Abstract
Much effort has been devoted to the identification of immunologically important antigens of Mycobacterium tuberculosis and to the combination of target antigens to which antibodies from serum of tuberculous patients could react specifically. We searched for IgG antibodies specific for antigens of 45 to 6 kDa obtained after sonication of the well-characterized wild M. tuberculosis strain in order to detect differences in the antibody response to low molecular weight antigens from M. tuberculosis between patients with pulmonary tuberculosis and contacts. Specific IgG antibodies for these antigens were detected by Western blot analysis of 153 serum samples collected from 51 patients with confirmed pulmonary tuberculosis. Three samples were collected from each patient: before therapy, and after 2 and 6 months of treatment. We also analyzed 25 samples obtained from contacts, as well as 30 samples from healthy individuals with known tuberculin status, 50 samples from patients with other lung diseases and 200 samples from healthy blood donors. The positive predictive value for associated IgG reactivity against the 6-kDa and 16-kDa antigens, 6 and 38 kDa, and 16 and 38 kDa was 100% since simultaneous reactivity for these antigens was absent in healthy individuals and individuals with other lung diseases. This association was observed in 67% of the patients, but in only 8% of the contacts. The humoral response against antigens of 16 and 6 kDa seems to be important for the detection of latent tuberculosis since the associated reactivity to these antigens is mainly present in individuals with active disease.
Western blot; Profile; Tuberculosis; Antigens
Braz J Med Biol Res, April 2005, Volume 38(04) 587-596
Humoral response to low molecular weight antigens of Mycobacterium tuberculosis by tuberculosis patients and contacts
S.T. Beck1*, O.M. Leite2*, R.S. Arruda3* and A.W. Ferreira4*
1Departamento de Análises Clínicas e Toxicológicas, Universidade Federal de Santa Maria, Santa Maria, RS, Brasil
2Divisão de Clínica de Moléstias Infecciosas e Parasitárias, and 3Laboratório Central, Hospital das Clínicas, Faculdade de Medicina, Universidade de São Paulo, São Paulo, SP, Brasil
4Bio-Merieux do Brasil S.A., and Instituto de Medicina Tropical, São Paulo, SP, Brasil
 References
References
 Correspondence and Footnotes
Correspondence and Footnotes
Correspondence and Footnotes
Correspondence and Footnotes
Correspondence and Footnotes
Correspondence and Footnotes
Abstract
Much effort has been devoted to the identification of immunologically important antigens of Mycobacterium tuberculosis and to the combination of target antigens to which antibodies from serum of tuberculous patients could react specifically. We searched for IgG antibodies specific for antigens of 45 to 6 kDa obtained after sonication of the well-characterized wild M. tuberculosis strain in order to detect differences in the antibody response to low molecular weight antigens from M. tuberculosis between patients with pulmonary tuberculosis and contacts. Specific IgG antibodies for these antigens were detected by Western blot analysis of 153 serum samples collected from 51 patients with confirmed pulmonary tuberculosis. Three samples were collected from each patient: before therapy, and after 2 and 6 months of treatment. We also analyzed 25 samples obtained from contacts, as well as 30 samples from healthy individuals with known tuberculin status, 50 samples from patients with other lung diseases and 200 samples from healthy blood donors. The positive predictive value for associated IgG reactivity against the 6-kDa and 16-kDa antigens, 6 and 38 kDa, and 16 and 38 kDa was 100% since simultaneous reactivity for these antigens was absent in healthy individuals and individuals with other lung diseases. This association was observed in 67% of the patients, but in only 8% of the contacts. The humoral response against antigens of 16 and 6 kDa seems to be important for the detection of latent tuberculosis since the associated reactivity to these antigens is mainly present in individuals with active disease.
Key words: Western blot, Profile, Tuberculosis, Antigens
Introduction
Mycobacterium tuberculosis causes pulmonary tuberculosis, and the clinical manifestations of infection can be either acute or latent and asymptomatic, depending on the intensity of the immune response set up by the infected patient (1).
Since one third of the world's population carries M. tuberculosis in its latent form, and that 5% develop active disease during the first years of infection, it is estimated that 8 million new cases of tuberculosis and 3 million deaths occur each year (2). Furthermore, the risk of reactivation increases as a consequence of associated pathologies, immunosuppressive therapy, malnutrition and, mainly, coinfection with human immunodeficiency virus (HIV) (3). Identification of these individuals is crucial for the control of disease transmission and elimination of tuberculosis.
