Abstract
We have observed that intracerebroventricular (icv) injection of selective N-methyl-D-aspartic acid (NMDA)-type glutamatergic receptor antagonists inhibits lordosis in ovariectomized (OVX), estrogen-primed rats receiving progesterone or luteinizing hormone-releasing hormone (LHRH). When NMDA was injected into OVX estrogen-primed rats, it induced a significant increase in lordosis. The interaction between LHRH and glutamate was previously explored by us and another groups. The noradrenergic systems have a functional role in the regulation of LHRH release. The purpose of the present study was to explore the interaction between glutamatergic and noradrenergic transmission. The action of prazosin, an alpha1- and alpha2b-noradrenergic antagonist, was studied here by injecting it icv (1.75 and 3.5 µg/6 µL) prior to NMDA administration (1 µg/2 µL) in OVX estrogen-primed Sprague-Dawley rats (240-270 g). Rats manually restrained were injected over a period of 2 min, and tested 1.5 h later. The enhancing effect induced by NMDA on the lordosis/mount ratio at high doses (67.06 ± 3.28, N = 28) when compared to saline controls (6 and 2 µL, 16.59 ± 3.20, N = 27) was abolished by prazosin administration (17.04 ± 5.52, N = 17, and 9.33 ± 3.21, N = 20, P < 0.001 for both doses). Plasma LH levels decreased significantly only with the higher dose of prazosin (1.99 ± 0.24 ng/mL, N = 18, compared to saline-NMDA effect, 5.96 ± 2.01 ng/mL, N = 13, P < 0.05). Behavioral effects seem to be more sensitive to the alpha-blockade than hormonal effects. These findings strongly suggest that the facilitatory effects of NMDA on both lordosis and LH secretion in this model are mediated by alpha-noradrenergic transmission.
Sexual behavior; Glutamate; Luteinizing hormone; Noradrenergic transmission; Prazosin; N-methyl-D-aspartic acid
Braz J Med Biol Res, March 2006, Volume 39(3) 365-370
Prazosin blocks the glutamatergic effects of N-methyl-D-aspartic acid on lordosis behavior and luteinizing hormone secretion in the estrogen-primed female rat
A.I. Landa1, R.J. Cabrera2 and  Correspondence and Footnotes
Correspondence and Footnotes
 P.A. Gargiulo1
P.A. Gargiulo1
1Laboratorio de Neurociencias y Psicología Experimental, Cátedra de Psicopatología, Facultad de Humanidades y Ciencias de la Educación, Universidad Católica Argentina, Área de Farmacología, Facultad de Ciencias Médicas, Universidad Nacional de Cuyo, Instituto de Medicina y Biología Experimental de Cuyo, Mendoza, Argentina
2Laboratorio de Investigaciones Neuroquímicas, Comportamentales y Endocrinas, Área de Farmacología, Facultad de Ciencias Médicas, Universidad Nacional de Cuyo, Instituto de Medicina y Biología Experimental de Cuyo, Mendoza, Argentina
 References
References
 Correspondence and Footnotes
Correspondence and Footnotes
Correspondence and Footnotes
Correspondence and Footnotes
Correspondence and Footnotes
Correspondence and Footnotes
Abstract
We have observed that intracerebroventricular (icv) injection of selective N-methyl-D-aspartic acid (NMDA)-type glutamatergic receptor antagonists inhibits lordosis in ovariectomized (OVX), estrogen-primed rats receiving progesterone or luteinizing hormone-releasing hormone (LHRH). When NMDA was injected into OVX estrogen-primed rats, it induced a significant increase in lordosis. The interaction between LHRH and glutamate was previously explored by us and another groups. The noradrenergic systems have a functional role in the regulation of LHRH release. The purpose of the present study was to explore the interaction between glutamatergic and noradrenergic transmission. The action of prazosin, an a1- and a2b-noradrenergic antagonist, was studied here by injecting it icv (1.75 and 3.5 µg/6 µL) prior to NMDA administration (1 µg/2 µL) in OVX estrogen-primed Sprague-Dawley rats (240-270 g). Rats manually restrained were injected over a period of 2 min, and tested 1.5 h later. The enhancing effect induced by NMDA on the lordosis/mount ratio at high doses (67.06 ± 3.28, N = 28) when compared to saline controls (6 and 2 µL, 16.59 ± 3.20, N = 27) was abolished by prazosin administration (17.04 ± 5.52, N = 17, and 9.33 ± 3.21, N = 20, P < 0.001 for both doses). Plasma LH levels decreased significantly only with the higher dose of prazosin (1.99 ± 0.24 ng/mL, N = 18, compared to saline-NMDA effect, 5.96 ± 2.01 ng/mL, N = 13, P < 0.05). Behavioral effects seem to be more sensitive to the a-blockade than hormonal effects. These findings strongly suggest that the facilitatory effects of NMDA on both lordosis and LH secretion in this model are mediated by a-noradrenergic transmission.
