Abstract
The C/T-13910 mutation is the major factor responsible for the persistence of the lactase-phlorizin hydrolase (LCT) gene expression. Mutation G/A-22018 appears to be only in co-segregation with C/T-13910. The objective of the present study was to assess the presence of these two mutations in Brazilian individuals with and without lactose malabsorption diagnosed by the hydrogen breath test (HBT). Ten milk-tolerant and 10 milk-intolerant individuals underwent the HBT after oral ingestion of 50 g lactose (equivalent to 1 L of milk). Analyses for C/T-13910 and G/A-22018 mutations were performed using a PCR-based method. Primers were designed for this study based on the GenBank sequence. The CT/GA, CT/AA, and TT/AA genotypes (lactase persistence) were found in 10 individuals with negative HBT. The CC/GG genotype (lactase non-persistence) was found in 10 individuals, 9 of them with positive HBT results. There was a significant agreement between the presence of mutations in the LCT gene promoter and HBT results (kappa = -0.9, P < 0.001). The CT/AA genotype has not been described previously and seems to be related to lactase persistence. The present study showed a significant agreement between the occurrence of mutations G/A-22018 and C/T-13910 and lactose absorption in Brazilian subjects, suggesting that the molecular test used here could be proposed for the laboratory diagnosis of adult-type primary hypolactasia.
Hydrogen breath test; Lactase-phlorizin hydrolase; Lactose intolerance; Milk intolerance; LCT polymorphism
Braz J Med Biol Res, November 2007, Volume 40(11) 1441-1446
Correlation between lactose absorption and the C/T-13910 and G/A-22018 mutations of the lactase-phlorizin hydrolase (LCT) gene in adult-type hypolactasia
 Correspondence and Footnotes
Correspondence and Footnotes
 A.C. Bulhões1, H.A.S. Goldani1,2, F.S. Oliveira3, U.S. Matte3, R.B. Mazzuca1 and T.R. Silveira1,2
A.C. Bulhões1, H.A.S. Goldani1,2, F.S. Oliveira3, U.S. Matte3, R.B. Mazzuca1 and T.R. Silveira1,2
1Laboratório Experimental de Gastroenterologia e Hepatologia, 2Unidade de Gastroenterologia Pediátrica, 3Centro de Terapia Gênica, Hospital de Clínicas de Porto Alegre, Universidade Federal do Rio Grande do Sul, Porto Alegre, RS, Brasil
 Correspondence and Footnotes
Correspondence and Footnotes
Correspondence and Footnotes
Correspondence and Footnotes
Correspondence and Footnotes
Correspondence and Footnotes
Abstract
The C/T-13910 mutation is the major factor responsible for the persistence of the lactase-phlorizin hydrolase (LCT) gene expression. Mutation G/A-22018 appears to be only in co-segregation with C/T-13910. The objective of the present study was to assess the presence of these two mutations in Brazilian individuals with and without lactose malabsorption diagnosed by the hydrogen breath test (HBT). Ten milk-tolerant and 10 milk-intolerant individuals underwent the HBT after oral ingestion of 50 g lactose (equivalent to 1 L of milk). Analyses for C/T-13910 and G/A-22018 mutations were performed using a PCR-based method. Primers were designed for this study based on the GenBank sequence. The CT/GA, CT/AA, and TT/AA genotypes (lactase persistence) were found in 10 individuals with negative HBT. The CC/GG genotype (lactase non-persistence) was found in 10 individuals, 9 of them with positive HBT results. There was a significant agreement between the presence of mutations in the LCT gene promoter and HBT results (kappa = -0.9, P < 0.001). The CT/AA genotype has not been described previously and seems to be related to lactase persistence. The present study showed a significant agreement between the occurrence of mutations G/A-22018 and C/T-13910 and lactose absorption in Brazilian subjects, suggesting that the molecular test used here could be proposed for the laboratory diagnosis of adult-type primary hypolactasia.
Key words: Hydrogen breath test, Lactase-phlorizin hydrolase, Lactose intolerance, Milk intolerance, LCT polymorphism
Introduction
Lactose is hydrolyzed in the small intestine into glucose and galactose by the lactase-phlorizin hydrolase (LPH) found in the intestinal villi. Lactose malabsorption consists of a failure of the mechanisms of digestion and absorption of lactose that can be shown by laboratory tests, whereas lactose intolerance is defined as a clinical syndrome caused by lactose ingestion (1).