The standard diagnosis is still made by clinical examination, direct sputum microscopy, and bacterial culture. The traditional laboratory methods used for complementation of diagnosis have limitations such as low sensitivity of acid-fast smears in paucibacillary patients, the time needed for cultivation, with undetectable growth in 10 to 20% of cases, or the high costs involved in molecular detection methods such as PCR.
The purified protein derivative used as a skin test (TST) has limitations in distinguishing between active disease and latent tuberculosis infection. This test can turn positive in people who have been vaccinated with bacillus Calmette-Guerin (BCG) and in individuals who came in contact with other mycobacteria (4). Serologic tests have not demonstrated sufficient performance but have been extensively investigated, leading to an improved understanding of the humoral response in tuberculosis and identifying novel M. tuberculosis-specific proteins.
Mycobacterial antigens such as MPT63 (5), MPT64 (5), 38 kDa (5), 30 kDa (6), 16 kDa (7), A60 (8), and Mtb81 (9) have been characterized, purified and independently tested with sera obtained from patients with tuberculosis.
The heterogeneous immune response observed, together with the absence of reactivity to a single antigen or to a specific group of antigens, suggest the existence of variations among individuals as well as an influence of disease stage (5).
Many investigators have suggested that one possibility to compensate for the variation of the immune response and the influence of disease stage would be to combine the antigens with the highest individual sensitivities into a single test. This antigen mixture would improve the test performance by allowing simultaneous recognition of the different epitopes displayed on each of the proteins employed (10). The profile of antibody reactivity has been exhaustively studied using different antigen preparations and Western blotting (11).
Not long ago, research focused on low-molecular weight antigens that are specific and potentially useful for the diagnosis of tuberculosis. These include the antigens of mass 38, 23/24, 19, and 16 kDa (14 kDa); Mtb8 and ESAT-6 (12-14).
The 38-kDa antigen is the antigen most extensively studied. It is a secretory lipoprotein possessing epitopes that are specific for the M. tuberculosis complex. Several serological assays using this antigen have been evaluated, showing good sensitivity and specificity for diagnosis of the disease (12). A 16-kDa protein, frequently called 14 kDa, contains several M. tuberculosis complex-specific B cell epitopes, showing homology with various antigens belonging to the alpha-crystallin family of low-molecular weight heat shock proteins (7). ESAT-6, characterized and purified in 1995 by Sorensen et al. (15), was subsequently used in different studies as an antigen with diagnostic potential since it was found to be specific for M. tuberculosis and absent in M. bovis BCG and in most environmental mycobacteria. ESAT-6 stimulates T-cells from patients with active tuberculosis leading to an increase in interferon-g production (13). These two latter purified proteins, 16 kDa and ESAT-6, are among the several proteins produced by the bacteria under low oxygen conditions and within artificial and animal granulomas, conditions suspected to be analogous to those present within human granulomas - but not during the logarithmic phase growth (16).
In the present study, we defined by Western blotting the serological IgG profiles of tuberculosis patients and household contacts using a whole protein extract from a wild M. tuberculosis isolate as antigen. The association of different low-molecular weight bands could be useful for the discrimination of pulmonary tuberculosis patients, household contacts and healthy individuals.
Material and Methods
Mycobacteria
The M. tuberculosis strain employed in this study was isolated from a sputum sample obtained from a patient with tuberculosis in 2000. Bacteria were cultivated on Löwenstein-Jensen medium at 37ºC for 30 days. Species identification was confirmed by microbiological assays and a DNA hybridization test using a DNA probe specific for the M. tuberculosis complex (Accuprobe M. tuberculosis complex, Gen-Probe®, cat. No. 2860; Incorporated Genetic Center Drive, San Diego, CA, USA).
Bacteria grown on Löwenstein-Jensen medium were inoculated into Sauton liquid medium (0.5 g MgSO4, 2 g citric acid, 0.65 g K2HPO4, 4 g asparagine, 0.05 g ammonium ferric citrate, and 50 ml glycerol in 1.0 liter Milli-Q H2O with a final pH of 7.2). After incubation at 37ºC for 5 weeks, the bacterial suspension was transferred to a 1-liter flask containing 250 ml Sauton liquid medium and incubated at 37ºC for another 12 weeks, without shaking.
