Abstract
The present work characterized and compared the anatomical structures of the leaves of Bactris gasipaes (Arecaceae) plants grown under different cultivation conditions (in vitro, ex vitro and in vivo) with the goal of identifying the origins of the difficulties encountered in acclimatizing micro-plants. The Quant program was used to determine leaf tissue thicknesses and areas, and histochemical tests were performed on leaf sections and analyzed using light microscopy. Stomatal and trichome densities were determined using the epidermal impression method and by scanning electronic microscopy. Our results indicated that there were no discernible alterations of the anatomical characteristics of the leaves of micro-plants cultivated under differing conditions and that the thickening of the mesophyll and the vascular fibers indicated adaptive responses to ex vitro conditions. As such, the observed difficulties in acclimatizing peach palm micro-plants to ex vitro conditions cannot be attributed to plant anatomical characteristics acquired during in vitro cultivation.
acclimatization; adaptation; plant anatomy
STRUCTURAL BOTANY
Anatomical analysis of peach palm (Bactris gasipaes) leaves cultivated in vitro, ex vitro and in vivo11. Part of the master's dissertation of the first author, Programa de Pós-Graduação em Fisiologia e Bioquímica Vegetal, Universidade de São Paulo, Escola Superior de Agricultura "Luiz de Queiroz", Piracicaba, SP, Brazil.
Katherine Derlene Batagin-PiottoI,III,22. Corresponding author: kabatagin@hotmail.com; Cristina Vieira de AlmeidaII; Fernando Angelo PiottoIII; Marcílio de AlmeidaI
IUniversidade de São Paulo, Departamento de Ciências Biológicas, Escola Superior de Agricultura "Luiz de Queiroz", Avenida Pádua Dias, 11, 13418-900 Piracicaba, SP, Brazil
IIInVitroPalm Consultoria, Estudo e Desenvolvimento Biológico Ltda, Rua Itajobi, 421, Colinas do Piracicaba, 13432-566 Piracicaba, SP, Brazil
IIIUniversidade de São Paulo, Departamento de Genética, Escola Superior de Agricultura "Luiz de Queiroz", Avenida Pádua Dias, 11, 13418-90 Piracicaba, SP, Brazil
ABSTRACT
The present work characterized and compared the anatomical structures of the leaves of Bactris gasipaes (Arecaceae) plants grown under different cultivation conditions (in vitro, ex vitro and in vivo) with the goal of identifying the origins of the difficulties encountered in acclimatizing micro-plants. The Quant program was used to determine leaf tissue thicknesses and areas, and histochemical tests were performed on leaf sections and analyzed using light microscopy. Stomatal and trichome densities were determined using the epidermal impression method and by scanning electronic microscopy. Our results indicated that there were no discernible alterations of the anatomical characteristics of the leaves of micro-plants cultivated under differing conditions and that the thickening of the mesophyll and the vascular fibers indicated adaptive responses to ex vitro conditions. As such, the observed difficulties in acclimatizing peach palm micro-plants to ex vitro conditions cannot be attributed to plant anatomical characteristics acquired during in vitro cultivation.
Key-words: acclimatization, adaptation, plant anatomy
INTRODUCTION
Palm hearts are largely obtained from illegal harvests of wild Euterpe oleracea Mart. and E. edulis Mart. plants, which threaten the survival of this species. As result, more ecologically correct manners of commercially cultivating peach palms (Bactris gasipaes Kunth Arecaceae) have been expanding, aided by the ease of cultivation of this plant, and its rapid growth and ability to grow in direct sunlight.
Nonetheless, many difficulties have been encountered during attempts to expand plantings of this cultivar (such as the lack of ready availability of seeds from selected stocks, which results in heterogeneous plantations with varying numbers of shoots, the presence of spines, irregular stem diameters, and low productivity and quality of the palm hearts) in addition to the fact that peach palm seeds are recalcitrant, which makes this form of propagation more difficult (Bovi et al. 2004).
Micro-propagation may represent a viable option for reproducing this palm in light of the difficulties encountered in conventional propagation (Bunn et al. 2007), allowing extensive propagation of these plants as well as the cloning of selected stocks.
Although a number of workers have investigated the in vitro multiplication of this species, including Almeida & Kerbauy (1996), Almeida et al. (2005), Almeida & Almeida (2006), Steinmacher et al. (2007), micro-propagated seedlings are not easily encountered on a commercial scale due to high losses during their transfer to ex vitro conditions.
