. Rio de Janeiro: LTC; 2007. 490p.). The anthraquinones are not common in species of the genus Eucalyptus, however Adeniyi et al. (2009Adeniyi CBA, Lawal TO, Mahady GB. In vitro susceptibility of Helicobacter pylori to extracts of Eucalyptus camaldulensis and Eucalyptus torelliana. Pharm Biol. 2009;47(1):99-102.) detected in the ethanolic crude extract of the leaves of E. torelliana F. Muell., in addition to the glycosides, tannins, saponins and cardiotonic heterosides, this extract was active for different strains of Helicobacter pylori. Whereas for the aqueous extract and ethanol extracts of the leaves of E. camaldulensis, Sani et al. (2014Sani I, Abdulhamid A., Bello F. Eucalyptus camaldulensis: phytochemical composition of ethanolic and aqueous extracts of the leaves, stem-bark, root, fruits, and seeds. J Sci Innov Res. 2014;3(5):523-6.) evidenced the anthraquinones and glycosides in the aqueous extract, in the two extracts the tannins, saponins and steroids and alkaloids only in the ethanolic extract. All this information corroborates the phytochemicals results obtained in the present study, for the aqueous extract of the leaves extract FreshExtH2O and DriedExtH2O of tolleriodora eucalyptus and in particular for the class of anthraquinones, that were only found in some species of Eucalyptus.
The content of phenolic compounds (mg of gallic acid/g of sample) was statistically equal to Extract FreshExtH2O and DriedExtH2O, whereas for the flavonoids the behavior differed between the extracts, with higher values for the extract DriedExtH2O (Table 1). The values of phenolic compounds were lower than those found in the aqueous extract of E. globulus (235.87 ± 4.38 mg gallic acid. g-1) and superior to contents of flavonoids (35.76 ± 0.95 mg rutin g-1) (Dezsi et al., 2015Dezsi‘, Bãdãrãu AS, Bischin C, Vodnar DC, Silaghi-Dumitrescu R, Gheldiu AM, et al. Antimicrobial and antioxidant activities and phenolic profile of Eucalyptus globulus Labill. and Corymbia ficifolia (F. Muell.) KD Hill & LAS Johnson leaves. Molecules. 2015;20(3):4720-34.).
As pointed out earlier the flavonoids are one of the most investigated class of allelochemicals. They act negatively on the permeability of the mitochondria membrane and of the chloroplast (Moreland and Novitzky, 1987Moreland DE, Novitzky WP. Effects of phenolic acids, coumarins, and flavonoids on isolated chloroplasts and mitochondria. ACS Symp Ser. 1987;330:247-61.) and inhibiting the polar transport of auxin (IAA) and in the degradation of the IAA oxidases and peroxidases; with this, there are changes in the plant growth and its ability to compete in the environment. However, depending on the metabolic sub-class and structure, they act positively in regulating the plant development, as modulars of auxin polar transport (Taiz and Zeiger, 2009Taiz L, Zeiger E. Fisiologia vegetal. 5ª. ed. Porto Alegre: Artmed; 2009. p.321-3.). Another important feature given to some groups of flavonoids is the affinity for many enzymes, proteins, in addition to acting on the inhibition of NADH oxidase and the balance of reactive oxygen species, altering the mitochondrial respiration and affecting the plants development (Weston and Mathesius, 2013Weston LA, Mathesius U. Flavonoids: their structure, biosynthesis and role in the rhizosphere, including allelopathy. J Chem Ecol. 2013;39:283-97.).
Although the mechanism of action by which these molecules participate in allelopathy remains unknown, it is a fact that the positive or negative interactions plant-plant may depend on the concentrations of flavonoids (Mierziak et al., 2014Mierziak J, Kostyn K, Kulma A. Flavonoids as important molecules of plant interactions with the environment. Molecules. 2014;19(10):16240-65.). According to Levizou et al. (2004Levizou E, Karageorgou P, Petropoulou Y, Grammatikopoulos G, Manetas Y. Induction of ageotropic response in lettuce radical growth by epicuticular flavonoid aglycones of Dittrichia viscosa. Biol Plant. 2004;48(2):305-7.) the allelopathic activity of many flavonoids is dependent on the dose and the authors suggest that this is highly dependent on the concentration; some of these compounds can be inhibitory or stimulant, depending on the availability, concentration in soil/water solution in the rhizosphere and its impact on growth and morphology of the roots.