Crude bacterial antigen
Approximately 2 g of wet bacteria were harvested by centrifugation from the surface of the liquid culture, washed and resuspended in PBS containing PMSF, EDTA and DTT at final concentrations of 1 mM each. The bacilli were subjected to sonication for 1 h at 80 W using a Vibra Cell 72405 sonicator (BioBlock Scientific, Illkirch, France) in ice and subsequently centrifuged at 10,000 g for 30 min at 4ºC. Proteins were precipitated by adding ammonium sulfate (80% saturation) to the supernatant and resuspended in saline after centrifugation at 10,000 g for 30 min at 4ºC. After dialysis for 24 h against saline using a D-7884 membrane (Sigma, St. Louis, MO, USA), the protein content was determined by the method of Lowry et al. (17). All steps were conducted according to biosafety level 3 criteria.
SDS-PAGE
Protein electrophoresis was carried out as described by Laemmli (18) using 18% acrylamide/bisacrylamide gels. The antigens were diluted in sample loading buffer without ß-mercaptoethanol, heated to 95ºC for 2 min in loading buffer and loaded onto the gel (10 µg/slot). Prestained Rainbow was used as a molecular weight marker in the 2,500- to 45,000-kDa range (Amersham Pharmacia Biotech, RPN 755 Piscataway, NJ, USA).
Western blotting
The SDS-PAGE-fractionated antigen mixture was transferred to a nitrocellulose membrane (0.45 µm, BioAgency, cat 162-0115n, São Paulo, SP, Brazil) as described by Towbin et al. (19). Transfer efficiency was monitored by checking for the presence of prestained marker bands on the membrane. After transfer, the membrane was blocked for 15 min in PBS/Tween-20 (0.05%; PBST) containing 5% skim milk, washed in PBST and cut into 4-mm wide strips. The strips were incubated with patient serum samples diluted 1/50 in PBST containing 0.5% skim milk for 18 h at 4ºC. After additional washes, the blots were incubated for 2 h at room temperature with peroxidase-conjugated anti-human IgG antibody (1/1,000 in PBST/0.5% skim milk). After another wash cycle, the blots were developed by adding 2 ml 4-chloro-1-naphthol (3 mg/ml in methanol) in 10 ml PBS and 4 µl H2O2 (30%) and incubated for 10 min in the dark. The reaction was stopped by washing the blot in distilled water. All incubations were carried out under constant shaking.
Human sera
All samples were collected after approval by the local Ethics Committee and after written informed consent was obtained from all patients or their parents.
Patients. A total of 153 serum samples were collected from 51 patients with a confirmed diagnosis of pulmonary tuberculosis at the University Hospital of São Paulo between 2000 and 2002. Forty-one patients were smear and culture positive and 10 patients were smear negative and culture positive. The patients had a median age of 37 years (age range, 14 to 75 years), 41 (80.5%) had radiological evidence of infiltration, and 22 (43.1%) of cavitary lesions. Samples were collected before treatment and after 2 and 6 months of therapy. All patients were HIV negative.
Contacts. Sixteen samples were collected from household contacts of tuberculosis patients, and another nine samples from individuals occupationally exposed to tuberculosis.
Control group. Serum samples were obtained from healthy individuals with known results of the TST (positive, N = 22; negative, N = 8), from patients with other lung diseases (lung carcinoma, silicosis, asthma, or bronchitis) (N = 50) with tuberculous diagnoses ruled out, and from an anonymous control group including sera from healthy blood donors whose tuberculin status was not known (N = 200).
Results
The serological profiles of IgG antibodies against low-molecular weight antigen fractions of M. tuberculosis were determined in different groups.