Few workers have investigated acclimation of palm trees, and even fewer yet of peach palms. Studies examining the anatomy of this species, especially its leaves, may be extremely important in acclimation research as leaf structure is highly responsive to environmental modifications and leaves are the principal sites of plant metabolite production (Taiz & Zeiger 2006).
Within this context, the present work examined if there are of anatomical alterations in the leaves of micro-plants of peach palms when they were transferred to ex vitro environments. The leaves of plants that had been cultivated under in vitro, ex vitro and in vivo conditions were analyzed and compared to determine the existence of alterations that could be related to the difficulties encountered in acclimatizing this species.
MATERIAL AND METHODS
The treatments consisted of raising peach palm plants under three different cultivation conditions (in vitro, ex vitro and in vivo) using 50 micro-plants of Bactris gasipaes that had been cultivated in vitro and 50 seedlings grown from seed (in vivo). Both the seeds and the stem apices were derived from selected matrixes from Yurimáguas (Peruvian Amazon).
In vitro culture conditions
Micro-plants of B. gasipaes derived from stem apices were cultivated in test tubes containing 10 mL of Murashige and Skoog (1962) culture medium with 2.4 mg L-1 of naphthylacetic acid (ANA) and 0.8 mg L-1 of 6- benzylaminopurine (6-BAP) and kept in growth chambers with controlled temperature and illumination (26 ± 2 ºC; luminosity 42 µmol m-2?s-1 of photosynthetically active radiation PAR) and a photoperiod of 16 hours, during 18 months; the culture medium was renewed every eight weeks (figure 1).
Ex vitro culture conditions
Micro-plants cultivated under in vitro conditions were transferred to plastic sacks (5 × 7.5 × 13 cm) containing red-yellow latosol, and maintained for 50 days in a growth chamber under the same conditions of luminosity, temperature, and photoperiod as the in vitro micro-plants; these were subsequently transferred to greenhouse conditions (with 50% natural illumination) for 130 days (figure 2), totaling 180 days of acclimation.
In vivo culture conditions
Peach palm seeds were sown onto planting plots containing 10 cm of sand and 5 cm of fermented sawdust and then covered with sawdust in a greenhouse with 50% natural illumination for 90 days, and subsequently transferred to plastic sacks containing red-yellow latosol, and then kept for 90 days under the same conditions as the ex vitro micro-plants (figure 3).
Determination of leaf area and thickness
The median third of the left leaflet (R1) of the first totally expanded leaf (+1) was used to supply histological sections to determine leaf area and thickness, as detailed in figures 4 and 5. The specimens were fixed in Karnovsky solution (1965), dehydrated in an ethanol series, and subsequently embedded in hydroxyethyl-methacrylate resin (Leica-historesin) according to the manufacturer's instructions. The blocks were sectioned transversely using a rotary microtome (~5 µm thickness) and the sections stained with toluidine blue (0.05%) in phosphate buffer and citric acid (Sakai 1973). Permanent slides were mounted in synthetic resin (Entellan®) and examined using a light microscope (Zeiss-Jenemed2) and photomicrographed at the same scales using a Samsung (SDC-313) digital camera.
The leaf structures corresponding to the epidermis, hypodermis, mesophyll, vascular sheath fibers, and vascular tissue were evaluated using adaptations to the standard Quant® program, and the thicknesses of the tissues were measured at 45X using an ocular micrometer. Five repetitions per sample (1 leaf per plant) were made for each culture condition.
Analyses of fiber composition
To determine the composition of the fibers observed in the mesophyll and vascular bundles, sections were made in fresh tissue in the bottom portion of the apical third of the left leaflet (R2) of plants maintained under different culture conditions (figure 5). The samples were clarified using 20% (v/v) sodium hypochlorite and subsequently stained with 0.0125% basic fuchsin and 1% Astra Blue, following Alves de Brito & Alquini (1996).
Scanning electron microscopic analysis (SEM)
Samples were taken from the median third of the left leaflet (R1) of plants growing under different environmental conditions (figure 5). The samples were fixed in modified Karnovsky (1965) solution (2.5% glutaraldehyde, 2.5% formaldehyde in 0.05 M sodium cacodylate buffer, pH 7.2) for 24 hours. The samples were then dehydrated in a increasing acetone series, subjected to critical point drying (Balzers CPD 030) using CO2 as the drying medium, followed by splutter gold coating (Med 010 Balzers metalizer), and then examined using a Zeiss SEM 940 scanning electron microscope.