Key words: Sexual behavior, Glutamate, Luteinizing hormone, Noradrenergic transmission, Prazosin, N-methyl-D-aspartic acid
Introduction
Glutamic acid is the main excitatory amino acid neurotransmitter in the mammalian brain (1). In previous studies, we observed that the activation of lordosis induced by progesterone and luteinizing hormone-releasing hormone (LHRH) in ovariectomized (OVX) estrogen-primed rats can be blocked by the intracerebroventricular (icv) administration of 7-amino-phosphonoheptanoic acid (AP-7), a selective N-methyl-D-aspartic-acid (NMDA)-glutamatergic antagonist (2). The icv administration of NMDA to OVX estrogen-primed rats induces an increase in lordosis and plasma LH (3). This effect on lordosis appears not to be mediated by LHRH, since a selective LHRH antagonist did not affect the lordosis/mount ratio (L/M), but the effect was decreased by AP-7 (3). The endocrine effect appears to be mediated by LHRH, since icv administration of a selective LHRH antagonist blunted the secretion of LH induced by NMDA (3). We previously raised the possibility that the NMDA effects might be mediated by other neurotransmitters (3).
On the basis of these considerations and of the relevance of noradrenergic transmission in lordosis and LH release (4), we investigated the possible involvement of noradrenergic transmission in NMDA-induced effects on these two variables. The aim of the experiments described here was to identify possible interactions between glutamatergic and noradrenergic transmission in lordosis. These interactions were considered because noradrenaline is released by glutamate (5-7). The stimulatory actions of NMDA appear to be hormone dependent (3). Estradiol appears to regulate the number of a1- but not ß- or a2-noradrenergic receptors in the hypothalamus of female rats (8). For this reason, we studied the sexual behavior and LH release of female rats previously treated with different doses of a noradrenergic receptor antagonist in order to establish or not a relationship between behavioral and hormonal effects.
Material and Methods
Animals
Female Sprague-Dawley rats (240-270 g) were ovariectomized under ether anesthesia and kept on a reverse light cycle (lights on from 19:00-7:00 h) in a temperature-controlled (22 ± 2ºC) room. Animals were allowed access to food and water ad libitum. Two weeks prior to the experiments, the receptivity of the rats was determined. Animals were injected subcutaneously (sc) with 20 µg estradiol benzoate (EB) 48 h before testing, and 1 mg progesterone sc 4 h before testing in order to select responsive rats. In the experiments, only EB sc 48 h before testing was used for priming, and progesterone was not administered. Mating tests were conducted in a rectangular arena illuminated by a red light during the dark period, in the morning (8:00-10:00 h). Two stud males (300-400 g) were introduced into the arena at least 10 min before the female. The behavioral tests ended when the test female had received 30 male copulatory acts or after 15 min.
The L/M ratio was established for each rat. Only responsive rats with an L/M of at least 80% (criterion of inclusion) in previous tests were selected for stereotaxic implantation of a 23-gauge stainless steel guide cannula into the 3rd brain ventricle under ether anesthesia. Coordinates for cannula implantation were: A-C: bregma; V: 8.5 mm; lateral: midline (9). Localization of the cannula was confirmed when cerebrospinal fluid flowed from the cannula after removal of an inner stylet used to prevent such leakage. The cannula was cemented to the skull. After surgery, the animals were housed individually and left undisturbed during recovery. Drugs were injected icv through a 30-gauge stainless steel tube fitted into the guide cannula and connected to a 10-µL Hamilton syringe with a polyethylene catheter. In all cases, a volume between 2 and 6 µL of solution was delivered over a period of 2 min. During icv injections, animals were manually restrained. Each animal was tested only once.
