Abstract
Glutathione is the major intracellular antioxidant thiol protecting mammalian cells against oxidative stress induced by oxygen- and nitrogen-derived reactive species. In trypanosomes and leishmanias, trypanothione plays a central role in parasite protection against mammalian host defence systems by recycling trypanothione disulphide by the enzyme trypanothione reductase. Although Kinetoplastida parasites lack glutathione reductase, they maintain significant levels of glutathione. The aim of this study was to use Leishmania donovani trypanothione reductase gene mutant clones and different Leishmania species to examine the role of these two individual thiol systems in the protection mechanism against S-nitroso-N-acetyl-D,L-penicillamine (SNAP), a nitrogen-derived reactive species donor. We found that the resistance to SNAP of different species of Leishmania was inversely correlated with their glutathione concentration but not with their total low-molecular weight thiol content (about 0.18 nmol/10(7) parasites, regardless Leishmania species). The glutathione concentration in L. amazonensis, L. donovani, L. major, and L. braziliensis were 0.12, 0.10, 0.08, and 0.04 nmol/10(7) parasites, respectively. L. amazonensis, that have a higher level of glutathione, were less susceptible to SNAP (30 and 100 µM). The IC50 values of SNAP determined to L. amazonensis, L. donovani, L. major, and L. braziliensis were 207.8, 188.5, 160.9, and 83 µM, respectively. We also observed that L. donovani mutants carrying only one trypanothione reductase allele had a decreased capacity to survive (~40%) in the presence of SNAP (30-150 µM). In conclusion, the present data suggest that both antioxidant systems, glutathione and trypanothione/trypanothione reductase, participate in protection of Leishmania against the toxic effect of nitrogen-derived reactive species.
Leishmania; Glutathione; Trypanothione; Trypanothione reductase; Nitric oxide; Free radicals
Braz J Med Biol Res, March 2006, Volume 39(3) 355-363
Glutathione and the redox control system trypanothione/trypanothione reductase are involved in the protection of Leishmania spp. against nitrosothiol-induced cytotoxicity
 Correspondence and Footnotes
Correspondence and Footnotes
 P.R.T. Romão1, J. Tovar2, S.G. Fonseca3, R.H. Moraes4, A.K. Cruz5, J.S. Hothersall6, A.A. Noronha-Dutra6, S.H. Ferreira4 and F.Q. Cunha4
P.R.T. Romão1, J. Tovar2, S.G. Fonseca3, R.H. Moraes4, A.K. Cruz5, J.S. Hothersall6, A.A. Noronha-Dutra6, S.H. Ferreira4 and F.Q. Cunha4
1Laboratório de Imunoparasitologia, Curso de Medicina, Universidade do Sul de Santa Catarina (UNISUL), Tubarão, SC, Brasil
2School of Biological Sciences, Royal Holloway University of London, Egham, UK
3Laboratório de Imunologia, Instituto do Coração, Faculdade de Medicina, Universidade de São Paulo, São Paulo, SP, Brasil
4Departamento de Farmacologia, 5Departamento de Biologia Celular e Molecular e Bioagentes Patogênicos, Faculdade de Medicina de Ribeirão Preto, de São Paulo, Ribeirão Preto, SP, Brasil
6The Institute of Urology and Nephrology, University College of London, London, UK
 References
References
 Correspondence and Footnotes
Correspondence and Footnotes
Correspondence and Footnotes
Correspondence and Footnotes
Correspondence and Footnotes
Correspondence and Footnotes
Abstract
Glutathione is the major intracellular antioxidant thiol protecting mammalian cells against oxidative stress induced by oxygen- and nitrogen-derived reactive species. In trypanosomes and leishmanias, trypanothione plays a central role in parasite protection against mammalian host defence systems by recycling trypanothione disulphide by the enzyme trypanothione reductase. Although Kinetoplastida parasites lack glutathione reductase, they maintain significant levels of glutathione. The aim of this study was to use Leishmania donovani trypanothione reductase gene mutant clones and different Leishmania species to examine the role of these two individual thiol systems in the protection mechanism against S-nitroso-N-acetyl-D,L-penicillamine (SNAP), a nitrogen-derived reactive species donor. We found that the resistance to SNAP of different species of Leishmania was inversely correlated with their glutathione concentration but not with their total low-molecular weight thiol content (about 0.18 nmol/107 parasites, regardless Leishmania species). The glutathione concentration in L. amazonensis, L. donovani, L. major, and L. braziliensis were 0.12, 0.10, 0.08, and 0.04 nmol/107 parasites, respectively. L. amazonensis, that have a higher level of glutathione, were less susceptible to SNAP (30 and 100 µM). The IC50 values of SNAP determined to L. amazonensis, L. donovani, L. major, and L. braziliensis were 207.8, 188.5, 160.9, and 83 µM, respectively. We also observed that L. donovani mutants carrying only one trypanothione reductase allele had a decreased capacity to survive (~40%) in the presence of SNAP (30-150 µM). In conclusion, the present data suggest that both antioxidant systems, glutathione and trypanothione/trypanothione reductase, participate in protection of Leishmania against the toxic effect of nitrogen-derived reactive species.