Adult-type hypolactasia is characterized by a fall of lactase activity levels to 5 to 10% of birth levels occurring during childhood and adolescence. The condition affects more than 75% of the population worldwide, with regional frequencies ranging from nearly 5% in northern Europe to more than 90% in some Asian and African countries (2).
Deficiency of lactase activity measured in enterocytes is the gold standard for the diagnosis of lactose malabsorption (3,4). However, it is an invasive test (5). Among non-invasive laboratory exams for the diagnosis of lactose malabsorption, the hydrogen breath test (HBT) is widely used. Nevertheless, it can cause symptoms such as bloating, vomiting, abdominal distension, abdominal pain, and diarrhea in intolerant patients. Sensitivity ranges from 69 to 100% and specificity from 96 to 100% when compared to lactase activity measurement in the intestinal biopsy (3,6,7).
Lactase activity is determined by lactase-phlorizin hydrolase (LCT), the gene encoding LPH, which has been mapped to chromosome 2q21-22 (8). Its persistence in adult life is of autosomal dominant inheritance (2, 9). Recently, it has been found that this persistence is due to two mutations (C/T-13910 and G/A-22018) in the LCT gene promoter region (10,11). The presence of the mutant alleles T-13910 and A-22018 has been related to persistent levels of lactase activity in duodenal biopsy material (12).
The objective of the present study was to determine the presence of the C/T-13910 and G/A-22018 mutations in the LCT gene in Brazilian individuals with and without lactose malabsorption diagnosed by HBT.
Subjects and Methods
The study was approved by the Ethics Research Committee of Hospital de Clínicas de Porto Alegre. Written informed consent was obtained from all subjects.
Twenty healthy individuals above 18 years of age from Porto Alegre, State of Rio Grande do Sul, Southern Brazil, were enrolled, 10 of them believed to be milk tolerant and 10 milk intolerant. Of the 20 individuals participating in the study, 18 (90%) were females. All were Caucasians, except for 1 Asian descendent. Overall mean age was 32.7 ± 7.3 years (range: 22-53 years). The mean ages of lactose-tolerant and -intolerant subjects were 32.2 ± 6.2 and 33.2 ± 8.6 years, respectively.
All participants were volunteer medical students. Sample size was calculated based on the frequency of the C allele in individuals with 100% lactose malabsorption (10). Since there are no data regarding the prevalence of LCT gene mutations in the Brazilian population, we calculated 10 subjects for each group, considering a 95% confidence interval, 0.9 power, and a standard error of 0.1 (Epi-Info, version 6.04).
Lactose-intolerant individuals were defined as subjects unable to drink any quantity of milk due to the presence of symptoms (nausea, abdominal pain, flatulence) or as subjects able to drink up to 2 glasses of milk (approximately 500 mL) a day (13). Tolerant individuals were those who reported being able to drink more than 3 glasses of milk a day without symptoms.
None of the subjects reported a history of gastrointestinal disorders associated with secondary lactose intolerance, e.g., Crohn's disease, ulcerative colitis, celiac disease, irritable bowel syndrome, diarrhea, or the use of antibiotics, enemas or laxatives during the 15 days preceding the exam. All subjects underwent the HBT and were classified as having lactose malabsorption or not.
H2 breath test
Subjects were instructed to fast overnight for 12 h and to avoid smoking for the same period of time before the HBT. The test was performed after oral ingestion of 50 g lactose (equivalent to 1 L of milk) diluted in 250 mL tap water.
Breath H2 excretion was measured in parts per million (ppm) using a gas chromatographer (Model 12i Quintron®, Quintron Instrument Company Inc., Milwaukee, WI, USA) at baseline and every 60 min after the ingestion of lactose over a period of 180 min. The HBT was considered to be positive (lactose malabsorption) when excretion was higher than 20 ppm compared to baseline. All subjects were observed regarding the occurrence of gastrointestinal symptoms such as diarrhea, bloating, abdominal pain, flatulence, and cramping over a period of 24 h after the test.