The results of pH, electrical conductivity and soluble solids of extracts FreshExtH2O and DriedExtH2O (Table 1) indicated that these physical-chemical characteristics did not affect negatively the processes of germination and seedling growth. According to Rice (1984Rice EL. Allelopathy. New York: Academic Press; 1984. 422p.) and Ferreira and Aquila (2000Ferreira AG, Aquila MEA. Alelopatia: uma área emergente da ecofisiologia. Rev Bras Fisiol Veg. 2000;12:175-204.) only in extreme conditions of alkaline pH, exceeding 11, and acid pH less than 3, the allelopathic tests are affected. The pH of the extracts did not differ statistically and are in the range of 4.5, compatible with the information above, not influencing the development of the tested species (Table 1). Regarding the pH values obtained for the extract FreshExtH2O and DriedExtH2O similar data were also obtained by Borella et al. (2009Borella J, Wandscheer ACD, Bonatti LC, Hernandez Pastorini L. Efeito alelopático de extratos aquosos de Persea americana Mill. sobre Lactuca sativa L. Rev Bras Biocienc. 2009;7(3):260-5.), for the aqueous extracts of fresh leaves (pH of 5.51 - 5.61) and dried (pH of 5.43 - 6.07) of Persea americana Mill (avocado tree). The authors also attribute to the flavonoids and tannins the allelopathic activity of fresh and dried leaves of avocado tree.
The electrical conductivity, expressed by the number of positive and negative electrolyte solution, directly influences the germination of seeds, once that elevated concentrations of electrolytes reflect the loss of integrity of the cell membrane. The values obtained for both extracts did not differ statistically and are out of bounds harmful to germination and seedlings development (Table 1). For Souza (1999Souza CLM, Morais V, Silva ER, Lopes HM, Tozani R, Parraga MS, et al. Efeito inibidor dos extratos hidroalcóolicos de coberturas mortas sobre a germinação de sementes de cenoura e alface. Planta Daninha. 1999;17(2):263-72.), values lower than 200 µS cm-1 do not cause deleterious effects on germination and vigor of seeds.
The soluble solids are employed in tests of allelopathy as a way of estimating the osmotic potential of the investigated extracts and represent a measure of reducing and non-reducing sugars and other solids diluted in solution. The values of Brix differed among themselves for the two extracts, being higher for the fresh leaves (FreshExtH2O). On the fresh leaves the water contributes to ions solubilization and consequently affects the percentage of soluble solids in relation to the dried leaves (DriedExtH2O). However, the values of soluble solids are within the values reported by Oliveira et al. (2013Oliveira AKM, Ribeiro JWF, Fontoura FM, Matias R. Leaf extracteffects of Vochysia divergens on lettuce and tomato. Allelopathy J. 2013;31:129-38.) that was 0.8 (oBrix at 20 oC), Oliveira et al. (2014Oliveira AKM, Pereira KCL, Muller JA, Matias R. Análise fitoquímica e potencial alelopático das cascas de Pouteria ramiflora na germinação de alface. Hortic Bras. 2014;32:41-7.) 0.3 (oBrix at 20 oC) and by Rizzi et al. (2016Rizzi ES, Pereira KCL, Abreu CAA, Silva BCFL, Fernandes RM, Oliveira AKM, et al. Allelopathic potential and phytochemistry of cambarazinho (Vochysia haenkeana (Spreng.) Mart.) leaves in the germination and development of lettuce and tomato. Biosci J. 2016;32(1):98-107.), that point out that values tending to zero do not interfere with the osmotic potential and are not able to mask the allelopathic effect of the investigated extracts.