Experiments
In the experiments, the effect of prazosin on lordosis induced by NMDA was studied. Rats implanted with an icv guide cannula received an sc injection of 20 µg EB in 0.2 mL corn oil. They were injected 48 h later with saline (SAL, two injections of 6 and 2 µL, respectively), SAL (6 µL) and NMDA (1 µg/2 µL), or prazosin (1.75 and 3.50 µg/6 µL) and NMDA (1 µg/2 µL), and tested for lordosis 1.5 h later. In all cases, the injections were separated by a 10-min interval. In each mating session, rats injected with drugs and controls were tested simultaneously.
Lordosis was evaluated 1.5 h after administration of the drugs. To determine LH release during copulatory behavior, rats were sacrificed by decapitation immediately after completion of the lordosis tests, and blood was collected from the trunk. Rats used for LH determination in blood samples were randomly selected. Plasma was separated and stored frozen (-70ºC) until assay for LH by radioimmunoassay. LH was measured with a double-antibody radioimmunoassay performed with materials and according to the instructions of the kits provided by the National Hormone & Pituitary Program (Harbor-UCLA Medical Center, Torrance, CA, USA). The intra- and inter-assay coefficients of variation were 9 and 11%, respectively. Data are reported as ng/mL serum after comparison to NIDDK-rLH-RP1 reference samples.
Drugs
The following drugs were used: NMDA (Research Biochemicals International, Natick, MA, USA), and an a1- and a2b-adrenoceptor antagonist (10), prazosin (1-(4-amino-6,7-dimethoxy-2-quinazolinyl)-4-(2-furanylcarbonyl) piperazine hydrochloride (Research Biochemicals International). They were dissolved in saline and the pH was adjusted to near physiological pH. EB and progesterone (Sigma-Aldrich, St. Louis, MO, USA) were dissolved in corn oil.
Data analysis
The L/M data were analyzed by the Kruskal-Wallis test followed by the Dunn test. Hormone values were analyzed by ANOVA 1 followed by the Student Newman-Keuls test. In all cases, the level of significance was set at P < 0.05. Data are reported as means ± SEM.
Results
L/M ratios were modified by injection of the drugs tested (Kruskal-Wallis = 53.89, d.f. = 3). The injection of NMDA (SAL-NMDA, N = 28) increased the L/M ratio to a very significant extent compared to control (SAL-SAL, N = 27, P < 0.001, Figure 1). The injection of both doses of prazosin (N = 17 for the lower dose, and N = 20 for the higher dose) decreased the scores to control levels (P < 0.001, Figure 1).
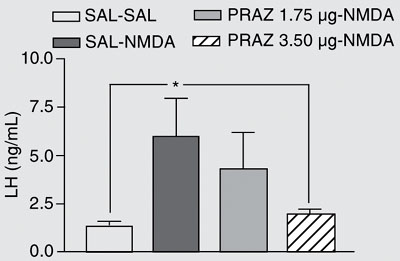
Plasma LH concentrations were modified by the treatments [(F = 3,461, d.f. treatment = 3, d.f. residual = 45)]. They were significantly increased by NMDA (SAL-NMDA, N = 13, P < 0.05) compared to control (SAL-SAL, N = 12, Figure 2). The administration of prazosin at the lower dose (1.75 µg prazosin-NMDA, N = 6) did not significantly reduce the increase of LH values induced by NMDA. No differences were observed between the lower dose of prazosin and the SAL-NMDA group, or the SAL-SAL group. However, a significant decrease in LH values was observed when the higher dose of prazosin was administered (3.5 µg prazosin-NMDA, N = 18, P < 0.05) compared to the NMDA group (SAL-NMDA), leading to plasma concentrations of LH that were equivalent to those of controls.