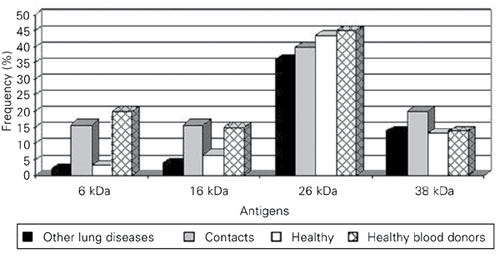
Serum from one of 51 patients with proven pulmonary tuberculosis did not react with any protein fraction between 45 and 6.0 kDa. The reagent serum from tuberculosis patients predominantly recognized antigen fractions with a molecular weight between 38 and 6 kDa. Western blot analysis showed that 57% of patients produced, before treatment, specific IgG antibodies against antigens in the 6-kDa range, 50.9% against antigens in the 16-kDa range, 59% against antigens in the 38-kDa range, and 98% against antigens in the 26-kDa range. After 2 months of therapy, the number of patients who recognized these antigens increased for the protein in the 6-kDa (D16%), 16-kDa (D15.7%) and 38-kDa (D4.1%) range. The number of reactive patients remained constant from the second to the 6th month of therapy (Figure 1). The frequency of reactivity to these antigens in the other groups was very low and significantly different (P < 0.05, Dunnett's test; Figure 2).
Among healthy TST-negative individuals, no one showed reactivity to proteins in the 6-, 16- or 38-kDa range.
Western blotting analysis of the sample obtained before treatment from 51 tuberculous patients showed four reactivity profiles that were not present in healthy controls or in patients with other lung diseases. These profiles were: simultaneous reactivity with antigenic fractions 38-26-16 kDa (profile I), 38-26-6 kDa (profile II); 38-26-16-6 kDa (profile III), and 26-16-6 kDa (profile IV; Figure 3 and Table 1). When the presence of any one of the cited profiles was considered to be a positive reaction, the sensitivity obtained for the group of positive smear patients was 75%, with 19.5% for profile I, 22% for profile II, 14.6% for profile III, and 19.5% for profile IV. In the group of negative smear patients, sensitivity was 30%, with 20% for profile II and 10% for profile III (Table 2).
Only two of 25 (8%) sera obtained from contacts presented profile IV. These individuals were TST-positive (strips 1, 3; Figure 4).
Of the 200 anonymous controls, 1 (0.5%) showed profile II, 4 (2%) profile III, and 6 (3%) profile IV (Table 1). The specificity detected for this population was 94.5. (Table 2). Other simultaneous reactivities (26-6, 26-38, 26-16) appeared not to be important, since they were present in all groups (Table 1) including TST-positive healthy individuals (Figure 2).
Therefore, the positive predictive values for the combined analyses of the reactivity profiles were 100% for the 38+16+6-kDa mass range, 38+16-kDa mass range and 38+6-kDa mass range, since these profiles were not present in healthy individuals or in patients with other lung diseases. The negative predictive values for the same combinations were 64% (38+6+16 kDa), 65% (38+16 kDa) and 66% (6+38 kDa).
Frequency of the reactivity of IgG antibodies against different antigenic fractions of Mycobacterium tuberculosis in tuberculosis patients before and during treatment.
Frequency of the reactivity of IgG antibodies against different antigenic fractions of contacts of Mycobacterium tuberculosis patients and control groups.
Western blot profile shown by tuberculosis patients. A, Sample before patient treatment; B, sample after two months of treatment; C, sample after 6 months of treatment. Simultaneous reactivity with antigenic fractions 38-26-16 kDa (profile I), 38-26-6 kDa (profile II); 38-26-16-6 kDa (profile III), and 26-16-6 kDa (profile IV). MM = molecular mass; C+ = positive control serum from tuberculosis patients.
Western blot profile shown by contacts, other lung diseases and healthy individuals. MM = molecular mass; C+ = positive control serum from tuberculosis patients.
Profiles of the reactivities shown by tuberculosis patients, patients with other lung diseases, healthy individuals, contacts, and healthy blood donors.
Discussion
The ability of a protein to detect antibodies present during subclinical disease is as important as the sensitivity of a protein in detecting antibodies formed during active tuberculosis, since therapy for latent tuberculosis infection can prevent the development of active disease.
We investigated the humoral response of tuberculosis patients and their contacts to proteins with a molecular mass between 45 and 2.5 kDa that are present in the soluble fraction of sonicated M. tuberculosis, including membrane and cytoplasm proteins. The use of a clinical M. tuberculosis isolate obtained from a patient with active disease instead of a standard laboratory strain, H37rv, is an important consideration. Lopes et al. (20) investigated the immune response produced by different M. tuberculosis strains and important differences were observed. Although the strain used in the present study to obtain the antigen extract has not been genetically characterized, it probably represents the predominant strain of M. tuberculosis in the geographical area studied.