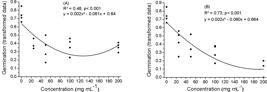
The germination percentage was not influenced by the extracts FreshExtH2O and DriedExtH2O of the leaves of torelliodora eucalyptus. For the extract FreshExtH2O, it was observed a reduction in the germination percentage until the concentration of 127.5 mg mL-1, being that above this the reduction of the allelochemicals action is evidenced (Figure 3A). The same was observed for dried leaves, however the negative influence on germination occurred up to a higher concentration of 190.8 mg mL-1 (Figure 3B). Correlating these results with the phytochemistry, it is verified that the dehydration, between 20 and 40 oC, favored an increase in the levels of flavonoids and tannins (Figure 1 and Table 1) in relation to the fresh plant material. Therefore, these allelochemicals present on dried leaves, may be released in higher concentrations with the decomposition of their residues or other processes release the constituents to the soil after the fall of the same, limiting more strongly the germination of native species.
Effect of different concentrations of aqueous extracts of torelliodora eucalyptus on germination of Guazuma ulmifolia: (A) fresh leaves and (B) dried leaves.
Similarly, the Average Germination Time (AGT) was influenced by the eucalyptus extract ExtFreshH2O (Figure 4), being that all the evaluated concentrations influenced negatively the AGT, resulting in an increase in the germination time. However, this relationship was not linear, being that after the concentration of 13.3% a reduction was observed of the allelochemicals action (Figure 4). The extracts ExtDriedH2O of Eucalyptus did not influence AGT of G. ulmifolia. Concerning the initial seedling growth, the increase in the concentration of the extract ExDriedH2O of the eucalyptus affected negatively the initial growth both of the hypocotyl (Figure 5A) and the primary root of seedlings of G. ulmifolia (Figure 5B). However, the extract ExtFreshH2O did not influence the growth of seedlings of native species.
Effect of different concentrations of the extract of fresh leaves of Eucalyptus torelliodora at the Average Germination Time of Guazuma ulmifolia.
Effect of different concentrations of extract of dried leaves of torelliodora eucalyptus on initial growth of seedlings of Guazuma ulmifolia: (A) hypocotyl length and (B) radicle length.
The allelopathy of eucalyptus has been considered an important mechanism of reduction of local biodiversity under pure crops involving the different species (Zhang et al., 2010Zhang D, Zhang J, Wu F-Z. Potential allelopathic effect of Eucalyptus grandis across a range of plantation ages. Ecol Res. 2010;25:13-23.). However, little is known if this relationship would be maintained in the intercropped plantations with native species, as in the case allowed by the New Forest Code for the restoration of Legal Reserve Areas (Brasil, 2012). In this new situation, the exotic species could interfere with both the initial growth of seedlings of reintroduced native species, and in the germination and establishment of individuals originated from seeds, either by direct seeding, seed dispersal of adult individuals already established over time in the area or from fragments close to the landscape.
The results found in the present study, however, did not show a linear relationship between the reduction in the percentage and average time of germination in relation to the increase in the extract concentration. In general, it is expected that the allelochemicals actions have fluctuations due to the concentrations; however, if their absorption and distribution were linear and instant investigations would not be necessary of different concentrations, which is usual in studies evaluating the allelopathic potential of different species and investigation with new herbicides. In the case of herbicides, the active principle has a long half-life and the dose indicated for each target species. Thus, it is possible to infer that from a certain concentration the effect of allelochemicals present in the extracts are reduced or canceled.
The increase in the concentration of allelochemicals originated from leaves of Eucalyptus plantations on the soil, when in the field, will be due to the increase in the deposition of leaves on the same, being related to the increase of the size/age of individuals of exotic species. Zhang et al. (2010Zhang D, Zhang J, Wu F-Z. Potential allelopathic effect of Eucalyptus grandis across a range of plantation ages. Ecol Res. 2010;25:13-23.) analyzed the allelopathic potential of Eucalyptus grandis in the germination and growth of three species (Raphanus sativus, Phaseolus aureus and Lolium perenne) growing near or under eucalyptus plantations with different ages. Their results showed a strong inhibitory effect on rates of germination of the evaluated species in young plantations; however, this effect is reduced with the age increase of the planting of the tree species. According to the authors, the found results suggest that the soil can inhibit or dilute the allelopathic agents with the increase of the age of the planting.