Prazosin inhibition of the NMDA-induced increase of lordosis in ovariectomized estradiol-primed female rats. SAL = saline; NMDA = N-methyl-D-aspartic acid; PRAZ = prazosin. Groups: SAL (6 µL)-SAL (2 µL), N = 27; SAL (6 µL)-NMDA (1 µg/2 µL), N = 28; PRAZ (1.75 µg/6 µL)-NMDA (1 µg/2 µL), N = 17; PRAZ (3.50 µg/6 µL)-NMDA (1 µg/2 µL), N = 20. *P < 0.001 compared to other groups (Kruskal-Wallis test followed by Dunn test).
Prazosin inhibition of the NMDA-induced increase in plasma luteinizing hormone (LH) values in ovariectomized estradiol-primed female rats. SAL = saline; NMDA = N-methyl-D-aspartic acid; PRAZ = prazosin. Groups: SAL (6 µL)-SAL (2 µL), N = 12; SAL (6 µL)-NMDA (1 µg/2 µL), N = 13; PRAZ (1.75 µg/6 µL)-NMDA (1 µg/2 µL), N = 6; PRAZ (3.50 µg/6 µL)-NMDA (1 µg/2 µL), N = 18. *P < 0.05 compared to SAL-SAL and PRAZ 3.50 µg-NMDA groups (ANOVA 1 test followed by Student Newman-Keuls test).
Discussion
The results presented here support the view that the enhancing effects of NMDA glutamatergic neurotransmission on mating behavior and LH release are mediated by a- noradrenergic transmission, thus extending our previous observations (3). The increase in L/M ratio induced by NMDA was clearly decreased by prazosin administration (P < 0.001 for both doses). Plasma LH levels were decreased only by the higher dose of prazosin (P < 0.05). Another interesting finding is that behavioral effects seem to be more sensitive to adrenergic blockade than the hormonal effects. In other studies, the role of a1-receptors has been linked to lordosis and LH release (4). Prazosin has been used in female Rhesus macaques to antagonize NMDA-induced LH release (11). In the cited study (11), however, copulatory behavior was not investigated and drugs were administered during the luteal phase of the menstrual cycle, before administration of NMDA. The present study extends these results, establishing a comparison between behavioral and hormonal effects.
We have observed that the enhancement of lordosis induced by NMDA is not mediated by LHRH, which is involved in NMDA-mediated endocrine effects (3). The present findings support the idea that a noradrenergic mechanism could mediate these effects. The hormonal status of the animals appears to play an important role since, under our previous experimental conditions, in OVX female rats not primed with EB, NMDA had no effects on lordosis and decreased plasma LH levels (3). Estradiol priming increases a1-receptors, which have facilitatory effects on the parameters considered here (4,8,12). The effects of NMDA could be mediated by noradrenaline release since a1-blockade interferes with the facilitatory effects on mating behavior and plasma LH values (4). Excitatory pathways appear to mediate these effects by a1-adrenoceptors, and these receptors use cyclic adenosine monophosphate as second messenger (12). The blockade of LH secretion by a1-adrenergic receptor antagonists has been described (13), and brainstem catecholaminergic neurons are activated by mating in the female rat (14). The different influences of hormonal status on noradrenergic receptors may explain why both stimulatory (3) and inhibitory (15,16) effects of excitatory amino acids on lordosis have been reported.
Axo-axonic contacts and stimulation of distal neurones could be mediating these effects. Excitatory amino acids are present in the presynaptic boutons of several important hypothalamic nuclei (1), and noradrenaline is released in response to excitatory amino acids in the rat mediobasal hypothalamus (5-7) and at other brain sites. Glutamate-stimulated noradrenaline release has been described in minislices of the bed nucleus of the stria terminalis (17) and superfused rat hippocampal synaptosomes, suggesting the presence of NMDA receptors in hippocampal noradrenergic axon terminals (18). Similar results have also been observed in slices of the prefrontal cortex (19). These findings support the idea that functions elicited by NMDA receptors are mediated by noradrenergic release in several brain areas.