For this study we used Western blot because it is a method that provides a safe reading and interpretation of the results when duly standardized. In the present study, we produced industrial lots of antigen fractions that were assessed for reproducibility with control serum samples collected from patients with confirmed pulmonary tuberculosis and healthy individuals (data not shown). Compared to molecular methods, Western blot has the advantage of being easy to perform, considering that small laboratories are better equipped for the execution of serological tests than of molecular tests. The stability of the nitrocellulose strips, that can be stored for more than 12 months, permits the execution of the test in laboratories with small routines. The Western blot method has been widely used to confirm serologic results and for the identification of the immune response against different protein fractions of viruses, fungi, and parasites (21).
Studies employing Western blot analysis have shown that M. tuberculosis antigens in the range of 65, 61, 58, 30, and 24 kDa frequently react with serum from controls (22), while sera from tuberculosis patients and healthy controls differ in their reactivity with antigens in the 43- and 12-kDa range (23).
In the present study, we demonstrated that before the initiation of treatment 57% of the patients produced antibodies against the 6-kDa range, and after two months of therapy 73% of the patients produced specific antibodies. The same was observed for antibodies specific for the 16- and 38-kDa fractions. After this initial increase, antibody levels remained constant until the end of therapy (Figure 1). Similar data were obtained in studies carried out by Imaz and Zerbini (24) to determine the antibody response to culture filtrate antigens in tuberculosis patients, and by Bothamley and Rudd (25) to determine the specific response to the 38-kDa antigen. A possible explanation for this phenomenon may be the intense stimulation of the immune response by antigens released by dead bacteria, and/or by the release of antibodies that were previously part of immune complexes. No change in the reactivity profile that would allow to determine the evolution of treatment was observed from the second month to the end of therapy.
In the investigated molecular mass range, a 26-kDa protein was recognized before and after treatment in 98% of the patients studied. Despite this high sensitivity, 40% of the contacts and 33% of the patients with other lung diseases also reacted with antigens in this mass range (Figure 2).
The frequency of reactivity observed for a protein of approximately 38 kDa (59%) was similar to that reported elsewhere (14,26) and more common in patients with cavitary processes, as also reported by others (27,28).
The response to the 16- or 6-kDa antigens was higher in patients (50.9 and 57%, respectively) than in contacts (both 16%) or healthy subjects (6.6 and 3.3%, respectively) (Table 1). The presence of these antibodies in tuberculosis patients has been associated with a more favorable prognosis or with spontaneously cured tuberculosis, since these antibodies are the first to disappear after 2 years of treatment (28). An increase in anti-14-kDa (16-kDa) antibodies has been reported for healthy individuals after occupational exposure, and for household contacts of tuberculosis patients (12,29). Thus, the presence of these antibodies in contacts may suggest a tuberculosis infection that is not clinically evident.
Little is known about the antibody response against antigens in the 6-kDa range. ESAT-6 (a 6-kDa early secretory antigenic target) has been investigated for diagnostic purposes. The antigen possesses various specific T-cell epitopes that induce a cellular response and lead to increased interferon g production in patients with subclinical or active tuberculosis but not in unexposed healthy individuals (13). This cell-mediated response has also been associated with increased risk of disease (30). A recent study showed that the humoral response to ESAT-6 may be associated with inactive tuberculosis but not with active tuberculosis (31).
In the present study, Western blot analysis showed that serum from 45 (88%) patients reacted simultaneously with at least two proteins (Figure 3). Franco et al. (11) studied the serological response to fractions of M. tuberculosis sonicated antigen and showed that 52.2% of positive smear pulmonary tuberculosis patients had full responses against 4 antigenic fractions (71, 65, 26-38, and 19 kDa) and 41.2% of negative smear patients had partial responses.
In our study, when the humoral response was analyzed in samples obtained before treatment, 34 (66.6%) of 51 patients showed simultaneous reactivity against antigenic fractions, that was absent in samples from healthy individuals or in patients with other lung diseases. In these latter two groups, the presence of tuberculosis was excluded by clinical and laboratory examination (Table 1).