Key words: Leishmania, Glutathione, Trypanothione, Trypanothione reductase, Nitric oxide, Free radicals
Introduction
The trypanosomatids, members of the order Kinetoplastida, include parasitic protozoa of importance to public health such as Leishmania spp. Leishmania cause a spectrum of diseases ranging from self-healing ulcers to disseminated and often fatal infections, depending on the species involved and the host's immune response. Adequate vaccines against trypanosomatid infections have yet to be developed, and drugs currently available for chemotherapeutic intervention are mostly unsatisfactory mainly because of their lack of specificity, toxicity to humans, and, in many cases, developed parasite resistance (1). Thus, one of the priorities in tropical medicine research has been the identification and characterisation of parasite-specific biomolecules, which play relevant physiological roles and thus might be exploited as selective targets.
Among many other metabolic distinctions, trypanosomatids maintain their intracellular redox balance by a mechanism that is different from that of their insect vectors and mammalian hosts. They lack glutathione reductase, which in nearly all other organisms is responsible for the maintenance of an intracellular thiol-reducing environment, and thus for the reduction of disulphides, detoxification of peroxides and synthesis of DNA precursors (2). Instead, they possess a unique system using trypanothione [T(SH)2] that is the major reduced thiol of Kinetoplastida parasites (3) and comprises a spermidine moiety linked to two glutathione molecules (2). Together with three thiol-redox proteins, trypanothione reductase (TryR), tryparedoxin and tryparedoxin peroxidase (4,5), T(SH)2 is thought to provide defence against oxidants, certain heavy metals (6) and xenobiotics (3). Thus, TryR has a vital physiological role in maintaining T(SH)2 redox, particularly within the highly oxidative intracellular environment of the host cells which is generated during the antimicrobial defence response.
In murine leishmaniasis, nitric oxide (NO) plays a crucial role in the killing of parasites both in vitro (7) and in vivo (8,9). In vitro macrophage microbicidal activity correlates with NO production, and both in vivo and in vitro microbicidal activities are completely inhibited by the NO synthase inhibitor L-arginine analogue NG-monomethyl-L-arginine (L-NMMA) but not by its enantiomer D-NMMA (8). In addition, the NO donors, S-nitroso-N-acetyl-D,L-penicillamine (SNAP) and 3-morpholino-sydnonimine hydrochloride are able to kill Leishmania parasites in a cell-free model system (10).
We have reported that glutathione is involved in the protection of mammalian macrophages against the cytotoxic effects of NO. Furthermore, despite evidence that glutathione in Leishmania appears not to be the major antioxidant, we have demonstrated that it protects L. major from the toxic effects of NO (11).
In the present study, we have extended these observations by comparing the glutathione levels and SNAP sensitivity of different Leishmania species: L. amazonensis, L. braziliensis, L. donovani, and L. major. Our results demonstrate that the sensitivity of distinct species of Leishmania to SNAP is inversely correlated with their glutathione concentration. When we extended our investigation to the role of TryR in the protection against SNAP using mutants of L. donovani for the TryR gene (tryR, formerly tryA) (12), we found
that, compared to control parasites (tryR genotype+/+/+), a double mutant clone (tryR genotype-/-/+) was significantly more sensitive to the cytotoxic effect of SNAP. Overall, these results demonstrate that glutathione as well as the T(SH)2/TryR redox system are essential protective components against NO cytotoxicity in Leishmania.