This information seem to converge with studies that assessed the regeneration of Brazilian native species in plantations of exotic species. Abreu et al. (2011Abreu RCR, Assis GB, Frison S, Aguirre A, Durigan G. Can native vegetation recover after slash pine cultivation in the Brazilian Savanna? For Ecol Manage.2011;262(8):1452-9. ) found a greater diversity of Cerrado natives, including endemic species, under stands of Pinus approximately 45 years of age than in reference areas of Cerradão, suggesting that the plantations of exotic species can be a good starting point for the savannas restoration. Carmo et al. (2012Carmo FMS, Poeiras LM, Gonçalves AB, Mello SM, Meira Neto JAAlves, Borges EEL, Silva AF. Germinação do banco de sementes de espécies nativas sob dossel de espécies exóticas. Rev Árvore. 2012;36(4):583-91.) evaluated the germination of the seed bank of a Seasonal Semideciduous Forest under the canopy of Pinus and Eucalyptus, and concluded that the low density and diversity of plants often found in the understorey of these species are not directly related with suppressive effects promoted by the presence of these, among them the allelopathy.
Oliveira et al. (2011bOliveira EB, Sousa LP, Radomski MI. Regeneração natural em sub-bosque de Corymbia citriodora no noroeste do Estado do Paraná. Rev Floresta. 2011b;41(2):377-86.) evaluated the potential of recomposition of native vegetation in understory of regrowth of Corymbia citriodora with seven years in the northwestern region of Paraná. These authors found a high potential of natural regeneration and growth of vegetation in the understory of exotic species, indicating that, with proper management, this species can be used as a facilitator for the purposes of restoration of native vegetation. Thus, a strategy for the reintroduction of native species in consortium with exotic species could be prior planting of exotics and subsequent reintroduction of the natives, when the allelochemicals action could be reduced or neutralized on the soil.
The phytochemical analysis indicated that the phenolic compounds and anthraquinones are present in aqueous extracts of fresh and dried leaves of tolleriodora eucalyptus with the same intensity, whereas flavonoids and tannins were higher on the leaves for the dried leaves and for the fresh leaves the steroids, triterpenes, cardiotonic heterosides, saponins and reducing sugars were higher on fresh leaves.
Dried and fresh leaves extracts of torelliodora eucalyptus influenced the G. ulmifolia germination, but this effect was dependent on the extract concentration.
Seedlings initial growth of G. ulmifolia was negatively influenced only the dried leaves extract or the exotic species.
REFERENCES
- Abreu RCR, Assis GB, Frison S, Aguirre A, Durigan G. Can native vegetation recover after slash pine cultivation in the Brazilian Savanna? For Ecol Manage.2011;262(8):1452-9.
- Adeniyi BA, Ayepola OO.The phytochemical screening and antimicrobial activity of leaf extracts of Eucalyptus camaldulensis and Eucalyptus torelliana (Myrtaceae). J Med Plants Res. 2008;2(1):34-8.
- Adeniyi CBA, Lawal TO, Mahady GB. In vitro susceptibility of Helicobacter pylori to extracts of Eucalyptus camaldulensis and Eucalyptus torelliana Pharm Biol. 2009;47(1):99-102.
- Avello M, Fernández Pola, Fernández M, Schulz B, De Diego M, Sigrid M et al. Efecto pediculicida de una formulación en base a Eucaliptus globulus L. Rev Chil Infectol. 2016;33(4):433-7.
- Bedin C, Mendes LB, Trecente VC, Silva JMS. Efeito Alelopático de extrato de Eucalyptus citriodora na germinação de sementes de tomate (Lycopersicum esculentum M.). R Cient Eletr Agron. 2006;5:1-7.
- Bhagat M, Sharma V, Saxena AK. Anti-proliferative effect of leaf extracts of Eucalyptus citriodora against human cancer cells in vitro and in vivo. Indian J Biochem Biophys. 2012;49(6):451-7.
- Borella J, Wandscheer ACD, Bonatti LC, Hernandez Pastorini L. Efeito alelopático de extratos aquosos de Persea americana Mill. sobre Lactuca sativa L. Rev Bras Biocienc. 2009;7(3):260-5.