These reactivity profiles showed sensitivity similar to that observed by other investigators. However, in the general population, especially in developing countries, with a high prevalence of M. tuberculosis infection, there is a percentage of persons who do not present clinical signs and symptoms of tuberculosis and who must be identified and treated. For this purpose, a highly specific test is needed in order to detect suspected cases. Analysis of Western blot tests performed on samples from healthy individuals and from patients with other lung diseases, showing the absence of reactivity for the profiles described earlier, revealed that this is a result of great clinical and epidemiological importance, since blood donors from the general population presented 5.5% positivity. These individuals, like the contacts, should be investigated more extensively in terms of a possible history of M. tuberculosis infection. It is important to point out that healthy individuals with a negative TST did not show reactivity to any of the antigen fractions studied. Thus, the occurrence of I, II, III, and IV reactivity profiles in individuals with a positive TST with no apparent clinical symptoms may represent an important prognostic factor. These individuals may eventually present a course of latent infection differing from that of healthy individuals with a positive TST who only showed less important reactivity profiles.
The simultaneous reactivity against 16- and 6-kDa proteins present in 2 of 25 contacts and in 10 of 200 healthy blood donors is an interesting finding. Both proteins are expressed by M. tuberculosis when subjected to oxygen deprivation (32,33). As containment within the granuloma may induce similar conditions these proteins may be an important antigenic target during bacillary latency (34). Thus, these data may suggest detection of latent tuberculosis in 8% of the contacts and 5% of the anonymous control group. This is possibly due to the high prevalence of tuberculosis infection in the Brazilian population.
Earlier studies have suggested that the humoral response to M. tuberculosis antigens was increased in patients with an inadequate cell-mediated response to the same antigens (35) and in patients that showed a strong humoral response after the infection led to the development of active tuberculosis (36). This evidence, together with a recent study that evaluated the humoral response to recombinant M. tuberculosis antigens, 38 kDa, 14 kDa and ESAT-6, showing association of the latter two antigens with risk factors for future active, but not current disease (31), suggest the possibility of identifying the subset of persons with latent tuberculosis infection who may be at high risk to develop active disease.
These data provide important evidence even though they cannot be considered definitive, since significant geographical variation in antibody titer to some antigens exists (37). Heterogeneous recognition of antigens by serum antibodies in tuberculosis can result from multiple factors (5), and characteristics such as the use of BCG vaccination (38) should also be assessed.
We conclude that the humoral response mainly against antigens of mass 16 and 6 kDa seems to be important for the detection of latent tuberculosis. The presence of reactivity profiles III and IV in a person with prior contact with tuberculosis suggests the presence of latent tuberculosis infection with an increased future risk for active disease, since this profile shows associated reactivity to these antigens and is mainly present in individuals with active disease, with a positive predictive value of 100%. However, this possibility can only be confirmed by prospective studies and deserves further evaluation.
Address for correspondence: S.T. Beck, Departamento de Análises Clínicas e Toxicológicas, CCS, Universidade Federal de Santa Maria, 97105-900, Santa Maria, RS, Brasil. E-mail: sbeck@ig.com.br
Publication supported by FAPESP. *All authors contributed equally to this study. Received April 22, 2004. Accepted January 11, 2005.
- 1. Orme IM (2001). The latent tuberculosis bacillus (I'll let you know if I ever meet one). International Journal of Tuberculosis and Lung Disease, 5: 589-593.
- 2. Dye C, Scheele S, Dolin P, Pathania V & Raviglione MC (1999). Global burden of tuberculosis: estimated incidence, prevalence, and mortality by country. Journal of the American Medical Association, 282: 677-686.
- 3. Rook GAW & Pando RH (1996). The pathogenesis of tuberculosis. Annual Review of Microbiology, 50: 259-284.
- 4. Pottumarthy S, Morris AJ, Harrison AC & Wells VC (1999). Evaluation of the tuberculin gamma interferon assay: potential to replace the Mantoux skin test. Journal of Clinical Microbiology, 37: 3229-3232.
- 5. Lyashchenko K, Colangeli R, Houde M, Jahdali HA, Menzies D & Gennaro ML (1998). Heterogeneous antibody responses in tuberculosis. Infection and Immunity, 66: 3936-3940.