Material and Methods
Leishmania strains and culture conditions
The Leishmania species used in this study were L. braziliensis (MHOM/BR/75/M2904), L. amazonensis (MPRO/BR/72/M1841-LV-79), L. major (LV-39, clone 5-Rho-SU/59/P), and L. donovani (clone LV9-3 from MHOM/ET/67/HU3). The L. donovani clones used were: wild-type LV9-3, which possesses three copies of the tryR (formerly tryA) gene (tryR+/+/+) and the mutants of TryR: clones H2-tryR-/+/+ (LV9-3 submitted to single replacement), clone HB3-tryR-/-/+ (LV9-3 submitted to double replacement (12) and clone HB3-pTTcTR (Tovar J and Fairlamb AH, unpublished results) that is identical to clone HB3 (tryR-/-/+) but harbours plasmid pTTcTR (13). Promastigote forms of all Leishmania species were grown in M199 medium supplemented with 40 mM HEPES, pH 7.4, 0.1 mM adenine, 7.7 mM hemin, 10% (v/v) heat-inactivated foetal calf serum, 50 U/mL penicillin, and 50 µg/mL streptomycin. Cultures were incubated at 26ºC, and cells were kept at densities ranging between 5 x 105 and 3 x 107 parasites/mL. Transfectants were cultured in the presence of selective drugs (12). The mutant H2-tryR-/+/+ was cultured in the presence of 16 µg/mL hygromycin B, the clone HB3- tryR-/-/+ in the presence of 16 µg/mL hygromycin B plus 2.5 µg /mL phleomycin, and the HB3-tryR-/-/+ clone plus episomal pTTcTR in the presence of 16 µg/mL hygromycin B, 25 µg/mL G418 and 2.5 µg/mL phleomycin.
Growth curves
Promastigotes of wild-type or L. donovani mutants were cultured in M199 medium prepared as described previously (14). Cell density in the inoculum was 1 x 105/mL. Viability was evaluated from motility and cell density was determined daily using a hemocytometer.
Glutathione and non-protein low-molecular weight thiol measurement
Non-protein low-molecular weight thiols and glutathione (reduced plus disulphide forms) were measured in lysates of promastigote forms (stationary phase) of different Leishmania species (L. amazonensis, L. braziliensis, L. donovani, and L. major) including the different clones of L. donovani. Low-molecular weight thiols were measured using Ellman's reagent (15). To measure soluble thiols the samples were deproteinised with 1% sulfosalicylic acid in the presence of 5 mM EDTA. The concentration of SH groups was calculated from a standard curve of 0.01 to 2 nmol cysteine. Glutathione levels were measured by the glutathione reductase enzyme recycling method (16). These assays were adapted for use in a microtitre plate using a microplate spectrophotometer system spectra MAX 250 (Molecular Devices, Union City, CA, USA). Cells were lysed by the addition of 100 µL 1 mM EDTA to each well and freezing immediately. Following thawing, plates were shaken for 30 s and then sonicated for 60 s. Assays were carried out immediately (17). Although this assay provides a measure of both oxidised and reduced glutathione, in non-oxidative stress equilibrium, the cellular condition under which we have measured glutathione, the thiol content is 95-99% reduced glutathione.
Cytotoxic effect of S-nitroso-N-acetyl-D,L-penicillamine on Leishmania viability
The direct cytotoxic effect of the nitrosothiol SNAP on Leishmania species was measured. Briefly, parasites (3 x 106/well) were incubated in M199 medium supplemented with 10% heat-inactivated foetal calf serum in the presence or absence of SNAP (30-1000 µM) for 12 h. The incubation medium contains L-cystine which allows the membrane transport of SNAP into cells (18). Parasites were then pulsed with 1 µCi/well [3H]-thymidine, and the incorporation of radioactivity by viable parasites was determined after 24 h in a ß-counter (11). The 50% inhibitory concentration (IC50) values for each Leishmania species were determined using Sigma-Plot software, Version 5.0.
Statistical analysis
Data are reported as means ± SEM and statistical significance (P < 0.05) was assessed by ANOVA followed by Bonferroni's t-test.
Results
Non-protein low-molecular weight thiol and glutathione concentration in different Leishmania species
Glutathione concentrations of different species of Leishmania (L. amazonensis, L. braziliensis, L. major, and L. donovani) are shown in Figure 1. Statistically significant differences in glutathione levels were observed between all Leishmania species, with promastigotes of L. braziliensis having the lowest level. Glutathione concentration was L. amazonensis > L. donovani > L. major > L. braziliensis. No significant differences in the non-protein low-molecular weight thiol levels were detected between different Leishmania species (Figure 2).