- Brancalion PHS, Garcia LC, Loyola R, Rodrigues RR, Pillar VD, Lewinsohn TM. Análise crítica da Lei de Proteção da Vegetação Nativa (2012), que substituiu o antigo Código Florestal: atualizações e ações em curso. Nat Conserv. 2016;14:1-16.
- Brasil. Lei 12.651, de 25 de maio de 2012. Dispõe sobre a proteção da vegetação nativa; altera as Leis nos 6.938, de 31 de agosto de 1981, 9.393, de 19 de dezembro de 1996, e 11.428, de 22 de dezembro de 2006; revoga as Leis nos 4.771, de 15 de setembro de 1965, e 7.754, de 14 de abril de 1989, e a Medida Provisória no 2.166- 67, de 24 de agosto de 2001; e dá outras providências. Diário Oficial da República Federativa do Brasil, Brasília, DF, 25 maio de 2012.
- Brasil. Ministério da Agricultura, Pecuária e Abastecimento. Instruções para análise de sementes florestais. Brasília: 2013. 97p.
- Carmo FMS, Poeiras LM, Gonçalves AB, Mello SM, Meira Neto JAAlves, Borges EEL, Silva AF. Germinação do banco de sementes de espécies nativas sob dossel de espécies exóticas. Rev Árvore. 2012;36(4):583-91.
- Chen Y, Wang J, Ou Y, Chen H, Xiao S, Liu G, et al. Cellular antioxidant activities of polyphenols isolated from Eucalyptus leaves (Eucalyptus grandis × Eucalyptus urophylla GL9). J Funct Foods. 2014;7:737-45.
- Dezsi‘, Bãdãrãu AS, Bischin C, Vodnar DC, Silaghi-Dumitrescu R, Gheldiu AM, et al. Antimicrobial and antioxidant activities and phenolic profile of Eucalyptus globulus Labill. and Corymbia ficifolia (F. Muell.) KD Hill & LAS Johnson leaves. Molecules. 2015;20(3):4720-34.
- Döll-Boscardin PM, Farago PV, Nakashima T, Santos PET, Paula JFP. Estudo anatômico e prospecção fitoquímica de folhas de Eucalyptus benthamii Maiden et Cambage. Lat Am J Pharm. 2010;29:94-101.
- Falkenberg MB. Quinonas. In: Simões CMO, Schenkel EP, Mello JCP, Mentz LA, Petrovick PR. Farmacognosia: da planta ao medicamento. Florianopolis: UFSC; 2010. p.254-255.
- Ferrarese MLL, Ferrarese-Filho O, Rodrigues JD. Ferulic acid uptake by soybean root in nutrient culture. Acta Physiol Plant. 2000;22(2):121-4.
- Ferreira AG, Aquila MEA. Alelopatia: uma área emergente da ecofisiologia. Rev Bras Fisiol Veg. 2000;12:175-204.
- Florentine SK, Fox JED. Allelopathic effects of Eucalyptus victrix L. on Eucalyptus species and grasses. Allelopathy J. 2003;11:77-83.
- Fontoura FM, Matias R, Ludwig J, Oliveira AKM, Bono JAMaior, Martins PFRB, et al. Seasonal effects and antifungal activity from bark chemical constituents of Sterculia apetala (Malvaceae) at Pantanal of Miranda, Mato Grosso do Sul, Brazil. Acta Amaz. 2015;45(3):283-92.
- Goetze M, Thomé G. Efeito alelopático de extratos de Nicotiana tabacum e Eucalyptus grandis sobre a germinação de três espécies de hortaliças. Rev Bras Agrocienc. 2004;10:43-50.
- Instituto Brasileiro de Geografia e Estatística - IBGE. Produção da extração vegetal e silvicultura. Rio de Janeiro: 2015. 48p.
- Javed S, Shoaib A, Mahmood Z, Mushtaq S, Iftikhar S. Analysis of phytochemical constituents of Eucalyptus citriodora L. responsible for antifungal activity against post-harvest fungi. Nat Prod Res. 2012;26(18):1732-6.
- Juntila O. Seed and embryo germination in S. vulgaris and S. reflexa as affected by temperature during seed development. Physiol Plant. 1976;29:264-8.