DNA
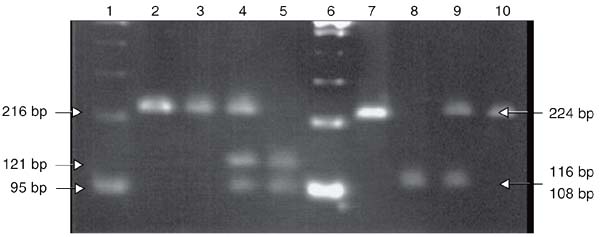
For DNA analysis, genomic DNA was isolated from venous blood (Easy-DNA, Invitrogen, Carlsbad, CA, USA). To test for the C/T-13910 variant, the fragment containing the polymorphic site was amplified by the polymerase chain reaction (PCR) using the primers C/T-for 5'-AAGACGTAA GTTACCATTTAATAC-3' and C/T-rev 5'-CGTTAATACCCACTGACCTATCCT-3' and digested with the BsmFI endonuclease (New England Biolabs, Beverly, MA, USA), generating fragments of 121 and 95 bp. For the detection of G/A-22018 polymorphism, the primers used were G/A-for 5'-TAAGA ACATTTTACACTCTTC-3' and G/A-rev 5'-AGAAAATGGGTTTTCGCCATG-3' and the PCR product was digested with HhaI (Invitrogen), generating fragments of 116 and 108 bp. Primers were designed for this study based on the GenBank sequence AC011893. PCR was performed using an Eppendorf Personal ThermoCycler (Eppendorf, Hamburg, Germany), in a final volume of 25 µL, using 100 ng genomic DNA, 1X 500 mM KCl buffer, pH 8.4, 5 mM MgCl2, 0.2 mM dNTPset, 20 pmol of each primer, and 1 U Taq DNA polymerase. All reagents were purchased from Invitrogen. The annealing temperature was 56ºC for both polymorphisms. PCR products and digestion fragments were analyzed on 2.5% agarose gel stained with ethidium bromide. A DNA test suggestive of lactase non-persistence was interpreted as a CC/GG genotype, whereas CT or TT and GA or AA genotypes were indicative of lactase persistence.
Statistical analysis
Data were analyzed statistically using the SPSS software, version 10.0. HBT values at baseline and at 60, 120, and 180 min for subjects with and without lactose malabsorption were compared by the Mann-Whitney test. The agreement between HBT and molecular analysis was assessed by the kappa coefficient (95%CI), with the level of significance set at 5%.
Results
All 10 lactose-tolerant subjects (100%) had a negative HBT and no evidence of gastrointestinal symptoms of lactose intolerance. Of the 10 lactose-intolerant subjects, 9 had a positive HBT and 1 had a negative result. However, all of these subjects presented some of the following symptoms during the 24-h period after the test: abdominal distention (N = 5), nausea (N = 5), flatulence (N = 6), and diarrhea (N = 5). HBT levels at baseline and at 60, 120, and 180 min for subjects with and without lactose malabsorption are shown in Figure 1. Hydrogen breath excretion levels in subjects with lactose malabsorption were significantly higher than in subjects without lactose malabsorption at 60, 120, and 180 min (P < 0.001). The maximum H2 breath excretion in subjects with lactose malabsorption occurred at 120 min.
The CT/GA, CT/AA, and TT/AA genotypes, related to the lactase persistence phenotype, were found in 10 individuals (CTGA in 6, CTAA in 1, and TTAA in 3). All of them showed negative HBT results. The CC/GG genotype, related to lactase non-persistence, was found in 10 individuals, 9 of whom showed positive HBT results. There was high agreement between the presence of mutations in the LCT gene promoter and HBT results: kappa = -0.9 (95%CI = -1.0 to -0.71), P < 0.001. A photograph of 2.5% agarose gel with different genotype patterns is shown in Figure 2.
Results of the hydrogen breath test (HBT) in subjects with (positive HBT) and without lactose malabsorption (negative HBT). Results are presented as median and 25-75% quartiles. *P < 0.001, HBT level of subjects with malabsorption compared with subjects without malabsorption (Mann-Whitney test).
PCR products digested with BsmFI (lanes 3, 4, 5) and HhaI (lanes 8, 9, 10) were separated on 2.5% agarose gel stained with ethidium bromide. Lanes 1 and 6, 100-bp DNA ladder. Lanes 2 and 7, undigested PCR products. Lane 3, CC homozygote. Lane 4, C/T heterozygote. Lane 5, TT homozygote. Lane 8, GG homozygote. Lane 9, G/A heterozygote. Lane 10, AA homozygote. Note that the 116- and 108-bp fragments cannot be distinguished on the 2.5% agarose gel but genotyping is clear by comparison with undigested PCR.