It is possible that glutamatergic neurons act on distal noradrenergic neurons (through axo-somatic contacts), since applying NMDA to the locus ceruleus (LC) increases noradrenaline release in other brain areas (20). Conversely, LC lesions decrease noradrenaline input into the medial preoptic area and medial basal hypothalamus, blocking the LH, follicle-stimulating hormone and prolactin preovulatory surge (21). The activation by genital-somatosensory signals (22) of midbrain and brainstem noradrenergic neurons, which project to the mediobasal hypothalamus, promotes the release of LHRH from nerve terminals in the median eminence. Similarly, it has recently been pointed out that LHRH release depends, at least in part, on LC noradrenergic inputs to the medial preoptic area and median eminence by activation of LHRH neurons (23). Furthermore, the LC appears to have an intrinsic cyclic activity which is amplified by ovarian steroids (23). Interaction of NMDA and noradrenaline, and noradrenaline and LHRH, appears to occur in different brain areas. In the present study we injected the drugs icv, but in future studies it would be enlightening to define the zones involved more precisely.
The present results strongly suggest the noradrenergic mediation of facilitatory NMDA-induced effects on lordosis and plasma LH values. Our findings support the idea that the stimulatory effect of NMDA in this model could be mediated by a-noradrenergic receptors.
Acknowledgments
We thank E. Morsucci, D.M. Videla, A. Funes, A. Morón, L. Peñafort, and J. González (Laboratorio de Neurociencias y Psicología Experimental) for technical support, and Dr. Graciela Jahn (IMBECU-CONICET) for the LH assay. We thank Dr. Yunes (IMBECU-CONICET, and Area de Farmacología, FCM, UNC) for his comments on the present paper. We thank Dr. Juan Delius (University of Konstanz, Germany) for his constant support and counseling. We thank Prof. Dr. Sean Patterson (Area de Fisiología, FCM, UNC) for style corrections.
 Address for correspondence: P.A. Gargiulo, Área de Farmacología, Facultad de Ciencias Médicas, Instituto de Medicina y Biología, Experimental de Cuyo, Universidad Nacional de Cuyo, Casilla de Correo 7, Mendoza (5500), Argentina. E-mail: gargiulo@lab.cricyt.edu.ar
Address for correspondence: P.A. Gargiulo, Área de Farmacología, Facultad de Ciencias Médicas, Instituto de Medicina y Biología, Experimental de Cuyo, Universidad Nacional de Cuyo, Casilla de Correo 7, Mendoza (5500), Argentina. E-mail: gargiulo@lab.cricyt.edu.ar
P.A. Gargiulo and R.J. Cabrera are established researchers of the National Council of Research (CONICET) of Argentina. A.I. Landa is Research Coordinator of the Laboratory of Neurosciences and Experimental Psychology. Research supported by CONICET (PEI No. 0386/97) and by a grant from the Volkswagen Foundation, Germany (Project "Nucleus Accumbens Septi und kognitives Verhalten", Delius-Gargiulo). This study is part of a cooperation with the Laboratory of General Psychology, Universitiy of Konstanz, Germany. Received January 31, 2005. Accepted December 6, 2005.
- 1. Brann DW & Mahesh VB (1994). Excitatory amino acids: function and significance in reproduction and neuroendocrine regulation. Frontiers in Neuroendocrinology, 15: 3-49.
- 2. Gargiulo PA, Muñoz V & Donoso AO (1992). Inhibition by NmethylDaspartic acid (NMDA) receptor antagonist of lordosis behavior induced by estrogen followed by progesterone or luteinizing hormone releasing hormone (LHRH) in the rat. Physiology and Behavior, 52: 737739.
- 3. Gargiulo PA & Donoso AO (1995). Interaction between glutamate and luteinizing hormone releasing hormone (LHRH) in lordosis behavior and luteinizing hormone release (LH): further studies on NMDA receptor mediation. Physiology and Behavior, 58: 169173.
- 4. Etgen AM (2003). Ovarian steroid and growth factor regulation of female reproductive function involves modification of hypothalamic alpha 1-adrenoceptor signaling. Annals of the New York Academy of Sciences, 1007: 153-161.
- 5. Navarro CE, Cabrera RJ & Donoso AO (1994). Release of 3H-noradrenaline by excitatory amino acids from rat mediobasal hypothalamus and the influence of aging. Brain Research Bulletin, 33: 677-682.