- Klein Junior LC, Henriques AT. Alcaloides: generalidades e aspectos básicos. In: Simões CMO, Schenkel EP, Mello JCP, Mentz LA, Petrovick PR, editores. Farmacognosia: da planta ao medicamento. Florianopolis: UFSC; 2010. p.223-6.
- Krzyzanowski FC, Vieira RD, França Neto JB. Vigor de sementes: conceitos e testes. Londrina: ABRATES; 1999. 218p.
- Kumar S. Organic chemistry: Spectroscopy of organic compounds. Amritsar: Department of Chemistry, Guru Nanak Dev University; 2006. 36p.
- Lawal TO, Adeniyi BA, Adegoke AO, Franzblau SG, Mahady GB. In vitro susceptibility of Mycobacterium tuberculosis to extracts of Eucalyptus camaldulensis and Eucalyptus torelliana and isolated compounds. Pharm Biol. 2012;50:92-8.
- Levizou E, Karageorgou P, Petropoulou Y, Grammatikopoulos G, Manetas Y. Induction of ageotropic response in lettuce radical growth by epicuticular flavonoid aglycones of Dittrichia viscosa Biol Plant. 2004;48(2):305-7.
- Mabry TJ, Markham KR, Thomas MB. The systematic identification of Flavonoids. Austin: University of Texas; 1970.
- Macias FA, Galindo JC, Castellano D, Velasco RF Sesquiterpene lactones with potencial useas natural herbicides models. 2. guaianolides. J Agric Food Chem. 2000;48(11):5288-96.
- Macías-Rubalcava ML, Hernández-Bautista BE, Jiménez-Estrada M, Cruz-Ortega R, Anaya AL. Pentacyclic triterpenes with selective bioactivity from Sebastiania adenophora leaves, Euphorbiaceae. J Chem Ecol. 2007;33(1):147-56.
- Matos JFA. Introdução a fitoquímica experimental. 2ª. ed. Fortaleza: UFC.; 2009. 150p.
- Maurya A, Srivastava SK. Determination of ursolic acid and ursolic acid lactone in the leaves of Eucalyptus tereticornis by HPLC. J Braz Chem Soc. 2012;23(2):468-72.
- May FE, Ash JE. An assessment of the Allelopathic Potential of Eucalyptus Aust J Bot. 1990;38:245-54.
- Mierziak J, Kostyn K, Kulma A. Flavonoids as important molecules of plant interactions with the environment. Molecules. 2014;19(10):16240-65.
- Moreland DE, Novitzky WP. Effects of phenolic acids, coumarins, and flavonoids on isolated chloroplasts and mitochondria. ACS Symp Ser. 1987;330:247-61.
- Moses DJ, Albert DD, Uwhomagbejo IR. Gas chromatography-mass spectral structural analysis, phytochemical screening and antimicrobial activity of n-hexane leaf extract of Corymbia torelliana Inter J Adv Chem.2017;5(1):39-53.
- Oliveira AKM, Pereira KCL, Muller JA, Matias R. Análise fitoquímica e potencial alelopático das cascas de Pouteria ramiflora na germinação de alface. Hortic Bras. 2014;32:41-7.
- Oliveira AKM, Ribeiro JWF, Fontoura FM, Matias R. Leaf extracteffects of Vochysia divergens on lettuce and tomato. Allelopathy J. 2013;31:129-38.
- Oliveira AKM, Ribeiro JWF, Matias R, Gusmão DDH, Pereira KCL. Potencial alelopático de folhas frescas de bacupari (Rheedia brasiliensis (Mart.) Planch. & Triana) na germinação de alface. Rev Bras Biocienc. 2011a;4(9):550-3.
- Oliveira APP, Pereira SR, Cândido ACS, Laura VA Peres MTLP. Can allelopathic grasses limit seed germination and seedling growth of Mutambo? A test with two species of Brachiaria grasses. Planta Daninha. 2016;34(4):639-48.
- Oliveira EB, Sousa LP, Radomski MI. Regeneração natural em sub-bosque de Corymbia citriodora no noroeste do Estado do Paraná. Rev Floresta. 2011b;41(2):377-86.