Discussion
The present study assessed the relationship between the presence of mutations C/T-13910 and G/A-22018 in the LCT gene and individuals with and without lactose malabsorption. There was significant agreement between the presence of these mutations and a negative HBT (i.e., without lactose malabsorption). Likewise, the absence of mutations significantly agreed with a positive HBT (lactose malabsorption).
The criterion of milk tolerance and intolerance was used only to enroll eligible individuals for lactose absorption or malabsorption, which was targeted for the study. Additionally, a weak correlation between self-reported lactose tolerance and presence of the LCT mutation has been previously shown (14), also in a population of equal gender distribution (15).
Enattah et al. (10) studied 9 Finnish families and identified two mutations related to lactase persistence (C/T-13910 and G/A-22018). All non-persistent lactase individuals were CC/GG homozygotes. Later, Kuokkanen et al. (12) described 4 genotypes when measuring LPH activity in intestinal biopsy material: CCGG and CCGA (lactase non-persistence), CTGA and TTAA (lactase persistence).
In the present study, we identified one individual with a CTAA genotype not previously described in the literature. As expected, this individual had a negative HBT, suggesting lactase persistence. In addition, one of the 10 subjects with the CCGG genotype (lactase non-persistence) had negative HBT, without symptoms. This could be explained by a slow decrease of lactase levels, with hypolactasia developing later in life. Another explanation could be the possible presence of an intestinal flora that did not produce H2 (16).
Among all subjects who presented genotypes of lactase persistence (CTGA, TTAA and CTAA), one CTGA subject presented nausea and abdominal distension during the test despite a negative HBT. The presence of symptoms may suggest a possible secondary cause for lactose intolerance; however, no further investigation such as gastrointestinal endoscopy was performed.
There are few studies comparing the presence of mutations and HBT, and all of them were conducted in European countries. This is the first study conducted on Brazilian subjects. The prevalence of adult-type primary hypolactasia, assessed by the lactose tolerance test, ranges from 38% in Caucasians to 95% in Blacks (17,18), with high regional differences due to the ethnic heterogeneity of the Brazilian population. An 8.4% prevalence of lactose malabsorption assessed by the HBT has been shown in the Brazilian pediatric population (19). Recently, a 53% prevalence of hypolactasia was identified in white individuals and a 91% prevalence was identified in nonwhite individuals by measuring lactase activity by immunohistochemistry in the duodenal mucosa (13). Regarding the frequency of C/T-13910 and G/A-22018 mutations compared to milk tolerance in the general population, a T allele frequency ranging from 0 to 0.112 has been shown in African Sub-Saharan populations and a frequency of 0.872 has been shown in Ireland (20).
It has been recognized that the C/T-13910 mutation is the major factor responsible for LPH persistence (11,21) and the G/A-22018 mutation appears to be only in co-segregation with C/T-13910 (22). Polymorphism of C/T-13910 has been used as a diagnostic predictor of adult-type hypolactasia in some European countries (5,23,24). However, this polymorphism has not been associated with lactase persistence in some African Sub-Saharan populations since several milk-drinking groups did not show the T-13910 allele. This suggests that this polymorphism may not be the only causative factor for lactase persistence (20). Therefore, the present study assessed the two mutations in a non-European country, showing a significant agreement between mutations G/A-22018 and C/T-13910 and HBT results.
Recent studies have demonstrated a 91-97% agreement of CC genotype with positive HBT results, and an 86-95% agreement between CT and TT genotypes and negative HBT results (5,7). Mutation analysis for the diagnosis of lactose malabsorption related to HBT presented a 97% positive and an 86% negative predictive value (7). In addition, this procedure has an advantage over the HBT by causing less discomfort to the patient (25).
The present study showed a high level of agreement between LCT mutation analysis and lactose absorption in Brazilian subjects. We suggest that this genetic test could be used for the diagnosis of adult-type primary hypolactasia.