- 6. Navarro CE, Cabrera RJ & Donoso AO (1995). Interaction between glutamate and GABA on 3H-noradrenaline release from rat hypothalamus. Brain Research Bulletin, 37: 119-122.
- 7. Donoso AO, Seltzer AM, Navarro CE et al. (1994). Regulation of luteinizing hormone-releasing hormone and luteinizing hormone secretion by hypothalamic amino acids. Brazilian Journal of Medical and Biological Research, 27: 921-932.
- 8. Etgen AM & Karkanias GB (1990). Estradiol regulates the number of a1 but not ß or a2 noradrenergic receptors in hypothalamus of female rats. Neurochemistry International, 16: 1-9.
- 9. Pellegrino LJ, Pellegrino AS & Cushman AJ (1979). A Stereotaxic Atlas of the Rat Brain Plenum Press, New York.
- 10. Mathe JM, Nomikos GG, Hildebrand BE et al. (1996). Prazosin inhibits MK-801-induced hyperlocomotion and dopamine release in the nucleus accumbens. European Journal of Pharmacology, 309: 1-11.
- 11. Urbanski HF, Garyfallou VT, Kohama SG et al. (1997). Alpha-adrenergic receptor antagonism and N-methyl-D-aspartate (NMDA) induced luteinizing hormone release in female rhesus macaques. Brain Research, 744: 96-104.
- 12. Etgen AM, Ungar S & Petitti N (1992). Estradiol and progesterone modulation of norepinephrine neurotransmission: implications for the regulation of female reproductive behavior. Journal of Neuroendocrinology, 4: 255-271.
- 13. Le WW, Berghorn KA, Smith MS et al. (1997). Alpha 1-adrenergic receptor blockade blocks LH secretion but not LHRH cFos activation. Brain Research, 747: 236-245.
- 14. Yang SP & Voogt JL (2001). Mating-activated brainstem catecholaminergic neurons in the female rat. Brain Research, 894: 159-166.
- 15. Kow LM, Harlan RE, Shivers BD et al. (1985). Inhibition of the lordosis reflex in rats by intrahypothalamic infusion of neural excitatory agents: Evidence that the hypothalamus contains separate inhibitory and facilitatory elements. Brain Research, 341: 26-34.
- 16. McCarthy MM, Curran GH & Feder HH (1991). Excitatory amino acid modulation of lordosis in the rat. Neuroscience Letters, 126: 94-97.
- 17. Forray MI, Andres ME, Bustos G et al. (1995). Regulation of endogenous noradrenaline release from the bed nucleus of stria terminalis. Biochemical Pharmacology, 49: 687-692.
- 18. Risso F, Grilli M, Parodi M et al. (2004). Nicotine exerts a permissive role on NMDA receptor function in hippocampal noradrenergic terminals. Neuropharmacology, 47: 65-71.
- 19. Russell VA & Wiggins TM (2000). Increased glutamate-stimulated norepinephrine release from prefrontal cortex slices of spontaneously hypertensive rats. Metabolic Brain Disease, 15: 297-304.
- 20. Kawahara H, Kawahara Y & Westerink BH (2001). The noradrenaline-dopamine interaction in the rat medial prefrontal cortex studied by multi-probe microdialysis. European Journal of Pharmacology, 418: 177-186.
- 21. Anselmo-Franci JA, Franci CR, Krulich L et al. (1997). Locus coeruleus lesions decrease norepinephrine input into the medial preoptic area and medial basal hypothalamus and block the LH, FSH and prolactin preovulatory surge. Brain Research, 767: 289-296.
- 22. Bakker J & Baum MJ (2000). Neuroendocrine regulation of GnRH release in induced ovulators. Frontiers in Neuroendocrinology, 21: 220-262.
- 23. Martins-Afferri MP, Ferreira-Silva IA, Franci CR et al. (2003). LHRH release depends on locus coeruleus noradrenergic inputs to the medial preoptic area and median eminence. Brain Research Bulletin, 61: 521-527.
Correspondence and Footnotes
Publication Dates
-
Publication in this collection
06 Mar 2006 -
Date of issue
Mar 2006
History
-
Accepted
06 Dec 2005 -
Received
31 Jan 2005