- Peixoto Sobrinho TJS, Silva CHTP, Nascimento JE, Monteiro JM, Albuquerque UP, Amorim ELC. Validação de metodologia espectrofotométrica para quantificação dos flavonóides de Bauhinia cheilantha (Bongard) Steudel. Rev Bras Cienc Farm. 2008;44(4):683-9.
- Rice EL. Allelopathy. New York: Academic Press; 1984. 422p.
- Rizzi ES, Pereira KCL, Abreu CAA, Silva BCFL, Fernandes RM, Oliveira AKM, et al. Allelopathic potential and phytochemistry of cambarazinho (Vochysia haenkeana (Spreng.) Mart.) leaves in the germination and development of lettuce and tomato. Biosci J. 2016;32(1):98-107.
- Sani I, Abdulhamid A., Bello F. Eucalyptus camaldulensis: phytochemical composition of ethanolic and aqueous extracts of the leaves, stem-bark, root, fruits, and seeds. J Sci Innov Res. 2014;3(5):523-6.
- Santana DG, Ranal MA. Análise da germinação: um enfoque estatístico. Brasília: UnB; 2004. 248p.
- Sasikumar K. Allelopathic effects ofeucalyptus on blackgram (Phaseolus mungo L.). Allelopathy J. 2002;9:205-14.
- Schenkel EP, Gosmann G, Athayde ML. Saponinas. In: Simões CMO, Schenkel EP, Mello JCP, Mentz LA, Petrovick PR. editores. Farmacognosia: da planta ao medicamento. Florianopolis: UFSC; 2010. p.711-34.
- Silverstein RM, Webster FX, Kiemle DJ. Identificação espectrométrica de compostos orgânicos. 7ª. ed. Rio de Janeiro: LTC; 2007. 490p.
- Sousa CMM, Silva HR, Vieira-Jr GM, Ayres MCC, Costa CLS, Araújo DS, et al. Fenóis totais e atividade antioxidante de cinco plantas medicinais. Quím Nova. 2007;30:351-5.
- Souza CLM, Morais V, Silva ER, Lopes HM, Tozani R, Parraga MS, et al. Efeito inibidor dos extratos hidroalcóolicos de coberturas mortas sobre a germinação de sementes de cenoura e alface. Planta Daninha. 1999;17(2):263-72.
- Taiz L, Zeiger E. Fisiologia vegetal. 5ª. ed. Porto Alegre: Artmed; 2009. p.321-3.
- Tuffi Santos LD, Ferreira FA, Meira RMSA, Barros NF, Ferreira LR, Machado AFL. Crescimento e morfoanatomia foliar de eucalipto sob efeito de deriva do glyphosate. Planta Daninha. 2005;23(1);133-42.
- Ugaz O. Investigación fitoquímica: Métodos en el estudio de produtos naturales. Lima: Fondo Editorial de la Pontifícia de la Universidad Católica del Perú; 1994. p.23-42.
- Weir TL, Park S-W, Vivanco JM. Biochemical and physiological mechanisms mediated by allelochemicals. Curr Opin Plant Biol. 2004;7(4):472-9.
- Weston LA, Mathesius U. Flavonoids: their structure, biosynthesis and role in the rhizosphere, including allelopathy. J Chem Ecol. 2013;39:283-97.
- Yamagushi MQ, Gusman GS, Vestena S. Efeito alelopático de extratos aquosos de Eucalyptus globulus Labill. e de Casearia sylvestris Sw. sobre espécies cultivadas. Semina: Cienc Agrar 2011;32(4):1361-74.
- Zhang D, Zhang J, Wu F-Z. Potential allelopathic effect of Eucalyptus grandis across a range of plantation ages. Ecol Res. 2010;25:13-23.
- Zuanazzi JAS, Montanha JA, Zucolotto SM. Flavonoides. In: Simões CMO, Schenkel EP, Mello JCP, Mentz LA, Petrovick PR, editores. Farmacognosia: do produto natural ao medicamento. Porto Alegre: Artmed; 2017. p.209-33.
Publication Dates
-
Publication in this collection
2018
History
-
Received
26 Mar 2018 -
Accepted
15 June 2018