References
1. Auricchio S, Troncone R. Genetically determined disaccharidase deficiences. In: Walker WA, Durie P, Hamilton JR, Walker-Smith JA, Watkins JB (Editors), Pediatric gastrointestinal disease. Ontario: BC Decker Inc.; 2000. p 677-700.
2. Sahi T. Hypolactasia and lactase persistence. Historical review and the terminology. Scand J Gastroenterol Suppl 1994; 202: 1-6.
3. Newcomer AD, McGill DB, Thomas PJ, Hofmann AF. Prospective comparison of indirect methods for detecting lactase deficiency. N Engl J Med 1975; 293: 1232-1236.
4. Vieira MR, Galvao LC, Fernandes MI. Relation of the disaccharidases in the small intestine of the rat to the degree of experimentally induced iron-deficiency anemia. Braz J Med Biol Res 2000; 33: 539-544.
5. Buning C, Genschel J, Jurga J, Fiedler T, Voderholzer W, Fiedler EM, et al. Introducing genetic testing for adult-type hypolactasia. Digestion 2005; 71: 245-250.
6. Romagnuolo J, Schiller D, Bailey RJ. Using breath tests wisely in a gastroenterology practice: an evidence-based review of indications and pitfalls in interpretation. Am J Gastroenterol 2002; 97: 1113-1126.
7. Hogenauer C, Hammer HF, Mellitzer K, Renner W, Krejs GJ, Toplak H. Evaluation of a new DNA test compared with the lactose hydrogen breath test for the diagnosis of lactase non-persistence. Eur J Gastroenterol Hepatol 2005; 17: 371-376.
8. Arola H, Tamm A. Metabolism of lactose in the human body. Scand J Gastroenterol Suppl 1994; 202: 21-25.
9. Sahi T. Genetics and epidemiology of adult-type hypolactasia. Scand J Gastroenterol Suppl 1994; 202: 7-20.
10. Enattah NS, Sahi T, Savilahti E, Terwilliger JD, Peltonen L, Jarvela I. Identification of a variant associated with adult-type hypolactasia. Nat Genet 2002; 30: 233-237.
11. Olds LC, Sibley E. Lactase persistence DNA variant enhances lactase promoter activity in vitro: functional role as a cis regulatory element. Hum Mol Genet 2003; 12: 2333-2340.
12. Kuokkanen M, Enattah NS, Oksanen A, Savilahti E, Orpana A, Jarvela I. Transcriptional regulation of the lactase-phlorizin hydrolase gene by polymorphisms associated with adult-type hypolactasia. Gut 2003; 52: 647-652.
13. Escoboza PM, Fernandes MI, Peres LC, Einerhand AW, Galvao LC. Adult-type hypolactasia: clinical, morphologic and functional characteristics in Brazilian patients at a university hospital. J Pediatr Gastroenterol Nutr 2004; 39: 361-365.
14. Enattah N, Pekkarinen T, Valimaki MJ, Loyttyniemi E, Jarvela I. Genetically defined adult-type hypolactasia and self-reported lactose intolerance as risk factors of osteoporosis in Finnish postmenopausal women. Eur J Clin Nutr 2005; 59: 1105-1111.
15. Carroccio A, Montalto G, Cavera G, Notarbatolo A. Lactose intolerance and self-reported milk intolerance: relationship with lactose maldigestion and nutrient intake. Lactase Deficiency Study Group. J Am Coll Nutr 1998; 17: 631-636.
16. Seva-Pereira A, da Silva RC, Pereira-Filho RA. Lactose malabsorption diagnosis with H2 breath test. Arq Gastroenterol 1999; 36: 18-26.
17. Troncon LE, Collares EF, Oliveira RB, Padovan W, Meneghelli UG. Lactose malabsorption in adult patients at the Hospital das Clínicas de Ribeirão Preto. Arq Gastroenterol 1981; 18: 106-112.
18. Seva-Pereira A, Beiguelman B. Primary lactose malabsorption in healthy Brazilian adult Caucasoid, Negroid and Mongoloid subjects. Arq Gastroenterol 1982; 19: 133-138.
19. Pretto FM, Silveira TR, Menegaz V, de Oliveira J. Lactose malabsorption in children and adolescents: diagnosis through breath hydrogen test using cow milk. J Pediatr 2002; 78: 213-218.