Cytotoxic effect of S-nitroso-N-acetyl-D, L-penicillamine on different Leishmania species
To ascertain whether the glutathione levels in the different Leishmania species (Figure 1) correlate with their sensitivity to reactive nitrogen species, we investigated the cellular viability of L. amazonensis, L. braziliensis, L. donovani, and L. major after treatment with SNAP. The addition of a SNAP directly to promastigotes of different species of Leishmania resulted in dose-dependent parasite killing (Figure 3). The sensitivity of Leishmania species to SNAP correlated inversely with the glutathione levels. L. amazonensis, that has a higher glutathione concentration, was more resistant to the cytotoxic effect of SNAP at concentrations of 30 and 100 µM, with an IC50 of 207.8 (200.2-215.7 µM). In contrast, L. braziliensis, that shows lower levels of glutathione, was more susceptible to the toxic effect of SNAP (IC50 of 83 (79.7-85.5 µM)). The IC50 values determined for L. donovani and L. major were 188.5 and 160.9 µM, respectively.
Effect of S-nitroso-N-acetyl-D,L-penicillamine on the viability of Leishmania donovani tryR gene mutants
To ascertain whether the T(SH)2/TryR antioxidant system is involved in the protection of Leishmania against nitrogen-derived reactive species, we investigated the effect of SNAP on the viability of targeted L. donovani TryR mutants that have been generated by gene disruption. Wild-type L. donovani (LV9-3, genotype of tryR+/+/+) and clones submitted to a single (clone H2 genotype of tryR-/+/+) or double (clone HB3 genotype of tryR-/-/+) experiment for the replacement of the tryR locus were utilised. It is important to mention that a null mutant has not yet been obtained for tryR (12,19). The addition of SNAP directly to promastigotes of different L. donovani clones resulted in partial parasite killing (Figure 4). Recombinant HB3 possessing only one tryR allele was more sensitive to the toxic effect of SNAP at concentrations of 30-150 µM compared with recombinant H2 or L. donovani parental clone (LV9-3). The sensitivity to high concentration of SNAP (300 µM) was unchanged. To demonstrate that the decrease in the sensitivity was solely due to the disruption of the tryR gene, we tested the viability of clone HB3 (tryR-/-/+) supplemented with the plasmid pTTcTR that harbours a functional T. cruzi tryR gene (13). This recombinant strain, whose levels of TryR are higher than those of wild-type parasites, regained wild-type levels of resistance to SNAP-generated stress (Figure 4).
Glutathione concentration in Leishmania donovani TryR mutants
Our previous results (11) and the data presented here suggest that glutathione is involved in the protection of Leishmania against the toxic effects of SNAP. As TryR is the enzyme that maintains T(SH)2 in its reduced form and plays a central role in oxidant detoxification through the enzymatic regeneration of the thiol pool, we examined whether the effect of the loss of the tryR copy in L. donovani mutant clones changed the glutathione levels. No significant differences in glutathione were detected between the wild-type and single- or double-targeted L. donovani tryR mutants or with the HB3-pTTcTR transfected clone (Figure 5).
Growth of Leishmania donovani clones
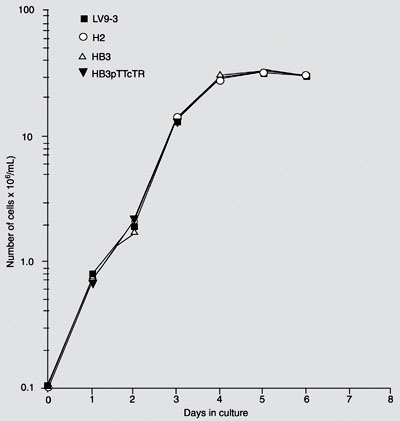
Growth curves of both wild-type (LV9-3) and L. donovani mutants (clones H2-tryR-/+/+, HB3-tryR-/-/+ and HB3-pTTcTR) show the typical log and stationary growth phases. A similar pattern of continual growth was observed for all transfectants (Figure 6).
Intracellular glutathione (GSH) concentrations in different Leishmania species. Total glutathione (GSH + oxidized glutathione (GSSG)) was measured in lysates of 1 x 107 promastigotes of different Leishmania species (L. amazonensis, L. braziliensis, L. donovani, and L. major). Data are reported as means ± SEM of four replicate cultures and are representative of three experiments. *P < 0.05 compared with L. amazonensis; #P < 0.05 compared with L. donovani; +P < 0.05 compared with L. major (ANOVA followed by Bonferroni's t-test).
Concentrations of non-protein low-molecular weight thiols in different Leishmania species. Total non-protein thiols (glutathione, T(SH)2, cysteine, and ovothiol) were measured in lysates of 1 x 107 promastigotes of Leishmania species (L. amazonensis, L. braziliensis, L. donovani, and L. major). Data are reported as means ± SEM of four replicate cultures and are representative of three experiments.