Analyses of stomata and trichomes
The epidermal impression method (Segatto et al. 2004) was used employing cyanoacrylate (Loctite®) to determine stomatal and trichome densities, as well as the sizes and diameters of the stomata. These analyses were carried out the median third of the right-hand leaflets (R3) of plants maintained under different environmental conditions (figure 5). The leaf blade areas of 1 mm2 were analyzed using a light microscope equipped with an ocular grid; the sizes of the stomata and their polar and equatorial diameters were also measured using a light microscope (45x) equipped with a micrometer eyepiece.
Stomatal and trichome densities were determined using 10 plants (1 leaflet per plant) from each cultivation condition and five microscopic fields per sample-region were examined (adaxial and abaxial) per leaflet, totaling 200 counts per cultivation condition. To determine the sizes of stomata, an average of 10 specimens (1 leaf per plant) from each cultivation condition were examined and the polar and equatorial diameters of five individual stomata on each were measured.
Statistical analyses
The data was submitted to variance analysis and the averages were compared using the Tukey test at a 5% probability level, employing the SAS software program.
RESULTS AND DISCUSSION
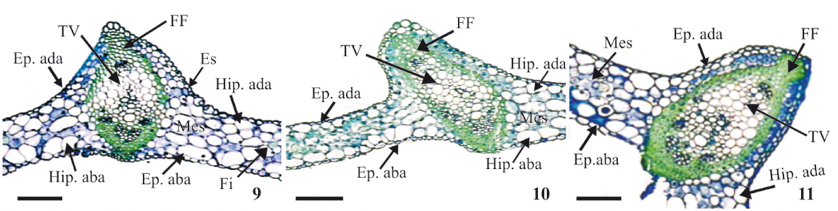
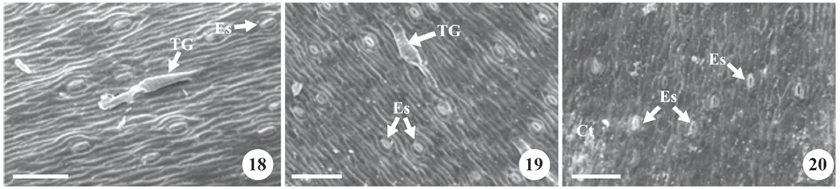
SEM and light microscopic analyses of the leaf anatomy of peach palm seedlings under all three cultivation conditions (in vitro, ex vitro and in vivo) demonstrated that they all had the same basic structural characteristics (figures 6-14 and 15-26) which are common to palm trees in general and have been described in classical publications such as Tomlinson (1961).
All of the cultivation conditions showed the presence of irregular and hetero-dimensional epidermal cells of varying sizes on both the adaxial and abaxial surfaces, with straight walls and with both cell extremities being pointed, juxtaposed, and aligned with the major axes of the leaf veins (figures 15-20); the epidermis was uniseriate on both leaf surfaces (figures 6-12).
The peach palm seedlings cultivated in vitro and the micro-plants transferred to ex vitro conditions demonstrated the absence (or reduced deposition) of epicuticular wax on both surfaces in comparison with plants cultivated in vivo (figures 15-20). This fine (or nonexistent) layer of epicuticular wax on micro-plants grown under in vitro conditions is apparently related to the fact that these plantlets grew under environmental conditions did not demand the formation of a thick wax layer as the humidity remained consistently high inside the test tubes. It is important to emphasize, however, that epicuticular wax is extremely important to the acclimation of the micro-plants, as it helps control transpiration and passive water losses (Taiz & Zeiger 2006).
The stomata of the peach palm seedlings analyzed had reniform guard cells with sub-stomatal chambers below them on both the adaxial and abaxial surfaces of the leaves (figure 12), characterizing a tetracytic stoma pattern (figure 13) commonly seen in other palms (Leite & Scatema 2001; El-Bahr et al. 2004) (figures 15-20).
As such, these leaves would be characterized as amphistomatic; however, as stomatal density was greater on the abaxial leaf surface (table 1) it is more adequate to characterize them as amphi-hypostomatic.
The types of environments under which the peach palm seedlings were grown were found to statistically influence only the numbers of stomata on the adaxial surfaces of the leaflets (table 1), with the greatest stomata densities being observed on plantlets cultivated in vivo.
No significant differences were observed in terms of the relationships between the polar and equatorial diameters of the stomata under different cultivation conditions; these differences were significant only when compared in terms of the upper and lower leaf surfaces, as the stomata on the abaxial surface were observed to be larger than those on the adaxial surface in all environments.