- 6. Salata RA, Sanson AJ, Malhotra IJ, Wiker HG, Harboe M, Philips NB & Daniel TM (1991). Purification and characterization of the 30,000 kilodalton native antigen of M. tuberculosis and characterization of six monoclonal antibodies reactive with a major epitope of this antigen. Journal of Laboratory and Clinical Medicine, 118: 589-598.
- 7. Verbon A, Hartskeeri RA, Moreno C & Kolk AH (1992). Characterization of B cell epitopes on the 16 K antigen of Mycobacterium tuberculosis. Clinical and Experimental Immunology, 89: 395-401.
- 8. Cocito C & Vanlinden F (1986). Preparation and properties of antigen 60 from Mycobacterium bovis BCG. Clinical and Experimental Immunology, 66: 262-272.
- 9. Hendrickson RC, Douglass JF, Reynolds LD, McNeill PD, Carter D, Reed SG & Houghton RL (2000). Mass spectrometric identification of Mtb 81, a novel serological marker for tuberculosis. Journal of Clinical Microbiology, 38: 2354-2361.
- 10. Lyashchenko KP, Singh M, Colangeli R & Gennaro ML (2000). A multi-antigen print immunoassay for the development of serological diagnosis of infectious diseases. Journal of Immunological Methods, 242: 91-100.
- 11. Franco J, Camarena JJ, Nogueira JM, Blanquer R, Ruiz MJ & Marin J (2001). Serological response (Western blot) to fractions of Mycobacterium tuberculosis sonicate antigen in tuberculosis patients and contacts. International Journal of Tuberculosis and Lung Disease, 5: 958-962.
- 12. Jackett PS, Bothamley GH, Batra HV, Mistry A, Young DB & Ivanyl J (1988). Immunodominance and specificity of mycobacterial antigens and their epitopes in serology of tuberculosis. Journal of Clinical Microbiology, 26: 2313 2318.
- 13. Arend SM, Ottenhoff THM, Andersen P & Dissel JTV (2001). Uncommon presentation of tuberculosis: the potential value of a novel diagnostic assay based on the Mycobacterium tuberculosis-specific antigens ESAT-6 and CPF-10. International Journal of Tuberculosis and Lung Disease, 7: 680-686.
- 14. Espitia C, Cervera I, Gonzales R & Mancilla R (1989). A 38 kDa Mycobacterium tuberculosis antigen associated with infection. Its isolation and serological evaluation. Clinical and Experimental Immunology, 77: 373-377.
- 15. Sorensen AL, Nagai S, Houen G, Andersen P & Andersen AB (1995). Purification and characterization of a low-molecular-mass T-cell antigen secreted by Mycobacterium tuberculosis. Infection and Immunity, 63: 1710-1717.
- 16. Quinn FD, Birkness KA & King PJ (2002). Alpha-crystallin as a potential marker of Mycobacterium tuberculosis latency. ASM News, 68: 612-617.
- 17. Lowry OH, Rosebrough D, Farr C & Randall RJ (1951). Protein measurement with the Folin phenol reagent. Journal of Biochemistry, 193: 265-275.
- 18. Laemmli UK (1970). Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature, 226: 680-685.
- 19. Towbin H, Staehelin T & Gordon J (1979). Electrophoretic transfer of proteins from polyacrylamide gels to nitrocellulose sheets: procedure and some applications. Proceedings of the National Academy of Sciences, USA, 76: 4350-4354.
- 20. Lopes B, Aguilar, D, Orozco H et al. (2003). A marked difference in pathogenesis and immune response induced by different Mycobacterium tuberculosis genotypes. Clinical and Experimental Immunology, 133: 30-37.
- 21. Matsumoto TK, Cotrim PC, da Silveira JF, Stolf AM & Umezawa ES (2002). Trypanosoma cruzi: isolation of an immunodominant peptide of TESA (trypomastigote excreted-secreted antigens) by gene cloning. Diagnostic Microbiology and Infectious Disease, 42: 187-192.
- 22. Verbon A, Kuijper S, Jansen HM, Speelman P & Kolk AH (1990). Antigens in culture supernatant of Mycobacterium tuberculosis: epitopes defined by monoclonal and human antibodies. Journal of General Microbiology, 136: 955-964.
- 23. Olsen I, Reitan LJ & Wiker HG (2000). Distinct differences in repertoires of low-molecular-mass secreted antigens of Mycobacterium avium complex and Mycobacterium tuberculosis Journal of Clinical Microbiology, 38: 4453-4458.