20. Mulcare CA, Weale ME, Jones AL, Connell B, Zeitlyn D, Tarekegn A, et al. The T allele of a single-nucleotide polymorphism 13.9 kb upstream of the lactase gene (LCT) (C-13.9kbT) does not predict or cause the lactase-persistence phenotype in Africans. Am J Hum Genet 2004; 74: 1102-1110.
21. Troelsen JT, Olsen J, Moller J, Sjostrom H. An upstream polymorphism associated with lactase persistence has increased enhancer activity. Gastroenterology 2003; 125: 1686-1694.
22. Rasinpera H, Savilahti E, Enattah NS, Kuokkanen M, Totterman N, Lindahl H, et al. A genetic test which can be used to diagnose adult-type hypolactasia in children. Gut 2004; 53: 1571-1576.
23. Jarvela IE. Molecular genetics of adult-type hypolactasia. Ann Med 2005; 37: 179-185.
24. Bodlaj G, Stocher M, Hufnagl P, Hubmann R, Biesenbach G, Stekel H, et al. Genotyping of the lactase-phlorizin hydrolase-13910 polymorphism by LightCycler PCR and implications for the diagnosis of lactose intolerance. Clin Chem 2006; 52: 148-151.
25. Matthews SB, Waud JP, Roberts AG, Campbell AK. Systemic lactose intolerance: a new perspective on an old problem. Postgrad Med J 2005; 81: 167-173.
 Address for correspondence: A.C. Bulhões, Centro de Pesquisas, Hospital de Clínicas de Porto Alegre, Rua Ramiro Barcelos, 2350, 90035-903 Porto Alegre, RS, Brasil. Fax: +55-51-2101-8760. E-mail: andreabulhoes@yahoo.com.br
Address for correspondence: A.C. Bulhões, Centro de Pesquisas, Hospital de Clínicas de Porto Alegre, Rua Ramiro Barcelos, 2350, 90035-903 Porto Alegre, RS, Brasil. Fax: +55-51-2101-8760. E-mail: andreabulhoes@yahoo.com.br
Part of this study was presented at the Digestive Disease Week 2007, May 20-25, 2007, Washington, DC, USA. Researh supported by Fundo de Incentivo à Pesquisa (FIPE) and Grupo de Pesquisa e Pós-graduação (GPPG, project No. 2004/04-123), Hospital de Clínicas de Porto Alegre, Porto Alegre, RS, Brazil. R.B. Mazzuca was the recipient of a FAPERGS-PIBIC grant. Received March 2, 2007. Accepted August 27, 2007.
- 1. Auricchio S, Troncone R. Genetically determined disaccharidase deficiences. In: Walker WA, Durie P, Hamilton JR, Walker-Smith JA, Watkins JB (Editors), Pediatric gastrointestinal disease Ontario: BC Decker Inc.; 2000. p 677-700.
- 2. Sahi T. Hypolactasia and lactase persistence. Historical review and the terminology. Scand J Gastroenterol Suppl 1994; 202: 1-6.
- 3. Newcomer AD, McGill DB, Thomas PJ, Hofmann AF. Prospective comparison of indirect methods for detecting lactase deficiency. N Engl J Med 1975; 293: 1232-1236.
- 4. Vieira MR, Galvao LC, Fernandes MI. Relation of the disaccharidases in the small intestine of the rat to the degree of experimentally induced iron-deficiency anemia. Braz J Med Biol Res 2000; 33: 539-544.
- 5. Buning C, Genschel J, Jurga J, Fiedler T, Voderholzer W, Fiedler EM, et al. Introducing genetic testing for adult-type hypolactasia. Digestion 2005; 71: 245-250.
- 6. Romagnuolo J, Schiller D, Bailey RJ. Using breath tests wisely in a gastroenterology practice: an evidence-based review of indications and pitfalls in interpretation. Am J Gastroenterol 2002; 97: 1113-1126.
- 7. Hogenauer C, Hammer HF, Mellitzer K, Renner W, Krejs GJ, Toplak H. Evaluation of a new DNA test compared with the lactose hydrogen breath test for the diagnosis of lactase non-persistence. Eur J Gastroenterol Hepatol 2005; 17: 371-376.
- 8. Arola H, Tamm A. Metabolism of lactose in the human body. Scand J Gastroenterol Suppl 1994; 202: 21-25.