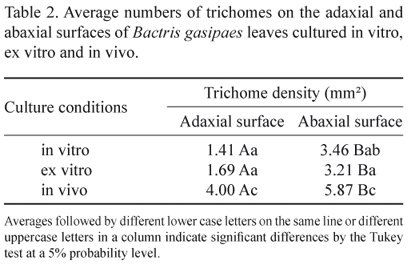
Glandular trichomes were observed with multicellular bases and unicellular apices (figure 21), as well as multicellular aculei (figure 22) on both epidermal surfaces under all cultivation conditions. These organs were severed when the epidermal mold was removed and it was not possible to distinguish or count them separately as only impression scars of their attachment sites remained. In this analysis, it was possible to observe significant differences in the average densities of trichomes on in vivo cultivated plants as compared to the other two cultivation conditions (table 2).
The density of the stomata and trichomes is inversely proportional, that is, when stomatal density decreased the numbers of trichomes increased (tables 1 and 2). This phenomenon can be attributed to the functions of trichomes, which, in addition to secreting substances that can protect plants against predators, can also indirectly influence the water economy of plants by reflecting back solar radiation and thus reducing leaf temperatures (Larcher 2000).
In relation to the areas occupied by the different leaf tissues, significant differences were seen among the epidermis of plants cultivated in vivo and the others (in vitro and ex vitro), and among the bundle fibers in all of the different cultivation regimes.
Even though the other types of leaf tissues did not demonstrate significant differences, it could be seen that the area occupied by them in the leaves of plants cultivated in vivo was greater than under the other environmental conditions (table 3). The same result was observed in terms of the thickness of these tissues, with the tissues of the plants cultivated in vivo being thicker, except for the epidermal layers (both adaxial and abaxial), as seen in table 4.
Adjacent to the epidermis, a uniseriate hypodermis composed of large irregularly shaped cells with thin cell walls on both the abaxial and adaxial surfaces of leaves could be seen under all cultivation conditions (figures 6-11).
The mesophyll was homogeneous, and composed of uniform and compact lacunar parenchyma cells, usually in four layers (figures 6-11). Dispersed throughout the mesophyll were nonvascular fibers (gelatinous fibers) that were different from the fibers associated with the vascular bundles (which are composed of cellulose) (figures 14 and 23).
Tomlinson (1961) noted that the frequency and distribution of fibers have great taxonomic importance in palms, as they are common in palms and distinct in each species. According to Esau (1959), these fibers represent adaptations to stress conditions (such as the lack of water or low light conditions) and aid the plant in resisting strong winds and rain. Paviani (1978) suggested that these gelatinous fibers could have an important role in water storage.
The areas of various tissues composing the vascular bundle were measured: the sclerenchyma fibers (bundle fibers); the vascular sheath; and the xylem and phloem (which were measured together, considering them as the vascular tissue) (figures 9-11). Only the bundle fibers demonstrated significant differences in terms of the area they occupied within the leaf blade under different culture conditions (table 3). According to Tomlinson (1961), these fibers, which are internal to the endoderm, are lignified and function to strengthen the vascular bundle.
The vascular bundles varied in size, with the smallest ones being found in the mesophyll and the largest bundles in the midrib (this being most prominently raised on the adaxial surface) (figures 24-26). According to Tomlinson (1990), the smaller vascular bundles have structural/support functions, while the larger bundles serve as conduits for water and nutrients.
The larger vascular bundles seen here were classified as bicollateral, similar to those described by Leite and Scatena (2001) for the species Syagrus harleyi, S. microphylla, S. vagans, S. werdermannii, S. coronata, and S. flexuosa, disagreeing with Pereira and Esemann Quadros (2007) and Steinmacher (2007) who classified the vascular bundles of the peach palm as being collateral. However, one must consider the development stage of the seedlings and the leaf regions examined as the apparent divergence in these classifications may well be due to the fact that the latter authors analyzed plants at the beginning of in vitro development and the vascular sheaths of adult leaves.
El-Bahr et al. (2004) reported that seedlings of the cultivar Phoenix dactylifera L. 'Zaghlool' obtained by micro-propagation showed less developed vascular tissues, with less differentiation than those seen in plants cultivated in vivo and ex vitro. This characteristic was not noted in the present work, with the bundles being very similar under all three cultivation conditions (table 2).