Cytotoxic effect of S-nitroso-N-acetyl-D,L-penicillamine (SNAP) on different Leishmania species. Promastigote forms of L. amazonensis, L. braziliensis, L. donovani, and L. major (3 x 106 cells/well) were incubated in M199 medium (control) or M199 plus SNAP (30-1000 µM). The cultures were pulsed with 3[H]-thymidine 12 h after SNAP treatment. Leishmania survival was determined after 24 h of further culture by the ability of residual live parasites to incorporate 3[H]-thymidine. Data are reported are means ± SEM of four replicates and are representative of three experiments. Statistically significant different with *P < 0.05 compared with L. amazonensis, #P < 0.05 compared with L. donovani, and +P < 0.05 compared with L. major (ANOVA followed by Bonferroni's t-test).
Cytotoxic effect of S-nitroso-N-acetyl-D,L-penicillamine (SNAP) on L. donovani viability. Effects of the number of tryR allele promastigotes (3 x 106) of wild-type L. donovani (LV9-3: tryR+/+/+) and clones H2 (tryR-/+/+), HB3 (tryR-/-/+) and HB3-pTTcTR (tryR-/-/+ plus pTEXTcTR episomic) were incubated in M199 medium (control) or M199 plus SNAP (30-300 µM) for 12 h. Leishmania survival was determined after 24 h of further culture by the ability of residual live parasites to incorporate 3[H]-thymidine. Data are reported as means ± SEM of four replicates and are representative of three experiments. *P < 0.05 compared with the parental clone (LV9-3; ANOVA followed by Bonferroni's t-test).
Intracellular glutathione (GSH) levels in L. donovani trypanothione reductase (tryR) mutants. Total glutathione (GSH + oxidized glutathione (GSSG)) was measured in lysates of 1 x 107 promastigotes of wild-type L. donovani (LV9-3: tryR+/+/+) and clones H2 (tryR-/+/+), HB3 (tryR-/-/+), and HB3-pTTcTR (tryR-/-/+ plus pTEXTcTR episomic). Data are reported as means ± SEM of four replicate cultures and are representative of three experiments.
Growth curve of wild-type L. donovani (LV9-3: tryR+/+/+) and clones H2 (tryR-/+/+), HB3 (tryR-/-/+), and HB3-pTTcTR (tryR-/-/+ plus pTEXTcTR episomic). Each point represents the average of counts from two cultures, initially inoculated with 1 x 105 organisms/mL in M199 medium.
Discussion
The results presented in this study suggest that both glutathione and T(SH)2/TryR antioxidant systems are important components of the protective mechanisms of Leishmania against the cytotoxic effects of nitrogen-derived reactive species. This conclusion is supported by the following observations: a) the sensitivity of distinct species of Leishmania (L. amazonensis, L. braziliensis, L. donovani, and L. major) to SNAP, a nitrogen-derived reactive species donor, correlated inversely with their cellular glutathione levels but not with their total non-protein low-molecular weight thiol content; b) mutants of L. donovani possessing only one wild-type tryR allele, tryR-/-/+ (submitted to a double replacement of the tryR gene locus), had a decreased capacity to survive in the presence of SNAP. Nevertheless, under normal conditions of in vitro growth, the L. donovani wild-type and recombinant mutants have similar levels of
glutathione.
Our data are consistent with Lemesre et al. (20) who demonstrated that amastigote forms of L. amazonensis, which have higher levels of glutathione, are particularly resistant to NO killing. It is recognised that similar molecular targets of NO are present in microorganisms and in mammalian cells (21,22) and, it has been demonstrated that glutathione protects several mammalian cells, including macrophages, against the oxidative stress induced by oxygen (23) and nitrogen-derived oxidants (11,24,25). Thus, the different intracellular glutathione levels may explain the large differences in susceptibility to SNAP among different species of Leishmania. The observation that the levels of non-protein thiols were similar in all Leishmania species analysed supports this conclusion. Furthermore, it has been demonstrated that buthionine sulfoximine, an inhibitor of glutathione synthesis, protects mice from Trypanosoma brucei infections by lowering the parasites' glutathione and protein thiol levels (26). In addition, conjugation of electrophilic drugs with glutathione via glutathione S-transferase has been shown to be an important detoxification system in T. cruzi epimastigotes (27).
The mechanism by which glutathione is implicated in the protection of trypanosomatid protozoa against the cytotoxic effects of nitrogen-derived oxidants is not clear. However, there is evidence that biological NO-derived oxidants such as nitrogen dioxide and carbonate radical anion dioxide promote cell injury (28-30). These events depend on the intracellular concentration of low-molecular weight thiols such as glutathione (24,31,32). Furthermore, glutathione and protein-cysteines are the preferred targets of NO-derived oxidants being oxidised to the respective thyil radicals (24,33).