- 9. Sahi T. Genetics and epidemiology of adult-type hypolactasia. Scand J Gastroenterol Suppl 1994; 202: 7-20.
- 10. Enattah NS, Sahi T, Savilahti E, Terwilliger JD, Peltonen L, Jarvela I. Identification of a variant associated with adult-type hypolactasia. Nat Genet 2002; 30: 233-237.
- 11. Olds LC, Sibley E. Lactase persistence DNA variant enhances lactase promoter activity in vitro: functional role as a cis regulatory element. Hum Mol Genet 2003; 12: 2333-2340.
- 12. Kuokkanen M, Enattah NS, Oksanen A, Savilahti E, Orpana A, Jarvela I. Transcriptional regulation of the lactase-phlorizin hydrolase gene by polymorphisms associated with adult-type hypolactasia. Gut 2003; 52: 647-652.
- 13. Escoboza PM, Fernandes MI, Peres LC, Einerhand AW, Galvao LC. Adult-type hypolactasia: clinical, morphologic and functional characteristics in Brazilian patients at a university hospital. J Pediatr Gastroenterol Nutr 2004; 39: 361-365.
- 14. Enattah N, Pekkarinen T, Valimaki MJ, Loyttyniemi E, Jarvela I. Genetically defined adult-type hypolactasia and self-reported lactose intolerance as risk factors of osteoporosis in Finnish postmenopausal women. Eur J Clin Nutr 2005; 59: 1105-1111.
- 15. Carroccio A, Montalto G, Cavera G, Notarbatolo A. Lactose intolerance and self-reported milk intolerance: relationship with lactose maldigestion and nutrient intake. Lactase Deficiency Study Group. J Am Coll Nutr 1998; 17: 631-636.
- 16. Seva-Pereira A, da Silva RC, Pereira-Filho RA. Lactose malabsorption diagnosis with H2 breath test. Arq Gastroenterol 1999; 36: 18-26.
- 17. Troncon LE, Collares EF, Oliveira RB, Padovan W, Meneghelli UG. Lactose malabsorption in adult patients at the Hospital das Clínicas de Ribeirão Preto. Arq Gastroenterol 1981; 18: 106-112.
- 18. Seva-Pereira A, Beiguelman B. Primary lactose malabsorption in healthy Brazilian adult Caucasoid, Negroid and Mongoloid subjects. Arq Gastroenterol 1982; 19: 133-138.
- 19. Pretto FM, Silveira TR, Menegaz V, de Oliveira J. Lactose malabsorption in children and adolescents: diagnosis through breath hydrogen test using cow milk. J Pediatr 2002; 78: 213-218.
- 20. Mulcare CA, Weale ME, Jones AL, Connell B, Zeitlyn D, Tarekegn A, et al. The T allele of a single-nucleotide polymorphism 13.9 kb upstream of the lactase gene (LCT) (C-13.9kbT) does not predict or cause the lactase-persistence phenotype in Africans. Am J Hum Genet 2004; 74: 1102-1110.
- 21. Troelsen JT, Olsen J, Moller J, Sjostrom H. An upstream polymorphism associated with lactase persistence has increased enhancer activity. Gastroenterology 2003; 125: 1686-1694.
- 22. Rasinpera H, Savilahti E, Enattah NS, Kuokkanen M, Totterman N, Lindahl H, et al. A genetic test which can be used to diagnose adult-type hypolactasia in children. Gut 2004; 53: 1571-1576.
- 23. Jarvela IE. Molecular genetics of adult-type hypolactasia. Ann Med 2005; 37: 179-185.
- 24. Bodlaj G, Stocher M, Hufnagl P, Hubmann R, Biesenbach G, Stekel H, et al. Genotyping of the lactase-phlorizin hydrolase-13910 polymorphism by LightCycler PCR and implications for the diagnosis of lactose intolerance. Clin Chem 2006; 52: 148-151.
- 25. Matthews SB, Waud JP, Roberts AG, Campbell AK. Systemic lactose intolerance: a new perspective on an old problem. Postgrad Med J 2005; 81: 167-173.
Correspondence and Footnotes
Publication Dates
-
Publication in this collection
09 Nov 2007 -
Date of issue
Nov 2007
History
-
Received
02 Mar 2007 -
Accepted
27 Aug 2007