It can be included that there were no visible alterations in the structural characteristics of the leaves of Bactris gasipaes under the different cultivation conditions tested (in vitro, ex vitro and in vivo). Increases in the thicknesses of the mesophyll and the vascular fibers appear to be adaptive responses of peach palm micro-plants to ex vitro conditions. As such, the anatomical characteristics of the leaves of peach palm micro-plants cultivated in vitro cannot explain the difficulties encountered in their acclimation.
Acknowledgments The authors would like to thank Capes for the study grant awarded to the first author, and Prof. Dr. Elliot Watanabe Kitajima of NAP/MEPA-ESALQ-USP Piracicaba-SP for the use of the equipment employed in the scanning electron microscopy analyses.
(received: August 12, 2011; accepted: December 7, 2011)
- Almeida CV, Yara R, Almeida M. 2005. Fungos endofíticos isolados de ápices caulinares de pupunheira cultivada in vivo e in vitro Pesquisa Agropecuária Brasileira 40: 467-470
- Almeida M, Almeida C.V. 2006. Somatic embryogenesis and in vitro plant regeneration from pejibaye adult plant leaf primordia. Pesquisa Agropecuária Brasileira 41: 1449-1452.
- Almeida M, Kerbauy GB. 1996. Micropropagation of Bactris gasipaes H.B.K. (Palmae) through flower bud culture. Revista Brasileira de Fisiologia Vegetal 8:215-217.
- Alves de Brito CJF, Alquini Y. 1996. A new method for staining botanical material embedded in glycol methacrylate. Arquivos de Biologia e Tecnologia 39:949-951.
- Bovi MLA, Martins CC, Spiering SH. 2004. Dehydration of seeds of four lots of peach: effect on germination and vigor. Horticultura Brasileira 22:109-112.
- Bunn E, Turner S, Panaia M, Dixon KW. 2007. The contribution of in vitro technology and cryogenic storage to conservation of indigenous plants. Australian Journal of Botany 55:345-355.
- El-Bahr MK, Ali ZA, Mahmoud M, Saker MM. 2004. A comparative anatomical study of date palm vitroplants. Arab Journal of Biotechnology 7:219-228.
- Esau K. 1959. Anatomía vegetal. Ediciones Omega, Barcelona.
- Karnovsky MJ. 1965. A formaldehyde-glutaraldehyde fixative of high osmolality for use in electron microscopy. Journal of Cell Biology 27:137-138.
- Larcher W. 2000. Ecofisiolgia vegetal. Rima, São Carlos.
- Leite KRB, Scatena VL. 2001. Anatomia do segmento foliar de espécies de Syagrus Mart. (Arecaceae) da Chapada Diamantina. Sitientibus, Série Ciências Biológicas 1:3-14.
- Murashige T, Skoog F. 1962. Revised medium for rapid growth and bioassays with tobacco tissue culture. Physiologia Plantarum 15:473-497.
- Paviani TI. 1978. Anatomia vegetal e cerrado. Ciência e Cultura 30:1076-1086
- Pereira RA, Esemann Quadros K. 2007. Euterpe edulis Mart. e E. oleracea Mart. (Arecaceae): identificação pela análise de palmitos em conserva. Revista Brasileira de Biociências 5:333-335.
- Sakai WS. 1973. Simple method for differential staining of paraffin embedded plant material using toluidine blue O. Stain Technology 48:247-249.
- Segatto FB, Bisognin DA, Benedetti M. 2004. Técnica para o estudo da anatomia da epiderme foliar de batata. Ciência Rural 34:1597-1601.
- Steinmacher DA, Clement C, Saldanha CW, Guerra MP. 2007. Cryopreservation of peach palm zygotic embryos. CryoLetters 28:13-22.
- Steinmacher DA, Clement CR, Guerra MP. 2007. Somatic embryogenesis from immature peach palm inflorescence explants: towards development of an efficient protocol. Plant Cell, Tissue and Organ Culture 89:15-22.
- Taiz L, Zeiger E. 2006. Fisiologia vegetal. Artmed, Porto Alegre.
- Tomlinson PB. 1961. Anatomy of the monocotyledons II. Palmae. Claredon Press, Oxford.
- Tomlinson PB. 1990. The structural biology of palms. Clarendon Press, Oxford.
Publication Dates
-
Publication in this collection
18 Oct 2012 -
Date of issue
2012
History
-
Received
12 Aug 2011 -
Accepted
07 Dec 2011