T(SH)2, a glutathione and spermidine conjugate, is one of the unique features of Kinetoplastida. In trypanosomatids, the existence of TryR instead of glutathione reductase suggested that the thiol pool in these organisms is dependent on TryR (3). Thus, TryR has long been regarded as a target molecule for selective attack of the trypanosomatids. Our data demonstrate that the L. donovani recombinant HB3 clone possessing only one wild-type tryR allele, retaining only one-third of its original TryR activity (12), has a partially decreased capacity to survive in the presence of SNAP. Furthermore, this mutant and the L. donovani mutants TR/TR::hyg and TR/TR::hyg/TR::neo, which also carry a single tryR allele, have been shown to be impaired in their ability to survive inside cytokine-activated mouse and human macrophages (12,19). Significantly, when the HB3 clone was electroporated with the plasmid pTTcTR, this recombinant strain (HB3-pTTcTR) regained its ability to resist SNAP cytotoxicity to approximately wild-type levels. It is important to note that the HB3 mutant clone possesses only one tryR allele, whilst the parental clone (LV9-3) possesses three allelic copies of the gene (12). Despite this, the parental and all mutant lines used in the present study proliferated at equivalent rates under the conditions used.
The fact that the tryR mutant HB3 showed only a partial decrease in the survival rate (~40%) when incubated with low doses of SNAP suggests that the remaining TryR activity present in this clone may still provide important antioxidant protection. This finding is consistent with the observation that Leishmania cells that overexpress a trans-dominant mutant version of tryR and as a result retain only about 15% of TryR activity, are not significantly impaired in their ability to metabolise hydrogen peroxide in vitro and are able to maintain wild-type levels of thiols under normal conditions of growth (34). It is possible that GSH may also contribute significantly to the residual antioxidant activity observed in clone HB3, as discussed above. In this context, we observed that all tryR mutant clones, despite having a reduction in TryR activities, present similar glutathione levels compared to L. donovani wild-type cells. This finding is in accordance with previous data demonstrating that L. donovani wild-type and tryR mutants with low levels of TryR catalytic activity did not differ in their levels of T(SH)2, glutathionyl-spermidine or glutathione (19, 35). Levels of reduced thiols (e.g., GSH, T(SH)2) may only become limiting under conditions of oxidative stress in the context of a compromised T(SH)2/TryR antioxidant system (12,19). An intriguing aspect of the L. donovani mutants that needs further investigation is that, in contrast to wild-type L. donovani (Figure 3), their sensitivity to SNAP was not dose-dependent.
Trypanosomatids also produce significant amounts of ovothiol A (36) which has recently been shown to catalytically accelerate nitrosothiol, both GSNO and T(SNO)2, decomposition to NO (37). The rate-limiting step in this reaction is the reduction of oxidised ovothiol which in Leishmania is mediated indirectly via trypanothione/trypanothione reductase, as is also the case for oxidized glutathione (GSSG) (5). Thus, we cannot exclude the possibility that the difference in cytotoxic potency of SNAP observed here between different tryR clones of Leishmania is dependent on variable ovothiol A concentrations and hence on nitrogen-free radical production.
The present data suggest that both antioxidant systems, glutathione and T(SH)2/TryR, are involved in protecting Leishmania against the toxic effect of nitrogen-derived reactive species.
Acknowledgments
We thank Ms. Giuliana Bertozi, Ms. Ana Kátia dos Santos, and Ms. Fabíola Leslie Mestriner for technical assistance (FMRP-USP) and Jamil Assreuy (Department of Pharmacology, UFSC) for helping with non-protein low-molecular weight thiol measurement. We are grateful to Professor A.H. Fairlamb (University of Dundee, UK) for providing L. donovani TryR mutants.
 Address for correspondence: P.R.T. Romão, Laboratório de Imunoparasitologia, Curso de Medicina, UNISUL, Rua José Acácio Moreira, 787, DEHON, 88704-900 Tubarão, SC, Brasil. Fax: +55-48-621-3067. E-mail: ptorres@unisul.br
Address for correspondence: P.R.T. Romão, Laboratório de Imunoparasitologia, Curso de Medicina, UNISUL, Rua José Acácio Moreira, 787, DEHON, 88704-900 Tubarão, SC, Brasil. Fax: +55-48-621-3067. E-mail: ptorres@unisul.br
Research supported by FAPESP (No. 97/01938-8). P.R.T. Romão and S.G. Fonseca were recipients of FAPESP fellowships. F.Q. Cunha, S.H. Ferreira, and R.H. Moraes were recipients of CNPq fellowships. Received January 21, 2005. Accepted November 11, 2005.