- 24. Imaz M & Zerbini SE (2000). Antibody response to culture filtrate antigens of Mycobacterium tuberculosis during and after treatment of tuberculosis patients. International Journal of Tuberculosis and Lung Disease, 4: 562-569.
- 25. Bothamley GH & Rudd RM (1994). Clinical evaluation of a serological assay using a monoclonal antibody (TB72) to the 38 kDa antigen of Mycobacterium tuberculosis European Respiratory Journal, 7: 240-246.
- 26. Verbon A (1994). Development of serological tests for tuberculosis - problems and potential. Tropical and Geographical Medicine, 46: 275-279.
- 27. Laal S, Samanich KM, Sonnenberg MG, Zolla-Pazner S, Phadtare JM & Belisle J (1997). Human humoral responses to antigens of Mycobacterium tuberculosis: immunodominance of high-molecular-mass antigens. Clinical and Diagnostic Laboratory Immunology, 4: 49-56.
- 28. Bothamley GH, Rudd R, Festenstein F & Ivanyi J (1992). Clinical value of the measurement of Mycobacterium tuberculosis specific antibody in pulmonary tuberculosis. Thorax, 47: 270-275.
- 29. Bothamley GH, Beck JS, Potts RC, Grange JM, Kardjito T & Ivanyi J (1992). Specificity of antibodies and tuberculin response after occupational exposure to tuberculosis. Journal of Infectious Diseases, 166: 182-186.
- 30. Doherty TM, Demissie A, Olobo J, Wolday D, Britton S, Eguale T, Ravn P & Andersen P (2002). Immune responses to the Mycobacterium tuberculosis-specific antigen ESAT-6 signal subclinical infection among contacts of tuberculosis patients. Journal of Clinical Microbiology, 40: 704-706.
- 31. Silva VMC, Kanujia G, Gennaro ML & Menzies D (2003). Factors associated with humoral response to ESAT-6, 38 kDa and 14 kDa in patients with a spectrum of tuberculosis. International Journal of Tuberculosis and Lung Disease, 7: 478-484.
- 32. Cunninghamm A & Spreadbury C (1998). Mycobacterial stationary phase induced by low oxygen tension: cell wall thickening and localization of the 16-kilodalton alpha-crystallin homolog. Journal of Bacteriology, 180: 801-808.
- 33. Haile Y, Bjune G & Wiker HG (2002). Expression of the mce-A, esat-6 and hspX genes in Mycobacterium tuberculosis and their responses to aerobic conditions and to restricted oxygen supply. Microbiology, 148: 3881-3886.
- 34. Agrewala JN & Wilkinson RJ (1998). Differential regulation of Th1 and Th2 cells by p91-110 and p21-40 peptides of the 16 kDa a crystallin antigen of Mycobacterium tuberculosis. Clinical and Experimental Immunology, 114: 392-397.
- 35. Bhantnagar R, Malaviya AN, Narayanan S, Rajgopalan P, Kumar R & Baharadwaj OP (1977). Spectrum of immune response abnormalities in different clinical forms of tuberculosis. American Review of Respiratory Disease, 115: 207-212.
- 36. David HL, Papa F & Cruaud P (1992). Relationships between titer of antibodies immunoreacting against glycolipid antigens from Mycobacterium leprae and M. tuberculosis, the Mitsuda and Mantoux reactions, and bacteriologic loads: implications in the pathogenesis, epidemiology and serodiagnosis of leprosy and tuberculosis. International Journal of Leprosy and Other Mycobacterial Diseases, 60: 208-224.
- 37. Bothamley G, Batra H, Ramesh V, Chandramuki A & Ivanyi J (1992). Serodiagnostic value of the 19 kilodalton antigen of Mycobacterium tuberculosis in Indian patients. European Journal of Clinical Microbiology and Infectious Diseases, 11: 6-10.
- 38. Pottumarthy S, Wells VC & Morris AJ (2000). A comparison of seven tests for serological diagnosis of tuberculosis. Journal of Clinical Microbiology, 38: 2227-2231.
Correspondence and Footnotes
Publication Dates
-
Publication in this collection
13 Apr 2005 -
Date of issue
Apr 2005
History
-
Accepted
11 Jan 2005 -
Received
22 Apr 2004