- 1. Molyneux DH (1997). Current public health status of the Trypanosomiases and Leishmaniases. In: Hide G, Mottran JC, Coombs GH et al. (Editors), Trypanosomiasis and Leishmaniasis: Biology and Control CAB International, Oxford, UK.
- 2. Fairlamb AH, Blackburn P, Ulrich P et al. (1985). Trypanothione: a novel bis (glutathionyl) permidine cofactor for glutathione reductase in trypanosomatids. Science, 227: 1485-1487.
- 3. Fairlamb AH & Cerami A (1992). Metabolism and functions of trypanothione in the kinetoplastida. Annual Review of Microbiology, 46: 695-729.
- 4. Nogoceke E, Gommel DU, Kiess M et al. (1997). A unique cascade of oxidoreductases catalyses trypanothione-mediated peroxide metabolism in Crithidia fasciculata Biological Chemistry, 378: 827-836.
- 5. Ariyanayagam MR & Fairlamb AH (2001). Ovothiol and trypanothione as antioxidants in trypanosomatids. Molecular and Biochemical Parasitology,115: 189-198.
- 6. Mukhopadhyay R, Dey S, Xu N et al. (1996). Trypanothione overproduction and resistance to antimonials and arsenicals in Leishmania tarentolae Proceedings of the National Academy of Sciences, USA, 93: 10383-10387.
- 7. Liew FY, Li Y & Millott S (1990). Tumor necrosis factor-a synergizes with IFN-g in mediating killing of Leishmania major through the induction of nitric oxide. Journal of Immunology, 145: 4306-4310.
- 8. Liew FY, Millott S, Parkinson C et al. (1990). Macrophage killing of Leishmania parasite in vivo is mediated by nitric oxide from L-arginine. Journal of Immunology, 144: 4794-4797.
- 9. Stenger N, Donhauser H, Thüring M et al. (1996). Reactivation of latent leishmaniasis by inhibition of inducible nitric oxide synthase. Journal of Experimental Medicine, 183: 1501-1514.
- 10. Assreuy J, Cunha FQ, Epperlein M et al. (1994). Production of nitric oxide and superoxide by activated macrophages and killing of Leishmania major. European Journal of Immunology, 24: 672-676.
- 11. Romão PRT, Fonseca SG, Hothersall JS et al. (1999). Glutathione protects macrophages and Leishmania major against nitric oxide-mediated cytotoxicity. Parasitology, 118: 559-566.
- 12. Tovar J, Wilkinson S, Mottram JC et al. (1998). Evidence that trypanothione reductase is an essential enzyme in Leishmania by targeted replacement of the tryA locus. Molecular Microbiology, 29: 653-660.
- 13. Tovar J & Fairlamb AH (1986). Extrachromosomal, homologous expression of trypanothione reductase and its complementary mRNA in Trypanosoma cruzi. Nucleic Acids Research, 24: 2942-2949.
- 14. Kapler GM, Coburn CM & Beverley SM (1990). Stable transfection of the human parasite Leishmania major delineates a 30-kilobase region sufficient for extrachromosomal replication and expression. Molecular and Cellular Biology, 10: 1084-1094.
- 15. Ellman GL & Lysko H (1967). Disulphide and sulfhydryl compounds in TCA extracts of human blood and plasma. Journal of Laboratory and Clinical Medicine, 70: 518-527.
- 16. Tietze F (1969). Enzymatic method for quantitative determination of nanogram amounts of total and oxidised glutathione: applications to mammalian blood and other tissues. Analytical Biochemistry, 27: 502-522.
- 17. Hothersall JS, Cunha FQ, Neild GH et al. (1997). Induction of nitric oxide synthesis in J774 cells lowers intracellular glutathione: effect of modulated glutathione redox status on nitric oxide synthase induction. Biochemical Journal, 322: 477-481.
- 18. Zhang Y & Hogg N (2004). The mechanism of transmembrane S-nitrosothiol transport. Proceedings of the National Academy of Sciences, USA, 101: 7891-7896.
- 19. Dumas C, Ouellette M, Tovar J et al. (1997). Disruption of the trypanothione reductase gene of Leishmania decreases its ability to survive oxidative stress in macrophages. EMBO Journal, 16: 2590-2598.
- 20. Lemesre JL, Sereno D, Daulouède S et al. (1997). Leishmania spp: nitric oxide metabolic inhibition of promastigote and axenically grown amastigote forms. Experimental Parasitology, 86: 58-68.
- 21. Granger DL, Taintor R, Cook J et al. (1980). Injury of neoplastic cells by murine macrophages leads to inhibition of mitochondrial respiration. Journal of Clinical Investigation, 65: 357-370.
- 22. Hibbs Jr JB, Vavrin Z & Taintor RR (1987). L-arginine is required for expression of the activated macrophage effector mechanism causing selective metabolic inhibition in target cells. Journal of Immunology, 138: 550-565.
- 23. Minhas HS & Thornalley PJ (1995). Reduced glutathione esters antidotes to toxicity. Cytotoxicity induced by hydrogen peroxide, 1-chloro-2-4-dinitrobenzene, and menadione in murine P388D1 macrophages in vitro Journal of Biochemical Toxicology, 10: 245-250.
- 24. Augusto O, Bonini MG & Trindade DF (2004). Spin trapping of glutathiyl and protein radicals produced from nitric oxide-derived oxidants. Free Radical Biology and Medicine, 36: 1224-1232.
- 25. Grinberg L, Fibach E, Amer J et al. (2005). N-acetylcysteine amide, a novel cell-permeating thiol, restores cellular glutathione and protects human red blood cells from oxidative stress. Free Radical Biology and Medicine, 38: 136-145.
- 26. Arrick BA, Griffith OW & Cerami A (1981). Inhibition of glutathione synthesis as a chemotherapeutic strategy for trypanosomiasis. Journal of Experimental Medicine, 153: 720-725.
- 27. Yawetz A & Agosin M (1980). Glutathione S-transferase and drug metabolism in Trypanosoma cruzi: in vivo and in vitro formation of thioethers. Comparative Biochemistry and Physiology. C, Comparative Pharmacology and Toxicology, 66C: 265-267.
- 28. Hurst JK & Lymar SV (1995). Rapid reaction between peroxynitrite and carbon dioxide: implications for biological activity. Journal of the American Chemical Society, 117: 8867-8868.
- 29. Bonini MG, Radi R, Ferrer-Sueta et al. (1999). Direct EPR detection of the carbonate radical anion produced from peroxynitrite and carbon dioxide. Journal of Biological Chemistry, 274: 10802-10806.
- 30. Linares E, Giorgio S, Mortara RA et al. (2001). Role of peroxynitrite in macrophage microbicidal mechanisms in vivo revealed by protein nitration and hydroxylation. Free Radical Biology and Medicine, 30: 1234-1242.
- 31. Bonini MG & Augusto O (2001). Carbon dioxide stimulates the production of thyil, sulfinyl and disulphide radical anion from thiol oxidation by peroxynitrite. Journal of Biological Chemistry, 276: 9749-9754.
- 32. Radi R (2004). Nitric oxide, oxidants, and protein tyrosine nitration. Proceedings of the National Academy of Sciences, USA, 101: 4003-4008.
- 33. Lopes de Menezes S & Augusto O (2001). EPR detection of glutathionyl and protein-tyrosyl radicals during the interaction of peroxynitrite with macrophages (J774). Journal of Biological Chemistry, 276: 39879-39884.
- 34. Tovar J, Cunningham ML, Smith AC et al. (1998). Down-regulation of Leishmania donovani trypanothione reductase by heterologous expression of a trans-dominant mutant homologue: Effect on parasite intracellular survival. Proceedings of the National Academy of Sciences, USA, 95: 5311-5316.
- 35. Kelly JM, Taylor MC, Smith K et al. (1993). Phenotype of recombinant Leishmania donovani and Trypanosoma cruzi which overexpress trypanothione reductase: sensitivity towards agents that are thought to induce oxidative stress. European Journal of Biochemistry, 218: 29-37.
- 36. Steenkamp DJ & Spies HS (1994). Identification of a major low-molecular-mass thiol of the trypanosomatid Crithidia fasciculata as ovothiol A. Facile isolation and structural analysis of the bimane derivative. European Journal of Biochemistry, 223: 43-50.
- 37. Vogt RN & Steenkamp DJ (2003). The metabolism of S-nitrosothiols in the trypanosomatids: the role of ovothiol A and trypanothione. Biochemical Journal, 371: 49-59.
Correspondence and Footnotes
Publication Dates
-
Publication in this collection
06 Mar 2006 -
Date of issue
Mar 2006
History
-
Accepted
11 Nov 2005 -
Received
21 Jan 2005